Abstract
Introduction:
A better understanding of etiology might improve poor outcomes of trochlear headaches (TRHs).
Aims:
To study clinical spectrum, etiology, and therapeutic response of TRH.
Methods:
Fifty-three TRH patients seen in a single center between 2015 and 2020 were included, excluding Trigeminal Autonomic Cephalalgia (TAC).
Results:
Mean age was 36.45 years (range 11–85 years), with 77.35% being females. Twenty-five patients had continuous trochlear headache (CTRH) and 28 episodic trochlear headache (ETRH). Tension-type headache (TTH) occurred in 9 ETRH patients and 24 of 25 CTRH patients, and migraine-like headaches occurred in 19 ETRH patients and 8 CTRH (trochlear migraine) patients. Prior history of headaches was noted in 22 of 28 ETRH and 11 of 25 CTRH patients. Twenty-eight responded to migraine/TTH prophylaxis, 25 being nonresponders (partial/no response). Fourteen of 25 nonresponders, 4 of 28 responders (4 of 4 secondary and 5 of 9 idiopathic trochleitis (IT), 3 of 9 primary TRH (PTRH), and 6 of 28 ETRH) had autoantibodies, that is, 11 antinuclear antibodies (ANAs) and 7 antithyroid antibodies. Ten of 14 (71.42%) antibody-positive nonresponders improved with immunosuppressants including steroids/hydroxychloroquine and only 11 required local injections. Finally, 38 patients had good response, 13 partial, and 2 no response. The etiology and refractoriness of IT can be attributed to underlying autoimmunity and a minor contribution by primary headaches, vice versa being the case for PTRH and ETRH. Refractory TRHs should be evaluated for underlying autoimmunity and primary headaches.
Conclusion:
Identification and treatment of underlying autoimmunity and primary headaches can help improve outcome of TRH.
Keywords
Introduction
Trochlear headache (TRH) or trochleodynia is an important but underdiagnosed cause of refractory headache that generally requires local steroid injections. 1 Trochlea is a cartilaginous structure situated in the superomedial orbit that supports movement of the superior oblique tendon, other nociceptive structures in its vicinity being fibrovascular sheath and supraorbital or supratrochlear nerves (Figure 1). TRH may occur in isolation or can cause exacerbation in episodic migraine and tension-type headaches (TTHs). It has been attributed to trochleitis or primary headaches, and the proposed mechanisms are neuropathic, neuromuscular, and inflammatory. Headache attributed to trochleitis is characterized by continuous local pain and swelling often aggravated by vertical eye movements or reading. 1,2 When secondary to an autoimmune connective tissue disorder it is called secondary trochleitis (ST), otherwise it is termed idiopathic trochleitis (IT). Primary trochlear headache (PTRH), a noninflammatory condition can also cause continuous trochlear headache (CTRH) and tenderness, but without any local inflammation or swelling. 3 –5 In majority of cases, pain is initially intermittent and becomes continuous within months, but many patients get only episodic trochlear headaches (ETRHs), and the patients of this group have not received much attention. Trochlear migraine (TM) refers to unilateral migraine and ipsilateral trochlear pain. A clear understanding of the various clinical entities and mechanisms responsible for them is indeed lacking. While the orbital inflammation of ST is caused by autoimmune disorder, it is likely that secondary mechanisms involved in causation of IT are yet unidentified. It is also not clear whether PTRH is related to other primary headaches or autoimmune disorders. A better understanding of these aspects might lead to improved outcome of this refractory condition. To address this knowledge gap, we conducted a study of clinical spectrum, investigations, and therapeutic response of patients with continuous or ETRHs.
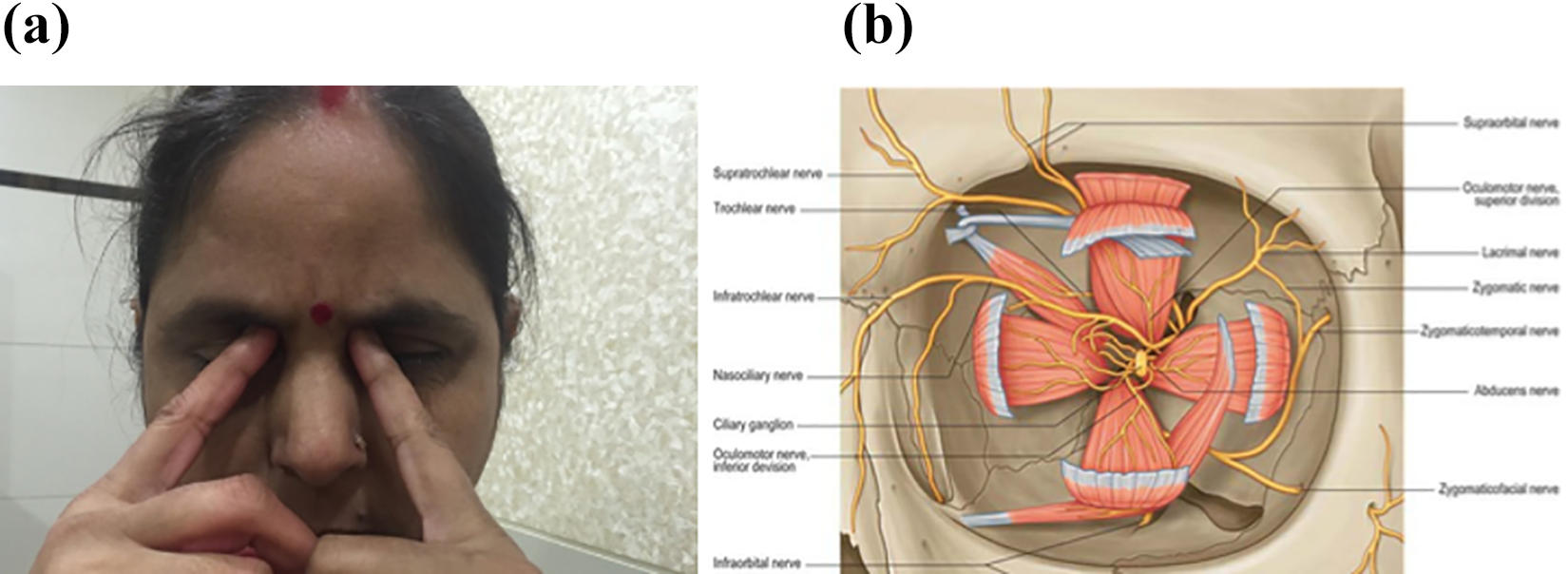
(a) Location of TRH. (b) Anatomy of trochlear region. TRH: trochlear headache.
Aims and objectives
To study the clinical spectrum, investigations and response of TRHs to prophylactic medications and need for local injections.
To understand the etiology of poorly characterized TRH entities like PTRH and IT.
To try and identify factors associated with refractoriness of TRHs.
Methods
The electronic database of the headache clinic was searched from January 1, 2015, to February 29, 2020, and 53 patients who had presented with either CTRH or ETRH were identified out of a total of 1745 registered patients. Patients were excluded if their headaches were explained by another condition, that is sinusitis, periorbital cranial neuralgia, and trigeminal autonomic cephalalgia. Ethics committee approval was taken from Independent Ethics Committee, Navi Mumbai (ref no: IEC/120/2014) prior to initiation of the study.
The term TRH used in the context of the study denoted headache and tenderness in the region of trochlear apparatus, aggravated, and/or reproduced by the action of superior oblique muscle. The term Continuous trochlear headache was assigned for daily occurrence of TRH which generally persisted throughout the day, whereas the term Episodic trochlear headaches was assigned for intermittent trochlear pain separated by pain-free day(s).
Patients having CTRH had a diagnosis of either trochleitis or PTRH. The diagnostic criteria for headache attributed to trochleitis was according to the International Classification of Headache Disorders, third edition. 5 Presence of swelling and inflammation in the trochlear region helped in the diagnosis of trochleitis and their absence favored the diagnosis of PTRH. ST was diagnosed if there was clinical, imaging, or histopathological evidence of orbital involvement by an inflammatory or neoplastic pathology, otherwise trochleitis was considered to be IT. The local inflammation in IT is restricted to tendon of superior oblique, while it extends in the orbit to the muscle and beyond in ST. 3 –5 TRH that triggered a secondary migraine attack in a patient with CTRH was termed as TM. 2,5 ETRH beginning from trochlea was termed episodic trochlear migraine (ETM). On the other hand, episodic headache spreading from head to trochlea was termed as migraine-associated TRH (MAT) or TTH-associated TRH (TAT) (Figure 2).
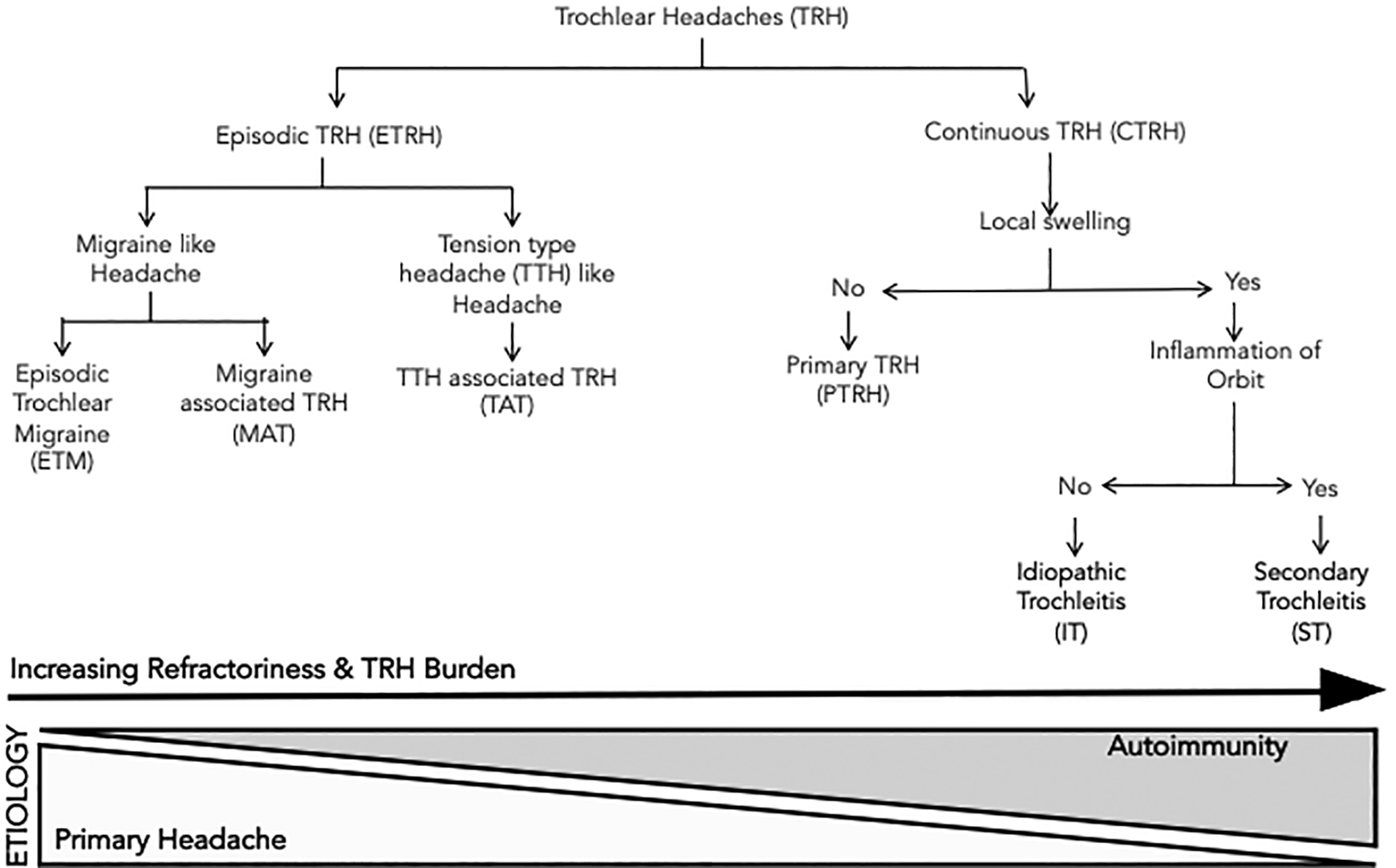
Proposed classification of TRHs. TRH: trochlear headache.
Onset of TRH (TRH onset) was considered acute if the headaches became continuous or severe from the onset, subacute if within a month and insidious if after a month to within a year from onset. Headache location, quality, quantity, associated features like nausea and photophobia, and aggravating and relieving factors were also noted. TRHs were considered as migraine-like if they had throbbing/aching nature, nausea or sensitivity to light/sound and aggravated by routine activities. They were considered as TTH-like if the TRH was constricting in nature and not aggravated by routine activities. 5 Patient-reported mild, moderate, and severe intensities of TRHs were graded as 1, 2, and 3, respectively. Burden of TRH (TRH burden) was a product of total TRH days/month and the reported intensity of headache (range 0–90). Blood investigations, antibodies testing (antinuclear antibodies (ANAs), ANA blot, antithyroid antibodies (ATABs) i.e. antimicrosomal antibody (AMA) and antithyroglobulin antibody (ATG)), and magnetic resonance imaging (MRI) of brain and orbit were also noted. Values of ANA (by immunofluorescence) levels ≥ 1:100, AMA levels ≥ 5.61 IU/mL, and ATG levels ≥ 4.11 IU/mL done by Chemiluminescent microparticle immunoassay (CMIA) were considered elevated, such patients being termed as antibody positive. Examination findings, especially tenderness and swelling in the trochlear region, were noted.
Details of treatment with prophylactic dugs, nonsteroidal anti-inflammatory drugs (NSAIDs), immunosuppressants, and local injections were recorded. All patients received headache prophylaxis for migraine/TTH, with/without NSAIDs based on the nature of headaches, prior headache history, and individual profiles for a minimum duration of 3 months and continued for 6–12 months or longer depending on the extent of relief. Drugs commonly prescribed for migraine prophylaxis included propranolol (maximum 160 mg/day), gabapentin (maximum 900 mg/day), topiramate (maximum 100 mg/day), divalproex (maximum 1000 mg/day), and flunarizine (maximum 10 mg/day). Drugs usually prescribed for prophylaxis of TTH included venlafaxine (maximum 75 mg/day), mirtazapine (maximum 30 mg/day), and NSAIDs (ibuprofen, diclofenac, etoricoxib). Amitriptyline (maximum 75 mg/day) or nortriptyline (maximum 50 mg/day) was used for prophylaxis of both migraine and TTHs. Patients with ST received treatment specific for the etiology. Local trochlear injections containing 40 mg of methylprednisolone and 2–3 ml of 2% lignocaine were administered to patients having inadequate pain relief. Steroids, hydroxychloroquine sulfate (HCQS), or other immunosuppressants were added in nonresponders having evidence of autoimmunity (autoantibodies).
Efficacy of treatment was summarized as good response (R), partial response (P), or no response (N). “Good response” was assigned for more than 50% reduction in the severity/frequency of TRHs. Less than 50% reduction in pain was termed “partial response”, while no observable reduction in pain was recorded as “no response”. Patients who had good response to treatment were termed responders and others nonresponders. Refractoriness was defined as a partial or no response to at least two prophylactic drugs given over 3 months. The final outcome was considered as status of the patient at the last follow-up visit. Statistical analysis was performed using independent sample t-test for continuous variables and χ 2 test for categorical variables, and the level of significance used was p ≤ 0.05.
Results
The study group comprised 3% of patients attending headache clinic but accounted for nearly 10% of patients with refractory headaches in the clinic. The mean age of the cohort was 36.45 years (SD 2.08, range 11–85 years), and 41 patients (77.35%) were female. The mean headache duration at presentation was 5.01 months (SD 1.19, range 1–60 months), and the mean follow-up duration was 15.90 months (SD 2.37 months, range 2–68 months). Of the 25 patients with CTRHs, 12 had PTRH, 9 had IT, and 4 had ST, which included 1 patient each of immunoglobulin G4 (IgG4)-related disease and Tolosa–Hunt syndrome and 2 patients with Wegener’s granulomatosis. Of the 28 patients with ETRHs, in 19 of them TRHs were related to migraine and to TTH (TAT) in 9 patients. Of these 19 patients, in 7 the headache began from the trochlear region and spread to head (ETM), whereas in 12 it started from head and then involved the trochlea (MAT) (Table 1).
Summary.

TRH: trochlear headache; TM: trochlear migraine; ETM: episodic trochlear migraine; MAT: migraine-associated trochlear headache; TAT: tension-type headache-associated trochlear headache; TTH: tension-type headache; PTRH: primary trochlear headache; CTRH: continuous trochlear headache; ETRH: episodic trochlear headache; IT: idiopathic trochleitis; seropos: autoantibody positive; seroneg: autoantibody negative AS: acute/subacute; IN: insidious.
Most patients (51 of 53) had moderate-to-severe intensity headaches in the region of medial eyebrow or orbit that was aggravated in two-thirds by vertical eye movement or reading. Headache was of constricting nature (TTH-like) in the CTRH group, while it was more often like migraine than TTH in the ETRH group. The mean duration (TRH duration) and burden (TRH burden) of TRHs was more in the CTRH group than in the ETRH group (6.24 vs. 3.92 months and 72.40 vs. 30.21). Prior history of headaches was noted in 17 of 19 ETRH patients who had migraine-like headaches, 5 of 9 ETRH patients having TTH-like headaches, 7 of 12 patients with PTRH, and 4 of 9 patients with IT which is presumably an inflammatory condition. TRHs were associated more often with acute–subacute onset, bilateral, TTH-like headaches, and diplopia in the CTRH group and with insidious onset, unilateral, migraine-like headaches, and prior history of headaches in the ETRH group (p < 0.05) (Tables 1 to 3 and Figure 2). Clinical examination showed local tenderness in all patients, but local swelling was seen only in those having trochleitis. Brain and orbit MRI revealed trochlear apparatus abnormalities only in patients with ST.
Response to local injection in CTRH.

ST: secondary trochleitis; PTRH: primary trochlear headache; TRH: trochlear headache; CTRH: continuous trochlear headache; IT: idiopathic trochleitis.
Response to addition of immunotherapy.

ST: secondary trochleitis; PTRH: primary trochlear headache; ETRH: episodic trochlear headache; IT: idiopathic trochleitis.
Twenty-eight patients had good response to headache prophylaxis, 16 having episodic and 12 continuous TRHs. Of the 25 patients having partial or no response to prophylaxis (nonresponders), 12 had ETRHs and 13 CTRHs. Six of the 13 nonresponders having CTRH had no response at all to prophylaxis, whereas all ETRH nonresponders had at least partial response (p < 0.05). Refractoriness to prophylaxis was more often associated with trochleitis than PTRH (10 of 13 vs. 3 of 12; Table 4 and Figures 2 to 4). Migraine prophylaxis was found useful in ETRH patients with migraine-like headaches and in those having TM exacerbation of CTRH. NSAIDs and TTH prophylaxis were found to be less useful in patients having TTH-like headaches in the CTRH group than in the ETRH group. Local steroid injections were required in 11 nonresponders with CTRH, 4 of these showed good response and another 4 had partial response. Injection benefit usually began in 3 days and lasted for at least a few months (Table 4 and Figures 4 and 5).
Continuous TRHs.
TRH: trochlear headache; PTRH: primary trochlear headache; IT: idiopathic trochleitis; ST: secondary trochleitis; ATAB: antithyroid antibodies; ANA: antinuclear antibodies; TM: trochlear migraine; TTH: tension-type headache; HCQS: hydroxychloroquine sulfate.

Headache characteristics according to TRH subtype and autoimmunity. TRH: trochlear headache; TTH: tension-type headache; Troch: trochlear; seropos: autoantibody positive; seroneg: autoantibody negative; ETRH: episodic trochlear headache; CTRH: continuous trochlear headache; PTRH: primary trochlear headache; IT: idiopathic trochleitis; ST: secondary trochleitis.

Correlation of TRH type and prior headache history with response to prophylaxis and autoimmunity. TRH: trochlear headache; TTH: tension-type headache.
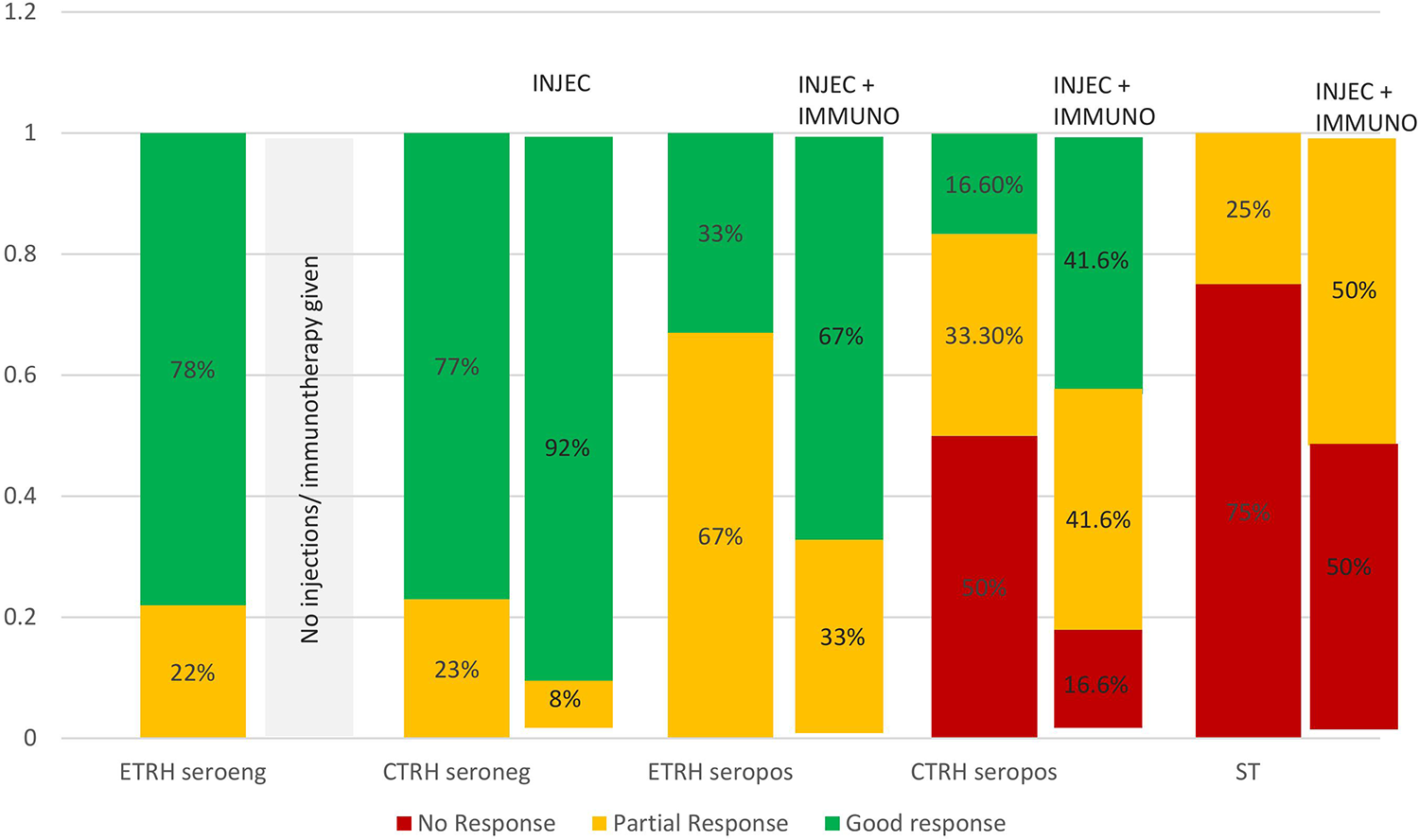
Response to escalation of therapy in prophylaxis nonresponders. Injec: local steroid injection; Immuno: immunosuppressants; ETRH: episodic trochlear headache; CTRH: continuous trochlear headache; ST: secondary trochleitis; seropos: autoantibody positive; seroneg: autoantibody negative.
Autoantibodies were detected in 14 of 25 nonresponders as compared to only 4 of 28 responders. Autoimmunity was found to be associated with acute–subacute onset, drug refractoriness, and benefit of local injections (p < 0.05). In patients with IT, 5 of 6 nonresponders had autoantibodies compared to none of the responders. Similarly, in patients with PTRH, more nonresponders were detected to have autoantibodies as compared to responders (1 of 3 vs. 2 of 9). Of a total of 18 antibody-positive patients, 9 had ANA, 7 had ATAB, and 2 had both antibodies. Two patients of IT were subsequently diagnosed as primary Sjogren’s syndrome (SSA/SSB antibody positive) and two patients with PTRH were diagnosed as secondary Sjogren’s syndrome (dry eyes and elevated ATAB). All other antibody-positive patients were thought to have undifferentiated autoimmune disorder.
Antibody-positive patients were treated with oral steroids (n = 5), HCQS (n = 7) or other immunosuppressive agents and improved therapeutic outcomes were seen in 10 of 14 (71.42%) patients. Seven patients were treated with HCQS 400 mg daily for 3–6 months and five patients with oral deflazacort 30 mg daily for 2–3 weeks, followed by a gradual taper. Patients with ST were administered intravenous methylprednisolone 1 g daily for 3–5 days, followed by oral steroids. Intravenous rituximab 1 g was given 2 weeks apart in two patients having secondary trochleitis, one IgG4-related disease and the other Wegener’s granulomatosis) (Tables 1 and 4 and Figures 4 and 5).
Final outcome recorded at mean 15.90 months showed that 38 patients had good response, 13 had partial response, and only 2 patients had no response despite treatment. Both these patients had secondary trochleitis, with one patient having Tolosa–Hunt syndrome and another Wegener’s granulomatosis. (Tables 4 and 5 and Figure 5).
Episodic TRHs.

TRH: trochlear headache; TAT: tension-type headache-associated episodic trochlear headache; MAT: migraine spreading from head to trochlea in episodic trochlear headache; ETM: migraine spreading from trochlea to head in episodic trochlear headache; ATAB: antithyroid antibody; ANA: antinuclear antibody; TM: trochlear migraine; HCQS: hydroxychloroquine sulfate; TTH: tension-type headache.
Discussion
TRH is an important but underdiagnosed cause of refractory headaches that causes moderate-to-severe pain and usually necessitates local steroid injections for pain relief. It accounted for 3% of all patients and nearly 10% of those having refractory headaches in our clinic. Our study helps to further understand about the etiology and reasons for refractoriness of various poorly understood entities causing TRH such as PTRHs and IT.
In most previous studies and a recently published meta-analysis, only patients having CTRHs have been included. Our patients with CTRH are comparable with the available data. Patients having ETRHs have not been studied earlier, so our ETRH group could only be compared with data available on episodic migraine or TTH. The mean age of our subjects of 36.45 years is lower than that reported in the meta-analysis, but we had similar female preponderance, that is, 77.35%. 6 In our study, CTRH was found to be significantly associated with acute–subacute onset, bilateral, TTH-like headache, diplopia, and refractoriness to prophylaxis, whereas ETRH was associated more with insidious onset, unilateral, migraine-like headaches, prior history of headaches, and good response to prophylaxis (p < 0.05). Bilateral headaches were reported by nearly half of our patients, an observation similar to the study conducted by Smith et al. 7 Similar to earlier studies, we too found that nausea, photophobia, giddiness, and prior history of headaches were not common in patients with CTRH. Similar to our study, previous studies have also reported a variable (10–60%) prevalence of prior history of headaches in patients with CTRH. 6,7 In our CTRH group, binocular diplopia was reported by nearly 50% patients, local swelling was noted only with trochleitis and MRI orbit abnormalities only with ST. These findings are similar to the study conducted by Smith et al. 7
Refractoriness to prophylaxis was found to be associated with acute–subacute onset, TTH-like headaches, local swelling, and presence of autoantibodies. All patients with ST and two-thirds with IT were refractory to prophylaxis compared to only one-fourths of patients having ETRH and PTRH. We found that as much as one-thirds of patients with IT had good response to prophylaxis, a figure higher than that reported in the literature. A likely explanation for this observation is that most of these studies included only severe cases requiring local injections. 2,6,7 Local injections were found necessary in less than half of our refractory patients.
Autoantibodies were detected in 18 of 53 (39.96%) patients, a higher incidence than reported in previous studies. Autoantibodies were seen more often in nonresponders than responders (14 of 25 vs. 4 of 28). They were detected not only in IT but also in episodic and PTRHs. Over 70% of antibody-positive nonresponders showed good response on treatment with immunosuppressants. This indicates that undiscovered autoimmunity could be a reason for presumed refractoriness of TRHs. Autoimmunity was found to be associated with acute–subacute onset, bilateral, continuous, TTH-like constricting headache, longer disease duration, higher disease burden, and refractoriness to prophylaxis. Our findings of autoimmunity in episodic and PTRHs are supported by known benefit of steroids in recurrent acute migraine attacks. Some recent studies have also proposed autoimmune basis for exacerbations of primary headaches. 8,9
In nonresponders having IT, 5 of 6 were detected to have autoantibodies and 3 of 6 had underlying primary headaches. These patients showed improvement with treatment of autoimmunity and with appropriately chosen headache prophylaxis. None of the responders having IT, however, had autoantibodies and only one patient had prior headache history. It is apparent that all cases of IT might not be truly idiopathic. Failure to recognize underlying autoimmunity or primary headache disorder might be a reason for its presumed refractoriness. PTRH and ETRH being primary headache disorders respond generally well to headache prophylaxis. However, among nonresponders having PTRH and ETRH, nearly one-thirds were detected to have autoantibodies and showed improvement on treatment with immunosuppressants. Prior history of headaches is also less frequently observed in nonresponder PTRH than responders (one-third vs. two-third). Therefore, refractoriness of PRTH and ETRH can be mostly attributed to unrecognized underlying autoimmunity.
We propose that all of these entities causing TRHs might belong to a spectrum starting with ETRH associated with migraine being at one end and immune-mediated secondary trochleitis at the other end. Their etiologies might have differing contributions from autoimmunity and primary headache disorders. In summary, PTRH appears to be more like ETRH, being predominantly a primary headache disorder and its refractoriness might be attributable to underlying autoimmunity. IT presumably lies between PTRH and ST, as it has a higher incidence of autoimmunity and a minor contribution of primary headaches than PTRH. The local tissue inflammation of antibody-positive IT is known to be restricted to trochlea and superior oblique tendon, a fact that can be used to differentiate it from ST where the inflammation extends beyond these structures in the orbit. Thus, a prompt identification and treatment of underlying autoimmunity and primary headaches can be crucial for successful management of TRHs.
Limitations of the study might be a possible selection bias and lack of standardized protocol for investigations and treatment.
Strengths of the study
Large number of patients with long-term follow-up data of treatment outcomes.
Introduces the concept of ETRH to better understand the entire spectrum of TRHs.
Provides insight into etiopathogenesis of TRHs, especially the poorly understood entities such as PTRH and IT.
Provides a new strategy for evaluation and treatment of refractory TRHs.
Recommendation
TRH can present as episodic or continuous medial orbital pain, aggravated by eye movements or reading. Generally, ETRH is responsive, while CTRH is refractory to prophylaxis based on the nature of headache and prior headache history. Patients with CTRH have modest benefit with NSAIDS or TTH prophylaxis, but occurrence of episodic exacerbations with TM often improved with the addition of migraine prophylaxis. Local steroid injections are generally required in patients who do not respond to prophylaxis. Nonresponders to prophylaxis should be evaluated and treated for underlying autoimmunity and primary headache disorder. Testing for autoimmunity is recommended in patients with refractory TRHs, especially those with acute–subacute onset of progression. Thus, identification of the etiological contributions of underlying autoimmunity and primary headache disorder can be crucial for achieving satisfactory outcomes in patients with refractory TRHs.
Clinical implications
TRH, an important but underdiagnosed cause of refractory headaches, has been ascribed to poorly understood entities such as PTRH and IT.
In our study, identification and treatment of unrecognized autoimmunity and primary headache disorder led to improved outcomes in many refractory patients. Therefore, identification of underlying autoimmunity and any primary headache disorder is recommended in refractory patients with TRHs.
Our findings shed new light on the etiopathogenesis of TRHs.
Footnotes
Author contributions
PO had full access to all data in this study and takes responsibility for the integrity of the data and the accuracy of analyses. PO contributed to study conception and design; all authors contributed to acquisition, analysis, and interpretation of data; drafting of the manuscript; approved the version to be published; critical revision of the manuscript for important intellectual content. PO, AV, and BS were involved in statistical analysis. PO and YJ helped in administrative, technical, and material support. PO and BS supervised the study progress.
Data sharing statement
Data used in this manuscript will be made available to qualified health care professionals in accordance with Partners Human Research Committee policies.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Notes
Research ethics and patient consent.
Approval for the study was obtained from the local ethics committee of Navi Mumbai.
