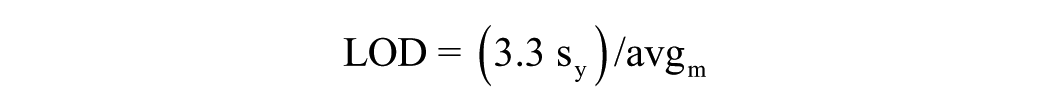Abstract
Catecholamines are integral neurotransmitters in the central and peripheral nervous system. Clinically, catecholamine levels are determined to help diagnose disease and measure corresponding therapeutic effectiveness. However, manual extraction of catecholamines and their metabolites may be labor-intensive and user-variable and require a variety of peripheral laboratory devices, especially at low sample concentrations. Here, we propose a novel solid-phase extraction (SPE) method using patented dispersive pipette extraction (DPX) tip technology. The tips are readily integrated into an automated workflow to extract these compounds from urine, which increases analytical throughput while removing human variability and error. Diphenylboronic acid (DPBA) forms a stable, negatively charged complex with catecholamines in the samples, and when aspirated into the DPX tip, the complexed analytes are retained on a styrene divinyl benzene sorbent. Wash buffers remove interfering compounds, after which the complex is eluted from the tip using an acidic aqueous solution and subsequently measured via liquid chromatography with tandem mass spectrometry (LC-MS/MS). The automated DPX method for catecholamine sample preparation from urine has excellent linearity over more than three orders of magnitude with concentrations ranging from 0.5 to 1000 ng/mL, with replicate analyses resulting in coefficients of variation of less than 8%. This high-throughput workflow is appropriate for use in regulated laboratories.
Introduction
The catecholamines, epinephrine, norepinephrine, and dopamine, are bioamines produced in the central nervous system, sympathetic nerves, and adrenal glands. Additionally, evidence suggests that lymphocytes and phagocytes are also capable of synthesizing catecholamines.
1
Catecholamines play an integral role as hormones and neurotransmitters in the central and peripheral nervous system. They are synthesized when
As essential compounds in the fight-or-flight response, catecholamine levels change in response to internal and external stimuli to influence body functions such as heart rate, blood pressure, and sugar and lipid metabolism. Additionally, catecholamines and their metabolites are implicated in a number of wide-ranging disease states, including pheochromocytomas, neurodegenerative disease, heart disease, and cancer.2–5 As such, robust and accurate screening of the catecholamines and their metabolites is a critical clinical tool to diagnose disease and track treatment efficacy.
Although catecholamines can be found throughout the body, blood plasma and 24 h pooled urine samples are typically used for catecholamine level determinations as their sampling methods are less invasive and more tolerated by patients. This eliminates stress-induced catecholamine production caused by the sample collection process. In the clinical laboratory, sensitivity to oxidative degradation and their highly polar nature present challenges during sample preparation and analysis of the catecholamines and their metabolites.
The structural properties listed above make sample preparation and analysis difficult. A wide range of sample preparation techniques have been employed to analyze catecholamines and metanephrines in urine, and most of these have been summarized by Bicker and coworkers. 6 Cation exchange mechanisms work well for metanephrines, but not the catecholamines, because of their susceptibility to oxidation during elution with base. Solid-phase extraction (SPE) with phenylboronic acid (PBA SPE) columns works well for sample preparation of catecholamines, because catechol groups bind with high affinity to the boronic acid groups to provide high recoveries. However, PBA SPE does not work well with metanephrines due to the absence of catechol groups. Whiting and coworkers compared published methods and reported reduced recoveries for catecholamines using cation exchange SPE with no recovery for metanephrines on PBA SPE. 7 Talwar and coworkers 8 developed an SPE method with the highest reported recoveries and discovered that diphenylboronic acid (DPBA) complexed and stabilized catecholamines in basic solution ( Fig. 1 ). This complex could then be extracted using C18 sorbent, followed by acidic media to disrupt the complex and elute the target compounds. DPBA forms reversible covalent bonds with the catechol of the catecholamines in basic solution, effectively minimizing oxidation and increasing lipophilicity for reverse-phase retention. Whiting and coworkers also noted that although metanephrines do not have the cis-diol moiety necessary for the complexation with DPBA, they exhibit high recoveries when the same extraction procedure is used. When DPBA is not present during extraction, metanephrine recovery is decreased by one-third. This suggests that DPBA interacts as an affinity-pairing agent. We have adapted the Whiting method to design a novel dispersive pipette extraction (DPX) that takes place within a pipette tip, which facilitates an easily automated alternative to traditional SPE requiring less sample and solvent volume. The objective of this study was to develop an automated sample preparation method utilizing the DPBA complexing agent to minimize sample preparation time (<15 min) and improve sensitivity (limit of detection [LOD] < 0.25 ng/mL) for the analysis of free catecholamines and metanephrines in urine with liquid chromatography with tandem mass spectrometry (LC-MS/MS).
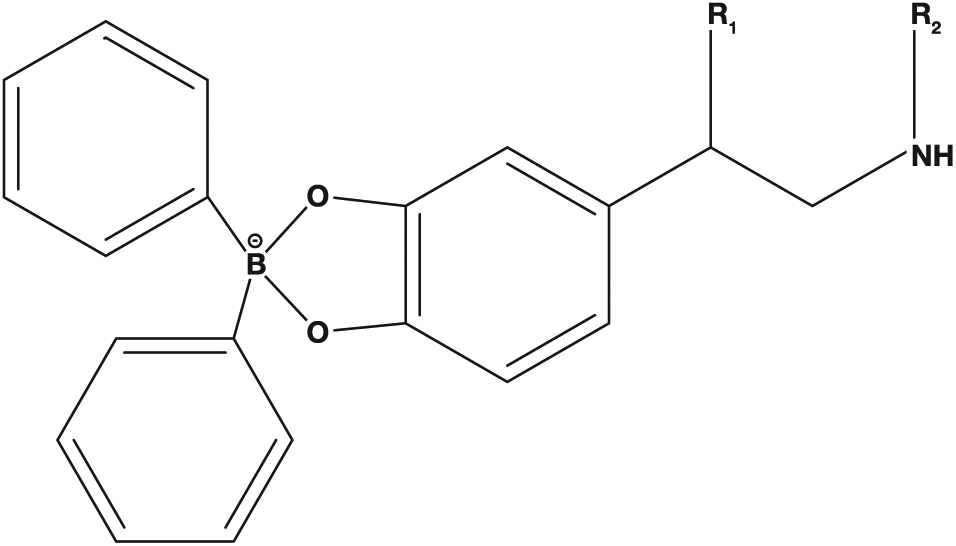
Diphenylboronate–catecholamine complex structure. (For norepinephrine: R1 = OH, R2 = H; for epinephrine: R1 = OH, R2 = CH3; for dopamine: R1 = H, R2 = H.)
The DPX method minimizes oxidation and maximizes analyte recoveries from biological samples intended for catecholamine analysis. Here, specialized pipette tips include an upper porous barrier (
Fig. 2A
) and a lower frit barrier (

DPX tips include an upper porous barrier (
In the example shown here, we apply the automated in-tip DPX technology workflow to the measurement of catecholamine levels in urine samples. DPBA is added to samples in a 96-well microplate to create a stable, negatively charged complex with the catecholamines in solution. When the complex is automatically aspirated into the specialized DPX tip, it is captured and retained on the aforementioned sorbent. Subsequent wash buffers remove any present interfering compounds, and an acidic aqueous/methanol solution is aspirated to disrupt the complex and disrupt any reverse-phase interactions. The low methanol content selectively elutes the catecholamines and eliminates the need for solvent evaporation. The purified sample is eluted from the tip into an assay plate and automatically transferred to an LC-MS system for analysis. The extraction process for 96 samples is performed in less than 15 min with no human involvement.
Materials and Methods
Materials
Samples and Reagents
The drug standards catecholamine mix 1, catecholamine mix 2, dopamine HCl, dopamine-D4 HCl, norepinephrine-D6 HCl, epinephrine-D6, normetanephrine-D3 HCl, and metanephrine- D3 HCl were purchased from Cerilliant Corporation (Round Rock, TX). Acros Organics ammonium chloride, Acros Organics diphenylboronic acid 2-aminoethyl ester (also called DPBA), 98%, and LC-MS-grade formic acid were sourced from Thermo Fisher Scientific (Waltham, MA). ClinChek Urine Controls, lyophilized, for biogenic amines, level I and ClinChek Urine Controls, lyophilized, for biogenic amines, level II were purchased from RECIPE (Munich, Germany). Surine synthetic urine was purchased from Dyna-Tek Industries (Lenexa, KS).
Consumables
DPX CO-RE RP tips (DPX170039) were donated by DPX Technologies, LLC (Columbia, SC). Hamilton sterile filtered 1 mL CO-RE tips were donated by Hamilton Company (Reno, NV). ABgene 96-well 1.2 mL polypropylene deep-well storage plates and Axygen single-well high-profile reagent reservoirs were purchased from Thermo Fisher Scientific.
Instrumentation
The Microlab NIMBUS96 from Hamilton Robotics is a personal pipetting workstation, configured with a 96-channel multipipetting head, choice of two deck styles, and air displacement pipetting volumes from 1 µL to 1 mL. NIMBUS96 was used to pipette all reagents and facilitate the DPX workflow ( Fig. 3 ). The NIMBUS96 deck was laid out according to Figure 4 . Off-deck analyses were performed using a Thermo TSQ Vantage triple quadrupole mass spectrometer (Milwaukee, WI) coupled to an Agilent 1260 Series HPLC (Agilent Technologies, Santa Clara, CA) equipped with a Restek 3 µm Ultra PFPP column (100 × 2.1 mm; Restek Corporation, Bellefonte, PA) with column temperature held at 40 °C. Sample injections were 10 µL.

Automated workflow describing the extraction of catecholamines from urine.
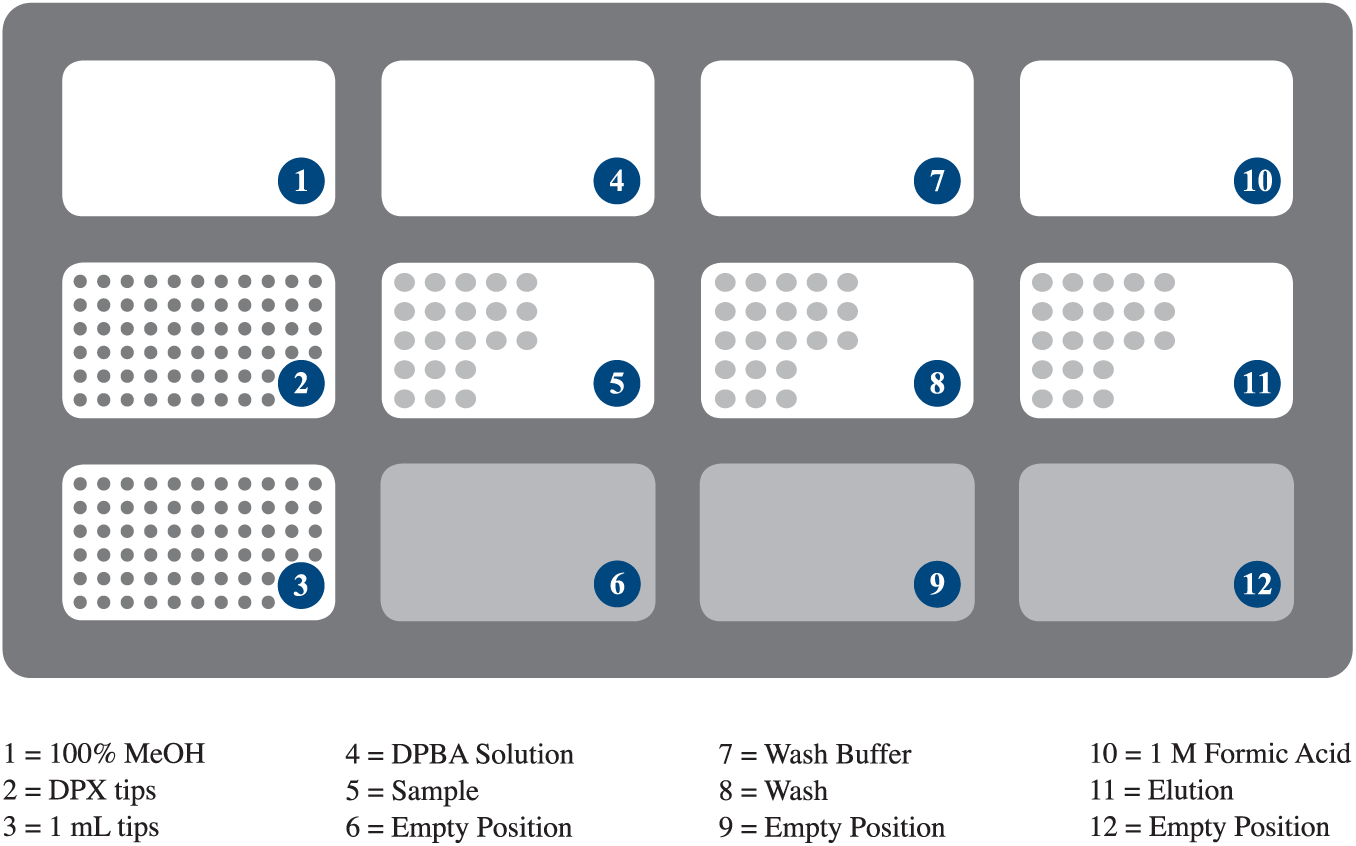
Microlab NIMBUS96 deck layout, showing reagent and sample positions, and tip stations.
The mobile phase was composed of 0.1% formic acid in water (A) and 0.1% formic acid in methanol (B). The gradient started at 4% B and was held for 2 min, and then ramped to 70% B at 3 min, where it remained until 3.75 min, when it was reequilibrated to 4% B, for a total run time of 5.5 min. Eluent was diverted to waste during the intervals of 0–0.5 and 4.0–5.5 min. The column flow rate was 0.3 mL/min. Mass spectrometer parameters were electrospray voltage, 3000 V; auxiliary gas pressure, 8 psi; sheath gas pressure, 30 psi; vaporizer temperature, 325 °C; and capillary temperature, 300 °C. The most abundant m/z was monitored, which for norepinephrine, normetanephrine, and metanephrine was the water-loss (M + H – H2O) product ion.
Methods
Level I and level II urine control samples were reconstituted according to manufacturer instructions. A total of 300 µL of urine sample and 10 µL of internal standard were manually added to each well of a 96-well assay plate. The assay plate was then loaded onto the deck of the Hamilton Microlab NIMBUS96 personal pipetting workstation. Disposable reservoirs containing assay reagents were also added to the deck. The NIMBUS96 used 1 mL CO-RE tips to automatically dispense 600 µL of complexing agent (0.2% [w/v] DPBA, 5 g/L EDTA in a 2 M NH4Cl/NH4OH, pH 8.5, buffer) to the urine sample well plate, 500 µL of wash buffer to a second plate for washing, and 270 µL of 1 M formic acid plus 30 µL of methanol to a third plate for elution. New tips were used to add each reagent and were then discarded. NIMBUS96 then picked up the 1 mL DPX reverse-phase tips. The tips were conditioned by aspirating and dispensing 100% methanol twice and aspirating and dispensing the wash buffer once. The sample solution was then aspirated and dispensed four times to ensure complexation and efficient retention on the styrene divinyl benzene resin within the DPX tip. The tips were rinsed with wash buffer, and then analytes were eluted with the 1 M formic acid/10% methanol solution into an elution plate. The elution plate was then moved to the autosampler where 10 µL sample volumes were injected into the LC-MS/MS for analysis.
Method Development
Linearity and Sensitivity
Linear least-squares regression with a 1/x weighting, where x denotes concentration, was used to fit calibration models based on nine calibration points for each analyte spanning the concentration range from 0.5 to 1000 ng/mL for norepinephrine and epinephrine and 0.1 to 1000 ng/mL for dopamine, normetanephrine, and metanephrine. Carryover was evaluated by running three replicate blank samples after each high calibrator. The LOD was calculated using the estimated standard deviation of the y intercept (sy0) and the average slope of the calibration (avg m ):
The limit of quantitation (LOQ) was similarly estimated with a multiple of 10 instead of 3.3. The average slope and standard deviation of the y intercept were taken from the analysis of three separate sets of extracted calibrators.
Accuracy and Precision
The accuracy and precision of the method were determined by evaluating two external quality controls. The level I control had mean values of 64 ng/mL for norepinephrine, 20 ng/mL for epinephrine, 194 ng/mL for dopamine, 325 ng/mL for normetanephrine, and 172 ng/mL for metanephrine. Level II had mean values of 174 ng/mL for norepinephrine, 39.8 ng/mL for epinephrine, 293 ng/mL for dopamine, 1568 ng/mL for normetanephrine, and 1017 ng/mL for metanephrine. Each control was analyzed in triplicate over three separate runs. The accuracy was calculated as shown below.
The within-run precision was determined by taking the standard deviation of a single run of triplicate quality control samples at a single concentration divided by the mean calculated value of that single run multiplied by 100%. The between-run precision was determined by taking the standard deviation of all observations (9 total) for each concentration divided by the grand mean for each concentration multiplied by 100%.
Extraction Efficiency and Matrix Effects
Matrix effects were determined using a postextraction addition technique, as referenced in the 2013 Scientific Working Group for Forensic Toxicology (SWGTOX) “Standard Practices for Method Validation in Forensic Toxicology.” 9 An unextracted neat solution of 100 ng/mL of each analyte was injected four times (set 1) into the LC-MS/MS. These results were compared with the results from four replicate samples of extracted synthetic urine spiked containing analyte at the appropriate concentration postextraction (set 2). Matrix effects were calculated as the mean area of set 2 divided by the mean area of set 1 subtracted from 1 and multiplied by 100%. A negative value represents ion suppression, while a positive value represents ion enhancement. Extraction efficiency was determined by comparing the matrix matched samples (set 2) to a set of synthetic urine samples that were fortified before extraction (set 3). Extraction efficiency was calculated as the mean area of set 3 divided by the mean area of set 2 multiplied by 100%.
Stability
Stability studies are beyond the scope of this paper, so data are not included here. The stability of catecholamines and metanephrines is discussed in the work of Grouzmann and Roberts.10,11
Results and Discussion
Linearity and Sensitivity
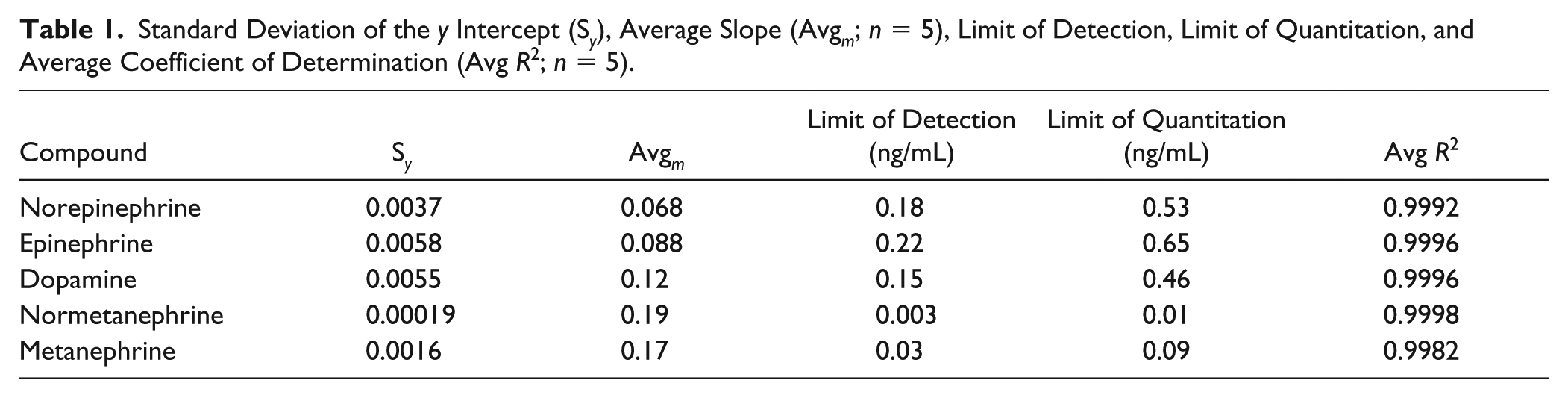
Calibrations resulted in average coefficient of determination (R2) values of 0.9992 for norepinephrine, 0.9996 for epinephrine, 0.9996 for dopamine, 0.9998 for normetanephrine, and 0.9982 for metanephrine. The average slope and y-intercept standard deviation values were used to determine the LODs and LOQs for each compound as described above and shown in Table 1 . The LOQ for each compound was similar to or better than those recently reported values.12–16
Standard Deviation of the y Intercept (S y ), Average Slope (Avg m ; n = 5), Limit of Detection, Limit of Quantitation, and Average Coefficient of Determination (Avg R2; n = 5).
Accuracy and Precision
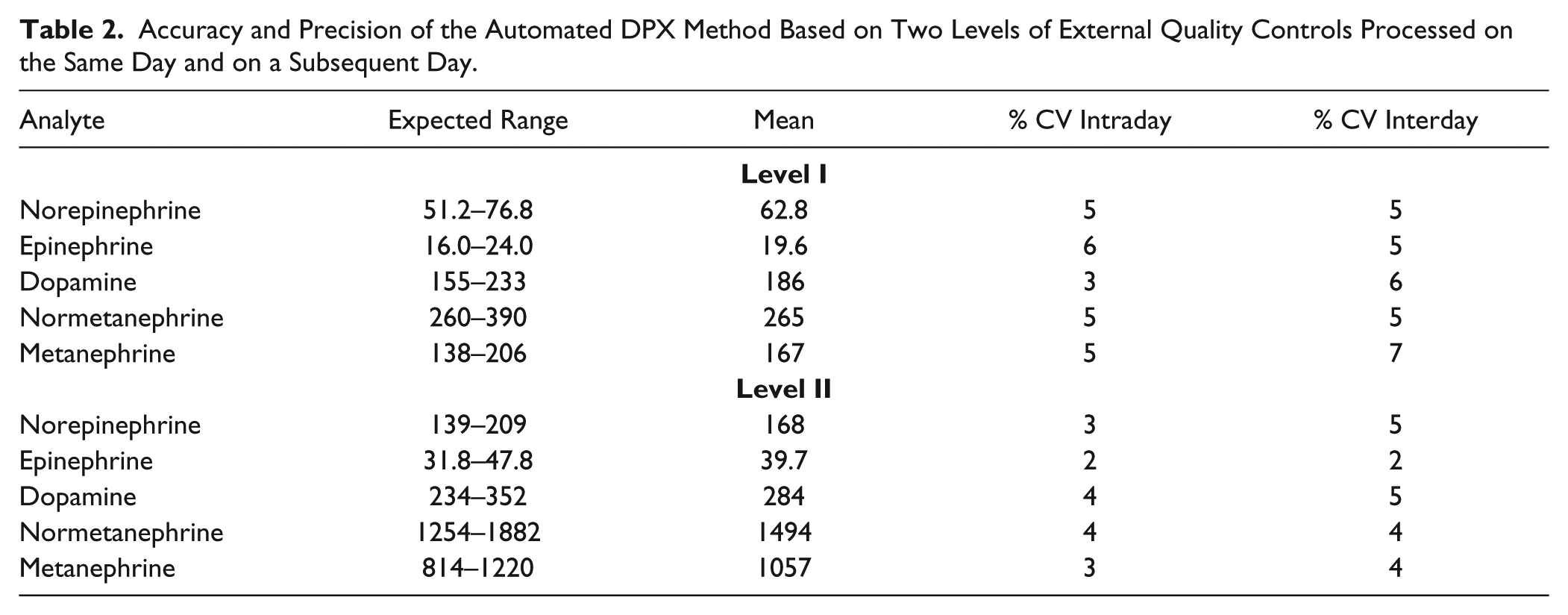
Accuracy, as well as within-run and between-run precision (% CV), is provided in Table 2 . The method was very accurate for quantitation of quality control samples, with each average analyte concentration falling within the manufacturer’s listed expected range of concentrations. The average within-run precision had a maximum CV of 6% for the level I epinephrine control, and the between-run precision had a maximum CV of 7% for the level I metanephrine control.
Accuracy and Precision of the Automated DPX Method Based on Two Levels of External Quality Controls Processed on the Same Day and on a Subsequent Day.
Extraction Efficiency and Matrix Effects
A sample chromatogram of the separation of analytes at 10 ng/mL is illustrated in Figure 5 . Matrix effects were relatively low for epinephrine, dopamine, normetanephrine, and metanephrine, with a range of ion suppression from 1% to 14%. Norepinephrine exhibited greater matrix effects with ion suppression at 39%, likely because it is the most polar compound with little to no retention on the PFPP column. Extraction efficiencies were higher than 96% for all analytes except dopamine, which resulted in 81% extraction recovery ( Fig. 6 ).
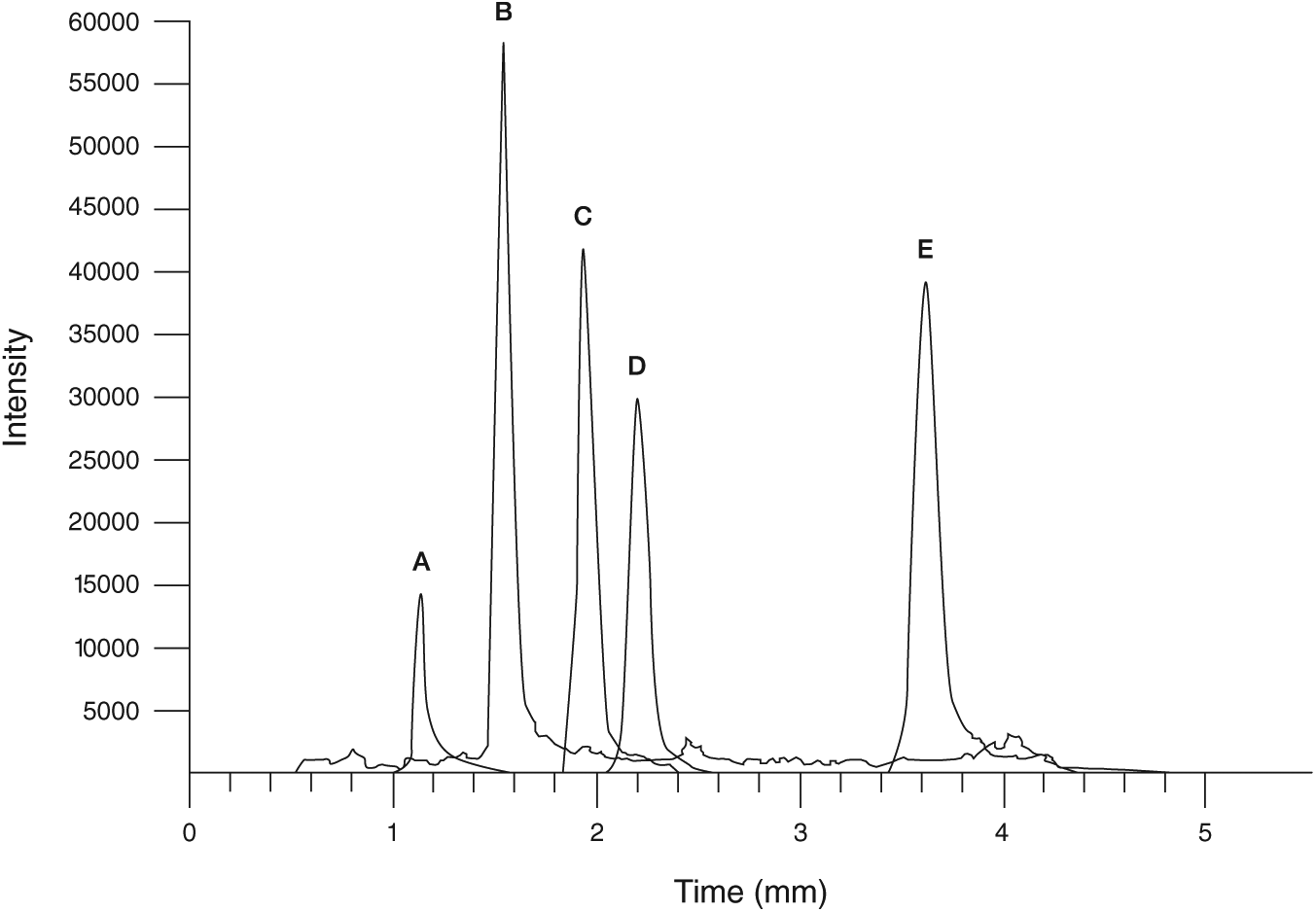
Separation of analytes at 10 ng/mL: (
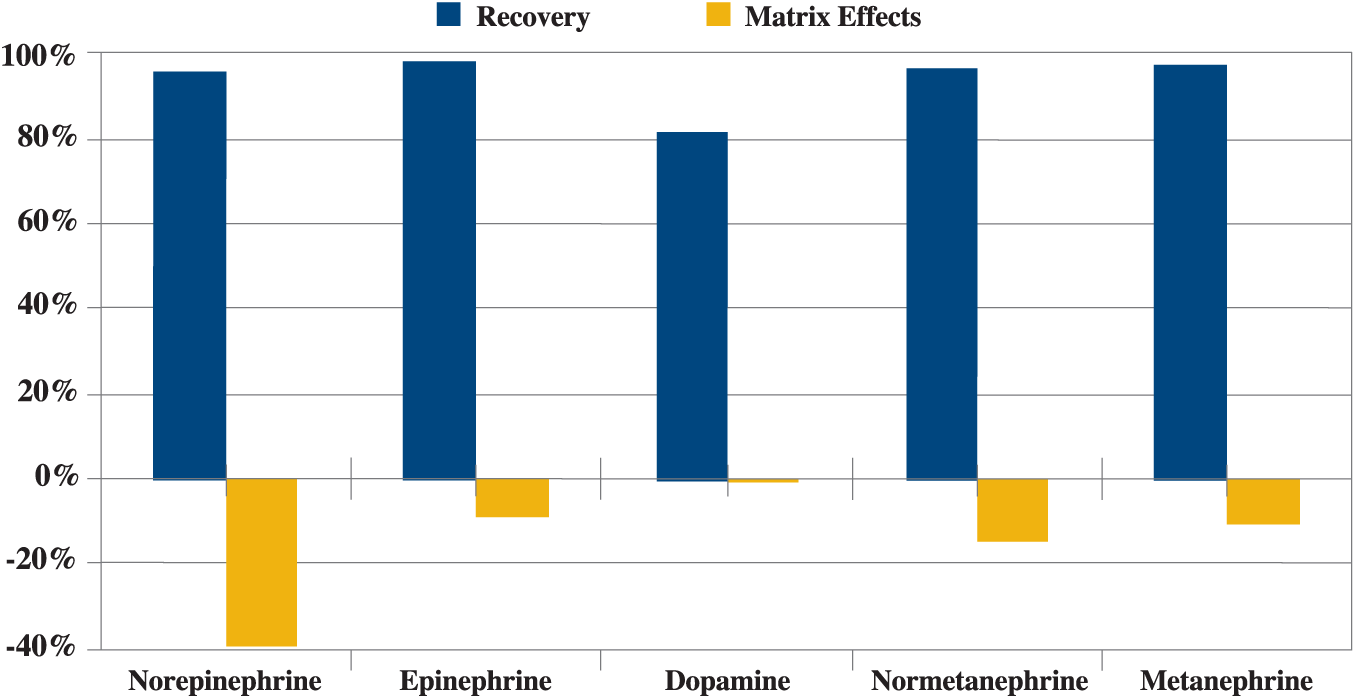
Recovery and matrix effects of catecholamines and metanephrines extracted using the automated DPX method.
DPX technology facilitates extraction of DPBA complexed catecholamines and metanephrines from urine in an automated workflow. By combining this novel SPE technique with a personal pipetting workstation such as the Hamilton Microlab NIMBUS96, users can extract up to 96 samples in less than 15 min. The method to purify samples prior to LC-MS/MS analysis provides the necessary analytical sensitivities without additional solvent evaporation steps, centrifuges, or separate filtration systems.
The combination of automation and tip technology was robust, with linear calibration (R2 > 0.998) over more than four orders of magnitude with concentrations ranging from 0.5 to 1000 ng/mL. Replicate analyses of level I and level II urine controls demonstrated less than 8% CV. The method is an excellent alternative to those previously published, given its ease of implementation, robustness, and high sensitivity, due to enhanced sample cleanup.
Footnotes
Acknowledgements
The data were previously presented at the SLAS 2018 International Conference & Exhibition, February 3–7, 2018, poster ID 1066-B, “Automated Dispersive Pipette Extraction of Catecholamines and Metabolites in Urine Using LC-MS/MS.”
Declaration of Conflicting Interests
The authors disclosed the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kaylee Mastrianni and William E. Kemnitzer are employed full-time by DPX Technologies in their respective positions. DPX Technologies manufactures and sells DPX technologies such as the type discussed in this paper. Kevin Miller is employed full-time by Hamilton Robotics as the Market Segment Leader, Government and Regulated Laboratories. Hamilton Robotics manufactures and sells automated liquid handling platforms such as the one discussed in this paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.