Abstract
Bacterial motility in response to chemicals, also called bacterial chemotaxis, is a critical ability to search for the optimal environment to ensure the survival of bacterial species. Recent advances in microbiology have allowed the engineering of bacterial chemotactic properties. Conventional methods for characterizing bacterial motility are not able to fully monitor chemotactic behavior. Developments in microfluidic technology have enabled the designing of new experimental protocols in which spatiotemporal control of the cellular microenvironment can be achieved, and in which bacterial motility can be precisely and quantitatively measured and compared. This review provides an overview of recent developments of and new insights into microfluidic systems for chemotaxis assay.
Introduction
The activity of bacterial flagella for determining the direction of swimming is regulated by signaling pathways of biomolecular motor systems, wherein the chemical source acts as the chemoeffector, and the process is called bacterial chemotaxis. 1 Chemoreceptors expressed on a bacterial surface can sense the chemical gradient to direct the move toward high or low concentrations of the chemoattractant or chemorepellent, respectively. Most of the chemotactic behavior of bacteria is closely associated with marine microbial food webs, metabolism, and disease pathogenesis. Specifically, chemotaxis of Pseudomonas aeruginosa, an opportunistic pathogen, plays a critical role in biofilm formation and invasive infections. 2 The analysis of chemotaxis in such cases via a rapid, effective, and high-performance method is needed because of the existence of many bacterial strains and their chemoeffectors. Compared with the conventional chemotaxis assays, a microfluidic device is a promising experimental assay platform for the monitoring and quantitative evaluation of cell migration.3–5 The microfluidic device can be advantageous due to its low cost, rapid assay time, better environmental control, and enhanced sensitivity. The high sensitivity of this device compared with conventional methods was demonstrated two decades ago. 6 At present, the development of microfluidics for chemotaxis assay is focused on user-friendly operation, enhancing assay ability, and the precise control of gradient shapes. This review provides a brief summary of recent advances in microfluidic-based bacterial chemotaxis assay.
Bacterial Chemotaxis Signal Transduction
Specific receptors on the bacterial surface are able to recognize the attractant and repellent chemoeffectors.
7
This signal is transferred via several molecules to the flagella motors to rotate either clockwise (CW) for straight runs or counterclockwise (CCW) for tumbling motion (
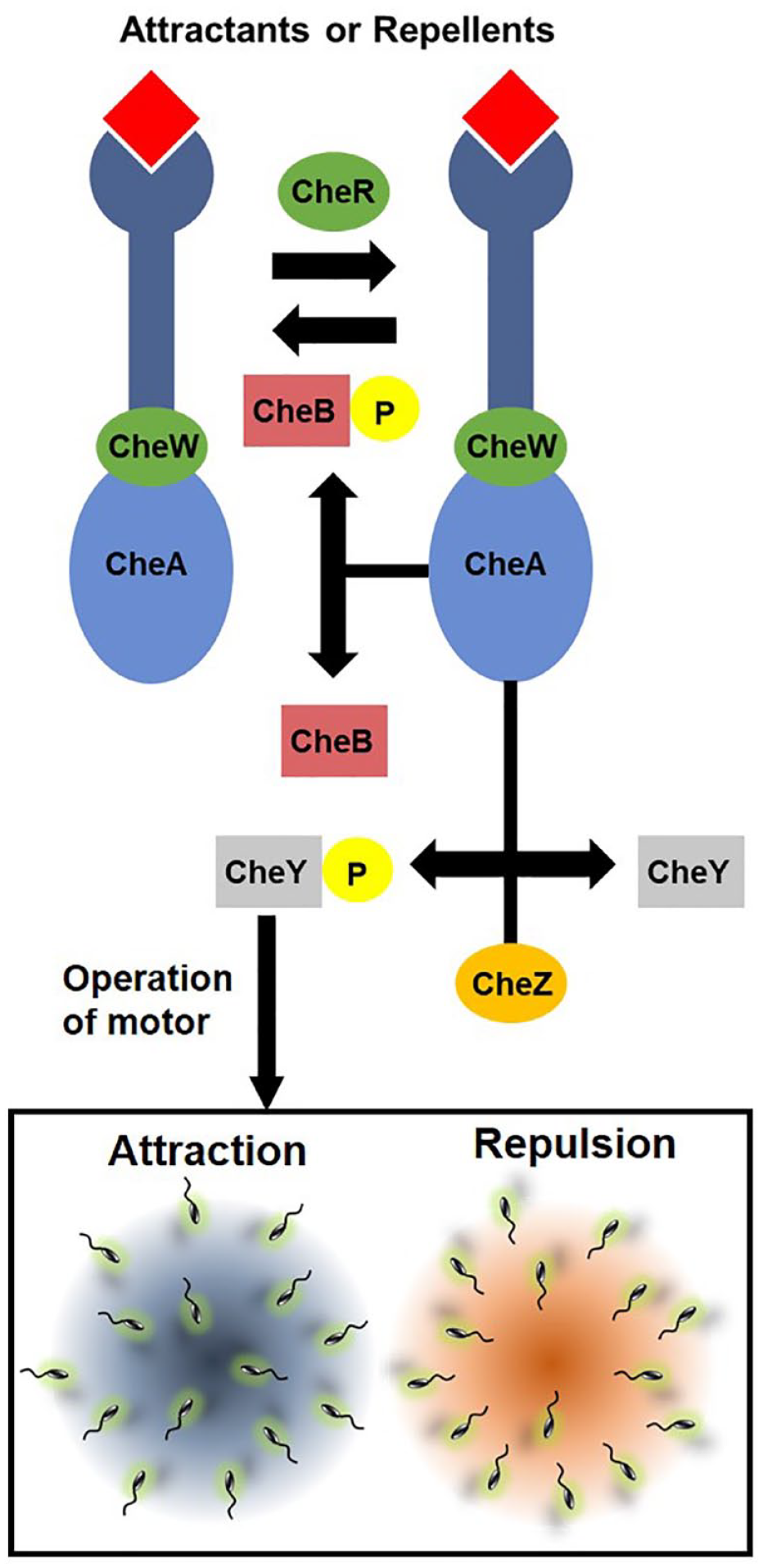
Schematic representation of bacterial signaling and chemotaxis. Depending on the activation of Che proteins, bacteria can move toward high concentrations of the attractant or move away from high concentrations of the repellent.
Methods for Chemical Gradient Generation
The chemical concentration gradient is essential for inducing bacterial chemotaxis. Thus, understanding the fluid dynamics of such a behavior in a microfluidic system is critical. In fluid dynamics, there are two fluid regimes, laminar or turbulent, defined by the Reynolds number (Re, ρUL/μ), a dimensionless parameter defined by the relative contribution of inertial and viscous forces in a channel. The parameters determining the Reynolds number are the density (ρ) and viscosity (μ) of the fluid, velocity (U) of fluid flow, and characteristic length of the channel (L). The major fluid regime in a microfluidic channel is laminar flow, having smooth layer-by-layer movement without turbulent mixing, due to the very low Reynolds number (Re < 2100) resulting from small channel dimensions. As the Reynolds number is used for determining whether the flow regime is laminar or turbulent, the Peclet number (Pé, UL/D) can be used to characterize whether the gradient generation is based on diffusion or convection; here, D is the molecular diffusivity. Dominant mass transport is by convection when Pé >> 1 or by diffusion when Pé << 1. In a flow-based system, the gradient is generated by both convection and diffusion, whereas gradient generation in a flow-free system is based only on diffusion.
One of the key functions of a chemotaxis assay device is the generation of a stable concentration gradient. Conventional methods for the analysis of bacterial chemotaxis, including the Boyden chamber, Zigmond chamber, Dunn chamber, and pipette injection, can determine which chemoeffectors play the role of chemoattractants or chemorepellents under the experimental conditions.8–10 However, inaccurate microscale control of chemical gradient and fluctuations of the chemoeffector solution pose a challenge in obtaining reliable quantification of bacterial chemotaxis.
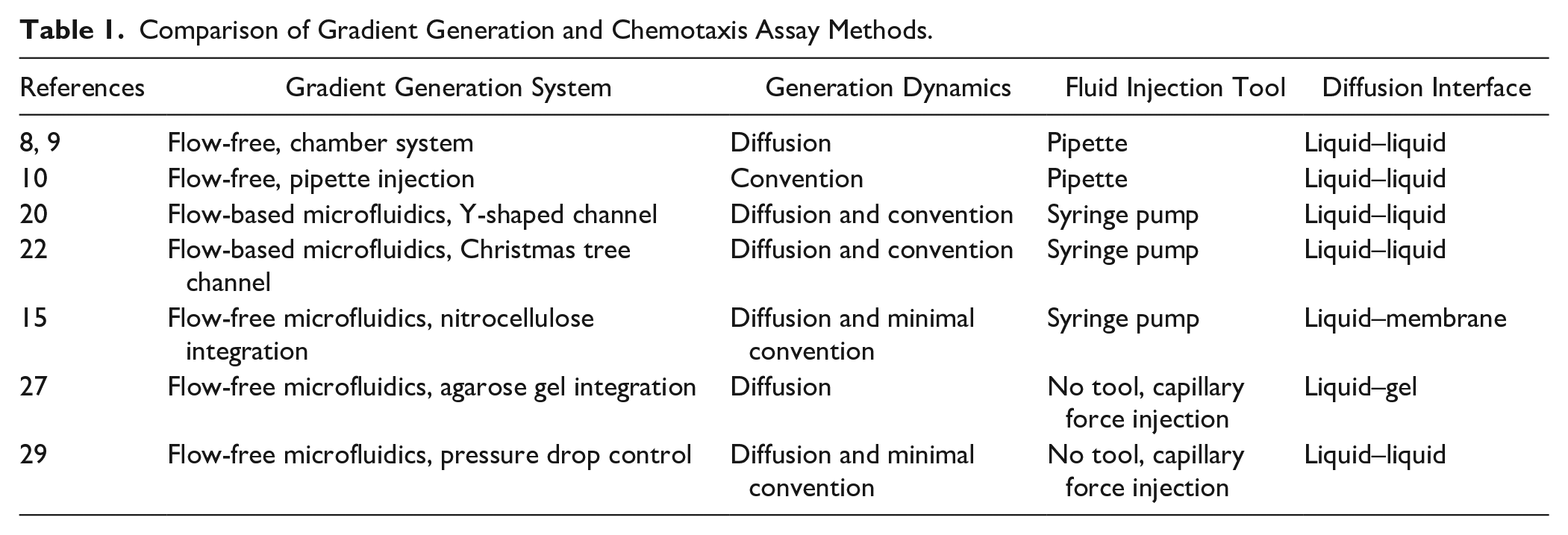
The microfluidic system was developed over 20 years ago to translate conventional bulk models into miniaturized systems in diverse areas, including chemical and biological experiments.11,12 The rapid acceptance of miniaturized microfluidic systems has largely been driven by the advances in various cell-based assays, including direct and indirect interactions, the release and reception of soluble factors, and the transmission of electrical signals. 13 Microfluidic systems provide significant advantages for these research fields, such as the precise control of microscale structures and fluid flow, as well as spatiotemporal control of cells, chemical concentration, and temperature or oxygen gradients. 14 In addition, the ability to combine multiple chemical and physical signals and the cost-effectiveness of using small volumes enhance the benefits of microfluidic devices. Due to its numerous advantages, the microfluidic system is suitable for the precise generation and control of concentration gradient for quantitative chemotaxis experiments. A comparison of conventional and microfluidic approaches for gradient generation and chemotaxis analysis is given in Table 1. Recently developed microfluidic-based chemotaxis assays can be categorized into flow-based and flow-free systems, both of which have challenges that need to be overcome with respect to their limitations. The key benefits of using microfluidic systems for bacterial chemotaxis assay include enhanced analytical performance and reduced operation times and instrument footprints compared with conventional methods. Importantly, in the aspect of analytical performance, the microfluidic device is especially helpful for generating one-, two-, and three-dimensional (3D) chemical gradients. Most microfluidic devices used for chemotaxis assay use the one-dimensional gradient that is formed in a straight channel using source and sink solutions. 15 More complex assays are possible in a two-dimensional plane gradient by precisely controlling chemical diffusion using microfluidic devices.16,17 Multiple gradients using more than two chemoeffectors can be generated and superimposed to perform bacterial chemotaxis assays with more complex and dynamic environments. For the generation of 3D gradients, 3D hydrogels are incorporated into the microfluidic channel, which enables the user to control chemical diffusion from the source/sink microchannels. The 3D environment gradient is usually applied to study adherent cell chemotaxis.18,19
Comparison of Gradient Generation and Chemotaxis Assay Methods.
Flow-Based Microfluidic System
The initial microfluidic system, designed in 2003, for bacterial chemotaxis assay with Y-shaped microchannels was a flow-based system.
20
This design is composed of three inlets; the bacterial solution is introduced into the middle inlet, and the chemoeffector and buffer solution are introduced into the upper and lower inlets, respectively. A chemoeffector concentration gradient is formed perpendicular to the direction of fluid flow in the gradient channel. At the end of the gradient channel, cells that move perpendicularly, depending on the gradient, enter one of the 22 outlets. Thus, bacterial chemotaxis behavior in response to chemoeffector concentration can be quantified by measuring the bacterial density within each channel. Using this flow-based microfluidic device, Mao et al. found that Escherichia coli is attracted to low concentrations of
In contrast to these simple designs, another approach aims at further realizing the potential of microfluidic devices for flexible gradient generation. As an example, a highly branched design, called the Christmas tree channel, was developed to generate a gradient that is more precisely controllable compared with the simple Y-shaped channel, and was first reported by Jeon et al. in 2000. 21 The buffer encounters the chemoeffector solution at the first branched channel and gets fully mixed while passing through the curved channel. Then, the mixed solution is transported to the next branched channel. After passing through several branched channels, parallel laminar flows with different concentrations are generated in a single main channel that joins all the branched channels. Compared with the simple Y-shaped channel, rapid and accurate generation of gradient with high linearity was possible by diffusional mixing of serially diluted solution in the microfluidic channel. Depending on the number of branched channels, the concentration gradient profile can be changed, and a bacterial chemotaxis experiment can be conducted under various conditions.
In 2009, Englert et al. applied the Christmas tree design for generating the intricate gradient for bacterial chemotaxis assay. 22 In this design, bacteria samples can be directly injected into the main channel by combining Y-shaped microchannels, thus preventing sudden exposure to high concentrations of chemicals. Lamanna et al. previously reported that sudden, large increments in attractant concentrations may disrupt the localization of chemoreceptors and inhibit signal transduction. 23 Roggo et al. designed filter channels (0.6 µm) between the detection channel and the source/sink channels to allow chemical diffusion but prevent the passage of cells and sudden exposure to high concentrations of chemicals. 24 They applied a lower flow rate for the bacterial solution than the source and sink channel flow rates, which minimized the disturbance of bacterial motility by flow. When chemoeffectors are slowly transported to the bacteria, movement of bacteria in response to chemoeffectors can be observed downstream of the detection channel. Thus, this ensures that the chemotaxis assay accuracy is improved, by providing enough time for the bacteria to respond to the concentration gradient. In addition, the high sensitivity in detecting chemotactic behavior in microfluidics enables users to accurately determine an effective response to a broad range of chemoeffector concentrations by comparing linear gradient profiles, which is the key advantage over the conventional endpoint assay methods. 25
However, since the concentration gradient profile can be easily altered by parameters related to the rate of diffusion (e.g., concentration difference, temperature, and viscosity), flow rate, bacterial motility, and prediction of the gradient profile are very complex. The lack of a consistent concentration gradient profile from inlet to outlet may lead to inefficient and inaccurate analysis, which is not suitable for the assay of bacterial chemotaxis indicating a sensitive response to temporal changes in chemical gradient. Additionally, the influence of bacterial motility by flow has been another challenge.
Flow-Free Microfluidic System
A diffusion-based chemical gradient in flow-free microfluidics could overcome the issues of flow-based systems. The gradient generation in a flow-free system is based only on the diffusion from a high concentration to a low concentration. This device does not affect bacterial motility and is more suitable for bacterial chemotaxis assays as it provides sufficient time to respond to the chemical gradient due to the slow diffusion rate. Although conventional chemotaxis assay methods were based on a static flow-free system, more efficient and accurate devices have been developed by integrating microfluidics. In this system, the integration of porous materials, such as membranes and hydrogel, plays an important role in generating the chemical gradient without fluid convection. Since the pore size of physical barriers is sufficiently small to prevent bacterial movement, only signal molecules can pass through the barrier to the cells that receive the signal. The fluid channels are composed of sink and source channels, with the porous barrier positioned between them. Two different chemical gradient profiles are possible by integrating the hydrogel system in the microfluidic device, namely, linear and nonlinear gradient profiles.
Diao et al. showed the generation of a static linear gradient using a nitrocellulose membrane-based microfluidic device and demonstrated it for bacterial chemotaxis assay.
15
They designed three parallel channels, containing two outer channels as the source and sink channels, and the central channel as the gradient channel. The chemoeffectors were loaded into one of the outer channels (source channel), while the blank fluid was loaded into the other outer channel (sink channel) to remove the chemoeffectors. A linear concentration gradient was confirmed using fluorescein dye, which was formed for about 20 min. This linear profile was highly sensitive to the flow rate of sink and source fluids. Based on this gradient profile, they found that wild-type E. coli RP437 cells move toward high concentrations of the attractant (10−4 M
As another example of this approach, Si et al. also developed an agarose-based assay device with eight parallel channels, operated using a pipette. 27 First, agarose gels were selectively formed by filling agarose solution in 20 parallel narrow channels (5 μm width × 300 μm length) connected to the source channel. Linear gradient generation and chemotaxis assay were performed in an observation channel that was connected to the sink channel. Compared with the previous method, this pumpless parallel system was convenient and could yield more chemotactic data from a single experiment. Berendsen et al. applied the 3D printing technique to fabricate a hydrogel-based microfluidic device using a double-casting method. A mold with positive patterns was 3D-printed, and a negative polydimethylsiloxane (PDMS) mold was fabricated. Based on this mold, the hybrid hydrogel (8% gelatin and 1% agarose) microfluidic device was fabricated, which was able to generate a stable chemical gradient. 28
There are, however, several limitations of membrane-based devices. The hydrogel or porous membrane-based diffusion generator poses various challenges, including complex procedures for forming the porous structure, a nonreproducible chemical gradient in heterogeneous pore size distributions, and the need for a relatively longer time to form a linear chemical gradient. The slow diffusion rate might also result in different chemical gradients with unexpected results due to the mixing of chemicals by the motility of high-density bacteria.
To develop more convenient and easy-to-use microfluidic devices for bacterial chemotaxis assays, hydrogel-free and pumpless systems have been developed.29,30 The basic principle for generating a gradient without hydrogel and pump is the enhancement of pressure drop by a liquid–gas interface. In such a system, a chemoeffector solution is injected, driven by capillary force, and stopped by surface tension at the junction channel at a different height. Subsequently, a bacterial solution is injected, which comes in direct contact with the chemoeffector, forming a stable liquid–liquid interface. A linear chemical gradient is formed after the contact of the two solutions. Dominant mass is transport in this method by diffusion and convection. However, convection is greatly minimized because of the difference in hydrodynamic pressure as the balance of liquid volume in each reservoir is reduced by the high pressure drop. In the P. aeruginosa chemotaxis assay, a binary dynamic motion was observed, which showed attraction and repulsion to a single chemoeffecor. 29 When the initial injection concentration of methyl-Asp was 10 mM, the bacterial population increased until 5.7 min. However, after this, the bacterial population decreased rapidly in the detection area, in response to the chemoeffector concentration. The switching threshold concentration of the chemoeffector (8.77 mM methyl-Asp) was the critical factor for binary dynamic motion of P. aeruginosa chemotaxis. Direct and real-time monitoring of attraction and repulsion by only one chemoeffector has not been reported using conventional methods.
In another example, a device consisting of two glass plates with microwells and ducts was designed. The sample solutions were loaded into separated nanoliter microwells. These microwells were interconnected by an assembly of ducts when the two glass plates were aligned by a simple slipping operation. Chemical diffusion through the duct resulted in the generation of concentration gradients.31,32 Coluccio et al. developed a microfluidic device composed of three layers, including solution reservoir, microfluidic channels, and alignment/observation layers. 33 The concentration gradient was formed in the transverse channels that were designed between the cell and the medium channels. Here, the working principle for gradient formation was guided by the precise control of differences in channel resistance between the cell and the medium channels.
Conclusions and Future Perspectives
Microfluidic devices use a variety of designs and materials that can provide flexibility for the user and facilitate new findings in biological research. The emerging microfluidic technology exhibits great potential for exploring complex biological systems by changing the cellular microenvironment. This article reviewed recent developments in flow-based and flow-free microfluidic chemotaxis assays. Many previous reports have demonstrated that both methods can generate linear and stable concentration gradients. Since both gradient generation systems have various advantages and disadvantages, a particular gradient system must be selected depending on the experimental requirements. For example, the flow-based microfluidic technique can maintain a constant chemical gradient for a long time at specific regions and is suitable for long-term monitoring of chemotaxis in adhesive cells or an antibiotic response of biofilms. 34 Recently, the development of microfluidics for bacterial chemotaxis assays has focused more on the flow-free systems, which have critical advantages for users, including no requirement of an external pump and easy operation by simple pipetting. With advances in the design of microfluidic systems, the advent of 3D printing and its use for simple fabrication of this device for commercial application are extremely exciting developments. In addition to the bacterial chemotaxis, a novel aspect of research on microscale motion engineering has been addressed using bacterial flagella, also called biological motors. For instance, attaching bacteria to the surface of a microfluidic channel can increase the effective diffusion coefficient, resulting in enhanced fluid mixing. These bacterial motors could be enhanced by controlling the pattern of attached bacteria, use of chemoeffectors, and external magnetic fields.35–37 A combined effort to develop the usability and capabilities of microfluidic systems for bacterial application will continue to expand this field of research.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (No. NRF-2020R1C1C1005505).
