Abstract
An immunochromatographic strip was developed for the detection of an antibody against Porcine hemagglutinating encephalomyelitis virus (PHEV). Colloidal gold–labeled rabbit anti-pig immunoglobulin G (IgG) was used as the detection reagent, and the PHEV recombinant antigens and goat anti-rabbit IgG were coated on the prototype strip and the control lines, respectively. The immunochromatographic strip was capable of specifically detecting PHEV antibodies in serum with a hemagglutination inhibition (HI) titer of 2 within 10 min. Storage of the strips at room temperature for 6 months or at 4°C for 12 months did not change their sensitivity and specificity. Using HI as a reference test, the relative specificity and sensitivity of the immunochromatographic strip were determined to be 93.41% and 98.42%, respectively. There was a strong agreement between the results obtained by HI and the immunochromatographic strips (κ = 0.926). Additionally, there was a strong agreement between enzyme-linked immunosorbent assay and immunochromatographic strips (κ = 0.929). When the immunochromatographic strip was used for serological diagnosis of 1,117 serum samples in Jilin Province in China, the seropositivity ranged from 6.5% in the Liaoyuan District to 81.6% in the Changchun District. Furthermore, many piglets were seropositive to PHEV, indicating the possible transfer of maternal antibodies via the colostrum. Based on the high specificity, sensitivity, and stability of the immunochromatographic strip, it would be suitable for on-site detection of PHEV antibodies in order to monitor PHEV infections in an animal population.
Keywords
Introduction
Porcine hemagglutinating encephalomyelitis is an acute, highly contagious disease in piglets caused by the Porcine hemagglutinating encephalomyelitis virus (PHEV), 16 which mainly infects 1- to 3-week-old piglets and causes vomiting, exhaustion, and neurological signs. The mortality rate varies from 20% to 100%. 7 In 1962, the pathogen was isolated for the first time from suckling piglets suffering from encephalomyelitis in Canada. 17 In 1972, the PHEV-VW572 strain was isolated from the tonsils of pigs that exhibited only vomiting and exhaustion symptoms, but no neurological signs. 26 Infections have been reported in the major pig raising countries of Europe and Asia as well as North America where PHEV seemed to be endemic without any clinical outbreaks. 2,6,25 In China, PHEV was first reported in 1986 on the mainland and later in Taiwan Province. 7,10
Serological surveys revealed that PHEV infections in pigs are very common worldwide, so there are different serum antibodies in different countries. The reported seropositivity rate of pigs for PHEV is 31% in Canada, 13 46% in Northern Ireland, 3 49% in England, 6 52–82% in Japan, 20 and 10.9–98.6% in the United States. 23 Currently, the incidence of the disease among pigs in many countries is on the rise, and it has caused great economic losses to the pig industry. 31 In August 2006, various pig farms in Argentina experienced outbreaks of this disease, leading to 1,226 swine deaths and a morbidity rate as high as 52.6% as well as a mortality rate of 16.9%. 28 In 2000, the occurrence of encephalomyelitis in piglets caused by PHEV on 3 farms in Jilin Province in China was reported, and such cases were characterized by high mortality (80.6%). 12 In 2007, PHEV occurred twice at pig farms in Jilin Province, with incidence rates among 20-day-old piglets as high as 100% and reported mortality rates of 48% and 100%. 18 Therefore, early detection and control of PHEV infection potentially can reduce the economic and health effect of the disease.
At present, various laboratory methods are available for the detection and surveillance of PHEV antibodies, including the hemagglutination (HA) and hemagglutination inhibition (HI) tests, 32 the enzyme-linked immunosorbent assay (ELISA), 10 and the virus neutralization (VN) test. 30 However, these detection methods are laborious, time-consuming, and require laboratory operations or special equipment, which makes these methods unsuitable for on-site inspection. As such, the current methods would not be useful for managing an outbreak of the disease. Therefore, it was necessary to develop a sensitive, specific, and easily performed detection method for on-site detection of PHEV antibodies in order to increase the surveillance of PHEV infections.
An immunochromatographic assay combines the immune response with chromatographic theory in a test that can also facilitate case diagnosis and disease management in remote areas where laboratory facilities are not readily available. This technique has been widely used in human medical science and animal medicine. 19,22,37 In the current study, an immunochromatographic strip was developed for the detection of swine antibodies and serological surveillance of PHEV infection.
Materials and methods
Virus strain
The virus strain used in the current study was PHEV-67N (GenBank accession no. AY078417). 24 The viruses were propagated and passaged in porcine kidney epithelial (PK)-15 cells, as reported previously, 10 and purified by sucrose density gradient centrifugation. The viruses were stored at −80°C until needed.
Recombinant protein
The materials and methods used for obtaining the hemagglutinin esterase (HE) protein of PHEV are described in detail elsewhere. 8 The gene encoding epitope-rich regions of HE protein of PHEV was cloned into the recombinant plasmid pET-HE and expressed in Escherichia coli. The expressed product, in the form of an inclusion body, was washed, lysed, and further purified by using affinity columns. a The identity of the protein was confirmed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and Western blot. The results showed that the expressed HE protein reached a protein content of 1.18 mg/ml and had a purity of 85.14% after purification. The protein was also recognized by monoclonal antibody against HE protein of PHEV. 10
Serum specimens
Four hundred and eighty-four serum samples were collected from sick pigs with suspected PHEV infection and used to evaluate the specificity and sensitivity of the immunochromatographic strips compared with HI. A total of 1,117 serum specimens were collected from approximately 1-month-old pigs that lacked the typical signs of PHEV infection, such as vomiting, exhaustion, and neurological signs. The samples were collected from 36 herds in the Changchun, Jilin, Baicheng, Songyuan, and Liaoyuan districts of Jilin Province, China, from 2009 to 2010. A total of 500 serum samples from pigs in different age groups were obtained from 8 pig farms with histories of PHEV infection in the Jilin Province in order to be tested by the immunochromatographic strip. All of the sera were heat inactivated at 56°C for 30 min and then stored at −70°C until needed.
Hemagglutination and hemagglutination inhibition test
The HA and HI tests were performed using a microtiter system as previously described. 30 All sera for the HI test were pretreated with a 25% (wt/vol) kaolin solution of equal volume at room temperature for 30 min in order to remove the nonspecific hemagglutination inhibitor. After centrifugation at 4,000 × g for 10 min, the supernatants were treated with 10% (vol/vol) chicken red blood cell suspension and an equal volume of phosphate buffered saline (PBS) solution for 30 min at room temperature. After the samples were centrifuged at 450 × g for 10 min, the treated sera samples were used for detecting the antibodies. The sera were diluted with PBS with a 2-fold step from 1:8 to 1:1,024. An HI titer of 8 or higher was recorded as positive according to established standards. 21
Enzyme-linked immunosorbent assay
The ELISA, which was based on HE recombinant antigens for detecting antibodies against PHEV, was performed as previously described 10 with some modifications. In brief, the 96-well polystyrene microtiter plates b were coated with purified HE protein diluted to a final concentration of 10 μg/ml in 50 mM of carbonate buffer (pH 9.2) at a volume of 100 μl per well. The plates were incubated overnight (16–18 hr) at 4°C. The plates were washed 3 times with PBS containing 0.1% Tween-20 (PBST). The remaining binding sites were blocked with 200 μl (per well) of 5% (wt/vol) bovine serum albumin (BSA) in PBST at 37°C for 2 hr. After the plates were washed, 100 μl of the serum samples was added per well. The standard serums for PHEV c and PBST diluted to 1:100 were used as positive and negative controls, respectively. The samples were held at 37°C for 1.5 hr. Horseradish peroxidase–labeled rabbit anti-pig IgG d (1:500 dilution) was added at a volume of 100 μl per well, and the plate was incubated for 1 hr at 37°C. The plates were washed 4 times with PBST, and color development was induced with the addition of the enzyme substrate o-phenylenediamine (OPD containing H2O2). The reaction was stopped by adding 50 μl of 2 M of sulfuric acid per well. The absorbance was read at optical density of 490 nm (OD490) with a universal microplate reader. e The ELISA detected 30 negative and 6 positive serum samples (Fig. 1A). The threshold value of 0.241 was evaluated using a receiver operating characteristic curve (Fig. 1B). An OD490 > 0.241 indicated a positive result and ≤0.241 indicated a negative result.
Preparation of colloidal gold and colloidal gold–labeled rabbit anti-pig IgG conjugate
Colloidal gold was prepared as previously reported 15 with minor modifications. In brief, 100 ml of 0.01% (wt/vol) chloroauric acid or gold chloride d in double-distilled water was poured into a 250-ml siliconized conical flask and heated to boiling in a microwave oven. Then, 1.4 ml of 1% trisodium citrate was added to the solution. After the colloidal gold solution was boiled for an additional 14 min, it turned a cardinal red color and was allowed to cool gradually. The pH of the solution was adjusted to 8.4 by using 1% potassium carbonate (wt/vol), and then the solution was stored at 4°C in a dark-colored glass bottle.
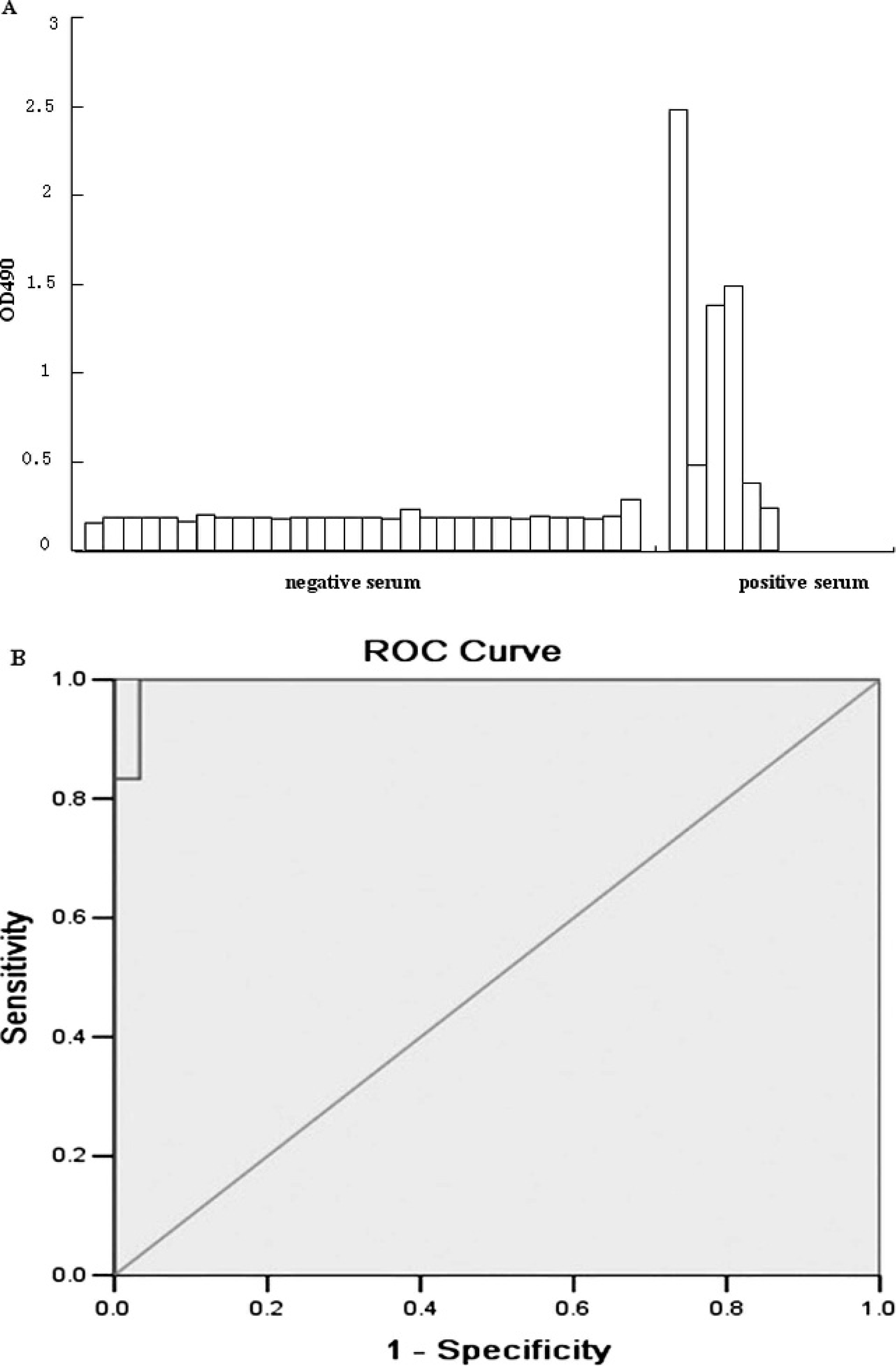
Antibody of porcine hemagglutinating encephalo-myelitis virus titers from the enzyme-linked immunosorbent assay (ELISA).
The rabbit anti-pig IgG (100 μl; 1 mg/ml) was slowly added dropwise into 10 ml of colloidal gold solution while mixing in a magnetic stirring apparatus for 30 min. Then, the solution was kept at 4°C for 30 min, and 1 ml of 10% (wt/vol) BSA was added to block excess reactivity of the gold colloid. The mixture was stirred on the magnetic stirring apparatus for an additional 30 min and stored at 4°C for 2 hr. After the mixture was centrifuged at 3,000 × g at 4°C for 30 min, the supernatant fluid was centrifuged at 14,000 × g at 4°C for 45 min, and the resulting conjugate pellet was suspended with 10 mM of borax buffer (pH 8.0) containing 2% (wt/vol) BSA and 0.02% sodium azide. The sizes and shapes of the unconjugated colloidal gold and colloidal gold conjugated to antibodies were characterized using transmission electron microscopy f measurements according to a standard procedure. 35
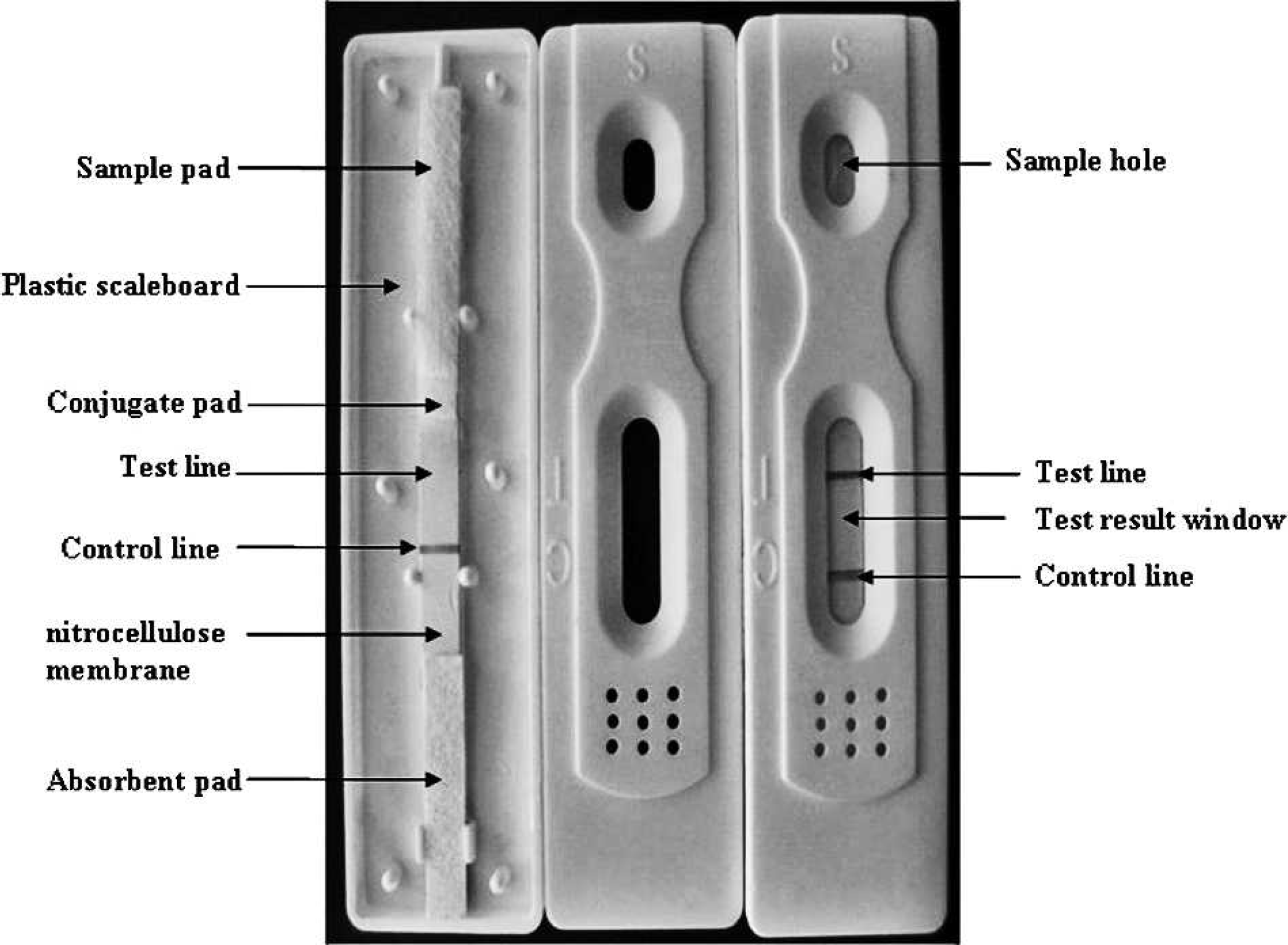
Schematic diagram of an immunochromatographic strip. The conjugate pad was coated with colloidal gold–labeled rabbit anti-pig immunoglobulin G (IgG). At the test line and control line positions, the purified hemagglutinin esterase proteins and the goat anti-rabbit IgG, respectively, were immobilized.
Preparation of the immunochromatographic strip
The immunochromatographic test device consisted of a plastic support attached to an immunochromatographic strip composed of a sample pad, g a conjugate pad, g a nitrocellulose (NC) membrane, g and an absorbent pad. g The sample pads and the conjugate pads were treated with 20 mM of phosphate buffer containing 2% BSA, 2.5% sucrose, 1% Tween-20, and 0.02% sodium azide (pH 7.4). The strips were dried under vacuum. The colloidal gold–labeled rabbit anti-pig IgG solution was dispensed onto glass fiber paper at a speed of 50 μl per cm using an automated dispenser. e The conjugate pad was then dried for 2 hr at 37°C and stored at 4°C. The purified HE proteins or the goat anti-rabbit antibody diluted in PBS was dispensed on the test or the control line on the NC membrane at a speed of 0.80 μl solution per cm using an automated dispenser. The membrane was dried for 2 hr at 37°C and stored at 4°C. The sample pad, pretreated conjugate pad, NC membrane, and absorbent pads were glued together on a support board and assembled into a test strip plate. The strip plate was then cut into 4-mm-wide pieces using a guillotine cutter. e In addition, a sample pad completed the assembly with 1.0–1.5 mm overlap mounted on the conjugate plastic card (Fig. 2). The dry strips were stored at 4°C until needed.
The principle and procedure of the immunochromatographic assay
During the assay process, 25 μl of undiluted serum sample was added to the specimen window of the assay device. The sample was allowed to migrate laterally and cover part of the membrane. For positive samples, rabbit anti-pig IgG through percolation in the NC membrane captured the pig IgG. For the test line, the formation of a colloidal gold–labeled rabbit anti-pig IgG–HE protein–pig IgG complex caused the appearance of a red line. For negative samples, the test line zone did not form a colloidal gold–labeled rabbit anti-pig IgG–HE protein–pig IgG complex, and therefore no red line appeared. As a control, both negative and positive samples caused the sample control line to turn red by forming a colloidal gold–labeled rabbit anti-pig IgG goat anti-rabbit IgG complex. The test strip is illustrated in Figure 3.
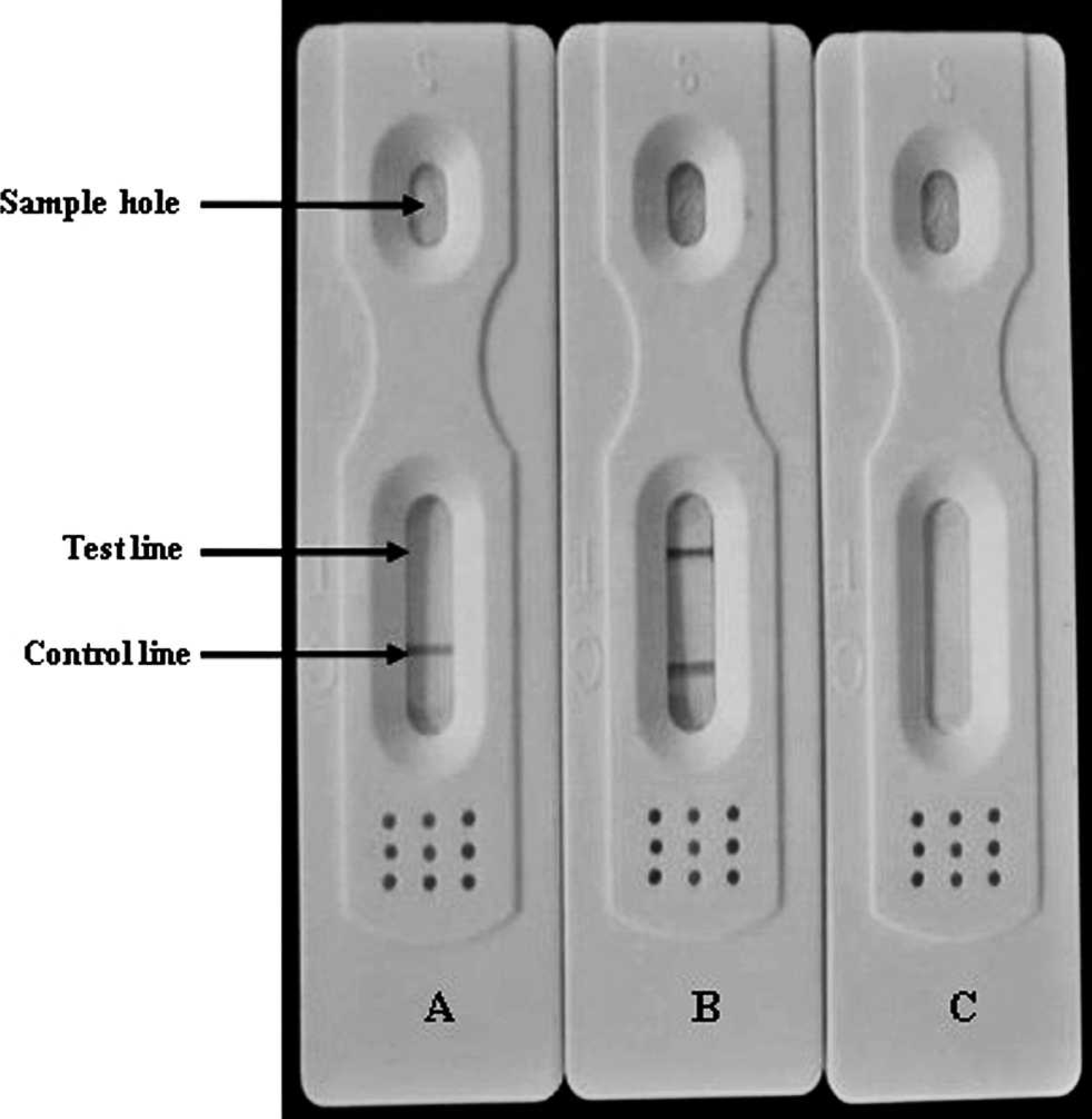
Interpretation of the results of the immunochromatographic strip test.
Specificity, sensitivity, reproducibility, and stability of the immunochromatographic strip
The serum sample of PHEV was diluted to an HI titer of 16 with 10 mmol/l of borate buffer solution (pH 8.0), and other serum samples, including Transmissible gastroenteritis virus (TGEV), c Porcine epidemic diarrhea virus (PEDV), c Hog cholera virus (HCV), h and Pseudorabies virus (PRV), h were simultaneously tested by using the immunochromatographic strips. In addition, G protein h from Vesicular stomatitis virus (VSV) Indiana serotype was expressed in E. coli and purified using affinity columns. a The purified G protein was dispensed on the test line of the immunochromatographic strips. To assure that the residual E. coli proteins generated sufficient background signals, the test strip detected the serum sample of PHEV. The serum sample with a PHEV HI titer at 256 was diluted to 1:2, 1:4, 1:8, 1:16, 1:32, 1:64, 1:128, 1:256, and 1:512 with 10 mmol/l borate buffer solution (pH 8.0). The dilutions were simultaneously tested using the immunochromatographic strips. To determine the reproducibility, the same batch and 5 different batches of the immunochromatographic strips were used to detect the positive serum samples with HI titers at 16. Testing of all of the samples was repeated 5 times. To determine the stability of the strips, immunochromatographic strips were stored at room temperature or at 4°C and used for testing positive and negative serum samples every 2 months.
Agreement between the immunochromatographic strip and reference methods
To evaluate the agreement between the immunochromatographic strip and reference methods, a total of 484 serum samples were collected from sick pigs with suspected PHEV infection and used to evaluate the specificity and sensitivity of the immunochromatographic strips compared with the HI and ELISA methods. The κ statistic 33 was used to measure the strength of agreement between the results from the immunochromatographic strip, HI, and ELISA. Kappa statistic values of 0.75, 0.40–0.75, and 0.40 represented strong agreement, good to fair agreement, and poor agreement, respectively. 27
Serological diagnosis of PHEV infection in the field
The immunochromatographic strip was applied in the field for serological diagnosis of PHEV. A total of 1,117 serum samples were collected from approximately 1-month-old pigs from 36 herds in the Changchun, Jilin, Baicheng, Songyuan, and Liaoyuan districts of Jilin Province, China, from 2009 to 2010. In addition, a total of 500 serum samples from pigs in different age groups (10-to 12-month-old brood sows and stock boars, 2-week-old pigs, 1-week-old pigs, and neonatal piglets) from 8 pig farms with histories of PHEV infection in Jilin Province were tested with the immunochromatographic strip.
Results
Colloidal gold and colloidal gold–labeled protein identification
The appearance of the colloidal gold solution was deep red and translucent. A bright band was visible when the test strip faced a light source. The colloidal gold particles were consistent in size and uniformly distributed with a mean diameter of 30 nm when observed under a transmission electron microscope. The colloidal gold–labeled rabbit anti-pig IgG was evenly distributed, and the particle size was consistent. The colloidal gold particles also had a visible clear space around the halo, the surface of proteins, and other adsorbed particles (Fig. 4).
Specificity and sensitivity of the immunochromatographic strip
To determine the specificity of the immunochromatographic strip, positive serum samples together with TGEV, HCV, PEDV, and PRV samples were characterized simultaneously with the test strip. Every sample resulted in 1 strong band on the control line, but the PHEV-positive serum displayed an additional band on the test line of the immunochromatographic strips. Notably, a negative result was observed in the test strip that was coated with G protein from VSV on the test line (Fig. 5). This result suggested that the immunochromatographic strip was able to detect PHEV antibodies specifically without any reactivity to other serum samples. Additionally, the residual E. coli proteins did not generate a sufficient background signal.
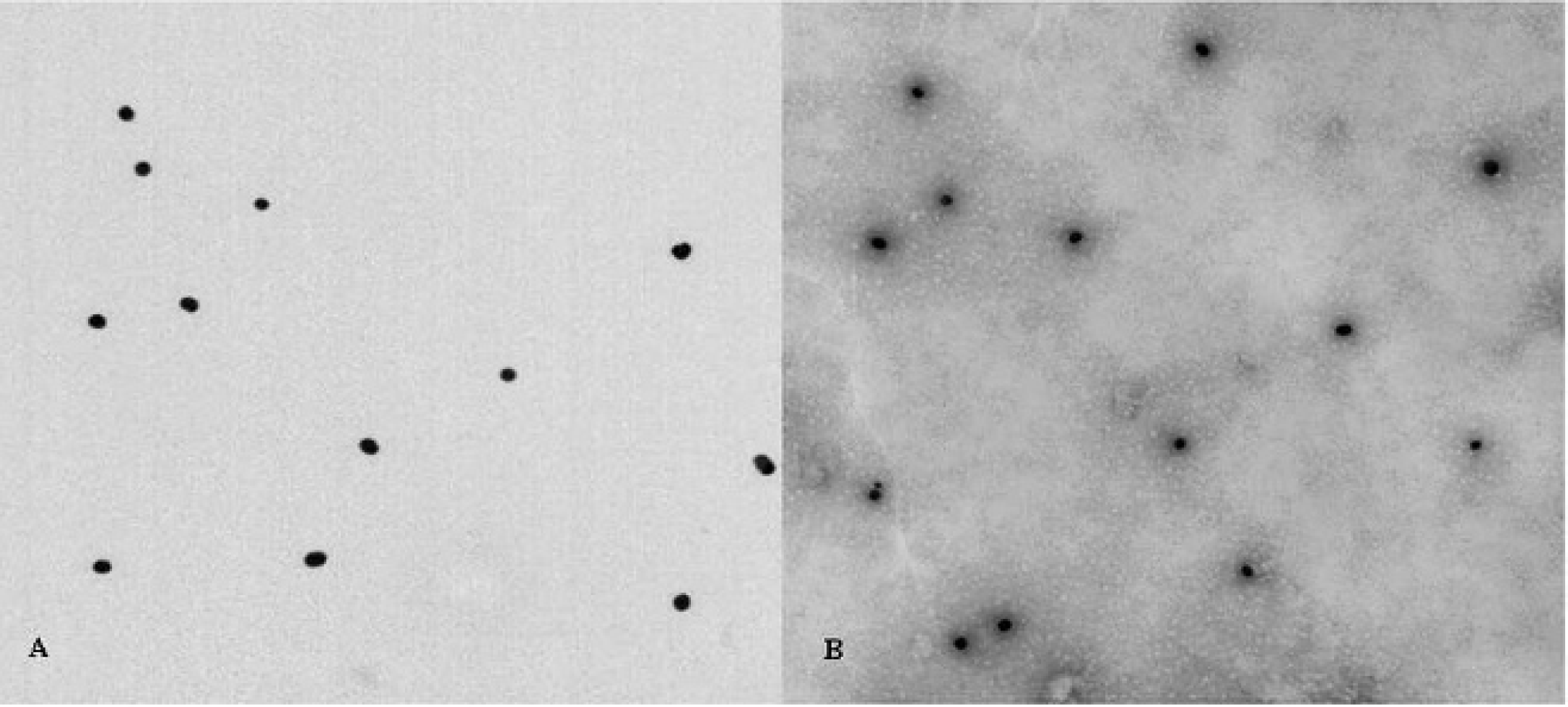
The result of transmission electron microscope observation of colloidal gold and colloidal gold–labeled rabbit anti-pig immunoglobulin G.
The sera of PHEV at a 28 titer, according to the HI test, were serially diluted and used to evaluate the sensitivity of the immunochromatographic strip (Fig. 6). When the serum samples were diluted from 1:2 to 1:128 (from 27 to 21 HI titers, respectively), 2 clear bands at the test and control lines were observed. Therefore, the sensitivity of the immunochromatographic strip reached a level of a 21 HI titer. Notably, the densities of the bands for various concentrations of HI antibodies on the test lines gradually declined from the strongest band at the 27 titer to a weaker band at the 21 titer, suggesting that detecting PHEV antibodies with this strip was dose dependent. Furthermore, similar results were observed by repeating the test of these samples 8 times, thereby indicating the high reproducibility of this experimental system. As such, using the immunochromatographic strip for the detection of PHEV antibodies was highly specific and sensitive as well as reproducible.
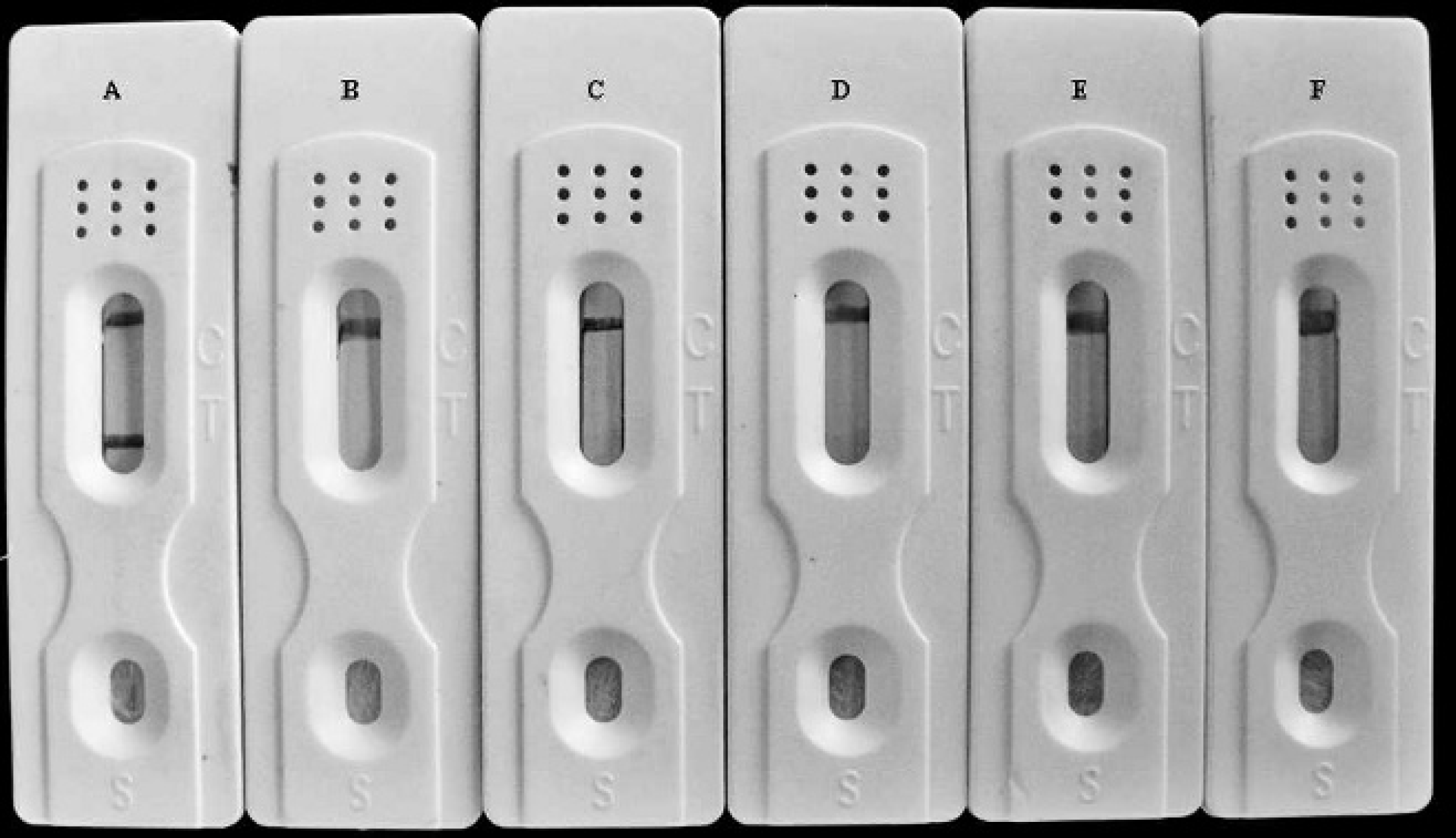
To determine the specificity of the immunochromatographic strips, positive serum Porcine hemagglutinating encephalomyelitis virus (PHEV), Porcine pseudorabies virus (PRV), Transmissible gastroenteritis virus (TGEV), Hog cholera virus (HCV), and Porcine epidemic diarrhea virus (PEDV) were characterized with the immunochromatographic strips.

The immunochromatographic strips displayed good sensitivity for the detection of porcine hemagglutinating encephalomyelitis virus antibodies: the immunochromatographic strips were still positive for 1:128 diluted Porcine hemagglutinating encephalomyelitis virus–positive sera (hemagglutinating inhibition, 28).
Reproducibility and stability of the immunochromatographic strip
This procedure was repeated 5 times to detect positive serum samples with an HI titer at 16 using the same batch and 5 different batches of the immunochromatographic strips to determine the reproducibility. As shown in Table 1, both the same batch and different batches had little variation in the detection of the positive serum samples with high reproducibility. In addition, the stability of the immunochromatographic strip under various storage conditions was determined. The immunochromatographic strips stored at 4°C for 12 months could detect 21 HI titers of PHEV antibodies, which was the same sensitivity level as the sensitivity for a strip that was freshly produced. In contrast, the strips stored at room temperature for 8 months displayed a 50% reduction in sensitivity, and the extension of the storage time to 12 months further decreased the sensitivity of the immunochromatographic strips. Notably, the specificity of the immunochromatographic strip for the detection of PHEV antibodies did not change under various storage conditions. Thus, the immunochromatographic strips can be stored at room temperature for 6 months with their sensitivity reduced by 50% and at 4°C for more than 12 months without any loss in sensitivity or specificity for the detection of PHEV antibodies.
Reproducibility of the immunochromatographic strips (ICS) for the detection of Porcine hemagglutinating encephalomyelitis virus antibodies.*
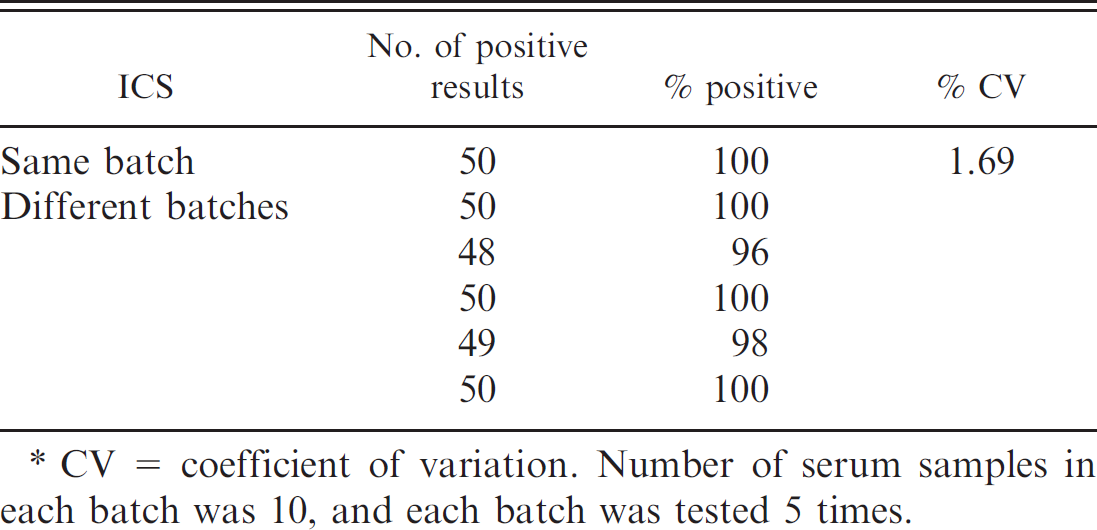
CV = coefficient of variation. Number of serum samples in each batch was 10, and each batch was tested 5 times.
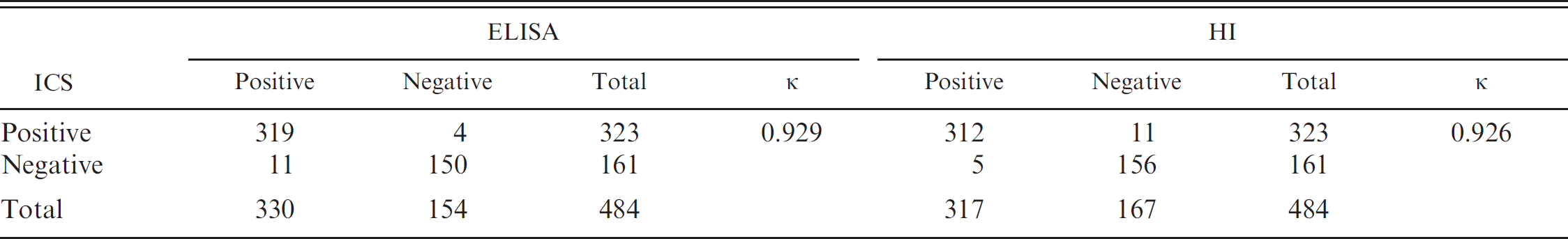
Agreement between the immunochromatographic strips (ICS), enzyme-linked immunosorbent assay (ELISA), and hemagglutination inhibition (HI) assay from testing clinical serum samples for the detection of Porcine hemagglutinating encephalomyelitis virus antibodies.
Agreement between the immunochromatographic strip and reference methods
To evaluate the agreement between the immunochromatographic strip and reference methods, the 484 serum samples collected from sick pigs were tested with the ELISA, HI, and immunochromatographic strips. The results are shown in Table 2. The specificity and sensitivity of the immunochromatographic strip, compared with HI, were 93.41% and 98.42%, respectively. There was a strong agreement (κ = 0.926) between the immunochromatographic strip and HI. A strong agreement (κ = 0.929) was also found between the ELISA and the immunochromatographic strip test results. Above all, the results suggested that the immunochromatographic strips had high sensitivity and specificity for detecting PHEV antibodies, which could be used for clinical applications.
Application of the immunochromatographic strip for serological diagnosis of PHEV in the field
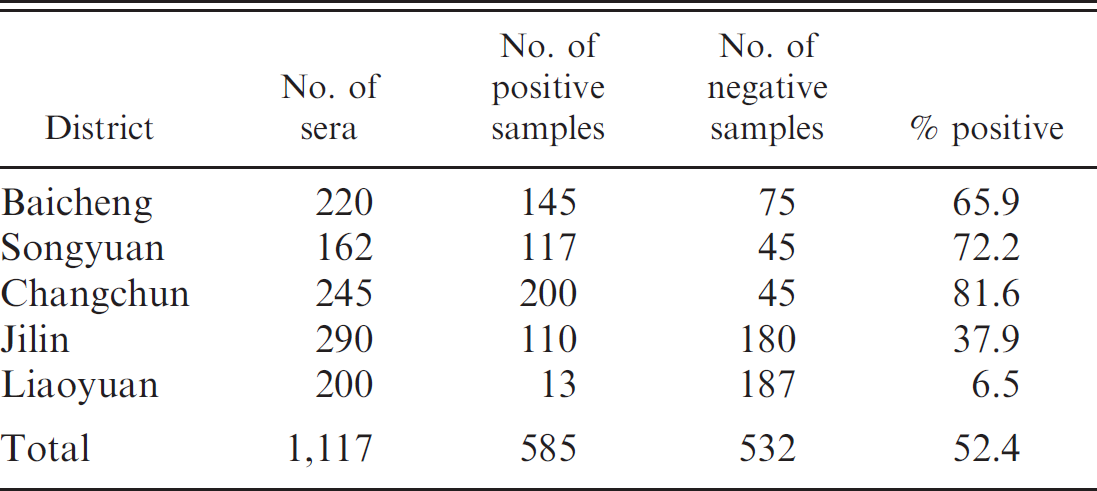
The immunochromatographic strip was used in the field for serological diagnosis of PHEV, and the results are shown in Table 3. A total of 585 out of 1,117 (52.4%) sera were positive for PHEV, and the seropositivity ranged from 6.5% in the Liaoyuan District to 81.6% in the Changchun District. Remarkably, 200 out of 245 (81.6%) sera in the Changchun District were positive, while 117 out of 162 (72.2%) sera were positive in the Songyuan District. In the Jilin District, PHEV antibodies were detected in 110 out of 290 (37.9%) sera. However, 8 farms in the Liaoyuan District exhibited seronegative results.
In addition, serum samples from pigs in different age groups were tested with the immunochromatographic strip, and the results are shown in Table 4. The PHEV antibodies were never detected in neonatal piglets, and the brood sows showed the highest seropositivity rate (78%), followed by the 2-week-old pigs (74%), stock boars (56%), and 1-week-old pigs (48%). Thus, many piglets were seropositive for PHEV, indicating the possible transfer of maternal antibodies via colostrum.
Discussion
Porcine hemagglutinating encephalomyelitis virus belongs to Group 2 of the family Coronaviridae, a virus group characterized by the presence of a gene encoding for the HE protein. 14 Nucleotide sequence analysis of the region covering the S2 probe revealed 92.6% nucleotide sequence homology to Bovine coronavirus (BCV) and 91.9% to Human respiratory coronavirus (HCV-OC43). 34 Moderate cross-reactivity was also observed between PHEV and Turkey enteric coronavirus (TEV). 11 Remarkably, although PHEV is known to be the cause of 2 distinct clinical syndromes in pigs, only 1 serotype of the virus is known to exist. In the past 30 years, with the exception of Taiwan Province, no serological survey of PHEV infection in pig herds has been conducted in Mainland China. However, during this period, outbreaks of disease caused by PHEV have been reported in China, including Jilin Province. 12,18 Currently, antibodies to PHEV are detected using the HI test and the VN test, which are almost equally sensitive. 23 However, these assays usually require a laboratory, skilled technicians, and specialized equipment, which makes these methods difficult to use for rapid and on-site detection of the virus in the field. Therefore, the development of a sensitive, specific, and easily performed assay is crucial for the rapid detection and surveillance of PHEV antibodies.
Testing the immunochromatographic strips for serological diagnosis of Porcine hemagglutinating encephalomyelitis virus in Jilin Province, China.
Detection of Porcine hemagglutinating encephalomyelitis virus antibodies among different age groups of pigs using the immunochromatographic strips.*
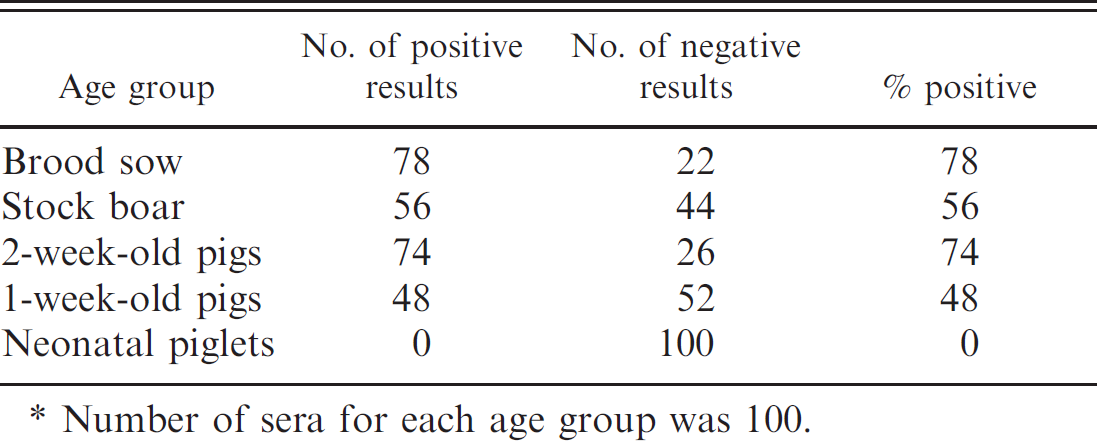
Number of sera for each age group was 100.
A colloidal gold immunochromatographic assay (GICA) is a solid-phase immunoassay developed in the 1980s that combines the techniques of colloidal gold labeling, chromatographic analysis, immunodetection, and other methods. 36 Because of GICA's convenience and speed as well as its specificity, sensitivity, and the ability to use the test without instruments or with only a simple instrument, it is suitable for clinical diagnosis and drug testing purposes at any location. 4,5 Most proteins and antibodies can be easily coupled to colloidal gold particles. In the current study, rabbit anti-pig IgG was used with gold particles as a detector reagent. Such antibodies d have a high affinity for their antigen epitopes. In addition, the key to successful colloidal gold labeling was the preparation of a homogeneous mixture containing monodispersed colloidal gold particles. 9 The microwave oven method was chosen in the current experiment because it provides even heating without manual shaking. As a result, the colloidal gold particles were consistent in size and uniformly distributed.
The specificity and sensitivity of the immunochromatographic strips were largely dependent on the antigen used in the test strip. In the current study, the HE protein of PHEV was used as detection antigen since the HE protein can induce antibodies and neutralize viral infection. Also, other coronaviruses in pigs, such as TGEV and PEDV, do not have the HE protein. Analysis of the strip specificity showed that the strip was specific for the detection of PHEV antibodies and did not cross-react with TGEV and PEDV or with other common viruses (HCV and PRV) that can produce similar clinical signs of disease. In addition, the choice of a membrane was critical for high specificity, sensitivity, and rapid detection of PHEV since the wicking rate and the speed of liquid diffusion on the membrane are key characteristics for the suitability of membranes. 37 The membrane used in the present experiment was an NC membrane g with a membrane pore size of 0.5–1.0 μm, an IgG binding force constant of 60–100 μg/cm2, and a chromatography rate of 15–20 sec/cm. The membrane ensured antibody adsorption as well as the leaching and binding of gold–labeled proteins. These advantages enabled the test results to be sensitive and easy to read.
Furthermore, the quantifying test results indicated that the sensitivity of the strip test was close to or slightly less than the sensitivity of ELISA. Taken together, an immunochromatographic strip for the detection of PHEV antibodies was successfully developed that specifically detected PHEV antibodies from serum with an HI titer of 2 within 10 min. Although ELISA has been developed to detect PHEV with high specificity and sensitivity, the test is not suitable for use outside of a research laboratory. In contrast, the immunochromatographic strip described in the current study was less laborious and time-consuming than other methods. Thus, the immunochromatographic strip test was capable of detecting PHEV antibodies in the field.
In addition, using the immunochromatographic strip for serological diagnosis of PHEV infection in Jilin Province, the seropositivity ranged from 6.5% in the Liaoyuan District to 81.6% in the Changchun District. Thus, PHEV infection, while prevalent, might be asymptomatic in pigs. Furthermore, seropositivity to PHEV in pigs of different age groups that were tested with the immunochromatographic strip indicates that many piglets might have received maternal antibodies against PHEV through the colostrum, which can protect against PHEV infection and disease. Notably, specific pathogen-free pigs derived from germ-free pigs that were given artificial milk have been introduced extensively in many farms. 1,29 Since these animals lack antibodies to certain pathogenic agents, including PHEV, the risk of infection by these agents may be greater. Therefore, there is a need for testing methods for the diagnosis and prevention of PHEV, which can cause fatal outbreaks on PHEV-seronegative farms.
In summary, an immunochromatographic strip was successfully developed for serological diagnosis of PHEV. The immunochromatographic strip is a simple and quick test that requires no special training to use, and the sensitivity and specificity of the strip were similar to the ELISA test. This test would be a valuable addition to clinical detection techniques for PHEV antibodies, especially in areas where laboratory facilities are not available.
Acknowledgements
The authors are extremely grateful to the staff working in the veterinary stations in the Jilin Province for collecting porcine sera for this study. The National Natural Science Foundation of China (31072134, 30871849, 30671551), the Young Scientist Program of Science and Technology Development Plan in the Jilin Province (20090154), and the International Cooperation Program of Science and Technology Development Plan in the Jilin Province (20080722) supported the study. Keyan Chen and Wenqi He contributed equally to the manuscript.
Footnotes
a.
HisTrap™ HP affinity columns, Amersham Biosciences Corp., Piscataway, NJ.
b.
Costar Corning Inc., Corning, NY.
c.
State Key Laboratory of Molecular Pathology and Immunopathology, Jilin University, Changchun, P. R. China.
d.
Sigma-Aldrich Inc., St. Louis, MO.
e.
XYZ3000 Dispense Workstation, CM4000 guillotine cutter; Beckman Coulter Inc., Fullerton, CA.
f.
JEM-1200EX transmission electron microscope, JEOL Ltd., Tokyo, Japan.
g.
SHF135, Millipore Inc., Billerica, MA.
h.
State Key Laboratory of Zoonosis, Ministry of Education, Institute of Zoonosis and Animal Research Center, Jilin University, Changchun, P. R. China.
i.
SPSS Inc., Chicago, IL.
