Abstract
Microfluidic devices offer new technical possibilities for a precise manipulation of
Introduction
In drug screening, vertebrate animals such as mice have widely been used as model organisms to test potential therapeutics before their translation into human clinical trials.
1
Recently, the relatively simple invertebrate animal
To dispense
Microfluidic systems with a channel size comparable to that of a
Materials and Methods
Fabrication of the Microfluidic Chip
The microfluidic chip consisted of two PDMS layers: a fluidic layer on the top and a pneumatic control layer on the bottom, as described previously. 10 A detailed description of the fabrication steps can be found in Supplemental Figure 1S of supporting information.
On-Chip Integration of Optical Fibers
The detection mechanism in our device was based on the light scattering when a worm passes by the optical gate
11
in the constriction channel that was required to decelerate the flow of
Trapping and Dispensing Method
The trapping mechanism for this device was based on the PDMS valving system, and this technique has successfully been applied to worm manipulation, such as immobilization. 10 After trapping, a syringe pump (Era Pump Systems Inc., Farmingdale, NY, NE-1002X) was turned on to pump M9 buffer solution for 20 s at a flow rate of 30 mL/min and dispensed the worm into the microplate via tubing with a dead volume of ~10 mL connected to the outlet of the microfluidic chip.
Characterization of Dispensing Performance
In order to characterize the device, we desired a uniform size of worms. Following the standard lab procedure for the synchronizing developmental stage, adult wild-type (N2 strain) worms were treated with bleach and unhatched embryos, which are protected from this treatment by an impermeable eggshell, were collected. Animals synchronized in this way were allowed to grow on agar plates supplemented with OP50 bacteria. After reaching the fourth larval (L4) stage, worms were washed from agar plates with M9 buffer and diluted to a concentration of ~25 worms/10 mL droplet. To characterize the dispensing accuracy and speed, two dispensing experiments were conducted in 10 microplate wells at three different flow rates (3, 5, and 7 µL/min) using two different constriction channel sizes (30 and 40 µm). The dispensing accuracy was measured by counting the average number of wells with a single worm. Once the dispensing in 10 wells was completed, a rapid and accurate high-throughput image analysis was performed using the image analysis system DevStaR to measure the dispensing accuracy.
Results and Discussion
The microfluidic dispensing system with a microfluidic chip and a dispensing tip that was connected to the chip outlet via tubing is shown in Figure 1a . A robotic arm was used to move the tip from well to well. The PDMS microfluidic chip had five integrated pneumatic microvalves (marked as green lines in the chip), which were controlled by a manifold, as well as a pair of the optical fibers connected to a laser source and a photodiode. To monitor the detection and trapping of worms, the microfluidic chip was initially mounted underneath a stereoscope. When improving the dispensing speed, it was mounted directly on the robotic arm so that the amount of dead volume between the outlet and the dispensing tip could be minimized. The worm suspension and M9 buffer solutions were supplied separately to the microfluidic dispensing chip via two syringe pumps. The syringe supplying the worm suspension had two vibrating actuators attached to its bottom to prevent the worms from settling down. The dispensing operation is shown in Supplemental Video 1S in supporting information. In Figure 1b , the integration of optical fibers toward the microfluidic channel, as described in the Materials and Methods section, is shown schematically. The flexible PDMS layer acting as a valve is directly underneath the fluidic channel. In Figure 1c , the closing of the microfluidic channel with a PDMS valve upon optical detection is shown schematically. The operation of the dispensing system can be seen in a video Supplemental Video S1 .
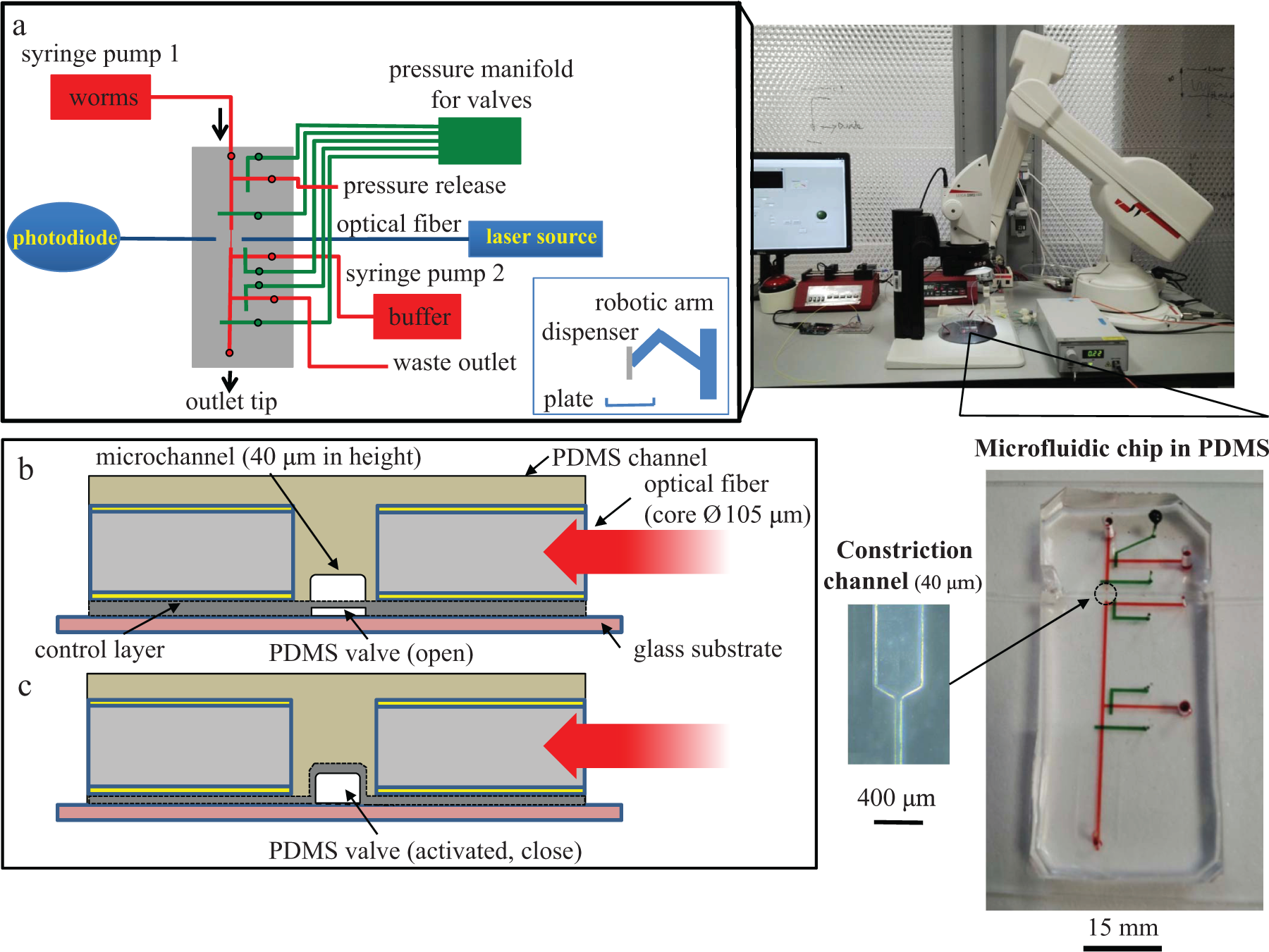
Microfluidic dispensing system for
For trapping and dispensing of the
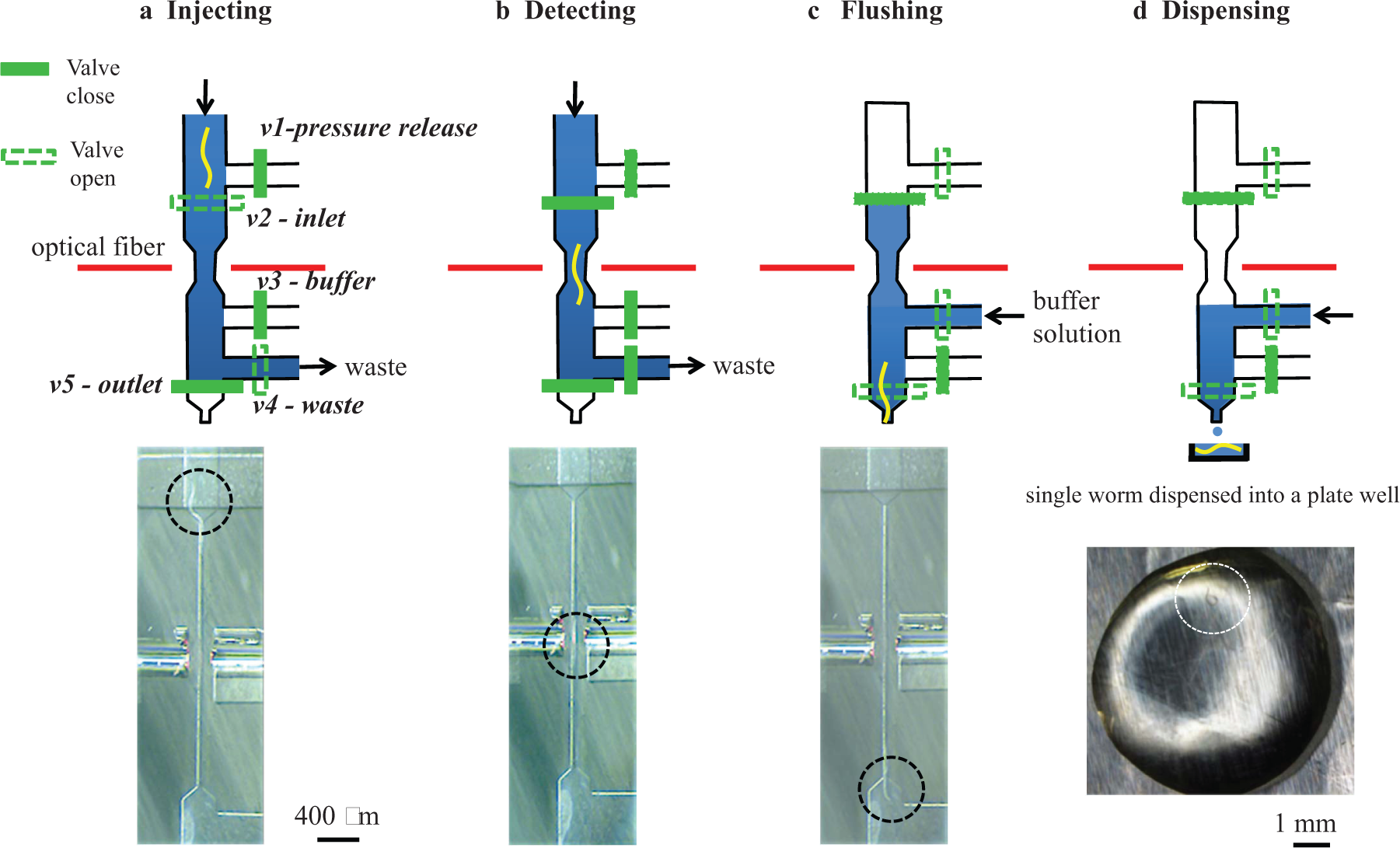
Schematics and images showing (
The delay time for switching off pump 1 had to be at least 2 s in order to make sure that the worm passed through the constriction completely. However, within this delay time, a second worm could pass through the constriction channel before pump 1 was turned off, resulting in a variation in the number of worms dispensed. To deal with this problem, the LabVIEW software scanned for a second drop of the laser signal intensity after 1 s of detection. If a second drop of the signal intensity was detected after 1 s, then these two worms were flushed out to the waste outlet. If, however, two worms followed each other very closely, which often was observed in the microchannel, then the current detection system could not differentiate between a single worm and two worms.
The most critical parameters when improving the dispensing accuracy were the flow rate, the constriction channel size, and the concentration of worms in the suspension. Using ~20–25 worms/10 µL worm suspension and a 30 µm constriction channel in a microfluidic chip mounted directly on the robotic arm (
Fig. 3a
), a dispensing accuracy of ~83.33% was achieved at a flow rate of 3 µL/min for a total dispensing time of 19.27 min (
Fig. 3b
). The total dispensing time was measured as the average time it took to dispense in 10 wells. Constriction channel sizes below 30 µm caused frequent clogging issues and therefore were not practical for dispensing. As the flow rate was increased to 7 µL/min, the dispensing accuracy decreased down to ~70% at a total dispensing time of 8.03 min. The main reason for the higher dispensing accuracy at 3 µL/min was the longer transition time of worms in the constriction channel for optical detection and trapping with the microvalves. In the case of the 40 µm constriction channel, a dispensing accuracy of 80% was achieved at a flow rate of 3 µL/min for a total dispensing time of 15.04 min (
Fig. 3c
). As implied by these dispensing results, a trade-off between accuracy and speed was necessary. In this regard, the 40 µm constriction channel was selected as the preferred channel size. Dispensing of two or more worms into a well was caused by the characteristic behavior of
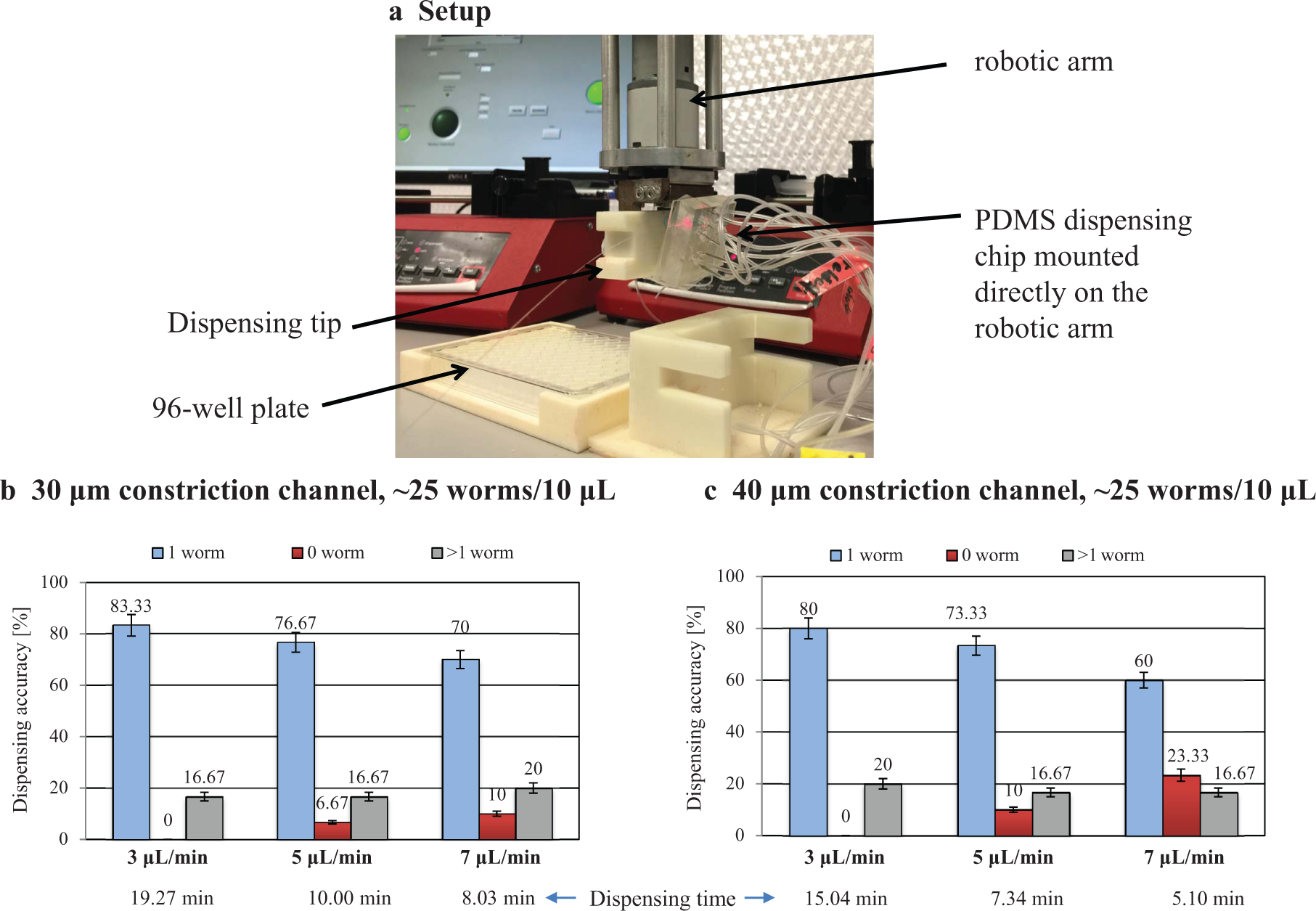
Characterization of the microfluidic dispensing chip directly mounted on the robotic arm in terms of accuracy for a single worm dispensing at three different flow rates and two constriction channel sizes, 30 and 40 µm. (
Conclusions
We developed a prototype of a disposable microfluidic chip for counting and dispensing a single
Footnotes
Acknowledgements
We thank Ms. Fathima Shaffra and Dr. Hala Fahs from NYUAD for providing us with the worms for the dispensing experiment. The device fabrication was performed in the microfabrication core facility at NYUAD. This work was conducted with the support of the NYU Whitehead Fellowship for Young Investigators in Biomedical and Biological Sciences 2014.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
