Abstract
Chronic migraine (CM) patients frequently overuse symptomatic medications (SM). These medications may create a cycle of rebound, worsening of headache and withdrawal symptoms that perpetuate the headache itself. In addition, the overuse of such substances is believed to counteract the efficacy of preventive treatments. We conducted a prospective randomized open-label trial comparing approaches to out-patient management in 150 CM patients (125 women, 25 men; ages 18-80 years, mean 40.3 ± 13.8) with overuse of SM. In each group, 50 patients received education and orientation and were then abruptly withdrawn from all SM. Immediately following withdrawal, the first group took prednisone (60 mg/day 2 days, 40 mg/day 2 days and 20 mg/day 2 days) for 6 days, the second group did not have any regular medications to take and the third group took naratriptan (2.5 mg twice a day) during this initial period. All patients had similar profiles of headache characteristics and consumption (quality and quantity) of SM before initiation of the treatment, but most were not severe headache sufferers, heavy SM overusers or were overusing opioids. After 5 weeks the headache frequency and intensity, the prevalence and frequency of withdrawal symptoms and consumption of rescue medications during the first 6 days were compared between groups. In addition, adherence to treatment (who returned or not and for which reasons, between groups) and headache frequency, week by week, among the groups of patients were also compared. Forty-four (88%) patients from the prednisone group, 41 (82%) from the ‘nothing’ group and 35 (70%) from the naratriptan group adhered to the treatment and returned. The were no differences between groups with regard to treatment adherence (P = 0.072), headache frequency as well as intensity (P = 0.311) and decreasing of days with headache after 5 weeks and weekly (P = 0.275). However, the incidence of withdrawal symptoms and consumption of rescue drugs was higher among the patients who did not take regular medications during the first 6 days (P = 0.0001 and P = 0.006). We concluded that CM patients with moderate overuse of SM other than opioids may be detoxified on an out-patient basis regardless of the strategy adopted with regard to the use of regular drugs during the initial days of withdrawal, but prednisone and naratriptan may be useful for reducing withdrawal symptoms and rescue medication consumption. Further controlled studies are necessary to confirm these observations.
Introduction
Chronic migraine (CM) represents a common disorder among patients from neurological clinics (1, 2). Its prevalence in the general population ranges from 1.6 to 2.4% (3, 4) and frequently these patients had episodic migraine in the past which, over time, transformed into daily or near-daily presentation with features resembling those of tension-type headache such as moderate severity, pressure-type head pain and bilateral location (2). The patients with CM commonly overuse symptomatic medications (SM) of various kinds, which may contribute to the pattern transformation and even counteract the effects of the preventive treatment with regard to decreasing the frequency and intensity of headache attacks (5–9).
Complete withdrawal from headache medication is the treatment of choice for medication overuse in patients with CM (10). The main obstacle to stopping the overused medications is the headache escalation and withdrawal symptoms mostly observed with heavy overusers of SM and overusers of opioids. Nausea, vomiting, nervousness, sleep disturbance and even seizures may be present and challenge efforts to keep patients from drug abuse (11–13). The worsening of headache along with the withdrawal symptoms may last for up to 10 days (or even longer) and typically, the intensity of withdrawal headache increases between the second and fourth days (11, 12). Some patients cannot tolerate these symptoms along with the headache exacerbation and require hospitalization and the parenteral administration of dihydroergotamine, neuroleptics and steroids (13–15). Replacement therapy with substances such as dihydroergotamine, sumatriptan, naproxen and prednisone for several days has been advocated (13, 14, 16–19). Others simply recommend no medication at all during the first days of withdrawal, in order to reverse previously learned illness behaviour (20).
The aim of this study was to compare three different strategies of out-patient detoxification with regard to adherence to treatment, headache presentation, withdrawal symptoms and rescue drug consumption during the initial withdrawal phase as well as the frequency of headache after a 5-week period following the sudden interruption of all kinds of SM previously overused. We also aimed to compare the gradual decreasing of headache, week by week, among the three groups studied and decided to evaluate the endpoint known as decreasing of headache frequency > 50%, week by week, in order to detect possible differences after the detoxification period.
Patients and methods
The 150 patients (125 women and 25 men) from a headache subspecialty centre were included according to the criteria proposed by Silberstein et al. (21, 22), and all had daily or near-daily headache for > 6 months. The ages varied from 18 to 80 years (mean ± SD 40.3 ± 13.8) and 141 (94%) patients had migraine without aura and nine (6%) migraine with and without aura as primary headaches. The patients were randomized into three groups according to the order of arrival in the clinic, alternating groups at the time of inclusion of each patient. The first patient was allocated to the prednisone group, the following patient to the nothing group, the third patient to the naratriptan group, and so on. The first group was clearly and emphatically orientated to withdraw all SM, took prednisone during the first 6 days (60 mg/day, 2 days; 40 mg/day, 2 days; 20 mg/day, 2 days) and initiated the preventive treatment composed of two capsules/day of atenolol, nortriptilyne and flunarizine (doses were increased after 6 and 12 days, Table 1) from the day 7 on. The second group received the same orientation and prevention but did not have any regular medication to take during the first 6 days (hereafter designated ‘nothing group or no prophylactic intervention’ group). The third group took naratriptan 2.5 mg bid during the first 6 days and had the same approach and prevention as the other two groups (Table 1).
Strategies undertaken for the groups of patients
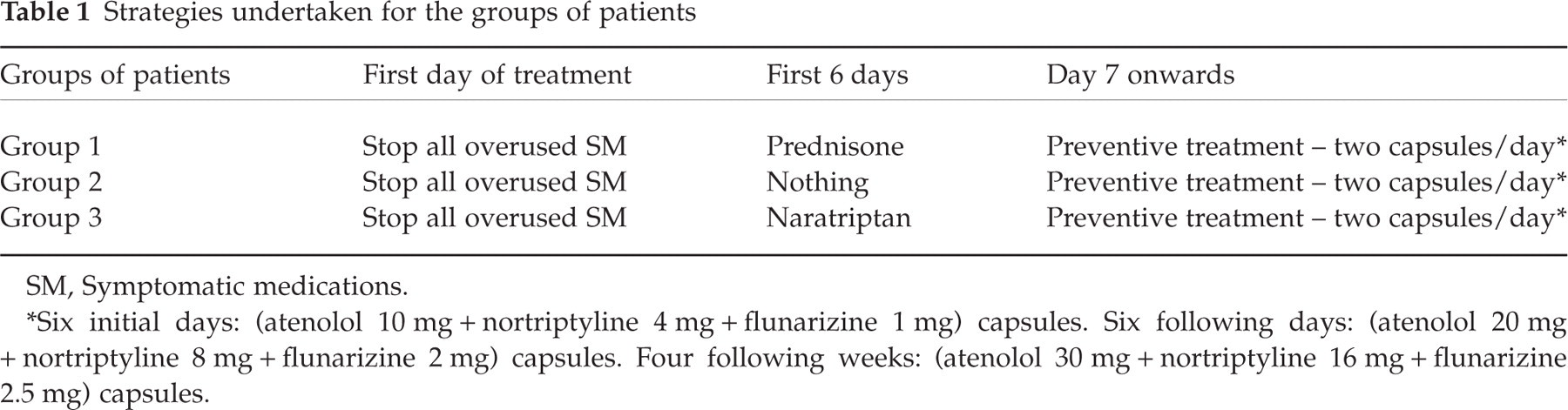
SM, Symptomatic medications.
∗Six initial days: (atenolol 10 mg + nortriptyline 4 mg + flunarizine 1 mg) capsules. Six following days: (atenolol 20 mg + nortriptyline 8 mg + flunarizine 2 mg) capsules. Four following weeks: (atenolol 30 mg + nortriptyline 16 mg + flunarizine 2.5 mg) capsules.
None of the patients was informed that they were taking part in an out-patient study of outcome in CM with overuse of SM, since the abrupt discontinuation of overused medications for headache is the treatment of choice for such a condition (10, 20). This approach was the same as that of Warner (23) and was authorized by the ethics committee of our institution. Therefore, no signed informed consent was obtained. Only patients belonging to the prednisone and the naratriptan groups received the explanation that these drugs could decrease the expected worsening of headache and withdrawal symptoms.
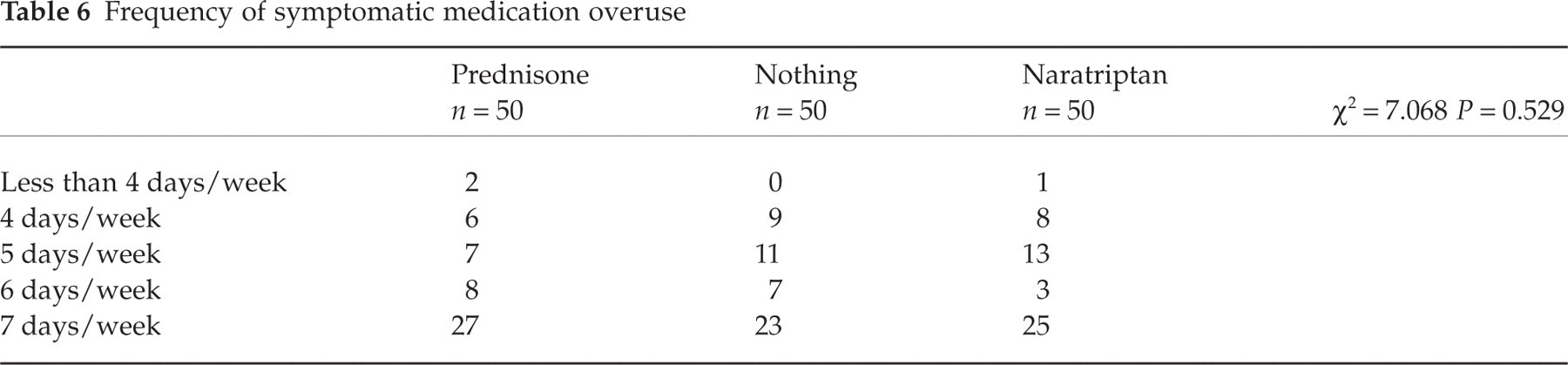
All patients had similar profile of headache characteristics (Table 2) and consumption (quality and quantity) of SM (Tables 3, 4 and 5) before the initiation of treatment and no statistical differences were found between groups with regard to these parameters. However, most patients were not suffering from severe headache most of the time and were not overusing opioids or tranquilizers. In addition, SM overuse was generally moderate. The frequency of usage was also compared between groups (Table 6) and no statistical differences were found.
Headache and patients characteristics
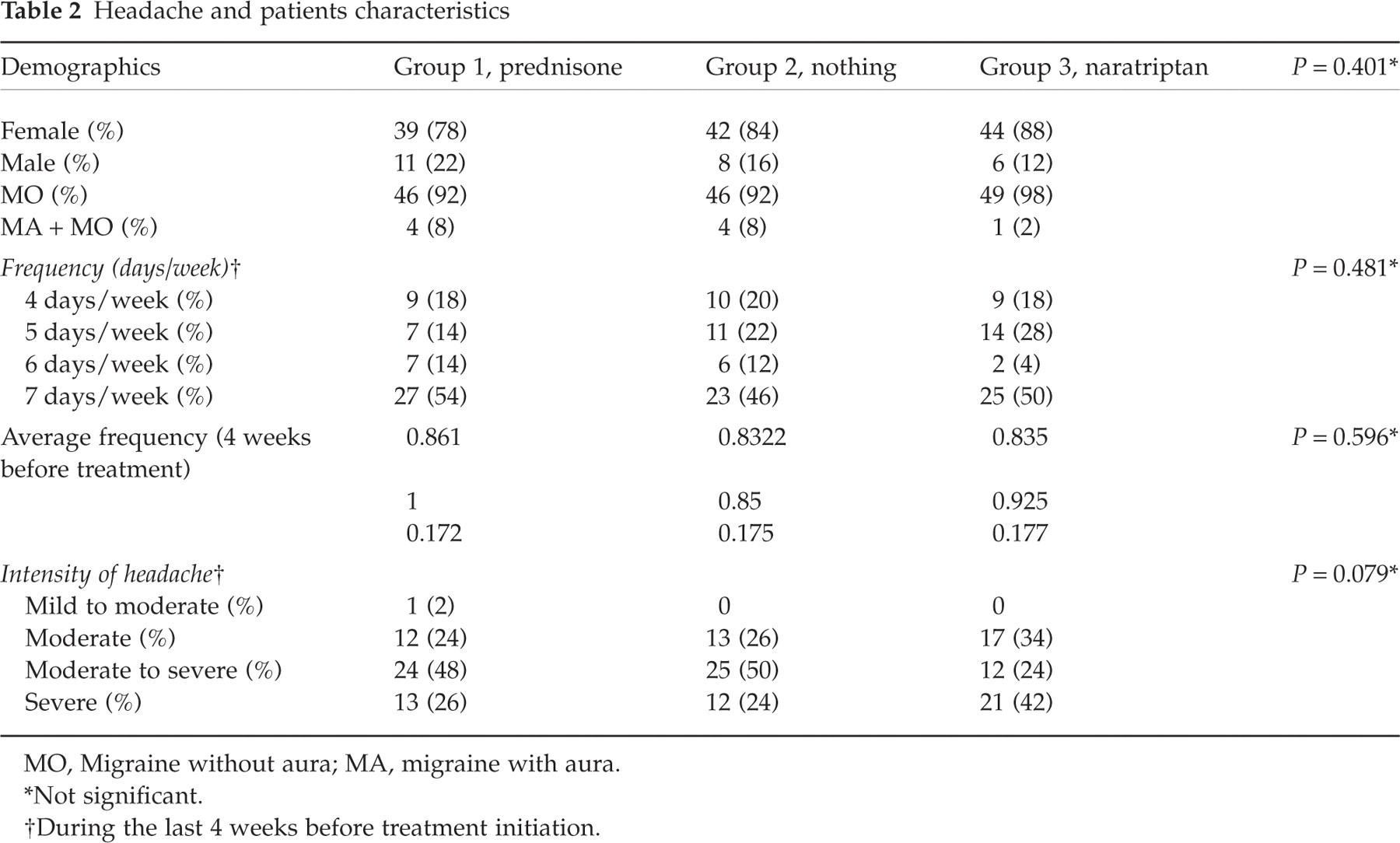
MO, Migraine without aura; MA, migraine with aura.
∗Not significant.
†During the last 4 weeks before treatment initiation.
Profile of symptomatic medications overuse – types of substances overused

Types of medications overused by patients

NSAID, Non-steroidal anti-inflammatory drug.
Amount of medications overused

Frequency of symptomatic medication overuse
From the first day of treatment, all patients received indomethacin suppositories 100 mg to be taken at a maximum frequency of one suppository per week, and were clearly informed about the relationship between overuse of SM and pattern transformation of their migraine. The patients were also informed about an emergency service available 24 h/day with the possibility of i.v. administration of chlorpromazine in a clinic-based environment, as needed.
In addition, all patients had to fill out a detailed daily diary divided into two parts. The first part was related to the first 6 days and requested information about the headache itself (presence, intensity, time of day), about the presence and quality of withdrawal symptoms (listed and not listed, requiring the patients to point out unusual features) and about consumption of the rescue medications (prescribed, not prescribed and use of the emergency service). We called all the patients weekly to emphasize the necessity of adhering to the treatment despite worsening of headache or development of withdrawal symptoms.
After 5 weeks (some patients were allowed to return after 6 weeks for logistic reasons, but only the information related to the first 5 weeks was considered) the patients had to return. Headache frequency and intensity during the first 6 days, prevalence and frequency of withdrawal symptoms and consumption of rescue medications during the first 6 days were compared between groups. In addition, adherence to treatment (who returned or not and for which reasons, between groups) and headache frequency, week by week, among the groups were also compared. We also evaluated the endpoint known as decreasing of headache frequency > 50%, week by week, among the three groups in order to detect possible differences with regard to evolution after the detoxification period.
Statistical analysis
Statistical comparisons were done by non-parametric F-test from Snedecor to compare mean numbers among the three groups and by non-parametric χ2 test or Fisher's exact test (when applicable), to verify the association differences between characteristics found in the three patient groups. In addition, we used the non-parametric tests of Friedman and Kruskal–Wallis, to compare the numeric variables and the t-test to make comparisons pre- and post-treatment among studied groups. Finally, Z-test was performed to compare percentages of frequency reduction between the three groups. We adopted two-tailed P-value < 0.05 for significance levels.
Results
The chronological headache characteristics of the patients belonging to the three groups are listed in Tables 7–9. After 5 weeks, 44 (88%) patients from the prednisone group, 41 (82%) of the nothing group and 35 (70%) of the naratriptan group returned (P = 0.072, NS). Among the prednisone patients, the ages varied from 19 to 68 years (mean ± SD 42.3 ± 12.8) and the daily headache lasted from 6 months to 30 years (4.6 ± 7); the patients from the ‘no prophylactic intervention or nothing group’ were 18–76 years old (38.7 ± 12.7) and the daily headache lasted from 6 months to 30 years (3.8 ± 5.3); the naratriptan patients were 18–80 years old (40.3 ± 16.2) with a daily headache duration of 6 months to 20 years (2.9 ± 3.7). There were no differences in these parameters among patients from the three groups (P = 0.450 for age and P = 0.288 for daily headache duration). Because all patients were called by phone weekly during the first 5 weeks to minimize the risk of a return to medication overuse, we assessed reasons for loss to follow-up. There were no statistical differences between treatment groups (Table 10).
Chronological characteristics of headache – the prednisone group
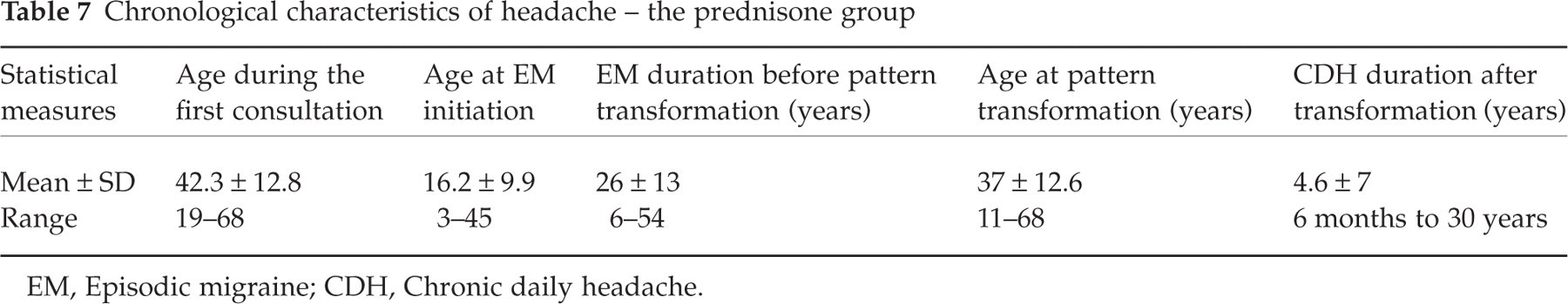
EM, Episodic migraine; CDH, Chronic daily headache.
Chronological characteristics of headache – the nothing group

EM, Episodic migraine; CDH, Chronic daily headache.
Chronological characteristics of headache – the naratriptan group

EM, Episodic migraine; CDH, Chronic daily headache.
Reasons for not returning

∗There was no information whether the initial daily drugs or the preventive treatment brought on the side-effects.
The evolution of headache presentation during the first 6 days is shown in Table 11. There was no difference between the three groups (P = 0.311).
Headache presentation during the first 6 days

∗ n refers to the total number of days considering the total number of 6-day periods among all the patients who returned in all the three groups.
†Not significant.
Withdrawal symptoms presented by patients in the three groups during the first 6 days are shown in Table 12. There was a statistically significant difference between the three groups (P = 0.003) in the number of patients manifested withdrawal symptoms. The differences observed were between the prednisone group and the ‘nothing’ group (P = 0.000) as well as between the naratriptan group and the ‘nothing’ group (P = 0.026). There was no difference between the prednisone and naratriptan groups (P = 0.139).
Presentation of withdrawal symptoms during the first 6 days

∗Statistically significant.
†Asthenia, drop in blood pressure, cold sweating, general feeling of sickness, tachycardia, abdominal pain, dry mouth, somnolence, palpitations, photophobia.
On the other hand, considering the initial 6-day period for all patients, the number of days in which there were withdrawal symptoms was significantly different among patients from the three groups. Insomnia [P = 0.000 for the three groups, between prednisone and nothing groups (P = 0.000), between naratriptan and nothing groups (P = 0.004) and between prednisone and naratriptan groups (P = 0.043)] and dizziness [P = 0.000 for the three groups, between prednisone and nothing groups of patients (P = 0.024) as well as between prednisone and naratriptan groups (P = 0.000)] were the symptoms with different days of incidence among the patients during this period. The category ‘other symptoms’ also revealed statistically significant differences with regard to days of incidence among studied patients (P = 0.005) (Table 13).
Numbers of days patients manifested withdrawal symptoms during the initial 6-day periods for all patients

∗Statistically significant.
†Asthenia, drop in blood pressure, cold sweating, general feeling of sickness, tachycardia, abdominal pain, dry mouth, somnolence, palpitations, photophobia.
Use of rescue medications during the first 6 days of treatment is shown in Table 14. Rescue medications were divided into three categories: indomethacin suppositories (100 mg), in-patient i.v. administration of chlorpromazine (0.4 mg/kg), and use of other drugs not prescribed. With regard to indomethacin use, eight (18.2%) patients from the prednisone group took one suppository 1 day; of the nothing group, 12 (24.4%) patients used it, 10 taking one suppository 1 day and two taking two suppositories in 1 day; in the naratriptan group, five (14.3%) patients used one suppository 1 day. There was no statistically significant difference in consumption of indomethacin (P = 0.239).
Pattern of rescue medication consumption during the first 6 days
∗From the ‘nothing’ group, three patients used symptomatic medication (SM) different from the prescribed ones, besides the use of indomethacin and/or the i.v. chlorpromazine.
The i.v. administration of clorpromazine was different among the three patient groups (P = 0.002). Only patients from the nothing group (n = 6; 14.6%) required administration of this drug. There was difference in such use when comparing the prednisone and nothing groups (P = 0.027) as well as the naratriptan and nothing groups (P = 0.028).
In the utilization of other non-prescribed medications the following profile was observed. In the prednisone group, one (2.3%) patient took one tablet of a triptan once. In the nothing group, three (7.3%) patients took one tablet of a triptan 1 day, one (2.4%) patient took two tablets of simple analgesics 2 days and one (2.4%) patient took one tablet of a non-steroidal anti-inflammatory drug 1 day. In this group of patients, three (7.3%) patients used these drugs in addition to the use of one suppository of indomethacin and one (2.4%) patient in addition to the emergency care provided with chlorpromazine. Moreover, the patient who used two tablets of simple analgesics did not use the options provided. The patients from the naratriptan included one (2.8%) patient taking two tablets of simple analgesics 1 day. There was no difference between the three groups in consumption of other SMs during the 6-day period (P = 0.457). On the other hand, the analysis of the days in which the patients used rescue medicines showed a statistically significant difference (P = 0.0002). In addition, the patients from the prednisone group consumed less rescue drugs than those from the nothing group (P = 0.0009), as did those from the naratriptan group (P = 0.002). Patients from the prednisone and naratriptan groups showed no difference in number of days on which these drugs were used (P = 0.963) (Table 14). With patients who did not use any rescue medications during the initial 6 days, there were statistically significant differences (P = 0.006) between the three groups and between the prednisone and nothing groups (P = 0.011) as well as the naratriptan and nothing groups (P = 0.007) (Table 14).

Evolution of weekly headache frequency in patients of the three groups. •, Prednisone; □, ‘nothing’; ○, naratriptan.
A decrease in headache frequency was also observed among patients in this study. In the prednisone group, the final average frequency was 0.52, significantly lower that the initial frequency (0.86; P = 0.000); in the nothing group, the final frequency was 0.47, also statistically significant in terms of reduction (initial frequency 0.83; P = 0.000); patients from the naratriptan group also demonstrated a significantly reduced frequency of 0.57 after 5 weeks (initial frequency 0.83; P = 0.000). For the whole group of 120 patients that returned, the final headache frequency was 0.52, a significant reduction (initial frequency 0.85; P = 0.000). The final frequencies among the three groups did not differ significantly (P = 0.275) as it did not at the beginning of treatment (P = 0.596).
The prednisone patients presented an endpoint frequency reduction of > 50% from week 3 of treatment onwards; the nothing group, from week 2 and the naratriptan group from week 3. After 5 weeks 62.5% of patients of the whole group showed a reduction of > 50%, with 66% of patients from the prednisone, 63.4% from the nothing, and 57.1% from the naratriptan group. No difference was found between groups (P = 0.250). The total reduction of headache frequency for the 120 patients was 41.2%, 39.5% for the prednisone group, 43.4% for the nothing group, and 33.7% for the naratriptan group, again with no differences between groups (P = 0.878, prednisone vs. nothing; P = 0.824, prednisone vs. naratriptan; P = 0.571, naratriptan vs. nothing). The comparison between groups, week by week, also did not reveal differences of percentage in patients who achieved > 50% frequency reduction (P = 0772 for week 1; P = 0439 for week 2; P = 0179 for week 3; P = 0077 for week 4; and P = 0407 for the last week of treatment) (Fig. 2).
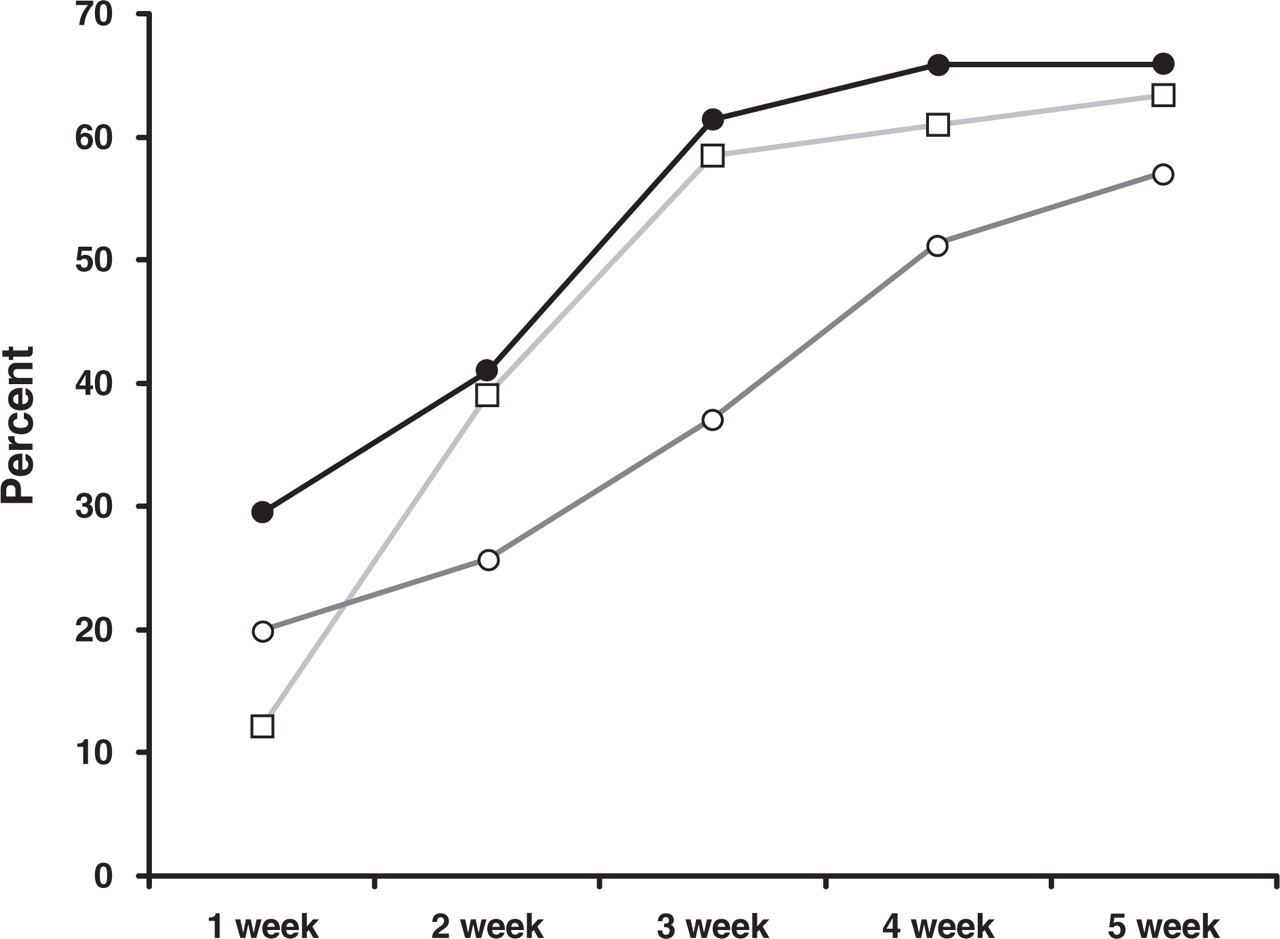
Percentage of patients from the three groups that achieved, week by week, a frequency reduction of > 50%. •, Prednisone; □, ‘nothing’; ○, naratriptan.
Discussion
The literature concerning out-patient strategies to detoxify these patients is scant and few prospective studies had been carried out to demonstrate the best options (10). We previously suggested (13) that prednisone might be an effective way to handle these subpopulations on an out-patient basis. In our previous study, 400 patients with CM and SM overuse were submitted to an abrupt discontinuation of overused medications, received prednisone in the same way as in the present study and initiated their prophylactic medication, composed of different drugs, from day 7 on. During the first 16 days, 12% (n = 48) presented withdrawal symptoms characterized by nausea, vomiting, nervousness, insomnia, tremor and diarrhoea but we did not specify the number of patients that showed each of these symptoms.
These numbers are far less than those seen in this study (81.8%, n = 36 for the prednisone group and 83.4%, n = 100 for the whole sample), perhaps due to the fact that 85.8% (n = 343) of the patients from the former study were under preventive treatment when initiating their treatment.
On the other hand, the numbers of these authors are similar to those of Diener et al. (24) who described withdrawal symptoms in 18% (n = 5) of 27 patients submitting to an in-patient strategy of detoxification. However, the authors stated that the remaining 82% (n = 22) of their patients during the initial 4–6 days manifested headache exacerbation, nausea, vomiting, cold sweat, dizziness and insomnia which were accompaniments, in their view, of the rebound headache and not withdrawal symptoms. If we consider these manifestations as symptoms of abstinence after abrupt discontinuation of overused medications, our numbers are similar.
The use of prednisone in our first study suggested that it could work well as a bridge to detoxification, since none of the patients returned to the pattern of SM overuse. However, the fear of steroid use, even short term, and the huge list of contraindications in some subpopulations of patients, led us to investigate the possibility of using other approaches to manage our patients (25–27). The presence of a non-drug group of patients was due to the fact that there is previous experience suggesting the success of such a strategy in detoxifying chronic daily headache (CDH) patients with SM overuse (28–30) and because we wanted to make a control group that could prove the possibility of detoxifying certain subsets of patients with the simple suspension of overused SM.
We decided to use naratriptan as a third group because it might well be a safe and useful option for patients unable to take prednisone, since it is used short term, as demonstrated in a small number of patients by Sheftell and Pascual (31, 32).
Drucker and Tepper (33) also used a triptan as a bridge to detoxification. In their study, 26 patients with CDH and medication overuse were submitted to withdrawal of these drugs and were started on 25 mg of sumatriptan three times a day for 10 days or until the headache was absent for 24 h, with various preventive medications as well. After 1 month, 58% of the patients were better in terms of headache frequency and reverted to the pattern of daily headache. These numbers are similar to those encountered in the present study, where 62.5% of our sample presented a frequency reduction of > 50%.
With regard to headache reduction, in our sample an overall decrease of 41.2% was observed. The prednisone group showed a reduction of 39.5%, the nothing group 43.4% and the naratriptan group 33.7% after 5 weeks. These numbers are different from those encountered by Kudrow (30), who described an overall 60% reduction in headache frequency in his sample of 200 patients submitted either to suspension of SM and amitriptyline, or continuation of such overuse with or without amitriptyline. In his study, the group of 50 patients who interrupted the overused SM and took amitriptyline showed a reduction of 72%, and the patients who interrupted the overused SM but did not take this tricyclic antidepressant showed a reduction of 43%. However, Kudrow did not mention that among the 100 patients that submitted to withdrawal of SM the adherence was not more than 38%, therefore noticeably lower than the 80% adherence we obtained. In addition, most of the 62 patients from Kudrow study that did not adhere to the strategy of abrupt SM discontinuation did so during the first 2 weeks (34). In our study, the reduction of frequency, week by week, in the three groups was similar. There was no difference in this regard comparing the mean week frequency and the percentage of patients, belonging to all groups, that achieved a frequency reduction of > 50% every week of treatment (week 1, P = 0.221; week 2, P = 0.392; week 3, P = 0.072; week 4, P = 0.422; and week 5, P = 0.718).
It is interesting to note that, even though the group using naratriptan as an out-patient bridge for detoxification did as well as the other groups in most of the endpoints we compared, more patients from this group did not return. Although this difference was not statistically significant (P = 0.072) and the reasons why the patients did not return were acceptable as justification, one could speculate that naratriptan would not be an effective way to assist the out-patient detoxification of these subjects. However, even though not statistically significant, the patients from this group had more intense headache (Table 2) and were overusing more opioids and tranquilizers (Table 3), which could have provoked this difference in adherence. Despite this, the analysis of headache behaviour as well as the prevalence of the withdrawal symptoms and consumption of rescue medications suggested that naratriptan did as well as prednisone.
When the characteristics of the three subpopulations of patients were analysed, nothing different was encountered regarding headache and drug use profile (Tables 2 and 3), with the exception of a non-significant difference in the higher usage of ansiolitics and/or opioids, by the naratriptan group. However, among the 15 patients of this group that did not return, none was taking these substances. According to a consensus paper by the German Migraine Society (35), out-patient detoxification has to be performed in patients that do not overuse tranquilizers and barbiturates, and if it was the case here, it could have explained such a discrepancy, even though not significant, in terms of percentages of patients that did and did not return (35).
Moreover, we expected to fail in most of the patients that did not take any regular medications during the first 6 days, as experienced by Diener et al. (24), but surprisingly, they did as well as the other two groups, with the exception of the presentation of withdrawal symptoms (P = 0.001) (Table 12) and consumption of rescue medications (P = 0.0002) (Table 14), although most of these patients were moderate SM over-users, did not manifest severe headache most of the time and were not opioid and tranquilizer users.
In Diener et al.'s study, the authors were able to detoxify only 1.5% (n = 3) of 200 patients on an out-patient basis, suggesting it is impossible to detoxify severe cases using this strategy. They also recommended that hospitalization for 10–14 days is a valid means of interrupting the overuse of SM.
On the other hand, Suhr et al. (36) evaluated 247 patients submitted either to out-patient suspension of overused medications (n = 110) or to an in-patient strategy for such an interruption (n = 147). The adherence of the two groups was similar (P < 0.2), with 85.4% of the out-patients and 75% of the in-patients adhering to the prescribed treatment. The numbers of the out-patient group are similar to those in the present study, where 80% (n = 120) of our patients followed the strategies of detoxification.
One might speculate that our results were also influenced by the fact the author was calling all the patients each week, which may have been a powerful tool in maintaining their adherence, despite the initial worsening observed even with the prednisone group, since highly motivated patients are the best target for out-patient approaches of detoxification (22, 37). Also, we cannot extrapolate into subsets of patients with more severe headache and using high dosages of opioids since only few patients in the present study were on opioids in each of the groups and most of them did not refer severe headache before treatment initiation (only 24–42% of each group were identified as having severe headache).
In conclusion, this study suggests, despite the questionable open design, that it is possible to withdraw overused SM in a subset of out-patients with chronic migraine and moderate overuse of SM other than opioids that do not present with severe headache most of the time; that the strategy chosen may vary from no medication at all to the use of a triptan or steroids; that, as expected, a noticeable occurrence of headache escalation and withdrawal symptoms is real, despite the approach taken; that the intensity of such exacerbation and withdrawal symptoms was bearable for most of these patients (although those patients using high dosages of opioids and perhaps higher amounts of SM are much less likely to be effectively treated on an out-patient basis); and finally, that most of these patients were significantly improved after 5 weeks. The evolution through the initial weeks after detoxification did not vary according to the strategy used, but we observed a non-significant slower pace in the naratriptan group.
Further controlled studies are necessary to confirm these observations as well for guidance as to the best option for these patients.
