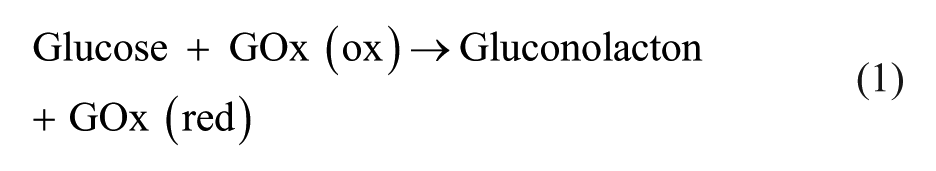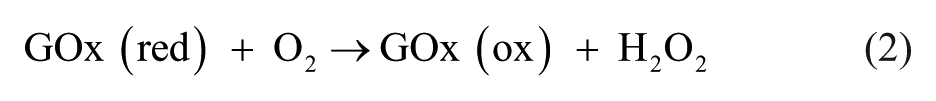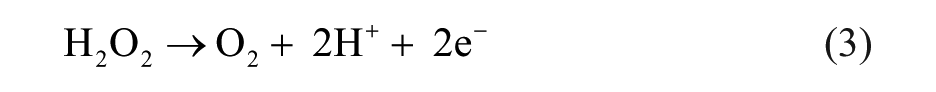Abstract
A novel electrochemical glucose sensor was created for a simple but semiquantitative visual screening of specific glucose concentrations in urine. This noninvasive glucose biosensor integrated a disposable, paper-based sensing strip and a simple amplifier circuit with a visual readout. The paper strip consisted of five enzyme-activated electrodes. Each electrode was connected to a specific indicator circuit that triggered a light-emitting diode (LED) when a predefined glucose concentration was reached. The device features (1) low-cost, disposable, paper-based glucose oxidase (GOx)/poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) sensing electrodes, (2) simple signal amplification, and (3) on-site, rapid, and visual detection. The sensor generated reliable, discrete visual responses to determine five glucose levels (1, 2, 3, 4, and higher than 4 mM) in urine in less than 2 min. This innovative approach will provide a simple but powerful glucose sensing paradigm for use in POC diagnostics.
Introduction
Urine has been widely studied as a glucose diagnostic sample because it is easily and noninvasively collected.1–3 Persistent high glucose levels in urine may increase the complications from diabetes or indicate a poor response to treatment. Urine glucose in the 2.78–5.55 mM range reliably indicates that blood sugar levels are high.2,4 Extensive research efforts have been made by using glucose oxidase (GOx) enzymes for amperometric detection of glucose levels in urine.5–8 However, until now no research has shown the possibility of using point-of-care (POC) diagnostics for quantitative or semiquantitative monitoring of urinary glucose. Most conventional POC devices for urinary glucose detection are limited to colorimetric or yes-no platforms based on monitoring the color intensity of the sensors by the naked eye. Although these subjective detection systems have been improved to provide great sensitivity with accuracy and precision by applying electrochemical techniques, their practicability is questioned. This is mainly because the electrochemical sensing platforms require (1) bulky instrumentation, (2) additional reference electrodes, (3) external readouts, and (4) extensive data analysis. 9 Recently, a simple, low-cost light-emitting diode (LED)-based readout technique has been demonstrated for glucose monitoring.7,8,10,11 Even though the LED-based sensor provides a simple and easy readout platform compared with conventional electrochemical methods in POC applications, it requires a camera-equipped smartphone or other handheld optical sensor to clearly determine different light intensities.
In this work, we demonstrate multiple-threshold urinary glucose detection with a visual readout. This work eliminates the need for interpretation of optical output intensity. The sensor consisted of a paper-based, disposable sensing strip assay integrated with discrete optical readout circuits. Poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) was used to conductively functionalize the paper structure because of its high electrical conductivity and excellent electrochemical stability for the enzyme immobilization.12,13 A schematic illustration of the proposed glucose sensor is depicted in Figure 1 . The sensing strip contained five glucose-specific enzyme-attached electrodes and a common cathode electrode. The currents generated by enzymatic reactions went from each of the five electrodes to their individual amplification circuit. The resistors were adjusted so that each LED indicator in the array corresponded to a certain glucose concentration in the urine sample ( Fig. 1 ). If all the LEDs light up, then the concentration is greater than 4 mM. If only four light up, then the concentration is less than or equal to 4 mM; three lights less than or equal to 3 mM; two less than or equal to 2 mM, and one less than or equal to 1 mM. This innovative approach will provide an accessible alternative way to conventional electrochemical glucose sensing measurements that typically use complex, bulky, and expensive potentiostat or galvanostat instrumentation that is inconvenient for use in POC diagnostics.
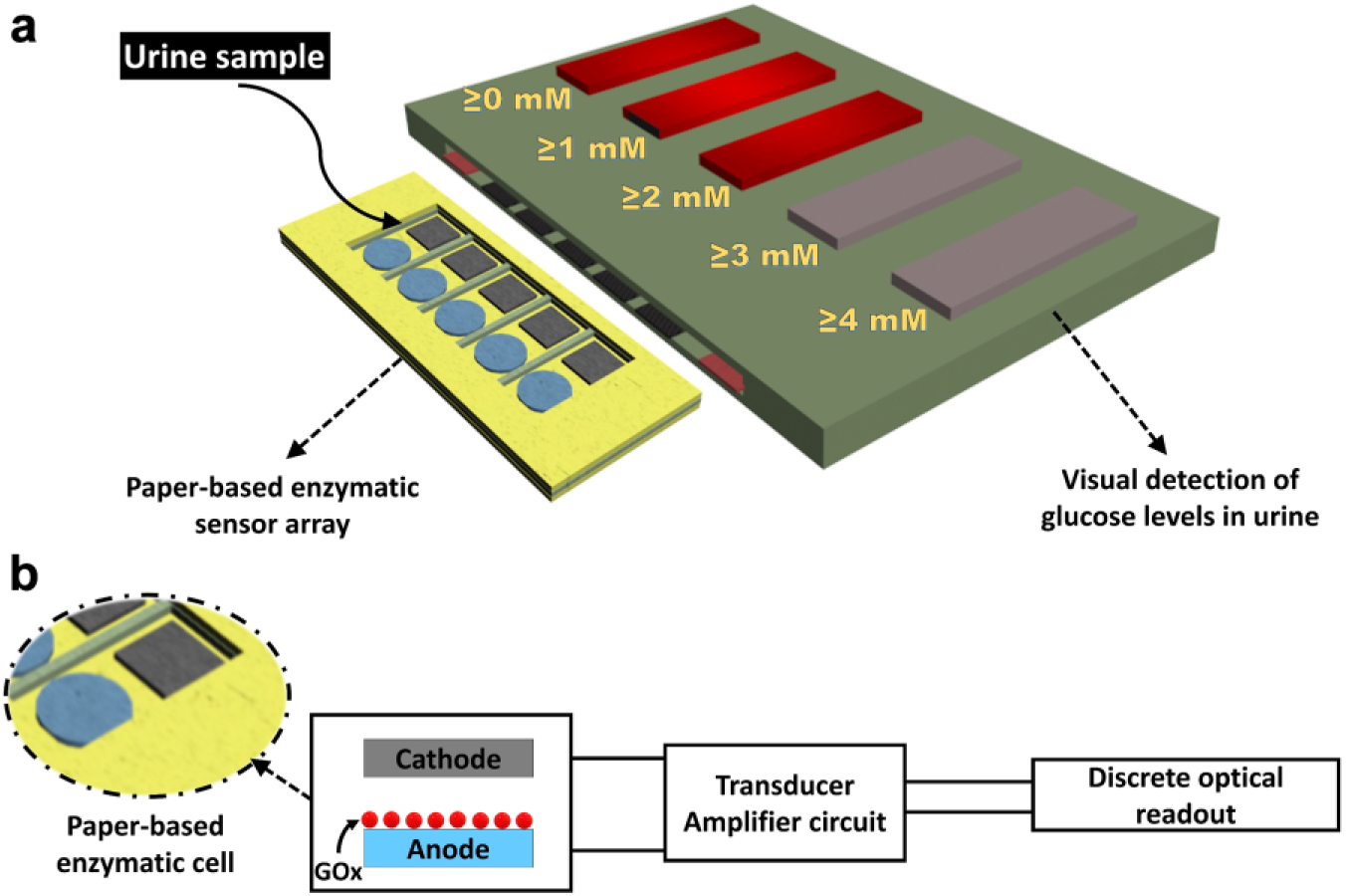
(
Materials and Methods
Reagents
GOx, PEDOT:PSS, Nafion solution, phosphate-buffered saline (PBS), and
Fabrication of Paper-Based Sensors
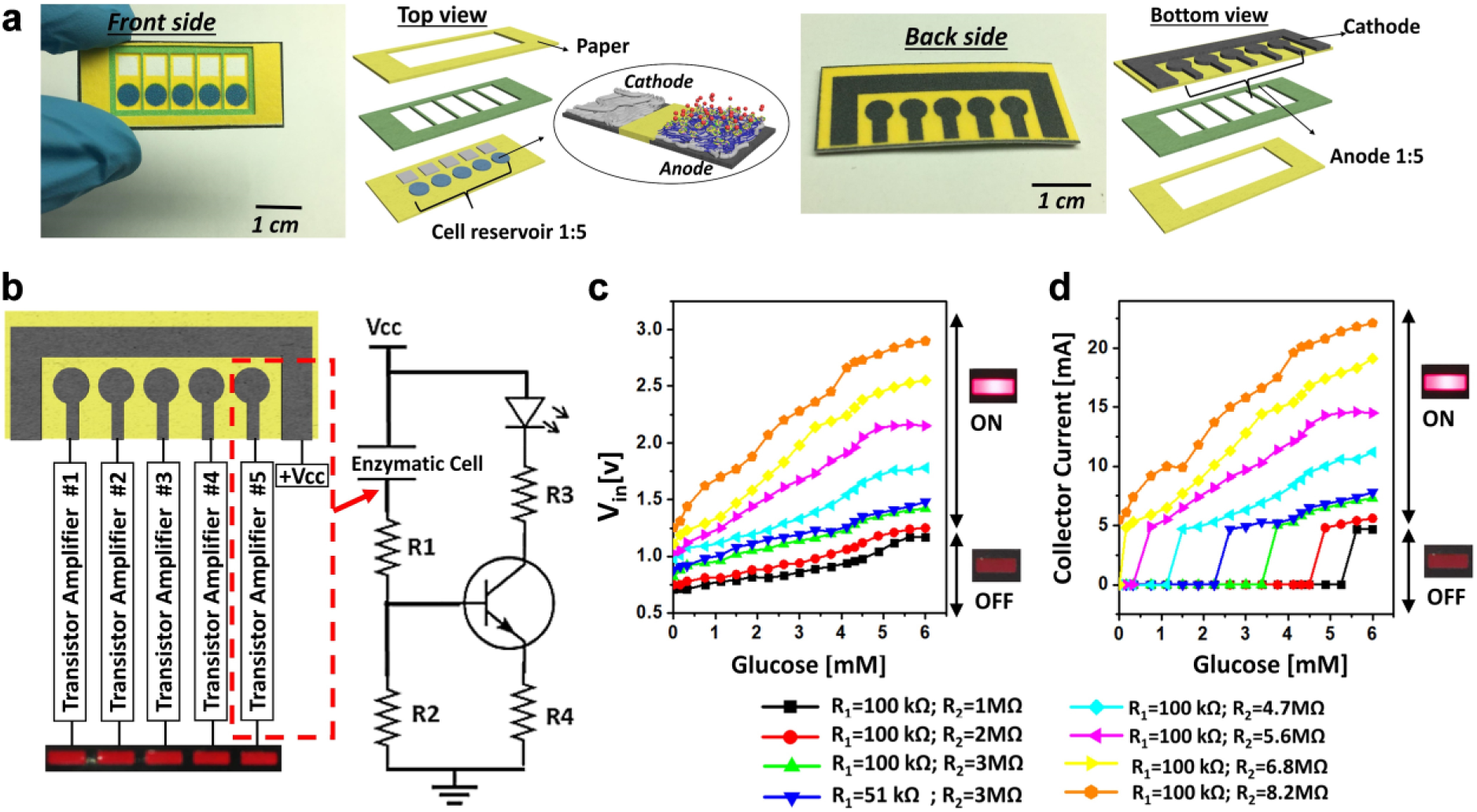
The electrodes were fabricated on Whatman #1 filter paper (001-329 Grade 1) by patterning five hydrophilic regions for the anodic enzyme reservoirs as well as the corresponding hydrophilic cathodic reservoirs ( Fig. 2 ). Five regions were well defined by printing wax boundaries with a solid-wax printer (Xerox Phaser 8570dn, Xerox Corporation, Norwalk, CT) ( Fig. 3a ). The wax patterns were then heat treated at 130 °C for 60 s to allow complete penetration of the wax through the paper. GOx-functionalized anodic enzyme reservoirs were prepared by introducing a 5 µL mixture of 1 wt% PEDOT:PSS and 5 wt% DMSO onto each hydrophilic anodic reservoir. The injection was performed on both sides of the paper to ensure back-to-back high conductivity (<30 Ω). Each injection step was followed by thermal drying at 100 °C for 20 min. After ensuring the high conductivity, the graphite-ink electrodes were screen printed on the back side of the sensing strip, followed by 24 h of air drying. A magnetic stirrer was used to mix 1 mL of 0.1 M (pH 7.0) PBS with 14 mg of GOx. PEDOT:PSS was added to encapsulate the GOx. The resulting solution, about 30% PEDOT:PSS, was used to immobilize the GOx on the conductive reservoir, preventing decay in connectivity with the paper matrix. To ensure uniform distribution of the enzyme, the immobilization was performed laterally via a sacrificial microfluidic patterned paper layer. The anodic reservoir with the GOx was air-dried at 4 °C.
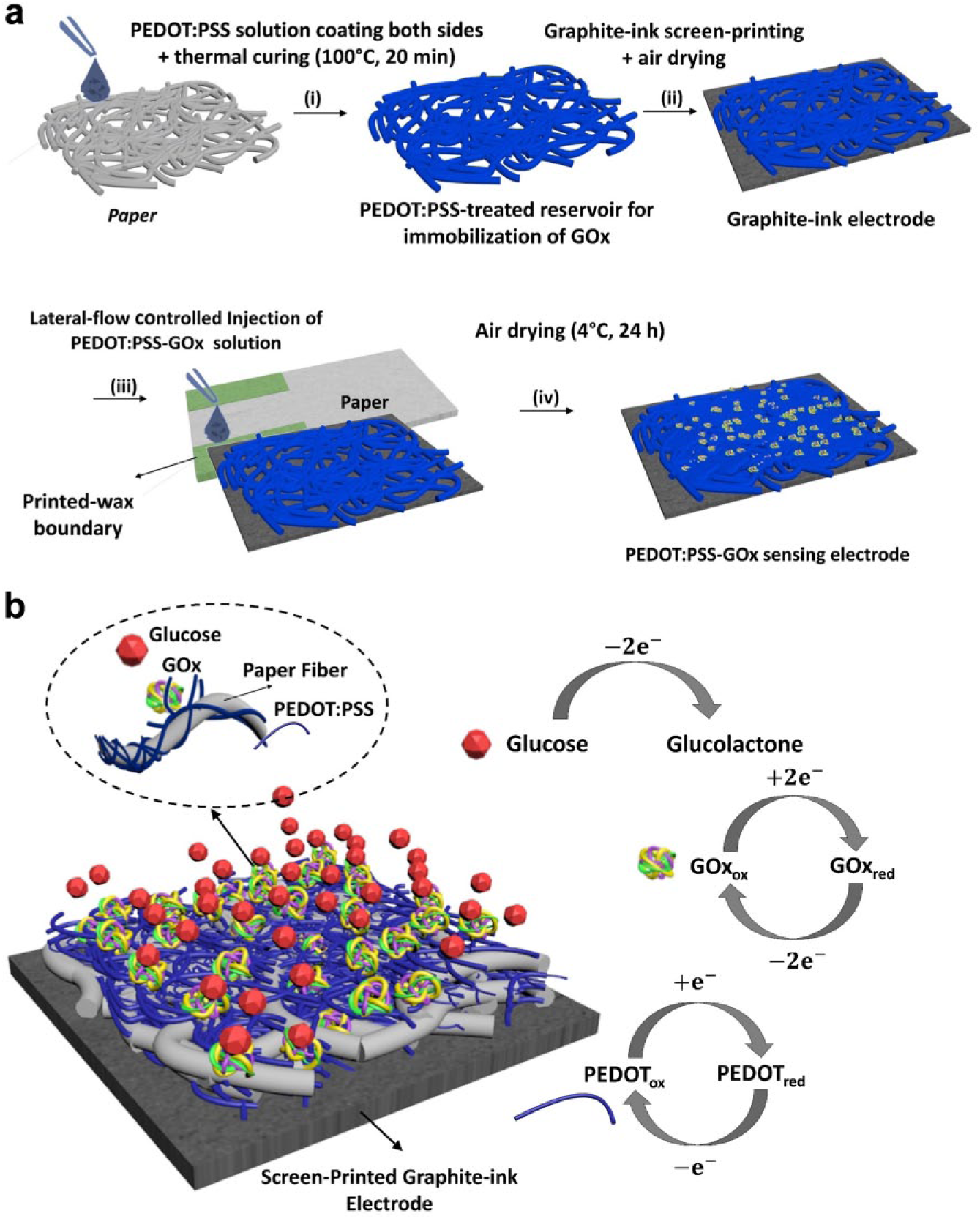
Schematic illustrations of (
(
Transistor Amplifier with Discrete Optical Readouts
The amplifier circuit consisted of five BC517 Darlington transistors (β = 30,000) with different values of R1 and R2 resistors ( Fig. 3b ). The enzymatic-electrochemical cell was regarded as an active electronic element, and the R1 and R2 resistors were carefully chosen to obtain a signal specific to a glucose concentration in the sample. Various combinations of R1 and R2 were tested. After the addition of an artificial urine sample with glucose to the cell, the glucose is oxidized on the conductive electrode and the current flowing through the cell is amplified and increases above the background current that exists in the absence of glucose. With the increasing concentration of glucose in urine, the resistance of the enzymatic cell decreased, resulting in higher transistor input voltage (Vin) and consequently brighter LED lighting ( Fig. 3c ). In all circuit configurations, 1 and 100 Ω resistors were used for R3 and R4, respectively, and the applied voltage (Vcc) was 3 V. The corresponding collector current (Ic) was measured by monitoring the voltage on R3 ( Fig. 3d ). A five-LED array (SSA-LXB525SRD purchased from Digi-Key) was used in all experiments.
Results and Discussion
The need for complex instrumentation such as a potentiostat and sophisticated external readers is a barrier for all existing electrochemical sensing techniques. 14 In 2017, our group reported a self-powered, paper-based glucose enzymatic biofuel cell for glucose monitoring in human sweat and showed its potential as a disposable, inexpensive, paper-based device platform. 9 Although the sensor showed a high sensitivity of 1.35 µA/mM and produced enough power and current to monitor glucose concentrations in sweat (0.02–1.0 mg/mL), the need for a voltage reader and different external loads to measure the maximum power and current was on the opposite side of the user-friendly spectrum of POC devices.
In this work, we report a simple glucose sensor without external readout instrumentation. Our sensor is the first demonstration of on-chip visual quantification of urine glucose concentrations. The device consists of five PEDOT:PSS/GOx paper electrodes, each of which functions as an active current source or resistor in a simple transistor-based amplifier circuit that is specifically tuned to detect a glucose threshold and correspondingly supply current to power the LED. The physiological threshold concentrations of glucose in urine were reliably detected with the developed semiquantitative electrochemical assay.
Sample Introduction and Working Principle
After the amplifier circuit and the disposable sensing strip were constructed and connected, various artificial urine samples with different glucose concentrations were introduced onto the five reservoirs on the paper strip. The sample volume was about 200 µL (approximately 40 µL for each electrode), ensuring that the whole surface of the electrode was covered. The samples wicked through the anodic enzyme reservoirs via capillary force, eliminating the need for any external pumps and electrical energy. The adsorption of the sample facilitated rapid attachment of the glucose in the sample to the conductive anode and promoted immediate electron transfer from the GOx. Because of the high conductivity and the unique electrochemical stability of the PEDOT:PSS in aqueous electrolytes, 15 the GOx was successfully encapsulated with the PEDOT:PSS matrix, resulting in effective GOx immobilization and promoting direct electron transfer between the center of the redox enzyme and the electrode matrix. Moreover, the large pores (11 μm) of the filter paper allowed a higher volume of the enzyme to be absorbed into the reservoir and, consequently, a greater sensitivity and a faster response time were achieved.4,16
The system we developed was based on a two-electrode amperometric configuration, five of which were connected to a specifically tuned transistor-based circuit and an optical sensing array. The number of active LEDs in the optical array was proportional to the discrete ranges of glucose concentration. The screen-printed electrodes were exposed to capillary-driven flow of artificial urine samples with different glucose concentrations. In the absence of an external voltage, the two-electrode enzymatic cell can be regarded as a simple resistor. However, as the voltage is applied, the glucose molecules in the PEDOT:PSS/GOx electrode can be oxidized to gluconolactone and deliver electrons by further electrochemical oxidation of H2O2 to O2 and the redox behavior of PEDOT:PSS.17,18
The flow of current through the enzymatic electrode resulted in voltage drop in the electrochemical cell (or a decrease in electrochemical cell resistance), leading to an increase in the current flow through the R1 and the base of the transistor. The appropriate choice of resistors R1 and R2 provided the desired limit of detection for each individual cell by adjusting the transistor threshold voltage and corresponding optical signal. Theoretically, when lower concentrations of redox compounds need to be detected, lower values of R1 and R2 and lower ratios of R2/R1 are required to increase the applied potential to the electrochemical cell, and vice versa, for detection of higher currents.
Semiquantitative Visual Screening of Glucose in Urine
Figure 4 shows collector current outputs of five transistors and the corresponding LED responses with different combinations of R1 and R2 resistors and varying concentrations of glucose in artificial urine (9 mg/dL [~0.5 mM], 27 mg/dL [~1.5 mM], 45 mg/dL [~2.5 mM], 63 mg/dL [~3.5 mM], and 81 mg/dL [~4.5 mM]). The resistor R1 adjusted the voltage across the enzymatic cell, while the resistor R2 controlled the minimum input voltage and current to obtain the desirable transistor operating point proportional to glucose concentration (R1 = 100 KΩ, R2 = 5.6 MΩ for ≥0 mM; R1 = 100 KΩ, R2 = 4.7 MΩ for ≥1 mM; R1 = 51 KΩ, R2 = 4.7 MΩ for ≥2 mM; R1 = 220 KΩ, R2 = 2.4 MΩ for ≥3 mM; R1 = 80 KΩ, R2 = 2 MΩ for ≥4 mM). The first LED (LED 1) corresponded to the 0–1 mM glucose concentration, which is a typical concentration in normal urine samples ( Fig. 4a ). When the concentration of glucose was in the normal range, the current was not enough to light up the remaining LEDs in the array. Simultaneous responses of LEDs 1–5 indicate the highest concentration of glucose in urine, larger than 4 mM. The LED responses were conducted in less than 2 min, which holds considerable promise for rapid and noninvasive glucose measurements, demonstrating that the entirely integrated sensing system with a simple readout platform can be realized toward POC diagnosis even in resource-limited countries.
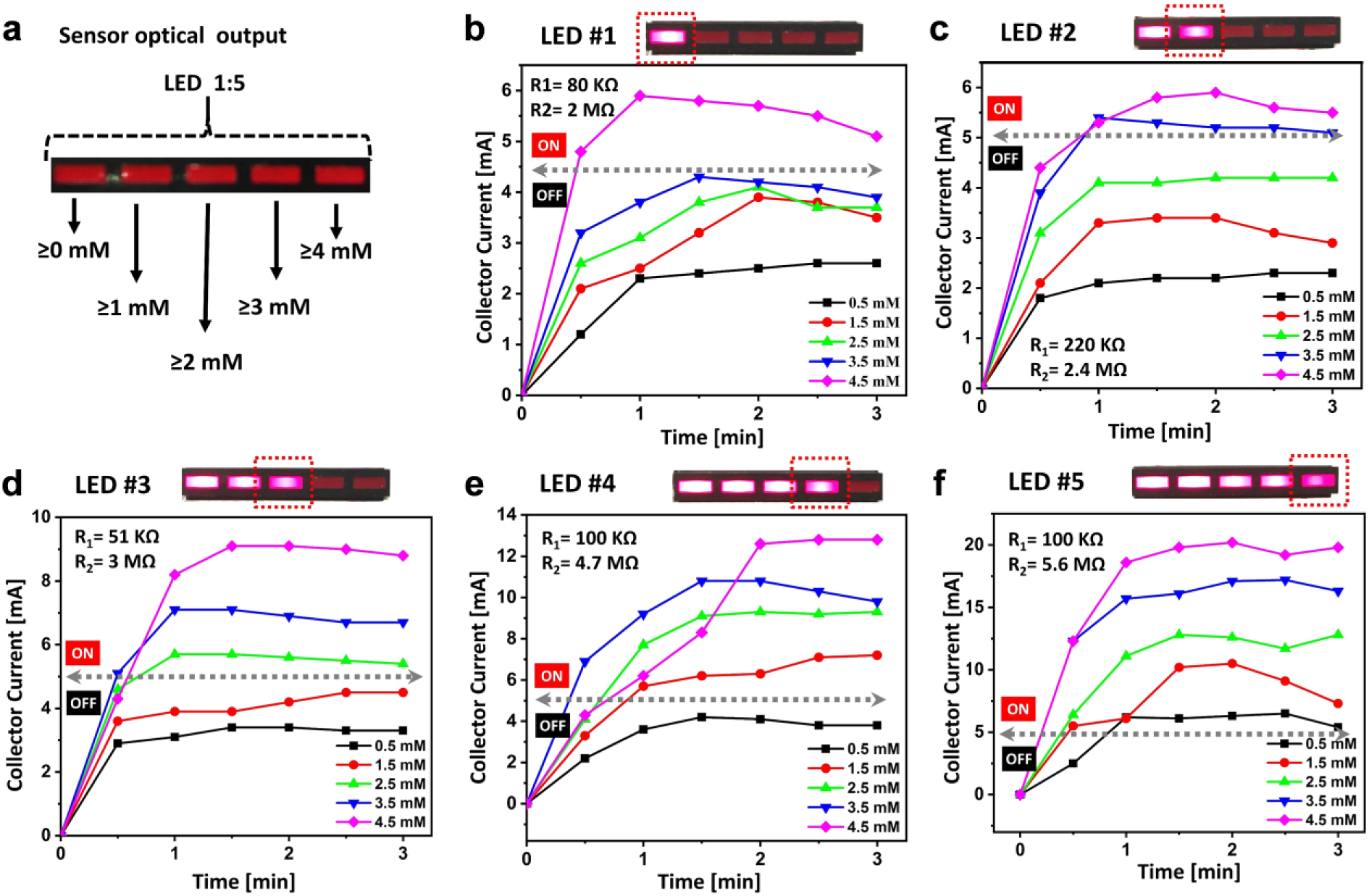
(
Glucose Monitoring
After demonstrating the circuit configuration to sensitively measure five glucose levels, we examined the reliability of the device with various unknown glucose levels in artificial urine. Repeated 8–10 samples were tested for each of the five detection levels, and the numbers of correct and incorrect classifications, as well as the invalid results, were recorded ( Fig. 5 ). More than 80% successful biosensing classification was achieved. This demonstration indicates that our sensor has a high potential for monitoring glucose concentrations with substantial performance improvement from conventional urinary glucose sensors.
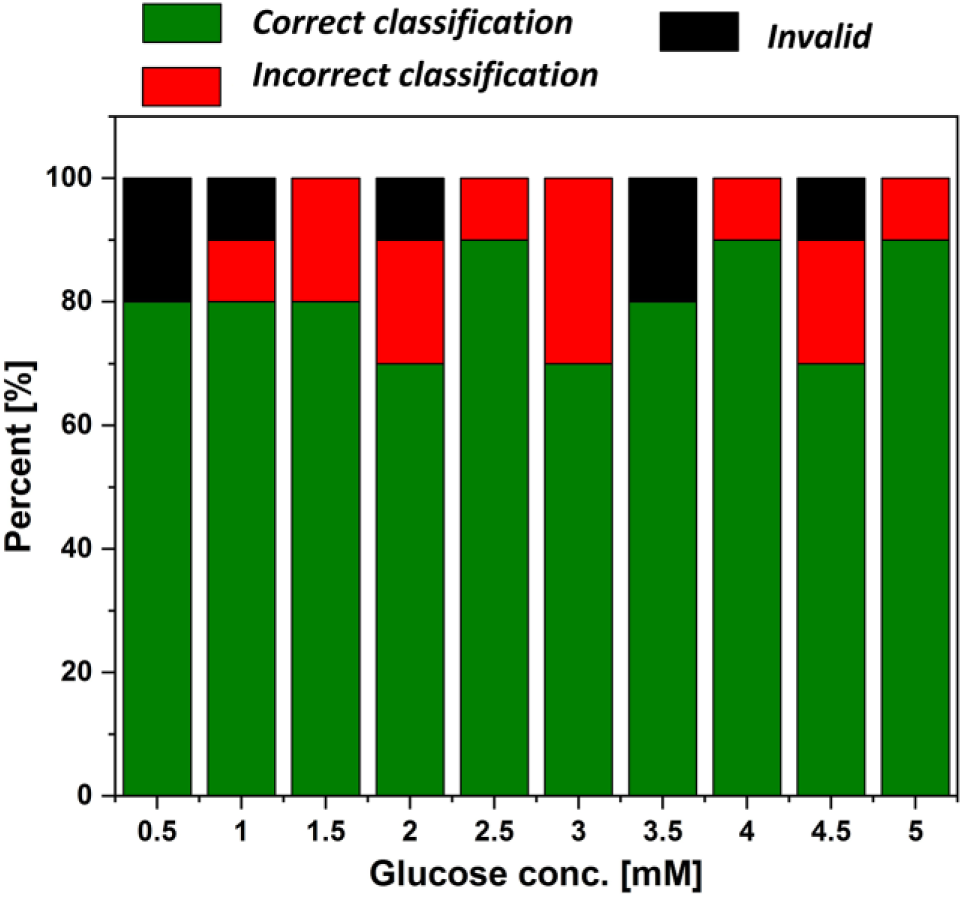
Percent of sensor responses to various glucose concentrations.
In this work, we reported a simple urinary glucose sensor by using the direct conversion of the amplified electrochemical signal to discrete visual perception to address the current limitations for developing low-cost, noninvasive, amperometric biosensing of glucose. These limitations mainly include the requirements for (1) complex instrumentation, (2) additional reference electrodes, (3) sophisticated external readouts, and (4) extensive data analysis. The proposed device allowed rapid, easy-to-use, low-cost, and noninvasive glucose monitoring in urine by using a paper-based PEDOT:PSS/GOx assay and a simple amplification circuit with a 5-LED indicator array. The proposed sensor platform will offer flexibility for more updated designs and will be transformative for various biological and chemical measurements.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Science Foundation (ECCS no. 1703394), Office of Naval Research (no. N00014-81-1-2422), Integrated Electronics Engineering Center (IEEC), and SUNY Binghamton Research Foundation (SE-TAE).