Abstract
We have implemented a simple, inexpensive, and fast procedure for validation and verification of the performance of pipettes mounted on automated liquid handlers (ALHs) as necessary for laboratories accredited under ISO 17025. A six- or seven-step serial dilution of OrangeG was prepared in quadruplicates in a flat-bottom 96-well microtiter plate, manually using calibrated pipettes. Each pipette of the liquid handler (1–8) dispensed a selected volume (1–200μL) of OrangeG eight times into the wells of the microtiter plate. All wells contained a total of 200μL liquid. The absorbance was read, and the dispensed volume of each pipette was calculated based on a plot of volume and absorbance of a known set of OrangeG dilutions. Finally, the percent inaccuracy (%d) and the imprecision (%CV) of each pipette were calculated. Using predefined acceptance criteria, each pipette was then either approved or failed. Failed pipettes were either repaired or the volume deviation was compensated for by applying a calibration curve in the liquid-handler software. We have implemented the procedure on a Sias Xantus, an MWGt The ONYX, four Tecan Freedom EVO, a Biomek NX Span-8, and four Biomek 3000 robots, and the methods are freely available.
In conclusion, we have set up a simple, inexpensive, and fast solution for the continuous validation of ALHs used for accredited work according to the ISO 17025 standard. The method is easy to use for aqueous solutions but requires a spectrophotometer that can read microtiter plates.
Introduction
Automated liquid handlers (ALHs) are increasingly used to improve throughput, pipetting accuracy and prevent occupational injuries to the technical staff due to intensive manual pipetting. 1 Standardized procedures for volumetric verification of standard piston operated pipettes exist and many laboratories verify their pipettes with defined intervals. 2 However, less standardized is the routine verification and validation of ALHs although various methods have been suggested for verification of the actual volume dispensed by ALHs, such as gravimetric, 3 –5 fluorescence, 6,7 photometric, 8 and combinations of different methods. 6 Commercial solutions have also been introduced. 8 –10 Testing laboratories that work under the ISO 17025 standard and perform its own calibration shall have and shall apply a procedure to estimate the uncertainty of measurement for all calibration and types of calibrations (ISO 17025:2005 § 5.4.6.1). 11 Furthermore, equipment and its software used for testing, calibration, and sampling shall be capable of achieving the accuracy required and shall comply with specifications relevant to the tests and/or calibrations concerned (§ 5.5.2). 11 Such calibrations must be performed with regular intervals and whenever needed. The vendors of ALHs have chosen various methods for validating and calibrating their instruments. The methods are often costly, require to be installed on each instrument and are not always well documented. More critically, methods applied by most vendors do not necessarily fulfill the demands of ISO 17025 because the calibration has to be performed on the actual pipetting techniques and liquid classes applied in the validated methods. Furthermore, if the laboratory has a number of different ALHs, implementation, calibration, optimization, validation, and trouble shooting concerning volume deviations may be time-consuming if the calibration methods vary too much. We have implemented a simple, inexpensive, fast, and easy-to-use procedure for verification and validation of dispensation of aqueous solutions by ALHs equipped with one through eight pipettes. The procedure is suitable for volumes between 1 and 200μL and requires a spectrophotometer that can read microtiter plates.
Experimental
Preparation of Solutions
A 10 mg/mL stock solution of OrangeG (O7252, Sigma) in MilliQ water was prepared in a 500 mL blue cap flask wrapped in aluminum foil to prevent photo-induced decomposition. The stock solution was stirred vigorously for 24 h on a magnetic stirrer. From the stock solution, two working solutions were prepared. Working Solution A was a 1:7.5 dilution of the stock solution used for volumes less than or equal to 10μL. Working Solution B was a 1:75 dilution of the stock solution used for volumes greater than 10μL.
Preparation of Standard Row
For volumes less than or equal to 10μL, a six-step standard row was prepared in 1.5 mL Eppendorf tubes using Working Solution A according to Table 1. For volumes greater than 10μL, a seven-step standard row was prepared in 1.5 mL Eppendorf tubes using Working Solution B according to Table 1. Standard rows were prepared either by manual pipetting or by means of a calibrated ALH.
Preparation of standard rows

The standard rows were distributed into 96-well flat-bottomed microtiterplates (BD Falcon microtest, Franklin Lakes, NJ) according to the layout presented in Figure 1. The remaining wells of the microtiterplates were then filled with MilliQ water. All used wells contained a total of 200μL after addition of OrangeG on the ALHs. Hence, if an ALH was to be tested for pipetting a fixed volume of 50 μL, the wells in columns one through eight were filled with 150 μL MilliQ water.
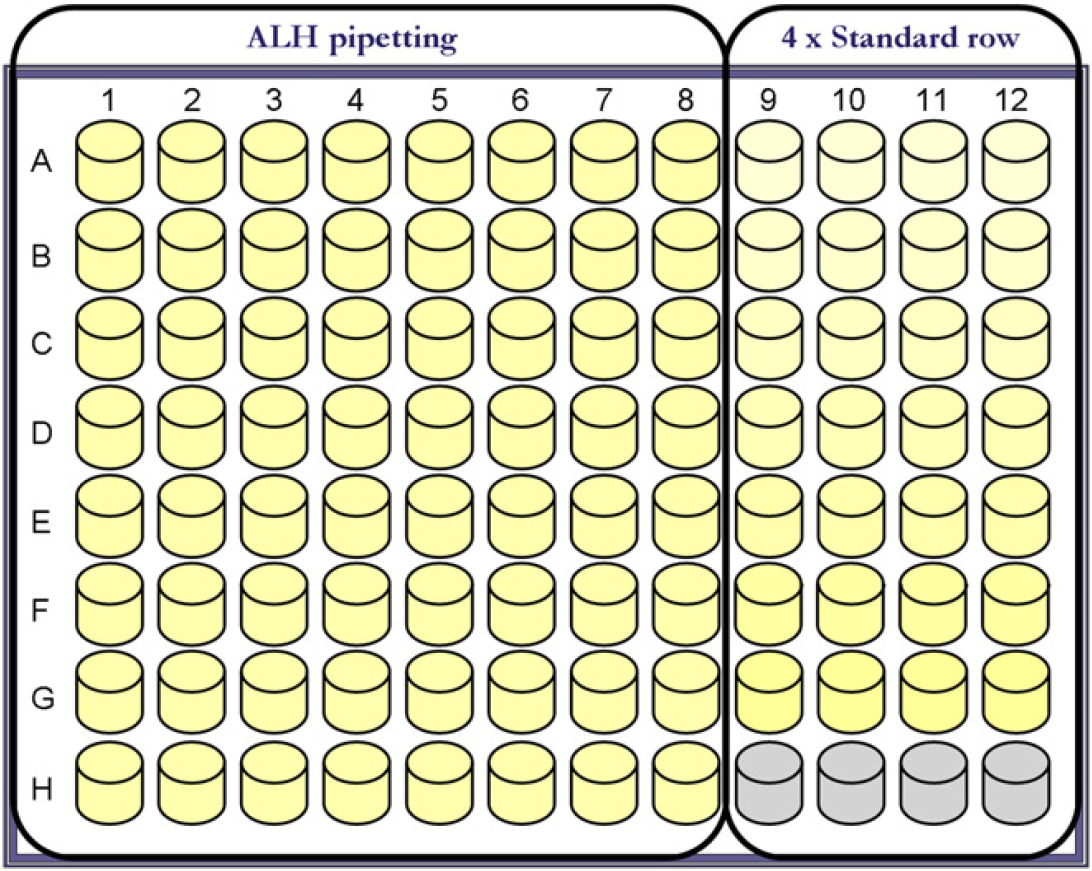
Plate layout. Columns one through eight were used for volume verification on automated liquid handlers. Columns nine through twelve were used for standard rows. The illustration indicates the plate layout used for volumes greater than 10μL. For volumes equal to or less than 10μL, only six standards were used.
Addition of OrangeG on ALHs
On each ALH, we constructed methods for the various volumes to be tested. Whenever possible, the volume was made a variable enabling the same method to be used for a series of volumes. Volumes less than or equal to 10μL were dispensed using the same method. Volumes greater than 10μL were dispensed using a separate method. Whenever appropriate, the methods used the same pipetting techniques, pipetting templates, and liquid classes or types, as methods used for routine work. For liquids with physically different properties than the OrangeG solution, for example, viscous, volatile, or detergent containing liquids, a more appropriate liquid type or class was used.
Absorbance Measurements
After addition of OrangeG, the plates were removed from the ALH and covered by a plate seal (AB-0558, ABgene, Epsom, UK) and incubated on a shaker at 150 RPM (Labline Orbit Shaker model 3520, Melrose Park, IL) for 1 h before absorbance measurements. The absorbance was read in a Sirio S spectrophotometer (Radim, Pomezia, Italy) at 450 nm using the accompanying software BrioWin Ver. 2.10 (SEAC, Firenze, Italy). The plates were read twice and the results were compared. If results from the two readings were inconsistent, the plate was read again.
Data Analysis
The absorbance readings from the standards were averaged and a standard curve was calculated using Excel and linear regression. Only plates having a standard curve with R
2 greater than 0.99 were further processed. Plates having a standard curve with R
2 less than 0.99 were inspected and, if necessary repeated. Using the standard curve, the volumes dispensed by the individual pipettes on the ALHs were calculated from averaged values of the eight or more replicates in columns one through eight. The calculated volumes were compared with the intended volume and an inaccuracy identifier, %d and an imprecision identifier, the coefficient of variance (%CV) were calculated for each pipette signifying the deviation from the intended volume and the repeatability of the individual pipette on the individual ALH. Two sets of criteria were used to approve different volumes on different ALHs. For volumes less than or equal to 10μL, values less than |20| for %d and %CV were accepted. For volumes greater than 10μL, values less than |10| for %d and %CV were accepted. The process flow is summarized in Figure 2. The values of %d were calculated using eq 1, where the observed averaged value (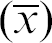

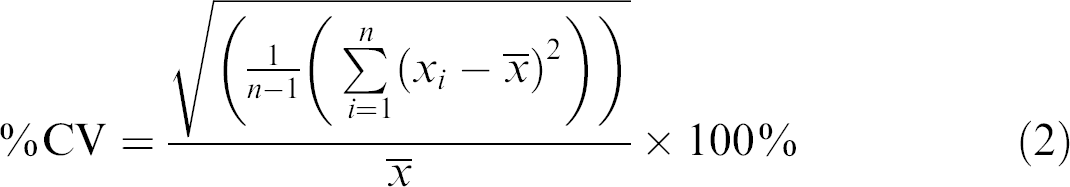
An Excel template containing the above-mentioned calculations and an accompanying guide is available as Supplementary data.
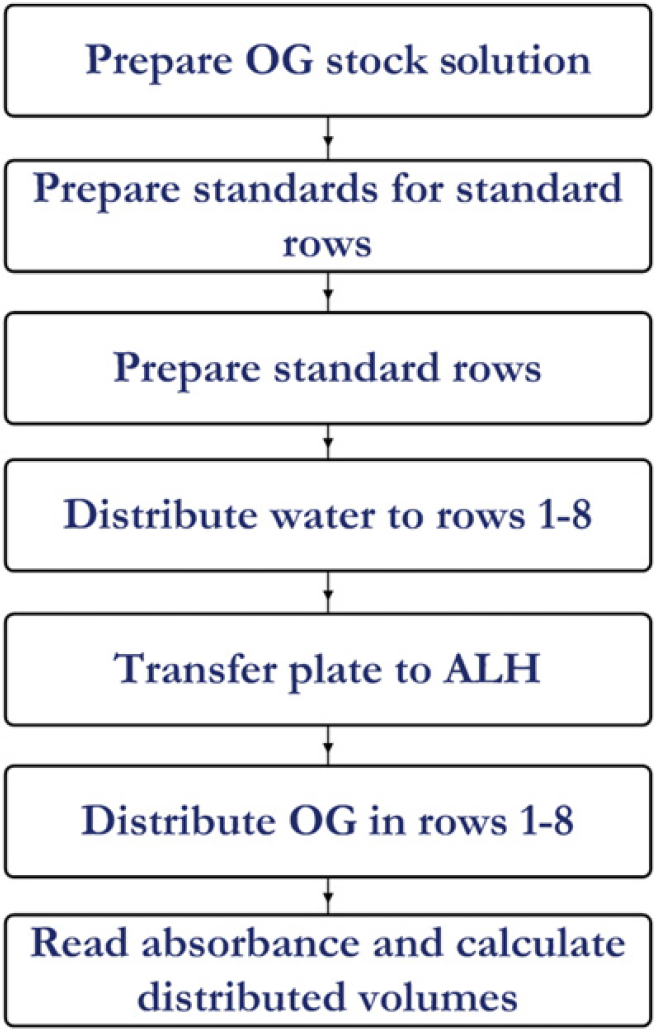
Process flow for the volume verification technique.
Results and Discussion
ALHs rely on pipetting techniques and/or liquid classes for accurate pipetting of various liquids in various volumes. The pipetting techniques in combination with liquid classes are used to compensate for variances in liquid viscosity, vapor pressure, hydrophobicity, and the volume dispensed. A general rule of thumb for accurate pipetting dictates slow aspiration and semifast to fast dispense speeds in combination with pauses of varying lengths. These pipetting properties can be fine-tuned in most liquid-handler software by using custom-optimized pipetting techniques for individual pipetting operations. Consequently, an ALH may use many different liquid classes and pipetting techniques during the execution of a single method. As the pipetting performance of the ALH is inevitably changed by altering the aspirate or dispense speeds, the validated pipetting techniques and/or liquid classes should in principle all be verified on routine basis.
As no suitable procedure for verifying the volumes dispensed by different ALHs using different pipetting techniques or liquid types exists, we developed one. Initially, the procedure tested using manual pipetting with a conventional plunger–operated pipette to determine the usefulness of the technique. The results were 0.3–0.2 μL lower than expected (Fig. 3). This deviation may be the result of heating of the pipette by simple transfer of body heat during the pipetting operation or bad manual pipetting performance. Hence, the procedure was found acceptable for use on ALHs.
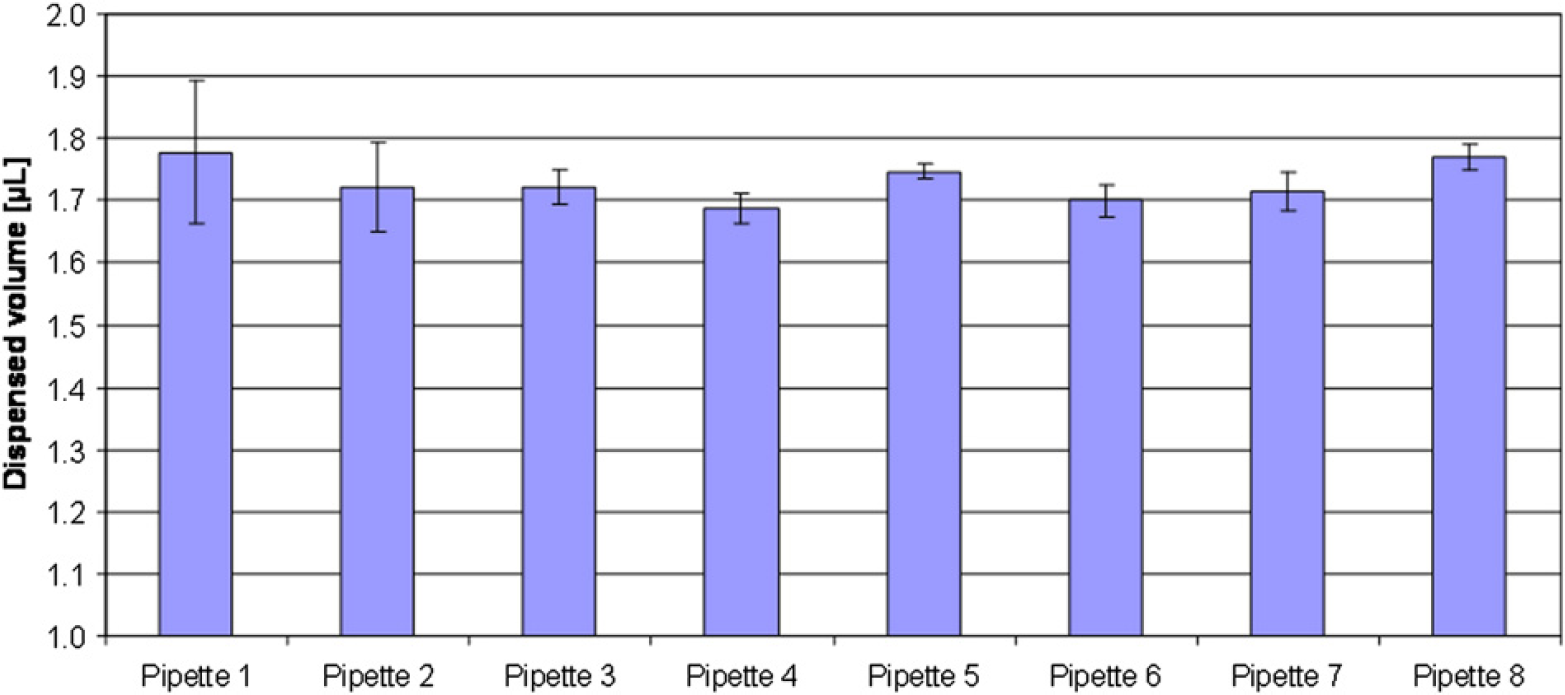
Manual pipetting. A manual plunger–operated pipette was set to 2 μL target volume, and 2 μL Working Solution B was distributed into columns one through eight that were prefilled with 198 μL MilliQ water. Columns nine through twelve all contained a six-step dilution row of OrangeG.
Validation of a new method or optimization of an existing method on an ALH under the ISO 17025 standard entails among other actions that the actual volumes transferred by the individual ALH must be verified and optimized if required. Utilization of the absorbance-based procedure described here makes it possible to asses and optimize the performance of the individual pipetting techniques for the desired volumes using the actual ALH and instrument-specific settings and in the physical environment in which the ALH is to operate under the ISO 17025 accreditation. As presented in Figure 4, utilization of two different pipetting techniques for pipetting the same volume resulted in two different volumes delivered. However, it should be noted that it is difficult, in most ALH softwares, to compensate for inherent differences between the instrument pumps. As an example, an MWGt The ONYX fitted with four fixed teflon-coated pipette tips was found to have a high degree of variation between the various pipettes (Fig. 5 A). After exchange of filters for system liquid and testing of pipette pumps, the pipetting performance was improved (Fig. 5B).
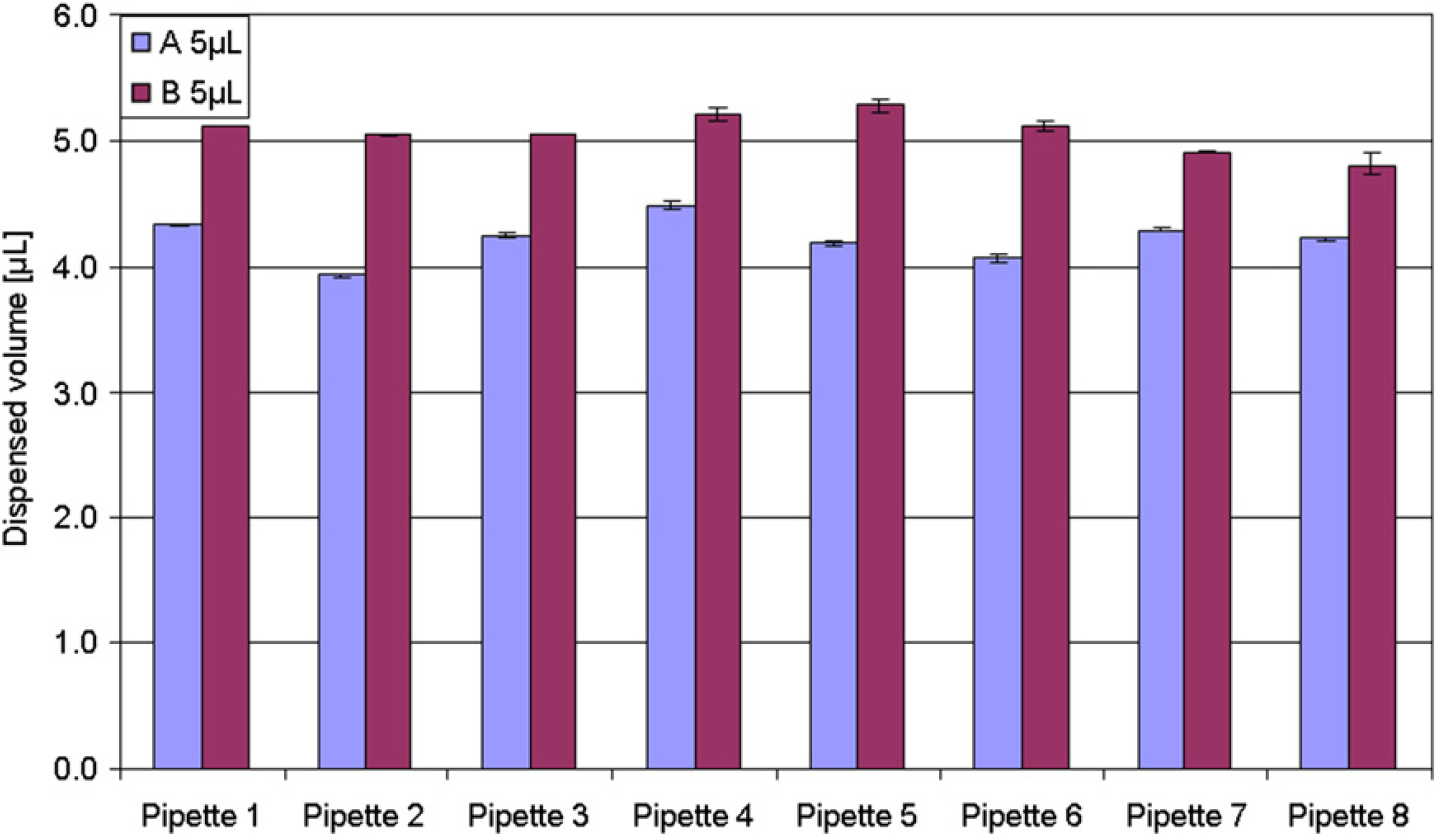
Pipetting technique optimization. Using a Sias Xantus ALH equipped with eight-channeled teflon-coated fixed tips, the procedure was used to optimize the pipetting technique that was used to deliver 5.0 μL.
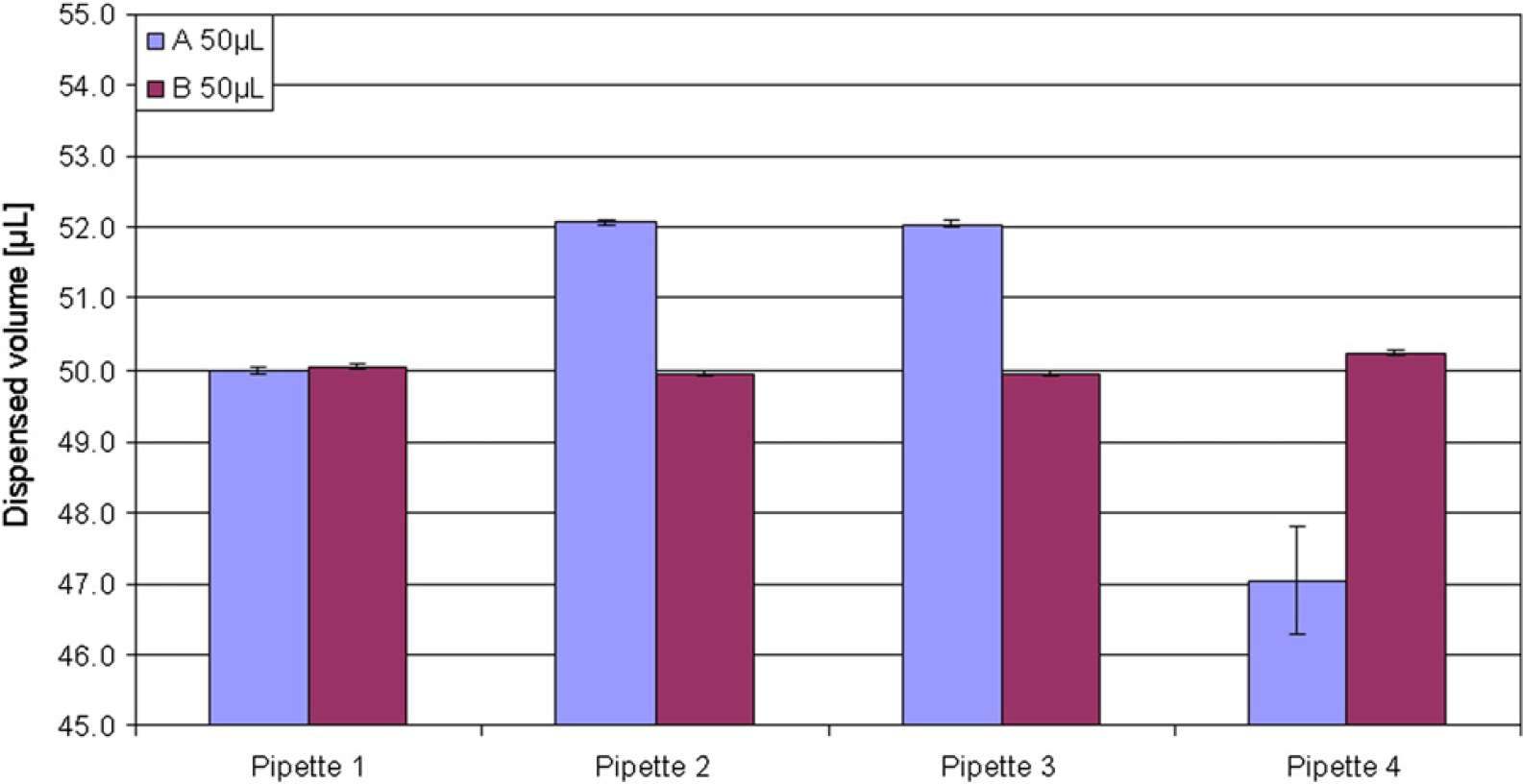
Identification of large variation between pipettes on an MWGt The ONYX liquid handler. The automated liquid handler was in both A and B set to dispense 50.0 μL.
Absorbance-based method relies on a standard curve of OrangeG. 3 In the present procedure, the standard rows were prepared in quadroplicates either by manual pipetting or with another recently verified ALH. Unlike another method, 6,7 the present method does not rely on fluorescent dyes but simply relies on OrangeG absorbance readings. This reduces the technical requirements to the platereader and eliminates precautions taken to prevent bleaching of the fluorescent dyes after exposure to light. OrangeG has been reported to photo-decompose slowly without catalysts at standard pH under visible or sunlight exposure. 12 The slow rate of decomposition without the catalyst eliminates the requirement for wrapping the plate in photo protective material, resulting in easy handling of the prepared plates before absorbance reading. Reading of a plate after exposure to light for a total of 75 h interrupted by periodic reading of the plate only showed signs of photobleaching following days of exposure (Fig. 6). As the procedure easily can be completed within one working day, photobleaching is unlikely to influence the results significantly. Some ALH systems are available with software controlled microscale that can be used to calibrate the unit. 3 Unlike the absorbance-based method, it can be applied to volumes >200μL and to liquids with low viscosity. However, this simple but laborious Gravimetric method 5,6 requires a software-controlled integrated microscale on each ALH. The absorbance-based method is a simple, fast, and inexpensive technique that enables volume verification of most ALHs and does not require purchase of additional instrumentation whenever a new ALH is purchased. Furthermore, the method can be applied to ALHs in pre- and post-PCR laboratory environments using the same platereader.
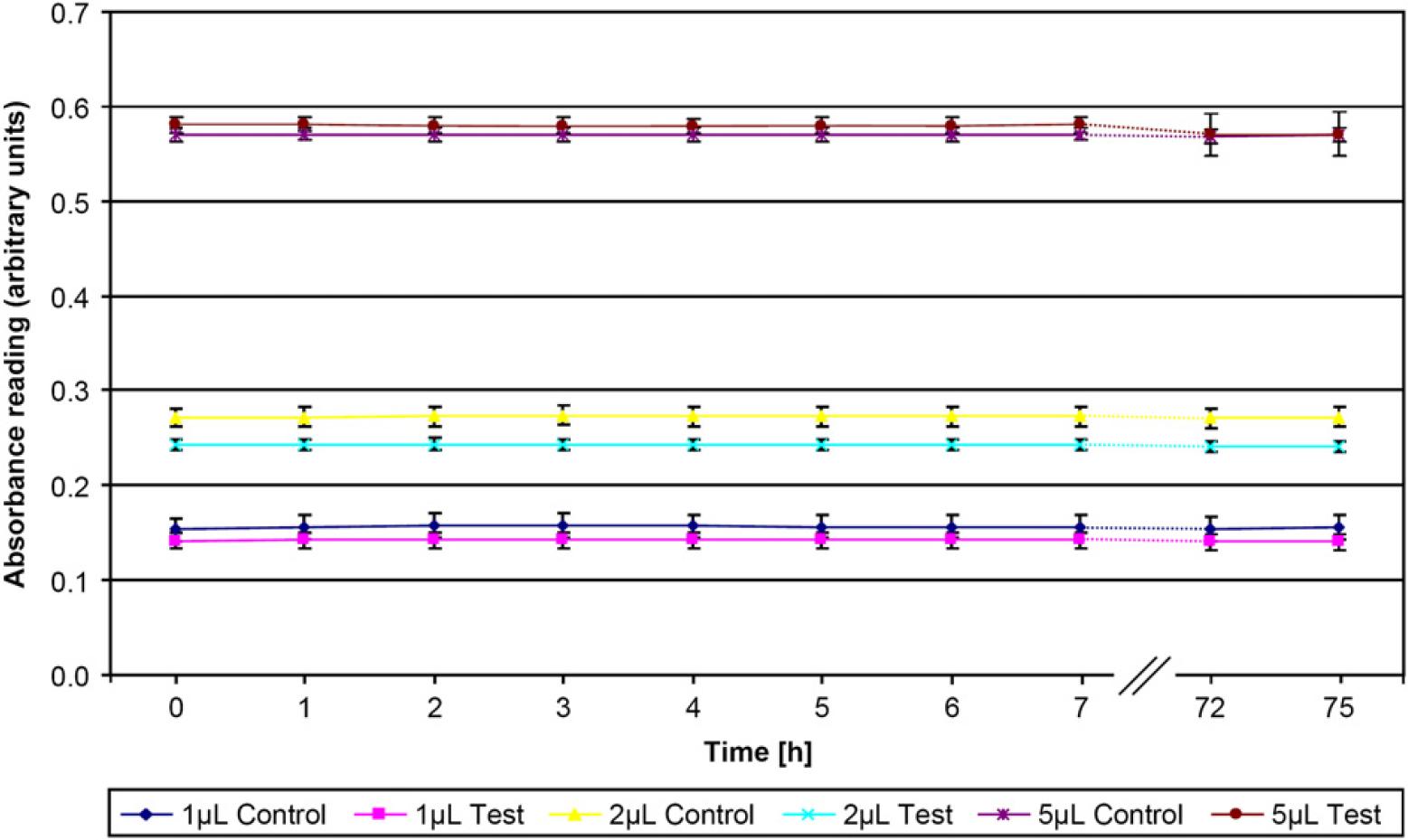
Time course experiment of photodecomposition rates. Six different plates were prepared for testing with two different Biomek 3000 automated liquid handlers. After initial absorbance reading, plates from one liquid handler were left exposed to the sun and room lighting without photo protective material. Plates from the other liquid handler were following absorbance readings wrapped in photo protective material and left adjacent to the other plates. The absorbance readings were repeated every hour for one working day. The plates were then left exposed to the sun and room lighting for an additional 65 h before additional absorbance readings.
The absorbance-based technique presented here is routinely used at the Section of Forensic Genetics at the University of Copenhagen for validating new ALHs. Furthermore, over time the performance of pumping devices on individual ALHs may perform differently. Hence, the technique is used to verify pipetting performance with predefined intervals according to the ISO 17025 standard.
We have no experience with ALHs equipped with 96-well or 384-well heads, but we think that only minor modifications will be needed for the application on ALHs with, for example, 96- or 384-well heads.
Conclusions
Advanced and precise pipetting can be performed using ALHs. To ensure the reliability of the systems, ALHs need frequent verification of the actual dispensed volume similar to manually operated pipettes. The technique presented here allows for fast and inexpensive testing of most ALH pipetting in microtiterplates. The technique in its present form does not easily allow for verification of dispensed volumes in automatic liquid handlers equipped with a 96-pipette or 384-pipette head. The technique is simple to use and does not require extensive training of the technical staff. The method allows testing laboratories that work under the ISO 17025 standard to perform their own calibration of ALHs and calculate the uncertainty of the dispensed volumes.
Supplementary Data
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.jala.2009.06.004.
Competing Interests Statement: The authors certify that they have no relevant financial interests in this manuscript.
