Abstract
The CIR Expert Panel assessed the safety of 6 Cucumis sativus (cucumber)-derived ingredients and found them safe in cosmetic formulations in the present practices of use and concentration. These ingredients are reported to function in cosmetics as skin-conditioning agents. Cucumber is a commonly consumed food with no history of significant adverse effects, suggesting that its ingredients should not pose any major safety issues following oral exposure. This assessment focused on the dermal exposure to the low concentrations of these ingredients as used in cosmetics. Some of the constituents of cucumbers have been assessed previously for safe use as cosmetic ingredients.
Introduction
This document is a safety assessment of the following 6 Cucumis sativus (cucumber)-derived ingredients defined in the International Cosmetic Ingredient Dictionary and Handbook
1
as cosmetic ingredients:
Cucumis sativus (cucumber) fruit extract;
Cucumis sativus (cucumber) extract;
Cucumis sativus (cucumber) fruit;
Cucumis sativus (cucumber) fruit water;
Cucumis sativus (cucumber) juice;
Cucumis sativus (cucumber) seed extract.
All of the ingredients included in this safety assessment are reported to function in cosmetics as skin-conditioning agents.
Cucumis sativus (cucumber) seed oil is not included in this safety assessment because it was previously reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel). In 2011, in the safety assessment of plant-derived fatty acid oils as used in cosmetics, it was concluded that C sativus (cucumber) seed oil is safe as used in cosmetics. 2
The chemical composition of cucumber is provided in this safety assessment, and some of the components of cucumber are cosmetic ingredients for which a CIR safety assessment is available. Others that are not the subject of a CIR safety assessment are compounds that have been discussed in previous CIR safety assessments; for example, some phytosterols were discussed in the safety assessment of polyethylene glycol soy sterols. 3
Published toxicity data were not readily available. However, according to the Food and Drug Administration (FDA), cucumbers are one of the 20 most frequently consumed raw vegetables (21CFR101.44). The fact that cucumber is a commonly consumed food suggests that its ingredients should not pose any major safety issue following oral exposure and argues against the need for oral safety data. Dermal irritation, sensitization, and phototoxicity data were available and are included in this assessment.
Chemistry
Definition
The definition, chemical class, and reported functions of these ingredients are provided in Table 1.
Definitions, Functions, and Chemical Class. 1
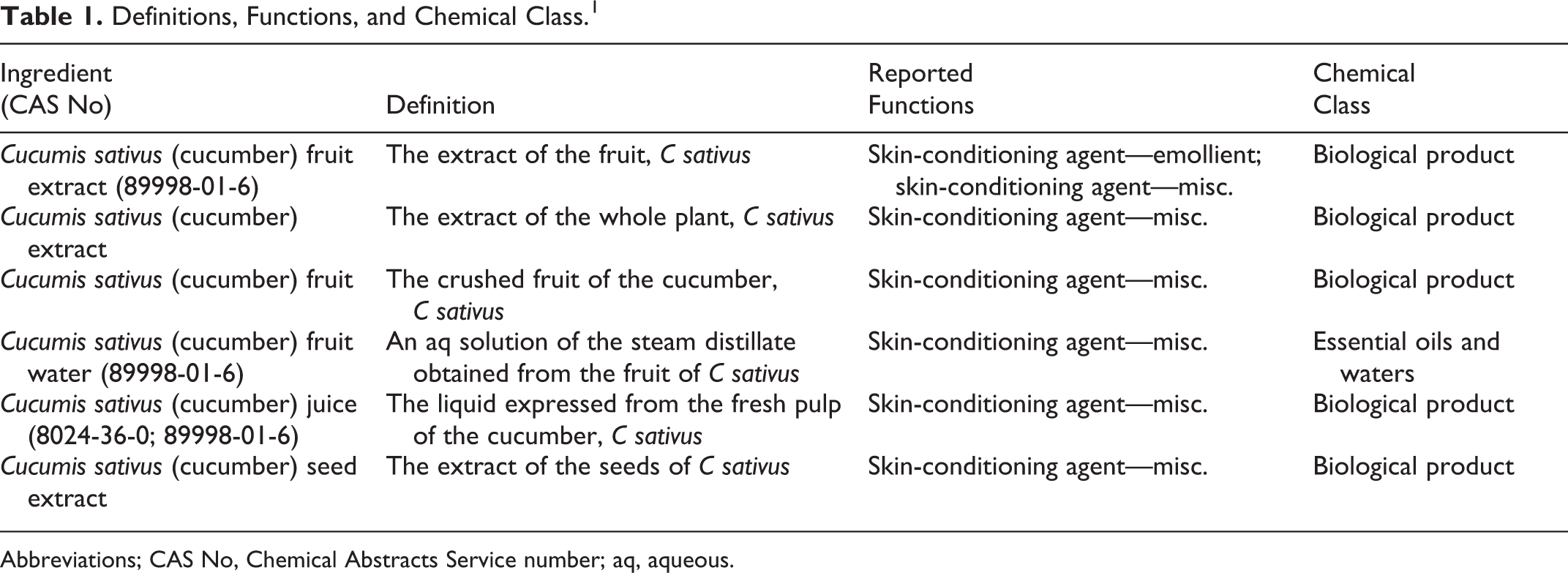
Abbreviations; CAS No, Chemical Abstracts Service number; aq, aqueous.
Chemical and Physical Properties
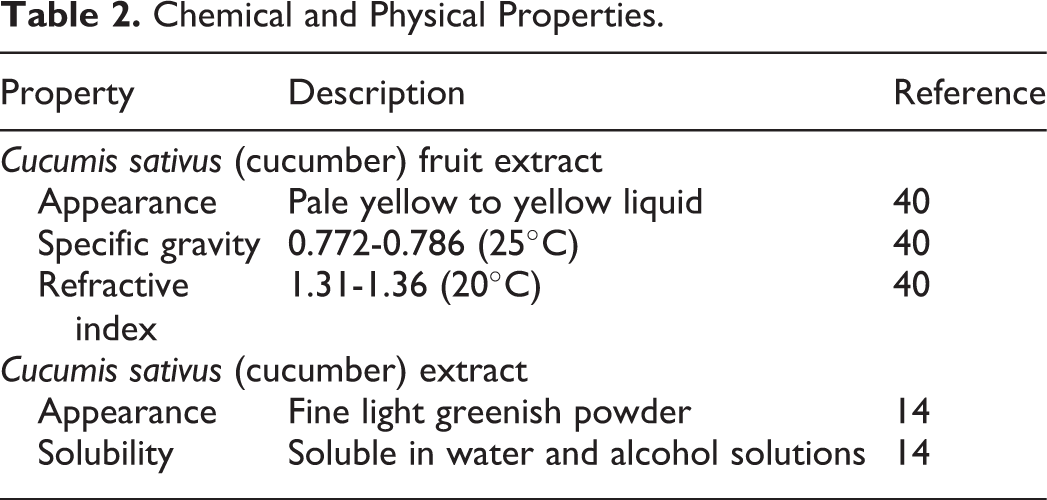
Chemical and physical properties are listed in Table 2.
Chemical and Physical Properties.
Composition
Cucumber fruit is composed mostly of water; that is, more than 96% of the edible unpeeled fruit is water. 4 Other constituents of C sativus L, according to 1 source, are vitamins, minerals, amino acids, phytosterols, phenolic acids, fatty acids, and cucurbitacins. 5 According to another source, traces of essential oil, amino acids, pectins, starch, sugars, vitamin C, and cucurbitacins are found in cucumbers. 6 Glycosides, steroids, flavonoids, carbohydrates, terpenoids, and tannins were identified in an aqueous extract of the cucumber fruit. 7 A comprehensive list of chemical constituents by plant part is presented in Table 3.
Chemical Constituents by Plant Part. 41

Abbreviation: NS, not specified.
Liquid chromatography–mass spectrometry that incorporated 13 C3-labeled standards determined that cucumber contained 12 to 13 μg phytoestrogens/100 g wet weight (wt) cucumber. 8 In the breakdown of the phytoestrogen composition, the content was primarily the lignan secoisolariciresinol; the lignan matairesinol, the isoflavones daidzein, genistein, glycitein, biochanin A, and formononetin, and coumestrol comprised <1 µg/100 g wet wt of the fruit. Another source reports the following phytosterols in cucumber fruit (amount is per 100 g edible portion): 3800 µg β-sitosterol, 200 µg campesterol, 2900 µg stigmasterol, 300 µg β-sitostanol, and 100 µg campestanol, giving a total plant sterol content of 7300 µg/100 g edible portion. 9
The lipid fatty acid content of C sativus (cucumber) has been described, and Table 4 provides information on cucumber lipids and their fatty acid composition. The major fatty acids in cucumbers are palmitic acid (23.6%-27.5%), linoleic acid (22.7%-26.3%), and linolenic acid (40%-46%). 10,11
Cucumis sativus (Cucumber) Lipids and Their Fatty Acid Composition.
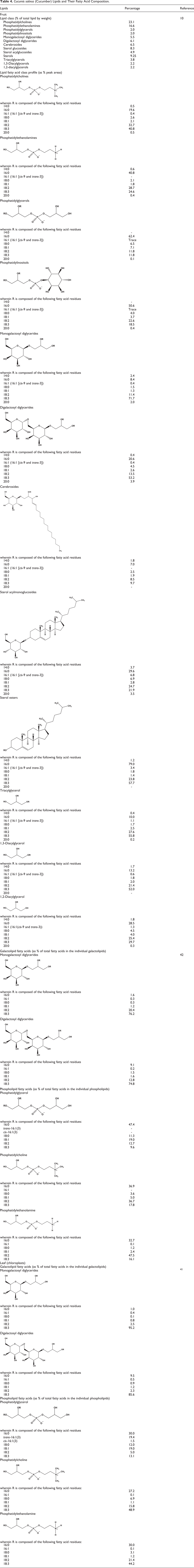
The mixed fatty acid and triterpene alcohol composition of C sativus seeds is presented in Table 5. For extrapolation purposes, according to the CIR safety assessment on plant-derived oils as used in cosmetics, the fatty acid composition of C sativus (cucumber) seed oil is 9% to 13% palmitic acid, 6% to 9% stearic acid, 14% to 20% oleic acid, 60% to 68% linoleic acid, and <1% linolenic acid. 2
Cucumber Seed Composition.

Table 6 provides the conclusions from published CIR safety assessments that exist for some of the constituents of cucumber. Table 7 references information on the safety of some components of cucumber that were discussed in previous CIR reports.
Conclusions of CIR Safety Assessments on Ingredients That Are Constituents of Cucumber.

Abbreviation: CIR, Cosmetic Ingredient Review.
Toxicity Information on Some Components of Cucumber as Discussed in Previous CIR Reports.

Abbreviations: CIR, Cosmetic Ingredient Review; IARC, International Agency for Research on Cancer; UV, ultraviolet.
Preparation/Extraction
Cucumis sativus (cucumber) fruit extract is reported to be manufactured by extracting cucumber fruit in mixtures of glycerin and water, 12 water and butylene glycol, or water and propylene glycol 13 or by hydroalcoholic extraction. 14
Use
Cosmetic
The C sativus-derived ingredients included in this safety assessment are reported to function in cosmetics as skin-conditioning agents. 1 The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2012 indicate that C sativus (cucumber) fruit extract is used in 575 cosmetic formulations, 377 of which are leave on. 15 The VCRP data indicate that the other C sativus-derived ingredients are each used in no more than 11 cosmetic formulations. A Personal Care Products Council (Council) survey of the maximum reported use concentrations found that the C sativus (cucumber) fruit water had the highest concentration of use, at 3% in foundations, and that C sativus (cucumber) fruit extract had the next highest concentration of use, at up to 1% in eye lotions and face and neck. 16
Frequency and concentration of use data categorized by exposure and duration of use are provided in Table 8. In some cases, reported use was received by the VCRP, but no concentration of use data were reported in the Council survey; that is, C sativus (cucumber) fruit and C sativus (cucumber) juice are reported to be used according to VCRP data but no concentration of use data were submitted in response to the Council survey. In another case, C sativus (cucumber) fruit water had only 1 use reported in the VCRP but the industry survey indicates that it is used at 3% in foundations and at 0.05% in bath soaps and detergents and in body and hand product formulations. It should be presumed that C sativus (cucumber) fruit water is used in at least 1 formulation in each of these categories.
Frequency and Concentration of Use According to Duration and Type of Exposure.
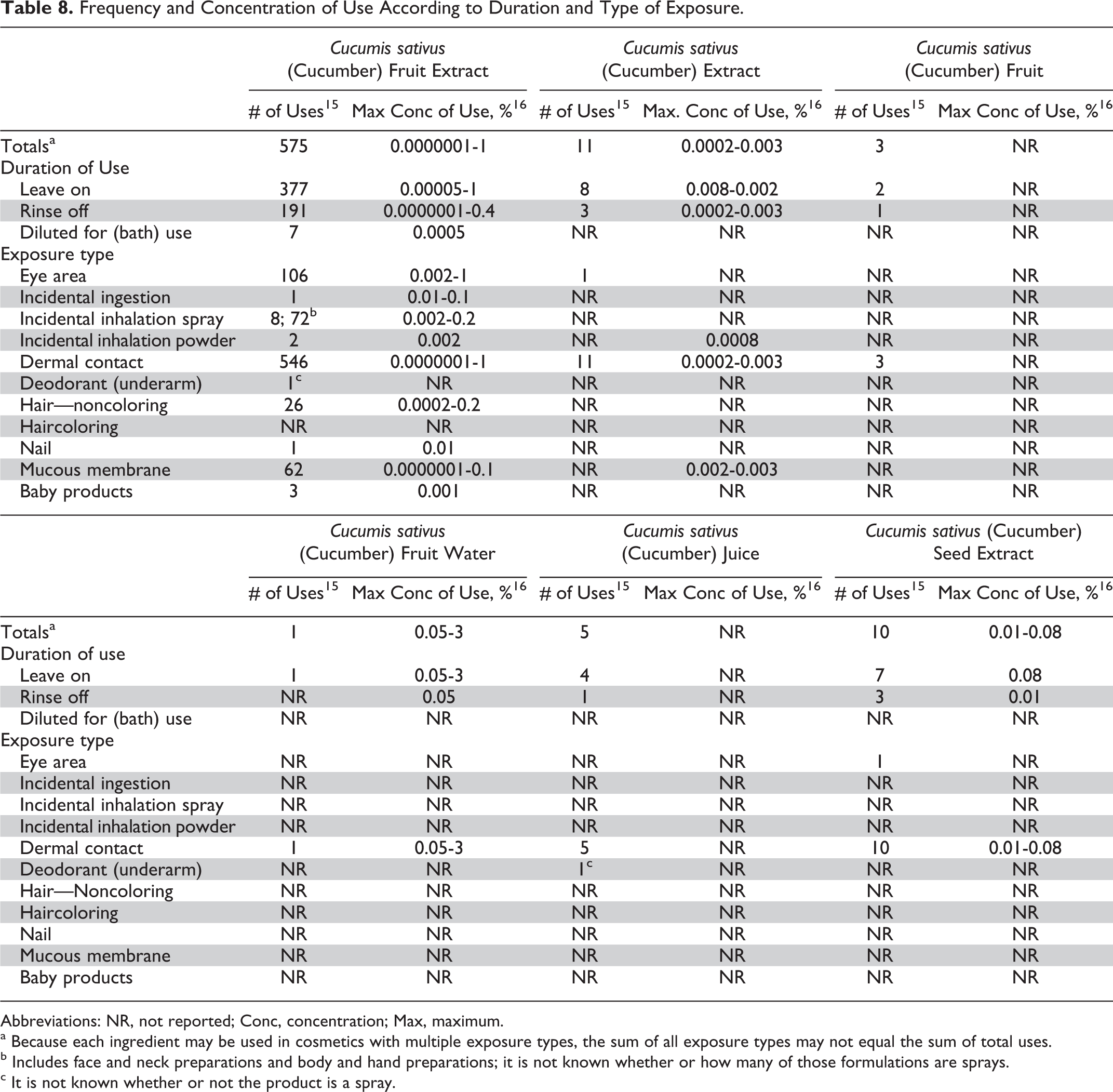
Abbreviations: NR, not reported; Conc, concentration; Max, maximum.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b Includes face and neck preparations and body and hand preparations; it is not known whether or how many of those formulations are sprays.
c It is not known whether or not the product is a spray.
Products containing C sativus (cucumber) fruit extract are reported to be used on baby skin, may be applied to the eye area or mucous membranes, or could be incidentally ingested. Cucumis sativus (cucumber) fruit extract is also used in cosmetic spray products such as face and neck and body and hand sprays and could possibly be inhaled. This ingredient is reportedly used at concentrations up to 0.2% in these cosmetic sprays. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 17 -20 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 17,20 Additionally, C sativus (cucumber) fruit extract is listed in the VCRP as having 1 use in a deodorant and it is not known whether this product is a spray. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 17 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
All of the C sativus-derived ingredients named in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 21
Toxicological Studies
Published toxicity studies were not found.
Reproductive and Developmental Toxicity
Published reproductive and developmental toxicity studies were not found.
Genotoxicity
Cucumis sativus (Cucumber) Fruit Extract
An Ames test was performed with C sativus (cucumber) fruit extract composed of 54.8% water, 45% butylene glycol, and 0.2% cucumber. 22 Doses of 156 to 5000 µg/plate were assayed using Salmonella typhimurium strains TA100 and TA98 with and without metabolic activation. Negative and positive controls gave valid results. Cucumis sativus (cucumber) fruit extract was not mutagenic in this assay.
Cucumis sativus (Cucumber) Fruit
The mutagenic potential of raw cucumber was evaluated in an Ames test using S typhimurium TA98 and TA100 with and without metabolic activation. 23 The cucumber was washed, peeled, trimmed, and cored. A 250-g sample was blended with 500 mL water and fractionated; fractions were obtained with dimethyl sulfoxide (fraction 1) or chloroform and n-butanol (fraction 5), water (fraction 7), methanol (fraction 3), or hexane (fraction 4). None of the cucumber fractions were mutagenic in this assay.
Carcinogenicity
Antitumor Promotion
The effect of C sativus (cucumber) on dermal tumor promotion was examined in Swiss Webster albino mice. 24 The test article was prepared by homogenization of the fruit and expressing the juice. Ten mice (sex not specified) were used per group. The mice were shaved and 6 days later 0.2 mL of 410 µg of dimethylbenz[a]anthracene (DMBA) in acetone was applied to the back of each mouse. Four days after DMBA application, 0.2 mL of 0.03% croton oil in acetone was applied to the shaved back of each animal; this application was made 3 times per week for 20 weeks. Three protocols were used for the application of the cucumber extract. In protocol 1, the extract was applied for 5 days prior to application of DMBA and 1 hour before the croton oil. In protocol 2, the extract was applied 1 hour before the croton oil. In protocol 3, the extract was applied immediately after the croton oil dried. Initially, a dose of 5.0 mg cucumber extract/0.2 mL acetone was “splashed on” the back of each animal. However, this reportedly caused 60% to 80% mortality prior to tumor development. (This unexplained outcome was observed with the 3 other test articles, that is, sugar beet roots, New Zealand spinach leaves, and turmeric rhizomes, at this dose as well. It was the opinion of the Panel that the observed mortality was due to a flaw in the study and not the test article). As a result, the dose was changed to 2.5 mg cucumber extract/0.2 mL acetone. The positive control group was exposed to DMBA + croton oil and the negative control group was untreated.
In the positive control group, none of the mice died before developing tumors (0% mortality) and the first tumor appeared at week 9. The tumor incidence was 100%, and the average number of tumors/mouse was 4.7 ± 3.3. The time for the first tumor to appear in all 3 test groups was delayed, appearing at week 12, for all 3 protocols using cucumber extract. Also, the tumor incidence was lower in all 3 groups exposed to cucumber extract. Using protocol 1, mortality was 0%, the tumor incidence was 70%, and the average number of tumors per mouse was 2.3 ± 1.1 (using Kruskal-Wallis analysis of variance by ranks, statistical difference from positive control, α = 0.20). Using protocols 2 and 3, mortality was 10% for both, the tumor incidence was 55.6% and 66.7%, respectively, and the average number of tumors/mouse was 4.2 ± 0.8 and 3.8 ± 1.3, respectively (statistical difference from positive control, α = 0.70 and 0.90, respectively). No tumors were reported in the negative control group.
Irritation and Sensitization
Skin Irritation/Sensitization
Human
Cucumis sativus (cucumber) fruit extract
A single insult patch test was performed on 20 patients with C sativus (cucumber) fruit extract composed of 54.8% water, 45% butylene glycol, and 0.2% cucumber. 22 The extract was diluted in water to 1% and 15 µL were applied for 24 hours under an occlusive patch. No erythema or edema was observed at 24 or 48 hours.
To test for dermal irritation, cosmetic formulations containing 0.5% to 2.5% ethanol extract of C sativus were prepared as oil-in-water emulsion-based creams, with stearic acid as the emulsifier. 25 The pH of the 7 formulations that were prepared ranged from 6.4 to 6.9. The irritancy was evaluated by applying the creams to a 1 cm2 area on the dorsal surface of the hand and observing signs of irritation for 24 hours. (The number of patients tested was not stated.) No irritation, erythema, or edema was observed.
Summary data from a 21-day use study in which 21 patients applied an eye gel containing 5% C sativus (cucumber) fruit extract 1 to 2 times daily were provided to the CIR. 26 Eight patients reported sensations of discomfort (primarily stretching) after application but no clinically significant cutaneous reactions were observed. Dermal assessments were performed, and it was concluded that the eye gel was well tolerated. (No other details or raw data were provided).
A 28-day use study was performed to determine the dermal irritation potential of an eye lotion containing 1% C sativus (cucumber) fruit extract. 27 Thirty female patients were instructed to apply the test material under the eye area and to the eyebrow area, avoiding the eyelid, up to 2 times daily for 4 weeks. The skin in the eye area was evaluated for dermal effects. One patient reported slight itching on the eyelids almost daily. The eye lotion containing 1% C sativus (cucumber) fruit extract did not demonstrate a potential for eliciting dermal irritation in the eye area.
The irritation and sensitization potential of 2 formulations containing 0.00055% C sativus (cucumber) fruit extract was evaluated in a modified occlusive human repeat insult patch test (HRIPT). A 21-day induction phase, 10- to 24-day nontreatment period, and a 4-day challenge phase were used. Distilled water was the negative control and sodium lauryl sulfate (SLS) was the positive control in both the studies. In the first study, a moisturizer containing 0.00055% C sativus (cucumber) fruit extract was applied neat to 101 patients. 28 The standardized cumulative irritation score was 0 for both the test material and distilled water and was 2430 for 0.5% SLS. (The scoring scale was not defined.) The formulation containing 0.00055% C sativus (cucumber) fruit extract was not predicted to be a significant skin irritant and it was not a sensitizer.
In the second study, a facial cleanser containing 0.00055% C sativus (cucumber) fruit extract was applied to 104 patients at a concentration of 1%. 28 The standardized cumulative irritation index was 96.15 for the test material, 58.65 for distilled water, and 1659.62 for 1% SLS. (The scoring scale was not defined.) The formulation containing 0.00055% C sativus (cucumber) fruit extract, tested at 1%, was not predicted to be a significant skin irritant and it was not a sensitizer.
An HRIPT was completed in 103 patients to determine the dermal irritation and sensitization potential of an eye lotion containing 1% C sativus (cucumber) fruit extract. 29 The test article was applied neat. During induction, a 24-hour semiocclusive patch was applied to the upper back of each patient 3 times per week for 3 weeks. Challenge patches were applied after a 2-week nontreatment period and the test sites were evaluated upon patch removal and at 48 and 72 hours. No reactions were observed during induction. In all, 5 patients (2 at 48 hours, 2 at 72 hours, and 1 at 48 and 72 hours) had a ± reaction (barely perceptible erythema) and 1 patient had a 1+ reaction (mild diffuse erythema) observed at 24 hours, which subsided to a ± reaction at 48 hours and no reaction at 72 hours. The researchers concluded that the eye lotion containing 1% C sativus (cucumber) fruit extract demonstrated no potential for eliciting dermal irritation or sensitization.
Another HRIPT was performed in 108 patients to determine the irritation and sensitization potential of an eye lotion containing 1% C sativus (cucumber) fruit extract. 30 Approximately 0.2 mL of the test material was applied neat to a 1 × 1 in semiocclusive patch, and the induction patches were applied for 24 hours to the upper back of each patient 3 times per week for 3 weeks for a total of 9 applications. Challenge patches were applied after a 2-week nontreatment period. Two patients had reactions at challenge. One had a mild response 24 hours, but not 72 hours, postchallenge; this was considered a transitory response and therefore clinically insignificant. The second patient had a moderate to mild response 24 and 72 hours postchallenge. A rechallenge was performed and included an open repetitive application to the forearm for 4 consecutive days. No reactions were observed at rechallenge. An eye lotion containing 1% C sativus fruit extract was not considered a dermal irritant or sensitizer.
The irritation and sensitization potential of an eye treatment mask containing 1% C sativus (cucumber) fruit extract was evaluated in a modified HRIPT that was completed in 600 patients. 31 The product was applied neat. 32 During induction, which consisted of ten 48-hour occlusive patches on the back of each patient, the test sites were scored for immediate reactions and then 1 to 2 hours after patch removal for delayed reactions. 31 Challenge patches (48 hours) were applied after a 2-week nontreatment period and again 1 week later. During challenge, the test sites were evaluated for immediate and delayed reactions. No reactions were observed and an eye formulation containing 1% C sativus (cucumber) fruit extract was not a dermal irritant or a sensitizer.
Summary data from an HRIPT that was completed in 100 patients to examine the irritation and sensitization potential of an eye hydrogel containing 5% C sativus (cucumber) fruit extract were provided to the CIR. 33 The undiluted test material was applied to the upper backs of patients using an occlusive patch. Mild erythema with or without edema was observed in 6 patients; these responses either decreased from the 48- to 96-hour evaluation or were not confirmed at both challenge sites. The responses were considered irritant responses, and the formulation did not induce clinically identifiable evidence of contact hypersensitivity (no other details or raw data were provided).
Phototoxicity
Human
Cucumis sativus (cucumber) fruit extract. Summary data from a study that was initiated in 11 patients to examine the phototoxicity potential of an eye gel containing 5% C sativus (cucumber) fruit extract were provided to the CIR. 34 Duplicate 24-hour occlusive patches were applied to the mid-back of each patient. Upon patch removal, 1 site was irradiated with 2/3 of the minimal erythema dose of ultraviolet (UV) A (UVA) and UVB, supplemented with 10 J/cm2 UVA. Both sites were evaluated for erythema at 10 minutes and 24, 48, and 72 hours after irradiation. In all, 9 patients completed the study; 2 withdrew for reasons not related to the study. An eye gel containing 5% C sativus (cucumber) fruit extract was not phototoxic (no raw data or other details were provided).
Cross-Allergenicity
Cross-allergenicity among cucumber, celery, carrot, and watermelon was investigated. 35 The pooled sera of 6 individuals that had demonstrated allergy to one or more of these foods in an enzyme-linked immunosorbent assay (ELISA) were used. At least 2 of the patients were symptomatic and also skin test—or radioallergosorbent test (RAST)—was positive to cucumber, celery, carrot, and watermelon as index foods. A strong allergenic cross-reactivity among these 4 foods was demonstrated by both ELISA inhibition and immunoblot inhibition studies.
Researchers studied the relationship between ragweed allergens and allergens found to the gourd family (including cucumber) in patients with sensitization to ragweed. 36 The researchers suggested that most oropharyngeal symptoms associated with these foods are mediated in part by an immunoglobulin E (IgE) mechanism and that ragweed most likely shares allergens with the entire gourd family. A case of cross-reactivity of cucumber with latex has been reported. 37 Details are provided in the section on “Case Studies”.
Case Studies
A male greenhouse employee who worked with cucumber plants developed severe eczema 5 months after starting work. 38 Patch testing was performed with cucumber leaves, stem, and peel and with a cucumber ethanol extract. A positive reaction (++) to the upper and under side of the cucumber leaf was reported on days 2, 3, and 7. A follicular reaction to the stem and ethanol extract was reported on days 3 and 7. No reaction was reported with the cucumber peel. In follow-up testing with cucumber leaves in 10 healthy individuals, slight redness was reported for 2 patients.
Cucumber anaphylaxis, demonstrated by dizziness, vomiting, dyspnea, thoracic erythema, and vaginal itching, was reported in a female patient within 5 minutes of eating an incompletely peeled cucumber. 37 Three months prior to this reaction, the patient had presented with an episode of papaya urticaria and a sensitization to latex was found. Prick-by-prick skin test results were positive for cucumber (peel and pulp) as well as for papaya and some other fruits and vegetables. Immunoblot inhibition confirmed latex-cucumber (and latex-papaya) cross-reactivity.
Ocular Irritation
In Vitro
Cucumis sativus (cucumber) fruit extract
The ocular irritation potential of a moisturizer containing 0.00055% C sativus (cucumber) fruit extract was evaluated in a chorioallantoic membrane vascular assay (CAMVA) and a bovine corneal opacity and permeability test (BCOP). 28 The material was tested undiluted in both assays. In the CAMVA, the RC50 was 66%. In the BCOP, the in vitro score was 2.62, the opacity score was 2.6, and the permeability score was 0.001. (No details were provided). The results of both of these assays predict the test material is not to irritating to the eye.
The potential ocular toxicity of an eye lotion containing 1% C sativus (cucumber) fruit extract was determined in a screening assay using the EpiOcular human cell construct. 39 The test material was tested as supplied. Sterile deionized water served as the negative control and 0.3% triton-X-100 served as the positive control. The duration of exposure resulting in a 50% decrease (ET50) in 3-[4,5-dimetylthiazol-2-yl]-2,5-diphenlytetrazolium bromide conversion) was >1440 minutes. (The ET50 of the positive control was 27.5 minutes).
Human
Cucumis sativus (cucumber) fruit extract
The 2 use studies described in the section on “Dermal Irritation and Sensitization” also examined the ocular irritation potential of those products. The summary from the 21-day use study in which 21 patients applied an eye gel containing 5% C sativus (cucumber) fruit extract reported that the eye gel was well tolerated.
In the 28-day in-use study of an eye lotion containing 1% C sativus (cucumber) fruit extract in 30 female patients, half of which wore contact lenses, an ophthalmic examination was made prior to and at the termination of testing. 27 Trace increases in redness of the palpebral conjunctivae were observed in 2 patients and of the bulbar conjunctivae were observed one patient; these observations were not attributed to the test product. The eye lotion containing 1% C sativus (cucumber) fruit extract did not demonstrate a potential for eliciting ophthalmic irritation.
Summary
This assessment pertains to the safety of the following 6 ingredients as used in cosmetic formulations: C sativus (cucumber) fruit extract, C sativus (cucumber) extract, C sativus (cucumber) fruit, C sativus (cucumber) fruit water, C sativus (cucumber) juice, and C sativus (cucumber) seed extract. These ingredients are reported to function in cosmetics as skin-conditioning agents. Cucumis sativus (cucumber) fruit extract is used in 575 cosmetic formulations; the other C sativus (cucumber)-derived ingredients are used in 11 formulations or less each. The highest reported use concentrations were 3% C sativus (cucumber) fruit water in foundations and 1% C sativus (cucumber) fruit extract in eye lotions and face and neck products; all other reported use concentrations (in leave on, rinse off, and diluted for [bath] use formulations) were less than 0.4%.
Cucumis sativus contains, among other constituents, fatty acids, vitamins, amino acids, phytosterols, phenolic acids, and cucurbitacins. Glycosides, steroids, flavonoids, carbohydrates, terpenoids, and tannins were isolated in an aqueous extract. The major fatty acids in cucumbers are palmitic acid (23.6%-27.5%), linoleic acid (22.7%-26.3%), and linolenic acid (40%-46%). Several of the chemical constituents have previously been assessed for safety as used in cosmetics.
Results of a dermal application study with C sativus (cucumber), prepared by homogenization of the fruit and expressing the juice, suggested that the exposure to cucumber delayed the onset and decreased the incidence of tumors in a tumor promotion assay using DMBA and croton oil in mice. However, this study also reported a high unexplained mortality rate when a higher dose of cucumber (and other test materials) was tested.
Cucumis sativus (cucumber) fruit extract was not mutagenic in an Ames assay when tested at doses of 156 to 5000 µg/plate with and without metabolic activation. Five fractions of raw cucumber were not mutagenic in an Ames test.
Cucumis sativus (cucumber) fruit extract, containing 0.2% cucumber, was not an irritant in a single insult patch test when tested as a 1% aqueous dilution, and cosmetic formulations containing 0.5% to 2.5% ethanol extract of C sativus, prepared as oil-in-water emulsion-based creams, were not irritants when applied for 24 hours. In a 21-day use study and a 28-day use study of an eye gel containing 5% C sativus (cucumber) fruit extract and an eye lotion containing 1% C sativus (cucumber) fruit extract, respectively, no dermal irritation was reported. Cosmetic formulations containing up to 1% C sativus (cucumber) fruit extract were not dermal irritants or sensitizers in clinical testing. In an HRIPT with a formulation containing 5% C sativus (cucumber) fruit extract, reactions considered an irritant response were observed during challenge, but the formulation did not induce clinically identifiable evidence of contact hypersensitivity.
In a clinical phototoxicity study completed in 9 patients, a formulation containing 5% C sativus (cucumber) fruit extract was not phototoxic.
Cross-allergenicity among cucumber, celery, carrot, and watermelon has been demonstrated as has a correlation between the ragweed pollen-specific IgE and the Cucurbitaceae-specific IgE and the specific IgE to Cucurbitaceae and to banana. A case of cross-reactivity of cucumber with latex has been reported.
In vitro ocular irritation testing predicted that C sativus (cucumber) fruit extract would not be an ocular irritant. In 21- and 28-day use studies, an eye gel containing 5% C sativus (cucumber) fruit extract and an eye lotion containing 1% C sativus (cucumber) fruit extract, respectively, did not demonstrate a potential for eliciting ocular irritation.
Discussion
The Panel recognized that cucumber is a commonly consumed food with no history of significant adverse effects. This fact suggests that its ingredients should not pose any major safety issue following oral exposure and argued against the need for oral toxicity data. Therefore, the focus of this safety assessment was on the dermal exposure to these C sativus (cucumber)-derived ingredients. The Panel noted that many of the constituent chemicals previously have been assessed for safe use in cosmetic formulations.
Skin sensitization and phototoxicity testing of a formulation containing 5% C sativus (cucumber) fruit extract (which is greater than the highest reported use concentration of 1%) demonstrated an absence of sensitization and phototoxicity potential. An irritant response to the formulation containing 5% C sativus (cucumber) fruit extract was observed in some patients, but no irritation was observed with cosmetic formulations containing up to 2.5% of an ethanol extract of C sativus prepared as an oil-in-water emulsion-based cream or with a formulation containing 1% C sativus (cucumber) fruit extract.
Because C sativus (cucumber) fruit extract can be used in products that may be aerosolized, including face and neck sprays and body and hand sprays, the Panel discussed the issue of incidental inhalation exposure. In the absence of inhalation data, the Panel noted that C sativus (cucumber) fruit extract caused no irritation at concentrations up to 2.5% or sensitization. Further, this ingredient is reportedly used at concentrations of ≤0.2% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. Nevertheless, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Cucumis sativus, and therefore derived extracts, contains a variety of phytochemicals, all present at relatively low concentrations. Although certain components of these extracts could exert significant biological effects (eg, isoflavones), the low levels that are present and the low use concentrations of the ingredients preclude significant effects. Also, although no dermal absorption data were available, it is the experience of the Panel that phytosterols and phytosterol esters are not significantly absorbed and do not result in systemic exposure. Additionally, the Panel noted that diacylglycerols are present as components of the lipids of cucumber fruit, but in an amount that is below the threshold for toxicological concern.
The Panel discussed the published tumor promotion study that reported a high level of mortality in mice after a dose of 5.0 mg cucumber extract in 0.2 mL acetone was applied to the skin, noting that the high mortality was also observed with other test articles that were evaluated. After extensive evaluation, the Panel stated that this study had sufficient methodological flaws to render the results not relevant to assessing the safety of cucumber extract in cosmetics.
Finally, the Expert Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
Conclusion
The Panel concluded that C sativus (cucumber) fruit extract, C sativus (cucumber) extract, C sativus (cucumber) fruit, C sativus (cucumber) fruit water, C sativus (cucumber) juice, and C sativus (cucumber) seed extract are safe in cosmetic formulations in the present practices of use and concentration.
Footnotes
Auhtor’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
