Abstract
Background
We characterise the history, vestibular tests, ictal and interictal nystagmus in vestibular migraine.
Method
We present our observations on 101 adult-patients presenting to an outpatient facility with recurrent spontaneous and/or positional vertigo whose final diagnosis was vestibular migraine (n = 27) or probable vestibular migraine (n = 74). Ictal and interictal video-oculography, caloric and video head impulse tests, vestibular-evoked myogenic potentials and audiometry were performed.
Results
Common presenting symptoms were headache (81.2%), spinning vertigo (72.3%), Mal de Débarquement (58.4%), and motion sensitivity (30.7%). With fixation denied, ictal and interictal spontaneous nystagmus was observed in 71.3 and 14.9%, and purely positional nystagmus in 25.8 and 55.4%. Spontaneous ictal nystagmus was horizontal in 49.5%, and vertical in 21.8%. Ictal spontaneous and positional nystagmus velocities were 5.3 ± 9.0°/s (range 0.0–57.4), and 10.4 ± 5.8°/s (0.0–99.9). Interictal spontaneous and positional nystagmus velocities were <3°/s in 91.8 and 23.3%. Nystagmus velocities were significantly higher when ictal (p < 0.001/confidence interval: 2.908‒6.733, p < 0.001/confidence interval: 5.308‒10.085). Normal lateral video head impulse test gains were found in 97.8% (mean gain 0.95 ± 0.12) and symmetric caloric results in 84.2% (mean canal paresis 7.0 ± 23.3%). Air- and bone-conducted cervical-vestibular-evoked myogenic potential amplitudes were symmetric in 88.4 and 93.4% (mean corrected amplitude 1.6 ± 0.7, 1.6 ± 0.8) with mean asymmetry ratios of 13.0 and 9.0%. Air- and bone-conducted ocular-vestibular-evoked myogenic potentials were symmetric in 67.7 and 97.2% (mean amplitude 9.2 ± 6.4 and 20.3 ± 12.8 µV) with mean asymmetry ratios of 15.7 and 9.9%. Audiometry was age consistent and symmetric in 85.5%.
Conclusion
Vestibular migraine is characterised by low velocity ictal spontaneous nystagmus, which can be horizontal, vertical, or torsional, and normal audiovestibular test results.
Introduction
Vestibular migraine (VM) is characterised by recurrent episodes of vertigo or motion-induced dizziness which last minutes to days and is associated with a migrainous headache at least half of the time (1). VM represents a prevalence of 5‒7% of patients assessed in neuro-otology outpatient facilities (2,3), constitutes a significant burden to society and the healthcare system, and is associated with loss of work and reduced quality of life; its rapid and accurate diagnosis is paramount in improving patient wellbeing, and reducing healthcare costs (4).
Unlike other causes of vertigo commonly encountered in the outpatient clinic and emergency room that often present as a single syndrome (e.g. benign paroxysmal positional vertigo, Menière’s disease, and vestibular neuritis, which present as episodic positional vertigo, episodic spontaneous vertigo, and acute vestibular syndrome respectively), VM has diverse manifestations and can present with spontaneous or positional vertigo of brief or prolonged duration, which may or may not be associated with a migraine headache. In the patient presenting with episodic spontaneous vertigo (ESV) both VM and Menière’s disease need to be considered. Here, conventional vestibular tests like the bithermal caloric test (5), ictal nystagmus characteristics (6), and audiometry (7) assist diagnosis. When VM presents as episodic positional vertigo (EPV), BPV becomes a key differential diagnosis; positional nystagmus recorded during provocative (Dix-Hallpike) testing could demonstrate paroxysmal positional nystagmus in the plane of a given canal, which identifies and separates benign positional nystagmus from the central nystagmus observed in VM (8,9). Rarely, VM can also present as an acute vestibular syndrome (AVS) (10), in which case investigation for vestibular neuritis and posterior circulation stroke using head impulse, ictal nystagmus, test of skew and imaging becomes compulsory. Thus, depending on the presentation, ictal nystagmus, vestibular function testing and audiometry could prove useful when seeking to separate VM from its mimics.
In the present study, our aim was to profile VM using ictal and interictal nystagmus characteristics, detailed tests of semicircular canal and otolith function, and audiometry in a large cohort of patients diagnosed by clinical criteria.
Methods
Standard protocol approvals and patient consents
This study has received approval by the Sydney Local Health District Ethics Review Committee (Royal Prince Alfred Hospital zone, Sydney, Australia) for the use of human participants (protocol number X18-0087). Written informed consent was obtained from all participants according to the 1964 Declaration of Helsinki and its later amendments. All figures and/or videos of research patients have been anonymised. Anonymised data will be shared by request from any qualified investigator.
Participants: Eligibility criteria
Between August 2014 and June 2020, adults over the age of 18 years were prospectively convenience recruited from a neurology outpatient clinic. Eligibility criteria for participation in this observational cohort study included a history of spontaneous vertigo lasting at least 5 min, or positional vertigo lasting at least 10 sec, recurring more than once per month. Recruited participants were offered a take-home video oculography (VOG) device and were trained in conducting a routine of fixation-denied eye recordings during episodes of vertigo. In another group, patients presenting to the Emergency Department (ED) of the Royal Prince Alfred Hospital (Sydney, Australia) with an acute vestibular event and rotatory vertigo underwent ictal VOG conducted by a medical professional (March 2018 to May 2020). In both groups, patients’ results were included in the final study cohort if their final diagnosis was VM, and had ictal VOG samples either self-recorded at home, or recorded in the hospital ED.
The research design goal was to include the results of a minimum of 100 patients with a final diagnosis of VM, to help reduce the influence of outliers. Individual patient follow-up and further audiovestibular testing was arranged by their respective consulting neurologists or general practitioner as deemed appropriate, irrespective of participation in the research study.
Based on the 2012 Bárány Society criteria, patients were classified as having VM when they reported current or previous headache, with current headache features time-locked to multiple recurring vertiginous events lasting minutes to days (11). When patients showed only one migraine headache feature with acute recurring vertigo, a diagnosis of probable (p) VM was made.
Home-video oculography
Frenzel style nystagmus recording goggles (the VOG ‘device’) were custom made (Neuromed Electronics, Sydney Australia) and consisted of a small video camera attached to the fenestrated left eye of opacified swimmers’ goggles illuminated by infrared lights. With the complete left eye in view of the video camera, both eye pieces of the goggles were blocked from light, creating a fixation-denied visual field.
Home VOG patients were advised to make recordings at the onset of a vertigo attack in a series of positions, including sitting upright looking forward for 15 sec, lying supine, and in either lateral lying position for 30 sec each. Patients were asked to verbally describe their current position for the audio recording, and to maintain their eyes in a centre gaze position. Patients who experienced episodes of vertigo lasting more than 60 min were instructed to make successive recordings every 15 min from symptom onset for the first hour of vertigo, when possible. When one or more episodes of vertigo had been recorded, or after a 3-month loan period, whichever came first, the participants were asked to return their VOG device at their next follow-up appointment or to upload the video files to a secure, encrypted file-sharing website hosted by the University of Sydney, Australia. When antiemetics or vestibular suppressants had been prescribed by the consulting neurologists, patients were advised to take their medication(s) before conducting a video recording. Patients presenting to the hospital ED with an acute vestibular syndrome had VOG conducted by the neurologist using vision-denied Frenzel goggles with video recording.
Nystagmus analysis
Videos were analysed for nystagmus after the return of each patient device by authors who were blinded to the patient’s final diagnosis. Eye videos were recorded at 30 Hz and analysed offline with a custom LabVIEW nystagmus analysis program (detailed nystagmus analysis methods can be found in Young et al. 2019 (Appendix) (6)). The patients’ eye movements were analysed in both the vertical and horizontal planes. Nystagmus velocity during the slow phase of eye movement was measured in degrees per second (°/s). In cases of multiple video recordings per patient, one video was chosen that represented the fastest nystagmus SPV while sitting upright, lying supine and lying laterally on either side. When no nystagmus was visible on analysis, the video result was still included in the study if the patient reported vertigo during the recording and thus was included as an SPV of 0°/s in the statistical analysis. There was no minimum or maximum nystagmus velocity imposed for reporting, however a minimum of three nystagmus beats within 15 sec of recording was required for analysis. When nystagmus was observed in both the horizontal and vertical planes, the plane of the fastest nystagmus SPV was used for statistical analysis and reporting.
Audiovestibular testing
We endeavoured to perform vHIT and magnetic resonance imaging on all patients presenting with AVS, caloric testing and audiometry on all patients with prolonged spontaneous vertigo lasting > 10 min, and positional testing on all patients presenting with episodic positional vertigo. Patients received additional imaging and/or audiovestibular testing as requested by their referring specialist. Audiovestibular testing was not a pre-requisite for study participation, not all patients received all audiovestibular tests, and total patients tested are listed for each test. All patients included in this study received ictal and interictal video oculography. Sixty-four (63.4%) patients underwent magnetic resonance imaging. All audiovestibular tests were conducted interictally unless noted otherwise.
Audiometry
Audiometry pure-tone hearing threshold asymmetry was defined as an interaural difference ≥20 decibels hearing level (dBHL, ISO) at one frequency, ≥15 dBHL at two frequencies, or ≥10 dBHL at three or more frequencies (12).
Bithermal caloric test
Bithermal caloric testing was conducted with water irrigations of 30° and 44°C for 25–40 sec. Canal paresis (CP), also known as unilateral weakness, was calculated with the Jongkees formula using the peak slow-phase velocity (SPV) of nystagmus produced during each irrigation. CP was considered within normal limits if ≤25% (13).
Vestibular-evoked myogenic potentials (VEMPs)
Vestibular-evoked myogenic potential (VEMP) testing was conducted with a Natus Medelec Synergy evoked-potential device (version 20.0, CA, USA). Cervical (c) and ocular (o) VEMPs were recorded in response to monaural air-conducted (AC) clicks and bone-conducted (BC) minishaker taps to the forehead midline. AC 0.1 millisecond (ms) clicks were delivered monaurally using TDH-49 headphones, at a rate of 5/sec, alternating polarity, at 105 decibels normal hearing level (dBnHL) (140 dB peak sound-pressure level). BC 1 ms, 20-volt amplitude taps were delivered by a hand-held bone vibrator (Bruel and Kjaer 4810) with condensation polarity at a rate of 5/sec. VEMP symmetry was compared to age-matched normal controls tested on the same equipment (13). Reflex amplitudes were measured in peak-to-peak microvolts (µV) for oVEMPs, and corrected amplitude (CA) for cVEMPs. Corrected amplitude was calculated using the peak-to-peak cVEMP reflex amplitude against the baseline sternocleidomastoid muscle activation, and is therefore a ratio without unit. Asymmetry ratios were calculated using the Jongkees formula.
Video head impulse test (vHIT)
Lateral video head impulse tests (vHITs) were conducted using ICS Impulse USB goggles (Otometrics, Taastrup, Denmark). Vestibulo-ocular reflex (VOR) gains were calculated for each canal after 20 impulses were performed in each canal plane. vHIT gains were compared against the means (± two standard deviations) of age-matched normal controls (14). Lateral vHIT saccade analysis was processed and analysed offline using custom software designed by author AP Bradshaw (LabView v2012; National Instruments, Austin, TX).
Statistics
Statistical analysis was performed by Allison S Young using IBM SPSS Statistics for Windows, Version 26.0 (Armonk, NY: IBM Corp), with assistance from Mario D’Souza (Senior Statistician, Central Clinical School of Medicine, University of Sydney), between February 2020 to July 2021. Linear regression analyses were conducted to compare dependent variables including nystagmus SPV in °/s, (ictal, interictal, spontaneous, and positional), lateral vHIT analysis (gain, saccade frequency (%), cumulative saccade amplitude (°), first saccade (S1) amplitude (°), peak velocity (°/s), onset (ms), and duration (ms)), and VEMP reflex symmetry (%), latency (ms), and amplitude (µV/CA). This analysis was conducted within the framework of generalised estimating equations, which accounts for the correlation in data when more than one observation per person is included. The output provided was the mean pairwise difference between conditions with a 95% confidence interval (CI), while the significance cut-off value was p < 0.05. As peripheral vestibular function may change over time, patient age was separated into three decade groups (<40 years, 40–60 years, and >60 years) and was included as a possible confounding independent variable in all of the above calculations (15). Comparisons in test results were made between the VM and pVM groups and age-matched normal controls. Descriptive statistics include means ± 1 standard deviation.
Results excluded from analysis
Patients with a diagnosis of VM or pVM comorbid with another audiovestibular pathology were excluded from the study, including those with Menière’s disease, delayed endolymphatic hydrops, superior semicircular canal dehiscence, and autoimmune inner ear disease.
Results
Patient characteristics
In the home VOG group, over a period of 6 years, 633 patients with reports of recurrent spontaneous or positional vertigo were prospectively recruited and were taught how to record home-videos on a portable VOG device. The results of 92 patients who made ictal fixation-denied home-video recordings and whose clinical assessment, imaging, and audiovestibular test results were consistent with a diagnosis of VM or pVM were included in the study. The mean home VOG device loan period was 78 days (range 7–316 days), and the mean number of videos recorded per patient was seven (range 1–45 videos). In the hospital ED group, over a period of 2 years 773 patients received ictal VOG recordings after presenting with an acute vestibular syndrome (i.e. continuous vertigo or imbalance for >24 h), and the results of nine patients with a final diagnosis of VM were included in the study.
Based on the Bárány Society criteria, in both groups combined, 73.3% (n = 74) had a diagnosis of pVM, and 26.7% (n = 27) with VM. The mean age was 48 ± 15 years, with 35 men and 66 women. No adverse effects were reported by participants after using the home-video recording device.
Symptoms
Of the 101 patients included in the study, 8.9% (n = 9) presented with acute vestibular syndrome to the emergency room. From the home VOG group, 30.7% (n = 31) reported episodic spontaneous vertigo (recurrent vertigo without trigger), 6.9% (n = 7) reported episodic positional vertigo (recurrent vertigo triggered by lying down in one or more positions), and 53.5% (n = 54) reported episodic spontaneous and positional vertigo (16).
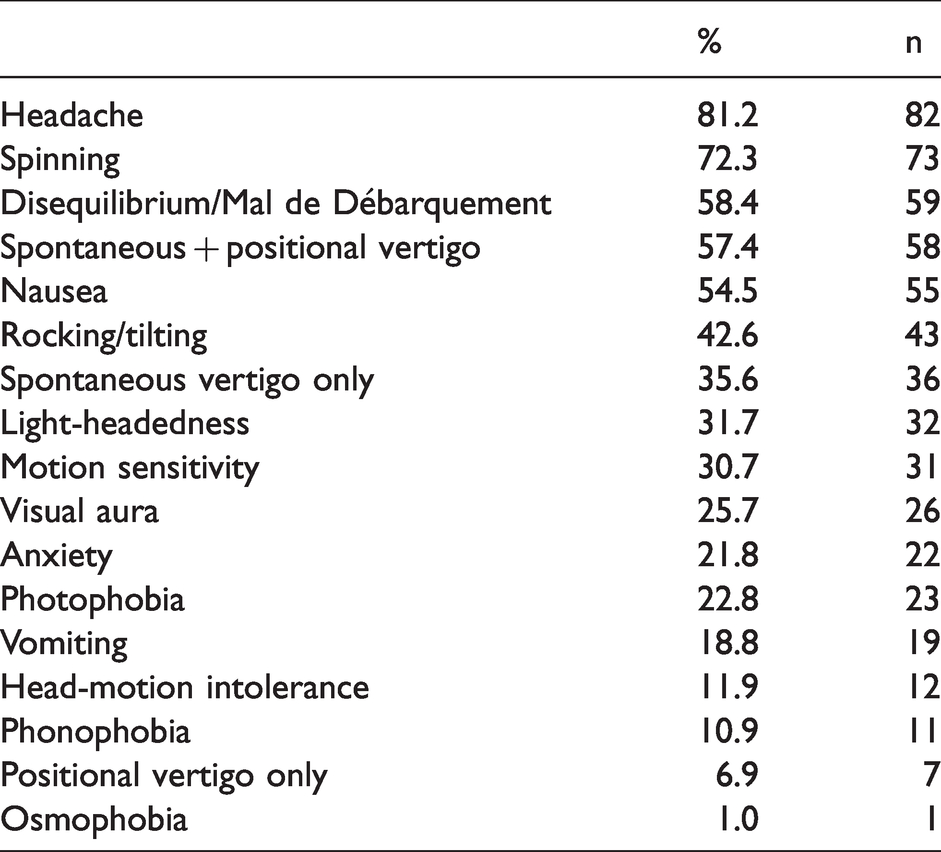
The presenting vestibular symptoms (some patients had more than one symptom) were true spinning vertigo in 72.3% (n = 73), followed by disequilibrium and/or Mal de Débarquement in 58.4% (n = 59), rocking/tilting sensation in 42.6% (n = 43), motion-sensitivity in 30.7% (n = 31) and head-motion intolerance in 11.9% (n = 12). Migraine symptoms included headache in 81.2% (n = 82), visual aura in 25.7% (n = 26), photophobia in 22.8% (n = 23), phonophobia in 10.9% (n = 11), and osmophobia in 1.0% (n = 1) (Table 1). Nausea was reported in 54.5% (n = 55), with vomiting in 18.8% (n = 19).
Vestibular and migraine-related symptoms in vestibular nigraine patients.
The reported duration of vertigo symptoms ranged from seconds to weeks, and included some with persistent symptoms (Figure 1). Anxiety was self-reported or reported by a treating psychologist in 21.8% (n = 22). Vertigo provocation was reported to be spontaneous in 35.7% (n = 36), purely positional in 6.9% (n = 7), and a combination of spontaneous and positional in 57.4% (n = 58). Bilateral or unilateral tinnitus was reported in 28.7% and 17.8% of patients (n = 29 and 18), with aural fullness reported unilaterally or bilaterally in 16.8% and 11.9% of patients (n = 17 and 12) (Table 2).
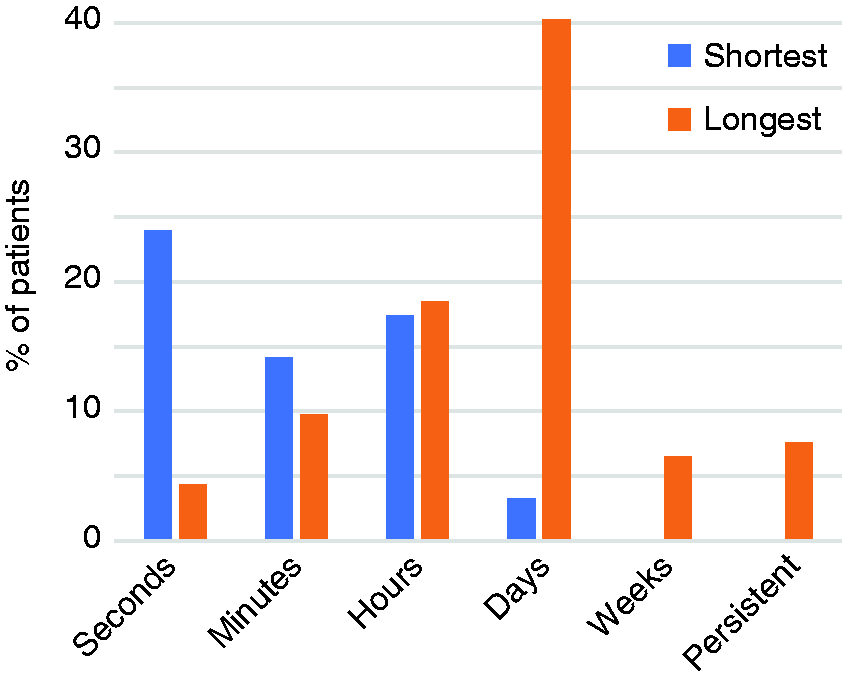
Reported duration of recurrent vertigo episodes in vestibular migraine patients.
Aural symptoms reported in vestibular migraine patients.

Ictal spontaneous nystagmus
During their episodes of vertigo (ictally), 71.3% (n = 72) of patients displayed spontaneous nystagmus while sitting upright, with a mean SPV of 5.3 ± 9.0°/s (range 0.0–57.4), with fixation denied (Table 3). The ictal spontaneous nystagmus SPV range was not normally distributed, and only six patients (5.9%) displayed spontaneous nystagmus SPV > 15°/s, with the 95 remaining patients (94.1%) displaying spontaneous nystagmus which was <15°/s. Spontaneous nystagmus fast-phase direction in the upright position was horizontal in 49.5% (n = 50) (Video 1), down-beating in 11.9% (n = 12) and up-beating in 9.9% (n = 10) (Video 2, Figure 2). A small portion of patients (4.0%, n = 4) also displayed an additional persistent torsional component in their eye movements. There was no significant difference in the spontaneous nystagmus SPVs between the VM and the pVM diagnostic groups (p = 0.574).
Ictal spontaneous and positional nystagmus slow-phase velocity (°/s) by body position in vestibular migraine patients.

L: left; Max: maximum; Min: minimum; Q1: first quartile; Q3: third quartile; R: right; SD: standard deviation; °/s: degrees per second.

Spontaneous and positional nystagmus in vestibular migraine patients. (a) Spontaneous horizontal left-beating nystagmus while sitting upright, which mildly enhances (becomes faster) in the supine and right lateral position. (b) Spontaneous vertical up-beating nystagmus while sitting upright, which enhances in the supine and lateral lying positions. (c) Low velocity spontaneous right-beating nystagmus while sitting upright, which changes into down-beating nystagmus in the supine and lateral lying positions. (d) Spontaneous horizontal left-beating nystagmus visible while sitting upright, which changes direction in the left lateral position; left-beating in the right lateral position, and right-beating in the left lateral position (apogeotropic).
Direction-reversal nystagmus
Most patients (86%) recorded more than one ictal video and showed unidirectional nystagmus only (i.e. the nystagmus was always beating in the same direction). However, five patients showed a spontaneous nystagmus direction-reversal over different recordings whilst sitting upright with forward gaze; four showed a nystagmus direction-reversal over different days, and one within the same day over a 4-h recording window. Four patients displayed a horizontal direction-reversal, and one displayed a vertical direction-reversal. One of these patients displayed a pure-tone hearing threshold asymmetry, which was in the high frequencies. Three of these patients also displayed a significant asymmetry on caloric testing, but did not meet the diagnostic criteria for Menière’s disease due to lack of unilateral low-/mid-frequency hearing loss on the audiogram, subjective hearing fluctuations, or tinnitus.
Ictal positional nystagmus
A total of 89 patients conducted positional recordings whilst ictal. Positional nystagmus without spontaneous nystagmus was observed in at least one lying position (supine or lying laterally on either side) in 25.8% of patients (n = 23), while 69.7% (n = 62) displayed nystagmus in both the upright and lying positions (Video 3). Only 3.4% (n = 3) showed no nystagmus in any position, and one patient displayed spontaneous nystagmus while sitting upright that disappeared when lying down. Ictal supine, right and left lateral nystagmus mean SPVs (and ranges) were 11.5°/s (0.0–99.9), 10.3°/s (0.0–57.7), and 9.4°/s (0.0–91.9), respectively (Table 3).
Ictal nystagmus recorded while supine had a significantly faster SPV when compared with nystagmus recorded in the upright position (p = 0.003, CI: 2.158–10.533) Video 2). The same was found for lying laterally on either side in comparison with the upright position (right and left lateral positions: p = 0.017 and 0.047, CI: 0.899–9.361 and 0.063–8.466). There was no significant difference in nystagmus SPV between the supine and the right/left lateral positions (p = 0.578 and 0.338). When lying on either side, 14 patients showed positional direction-changing nystagmus with respect to gravity; 10 patients showed a geotropic pattern in the lateral lying position (horizontal positional nystagmus with a fast-phase beating towards the ground) (Video 4), while four showed apogeotropic positional nystagmus (Video 5). All nystagmus observed in the lying positions was persistent.
The nystagmus of the nine patients first presenting with an acute vestibular syndrome to the hospital ED (and later diagnosed with VM) was significantly slower than in those who self-recorded ictally at home (p = 0.013, CI: 1.0982–9.1893), presumably due to the ED recordings occurring further from the onset of vertigo.
Ictal vs. interictal nystagmus
All but five patients underwent interictal fixation-denied VOG during clinical assessment, with the majority (90.2%, n = 83) having recorded videos with nystagmus SPV analysis. The remaining patients underwent clinical VOG without video recording, therefore nystagmus SPV analysis was not possible; however, the presence or absence of nystagmus was reported by the consultant neurologist. In the home VOG group, 14.9% (n = 15) showed interictal spontaneous nystagmus in the upright position, while 55.4% (n = 56) showed nystagmus in at least one of the three lying positions. Of those patients with interictal nystagmus, velocities were generally low, with spontaneous nystagmus SPVs < 3°/s in 91.8% of patients, and positional nystagmus SPVs < 5°/s in 81.5%. The mean interictal upright nystagmus SPV was 0.5 ± 1.5°/s (range 0.0–11.0) (Table 4). The mean interictal positional nystagmus SPV for all three positions combined (supine, left and right lateral) was 2.6 ± 4.2°/s (range 0.0–26.3). The ictal spontaneous nystagmus SPV while upright was significantly faster than interictal (p < 0.001, CI: 2.908–6.733). Similarly, the ictal positional nystagmus SPVs, grouped together, were significantly faster than interictal (p < 0.001, CI: 5.308–10.085) (Figure 3).
Interictal spontaneous and positional nystagmus slow-phase velocities in vestibular migraine patients.

Max: maximum; Min: minimum; Q1: first quartile; Q3: third quartile; SD: standard deviation; °/s: degrees per second.
Note: “Positional” indicates nystagmus recorded in the supine, left- and right-lateral positions, combined.

Ictal vs. interictal nystagmus slow-phase velocities in vestibular migraine patients. “Primary” indicates nystagmus recording in the upright position, gaze forward, and “positional” indicates the supine, left and right lateral position nystagmus velocity combined. “Ictal” indicates nystagmus recorded during an attack of vertigo, and “interictal” indicates nystagmus recorded between attacks.
Audiometry
Pure-tone audiometry was conducted in 75 patients; 80.0% with ESV were tested, 57.1% with EPV, and 22.2% with AVS. Results showed symmetric thresholds within the normal limits in 62.7% (n = 47), and a further 22.7% (n = 17) with age-consistent symmetric hearing loss in the high frequencies (presbycusis). Of those with asymmetries, 8.0% (n = 6) involved the high frequencies, 4.0% (n = 3) showed a flat pattern (including both the high, mid and low frequencies), and 2.7% (n = 2) showed a conductive asymmetry (Figure 4).

Audiovestibular test results in vestibular migraine patients.
Caloric test
Bithermal caloric testing was undertaken in 64 and completed in 57 patients; 61.2% with ESV were tested, 57.1% with EPV, and 11.1% with AVS; 66.2% of patients with ESV lasting hours underwent caloric testing. Normal and symmetric caloric results were found in 84.2% (n = 48) of all patients. Of the nine patients with canal paresis, four had normal hearing thresholds, and five had a symmetric presbycusis pattern of hearing loss. Stimulus durations of 40, 30 and 25 sec were used in 14.1%, 48.4%, and 26.6% of tests. The average peak SPV recorded was 20.8°/s for warm water irrigation (44°C), and 15.1°/s for cool water (30°C). There was no significant difference in the nystagmus SPVs based on irrigation times (p = 0.655). In seven VM patients, the test was abandoned due to severe nausea, and they were not included in the analysis.
Video head impulse test
Lateral canal vHIT was conducted interictally for 89 patients; 85.9% with ESV were tested, 100% with EPV, and 88.9% with AVS. Three-dimensional vHIT was conducted for 28 patients. With both left and right canal values combined, mean lateral vHIT gain was 0.95 (±0.12), mean anterior gain was 0.90 (±0.19), and mean posterior gain was 0.85 (±0.16). One patient showed reduced gains in both lateral canals, one patient in one lateral canal, one patient showed reduced gain in one anterior canal, and all patients (100%) showed posterior canal gains within normal limits. Compared with normal controls, there were no significant differences in lateral vHIT gains, saccade frequency, cumulative saccade amplitude, first saccade (S1) amplitude, or duration (p = 0.205, 0.177, 0.222, 0.173, and 0.476). However, S1 peak velocity and S1 onset in those with VM had significantly higher velocities and later onset compared to normal controls (p = 0.027, CI: 1.064–17.459, and p = 0.004, CI: 9.755–50.151 respectively) (Table 5).
Interictal lateral video head impulse test results in patients with vestibular migraine (VM), and in normal controls (NC).

°: degrees; °/s: degrees per second; ms: milliseconds; SD: standard deviation; S1: first saccade.
Ictal vHIT showing peripheral dysfunction
Three patients in this study underwent vHIT during an acute episode of vertigo. Of these, all showed a unilateral reduction in semicircular canal function. The first patient was previously found to have normal interictal lateral vHIT gain (right ear 1.02, and left ear 0.95). However, during an ictal presentation to an outpatient clinic 3 months later, spontaneous right-beating nystagmus with SPV of 7.3°/s was observed, and a reduced left vHIT gain with significant catchup saccades was recorded (0.72), with normal right ear gain (0.90). The left lateral canal VOR function slowly returned to normal over 1 month, with an improved vHIT gain of 0.88 (right ear 0.92). The second patient had an initial visit to the outpatient facility during an attack of vertigo, showing a slight reduction in the left lateral canal and vHIT gain (0.83) as compared to the right ear (0.91), and a significant reduction in the left anterior canal gain (0.48) as compared to the right ear (0.91). Ictal home VOG conducted 17 months earlier captured spontaneous left-beating nystagmus with SPV of 13.9°/s. Repeat testing was not conducted. During admission to the ED for acute vertigo, a third patient displayed spontaneous right-beating nystagmus with SPV of 5.9°/s, and received vHIT on the same day, which demonstrated slightly reduced right lateral canal gains (0.83) that increased to 0.96 7 days later (left ear gains were 1.04 and 0.92 respectively).
Vestibular-evoked myogenic potentials
At least one VEMP test was conducted in 95.3% of patients with ESV, 85.7% with EPV, and 44.4% with AVS. AC and BC cVEMP reflexes were conducted in 86 and 76 patients, and were present in 94.2% and 98.7%, with mean corrected reflex amplitudes of 1.6 ± 0.7 and 1.7 ± 0.8 respectively (Table 6). To AC and BC stimuli, asymmetry ratios were within normal limits in 88.4% and 93.4% of patients. AC cVEMP corrected reflex amplitudes, and p13 and n23 peak latencies showed no significant difference to those of controls (p = 0.466 and 0.330). BC cVEMP amplitudes were significantly smaller in VM patients compared with those of controls (p = 0.047, CI: 0.003–0.512), with no significant difference in peak latencies (p = 0.266).
Vestibular evoked myogenic potential results in patients with vestibular migraine compared with those of normal controls.

Note: cVEMP amplitudes are presented as a corrected amplitude calculated upon baseline electromyogenic activity (this ratio has no unit value). Statistical comparison between results of healthy controls was calculated controlling for age. p-values with an asterisk (*) indicate a significant difference from those of healthy controls (BC cVEMP amplitude: 95% confidence interval 0.003–0.512).
AC and BC oVEMP reflexes were conducted in 65 and 71 patients, and were present in 83.1% and 100%, with mean n10 peak latencies of 8.7 ± 0.5 ms and 9.2 ±0.5 ms, and peak-to-peak reflex amplitudes of 9.2 ± 6.4 µV and 20.3 ± 12.8 µV, respectively. Asymmetry ratios were within the normal limits in 67.7% and 97.2% of patients, and bilaterally absent reflexes were observed in 16.9% and 0.0% (to AC and BC stimuli respectively). Four ears (in three patients) displayed AC oVEMP amplitudes enlarged beyond the normal limits (range 24.0–35.8 µV). Twelve ears (in eight patients) displayed enlarged BC oVEMP reflex amplitudes beyond the normal limits (range 42.6–79.8 µV). These patients were investigated for a third mobile window syndrome; however, no radiological evidence of superior semicircular canal dehiscence or large vestibular aqueduct syndrome was found. Overall, there was no significant difference in any mean oVEMP parameter compared with the results of normal controls.
Electrocochleography
Due to reports of unilateral tinnitus, aural fullness and/or subjective hearing loss or fluctuation, transtympanic electrocochleography to test for endolymphatic hydrops was conducted for eight patients (15 ears tested). Mean SP/AP ratios for AC click stimuli was 16.9%. Mean SP values to 0.5, 1, 2 and 8 kHz tone-bursts were 1.1 ± 1.9, 1.1 ± 1.7, 0.8 ± 1.6, and 1.2 ± 0.8 µV. There were no recorded summating potentials < −6.0 µV to 1 kHz tone bursts, which is consistent with normal results (i.e. no evidence of endolymphatic hydrops) (17).
Vestibular test characteristics by syndrome
In the nine patients presenting to the hospital emergency department with AVS, only four displayed spontaneous nystagmus, all horizontal, with a mean SPV of 5.4°/s. Eight of these patients had interictal lateral vHITs conducted, and one patient additionally had an ictal vHIT conducted that showed the right ear to have lower gain (0.83) with refixation saccades, compared to the normal left ear (1.04 gain). Seven patients demonstrated interictal lateral vHIT gains that were normal, while one demonstrated reduced gains in the left ear (0.69), with normal gains in the right ear (0.79). Five AVS patients had BC VEMPs conducted, the results of which were all within normal limits. HINTS testing (head-impulse, nystagmus, test of skew) indicated a central pattern in four patients, a peripheral pattern in two, and was not applicable in the remaining four, because they were asymptomatic at the time of assessment. All AVS patients all underwent MRI, which showed no infarcts, and were followed up in the outpatient clinic. A final diagnosis of VM was made after subsequent presentations with episodic spontaneous vertigo.
In 85 patients with ESV, 71.8% showed ictal spontaneous nystagmus. Four outliers displayed significantly higher velocity nystagmus > 25°/s (range 28.7–57.4°/s), and excluding these four and those without nystagmus, the mean SPV for this group was 5.5°/s. In this group, asymmetric AC cVEMP and BC oVEMPs were found in only 5.7 and 3.2%. While 11.5% had a caloric asymmetry, only one patient had reduced lateral vHIT gains. Sixty-two ESV patients (72.9%) underwent MRI with normal results or minor findings not relevant to vestibular symptoms.
In seven patients reporting EPV, all displayed mild spontaneous nystagmus (mean SPV 4.0°/s), and all displayed persistent positional nystagmus in all three lying positions, which continued until the end of the recording. Two patients demonstrated vertical nystagmus in all lying positions (one down-beating, one up-beating) while the rest showed horizontal nystagmus with either ear down (two geotropic, two left-beating, and one apogeotropic) with a mean SPV 12.3°/s. Nystagmus SPV fell between 20–30°/s in four patients, 10–20°/s in one, and 5–10°/s in two. The mean time spent in each lying position was 32.2 sec. None displayed paroxysmal nystagmus. All seven EPV patients underwent MRI with normal results or minor findings not relevant to vestibular symptoms.
Treatment
A subset of patients (50.5%, n = 51) were prescribed emergency medications to be taken for acute symptomatic relief, including cinnarizine (35.6%, n = 36), ondansetron (25.7%, n = 26), and prochlorperazine (18.8%, n = 19). Migraine preventative therapies were trialled in 79.2% (n = 80) of patients, with 20.8% (n = 21) trialling more than three preventative migraine medications. Nutraceutical (including vitamin B2 and magnesium) were offered to 48.5% (n = 49) as a first-line therapy, with 19.8% (n = 20) not requiring further medications. Other commonly prescribed VM preventative medications included pizotifen in 24.8% (n = 25), dothiepin in 18.8% (n = 19), and topiramate in 17.8% (n = 18) (Table 7).
Medications trialled in patients with vestibular migraine.

PRN: as needed (pro re nata).
Discussion
In the present study, we describe the characteristics of vestibular migraine presenting as three distinct syndromes: Acute vestibular syndrome (AVS), episodic spontaneous vertigo (ESV), and episodic positional vertigo (EPV), of which ESV and EPV were most frequent. Overall, most patients demonstrated low amplitude spontaneous nystagmus and/or positional nystagmus while experiencing vertigo (18). Most patients also demonstrated normal and symmetrical interictal audiometric, canal, and otolith vestibular function tests. In patients with AVS, some demonstrated “central” HINTS assessments and were investigated for stroke, and subsequently presented with episodic vertigo before receiving a final diagnosis of VM. In ESV, the history, complemented by normal audiometry assisted the final diagnosis. In isolated EPV, the history, central positional nystagmus and normal imaging helped diagnose VM.
Comparison with earlier studies
Our observations are similar to those of other studies, which have shown that while subjective auditory symptoms such as aural fullness, tinnitus and hearing loss may be reported in a subset of patients with VM, audiovestibular test results are generally normal and thus may be useful when seeking to exclude other pathologies such as Menière’s disease (19,20). Other studies have also shown that, rarely, those with VM may show reduced vHIT responses when ictal, indicating an acute peripheral vestibular loss (10). While our study demonstrated three patients with ictally reduced vHIT gains, this may not be generalisable to the broader VM population, as these patients were being investigated for an acute vestibular syndrome at the time of testing while patients with known VM do not routinely have ictal vHIT testing conducted.
Our observations differ from some studies which found that between 11–68% of those with VM had reduced AC cVEMP amplitudes with a statistically significant difference when compared with normal controls, whereas our group displayed normal mean AC cVEMP amplitudes, latencies, and asymmetry ratios (22,42). However, our standard AC VEMP protocol uses a broadband click stimulus delivered at 105 dBnHL, whereas other studies used a 400 or 500 Hz tone-pip of between 90–100 dBnHL, which may yield different results.
Horizontal canal dysfunction in VM
Unilateral caloric weakness is a surprisingly common finding in patients with VM, with reported rates of abnormality ranging between 8–22% in other studies, and 15.8% in ours (21–24). This may be due to observations that a portion (31.8%) of migraine patients show a hyperresponsive caloric response pattern (25), which may be hypothesised to increase detection of a relative canal paresis or asymmetric response. Lateral vHIT results demonstrated normal gains in most patients, with a small subset (9–11% in other studies, 2.4% in ours) showing reductions (22,23).
Ictal and interictal nystagmus in VM and healthy controls
Previous investigators also demonstrated a high prevalence of spontaneous ictal nystagmus (70%) comparable with our study (71.3%) (29). Persistent positional nystagmus while symptomatic was found in 100% of patients in two separate studies, comparable with our findings in 97.3% of patients (30,31). Occurrence of interictal spontaneous nystagmus in earlier reports of VM range between 2–11% (14.9% in our cohort) (21,32). A high prevalence of interictal positional nystagmus has also been reported (28–57.9% to our 55.4%) (32). Another observation made by prior investigators and replicated in our study is that despite the presence of moderate interictal spontaneous or positional nystagmus, there were no subjective complaints of vertigo in some VM patients during interictal assessment (21). Our VM patients exhibited interictal nystagmus at rates similar to studies of normal controls without vertigo. Some studies report a high prevalence of spontaneous nystagmus while sitting upright without visual fixation ranging between 20–40% of healthy controls (33–37), while other studies cite a lower rate of 5–6.6% (38–40). In the present study, the prevalence of interictal spontaneous nystagmus was 14.9%. The occurrence of positional nystagmus in healthy controls is reported to be between 22.5–88% (33–38,40,41); the prevalence of interictal positional nystagmus in VM was 55.4%.
Outliers with high velocity nystagmus
The maximum SPV of spontaneous ictal nystagmus observed in this study (57.4°/s) was somewhat faster than other reports (33.3°/s), which may reflect the ability of our patients to record their eye movements closer to symptom onset while at home, as well as larger patient numbers (29,31). While one study found the average lateral lying positional nystagmus SPV in symptomatic VM patients to be 12.7–19.4°/s, which is faster than our values (mean combined positional SPV 10.4°/s), these differences may reflect the smaller sample size and the inclusion of only patients with positional vertigo (43). While our study included all positional results regardless of SPV, the other study included only visible positional nystagmus (43). This may also explain the higher incidence of interictal positional nystagmus in our study (55.4%), compared to other studies (11%) (21).
Anxiety in VM
The occurrence of anxiety in 21.8% of this study group replicates the observations of earlier investigators; one group showed that those with VM are significantly more likely to show agoraphobia, separation anxiety, illness phobia, and reassurance seeking compared with normal controls without migraine or vertigo (26). Due to the prevalence of unpredictable and disabling vertigo in this group, it has been hypothesised that VM may enhance anxiety and/or trigger panic and spatial phobias with associated avoidance behaviours (27,28).
Study limitations
Our study limitations were that not all patients received all tests. Clinicians classifying the study patients were not blinded to their ictal nystagmus results or audiovestibular test results, and therefore their diagnoses may have been biased. While all patients within the study received treatments and clinical follow-up based on their individual needs, there was no formal assessment of treatment responses, such as questionnaires or self-rated improvement over time. As this was a single-centre study, results may not be generalizable to a wider population. The video recordings were all conducted with fixation denied (i.e. in the dark), and therefore the effects of fixation are unknown. The effect of gaze direction was not investigated.
Future research should examine the effects of pharmacological treatments on recurrent and/or chronic vertigo (44) and both ictal and interictal nystagmus, the characteristics of audiovestibular tests conducted on the ictal dizzy patient, and the prevalence of vestibular asymmetry as a function of disease duration.
Conclusion
Patients with VM can present as an acute vestibular syndrome, or with recurrent spontaneous or positional vertigo. Most patients with VM show normal interictal test results, including symmetrical hearing thresholds, normal caloric, vHIT, and VEMP results. Spontaneous ictal nystagmus occurs in most patients and is significantly enhanced in the supine and lateral positions, which may explain the large subset of patients who report significant positional vertigo in the absence of benign paroxysmal positional vertigo. Between attacks, most patients with VM either display no nystagmus, or very low velocity nystagmus.
Key findings
Most patients with vestibular migraine show spontaneous nystagmus during vertigo attacks. A small portion of patients with vestibular migraine show asymmetric caloric test results. Patients with vestibular migraine typically have normal lateral video head impulse test results, interictally.
Footnotes
Author contributions
AY drafted the manuscript, collected and analysed patient data, created all original figures, and approved the final version of the manuscript. AB designed the video analysis software, created and edited the videos, critically revised and approved the final version of the manuscript. ZC, BN, and JP collected and analysed patient data, critically revised and approved the final version of the manuscript. MD’S advised on the statistical analysis, critically revised and approved the final version of the manuscript. GMH consulted with and recruited research patients, critically revised and approved the final version of the manuscript. MW designed the study, consulted with and recruited research patients, critically revised and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AY receives scholarship funding from the University of Sydney, and reports no conflict of interest. JP receives funding from the Garnett Passe and Rodney Williams Memorial Foundation, and reports no conflict of interest. AB, ZC, and MD’S report no conflicts of interest. GMH is an unpaid consultant to Natus Otometrics, and reports no conflicts of interest. MW receives funding from the Garnett Passe and Rodney Williams Memorial Foundation, and the National Health and Medical Research Council of Australia, and reports no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Garnett Passe and Rodney Williams Memorial Foundation [grant number RP554]; and the National Health and Medical Research Council of Australia [grant number APP1126976]. These funding sources have no role in any aspect of the study or the decision to submit for publication.
