Abstract
Objective:
This trial explored the therapeutic utility and safety of external trigeminal neurostimulation (eTNS) as a preventive treatment in patients suffering from chronic migraine (CM).
Methods:
It was a monocenter, prospective, open-label, pilot trial conducted at the University of Colorado, Anschutz Medical Campus (Aurora, CO, USA). Participants were adult patients with a history of CM meeting International Classification of Headache Disorder-3 beta (2013) diagnostic criteria with or without medication overuse. After a 1-month baseline period, the patients still fulfilling the inclusion criteria applied at least one daily 20-min session of eTNS for 3 months. Primary outcomes were mean monthly changes in frequency of headache days and in overall acute headache medication intake.
Results:
Eighty patients were assessed for eligibility, 73 were included in the baseline period after which 58 entered the treatment period and were included in the intention-to-treat analysis. Compared to baseline, frequency of headache days decreased by −3.12 days (−16.21%, p < 0.001) and acute medication intake decreased from 26.33 to 18.22 (−30.81%, p < 0.001) during the third month of treatment. Twenty-six patients reported 47 minor adverse events, of which only 2 were related to the use of the device (skin irritation under the electrode and headache worsening with vertigo).
Conclusions:
This trial suggests that eTNS is safe and effective as prophylactic treatment for CM and indicates that a randomized sham-controlled trial is worthwhile.
Trial registration:
ClinicalTrials.gov (identifier: NCT02342743).
Keywords
Introduction
Chronic migraine (CM) is the most disabling form of migraine, individualized as a migraine subtype (code 1.3) in the most recent International Classification of Headache Disorder (ICHD)-3 beta (2013) and ICHD-3 (2018). 1,2 According to the latter, CM patients have at least 3 months ≥ 15 headache days per month, of which at least 8 days fulfill the criteria for migraine or are believed by the patient to be migraine at onset and relieved by a triptan or ergot derivative. A subgroup of CM patients has continuous headache and is tentatively classified in the ICHD-3 appendix as “chronic migraine with continuous pain” (code A1.3.2) as long as they fulfill the criteria for CM and have no pain-free periods >3 h on ≥5 days per month, while such periods occur in “chronic migraine with pain-free periods” (code A1.3.1). In several trials, treatment outcome was better in noncontinuous than in continuous headache. 3 –5
CM affects up to 3% of the general population and is frequently associated with acute antimigraine medication overuse headache (MOH) (ICHD-3, code 8.2). In the strict sense, it can thus be diagnosed only if persisting after drug withdrawal, which is the case in approximately 50% of patients. 6 CM is thus a complex and dynamic condition; 3% of episodic migraine (EM) patients per year evolve to CM, 7 while an estimated 25% of CM patients reverse to EM over 2 years. 8 Observational studies have shown differences in clinical features, comorbidity profiles, pathophysiological abnormalities, and treatment response between EM and CM. 5 For instance, with regard to treatment response, there were only 20% of CM patients with ≥50% reduction in monthly migraine days in a topiramate trial, 9 as compared to 45.3% in the pooled trials of EM patients. 10 Hepp et al. 11 found that 50% of CM patients abandon oral preventive drugs within 2 months because of the lack of efficacy and/or because of adverse effects, while adherence in EM reached up to 80% at 3 months in several studies. 12 In the pivotal PREEMPT trials in CM, pooled data show that monthly migraine days after 24 weeks decreased by 43% with onabotulinumtoxinA (n = 680) and by 33% with placebo injections (n = 696), which is a modest therapeutic gain of 10%. 13 OnabotulinumtoxinA stands out, however, by its excellent tolerability and safety. 14 Taken together, available trial data and clinical practice clearly indicate a need for more efficient and well-tolerated preventive therapies in CM.
Neurostimulation methods have raised hope for a better management of chronic headaches. Among the minimally invasive techniques, percutaneous occipital nerve stimulation (ONS) has not held its promises in CM, yielding negative or marginally beneficial results in randomized sham-controlled trials, contrary to chronic cluster headache, where it benefits around 60% of patients. 15 ONS is furthermore endowed with numerous device-related adverse events (AEs). Noninvasive neurostimulation methods have the advantage of being quasi devoid of side effects. 16 Cervical vagus nerve stimulation with the Gammacore® device is up to now the only noninvasive method that was assessed for CM in a sham-controlled trial, the EVENT trial. 17 It did not reach the primary endpoint of headache day reduction after 2 months of daily treatment, but a small subgroup of patients who continued the stimulation in an open-label phase had an average reduction of −7.9 monthly headache days after 8 months. External trigeminal neurostimulation (eTNS) with the Cefaly® device was found superior to sham stimulation for the prevention of EM in the PREMICE trial 18 and for the abortive attack treatment in the ACME trial. 19 Although, hitherto, no large trial of eTNS was performed in CM, there is circumstantial evidence that it could also be of benefit in these more disabled patients. In the PREMICE trial, indeed, a subanalysis showed that the effect size of eTNS was greatest in patients with a higher attack frequency. 20 By the time this study was initiated, there were no published data on eTNS in CM. Meanwhile, two open-label studies in a small total sample of 29 CM patients have reported a favorable effect of eTNS. 21,22
These elements indicate the interest for exploring the therapeutic potential and safety of eTNS with the Cefaly® in a larger population of CM patients before eventually considering a randomized sham-controlled trial. The objective of this open-label trial was therefore to assess the efficacy and safety of eTNS as a preventive treatment in patients suffering from CM.
Methods
Trial design
This study was a monocenter, prospective, open-label, pilot clinical trial conducted at the University of Colorado, Anschutz Medical Campus (Aurora, CO, USA). The protocol was reviewed and approved by the Colorado Multiple Institutional Review Board (protocol 14-2159). Written informed consent was obtained from all participants. The trial has been registered on ClinicalTrials.gov (identifier: NCT02342743).
During the screening visit (visit 1), participants were assessed for eligibility and were tested for tolerance to the neurostimulation during a training eTNS session. Only patients who were able to bear the paraesthesias induced by the neurostimulation for 6 min, that is, who had a nociceptive threshold >7 mA, were included in a 1-month baseline period and were asked to fill in a paper headache diary reporting headache intensity (on a scale of 1 = mild to 3 = severe), time when the headache started and stopped, accompanying symptoms (nausea, vomiting, photophobia, and phonophobia), and acute antimigraine medication intake during this period.
At the end of the baseline period (visit 2), the headache diaries were collected and analyzed. Patients who still fulfilled the criteria for CM (i.e. having at least 15 headache days and 8 migraine days during the baseline month) were included in the treatment period for 3 months and were asked to continue filling in the diary and to apply every day one, or two, 20-min sessions of eTNS with the Cefaly® device (CEFALY Technology, Seraing, Belgium). Patients were instructed on how to apply the device. Instructions were the same as those given in the PREMICE trial 18 and so were the stimulation parameters: pulse width 250 μs, frequency 60 Hz, incremental increase in stimulation intensity up to 16 mA, and session duration of 20 min.
Research personnel communicated with subjects regularly to assess any immediate safety concerns. The patients were asked to report spontaneously the occurrence of any adverse effect in an adverse effect report form on a daily basis.
At the end of the treatment period (visit 3), headache diaries were collected and reported adverse effects listed in the clinical report form. The devices were retrieved to assess number of sessions and time of use thanks to an inbuilt software.
Eligibility criteria
Patients had to fulfill the following criteria: age 18 to 65 years, a history of CM (ICHD-3 beta 1.3) with (ICHD-3 beta 8.2) or without MOH, no other headache disorder, a Beck’s Depression Inventory score ≤24, not pregnant, not lactating, and not <6 months postpartum. Oral preventive antimigraine medications were allowed as long they were started ≥3 months before baseline and their dose remained unchanged during the entire trial. Subjects treated with botulinum toxin within the last 4 months were not included. Intake of acute antimigraine rescue medication was allowed at any time of the study and had to be reported in the headache diary. Both subjects with continuous pain (ICHD-3 beta A1.3.2) and those with pain-free periods of >3 h on ≥5 days per month (ICHD-3 beta A1.3.1) were included, as long as they met the criteria for CM.
Outcome measures
The following outcome measures were computed per patient from the data reported in the diaries: – frequency of headache days, defined as the number of calendar days with at least one headache episode; – acute medication intake, computed as the number of abortive antimigraine medication doses taken; – frequency of migraine days, defined as the number of headache days meeting ICHD-3 beta criteria for migraine headache 1.1 or 1.2 as well as headache days where the intensity was 1 and there was acute medication intake; – frequency of moderate/severe headache days, defined as the number of calendar days with at least one headache episode with intensity of 2 or 3; – cumulative headache hours on headache days; – frequency of headache episodes, defined as the number of patient-reported headache attacks; – mean headache intensity.
Changes were expressed as the difference between the baseline measures and the measures during the third month of the treatment period.
Primary outcomes were mean changes in frequency of headache days and in overall acute headache medication use (all pharmacological classes).
Secondary outcomes were mean changes in frequency of migraine days, in frequency of moderate/severe headache days, in monthly cumulative headache hours, in frequency of headache episodes, and in average headache intensity, as well as the 50 and 30% responder rates for migraine days (i.e. the percentage of patients having a reduction in migraine days of at least 50 and 30%, respectively).
Data analysis and statistics
The sample size was computed based on the primary outcome measure, that is frequency of headache days. Based on the hypothesis that eTNS with the Cefaly® produces a 25% reduction in the number of headache days, inclusion of 50 patients (25 without and 25 with acute MOH) would allow to detect a significant change with sufficient statistical power.
All relevant general, safety, and efficacy data were descriptively summarized at each time point. Data analysis was conducted on an intention-to-treat (ITT) basis, that is, on all subjects who were included in the treatment period and used the Cefaly® device at least once. For each patient, the outcome was calculated according to all data available. Missing data were handled according to the last value carried-forward method. Of the 58 included patients, 10 (17%) did not return their diary at the end of the treatment period because they were lost to follow-up (n = 8) or because they withdrew from the study (n = 2) during this period. For these subjects, baseline data were carried forward for the treatment period.
A post hoc analysis was performed on the subgroups of patients with noncontinuous and with continuous headache.
Wilcoxon’s signed rank test for paired samples was used to compare outcome measures between the baseline and the third month of the treatment period.
Device safety and tolerance were assessed by the number of reported AEs and their severity.
Results
Patients repartition and demographic data
The repartition of patients is shown in Figure 1. Between February 2015 and April 2017, a total of 80 patients were assessed for eligibility; 73 patients were included in the baseline period; 58 patients entered the treatment period and were eligible for the ITT analysis. Among the latter, 34 (58.6%) had noncontinuous headache, while 24 (41.4%) suffered from continuous (permanent) headache, that is, headache fulfilling criteria for CM and not interrupted by pain-free periods of >3 h on ≥5 days/month unless these are attributed to drug treatment (ICHD-3 beta A1.3.2). Thirty-three patients (56.9%) fulfilled ICHD-3 beta criteria for acute MOH (code 8.2): 18 in the noncontinuous headache group and 15 in the other group. Demographic data are shown in Table 1. Among the 58 patients who were included in the treatment period, 25 were taking preventive antimigraine drugs; most of them (n = 16) were taking beta-blockers and/or anticonvulsants, 5 magnesium, and 4 antidepressants. The proportion of patients taking preventive drugs was not significantly different between those with continuous (n = 11/24) or noncontinuous headache (n = 14/34).
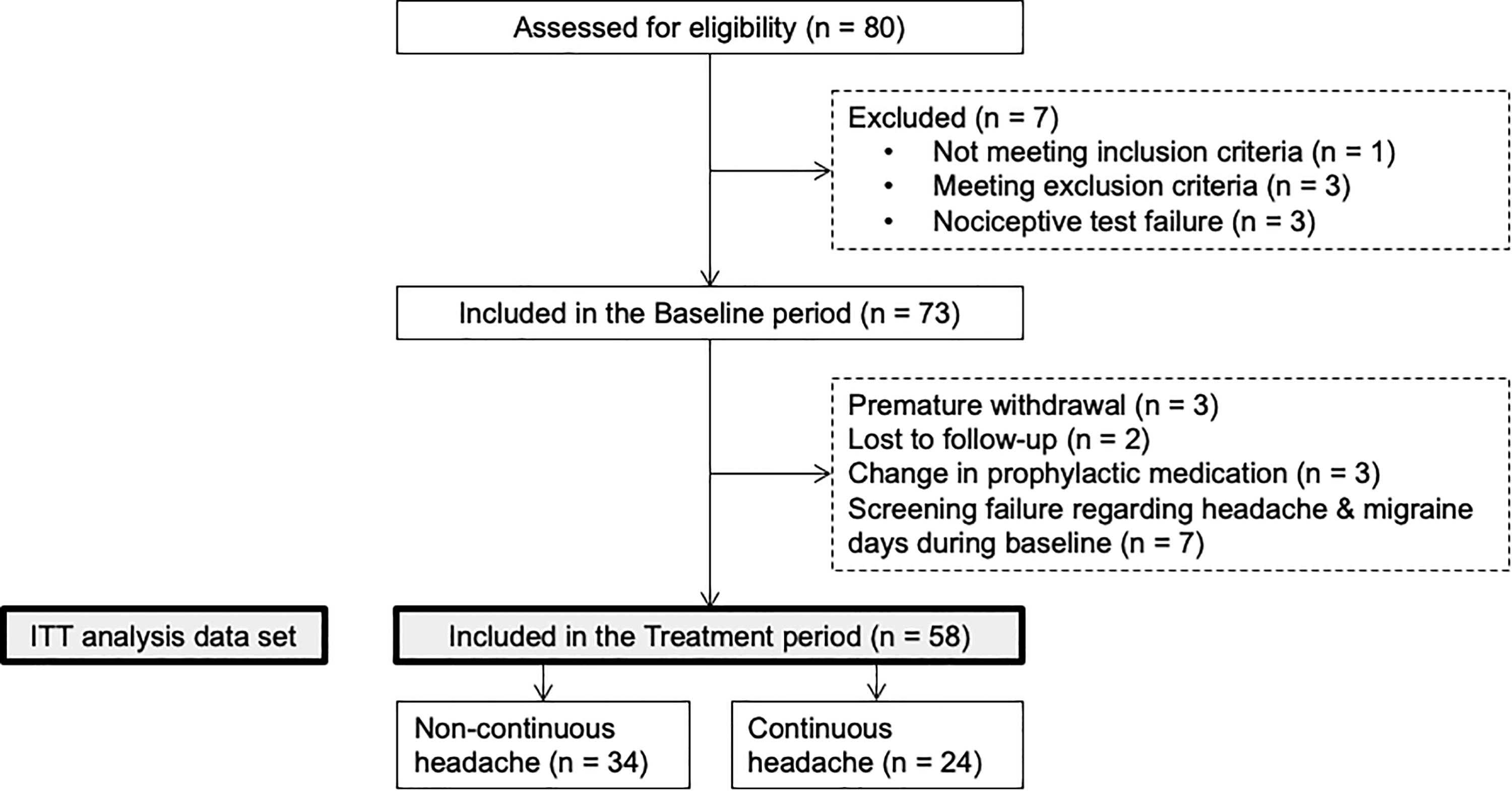
Repartition of patients. ITT: intention-to-treat.
Demographic data.a

MOH: medication overuse headache; SD: standard deviation.
aData are expressed as n, n (%) or mean ± SD.
Primary outcome measures
Changes in outcome measures between the 1-month baseline and the third month of eTNS with the Cefaly® are shown in Table 2.
Outcome measures before (baseline) and after 3 months of eTNS with the Cefaly® and mean change (absolute and relative) in outcome measures for the ITT population.a

eTNS: external trigeminal nerve stimulation; ITT: intention-to treat; SD: standard deviation.
aPrimary outcome measures and significant changes are highlighted in bold. Data are expressed as n (%) or mean ± SD.
b p < 0.001 (Wilcoxon’s signed rank test for paired samples).
d p < 0.05.
eAs some patients reported no medication use at baseline, relative change in medication was not calculable for them. The mean relative change in medication was therefore assessed based on the relative change between the mean baseline and treatment values and not as the mean of the relative changes of all patients.
Monthly frequency of headache days significantly decreased from 22.55 to 19.43 days (−16.21%, p < 0.001) in the total patient cohort. When considering only patients with noncontinuous headache, frequency of headache days was reduced by 24.38% (p < 0.001), while a reduction of only 4.62% was observed in patients with continuous headache (Figure 2 and Table 2). In the subgroup of 34 patients with noncontinuous headache, 15 (44.12%) had reversed to an EM pattern at the end of the treatment period. These patients had on average 17.2 headache days pretreatment and moved to 7.5 headache days on average posttreatment. All of them had a reduction of at least 5 headache days between the baseline and the third month of treatment, except one patient who had a 3-day reduction (from 16 to 13 headache days).
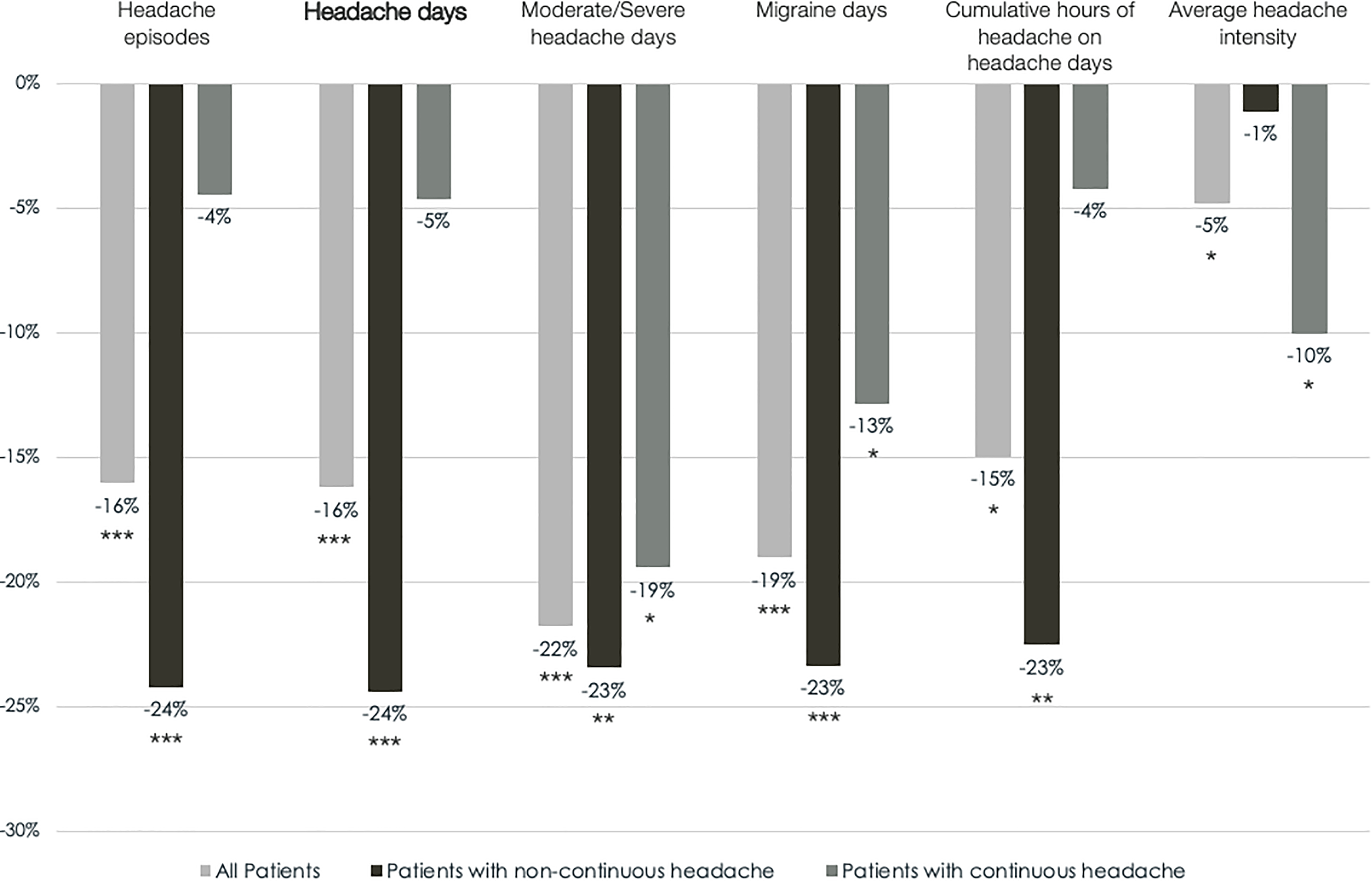
Mean relative change in outcome measures between the 1-month baseline and the third month of eTNS with the Cefaly® in the ITT population. *p < 0.05, **p < 0.01, and ***p < 0.001 (Wilcoxon’s signed rank test for paired samples). eTNS: external trigeminal nerve stimulation; ITT: intention-to-treat.
Mean monthly acute medication intake was significantly reduced from 26.33 to 18.22 (−30.81%, p < 0.001) (Figure 3 and Table 2). This reduction was greater in the subgroup of patients with continuous headache than in those with noncontinuous headache (−11.85 vs. −5.47). However, medication use at baseline was higher in patients with continuous headache (32.92 vs. 21.68) (Table 2). When considering separately triptans and nontriptans, intake was significantly reduced for triptans in patients with noncontinuous headache but the reduction did not reach the level of significance in those with continuous headache (Figure 3 and Table 2).
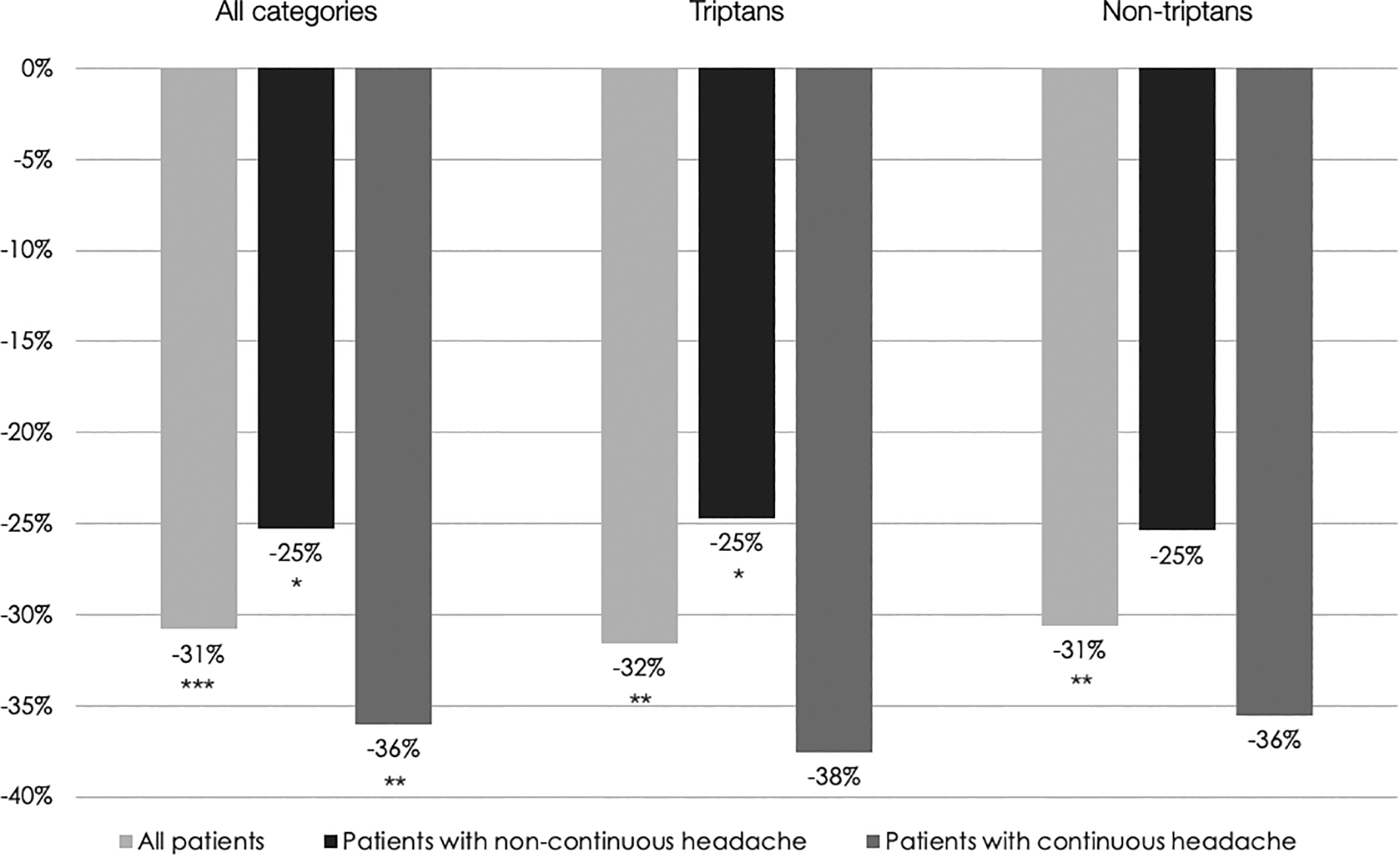
Mean relative change in monthly acute medication intake between the 1-month baseline and the third month of eTNS with the Cefaly® in the ITT population. *p < 0.05, **p < 0.01, and ***p < 0.001 (Wilcoxon’s signed rank test for paired samples). eTNS: external trigeminal nerve stimulation; ITT: intention-to-treat.
Secondary outcome measures
In the total group of patients, all secondary outcome measures were significantly improved after treatment (Figure 2 and Table 2). When only patients with noncontinuous headache were considered, the reduction in headache episodes, cumulative hours of headache, triptan and nontriptan consumption reached the level of significance, but reduction in average headache intensity and nontriptan intake did not (Table 2).
The 50% responder rate for migraine days in all subjects was 18.97%. The responder rate was higher in patients with noncontinuous headache, compared to those with continuous headache (29.41% vs. 4.17%). The 30% responder rate for migraine days was 24.14% overall, 38.24 and 4.17% in noncontinuous and continuous headache, respectively.
Medication overuse
In the 33 patients (56.90%) included in the ITT analysis and fulfilling ICHD-3 beta criteria for MOH (n = 18 with noncontinuous headache, n = 15 with continuous headache), frequency of headache days was reduced by 15.82% (vs. 16.21% in the whole cohort), of headache episodes by 15.42% (vs. 16.03%), of moderate/severe headache days by 26.67% (vs. 21.78%), and of migraine days by 21.15% (vs. 19.02%). Average headache intensity decreased by 8.30% (vs. 4.81%) and cumulative headache hours by 14.00% (vs. 14.95%). The treatment significantly reduced acute medication intake in the overuser group from 41.52 to 28.45 per month (p < 0.001). At the end of the third eTNS treatment month, the number of patients in the group with noncontinuous headache overusing acute medication had dropped from 18 at baseline to 10, that is, a 44% decrease.
Compliance
Compliance results are shown in Figure 4. On average, subjects included in the ITT analysis used the Cefaly® device during 46.44 h over the 3-month treatment period, which corresponds to 139.32 sessions. Subjects with continuous headache used the device more often than patients with noncontinuous headache.

Mean number of eTNS sessions performed by all patients and the two patient subgroups during the treatment period in the ITT population. Dashed lines indicate the total theoretical number for applying one and two sessions per day, respectively. eTNS: external trigeminal nerve stimulation; ITT: intention-to-treat.
Adverse events
A total of 78 minor AEs were reported by 34 patients: 29 AEs occurred during the baseline period and thus before use of the Cefaly® device; 2 AEs occurred on the day that the patients entered the treatment period but before they used the Cefaly®; and 47 AEs occurred during the treatment period. Only two AEs were related to the use of the device. One patient had skin irritation at the electrode site on the forehead. Another patient reported worsening headaches and vertigo and discontinued using the device. The latter AE was considered to be possibly related to the eTNS but without a definitive proof.
One serious AE occurred during the trial. A patient was hospitalized for worsening of myasthenia gravis and migraine headaches on the day she was included in the treatment period but before using the device. This serious AE was considered not related to the treatment and the patient resumed eTNS after discharge from the hospital.
Discussion
This study shows that 3-month daily eTNS with the Cefaly® device improves headache in patients suffering from CM. Regarding the primary outcome measures, eTNS significantly reduces the number of headache days by 16.21% and the intake of acute antimigraine drugs, both nonspecific and specific ones, by more than 30%. When considering the secondary outcomes, results show that eTNS significantly reduces the number of migraine days, days with moderate/severe headache, headache duration, and intensity. In 44% of patients with noncontinuous headache, eTNS switches the CM pattern to an episodic pattern. In the total patient group, eTNS with the Cefaly® decreased monthly migraine days by more than 50% in 18.97% of patients and by more than 30% in 24.14% of them.
The reduction in headache frequency and duration was clearly smaller for the subgroup of patients suffering from continuous headache (ICHD-3 beta A1.3.2). By contrast, the average intensity of headaches decreased significantly after eTNS in patients with continuous headache, but not in those with noncontinuous headache, and the former also had numerically greater reductions in acute medication intake. These findings suggest that, in the continuous headache subgroup, treatment with the Cefaly® clearly reduces pain intensity and abortive medication intake. The fact that these two CM subgroups may respond differently to therapeutic interventions was previously reported. 3 –5 Our results support the clinical distinction between the two subgroups, as proposed in the appendix of ICHD-3 beta (2013) and ICHD-3 (2018).
Acute medication overuse is a known chronifying factor for migraine. In our ITT patient cohort, 33 of 58 patients (56.90%) had medication overuse according to ICHD criteria. In this subgroup, the frequency of headache days decreased by 15.82%, the frequency of moderate/severe headache days by 26.67%, the frequency of migraine days by 21.15%, and the average headache intensity by 8.30%. Importantly, the acute medication use was highly significantly reduced. After eTNS, the number of overusers in the subgroup with noncontinuous headache was reduced by 44% (8/18), suggesting that the treatment may contribute to interrupt the vicious circle of medication overuse. For comparison, in the US CM trials with topiramate, 23 the subgroup of patients overusing any combination of acute drugs for >15 days per month had a reduction of monthly migraine days of −8.0, but this only tended to be different from placebo (−5.8 days, p = 0.081) and there was no significant difference in mean reduction of acute medication intake between the two treatment groups (p = 0.269). In a European topiramate trial in CM, 24 where the number of migraine days decreased by −3.5 days (vs. placebo +0.8 days) in patients overusing acute medication, there was no decrease in average monthly medication intake. In CM trials with onabotulinumtoxinA, the subgroup of patients overusing acute medication had a significant reduction in headache and migraine days. 25 In another trial with onabotulinumtoxinA, the primary outcome measure, reduction in headache days, was not significant in CM patients overusing acute medication, but the injection protocol was different from the PREEMPT one. 26
The present study was conceived as a pilot trial and its results must be taken with reservation because of the lack of a sham stimulation arm. The contribution of a placebo response can nonetheless be grossly estimated by comparing the results with those from other sham-controlled trials. In the PREMICE trial, 18 where the preventive effect of eTNS with the Cefaly® was assessed in EM, the reduction of headache days in the placebo group was clearly smaller (−3.42%) than the effect observed in our noncontinuous headache subgroup (−23.62%), but similar to that found in patients with continuous headache (−4.61%). Migraine days decreased by −4.89% in the sham arm of PREMICE, which differs clearly from the −22.62 and −13.14% reductions found, respectively, in our noncontinuous and continuous headache groups. Concordantly, acute medication use was increased by +0.43% in the placebo group of PREMICE, contrasting with reductions of −25.24 and −36.01% in the respective subgroups of the present study. During the blinded randomized phase of the EVENT trial that explored the preventive effect in CM of three daily cervical noninvasive vagus nerve stimulations with the Gammacore®, 17 the number of headache days during the second month of treatment decreased on average by 0.2 days in the sham arm, contrasting with –3.12 at month 3 in our total ITT patient group (−4.42 in patients with noncontinuous headache). Taken together, the outcomes in our pilot trial appear markedly better than the placebo response reported in two noninvasive neurostimulation studies. However, the difference in the trial protocol (open-label vs. placebo-controlled) limits this comparison. Nevertheless, the outcomes of our study strongly suggest that eTNS is effective for the preventive treatment of CM and indicate that a sham-controlled trial of eTNS with the Cefaly® in CM is not only necessary but also worthwhile.
While awaiting the results of such a trial, the treatment effect observed in our study can be tentatively compared to those found in other preventive migraine studies. In the abovementioned PREMICE trial that included EM patients, 18 migraine days decreased by −29.68% in the active arm, as compared to −22.62% in the present study; 50% responder rate for migraine days was 38.1% in PREMICE versus 29.4% in CM patients with noncontinuous headache included in the ITT analysis of the present trial. Corresponding 50% responder rates in CM are 10% for noninvasive vagus nerve stimulation, 17 22% for topiramate 9 and 38% for fremanezumab, 27 40% for erenumab, 28 the novel monoclonal antibodies, respectively, against calcitonin gene-related peptide (CGRP) and its receptor.
No safety concerns arose for eTNS with the Cefaly® in this trial. Triggering action potentials by electrical impulses is physiologically associated with paraesthesias that were thus not considered as an adverse effect. However, as mentioned before, patients who did not tolerate the stimulation during the training session because of painful paraesthesias were excluded from the trial (see below). Thirty-four patients reported a total of 78 minor AEs among which only 2 were device-related. Local skin irritation reported by one patient is a well-known side effect of eTNS and fully reversible or avoidable by an anergic electrode gel if due to allergy. Transient worsening of headache, as mentioned by one patient, has been reported previously 29 but not vertigo that occurred in the same patient. Relation of the latter with eTNS is uncertain. Overall, these results confirm the high safety level and excellent tolerance of the Cefaly® device, confirming those of a retrospective survey of 2.313 device users for migraine prevention of whom 4.3% reported minor AEs. 29 For comparison, in the EVENT trial of noninvasive Gammacore® vagus nerve stimulation for CM, 17 minor AEs were reported during the randomized phase by 24 of 30 patients of whom 6 had device-related events. In the OnabotulinumtoxinA PREEMPT 2 trial in CM, 30 116 of 347 subjects in the active group (33.4%) reported AEs related to the use of the drug and 12 subjects (3.5%) discontinued the study due to AEs. By contrast, topiramate induced adverse effects in 75% of CM patients, mostly paraesthesias (53%). 10
Our study has some limitations. An obvious one is its open-label design and the lack of pacebo arm, as already discussed above. We decided to exclude patients who were not able to tolerate the paraesthesias induced by the electrical stimulation for ≥6 min, that is, up to 7 mA of intensity, during the initial training session. Only three patients, however, were intolerant to the stimulation. Given the high prevalence of cutaneous allodynia in CM patients, 5 this suggests that eTNS may be well tolerated even in the presence of some degree of allodynia, but mechanical cutaneous allodynia was not tested in our study and is not comparable to electrically induced paraesthesias.
The fact that the number of daily eTNS sessions was not the same for all patients could be considered another weakness of our trial protocol, producing unnecessary variability. One important challenge in eTNS is compliance. Daily repetition of the treatment is necessary for optimal efficacy. We instructed, therefore, the subjects to have one to two sessions each day to maximize the chance of having at least one session performed per day. In practice, patients should have used the device at least once a day for 84 days, that is, a total of 28 h of stimulation, but they could have used it twice a day, that is, 168 sessions or 56 h of stimulation. Control of use with the in-built software of the Cefaly® device indicated that subjects were highly compliant and used on average the device during 46.44 h over the treatment period, which is more than the minimum required of 28 h and could have been favored by the instruction on how to use the device.
It would have been interesting to search for a correlation between the frequency of daily sessions and outcomes. Unfortunately, this was not possible in our study because the device recorded the total number of treatment sessions and the total time of use over the 3-month treatment period, but not the number of treatment sessions per day. In future studies, a two-arm trial protocol, one with patients instructed to use the device once a day and the other with an instruction for two daily sessions, would allow to determine if such a correlation exists.
To conclude, this open-label pilot trial suggests that eTNS with the Cefaly® device is safe and effective as prophylactic treatment for CM in adult patients. The treatment effect is greatest in patients with noncontinuous headache; it is hardly significant in those with continuous headache. The treatment is also effective in patients with acute medication overuse and allows overall an important reduction in acute antimigraine drug intake. These results indicate that a randomized sham-controlled trial is worthwhile. Such a trial should plan to stratify the results by continuous headache or not, and, if possible, by the number of daily treatment sessions applied.
Clinical implications
In this open-label, pilot trial of CM patients, daily eTNS with the Cefaly® device for 3 months significantly reduced headache days (−16.2%) and acute medication intake (−30.8%). The 30% responder rate was 38.2% in patients with noncontinuous headache, but only 4.2% in those with continuous headache. The effect was similar in patients with or without medication overuse. The treatment was safe and there were only two device-related AEs, both benign and transient. Compliance was excellent. The outcomes in this trial are superior to the placebo response expected with noninvasive neurostimulation and compare favorably with those published for cervical vagus nerve stimulation or for topiramate. A randomized sham-controlled trial is thus worthwhile.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB and KC have no conflict of interest. JS has received honoraria from CEFALY Technology as an external consultant; outside of the submitted work, he has received research grants from Electrocore and Medtronic, as well as personal fees from Teva and Novartis. SP is an employee of CEFALY Technology. None of the authors are on the Sponsor’s medical board.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from CEFALY Technology.
