Abstract
Background
Vestibular symptoms are a frequent and disabling manifestation of migraine, yet their underlying pathophysiology remains incompletely understood. Although subtle vestibular abnormalities have been described, their clinical relevance and relationship with dizziness-related disability in migraine are still unclear.
Methods
In this cross-sectional study, consecutive participants with episodic migraine (EM) or chronic migraine (CM) and vestibular symptoms, as well as healthy controls (HC), were recruited. All subjects underwent a standardized bedside vestibular examination and video head impulse testing (vHIT). Vestibulo-ocular reflex (VOR) gain was measured bilaterally, and gain asymmetry was calculated as a continuous variable. Vestibular-related disability was assessed using the Dizziness Handicap Inventory (DHI). Between-group comparisons were performed, and correlations between vHIT asymmetry and DHI scores were explored within the migraine group.
Results
A total of 101 participants were included (30 HC, 30 EM, and 41 CM). Median vHIT gain asymmetry was comparable between groups (HC: 3.75% [IQR 2.58–5.70]; EM: 3.26% [2.37–5.57]; CM: 3.16% [1.35–5.05]; p = 0.514), with no significant differences in VOR gain. Within the migraine population, greater vHIT asymmetry was weakly but significantly associated with higher dizziness-related disability (Spearman's ρ = 0.274, p = 0.021), despite asymmetry values remaining within normative ranges.
Conclusions
Peripheral vestibular function, as assessed by vHIT, appears preserved in migraine. However, subtle vestibular asymmetry which is clinically silent in healthy individuals may contribute to dizziness-related disability in migraine, likely reflecting altered central sensory processing rather than peripheral vestibular dysfunction.
Keywords
Introduction
Vestibular symptoms are highly prevalent in individuals with migraine, affecting up to half of patients at some point during the course of the disease.1–3 Vestibular migraine represents one of the most common causes of episodic vertigo 4 ; however, dizziness and vertigo are also frequently reported by patients who do not meet diagnostic criteria for vestibular migraine. These symptoms contribute substantially to functional impairment and reduced quality of life, which can be captured using patient-reported outcome measures such as the Dizziness Handicap Inventory (DHI).5,6 Clinical manifestations range from rotational vertigo and positional dizziness to persistent unsteadiness and visual dependence, and may be present not only during attacks but also in interictal periods.7,8 Notably, accumulating evidence suggests that the burden of vestibular symptoms is closely linked to interictal sensory hypersensitivity: patients exhibiting a greater number of hypersensitive sensory modalities show a higher likelihood of experiencing vestibular symptoms and a greater degree of dizziness-related disability. 9
Understanding the origin of vestibular symptoms in migraine remains challenging. Such symptoms may reflect an intrinsic expression of the migraine disorder itself or arise from concomitant peripheral or central vestibular dysfunction. Peripheral mechanisms include altered trigeminovascular input to vestibular nuclei and brainstem circuits,9–11 whereas central mechanisms involve dysfunctional sensory integration within thalamo-cortical and cerebellar networks.12–15 These alterations have been demonstrated both during vestibular stimulation 16 and migraine attacks. 16 Consistent with this model, functional studies have shown that patients with migraine—particularly those with aura or chronic migraine (CM)—exhibit impaired postural control under conditions of sensory conflict, despite largely normal findings on routine bedside vestibular examination.17,18
A growing body of evidence implicates calcitonin gene-related peptide (CGRP) as a potential mediator of vestibular symptoms in migraine. CGRP plays a central role in migraine pathophysiology and is elevated both during attacks and interictally.19–21 CGRP-expressing neurons and receptors have been identified in vestibular-related regions of the brainstem and cerebellum,22–25 as well as in efferent terminals innervating type I hair cells of the vestibular end organs 26 where they may modulate afferent sensitivity. 27 In animal models, CGRP administration increases postural sway and alters vestibular responses, whereas CGRP-deficient mice show reduced VOR gain.27–29 In humans, systemic CGRP infusion can induce dizziness or vestibular symptoms in up to one-third of patients with migraine.30,31 Notably, patients with migraine exhibit higher circulating CGRP levels than healthy controls (HC),32,33 supporting the hypothesis that migraine-related CGRP activity may induce modulation of vestibular function rather than frank bilateral deficits.
Objective vestibular testing has yielded heterogeneous results. Video head impulse test (vHIT) studies have reported overt abnormalities in a subset of migraine patients, especially in those with prominent vestibular symptoms.33–35 In contrast, other investigations describe only subtle or subclinical deviations, including mildly increased vestibulo-ocular reflex (VOR) gains or inter-aural differences that remain within conventional normal ranges.36–39 These inconsistencies suggest that standard measures of absolute gain may not fully capture migraine-related vestibular dysfunction.
In this context, VOR asymmetry assessed by vHIT may represent a sensitive marker of migraine-related vestibular involvement. Previous work has shown that reductions in vHIT asymmetry are associated with lower dizziness-related disability, 40 supporting an association between VOR imbalance and the subjective burden of vestibular symptoms.
Therefore, the aim of the present cross-sectional study was to compare vHIT gain asymmetry between individuals with migraine and HC, and to evaluate whether such asymmetry is associated with the severity of vestibular symptoms as measured by the DHI. We hypothesized that patients with migraine would exhibit greater interictal vHIT asymmetry and that inter-aural vHIT asymmetry would be associated with dizziness-related disability.
Methods
Design and setting
This was a cross-sectional study conducted at a tertiary referral headache center between May 2022 and May 2025. This report follows the recommendations of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Participants
Participants were consecutively enrolled. The migraine group included individuals aged 18–65 years with a diagnosis of migraine, with or without aura, and in episodic migraine (EM) or chronic type (CM), according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) 41 ; and the presence of vestibular symptoms occurring during migraine attacks or in interictal periods, regardless of whether formal diagnostic criteria for vestibular migraine were fulfilled. Accordingly, the study population represented a broad continuum of vestibular involvement in migraine, ranging from mild symptoms to established vestibular migraine.
HC were recruited from the hospital clinical staff, with no personal history of migraine or other primary headache disorders, no first-degree family history of migraine, and no history of vestibular symptoms.
Participants were excluded if they had a known primary peripheral or central vestibular disorder, another primary headache diagnosis, or were receiving medications at baseline that could interfere with vestibular function (including antihistamines, benzodiazepines, anticholinergic agents, antiemetics, aminopyridines, betahistine, or anticonvulsants). Pregnancy was also considered an exclusion criterion. All participants provided written informed consent prior to inclusion.
Variables
Demographic and headache-related variables
Demographic variables included age and sex. Headache-related variables comprised migraine subtype (episodic or chronic; with or without aura; vestibular migraine and probable vestibular migraine when applicable, according to ICHD-3 criteria 41 and the consensus document of the Bárány Society and the International Headache Society), 41 age at migraine onset, and monthly headache days (MHDs), defined as any calendar day on which the participant reported headache in a paper diary. Headache-related impact was assessed using the Spanish-validated version of the Headache Impact Test (HIT-6). 42 Higher scores indicate greater headache-related disability.
Vestibular symptom characterization
Vestibular symptoms were systematically classified as vertigo or dizziness according to international consensus definitions. 43 Vertigo was defined as a sensation of self-motion of the head or body in the absence of actual movement, or as a distorted perception of self-motion during normal head movements. Dizziness was defined as a sensation of impaired or disturbed spatial orientation without a false or distorted sense of motion, excluding vertigo. This operational definition was used for symptom characterization and does not necessarily correspond to the qualifying vestibular symptom categories defined in the Bárány Society / ICHD-3 criteria for vestibular migraine. 41 The temporal relationship between vestibular symptoms and migraine attacks was categorized as occurring before, during, after, or independently of migraine attacks.
Vestibular disability
Vestibular-related disability was assessed using the Spanish-validated DHI. 44 This 25-item questionnaire provides a total score ranging from 0 to 100, with higher scores indicating greater disability, and includes three subscales: physical (7 items; maximum 28 points), functional (9 items; maximum 36 points), and emotional (9 items; maximum 36 points). The DHI total score was considered the primary patient-reported outcome reflecting vestibular symptom burden.
Bedside vestibular examination variables
A standardized bedside vestibular examination was performed in all participants. The presence of spontaneous nystagmus was assessed in primary gaze using Frenzel or infrared goggles to suppress visual fixation. Oculomotor function was evaluated through assessment of saccades and smooth pursuit.
Dynamic visual acuity (DVA) was assessed by comparing static visual acuity, measured with a standardized Snellen chart, with visual acuity during passive horizontal head oscillations (approximately 10–15° amplitude at 2 Hz). DVA decrement was calculated as the loss in logMAR units relative to static acuity; a decrement ≥0.2 logMAR was considered abnormal. 45
Postural stability was evaluated using the Romberg test performed on a firm surface with eyes open and eyes closed. Failure of the eyes-closed condition or postural sway graded ≥2 on a 0 to 3 scale was considered abnormal. 46 The Unterberger (Fukuda) stepping test was performed with eyes closed for 50 steps; rotation >45° or inability to complete the test without assistance was classified as abnormal. 47 Cerebellar function was screened using finger-to-nose and heel-to-shin testing. The Dix–Hallpike maneuver was performed to exclude benign paroxysmal positional vertigo.
Instrumented vestibular variables
VOR function was assessed using the vHIT with the ICS Impulse® system (Otometrics, Natus Medical Inc., Denmark). Horizontal vHIT quantified semicircular-canal VOR gain. 48 Participants fixated on an earth-fixed optotype at 1.5 m while an examiner delivered unpredictable, passive head impulses (10–20°; peak 150–250°/s; acceleration >3000°/s2) with ∼20° neck flexion. At least 15 artifact-free impulses per side were required. Blinks, anticipatory movements, or goggle slippage led to trial exclusion and repetition. The device algorithm computed median VOR gain per side. Outcomes included the median VOR gain for each side, the bilateral average median gain ([left gain + right gain]/2), and a signed percentage gain asymmetry, calculated as [(left gain − right gain)/(left gain + right gain)] × 100%. The vHIT gain asymmetry was considered the primary objective vestibular variable.
This study did not include data linkage across multiple databases.
Endpoint
The primary endpoint was the vHIT gain asymmetry between individuals with migraine and HC, and its association with vestibular-related disability as measured by the DHI.
Statistical analysis
Sample size estimation was based on the primary outcome. Assuming a two-sided α level of 0.05 and 80% power, the required sample size was calculated for a between-group comparison using a standard deviation of 1.96%, derived from normative data in healthy adults. 49 Under these assumptions, the study was powered to detect a moderate between-group difference in asymmetry (Δ ≈ 1.5%).
Descriptive statistics were used to summarize baseline demographic and clinical characteristics. Continuous variables were reported as mean ± standard deviation (SD) or median with interquartile range (IQR) and range, depending on their distribution, while categorical variables were presented as absolute counts and percentages. Normality of continuous variables was assessed using the Kolmogorov–Smirnov test.
Between-group comparisons were performed using one-way ANOVA or the Kruskal–Wallis test for continuous variables, as appropriate. Categorical variables were compared using the Chi-squared test or Fisher's exact test. Pairwise comparisons for onset age and MHDs were restricted to migraine groups and performed using the Mann–Whitney U test. Associations between vestibular-related disability (DHI total score) and vestibular function measures were assessed using Spearman's rank correlation coefficient.
No missing data were observed; therefore, no imputation procedures were applied. All statistical tests were two-tailed, and a p-value < 0.05 was considered statistically significant. No formal correction for multiple comparisons was applied, as analyses of secondary outcomes and DHI subscales were considered exploratory. Accordingly, p-values derived from these comparisons should be interpreted with caution. Statistical analyses were conducted using SPSS Statistics (version 30.0, IBM Corp., Armonk, NY, USA).
Results
Sample characteristics
A total of 101 participants were included: 30 (29.7%) HC, 30 with EM (29.7%), and 41 with CM (40.6%) (Figure 1). The mean age was 37.9 years (SD 12.9), with no significant differences among groups (p = 0.136) (Table 1). Almost all participants were women (99.0%). No HC reported vestibular symptoms, whereas all participants with migraine did, as this was an inclusion criterion. The median age at migraine onset was 14 years (IQR 11–20), with no significant difference between EM (14.5 [IQR 11.5–21.7]) and CM (14 [IQR 11–18]; p = 0.439). Vestibular migraine was diagnosed in 71.8% of participants with migraine (EM 24 [80.0%]; CM 27 [65.9%]), whereas the remaining 28.2% fulfilled criteria for probable vestibular migraine (EM: 6 [20.0%]; CM: 14 [34.1%]; p = 0.286). Interictal photophobia was observed in 33 participants (32.7%) overall and was more frequent in CM (22 [53.7%]) than in EM (11 [36.7%]; p < 0.001). The median number of MHDs was 8 (IQR 5.5–12) in EM and 30 (IQR 17.5–30) in CM (p < 0.001). Headache-related disability, assessed using the HIT-6, was comparable between EM (median 66 [IQR 63–70.5]) and CM (median 67 [IQR 65.5–72.0]; p = 0.147). At the time of analysis, 11 participants (15.5%) were receiving preventive treatment, predominantly among those with CM (10 [24.4%]) compared with EM (1 [3.3%]). The preventive agents used were topiramate (4 [5.6%]), flunarizine (3 [4.2%]), zonisamide (2 [2.8%]), lisinopril (1 [1.4%]), and amitriptyline (1 [1.4%]).
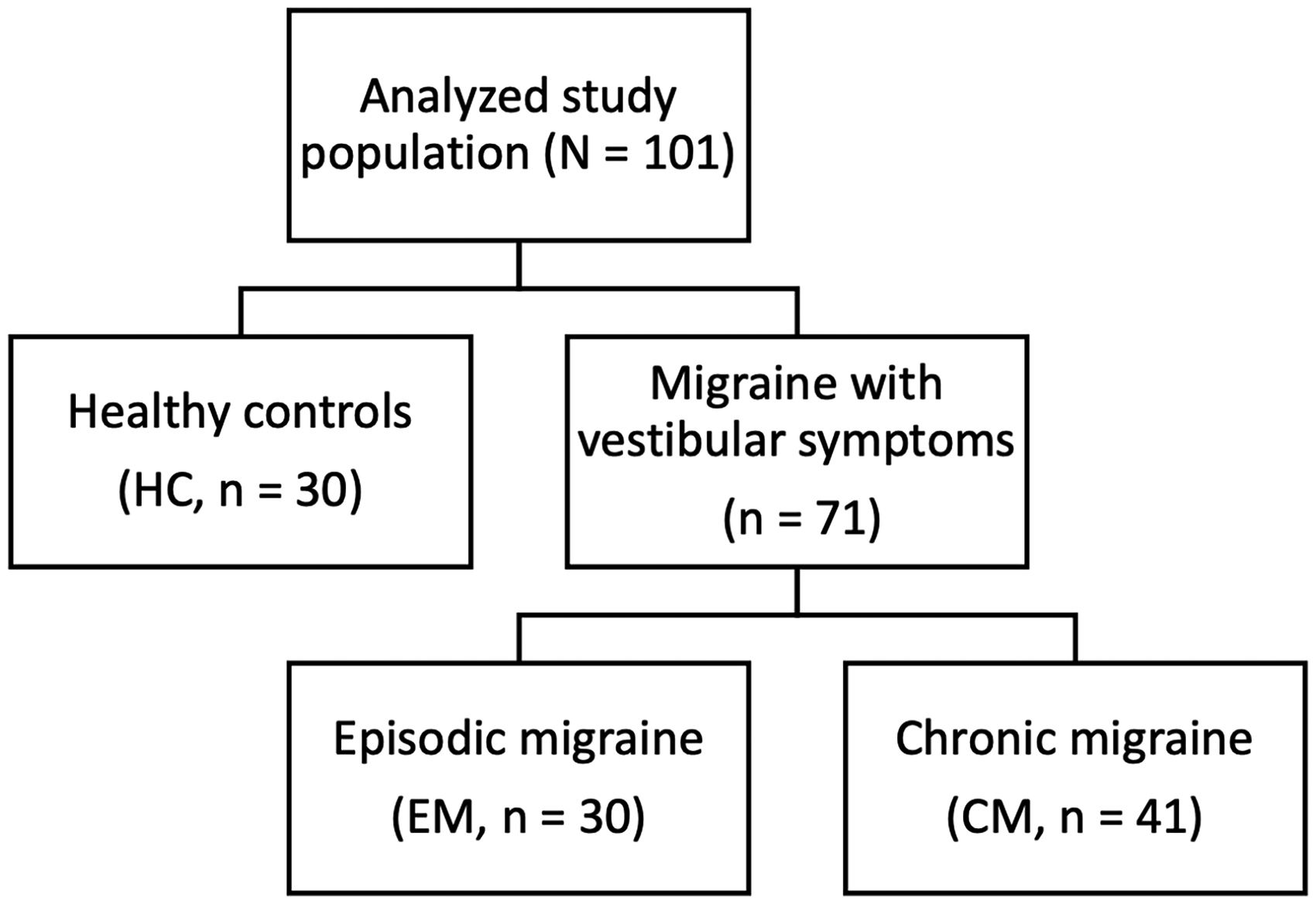
Study population.
Demographic and clinical characteristics of the study population.

Comparisons for age at migraine onset, vestibular migraine, interictal photophobia, monthly headache days, HIT-6 were restricted to participants with migraine.
Abbreviations: HC = healthy controls; EM = episodic migraine; CM = chronic migraine; IQR = interquartile range; HIT-6 = Headache Impact Test-6.
Vestibular symptom characteristics
Among participants with migraine and vestibular symptoms (n = 71), dizziness was the most frequently reported manifestation, present in 59 subjects (83.1%), whereas vertigo was reported by 37 participants (52.1%) (Table 2). Dizziness was more commonly reported in EM (27/30, 90.0%) than in CM (32/41, 78.0%). Vertigo was reported by 19 patients with EM (63.3%) and 18 patients with CM (43.9%).
Characteristics and temporal relationship of vestibular symptoms in participants with migraine.

Abbreviations: EM = episodic migraine; CM = chronic migraine.
Regarding the temporal relationship with migraine attacks, vestibular symptoms showed substantial variability. Dizziness most frequently occurred during migraine attacks (45/71, 63.4%), although a relevant proportion of patients reported symptoms independent of migraine attacks (25/71, 35.2%). Similarly, vertigo was most commonly reported during migraine attacks (23/71, 32.4%), but interictal vertigo was also frequent, occurring independently of migraine attacks in 16 patients (22.5%). Vestibular symptoms occurring exclusively before or after migraine attacks were less common in both dizziness and vertigo presentations.
Vestibular examination
No participants exhibited spontaneous nystagmus, oculomotor abnormalities, cerebellar signs, or positional vertigo on Dix–Hallpike testing. Pathological DVA was observed in 44 participants (43.6%), with no significant differences between groups (HC: 12 [40.0%]; EM: 12 [40.0%]; CM: 20 [48.8%]; p = 0.683). The Romberg test was normal in all participants. Abnormal results on the Unterberger test were observed in 46 individuals (45.5%), with a lower proportion in HC (8 [26.7%]) compared with EM (14 [46.7%]) and CM (24 [58.5%]) groups (p = 0.028).
DHI
Mean total DHI scores (DHI-T) were significantly lower in EM than in CM (36.4 ± 20.0 vs. 44.2 ± 23.8, respectively; p < 0.001) (Table 3). This difference was consistent across all subscales, including the emotional domain (DHI-E: 12.5 ± 7.8 vs. 15.3 ± 9.3; p < 0.001), the functional domain (DHI-F: 13.1 ± 7.2 vs. 15.0 ± 9.9; p < 0.001), and the physical domain (DHI-P: 10.9 ± 7.4 vs. 13.9 ± 6.4; p < 0.001).
Dizziness Handicap Inventory (DHI) scores in participants with migraine.
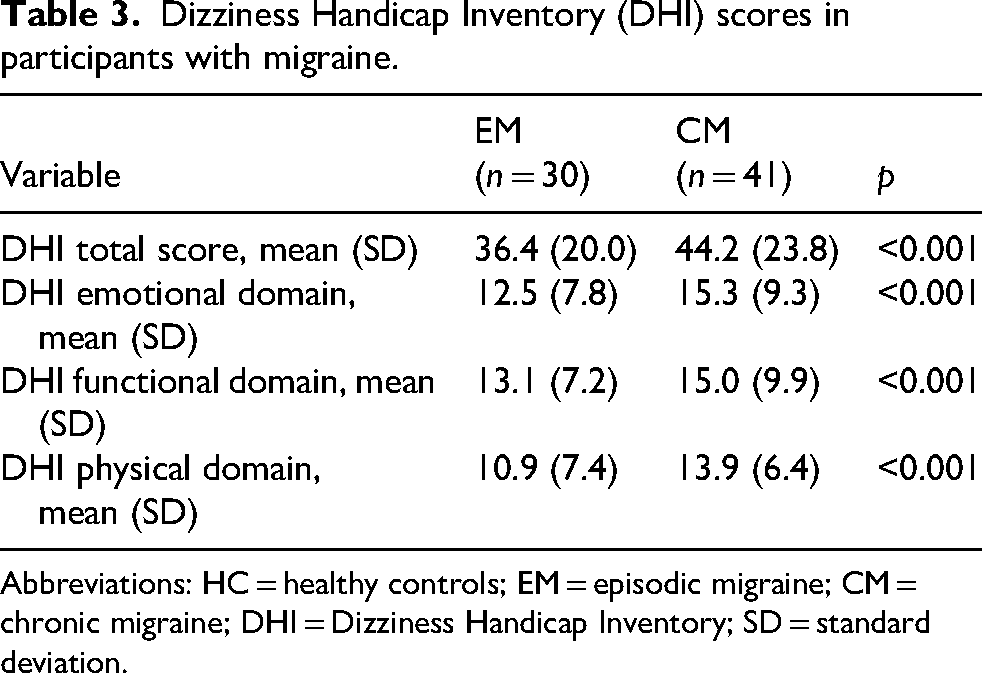
Abbreviations: HC = healthy controls; EM = episodic migraine; CM = chronic migraine; DHI = Dizziness Handicap Inventory; SD = standard deviation.
VHIT
There were no significant differences in vHIT measures between groups (Table 4). Median gains were 0.89 [IQR 0.87–0.94] for the left side (HC 0.89 [IQR 0.87–0.92], EM 0.89 [IQR 0.88–0.96], CM 0.90 [IQR 0.86–0.93]; p = 0.623) and 0.96 [IQR 0.93–1.00] for the right (HC 0.95 [IQR 0.92–1.00], EM 0.96 [IQR 0.95–1.03], CM 0.95 [IQR 0.91–0.99]; p = 0.154). The average median gain was 0.93 (IQR 0.90–0.96), with no differences between groups (HC 0.92 [IQR 0.90–0.97], EM 0.93 [IQR 0.92–0.99], CM 0.92 [IQR 0.89–0.96]; p = 0.174) (Figure 2). Median asymmetry in the total sample was 3.26% (IQR 2.22–5.27), with no differences between groups (HC 3.75 [IQR 2.58–5.70], EM 3.26 [IQR 2.37–5.57], CM 3.16 [IQR 1.35–5.05]; p = 0.514) (Figure 3).
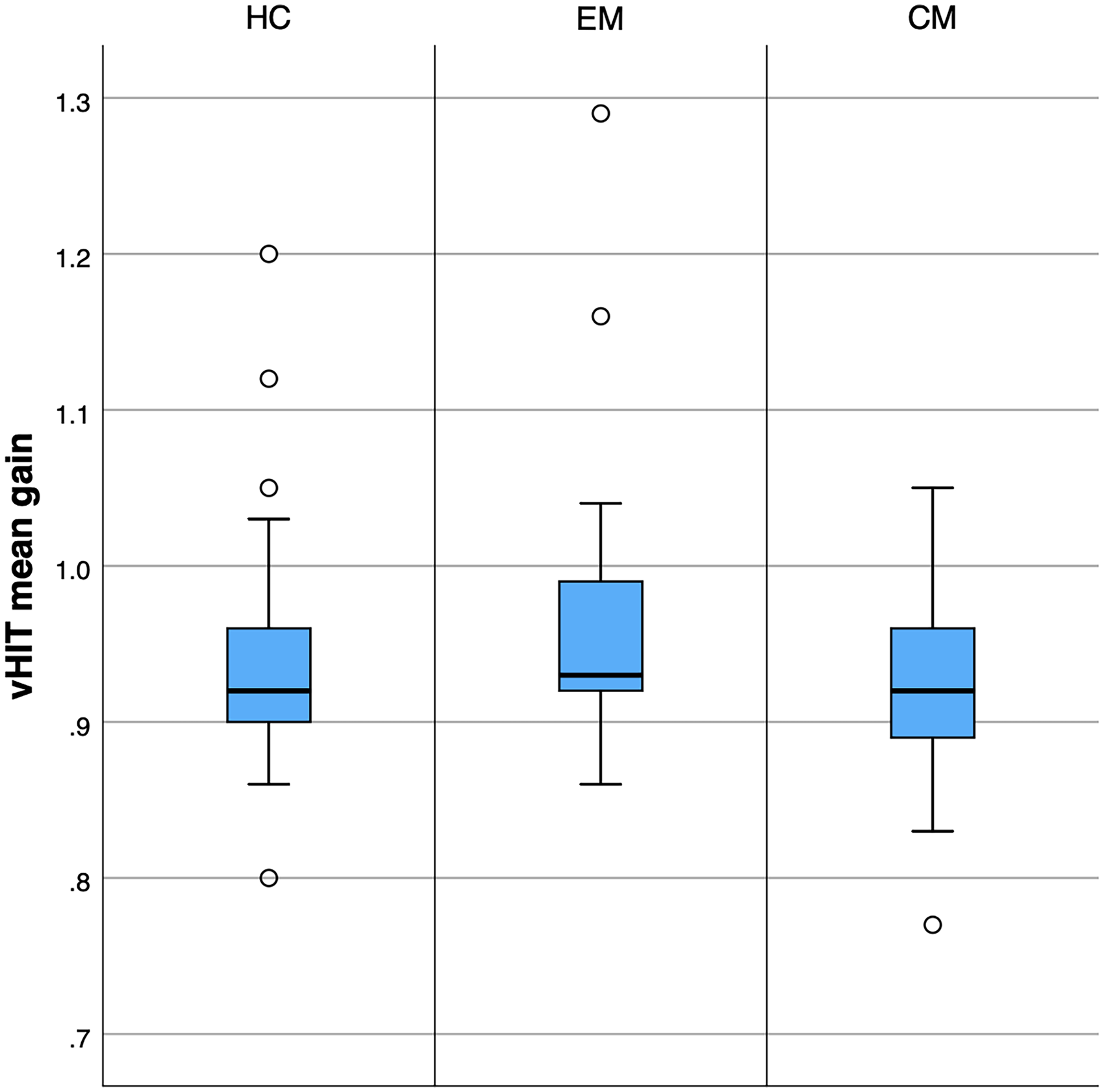
Mean vestibulo-ocular reflex (VOR) gain assessed by video head impulse test (vHIT) across groups.

Video head impulse test (vHIT) gain asymmetry across study groups.
Video head impulse test (vHIT) results.
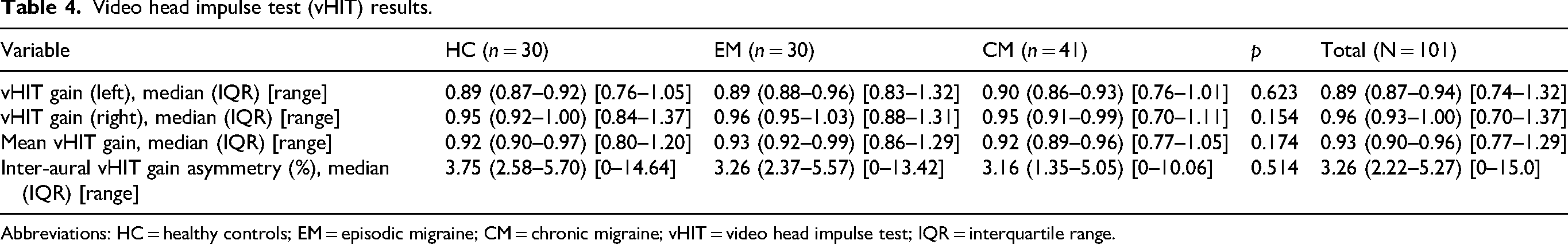
Abbreviations: HC = healthy controls; EM = episodic migraine; CM = chronic migraine; vHIT = video head impulse test; IQR = interquartile range.
Correlation analysis
Spearman's correlation analyses restricted to participants with migraine showed a weak but statistically significant positive association between mean DHI-T and vHIT gain asymmetry (ρ = 0.274, p = 0.021) (Figure 4).
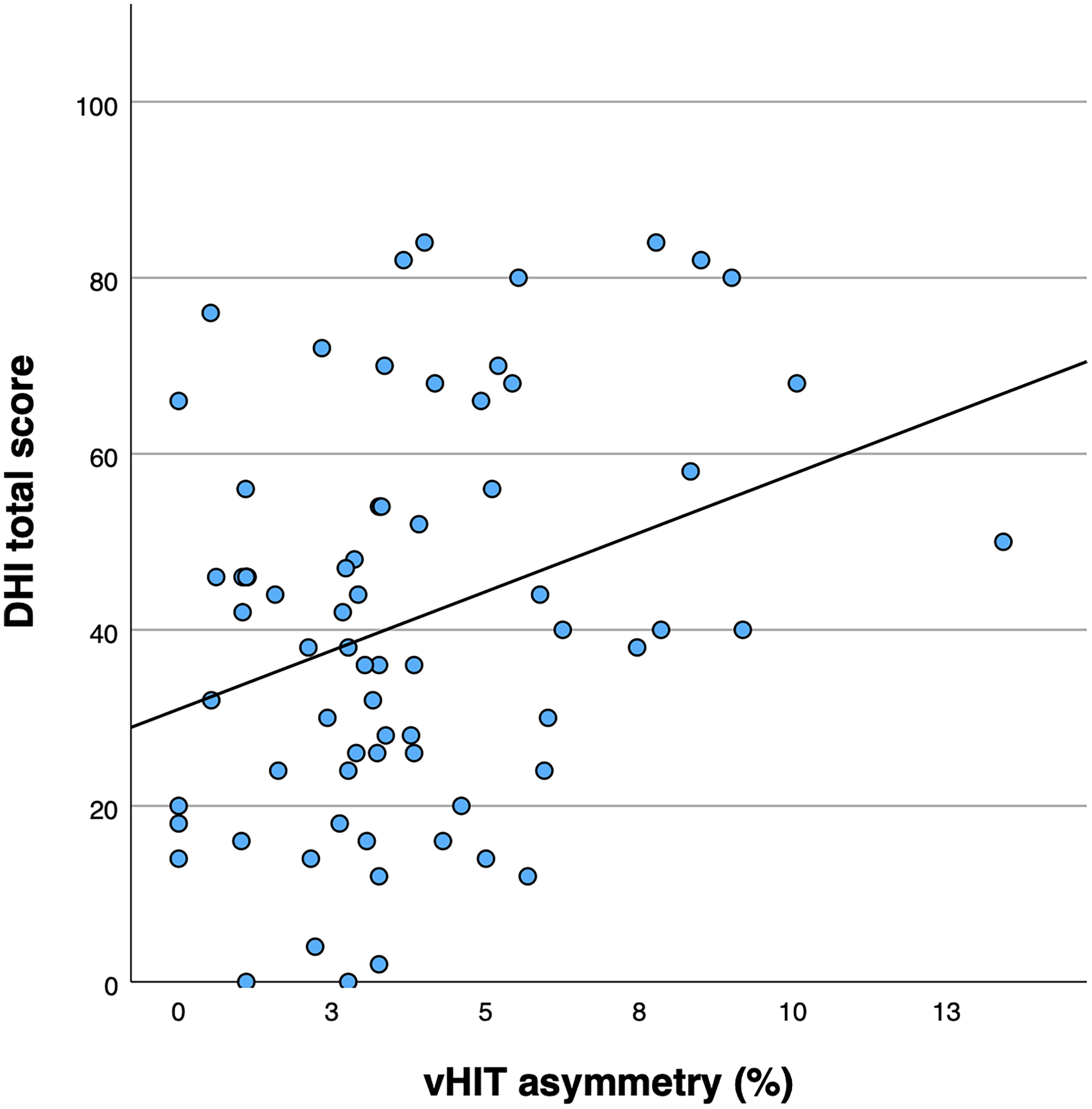
Association between video head impulse test (vHIT) gain asymmetry and dizziness-related disability in migraine.
Discussion
In this cross-sectional study, we found that patients with migraine did not differ from HC in VOR gain asymmetry as measured by vHIT, indicating preserved high-frequency horizontal peripheral vestibular function. However, within the migraine group, greater vHIT asymmetry was associated with higher dizziness-related disability, as reflected by total DHI scores. Taken together, these findings suggest that vestibular symptoms in migraine are not driven by overt semicircular canal dysfunction, but rather by an increased susceptibility to otherwise subtle vestibular imbalances.
The absence of differences in VOR gain between migraine patients and HC is consistent with the notion that migraine is not typically associated with frank vestibular hypofunction. In line with previous reports showing heterogeneous or subtle vHIT findings in migraine, our results support the idea that standard measures of absolute gain may not adequately capture migraine-related vestibular involvement. Importantly, the lack of between-group differences in vHIT asymmetry further argues against a primary vestibular deficit in migraine and highlights the need to consider alternative mechanisms underlying vestibular symptom generation.
A key finding of this study is the positive association between vHIT asymmetry and dizziness-related disability within the migraine group, despite asymmetry values remaining within normative ranges and being comparable to those observed in HC. Although this association explains only a modest proportion of the variance in DHI scores, it is noteworthy that vHIT asymmetry represents an objective, observer-independent measure of vestibular function. This observation suggests that small vestibular imbalances that are clinically silent in healthy individuals may become symptomatically relevant in subjects with migraine. Rather than reflecting vestibular dysfunction per se, this relationship likely reflects altered central processing of vestibular input and reduced tolerance to sensory mismatch.
Migraine is increasingly conceptualized as a disorder characterized by abnormal sensory processing and impaired multisensory integration. 50 Alterations in thalamo-cortical, cerebellar, and brainstem networks involved in vestibular and visual processing may amplify the perceptual impact of minor vestibular asymmetries, thereby contributing to dizziness, imbalance, and functional impairment.12–15 In this context, VOR asymmetry may reflect subtle vestibular imbalance that, in individuals with migraine, could be associated with increased symptom perception. Rather than indicating peripheral vestibular dysfunction, this relationship may be consistent with altered central sensory processing and reduced tolerance to sensory mismatch. However, given the cross-sectional design, these interpretations remain speculative and should be confirmed in longitudinal mechanistic studies.
CGRP provides a plausible biological substrate linking migraine pathophysiology and vestibular symptom susceptibility.19–21 CGRP is known to modulate sensory processing and is expressed in vestibular-related brainstem and cerebellar structures,22–25 as well as in vestibular efferent pathways. 26 Elevated CGRP levels in migraine may lower the threshold for vestibular symptom perception by enhancing central responsiveness to vestibular input, without inducing structural or functional damage to the peripheral vestibular apparatus. 31 This framework is consistent with experimental data showing CGRP-mediated modulation of vestibular responses27–29 and with clinical observations that CGRP infusion can induce dizziness in patients with migraine. 31
From a clinical perspective, our findings suggest that vHIT should not be interpreted in migraine as a diagnostic tool to identify peripheral vestibular deficits. Instead, vHIT asymmetry—when analyzed as a continuous variable—may serve as a marker of vulnerability to vestibular symptom burden in the context of altered central sensory processing. In line with this interpretation, in the ESPIRAL study 40 we previously observed that reductions in vHIT asymmetry following preventive migraine treatment were associated with improvements in dizziness-related disability, supporting the clinical relevance of asymmetry as a dynamic, modifiable marker rather than a static indicator of peripheral vestibular dysfunction. This distinction is clinically relevant, as it may help avoid misclassification of migraine-related vestibular symptoms as peripheral vestibulopathy and supports a more integrated, central interpretation of vestibular complaints in these patients.
This study has several strengths, including the use of a well-characterized migraine sample, the inclusion of HC, and the combined assessment of objective vestibular measures and patient-reported disability. The decision to analyze vHIT asymmetry as a continuous variable preserved information across the full range of values and avoided arbitrary dichotomization.
However, several limitations should be acknowledged. First, the cross-sectional design of the present study precludes causal inference regarding the relationship between vestibular asymmetry and symptom burden; therefore, prospective longitudinal studies are needed to confirm the temporal dynamics of this association across different migraine phenotypes. Second, participants were recruited from a tertiary headache center and were predominantly women with migraine and vestibular symptoms, which reflects clinical practice but may limit extrapolation to male patients or community-based populations. Moreover, all migraine participants reported vestibular symptoms, and therefore the results may not apply to individuals with migraine without vestibular complaints.
In addition, vestibular assessment was limited to horizontal semicircular canal function as measured by vHIT, which primarily evaluates high-frequency VOR responses. Anterior and posterior canal functions were not evaluated, and otolith organ function was not assessed. Therefore, subtle dysfunction affecting vertical canals, otolith pathways, or lower-frequency peripheral vestibular function cannot be excluded. Our conclusions regarding preserved vestibular function thus apply specifically to high-frequency horizontal VOR parameters rather than to the vestibular system as a whole.
Moreover, other vestibular assessments—such as caloric testing, vestibular-evoked myogenic potentials, or vestibular perceptual threshold testing—were not performed and could provide complementary information regarding frequency-specific canal function, otolith integrity, and central vestibular processing. Finally, both physiological and technical factors may have contributed to variability in vHIT measurements. Migraine-related alterations in attentional and oculomotor processing, which have been described even during interictal phases, 51 may introduce subtle variability in task performance despite standardized testing procedures. Likewise, vHIT outcomes are sensitive to factors such as head impulse velocity, calibration accuracy, and fixation stability, which were carefully controlled but cannot be entirely excluded as sources of residual variability.
Additionally, a minority of participants—predominantly those with CM—were receiving preventive treatment at the time of evaluation. Although the proportion was modest, preventive medications may influence central sensory processing and vestibular excitability, potentially affecting vHIT parameters, vestibular symptom burden, or dizziness-related disability. Finally, another aspect that should be considered when interpreting these findings is that the study was not restricted to patients with the diagnosis of definite vestibular migraine. While this may have reduced the likelihood of identifying overt pathological vestibular findings, it allowed the inclusion of a broader and clinically relevant spectrum of patients with migraine and vestibular symptoms. This approach enabled the exploration of subtle vestibular imbalances and their relationship with dizziness-related disability beyond established diagnostic categories, supporting the interpretation of vestibular symptoms in migraine as a dimensional rather than categorical phenomenon.
Conclusions
In this cross-sectional study, migraine was not associated with increased vHIT asymmetry compared with healthy controls, supporting preserved high-frequency horizontal vestibular function. Nevertheless, greater vHIT asymmetry—even within the normal range—was associated with higher dizziness-related disability among patients with migraine. This association should be interpreted cautiously and does not establish causality or mechanism; rather, it highlights a potential relationship that warrants further investigation in longitudinal studies.
Clinical implications
No differences in vHIT gain asymmetry were observed between participants with migraine and healthy controls, supporting preserved peripheral vestibular function.
vHIT gain asymmetry, even within normative ranges, was associated with greater dizziness-related disability in patients with migraine.
These findings support a central, integrative interpretation of vestibular symptoms in migraine, with implications for clinical assessment and patient counseling.
Footnotes
Acknowledgments
The authors would like to thank the patients and healthy volunteers for their participation in this study.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the local Ethics Committee.
Consent to participate
All participants provided written informed consent prior to inclusion.
Consent for publication
All authors have approved the submitted version and agree with its submission to Cephalalgia Reports.
Author contributions
Conceptualization was done by AJ, JPE.
Data curation and formal analysis were done by AJ.
Funding acquisition: JRV, JPE;
Investigation: AJ, JRV, OP, PVV, AG, JPE;
Methodology and software were done by AJ, JPE.
Project administration was done by AJ.
Resources did by AJ, JRV, AG, JPE.
Supervision and validation were done by JPE.
Visualization and writing—original draft were done by AJ.
Writing—review and editing was done by AJ, JRV, OP, PVV, AG, JPE.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AJ has received honoraria from Lilly, TEVA, Organon, Allergan-Abbvie and Lundbeck. JRV has received honoraria from Lilly, TEVA, Novartis, Allergan-Abbvie, and Exeltis, and research support from Allergan-Abbvie. AG has received honoraria from Organon. The remaining authors declare no conflicts of interest.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
