Abstract
An immunochromatographic assay was developed to detect Salmonella enterica serovars Typhimurium and Enteritidis in a single strip. The assay was constructed in the form of a sandwich, using 2 specific anti–S. Typhimurium and anti–S. Enteritidis antibodies immobilized on a nitrocellulose membrane at separated test lines, while the other specific antibody to Salmonella spp. was conjugated with gold nanoparticles. The test strips can immediately detect S. Typhimurium and S. Enteritidis specifically in a culture medium at levels as low as 104 and 106 cfu/ml, respectively. The contamination of S. Typhimurium and S. Enteritidis at 1 cfu/ml or greater can be detected by the test strips after 6–24 hr incubation. The specificities of the test strips to detect S. Typhimurium and S. Enteritidis, in spiked samples, were obtained at 100%; the sensitivities were at 98.89% (89/90) and 87.50% (70/80), respectively, compared with the conventional method. The newly developed multiplex immunochromatographic assay is the first report on the efficient detection of both S. Typhimurium and S. Enteritidis simultaneously in single strip. This test strip also provides advantages of simplicity and very rapid detection of these specific bacterial contaminants in chicken and can be useful for mass detection on chicken farms and in other veterinary products.
Keywords
Salmonella is a well-documented pathogen known to occur in a wide range of foods, especially poultry products, which are implicated in the majority of traceable food-borne illnesses reported in Thailand and worldwide (U.S. Department of Agriculture, Foreign Agricultural Service: 2008, EU-27 poultry and products annual report 2008. Global Agriculture Information Network. Available at: http://www.fas.usda.gov/gainfiles/200803/146293877.pdf. Accessed on December 20, 2010; National Institute of Health, Department of Medical Sciences, Ministry of Public Health, Thailand: 2006, Annual Report of Confirmed Salmonella and Shigella in Thailand 2006. Available at: http://webdb.dmsc.moph.go.th/ifc_nih/applications/files/Annual_report_2006.pdf. Accessed on December 20, 2010). 16,18 Several steps involved in raising and processing poultry can create various potential risks for contamination with Salmonella.
Salmonella enterica comprises more than 1,400 non-typhoid serotypes, of which approximately 29% have been isolated from poultry (World Health Organization: 2009, Salmonella and Campylobacter in chicken meat: meeting report. Microbiological Risk Assessment Series 19. Available at: http://www.who.int/foodsafety/publications/micro/MRA19.pdf. Accessed on December 21, 2010). 1,11,12,17 A wide range of serotypes that cause human disease can be broadly grouped into the typhoidal species that are specific human pathogens and include serotypes Typhi and Paratyphi. The non-typhoid serotypes are primarily spread to human beings from animal sources. 1,5,8
Salmonella Typhimurium and S. Enteritidis emerged as important pathogens in poultry and human beings and remain the serovars most frequently isolated from patients suffering from chicken food poisoning. 15 The minimum contamination of Salmonella that consequently causes disease has not been established. The food law of the European Union insists on the absence of Salmonella (i.e., not detectable in 25 g of food product; the Commission Regulation (EC) nos. 2160/2003 [http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2003:325:0001:0015:EN:PDF] and 1441/2007 [http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007:322:0012:0029:EN:PDF]; Department of Livestock Development, Ministry of Agriculture and Cooperatives, Thailand: 2009, Control of Salmonella in poultry. Available at http://www.dld.go.th/certify/certify/page/page_law/standards-farm/data/Salmonellosis.pdf). Commission Regulation no. 646/2007 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007:151:0021:0025:EN:pdf) amended the Community target, as referred by Article 4(1) of Commission Regulation (EC) no. 2160/2003 and repealing Regulation (EC) no. 1091/2005 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2005:182:0003:0004:EN:PDF). These regulations aim to control contamination of flocks of broilers in farms by S. Enteritidis and S. Typhimurium by reducing the prevalence of these 2 serovars to a maximum to 1% or less by December 31, 2011. Therefore, countries that export chicken products to the European Union are required to strictly control their products according to the above regulations. In the past 10 years, Thailand has been one of the leading exporters of agricultural products, particularly poultry and poultry meat products, shrimp, seafood, rice grain and cereal products, and fruits and vegetables. (Proportion of export value of livestock goods, 2008. Available at: http://www.dld.go.th/ict/stat_web/yearly/yearly51/imex51/graph/graph_2.pdf. Accessed on December 23, 2010; Statistics of cooked chicken meat for export by destination 2004-2008. Available at: http://www.dld.go.th/ict/stat_web/yearly/yearly51/imex51/page/imex51-31.pdf. Accessed on December 23, 2010; The determinants of successful export marketing strategy in Thai processed agricultural products. Available at http://www.iis.ru.ac.th/download/Journal/pdfvol3(1)/5.%20Woraphot%20Kantapipat.pdf. Accessed on December 23, 2010). 14 Currently, the Department of Livestock Development of Thailand routinely checks for the presence of Salmonella spp., S. Enteritidis, and S. Typhimurium in farms to control the quality of chicken products in order to comply with international regulations. 5 Therefore, a rapid, simple, and accurate Salmonella screening method that can be carried out on-site is necessary, and has been in increasing demand, not only on chicken farms but also in various food industries.
Lateral flow assay or simple strip assay is based on an immunochromatographic procedure that utilizes antigen–antibody properties in a novel manner that provides rapid detection of analyte. The strip has the advantage and benefit of being in a user-friendly format, taking a very short time to get test results, providing long-term stability over a wide range of climates, and being relatively inexpensive to produce. These characteristic advantages render the strip assay ideally suited for on-site testing, even by untrained personnel, and to become a common diagnostic tool for bacteria, such as Salmonella spp., Listeria spp., and Escherichia coli. 7,10
Various commercially available antibodies were screened and selected to develop the test system. The specific rabbit polyclonal antibody to Salmonella spp. a was conjugated to colloidal gold (diameter = 40 nm) and applied on a conjugate pad using planar chromatography. b The mouse anti–S. Typhimurium a and mouse anti–S. Enteritidis a antibodies were applied onto a nitrocellulose membrane at separated positions that would become the capture test lines of the strip. Swine anti-rabbit antibody c was applied on the same nitrocellulose membrane at a position that would become the control line (Fig. 1). The membrane was dried for 1 hr at room temperature and stored in closed packaging with silica gel at 4°C. For kit assembly, the conjugate pad was attached at the end upstream of the test line of the nitrocellulose membrane, an absorption pad was placed at the opposite end downstream from the control line, and a sample pad was placed at the end upstream from the conjugate pad.
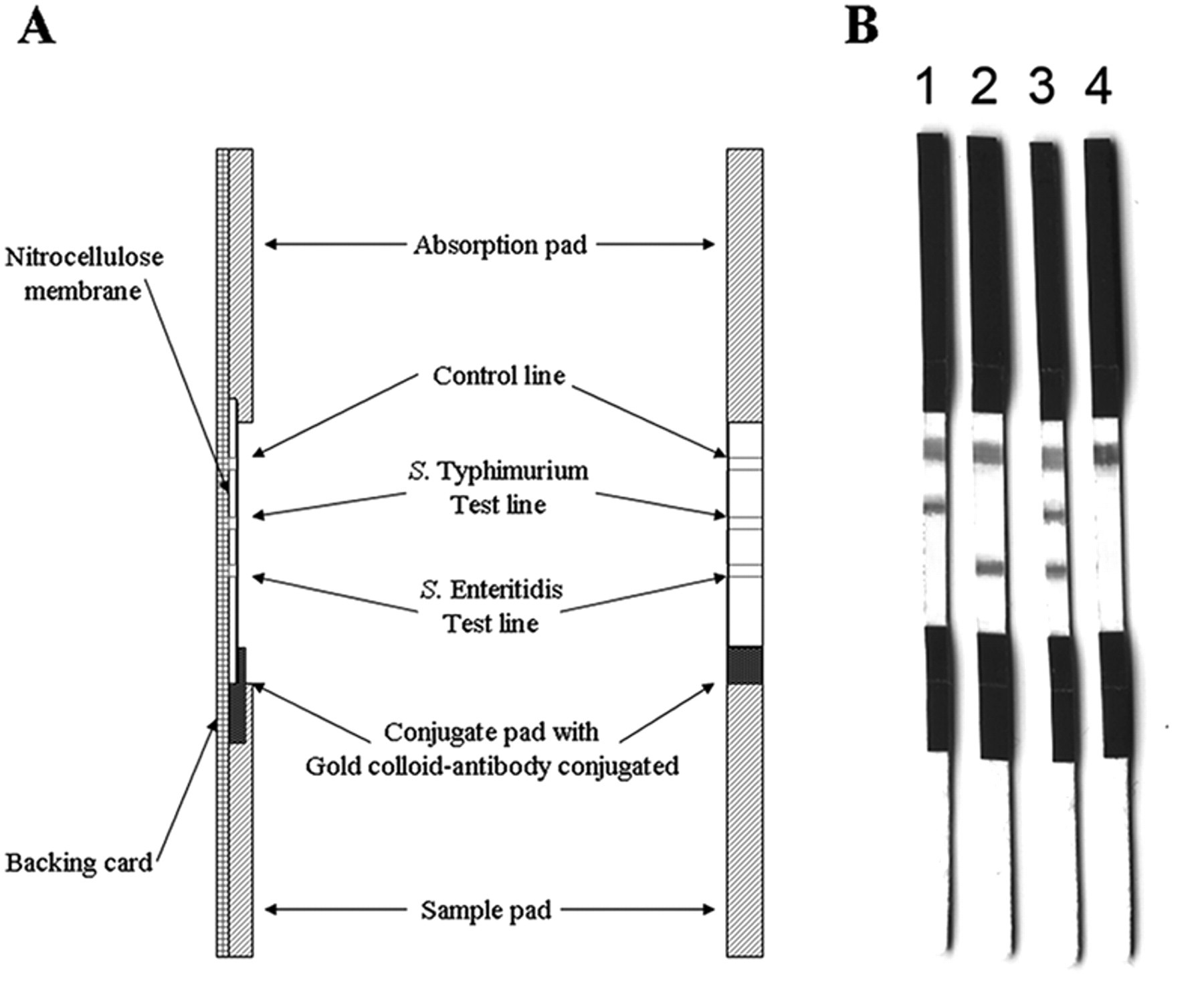
A, the schematic description of the immunochromatographic test device. B, result of newly developed test strips for detection of 1: Salmonella enterica serovar Typhimurium, 2: Salmonella enterica serovar Enteritidis, 3: mixture of S. Typhimurium and S. Enteritidis, and 4: Escherichia coli. All microorganisms at 108 cfu/ml were tested.
The developed immunochromatographic strips were tested for specificity, using various reference microorganisms obtained from the American Type Culture Collection (ATCC), the Culture Collection for Medical Microorganism, Department of Medical Sciences Thailand (DMST), and the Veterinary Public Health Laboratory, Department of Livestock Development Thailand (DLDT). The 44 reference strains were composed of 4 strains of Salmonella Typhimurium, 4 strains of S. Enteritidis, 27 strains of other Salmonella species, and 9 strains of non-Salmonella species. The identifications of reference Salmonella strains were confirmed by conventional methods and serotyping based on the antigenic structure of both somatic or cell wall (O) antigens, flagellar (H) antigens, and capsular (Vi) antigens. The standard procedure for Salmonella identification in poultry was also performed according to the Bacteriological Analytical Manual (http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/%20BacteriologicalAnalyticalManualBAM/ucm070149.htm#content) and International Standard ISO 6579:2002. The media consisted of nonselective pre-enrichment in buffered peptone water (BPW) d at 37°C for 24 hr, followed by selective enrichment in Rappaport–Vassiliadis (RV) medium d and Müller–Kauffmann tetrathionate–novobiocin (MKTTn) broth d at 42°C for 24 hr. The selective solid media xylose–lysine–deoxycholate (XLD) agar and brilliant green agar (BGA) were further used to identify the microorganisms. A final analysis of serological typing was then carried out to confirm the serovars. 4,6
One ml of each bacterial sample grown in BPW for 24 hr at 37°C was centrifuged at 6,000 × g e and adjusted to 1 × 108 cfu/ml. The supernatant was discarded, and the packed cells were re-suspended with 1 ml of 0.01 M phosphate buffered saline (PBS; pH 7.4). Each sample was boiled at 100°C for 10 min, then immersed in an ice bath immediately. 10 The heat lysed bacteria was reported to maintain the antigen required for the test and would normally render clear results. Therefore, it is suitable to perform by strip test. The lysed sample (200 µl) was transferred into a new tube. The test strip was placed into the tube with the sample pad end down, soaking into the sample, allowing at least a 5–15 min time interval for adequate absorption and migration of the liquid along the strip. The strip was then examined for a positive red color presented on the S. Typhimurium test line or S. Enteritidis test line (Fig. 1A). If there was combined contamination of both S. Typhimurium and S. Enteritidis, a positive red color of the 2 test lines would be obtained. The presence of a red line on the control line confirmed the acceptable performance of the test strips (Fig. 1B)
The specificity of the immunochromatographic strip was identified using the 44 reference strains of microorganisms. The test strips showed positive results with 4 strains of S. Typhimurium and 4 strains of S. Enteritidis and showed clearly negative results with 26 strains of other Salmonella species and 9 of non-Salmonella species. However, the test strip could not differentiate between S. Typhi and S. Enteritidis, since both of them were in Salmonella group D, and the somatic O antigens were very similar with 1, 9, 12 for S. Enteritidis and 9, 12 for S. Typhi. 8 Nevertheless, S. Typhi is the serovar that causes the disease in human beings, and not in chickens, and very rarely causes contamination in chicken meat. Therefore, positive results on the S. Enteritidis test line in chicken sample identification would be presumably interpreted as positive for S. Enteritidis. Moreover, both S. Typhi and S. Enteritidis could be further differentiated from the conventional method by biochemical reactions. In order to perform a quantitative assessment of Salmonella detection or the detection limit of the immunochromatographic strip, pure culture of S. Typhimurium ATCC 13311 and S. Enteritidis ATCC 13076 were grown in BPW. The number of bacteria had to be quantified using the most probable number (MPN) and the total aerobic viable counts methods before adjusting to certain number and lysing cells. 9,10,19 Bacteria starting at 1 × 108 cfu/ml were then ten-fold serially diluted (10-1–10-8 dilution) with 0.01 M of PBS (pH 7.4). Each sample was lysed as described previously and tested with the strips. The last dilution that yielded an observable positive result in the test strip was recorded as the sensitivity or detection limit of the test strip. The minimal concentrations of S. Typhimurium and S. Enteritidis obtained from pure culturing could generate positive bands on the test strip at 104 cfu/ml and 106 cfu/ml, respectively (Fig. 2). By comparing results to previous reports of immunochromatographic test strips for Salmonella spp., the sensitivities were also obtained at range 104–106 cfu/ml. 3

The detection limit of the newly developed test strip obtained with (A) Salmonella enterica serovar Typhimurium at 104 cfu/ml and (B) Salmonella enterica serovar Enteritidis at 106 cfu/ml.
To determine the detection of microorganisms at only 1 cfu/ml, an enrichment step was necessary. Salmonella Typhimurium ATCC 13311 and S. Enteritidis ATCC 13076, at 1 cfu/ml in BPW, were incubated for 0, 2, 4, 6, and 24 hr, at 37°C. One ml of each collected culture was lysed and tested with the strip. After 6–24 hr of incubation, the strips could demonstrate clearly positive results. The generation time of S. Typhimurium and S. Enteritidis previously analyzed in the study was 16.8 and 17.4 min, respectively. Therefore, after only 6 hr of incubation, the numbers of microorganisms were increased high enough to reach the detection limit of the developed test strip.
The application of test strips to detect the targeted microorganisms in chicken samples was evaluated, and the sensitivity and specificity of the developed strips were determined. Each chicken meat, pre-identified as free of Salmonella spp., was spiked either with S. Typhimurium ATCC 13311 (90 samples), S. Enteritidis ATCC 13076 (80 samples), Campylobacter coli ATTC 43485 (5 samples), Campylobacter jejuni ATCC 33291(5 samples), or E. coli ATCC 35218 (5 samples) at 15–20 cfu/25 g in 225 ml of BPW. After an overnight incubation, 1 ml from each sample was lysed and tested with the strips. Identification by test strips was compared with the conventional method. The sensitivity and specificity of test strips were calculated, using the following results: TP (a true-positive result: conventional method), TN (a true-negative result: conventional method), FN (a false-negative result), and FP (a false-positive result). The diagnostic sensitivity was calculated in percentage as {[TP/(TP + FN)] × 100}, and the diagnostic specificity was calculated in percentage as {[TN/(TN + FP)] × 100}. 13 The sensitivities of test strips for detecting S. Typhimurium and S. Enteritidis in chicken meat samples were 98.89% (89/90) and 87.50% (70/80), respectively, and the specificities were obtained at 100% (15/15) and 100% (15/15), respectively (Table 1).
The sensitivity and specificity of the newly developed test strips for detection of Salmonella enterica serovars Typhimurium and Enteritidis in chicken samples were obtained by comparing the results from the test strips with conventional method.*
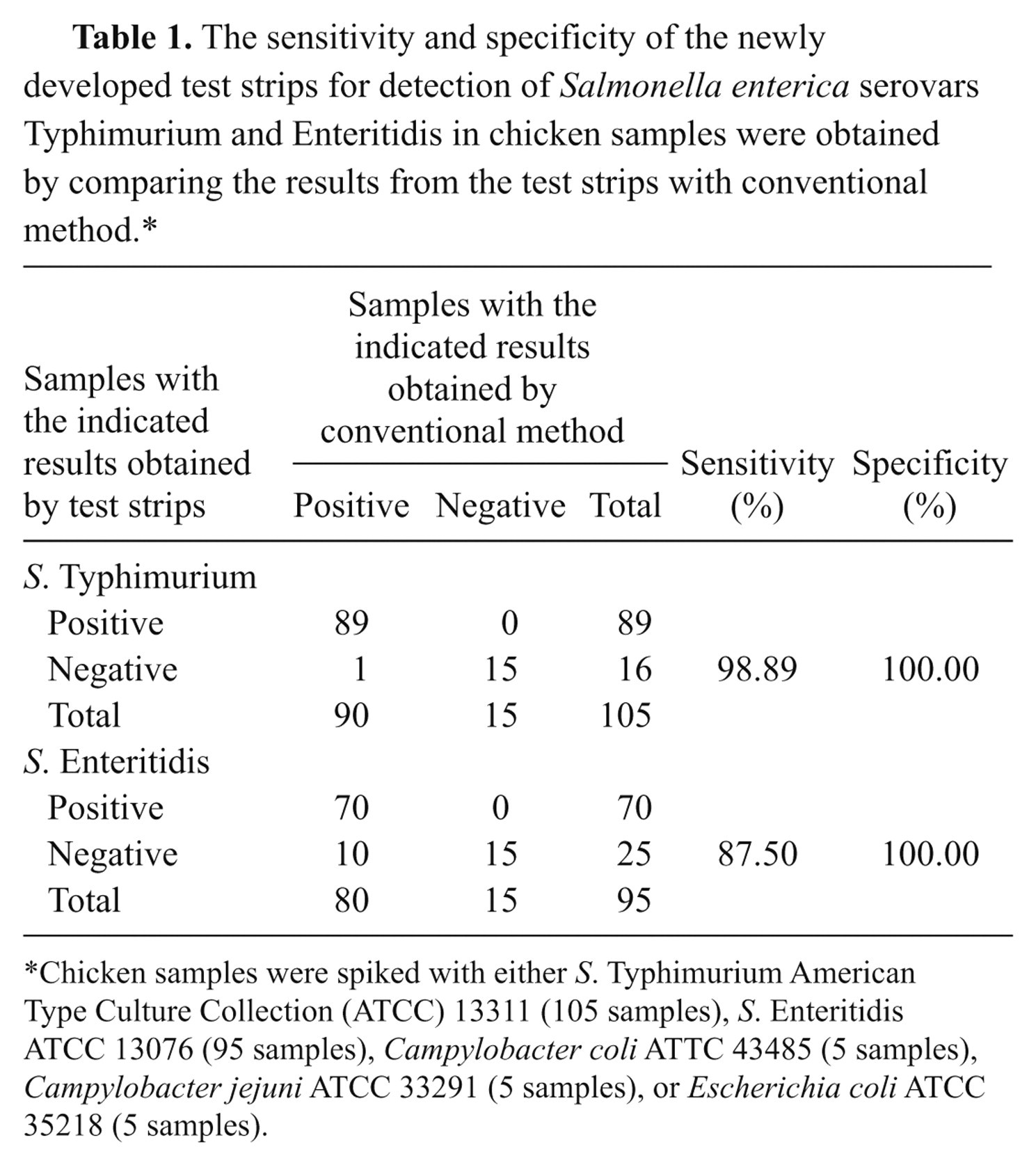
Chicken samples were spiked with either S. Typhimurium American Type Culture Collection (ATCC) 13311 (105 samples), S. Enteritidis ATCC 13076 (95 samples), Campylobacter coli ATTC 43485 (5 samples), Campylobacter jejuni ATCC 33291 (5 samples), or Escherichia coli ATCC 35218 (5 samples).
To evaluate the stability of the immunochromatographic strips during storage, the test strips were stored in closed packaging with silica gel at 4–8°C. Pure lysed cultures of S. Typhimurium ATCC 13311, S. Enteritidis ATCC 13076, C. coli ATTC 43485, C. jejuni ATCC 33291, and E. coli ATCC 35218 were used to evaluate the reaction and detection limit performance at 0, 1, 3, 6, and 12 months. The test strips showed no significant change of the immunorecognition bands for detecting the microorganism in months 0, 1, 3, 6, and 12. The detection limit was still at 104 cfu/ml and 106 cfu/ml for S. Typhimurium and S. Enteritidis, respectively. The test strips remained functional and stable under dry storage condition at 4–8°C for at least 12 months after preparation.
Immunochromatographic test kits for detecting pathogens in enriched food samples have been introduced and reported in recent years, particularly for the detection of E. coli O157:H7, Salmonella spp., and Listeria spp., using commercially available test kits. 2,8 Salmonella remains one of the most frequently-isolated human food-borne pathogens, causing significant problems in human as well as veterinary medicine. Rapid and specific isolates of Salmonella serovars are the keys to effective outbreak tracking and control. Currently, there are no immunochromatographic test kits available commercially for the detection of S. Typhimurium and S. Enteritidis, especially in the same test-strip format. The specificities and sensitivity of the newly-developed test strips described in the current study are also comparable with the conventional method.
In addition, the strip test kit is portable, does not require special tools or training, and yields rapid results within 5–15 min. After a 6–24-hr enrichment step, contamination as low as 1 cfu/ml can be determined. On the other hand, the standard conventional method is time-consuming, taking approximately a week to produce results, and requires specific technical skill, and careful, consecutive, and tedious work from microbiologists. The test strip described in the present study would be beneficial to poultry farms and in other veterinary production where contaminations of S. Typhimurium and/or S. Enteritidis need to be monitored.
Footnotes
Acknowledgements
The authors are grateful to the Department of Microbiology, Faculty of Pharmacy, Mahidol University for supporting facilities. The staff at the Veterinary Public Health Laboratory, Ministry of Agriculture and Cooperatives, Department of Livestock Development are appreciated for the assistance in samples collection and conventional identification. The authors wish to thank Professor Dr. Neelobol Neungton, Department of Biochemistry, Siriraj Hospital, Mahidol University, Thailand for her suggestions and comments on the manuscript.
a.
United States Biological Inc., Swampscott, MA.
b.
LINOMAT 5, Camag Co. Ltd., Muttenz, Switzerland.
c.
Dako North America Inc., Carpinteria, CA.
d.
Oxoid Ltd., Cambridge, United Kingdom.
e.
HETTICH-Zentrifugen GmbH & Co. KG, Tuttlingen, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
