Abstract
Introduction
Acute medication overuse is prevalent in patients with migraine.
Methods
In three phase 3, double-blind, randomized, placebo-controlled studies, patients with episodic migraine (EVOLVE-1 and EVOLVE-2) or chronic migraine (REGAIN) were randomized 2:1:1 to monthly subcutaneous injections of placebo or galcanezumab 120 or 240 mg for 3 or 6 months. This subgroup analysis evaluated mean changes in the number of monthly migraine headache days in each treatment among patients with versus without baseline acute medication overuse via mixing modelling with repeated measures.
Results
The percentages of patients with baseline medication overuse in placebo, galcanezumab 120-mg and 240-mg groups, respectively, were 19.4%, 17.3%, and 19.3% for EVOLVE-1/-2 (pooled; post hoc), and 63.4%, 64.3%, and 64.1% for REGAIN (a priori). Both galcanezumab doses demonstrated significant improvement compared with placebo for overall least squares mean change in monthly migraine headache days in patients with baseline medication overuse in both the episodic and chronic migraine studies (p ≤ 0.001). Furthermore, both galcanezumab doses reduced average monthly medication overuse rates compared to placebo (p < 0.001) in both patient populations with medication overuse at baseline.
Conclusions
Galcanezumab appears to be effective for the preventive treatment of episodic and chronic migraine in patients who overuse acute medications.
Keywords
Introduction
Medication overuse headache has an estimated worldwide incidence of 1% to 2% in individuals with migraine (1), affecting approximately 58 million people globally (2). The overuse of acute medications, including simple and combination analgesics, triptans, and opioids, by people with migraine may contribute to the development and/or maintenance of medication overuse headache (3). Diagnostically, medication overuse headache is defined as headache occurring ≥15 days per month in a patient with a pre-existing primary headache and developing as a consequence of regular overuse of acute or symptomatic headache medication (on ≥10 or ≥15 days per month, depending on the medication) for > 3 months (4).
Pooled analyses of the PREEMPT (Phase 3 Research Evaluating Migraine Prophylaxis Therapy) clinical program evaluating onabotulinumtoxinA suggest preventive medications can be effective in patients with chronic migraine and acute medication overuse (5–7), even in the absence of deliberate attempts at limiting or withdrawing the overused medication. In addition, a systematic review of different treatment strategies for medication overuse headache concluded that adding preventive medication to the withdrawal of overused medications led to better improvements than early discontinuation alone (8). More recently, a randomized clinical trial that directly compared treatment modalities found withdrawal of acute medication in combination with initiating preventive medication constituted a more effective treatment for medication overuse headache than either approach alone (9).
Galcanezumab is a humanized monoclonal antibody that selectively binds to the calcitonin gene-related peptide (CGRP) ligand and blocks its binding to the CGRP receptor (10,11); it is approved for the preventive treatment of migraine in adults. Three phase 3 studies demonstrated that monthly subcutaneous injections of galcanezumab 120 mg and 240 mg significantly reduced the number of monthly migraine headache days in patients with episodic migraine (EVOLVE-1 and EVOLVE-2) or chronic migraine (REGAIN) compared with placebo over 6 or 3 months of treatment, respectively (12–14). In each of these studies, galcanezumab was safe and well tolerated for the respective study population (12–14). This subgroup analysis of the three studies aimed to investigate the efficacy of galcanezumab in patients with episodic or chronic migraine and medication overuse.
Methods
Study design and patients
The study designs and eligibility criteria for EVOLVE-1 and -2 and REGAIN have been published previously (12–14). These studies were conducted in accordance with the International Conference on Harmonization Guidelines for Good Clinical Practice, the Declaration of Helsinki, and approved by each institution’s ethical review board. Patients provided written informed consent before enrolment. Each study has been registered with ClinicalTrials.gov, identifiers NCT02614183, NCT02614196, and NCT02614261, respectively.
Randomization and blinding
The randomization and blinding procedures for EVOLVE-1 and -2 and REGAIN have been published previously (12–14). Patients with episodic migraine (4–14 monthly migraine headache days; EVOLVE-1 and -2) and chronic migraine (≥15 monthly headache days, of which at least eight must have the features of migraine headache, for >3 months; REGAIN) were randomized 2:1:1 to monthly subcutaneous injections of placebo or galcanezumab 120 mg or 240 mg for 6 months (EVOLVE-1 and -2) or 3 months (REGAIN).
Assessments
The assessments and outcome measures for EVOLVE-1 and -2 and REGAIN have been published previously (12–14). This subgroup analysis evaluates patients with episodic or chronic migraine treated with galcanezumab (120 mg or 240 mg) versus placebo, and presents the overall mean change from baseline in monthly migraine headache days, mean monthly reduction in migraine headache days with acute medication use, the proportion of galcanezumab-treated patients who achieved ≥50% reduction in monthly migraine headache days, and the proportion of patients with medication overuse after randomization.
This analysis used data from patients with and without medication overuse based on frequency of acute headache medication intake during the baseline period, as defined by criteria according to International Headache Society guidelines for chronic migraine clinical trials and similar to those previously used in a study of onabotulinumtoxinA (7,15). Medication overuse was defined as the use of any of the following during the prospective 30-day baseline period as reported in the electronic patient-reported outcome diary: i) any triptan for ≥10 days, ii) any nonsteroidal anti-inflammatory drug (NSAID) or aspirin for ≥15 days, iii) any acetaminophen/paracetamol for ≥15 days, iv) any ergot or ergotamine derivative for ≥10 days, v) any combination drug containing two or more of the above medication classes for ≥10 days, or vi) a total of ≥10 days of drug use from at least two of the above categories. Opioid use for acute treatment of headache was limited to no more than 3 days per month during the initial studies. Patients with a diagnosis of medication overuse headache in the 3 months preceding screening were excluded from the EVOLVE studies, but not from the REGAIN study.
Statistical analyses
The statistical analysis methods for EVOLVE-1 and -2 and REGAIN have been published previously (12–14). Stratification of randomization by acute headache medication overuse (yes/no) was a priori for REGAIN, and this subgroup analysis was planned. Efficacy data from EVOLVE-1 and -2 were pooled and the subgroup analysis was post hoc. Subgroup analyses with treatment and subgroup interaction terms for comparison across subgroups for continuous endpoints, change from baseline in monthly migraine headache days, and change from baseline in monthly migraine headache days with acute medication use, were performed using a linear mixed model repeated measures analysis (identity link function). A subgroup analysis was also performed for binary endpoints, the proportion of patients with ≥50% reduction in monthly migraine headache days and the proportion of patients with medication overuse after randomization, using a generalized linear mixed model with a logit link function. All patients included in statistical analyses had received at least one dose of investigational product. Treatment effects from linear mixed models were presented as mean differences versus placebo, whereas effects from generalized linear mixed models were presented using odds ratios and associated 95% confidence intervals (95% CIs). The proportion of patients who changed status from baseline medication overuse to non-overuse of medication by acute medication categories (NSAIDs/aspirin, triptans, and multiple drugs) was descriptively summarized for each treatment group at each month. All statistical analyses were performed using SAS software (SAS Institute Inc., Cary, NC, USA), and assumed a two-sided significance level of 5%.
Results
Patient disposition and baseline characteristics
Figure 1 details the disposition of patients with baseline medication overuse. Among randomized patients, a total of 334 patients had baseline medication overuse in EVOLVE-1 and -2 (173/894 (19.4%) in the placebo group, 77/444 (17.3%) in the galcanezumab 120-mg group, and 84/435 (19.3%) in the galcanezumab 240-mg group), and a total of 708 patients had baseline medication overuse in REGAIN (353/557 (63.4%) in the placebo group, 178/277 (64.3%) in the galcanezumab 120-mg group, and 177/276 (64.1%) in the galcanezumab 240-mg group).

Disposition for patients with medication overuse.
Baseline demographic and clinical characteristics were generally well balanced between treatment groups in EVOLVE-1 and -2 and REGAIN (Tables 1 and 2); however, among patients with episodic migraine, mean migraine headache days per month and mean number of headache days were higher in patients with medication overuse at baseline than in those without (12 ± 2 vs. 9 ± 3, and 13 ± 3 vs. 10 ± 3, respectively). Rates of medication overuse at baseline varied somewhat by acute medication class. Among EVOLVE-1 and -2 episodic migraine patients, rates of overuse at baseline were higher for triptans and multiple drugs than for NSAIDs/aspirin (Table 1). Among REGAIN chronic migraine patients, rates of overuse at baseline were highest for triptans, followed by NSAIDs/aspirin, and multiple drugs (Table 2).
Baseline demographic and clinical characteristics for patients with episodic migraine (EVOLVE-1 and -2).

Note: Data are n (%) unless otherwise indicated.
MIDAS: migraine disability assessment; MSQ RF-R: migraine-specific quality of life questionnaire role function-restrictive; N/App: not applicable; PGI-S: patients’ global impression of severity; SD: standard deviation.
an = 171
bn = 82
cn = 716
dn = 366
en = 348
fn = 172
Baseline demographic and clinical characteristics for patients with chronic migraine (REGAIN).

Note: Data are n (%) unless otherwise indicated.
MIDAS: migraine disability assessment; MSQ RF-R: migraine-specific quality of life questionnaire role function-restrictive; N/App: not applicable; PGI-S: patients’ global impression of severity; SD: standard deviation.
an = 173
bn = 98
cn = 197
Efficacy analyses
Among patients with baseline medication overuse, both galcanezumab doses demonstrated a statistically significant improvement compared with placebo (p < 0.001) in overall mean change from baseline in monthly migraine headache days (Table 3). In EVOLVE-1 and -2, the overall least square mean change over the 6-month study period among patients with baseline medication overuse was placebo −2.7, galcanezumab 120 mg −6.3, and galcanezumab 240 mg −5.8; the improvements among galcanezumab-treated patient groups were statistically significant versus placebo (p < 0.001 for both 120-mg and 240-mg doses vs. placebo) (Table 3). Among patients without baseline medication overuse, the overall least square mean change over the 6-month study period was placebo −2.5, galcanezumab 120 mg −4.1, and galcanezumab 240 mg −4.0; again, the improvements in overall mean change from baseline in both galcanezumab-treated groups were statistically significant versus placebo (p < 0.001 for both galcanezumab doses vs. placebo) (Table 3). In the EVOLVE studies, the treatment-by-subgroup interaction for medication overuse-yes versus medication overuse-no was significant for galcanezumab-treated patients versus placebo (p < 0.001 for galcanezumab 120 mg, p = 0.001 for galcanezumab 240 mg).
Change from baseline in the number of monthly migraine headache days.
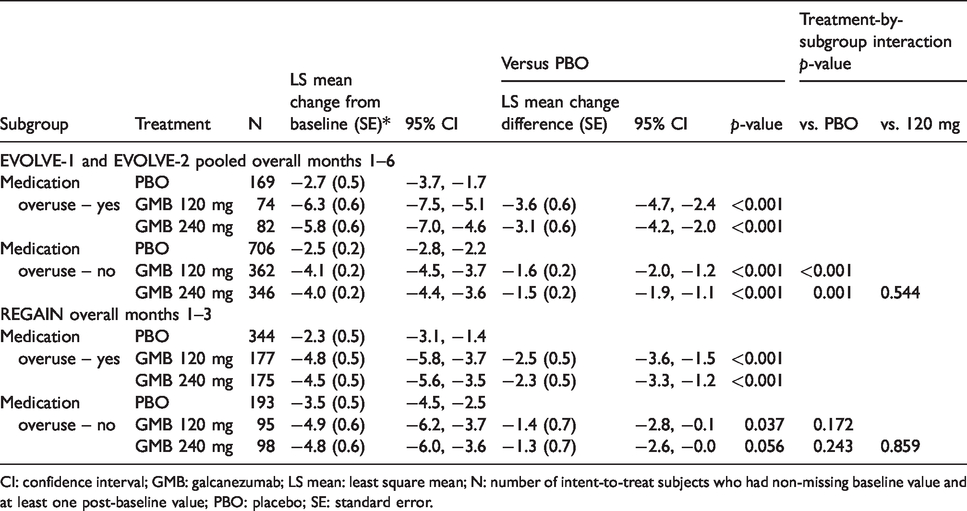
CI: confidence interval; GMB: galcanezumab; LS mean: least square mean; N: number of intent-to-treat subjects who had non-missing baseline value and at least one post-baseline value; PBO: placebo; SE: standard error.
In REGAIN, the overall least square mean change over the 3-month study period among patients with baseline medication overuse was placebo −2.3, galcanezumab 120 mg −4.8, and galcanezumab 240 mg −4.5; these improvements were also statistically significant for galcanezumab-treated patients versus placebo (p < 0.001 for both galcanezumab doses vs. placebo) (Table 3). Among patients without baseline medication overuse, the overall least square mean change over the 3-month study period was placebo −3.5, galcanezumab 120 mg −4.9, and galcanezumab 240 mg −4.8; this change was statistically significant in the galcanezumab 120-mg dose group versus placebo (p = 0.037), but not for the galcanezumab 240-mg dose group (p = 0.056) (Table 3). No significant treatment-by-subgroup interaction was observed for galcanezumab-treated patients versus placebo (p = 0.172 for galcanezumab 120 mg and p = 0.243 for galcanezumab 240 mg vs. placebo).
Onset of efficacy (the earliest month in which a statistically significant improvement in change from baseline of monthly migraine headache days was observed and maintained during treatment) occurred during month 1 for both galcanezumab dose groups in all three studies (Figure 2). At month 1, the least square mean change from baseline in EVOLVE-1 and -2 among patients with baseline medication overuse was placebo −2.0, galcanezumab 120 mg −5.6, and galcanezumab 240 mg −4.9 (p < 0.001 vs. placebo for both 120-mg and 240-mg doses) (Figure 2(a)). Mean change from baseline at month 1 among patients without medication overuse was placebo −1.3, galcanezumab 120 mg −3.4, and galcanezumab 240 mg −3.0 (p < 0.001 vs. placebo for both 120-mg and 240-mg doses) (Figure 2(c)). In REGAIN at month 1, the least square mean change from baseline among patients with baseline medication overuse was placebo −1.5, galcanezumab 120 mg −4.0, and galcanezumab 240 mg −4.1 (p < 0.001 vs. placebo for both 120-mg and 240-mg doses) (Figure 2(b)). Mean change from baseline at month 1 among patients without medication overuse was placebo −2.3, galcanezumab 120 mg −4.1, and galcanezumab 240 mg −4.3 (galcanezumab 120 mg: p = 0.005 vs. placebo; galcanezumab 240 mg: p = 0.003 vs. placebo) (Figure 2(d)).

Least squares mean change from baseline ± standard error in number of monthly headache days for the overall study durations in patients with episodic migraine (EVOLVE-1 and -2) and chronic migraine (REGAIN) with medication overuse at baseline ((a) and (b)) and without medication overuse at baseline ((c) and (d)).
Compared with placebo patients, galcanezumab-treated patients had significantly greater mean reductions in monthly migraine headache days with acute medication use, regardless of baseline medication overuse status. In EVOLVE-1 and -2, for patients with medication overuse, the overall mean reduction (95% CI) in monthly migraine headache days from month 1 to month 6 was placebo −2.7 (−3.7, −1.8), galcanezumab 120 mg −6.3 (−7.4, −5.2), and galcanezumab 240 mg −5.9 (−7.0, −4.8); for patients without medication overuse the reduction was placebo −1.9 (−2.1, −1.6), galcanezumab 120 mg −3.3 (−3.6, −3.0), and galcanezumab 240 mg −3.2 (−3.6, −2.9). There was a treatment-by-subgroup interaction for medication overuse-yes versus medication overuse-no observed for galcanezumab-treated patients versus placebo (p < 0.001 for both galcanezumab dose groups vs. placebo).
In REGAIN, for patients with medication overuse the reduction in monthly migraine headache days with acute medication use was placebo −2.5 (−3.3, −1.6), galcanezumab 120 mg −5.3 (−6.3, −4.3), and galcanezumab 240 mg −4.7 (−5.8, −3.7); for patients without medication overuse this reduction was placebo −1.5 (−2.4, −0.5), galcanezumab 120 mg −3.5 (−4.6, −2.3), and galcanezumab 240 mg −3.1 (−4.2, −1.9). No significant treatment-by-subgroup interaction was observed for galcanezumab-treated patients versus placebo (p = 0.229 for galcanezumab 120 mg and p = 0.301 for galcanezumab 240 mg vs. placebo).
In EVOLVE-1 and -2 and REGAIN, a significantly greater proportion of patients who received galcanezumab 120 mg or galcanezumab 240 mg achieved a ≥50% reduction in monthly migraine headache days compared with patients who received placebo (p < 0.001), irrespective of whether or not patients had medication overuse at baseline. The estimated percentage of patients with medication overuse who achieved a ≥50% reduction in monthly migraine headache days in the placebo, galcanezumab 120 mg, and galcanezumab 240 mg groups were 28.0% (95% CI 23.2%, 33.3%), 59.5% (51.1%, 67.5%), and 59.3% (51.2%, 66.9%), respectively, in EVOLVE-1 and -2 (Figure 3(a)), and 13.8% (10.7%, 17.6%), 27.1% (21.6%, 33.5%), and 27.4% (21.8%, 33.8%), respectively in REGAIN (Figure 3(b)).

Estimated percentage of patients with (a) episodic migraine (EVOLVE-1 and -2) and (b) chronic migraine (REGAIN) who had a 50% reduction in monthly migraine headache days, stratified by presence/absence of medication overuse at baseline. Error bars depict 95% confidence intervals.
The percentage of patients with medication overuse significantly decreased following galcanezumab administration relative to placebo (p < 0.001). On average across 6 months in EVOLVE-1 and -2, patients who received placebo observed a 64.4% (95% CI 59.4%, 69.1%) reduction in medication overuse rate, while patients who received galcanezumab 120 or 240 mg observed reductions of 85.4% (79.2%, 90.0%) and 81.0% (74.5%, 86.1%), respectively (Figure 4(a)). On average, across 3 months in REGAIN, the medication overuse rate decreased in placebo patients by 29.6% (95% CI 25.2%, 34.5%) while patients who received galcanezumab 120 or 240 mg observed reductions of 47.9% (41.2%, 54.8%) or 47.7% (41.0%, 54.5%), respectively (Figure 4(b)).

Estimated percentage reduction of patients with medication overuse from baseline after randomization in patients with episodic migraine (EVOLVE-1 and -2) (a) and chronic migraine (REGAIN) (b) for the overall study durations. Error bars depict 95% confidence intervals.
Transition from acute medication overuse to non-overuse of medication
Galcanezumab reduced the number of patients with acute medication overuse (Figures 4 and 5). The clinically meaningful difference in rates of medication overuse was driven by changes in triptan overuse. In EVOLVE-1 and -2, the percentage of patients with episodic migraine who transitioned from triptan overuse to non-overuse status at 1 month nearly doubled in galcanezumab-treated patients at both doses compared to placebo, and of the 85 galcanezumab-treated patients who switched to non-overuse of triptans by month 1, 76 (89.4%) maintained non-overuse status through all 6 months (Figure 5(b)). In contrast, percentages of galcanezumab-treated patients who transitioned from overuse to non-overuse of NSAIDs/aspirin or multiple drugs were comparable to placebo (Figure 5(a),(c)). In REGAIN, galcanezumab-treated patients with chronic migraine transitioned from triptan overuse to non-overuse status at approximately twice the rate of placebo patients across months 1–3 (Figure 5(e)); galcanezumab-treated patients transitioned from NSAID/aspirin overuse to non-overuse status at rates approximately 30% greater than the rate of placebo patients and switched from multiple drug overuse to non-overuse status at rates approximately 50% greater than the rate of placebo patients across months 1–3 (Figure 5(d), (f)).

Transition from medication overuse to non-overuse of medication: The percentage of patients with medication overuse at baseline who changed status to non-overuse of medication by acute medication category at months 1–6 in EVOLVE-1 and -2 ((a)–(c)), and at months 1–3 in REGAIN ((d)–(f)). For each graph, n = the number of patients meeting criteria for medication overuse for the treatment group within each drug class at baseline.
Discussion
This subgroup analysis of EVOLVE-1 and -2 and REGAIN demonstrates galcanezumab 120 mg or 240 mg significantly improved mean monthly migraine headache days compared with placebo in patients with baseline medication overuse. Additionally, among patients with medication overuse at baseline, the average overall monthly medication overuse rate significantly decreased with galcanezumab compared with placebo. Mean monthly reductions in migraine headache days with acute medication use were also significantly greater with galcanezumab compared with placebo in patients with baseline medication overuse.
Patients with episodic migraine and medication overuse had an average of 12 migraine headache days per month, and patients with chronic migraine and medication overuse had an average of 20 migraine headache days per month (Tables 1 and 2). It has also been noted that medication overuse was more common among patients with chronic migraine: only 19% of episodic migraine patients in EVOLVE-1 and -2 had medication overuse compared to 64% of chronic migraine patients in REGAIN. However, for both monthly migraine headache days and monthly migraine headache days with acute medication use, subgroup-by-treatment interactions were significant among episodic migraine patients for both galcanezumab dose groups compared with placebo, indicating a larger treatment effect in episodic migraine patients with medication overuse. Interestingly, the treatment effect appears to be driven by the patients with triptan overuse at baseline, which was the highest percentage of patients (55%) with medication overuse. Of the galcanezumab-treated patients, 77–89% reverted to triptan non-overuse at any time during the 6 months of the study compared to 44–53% of placebo patients. There have been several studies indicating that migraine patients overusing triptans are easier to treat and withdraw from acute medication (16,17). Additionally, simple analgesics (NSAIDs and aspirin) were the most commonly used acute treatments in the EVOLVE studies, but only 8% of the patients reported medication overuse with simple analgesics. This finding is consistent with the findings from the MAST study conducted in the United States showing that respondents who were overusing acute medications were two times more likely than those who were not overusing acute medications to be overusing triptans (18). These data, together with those from each of the pivotal parent trials, indicate galcanezumab is an effective treatment of both episodic and chronic migraine, regardless of medication overuse.
Data from REGAIN, published as part of an integrated analysis of galcanezumab safety and tolerability, showed no statistically significant differences in common treatment-emergent adverse events in patients with chronic migraine, with or without medication overuse (19). The pathophysiology of medication overuse headache is not well understood, and debate persists regarding its cause and effect (20). While there is experimental evidence indicating prolonged exposure to acute drugs can drive cephalic nociception, overuse of acute medications for headache may be the consequence rather than the cause in some patients (20). Current evidence indicates recidivism rates are high and only a modest proportion of patients with medication overuse derive long-term benefit from treatment limitation or discontinuation alone (20,21). There is a clinical belief that patients have to wean off or discontinue acute medications for preventive drugs to be effective (8). However, while some studies report high “cure” rates with complete cessation of acute medications, most patients require the addition of preventive treatment to successfully stop or reduce the overused medication(s) (22). In a head-to-head comparison of three differing treatment modalities, data from several secondary endpoints indicated that withdrawal of acute medication combined with prophylactic treatment was more effective than either treatment alone (9). The data from this post hoc analysis indicate patients with medication overuse, who were not weaned off or instructed to limit the use of acute medications, responded to galcanezumab equally as well as patients without medication overuse. These data suggest galcanezumab is effective as a preventive medication independent of whether acute medication is withdrawn; however, further studies are required in patients with medication overuse headache to determine the most effective treatment strategy.
These data are consistent with that from a 24-week pooled analysis of the PREEMPT studies, which demonstrated onabotulinumtoxinA had a similar effect in patients with chronic migraine with and without medication overuse (7). Furthermore, in a post hoc analysis of two similarly-designed studies conducted in Europe and the United States that investigated topiramate in patients with chronic migraine, frequency of migraine was reduced significantly more in patients with medication overuse in the European study, and trended downward but was not significantly different from patients without medication overuse in the United States study (23). Another monoclonal antibody against the canonical CGRP receptor, erenumab, has also been shown to be efficacious in patients with medication overuse (24). A subgroup analysis showed the erenumab 70-mg and 140-mg groups had greater reductions in mean monthly migraine headache days than the placebo group at month 3 in patients with chronic migraine and medication overuse (24). Furthermore, a ≥50% reduction in monthly migraine headache days was achieved by 18%, 36%, and 35% of patients in the placebo, erenumab 70-mg, and erenumab 140-mg groups, respectively (24). These data are consistent with that for patients with chronic migraine and medication overuse in REGAIN; that is, 13.8%, 27.1%, and 27.4% of patients had a ≥50% reduction in monthly migraine headache days in the placebo, galcanezumab 120-mg, and galcanezumab 240-mg groups, respectively.
Collectively, these findings suggest at least certain medications effective for the preventive treatment of chronic migraine are also effective for patients with acute medication overuse. We speculate galcanezumab may have been effective in patients with chronic migraine who overuse medications because the overused medication was not causing the increased headache frequency in those patients and/or because galcanezumab may have reversed the biology of medication overuse since CGRP has been shown preclinically to play a pivotal role in medication overuse (25,26). Furthermore, fremanezumab, another anti-CGRP monoclonal antibody, was shown to be effective in preventing and reversing the development of medication overuse in a preclinical animal model (27). These findings suggest acute medications may promote medication overuse through CGRP-dependent mechanisms and anti-CGRP antibodies may be efficacious for the treatment of medication overuse headache.
There are several aspects to consider regarding the study designs of EVOLVE-1 and -2 and REGAIN. For instance, patients were allowed to be on acute medications without restriction with the exception of opioids and barbiturates, which were limited to 3 days per month during the treatment phase of the study, and up to one-third of the patients in the REGAIN study were allowed to remain on stable doses of topiramate or propranolol, but only 15% of these patients continued on these medications during the study (14). In addition, during the EVOLVE-1 and -2 studies, medication overuse was documented in patients with episodic migraine (12–14), albeit this may not be unexpected since patients with high frequency episodic migraine can meet medication overuse criteria. Furthermore, this study investigated and found significant subgroup treatment interactions in patients with episodic migraine, indicating a larger treatment effect in patients with medication overuse.
There are several limitations with this analysis, including its post hoc nature for EVOLVE-1 and EVOLVE-2 and corresponding lack of controlling for multiple comparison bias (type I error). Another limitation of this subgroup analysis is that the study was not designed or powered to show significance in subgroups. However, the similarity of findings across the different study populations along with the similarity to results observed with erenumab despite a smaller percentage of patients overusing acute medications in the erenumab subgroup analysis suggest this is not a substantial limitation (24).
In conclusion, both doses of galcanezumab significantly improved mean monthly migraine headache days compared with placebo in patients with episodic and chronic migraine and baseline medication overuse. Furthermore, the proportion of those with monthly medication overuse decreased with galcanezumab compared with placebo in patients with baseline medication overuse. Galcanezumab, therefore, appears to be effective in patients with episodic and chronic migraine who overuse acute medications.
Clinical implications
Medication overuse is common among patients with migraine. Galcanezumab, a humanized monoclonal antibody that selectively binds to the calcitonin gene-related peptide, was superior to placebo in the prevention of episodic and chronic migraine in three phase 3 studies. This analysis of three phase 3 studies provides preliminary evidence that galcanezumab significantly reduces monthly migraine headache days and acute medication consumption in patients with episodic and chronic migraine who overuse acute medications.
Footnotes
Acknowledgements
We thank the patients, their families, the study sites, and the study personnel who participated in this clinical trial. Medical writing support was provided by Andrew Sakko, PhD, CMPP and Regina E Burris, PhD, and editorial support was provided by Angela Lorio, ELS, and Dana Schamberger, MA of Syneos Health, and funded by Eli Lilly and Company in accordance with Good Publication Practice (GPP3) guidelines (![]() ).
).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Erin Doty, Dustin D Ruff, Virginia L Stauffer, Jakub Jedynak, Yan Dong, and Eric Pearlman are full-time employees of Eli Lilly and Company, Indianapolis, IN, USA, and minority holders of company stock. At the time the study was conducted, Sheena K Aurora was a full-time employee of Eli Lilly and Company, Indianapolis, IN, USA, and a minority holder of company stock; Sheena K Aurora is currently affiliated with Impel NeuroPharma, Seattle, WA, USA.
David W Dodick reports the following conflicts within the past 12 months: Consulting: AEON, Amgen, Clexio, Cerecin, Allergan, Alder, Biohaven, Linpharma, Lundbeck, Promius, Eli Lilly, eNeura, Novartis, Impel, Theranica, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Majallin LLC, Medlogix Communications, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Ontologics (Options/Board), King-Devick Technologies (Options/Board), Precon Health (Options/Board). Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis.
Ethical approval and conduct
The study protocol was reviewed and approved by the appropriate institutional or ethical review board for each site. The study was conducted according to Good Clinical Practice and the Declaration of Helsinki guidelines. Patients provided written informed consent before undergoing study procedures.
Data sharing
Eli Lilly and Company provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request 6 months after the indication studied has been approved in the US and EU and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Eli Lilly and Company. The authors received no financial support for the research, authorship, and/or publication of this article.
