Abstract
To describe clinical, oculographic and vestibular test profiles in patients with vestibular migraine (VM) who presented with acute peripheral vestibulopathy. VM was diagnosed according to Bárány Society or Neuhauser criteria. Neuro-otological examination, video-head impulse tests (v-HIT), cervical and ocular vestibular-evoked myogenic potentials (cVEMP/oVEMP), subjective visual horizontal (SVH) and audiometry were undertaken. Ten patients presented with prolonged vertigo. All had primary position unidirectional horizontal spontaneous nystagmus (mean slow-phase velocity 9.6 ± 7.0°). Horizontal canal vestibulo-ocular reflex was reduced in all (mean gain 0.54 ± 0.2) with refixation saccades (cumulative amplitude 6.4 ± 3.2°). Abnormality rates for cVEMP, oVEMP and SVH were 30%, 80%, 78%, respectively. Magnetic resonance imaging brain was normal in all patients. Patients were followed up over 6 months to 8 years with no change in the final diagnosis. VM can rarely present as an acute peripheral vestibulopathy with findings that mimic vestibular neuritis and should be considered in the differential diagnosis of acute prolonged vertigo.
Keywords
Introduction
Migraine is considered the chameleon of neurology and can present with headache, vertigo, visual and other sensory symptoms. Vestibular migraine (VM) is a common cause of acute spontaneous vertigo lasting minutes to days 1 as well as positional vertigo that could mimic benign paroxysmal positional vertigo. 2 Spontaneous horizontal and vertical nystagmus and persistent positional nystagmus have been reported during episodes of acute VM. 2 –4 Spontaneous horizontal nystagmus has been reported in 19–25%, 3,4 and positive bedside head impulse test was reported in 15%. 4
Vestibular function tests in inter-ictal VM are said to yield a lower rate of abnormalities than Ménière’s disease (MD). Caloric asymmetry was found in 22% and abnormal video-head impulse test (v-HIT) in 9% of patients with VM, compared to 67% and 37% in MD. 5 Thus, persistent vestibular asymmetries in VM are considered an exception rather than the norm. Here we present the clinical, oculographic and vestibular test characteristics of 10 patients who presented to an outpatient facility over a 10-year period, with prolonged spontaneous vertigo exceeding 24 h, associated with migraine headache and acute peripheral vestibular loss.
Methods
In a period from January 2011 to January 2020, a total of 5305 patients evaluated at the neuro-otology clinic received a diagnosis of VM. We retrospectively studied five women and five men (age 47.4 ± 14.5 years) who presented with (1) acute spontaneous vertigo lasting >24 h, (2) peripheral spontaneous nystagmus and (3) a positive bedside head impulse test. At presentation, all patients were acutely symptomatic, nine patients fulfilled International Headache Society-3
1
criteria for migraine headaches. Eight patients met Bárány Society
1
or Neuhauser
6
diagnostic criteria for definite VM (
V-HIT was performed using ICS Impulse (GN Otometrics, Taastrup, Denmark). Patients were instructed to visually fixate on a small dark target against a blank wall while the examiner delivered between 20 and 30 passive head impulses in the plane of each semicircular canal (SCC). The vestibulo-ocular reflex (VOR) gain for SCCs was calculated as the ratio of the area under the curves for the eye to head movement using the Labview software. V-HIT VOR gain was considered reduced if mean VOR gain was <2SD of the mean gain determined in the age-matched normal controls (horizontal canal (HC) <0.86, anterior canal (AC) <0.65, posterior canal (PC) <0.68). A Medelec Synergy EMG/EP system (Viasys Healthcare Systems, Old Woking, UK) recorded cervical VEMPs (cVEMPs) to monaural air-conducted 0.1 ms clicks and ocular VEMPs (oVEMPs) to binaural stimulation with bone-conducted ‘mini-taps’ delivered at Fz with a mechanical Brüel and Kjaer 4810 mini-shaker using previously described methods. 7 VEMP amplitude asymmetry ratio (AR) was calculated using the Jongkees-Formula and compared against laboratory control data (mean ± 2SD of 77 controls; upper limit of normal 30.2% for AC-cVEMP, 38.1% for BC-oVEMP). By convention, rightward deficits would be represented with a positive AR and leftward deficits with a negative AR. Patients with a unilaterally absent response were assigned an amplitude of zero (AR = 100%).
SVH was performed in a dark room with the participant sitting in an upright position looking at an illuminated red line on a monitor at a 1.5 m distance. The participant aligned the line with horizontal using a joystick over 10 trials. SVH test deviation exceeding ±2.5° of the gravitational horizontal for binocular viewing was defined an abnormal response. By convention, a rightward tilt of SVH, observed in right vestibular loss is reported as a positive value and a leftward tilt as a negative value. All patients had inter-ictal studies for comparison. The study was approved by Royal Prince Alfred Hospital Research Ethics and Governance Office, approval number HREC/13/RPAH/59. All subjects provided written informed consent.
Statistical analysis
Statistical analyses were performed with SPSS software version 23 for Windows (SPSS, Inc., Chicago, Illinois, USA) and
Results
History
All patients reported spontaneous rotatory vertigo lasting >24 h, associated with headaches. Vertigo was associated with aural fullness in one patient and high-pitched tinnitus in two patients. Hearing loss was not reported by any. Their clinical characteristics are summarized in Table 1. Eight patients reported multiple previous spells of vertigo associated with headache, similar to the episode studied. Four reported motion sensitivity and two had a history of vertigo from childhood. All patients had subsequent episodes of spontaneous vertigo. None, to our knowledge, had a second episode of peripheral vestibular loss.
Clinical and vestibular characteristics of patients during acute vestibular migraine presenting with prolonged spontaneous vertigo.a
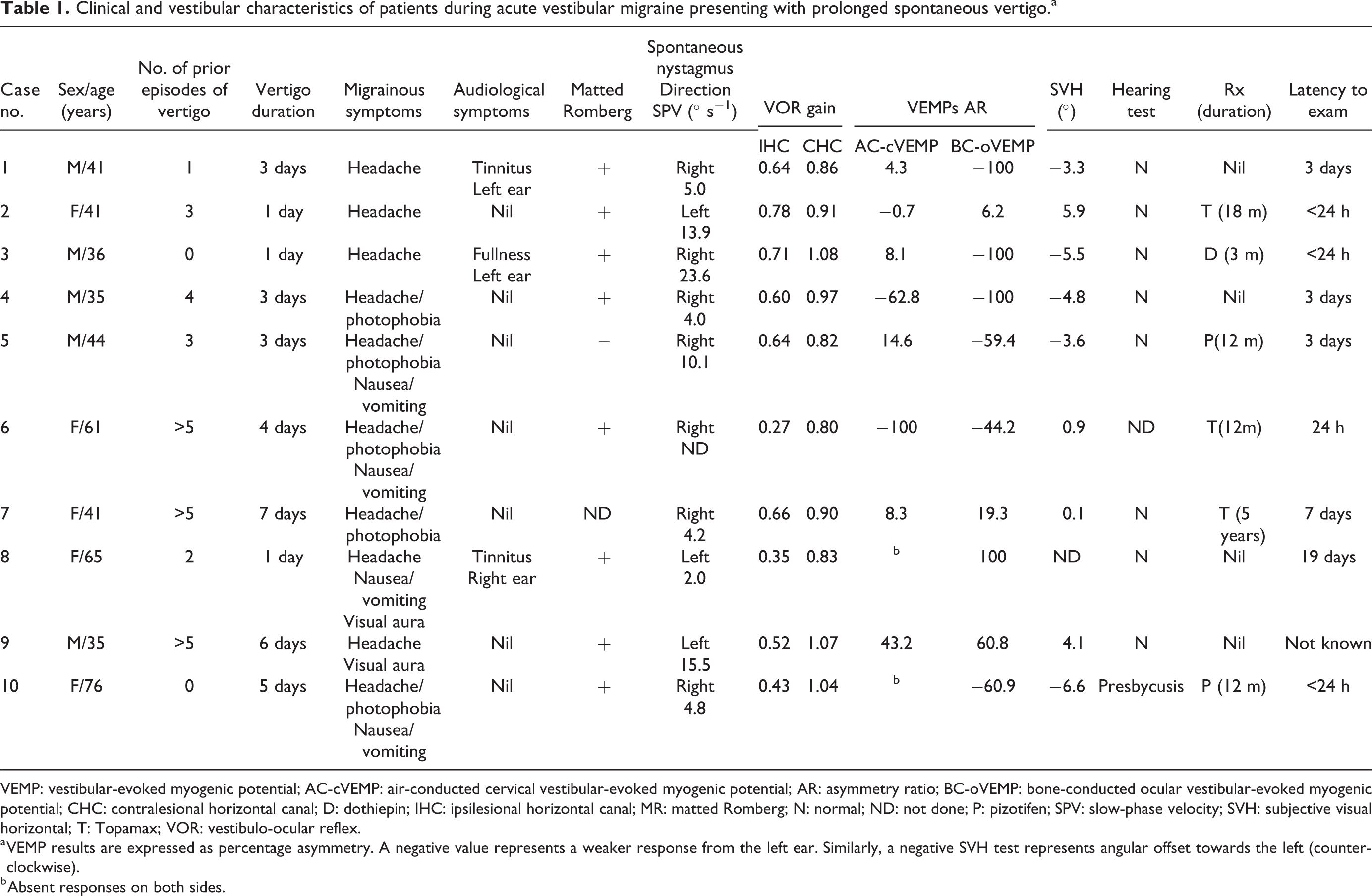
VEMP: vestibular-evoked myogenic potential; AC-cVEMP: air-conducted cervical vestibular-evoked myogenic potential; AR: asymmetry ratio; BC-oVEMP: bone-conducted ocular vestibular-evoked myogenic potential; CHC: contralesional horizontal canal; D: dothiepin; IHC: ipsilesional horizontal canal; MR: matted Romberg; N: normal; ND: not done; P: pizotifen; SPV: slow-phase velocity; SVH: subjective visual horizontal; T: Topamax; VOR: vestibulo-ocular reflex.
a VEMP results are expressed as percentage asymmetry. A negative value represents a weaker response from the left ear. Similarly, a negative SVH test represents angular offset towards the left (counter-clockwise).
b Absent responses on both sides.
Examination
In all patients, ictal assessment was indicative of an acute peripheral vestibular loss, including positive bedside head impulse test, unidirectional spontaneous horizontal nystagmus and no skew (peripheral HINTS examination). Average nystagmus slow-phase velocity (SPV) in the primary position was 9.6 ± 7.0° s−1 (range 2.0–23.6). As indicated in Table 1, v-HIT showed reduced HC VOR gain in all patients. Average affected VOR gain was 0.61 ± 0.2, 0.63 ± 0.2 and 0.70 ± 0.2 for the HC, AC and PC, respectively. Two patients had gains outside the normal range for all three canals, three for HC and AC, one for HC and PC and four for HC only. For HC impulses, average cumulative saccade amplitude was 6.4 ± 3.2°. First saccade amplitudes were 4.8 ± 2.7°, peak velocities were 180.5 ± 65.1° s−1, onset latencies were 166.1 ± 50.4 ms and duration was 47.0 ± 11.6 ms. Utricular dysfunction (asymmetric o-VEMPs) was present in 80% and saccular asymmetry (asymmetric cVEMPs) in 30% of patients. In all patients, unilateral loss was on the side implicated by the peripheral nystagmus (in the direction of the slow phase). We found no asymmetric hearing loss. The SVH test was undertaken in nine patients and showed abnormal ipsiversive bias towards the side of the vestibulopathy in seven patients. All patients underwent magnetic resonance imaging with diffusion weighted imaging sequences within 7 days of symptom onset which was found to be normal in all cases. Caloric testing was performed only in two cases (cases 1 and 3) and demonstrated no evidence of canal paresis. Audiometry was done in nine patients and was normal in all except for early presbycusis in case 10). Vestibular function test profile and spontaneous nystagmus for case 7 are shown in Figure 1 and Supplemental Video 1.

Vestibular function test profile in a patient with vestibular migraine (case 7). (a) Normal v-HITs; (b) ictal v-HITs shows reduced VOR gain with covert and overt refixation-saccades in the left HC; (c) v-HIT at 1 month follow-up shows recovery of VOR gain of the left HC; (d) ictal cervical and ocular vestibular-evoked myogenic potentials (cVEMP/oVEMP) are preserved bilaterally with normal amplitude asymmetry ratios; (e) normal ictal audiogram; (f) ictal right beating spontaneous horizontal nystagmus (black trace) with slow-phase velocity 7.3° s−1 and up-beating component (red trace) with slow-phase velocity 2.4° s−1. v-HITs: video-head impulse tests; VOR: vestibulo-ocular reflex; AC: anterior canal; AC-cVEMP: air-conducted cervical vestibular-evoked myogenic potential; AR: asymmetry ratio; BC-oVEMP: bone-conducted ocular vestibular-evoked myogenic potential; HC: horizontal canal; PC: posterior canal; *: reduced VOR gain.
Comparison with age-matched patients with VN
We compared spontaneous nystagmus SPV, HC VOR gain, saccade metrics and VEMP ARs between our VM group and 10 age-matched patients with acute VN (without headache). Average nystagmus SPV in the primary position was 14.0 ± 5.1° s−1 (range 7.8–24.3). For the VN group, the VOR gains were 0.36 ± 0.2, 0.36 ± 0.1 and 0.66 ± 0.2 for HC, AC and PC, respectively. HC cumulative saccade and first saccade amplitudes were 10.2 ± 3.5° and 6.4 ± 2.0°, peak velocity was 242.0 ± 60.3° s−1, onset latency was 194.7 ± 57.0 ms and duration was 49.6 ± 5.6 ms.
There were significant differences in VOR gain (
Follow-up
All but one patient were followed up for a mean period of 13 months (range 1–35 months) following the ictal episode. Six were commenced on migraine preventative therapies (topiramate in three, pizotifen in two and dothiepin in one patient), and at the time of follow-up all continued with the therapy. No patient reported history of fluctuating hearing loss or aural fullness. All experienced further episodes of vertigo, two patients developed ipsilesional posterior SCC BPV. No patient had recurrent episode of prolonged vertigo with acute peripheral vestibular loss. In seven patients, follow-up vestibular testing revealed improvement of peripheral vestibular function (average HC VOR gain = 0.77 ± 0.2, return of oVEMP to normal in six, return of cVEMP to normal in two).
Discussion
In this study, we identified a group of patients with VM presenting with headache time locked with prolonged spontaneous vertigo, spontaneous horizontal nystagmus, acute unilateral peripheral vestibular loss and vestibular function tests mimicking acute VN. In all cases vertigo lasted for days, patients had few associated cochlear symptoms and none hearing loss. We propose that acute unilateral vestibular loss observed in these patients was an uncommon manifestation of VM. Our cohort included a small subset of VM patients, and surprisingly, both genders were equally affected.
Mechanisms thought to underlie VM include vasospasm of the labyrinthine artery, reduced thresholds for trigeminovascular stimulation and dysfunctional activation of the brainstem, spreading depression affecting vestibular cortex, channelopathy and a sterile inflammation affecting the inner ear structures. 8,9 Although our patients’ presentation could be attributed to labyrinthine artery vasospasm, the low prevalence of auditory symptoms and absence of objective hearing loss makes labyrinthine ischaemia unlikely.
Could these patients have developed VM as a consequence of an initial episode of VN? Intense vestibular activation can serve as a migraine trigger. Development of migraine headache was found to be more prevalent in a group of migraine patients exposed to caloric testing during a 24h review period, when compared with a group of patients who did not receive the test. 10 In patients 3 and 10 with no prior history of migraine, it is conceivable that an initial attack of VN triggered subsequent migraines. The remaining patients reported shorter episodes of recurrent vertigo associated with headache or photophobia in the past. Lower VOR gain and larger cumulative saccade amplitude in patients with VN may be indicative of more profound vestibular loss in VN compared to the VM.
Could these patients represent early MD presenting with ictal v-HIT asymmetry? Reduced VOR gain has been described in MD patients during ictal period. 11 The prolonged duration of vertigo, absence of fluctuating aural symptoms or low- to medium-frequency sensorineural hearing loss on audiometry and the low nystagmus SPV 12 were more in keeping with VM than MD. Could these patients have been manifesting an autoimmune inner ear disorder? The absence of hearing loss on presentation or follow-up, normal imaging, non-recurrence of peripheral vestibular loss makes this explanation unlikely. Finally, the possibility of patients with a previous history of VM experiencing an isolated episode of VN cannot be excluded.
We describe the clinical, oculographic and vestibular function test profiles of 10 patients with VM who presented with an acute peripheral vestibular loss. While confirmation of cardinal signs of VN and their separation from stroke are a primary concern, VM should also be considered as a potential diagnosis in any patient with apparent VN who experiences either prior or subsequent attacks of vertigo.
Key findings
Vestibular migraine can rarely present with acute prolonged vertigo and oculographic and vestibular function test profiles consistent with acute peripheral vestibular loss.
Vestibular migraine should be considered as differential diagnosis in patients with acute peripheral vestibulopathy who have a history of either prior or subsequent attacks of vertigo.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AY acknowledges receipt of a scholarship grant from the University of Sydney, and reports to conflict of interest. MW receives grant funding from the National Health and Medical Research Council, and the Garnett Passe and Rodney Williams Memorial Foundation, and reports to conflict of interest. The remaining authors declare no conflicts of interest.
Funding
The author(s) disclosed no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
