Abstract
Droplet-based microfluidics holds enormous potential for transforming high-throughput drug screening. Miniaturization through droplets in combination with automation contributes to reduce reagent use and analysis time as well as minimizing or eliminating labor-intensive steps leading to associated reductions in cost. In this paper, we demonstrate the potential of automated and cost-effective microfluidic droplet-generating technology in the context of an enzymatic activity assay for screening collagenase inhibitors. Experimental results show reproducible and accurate creation and mixing of droplet combinations resulting in biochemical data comparable to data produced by an industry standard instrument. This microfluidic platform that can generate and combine multiple droplets represents a promising tool for high-throughput drug screening.
Introduction
The current cost of bringing a new drug to market is estimated to be in excess of $2.5 billion. 1 Interestingly, of the compounds that make it successfully through the preclinical phase, on average only 10% will make it through the clinical trials and on to market. For oncology drugs, this figure is as low as 5%. 2 Drug development involves several stages, such as drug discovery, preclinical tests, and clinical trials, to develop a new drug for commercialization. The first of these is the drug discovery and development phase, which requires rapid analysis of compound libraries containing tens of thousands of possible drug candidates and is undertaken using high-throughput screening (HTS). HTS is a complicated process that faces challenges from significant issues such as time consumption, cost, and data management.
A large proportion of the costs come from the high capital expenditures associated with the HTS equipment required to undertake compound screening (robotics for liquid and plate handling) as well as equipment suites for compound storage, liquid handling, assay incubation, and plate reading. 3 Biological samples and drug libraries further increase costs. Reducing costs at the preclinical phase would have a significant impact on the overall cost of drug development. Many autonomous researchers, whose work involves screening for potential new drug molecules, are constrained by the high cost of the current HTS systems. The rapid development of these technologies is in marked contrast to the decline in the rate of blockbuster drugs being brought to market in recent years.
There are also further disadvantages to using current HTS systems, including limitations in the accuracy of dispensing volumes less than 1 µL. 4 The 96-well microplate is currently the standard format of miniaturization and automation for assays in drug discovery. 5 Reducing assay volumes by increasing microplate formats in size, for example, from 96 wells to 384 or 1536 wells, is advantageous; 6 however, there are also further limitations with the use of microplates, such as evaporation and dead volume, while edge effects can become more pronounced as assay volumes decrease. Liquid handling problems from carryover of storage liquids such as DMSO and heating microplates from room temperature to 37 °C can also cause artifacts and increase assay coefficients of variation (CVs) as the temperature gradient across the plate will cause different reaction rates depending on the assay. 7 In addition, the logistics of the experimental setup of drug combination screening in microplates can significantly restrict the use of robust experimental designs
A low-cost HTS technology that has low sample and reagent consumption and is automated is an urgent requirement for the drug discovery industry. Droplet-based microfluidics has been proposed as an enticing technology for drug screening due to its low sample usage, low cost, and easy handling of nanoliter volumes, and it has been proven useful in high-throughput drug screening, gene analysis and sequencing, single-cell RNA sequencing, and vaccine screening.8–10 Droplets can be created from a range of fluids, which act as individual compartments transported by a carrier oil. Aqueous fluids immersed in an immiscible organic fluid, in this case silicone oil, will be referred to as droplets. There are many advantages to carrying out assays in droplets on the microliter, nanoliter, and picoliter scale. Furthermore, typical consumables (pipette tips, microplates) are often not required. Compared with microplates and robotic screening technology, droplet microfluidics have demonstrated up to a 1000-fold increase in the throughput and a million-fold reduction in cost for applications such as the directed evolution of enzymes. 11 Minimizing the volume of compounds/drugs screened has a substantial economic saving, while a reduction in biological sample volume is beneficial. Chip-based microfluidics are widely employed in the pharmaceutical industry as a consumable. 12 Although such devices are capable of generating droplets at high rates, 13 these droplets consist of only one or two fluids, which restricts applications of droplet microfluidics for high-throughput drug discovery. The Labcyte Echo (San Jose, CA) with its acoustic droplet ejection technology uses sound energy to transfer small-volume liquid droplets to microplates and is an interesting solution to droplet manipulations. Variations of the Echo liquid handlers have been used in many combination drug screening applications over the last decade.14–16 Automated microfluidic platforms such as DropLab 17 and SODA 18 systems are based on a tapered capillary–syringe pump module design coupled to an oil-covered 2D array and a 384 microplate on an X-Y-Z translation stage. Both can achieve automated droplet manipulations with picoliter precision. The Ismagilov group19,20 has developed a cartridge system preloaded with nanoliter plugs of reagents using two pumps and a merging junction. Even with the advancements of the microfluidic systems described, platforms with the ability to combine and mix multiple compounds are uncommon and highly sought after.
This study demonstrates a scalable, high-throughput, droplet-generating device, the Gap Switch Technology (GST), which can mix a variety of fluids. The ability to mix multiple fluids is a key feature that makes the system ideal for screening drug combinations or potential drug compounds. The linear system described in this paper has been developed using polytetrafluoroethylene (PTFE) tubing, which minimizes manufacturing costs and allows assay volumes of <1 µL. This technology has the ability to perform biochemical assays flexibly as demonstrated in this study using Molecular Probes EnzChek Collagenase Assay (Biosciences, Dublin, Ireland). The assay can be used as an indicator of the functionality of matrix metalloproteinase (MMP) inhibitors to be developed as therapeutics.
Materials and Methods
Device Fabrication and Materials
The GST is a four-part assembly consisting of wells, base, shuttle, and slider as shown in
Figure 1
. An image of this assembly is show in
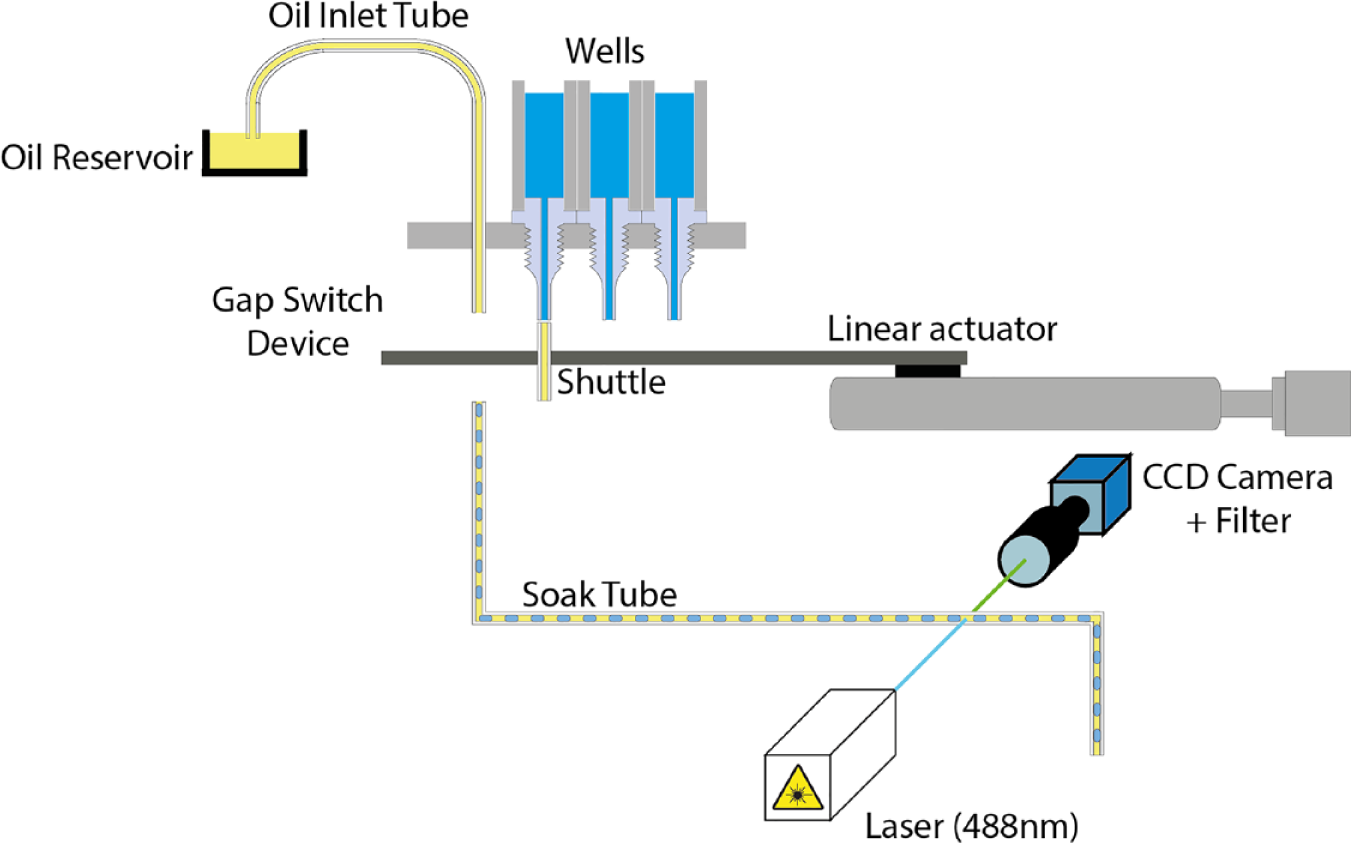
GST setup depicting droplet generation and optical detection. The droplets are created using the GST and are flowed into a soak tube for incubation.
Engineering Experimental Methodology
Droplet Reproducibility
Droplets of 200 nL were dispensed from each well. Fluids used on the GST vary in physical properties, including interfacial tension, viscosity, density, and fluid homogeneity. Variations in drug concentrations, cell types, and nutrient compositions will also contribute to changes in fluid physical properties. These fluidic changes affect the flow of fluid from the wells, creating inconsistent droplet volumes. To counteract this, the system is calibrated to produce 200 nL droplets by altering the length of time the shuttle passes underneath the well, that is, shuttle velocity, which is a function of droplet volume generated. Shuttle velocities ranged from 10 to 20 mm/s. Deionized water droplets were dispensed from each well individually and in combination (six droplets each) and repeated three times to demonstrate the accuracy of the droplet volumes generated. This accuracy is defined by the CV, which is the ratio of the standard deviation to the mean and is a measure of relative variability. It is calculated by

where σ is the standard deviation and
Droplet Mixing
Well 1 was filled with deionized water, while well 2 was filled with deionized water mixed with 5% (v/v) red food dye (Goodalls, Dublin, Ireland). The shuttle speed for well 1 was set to 20 mm/s and for well 2 was set to 10 mm/s. Both wells generate 200 nL droplets with this experimental setup. A 200 nL deionized water droplet was dispensed first into the shuttle followed by a second 200 nL red food dye droplet, which mixed into the first droplet. Images of these events are taken using a high-speed camera setup. Droplets were dispensed from each well individually and in combination (six droplets each) and repeated three times to demonstrate the accuracy of droplet mixing.
Biological Experimental Methodology
Collagenase Assay
The Molecular Probes EnzChek Gelatinase/Collagenase Assay Kit (Biosciences) was used to determine MMP proteolytic activity according to the manufacturer’s instructions. DQ-gelatin (E-12054) was used as a general MMP substrate and is heavily labeled with fluorescein.
Collagenase Assay Undertaken in Microplates on the Janus Automated Workstation
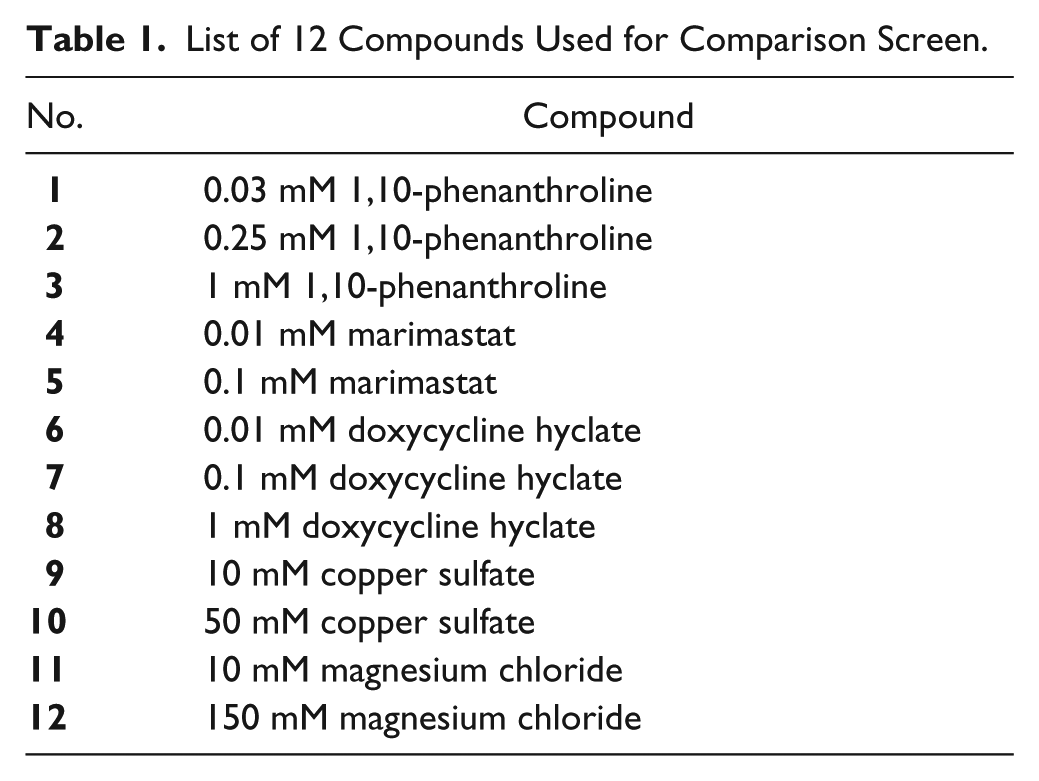
The HTS comparison was undertaken on a Nunc 384, black, flat-bottom microplate (Sigma-Aldrich, Dublin, Ireland). The well contained 20 µL of buffer (50 mM Tris-HCl, 5 mM CaCl2, pH 7.4) or drug/compound ( Table 1 ) and 5 μL of fluorescein-labeled DQ-gelatin substrate (120 µg/mL) to a final concentration of 12 µg/mL, along with 25 µL (0.2 U/mL) of collagenase to a final concentration of 0.1 U/mL. The HTS comparison screen was undertaken on the Janus Automated Workstation (PerkinElmer, Dublin, Ireland) and plates were imaged in the Victor X5 Multilabel Reader (PerkinElmer).
List of 12 Compounds Used for Comparison Screen.
Collagenase Assay Undertaken in Droplets Generated from the GST System
Analysis of proteolytic activity was performed in droplets that were created using the GST. Well 1 contained DQ-gelatin (36 μg/mL), well 2 held control enzyme (0.3 U/mL, purified collagenase type IV from
Inhibitors for High-Throughput Screening
A small library of 12 compounds were screened against the collagenase (MMP-1) enzyme (EnzChek Kit). The compound library contained two salts and three inhibitors at concentrations ranging from 10 to 150 mM for the salts and 0.01 to 1 mM for the inhibitors ( Table 1 ). 1,10-Phenanthroline, marimastat, and doxycycline hyclate (Sigma-Aldrich) inhibitors were selected as part of the screen as they are broad-spectrum MMP inhibitors. 1,10-Phenanthroline was used as a positive inhibitor to confirm the extent of the proteolytic activity specifically related to MMP activity as gelatin substrates can also be digested by other proteases. A Z factor of >0.5 was established on both systems indicating good assay quality.
Results and Discussion
The fluidics set up for droplet generation incorporating the GST and subsequent optical detection of droplets is outlined in Figure 1 . The GST accommodates a wide array of fluids including aqueous solutions and organic solvents for a diverse scope of bioengineering applications. Unlike other technologies, with the addition of several wells, droplets can be dispensed and rapidly mixed in a predefined combinational sequence.
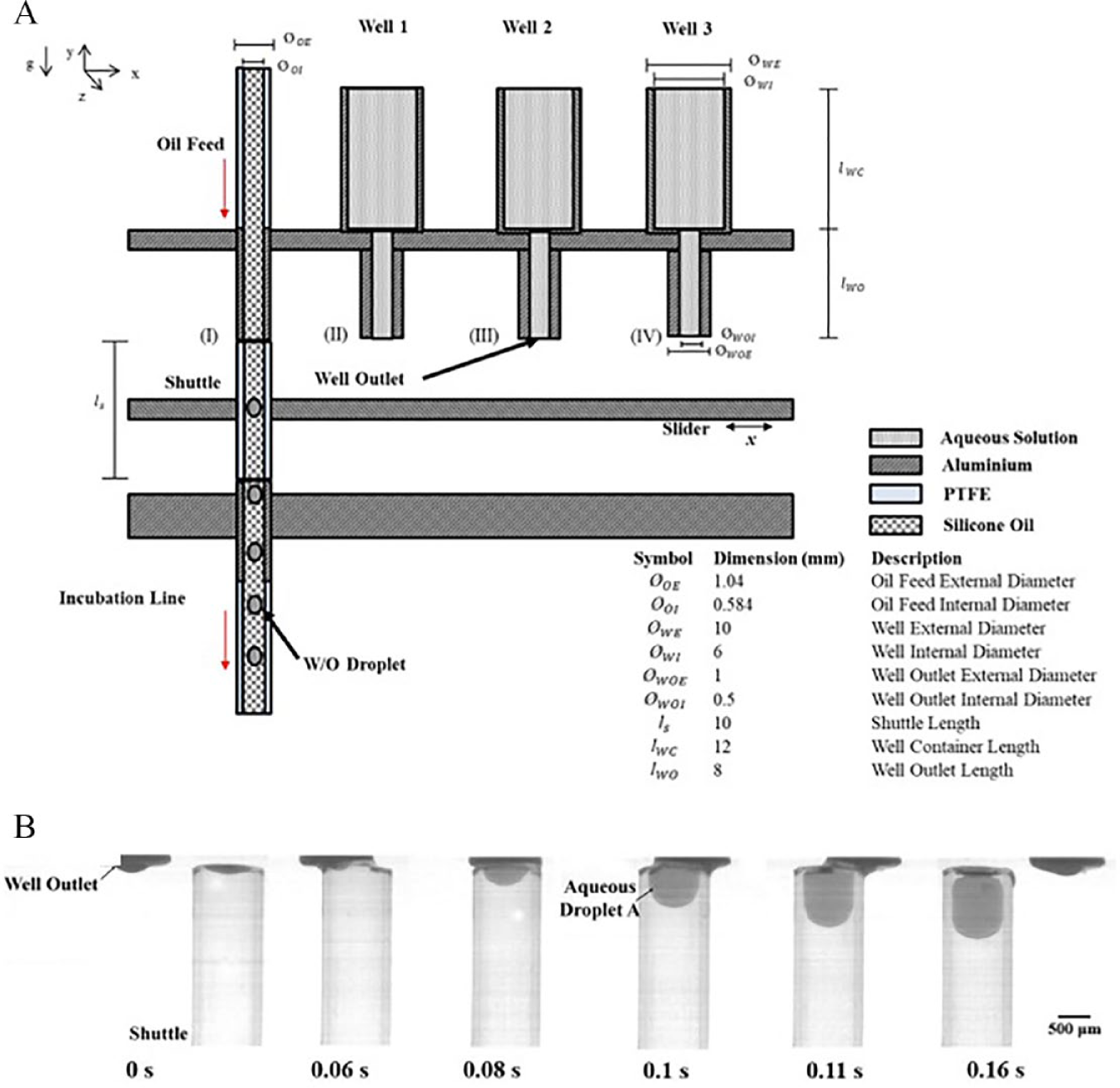
The GST consists of a moveable microfluidic channel, called the shuttle, which acts as a mechanism to collect and mix droplets from a series of wells containing fluid. Figure 2A shows the shuttle in series with the oil feed during flow, that is, position I. The shuttle is fixed to the slider, which is controlled by a stepper motor. The GST works on the principle of balancing surface tension and head pressure, thereby enabling droplet dispensing. A simplified version of the Young–Laplace equation (eq 2) can explain this.

where
(
The wells in Figure 2A are filled to their capacity with an aqueous fluid. All lines are primed with silicone oil and flow is initiated via siphon flow from the oil feed, through the shuttle, and into the incubation line. The fluid in each well is test dependent and can contain, for example, cells, bacteria, enzymes, reagents, and drugs. The pressure of the height of the fluid is counteracted by the surface tension forces at the outlet of the well, which allows the well to hold the fluid. 27
As the shuttle moves to positions II, III, and IV, the inlet of the shuttle makes contact with the outlet of each well and the overall head height is increased by a factor of the shuttle length. This increase in head height results in flow initiation, as the capillary force of the fluid in the well is overcome by the new pressure head and an aqueous droplet is segmented into the shuttle. The segmented aqueous droplet then becomes wrapped by the silicone oil in the shuttle. As the shuttle departs well 1 (position II,
Experimental Results
Mechanofluidic Validation
Fluids used on the GST will vary in physical properties including interfacial tension, viscosity, density, and fluid homogeneity. Varying drug concentrations, cell types, and nutrient compositions will see the largest of these property variations from fluid to fluid. Biological assays require precise droplet volumes and homogeneous mixing to ensure a high degree of reproducibility. Since the functionality of the GST is dependent on the fluid’s physical properties, validation of the GST to guarantee assay data quality and integrity is necessary.
Droplet Reproducibility
The reproducibility of droplets is shown in Figure 3 and results in a CV of 4.7% for three fluids dispensing approximately 200 nL to create approximately 600 nL of mixed droplet. Notably, droplet volumes dispensed and subsequently mixed will decline as the number of droplets in the combination increase due to an added resistance in the incubation line. 28 Shuttle velocity profiles are adjusted as required to compensate for resistance.
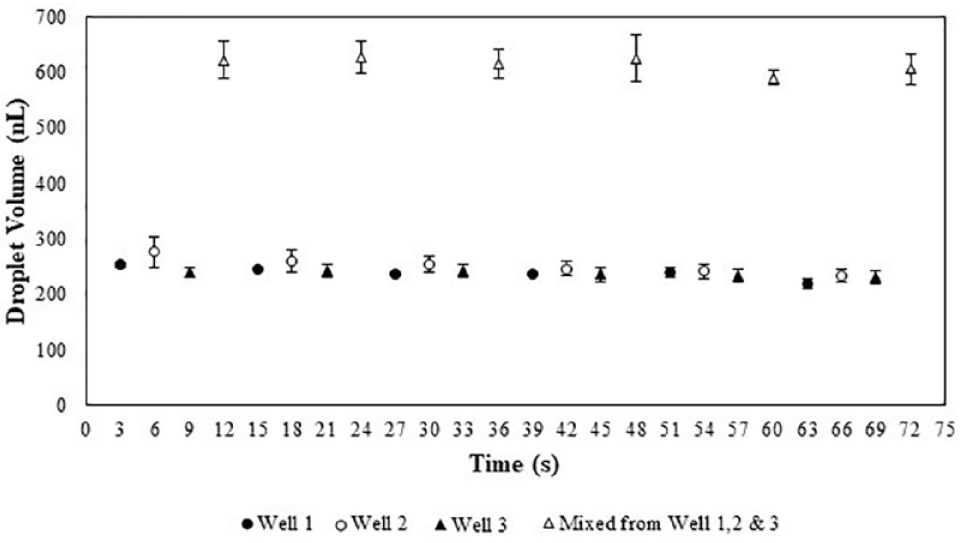
Consistency of droplets dispensed from each well, individually and in combination, per a 3 s cycle using the GST,
A typical HTS system, PerkinElmer’s Janus Automated Workstation is widely used in industry for drug screening.29–31 The Janus typically produces CVs of 3.8% using the blowout method with VersaTip Fixed Tips (PerkinElmer), dispensing 1 μL of single-fluid deionized water. 32 The CV of 4.7% using the GST is inferior to the CV of the PerkinElmer technology, but the authors are confident that with technical improvements the GST can achieve similar if not better CVs. The successful outcome of a biological assay is highly dependent on precise volumetric control, particularly in HTS.
Droplet Creation and Velocity Change
The operational performance of the GST system accommodates droplet volume adjustments as required through shuttle velocity modification. This is only achievable with a fixed wellhead pressure. Figure 4 shows the linear regression between droplet volume dispensing and increasing shuttle velocity. The fluids used in this experiment consisted of water droplets wrapped in silicone oil M5. Volume compensation factors will be developed for each fluid used on the GST. With such data, customized system parameters can be established for individual assays by varying the time of droplet dispersion based on individual fluid properties such as viscosity, surface tension, and density.
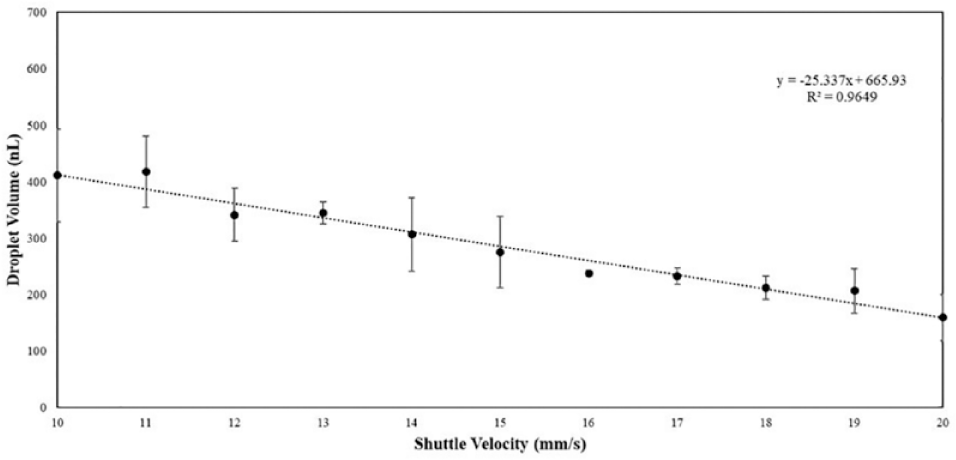
Droplet volume assessment based on shuttle velocity speed variation,
Droplet Mixing
Consistent droplet mixing for the biological assay ensures homogeneous reactions. Mixing commences when a droplet is dispensed into another by single-phase Poiseuille flow. Gravity-driven droplet transport advances the mixing by crossflow advection through recirculation paths within the droplet (

Mixing within a droplet. (
Figure 5A exhibits the dispensing of a 200 nL droplet containing red food dye into a 175 nL water droplet by way of Laplace mixing. The dispersion of the dye solution into the clear water droplet occurs over approximately 200 ms. In Figure 5B , the shuttle is positioned under the oil feed line and the newly formed droplet flows down the shuttle into the incubation line. The image sequence presented in Figure 5 occurs over approximately 100 ms. Poor droplet mixing will yield unreproducible assay results; therefore, biological validation was also used to verify successful droplet mixing for this platform.
Biological Validation
Collagenase Assay
MMPs digest gelatin, collagen, and strome lysins, which are crucial for healthy development but are also involved in a range of illnesses, including cancer and inflammatory diseases, and have been found to directly contribute to disease progression.33,34 Increased expression of MMPs is linked to increased cancer cell proliferation and increased tumor size. MMPs can influence tumor environment by promoting angiogenesis, tumor growth, and metastasis.
33
Due to their powerful degradative capacity, MMP activity is tightly controlled by a specific class of natural inhibitors known as tissue inhibitors of metalloproteinases (TIMPs). Several inhibitors of metalloproteinases are used within this study to inhibit the action of
Role of BSA in Stabilizing the Assay
BSA is used within the droplets to prevent the assay proteins (DQ-gelatin and collagenase) from adhering to the droplet-oil interface. Blocking proteins such as BSA can adsorb at fluid interfaces and are often used, for example, as emulsifiers for oil-in-water (O/W) emulsions. As the interfacial tension between water and oil is high, these blocking proteins adsorb at the interface and reduce interfacial tension, thereby imparting stability.37,38 In such systems, the interfacial adsorption of the protein is due to its amphiphilic character. The adsorption of blocking proteins at the O/W interface generally consists of three steps: (1) diffusion of the protein from the aqueous phase to the interface; (2) protein infiltration and subsequent unfolding of the proteins at the interface; and (3) establishment of a viscoelastic monolayer via structural reorganization of the adsorbed proteins at the interface. All of these changes will be dependent on environmental conditions such as protein concentration, pH, ionic strength, and temperature. Minimal unfolding occurs upon adsorption at the O/W interface.39,40
Droplet images are presented in Figure 6 . As seen, Figure 6A displays a bright halo at the O/W interface, while this is absent from the droplet in Figure 6B . A schematic representation of the droplets with and without the BSA aggregation at the O/W interface is also shown. These schematics are simple sketches and do not aim to reflect the true spatial arrangement of protein molecules on adsorption. It is theorized that DQ-gelatin is attracted to the droplet’s O/W interface, which is characterized by a distinct brightness at the boundary. This brightness at the O/W interface is absent when 0.0002% BSA is added, indicating that the presence of BSA reduces DQ-gelatin accumulation (and possibly collagenase attraction) at the boundary over the assay length of 20 min. From the distribution plot shown in Figure 6D , the plot representing the droplet without BSA shows peaks at approximately 120 and 680 pixels, while these are not present in the plot representing the droplet with BSA added.
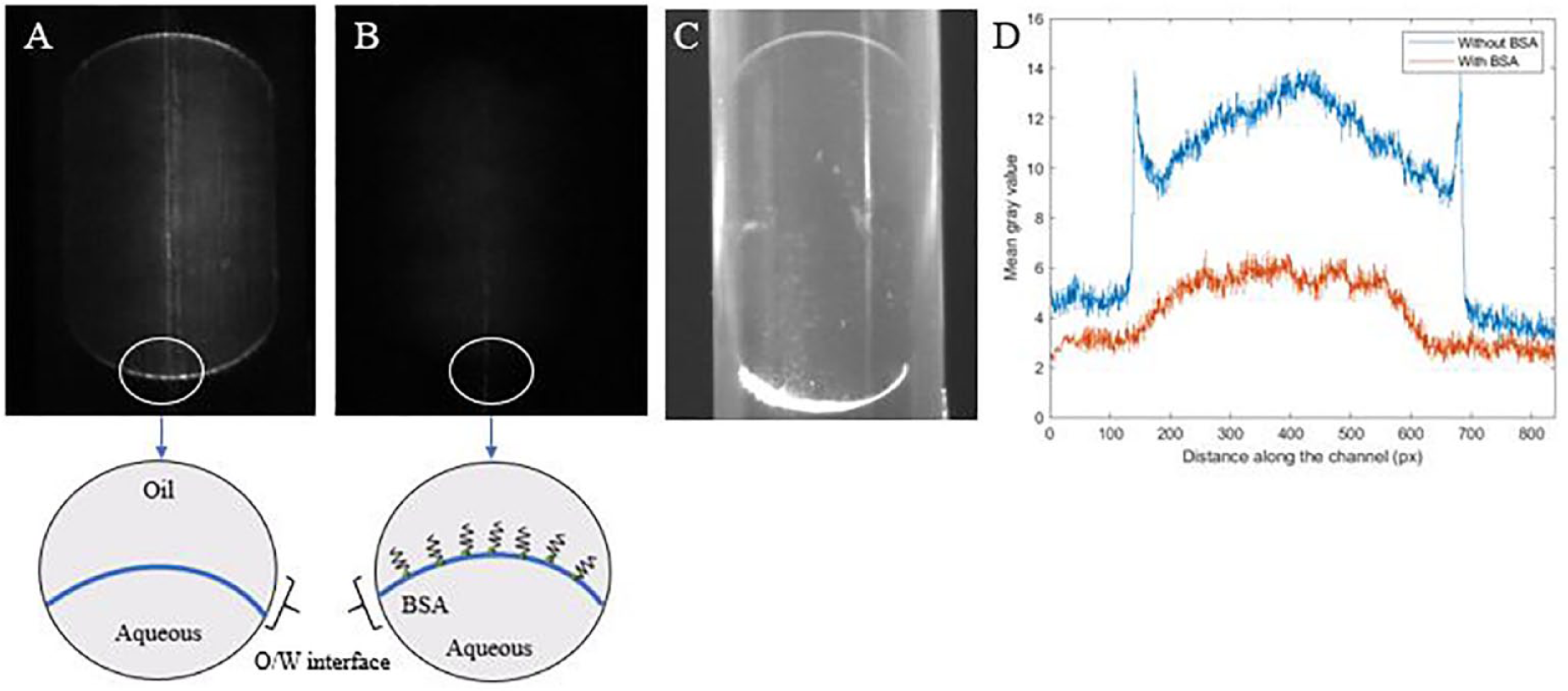
Effect of BSA addition on assay stabilization in droplets. (
In addition, the fluorescent signal elucidated from droplets containing 0.00025% BSA is more than threefold higher than the droplets that do not contain BSA (
Comparison of Janus Automated Workstation and GST System
A screen was undertaken to validate the GST by comparing the results of the biochemical reaction in droplets with that of a standard 384 microplate filled using the Janus Automated Workstation. The workstation is designed for the efficient automation of sample preparation procedures used in pharmaceutical, biotech, and research applications. 41 It is extensively used in the pharmaceutical industry for HTS assays as it provides scalable and accurate liquid dispensing.
A small library of 12 compounds ( Table 1 ) were screened against the collagenase (MMP-1) enzyme. Of the 12 compounds selected, 8 were known MMP inhibitors (1,10-phenanthroline, doxycycline hyclate, and marimastat) that were assessed at concentrations increasing 10-fold from 0.01 to 1 mM. 1,10-Phenanthroline is a metal chelator that inhibits a broad spectrum of MMPs, marimastat chelates the zinc ion at the active site of MMPs and is relatively nonspecific, 42 and doxycycline hyclate is an antibiotic that inhibits MMP activity independently of its antimicrobial properties, through its ability to chelate zinc ions essential for MMP activity. 43
Data were converted to percentage inhibition from raw fluorescence data as follows: % Inhibition = Activity (Control) – Activity (Inhibitor)/(Activity [Control]) × 100, where Activity (Control) refers to the fluorescence intensity of the enzyme control and Activity (Inhibitor) refers to the fluorescence intensity in the presence of an inhibitor. Results from the assay were normalized to between 0% and 100% inhibition for both systems. If values were higher than the linear dynamic range, they were capped at 100, while any values below the linear dynamic range were capped at 0. A 20% difference in inhibition between the GST and Janus system was set as a baseline error for data analysis; this error was selected due to differences in the systems’ imaging sensitivity and the GST’s surface tension-dependent droplet dispensing.
Out of the 12 compounds screened, only one, 0.25 mM 1,10-phenanthroline, fell outside of this parameter showing a 27% difference between the GST and the Janus (
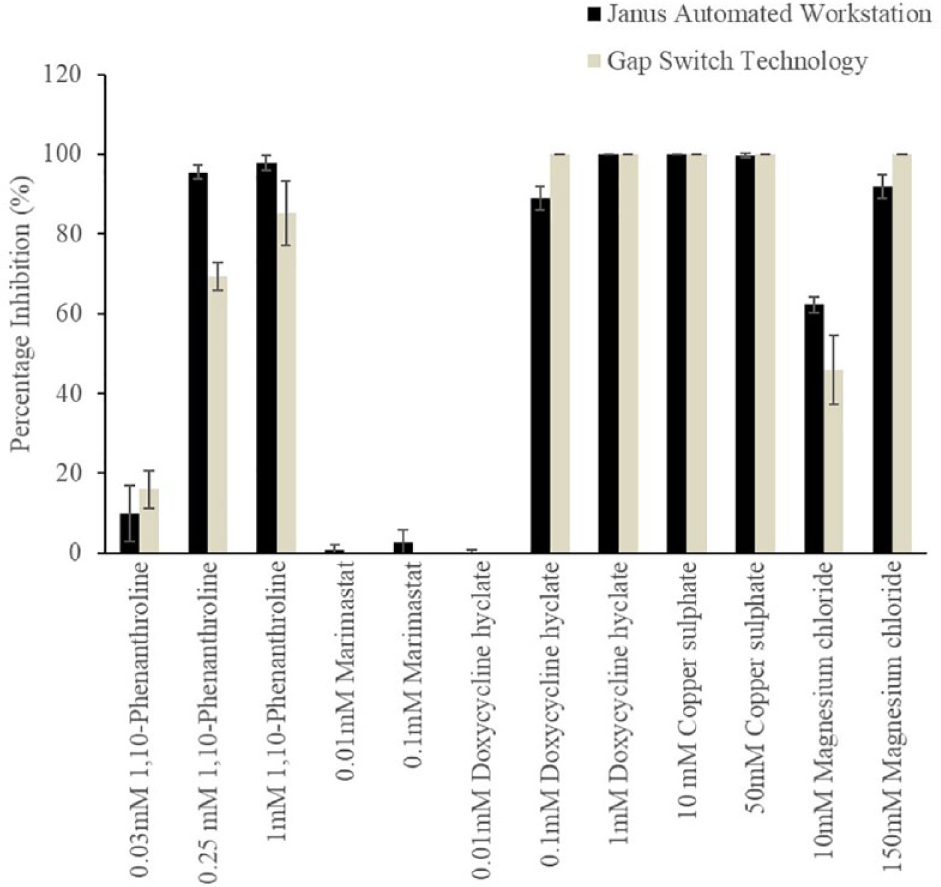
GST versus Janus system assay screen of percentage inhibition of compound,
Overall, the screening of the 12 compounds demonstrated that droplets created using the GST compare favorably with samples assessed in a 384 microplate on the Janus Automated Workstation, with less than a 20% difference between both (excluding 0.25 mM 1,10-phenanthroline). The GST has the advantage of an 80-fold reduction of reagent volume from 600 nL compared with 50 µL volume on the 384 microplate. HTS assays are characterized in several ways, including the use of the Z-factor metric. This metric is one of the most commonly used in the field of HTS to identify hits with high fidelity. The result is a statistical test where a value of >0.5 is seen as a good-quality assay.
46
A Z factor of >0.5 was established for both systems, 0.54 for the GST and 0.69 for the Janus Automated Workstation. The Z factor is calculated using eq 3, where
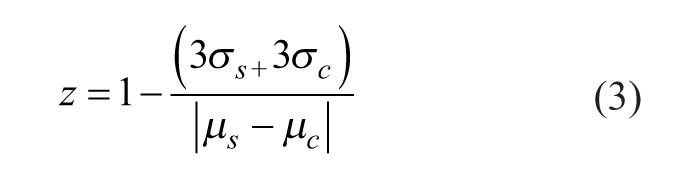
The scatterplot (
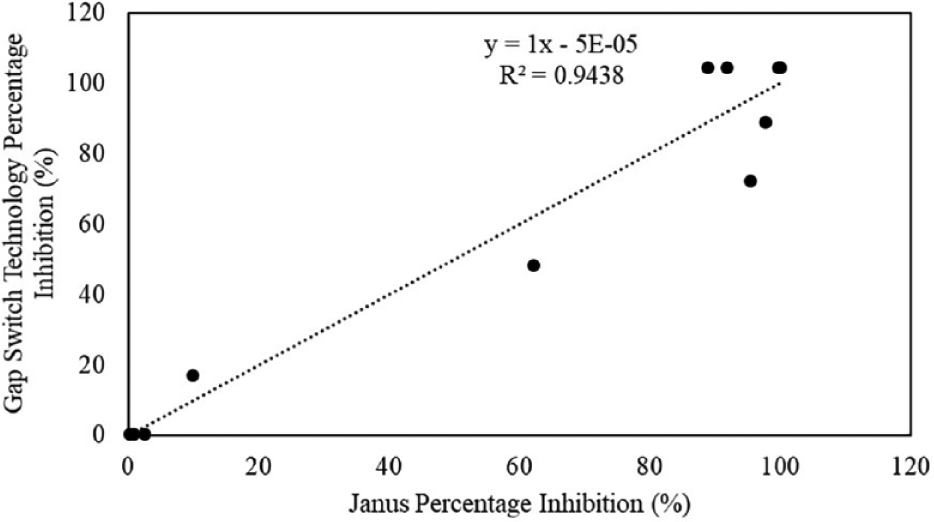
Standardized summary of percentage inhibition of 12 compounds assessed on GST versus the Janus system.
Limitations of GST and Strategy for Improvements
There are several limitations to the formation of droplets through operation of the GST. One of the main difficulties in HTS is the challenge of generating and handling multiple compound-containing droplets. Traditionally, it has been very difficult to generate screening droplets composed of multiple compound reagents. With the current GST prototype, only one compound can be assayed at a time, which is acceptable for proof of concept but is restrictive for actual compound screening. It is estimated that with the current design, one well can store approximately 340 µL of liquid, from which approximately 1700 droplets of 200 nL can be generated. Using three wells, the system has the potential to generate up to 5100 droplets (200 nL each in volume) in one run or 1700 mixed droplets (200 nL from each well) of 600 nL (until the well empties of fluid). To increase the throughput, it is envisioned that the linear GST system will be scaled to a rotational design. Such a design change would allow the implementation of multiple wells, thereby allowing the selection and mixing of multiple droplets via the shuttle as designated by the user. Droplets of different composition could also be generated in parallel. This would dramatically increase the throughput of the GST.
The effect of interfacial tension on the formation of reproducible droplets is another limitation that must be addressed. Interfacial tension can vary from compound to compound, altering the volume of droplets picked from the well. Any deviation in droplet volume can have a knock-on effect on compound concentration and in turn the efficacy of the assay. To offset this effect, calibrations of all fluids used on the GST will be required. In terms of optical detection, very sensitive equipment may be required to detect the analyte of interest and complications around image capture, for example, artifacts from lighting conditions, light scattering at the droplet interface, and bleaching, need to be tackled. Fluorescence intensities of multiple droplet reactions can be achieved by integrating an optical detection system or imaging platform that allows capture of the fluorescence signal from droplets in tubing at specified time points.
As the system scales upward, issues related to data collection, storage, standardization, and processing will need to be addressed. For application in industry and academia, a considerable amount of work is necessary to surmount these limitations. In terms of technical improvements, several changes could be implemented to improve the prototype, including the addition of a pumping system that would give greater control of fluid flow as well as steps such as improving manufacturing tolerances, optimizing well geometries, and integrating a programmable logic controller (PLC) for automation. With the ongoing research in this area, the authors believe that these issues can be solved and microfluidic platforms like the GST will soon become irreplaceable in drug screening, diagnostics, and other applications.
Conclusions
Droplet microfluidics are currently used in various fields such as pharmaceuticals, biotechnology, and chemical analysis/synthesis, and the market is expected to be worth approximately $14 billion by 2022. 47 HTS and compound profiling has the largest application, which is expected to account for nearly 59% of the total market by 2022 (compound annual growth rate [CAGR] of 20%). Within this study, a microfluidic HTS technology is presented as a platform that could support combinatorial drug discovery at the preclinical stages. The production of a continuous stream of droplets housing the biological assay circumvents the stop–start batch nature of conventional microfluidic chips. The GST presented demonstrates accurate droplet creation and mixing in the 200–600 nL range; however, larger and smaller volumes could be accommodated in the system. The aqueous droplets created were reproducible and stable as demonstrated by results achieved from the EnzChek Collagenase Assay, and this methodology is applicable to other biochemical assays that require multiple sample/reagent combinations. This assay also allowed comparison between the platform and an industry standard HTS system, PerkinElmer’s Automated Janus Workstation. The results indicated that with the suggested improvements, the microfluidic GST could be developed as a low-cost, automated, and high-throughput solution for drug combination screening. Further prototype development is ongoing with the aim of undertaking 3D cell-based assays particularly in the area of personalized medicine.
Supplemental Material
Supplemental_Material_Droplet_combinations_A_scalable_microfluidic_platform_for__biochem ical_assays_R.1 – Supplemental material for Droplet Combinations: A Scalable Microfluidic Platform for Biochemical Assays
Supplemental material, Supplemental_Material_Droplet_combinations_A_scalable_microfluidic_platform_for__biochemical_a ssays_R.1 for Droplet Combinations: A Scalable Microfluidic Platform for Biochemical Assays by Finola E. Cliffe, Mark Lyons, Daniel C. Murphy, Lisa McInerney, Niall Hurley, Michael A. Galvin, Jane Mulqueen, Lorraine B. Bible, Claudio Marella, Michael Kelleher, Anne O’Sullivan, Howard O. Fearnhead, Enda O’Connell and Mark Davies in SLAS Technology
Footnotes
Acknowledgements
The authors acknowledge financial assistance from Enterprise Ireland, who partially funded this research. The technology described in this article is in development and represents ongoing research and development efforts.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
