Abstract
Point-of-care (POC) microfluidic devices often lack the integration of common sample preparation steps, such as preconcentration, which can limit their utility in the field. In this technology brief, we describe a system that combines the necessary sample preparation methods to perform sample-to-result analysis of large-volume (20 mL) biopsy model samples with staining of captured cells. Our platform combines centrifugal-paper microfluidic filtration and an analysis system to process large, dilute biological samples. Utilizing commercialization-friendly manufacturing methods and materials, yielding a sample throughput of 20 mL/min, and allowing for on-chip staining and imaging bring together a practical, yet powerful approach to microfluidic diagnostics of large, dilute samples.
Introduction
The interdisciplinary field of microfluidics examines fluid flows in microchannels and has been used practically to develop systems for high-throughput screening, multiplexing, and automation. A wide variety of microfluidic technologies have been developed, such as centrifugal systems, which use centrifugal forces to manipulate fluid behavior, 1 and paper-based systems, which take advantage of the small capillary network in the paper matrix to filter and move fluid. 2 Microfluidic point-of-care (POC) devices have been well established as alternatives to standard diagnostic tests for their low cost, reduced sample and reagent consumption, rapid speed, small footprint, and no requirement for trained technicians or dedicated facilities. 3 These advantages of microfluidics have been widely applied to small sample analysis, yet studies have been lacking for analysis of larger-volume samples.4–6 The necessity of techniques to analyze large samples arises as ultra-rare cell types, such as circulating tumor cells, 7 and dilute biological samples, such as vitreous humor aspirates, 8 require additional sample preparation steps before analysis in a microfluidic device. Commonly, samples must be concentrated via centrifugation or filtration off-chip before a microfluidic device can be used. For example, red blood cell removal is necessary to improve capture of circulating tumor cells in peripheral blood, 9 and preconcentration of invading immune cells in ocular biopsy fluid is necessary for diagnostic analysis. 10 A major barrier to commercialization of point-of-care microfluidic devices is the lack of integration of sample preparation steps in microfluidic methods. 11 Recent work by Kloke et al. has developed an integrated POC sample preparation system using a modified standard centrifuge tube capable of reagent addition, mixing, and solid-phase extraction; however, processed samples still require additional analysis afterward and sample volume is limited. 12
A clinical example that could benefit from large-volume, integrated sample preparation microfluidics is the diagnosis of idiopathic eye diseases such as uveitis and primary intraocular lymphoma (PIOL). Uveitis is the general term for inflammation within the uvea of the eye and can be caused by a wide variety of ailments, such as infection or autoimmune disease. 13 On the other hand, PIOL is a type of cancer often related to lymphoma somewhere else within the primary nervous system. 14 Uveitis and PIOL are ocular diseases with similar symptoms, but very different prognoses. Diagnosis is achieved by immunophenotying invasive cell types in the vitreous humor of the eye. The gel-like fluid of the vitreous humor (approximately 4 mL) is extracted by aspiration using an irrigation solution known as BSS-plus, which produces a 50 to 100+ mL sample with fluidic properties similar to those of salt water. When a disease is present, the biopsy sample contains between 0 and 100,000 cells in total. 15 These idiopathic eye diseases can be categorized based on the majority of invasive cell type, typically T-cell predominance for uveitis and B-cell predominance for PIOL. While immunoassays quantifying ocular cytokine concentration and polymerase chain reaction testing for pathogens may offer some diagnostic information, the gold standard for diagnosing these diseases is flow cytometric analysis of the cells found in the biopsy. However, the reported diagnostic yield for flow cytometry is around 20% as cells ex vivo are fragile and undergo morphological changes within 60 min.16,17 There are currently no rapid tests for these diseases and no other cellular analysis tools used clinically besides flow cytometry for immunophenotyping. POC analysis of these samples would provide a higher yield of conclusive diagnostic results; however, a successful platform requires integration of preconcentration, antibody staining, and ability to directly image the cellular content.
The use of paper in microfluidics has been a widely discussed topic due to its low cost, ease of use, and strong commercialization potential. 18 Integration of filter paper into microfluidic devices has been shown to be a powerful method for sample separation.19–21 The major drawback of current systems is the inability to process large volumes. The throughput of recent reports on continuous-flow microfluidic filtration of biological samples is on the order of 1 mL/h. 22 To overcome this barrier, an “open” system alternative must be considered to allow rapid fluid analysis through a microfluidic device. Karle et al. have developed one of the first open centrifugal microfluidic filtration systems using high speeds (1000g) and low volumes (500 μL) per channel utilizing axial filtration. 23 This open system provides promise for larger-volume samples; however, its high speed is too strong for cells and relatively low volume still would require preconcentration. The Spinning Ocular Lymphocyte Immunotyping Diagnostic (SOLID) device described below provides a proof of concept of an integrated centrifugal-paper microfluidic filtration and analysis system for large, dilute biological samples. SOLID is ideal for clinical applications with large sample volume requirements.
Materials and Methods
Device and Setup Manufacturing
The rotating platform is composed of a custom-built aluminum stand with an integrated EC 90 flat 90 mm diameter, brushless, 90 W motor with hall sensors (no. 323772, Maxon Motor, Sachseln, Switzerland) and ESCON 50/5, 4-Q servocontroller (no. 409510, Maxon Motor). Depressions on top of the spinning aluminum stand allow a vacuum seal to form between the device and the stand, connected directly to a 230 VAC vacuum gas pump (no. 8907C-02, VWR, Radnor, PA). A custom, Plexiglas waste collection chamber is placed on top of the aluminum stand. Finally, a second custom, Plexiglas outer stand is placed on top to safely contain any leaks or spills during operation. A photograph of the rotating platform is shown in Figure 1a .
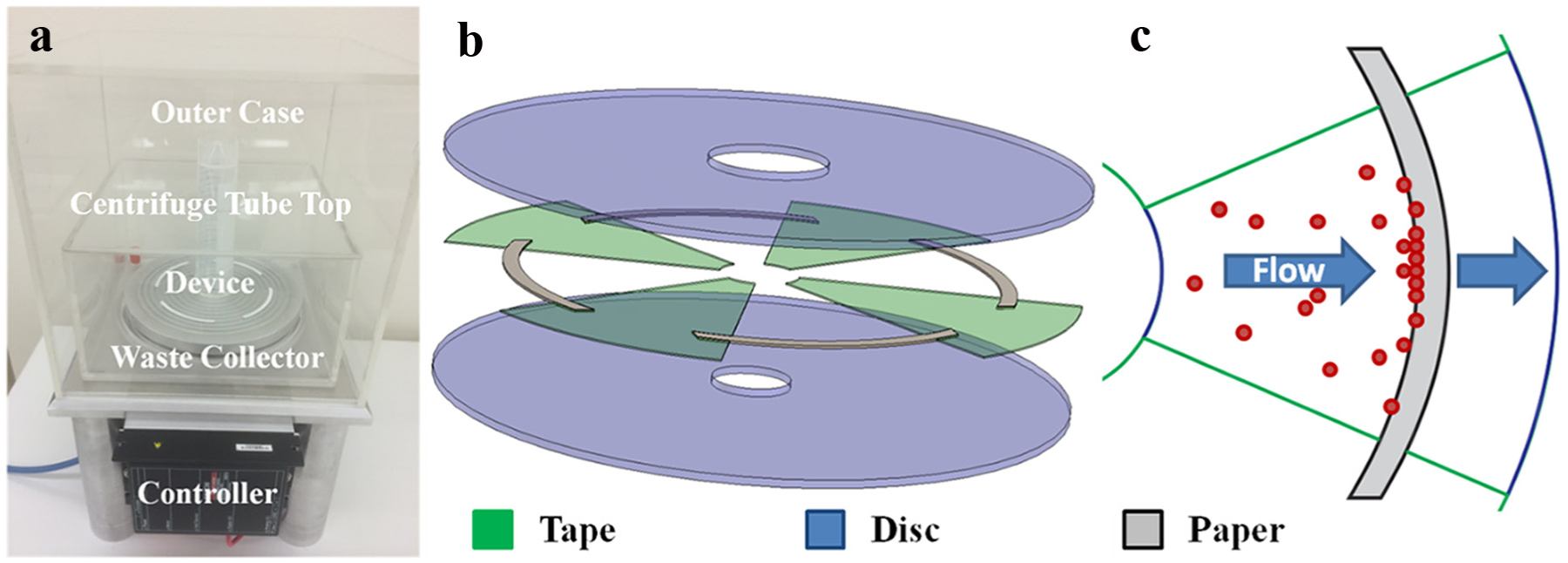
Overview of setup and device. (
The SOLID device is composed of precleaned, laser-cut, polypropylene, double-sided, pressure-sensitive adhesive (PSA) tape (no. 90880, Adhesives Research, Glen Rock, PA) and Whatman’s Fusion 5 (no. 8151-9915, GE Healthcare Life Sciences, Marlborough, MA) paper sandwiched between two polycarbonate (PC), DVD flat-shape, 0.6 mm thickness, 120 mm diameter discs with no stacking ring and a flat stamperholder (Axxicon, Eindhoven, Netherlands). The bottom disc has a standard 15 mm diameter center hole, while the top disc has a custom 22.5 mm diameter center hole to better match the diameter of the sample tube (26 mm inner diameter) above. Briefly, a 150 mm2 piece of PSA was laser cut with a Hypertronics HT6340 Synrad Laser cutter (Synrad, Mukilteo, WA) to have four microfluidic channels. A laminator was used to bind one side of the cut PSA to a PC disc. Four pieces of Fusion 5 paper were manually placed in each channel before placing the top disc on top using a custom disc aligner stand. Pressure was then applied using a hand roller to seal the device together. Next, an 18.5 mm diameter sticker (no. 9964, 3M, Saint Paul, MN) was placed over the smaller-diameter disc to block leakage from the sample tube. Afterward, two-component Araldite super glue (Araldite, Basel, Switzerland) was placed on the open end of a 50 mL Falcon centrifuge tube (no. 14-432-22, Fisher Scientific, Pittsburgh, PA) and placed upside down over the center of the disc and left for 12 h to seal. Finally, a handsaw was used to cut off the closed side of the centrifuge tube. A schematic of the device is shown in Figure 1b , c .
Vitreous Biopsy Model
Vitreous biopsy models were created with either fluorescent microspheres (no. G0500, Fisher Scientific, and no. C-MFRhB-10.0, Corpuscular, Cold Spring, NY) or the cell line Jurkat (no. TIB-152, ATCC, Manassas, VA) or Raji (no. CCL-86, ATCC). All cells were incubated in Corning T-75 flasks (no. 07-202-006, Fisher Scientific) at 37 °C with 5% CO2 in supplemented culture medium composed of RPMI 1640 (no. 10-040-CV, Fisher Scientific), with
Running the Device
After motor calibration with the ESCON studio software, a complete microfluidic device was placed on the free-spinning aluminum stand and the vacuum was turned on to seal the device down. After the waste chamber was placed, the vitreous biopsy model was poured into the device. The outer case was then placed over the setup and the motor was started by pressing and holding a manual switch with an acceleration of 20 rpm/s to minimize the torque requirement of the platform. Unless otherwise stated, all experiments were performed by spinning at 1000 rpm with 45 s on, 5 s off, and 30 s on to ensure all fluid had been processed. The on–off–on spin cycle was used to allow fluid trapped at the edge of the centrifuge tube to resettle and process out. When cells were used, a second step after the first spinning cycle was introduced by adding 1 mL of FxCycle PI/RNase (no. F10797, Fisher Scientific) (
Device Characterization and Optimization
The device was first characterized to optimize manufacturing, geometry, and analysis time. Four separate and equally sized radial channels provided the best fit for the paper. A 5 mm lip on each side of all the channels was included to best fit the 3 mm thick paper in the device. A 140 µm thickness tape was chosen as it best sealed the Fusion 5 paper (<200 µm compressed) and provided faster processing times of 20 mL PBS buffer than thinner adhesives, which limited volume flow. Processing times for 20 mL PBS were tested on the final design at different speeds and results are tabulated in
Table 1
. A sample video of the device analyzing fluid can be seen in
Rotational Speed and Corresponding Processing Time of 20 mL of PBS in the Device.

All speeds have a ramp acceleration of 20 rpm/s.
Fusion 5 paper was chosen as it provided the lowest and most uniform fluorescent background of various Whatman papers tested previously.19,24 Additional testing was performed on the Fusion 5 matrix to determine the effect of nonspecific binding of 1 μg/mL anti-IgE conjugated with phycoerythrin (no. NB110-55357, Novus Biologicals, Littleton, CO) to the paper, and results are shown in Figure 2 . At a gain of 1.0 and exposure time of 5 s, a weak fluorescent background can be seen on the dry paper in the device. Addition of PBS reduced the scattering, and the addition of a large amount of anti-IgE antibody conjugated with phycoerythrin then tripled the fluorescent signal present. An after-wash with 5 mL of PBS returned the paper within 12% of its previous wet background fluorescence level.
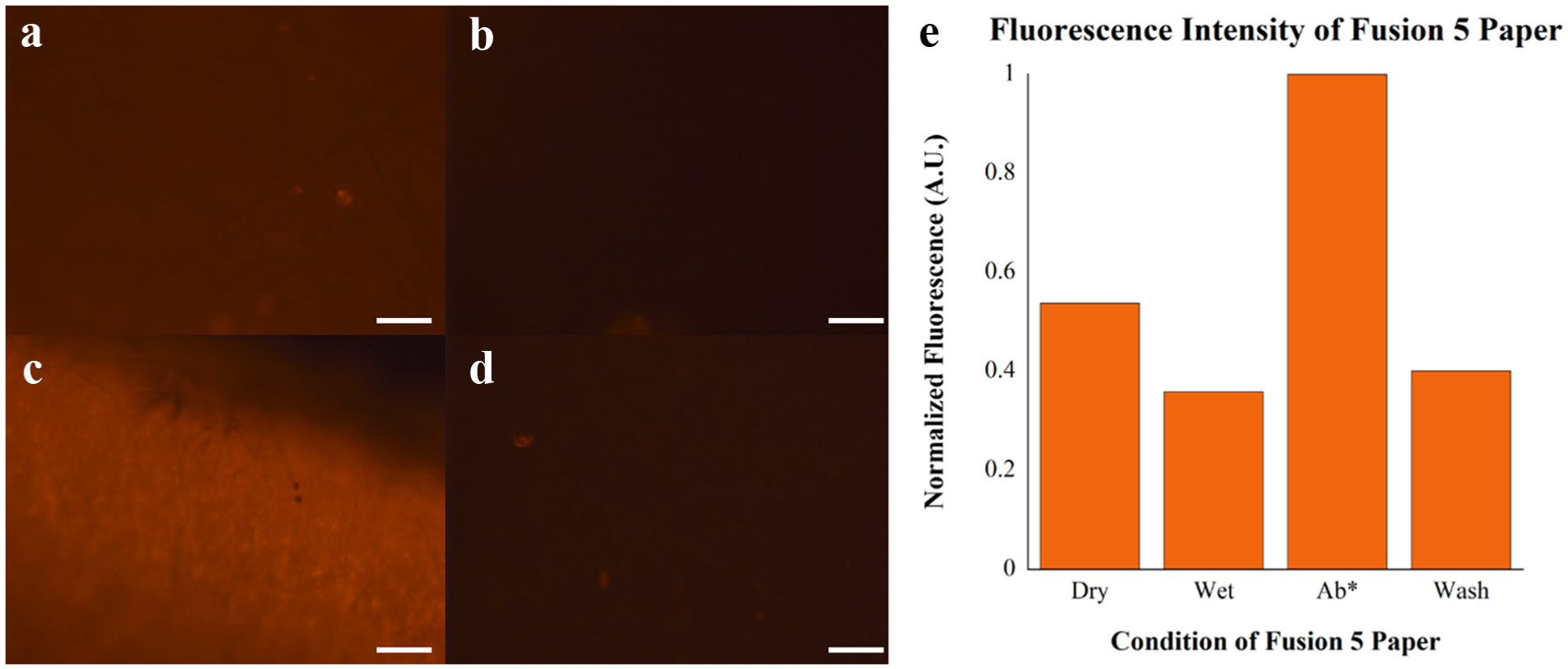
Analysis of fluorescent intensity on Fusion 5 paper under different conditions: (
Device complexity was kept to a minimum by utilizing simple and readily available materials, manufacturing, and methods. Standard polycarbonate discs used to make DVDs, double-sided pressure-sensitive adhesive, and filter paper are low-cost materials that are widely accessible. These materials provide multiple advantages over classic polydimethylsiloxane (PDMS) and glass materials, which are fragile, sharp, less available, and more expensive to make, as they require specially trained technicians and dedicated clean room space, the monthly membership and usage costs of which range in the thousands of dollars. Laser cutting and lamination provide faster, simpler, and cheaper manufacturing than traditional soft lithography methods employed in microfluidic device manufacturing. In addition, the use of centrifugation to provide all the forces necessary forgoes the need for complicated engineering equipment such as syringe pumps. Finally, the disc device also allows the user to place the device directly on a fluorescent microscope for imaging when complete.
Results and Discussion
Proof of Concept
Our first proof-of-concept experiment processed a large solution (20 mL) containing fluorescent microspheres. Microsphere capture was tested on the device using fluorescent polystyrene and melanin microspheres of 5 and 10 µm sizes, respectively. Analysis plots are shown in
Figure 3
(the method used to produce the plots is shown in
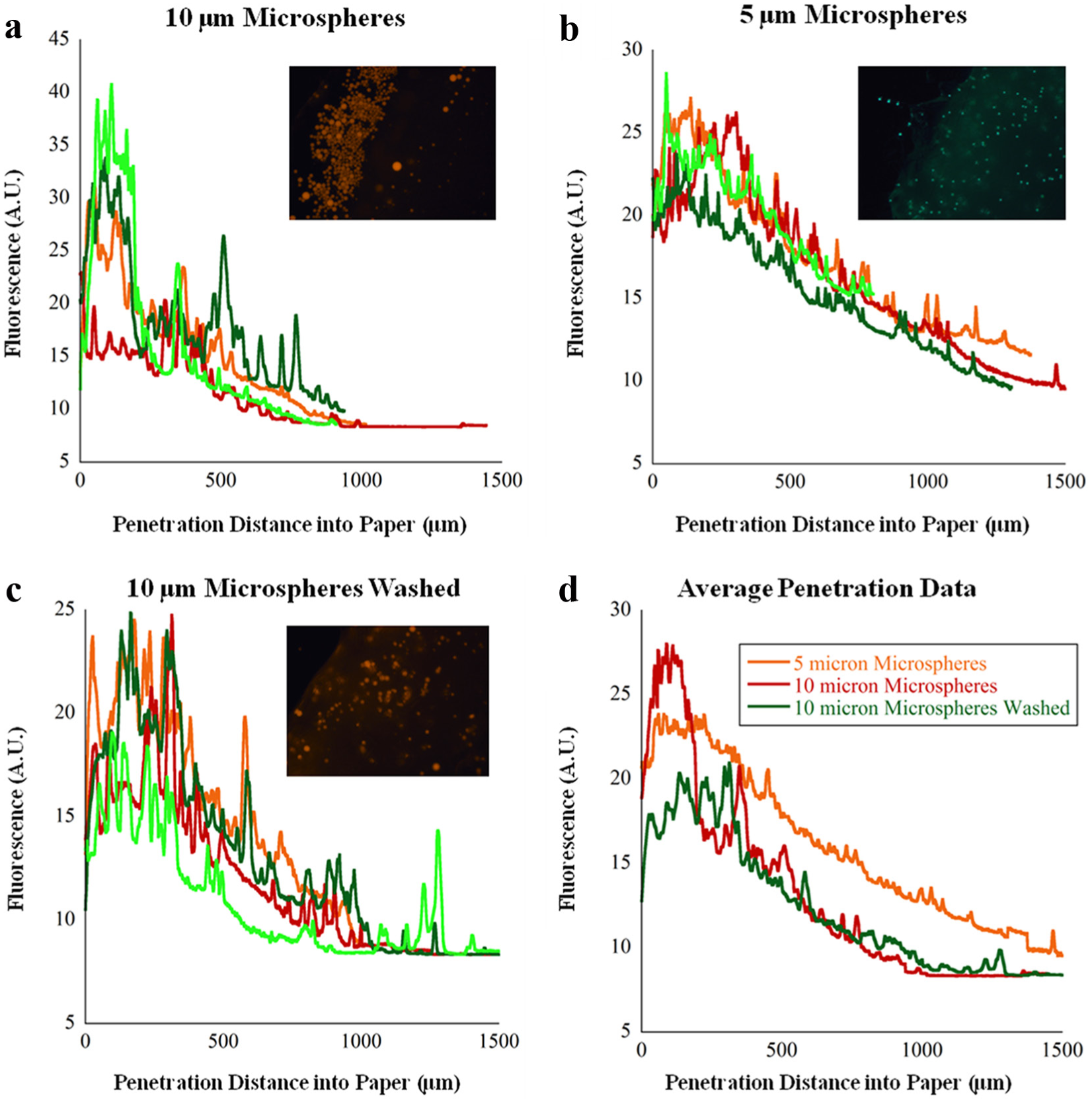
Sample images and distributions of different size particles in the filter paper. The method used to produce distribution graphs is shown in
The next proof-of-concept experiment examined on-chip staining of T cells using a live cell stain. Once the vitreous biopsy model as described in the Materials and Methods section was processed in the device, a calcein AM stain was prepared in 1 mL of culture medium as per the manufacturer’s instructions and spun for a few seconds into the device and incubated for 10 min. The device’s four channels were then imaged using the image stacking method shown in
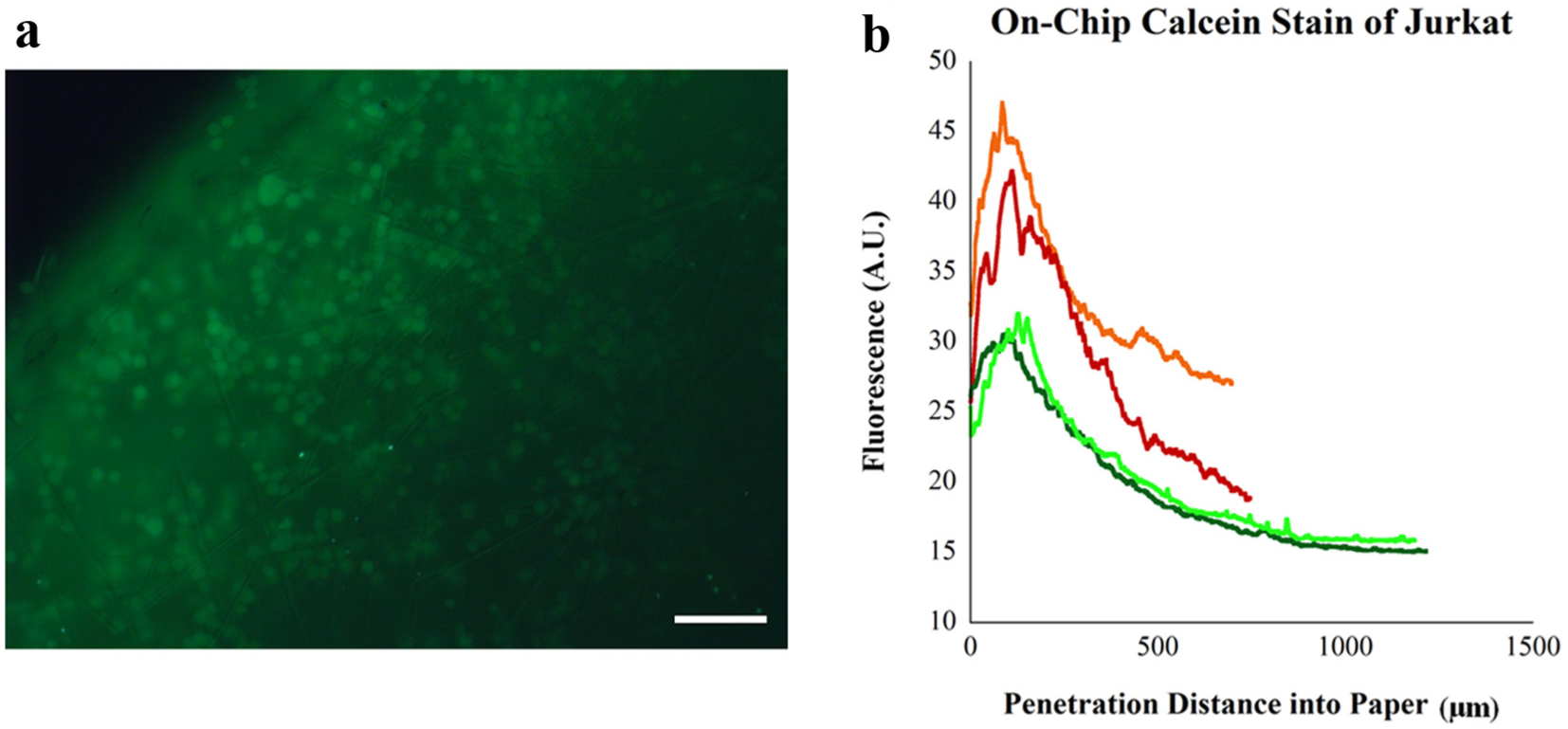
Proof-of-concept on-chip staining of T cells. (
A major consideration for this platform is particle loss through the Fusion 5 matrix. With a nominal pore size of 11 µm (specified by the manufacturer), this large pore size allows rapid ejection of fluid at the expense of smaller cells being able to travel through the matrix. We used a paper thickness of 3 mm to force cells to travel a long distance before escaping. Manipulation of this pore size could allow for a physical cell separation for dealing with more complex samples in the future.
Conclusions
This proof-of-concept work demonstrates an approach to integrate centrifugation with analysis of a large-volume (20 mL) sample containing a small number of cells (104 to 106 cells) for immediate imaging. By utilizing commercialization-friendly manufacturing methods and materials, this framework could help provide for a truly plug-and-play system where the clinician simply inputs the patient sample, and without any additional steps a readout is given. Future work of the SOLID system will examine staining methods for specific cell immunophenotyping and improvements to automation and include optics to provide a “black box” for point-of-care analysis. In addition, while cells may not be easily extracted from the device, on-chip lysis may be possible as an additional step and nucleic acid may be able to be flushed out for downstream analysis. Modifications to device design can also be implemented to change channel geometry to improve the concentration in smaller channels or use multiple paper filters with different capillary sizes for sequential capture. There is strong potential to incorporate this system into an opticocentrifugal platform as described by Burger et al. 26 And an even further end goal would be to incorporate this technology into a modified standard DVD drive used as a fluorescent laser scanning microscope, similar to the one described by Ramachandraiah et al. 27 Integration of sample preparation and analysis on a high-throughput (20 mL/min) microfluidic device via centrifugation is well suited for the point of care.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This material is based upon work supported by the National Science Foundation Graduate Research Fellowship awarded to D.I.W. under grant NSF/DGE-0946746, and by grants provided by the Royal Institute of Technology and the Stockholm County Council (KTH-SLL project).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
