Abstract
Fish species such as medaka or zebrafish are widely used as animal models to study physiology, disease development, and treatment efficacy. They are also used to study the rapidly growing field of behavior research, such as social interactions, anxiety, and the influence of environmental factors. Here we describe an automated experimental setup allowing the recording of general locomotor activity in combination with a food-on-demand system. It can simply be built with some basic electronic knowledge. Our setup enables the recording of locomotor and feeding activity of several fish for long-term studies, excluding disturbing external influences. A description of the automated recording system is given, as well as examples of recordings to illustrate its applicability for the study of fish behavior. The construction manual and operation instructions can be downloaded for free.
Introduction
Animal models provide important paradigms to study complex biological processes under precisely controlled experimental conditions. For example, they are widely used to analyze physiology, disease, and behavior.1,2 Small fish species such as medaka or zebrafish are used as animal models due to a number of advantages. Their economical husbandry, fecundity, and established genetics allow modeling of a wide spectrum of paradigms.3,4 More recently fish have also been used to study behavior such as social interactions, anxiety, and the influence of environmental factors.5,6
An important behavioral parameter is the rhythm of daily activity as it is indicative of basic patterns of behavior, such as diurnal or nocturnal activity. 7 Coupled to this activity is the feeding activity. Fish, like many other animal species, do not feed continuously and their feeding patterns often follow daily rhythms. 7 Both feeding and general locomotor activity are strongly influenced by environmental factors such as light and constitute key parameters of behavior that provide important information on a given species and the experimental conditions.
Generally, locomotor activity has to be studied over a long period of time to reveal hidden patterns such as daily rhythmicity. 7 Furthermore, studies that model seasonal changes require the observation of activity patterns over weeks or even months. Activity observations should not interfere with animal behavior to exclude experimental artifacts. Thus, in many cases manual observation by scientist is not possible. Considering the length of the experimental duration, the data volume and downstream analysis of activity recordings have to be taken into account. High-resolution observation as provided by video recordings requires complex and time-consuming data analysis, especially when groups of animals are recorded. Therefore, for many applications video analysis is not an option. When groups of animals are observed, often the general locomotor and feeding activities of the entire group are sufficient to determine behavioral patterns in a given experimental paradigm.
It has been demonstrated that fish are capable of associative learning. This feature of fish behavior can be exploited to use food-on-demand systems to efficiently record feeding behavior. 7 It is thus possible to circumvent the need for manual feeding, enabling experimental setups where animals are not influenced by external stimuli such as food that is provided by the experimenter.
Individual animals or groups often show considerable variation of behavioral parameters. 8 It is therefore important to consider replication to ensure solid statistics for the experimental validation. Often, it is desirable to run parallel recordings to ensure identical conditions, such as time of day, temperature, and water conditions. Thus, a device that can operate several parallel recording setups provides considerable improvement for the experimenter. Taking these requirements and limitations into account, a simple and sensitive automated setup is needed to record activity patterns of larger numbers of animals.
Here we describe a simple experimental system allowing the recording of general locomotor activity in combination with a food-on-demand system that is based on a prototype design.
7
This employs infrared (IR) sensors that record locomotor activity as well as activation of a feeder. Del Pozo and coworkers demonstrated its applicability to record activity from zebrafish,
7
but such sensor-based recording systems have been shown to be applicable for a wide range of aquatic species.9,10 Our setup enables the recording of locomotor and feeding activity of groups of animals that are kept in isolation for extended periods, thus excluding unwanted external stimuli. A description of the automated recording system is given. As an example of its applicability, we show recordings of the locomotor and feeding activity of the fish model system medaka (
Methods
Using a technically simple setup based on IR sensors,7,9,10 we designed an automated recording system that enables parallel recording of general locomotor and feeding activity from several fish tanks. Simple installation and a graphical user interface (GUI) ensure a user-friendly operation. Our system allows for the automated recording of fish locomotor activity and for the automated feeding of six independent fish tanks (
Figs. 1

System to control automated feeding of fish and recording of locomotor activity. F = feeder; S = IR sensor, DAQ = data acquisition and control board (here without protective housing).

Fish tanks were equipped with the automated feeding and recording system. F1–5 = feeder; S1-1 … 2-2 = IR sensors; DAQ = data acquisition and control board in a housing.
The system can also be used to monitor the feeding behavior. In this case, an additional IR sensor (model E3S-AD63, Omron, Kyoto, Japan) with short-range sensitivity is placed in front of a compartment in the upper corner of the fish tank that is accessible only from the lower side. When fish enter the compartment and pass by the IR sensor, it is activated and a signal is sent to a data acquisition device (model USB-1024HLS, Measurement Computing Corporation, Norton, MA), which is connected via USB interface with a notebook. The data acquisition device is also used to control the food dispensers (model 3581, Eheim GmbH, Deizisau, Germany) via switch relays that activate the dispensers upon activation of the short-range IR. This way, up to six fish tanks with food dispensers can be controlled automatically independent from each other. Fish learn within a few days to associate swimming into the compartment, and thus activating the sensor, with the resulting food reward from the coupled food dispenser.
We have developed a program code for a GUI ( Fig. 3 ) in a LabView environment (National Instruments, Austin, TX) that allows for controlling the feeding and motion recording system. It is possible to control the parameters for automatic feeding, such as delay, trigger stimulus, and repeated feeder activation. Additionally, the settings can be changed for automatic recording of fish locomotor activity in 12 fish tanks simultaneously. Typically, the locomotor activity is recorded every 10 min. However, the interval can be set anywhere between 1 min and 1 h by the user. The data are saved together with the associated timestamp in text files on the computer.
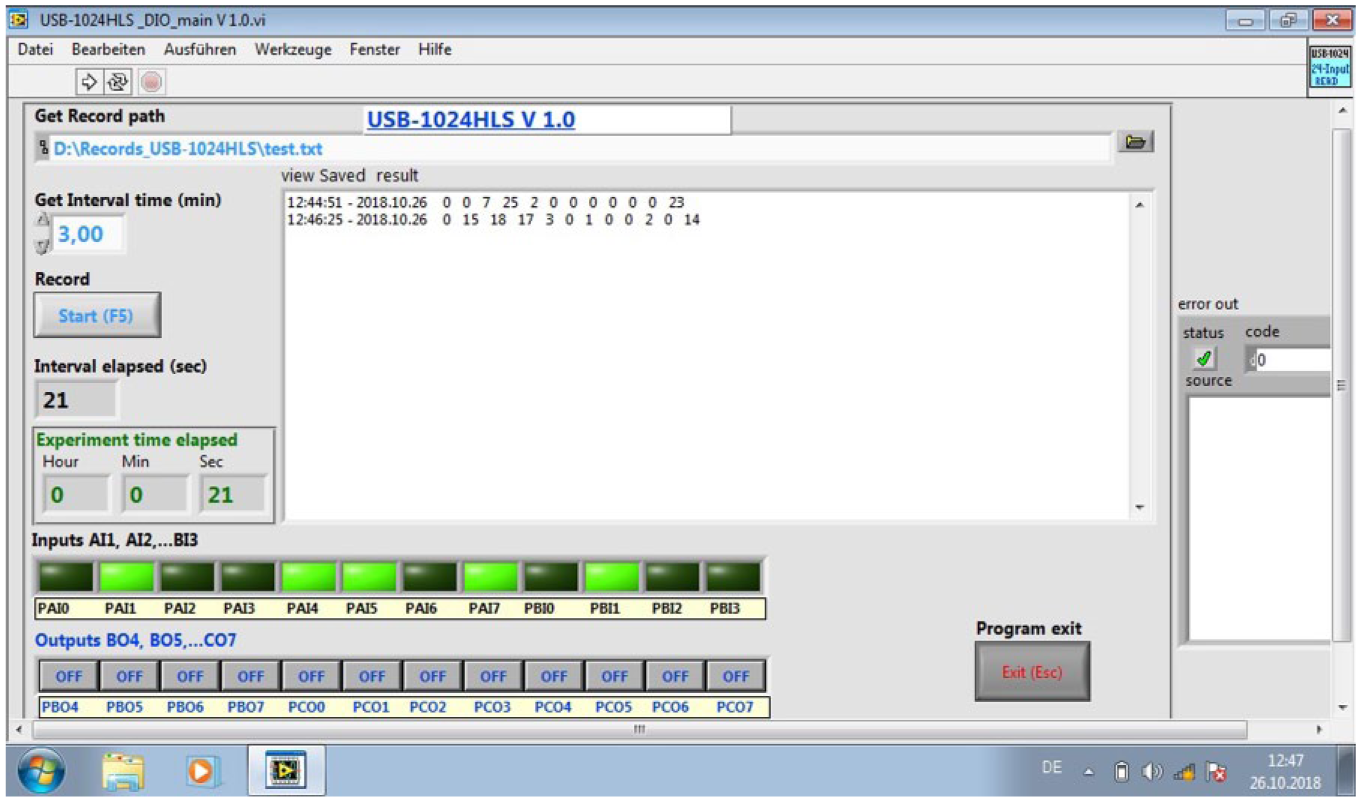
Graphical user interface. A folder can be selected to choose where to save the acquired data (top line). The recording interval time (minutes) can be set in the second line. The elapsed time since acquisition started and all motion events that were detected and saved are displayed (middle section). All channels (1–12) that are activated for detection and recording are shown below.
Adult medaka fish (
Results and Discussion
We have used two of the new systems in parallel with up to 12 IR sensors per system to detect patterns of general locomotor activity and feeder activation. The data volume generated by the system is very small. For example, a 1-month recording period results in a text file of <1 MB; thus, long-term activity observation is possible. We have successfully tested our setup in an experimental paradigm using small freshwater fish (zebrafish and medaka). In a typical experiment we recorded general locomotor activity and, in parallel, feeder activation in tanks containing up to 14 adult fish for several weeks. The saved data were processed by using the Chronobiology software “El Temps” (Antoni Díez-Noguera, University of Barcelona, Spain: http://www.el-temps.com/principal.html) and a circadian software plot for generating a graph of locomotor and feeding activity (actogram) ( Fig. 4 ). This software allows a user-friendly conversion of the primary data of activity recordings (sensor activations) into graphical representations (actograms). This software package also provides tools for statistical analysis (e.g., mean waveforms, analysis of periodicity patterns [not shown]) that enable the interpretation of activity recordings and activity pattern detection.
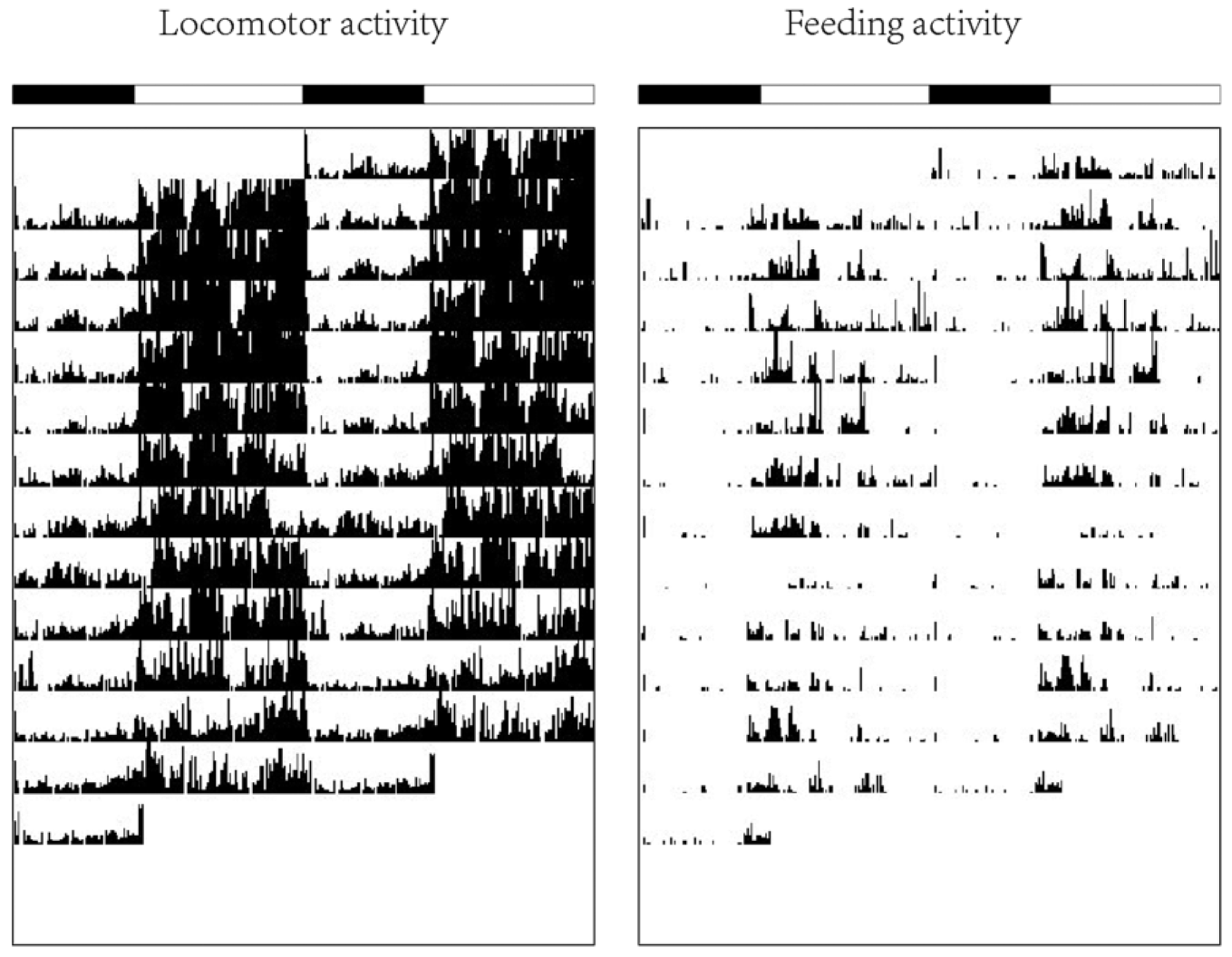
Representative actograms of locomotor and feeding activities of medaka groups. Actograms of locomotor (left panel) and self-feeding (right panel) activities of seven males and females during a 12 days self-feeding experiment. The height of each bar represents the number of sensor activation/10 min. Light and dark phase are indicated by white and black bars (x-axis top, 14:10 LD). An example of primarily diurnal locomotor and feediing activity is shown.
Figure 4 shows a representative example of a general locomotor and feeding activity recording from a group of 14 adult medaka fish over a period of 12 days. The binning period during the data acquisition was set to 10 min to allow a detailed graphical representation of the activity patterns. The height of each vertical black bar depicts the number of activations during this binning period. Thus, the plot is a quantitative representation of the group activity. In these actograms each 24 h period is plotted twice on consecutive lines of the plot to facilitate the detection of activity patterns. Since the activity of an entire group of fish is recorded, the plot does not display activities of individuals but rather an averaged pattern of the entire group. The left-hand panel depicting the recording of locomotor activity reveals that, consistent with our previous findings, 14 the activity of the medaka fish is predominantly restricted to the daytime (represented by white bars above the plot). Thus, the fish display diurnal locomotor activity. Minor bouts of activity during the nighttime represent brief periods of swimming that interrupt sleeping phases characterized by scant swimming activity. The simultaneously recorded feeding activity displayed in the right panel shows predominant diurnal feeding, with peak feeding starting at the onset of the light phase, which then subsequently declines during the remainder of the light period. Thus, our recording shows that both locomotor and feeding activities of medaka under a 14:10 h light-dark cycle are mainly diurnal. In addition, the recording shows a stable diurnal feeding pattern indicating that medaka learn rapidly how to activate the feeder system.
Our system enables simple recording of the activity patterns of groups or individual fish. Importantly for the activity recording, fish can be kept in isolation to prevent external stimuli from interfering with experimental data. The system allows simultaneous recording of both locomotor and feeding activity and thus provides important species-specific information on key behavioral parameters. For example, such a system would be appropriate to study the impact of environmental conditions such as seasonal changes in photoperiod, or the timing of feeding behavior in fish models. More generally, data obtained from this system could be relevant for optimizing experimental design and improving husbandry conditions.
The methodology is based on IR technology that does not interfere with fish behavior as it is below the spectrum that is detected by commonly used fish models. Furthermore, the setup does not require surgical manipulation of the animal as the external IR sensor system works noninvasively. Importantly, the noise of the IR sensor system is very low and thus provides a faithful recording of activity patterns.
It should be noted, however, that very low activity levels, for example, recordings from single fish in large tanks, may result in data sets that do not permit robust statistical analysis. Thus, tank size and sensitivity of the sensor system have to be adapted to the specific experimental purpose.
The simultaneous recording of general locomotor activity and feeding activity provides important additional information on basic behavioral patterns. Our food-on-demand system is easy to install and can be coupled with most commercially available feeder systems. It thus provides a simple, versatile setup to analyze key behavioral parameters as well as important information for the experimental use of laboratory fish models.
The printed circuit board (PCB) layout of the electronic circuit, the bill of materials, and the software file used in the design and operation of the hardware are freely available and can be downloaded from the approved online repository: https://osf.io/ndb65/.
Supplemental Material
DS_DISC841412 – Supplemental material for DIY Automated Feeding and Motion Recording System for the Analysis of Fish Behavior
Supplemental material, DS_DISC841412 for DIY Automated Feeding and Motion Recording System for the Analysis of Fish Behavior by Christian Pylatiuk, Haiyu Zhao, Eduard Gursky, Markus Reischl, Ravindra Peravali, Nicholas Foulkes and Felix Loosli in SLAS Technology
Footnotes
Acknowledgements
The authors acknowledge the excellent fish husbandry of Nadeshda Wolf and Natalja Kusminski. We also thank Jose Fernando Lopez Olmeda for installing an earlier version of the feeder system at KIT.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding provided by the Helmholtz Association program “BioInterfaces in Technology and Medicine—BIFTM.”
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
