Abstract
Background
Vestibular migraine is among the most common causes of recurrent vertigo in the general population. Despite its prevalence and high impact on healthcare cost and utilization, it has remained an under-recognized condition with largely unknown pathophysiology. In the present article, we aim to provide an overview of the current understanding of vestibular migraine.
Methods
We undertook a narrative literature review on the epidemiology, presentations, clinical and laboratory findings, pathophysiology, and treatments of vestibular migraine.
Results
Currently, the diagnosis of vestibular migraine relies solely on clinical symptoms since clinical tests of vestibular function are typically normal, or difficult to interpret based on inconsistent results reported in earlier studies. The challenges related to diagnosis of vestibular migraine lie in its relatively broad spectrum of manifestations, the absence of typical migraine headaches with vestibular symptoms, and its very recent definition as a distinct entity. Here, we highlight these challenges, discuss common vestibular symptoms and clinical presentations in vestibular migraine, and review the current aspects of its clinical diagnosis and evaluation. The concepts related to the pathophysiology and treatment of vestibular migraine are also discussed.
Conclusion
Vestibular migraine is still underdiagnosed clinically. Future studies are needed to address the pathophysiological mechanisms and investigate effective treatment regimens.
Introduction
Clinicians are no strangers to patients with migraine symptoms and frequent dizziness. As a common neurological disorder, migraine affects about 15% of the general population (1). Dizziness is also a common symptom, accounting for up to 15% of visits in frontline healthcare settings (2,3). Considering the prevalence of both conditions, an overlap in the clinical presentations of vestibular symptoms and migraine may not seem surprising at first. Several studies, however, have found a close association between dizziness and migraine beyond coincidence (4–15). Dizziness is more common in migraineurs compared with those suffering from other headache subtypes such as tension-type headache, suggesting a pathological link between migraine and dizziness (9,16,17). In some migraine patients, dizziness or vertigo is even more prominent and debilitating than the headache (12). The link between the vestibular symptoms and migraine is also reflected by a higher incidence of migraine in patients with recurrent dizziness who do not fulfill the criteria for other vestibular disorders (18–23). Such clinical findings have prompted efforts to recognize and classify migraine-associated dizziness under a distinct diagnostic entity.
Patients with VM symptoms frequently report sensitivity to head motion and visual surroundings or disabling misperceptions such as a sudden feeling of imbalance or tilt. When it comes to characterizing vestibular symptoms, dizziness or vertigo are often loosely used terms that may convey different symptom experiences in different people. According to the ICVD (28), vertigo is a sensation of self-motion when no self-motion is occurring or a sensation of distorted self-motion during an otherwise normal head movement. Dizziness is defined as a sensation of impaired spatial orientation without a distorted sense of motion. In fact, dizziness is a broader term that encompasses false spinning sensations (i.e. spinning vertigo) and also other false sensations such as swaying, tilting, or veering (i.e. non-spinning vertigo). In this classification, having dizziness does not preempt inclusion of vertigo if both symptoms are present as defined by their criteria. Vestibular symptoms do not include a pure sensation of impending fainting (presyncope), disordered thinking (mental confusion), or detachment from reality (depersonalization). Likewise, the term dizziness should not be applied if the patient experiences a generalized weakness or a non-specific sense of malaise or fatigue.
Unlike a typical migraine aura, which can last 5–60 minutes, vestibular symptoms in VM sufferers can persist for hours, leading to severe impairment of daily activities (25,26,29,30). This is a common clinical scenario in VM patients, which often becomes challenging for clinicians and costly for patients to treat, indicating an unmet need to delineate VM pathophysiology (31). In this article, we review common VM symptoms and clinical presentations. The current concepts related to VM pathophysiology and its treatment are discussed. Also, some of the barriers in addressing the neural mechanisms of dizziness, vertigo, and visuospatial symptoms in VM patients are highlighted.
Epidemiology and demographic factors
Vestibular migraine is a common cause of spontaneous vertigo in both children and adults (32–34). In children, benign paroxysmal vertigo of childhood is considered an early manifestation of migraine affecting about 3% of children between ages 6 and 12 (35,36). A recent nationwide study in the United States based on the ICHD-3 criteria found a VM prevalence of 2.7% in adults (37). In a large population study in Germany, VM lifetime prevalence was estimated at about 1%, and its one-year prevalence was about 1% (12). In a community-based study of middle-aged women, the one-year VM prevalence was higher, at about 5% (38). Despite classification efforts in recent years, VM has remained clinically underdiagnosed. Vestibular migraine is reported to account for 4–10% of diagnoses in specialized dizziness and headache clinics, although these numbers reflect different inclusion criteria among various studies (11,39–41). According to a study that examined data from a tertiary dizziness center, only 2% of patients were suspected to have VM by referring doctors, whereas 20% were later diagnosed as VM by specialists (42). In a Korean multicenter study, about 10% of migraineurs were diagnosed with VM in their first visit to neurology clinics (43). Another study found that only 10% of patients who met the VM diagnostic criteria were told that migraine was the cause of their vestibular symptoms (37).
Similar to other subtypes of migraine, VM has a female preponderance, with a reported female to male ratio of 1.5–5 to 1 (5,7,8,11,12,44,45). The reported age of onset for VM symptoms is between 8 and 50 years old or even older (median ages being the mid-30s to 40s) (12,21,39). Usually, migraine headache tends to present first, and patients may be headache-free for years before the onset of vestibular symptoms. In one study, the mean duration between the onset of headache and vestibular symptoms was about eight years (46). In women, vestibular symptoms can become more pronounced around the time of menopause (47).
In keeping with the heritability and genetic background of migraine disorders, familial occurrence has been reported in some VM patients, with an autosomal dominant pattern of inheritance and a decreased penetrance in men (48). Within a family, there could be multiple affected individuals, some with VM and some with other migraine variants. In a survey of family members in patients with chronic vertigo (and no auditory or neurologic disorders), half of the biological relatives who reported vertigo met the diagnostic criteria for migraine, as opposed to a small percentage of unrelated spouses who also reported vertigo (13).
Clinical presentation: Basis for VM diagnostic criteria
Common VM features: Vestibular migraine patients may have a history of migraine earlier on in their lives, and their migraine symptoms are often variable in nature, duration, or temporal relation to their vestibular symptoms (5,11,49). The core migraine features in VM patients are similar to those of migraine with or without aura. VM patients also report similar migraine triggers such as sleep disruption, menstruation, stress, or specific foods (e.g. aged cheese, red wine, or monosodium glutamate) (11,50,51). Not uncommonly, VM patients develop vestibular symptoms after the headache subsides, and in some patients headache and vestibular symptoms never occur together. Also, a minority of VM patients may experience vestibular symptoms in the time frame of 5–60 minutes, as defined for the duration of aura, and even fewer patients have these symptoms immediately before the headache starts. Apart from the headache and vestibular symptoms, VM patients may experience photophobia, phonophobia, or visual aura. In a case series, 60% of VM patients reported phonophobia, 70% photophobia, and 36% migraine auras (11). In another study, 87% of VM patients had photophobia, 86% phonophobia, 64% headache, and 13% migraine auras (52). Phonophobia refers to a sound-induced discomfort, which is a bilateral, transient phenomenon distinct from unilateral or persistent aural symptoms. Other intermittent auditory symptoms, such as a sense of pressure or fullness in the ears or tinnitus, have been reported in up to 40% of VM patients (5,8,9,52–55). However, since these symptoms are non-specific and also common in other vestibular disorders, they were not included in the VM diagnostic criteria (26). Of note, migraine features must be present at least half of the time along with vestibular symptoms in order to fulfill the criteria for VM diagnosis. At least one migraine feature is needed along with the vestibular symptoms, but different migraine features may be present at different times. Although the co-occurrence of migraine and vestibular symptoms is required by the ICHD-3 criteria for VM diagnosis, the Bárány classification ICVD has recognized the heterogeneity of migraine presentation in VM patients, and those patients who only have a history of migraine without co-occurrence of migraine features and vestibular symptoms are included under a subcategory of probable vestibular migraine (Table 1) (26).
Vestibular symptoms in vestibular migraine patients and relevant terminology defined by the International Classification of Vestibular Disorders (ICVD).

The duration of vestibular symptoms in VM patients is quite variable (9,11,39,52). About 30% of patients have symptoms lasting for minutes, 30% lasting for hours, and 30% lasting for several days at a time. The other 10% often report fluctuating daily symptoms. Nausea, vomiting, and susceptibility to motion sickness are also frequent in VM patients; however, these symptoms are non-specific and may also occur with other vestibular disorders (9,10,60–62). Vestibular symptoms are considered moderate when they interfere with daily activities and considered severe when they prevent daily activities.
Clinical overlap in VM presentations: Any vestibular disorder can be complicated by superimposed migraine attacks, in which cases VM is not the primary culprit. For example, migraine is more common in patients with benign paroxysmal positional vertigo (BPPV) than in age- and sex-matched controls (11,44,63–65), and having migraine could be associated with an increased risk of developing BPPV (66). Vertigo can also be a feature of migraine with brainstem aura, but other symptoms related to brainstem involvement (e.g. dysarthria) are typically not seen in VM patients. Thus, with such a wide range of clinical presentations, the co-occurrence of migraine and vestibular symptoms is not specific to VM patients, and other disorders with similar symptoms should always be considered in the differential diagnosis (Figure 1). In this scheme, the distinction between VM and other vestibular disorders such as Ménière's disease can become unclear, especially since the auditory and vestibular signs of labyrinthine pathology may be absent in the early stages of Ménière's disease (67,68). Therefore, it is not surprising that both conditions are often reported to coincide (55,68–71). Migraine is more commonly reported in patients with Ménière's disease than in otherwise healthy individuals, and about 13% of the patients meet the diagnostic criteria for both VM and Ménière's disease (72). Such association has prompted theories about a link between these two conditions versus those that attribute the overlap to the secondary migraine symptoms provoked by vestibular symptoms in Ménière's disease. Whether there is a pathological link between these two conditions remains unclear. Vestibular symptoms in VM patients are also similar to those of another clinical entity that was recently defined as persistent postural-perceptual dizziness (PPPD) (73,74). Similar to VM, PPPD patients have symptoms consistent with perceptual dysfunction in spatial orientation and also experience fluctuating dizziness or unsteadiness, provoked by postural changes or visual motion. Both VM and PPPD patients may have comorbid psychiatric conditions such as anxiety and depression (44,75–78). Also, migraine is one of the most common conditions associated with chronic visuospatial symptoms in PPPD patients (about 15–20% of cases) (44,73). Since VM and PPPD are both defined only by clinical symptoms and have unknown pathogenesis, it is not clear whether the overlap in their clinical presentations represents a pathological link between these two conditions.
Video head impulse testing (vHIT) shows bilateral vestibulopathy in a patient with clinical presentation consistent with vestibular migraine (VM): 37 year old Navy man with 2-year history of fluctuating dizziness, described as feeling of being drunk lasting for hours, and attacks with spinning sensation lasting for minutes without headaches. He reported frequent headaches, twice a month, associated with dizziness, nausea, photophobia and phonophobia since age 25. Bedside neurological and vestibular examinations were only remarkable for difficulty in tandem gait. The vHIT shows reduced vestibular gains in the horizontal canal planes at 0.7, calculated as eye velocity (black traces) divided by head velocity (red traces: Right side and blue traces: Left side). There are also corrective saccades in the eye traces. These “covert” saccades are embedded in the deficient vestibulo-ocular responses during head rotation and are difficult to discern with the naked eye. Cases like this show that vestibular dysfunctions can be complicated by superimposed migraine attacks; however, VM is not the primary culprit.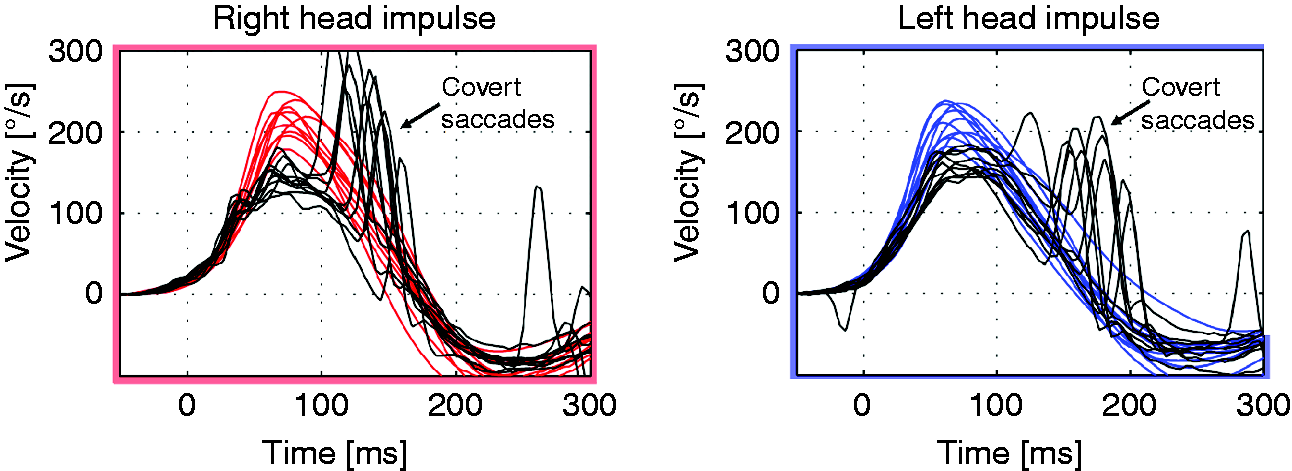
Clinical finding and laboratory testing
Vestibular migraine is a diagnosis primarily based on clinical history, with currently no pathognomonic clinical sign or laboratory test that can verify its diagnosis. Vestibular laboratory abnormalities are quite variable among VM patients, which may reflect inconsistent findings regarding the existence of a peripheral vestibular component. Nevertheless, vestibular testing is still helpful to rule out other disorders considered in the differential diagnosis (Figure 1). Neurological examination is usually normal in VM patients during symptom-free periods. However, some mild, non-specific vestibulo-ocular abnormalities have been reported in association with VM. Pursuit abnormality is reported in up to 48% of VM patients, spontaneous nystagmus in about 10% of patients, and central positional or gaze-evoked nystagmus in up to 28% of patients (5–7,45,59,79–84). These non-specific ocular motor abnormalities increased over time, from 16% to 41% of patients during a follow-up after 5.5 to 11 years (85). Apart from pursuit dysfunction, the most frequent ocular motor abnormality is central positional nystagmus. In a small study, about 70% of patients were found to have pathological nystagmus during their VM attacks (86). Another study found nystagmus was provoked by horizontal headshaking in 35% of VM patients, and spontaneous nystagmus in 19% of the patients during vestibular attacks (87). Head shaking-induced nystagmus is also reported in between the attacks (79,81). Vestibular migraine patients were also found to have positional nystagmus after removal of visual fixation (e.g. vestibulo-ocular examination under Frenzel goggles). The nystagmus was low-velocity and sustained during the vestibular attack, but it dissipated during the symptom-free period.
While some studies have found a higher incidence of central vestibular dysfunction in VM patients (39,86), others have found no significant central vestibulo-ocular findings (76,88), or have reported a higher incidence of peripheral vestibular dysfunction in VM patients (6,23,80,89). Such variability could be related to lack of standard diagnostic criteria among earlier studies or a sole reliance on clinical symptoms by the current VM criteria, which does not prevent inclusion of patients with other vestibular disorders who may have superimposed migraine symptoms. In this context, findings such as asymmetry in caloric responses (reported in up to 20% of VM patients), or vestibular loss with video head impulse testing (vHIT) cannot be specific to VM and may represent a secondary migraine syndrome triggered by uncompensated vestibular dysfunction (Figure 1) (5,6,39,80,85,90–92).
The cervical and ocular vestibular evoked myogenic potentials (cVEMP/oVEMP), the widely used laboratory tests of otolith function, have also shown conflicting results in VM patients. Some studies have reported reduced amplitude or delayed latencies of VEMP responses (93–98), whereas other studies have found asymmetrical VEMP responses with normal latencies and amplitudes (99,100). Three recent studies using ICHD criteria have reported abnormal oVEMP but normal cVEMP responses in VM patients, which differed from the pattern of VEMP responses in normal controls (101) or patients with Ménière's disease. A significant vestibulo-ocular finding that could be related to VM pathophysiology is a longer duration of post-rotatory nystagmus in VM patients compared with healthy controls or migraine patients without dizziness (i.e. increased time constant of the vestibulo-ocular reflex) (Figure 2) (81). VM patients were also found to have quicker modulation of the otolith-ocular response during off-axis rotation (i.e. body centrifugation) (76). These findings suggest an innate hypersensitivity of the vestibular system in these patients. Balance impairment is also commonly reported in VM patients, including abnormal Romberg and sensory organization test (SOT) (76,79,86,89,91,92). Studies using static posturography have found increased sway in VM patients compared with healthy controls (80,104). Hearing impairment is reported in about 8% of VM patients, which is often mild and non-progressive, as opposed to progressive low frequency hearing loss in Ménière's patients (40,85,105).
Vestibulo-ocular responses during step velocity rotations (60°/sec) in a VM patient versus a healthy control. Data points show the slow phase velocity of vestibular nystagmus induced by rotation around the body axis to the right (positive values) and left (negative values). The gains of vestibular responses (maximum eye velocity/60°/sec step velocity) are normal in both the VM patient and the healthy control; however, the durations of nystagmus measured as time constant (TC) are longer in the VM patient compared to the healthy control (30–33 sec versus 7 sec). See reference 106 for rotational chair testing.
Pathophysiology
To date, VM pathogenesis remains obscure as the wide ranges of clinical and laboratory findings do not seem consistent with a uniform pathology. Such variability comes from sole reliance on clinical symptoms and lack of objective measures for diagnosis, which have hampered efforts toward understanding VM pathophysiology. Various mechanisms have been put forward to explain VM symptoms and some are broadly inferred from other migraine disorders (Figure 3). In this context, altered neural activity within the trigeminovascular system (TVS) is considered the primary mechanism for headache in migraine patients (107). The TVS neuropeptides, such as substance P and calcitonin gene-related peptide (CGRP), can cause vasodilation and neurogenic inflammation, leading to throbbing pain or “central sensitization” that often presents as allodynia (108–110). Some of these neurotransmitters are also expressed in the vestibular system (e.g. CGRP and serotonin) and might be involved in VM pathophysiology (108,111,112). The trigeminal nucleus is connected to the contralateral thalamus, which in turn sends projections to the temporal, parietal, insular, and cingulate cortical regions. The nociceptive brainstem centers such as the nucleus raphe magnus, periaqueductal gray and hypothalamic areas are also connected with the TVS and vestibular nuclei (113,114,114). These reciprocal connections can modulate neural activity within both the TVS and the vestibular system (112,113,115–118). For example, trigeminal stimulation produced nystagmus in migraine patients, suggesting increased vestibular excitability in these patients compared with healthy controls (116,119). Similarly, spontaneous nystagmus during VM attacks or prolonged vestibular responses in some VM patients (i.e. long time constant of vestibular nystagmus) could be linked to vestibular hyperexcitability in these patients (76,81,120). It is, however, not clear whether such hyperexcitability is at the level of the peripheral vestibular system or the brainstem, or whether it could be related to the modulating effects of the cerebellum or cerebral hemispheres on the vestibular system.
Possible mechanisms involved in the pathogenesis of vestibular migraine. Abnormal sensory modulation or integration within the thalamo-cortical network could result in dizziness and spatial disorientation. Hyperactivity within the trigeminovascular system (TVS) and nociceptive brainstem centers could result in headache. Altered activity in the vestibular system could lead to transient vestibulo-ocular dysfunction or vestibular hypersensitivity associated with migraine features.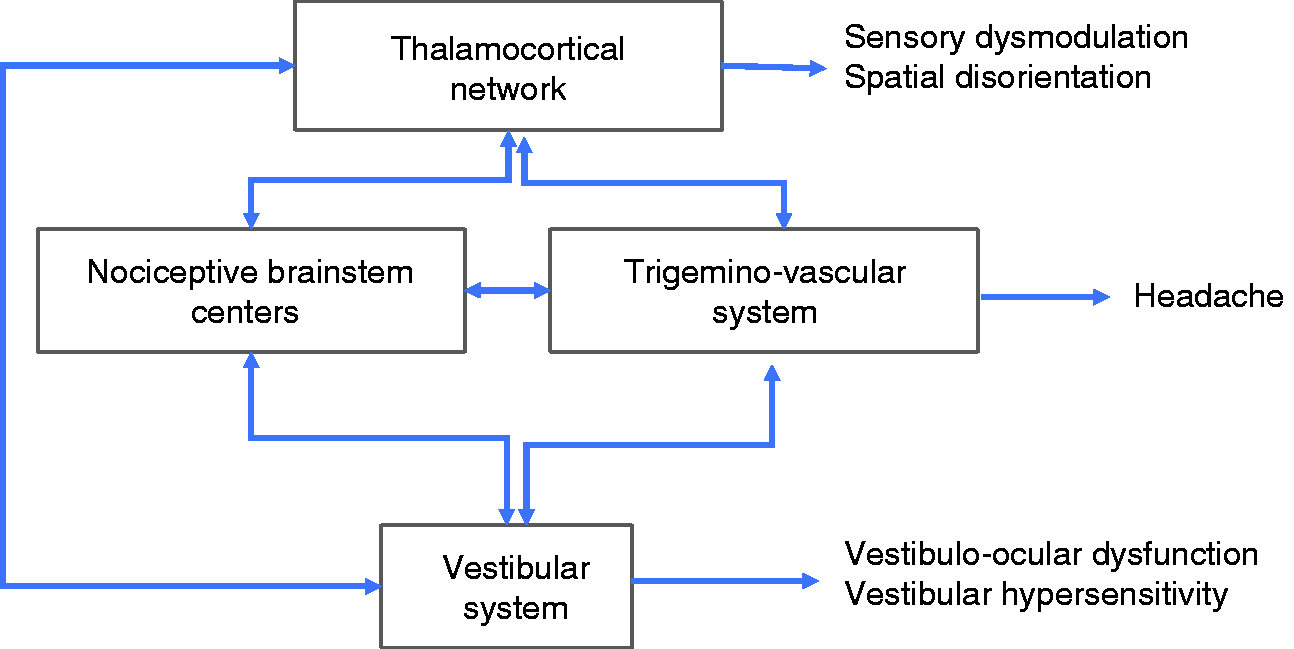
The phenomenon referred to as cortical spreading depression has been proposed as the neural correlate of migraine aura (107). This is a depolarization wave that slowly spreads across the cerebral cortex, followed by a prolonged suppression of cortical activity. The theory of cortical spreading depression cannot explain chronic or fluctuating symptoms in migraineurs. However, according to its proposed mechanism, symptoms such as hypersensitivity to light and sound could be linked to a generally hyperexcitable brain state in migraine patients (121). This has led to the concept of sensory dysmodulation, in which deficient habituation and potentiation of sensory responses are implicated in migraine pathogenesis (122,123). Accordingly, exposure to one sensory stimulus results in hypersensitivity that can extend to other sensory stimuli. VM patients, likewise, show higher sensitivity to motion in the roll plane (e.g. lateral head tilt with respect to gravity) (Figure 4) (120,124,125). Perception of tilt during whole body centrifugation is also altered in VM patients, suggesting that the integration of semicircular canal and otolith inputs is affected in these patients. During whole-body yaw rotations, VM patients were found to have altered perceptual thresholds while reporting direction of rotation (126). During lateral head tilts, VM patients had larger errors in spatial orientation compared with healthy controls (Figure 4) (127). When the head is tilted, the brain must integrate vestibular signals that encode head position with visual inputs in order to maintain spatial orientation. The larger errors of spatial perception in VM patients were in the opposite direction of the head tilt, consistent with overestimation of the head position in the process of sensory integration for spatial orientation. In keeping with this result, VM patients reported dizziness mostly in the same head direction that induced larger errors of spatial orientation. VM patients were also found to have high variability in spatial orientation with the head in the upright position (128,129). Altogether, these findings suggest that visuospatial symptoms in VM patients are related to altered sensory processing and integration that contribute to the perception of spatial orientation. Regarding the effect of visual motion, VM patients had larger visual-induced errors in spatial orientation during and after optokinetic stimulation compared with healthy controls (126,130,131). These errors also increased significantly in migraine patients with the duration of visual stimulation (132). In addition, VM patients had larger postural sway with visual motion compared with healthy controls (76,131,133). Regarding other sensory modalities, there is evidence for sensory dysmodulation affecting the auditory system in VM patients. In the presence of noise, otoacoustic emissions were less suppressed in VM patients compared with healthy controls, suggesting a link to phonophobia in these patients (134).
(a) Spatial orientation measured as subjective visual vertical (SVV) errors in a group of VM patients versus healthy controls (127). In the upright head position, there is no significant difference between VM patients and controls. During static lateral head tilt to the right (20°), SVV errors in VM patients are in the opposite direction of head tilt, consistent with overestimation of the head tilt position for spatial orientation. (b) During low-frequency dynamic head tilt, VM patients show a lower motion detection threshold compared with migraine patients without dizziness and healthy controls (modified with permission from Lewis et al. (125)).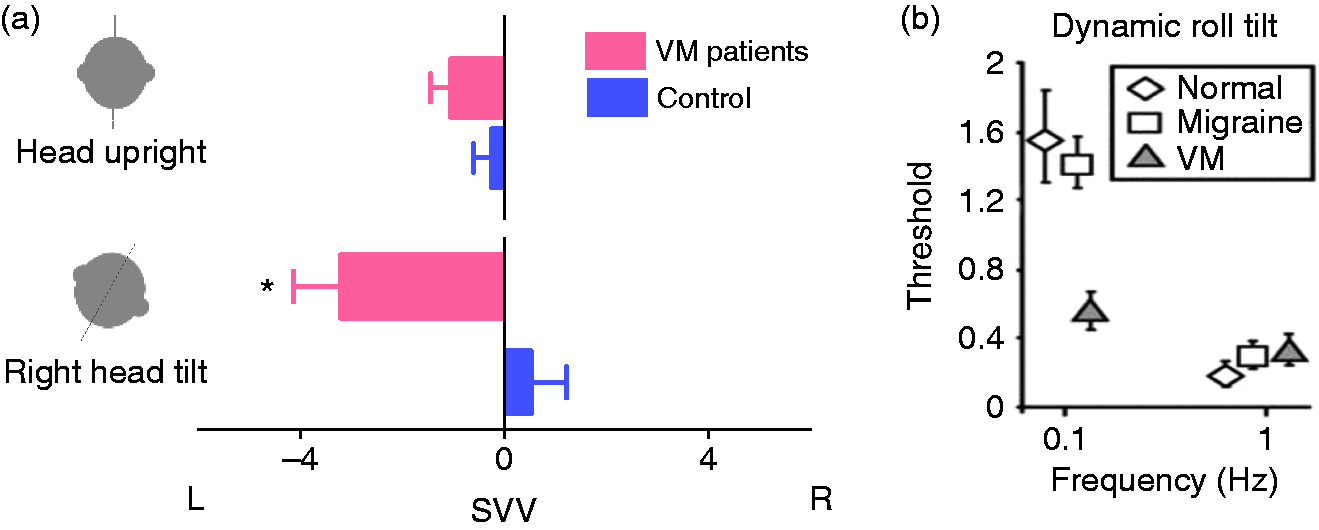
Imaging studies suggest structural and functional changes within the temporo-parietal regions in VM patients (117,135,136). The higher-order neural mechanisms within these cortical regions are involved in sensory integration for coherent spatial perception (137). In addition, VM patients were also found to have abnormal thalamic activities (117,138). These patients showed increased thalamic activations with cold water ear irrigation during fMRI studies, in comparison with migraine patients without aura or healthy controls (138). The magnitude of thalamic activation was positively correlated with the frequency of migraine attacks in VM patients. Also, increased ictal PET activities during vertigo attacks in VM patients were noted in the temporo-parieto-insular areas and bilateral thalami (117). These findings are in agreement with the studies that show the role of the thalamus as a major sensory relay within the vestibular pathways, and its involvement in multisensory processing and integration including vestibular, visual, and somatosensory inputs (139,140). However, little is known about these sensory processes and how these higher-level networks could be affected in VM patients.
The familial occurrence of VM suggests a genetic component in its pathogenesis. Although there are some familial migraine syndromes such as hemiplegic migraine or episodic ataxia, similar mutations (e.g. CACNA1A or ATP1A2) are not found in VM patients (141,142). The CACNA1A gene encodes a subunit of the voltage-gated P/Q-type calcium channels, and its mutation can cause at least three neurological disorders linked to calcium channelopathy: Episodic ataxia type 2, familial hemiplegic migraine type 1, and spinocerebellar ataxia type 6 (143). Vertigo and migraine symptoms are also common in patients with episodic ataxia type 2 (144). Despite the lack of evidence for monogenic inheritance, whether polygenic inheritance plays a role in VM patients warrants further research (48,145–150).
Treatment
The uncertainties surrounding VM diagnosis over the last few decades have also limited advances in treatment. In general, VM patients can be managed with lifestyle modification, dietary adjustments, medications, vestibular physical therapy, and activities that can enhance perception of spatial orientation, such as ping-pong or dancing. Given the considerable psychosocial impact of symptoms in VM patients, it is important to adjust treatment strategies based on each patient's needs. Those who are not significantly impaired by their symptoms may benefit from reassurance to allay their fear and prevent unnecessary medical costs. This is especially important for patients with prominent vestibular symptoms. Those who are significantly impaired by their symptoms often benefit from combined interventions. In this process, identifying triggers and implementing measures to circumvent them can be used as the first line of treatment. As a key intervention, dietary adjustment and eliminating triggers such as red wine, aged cheeses, artificial sweeteners, processed meats, chocolate, caffeine, MSG, and alcohol are found to be effective in reducing symptoms (14,151,152).
Summary of studies on preventive treatments for vestibular migraine defined based on the ICHD-3 criteria.

p < 0.05.
There are only a few randomized controlled studies on acute treatment for VM attacks. One small double-blind, placebo-controlled study showed 38% of VM patients improved from severe or moderate vertigo to mild or no vertigo in two hours after taking zolmitriptan, compared to 22% with placebo (165). However, due to the limited power of the study, the difference did not reach statistical significance. Rizatriptan reduced vestibular-induced motion sickness in migraineurs compared to the placebo (166). The authors suggested that rizatriptan may reduce motion sickness by influencing serotonergic vestibulo-autonomic pathways. Sumatriptan was also effective in improving both headache and vertigo regardless of their temporal relation (155). Steroid injections were reported to be effective in four patients with prolonged or frequent vertigo attacks in VM patients (167), similar to the effect of steroids on patients with status migrainosus (168).
Vestibular rehabilitation has been used to alleviate symptoms and promote recovery in VM patients, as in other vestibular disorders (169). Although most studies show that focused vestibular rehabilitation benefits patients, no randomized control study has evaluated the efficacy of vestibular physical therapy in VM patients (170). One early study compared the effect of vestibular rehabilitation in 14 VM patients to 25 patients with migraine who had unrelated vestibular dysfunction, and showed both groups improved significantly on subjective and objective outcome measures within four months (84). This finding was supported by another study of 34 VM patients (14). A prospective study in which 20 VM patients received a nine-week customized vestibular rehabilitation program also showed benefits, regardless of their medication regimen (171). In another study, 28 VM patients based on ICHD-3 beta criteria and 79 patients with tension-type headache and dizziness were recruited (172). Both groups were trained in vestibular therapy exercises during a five-day admission process and were assessed 6 months later. The results showed that vestibular rehabilitation contributed more prominently to clinical improvement in the VM group compared with the tension-type headache group. In practice, vestibular rehabilitation can be particularly beneficial to patients if secondary complications such as deconditioning or visual dependence have developed. VM patients often experience dizziness and disorientation with changes in head and body positions, which raises the possibility of a “higher level” dysfunction in multisensory integration for spatial orientation (i.e. visual, vestibular, and somatosensory inputs). Therefore, natural activities that can enhance spatial perception and body coordination, such as ping-pong and dancing, can be helpful to alleviate symptoms in these patients. Such an approach is supported by the effects of long-term training in disciplines such as ballet dancing and yoga. Experts in these fields are better at processing and weighting body perceptual information and are less visually dependent in their perception of spatial orientation (173,174).
Conclusions
Vestibular migraine is among the most common causes of recurrent vertigo in the general population, with largely unknown pathophysiology. Patients with VM experience disabling spatial misperceptions including unusual sensitivity to head motion or visual stimuli, or sudden feelings of imbalance or tilt. These symptoms often become chronic and lead to severe impairment of daily activities in VM sufferers. Unlike typical migraine aura, vestibular symptoms in VM patients are not temporary symptoms that occur with headaches, and their duration can last from hours to several days. Currently, the VM diagnosis is entirely based on clinical history, and according to the recent consensus of the International Headache and the Bárány Societies, the presence of episodic vestibular symptoms associated with at least one migraine feature can fulfill the VM diagnostic criteria. Neurological and ocular motor examination is usually normal in VM patients during symptom-free periods. However, some mild, nonspecific vestibulo-ocular abnormalities such as positional nystagmus have been reported, especially during VM attacks. So far, no pathognomonic clinical or laboratory finding exists for VM; however, vestibular testing is still helpful to rule out other disorders considered in the differential diagnosis. Vestibular laboratory abnormalities are quite variable among VM studies. Such variability could be related to the lack of standard diagnostic criteria used in earlier studies or the reliance on clinical symptoms alone in the current VM criteria, which does not prevent inclusion of patients with other vestibular disorders who may have superimposed, secondary migraine symptoms. Like other types of migraine, both abortive and preventive medications have been used in VM patients. However, the efficacy of these medications has not been verified by large-scale randomized placebo-controlled clinical trials. Non-pharmacological treatment including life-style adjustment, trigger avoidance and vestibular rehabilitation are also shown to be beneficial in VM patients. The pathophysiology of VM remains largely unknown; however, recent findings have provided some preliminary insights. These include reports of abnormal motion-detection thresholds during roll tilt or yaw rotation, or larger errors of spatial orientation during head tilt, which suggest high-level dysfunctions related to vestibular processing in these patients. So far there is no information about the plausible role of central neural processes in VM spatial disorientation and their link to visuospatial symptoms in these patients. Therefore, gaining insight into these processes and how they result in dizziness, vertigo, and spatial disorientation in VM patients is an important step towards devising effective treatment strategies.
Footnotes
Clinical implications
VM is among the most common causes of recurrent vertigo. In addition to vertigo, patients may experience disabling spatial misperceptions.
The diagnosis of VM is entirely based on clinical history, and no laboratory test is pathognomonic.
The pathophysiology of VM remains obscure, but recent studies suggest sensory dysfunction is related to processing vestibular inputs.
The treatment of VM requires lifestyle adjustment, trigger avoidance and vestibular rehabilitation as well as abortive and preventive medications, although solid evidence is still lacking.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the following grants: Ministry of Science and Technology of Taiwan [MOST 107-2321-B-010-001], Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, as well as the United States National Institute of Deafness and Other Communication Disorders (NIDCD); K23DC013552.

