Abstract
We developed and made a preliminary validation of a bead-based multiplexed immunoassay for simultaneous detection of porcine serum antibodies to Actinobacillus pleuropneumoniae serovars 1, 2, 6, 7, and 12. Magnetic fluorescent beads were coupled with A. pleuropneumoniae antigens and tested with a panel of serum samples from experimentally infected pigs and with serum samples from uninfected and naturally infected pigs. The multiplex assay was compared to in-house ELISAs and complement fixation (CF) tests, which have been used for decades as tools for herd classification in the Danish Specific Pathogen Free system. Assay specificities and sensitivities as well as the corresponding cutoff values were determined using receiver operating characteristic (ROC) curve analysis, and the A. pleuropneumoniae multiplex assay showed good correlation with the in-house ELISAs and CF tests with areas under ROC curves ≥ 0.988. Benefits of multiplexed assays compared to ELISAs and CF tests include reduced serum sample volumes needed for analysis, less labor, and shorter assay time.
Introduction
Actinobacillus pleuropneumoniae is a bacterium that can cause severe respiratory infections in pigs. Sixteen A. pleuropneumoniae serovars with variable pathogenicity have been described, and distinct patterns of predominant serovars exist within different countries.5,14 In Denmark, surveillance and detection of A. pleuropneumoniae infections is partly based on serology. The serologic analyses currently employed at the National Veterinary Institute (Technical University of Denmark) for detection of antibodies to A. pleuropneumoniae are indirect and blocking ELISAs as well as complement fixation (CF) tests (http://www.vet.dtu.dk/diagnostik; http://spfsus.dk/en).6,9–12 These assays are time-consuming to perform because they generally only measure antibodies toward a single serovar at a time. However, pigs and other production animals are often suspected of being infected with multiple pathogens, and surveillance programs often include testing for several pathogens or serovars at a time, which requires analysis of the same serum samples in various types of assays with different assay conditions.
Several laboratories worldwide have implemented commercial or in-house–developed bead-based multiplex assays in order to permit simultaneous detection of antibodies to multiple pathogens in a single serum sample. Most have been developed for human testing purposes, but some have also been developed for veterinary applications.1,4,8 The majority of bead-based multiplex assays are based on a commercial platform (xMAP Technology, Luminex, Austin, TX) that utilizes magnetic or non-magnetic polystyrene beads containing fluorescent dyes that differentiate the beads into multiple regions (80 regions for magnetic beads and 100 regions for non-magnetic beads). Sets of beads from various regions can be coated with different pathogen antigens. Serum antibody bound to the beads can be detected with layers of biotinylated anti-porcine IgG and phycoerythrin (PE)-conjugated streptavidin. Finally, a flow cytometer detects both the internal fluorescence of the bead (identifies the antigen) and the bound PE (measures bound antibody).
Herein, we describe the development and preliminary validation of a bead-based multiplexed immunoassay that detects and distinguishes between antibodies toward A. pleuropneumoniae serovars 1, 2, 6, 7, and 12 within a single serum sample volume. The A. pleuropneumoniae multiplex assay is compared with well-established in-house ELISAs and CF tests. Antigens used for the development of this multiplex assay include lipopolysaccharide (LPS) and capsular polysaccharide. Some A. pleuropneumoniae serovars share identical LPS structures, and this applies to serovars 1, 9, and 11, serovars 3, 6, and 8, as well as serovars 4 and 7. 13 Serum antibodies toward serovars with identical LPS structures cross-react, and therefore the developed multiplex assay for A. pleuropneumoniae serovars 1, 2, 6, 7, and 12 should in theory also detect antibodies to serovars 3, 4, 8, 9, and 11.
Materials and methods
Serum samples
Positive control sera were obtained from experimentally infected pigs. These sera have continuously, over many years, been used as part of a quality control (QC) panel for the monthly evaluation of the ELISAs and CF tests currently employed in routine laboratory activities at the National Veterinary Institute. The positive serum samples for each of the A. pleuropneumoniae serovars 1 (n = 4), 2 (n = 5), 5 (n = 4), 6 (n = 4), 7 (n = 5), 8 (n = 2), 10 (n = 4), and 12 (n = 5) were included in the QC panel for the multiplex assay (Table 1). These sera originated from experimentally infected pigs and were collected 4–5 wk after infection, as previously described (Sørensen V. Evaluation of laboratory diagnostic assays for monitoring respiratory infections in pigs. Thesis. The Royal Veterinary and Agricultural University, Denmark, 1997). Field samples were collected between 2010 and 2016 from Danish swine herds that were found positive or negative for A. pleuropneumoniae by the ELISAs or CF tests (Table 2).
Results of the Actinobacillus pleuropneumoniae multiplex assay, using a single mix of bead types coupled with antigen from serovars 1, 2, 6, 7, and 12, that were incubated with individual experimental serum samples positive for antibodies to serovars 1, 2, 5–8, 10, and 12 (quality control panel).
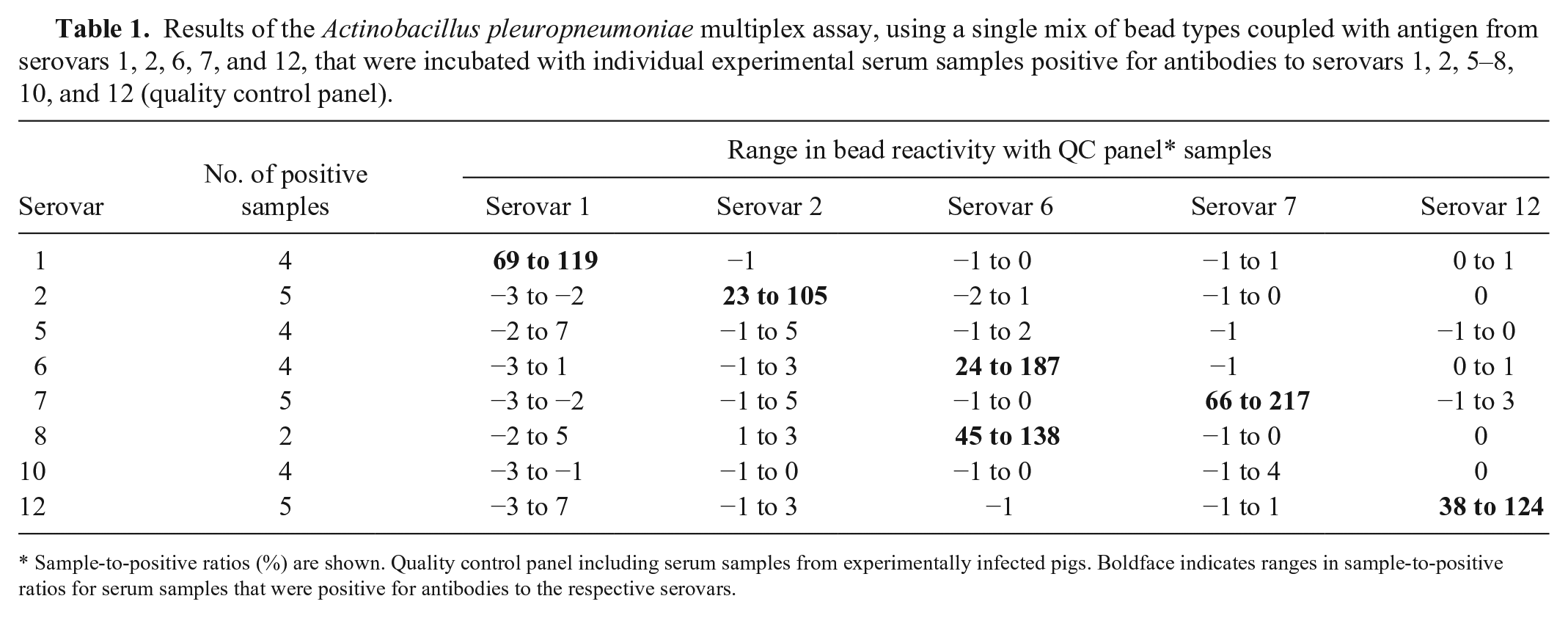
Sample-to-positive ratios (%) are shown. Quality control panel including serum samples from experimentally infected pigs. Boldface indicates ranges in sample-to-positive ratios for serum samples that were positive for antibodies to the respective serovars.
Swine serum samples included in the statistical analysis of the multiplex assay. Samples were from swine herds (Danish high-health, conventional, and foreign herds) that were positive or negative for antibodies to Actinobacillus pleuropneumoniae serovars 1, 2, 6, 7, 9, 11, and 12.
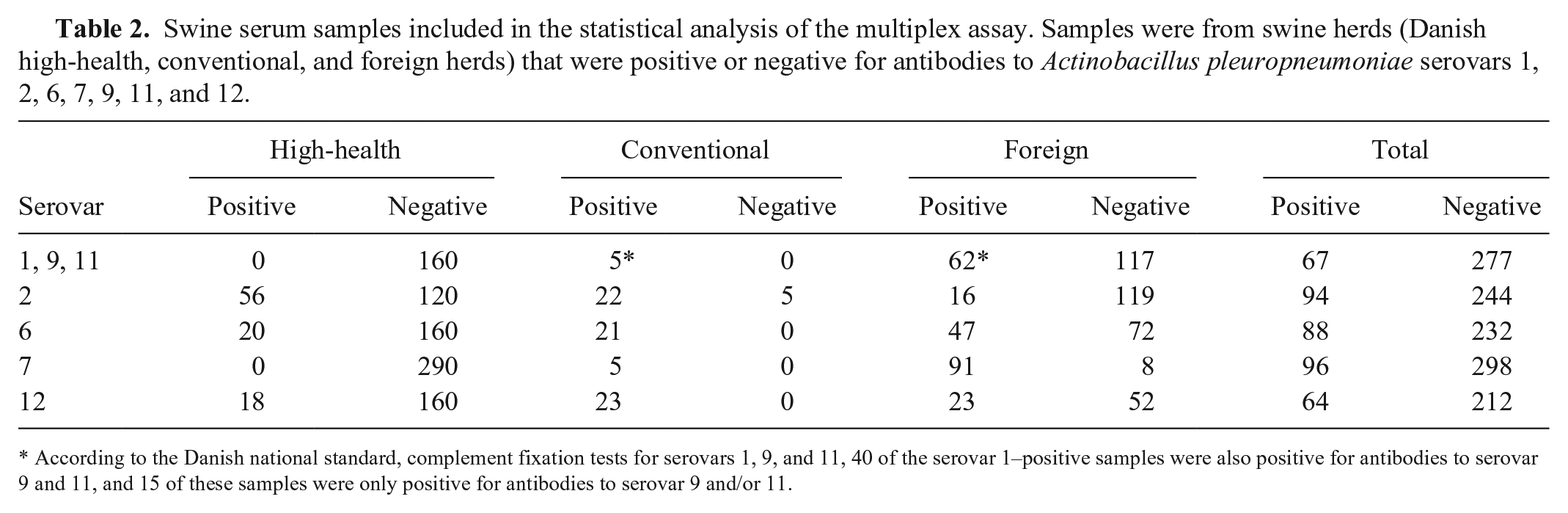
According to the Danish national standard, complement fixation tests for serovars 1, 9, and 11, 40 of the serovar 1–positive samples were also positive for antibodies to serovar 9 and 11, and 15 of these samples were only positive for antibodies to serovar 9 and/or 11.
Preparation of lipopolysaccharide and capsular polysaccharide
LPS from A. pleuropneumoniae serovars 1 (strain 4074), 2 (strain 1536), 6 (strain Femø), 7 (strain WF83), and 12 (strain 1096) was purified using methods described previously. 6 In brief, cultures were prepared by growth in a fermenter, and crude LPS was purified from aqueous suspensions of the bacterial cultures using repeated extractions with hot phenol. Aqueous and organic phases were separated by centrifugation followed by dialysis of the aqueous suspensions to remove residual phenol. The dialyzed aqueous phase was digested with proteinase K and then ultracentrifuged to obtain LPS as a gel. The crude LPS was dissolved in water and freeze-dried. The LPS preparations were then dissolved in a deoxycholate-containing buffer at a concentration of 30 mg/mL and, finally, subjected to gel filtration on an S100 Sephacryl column (GE Healthcare, Brondby, Denmark) using the same buffer. Fractions (5 mL) were collected and analyzed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Fractions from the gel filtration chromatography that contained material with molecular weights of >50 kDa as judged by silver-stained sodium dodecyl sulfate–polyacrylamide gel electrophoresis were pooled, dialyzed, and solubilized in high-quality water (MilliQ integral water purification system, Merck Millipore, Darmstadt, Germany).
Coupling of lipopolysaccharide and capsular polysaccharide to magnetic beads
LPS preparations from A. pleuropneumoniae serovars 1, 2, 6, 7, and 12 were coupled to magnetic beads (Bio-Plex Pro magnetic COOH beads, Luminex) using a method analogous to one described previously 7 ; a 3-step incubation protocol is used to ensure high levels of antigen saturation on the beads. Briefly, 1.25 × 107 beads were resuspended in 250 μL of 0.1 M 2-(N-morpholino)ethanesulfonic acid (MES buffer; pH 5.0; M 22933, Sigma-Aldrich, St. Louis, MO). Next, after vortexing (10 s) and sonication (20 s; Sonorex Digitec, Bandelin, Berlin, Germany), 750 μL of the LPS diluted in MES buffer and 25 μL of fresh N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide hydrochloride (EDC; 03449, Sigma-Aldrich) solution (50 mg/mL) were added. The suspension was vortexed and then incubated for 40 min in the dark at room temperature (RT) in a rotator (PTR-35, Grant Instruments, Cambridge, England). An additional 1,000 μL of LPS, together with 25 μL fresh EDC, was added and incubated with the beads for 40 min in the dark at RT in the rotator. The last incubation step was repeated once for a total of 3 incubations with LPS. A magnet (DynaMag-5, Thermo Fisher Scientific, Waltham, MA) for 5-mL tubes (Eppendorf Protein LoBind tube, Sigma-Aldrich) was used for washing the beads (3 times) with 3,000 μL of phosphate-buffered saline (PBS), 0.1% bovine serum albumin (BSA), 0.02% Tween 20, and 0.05% sodium azide (PBS-TBN; pH 7.4). The beads were stored in the dark at 2–8°C in 1 mL of PBS-TBN. The coupling procedure was optimized with regards to the amount of antigen and buffers and the incubation time as well as conditions providing the highest signal-to-noise ratio of median fluorescent intensities (MFIs) when testing the beads with serum from experimentally infected pigs.
Multiplex immunoassay
The 5 different A. pleuropneumoniae antigen–coated bead suspensions were vortexed (10 s) and diluted to a concentration of 8 × 104 beads/mL in a single volume of assay buffer A (PBS 0.05 M, 0.05% Tween, 1% BSA, 0.5 M NaCl). After vortexing (10 s) and sonication (20 s), 25 μL of bead suspension was added to black flat-bottom, 96-well plates (Bio-Plex Pro, Bio-Rad, Hercules, CA) together with 25 μL of pig serum diluted in assay buffer A (final serum dilution, 1:200). All incubations were performed at RT in the dark (covered with aluminum foil) on a rotating shaker (MTS 2/4 digital microtiter shaker, IKA-Werke, Staufen, Germany). The plates were incubated for 45 min and washed with wash buffer (PBS, 0.05% Tween) using an automated plate washer for magnetic beads (ELx405, BioTek, Winooski, VT). Next, biotin-conjugated rabbit anti-swine IgG (SAB3700429, Sigma-Aldrich) was diluted 1:1,500 in assay buffer B (PBS, 0.05% Tween, 1% BSA), and 25 μL was added per well and incubated with the beads for 30 min. The plates were washed in the automated plate washer and incubated for 30 min with 75 μL of streptavidin–PE (S21388, Thermo Fisher Scientific) diluted 1:700 in assay buffer B. The plates were shaken on the rotating shaker for 30 s, and samples were read in a flow cytometric platform dedicated to multiplex analysis (Bio-Plex 200 reader, Bio-Rad) that was adjusted to acquire a 50-μL sample and count a minimum of 50 beads/analyte. All samples were analyzed in duplicate, and data were acquired using the flow cytometric platform software (Bio-Plex Manager software v.6.1, Bio-Rad). Results for bound pig serum IgG were measured as MFIs of PE.
Assay optimization and investigation of reagent stability
Antigen-coupled beads were first evaluated separately with the A. pleuropneumoniae QC panel. Various assay parameters were tested in singleplex assays during optimization of the multiplex analyses, including bead, serum, and conjugate concentrations, temperature, reaction time, and buffer composition.
The repeatability of the assay was assessed by 10 repeated intra-plate duplicate analyses of 3 positive control sera that were positive for antibodies to serovars 1, 2, 6, 7, and 12 (sample 1: serovars 1 and 2 positive; sample 2: serovars 6 and 12 positive; and sample 3: serovar 7 positive). The same plate setup was used to determine reproducibility of the multiplex assay by testing the same samples over a period of 5 d. Both the repeatability and the reproducibility of the assay were defined as the mean coefficient of variation percentage (CV%). Furthermore, the A. pleuropneumoniae QC panel (Table 1), which included 2–5 positive samples per serovar, was tested monthly over a 3-mo period to evaluate the shelf life of coupled beads.
Data analysis
For each sample, the ratio between the sample MFI (S) and the positive sample MFI (P; S/P ratio) was calculated using the following formula:
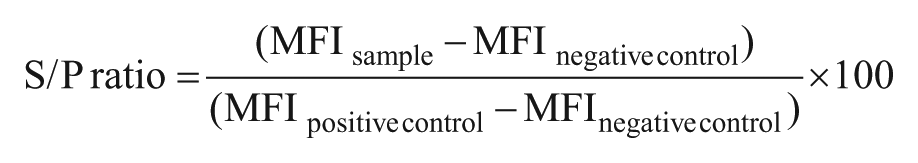
Receiver operating characteristic (ROC) curve analysis was used for the determination of test analysis quality compared to the existing in-house analyses, and differential positive rates (DPR) (defined as sensitivity + specificity – 1) were used together with dot plot correlation curves to determine cutoffs for the individual analytes in the multiplex analysis.3,15,16
Assay validation
When satisfactory assay conditions that could be applied to all analytes had been established, beads coupled with different antigens were mixed and tested in a 5-plex assay against field serum samples from both seropositive and seronegative pig herds.
The results of the multiplex subtests were compared with results obtained with existing routine analyses that have been used for >10 y as Danish national standards (DN standards) for herd classification in the Danish Specific Pathogen Free (SPF) system. These included CF tests for A. pleuropneumoniae serovars 1, 9, and 11 as described previously (Nielsen R. Haemophilus pleuropneumoniae infection in pigs. Thesis. Carl Fr. Mortensen A/S, Denmark, 1982), a blocking ELISA for A. pleuropneumoniae serovar 2, (Enøe 2001) 11 as well as in-house indirect ELISAs for A. pleuropneumoniae serovars 6 (unpublished), 7, 10 and 12 (unpublished). The DN standards, with the exception of the CF tests and the serovar 12 ELISA, have been accredited according to the international DS/EN ISO/IEC 17025 standard (https://goo.gl/OIh90u). The specificities and sensitivities of the DN standards are summarized in Table 3.
Specificity and sensitivity of the Danish national standard serologic assays for detection of antibodies to the indicated Actinobacillus pleuropneumoniae serovar.
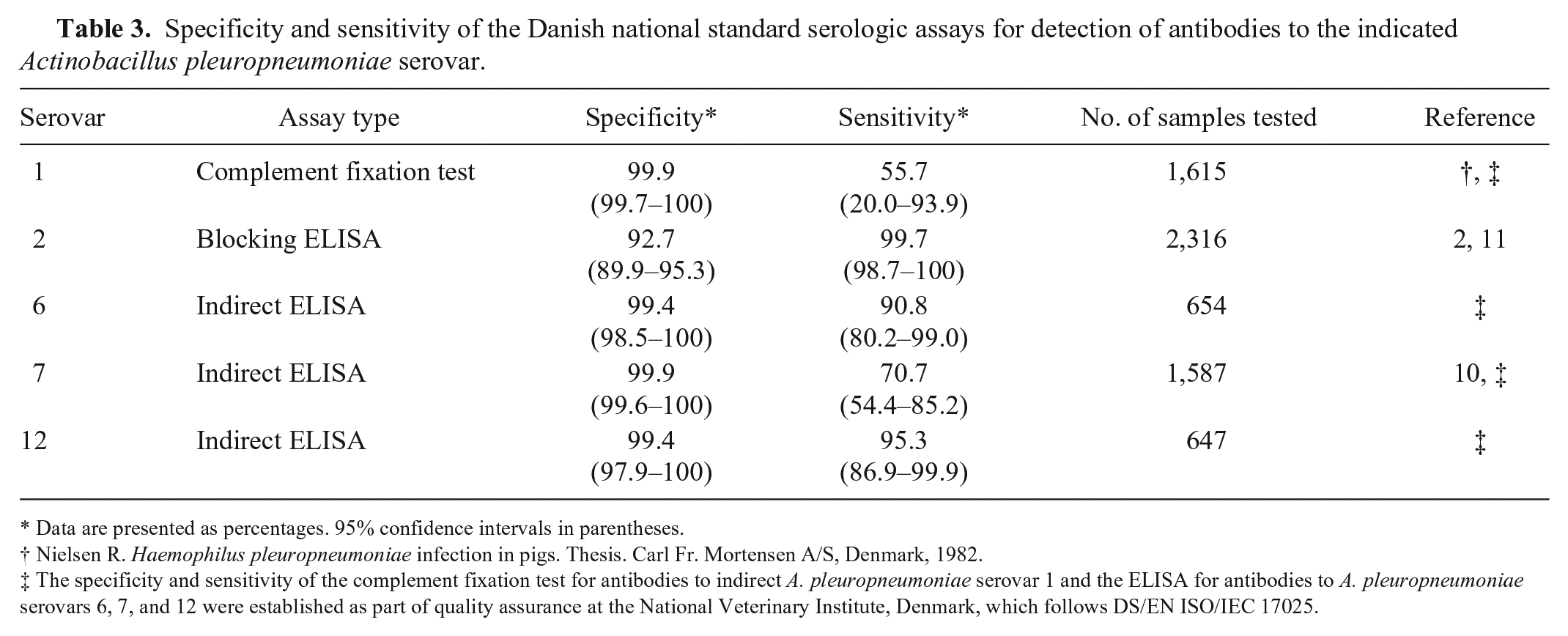
Data are presented as percentages. 95% confidence intervals in parentheses.
Nielsen R. Haemophilus pleuropneumoniae infection in pigs. Thesis. Carl Fr. Mortensen A/S, Denmark, 1982.
The specificity and sensitivity of the complement fixation test for antibodies to indirect A. pleuropneumoniae serovar 1 and the ELISA for antibodies to A. pleuropneumoniae serovars 6, 7, and 12 were established as part of quality assurance at the National Veterinary Institute, Denmark, which follows DS/EN ISO/IEC 17025.
Because of their identical LPS antigen structure, antibodies to LPS from A. pleuropneumoniae serovars 1, 9, and 11 will cross-react. 13 If sera only tested positive in the DN standard toward, for example, A. pleuropneumoniae serovars 9 and 11, these samples were regarded as positive for serovars 1, 9, and 11 in the statistical analysis. A commercial A. pleuropneumoniae serovars 1, 9, and 11 antibody ELISA kit (Swinecheck App 1, 9, 11, Biovet, Saint-Hyacinthe, Canada) was used to test 175 samples, including 36 that were positive and 105 that were negative to serovars 1, 9, or 11 in the DN standards as well as 34 samples that were negative in the DN standards but suspected as positive using multiplex analysis (S/P ratio ≥ 26).
Sera from high-health herds that are part of the Danish SPF system, as well as from conventional pig herds, were included in the study. Furthermore, to obtain samples positive to the A. pleuropneumoniae types that are not present in the Danish pig production, we included samples from other countries where pigs seropositive to the specific A. pleuropneumoniae types could be found. For validation of the test, we included 340 samples from 24 Danish high-health herds, 106 samples from 7 conventional Danish herds, as well as 398 samples from 30 foreign herds (from Bulgaria, The Netherlands, Norway, Poland, Russia, Spain, and Ukraine). All samples were tested for antibodies to the 5 A. pleuropneumoniae serovars using the multiplex assay, and to validate the results, a subset of these samples was tested in parallel in the ELISA and CF tests. Table 2 shows the number of A. pleuropneumoniae–positive and –negative sera from Danish high-health herds, conventional herds, and foreign herds that were included in the statistical analysis.
In the data analysis, if a herd was found A. pleuropneumoniae–positive in the DN standards, multiplex data were included for the positive sera; samples in the same herd that tested negative in the DN standards were excluded in order to minimize the influence of animals undergoing seroconversion that would give borderline reactions.
In a preliminary analysis of serum samples with the A. pleuropneumoniae multiplex assay, sera were identified that reacted positively in the multiplex assay but were negative in the in-house A. pleuropneumoniae serovars 1, 9, and 11 CF tests. To help define a cutoff, these samples were included in the final validation, and sample selection for serovars 1, 9, and 11 therefore presented a bias toward false-positive reactors.
Results
Assay optimization and investigation of reagent stability
Each antigen–antibody reaction was first tested separately with the A. pleuropneumoniae QC panel, and then optimized (based on the highest signal-to-noise ratios) in order to obtain the most optimal assay conditions for all analytes included. Antigen-specific reactivities with the QC panel were similar in the singleplex (separate A. pleuropneumoniae analyses) and multiplex (A. pleuropneumoniae serovars 1, 2, 6, 7, and 12) format. The multiplex assay could detect and distinguish between all of the serovar-specific antibodies included in the QC panel. Cross-reactivity of A. pleuropneumoniae serovars 6 and 8 serum antibodies, which is normally observed in ELISA given the identical LPS structures, 13 was also observed in the multiplex assay (Table 1).
Over a period of 3 mo, the coupled beads were tested with the A. pleuropneumoniae QC panel in order to determine the shelf-life of antigen-coupled beads. In general, antigen-coupled beads maintained stable interaction with serum antibodies during this period (Fig. 1). The CV% for the 4 average S/P ratios (day 1 to 3 mo) of the different serovars was 1.8–8.9%. Results obtained from repeated (10 times) intra-plate testing of duplicates of serum samples positive for each of the 5 A. pleuropneumoniae serovars showed an acceptable repeatability (intra-plate variation) with mean CV% for each serovar of 6.1–9.9%. By running the same samples on 5 separate days, the reproducibility (inter-plate variation) of the multiplex assay was also shown to be acceptable with a mean CV% of 2.4–11.5%.
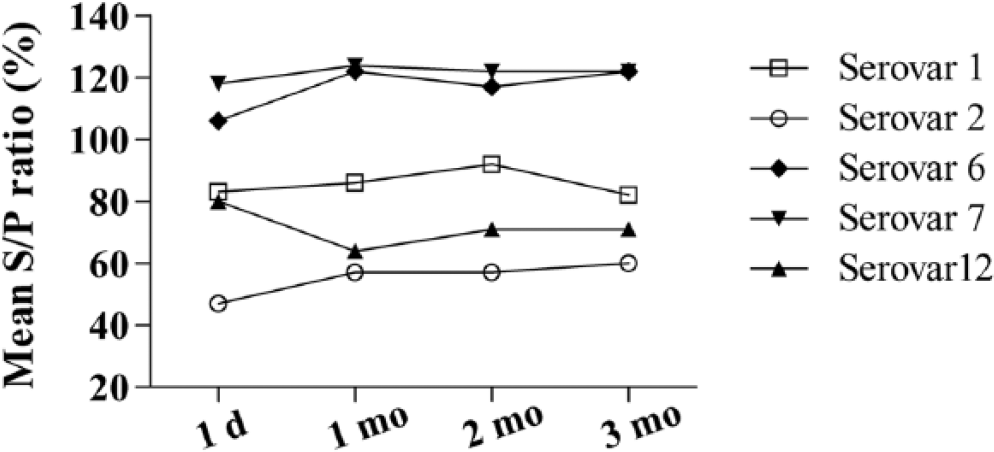
Time-dependent activity of Actinobacillus pleuropneumoniae antigen–coupled beads. Antigen-coupled beads were tested with the quality control (QC) panel after 1 d and then once a month over a 3-mo period. For each serovar, bead activity was calculated as the mean sample-to-positive (S/P) ratio of the positive control samples included in the QC panel.
Validation of the multiplex assay
The ROC curves showed that the results from the A. pleuropneumoniae serovars 1, 2, 6, 7, and 12 multiplex assay analysis of serologically positive and negative sera from non-infected and naturally infected herds were highly comparable to those of the DN standards because the area under the curve (AUC) for all analytes was ≥0.988 (Fig. 2).
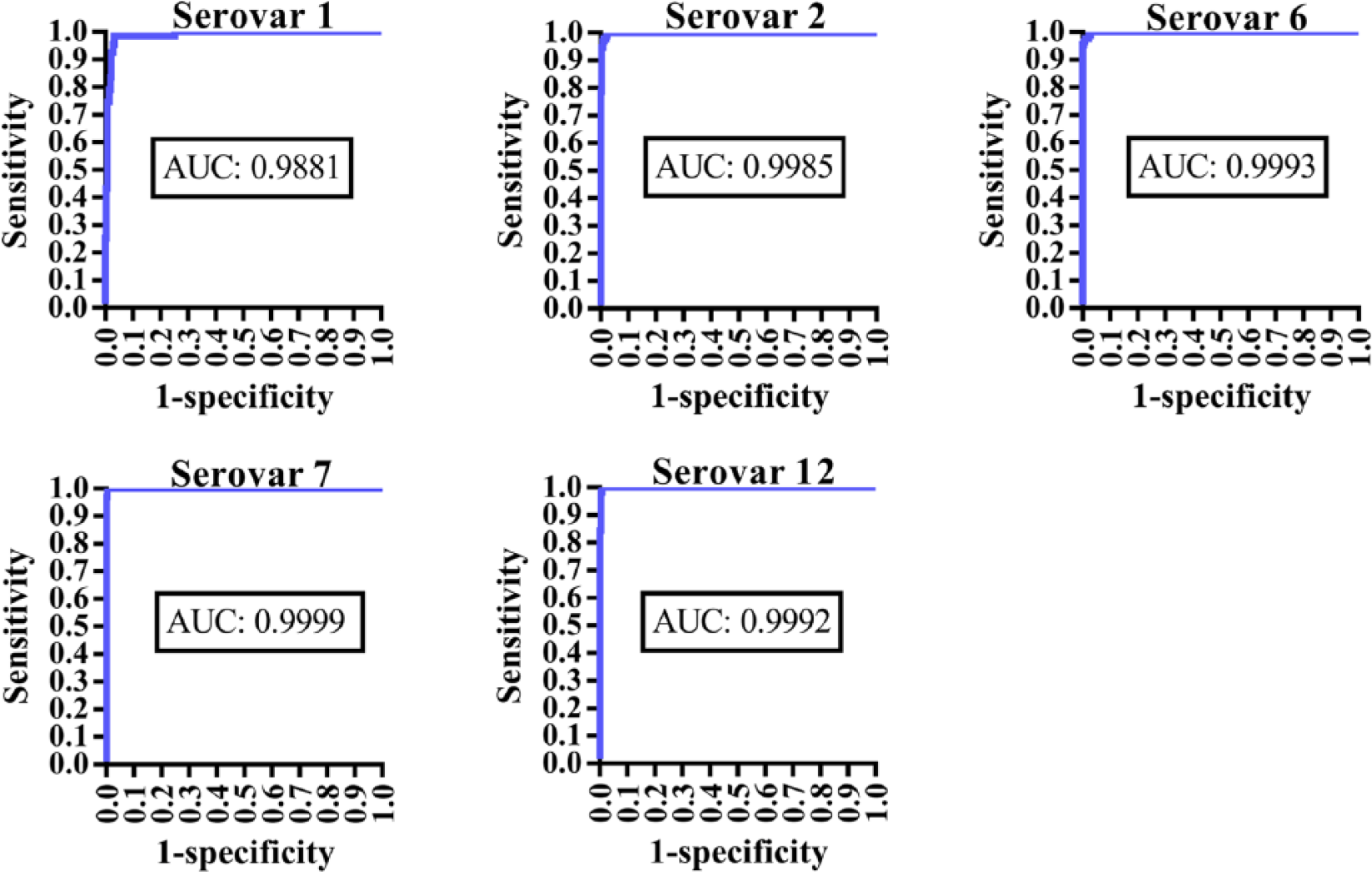
The receiver operating characteristic curves for Actinobacillus pleuropneumoniae serovar subtests included in the multiplex analysis. The results were compared to the Danish national standards, including complement fixation tests (serovars 1, 9, 11) and ELISAs (serovars 2, 6, 7, and 12). The area under the curve (AUC) is shown within each graph.
Cutoffs providing the highest DPRs are shown together with the optimal specificities and sensitivities in a dot plot (Fig. 3), which also illustrates the reactivity of antibodies from serum samples that were positive or negative for A. pleuropneumoniae serovars by ELISAs or CF tests. The statistically optimal cutoffs, sensitivities, and specificities as well as maximum DPRs are summarized in Table 4. The A. pleuropneumoniae multiplex assay generally seems to be more sensitive than the A. pleuropneumoniae serovars 1, 9, and 11 CF tests given that it detects the same or more positive samples within a seropositive herd compared to the CF test (Fig. 4A). Of the samples from Danish high-health herds, 2.5% had relatively high S/P ratios (S/P% ≥ 20) for serovar 1 in the multiplex assay. Because Danish high-health herds are very rarely infected with serovars 1, 9, or 11, we assume that the multiplex assay detects false-positive samples in these herds. To minimize the number of false-positive reactors, the cutoff value for serovars 1, 9, and 11 was increased from 26 to 35, and the specificity thereby became 100% for Danish high-health herds (Fig. 4B). Although sensitivity is decreased by increasing the cutoff to 35 (Fig. 4A), the sensitivity is still acceptable (92.5%) for detection of antibodies at herd level (Table 4). Some (5.1%) of the samples positive for serovars 1, 9, and 11 in the foreign herds that were negative in the CF tests will still be detected using the adjusted cutoff value (Fig. 4B).
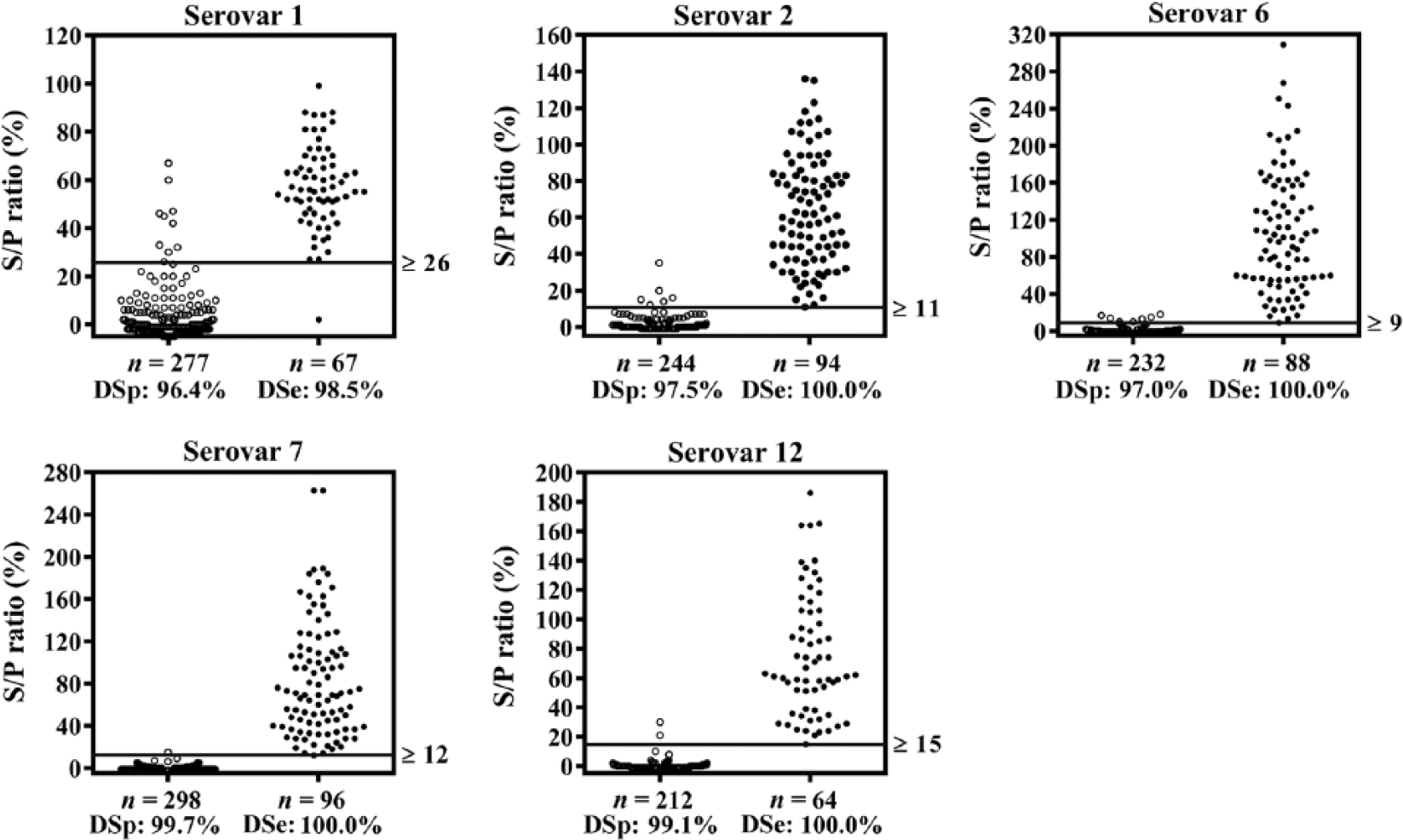
Dot plots showing the results of the Actinobacillus pleuropneumoniae multiplex assay validation. Reactivity of serovars 1, 2, 6, 7, and 12 coupled beads with antibodies in serum samples from ELISA or complement fixation test–negative pig herds (left) and –positive herds (right). The horizontal lines indicate the cutoff value determined at the maximum differential positive rates. The figure also shows results from the receiver operating characteristic curve analysis including diagnostic specificities (DSp) and sensitivities (DSe). S/P ratio = sample-to-positive ratio.
Diagnostic specificities and sensitivities as well as cutoffs and differential positive rate (DPR) of each serovar in the multiplex assay.*
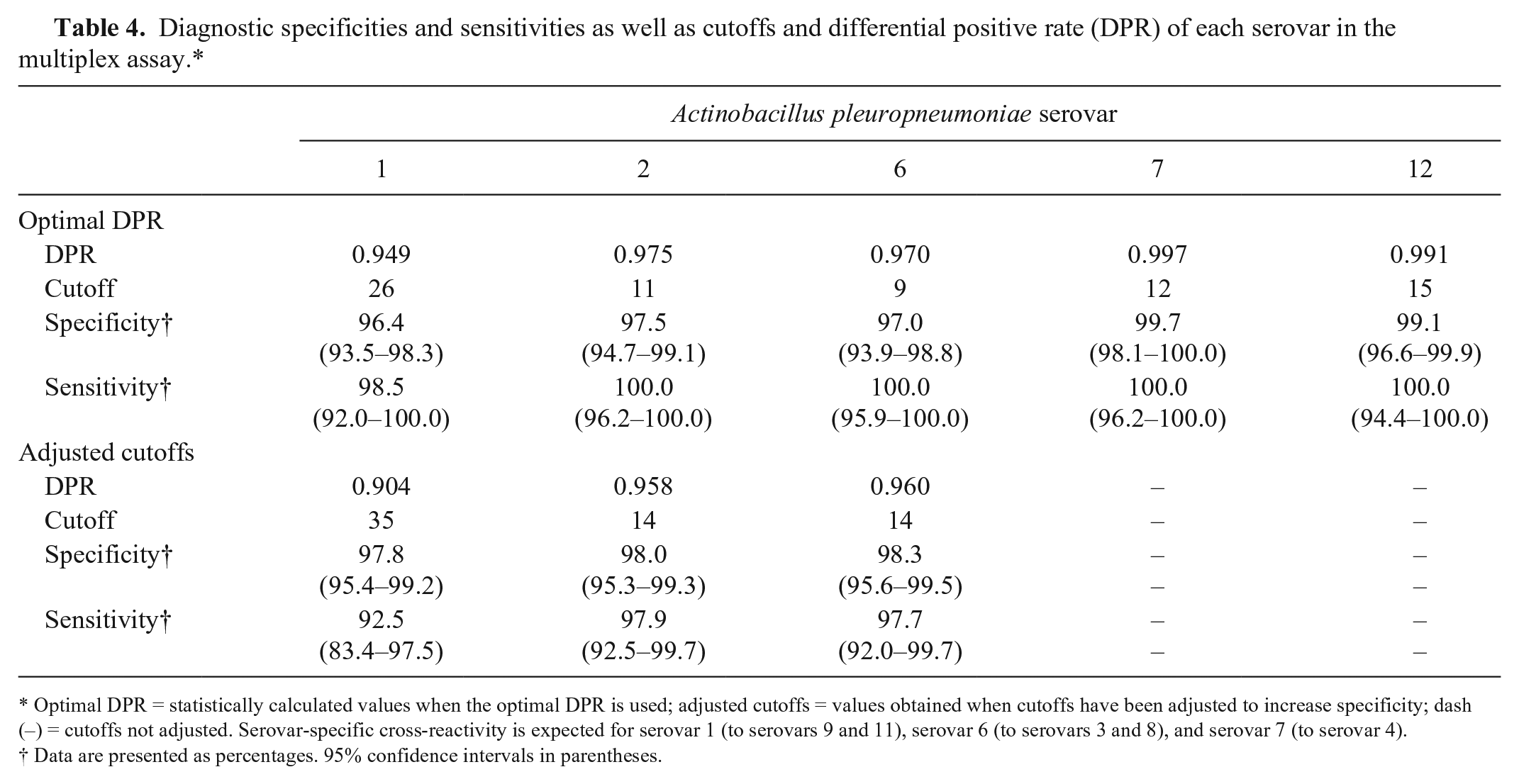
Optimal DPR = statistically calculated values when the optimal DPR is used; adjusted cutoffs = values obtained when cutoffs have been adjusted to increase specificity; dash (–) = cutoffs not adjusted. Serovar-specific cross-reactivity is expected for serovar 1 (to serovars 9 and 11), serovar 6 (to serovars 3 and 8), and serovar 7 (to serovar 4).
Data are presented as percentages. 95% confidence intervals in parentheses.
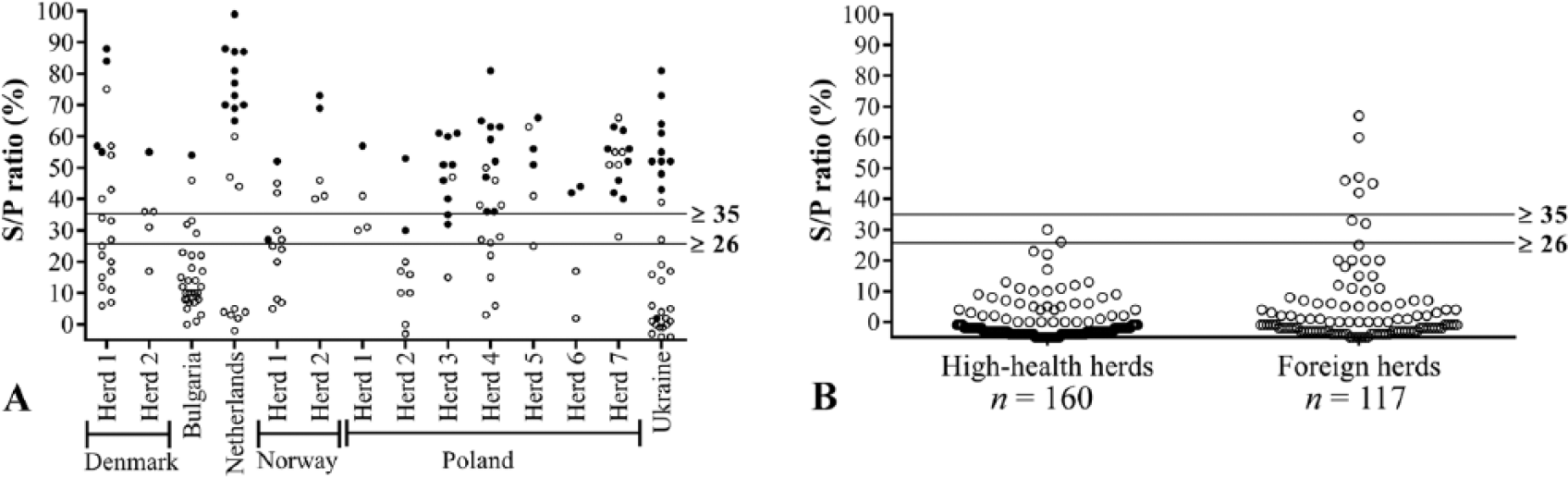
Dot plots showing multiplex analysis results for samples from herds that, according to the Danish national standards, were either positive or negative for Actinobacillus pleuropneumoniae serovars 1, 9, and 11.
A subset of 175 samples was tested with a commercial A. pleuropneumoniae serovar 1, 9, and 11 antibody ELISA, which confirmed the multiplex assay results. Compared to the in-house CF tests and similar to the multiplex assay (Fig. 4A, 4B), this A. pleuropneumoniae serovar 1, 9, and 11 ELISA detected more positive reactors within seropositive herds, as well as the assumed false-positive reactors in the Danish high-health herds and reactors suspected as positive within foreign herds.
Discussion
Serum samples from naturally infected and uninfected herds were used to validate the A. pleuropneumoniae multiplex assay, and the different multiplex subtests were shown to be excellent compared to the DN standards, with AUCs ranging from 0.988 to 1.000 (Fig. 2). Cutoff values for the different subtests were set according to the maximum DPR, but the cutoff for the A. pleuropneumoniae serovar 1 subtest was adjusted to increase specificity in Danish high-health herds to 100% thereby increasing overall specificity to 97.8%. Cutoffs were also adjusted for the serovar 2 and serovar 6 subtests in order to increase specificity to at least 98% (Table 4). This gives rise to decreased sensitivities. However, for Danish swine veterinarians and farmers, a high specificity is crucial given that a false suspicion of infection will force the farmer, unnecessarily, to stop selling pigs to other herds, until re-sampling and re-testing has provided data to reject the suspicion. The sample size from a herd is typically 10–20 samples, and given that the sensitivity of the multiplex assay is acceptable even after increasing the specificity, A. pleuropneumoniae antibodies will still be detected at the herd level. Even higher sensitivities can be achieved by increasing the number of samples tested and/or increasing the frequency of sampling within herds.
The multiplex assay exhibited good repeatability and reproducibility with low intra-assay and inter-assay variation. Fluctuations in bead activity (A. pleuropneumoniae serovar 12 beads: 11.5% reduction; A. pleuropneumoniae serovar 2 beads: 20.8% increase) were observed during 3 mo of storage (Fig. 1), but the average CV% of the individual tests run over 3 mo was low (1.8–8.9%). The fluctuations in bead activity probably represent day-to-day variation, and similar fluctuations in S/P ratios are observed in the in-house ELISAs. Because MFIs of both negative and positive samples would be affected by fluctuations in bead activity, the significance of such fluctuations is probably minor as long as activity remains relatively stable.
The A. pleuropneumoniae serovar 1 multiplex subtest tends to be more sensitive than the A. pleuropneumoniae serovars 1, 9, and 11 CF tests. This is in line with previous observations in which sensitivity was increased when transferring an A. pleuropneumoniae serovar 7 CF test to the ELISA platform. 12 Results similar to those of the multiplex test were found using a commercial A. pleuropneumoniae serovar 1, 9, and 11 ELISA.
Sample selection for the validation of the A. pleuropneumoniae serovar 1 multiplex subtest presented a bias toward false-positive samples. We expect to observe higher specificities and sensitivities for the A. pleuropneumoniae serovar 1 multiplex subtest in an unbiased population of samples because of a lower rate of false-positive results. The A. pleuropneumoniae multiplex assay is currently being further validated with an unbiased population of serum samples that will also be analyzed using the in-house ELISAs and CF tests.
The A. pleuropneumoniae multiplex assay should detect antibodies cross-reactive to serovars 3 and 8, to serovar 4, and to serovars 9 and 11 because of LPS structures identical to some of the serovars included in the multiplex assay (i.e., LPS from serovars 6, 7, and 1, respectively). 13 Unfortunately, the samples used for validating the assay only included a few samples that were positive for antibodies to these serovars, given that these are rarely detected in Danish herds. Of the 67 sera included in the validation that were positive for serovars 1, 9, and 11, only 15 were seropositive for serovar 9 and/or 11 alone (Table 2). Therefore, more samples must be analyzed to study to what extent antibodies to serovars 3, 4, 8, 9, and 11 are detected in the A. pleuropneumoniae multiplex assay.
Simultaneous detection of antibodies to multiple analytes within a serum sample reduces the required amount of serum sample, as well as the amount of time and labor needed to test the samples. This will lower the costs of analyses for serologic testing and surveillance as well as shorten the response time, which in turn can result in more effective serologic surveillance and detection of disease outbreaks in pig production.
Footnotes
Acknowledgements
We thank the laboratory staff at the Section for Diagnostics and Scientific Advice, National Veterinary Institute, Technical University of Denmark. Especially, we thank Natascha Morton, Henriette Vorsholt, and Jonathan Rogersen (laboratory technicians) for excellent technical assistance. We also thank Professor Nils Toft for his contribution to the statistical analysis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
