Abstract
In this study, an immunochromatographic strip (ICS) was developed for the detection of antibody against Streptococcus suis serotype 2 (SS2). Colloidal gold particles labeled with staphylococcal protein A (SPA), which can bind to the FC fragment of mammalian immunoglobulin, were used as the detector reagent. The capsular polysaccharide (CPS) of SS2 and affinity-purified IgG from a healthy naive pig were immobilized on test and control regions of a nitrocellulose membrane, respectively. The ICS was used to 1) detect anti-CPS antibody in 14 sera taken from 4 SS2-infected pigs, 24 sera from pigs hyperimmunized with SS2, and 68 sera from pigs inoculated or infected with bacteria other than SS2; 2) determine anti-CPS antibody titers of 20 positive sera for comparison with enzyme-linked immunosorbent assay (ELISA); and 3) detect anti-CPS antibody in 226 clinical sera taken from diseased pigs also for comparison with ELISA. An ELISA used as a reference test determined the specificity and sensitivity of the ICS to be 97.1% and 86.3%, respectively. There was excellent agreement between the results obtained by ELISA and the ICS (kappa = 0.843). Additionally, there was strong agreement between the results of bacterial isolation from pig tonsils and ICS test (kappa = 0.658). Because it is rapid and easy to use, the test is suitable for the serological surveillance of SS2 at farms.
Introduction
Streptococcus suis is a common pathogenic microorganism and can cause a variety of clinical diseases in swine, including meningitis, pneumonia, endocarditis, polyserositis, and arthritis. 4,13 Based on the capsular polysaccharide (CPS) antigens of S. suis, 35 serotypes or capsular types (type 1 to 34 and type 1/2) have been characterized. 12 S. suis serotype (SS2) is most commonly associated with severe disease and is the serotype most frequently isolated from pigs. 8,23
S. suis is also an important zoonotic agent for humans and can be transmitted from pigs to humans mainly by infecting open wounds. In humans, SS2 has been identified as a causative agent of meningitis, 3 septicemia, 7 and endocarditis 11,20 and can lead to permanent hearing loss. 17 An outbreak associated with SS2 in Ziyang county, Sichuan province, People's Republic of China, in the summer of 2005 resulted in the infection of 215 people, with 38 fatalities. Most of the cases were associated with meningitis or streptococcal toxic shock syndrome. 19
Crowding, poor ventilation, sudden weather changes, mixing, relocation, immunization, and the presence of concurrent infections could predispose pigs to SS2 infection and morbidity. 17 It has been suggested that S. suis is an opportunistic bacterium of pigs. 18 Therefore, serosurveillance may be useful in determining the infection status and can play an important role in the control of streptococcosis in pigs.
In this study, an immunochromatographic strip (ICS) with high sensitivity and specificity was successfully developed for the detection of swine antibodies against CPS of SS2. The test is rapid, easy to use, and suitable for the serological surveillance of SS2 infection.
Materials and methods
Bacteria strains and cultures
SS2 strain SCZY05 was isolated from the liver and spleen of a pig that had died of meningitis/septicemia during the SS2 outbreak in human and pigs in Ziyang County, China, in August 2005. Five grams of liver/spleen tissue in 10 ml of sterile physiological saline were homogenized in a sterile grinder and centrifuged at 1000 × g for 10 minutes. Fifty microliters of the supernatant was transferred to a tryptic soy agar a plate containing 5% sheep blood and spread with a bent glass rod. After incubation for 20 hours under aerobic conditions at 37°C, the colonies showing hemolysis were selected for further identification. An isolate, numbered SCZY05, was confirmed as SS2 by Gram staining and polymerase chain reaction assays for amplification of specific genes, including glutamate dehydrogenase (gdh), capsular synthesis gene 2J (cps2J), muramidase-released protein (mrp), extracellular protein factor (epf), and hemolysin (sly), were carried out as described, 14,16,22,24 as well as agglutination tests with specific rabbit antisera. 1
The other 2 strains of SS2, LT-01 and LT-02, isolated from a pig without any clinical symptoms and a pig that died from septicemia, respectively, were supplied by the Veterinary Hospital at Huazhong Agricultural University (HZAU). Strain 050412 of S. suis type 1 was isolated in the author's laboratory in 2005. Strains C55138 and C55162 of S. suis group C were provided by the China Institute of Veterinary Drug Control.
All strains were cultured on tryptic soy agar plates containing 5% heat-inactivated sterile bovine sera at 37°C for 18 hours or in 10 ml of Todd-Hewitt broth a for 12 to 14 hours under aerobic conditions at 37°C with shaking.
Preparation of capsular polysaccharide from Streptococcus suis serotype 2
Capsular polysaccharide extracted from SS2 of SCZY05 was purified by gel filtration using a Sepharose CL-6B column as previously described. 2 The purified CPS was free from protein as determined by Bradford protein assay.
Production of hyperimmune serum
Formalin-inactivated whole cells of the 6 S. suis strains used in this study were prepared as described previously. 2 A total of 56 5-week-old healthy piglets that were SS2 negative, as determined by nasal swab culture, were randomly divided into 7 groups of 8 and housed separately in 7 different rooms, with each pig staying in its own stainless steel cage. Pigs belonging to groups 1 to 6 were vaccinated with 1 ml of immunogen prepared from strains SCZY05, LT01, LT02, 050412, C55138, and C55162, respectively. Group 7 was the unvaccinated control. Groups 1 to 6 received booster vaccinations using the appropriate immunogen at 2 and 4 weeks post vaccination. Sera were obtained from blood (5 ml per animal) immediately before vaccination and at weekly intervals for 8 weeks. All sera were heat inactivated at 56°Cfor 30 minutes and stored at − 70°C until required.
Hyperimmune pig sera raised against Escherichia coli, Salmonella enterica serovar Typhi, Pasteuralla multocida, and Haemophilus parasuis were kindly provided by Dr. Zhang. b
Convalescent sera and clinical sera
Fourteen convalescent heat-inactivated sera, taken from 4 pigs 2 to 6 weeks post challenge with SCZY05, were obtained from a commercial source. c A total of 226 sera were collected from sick pigs seen at the Veterinary Hospital at HZAU between July 2005 and April 2006. All sera were inactivated at 56°C for 30 minutes and stored at − 70°C until required.
Preparation of detector and capture reagents
The detector reagent, 15-nm diameter colloidal gold particles labeled with staphylococcal protein A (SPA), was prepared as described previously, 25 except that the gold solution pH was adjusted to 6.5 instead of 8.5.
The test capture reagent, purified CPS, was diluted with 20 mM phosphate-buffered solution (PBS) (pH7.4) to 1, 1.5, 2.0, 2.5, and 3.0 mg/ml, respectively, and stored at − 20°C until required. Affinity-purified swine IgG was used as the control capture reagent. Ammonium sulfate was used to precipitate IgG from the serum taken from a 3-month-old healthy naive pig. The precipitated IgG was purified by a protein A affinity column a according to the manufacturer's specifications. The protein concentration of the purified IgG was determined with a DU 800 Nucleic Acid/Protein Analyzer d and was diluted to the working concentrations of 1.2, 1.4, 1.6, 1.8, and 2.0 mg/ml with PBS (pH7.2). The diluted IgG solutions were stored at − 70°C until required.
Preparation of the conjugate pad and immobilization of the capture reagents onto nitrocellulose membranes
The G-SPA solution was dispensed onto glass fiber paper (300 × 6 mm) at a speed of 50 μl per cm using an XYZ3050 Dispense Workstation, e and the conjugate pad was dried under vacuum. Different working concentrations of both the test and the control capture reagents were dispensed onto nitrocellulose membrane strips (300 × 25 mm) at a speed of 0.80 μl solution per cm. After drying for 2 hours at 37°C, the membrane strips were blocked by incubating in 20 mM PBS (pH 7.5) containing 2% (w/v) nonfat dried milk for 25 minutes and washed 3 times with PBS containing 0.1 % (v/v) Tween-20 for 3 minutes each time. The membrane was dried for 2 hours at 37°C and stored at 4°C.
The finished immunochromatographic strip product
The sample pad, conjugate pad, immobilized nitrocellulose membrane, and absorbent pad were glued together on a backing plate (300 × 70 mm), as shown in Figure 1, and then cut into 3-mm-wide strips using a CM-4000 cutter. e The strips were stored dried at 4°C until required.
Detection principle and test procedure
During the test, the sample or IgG reacts with the G-SPA conjugate to form a G-SPA-IgG complex that then flows laterally onto the nitrocellulose membrane by capillary action. For a positive sample, the specific antibody (IgG) binds the CPS, forming a red band at the test region. Excess G-SPA conjugate binds purified pig IgG at the control line, forming another red band. As a procedural control, the red band at the control line should appear regardless of the presence of specific IgG. Thus, the appearance of 2 red bands in the read-out zone indicates a positive result (Fig. 2A), and the appearance of only 1 red band at the control line indicates a negative result (Fig. 2B). The absence of a red band at the control position suggests an invalid test (Fig. 2C, D). Practically, the ICS is laid on a flat bench, and 150 μl of a 1/40 dilution of serum is added to the sample hole. The result is available in 15 minutes.
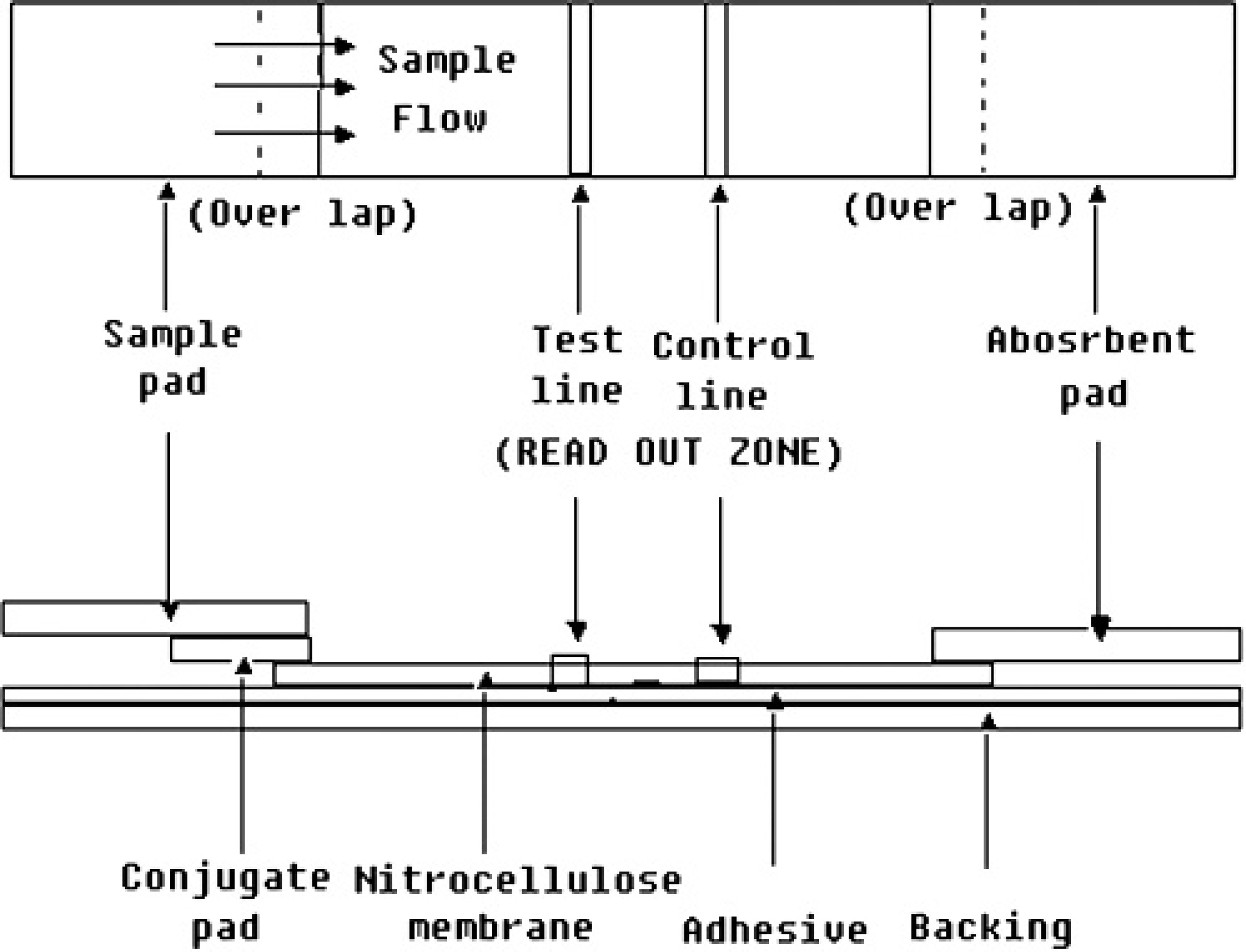
Schematic diagram of an immunochromatographic strip showing its several components. The conjugate pad was dispensed with IgG staphylococcal protein A solution. At the test line and control line position, purified capsular polysaccharide and pig IgG were immobilized, respectively.
Enzyme-linked immunosorbent assay (ELISA)
The ELISA assay based on purified CPS for detection antibody against SS2 was performed as previously described. 5 The purified CPS diluted to a final concentration of 5 μg/ml with 50 mM carbonate buffer (pH 9.2) was used as antigen in ELISA for coating 96-well microtiter plates f 100 μl per well. The plate was incubated overnight at room temperature (22-25°C). The solution was poured off, and the plate was washed 3 times with PBS containing 0.1% tween-20 (PBST). The remaining binding sites were blocked with 5% (w/v) bovine serum albumin in PBST (250 μl/well). The plates were incubated for 1 hour at 37°C and then washed well 3 times with PBST. The coated plates were stored at 4°C. During the test, 100 μl of a 40-fold diluted serum sample was added into the coated well, incubated for 30 minutes at room temperature, and then drained, and the plates were washed 3 times with PBST. One hundred microliters of horseradish peroxidase-labeled goat anti-pig IgG a at a working concentration of 1:1000 was added into each well. The plate was then incubated for 25 minutes at room temperature and washed 3 times again with PBST. One hundred microliters of substrate solution (TMB containing H2O2) was added into each well, and the color reaction was developed for 10 minutes at room temperature followed by stopping with 1 drop of 2 M H2SO4. The absorbance was read at 450 nm (OD450) with a microtiter plate reader. g An OD450 >0.157 indicated a positive and ≤0.157 a negative result. OD450 0.157 was the cut-off value calculated as the average value of the OD450 plus 3 standard deviations of results obtained from 20 healthy control sera.
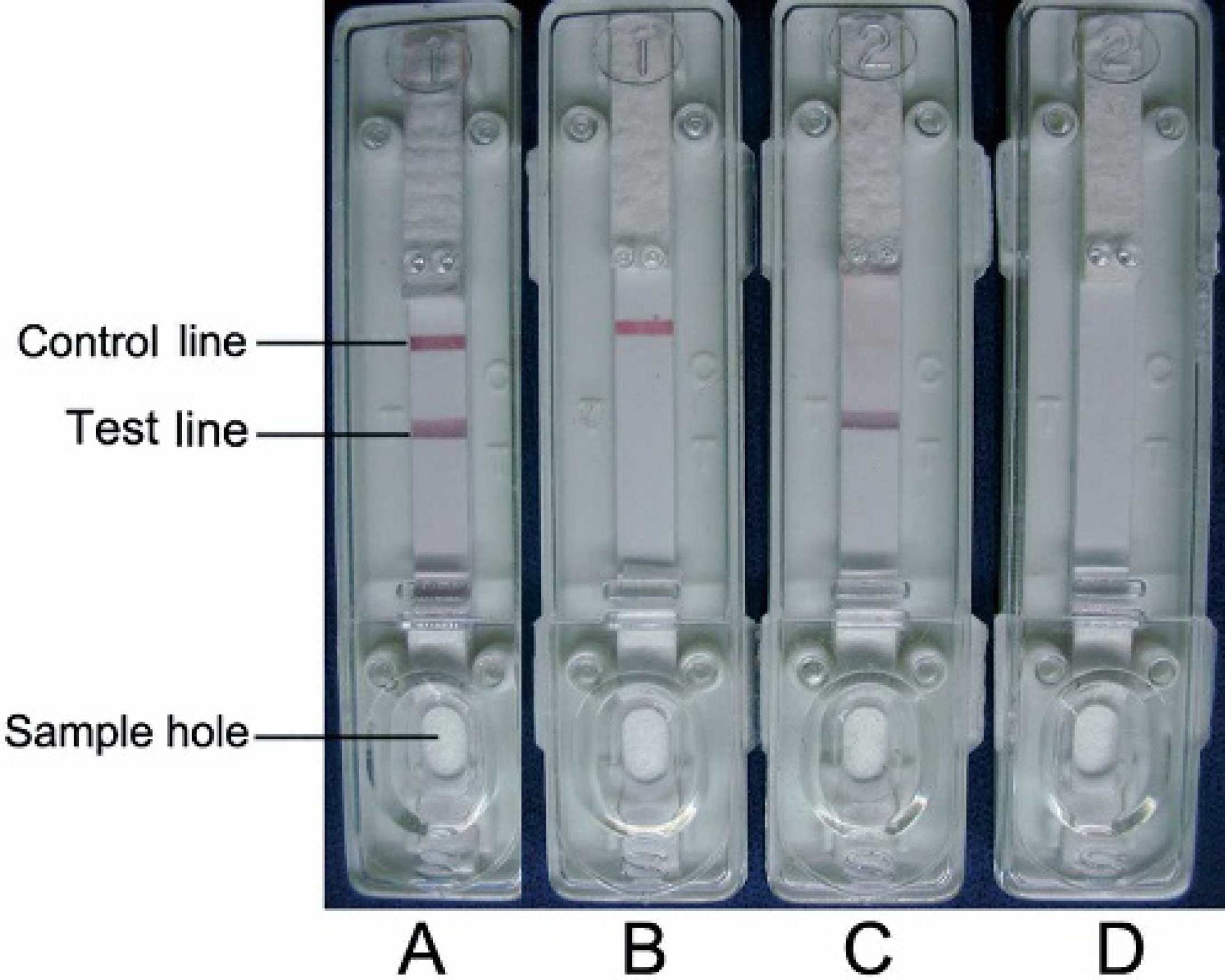
Interpretation of the results detected by immuno-chromatographic strip test.
Specificity and sensitivity of immunochromatographic strip and the agreement between immunochromatographic strip and reference methods
The strips were used to detect antibody in 14 sera from pigs that had survived challenge with SS2 and hyperimmune sera raised against SS2 (2 weeks post the third dose) and against bacteria other than SS2, as described above. All samples were tested by ICS and ELISA in triplicate. To evaluate the sensitivity of ICS compared with the ELISA, 20 hyperimmune sera were serially diluted (1:40-1:5,120) in 20 mM PBS (pH 7.6) and tested by ICS and ELISA simultaneously. Forty-fold dilutions of 226 clinical sera were tested by ICS and ELISA. The specificity and sensitivity of ICS were compared with those of ELISA. The specificity was calculated as the number of negative results in both ICS and ELISA tests divided by the number of negative results in ELISA. The sensitivity was calculated as the number of positive results in both tests divided by the number of positive results in ELISA. The agreement between ICS and ELISA was evaluated by kappa statistical analysis. 21 In order to investigate the correlation of antibody levels with SS2 infection status, the extent of bacterial isolation from the tonsils of 226 sick pigs was also used as a reference method. Bacterial isolation and serotype assay were performed as described above. The extent of correlation between ICS and bacterial isolation was determined by kappa statistical analysis. 21
Results
Optimal concentrations of capture reagents for immobilization
Preliminary experiments established that 2 and 1.6 mg/mL of CPS and pig IgG were the optimal concentrations for immobilization (data not shown).
Specificity of immunochromatographic strip (ICS) for hyperimmune serum samples compared with enzyme-linked immunosorbent assay (ELISA)
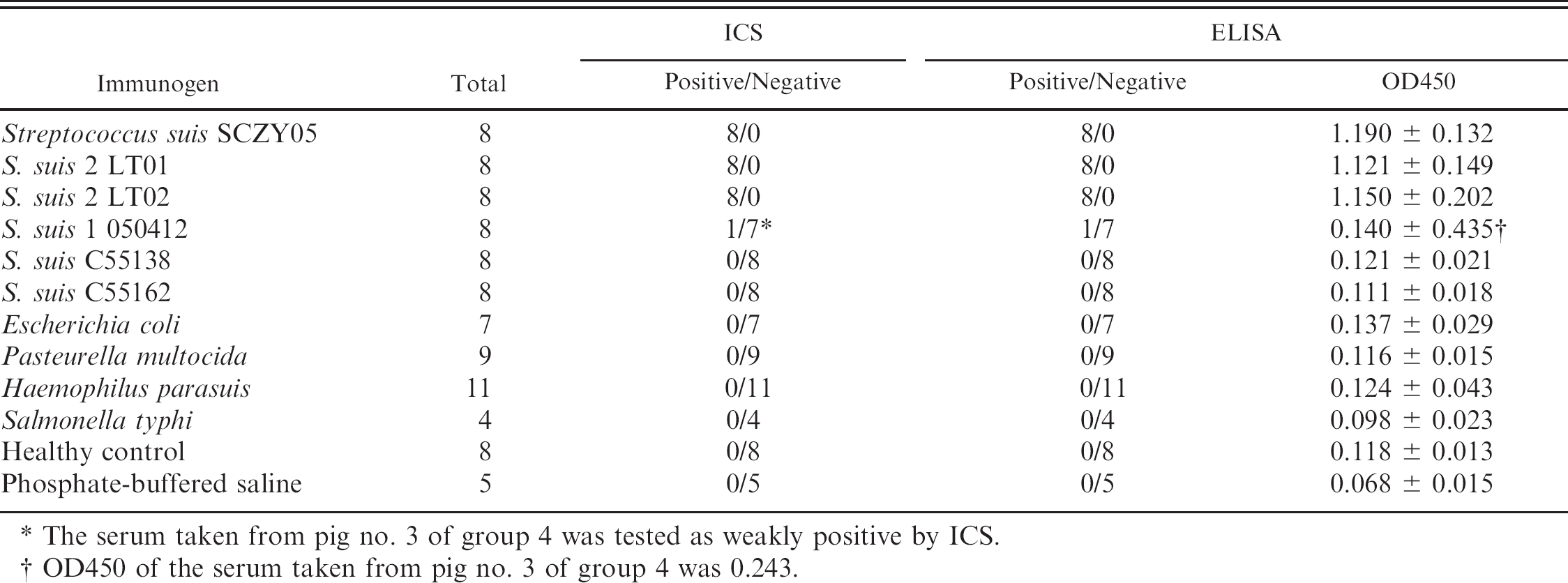
The serum taken from pig no. 3 of group 4 was tested as weakly positive by ICS.
OD450 of the serum taken from pig no. 3 of group 4 was 0.243.
Specificity and sensitivity of immunochromatographic strip
For 14 sera from pigs that had survived challenge with SS2, both ICS and ELISA gave all samples positive results. There was a perfect agreement between them (kappa = 1, data not shown). The antibody titers peaked at 3 weeks post challenge and dropped slowly from the fourth week post challenge. Both ICS and ELISA gave very low antibody titers (1:40) with sera taken at 6 weeks post challenge. For the 24 hyperimmune sera raised against SS2, there was also a 100% correlation between ELISA and ICS results. All of the 68 sera against non-SS2 bacteria, except one antisera against S. suis type 1 determined as weak positive by both ICS and ELISA, were negative in both ELISA and ICS tests (Table 1).
Antibody titers of 20 hyperimmune sera raised against SS2 strains SCZY05, LT01 and LT02 were tested by ICS and ELISA. For 12 of these sera, both tests gave the same antibody titers. The antibody titers of 3 sera were 1-fold higher by ICS than by ELISA. The antibody titers of 5 sera were 1-fold lower by ICS than by ELISA (Fig. 3).
The agreement between ICS and reference methods
Of the 226 clinical sera from sick pigs, 51 were antibody positive and 175 were negative by ELISA, whereas 49 were positive and 177 were negative by ICS. Additionally, 44 were positive in both assays and 170 were negative in both tests. Thus, the specificity and sensitivity of ICS, compared with ELISA, were 97.1 % and 86.3%, respectively. There was an excellent agreement (kappa = 0.843) between ICS and ELISA (Table 2). Bacterial isolation and serotype assay indicated that 37 pigs were SS2 positive and the other 189 pigs were SS2 negative. Thirty-one pigs were determined as seropositive by ICS and SS2 positive by bacterial isolation. One hundred and seventy-one pigs were seronegative and SS2 negative. In addition, ICS gave a positive result on 18 sera SS2-negative and a negative result on 6 SS2-positive pigs. A strong agreement (kappa = 0.658) was found between the extent of bacterial isolation and the ICS test results (Table 2).
Discussion
In this study, an ICS for antibody detection of SS2 was successfully developed. With ELISA used as a reference test, this test had good specificity (97.1%) and sensitivity (86.3%). There was excellent agreement between the results obtained by ELISA and the ICS (kappa = 0.843). The quantifying test result indicated that the sensitivity of the strip test was close to or slightly less than that of ELISA. Additionally, there was strong agreement between the results of bacterial isolation and ICS test (kappa = 0.658). These data indicate that there is a correlation between CPS antibody levels and SS2 infection status, suggesting that ICS would be a valuable tool for use in surveillance of SS2.
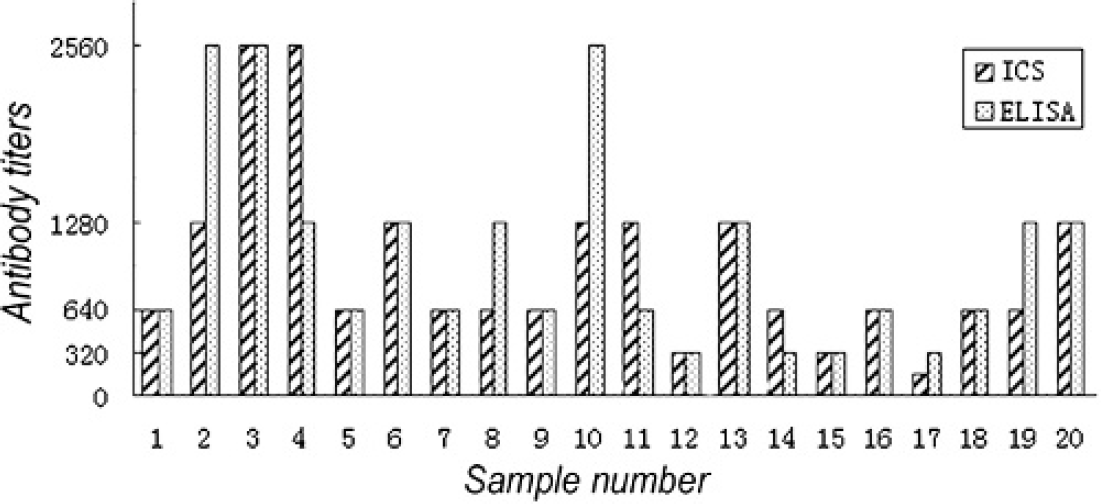
Antibody titers tested by immunochromatographic strip and enzyme-linked immunosorbent assay.
Results of antibody detection and bacterial isolation of clinical samples
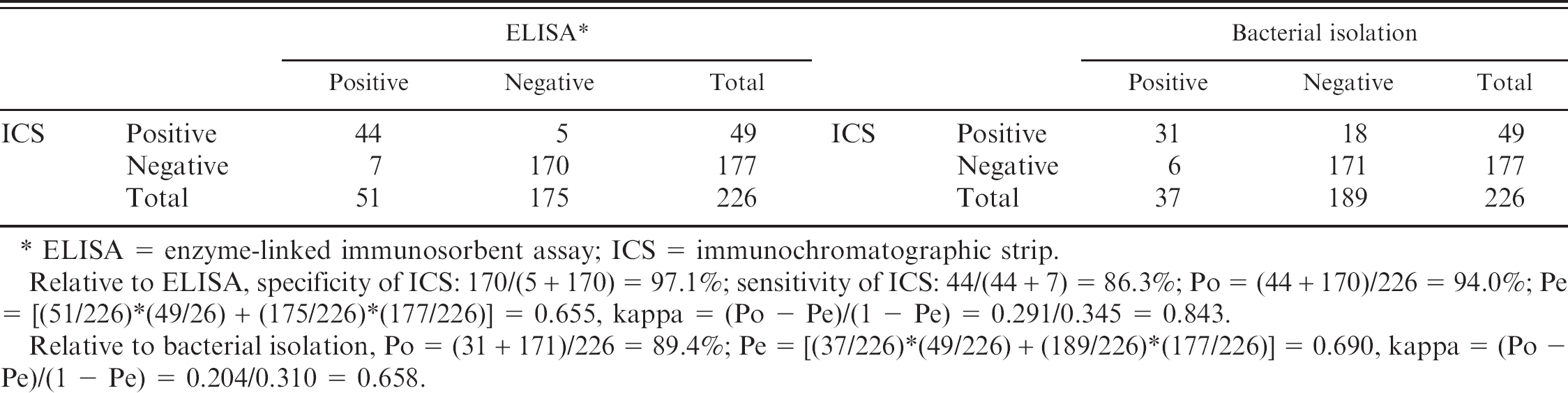
ELISA = enzyme-linked immunosorbent assay; ICS = immunochromatographic strip.
Relative to ELISA, specificity of ICS: 170/(5 + 170) = 97.1%; sensitivity of ICS: 44/(44 + 7) = 86.3%;Po = (44 + 170)/226 = 94.0%;Pe = [(51/226)*(49/26) + (175/226)*(177/226)] = 0.655, kappa = (Po - Pe)/(1 - Pe) = 0.291/0.345 = 0.843.
Relative to bacterial isolation, Po = (31 + 171)/226 = 89.4%;Pe = [(37/226)*(49/226) + (189/226)*(177/226)] = 0.690, kappa = (Po - Pe)/(1 - Pe) = 0.204/0.310 = 0.658.
Gold conjugates have increasingly been used in ICS for rapid diagnosis. Most proteins and antibody can be easily coupled to colloidal gold particles. When developing an ICS for antibody detection, the gold-antigen conjugate is an excellent detector reagent. Just as the ICS for trichinellosis detection, 25 excretory-secretory protein antigen of Trichinella spiralis conjugated with gold particles was used as the detector probe. In the study reported here, however, there was a failure to label gold particles with the antigen-CPS, even after many different conditions were evaluated. This may be due to the CPS's being a polysaccharide and not a protein. Hence, we used SPA, which can bind to the FC fragment of mammalian immunoglobulin, labeled with gold particles as the detector reagent. The pH of the gold solution and the quantity of the protein used are the most 2 important factors for the successful preparation of a gold-protein conjugate. The gold solution will bind proteins more efficiently when its pH is close to or 0.5 higher than the isoelectric point (pI) of the protein (the pI of SPA is about 6.0). Tighter binding occurs at a higher pH, but this may have a denaturing effect on the protein, making the probe less effective. Having excess protein coupled to the gold particles may also be disadvantageous: some of the weakly bound protein may detach from the particles, thus making the probe less effective because the free protein will compete for binding sites with the gold-labeled protein. Additionally, inadequately labeled protein will decrease the sensitivity. Because both specific and nonspecific antibodies in serum will bind to SPA, enough gold-SPA solution should be dispensed onto the glass fiber paper (conjugate pad) to avoid the nonspecific antibody's minimizing the sites available for binding of specific antibody.
The specificity and sensitivity of the ICS are largely dependent on the antigen used in the test strip. Many scientists have taken a great interest in CPS and S. suis infection. Enzyme-linked immunosorbent assay for antibody detection of SS2 based on purified CPS is much more specific than that based on whole-cell antigen. 5 The duration of bacterial culture is critical for obtaining high-quantity and -quality CPS. 9 In this study, cultures were incubated at 37°C for 8 hours with shaking at 250 rpm, and this was sufficient to obtain the necessary material. Based on the ability to detect antibodies to SS2 CPS in ELISA, an ICS test with CPS extracted from SS2 strain SCZY05 has been formulated. In order to reduce nonspecific background binding, contaminating proteins must be eliminated from the CPS preparation. In this study, the purified CPS was free from protein, as determined by the Bradford protein assay. In addition, the specificity and sensitivity of the strip are also affected by the materials, including the sample pad, conjugate pad, and nitrocellulose membrane. When developing an ICS test, it is advisable to evaluate materials from different manufacturers and select those that are optimal based on empirical data.
The cross-reaction of 1 serum sample, from an animal vaccinated with S. suis type 1, was probably due to common epitopes present in the capsule. It has previously been reported that CPS of SS2 cross-react with antisera raised against SS 1/2, 12, and 17 as determined by ELISA. 5 The capsule of SS2 and SS1/2 share some common sugar residues 6 or antigenic determinants. 16 The ICS could not differentiate the antisera raised against SS2 from that raised against SS1/2.
That SS2 could not be isolated from 18 seropositive pigs tested by ICS might be due to antibiotics treatment, which could result in failure to routine bacterial isolation. On the other hand, 6 pigs infected with SS2 were determined as antibody negative. These pigs might be infected a very short time before the sample collection, and their anti-CPS antibody levels were too low to be detected by ICS. In addition, anti-CPS antibodies are usually not long lasting at a high level in pig sera. We also found that anti-CPS antibody titers were at a low level (between 80 and 320, data not shown), which confirmed the data reported previously. 5 Despite these disadvantages, this experiment gave a strong agreement (kappa = 0.658) between bacterial isolation and ICS, which indicated that antibody monitoring might be useful in determining the SS2 infection status and that ICS developed in this study could be used as a useful tool for seroepidemiological surveillance, especially for screening large numbers of blood samples in pig stocks.
More interestingly, SPA has the capacity to bind to the Fc fragment of IgG of humans and other mammalian species. Furthermore, SPA can also react with IgM of humans 10 and some other animal species, such as the dog. 15 Whether SPA can react with IgM of swine is unclear. Theoretically, the ICS is also suitable for antibody detection at early stages of SS2 infection. In addition, many other nonporcine hosts, such as humans, can be infected with or carry SS2, and therefore the ICS may also be useful in serosurveillance with these species.
Although some assays, such as the indirect ELISA and agar gel precipitation tests, have been developed for antibody detection for S. suis, these methods are not suitable for use outside of the research laboratory. In contrast, the ICS is easy to use and rapid. In this context, the ICS can be used as a tool for seroepidemiological survey of pigs from different areas in China. This would in turn facilitate the formulation of effective control measures.
In summary, the results of this preliminary study indicate that the ICS has high specificity and sensitivity. The ICS will potentially be of great value in seroepidemiological surveys of streptococcosis associated with SS2.
Acknowledgements
This study was supported by the National 973 Program (2006CB504404), National 863 Program (2006AA10A206), and Chinese National S&T program (2004BA519A60). We thank Dr. Rui Zhou for helpful advice on the experiment design. We also thank Dr. Paul R. Langford, Dr. Chuanqing Wang, and Dr. Doug Begg for helpful discussion and English checking.
Footnotes
a.
Sigma-Aldrich, Inc., Spruce Street, St. Louis, MO.
b.
State Key Laboratory of Agricultural Microbiology, Wuhan, China.
c.
Keqian Biological Products Co., Ltd., Shizishan Street, Wuhan, China.
d.
Beckman Coulter, Inc., Harbor Boulevard, Fullerton, CA.
e.
BioDot, Inc., Sky Park, Irvine, CA.
f.
Costar Corning Inc., Corning, NY.
g.
Bio-Rad Laboratories Inc., Richmond, CA.
