Abstract
Escherichia coli O157:H7 (E. coli O157:H7) is a dangerous foodborne pathogen, mainly found in beef, milk, fruits, and their products, causing harm to human health or even death. Therefore, the detection of E. coli O157:H7 in food is particularly important. In this paper, we report a lateral flow immunoassay strip (LFIS) based on aggregation-induced emission (AIE) material labeling antigen as a fluorescent probe for the rapid detection of E. coli O157:H7. The detection sensitivity of the strip is 105 CFU/mL, which is 10 times higher than that of the colloidal gold test strip. This method has good specificity and stability and can be used to detect about 250 CFU of E. coli O157:H7 successfully in 25 g or 25 mL of beef, jelly, and milk. AIE-LFIS might be valuable in monitoring food pathogens for rapid detection.
Introduction
Escherichia coli O157:H7 is a notorious foodborne pathogen. It is spread by eating contaminated foods such as beef, milk, fruits, and their products. In people with low immunity to an infectious dose as little as 10 live cells, it may still cause hemorrhagic colitis, thrombotic thrombocytopenic purpura, and hemolytic uremic syndrome.1–3
Since it was first isolated and confirmed to be a serotype of enterohemorrhagic E. coli, there have been many outbreaks of E. coli O157:H7 infection in China, Canada, Japan, and other countries. Every year, E. coli O157:H7 causes more than 2 million cases of the disease worldwide. This is a serious food safety and public health problem. Therefore, the development of a rapid and accurate E. coli O157:H7 detection method is of great significance for the prevention of foodborne diseases. 4
In recent years, researchers have developed various techniques to detect E. coli O157:H7, including traditional microbial culture methods, molecular biology methods, and immunological methods. The traditional culture methods are considered to be the “gold standard” for detecting bacteria. This method has extremely high sensitivity according to a specific cultivation protocol. However, this method has a long detection cycle, high labor intensity, and lagging results. It is not suitable for the detection of a large number of samples and cannot monitor the epidemic in real time. 5 Methods based on molecular biology include polymerase chain reaction (PCR), 6 real-time PCR, 7 multiplex PCR, 8 loop-mediated isothermal amplification (LAMP), 9 rolling circle amplification (RCA), 10 and recombinase polymerase amplification (RPA). 11 Although molecular biology methods have high sensitivity, they require specialized equipment and skilled technicians to carry out complex operation procedures.
Immunological detection technologies included the enzyme-linked immunosorbent assay (ELISA), chemiluminescent immunoassay,12,13 electrochemical immunosensor,14,15 and lateral flow immunoassay strip (LFIS). LFIS is an accurate, cheap, and portable on-site testing technology that achieves reliable detection within minutes and is widely used for the rapid detection of foodborne pathogenic microorganisms in food production, processing, sales, and supervision. At present, most research on LFIS is focused on the development of new nanomaterials, such as flower-shaped gold nanoparticles,16,17 silver nanoparticles, 18 carbon nanoparticles, 19 paramagnetic nanoparticles, 20 quantum dots,21,22 and conventional luminogens. 23 These materials are coupled with antibodies as fluorescent probes to capture antigens and output detection signals. However, most of them require instruments for reading specialized signals, which limits the commercial application of LFIS in grassroot fields. Therefore, the development of a new LFIS without antibody labeling may bring about breakthroughs in the rapid detection of E. coli O157:H7 in food.
Aggregation-induced emission (AIE) materials refer to a class of materials in which the fluorescence is weak or even not observed in the single-molecule state, and the fluorescence is significantly enhanced in the aggregate state. Because AIE has unique superiority to the fluorescence quenching caused by the aggregation of traditional fluorescent chromophores, many research groups have applied it in the fields of fluorescence strips, bioimaging, and organic light-emitting diodes. 1,1,2,2-Tetra(4-carboxylbiphenyl)ethylene (TCBPE) is a kind of fluorescent material with AIE characteristics.
In this work, we first prepared fluorescent bacteria probes by labeling E. coli O157:H7 with carboxyl functional groups of TCBPE, instead of the conjugating antibody with nanomaterials. Then, a new LFIS based on AIE material labeling antigen (AIE-LFIS) was successfully developed. AIE-LFIS could directly detect E. coli O157:H7 in food with only one monoclonal antibody (McAb), which significantly reduced the cost of detection. Furthermore, we evaluated the sensitivity, stability, and specificity of AIE-LFIS and also verified the feasibility of AIE-LFIS for detecting E. coli O157:H7.
Materials and Methods
Materials and Equipment
The McAb against E. coli O157:H7 was supplied by Shanghai Prajna Biotech Co. Ltd. (Shanghai, China). 1,1,2,2-Tetra(4-carboxylbiphenyl) ethylene (TCBPE) was purchased from Alfa Chemical (Henan, China). Modified EC Novobiocin Broth Base was purchased from Land Bridge (Beijing, China). Dimethyl sulfoxide (DMSO), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), and N-hydroxysuccinimide (NHS) were purchased from Sigma-Aldrich (Darmstadt, Germany). All the solution used in this study was freshly prepared with ultrapure water (>18 MΩ).
The sample pad, absorption pad, and polyvinyl chloride (PVC) backing card were purchased from Kinbio Technology (Shanghai, China). The nitrocellulose (NC) membrane was purchased from Sartorius. The XYZ dispenser AD6010 was purchased from BIO-DOT (California, USA). The fluorescence microscope DM2500 was purchased from Leica (Wetzlar, Germany).
Preparation of AIE-LFIS
AIE-LFIS consists of one conjugate pad, one absorption pad, and one PVC backing card, and two pieces of NC membranes and sample pads. The structure of AIE-LFIS is shown in Figure 1 . The McAb against E. coli O157:H7 (1 mg/mL) was spotted on the NC membrane (2 × 30 cm) with 1 μL/cm as the test line (T line) and control line (C line). After drying at 37 °C for 1 h, the NC membrane was sealed and stored at 4 °C. The NC membranes, sample pads (2 × 30 cm), and absorption pad (1.8 cm×30 cm) were assembled by overlapping 1–2 mm on the PVC backing card (10 × 30 cm). The assembled semifinished product was cut into 3 mm wide strips; then the strips were collected in a silica sealed box and stored at 4 °C.
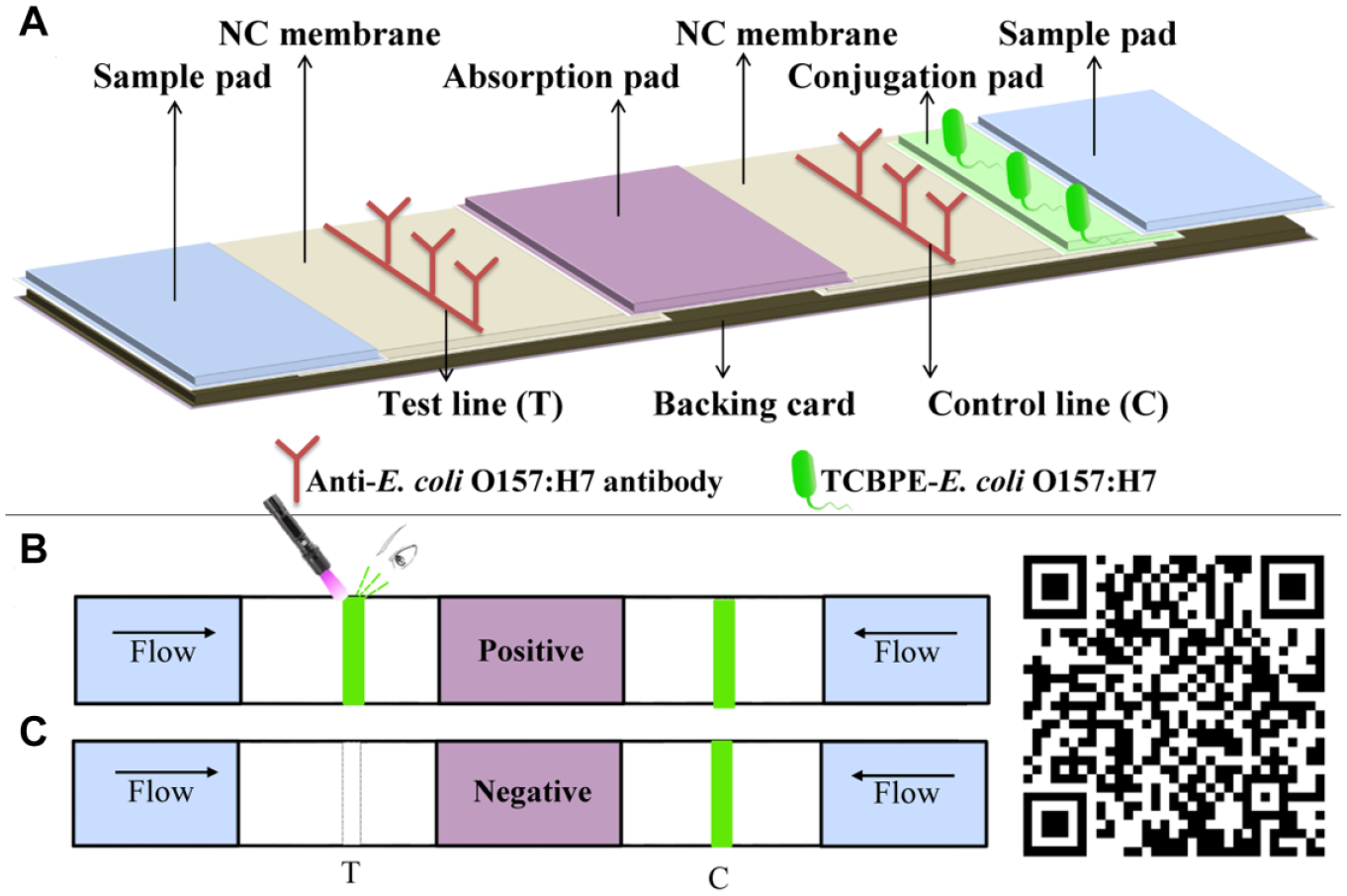
Schematic diagram of the principle for the detection of E. coli O157:H7 by AIE-LFIS.
Carboxyl Activation and Optimum Concentration of TCBPE
TCBPE (1.5 mL; 0.1 mol/L in DMSO) was added to 30-mL boric acid buffer (0.2 mol/L, pH 8.0) with 3-mL EDC solution (10 mg/mL) and 2.25-mL NHS (10 mg/mL) in the dark with stirring for 2 h to activate the carboxyl group. Following activation, the solution was centrifuged (12,000 rpm/min) for 20 min and the precipitate was dissolved in 1 mL of DMSO.
A single colony of E. coli O157:H7 (NCTC 12900) and different concentrations of activated TCBPE solution (0.01%, 0.05%, 0.1%, 0.5%, 1%, and 5%, v/v) were added to nutrient broth (NB) medium to culture at 37 °C for 12 h. During the incubation period, the optical density (OD) at 600 nm was measured every 30 min by a microplate reader. The sterile NB medium containing an equal volume of TCBPE solution without E. coli O157:H7 was used as a negative control. After incubation, the cultures were detected with simple AIE-LFIS (schematic diagram; see Suppl. Fig. S1 ).
Analytical Procedure
One hundred microliters of the sample solution was slowly dropped onto the sample pads at both ends of AIE-LFIS to detect E. coli O157:H7. After 10 min, the results of AIE-LFIS were directly determined by naked eye with a fluorescent flashlight and photographed using a digital camera with a 525 nm filter (FF01-525/15-25, Semrock, Rochester, NY). The signal intensity of the T line of AIE-LFIS was analyzed by the Plot Profile function of ImageJ software (National Institutes of Health, Bethesda, MD) on the green layer of the images.
Sensitivity, Specificity, and Stability of AIE-LFIS
To evaluate the sensitivity of the strip, the culture solution was diluted with sterile phosphate buffer to adjust to different concentrations of E. coli O157:H7 by the method of plate counting. The sensitivity measurement of AIE-LFIS was carried out by pipetting 103 to 109 CFU/mL concentrations of E. coli O157:H7 solution onto the sample pad of AIE-LFIS. The NB medium with TCBPE served as the negative control. Twenty-six foodborne pathogens ( Table 1 ) were incubated in their suitable medium with an optimum concentration of TCBPE and then tested by AIE-LFIS to evaluate the specificity. The stability of the AIE-LFIS stored for 3 months at room temperature under dry conditions was assessed by sensitivity evaluation. These assays were repeated at least three times.
Specificity of AIE-LFIS to the Strains Used in the Study.

, positive result; −, negative result.
Detection of Simulated Food Samples
To verify the feasibility of AIE-LFIS in the actual detection of food samples, artificially contaminated samples were used to simulate the AIE-LFIS for detecting E. coli O157:H7 in food. The E. coli O157:H7-free jelly, beef, and milk samples were purchased from a supermarket, and these food samples (25 mL or 25 g) were added into 225-mL NB medium, respectively. About 250 CFU of E. coli O157:H7 confirmed by plate count was added to the broth, along with 250-μL TCBPE solution. Subsequently, E. coli O157:H7 in these samples was propagated in a 37 °C incubator for 12 h. During the incubation period, 2 mL of culture solution was collected each hour, and 0.3% (v/v) formaldehyde was added to the collection at 4 °C to prevent E. coli O157:H7 growth. Finally, the cultured samples were centrifuged at 6000 rpm for 10 min to remove the food matrix and resuspended to the original volume with phosphate buffer and tested by AIE-LFIS.
Results and Discussion
Principle of AIE-LFIS
The principle of AIE-LFIS is based on the NC membrane as a solid-phase carrier, TCBPE-labeled bacteria as a fluorescent probe, and the specific binding of an antigen and an antibody to achieve detection. When the sample solution is dropped on the sample pads at both ends of the test strip, the fluorescent E. coli O157:H7 on the conjugate pad is transferred to the NC membrane by the sample solution. The anti-E. coli O157:H7 antibody on the C line captures E. coli O157:H7, and a fluorescent band appears on the C line. At the other end of the test strip, the labeled E. coli O157:H7 in the sample solution is captured by the antibody on the T line during the migration process on the NC membrane, leaving a green band in the T line area, indicating that the result was positive. ( Fig. 1B ). If there is no fluorescent band in the T line area of AIE-LFIS, this means the sample does not contain E. coli O157:H7 ( Fig. 1C ). The most significant difference between AIE-LFIS and traditional LFIS is that there is no detection antibody labeled with nanomaterials (such as colloidal gold or quantum dots) on the conjugate pad, which greatly reduces the difficulty and cost of LFIS development. After scanning the QR code with a mobile phone, animation of the detection process is demonstrated on the phone screen.
Optimal Concentration of the TCBPE for AIE-LIFS
E. coli O157:H7 was cultured in NB medium with different concentrations of TCBPE to form the fluorescent bacteria probe with varying intensities of fluorescence, and then the probe was detected by AIE-LFIS. The concentration of TCBPE solution may influence the signal intensity of the T line and background of AIE-LFIS. It can be seen from Figure 2A that as the concentration of TCBPE increases from 0.01% to 5%, the T line gradually becomes brighter. However, when the concentration is higher than 0.1%, the increased background signal of the strip affects judgment of the result. There is no fluorescent band at the T line of the negative control group ( Fig. 2B ). Therefore, the preferable concentration of TCBPE is 0.1%, with the strongest T line signal and the lowest background signal. It can be seen from Figure 2C that the largest difference in signal intensities between the positive and negative results is with the optimal concentration. At the same time, an OD of 600 nm of the bacterial culture solution shows that a 0.1% concentration of TCBPE solution has no effect on the growth of E. coli O157:H7 ( Suppl. Fig. S2 ). The morphology of E. coli O157:H7 labeled with the optimal concentration of TCBPE solution was photographed by fluorescence microscopy ( Fig. 2D ).
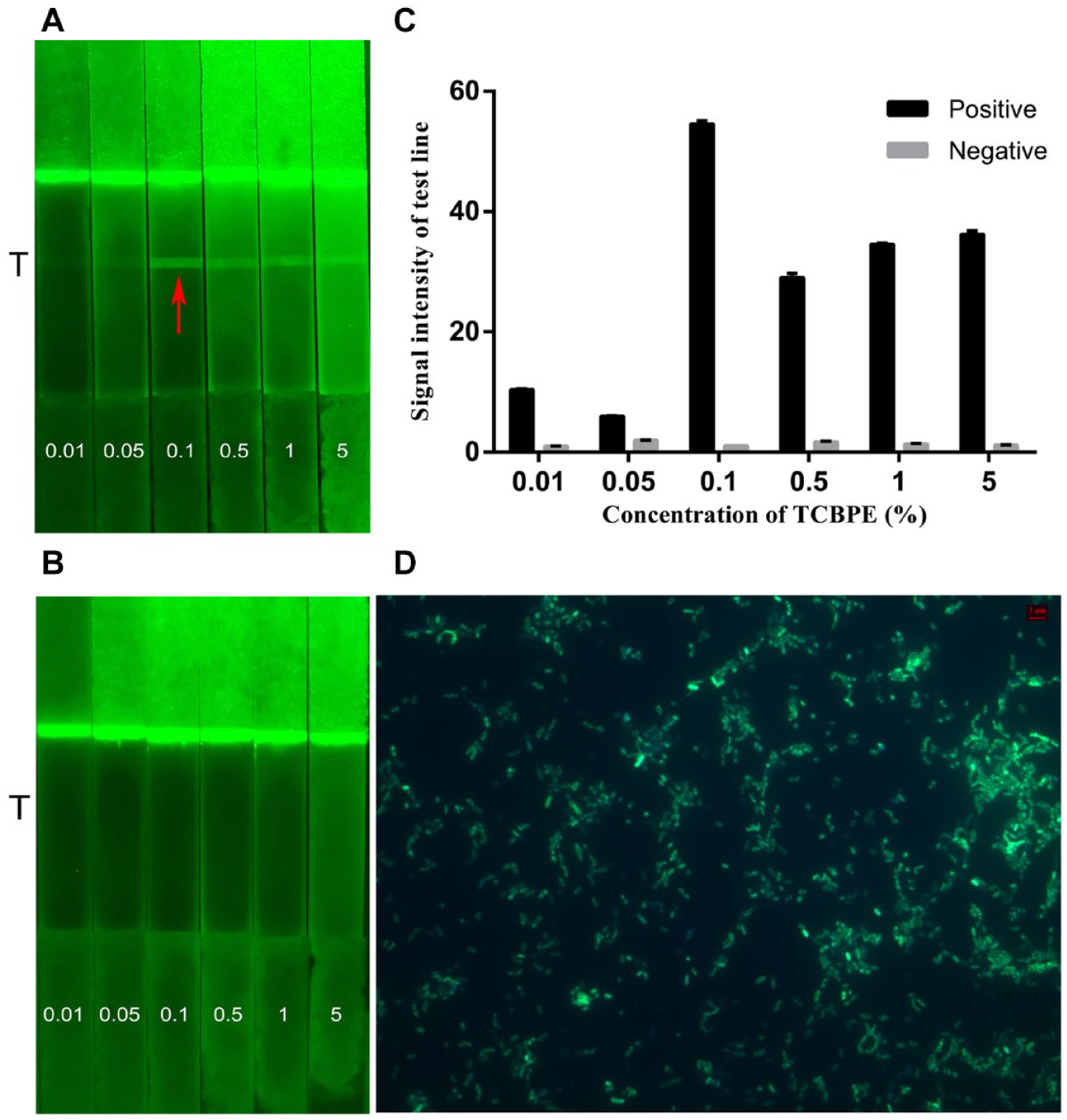
Results of the optimal concentration of TCBPE for simple AIE-LIFS. Images of (
Sensitivity and Stability of AIE-LFIS
To confirm the sensitivity of AIE-LFIS, 10-fold gradient dilution of E. coli O157:H7 solution was tested by AIE-LFIS. As shown in Figure 3A , when the concentrations ranged from 105 to 109 CFU/mL, the green fluorescent band on the T line was more evident under ultraviolet light. However, when E. coli O157:H7 with a concentration lower than 104 CFU/mL was detected, there was no fluorescent band in the T line area. Therefore, the experiment indicated that AIE-LFIS detection sensitivity was considered to be 105 CFU/mL. The signal of the T line area appears on the test strip that detected a 105 CFU/mL concentration of E. coli O157:H7 and increases with an increase in E. coli O157:H7 concentration, which is consistent with the detection result of direct observation ( Fig. 3B ). The detection method of labeling antigen with TCBPE is 10 times more sensitive than fluorescein isothiocyanate (FITC) as a marker. 24 The reason is that the fluorescence intensity of E. coli O157:H7 solution labeled with TCBPE at the same concentration is stronger than that labeled with FITC (microscope pictures and fluorescence intensity values are shown in Suppl. Fig. S3 ). Zhang et al. 25 prepared solid fluorescent microspheres based on TCBPE, poly(maleic anhydride-alt-1-octadecene), poly(methyl methacrylate), and sodium dodecyl sulfate as loading fluorescent dye, functional linker, polymer framework, and surfactant, respectively, for detecting of E. coli O157:H7. The sensitivity of this method was 3.98 × 103 CFU/mL. Although the method was 100 times more sensitive than AIE-LFIS, the process of synthesizing nanomaterials is cumbersome and not suitable for the commercial production of LFIS.
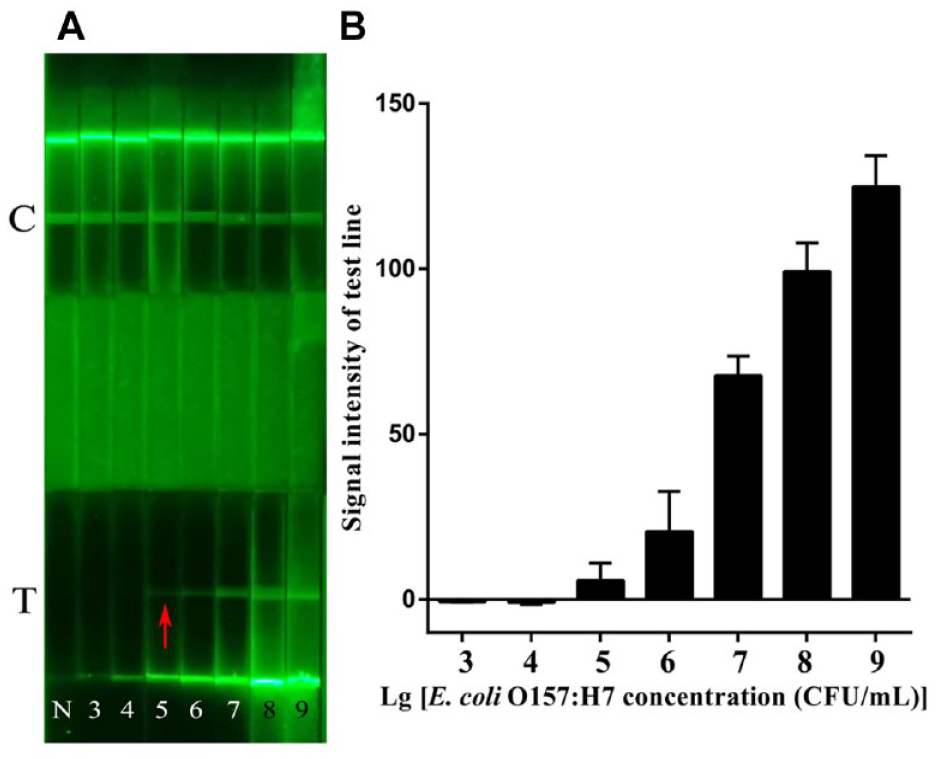
(
To intuitively reflect the change in sensitivity of AIE-LFIS, we developed a colloidal gold LFIS (GLFIS) using the same antibody. The sensitivity of the GLFIS detecting E. coli O157:H7 was 106 CFU/mL ( Suppl. Fig. S4 ). The detection sensitivity of AIE-LFIS was 10 times higher than that of GLFIS.
After storing AIE-LFISs at room temperature and dry conditions for 3 months, the stability was evaluated by confirming the sensitivity of AIE-LFIS. As can be seen from
Specificity of AIE-LFIS
As can be seen from Figure 4 , only E. coli O157:H7 showed green fluorescent bands in the T line area of AIE-LFIS, and the signal intensity of the T line was very high. However, when AIE-LFIS tested the other 23 bacteria in Table 1 , there was no fluorescent line at the test position, and they showed extremely low signal values. According to the test results, this indicated that the test strips had high specificity for the detection of E. coli without cross-reaction.
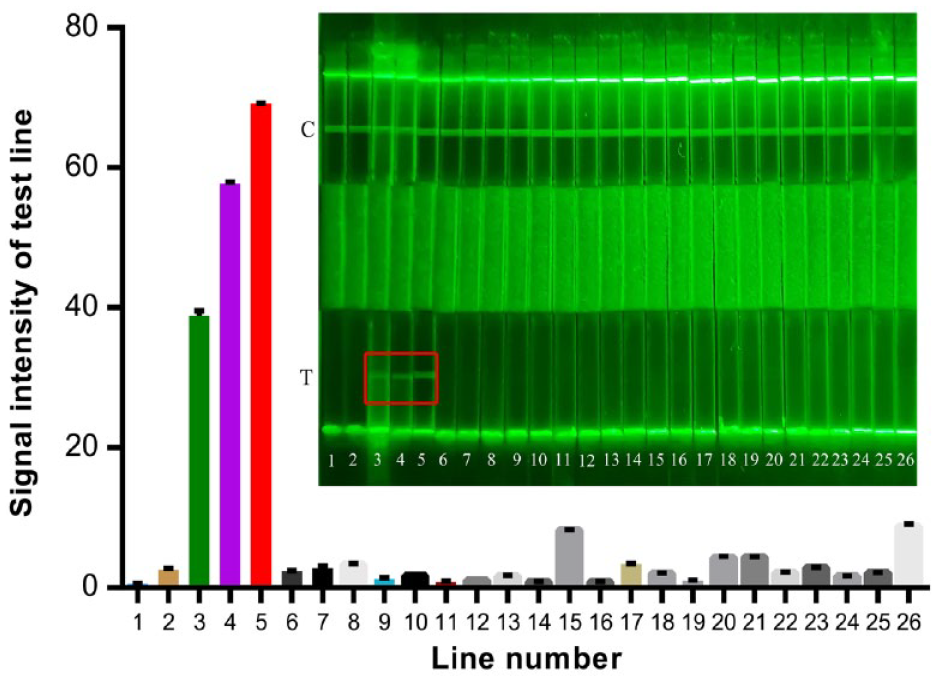
Specificity of AIE-LFIS; 1–26 correspond to the standard strains listed in Table 1.
AIE-LFIS for Detection of Artificially Contaminated Samples
To verify the feasibility and applicability of AIE-LFIS, milk, beef, and jelly food samples with artificial contamination during different cultivation periods were tested by AIE-LFIS. However, the complex and diverse food matrix was one of the main factors affecting the detection performance.26,27 AIE-LFIS could detect E. coli O157:H7 incubated 8 h in the untreated milk and jelly samples, but could not confirm the positive results of E. coli O157:H7 in the beef samples at any period ( Suppl. Fig. S6 ). The reason for this false-negative result may be that the complex food matrix in the sample affected the test process. The sample culture solution was centrifuged to remove the supernatant, and the precipitate was resuspended in phosphate buffer to the original volume and then detected by simple AIE-LFIS. The centrifugation could eliminate false-negative results caused by the food matrix ( Suppl. Fig. S7 ).
As shown in Figure 5 , the jelly, milk, and beef samples containing about 250 CFU of E. coli O157:H7 were tested with positive results on AIE-LFIS after culturing for 5, 6, and 5 h, respectively. The detection time of the jelly and milk samples removed from the matrix was 3 and 2 h earlier than the detection time of the untreated samples. More importantly, the removal of the food matrix from the beef sample eliminated the problem of false negatives, and the time to detect a positive result is consistent with 5 h of the jelly sample. We tested the sample cultures by ELISA to verify the accuracy of AIE-LFIS. The results showed that AIE-LFIS shortened the food sample detection time of E. coli O157:H7 by at least 3 h compared with the ELISA method ( Suppl. Table S1 ). It showed that the method could accurately and rapidly detect 250 CFU/25 g E. coli O157:H7 in jelly, milk, and beef samples.
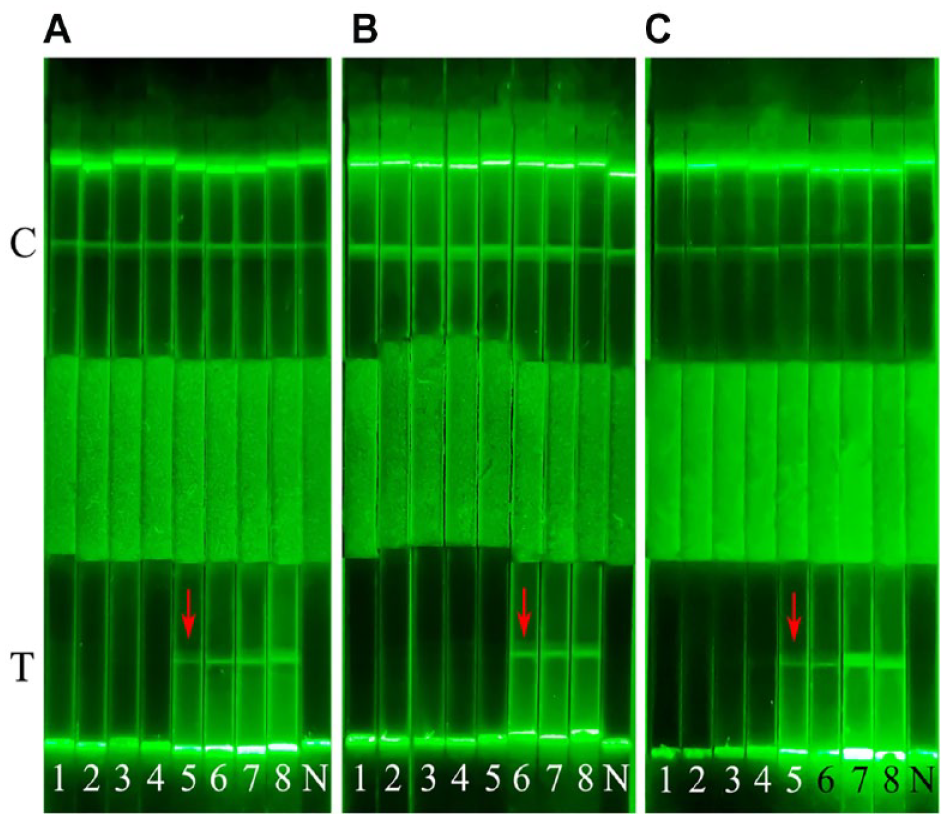
Results of AIE-LFIS for the detection of artificially contaminated samples: (
Conclusions
In this study, we developed a novel, simple, LFIS based on AIE material labeling antigens for the detection of E. coli O157:H7 in food. E. coli O157:H7 was labeled during the proliferation process to form fluorescent probes by TCBPE with AIE characteristics. The fluorescent probes were captured by the McAb on the detection line so that the bacteria in the sample gather on the T line and a fluorescent signal appears under the fluorescent flashlight. The detection sensitivity of AIE-LFIS (105 CFU/mL) was 10 times higher than that of the colloidal gold test strip. The test strip for detecting E. coli O157:H7 had good specificity. After precultivation of 250 CFU of E. coli O157:H7 in 25 g/25 mL jelly, milk, and beef samples, AIE-LFIS based on TCBPE-labeled antigen could successfully detect positive results and provided technical support for the rapid detection of E. coli O157:H7 in food.
Since AIE-LFIS requires neither nanomaterials nor sandwich-based antibodies, the development difficulty and cost of AIE-LFIS are low, and it may be easier to commercialize AIE-LFIS. Moreover, this method can expand the detection of other pathogens by simply replacing antibodies. Our research group is verifying the feasibility of this method for detecting other foodborne pathogens. AIE-LFIS may provide a new idea for the development of an LFIS for detecting foodborne pathogens.
Supplemental Material
sj-pdf-1-jla-10.1177_2472630320981935 – Supplemental material for An Aggregation-Induced Emission Material Labeling Antigen-Based Lateral Flow Immunoassay Strip for Rapid Detection of Escherichia coli O157:H7
Supplemental material, sj-pdf-1-jla-10.1177_2472630320981935 for An Aggregation-Induced Emission Material Labeling Antigen-Based Lateral Flow Immunoassay Strip for Rapid Detection of Escherichia coli O157:H7 by Cheng Liu, Shuiqin Fang, Yachen Tian, Youxue Wu, Meijiao Wu, Zheng Wang, Dongpo Xu, Dongjun Hou and Qing Liu in SLAS Technology
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 31871897) and the Science and Technology Innovation Plan of Shanghai (grant no. 19391902000).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
