Abstract
Chronic migraine has been linked to the excessive use of acute headache medications. Medication overuse (MO) is commonly considered the most significant risk factor for the progression of migraine from an episodic to a chronic condition. Managing MO is a challenge. Discontinuation of the acute medication can result in withdrawal headache, nausea, vomiting and sleep disturbances. This review summarizes the results from two similarly designed, randomized, placebo- controlled, multicentre studies of chronic migraine conducted in the USA and European Union. Both studies demonstrate the efficacy and safety of the migraine preventive medication, topiramate, for the treatment of chronic migraine in patient populations both with and without MO. These studies may have important implications for the future of chronic migraine management, suggesting that detoxification prior to initiating prophylactic therapy may not be required in all patients if MO is present.
Introduction
A subgroup of individuals with migraine may have a clinically progressive disorder that is characterized by a persistent increase in migraine attack frequency (1, 2). This subtype of chronic daily headache is a condition that has been referred to as chronic migraine (3). Chronic migraine is a serious, disabling disorder that has a considerable impact on patients' productivity and their ability to perform routine daily activities (4–6). Operational diagnostic criteria for chronic migraine are provided by the second edition of the International Classification of Headache Disorders (ICHD-II). According to ICHD-II, chronic migraine requires headaches occurring ≥ 15 days per month; on at least 8 days, the attacks should fulfill criteria for migraine or respond to acute migraine medications (7–11).
Topiramate is approved in the USA and European Union (EU) for the prophylaxis of migraine in adults. Several randomized, placebo-controlled trials have also demonstrated topiramate to be an effective migraine preventive therapy in adolescents (12) and children (13).
The utility of topiramate in migraine prevention has been shown to extend potentially beyond the treatment of episodic migraine and across a wide spectrum of migraine frequencies. In the 12-week, double-blind study conducted by Mei et al., topiramate significantly reduced the number of migraine days in patients with chronic migraine and medication overuse (MO) (14). A progressive reduction in the amount of acute medication was also observed. In a randomized, double-blind, placebo-controlled trial, topiramate at low doses (50 mg/day) proved to be an effective therapy to reduce headache frequency in patients with chronic migraine and analgesic overuse (15). A small, single-centre, randomized, double-blind, active comparator-controlled trial found that topiramate significantly lowered the mean monthly headache frequency in patients with chronic migraine (16). Furthermore, topiramate may prevent the escalation of migraine to chronic migraine (17) and be useful in reverting chronic migraine to episodic migraine (14).
The primary objective of this review was to evaluate the potential use of topiramate as an effective preventive treatment option for patients with chronic migraine with or without acute MO. The role of MO as a risk factor for the progression from episodic to chronic migraine is discussed. A comparison of the results from two randomized, placebo-controlled, multicentre studies of chronic migraine conducted in the USA (18) and EU (19) demonstrating the efficacy and safety of topiramate for the treatment of chronic migraine is also presented. Particular emphasis is placed on results in chronic migraine with MO.
Medication overuse in patients with chronic migraine
Diagnostic criteria for chronic migraine and MO are in flux and have been recently revised by ICHD-II. In brief, ICHD-II defines MO as the monthly use of ≥ 10 days of ergotamine, triptans, opioids or combination analgesics, or ≥ 15 days of simple analgesics (20). Medication overuse headache (MOH) is defined as headache on ≥ 15 days per month with overuse of acute treatment drugs for > 3 months, and headache that has developed or worsened during MO (20). MOH is now the default diagnosis if MO is present alongside the abovementioned criteria.
MO may contribute to the escalation from episodic migraine to chronic migraine and may increase not only the frequency of attacks in established chronic migraine but also the severity of attacks (21–23). A high initial frequency of headaches and MO are predominant factors for escalation from episodic migraine to chronic migraine. In fact, MO is the most common association in patients experiencing ≥ 15 headache days per month. Thirty per cent to 50% of patients who otherwise meet criteria for chronic migraine, and up to 80% of patients with chronic headache, overuse acute headache medications (22, 24, 25), although conversely, only about one-third of patients who overuse acute medicines have frequent headache (2, 22, 26).
Preventive medications traditionally have been considered to be ineffective in patients who overuse acute headache medications, and in this population, detoxification strategies (abrupt withdrawal of the acute medication) have been advocated in preference to the initiation of migraine preventive medications (27–29). There is evidence suggesting that terminating acute medication may aggravate headache, cause an abstinence syndrome (which may include nausea, vomiting, agitation, restlessness, diaphoresis and insomnia), or result in drug withdrawal seizures (30, 31). To the best of our knowledge, there are no randomized clinical trials that systematically evaluate the effectiveness of medication withdrawal as the only therapeutic intervention for the management of MOH, although detoxification is clearly helpful for some patients (32, 33).
Migraine preventive medications may be an important therapeutic option for migraine patients with concurrent MO. Strong supporting evidence, however, is limited. A small, randomized, double-blind, placebo-controlled, parallel-group study evaluated the efficacy of topiramate in the prevention of migraine in 28 patients with chronic migraine and MO (15). Patients were asked to reduce the frequency of analgesic use during the trial and were not allowed to take acute pain medications more than once per day. There was a statistically significant reduction in headache frequency in patients receiving topiramate; however, none of the patients was able to withdraw from analgesic use completely. Therefore, larger-scale, controlled trials are needed to evaluate the efficacy of preventive medications in patients with chronic migraine and MO. The results could help establish clear therapeutic guidelines on the effective management of this challenging patient population.
Efficacy and safety of topiramate in patients with chronic migraine in the presence and absence of acute medication overuse
Two multicentre, randomized, double-blind, placebo-controlled trials demonstrated that topiramate was effective for the treatment of patients with chronic migraine (18, 19). The intent-to-treat (ITT) population in the US study consisted of 306 subjects (topiramate, n = 153; placebo, n = 153), whereas the ITT population in the EU study consisted of 59 subjects (topiramate, n = 32; placebo, n = 27) (18). In the US trial, topiramate was effective in reducing the mean monthly number of migraine/migrainous days in subjects with chronic migraine compared with placebo (P = 0.010). Similarly, in the EU trial, topiramate significantly reduced the mean monthly number of migraine days compared with placebo (P = 0.02) (19). A post hoc analysis in the subset of patients with MO in the US trial trended towards significance but did not reach a statistical difference between topiramate and placebo in the reduction in migraine/migrainous days (P = 0.059). In the EU trial, topiramate-treated patients with MO experienced a significant reduction in the mean number of migraine days vs. placebo treatment (P = 0.03). Treatment-emergent adverse events (TEAEs) were similar to those observed in previous clinical trials with topiramate (34–36). TEAEs reported by at least 5% of patients in the US trial included paraesthesia, upper respiratory tract infection, fatigue, hypo-aesthesia, dry mouth, difficulty with concentration/attention, taste perversion, nausea, difficulty with memory, somnolence, injury and anorexia (18). TEAEs reported by at least 5% of placebo-treated patients included paraesthesia, upper respiratory tract infection, fatigue, nausea, difficulty with memory and anorexia. In the EU trial, TEAEs reported by at least 5% of topiramate-treated patients were paraesthesia, nausea, dizziness, dyspepsia, fatigue, anorexia and disturbance with attention (19). TEAEs reported by at least 5% of placebo-treated patients included paraesthesia.
There were several key differences between the patient populations participating in the two multicentre studies (Table 1). Notably, between-study differences included the number of subjects with MO and the proportion of patients using different classes of acute pain medications. In the US trial, 115 of 306 (37.6%) subjects vs. 46 of 59 (78%) subjects in the EU trial reported using acute medications for migraine that met the definitions of MO during the 28-day prospective baseline period. In the US trial, the most commonly overused medications were triptans and analgesics—40% of subjects overused non-steroidal anti-inflammatories and 6% overused opioids (data on file). In the EU trial, the vast majority of overused medications were triptans (Fig. 1). Of note, butalbital-containing analgesics were allowed (≤ 4 days per week) in the US trial, whereas no butalbital-containing analgesics were available or prescribed in the EU trial. It should also be noted that ‘any combination’ refers to any combination of triptans, opioids and analgesics, and should not be confused with the ICHD's definition of combination analgesic overuse headache.
Key differences between US and European Union trial patient populations
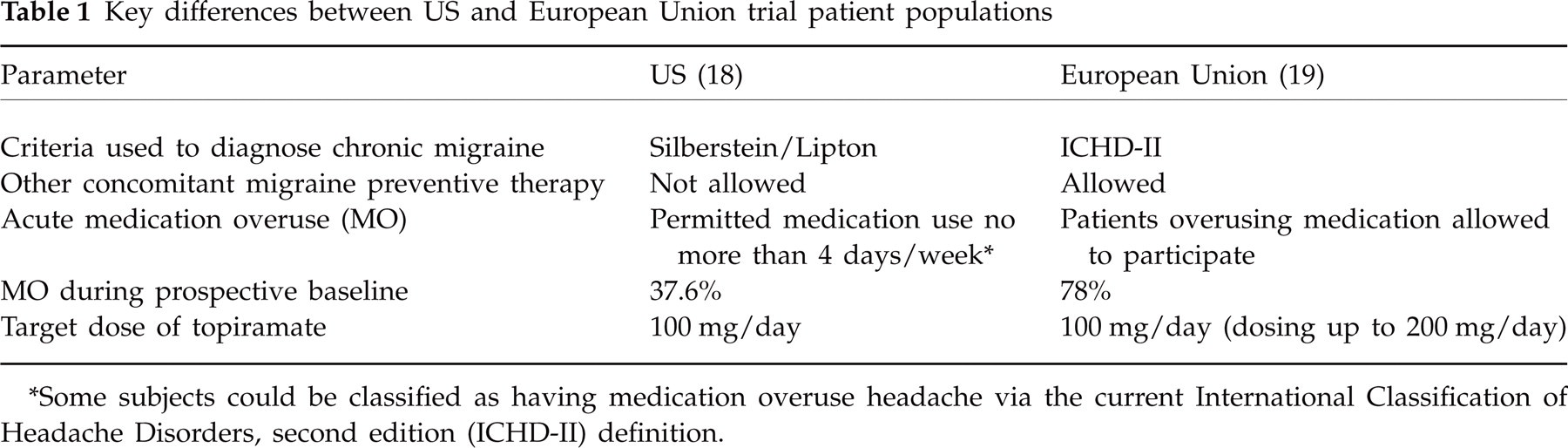
∗Some subjects could be classified as having medication overuse headache via the current International Classification of Headache Disorders, second edition (ICHD-II) definition.
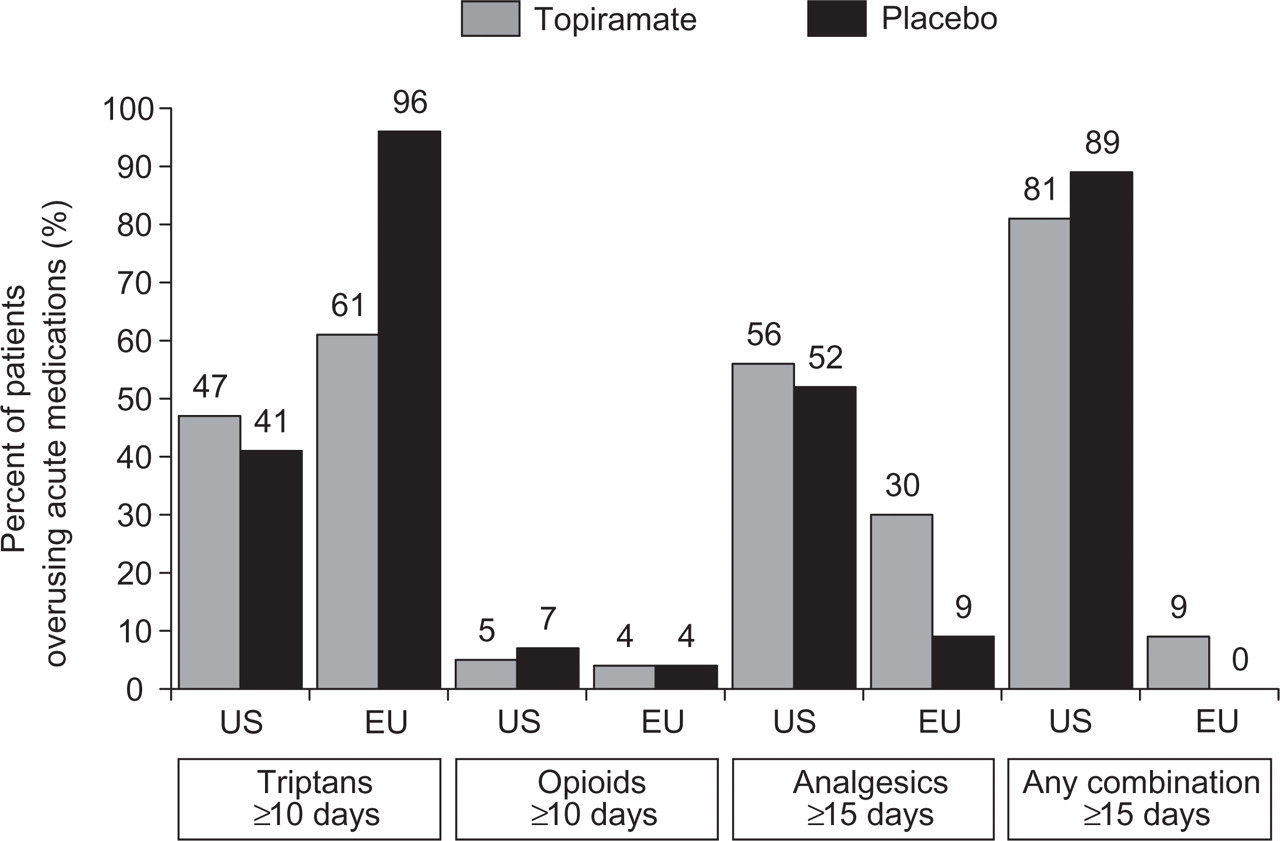
Acute medication overuse in the US and EU trials, within the topiramate- and placebo-treatment groups. In both trials, ergots were not overused in either of the treatment groups. ‘Any combination’ refers to any combination of triptans, opioids and analgesics.
Potential reasons for differences in placebo response in chronic migraine clinical trials
Placebo response rates in migraine clinical trials can vary greatly between studies and may depend upon the choice of primary efficacy measures in addition to patient characteristics and study methodology (37–41). A high placebo response with regard to mean reduction of migraine/migrainous days was observed in the MO subgroup in the US trial, whereas the placebo response was much less in the EU trial.
The difference in placebo response may be attributed to several factors or a combination of factors. For instance, the clinical conditions of patients with chronic migraine were held stable at baseline due to the lack of opportunity for any change in acute or preventive treatment regimens prior to trial entry. Although there was a run-in baseline in the EU trial, there was no opportunity for any change in treatment, either preventive or acute. Therefore, the clinical conditions of these patients remained stable, and this design may have contributed to the low placebo effect. It is possible that the continued intake of acute medications may have perpetuated the frequency of migraines in these patients, thereby counteracting any potential decrease in migraine frequency associated with medication withdrawal in placebo-treated patients. In addition, differences in trial methodology regarding allowable acute medication intake and/or the type of acute medication intake may have accounted for the lack of placebo response in the EU trial. The US trial imposed a limit on the number of medication use days per month allowed (up to 16 days/month), whereas the EU trial imposed no limitation. The baseline rates of medication use for topiramate-treated subjects in the US and EU trials were 11.9 ± 7.0 and 13.3 ± 6.8 days/month, respectively. During the trials, these rates were reduced by 4.4 ± 5.8 and 3.0 ± 5.9 days/month, respectively.
The differences in the types of medication that were overused also may have contributed to the between-group difference in placebo response. Of those patients with MO, 61% of topiramate-treated patients overused triptans in the EU trial, and 96% of placebo-treated patients overused triptans during the prospective baseline period. For patients with MO in the US trial, triptans were overused by 47% of patients in the topiramate-treated group and 41% of patients in the placebo group, whereas 56% of patients treated with topiramate and 52% of patients receiving placebo overused analgesics. In addition, a high percentage of patients in both treatment groups overused a combination of acute medications in the US but not in the EU trial. In the US trial, the most commonly overused medications were triptans and analgesics (Fig. 1).
The higher placebo response observed in the US trial may be attributed to a number of factors. One possibility is that the differences in selection criteria, including the withdrawal of other preventive medications prior to trial entry, may have had an impact on placebo-treated patients. Alternatively, there may have been an ‘amplification effect’, whereby reduction in headache frequency was accompanied by a proportional reduction in acute headache medication use. The withdrawal of overused headache pain medication may have effectively provided a level of treatment for the overuse component of the headache syndrome, resulting in a consequent additional, and therefore amplified, reduction in headache frequency. Investigators were not instructed to avoid discussing the risk of MO with their patients. Therefore, some patients may have been advised to minimize their acute medication intake, resulting in decreased migraine frequency, including in the placebo treatment group.
Table 2 presents the results of a subgroup analysis for the MOH population in the US trial. The change from baseline in mean monthly migraine/migrainous days is stratified as a function of the type of medication, the frequency of MO during baseline (categories include ≥ 10 days per month triptan intake, ≥ 15 days analgesic intake, any drug combination ≥ 15 days, any drug combination 15–18 days, any drug combination > 18 days), and the degree of reduction in acute abortive medications. Although no significant differences in migraine frequency were observed, whether or not patients had a reduction in acute medication intake vs. placebo (Table 2), the sample size used for this analysis is too small to draw a definitive conclusion regarding a possible amplification in migraine frequency reduction and decreased abortive medication intake. We have suggested that differences in the types of medication overused might account for differences in the observed placebo response between trials. Interestingly, when patients with MO were stratified by type and frequency of MO in the US trial, the cohort overusing triptans showed the lowest placebo response. Recall that in the EU trial, the vast majority of overused medications were triptans.
US trial: mean change in migraine/migrainous days as a function of type and frequency of medication overuse

∗Test for no difference between treatments from
Implications for the management of chronic migraine with medication overuse
The overuse of acute medications in patients with chronic migraine represents a significant challenge for the establishment of effective management strategies. The current consensus approach to management is built around initial detoxification or cessation of the overused medication(s). Many physicians agree that preventive migraine medication is less likely to be effective in the presence of acute MO (29, 33). Detoxification strategies may include the complete withdrawal of acute medication(s) before initiating migraine preventive treatment, or a more gradual detoxification following the onset of migraine prophylaxis.
In the results presented here from the EU trial, topiramate effectively reduced the frequency of migraine/migrainous days regardless of whether or not patients had a reduction in acute medication intake. These results suggest that the complete withdrawal of acute medication(s) may not be necessary before initiating topiramate treatment. A more gradual detoxification strategy concurrent with preventive topiramate therapy may be preferred in selected patients. Ongoing research and emerging therapeutic strategies for the chronic migraine patient with MO should take into account this conceptual model of migraine management, which represents a paradigm shift away from a two-stage model of detoxification followed by prophylaxis to a more integrated model of concurrent withdrawal with preventive treatment.
Although these studies demonstrate an important effect of topiramate in chronic migraine patients with MO, it is important to note that the majority of patients in the EU trial with MO were using triptans. It is well-known from previously published reports (23, 42) that migraine patients overusing triptans are among the least troublesome of the MO cohort and are easier to withdraw from acute medication and treat. The majority of MO patients in the US trial were overusing other medications that might render their condition more intractable and resistant to treatment with a migraine preventive. Clearly, a subgroup of patients with MO may not benefit from immediate topiramate treatment, which is consistent with the general impression that MO may impair the efficacy of migraine preventives (33). Future studies might be aimed at identifying differences between the subgroup of patients who do well if treated immediately and those who might benefit from initial detoxification. With the current level of evidence, until such subgroups are identified and characterized, prophylactic therapy combined with acute medication withdrawal may be the most sensible clinical approach to the management of these patients. If, in the ensuing months, the patient dramatically improves to the point where the preventive medication is considered no longer necessary, this medication also can be tapered.
Conclusions and future directions
There is an unmet clinical need for effective and safe treatments for patients with chronic migraine associated with the overuse of acute headache medications. Multicentre, randomized, double-blind, placebo-controlled trials have shown that topiramate is an effective treatment for patients with chronic migraine. Results presented here suggest that topiramate preventive treatment may be effective in a proportion of patients with chronic migraine with MO prior to detoxification or complete withdrawal of the overused acute headache medications. Migraine prophylaxis, without prior detoxification from acute medications but with concurrent withdrawal of the overused medications, may be considered as an option for effective management. Although there were between-study differences in the placebo response rates, these may be partly associated with differences in the types of medication overused, the limit on the number of acute headache medication use days imposed in the US study, and differences in expectations and instructions regarding minimizing acute medication use in the patient groups. These preliminary findings could be used to inform the design of future randomized, placebo-controlled studies of preventive migraine therapies in patients with MO.
Footnotes
Acknowledgements
Editorial support was provided by Ann Yeung, PhD, Phase Five Communications Inc., with funding provided by Ortho-McNeil Neurologics.
