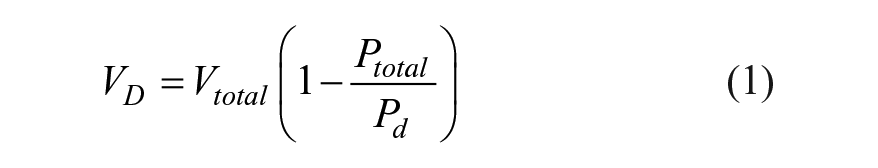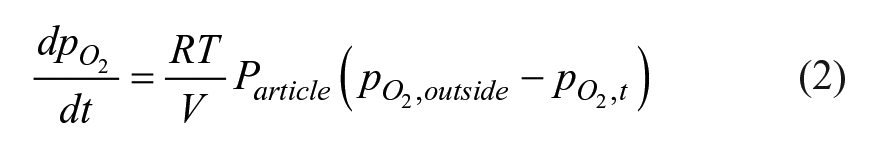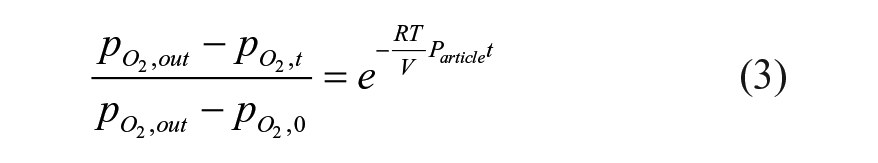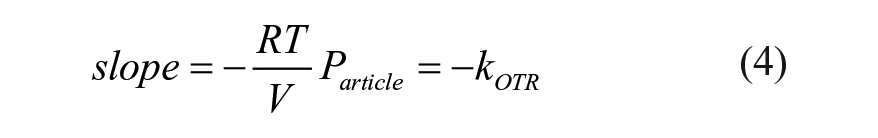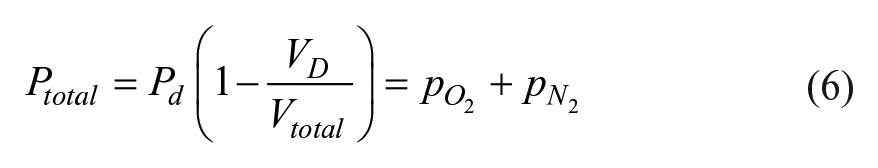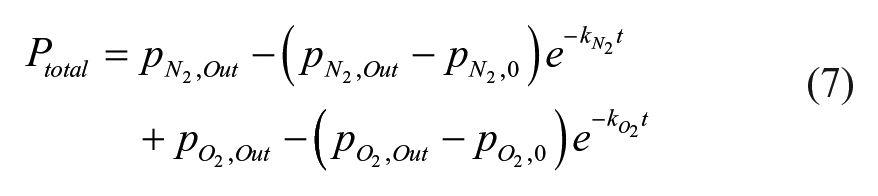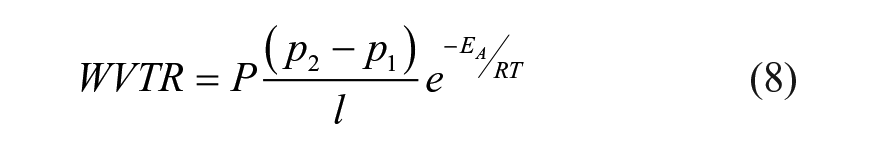Abstract
SiO2 Medical Products, Inc. developed hybrid blood collection tubes (BCTs) that combine the breakage resistance of plastic and a shelf life approaching that of glass. These blended attributes provide improved BCT safety and reliability for patients and clinical workers. A shelf life of at least 2 y with less than 10% draw volume variation was demonstrated on evacuated hybrid BCTs, which is approximately 7 times longer than standard polyethylene terephthalate (PET) BCTs. This translates into more consistent and reliable blood draw volumes over a longer shelf life. The moisture vapor barrier of hybrid BCTs is 5 times lower than that of PET BCTs, which significantly reduces preservative evaporation over their shelf life. As a result, the risk of preservative gelation and alteration to the blood-to-preservative ratio mix is practically eliminated. Cyclic olefin polymer (COP) exhibits superior impact resistance to breakage because of its high ductility and impact strength and is not influenced by defects and flaws as is glass. Although COP has a mechanical toughness comparable with that of PET, it maintains this over a wider range of temperatures (–70 to 121 °C). As a result, COP can tolerate steam sterilization and cold storage temperatures without mechanical fatigue, deformation, or breakage. Lastly, extreme centrifugation of water-filled BCTs did not impose breakage of any kind.
Introduction
Prior to the introduction of evacuated blood collection tubes (BCTs) in the 1950s, blood collection by a phlebotomist was an antiquated, time-consuming, painful, and hazardous practice. 1 The original design of the first evacuated BCT, invented and patented 2 by Joseph Kleiner in 1945, was an innovative breakthrough that revolutionized blood collection. Kleiner’s design enabled blood to be drawn from the vein directly into a glass collection tube that was partially evacuated of air. Becton Dickinson (BD) commercialized Kleiner’s evacuated BCT and trademarked it as the BD Vacutainer.
BD held a complete monopoly on the evacuated BCT market in the United States until the early 1970s. During this time, the introduction of standardized product specifications, good manufacturing practices, and Food & Drug Administration regulations significantly improved the safety, reliability, and quality of BD Vacutainer tubes. Concurrently, additives were developed for different functions including accelerated clotting, anticoagulation, and blood preservation prior to laboratory testing. Additives are applied either “dry” to the wall or “wet” as a fluid in the bottom of the BCT prior to evacuation and stoppering. The additive mixes with the blood during vacuum draw collection and ultimately improves testing reliability and assay throughput.
Despite numerous design improvements, the materials used for BCT construction remained practically unchanged for 40 y. The tubes themselves were molded from multicomponent silicate glass and the rubber stoppers first from natural rubber and then later from halogenated butyl rubbers. The 101st United States Congress enacted the Safety Medical Devices Act in 1990, which sparked a shift from glass to plastic materials in the manufacture of BCTs. The purpose of the change was to reduce and ultimately eliminate glass breakage linked to injury and exposure of patients and clinical workers to blood-borne pathogens.
The decision whether to use glass or plastic BCTs comes with tradeoffs and compromises of functional performance.3,4 For example, the shelf life of glass BCTs is dependent on the stability of the additive. Moisture loss through the wall of a glass BCT to the outside environment is essentially blocked, which helps preserve the additive and maintain a proper blood-to-additive ratio after blood draw. Similarly, blocked gas permeation through the glass wall helps maintain a consistent vacuum pressure inside the BCT during storage. Consistent vacuum pressure results in a consistent blood draw volume over the BCT shelf life.
Plastic BCTs exhibit unmatched impact toughness and resistance to breakage, but they suffer from higher gas and water vapor permeation, which can shorten shelf life. As air permeates through the polymer wall, the partial vacuum pressure increases inside the BCT, which decreases the draw volume. Similarly, water vapor loss from a liquid additive inside a plastic BCT could cause gelation, compromise stability, and affect the clinical results. 1 Subsequently, the shelf life of plastic BCTs is limited by both the additive stability and loss of partial vacuum. The shelf life of ordinary polyethylene terephthalate (PET) BCTs was reported to be more than 12 mo, 1 but our findings based on draw volume loss suggest that the real shelf life could be as short as 3 mo.
Until now, a hybrid BCT with the combined benefits of glass and plastic has been nonexistent in the marketplace. SiO2 Medical Products, Inc. first developed hybrid primary containers for biologic drugs through a blend of materials science and process engineering.5–8 Vials and syringes were precision molded from a medical-grade polymer and coated on the inside wall with a silica-based material less than half a micron in thickness. Combining the break resistance of plastic and the gas barrier of glass comes with some benefits. For example, the oxygen barrier performance of the coating is maintained after 1000 lb of compression force applied to coated vials. 5 Furthermore, coated containers have the optical clarity and appearance of glass but are less than half the weight. The dimensional variability is 10 to 100 times lower than glass, which improves volume accuracy and reduces the risk of poor sealing integrity with rubber stoppers. Lastly, the unique coating is chemically inert and resistant to a wide range of pH values and drug formulation excipients without the risk of metal ion and organic extractables and leachables (E&L).6,7 E&L studies have not been performed on hybrid BCTs. However, hybrid primary containers are composed of the same materials of construction and therefore a similar level of E&L cleanliness is expected.
SiO2 Medical Products, Inc. adapted and developed their hybrid coating technology for BCTs intended for genetic diagnostic testing. Hybrid BCTs containing a proprietary blood preservative are competitively priced compared with other commercial BCTs on the market for the same intended use. Here we review how the hybrid BCTs are fabricated and the material science behind their unique blend of performance attributes. Breakage resistance and vacuum retention as they relate to shelf life are the primary foci of this report, because many of the other performance attributes have been reported in the past.5–9
Materials and Methods
BCTs and Stoppers
BCTs were injection molded on a horizontal Milacron (Cincinnati, OH) Roboshot 165-ton electric molding machine. Zeonex 690R cyclic olefin polymer (COP) resin pellets were purchased from Zeon (Louisville, KY) 10 and fed into the screw and barrel, where the resin was melted at a barrel temperature of 290 °C. The plastic was forced into the injection mold at 135 MPa. The injection mold was used to create final geometry of the BCT and cool the plastic enough to eject the parts from the mold. An eight-cavity mold was used with a cycle time of 16 s to eject the finished BCT parts. Two volumes of BCTs were molded with nominal fill volumes of 4 mL (13 mm OD and 75 mm height) and 9 mL (100 mm height and 16 mm OD).
Commercial glass and PET BD (Franklin Lakes, NJ) Vacutainer tubes with similar volumes and dimensions were purchased from BD to serve as benchmarks. Standard halogenated butyl rubber stoppers (Streck, Omaha, NE) were used for both 4 mL and 9 mL BCTs.
Plasma-Enhanced Chemical Vapor Deposition
Barrier coatings were deposited on the inside of BCTs by plasma-enhanced chemical vapor deposition on proprietary customized production equipment. A vacuum pump reduced the pressure on the inside of the blood tube to about 1 torr. A mixture of hexamethyldisiloxane (Alfa Aesar, Tewksbury, MA) and oxygen gas was introduced to the inside of the vial by mass flow controllers. The gas mixture flows continuously in and out of the BCT under steady-state flow conditions. A voltage potential applied across the BCT from an outer powered electrode to an inner counter electrode, ignites the low-pressure gas mixture into a plasma discharge. The coating deposition is initiated as soon as the plasma discharge is ignited.
Oxygen Transmission Rate
The oxygen transmission rate (OTR) was determined by measuring the oxygen partial pressure over time inside a stoppered BCT with a Mocon (Brooklyn Park, MN) OpTech system.11,12 An oxygen sensor was affixed to the interior of a BCT. The BCT was sealed with epoxy and a glass slide under a reduced oxygen atmosphere (nominally 0.2% oxygen). Once the epoxy was set, the initial atmospheric composition within the BCT was measured. The BCTs were stored in a controlled temperature chamber at 25 °C. As the oxygen content within the BCT increased over time, the intensity of the light emitted by the sensor decreased in accordance with the Stern-Volmer relationship
12
for fluorescence. The oxygen partial pressure,
Water Vapor Transmission Rate
A modified version of the USP 671 test methodology 13 was employed to measure the water vapor transmission rate (WVTR) into BCTs. Each BCT was weighed on a microbalance with four significant digits. Four grams of 3 Å molecular sieve pellets (Delta Adsorbents, Roselle, IL) was weighed separately on a microbalance. Pellets were transferred into a blood tube, which was closed with a halogenated butyl rubber stopper. BCTs were stored in a controlled environmental chamber at 75% relative humidity (RH) and 40 °C. BCTs were weighed gravimetrically prior to placing in the environmental chamber. Moisture weight gain was determined by remeasuring the vials every 7 d within 30 to 60 min after removing BCTs from the environmental chamber. The moisture weight gain was plotted with time, and the slope of the linear regression was equal to the WVTR in units of milligram of moisture per day (i.e., mg/day).
Draw Volume Test
The analysis of draw volume of BCTs was conducted according to ISO 6710:2017. 14 BCTs were stoppered under partial vacuum, stored at ambient temperature, and tested at 1, 3, 6, 15, and 24 mo after stopper insertion. A total of 16 BCTs (i.e., four BCTs from four different production lots) were measured at each time point.
Drop Impact Test
BCTs were filled with water to the desired fill volume (i.e., 9 mL or 4 mL), evacuated, and then stoppered. A vacuum pump pulled a partial vacuum pressure of –26.8 in. Hg for 9 mL BCTs and –26.0 in. Hg for 4 mL BCTs. After 5 s of applied vacuum to the BCT, a rubber stopper was inserted. Each BCT was dropped in a vertical orientation inside a plexiglass tube to a height of 36 inches measured from the bottom of the tube to the surface of the floor (i.e., linoleum tile). Each dropped BCT was visually inspected for cracks. If no cracks were observed, then the drop test was repeated with a new set of BCTs but at a height of 48 inches. Visual inspection of cracks was repeated a second time.
Results and Discussion
Origin, Manufacture, and Materials Science of Hybrid BCTs
Combining two dissimilar materials like glass and plastic into a hybrid BCT has its challenges because their physical and chemical properties are so different. Moreover, blending the advantages of both materials without picking up their deficiencies is particularly difficult. The intent is to design and manufacture a BCT that has the break resistance of plastic, the vacuum retention (i.e., shelf life) of glass, and no leachable contaminants from either material. The solution is a blend of process engineering and materials science.
Prior to the development of hybrid BCTs, SiO2 Medical Products, Inc. first commercialized hybrid SiOPlas primary containers for packaging biologic drugs.5–9 Hybrid primary containers possess a similar blend of performance advantages that BCTs require. A range of hybrid syringe and vial sizes were manufactured from a medical-grade polymer resin and coated on the inside with a submicron-thick silica-based coating. This was made possible with the development and adaptation of two well-known manufacturing processes from two different industries, injection stretch blow molding (ISBM) or injection molding (IM) and plasma-enhanced chemical vapor deposition (PECVD). ISBM and IM processes produce polymer containers, which are then coated on the inside by a PECVD process. Both the molding and coating manufacturing processes had to be reengineered and optimized to fit the form factor of a BCT to replicate the performance advantages of hybrid primary containers for drug products.
Irrespective of the application, the materials of construction for each product are critical to the functional performance, and BCTs are no different. This starts with the selection of polymer resin for molding BCTs. Ordinary plastic BCTs are injection molded from polyethylene terephthalate. IM is a preferred process over ISBM because of the small size and geometric simplicity of BCTs compared with pharmaceutical vials and beverage bottles.
Cyclic olefin resins were selected for molding BCTs because of their utility in various medical applications including pharmaceutical drug packaging and disposable laboratory consumables. Cyclic olefins are a broad class of polyolefin resins that are unlike any of the more traditional commodity resins such as polyethylene and polypropylene (PP).
A total of eight resins were evaluated, including two from Zeon (i.e., Zeonex 690R, Zeonex 790R), two Mitsui resins (i.e., APEL, APL6015T), two from Ticona (i.e., TOPAS 6013 M-07, TOPAS 6013 S-04), and two from JSR (i.e., Arton D4540 and Arton F4520). Although many criteria were used for the evaluation, the most important were transparency, heat resistance, and barrier to moisture. Zeonex 690R COP resin was found to have the best balance of these three criteria as well as the best esthetics, color, and moldability. The only criterion that was inferior to ZEON 790R resin was heat resistance, but it was completely satisfactory for steam sterilization.
After molding COP BCTs, coatings were applied to the inside by PECVD. PECVD was first developed in the 1950s, and later developments in the 1960s led to the fabrication of the first microprocessor components.15–17 Early PECVD was strictly a batch process whereby silicon wafers were loaded into a vacuum chamber for either thin-film deposition, ion implantation, or chemical etching steps. PECVD processing has evolved and found utility in many other market segments such as flat panel displays, 18 medical implants, 19 contact lenses, 20 and food and beverage containers.21–23 Some of these applications require batch processing, whereas others have been developed into semicontinuous to continuous processing, as in the case of food and beverage containers. The application of PECVD to parenteral containers and BCTs is a matter of adapting the process to the form factor of the container. Because deposition is restricted to the inside of a container, a semicontinuous PECVD process is possible, as with beverage containers. A key benefit of BCTs is their rigidity, which enables vacuum pumping the inside without collapsing, which is not possible with flexible beverage containers.
The PECVD process for BCTs is shown in
The coating is composed of three individual layers, each with a specific function, as shown in
Gas Permeation, Draw Volume, and Shelf life
The shelf life prediction of a BCT starts with an understanding of the relationship between the inside pressure before filling and the volume of liquid drawn into the tube after filling. First, consider an empty, evacuated, and stoppered BCT with a total internal pressure at the storage time just prior to liquid draw, Ptotal, and a volume, Vtotal. Ptotal at this point is less than 1 atmosphere of pressure or a partial vacuum pressure. After puncturing the stopper and connecting it to a source of liquid at atmospheric pressure, Patm, the liquid flows freely into the BCT until the pressure reaches a final headspace pressure of Ph. After the BCT is filled, the headspace pressure above the liquid, Ph, is equal to the atmospheric pressure or draw pressure, Pd (i.e., 1 atm). The total BCT volume, Vt, is the sum of the final headspace volume, Vh, and the liquid volume or liquid draw volume, Vd. Applying Boyle’s law, PtotalVtotal = PhVh, substituting Ph = Pd and solving for the draw volume is as follows:
This equation indicates that the draw volume is directly related to the total internal pressure. During storage, air slowly enters both glass and plastic stoppered BCTs, which increases the total pressure over time. As the inside pressure increases, the amount of blood drawn in to the BCT decreases. The change of the total internal pressure over time can be related to the steady-state gas permeation in the BCT over time.
The instantaneous oxygen transmission through a sealed BCT is
where
Integration of eq 2 results in
where
The permeation rate constant
The OTR constant,
A comparison of the OTR constant for COP, barrier coated COP, PET, and glass 9 mL BCTs is shown in Figure 1 . A plain COP BCT has the highest OTR constant of 0.0129 day–1. A standard PET BCT has an OTR constant 8 times lower (i.e., 0.00159 day–1), and a coated COP BCT has an OTR constant 86 times lower (i.e., 0.00015 day–1) than an uncoated COP BCT. The application of the barrier coating to COP provides about 10 times lower OTR constant than the standard PET BCT. Because the relative shelf life, ignoring the stopper OTR, of PET and coated COP BCTs is directly proportional to the ratio of their OTR constants, the COP BCT shelf life is approximately 10 times longer than a PET BCT. The OTR of the glass BCT is below the lower detection limit of the test method or 0.00006 day–1.
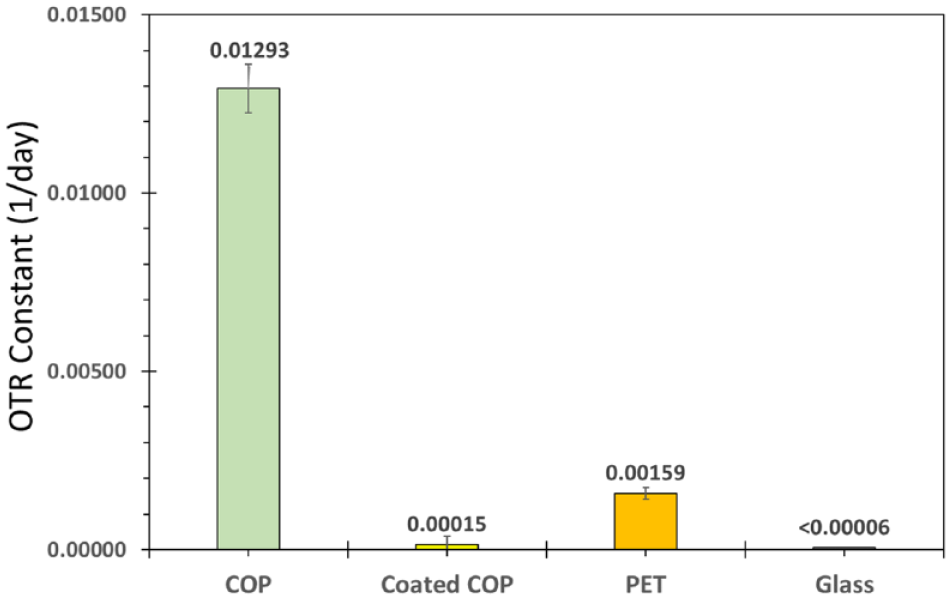
Mean oxygen transmission rate (OTR) constants of 9 mL blood collection tubes (BCTs) made from cyclic olefin polymer (COP), coated COP, polyethylene terephthalate, and glass. Ten replicates of each of the four BCT types were stored in a controlled chamber at 25 °C, and the oxygen partial pressure,
The permeation of air through the stopper and plastic BCT wall increases the internal pressure and ultimately decreases the draw volume over storage time. The relationship between draw volume and storage time can be derived by first realizing that the total internal pressure, Ptotal, in eq 1 is the sum of both nitrogen,
The permeation of nitrogen into the BCT follows a behavior analogous to eq 3 but with a nitrogen transmission rate constant,
This total internal pressure can then be used to estimate draw volume by substitution into eq 1. Although the change in internal pressure with relatively short storage times is approximately linear (using a Taylor series expansion approximation of eq 7), the internal pressure change is nonlinear with longer storage times. Similarly, draw volume decay during storage is approximately linear at only short storage times.
Draw volume was measured experimentally and plotted versus storage time for PET, COP, and coated COP 4 mL BCTs. A linear regression analysis was applied to the data for each of the three BCT types as shown in Figure 2 . It should be noted that the draw volume data for time 0 was purposely omitted from the plot and the linear regression analysis. This is particularly important for the PET and COP BCTs because the measured draw volume at time 0 (not shown) is much higher than an extrapolation of the linear regression analysis would predict. The reason for this is attributed to gas leaving the inside surface of the BCT and stopper while evacuated, also called degassing. The BCTs and stoppers are equilibrated with ambient air prior to evacuation, and as such, uncoated polymer, PET, and stoppers are saturated with dissolved oxygen and nitrogen at the time of evacuation. After evacuation and once stoppers are inserted, the dissolved gas diffuses out into the evacuated interior of the BCT. Degassing can take several days to establish a steady-state gas permeation through the BCT wall and stopper. Coated COP BCTs are far less susceptible to degassing because of the dense barrier coating. Initial draw volumes were collected on day 7 to minimize the effects of degassing on the linear regression analysis.
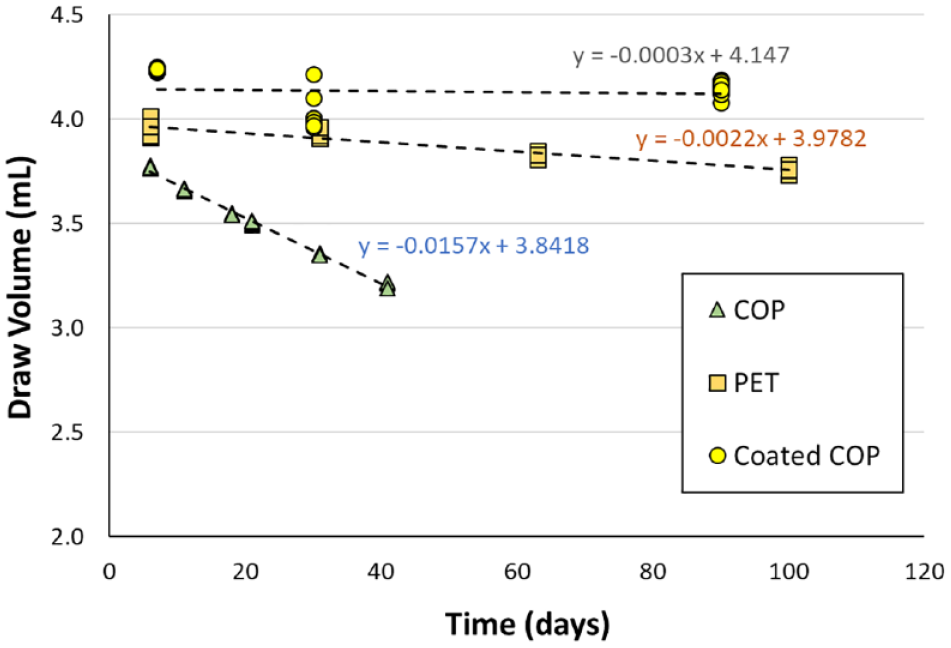
Draw volume decay rates for 4 mL blood collection tubes (BCTs) made from cyclic olefin polymer (COP), polyethylene terephthalate, and coated COP. All BCTs were evacuated according to ISO 6710 requirements and stoppered using a standard rubber stopper. Four replicates of each BCT type were measured at each time point over 100 d and plotted. A linear regression analysis was performed on the data for each BCT type. A linear equation (i.e., y = mx + b, where m is the slope and b is the y intercept) for each linear regression is shown beside each data set.
For relatively short storage times shown in Figure 2 , the slope of the linear regression approximates the draw volume decay rate. The relative shelf life of two different BCT types can be approximated from the ratio of the draw volume decay rates. For example, the shelf life of a coated COP BCT is approximately 7 times longer than PET BCTs and 55 times longer than COP BCTs.
The shelf life specifications for plastic BCTs can vary depending on the manufacturer and materials of construction and were reported to be greater than 12 mo. 1 According to the draw volume decay in Figure 2 , a 2-y shelf life is not achievable with a PET BCT because of its higher gas permeation. Our internal company specification for coated COP BCTs is a draw volume decay of no more than 10% over 2 y. In other words, if the BCT draw volume immediately after evacuation and stoppering is 4.0 mL, then after 2 y stored at ambient conditions, it cannot drop below 3.6 mL.
The draw volume decay of coated COP BCTs was measured over a period of 2 y. Two different BCT sizes with 9 mL and 4 mL nominal fill volumes were included in this testing. The targeted minimum nominal draw volumes for 9 mL and 4 mL BCTs after 2 y was 8.1 mL and 3.6 mL, respectively. Therefore, BCTs are manufactured with minimum actual blood draw volumes of 9.3 mL and 4.3 mL to ensure an 8.1 mL and 3.6 mL minimum draw volumes after 2 y. The bar graphs in Figure 3 show that the mean draw volumes after 2 y are at or above the targeted draw volume minimums after 2 y of storage.
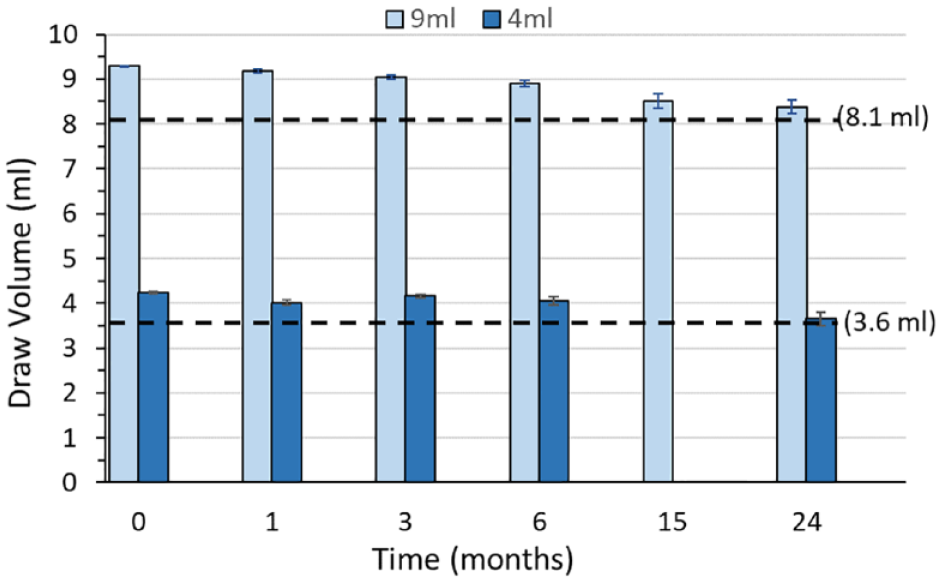
Draw volume decay rates for 9 mL and 4 mL coated cyclic olefin polymer blood collection tubes (BCTs) over 2 y of storage at ambient conditions. All BCTs were evacuated according to ISO 6710 requirements and stoppered using a standard rubber stopper. Four replicates of each BCT type were measured at each time point over 24 mo. The mean and standard deviation are plotted at each time point. Dotted lines correspond to a 10% drop of the nominal draw volume for 9 mL and 4 mL BCT, which correspond to 8.1 mL and 3.6 mL, respectively.
The draw volume decay of glass BCTs was not measured because this represents air permeation through the rubber stopper alone. The glass wall is essentially a perfect barrier with no contribution to the draw volume decay. A glass BCT would be practically a horizontal line at the top of Figure 2 . In other words, the shelf life of a rubber stoppered glass BCT would be expected to exceed well beyond 2 y.
Moisture Barrier and Absorption
Low water vapor permeation is an important requirement of polymeric containers for preserving drug formulations stored in prefilled syringes, slowing evaporation from storage microplates and maintaining preservatives in BCTs. Moisture loss can result in gelation of the preservative and alter the appropriate ratio with blood serum, which affects the shelf life of the BCT. Water vapor permeability is a constant used to compare different materials without a geometry in mind.
Cyclic olefins exhibit superior water vapor permeability compared with other commodity polyolefins such as PP and PET. For example, the permeability of COP film10 is 0.25 g/m2/24 h measured at 25 °C and 90% RH. PP and PET films of the same thickness have permeabilities of 0.5 and 1.9 g/m2/24 h, respectively, measured under the same temperature and RH conditions. The permeability, as with diffusivity, follows an Arrhenius temperature dependence whereby the permeability increases as the temperature increases. The extent of permeation and diffusion of water vapor at higher temperature depends on the molecular structure and chemistry of the polymer. The water vapor permeability of the COP film at 50 °C and 90% RH is 2 g/m2/24 h. PP and PET films are 3 to 5 times more permeable measured at the same temperature and RH. COP, therefore, is a better material choice for safeguarding against water vapor permeability loss based on data from the literature.
The WVTR is a more convenient metric to compare different materials of the same geometry such as BCTs. The permeability constant, P, is a material-specific property independent of the geometry, which is related to WVTR by the following equation:
where P is the permeability of water vapor, l is the thickness, p2 is the partial pressure of water vapor on one side of the film and p1 is on the other side, EA is the activation energy, R is the universal gas constant, and T is the temperature.
The water vapor barrier of BCTs with different materials of construction was verified by measuring the WVTR. WVTR was measured according to the USP 671 test method 13 on plastic 4 mL BCTs composed of COP, PET, and glass. COP BCTs exhibited about 5 times lower WVTR compared with PET, as shown in Figure 4 . The WVTR of a glass BCT is a measure of the rubber stopper because the WVTR of the glass is essentially zero. The WVTR of coated COP BCTs (data not shown) is extrapolated to be essentially equivalent to COP BCTs based on extensive comparative testing of coated and uncoated COP vials and syringes. The barrier coating provides no additional water vapor barrier protection; therefore, the rate-limiting step of water vapor permeation is through the COP polymer. This is not the case for oxygen permeation, which follows Fick’s law of diffusion for an ideal gas. Water vapor, by comparison, is a nonideal gas that deviates from Fick’s law because of interactions with the silicon oxide coating as it diffuses through.25,26
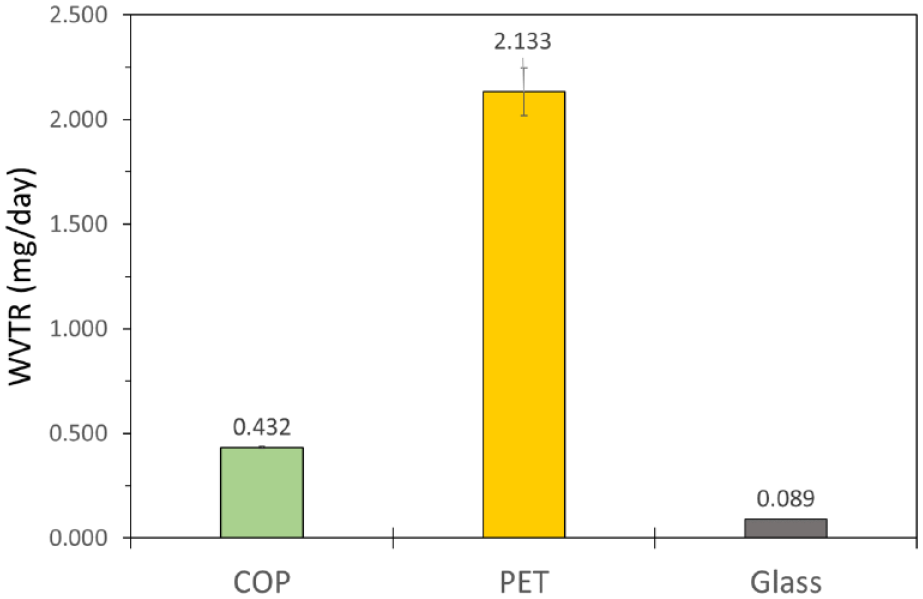
Water vapor transmission rate of 4 mL blood collection tubes made from cyclic olefin polymer and polyethylene terephthalate was measure according to a modified USP 671 test method described in the Materials and Methods section. All vials were stored in a controlled environmental chamber at 75% relative humidity and 40 °C. Weight gain was measured gravimetrically every 7 d within 30 to 60 min after removing from the chamber.
Breakage, Mechanical Toughness, and Heat Resistance
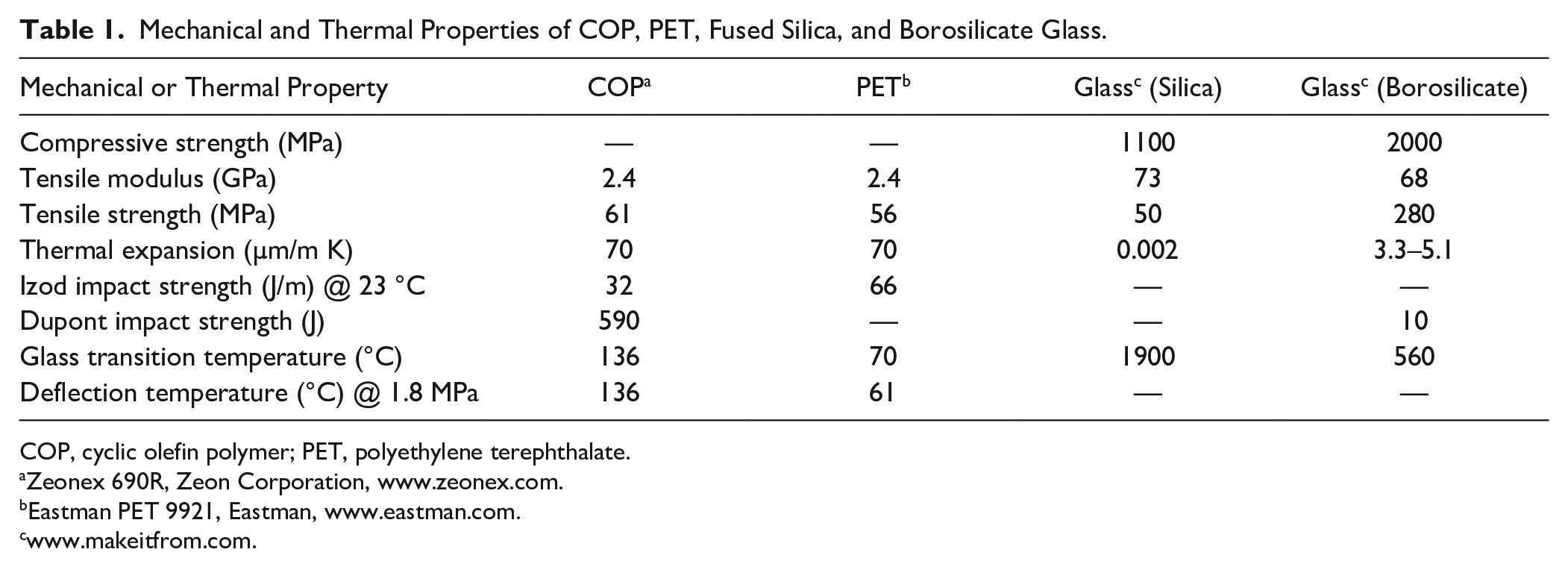
The undisputed Achilles heel of glass has long been breakage and is unlikely to change despite attempts to improve it.27,28 Although fused quartz has remarkably high thermal shock resistance, its mechanical toughness, as measured by its compressive and tensile strength, is inferior to borosilicate glass, as shown in Table 1 . Furthermore, pure silica is impractical for manufacturing high-volume commodity containers because of its extremely high glass transition temperature (Tg). The addition of metal oxides to pure silica (e.g., borosilicate glass) is essential to lower its Tg for easier forming and shaping into complex-shaped containers. This comes with improvements in mechanical toughness, particularly its ability to resist compressive and tensile stress compared with fused quartz. Nonetheless, borosilicate glass breakage and shattering from manufacturing and dropping is an omnipresent problem. This type of failure is exacerbated by defects and flaws in borosilicate glass, which cause it to fail well below its theoretical mechanical limits ( Table 1 ). For example, even a simple scratch on the surface of a glass container can concentrate the stress from an impact, causing it to fracture or shatter.
Mechanical and Thermal Properties of COP, PET, Fused Silica, and Borosilicate Glass.
COP, cyclic olefin polymer; PET, polyethylene terephthalate.
Zeonex 690R, Zeon Corporation, www.zeonex.com.
Eastman PET 9921, Eastman, www.eastman.com.
Polymers, in general, are inherently elastic materials that are less susceptible to impact-induced breakage compared with borosilicate glass. COP is no exception, with high elongation (i.e., 20%), low tensile modulus, and high impact strength. COP’s impact strength is even maintained at temperatures as low as –70 °C, which is advantageous for cold storage.
Drop impact testing was conducted on 10 mL COP and glass BCTs. All BCTs were filled with 9 mL of water and stoppered. BCTs were dropped in a vertical orientation (i.e., stopper up and tube bottom down) at a height of 36 inches onto a linoleum tiled laboratory floor. None of the 24 tested COP BCTs showed any visual signs of breakage or cracking after impact. Twenty-one of 24 glass BCTs filled with water, evacuated, stoppered, and dropped from 36 inches broke. All glass BCTs failed when dropped from a height of 48 inches ( Table 2 ).
Drop Impact Test Results on Water-Filled COP and Glass Blood Collection Tubes at 36-Inch and 48-Inch Drop Heights onto a Linoleum Tile Surface. a

COP, cyclic olefin polymer.
Visual evidence of breakage such as cracks or damage are tabulated in the “No. Break” column and those without in the “No. Pass” column.
Extreme centrifuge testing was conducted to ensure the durability of water-filled BCTs. Typical residual centrifugation forces for blood plasma are between 500 and 2000 × g. Three water-filled, evacuated, and stoppered COP BCTs were centrifuged for 20 min at a residual centrifugation force of 3000 × g according to ISO 6710 guidelines. Visual inspection showed signs of cracks or leakage in the COP BCTs. PET BCTs are expected to survive these centrifugation conditions according to the manufacturer. 29 Glass BCTs, however, are not recommended to exceed 2200 × g in a swing-out rotor and 1300 × g for a fixed-angle rotor. 29 This may be due to the potential for breakage.
PET has comparable thermal expansion, tensile strength, and impact strength to COP at room temperature, although its glass transition temperature and heat deflection temperature are much lower ( Table 1 ). COP’s higher heat deflection temperature helps maintain mechanical strength and reduce the risk of partial deformation at high temperatures. For example, COP maintains its dimensional integrity after 20 min of steam sterilization at 121 °C, which is not possible for many polymers, including PET. 10 Glass has no problem with steam. Steam is routinely used to sterilize a variety of medical devices and primary containers. Steam is not typical for finished BCTs because of the risk of altering the inside partial vacuum pressure and thus the draw volume. Gamma sterilization is most typical and compatible with either glass or plastic BCTs, and COP is no exception. Overall, COP can be used by a broad range of sterilization techniques and over a wider working temperature range compared with PET because of its excellent heat resistance and low temperature toughness.
Conclusions
SiO2 Medical Products Inc. developed hybrid BCTs that combine a shelf life approaching glass and the breakage resistance of plastic. A consistent and reliable blood draw volume is maintained over at least 2 y of shelf life because of improved gas barrier properties compared with ordinary plastic BCTs. Similarly, the moisture vapor barrier is so high that preservative evaporation is expected to be negligible over the BCT shelf life. As a result, the risk of preservative gelation and alteration to the blood-to-preservative ratio mix is practically eliminated.
Hybrid BCTs exhibited superior impact resistance to breakage because of their high ductility and impact strength. This is not influenced by defects and flaws as in glass. COP tolerates a broad range of temperatures (i.e., –70 to 121 °C) without mechanical fatigue, deformation, or breakage, unlike other plastics such as PET. Extreme centrifugation of water-filled hybrid BCTs did not impose breakage of any kind.
Supplemental Material
Weikart_SLAS_Technology_Supplemental_Material_1-2_Response_to_Reviewers2 – Supplemental material for Hybrid Blood Collection Tubes: Combining the Best Attributes of Glass and Plastic for Safety and Shelf life
Supplemental material, Weikart_SLAS_Technology_Supplemental_Material_1-2_Response_to_Reviewers2 for Hybrid Blood Collection Tubes: Combining the Best Attributes of Glass and Plastic for Safety and Shelf life by Christopher M. Weikart, Adam P. Breeland, Matt S. Wills and Martin E. Baltazar-Lopez in SLAS Technology
Footnotes
Acknowledgements
We are grateful to Dr. Steve Martin for deriving the permeation and draw volume equations. Dr. Martin also reviewed the article for technical accuracy and appropriate messaging. We also appreciate Dr. Dave Henton for providing sources of polymer mechanical properties and reviewing that section for technical content.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.