Abstract
The SpinVessel system provides a methodology using pulsed radial flow to gently mix and uniformly suspend particulates (cells, magnetic beads, silica beads, and microcarrier beads) for automated assays. SpinVessels are well suited for aliquoting on robotic liquid handlers and with robotic reagent dispensers, as well as manually. The SpinVessel system combines two critical features: (1) special internal side fins and projections in the bottom of the vessels and (2) an instrument that quickly spins the vessels and repeatedly reverses the spin direction. This rapid reversing motion sends multiple pulses of fluid up the side walls of the SpinVessel, creating a circular radial flow pattern. We tested five different particulates and six different SpinVessels with volume capacities varying from 50 mL to 1200 mL. SpinVessels are compatible with either single-, 8-, 12-, 96-, or 384-channel pipettors or with siphon tubing on robotic reagent dispensers. Experiments have demonstrated high viability of cells and undamaged morphology of microcarrier beads even after hours of constant agitation. The uniformity of aliquots collected at various vertical depths and horizontally across the SpinVessels demonstrated that cells, magnetic beads, and silica beads were uniformly suspended throughout the height and breadth of the SpinVessels, and uniformity of samples was consistent from the beginning to the end of the aliquoting procedure. Only 5 min of mixing is required to resuspend settled particulates. This novel mixing methodology has many applications in laboratory automation where particulate aliquot uniformity and/or particulate integrity are important to automating assays.
Keywords
Introduction
There is a great need for an automation-compatible method of gently keeping a wide variety of particulates in uniform homogeneous suspension. Most particulates will aggregate, clump, or sediment over time if not dispersed. Often, the ability to keep particulates in uniform suspension is a major hurdle to overcome in automated assays involving particulates and achieving large batch and lot-to-lot consistent aliquots. 1 Suspension methods used with manual assays (pipetting up and down, vortex mixing, conventional magnetic stirring, overhead stir propellers, roller tube mixing, rocker platforms, oscillating grids, 2 or large orbital shakers3–5) are simply not compatible with automated assays. In 2002, the bubble paddle stirrer system was introduced by V&P Scientific Inc. (San Diego, CA) 6 to keep heavy yttrium oxide beads in uniform suspensions for scintillation proximity assays (SPAs). Bubble paddle stirring worked well for suspending SPA beads 7 and for many other particulates, including chromatography resin slurries. 8 However, some fragile resins were damaged by the shear forces generated by the rapidly spinning bubble paddles. 9 The bubble paddle system was also limited to small-volume runs of under 450 mL. Another solution was to use conventional magnetic stirrers with magnetic stir bars. However, the centrifugal currents that stir bars produce spin the heavy particulates to the wall of the vessel, where they tend to congregate in greater numbers, and lighter particles like cream collect in the center (vortex) of the vessel, which is the principle that old-fashioned cream separators use. Fortunately, many bacterial cells have strong cell walls that are resistant to damage by magnetic stir bars. LaCroix et al. 10 demonstrated the ability of V&P Scientifics’s vertical magnetic disc tumble stirring system to aerate, uniformly suspend, and sequentially sample on a robotic liquid handler and generate an automated growth curve using Escherichia coli. Vertical tumble stirring with magnetic discs eliminates the problem of having particulates collecting on the walls of the vessel, as particulates with this method are propelled vertically to the top of the vessel rather than to the side of the vessel. Unfortunately, this method is limited to a 50 mL volume.
With the advent of magnetic bead solid-phase immunoassays and biomolecule purification (RNA, DNA, protein, and cell selection), several methods of suspending the magnetic beads have been employed, such as vortexing, roller bottle mixing, overhead propeller mixing, magnetic stirring, and sonication. 1 Martinez 1 rejects the first four mixing methods for magnetic beads as unworkable because they do not generate enough energy to pull aggregated beads apart. He also states that “sonication, although complex and difficult to master in large volumes, can pull the aggregates apart.”
Methods of magnetic bead suspension have been developed by Dexter Magnetics Technologies (Elk Grove Village, IL) using magnetism 11 and by V&P Scientific using direct drive bubble paddles and parylene-coated bubble paddles. 9 Unfortunately, both of these methods do not work with volumes more than 450 mL. Another approach to solving the magnetic bead suspension problem is to make magnetic beads so small and so close to the density of water that they stay uniformly suspended for long periods of time. 12 Stange et al. 13 introduced a method of mixing and suspending superparamagnetic beads using variable electromagnetic fields; however, it is only capable of suspending small volumes of material (less than 5 mL). Dexter Magnetic Technologies has recently introduced a device that resuspends magnetic beads and pulls aggregated magnetic beads apart. However, they do not specify the volume it is capable of handling, but it appears to be less than 300 mL 14 and is no longer on the market.
V&P Scientific has recently developed a new line of devices called SpinVessels (U.S. and foreign patents pending), which work without internal moving parts within the solution to be mixed and therefore produce less damaging shear forces. An added benefit is that SpinVessels will suspend both magnetic and nonmagnetic particulates in volumes up to 1200 mL and possibly beyond. The SpinVessel systems were designed specifically to work on robotic liquid handlers with 1-, 2-, 3-, 4-, 5-, 6-, 7-, 8-, 9-, 10-, 11-, 12-, or 96-channel pipette heads with 9 mm center-to-center spacing; 384-pipette tip heads; or robotic reagent dispensers that utilize siphon tubing to facilitate the automation of a variety of assays. These SpinVessel devices keep particulates in uniform suspension without damaging them by using a pulsed radial flow (PRF) methodology. PRF mixing is produced by rapidly spinning vessels with internal side fins at the vessel bottom to create a rapid current of circular flowing fluid and repeatedly reversing the spin direction. When the spin direction is reversed, the circular current strikes the reversing side fins and the fluid current vector is directed up the side walls of the vessel as a pulse. Each spin direction reversal sends another pulse of fluid up the vessel side wall. The number of direction reversals per minute times the number of side fins equals the number of pulses generated per minute. When spun rapidly enough and reversed often enough, a continuous stream of pulses moves the particulates up the wall of the vessel. PRF’s circular mixing loop is completed by the particulates flowing down the center of the vessel to the bottom, and this keeps the particulates in uniform suspension throughout the vessel, as shown in
Figure 1A
and
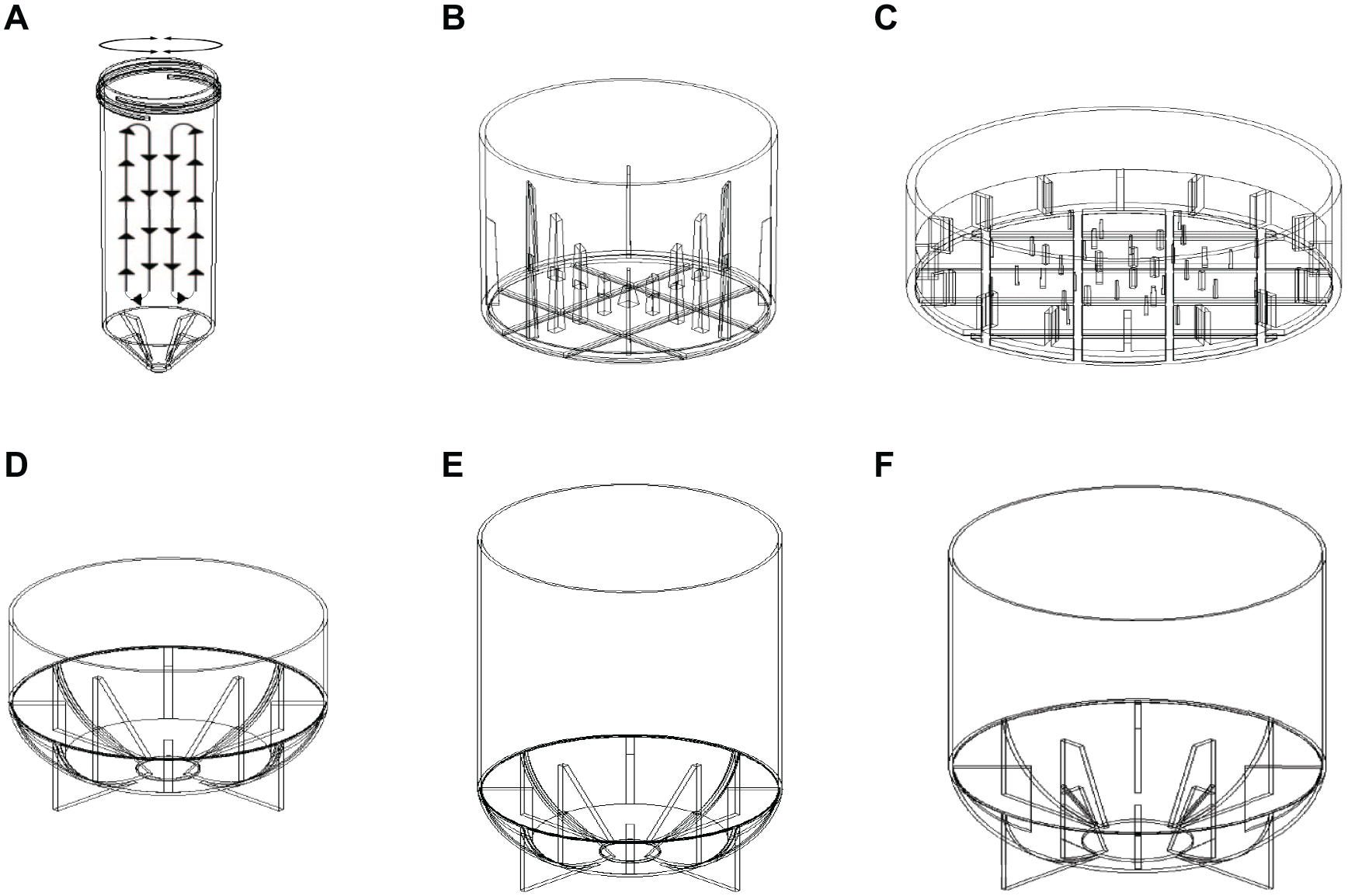
Schematic drawing of a 50 mL SpinVessel illustrating PRF and drawings of six SpinVessels. (
Materials and Methods
SpinVessel Systems
We performed a series of experiments to test the gentleness of the mixing and the uniformity of particulate aliquots with six different SpinVessels (V&P Scientific), shown in
The SpinVessel side fins and projections were designed so they will not contact a robotic liquid handler’s pipette tips when the SpinVessels are rotated. This allows continuous mixing while aliquots are withdrawn. Four of the six SpinVessel motor units (V&P Scientific) shown in

SpinVessels mated to a robotic liquid handler and a SpinVessel mated to a robotic reagent dispenser. (
The key to making PRF mixing work was coupling SpinVessels that have internal side fins to a positive engagement mechanism on a motor capable of spinning the SpinVessels and repeatedly reversing the spin direction after a few rotations with a smart controller. The speed in revolutions per minute (RPM) and the inner circumference of the SpinVessel in meters determine the linear speed of the circulating current in meters per minute, which is the critical measure that determines the force necessary to create sufficient PRF to establish a mixing circular loop. The VP 830SV-50-25 SpinVessel has the smallest internal circumference at 0.085 m and an optimal RPM of 375 RPM, yielding a circulating current speed of 31.8 m/min, while the largest-diameter SpinVessel (VP 830SV-650) has an internal circumference of 0.441 m and an optimal RPM of 155, yielding a linear circulating current speed of 68.4 m/min. The rule of thumb is the larger the SpinVessel circumference, the lower the RPM needs to be to keep the particulates in uniform suspension because the linear circulating current speed is most important and is determined by the RPM multiplied by the SpinVessel inner circumference. We found that linear circulating current speeds between 31.8 m/min and 68.4 m/min would produce good PFR mixing, while circulating currents at 88.2 m/min or greater speeds would produce frothing 15 (small bubble formation produced by mechanical agitation of polar proteins). Clarkson et al. 16 has demonstrated that foam or frothing denatures protein at the gas–liquid interface. However, if proteins are not present in the liquid vehicle, then greater speeds are possible.
Although we did not directly test the effect of particulate density on the PRF force necessary to produce effective PRF loop mixing, we did observe uniform particulate suspensions using all the different density particulates (silica beads, magnetic beads, CHO cells, HEK cells, and Cytodex dextran beads) in aqueous suspension. The linear circulating current speed was found to be the most important factor for PRF mixing. The number of rotations before direction reversal was best between 1 and 4 and needed to be determined for each SpinVessel, each particulate, and each liquid vehicle and its viscosity. The internal views of the SpinVessel side fins are shown in
The SpinVessel touch screen controllers allow easy user setting of speed (RPM), the number of rotations before reversing the spin direction, the length of pause between rotational reversal, and the length of time the repetitive program runs (
Five Different Particulates Tested
A human embryonic kidney (HEK) suspension cell line (FreeStyle 293-F, cat. R79007) was obtained from Thermo Fisher Scientific (Waltham, MA) and diluted to 420,000 cells/mL and 760,000 cells/mL in growth medium.
Chinese hamster ovary (CHO) adherent cell lines (CHO-K1 and ATCC CCL-61) were obtained from ATCC (Manassas, VA) and diluted to 178,000 cells/mL in growth medium.
Cytodex dextran microcarrier beads (190 µm diameter) were obtained from Sigma-Aldrich (St. Louis, MO) and diluted to 5 mg/mL in water.
Unlinked silica beads (1.2 µm diameter) were obtained from Illumina (San Diego, CA) and diluted in water to 10 mg/mL or 6.25 mg/mL.
MagneSil paramagnetic beads (10 µm diameter) were obtained from Promega (Madison, WI) and diluted in water to 50 ng/mL.
HEK Cell Counts and Viability Assays
The HEK suspension cell line (FreeStyle 293-F) was cultured in FreeStyle 293 expression medium in 125 mL Erlenmeyer cell culture vented flasks (CLS431143) (Corning, Corning, NY) and incubated on an orbital shaker at 37 °C. Cell counts and viability were determined using the Chemometec Via1-Cassette (Chemometec, Gydevang, Denmark) together with the NucleoCounter NC-200 (Chemometec). Viability and cell concentration were determined by drawing a 60 µL sample into the Via1-Cassette. The inside of the Via1-Cassette is coated with two different dyes, staining the entire population of cells and nonviable cells, respectively. The 60 µL volume of each Via1-Cassette had been calibrated to give a high precision of the resulting count.
Viability of HEK Cell Assays
Five different SpinVessels (VP 830SV-300FB, VP 830SV-350RB, VP 830SV-650FB, VP 830SV-850CB, and VP 830SV-1200CB) were filled to 50% of capacity with a suspension of HEK cells (420,000 cells/mL) in FreeStyle 293 expression medium and mixed by PRF for 3 h at room temperature, at a speed setting of 155 RPM and repeatedly reversing direction after three rotations. Samples were removed manually from the top 10 mm of the suspension and the cell count and viability determined as described above.
Time to Resuspend Settled HEK Cell Assays
A VP 830SV-1-650FB SpinVessel was filled with 325 mL of HEK cells (760,000 cells/mL) in FreeStyle 293 expression medium and mixed by PRF for 2 h at a speed setting of 155 RPM and repeatedly reversing direction after three rotations. A sample was removed manually for counting and viability from the top 10 mm of the suspension, and then the vessel was allowed to rest for 30 min and another sample was removed for counting and viability from the top 10 mm of the suspension. Next, the solution in the SpinVessel was mixed by PRF for 5 min at a speed setting of 155 RPM and repeatedly reversing direction after three rotations, and again a 60 µL sample was removed for counting and viability from the top 10 mm of the suspension.
HEK Cell Suspension Assays for Horizontal Uniformity
A VP 830SV-1-650FB SpinVessel was filled with 325 mL of HEK cells (620,000 cells/mL) in FreeStyle 293 expression medium and mixed by PRF for 2 h at a speed setting of 155 RPM and repeatedly reversing direction after three rotations; a sample was removed for counting and viability. Twelve samples were simultaneously collected with a 12-channel manual pipettor submerged 10 mm into the SpinVessel, and each sample was counted, and the viability determined as previously described.
CHO Cell Counts and Viability
CHO adherent cells were harvested according to the ATCC protocol using 0.25% (w/v) trypsin and then diluted in DiscoverX medium from the DiscoverX Assay Complete Cell Culture Kit-107 (DiscoverX, Fremont, CA). Cell counts and viability were determined using the Chemometec Via1-Cassette together with the NucleoCounter NC-200. Viability and cell concentration were determined by drawing a sample into the Via1-Cassette as previously described for HEK cells.
CHO Cell Suspension Assays for Horizontal Uniformity
A VP 830SV-1-650FB SpinVessel was filled with 350 mL of CHO cells (178,000 cells/mL) in DiscoverX medium and mixed by PRF for 1.5 h at a speed setting of 155 RPM while repeatedly reversing direction after three rotations, and a sample was removed for counting and viability. Because of the 130 mm Z-height pipetting restrictions on the FeliX robot, the SpinVessel was removed from the motor mechanism and placed stationary on the deck of a FeliX robot (Analytik Jena, Jena, Germany). The FeliX was then able to pipette 384 samples of 25 µL and transfer them to a 384-well microplate (Greiner Bio-One, Frickenhausen, Germany, cat. 781945) using 384 sterile 60 µL pipette tips submerged 10 mm into the SpinVessel (4450 CHO cells/well). The microplate was cultured for 24 h at 37 °C. The SpinVessel was not producing PRFs at this point. After 24 h of culture, 25 µL of CellTiter-Glo reagent (Promega) was added to each well and the microplates were incubated for 15 min and read on a PHERAstar plate reader (BMG Labtech, Ortenberg, Germany) to determine the relative luminescence units (RLU) in each well. A CellTiter-Glo cell standard curve was generated and used to convert RLU in the microplate wells to cells per well. The average number of cells per well, the standard deviation, and percent coefficient of variation for each 384-well plate, for each of the 24 columns of 16 pipettes, and for each of the 16 rows of 24 pipettes were calculated. Additionally, light micrographs of the cell monolayer in wells of the 384-well microplate were taken on a Model DM IL LED light microscope at 200× (Leica Microsystems, Buffalo Grove, IL).
Gentleness Assay for Cytodex Dextran Microcarrier Beads
A VP 830SV-50-25 SpinVessel was filled with 25 mL of Cytodex dextran microcarrier beads (0.125 g of beads in 25 mL of water) and the beads were allowed to swell to ~190 µm in diameter. The SpinVessel was spun at 70 RPM and the spin direction was repeatedly reversed after every three rotations. A comparison test was also performed on a similar suspension of Cytodex dextran microcarrier beads that were added to a VP 756B Bubble Paddle resuspension reservoir on a VP 710C5-7A Magnetic Tumble Stirrer at 650 RPM (V&P Scientific). Another comparison test was also performed on a similar suspension of Cytodex dextran microcarrier beads, which was placed in a conventional 50 mL centrifuge tube with a VP 772DP-N42-24-3 stir disc and stirring on a VP 710C5-7A Magnetic Tumble Stirrer at 650 RPM (the speed required to completely mix the beads). Samples of 25 µL were collected by pipette from each of the three different mixing vessels and the morphology was examined on slides under a coverslip and photographed at 100× magnification on a Model M150C-E5 digital microscope (AmScope, Irvine, CA) at 0 min, 15 min, 30 min, 60 min, 24 h, and 7 days of mixing.
Timed Silica Bead Suspension Assays for Settled Beads
Fifty milliliters of 1.2 µm diameter unlinked silica beads obtained from Illumina at 10 mg/mL were allowed to settle for 7 min to the bottom of a VP 830SV-50-25 SpinVessel mated to a VP 418SV1-1-50CB on the deck of an OT-2 automatic liquid handler (Opentrons, New York, NY). The OT-2 was equipped with a P50 pipette (Opentrons) with a 300 µL pipette tip, and once beads settled to the bottom, an initial 50 µL sample of settled bead solution was collected by pipette at the 40 mL level. Then, the SpinVessel was spun at 375 RPM and the spin direction repeatedly reversed after every 2.75 rotations. Fifty-microliter samples were taken at the 40 mL level every 16 s for the first 5.5 min and then every 5 min after that. The 50 µL samples were diluted into 50 µL of water in a 96-well polystyrene flat-bottom microplate (Costar cat. 3585, Corning), and absorbance was read at 405 nm on a Victor II spectrophotometer (PerkinElmer, Waltham, MA).
Silica Bead Suspension Assays for Vertical Uniformity
Two hundred milliliters of 1.2 µm diameter unlinked silica beads obtained from Illumina (10 mg/mL) were allowed 7 min to completely settle to the bottom of a VP 830SV-300FB SpinVessel on the deck of an OT-2 automatic liquid handler, equipped with an eight-channel P 300 µL pipette and then sampled at 35 mm from the bottom of vessel for initial sample of the settled bead solution. The sample was collected with the OT-2 eight-channel pipette, removing three replicates of 50 µL of beads per pipette. Each pipette was separated by 9 mm on a center-to-center spacing. The SpinVessel was then spun at 200 RPM and the spin direction repeatedly reversed after each single rotation. After 5 min of PRF mixing, three replicates of 50 µL robotic samples were taken across the vessel at 35 mm above the bottom of the vessel, followed by three replicates of 50 µL robotic samples taken at 25 mm, 15 mm, and 10 mm above the bottom of the vessel. Twenty-five minutes had elapsed by the time the last (10 mm samples) were taken. All samples were diluted in 50 µL of water in a 96-well microplate as previous described, and absorbance was read at 405 nm on a Victor II spectrophotometer.
Silica Bead Suspension Assays for Horizontal and Vertical Uniformity
Two hundred milliliters of 1.2 µm diameter unlinked silica beads obtained from Illumina (10 mg/mL) were added to a VP 830SV-300FB SpinVessel on the deck of an OT-2 automatic liquid handler, equipped with an 8xP 50 µL pipette. The SpinVessel was then spun at 200 RPM and the spin direction repeatedly reversed after each single rotation. After 5 min of PRF mixing, eight 50 µL samples spaced 9 mm apart were robotically collected across the vessel at 35 mm, 25 mm, 15 mm, and 10 mm above the bottom of the vessel, with the OT-2 eight-channel pipette. Three replicates of eight samples were taken in this manner and diluted in 50 µL of water in a 96-well microplate as previously described; absorbance was read at 405 nm on a Victor II spectrophotometer.
Magnetic Bead Suspension Assays to Determine Vertical Uniformity
Fifty microliters of 10 µm diameter MagneSil paramagnetic beads (50 ng/mL) was added to a VP 830SV-50-25 SpinVessel on the deck of an OT-2 automatic liquid handler, equipped with a P50 pipette and a 300 µL pipette tip. The SpinVessel was then spun at 375 RPM and the spin direction repeatedly reversed after every 2.75 rotations. After 5 min of PRF mixing, 24 serial 50 µL robotic samples were taken at 55 mm above the bottom of the vessel, followed by 24 serial 50 µL robotic samples taken at 45 mm, 35 mm, and 25 mm above the bottom of the vessel and diluted into 50 µL of water in a 96-well microplate, as previous described, and absorbance was read at 405 nm on a Victor II spectrophotometer.
Silica Bead Suspension Assays for Uniformity from Beginning to End of Aliquoting
A 250 mL volume of 1.2 µm diameter unlinked silica beads obtained from Illumina (6.25 mg/mL) was mixed by spinning a VP 830SV-300FB SpinVessel at 155 RPM and repeatedly reversing the spin direction after three rotations. After 5 min of PRF mixing on the deck of an OT-2 automatic liquid handler, equipped with an 8× P300 µL pipette with 200 µL filter barrier tips that were 77.4 mm long (Dragon Laboratories, Bath UK), 100 µL was removed from a sampling depth of 4 mm above the SpinVessel bottom for all eight pipette tips throughout the aliquoting process. The eight replicate 100 µL samples were then dispensed into a column of a 96-well microplate containing 100 µL of water. This process was repeated until all 12 columns of the microplate were filled and a new microplate was started to be filled. All the diluted samples had the absorbance read at 405 nm on a Victor II spectrophotometer
Results
Viability of Cells after PRF Mixing
The results from using five different SpinVessels that were 50% filled with a suspension of HEK cells and mixed by PRF for 3 h at a speed setting of 155 RPM while repeatedly reversing direction after three rotations are shown in Table 1 , along with the initial count and viability of the HEK cells prior to starting the mixing. The maximum working volumes for the five SpinVessels were 300 mL, 350 mL, 650 mL, 850 mL and 1200 mL. The five different SpinVessels all have different shapes—flat bottom versus conical bottoms or round bottoms—and hold different volumes of liquid. Neither the volume or shape of the first four SpinVessels appeared to significantly affect the cell count or viability, while the fifth SpinVessel (VP 830SV-1-1200) cell count actually increased significantly from 420,000 cells/mL to 500,000 cells/mL. This could be due to breaking up clumps of cells during the course of mixing. Also, the viability of the cells was excellent for all five SpinVessels even after 3 h of continuous PRF mixing.
Effect of 3 h of PRF Mixing on the Number and Viability of HEK Cells with Five Different SpinVessels.

None of the larger circumference SpinVessels (from 300 mL to 1200 mL) produced significant frothing of cell growth medium over the course of 3 h of PRF mixing when used at a speed setting of 155 RPM. We had previously observed that speeds of 200 RPM produced frothing of the culture medium. 15 Because frothing is associated with the denaturization of proteins, we avoided speeds of 200 RPM with the larger-circumference SpinVessels. 16
A VP 830SV-650FB SpinVessel was used to demonstrate how fast PRF mixing was able to resuspend settled HEK cells and again demonstrated the high cell viability of PRF mixing (see Table 2 ). In this experiment, the SpinVessel was mixed at 155 RPM and repeatedly reversed after three rotations for 2 h, and 60 µL samples were taken from the top 10 mm of the SpinVessel. Data were obtained for the initial count, after mixing for 2 h, after 30 min of no mixing, and again after 5 min of resuming mixing.
Effect of 2 h of PRF Mixing in a VP 830SV-1-650FB SpinVessel on the Number and Viability of HEK Cells after Allowing the Cells to Settle and Then Be Resuspended.

After letting the HEK cells settle for 30 min, another sample was taken to determine the number of cells remaining in suspension and their viability prior to resuming the mixing for 5 min to resuspend settled HEK cells.
Table 3 demonstrates that a VP 830SV-1-650FB SpinVessel was able to keep HEK cells uniformly suspended and at least 95.3% viable for 2 h with PRF mixing at 155 RPM and reversing direction after three rotations. Twelve samples were taken spaced 9 mm center-to-center apart horizontally across 99 mm of the SpinVessel with a 12-channel pipette.
Results of HEK Cell Uniformity after 2 h of PRF in a VP 830SV-1-650FB SpinVessel.

Samples were collected with a 12-channel pipette from the top 10 mm of the liquid.
In a separate experiment using a VP 830SV-650FB SpinVessel, CHO cells were PRF mixed at 155 RPM and reversed after three rotations for 1.5 h, and 25 µL samples were taken from the top 10 mm of the SpinVessel. The viability was greater than 96.9% at 178,000 cells/mL. The CHO cells were inoculated (25 µL/well) into the wells of a 384-well microplate (4450 cells/well) using a FeliX liquid handler equipped with a 384-channel pipette head. The number and viability of cells in the wells were determined after 24 h of incubation by RLU and a standard curve using a PHERAstar plate reader. The cells had nearly doubled to 8190 cells/well from the seeding number of 4450 CHO cells. Chida et al.
17
have reported that the doubling time for CHO cells is reported to be ~24 h. The standard deviation between all 384 wells was 278 cells, with a coefficient of variation of 3.9%. A photomicrograph of the well monolayers illustrated a normal morphology (
Effect of Three Mixing Methods on the Morphology of Cytodex Dextran Beads
PRF mixing with a VP 418SV-1-4-50CB mated to a VP 830SV-50-25 SpinVessel and spun at 70 RPM (the speed required to keep the beads suspended) and reversing spin direction after three rotations.
Bubble paddle mixing with a V&P Scientific Bubble Paddle reservoir (VP 756B), which was spun continuously by a VP 710C5-7A Tumble Stirrer at 650 RPM (the speed required to keep the beads suspended).
A magnetic stir disc (VP 772-DP-N42-24-3) in a 50 mL centrifuge tube was spun continuously by a VP 710C5-7A Tumble Stirrer at 650 RPM (the speed required to keep the beads suspended).
Samples of 25 µL from all three continuous mixing systems were collected from the top 10 mm of the liquid at 0 min, 15 min, 30 min, 60 min, 24 h, and 7 days. The samples were placed on glass slides with a coverslip and examined under a light microscope. Photographs were taken of the beads over time and are shown in Figure 3 .
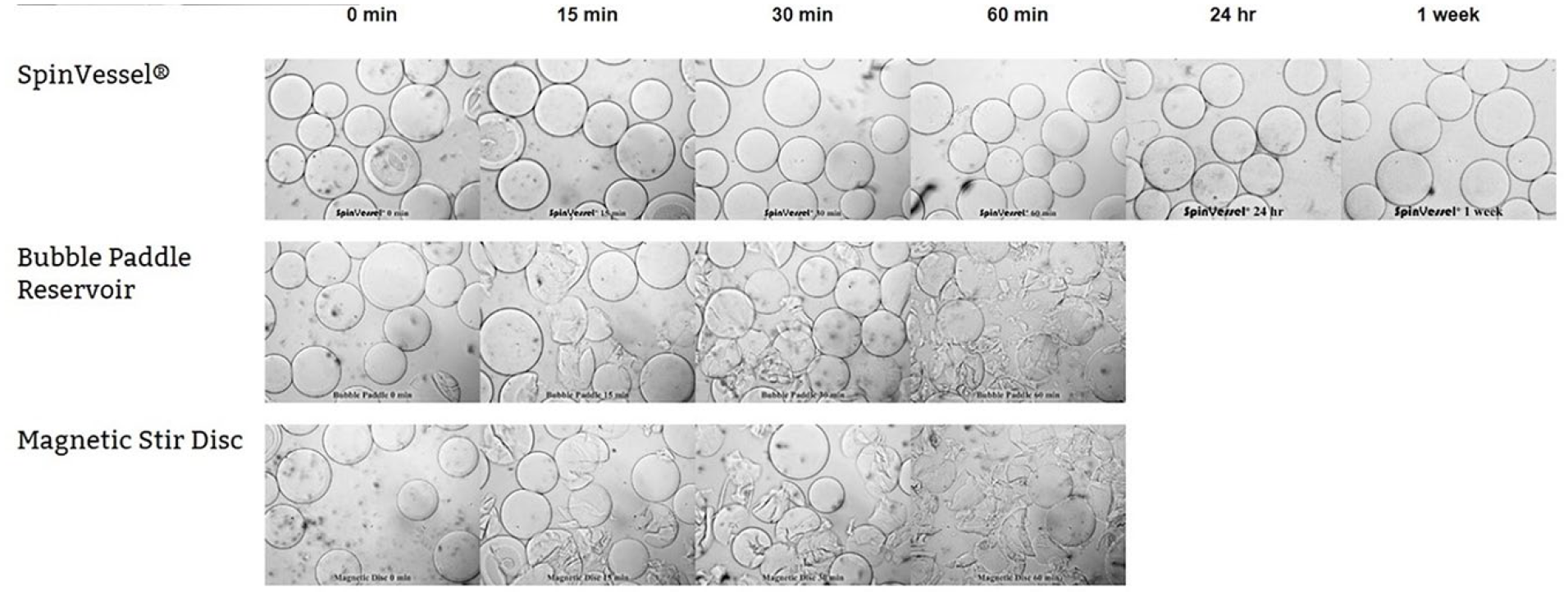
Comparison of SpinVessel PRF mixing to bubble paddle mixing and magnetic stirring of dextran microcarrier beads. A time course experiment demonstrated that both the bubble paddle and magnetic disc mixing destroyed the dextran microcarrier beads in 60 min, while the morphology of the dextran microcarrier beads had not changed even after a week of continuous mixing using PRF with a SpinVessel.
Results of CHO Cell Horizontal Uniformity Assays
The uniformity of the average cell numbers in both the columns and rows of microplate 1 (
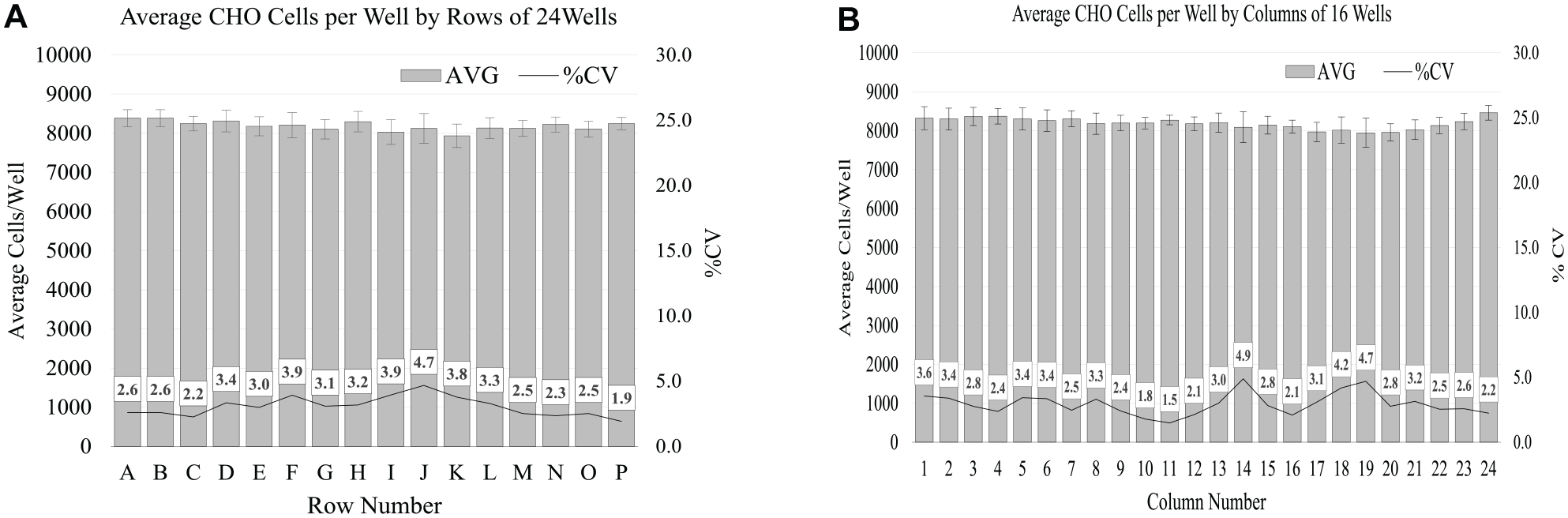
Results of adherent CHO cells’ horizontal uniformity after 1.5 h of PRF mixing. Samples of 25 µL were collected with a 384-channel pipette head from the top 10 mm of the liquid approximately 30 s after the VP 830SV-650FB SpinVessel was stationary and dispensed into a 384-well microplate. (
It should also be noted that the center projections in the VP 830SV1-650FB SpinVessel would interfere with the 384-channel pipette tips if they were lowered to the bottom of the vessel while the vessel was spinning. Even though it is possible to use this SpinVessel with a 384-channel pipette head, if the pipette tips are not lowered to the bottom, the dead volume of the cell solution would be larger than using a 96-channel pipette head and tips that can be lowered all the way to the bottom of the vessel.
Silica Bead Assays for Settled Beads
Experiments were performed to determine how quickly a VP 830SV-50-25 SpinVessel could uniformly suspend 1.2 µm silica beads. The data in Figure 5 demonstrate that the silica beads are resuspended to uniformity within 4 min of PRF mixing at 375 RPM and reversal after 2.75 rotations.
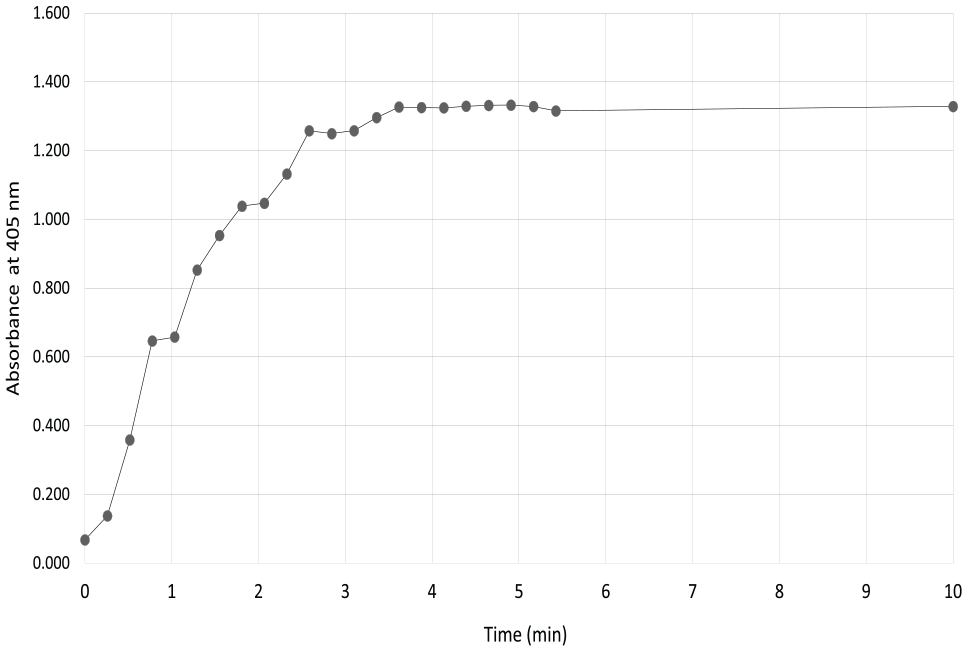
The time course to uniformity of a 1.2 µm silica bead suspension in a VP 830SV-50-25 SpinVessel was shown by 50 µL samples collected at 16 s intervals for the first 5.5 min by a single pipette sampling at a depth of 10 mm into the liquid of the SpinVessel that was filled with 50 mL of beads. Not shown are data points at 15, 20, and 25 min that were the same as the 10 min absorbance reading; they were left off the graph to better illustrate the suspension curve.
Vertical Uniformity Assays of Magnetic and Silica Bead Suspensions
Experiments were performed to determine the vertical uniformity of both a magnetic and 1.2 µm silica bead suspension in a VP 830SV-50-25 SpinVessel with a single pipette on the Opentrons OT-2 robotic liquid handler by collecting 24 replicate 50 µL samples at various vertical depths in the SpinVessel while PRF mixing. The data in
Vertical and Horizontal Uniformity Assays of Silica Bead Suspensions with an Eight-Channel Pipette
Experiments were performed to determine the vertical uniformity of a silica bead suspension in a VP 830SV-300FB SpinVessel with an eight-channel pipettor on the OT-2 robotic liquid handler, by collecting three replicate samples, eight at a time horizontally across the SpinVessel at various vertical depths. The data in
Silica Bead Suspension Assays for Uniformity from Beginning to End of Aliquoting
Experiments were performed to determine the uniformity of silica bead aliquoting from a VP 830SV-300FB SpinVessel with an eight-channel pipettor on the OT-2 robotic liquid handler, by collecting eight replicate 100 µL samples at 4 mm above the SpinVessel bottom with a starting volume at 250 mL in the SpinVessel. The mean absorbance and standard deviation for the aliquots were determined for each microplate as a whole.
The data in
The lower absorbance data in Table 4 , tips 3, 4, 5, and 6 in column 10 and tips 4, 5, and 6 in column 11 (noted by bold numerals), illustrate the curvature of the liquid-level surface characteristic of PRD mixing since the SpinVessel liquid height is higher at the SpinVessel edge and is depressed in the middle as fluid is being driven up the edge of the vessel and comes down in the center of the vessel. Consequently, the pipette tips near the center depression (tips 3–6) will be the first to not fill upon aspiration. The remaining volume where aliquoting is no longer uniform is therefore called the “dead volume.” In this case, when measured there was 25 mL of dead volume remaining in the SpinVessel. The turbulence of the mixing and the dead volume will be affected by the RPM speed and the inner circumference of the vessel, the number of rotations reversals, the number of side fins, and the viscosity of the liquid. Consequently, dead volume will have to be empirically determined for each SpinVessel and the experimental conditions.
Results of Aliquoting Experiment Using Silica Beads, a VP 830SV-300FB SpinVessel, PRF Mixed at 155 RPM, Reversal after Three Rotations, and Aliquots with an Eight-Channel Pipette Head into 24 Microplates until Aliquots Were not Uniform.

Shown are the row and column means and standard deviations of silica bead 450 nm absorbance readings of the last microplate to give consistent uniform results.
As can be seen in Table 4 , columns 1–9 have very small standard deviations, indicating very good uniformity of aliquots in those columns. However, columns 10 and 11 illustrate a sudden degradation of uniformity accentuated in the center wells. Again, this progressive degradation corresponds to the curvature of PRF mixing action, and it suggests that under the following conditions—a VP 830SV-300FB SpinVessel with a starting volume of 250 mL, using 100 µL aliquots and PRF mixed at 155 RPM, and reversed after three rotations—the maximum number of aliquots that can be safely taken is 2280, while leaving 25 mL of dead volume behind. The time to complete sampling of all 2280 aliquots was 42 min.
Discussion
Gentle Suspension
V&P Scientific SpinVessel systems using PRF mixing were able to keep both HEK and CHO cells uniformly suspended for hours (HEK cells, 3 h; CHO cells, 1.5 h) without loss of viability or significant reduction in the number of cells (
Quick Resuspensions
Not only were SpinVessels able to keep particulates in suspension for a long period of time, but also they were able to resuspend settled HEK cells after only 5 min of commencing PRF in a VP 830SV-300FB SpinVessel (
Table 2
), and after only 4 min using silica beads in a more comprehensive timing experiment (
Horizontal Suspension Uniformity
The horizontal uniformity of CHO cells/well across the 384 wells, and the small percent coefficient of variation differences between the columns and rows in microplate 1 were striking (
It should also be noted that the center projections in the VP 830SV1-650FB SpinVessel would interfere with the 384 pipette tips if they were lowered to the bottom of the vessel while the vessel was spinning. Thus, although it is possible to use this SpinVessel with a 384-channel pipette head if the pipette tips are not lowered to the bottom, the dead volume of the cell solution would be greater than if one used a 96-channel pipette head, which can be lowered all the way to the bottom of the vessel. A custom SpinVessel will have to be developed to address the dead-volume issue for 384-channel pipette heads. The VP 830SV-300FB SpinVessel can be sampled with either single-, 2-, 3-, 4-, 5-, 6-, 7-, or 8-channel pipette heads as long as they are separated by 9 mm center-to-center spacing. In the same fashion, the VP 830SV-650FB can be sampled with single-, 2-, 3-, 4-, 5-, 6-, 7-, 8-, 9-, 10-, 11-, 12-, or 96-channel pipette heads as long as they are separated by a 9 mm center-to-center spacing.
Vertical Suspension Uniformity
The results of
The results of
Sample Aliquoting Uniformity from Beginning to End
The results of
If achieving the smallest dead volume is important (think expensive reagents), then the effect of speed on PRF mixing curvature at the surface of a liquid would argue for one to use the lowest speed possible that would still achieve uniform suspension of the particulates and the most level surface liquid effect. This same effect may also be achieved by reducing the speed as the liquid level approaches the bottom of the SpinVessel, which could be accomplished by a computer control mechanism.
Conclusion
V&P Scientific’s SpinVessels provide, for the first time, a gentle method for keeping cells, magnetic beads, silica beads, and dextran microcarrier beads uniformly suspended for automated aliquoting to tubes, vials, or microplates, thus facilitating the automation of many assays.
The SpinVessel motor box was designed to fit into a microplate nest on a robotic deck (127.76 mm × 85.48 mm). This ubiquitous industry-wide footprint and low motor box profile facilitates locating the SpinVessels on many automation platforms and within the Z-height requirements of many robotic liquid handlers.
This novel mixing methodology was the winner of the 2020 SLAS “Best New Product Award” and has many applications in laboratory automation where particulate aliquot uniformity and/or particulate integrity are important to automating assays.
SpinVessel Compatibility
1. Compatible robotic liquid handlers
a. Agilent: Bravo
b. Eppendorf: epMotion
c. Beckman: FX and I series
d. Dynamic Devices: Lynx
e. Hamilton: Microlab Prep, NIMBUS, STAR, and VANTAGE
f. Opentrons: OT-2.
g. PerkinElmer: JANUS, Sciclone, and Zephyr
h. Tecan: Freedom EVO and Fluent
2. Compatible robotic reagent dispensers
a. BioTek: MultiFlo FX
b. Formulatrix: MANTIS and TEMPEST
c. INTEGRA: VIAFILL and ASSIST PLUS
d. Thermo Fisher: Multidrop Combi
Supplemental Material
sj-pdf-1-jla-10.1177_24726303211008864 – Supplemental material for A Novel Method to Gently Mix and Uniformly Suspend Particulates for Automated Assays
Supplemental material, sj-pdf-1-jla-10.1177_24726303211008864 for A Novel Method to Gently Mix and Uniformly Suspend Particulates for Automated Assays by Kristi K. Myers, John P. Herich, John E. Chavez, Kathryn G. Berkey, Alan J. Loi and Patrick H. Cleveland in SLAS Technology
Footnotes
Acknowledgements
Illumina (San Diego, CA) kindly provided the unlinked 1.2 µm silica beads used in the experiments.
Novo Nordisk (Seattle, WA) kindly provided equipment to quantitate and photograph cells
Pfizer (San Diego, CA) kindly provided the photograph (
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.K.M., J.E.C., K.G.B., A.J.L., and P.H.C. have financial interests in V&P Scientific Inc., a company commercializing the technology as well as the intellectual property described herein.
Five of the six authors work for V&P Scientific, which paid for the research done on the SpinVessel.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for all the experiments was provided by V&P Scientific Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
