Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of nylon polymers, which function in cosmetics primarily as bulking and opacifying agents. The Panel reviewed relevant animal and human data related to these large polymers and determined that they are not likely to penetrate the skin. Whatever residual monomers may be present were not present at a sufficient level to cause any reactions in test subjects at the maximum ingredient use concentration. Accordingly, the Panel concluded that these ingredients are safe in the present practices of use and concentration.
Introduction
In the 1930s, Carothers and coworkers pioneered the synthesis of the first commercially viable synthetic fibers, polyamides. 1 The initial commercial application of these polyamides, specifically nylon 6/6, was women’s hosiery. These polymers found use during World War II for parachutes, tire cord, thread, and rope.
In cosmetic formulations, nylon ingredients function primarily as bulking and opacifying agents. This safety assessment reviews the available scientific literature, including unpublished data provided by industry, for nylon-6, nylon-11, nylon-12, nylon 6/12, nylon-66, nylon-611, nylon-10/10 and nylon-12/6/66 copolymer.
The Cosmetic Ingredient Review Expert Panel (Panel) has previously reviewed the safety of 2 of the monomers used in the production of nylon, that is, decanedioic acid (also known as sebacic acid) and adipic acid. 2 The Panel concluded that these ingredients are safe in the present practices of use and concentration.
Chemistry
The definition and structure of these ingredients are presented in Table 1, and available information on the physical and chemical properties of nylon ingredients is presented in Table 2.
Definitions, Functions, and Structures of nylon Ingredients in This Safety Assessment. 13 ,a
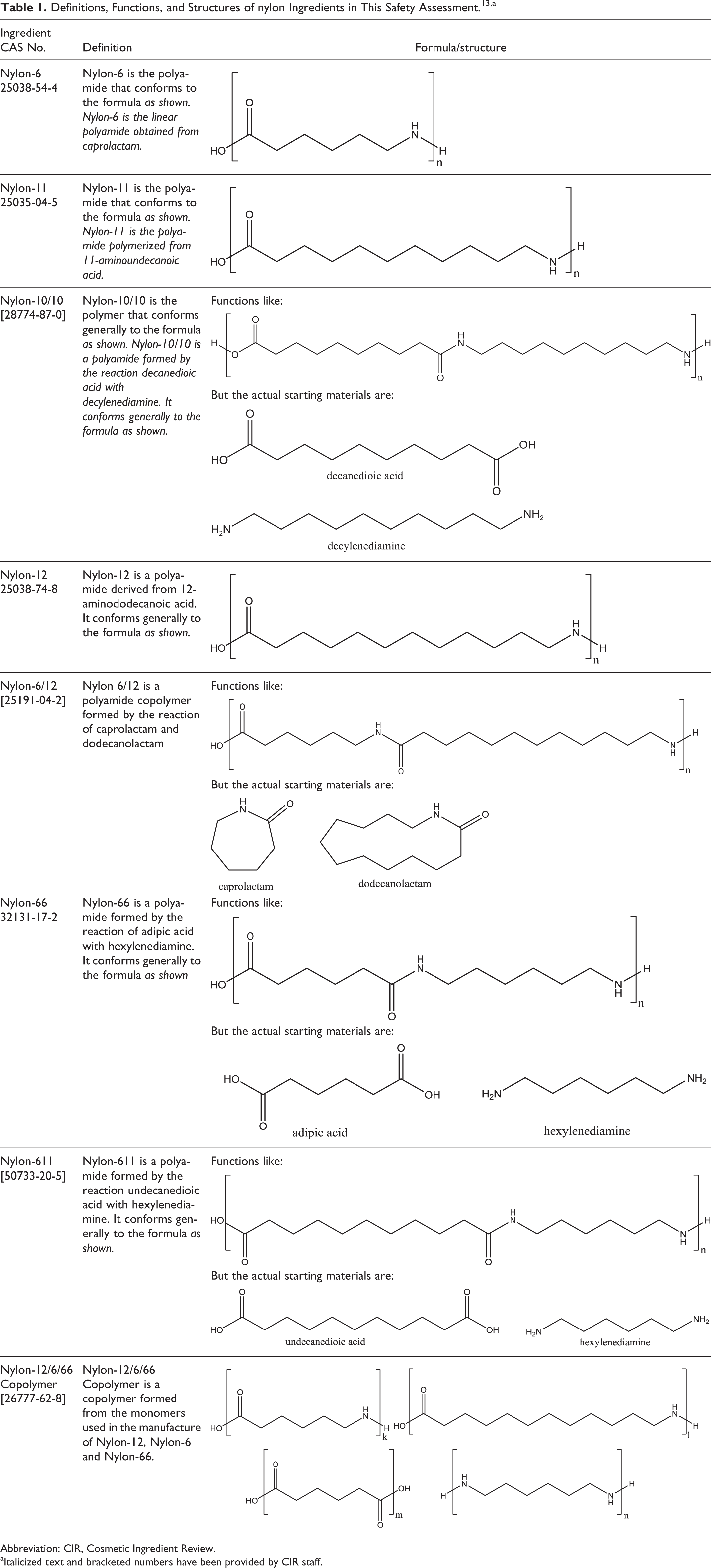
Abbreviation: CIR, Cosmetic Ingredient Review.
aItalicized text and bracketed numbers have been provided by CIR staff.
Physical and Chemical Properties.
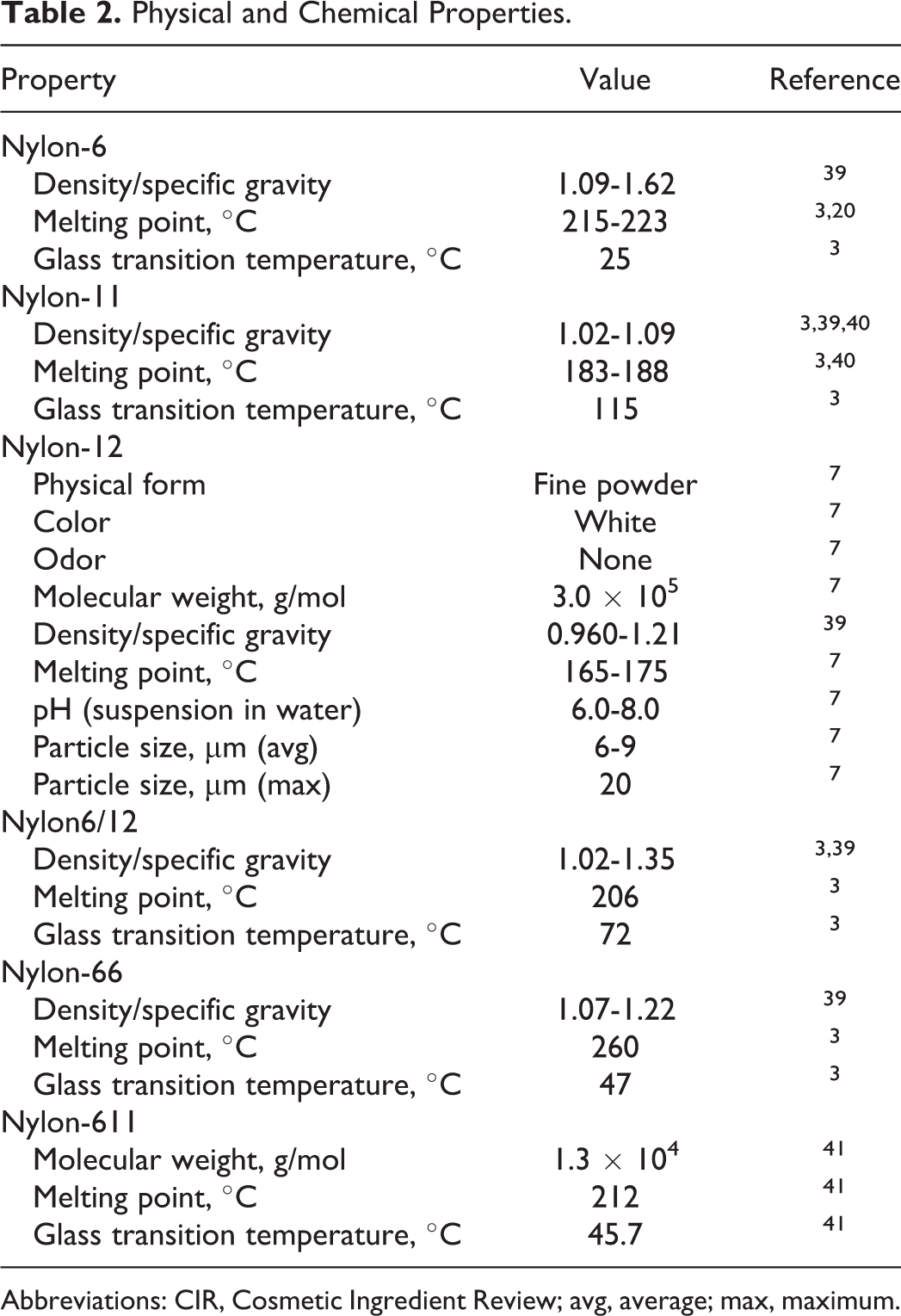
Abbreviations: CIR, Cosmetic Ingredient Review; avg, average; max, maximum.
The trade name terminology, nylon, has been firmly established as applying only to polyamides polymerized from unsubstituted, nonbranched aliphatic monomers. Unfortunately, variations in these names are dispersed throughout the literature, such as nylon-66, 66-nylon, 6,6-nylon, and 6-6-nylon, all indicating nylon 6/6. The numbers added to the word “nylon” are indicative of the number of methylene groups (wherein the acid carbonyl can be counted as a theoretical methylene) in a monomer. These nylon ingredients are commonly synthesized via 1 of the 3 ways: (1) polymerization of linear ω-amino acids, (2) ring-opening polymerization of lactams, or (3) copolymerization of a linear diacid and a linear diamine. For example, nylon-6 is a polyamide that can be synthesized from the unsubstituted, nonbranched, aliphatic monomer 6 aminocaproic acid (Figure 1). Therein, “n” is equal to the number of monomer units in the resulting polymer. Because manufacturers can control the polymerization process to produce virtually any size of polymer desired, n and, thus, molecular weights of these nylon ingredients can vary greatly.
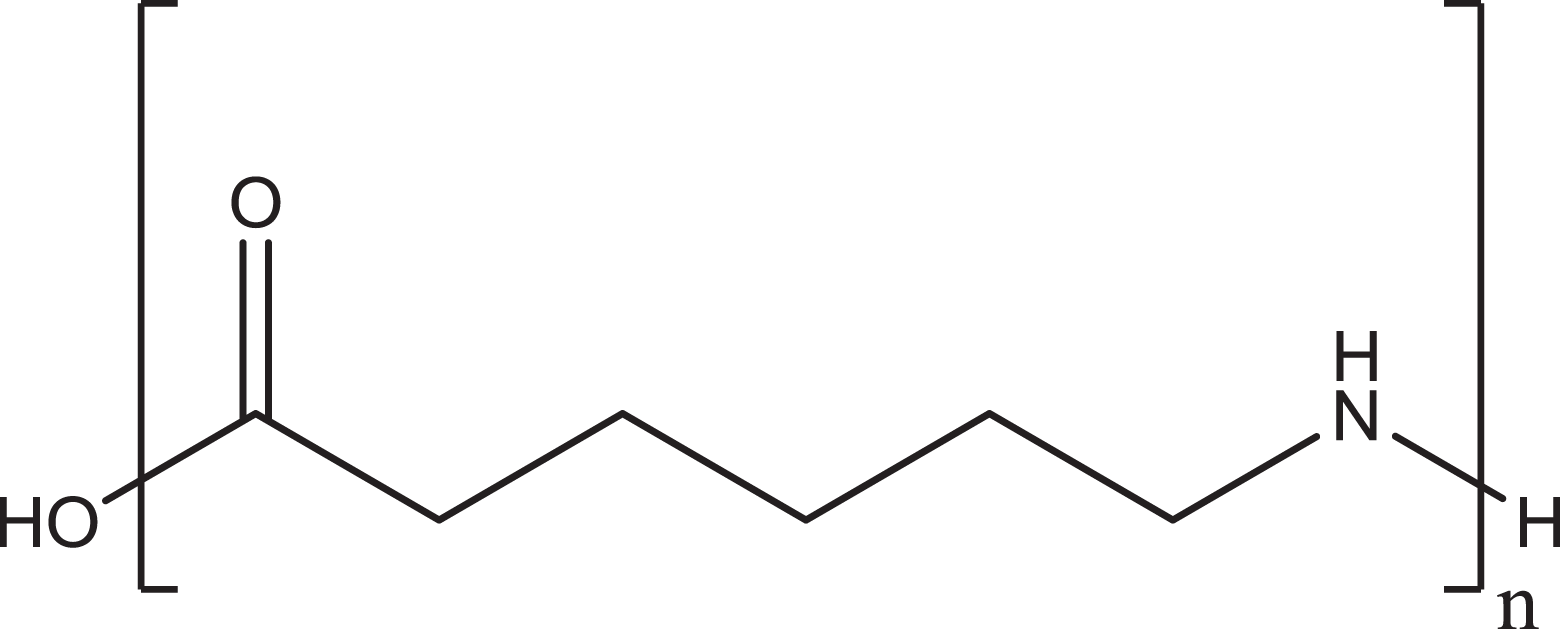
Nylon-6.
In practice, however, nylon-6 and other polyamides can also be synthesized from the ring-opening polymerization of the cyclic versions of these monomers, lactams. For instance, nylon-6 and nylon-12 can be synthesized from ∊ caprolactam and dodecanolactam (also known as laurolactam), respectively (Figure 2). 3 Ring-opening polymerization from the lactam monomer most likely occurs via a concerted ring-opening monomer addition. Essentially, this is a transamidation, converting from the amide of the lactam to the amide of a nylon product. The amide bond of a lactam (except in small, strained lactams that are not pertinent here) is a fairly high-energy bond, thus requiring reaction conditions above 500 K to initiate polymerization. However, this works in favor of processing and drawing the fibers, as they are ductile at this temperature.
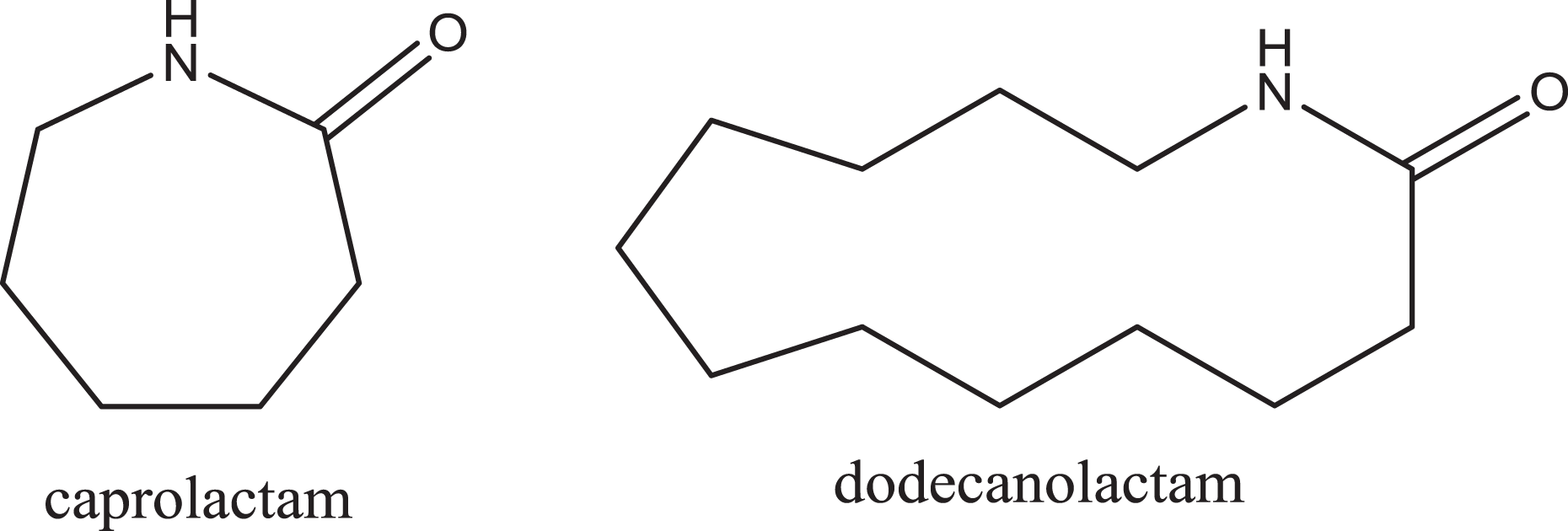
∊ Caprolactam and dodecanolactam.
Nylon analogues with 2 numbers added to the name represent polyamides synthesized from 2 distinct monomer units. Nylon 6/6 (International Nomenclature Cosmetic Ingredient name nylon-66), for example, is the polyamide synthesized from hexylenediamine and adipic acid (wherein each monomer has 6 methylene groups; Figure 3). 3 This polymer is an ordered, alternating copolymer of these monomers. As an example, starting with a molecule of adipic acid, the first step is the addition of 1 molecule of hexylenediamine. Therein, one of the nitrogens of hexylenediamine will form a bond with the carbon of one of the acid groups of adipic acid, releasing water (OH from the acid and H from the amine). This will result in a new molecule with the unreacted acid group of the adipic acid residue on one end, a newly formed amide in the middle, and the unreacted amine of the hexylenediamine at the other end. Next, either an additional molecule of adipic acid can be added to the unreacted amine of the hexylenediamine residue, or an additional molecule of hexylenediamine can react with the unreacted acid of the adipic acid residue. The polymerization will then continue in both directions, along the linear axis of the growing polymer, until one of the monomers is spent or a terminating group (eg, acetic acid) may be added to endcap the amines.
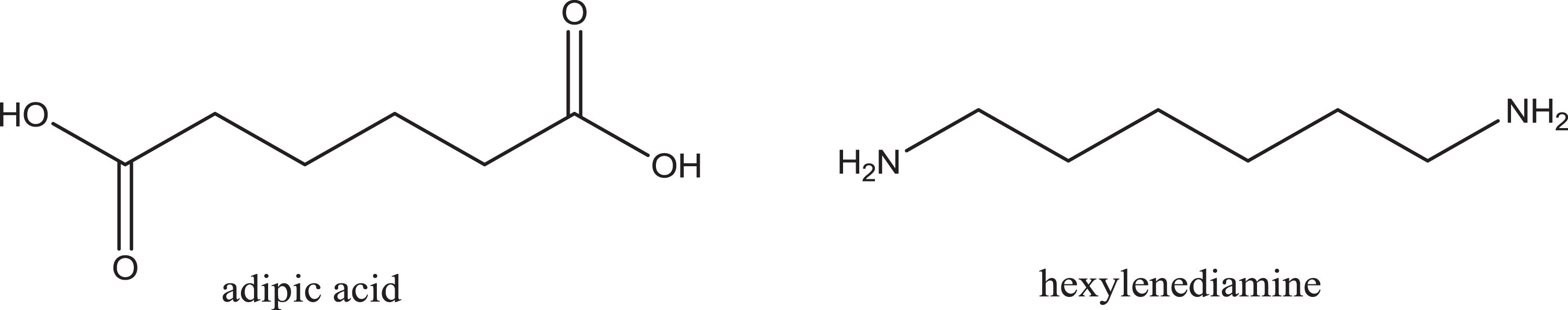
Adipic acid and hexylenediamine.
Traditionally, the first number listed in nylon nomenclature represents the diamine monomer and the second number represents the diacid, but this is not strictly followed. Indeed, nylon 6/12 is actually a polyamide synthesized via the copolymerization of caprolactam and dodecanolactam (neither of which is a diamine or a diacid). Nylon 6/12 is reported to have at least 85% (w/w) ∊ caprolactam residue and not more than 15% (w/w) dodecanolactam residue. 4
Nylon-6 and nylon-66 can undergo photooxidative degradation, resulting in loss of strength, following long-term exposure to ultraviolet radiation. 3
Method of Manufacture
The nylon analogues in this report, and polyamides in general, are typically manufactured by direct amidation of a diacid with a diamine, by self-amidation of amino acids or by ring-opening transamidation of lactams. 3 For example, a supplier reports that nylon-10/10 is obtained by melt polycondensation of sebacic acid (decanedioic acid) and decane diamine, and nylon-12 is synthesized by ring-opening polymerization using laurolactam. 5 A stoichiometric balance, in the case of direct amidation of a diacid, is readily obtained by the preliminary formation of a diammonium salt (referred to as a “nylon salt”). This balance can be adjusted simply by adjusting pH. This aqueous salt solution is then concentrated to a slurry and heated under pressure. The pressure is then slowly released to essentially perform a melt polymerization. Molecular weight control is often achieved by adding acetic acid as an end-capping unit.
Impurities
Nylon-6 may contain approximately 1% of the monomer, ∊ caprolactam. 6 Additionally, nylon-6 may contain 6-aminocaproic acid, as well as ε caprolactam, if synthesized from the ω-amino acid.
A product description of nylon-12 indicated that the maximum levels of heavy metals and arsenic were 10 ppm and 1 ppm, respectively; however, certificates of analysis for this product indicate that these substances were not detected (detection limits were not specified). 7 -9 Polybrominated biphenyls and polybrominated diphenyl ethers were also not detected (detection limits were not specified). 9 In further analyses of nylon-12, the residual solvent levels of isoparaffinic hydrocarbon ranged from 0.08% to 0.13% and the residual monomer of the nylon-12 powder was 100 ppm or less, which meets United States Pharmacopeia 33/ National Formulary 28. 10 -12 The initiator and catalyst in the manufacture of nylon-12 that is, potassium metal and phosphorus trichloride, were reported to be 2.0% to 2.5% and 1.0% to 2.0%, respectively. 12
A supplier of nylon-12 and nylon-10/10 reported the residual monomer content after ethanolic extraction to be 0.14% to 0.18%. 5
Inclusion of a variety of chemicals (including volatile liquids and even gasses) is a common occurrence in polymer manufacture, especially in large-scale production. Most commercial polymers have at least some monomer, solvent, initiator, or catalyst entrapped in the polymer superstructure, which may not be easily evaporated or released. The entrapment may be so inclusive that there is little concern of release under normal conditions of use, but time and solvents in a formulation may enable the escape/release of these nonpolymeric materials.
To further complicate matters, the glass transition temperature (Tg) of nylon-6 (25°C) is just above the room temperature but below the body temperature. A Tg is a unique properties-changing temperature threshold exclusive to polymers. Above this temperature, the polymer is more pliable and plastic like. Below this temperature, the polymer is more brittle and “glass-like.” Nylon-6 may be below the Tg in packaging, but on the skin it could rise above the Tg. Exceeding this threshold causes a change in the physical properties of the polymer, potentially increasing the rate of release of the monomers or other nonpolymeric materials from the polymer.
Use
Cosmetic
The nylon ingredients discussed in this safety assessment function primarily as bulking and opacifying agents in cosmetic formulations. 13 Additional functions may include absorbents (nylon 6/12 and -611) and film formers (nylon-12/6/66).
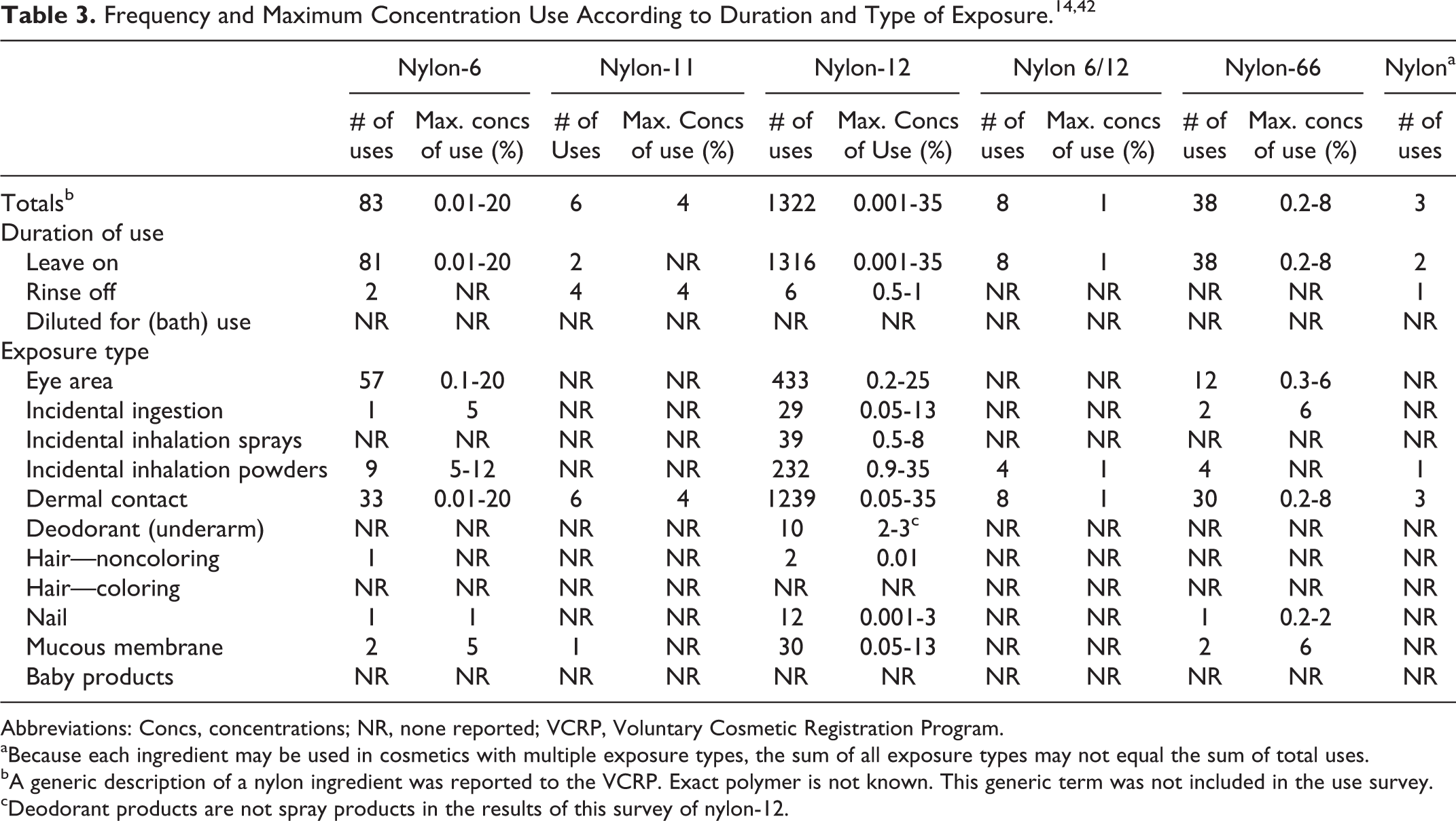
Table 3 presents the current product formulation data for the nylon ingredients. According to the information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), nylon-12 has the most reported uses in cosmetic and personal care products, with a total of 1322; 285 of those uses are in eye shadow formulations. 14 Nylon-6 has the second greatest number of overall uses reported, with a total of 83; 47 of those uses are in mascara formulations.
Abbreviations: Concs, concentrations; NR, none reported; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bA generic description of a nylon ingredient was reported to the VCRP. Exact polymer is not known. This generic term was not included in the use survey.
cDeodorant products are not spray products in the results of this survey of nylon-12.
In a survey of use concentrations conducted by the Personal Care Products Council, nylon 12 is reported to be used at a range of maximum concentrations of 0.001% to 35%, with 35% reported in face powder formulations. 42 For nylon-6, the range of maximum concentrations was reported to be 0.01% to 20%, with 20% reported in eyebrow pencil formulations. No uses or concentrations were reported for nylon-611 or nylon-12/6/66 copolymer. Nylon-10/10 is a new cosmetic ingredient for which there are no reported uses to the VCRP. No reported use concentrations are available.
Nylon 12 was reported to be used in cosmetic sprays (perfumes and other fragrance preparations) and face powders, and could possibly be inhaled. These ingredients are reportedly used at concentrations up to 8% in spray products and up to 35% in face powder products. 42 In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 15 -18 Likewise, particles from loose powder products would be expected to have aerodynamic diameters greater than 10 µm, although the size distributions of these powders have not been reported. Therefore, most droplets/particles incidentally inhaled from cosmetic aerosols would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 15,17 The nylon ingredients in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 19
Noncosmetic
Nylons, especially nylon-6 and nylon-66, have been used in textiles, such as hosiery, parachutes, tent cloth and other woven fabrics, thread, tire cord, fishing line, and rope, since the early 1940s. 1,20
Nylon-6, nylon-11, nylon-12, nylon 6/12, and nylon-66 have been approved by the FDA as indirect food additives used for food contact surfaces (21 CFR §177.1500).
Nylon-6 and nylon-66 are used in nonabsorbable surgical sutures, which are FDA-approved medical devices (21 CFR §878.5020).
Toxicokinetics
The major metabolites of the monomer of nylon-6 were studied in male Sprague-Dawley rats that received 3% caprolactam in their feed ad libitum for 2 to 3 weeks. 21 The metabolites were isolated by ion-exchange chromatography and characterized by infrared and nuclear magnetic resonance spectroscopy from urine samples collected at 24-hour intervals during the final week of feed administration. The majority of the caprolactam (˜16% of the dose) was excreted as 4-hydroxycaprolactam or the corresponding free acid, which rearranges in acidic solutions to yield an equilibrium mixture of 6-amino-γ-caprolactone and 6-amino-4-hydroxyhexanoic acid. In addition to these metabolites, a small amount of 6-aminohexanoic acid was excreted. No other relevant studies on the toxicokinetics of the nylon ingredients were discovered in the published literature.
Toxicological Studies
Acute Toxicity
Oral—nonhuman
Nylon-12 monomer
The acute oral median lethal dose (LD50) for dodecanolactam, the monomer of nylon-12, was 2330 mg/kg body weight in Wistar rats tested at dose levels of 1580, 1990, 2510, and 3160 mg/kg body weight, with central nervous system stimulation (trembling, convulsive twitches, and ataxia) as the main clinical sign appearing at about 1580 mg/kg body weight. 22
Nylon-12
The acute oral LD50 for nylon-12 was reported to be 1 g/kg in rats, mice, guinea pigs, and rabbits. 23 In cats, the acute oral LD50 was about 0.25 g/kg.
Nylon 6/12
The acute oral LD50 for nylon 6/12 in a corn oil suspension was reported to be greater than 10 g/kg in Sprague Dawley rats. 4
Dermal—non-human
Nylon-12 monomer
The dermal LD50 for dodecanolactam was reported to be greater than 2000 mg/kg body weight in Sprague-Dawley rats under occlusive conditions. 22 Decreased food consumption and stagnation of body weight development were noted.
Intraperitoneal—nonhuman
Nylon-6 monomer
In a study from 1954, the monomer of nylon-6, ∊ caprolactam, caused convulsions in rats following intraperitoneal (ip) injection (frequency and duration not described) at doses starting at 500 mg/kg.6 Deaths were observed at doses of 800 mg/kg and higher.
Nylon-11
In an acute study, groups of 25 male mice received ip injections (1.0 mL per 20 g of body weight) of nylon-11 macerated in normal saline, cottonseed oil, or 30% ethyl alcohol. 24 The mice were closely observed for the first hour after injection and then periodically for the next 7 days. No toxic responses were observed.
Nylon-12
The acute ip LD50 was reported to be 0.32 to 0.53 g/kg in rats. 23
Intravenous—nonhuman
Nylon-6 monomer
In a 1954 study, the monomer of nylon-6, ∊ caprolactam, caused convulsions in rabbits following intravenous (iv) injection (frequency and duration not described). 6 The animals were dosed with 100 to 300 mg/kg of the monomer, but the doses that caused the convulsions were not identified.
Nylon-11
In an acute study, groups of 25 male mice received iv injections (1.0 mL per 20 g of animal weight) of nylon-11 macerated in normal saline, cottonseed oil, or 30% ethyl alcohol. 24 The mice were closely observed for the first hour after injection and then periodically for the next 7 days. No toxic responses were observed.
Rabbits were cannulated in the jugular vein with nylon-11 macerated in normal saline, 30% ethyl alcohol, or harsh alcohol extract (HAE; the result of refluxing nylon pellets with absolute alcohol for 24 hours). 24 The internal carotid artery was cannulated for blood pressure measurement and the trachea was cannulated for differential respiratory measurement. No further details on doses or exposure durations were provided. A decrease in blood pressure was observed after dosing with nylon-11 in the ethyl alcohol extract. In some cases, there was a prolongation of the response to iv acetylcholine administered 4 minutes after the nylon-11, but this effect could not always be duplicated. No adverse effects regarding blood pressure or respiration were observed following dosing with nylon-11 in saline or in HAE.
Repeated Dose Toxicity
Oral—nonhuman
Nylon-12 monomer
In a 90-day oral gavage study, Sprague-Dawley rats in groups of 20/sex in the low and mid doses and 25/sex in the control and high doses received 0, 5, 25, and 125 mg/kg body weight/d of dodecanolactam.22 No adverse effects on general condition, behavior, body weight gain, or feed consumption were observed in the 5 and 25-mg/kg dose groups. In the 125-mg/kg dose group, a slight increase in total serum protein and albumin levels was observed in females and a moderate increase in potassium levels was observed in males. Sodium excretion was also increased in males. In a few males treated with 25 or 125 mg/kg had slight morphological changes in centrilobular hepatocytes (ground glass appearance) were noted, but this effect was reversible 4 weeks postexposure, and in the absence of any other sign of liver toxicity was considered adaptive rather than adverse. No histopathological changes were observed in the reproductive organs of these animals at any dose level. The no observed adverse effect level (NOAEL) was 25 mg/kg body weight/d.
In a subchronic study, male rats in groups of 10 were fed doses of 0, 0.06, 0.12, 0.25, or 0.5 g/kg of dodecanolactam via stomach tube 5 times per week for 12 weeks. 23 The no-effect level was 0.5 g/kg. No microscopic examination was performed on the major organs.
Dodecanolactam was evaluated for toxicity in a 90-day feeding study in Beagle dogs by the FDA. 23 Groups of 4 males and 4 females received 44 to 49, 350 to 352, or 969 to 989 mg/kg dodecanolactam in feed (high-dose group received capsules) 6 times per week. A positive control group received 306 to 353 mg/kg caprolactam in feed. (Results of the positive control group were not described.) A negative control group was also included in the study. The dogs were observed daily for ∼8 hours during the week and at least once daily on weekends. No abnormal behavior was observed during the treatment period in the controls and in the low- and medium-dose groups.
However, all dogs in the high-dose group demonstrated an intense resistance to the administration of the capsules at times. On the second day, the dogs exhibited apathy, ataxia, trembling, and sialorrhea. Reactions to auditory and visual stimuli were observed. These clinical signs occurred throughout the treatment period. One female dog of the high-dose group died after 5 weeks. The death was preceded by severe lateral decubitus, extremely high respiratory frequency, and intense howling on occasion. Diarrhea was observed during the entire study in the high-dose group and on occasion in the low- and medium-dose group. Vomiting was observed in all groups, except the positive control group, with the highest frequency occurring in the high dose group. Dogs in the high-dose group had body weight losses ranging from 20% to 25%. Dogs in the remaining dose groups had increases in body weights. Feed consumption was decreased in the high-dose group, but not in the remaining dose groups. Urine parameters were normal in all dogs. A decrease in erythrocyte number, hematocrit, and hemoglobin concentration was observed in 1 dog of the high-dose group, the reticulocyte count was increased after 3 months in another dog in the high-dose group, and the leukocyte count decreased with a statistically significant difference from the negative controls in the high-dose females after 1.5 months and in the high-dose males after 3 months. A slight increase in serum glutamate-pyruvate transaminase activity at 1.5 and 3 months was observed in the high-dose group. Increased alkaline phosphatase activity was observed in the middle-dose group at 1.5 months in females and at 3 months in males; these levels were increased in the high-dose male and female groups after 1.5 months. Liver and kidney function tests in all dogs during the duration of the study were normal.
Ophthalmoscopic examinations on all dogs were normal. At necropsy, a statistically significant increase in the liver weights occurred in the female high-dose dogs, as was a statistically significant increase in liver weight to brain weight ratio, when compared to the negative controls. A statistically significant increase in the liver weight to body weight ratio was observed in dogs in the middle- and high-dose groups. In the high dose males, statistically significant decreases in absolute testes weights, testes weight to body weight ratio, and testes weight to brain weight ratio were observed when compared to negative controls. Histopathological examination indicated changes in the prostate and testes in the high-dose group. Interruption of spermatozoa maturation was observed in this group.
This evaluation of a 90-day study in Beagle dogs determined that the no significant effect level was 44 mg/kg. The FDA calculated the estimated maximum acceptable daily intake of food for humans to be 2.6 mg/kg or 1.74 ppm. 23
Intraperitoneal—nonhuman
Nylon-11
In a subchronic study, groups of 10 female Holtzman rats received daily ip injections (2 mL) of nylon-11 macerated in HAE (doses not reported).24 A control group of 10 rats received daily ip injections of normal saline solution. The injections were given 5 days a week for 6 weeks. A blood study was conducted on each rat at the beginning and end of the experiment. The animals were weighed weekly. At the end of the study, all animals were killed and underwent histopathological examination. The hematological values indicated significantly higher white cell counts for the rats that received nylon-11 compared to the control rats; however, the authors of this study could not attribute this observation directly to the test material “because the terminal count was elevated over the initial count as would be expected.” No other signs of toxicity to nylon-11 were observed in this study.
Tissue implantation—nonhuman
Nylon-11
The potential toxicity of nylon-11 in pellet form was studied for up to 3 months in rabbits. 24 Groups of 2 to 10 animals received implantation of the samples in the paravertebral muscle, the brain, and the intestinal mesentery. Positive and negative control samples were implanted into the same animals (materials used were not specified). Animals were observed for behavioral changes and “signs of growths” throughout the 3-month period. No toxicity to the exposed tissues was observed, even in a histopathological examination.
This study also used albino Holtzman female rats to study toxicity to nylon-11. 24 The test material was implanted in the thigh, medial to the biceps femoris muscle, and bounded by the adductor femoris, gastrocnemius, and the biceps femoris. In several additional rats, implants were placed in the nape of the neck into or between fascia or fatty tissue, lying near the dorsal surface of the levator auris muscle. At various postimplantation times, the rats were killed and the implant sites were examined for evidence of toxicity. No evidence of toxicity was observed.
Reproductive and Developmental Toxicity
Nylon-6 Monomer
The potential for the nylon-6 monomer, caprolactam, to cause developmental toxicity was evaluated in Fischer 344 rats and New Zealand White rabbits. 25 Groups of 20 rats received 0, 100, 500, or 1000 mg/kg body weight/d via gavage on days 6 to 15 of gestation. All surviving dams were killed on day 20 of gestation and the dams and fetuses were examined. The high-dose group dams had significantly decreased (P ± 0.05) maternal survival rate and fetal viability. The remaining dose groups were comparable to controls. No fetal skeletal anomalies or major malformations were observed in any dose group.
Groups of 25 rabbits received 0, 50, 150, or 250 mg/kg body weight/d caprolactam via gavage on days 6 to 28 of gestation. An additional group of 21 rabbits received 3 mg/kg/d of the positive control, 6-aminonicotinamide, on gestation day 9. All surviving dams were killed on day 29 of gestation and the dams and fetuses were examined. No embryotoxicity or teratogenicity was observed. In the 150- and 250-mg/kg/d groups, fetal weights were decreased (P < 0.05 and p < 0.01, respectively), and in the 250-mg/kg/d group, an increased incidence of 13 ribs was observed (P < 0.05). The group that received 6-aminonicotinamide had significant loss of weight in the dams (P < 0.05), decreased fetal weight (P < 0.01), and the fetuses all had major malformations. This study concluded that caprolactam did not induce embryotoxicity or teratogenicity in rats or rabbits. 25
In a 3-generation reproduction study, Fischer 344 albino rats received feed containing 0, 1000, 5000, or 10 000 ppm caprolactam. 26 The dose groups consisted of 10 male rats/group and 20 female rats/group, and each generation was treated over a 10-week period. Decreased body weights and feed consumption were observed in the 5000- and 10 000-ppm dose groups of both the parental generations and the offspring (from in utero through weaning). Significantly decreased body weights were observed in mature rats that received 5000 ppm caprolactam. Additionally, kidney toxicity, which consisted of slightly increased severity of spontaneous nephropathy accompanied by granular casts, was observed in the males of the 10 000 ppm group. No treatment-related effects on gross appearance, gross pathology, survival rate, or number of pups were observed. The study concluded that the no-effect level for caprolactam toxicity was 1000 ppm and the minimum effect level was 5000 ppm. The reproductive toxicity no-effect level for caprolactam was 10 000 ppm.
Nylon-12 Monomer
In an oral gavage study performed in accordance with Organisation for Economic Co-operation and Development testing guideline (OECD TG) 414, pregnant Sprague-Dawley rats received doses of 50, 250, or 1000 mg/kg body weight/d dodecanolactam on days 6 through 19 postcoitum inclusive. 22 In dams of the 250 and 1000 mg/kg dose groups, decreased feed consumption (−7%/−11%) and body weight gains (−37%/−50%) were observed. Clinical signs of toxicity were observed in the high-dose group, resulting in the premature killing of 2 animals. No adverse related effects were observed in the fetuses, including body weight gain, pre- or postimplantation loss, sex ratio, external/skeletal/soft tissue malformations, and variations. The maternal NOAEL was determined to be 50-mg/kg body weight/d and the fetal NOAEL was determined to be 1000 mg/kg body weight/d.
Genotoxicity
No studies were found on the genotoxicity potential of the nylon polymer ingredients. However, the available genotoxicity data for caprolactam and dodecanolactam are summarized in Table 4. In the majority of the assays described, caprolactam and dodecanolactam tested negative. In a previous Cosmetic Ingredient Review (CIR) safety assessment of dicarboxylic acids, adipic acid was not genotoxic in bacterial assays at concentrations up to 10 mg/plate, with or without metabolic activation, or in a yeast gene assay at concentrations up to 200 mg/L. 2 In mammalian cells, adipic acid was not genotoxic at concentrations up to 200 mg/L in human embryonic lung fibroblast cells.
Genotoxicity of nylon Monomers.
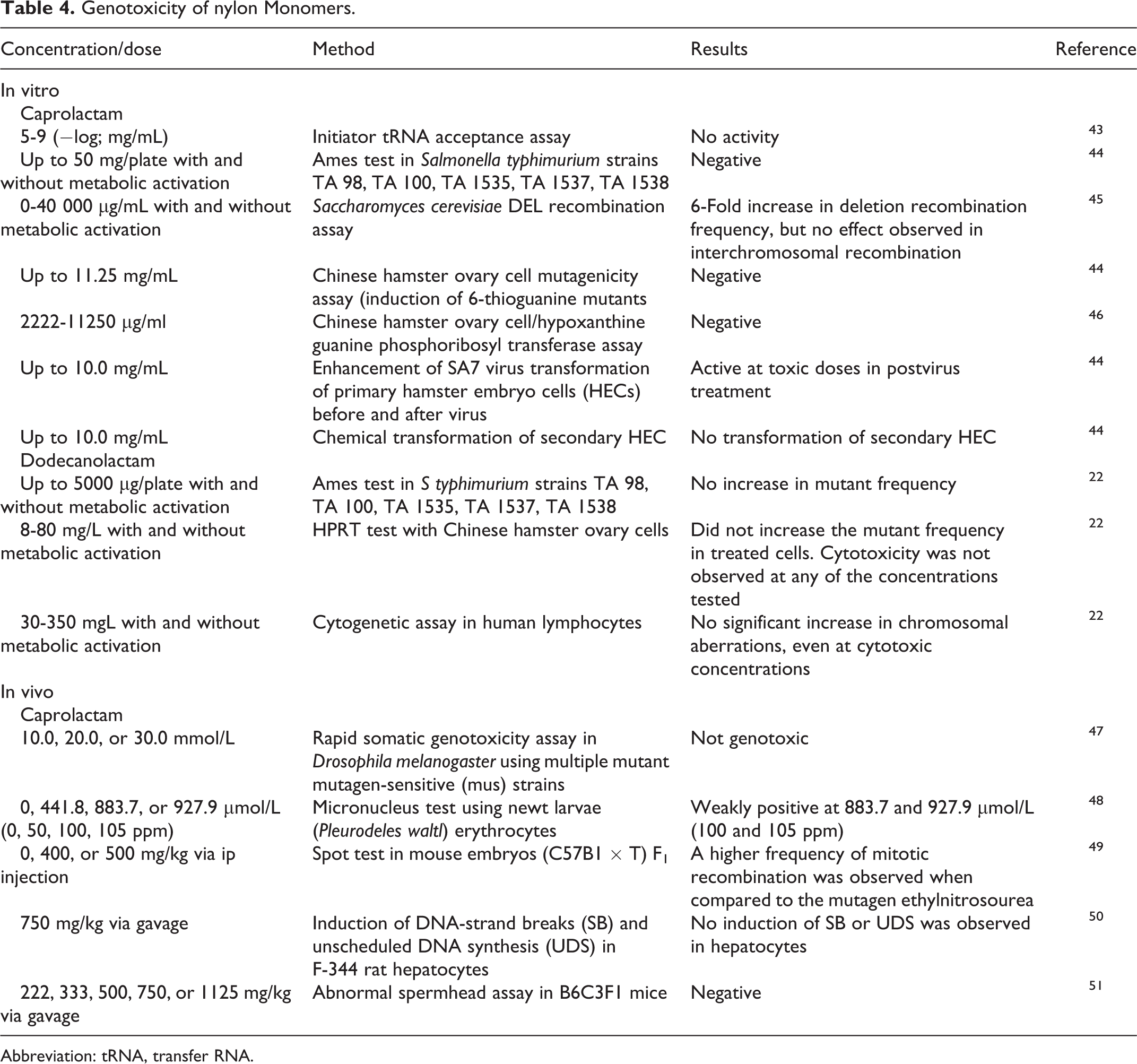
Abbreviation: tRNA, transfer RNA.
Carcinogenicity
Nylon-6 Monomer
The carcinogenic potential of the monomer of nylon-6, ∊ caprolactam, was studied in groups of 50 male and 50 female F344 rats and 50 male and 50 female B6C3F1 mice for 103 weeks. 27 In the feeding study, the rats received 3750 or 7500 ppm of the monomer and the mice received 0, 7500, or 15 000 ppm. During the study, mean body weight gains for rats and mice of either sex receiving the monomer were decreased when compared with those of the controls. No other treatment-related effects were observed. It was concluded that ∊ caprolactam was not carcinogenic for F344 rats and B6C3F1 mice.
The International Agency for Research on Cancer (IARC) determined that caprolactam is probably not carcinogenic to humans (Group 4). 28
Nylon-11 Monomer
The carcinogenic potential of the monomer of nylon-11, 11-aminoundecanoic acid, was studied in groups of 50 male and 50 female F344 rats and 50 male and 50 female B6C3F1 mice for 103 weeks. 29 In the feeding study, the animals received 0, 7500, or 15 000 ppm of the test material. During the study, mean body weight gains for male rats and mice of each sex receiving the monomer were decreased when compared with those of the controls. A dose-related decrease in survival was also observed in these animal groups. Also noted was a dose-related increased incidence of hyperplasia of the transitional epithelium of the kidney and urinary bladder in rats of both sexes. In the dosed mice, mineralization of the kidney was observed in both sexes. In high-dose male rats, neoplastic nodules of the liver and transitional-cell carcinomas of the urinary bladder were observed at significantly increased incidences (P < 0.01) when compared to controls. Low-dose male mice had significantly increased incidences (P < 0.05) of malignant lymphomas. It was concluded that 11-aminoundecanoic acid was carcinogenic in male F344 rats, but not carcinogenic in female F344 rats and female B6C3F1 mice. The results were equivocal for male B6C3F1 mice. The IARC determined that 11-aminoundecanoic acid is not classifiable as to its carcinogenicity to humans (Group 3). 30
Nylon-66 Monomer
As previously reported by CIR, adipic acid was not carcinogenic in a 2-year study in rats fed diets containing up to 5% adipic acid. 2
Irritation and Sensitization
Irritation
Dermal—nonhuman
Nylon-12 monomer
In a study performed according to OECD TG 404 under occlusive conditions, dodecanolactam wetted with paraffin oil caused very mild and transient effects to rabbit skin.22 The average Draize scores for erythema and edema were 0.44 and 0.17, respectively. It was concluded that dodecanolactam was not a skin irritant.
Ocular—nonhuman
Nylon-12 monomer
In an eye irritation study in rabbits following OECD TG 405, dodecanolactam (tested neat) elicited slight conjunctival effects that were most prominent at 48 hours after instillation and completely resolved within 6 days (average Draize scores 1.17 for redness and 0.56 for chemosis). 22 Of 6 animals, 5 showed a slight reddening of parts of the iris at 24 hours; this effect had completely subsided at 48 hours. It was concluded that dodecanolactam was not an ocular irritant.
Ocular—human
Nylon-12
In an ophthalmological in-use safety evaluation of an eye shadow containing 5% nylon-12, 33 female subjects were instructed to apply the product as they normally would at least once a day for 4 weeks. 31 Prior to the start of the study and at study completion, the subjects underwent a complete ophthalmic examination. The subjects kept a daily diary to record use. Approximately 50% of the panel wore hard or soft contact lenses. No adverse events were reported during the study. All ophthalmologic examinations found the subjects’ eyes to be within normal limits throughout the study. It was concluded that an eye shadow containing 5% nylon-12 did not produce ocular irritation and was considered safe for use by contact and non-contact lens wearers, individuals with self-perceived sensitive eyes, and individuals with normal eyes.
Other—nonhuman
Nylon-11
The intracutaneous toxicity of nylon-11 macerated in normal saline, cottonseed oil, 30% ethyl alcohol, or HAE was evaluated in rabbits. 24 Approximately 0.2 mL of each eluate was injected in the back of shaved animals. (No further details were provided). The injection sites were observed for 48 hours for signs of erythema or edema. A slight positive response was observed to the nylon-11 in HAE. No tissue responses were noted in the remaining eluates.
Sensitization
Dermal—nonhuman
Nylon-12 monomer
In a Guinea pig maximization test according to OECD TG 406, irritation was not induced and sensitization was not observed in any of the 20 animals treated with 25% dodecanolactam in corn oil.22 Positive controls were not used in this study.
Dermal—human
Nylon-12
A human repeat insult patch test (HRIPT) of a lip product containing 3% nylon-12 was performed on 30 subjects. 32 The type of patch was not described. No dermal irritation or allergic hypersensitivity to the test material was observed.
An HRIPT was performed on 103 subjects using an eye shadow containing 5% nylon-12. 33 The subjects received 0.2 g of the test material on a 12-in pad. The treatment site was then semioccluded. No adverse events were observed during the induction period or the challenge period. No dermal irritation or allergic contact sensitization was observed to the eye shadow.
In another HRIPT, a concealer containing 6% nylon-12 was evaluated in 103 subjects. 34 The subjects received 0.2 g of the test material on a 12-in pad, which was then semioccluded. No adverse events were observed during the test period. No dermal irritation or allergic contact sensitization was observed to the concealer.
The sensitization potential of a solid perfume containing 5.24% nylon-12 was studied in an HRIPT with 107 subjects. 35 The test material was applied as received under a semiocclusive patch. The study concluded that the solid perfume containing 5.24% nylon-12 did not demonstrate a clinically significant potential for eliciting dermal irritation or sensitization.
In a human maximization assay, the sensitization potential of a facial moisturizer containing 19.5% nylon-12 was evaluated in 26 subjects. 36 The subjects first received 0.1 mL of aqueous 0.25% sodium lauryl sulfate (SLS) on the volar forearm or back on a 15-mm Webril disc that was occluded. After 24 hours, the SLS patch was removed and 0.1 mL of the test material (neat) was applied to the same site and occluded. The induction patch was left in place for 48 to 72 hours and the site was examined for irritation after removal. If no irritation was present, another round of SLS and test material patching occurred for a total of 5 induction exposures. If irritation was observed, the 24-hour SLS patch was eliminated and only test material was reapplied. After a 10-day rest period, the subjects were challenged with a single application of the test material on a new skin site on the opposite arm or side of back. The test site was pretreated with an occluded patch containing 0.1 mL of a 5% SLS solution for 1 hour, which was then replaced with a patch containing the test material. The challenge patch was occluded and remained in place for 48 hours. After the patch was removed, the site was graded at 1 hour and again 24 hours later. No instances of adverse reactions were observed during induction or challenge, and it was concluded that the facial moisturizer containing 19.5% nylon-12 would not likely cause contact sensitivity reactions under normal use conditions.
An HRIPT of a face powder containing 35% nylon-12 was performed on 221 subjects. 37 The subjects received approximately 0.2 g of the test material on a ¾-in squared pad and the treatment site was occluded. The study concluded that the face powder containing 35% nylon-12 did not indicate a potential for dermal irritation or allergic contact sensitization.
Nylon 6/12
An HRIPT was performed on 103 subjects using a face powder containing 1.4% nylon 6/12. 38 The subjects received 0.2 g of the test material on a 12-in pad. The treatment site was then semioccluded. No adverse events were observed during the induction period or the challenge period. No dermal irritation or allergic contact sensitization was observed to the face powder.
Summary
The nylon ingredients evaluated in this safety assessment function primarily as bulking and opacifying agents in cosmetic formulations. Nylon-12 has the most reported uses in cosmetic and personal care products, with a total of 980; 213 of those uses are in eye shadow formulations. Nylon-6 has the second greatest number of overall uses reported, with a total of 61; 31 of those uses are in mascara formulations. Nylon 12 is reported to be used at a maximum concentration range of 0.001% to 35%, with 35% reported in face powder formulations. For nylon-6, the maximum concentration range was reported to be 0.01% to 20%, with 20% reported in eyebrow pencil formulations. No uses or concentrations were reported for nylon-611 or nylon-12/6/66 copolymer.
The nylon ingredients in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union.
Nylon has been used in textiles, such as hosiery, parachutes, tent cloth, and other woven fabrics, thread, tire cord, fishing line, and rope, since the early 1940s. Nylon-6, nylon-11, nylon-12, nylon 6/12, and nylon-66 have been approved as indirect food additives as polymers used for food contact surfaces, and nylon-6 and nylon-66, are used in nonabsorbable surgical sutures.
The acute oral LD50 for nylon-12 was reported to be 1 g/kg in rats, mice, guinea pigs, and rabbits. In cats, the acute oral LD50 for nylon-12 was about 0.25 mg/kg. For the monomer of nylon-12, dodecanolactam, the oral LD50 was 2330 mg/kg and the dermal LD50 was greater than 2000 mg/kg in rats. The acute ip LD50 for nylon-12 was reported to be 0.32 to 0.53 mg/kg in rats. The acute oral LD50 for nylon 6/12 in a corn oil suspension was reported to be greater than 10 g/kg in rats.
In acute studies, no toxic responses were observed in mice that received ip or iv injections of nylon-11 macerated in normal saline, cottonseed oil, or 30% ethyl alcohol. A fall in blood pressure was observed after dosing rabbits in the jugular vein with the nylon-11 in the ethyl alcohol extract.
The NOAEL for dodecanolactam in a 90-day oral gavage in rats was 25 mg/kg. In another repeated dose study, the no-effect level for dodecanolactam was 0.5 g/kg in male rats. The FDA evaluation of dodecanolactam in a 90-day feeding study in Beagle dogs determined that the no significant effect level was 44 mg/kg and the estimated maximum acceptable daily intake in food for man was calculated to be 2.6 mg/kg or 1.74 ppm.
In a subchronic study, rats that received daily ip injections of nylon-11 macerated in HAE had significantly higher white cell count. No signs of toxicity were observed in rabbits and rats that received implant of nylon-11 in pellet form. In rabbits, slight positive responses were observed in intracutaneous studies of nylon-11 in HAE, but negative results were observed in normal saline, cottonseed oil, and 30% ethanol.
While there were no data available on the nylon polymers, data were available on the monomers caprolactam and dodecanolactam. These data largely indicate that these 2 monomers are not genotoxic, with negative results in a bacterial cell assays up to 50 mg/plate in caprolactam and 5 mg/plate in dodecanolactam, and in Chinese hamster ovary cell assays up to 11.25 mg/mL in caprolactam and up to 80 mg/L in dodecanolactam. No significant chromosomal aberrations were observed in a human lymphocyte cytogenetic assay in dodecanolactam up to 350 mg/L. However, dose-dependent increases were observed in recombination frequency in a yeast cell assay when tested with caprolactam up to 40 000 µg/mL. Caprolactam also produced weakly positive results in a newt larvae micronucleus test. In a previous CIR safety assessment of dicarboxylic acids, adipic acid was not genotoxic in bacterial assays at concentrations up to 10 000 mg/plate, with or without metabolic activation, or in a yeast gene assay at concentrations up to 200 mg/L. In mammalian cells, adipic acid was not genotoxic at concentrations up to 200 mg/L in human embryonic lung fibroblast cells.
Caprolactam was not carcinogenic in a 103-week feed study in rats and mice. In a similar study, the monomer of nylon-11, 11-aminoundecanoic acid, neoplastic nodules of the liver, and transitional-cell carcinomas of the urinary bladder were observed in male rats, but not in female rats or mice. The results were equivocal for male mice. As previously reported by CIR, adipic acid was not carcinogenic in a 2-year study in rats fed diets containing up to 5% adipic acid.
Dodecanolactam was not a dermal or ocular irritant in rabbits, nor was it a dermal sensitizer at 25% in a guinea pig maximization test. In human irritation and sensitization studies, nylon-12 was not an ocular irritant nor was it a dermal sensitizer in products that contained the ingredient at concentrations up to 35%. nylon 6/12 was not a dermal sensitizer in a face powder at a concentration of 1.4%.
Discussion
The Panel discussed the issue of incidental inhalation exposure from fragrance spray preparations and face powders. There were no inhalation toxicity data available. The Panel believes that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range. These ingredients are reportedly used at concentrations up to 8% in cosmetic spray products that may be aerosolized and up to 35% in face powders that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered data available to characterize the potential for nylon ingredients or their monomers to cause systemic toxicity, irritation, sensitization, reproductive and developmental toxicity, and genotoxicity. They noted the lack of systemic toxicity at high doses in several acute and subchronic oral exposure studies and 1 chronic oral exposure study, little or no irritation or sensitization in multiple tests of dermal and ocular exposure, the absence of genotoxicity in multiple Ames tests and a Chinese hamster ovary test, and lack of carcinogenicity in a lifetime oral exposure study. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel noted that the size of the polymers would limit significant dermal penetration but expressed concern that limited residual monomer data were available for dermal absorption. The Panel reviewed human repeat patch test data on nylon-12 at its maximum use concentration of 35%. No sensitization or irritation was observed in this study. From these data, the Panel determined that whatever residual monomers may be present in nylon-12 were not present at a sufficient level to cause any biological reactions in test subjects at the maximum use concentration.
Conclusion
The Panel concluded that nylon-6, nylon-11, nylon-12, nylon 6/12, nylon-66, nylon-611, nylon-10/10, and nylon-12/6/66 copolymer are safe in the present practices of use and concentration in cosmetics.
Footnotes
Author Contributions
Burnett contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Heldreth contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript, critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Gill contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Bergfeld contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Belsito contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Hill contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Klaassen contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Liebler contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Marks contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Shank contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Slaga contributed to conception and design, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy. Snyder contributed to conception and design, analysis, and interpretation; critically revised the manuscript, gave final approval; and agreed to be accountable for all aspects of work ensuring itegrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The article in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.