Abstract
Since the advent of modern-day screening collections in the early 2000s, various aspects of our knowledge of good handling practices have continued to evolve. Some early practices, however, continue to prevail due to the absence of defining data that would bust the myths of tradition. The lack of defining data leads to a gap between plate-based screeners, on the one hand, and compound sample handling groups, on the other, with the latter being the default party to blame when an assay goes awry.
In this paper, we highlight recommended practices that ensure sample integrity and present myth busting data that can help determine the root cause of an assay gone bad. We show how a strong and collaborative relationship between screening and sample handling groups is the better state that leads to the accomplishment of the common goal of finding breakthrough medicines.
Keywords
Introduction
Around 15 years ago, several high-throughput screens at multiple Pfizer research sites across different targets encountered an issue where hits from the primary screen were not confirmed either in a single-dose follow-up or by IC50 confirmation. A massive internal study on the nature of the compound screening deck ensued.
Since then, several other studies have shaped our current compound handling practices, along with studies in the literature.1–5 In this paper, a distinction is made between a “compound” and a “compound sample.” A “compound” is the idea, existing with an identifier and a structure. It would have intrinsic properties such as potency against a target or thermodynamic solubility in a solvent. On the other hand, the “compound sample” is a manifestation of the compound; it physically exists in a container as either a solid or a solution. As such, a compound sample has history; for example, the date it was first registered, solvation date, and storage condition, among others. Compound management of today evolved from early efforts to plate many compound samples into wells for screening into a practice combining physicochemical science and technology along with supply chain and informatic tools. Compound management is the starting point to the finish line of discovering new medicines by supplying first assay-ready compound plates in early drug discovery.
The assay developer needs to be aware of the history of compound samples being screened to make the best possible data-driven decisions. Such knowledge would help not only in designing the assay, but also in the interpretation of screening data. Best practices in compound handling can also be directly applied in early stages of assay design and in the routine running of the assay.
Materials and Methods
Compound QC
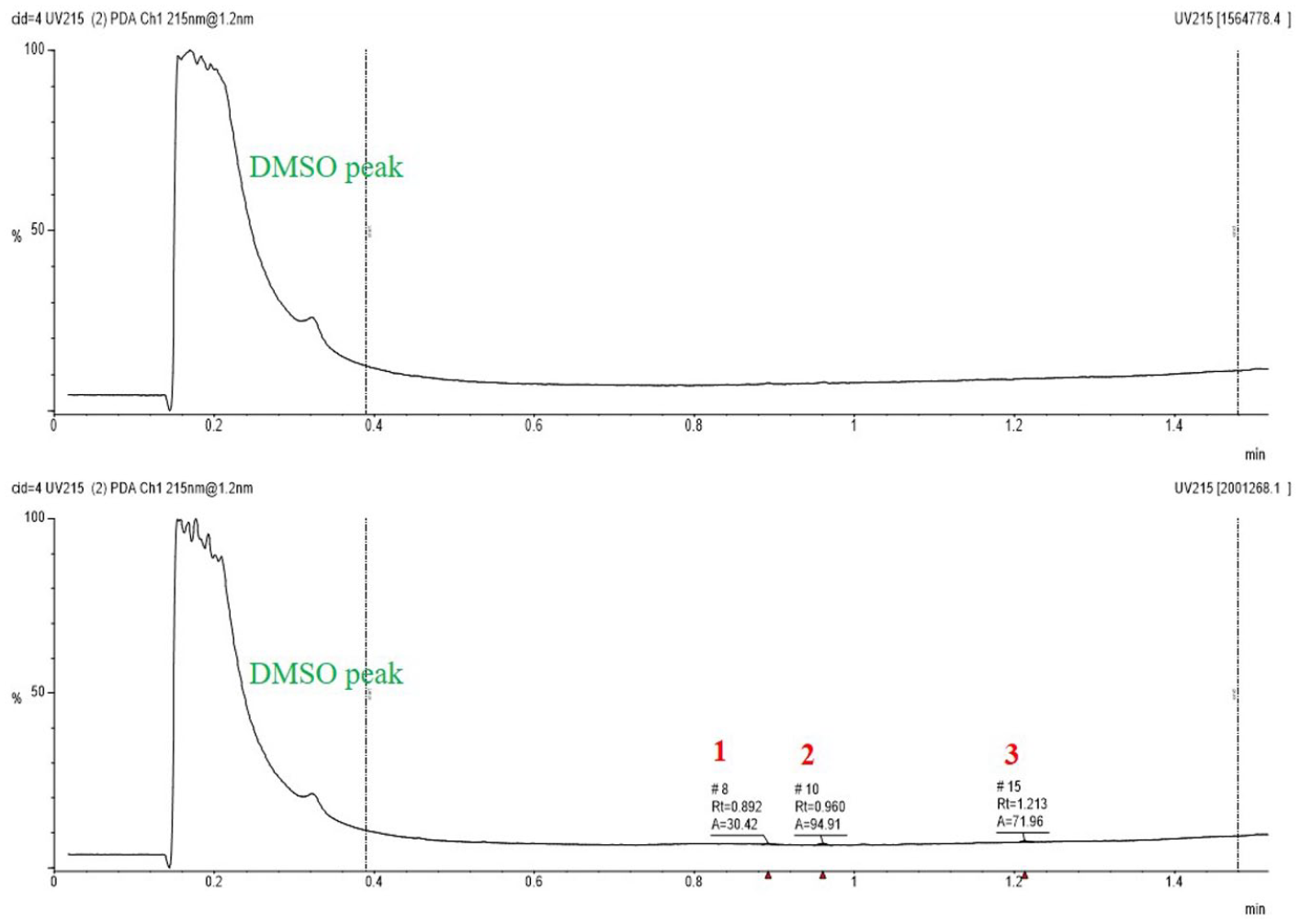
Sample analysis was conducted using a routine Pfizer compound quality control (QC) method employing liquid chromatography–mass spectrometry (LC-MS) as previously described, 6 with some modifications to further reduce compound sample consumption and increase throughput. The analysis is used to quantify the relative purity of a sample and confirm compound identity by comparing target mass to observed mass.
A Waters (Milford, MA) Acquity UPLC with photodiode array (PDA) and single quadrupole MS detection was integrated with Virscidian (Cary, NC) Analytical Studio software. Compound samples at 1 mM in DMSO were in 384-well polypropylene plates. A volume of 1 µL was injected into a Waters 2.1 × 50 mm CSH C18 1.7 µm column held at 45 °C. Sample was eluted at a flow rate of 1 mL/min for 1.2 min in a linear gradient of a 95:5 volume-by-volume ratio of 10 mM ammonium acetate (aqueous) and acetonitrile, and a 95:5 volume-by-volume ratio of acetonitrile and 10 mM ammonium acetate (aqueous). A short hold and reequilibration concluded each sample injection.
UV data were collected from the PDA at 215 nm; MS data were collected on a Waters sQD2 using electrospray ionization (ESI) with positive and negative ionization. Samples were scanned in the mass range of
Sample integration and analysis were conducted in Analytical Studio. Samples were designated as PASS, FAIL, or SUSPECT using the following defined criteria. The designation PASS indicated that the compound mass was confirmed with respect to the target. This included the presence of protonated or deprotonated molecular ions, adducts, or dimers. Additionally, the relative purity of a compound was observed to be ≥80.0% to receive the PASS designation. Compounds were designated as FAIL when no single peak with relative purity ≥80% was observed in a chromatogram. The exception to this FAIL designation occurred when early or late elution was presumed by chromatographic evidence or when proper mass was detected without confidence in a corresponding UV peak. This would drop the sample into the designation of SUSPECT. Additionally, SUSPECT was used to designate poor chromatography stemming from any number of reasons and failure to confirm target mass in the presence of a compound with a relative purity ≥80%.
Extractables and Leachables
Polypropylene and polystyrene plates commonly used as storage plates and assay plates were selected from different vendors. Three different solvents were added to the plates: 100% DMSO, 10 mM ammonium acetate in water, and 0.1% formic acid in water. The plates were stored at regular lab room temperature or at −20 °C. Over a 5-week period, wells were sampled at specific time points and analyzed by ultra-performance LC (UPLC)-MS as detailed above.
Sampling the Canary Set
A diverse set of 3108 compounds ( Fig. 1 ) were identified from the Pfizer compound collection with the requirement that each compound have a solid sample (Neat) and a corresponding batch-matched sample frozen at −20 °C in a REMP (Brooks Life Sciences, Manchester, UK) 384-tube with 25 µL at 30 mM (Frozen). After solvation to 30 mM, a 2 µL aliquot of the Neat samples was transferred and diluted to 1 mM for QC analysis as detailed above. Upon thawing, a 2 µL aliquot of the Frozen samples was also transferred and diluted for QC analysis. Two aliquots of 10 µL each were taken from the Frozen samples and transferred to acoustic 384-well plates (Beckman Labcyte LP-0200, Beckman Coulter, Indianapolis, IN). One plate was stored at −20 °C (Frozen), while the other plate was stored under nitrogen at room temperature (Inert). At specified time points, a 67 nL aliquot was taken from each plate and diluted to 4 µL, 0.5 mM for QC analysis as detailed above.
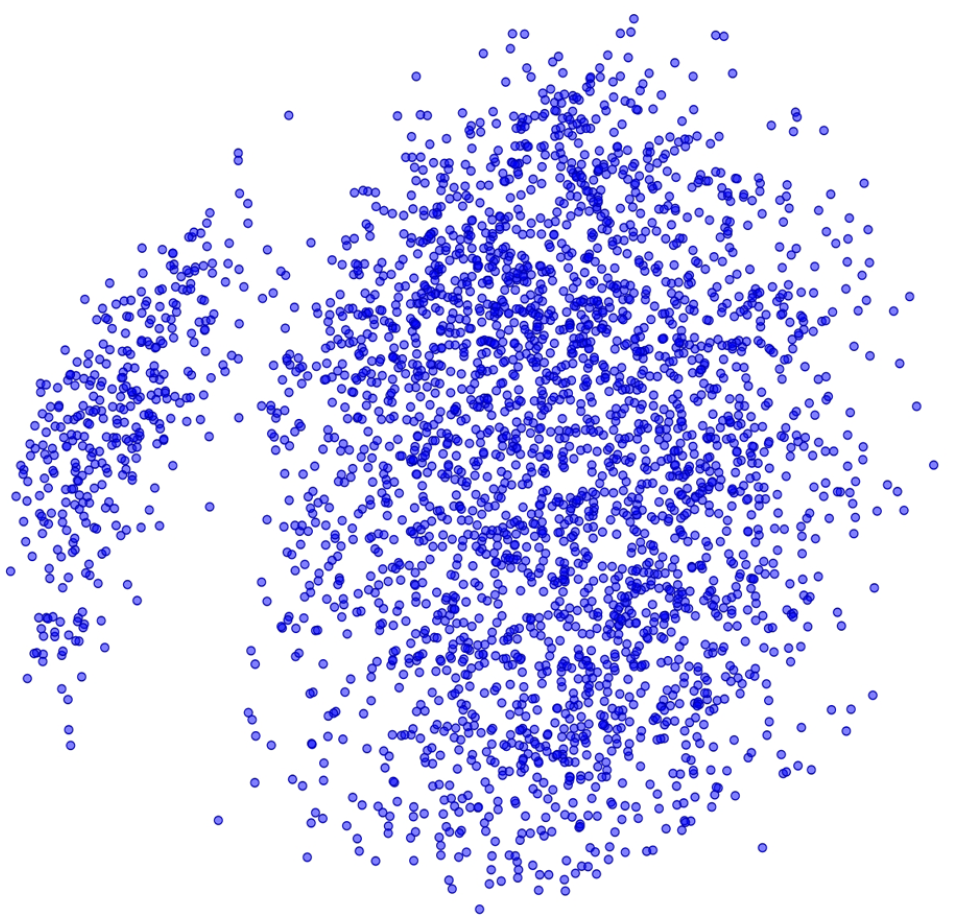
Canary Set compound diversity. Canary Set of 3108 compounds chosen from the existing Pfizer compound collection to make up the Canary Set. Shown is a similarity map of the structures in Tanimoto distance space (UNITY fingerprints) with an outlier radius of 20% and accuracy horizon of 20%.
Water Uptake
Due to DMSO hygroscopicity, samples dissolved in DMSO will accumulate water over time, particularly when exposed to air during routine use in the laboratory. The rate of water uptake is likely dependent on the container type, temperature, and relative humidity. Known amounts of DMSO were placed in tubes (FluidX AcoustiX SP-2727, Brooks Life Sciences) and exposed in air by removing the cap. Water content was assessed over an 80 h period using the Beckman Labcyte Echo 655T. Tubes were filled with starting volumes of 17, 30, 50, or 68 µL, 24 tubes for each starting volume to assess the full working range of the tube.
Tubes were surveyed for percent DMSO at 1 h intervals during normal business hours on days 1, 2, and 4 when all tubes had reached the Echo measurement limit of 70% DMSO composition. 7 This happened after about 24 h, although an increase in volume continued to be observed up to about 72 h, after which the total volume began to decrease.
Liquid Handling QC
The ARTEL (Westbrook, ME) MVS liquid handling verification system is routinely utilized to determine accuracy and precision at a given volume. In the absence of such a system, a suitable colorimetric or fluorimetric dye indicator can be used to measure accuracy.
A stock solution of 25 mM carboxyfluorescein (CF; Invitrogen (ThermoFisher Scientific, Waltham, MA) C-194 or equivalent) in DMSO was prepared. A 10 mM stock solution of sodium borate was also prepared from a 40 g/L solution (VWR (Radnor, PA) VW3869-2). A titration curve of CF was previously generated using the stock solution prepared in a 384-well plate and diluted with the sodium borate buffer. The resulting curve identified the linear part within which deviations from a test volume would be easily detected. Once the target concentrations were determined, standard solutions were prepared in volumetric flasks. The solution was dispensed into a 384-well plate to get the average fluorescence with 95% confidence limits. The upper 95% confidence limit of the top acceptance criteria of the test volume and the lower 95% confidence limit of the bottom acceptance criteria were plotted. Therefore, for a test volume of 500 nL with an acceptance criterion of ±10%, the lower acceptance criterion is 450 nL, while the upper acceptance criterion is 550 nL.
Results and Discussion
Best Practices in Hit ID
Store Compound Plates in 100% DMSO Only
Today, it is an accepted best practice to solvate compounds in DMSO, although that has not always been the case. 8 Despite its limitations, DMSO continues to be a universal solvent for small-molecule compounds for drug discovery. In our hands, DMSO stock solutions frozen at −20 °C for more than 15 years continue to be viable when compared with their freshly solvated solid counterparts ( Fig. 2 ). In the Canary Set that was frozen, 64% of the samples remained at ≥80% purity, compared with 63% of samples that were kept as solids. Interestingly, there were more compound samples that appear to have degraded to purity below 80% when kept as solids (−22% vs 18%).
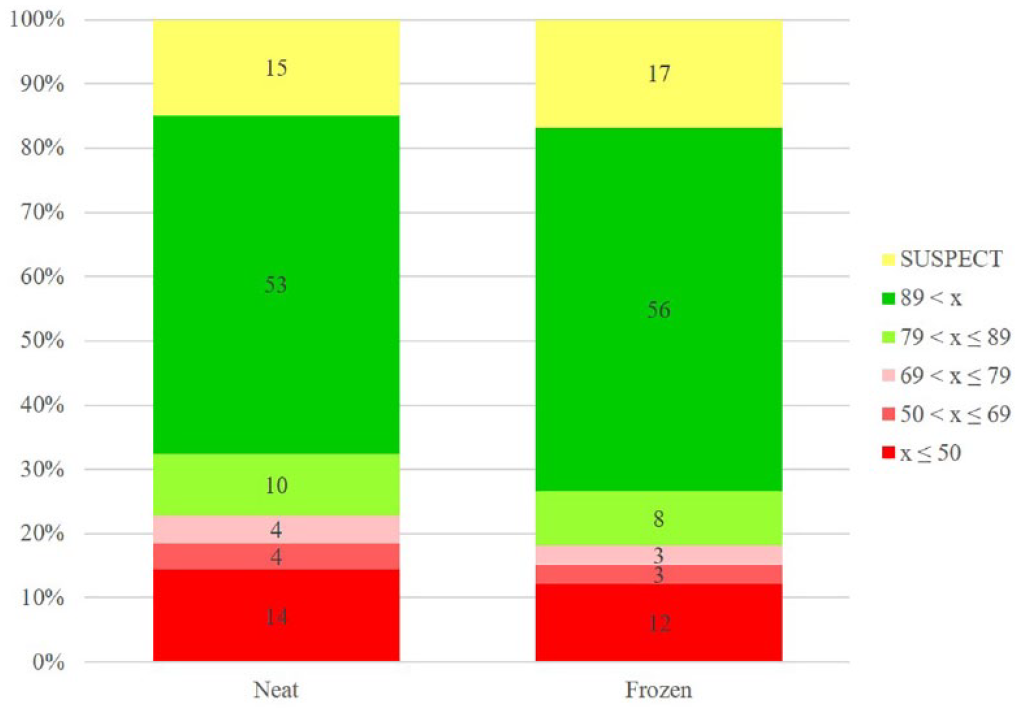
Comparison of freshly solvated solids of the Canary Set versus corresponding sample frozen long term (i.e., >10 years). Bar labels represent percent of compounds falling into a purity bin, as indicated in the legend. Green shading represents PASS by our criteria as outlined in the Materials and Methods section. Red shading represents FAIL, while yellow represents SUSPECT. See article online for color figure.
DMSO also has it downsides. Like most solvents, not all samples will dissolve in it and will consequently precipitate in solution. In addition, DMSO is hygroscopic, and what we consider to be pure DMSO in a bottle in the lab may have more than 30% water after the bottle is left open for several days or hours ( Fig. 3 ). Indeed, after leaving freshly aliquoted DMSO in an uncapped tube for just 3 days, it appears to have gained as much as 50% water by volume within 24 h. Being a universal solvent, DSMO might also extract leachates from the very containers that hold it ( Fig. 4 ). Fortunately, these limitations can be addressed through routine care and diligence. Among the recommended best practices to handling DMSO are the following:
Minimize the time that reservoirs of DMSO are open to the air. While counterproductive to enabling long uninterrupted runs on laboratory automation systems, using DMSO from reservoirs open to the air means that compound samples are being diluted in a mixed solvent rather than pure DMSO. This would increase the chances of precipitation and compound instability, which may lead to anomalous screening results. 9
Reservoirs should be made of inert material, like glass, and tubing made without plasticizers. This will avoid contamination of compound solutions with plasticizers and other extractable substances. Some of these leachates have been previously shown to affect assay results.10,11
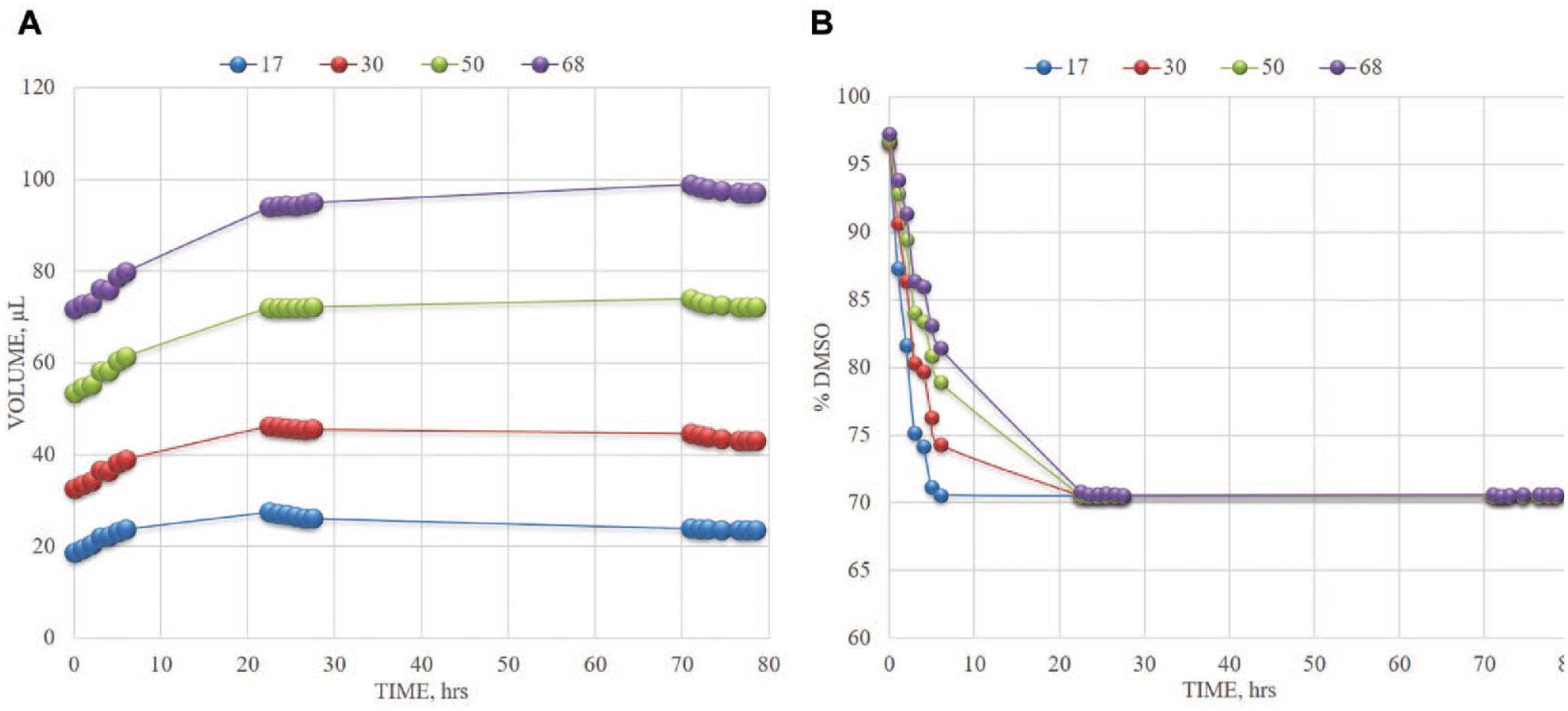
Rate of water uptake of DMSO stored in uncapped tubes for more than 72 h. (
Evidence of extractables in common laboratory containers.
Stay in DMSO for as Long as Possible
Compound samples eventually need to be tested in aqueous solutions with other assay reagents. Staying in DMSO for as long as possible minimizes the time that a compound is in mixed DMSO/water. Any compound processing steps done prior to mixing with other assay reagents, such as serial dilutions, should be done in DMSO.
Cell-based assays usually have low DMSO tolerance, typically below 5%. Serial dilutions in DMSO can be an issue for an assay with low DMSO tolerance. One way to address this is to use very low volumes of the compound stock. For example, in a 1536-well-based screen with a total volume of 4 µL and a DMSO tolerance of 1%, a compound stock volume of only 40 nL should be delivered to the assay. Low-volume liquid handlers, such as acoustic dispensers, can deliver these small volumes with good accuracy and precision. In cases where the compound stock volume is too low to be within the range of this equipment or in laboratories not equipped with them, the alternative is to do an intermediate dilution of the DMSO stock with water. In our experience, this is best done by ensuring an aqueous dilution of greater than 1:100 of DMSO to water and using the resulting solution immediately. In practice, this aqueous dilution should be done as part of the assay; the aqueous plate is discarded immediately and not reused.
Incorporate Compound Sample Information in Making Hit Triage Decisions
In a perfect world, several thousand hits would come out of a primary screening campaign of several million compounds. This would then reveal multiple series of structurally similar compounds whose activity may be further confirmed by screening additional members of each series. This quintessential hit triage scenario, however, is often marred by the reality that samples in the compound collection have a history that can confound the success of a screening funnel. The cost of ignoring such history is many months of chasing compounds that turn out to be false positives, many dollars spent resynthesizing fresh batches or making new libraries, and long hours of frustration and blame.
Compounds are made in batches, and each batch is made with different lots of monomers; over time, each batch may be purified using different methods and stored under different conditions. These batches may show different instability profiles that will cause some batches to degrade over time, but not others. In addition, compound structures themselves can have their own intrinsic instability profiles.
The impact of sample history on hit triage is best illustrated in the following cases:
These examples highlight the importance of knowing the quality of the compound sample being screened, particularly in identifying and following up on hits.
Best Practices in Hit to Lead
All compound testing begins with the weighing of solid material followed by the addition of DMSO to make a stock solution. This simple step determines the concentration of the resulting solution since it is routinely assumed that the compound in the sample is 100% pure. Furthermore, these DMSO stocks will be preserved by the compound management group to be used in the foreseeable future for screening against other targets. 12 These two simple steps of weighing and the addition of solvent have far-reaching consequences in early drug discovery; therefore, it is important to be as accurate as possible.
Weigh Carefully
The most accurate way to make a solution is to follow industry standard procedure, which might involve the use of a previously certified scale and liquid dispenser. Gram quantities of solid dissolved in a duly calibrated volumetric flask would ensure that relative weighing errors are minimized.
The typical compound management or screening laboratory in early drug discovery, however, may not routinely weigh gram quantities of compound samples in volumetric flasks. Higher-throughput weighing of milligram quantities into glass vials or small-volume tubes is more often the norm. Furthermore, compound samples made using parallel medicinal chemistry efforts yield even smaller quantities of purified material that go through solvent evaporation as part of the purification process. Fluctuations in tare weights heavily influence the resulting sample weight; therefore, greater uncertainty is associated with lower yields.
In a compound management laboratory supporting hit-to-lead screening, the use of 1.4 mL 2D barcoded tubes allows for automation of sample workflows. It is desirable to weigh samples in the “safe zone” of good analytical balances, in the range upward of 2 mg, to create highly concentrated stocks greater than 10 mM. This ensures that there is enough stock for all plate-based screening needs.
Good Liquid Handling Practices
In plate-based screens, the accuracy of dispensing can significantly affect the determination of compound activity.13,14 Regular monitoring of the accuracy and precision of a liquid handler is now easier to perform using commercially available kits and/or reagents. There are a few things to watch out for to ensure good liquid handling:
Measurement of accuracy and precision should be done in the solvent being used for the laboratory application. For compound handling, tests should be conducted using DMSO. For screening, tests may need to be done in aqueous solvents. Liquid handling manufacturers routinely specify accuracy and precision using water; therefore, before selecting a liquid handler, it is important to ensure that it can meet the requirements of the application in the right solvent. When testing small volumes, it is good practice to minimize the effect of air bubbles with a quick centrifugation step prior to the read step to avoid false fluorescence or absorbance readings.
Accuracy and precision are typically expressed as a plate-based statistic. It is important to see the data in the context of the number of independent tips in the device. Data from a bad tip can be smoothed over by summary statistics, thereby resulting in consistent mis-dispensing that will have significant implications on screening results. Plots like that in Figure 5A can show tips over- or underdispensing beyond the expected accuracy criteria, while plots like Figure 5B can identify specific tips that might be over- or underdispensing, resulting in higher or lower EC50 estimates.
Tip manifolds get blocked, O-rings and gaskets can degrade, and transducers can wear out over time. If a unit is heavily used, it is good practice to check dispensing performance every 1 or 2 weeks. It is also good practice to check performance in the right liquid after a service call is performed.
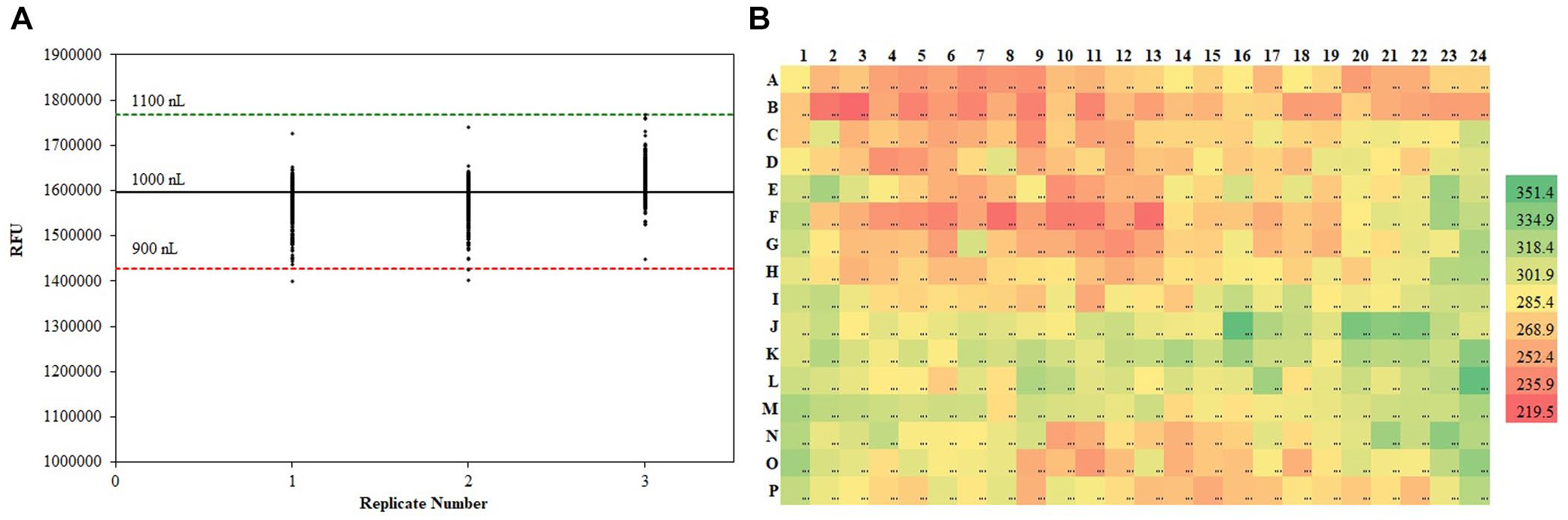
Visualizing accuracy and precision of liquid handlers. (
Best Practices for Storing DMSO Stocks
If a solution is to be used within 3−6 months, room temperature storage under dry conditions using a nitrogen blanket or a desiccator is recommended. Figure 6 shows the stability profile of our Canary Set under inert conditions. After 12 months, more than half of the compounds in the Canary Set still pass our QC criteria of being ≥80% pure.
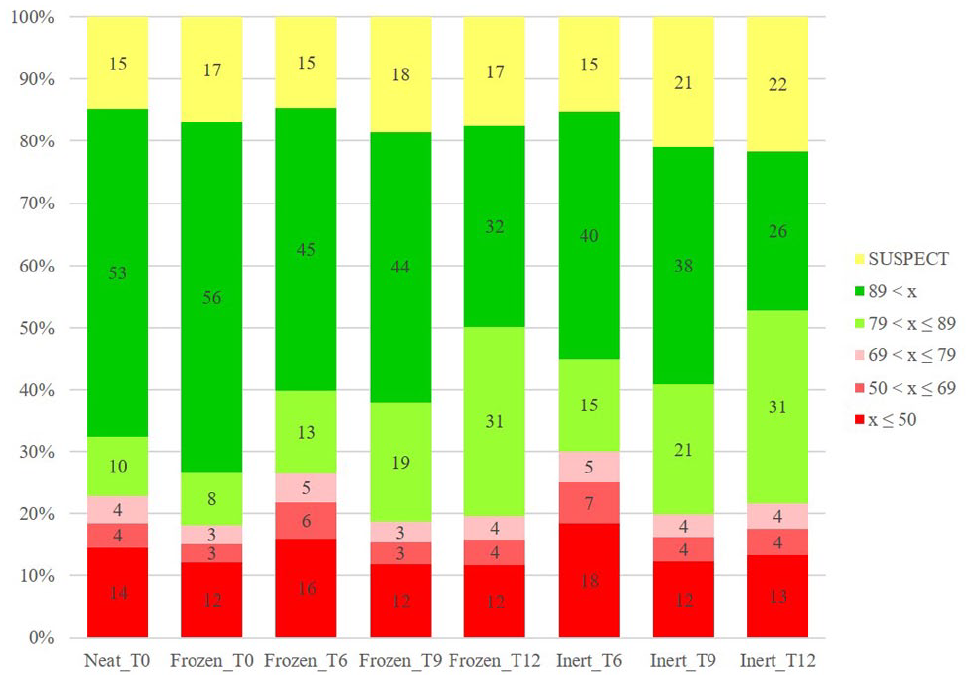
Stability over time (
Long-term storage of DMSO stocks is best done at −20 °C. Earlier studies at Pfizer indicated that storage in a regular lab refrigerator, typically at +4 °C, is not recommended. 15 More recent studies with our Canary Set show that compound stocks can be frozen at −20 °C for more than 10 years with no appreciable loss in quality. While repeated freeze−thaw cycles do negatively impact quality, the rate of degradation is much slower for frozen samples when compared with samples stored at room temperature or room temperature/inert.
When Things Go Wrong
A compound manager has a set of tools that can help troubleshoot an assay gone bad, particularly if the compound sample is a suspected cause. These tools build up from best practices that could be in place in the lab and help increase confidence that the assay plate did contain the correct compound sample:
Visual inspection—Inspection of an assay plate or its mother plate is one of the first steps in troubleshooting. Visual inspection can potentially confirm that there was liquid in the mother plate or the well is empty.
Instrument logs and software logs—Liquid handlers log movements and dispenses at varying levels of detail. Acoustic liquid handlers, for example, generate transfer logs that confirm a dispense of a certain volume took place. Workflow manager applications also provide logs that track the movement of samples from source to destination, thus generating a kind of genealogy of the well in an assay plate.
Analytical tools—If the source plate of the final assay deliverable is still intact, one might be able to confirm the presence or absence of a specified compound by MS.
Performance data—These data can provide confirmation that the liquid handling equipment was performing according to the specifications, when the last service date was, or what kind of repairs might have been performed recently. Historical data may also bring to light a record of mis-dispensing for a specific position.
Compound sample quality data—Records of purity assessment over time showing possible degradation, or the presence of precipitates might give indications of an unstable sample that could be affecting assay results.
Conclusion
The last 10−15 years has seen an increase in the knowledge base accumulated on how to work with small-molecule samples in early discovery, particularly in screening for potency or an equivalent endpoint. The opportunity and obligation to distill this knowledge into laboratory practices are the right thing to do. As new technologies and new data are employed and generated, respectively, laboratory practices should evolve and change.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employed by Pfizer Inc., and their research and authorship of this article was completed within the scope of their employment with Pfizer Inc.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
