Abstract
Fumarate hydratase (FH) is a metabolic enzyme that is part of the Krebs cycle and reversibly catalyzes the hydration of fumarate to malate. Mutations of the FH gene have been associated with fumarate hydratase deficiency (FHD), hereditary leiomyomatosis and renal cell cancer (HLRCC), and other diseases. Currently, there are no high-quality small-molecule probes for studying human FH. To address this, we developed a quantitative high-throughput screening (qHTS) FH assay and screened a total of 57,037 compounds from in-house libraries in dose–response. While no inhibitors of FH were confirmed, a series of phenyl-pyrrolo-pyrimidine-diones were identified as activators of human FH. These compounds were not substrates of FH, were inactive in a malate dehydrogenase counterscreen, and showed no detectable reduction–oxidation activity. The binding of two compounds from the series to human FH was confirmed by microscale thermophoresis. The low hit rate in this screening campaign confirmed that FH is a “tough target” to modulate, and the small-molecule activators of human FH reported here may serve as a starting point for further optimization and development into cellular probes of human FH and potential drug candidates.
Keywords
Introduction
Fumarate hydratase (FH) is an important metabolic enzyme in the Krebs cycle, which reversibly catalyzes the conversion of fumarate to malate (hydration). It is encoded by the FH gene, which produces two isoforms of protein by alternative splicing, a cytosolic form and a mitochondrial form. The mitochondrial form of FH contains an N-terminal mitochondrial targeting sequence, which is removed in the mitochondrion to generate the same protein as that in the cytoplasm. Despite decades of research, a detailed understanding of the mechanism of FH’s biochemical activity remains a matter of debate. The active enzyme is a homo-tetramer in which each active site is made up of residues at the interface of three of the four subunits. 1 FH participates in adenosine triphosphate (ATP) production in mitochondria and the metabolism of amino acids and fumarate in cytoplasm.
There are more than 100 known mutations in the human FH gene. The most common type is missense (57%), followed by frameshifts and nonsense (27%), and other mutations. 2 Mutations of the FH gene lead to defects of the enzymatic activity either by indirectly compromising the integrity of the protein’s core architecture or by directly affecting residues within the active site that regulate catalytic activity of the enzyme. 3 FH gene mutations have been mainly associated with two heritable diseases: fumarate hydratase deficiency (FHD) and hereditary leiomyomatosis and renal cell cancer (HLRCC). Homozygous germline mutations in the FH gene cause autosomal recessive FHD syndrome, which is characterized by early-onset hypotonia, profound psychomotor retardation, seizures, facial dysmorphism, and brain abnormalities. 4 Mutations of the FH gene severely impair the enzyme activity, which leads to the defect of energy production and the accumulation of fumarate, which is believed to be the cause of clinical symptoms. 5 Heterozygous germline mutations in the FH gene predispose individuals to HLRCC, characterized by benign leiomyomas of the skin and the uterus and early-onset type II papillary renal cell carcinoma. 6 In affected individuals, loss of heterozygosity (LOH) in the wild-type allele by somatic mutations leads to severe reduction or absence of FH activity, which is followed by an increased intracellular level of fumarate. Accumulated fumarate acts as an oncometabolite to induce tumorigenesis through competitively inhibiting α-ketoglutarate–dependent dioxygenase enzymes and posttranslationally modifying proteins by succination.7,8 Aside from FH deficiency and HLRCC, studies have also shown that FH is involved in hypertension, type 2 diabetes, and diabetic kidney disease.9 –11 For example, deletion of FH in mouse pancreatic β cells leads to progressive glucose intolerance and diabetes after 6–8 weeks. In humans, the fumarate level is higher in islets from donors with type 2 diabetes than from normal donors, and high glucose produces no further increase of fumarate. 11
Rescue of defected fumarase activity by re-expression of a normal FH gene could restore the fumarate level and respiratory function of mitochondria,12,13 which suggests that FH is a potential therapeutic target. The activators of fumarase would enhance the conversion of fumarate to malate, which may rescue the defect of FH. Currently, there are no biochemically characterized activators or inhibitors of FH. Two series of inhibitors have been reported to inhibit the activity of human
14
or Mycobacterium tuberculosis FH.
15
In an attempt to identify nutrient-dependent cytotoxic compounds, Takeuchi et al.
14
screened a collection of 6000 small molecules and discovered a new class of inhibitors of human FH (compounds
To identify small-molecule activators or inhibitors of human FH, we developed a fluorescence-based FH assay by coupling to malate dehydrogenase (MDH)–diaphorase–resazurin and optimized the assay for quantitative high-throughput screening (qHTS) in 1536-well plates. We proceeded to screen a collection of small-molecule libraries to identify new activators or inhibitors of human FH. A total of 57,037 small-molecule compounds were screened, and the majority of hits were triaged by MDH counterscreen assay. One series of the activators for human FH was confirmed and validated in the current study.
Materials and Methods
Reagents and Chemical Libraries
Recombinant human FH (with 6 histidine tag in the N-terminal of protein), dithiothreitol (DTT), and Hank’s Balanced Salt Solution (HBSS) were purchased from Thermo Fisher Scientific (Waltham, MA). L-malate dehydrogenase from pig heart, diaphorase from Clostridium kluyveri, β-NAD (NAD+), resazurin, Brij 35, sodium fumarate dibasic, malic acid, and horseradish peroxidase (HRP) were purchased from Sigma-Aldrich (St. Louis, MO). Fumarate hydratase-IN-2 was purchased from MedChem Express (Monmouth Junction, NJ). Compound
High-Throughput Screen and Counterscreen
For the primary high-throughput screen, 3 µL of FH solution (containing 13.33 nM human FH, 13.33 IU/mL malic dehydrogenase, 0.2 mM NAD+, 0.067 mg/mL diaphorase, and 0.067 mM resazurin) in the assay buffer (50 mM Tris pH 8.0, 5 mM MgCl2, and 0.01% Brij 3) was dispensed into each well in a black solid-bottom 1536-well assay plate (Greiner Bio-One, Monroe, NC) using a BioRAPTR FRD (Flying Reagent Dispenser; Beckman Coulter, Brea, CA). A 1536-well Pintool dispenser outfitted with 20 nL pins (Wako Automation, San Diego, CA) was used to transfer 20 nL of DMSO-solubilized compound (cherrypick plates) to each 1536-well assay plate. Each compound was screened at five concentrations: 15 nM, 151 nM, 1.5 µM, 15.4 µM, and 76.9 µM. Following compound transfer, plates were incubated at room temperature (RT) for 10 min. To initiate the reaction, 1 µL of substrate solution containing fumaric acid (160 µM) was dispensed via BioRAPTR FRD. Plates were immediately transferred to a ViewLux microplate imager (PerkinElmer, Waltham, MA), and any resulting resorufin fluorescence was measured (excitation/emission, 525/598 nm) at 0 and 15 min. The exposure time is 1 s. Fluorescence from each well was normalized using enzyme-free and DMSO-treated control wells on each plate, and changes in fluorescence (∆RFU) were calculated using the difference in fluorescent signal for each well at 15 min versus 0 min.
The MDH counterscreen was adapted from the primary high-throughput screen protocol. Three microliters of MDH solution [containing 13.33 IU/ml malic dehydrogenase, 0.2 mM NAD+, 0.067 mg/mL diaphorase, and 0.067 mM resazurin in assay buffer (50 mM Tris pH 8.0, 5 mM MgCl2, and 0.01% Brij 3)] were dispensed in each well in the 1536-well plate. Plates were incubated at RT for 10 min after compound transfer. To initiate the reaction, 1 µL of substrate solution containing malic acid (160 µM) was dispensed. Fluorescence was measured (ex540, em590 nm) at 0 and 5 min. The fluorescence was normalized using enzyme-free and DMSO-treated control wells on each plate, and ∆RFU was calculated using the difference in fluorescent signal for each well at 5 min versus 0 min.
The Reduction–Oxidation (Redox) Assay
The Amplex Red assay was used to detect the redox activity of small molecules. Briefly, 2.5 µL HBSS solution (1.26 mM CaCl2, 0.49 mM MgCl2, and 1 g/L D-glucose) was dispensed into each well in a black solid-bottom 1536-well assay plate. Twenty nanoliters of compounds were pinned into the plate, and the background fluorescence (RFU0min) was measured in the ViewLux (excitation/emission, 525/598 nm). Two and a half microliters freshly made 2× Amplex Red solution (100 µM Amplex Red, 200 µM DTT, and 2 U/mL HRP in 1× HBSS solution) were added to each well. After 15 min incubation at RT, fluorescence (RFU15min) was measured in the ViewLux with the same setting. Redox activity of each compound was calculated using corrected fluorescence values (ΔRFU = RFU15min − RFU0min). The negative control was DMSO, and the positive controls were walrycin B and chlorinal.
Microscale Thermophoresis (MST)
Recombinant human FH, which contains a polyhistidine tag at the C-terminus, was labeled with Monolith His-Tag Labeling Kit RED-tris-NTA kit (NanoTemper Technologies, Munich, Germany). Briefly, the protein was diluted to 200 nM in a phosphate-buffered saline and Tween (PBS-T) buffer (137 mM NaCl, 2.5 mM KCl, 10 mM Na2HPO4, 2 mM KH2PO4, and pH 7.4 in 0.05% Tween-20). The RED-tris-NTA dye was also diluted in PBS-T to 100 nM. The diluted protein and dye were mixed in a 1:1 volume ratio and incubated for 30 min at RT. After centrifuge 15,000 ×g for 10 min at 4 °C, the supernatant was transferred to a new tube and ready to use.
Compounds were serially diluted in DMSO (16 points, 1:2 dilutions, from 10 mM), and then further diluted 20 times in PBS-T buffer. Ten microliters of labeled protein and 10 µL diluted compounds were mixed and incubated at RT for 2–3 min. The samples were loaded in the standard treated capillaries and measured in a NanoTemper Monolith NT.115 LabelFree instrument (NanoTemper Technologies). The samples were measured at light-emitting diode (LED) power of 40%, and MST power of 40% and 60%. A laser-on time of 30 s and a laser-off time of 5 s were used in this experiment. Curve fitting was performed using GraphPad Prism 5 (La Jolla, CA).
In Vitro ADME Studies
The physicochemical and pharmacokinetics properties of compounds were measured using the following high-throughput in vitro assays: rat microsomal stability assay, parallel artificial membrane permeability assay (PAMPA), and aqueous kinetic solubility assays. The details of these assays are described in a previous study. 17
qHTS Data Analysis
Analysis of compound concentration–response data was performed as previously described. 18 Briefly, raw plate reads for each titration point were first normalized relative to the no-enzyme control and DMSO-only wells as follows: % Activity = [(Vcompound – VDMSO) / (VDMSO – Vno enzyme)] × 100, where Vcompound denotes the compound well values, Vno enzyme denotes the median values of the no-enzyme control wells, and VDMSO denotes the median values of the DMSO-only wells; and then corrected by applying an NCATS Chemical Genomics Center (NCGC) in-house pattern correction algorithm using compound-free control plates (i.e., DMSO-only plates) at the beginning and end of the compound plate stack. 19 Concentration–response titration points for each compound were fitted to a four-parameter Hill equation 20 yielding concentrations of half-maximal activity (AC50) and maximal response (efficacy) values. Compounds were designated as Classes 1–4 according to the type of concentration–response curve observed.18,21 Curve classes are heuristic measures of data confidence, classifying concentration–responses on the basis of efficacy, the number of data points observed higher than background activity, and the quality of fit. Compounds with curve classes 1.1, 1.2, 2.1, 2.2 (activators) and −1.1, −1.2, −2.1, or −2.2 (inhibitors) were considered active. Class 4 compounds were considered inactive. Compounds with other curve classes were deemed inconclusive. Active compounds were cherrypicked for follow-up experiments.
Results
Assay Design, Optimization, and Miniaturization
To identify activators or inhibitors of human FH, we developed a fluorescence-based FH assay, adapted from a previous study.
15
In our assay format, the FH reaction is coupled to two sequential enzymatic reactions: MDH and diaphorase reactions. In the first coupled MDH reaction, L-malate produced by FH is converted into oxaloacetate, removing L-malate from the system and driving the FH forward reaction. The reduced form of nicotinamide adenine dinucleotide (NADH) generated by MDH is used in the second coupled reaction by diaphorase to convert resazurin to resorufin (
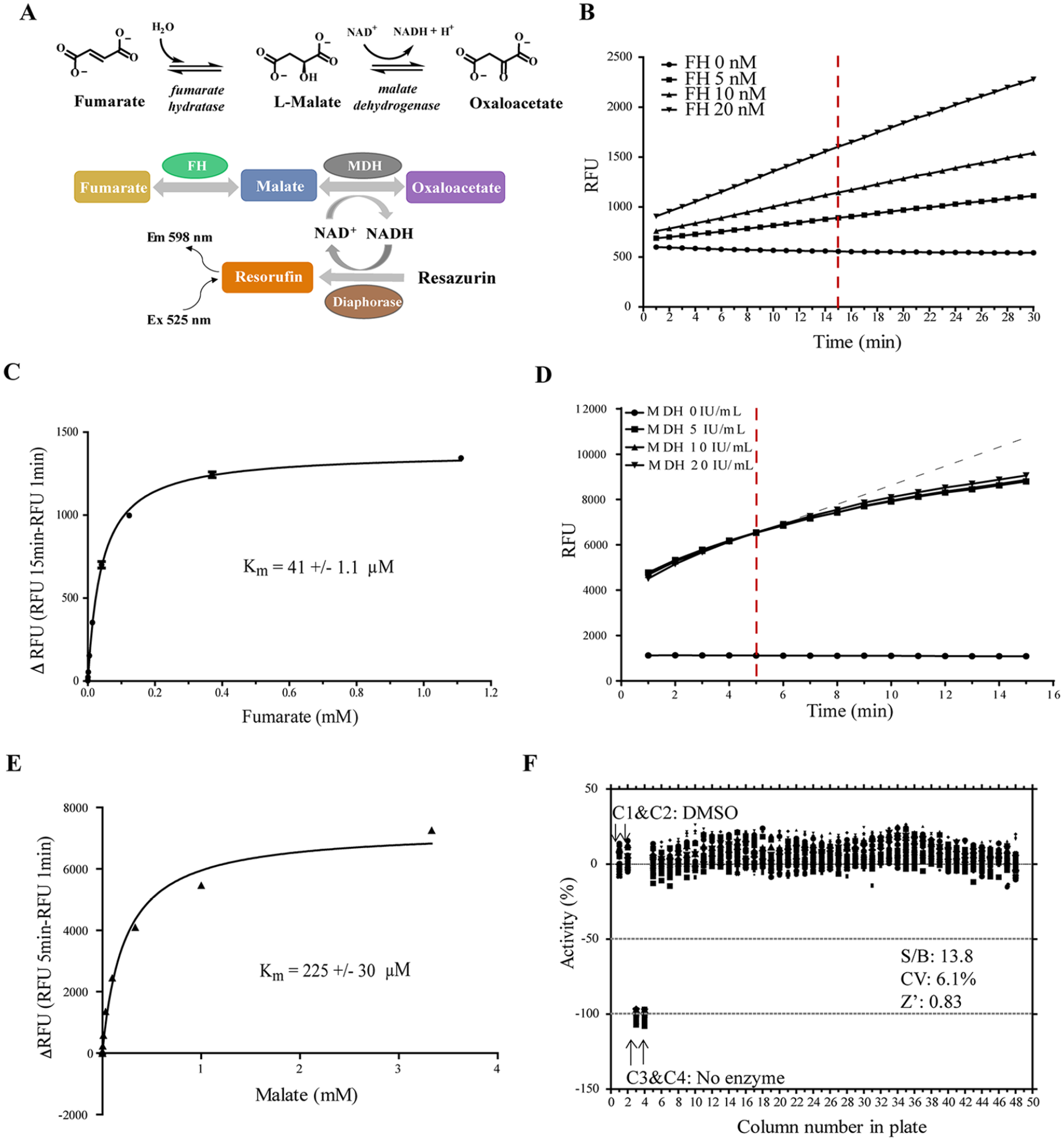
Validation of the fumarate hydratase (FH) primary assay and malate dehydrogenase (MDH) counterassay in quantitative high-throughput screening (qHTS). (
To optimize assay conditions, the initial linear velocity of the enzymatic reaction was determined in the presence of excess fumarate. We found that the enzymatic reaction was linear during a period of 30 min at a concentration of up to 20 nM FH enzyme (
To assess the reproducibility and robustness of the assay, the FH assay was performed in white solid-bottom 1536-well Greiner microplates, and statistics were analyzed. Buffer containing enzyme was dispensed into a microplate for columns 1, 2, and 5–48. The reagent without FH enzyme was dispensed into columns 3 and 4 as an enzyme-free negative control. DMSO (20 nL) was transferred into all wells, then incubated with enzyme for 10 min. The reaction was then initiated by adding fumarate. The kinetics of reaction was measured at t = 0 min and t = 15 min (
Protocols of Fumarate Hydratase (FH) Assay and Malate Dehydrogenase (MDH) Counterassay.
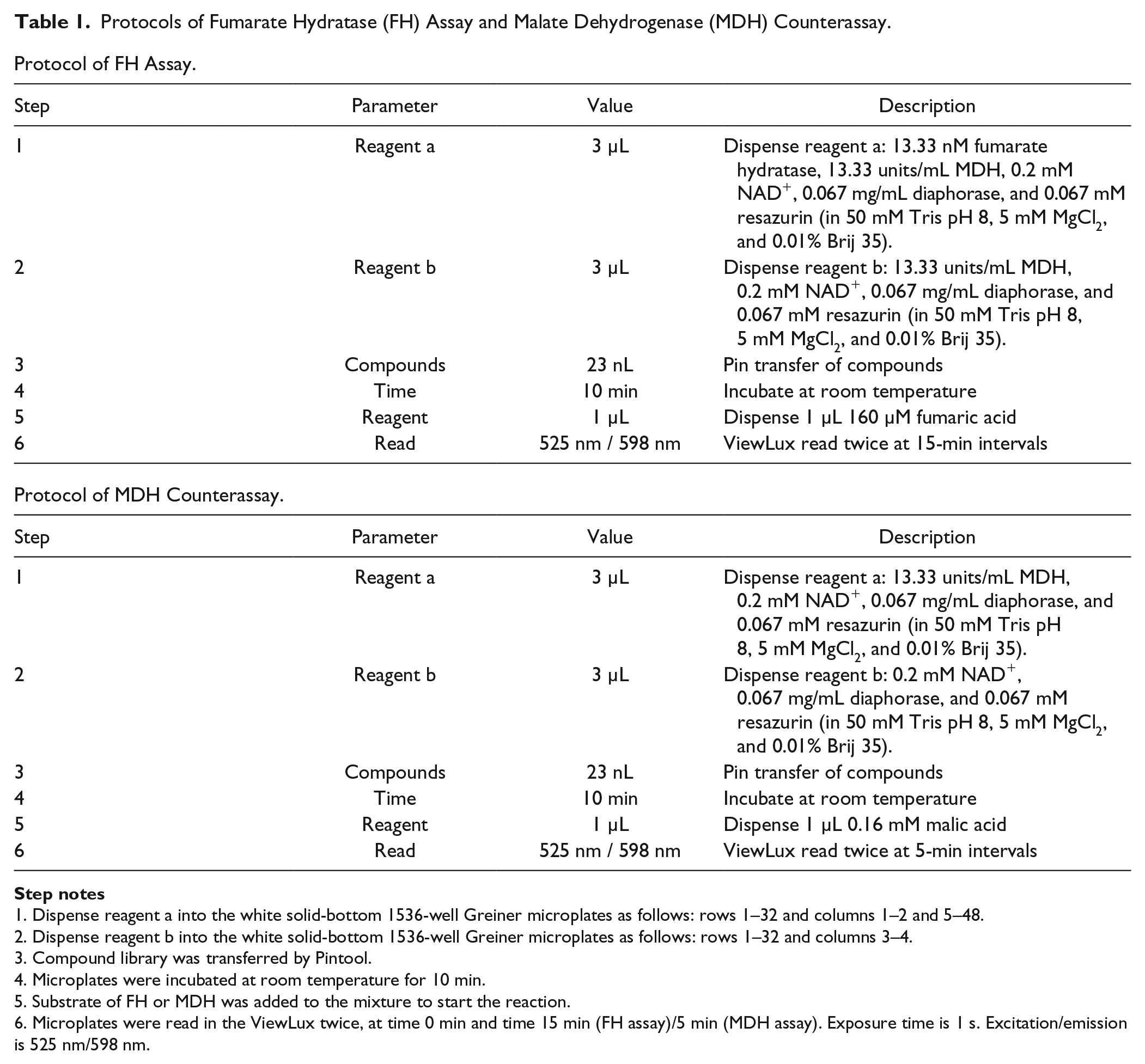
Dispense reagent a into the white solid-bottom 1536-well Greiner microplates as follows: rows 1–32 and columns 1–2 and 5–48.
Dispense reagent b into the white solid-bottom 1536-well Greiner microplates as follows: rows 1–32 and columns 3–4.
Compound library was transferred by Pintool.
Microplates were incubated at room temperature for 10 min.
Substrate of FH or MDH was added to the mixture to start the reaction.
Microplates were read in the ViewLux twice, at time 0 min and time 15 min (FH assay)/5 min (MDH assay). Exposure time is 1 s. Excitation/emission is 525 nm/598 nm.
qHTS Screening and Counterscreen
We screened six in-house libraries with a total number of 57,037 small-molecule compounds in 1536-well format (
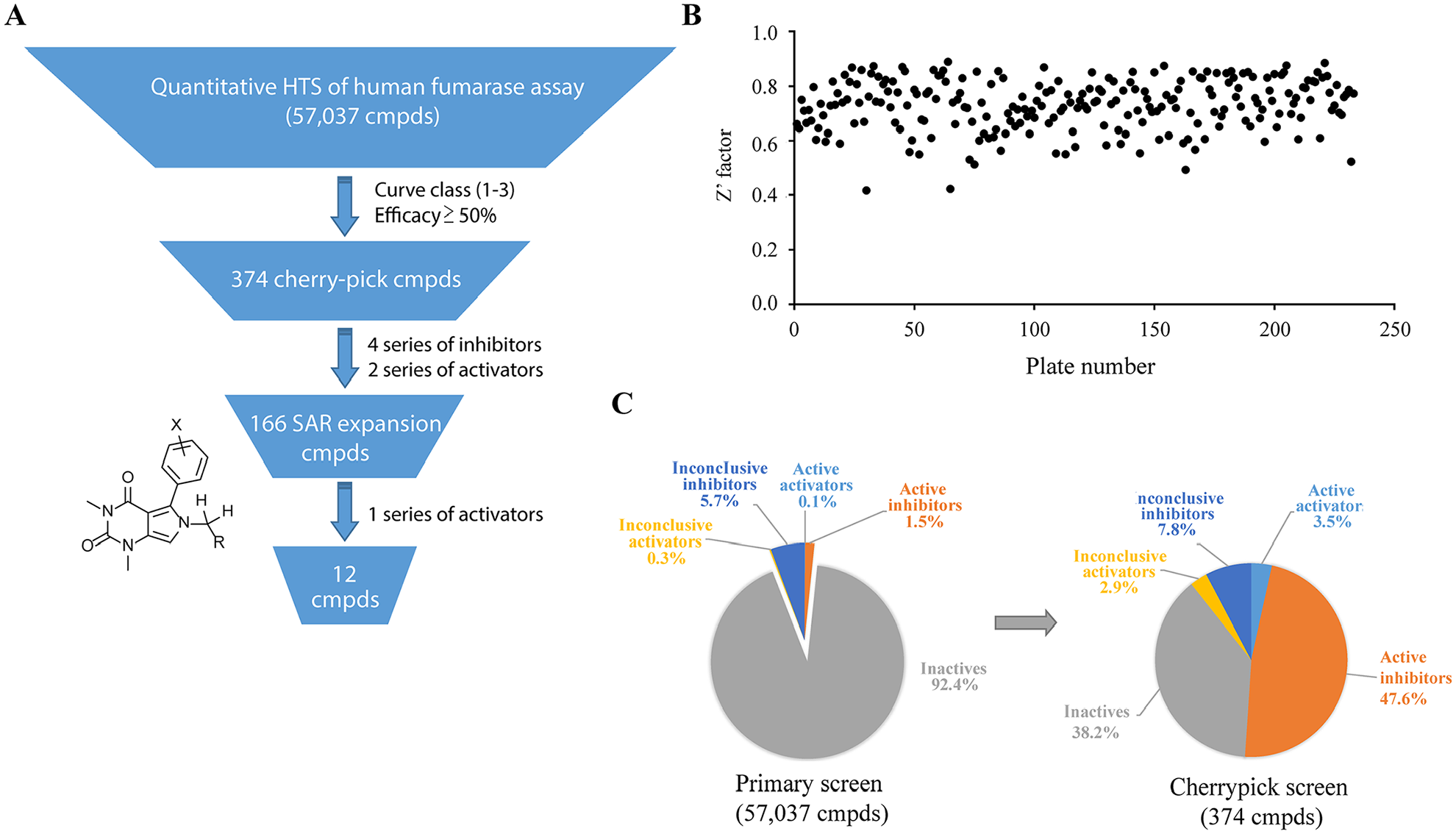
Statistics of quantitative high-throughput screen (qHTS) of a human fumarase assay. (
Only four series of inhibitors and two series of activators survived the counterscreening process. We then plated a total of 166 analogs structurally related to the hits from our in-house library for retesting. One series of activators was confirmed in our FH assay (
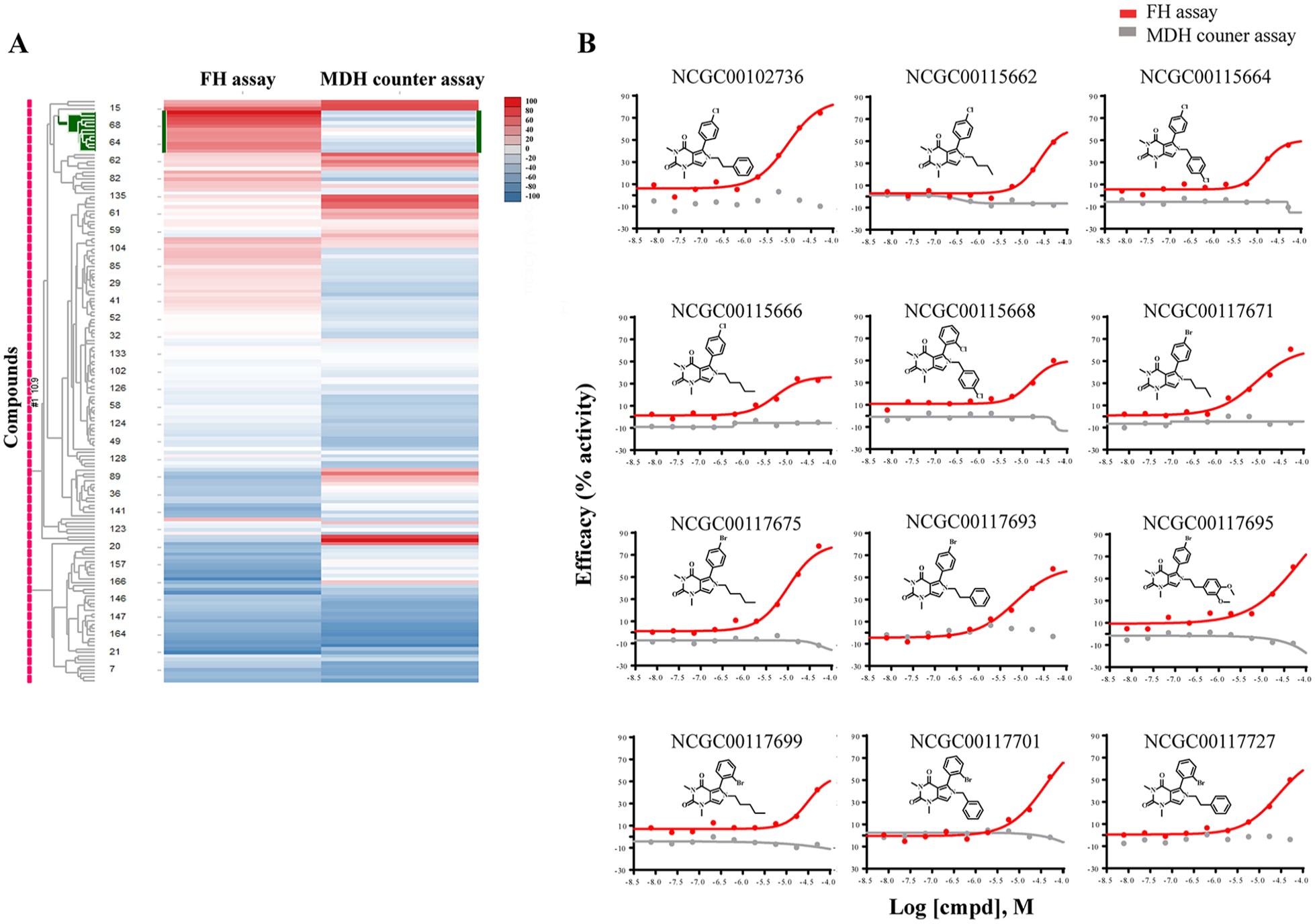
Heatmap of 166 compounds from six structural series. (
Identification and Validation of FH Activators
The activator chemotype is composed of a series of phenyl-pyrrolo-pyrimidine-diones, which are active against FH with AC50 ranging from 5.6 µM to 31.6 µM and efficacy ranging from 35% to 79% (
One Series of Activators of Fumarate Hydratase.
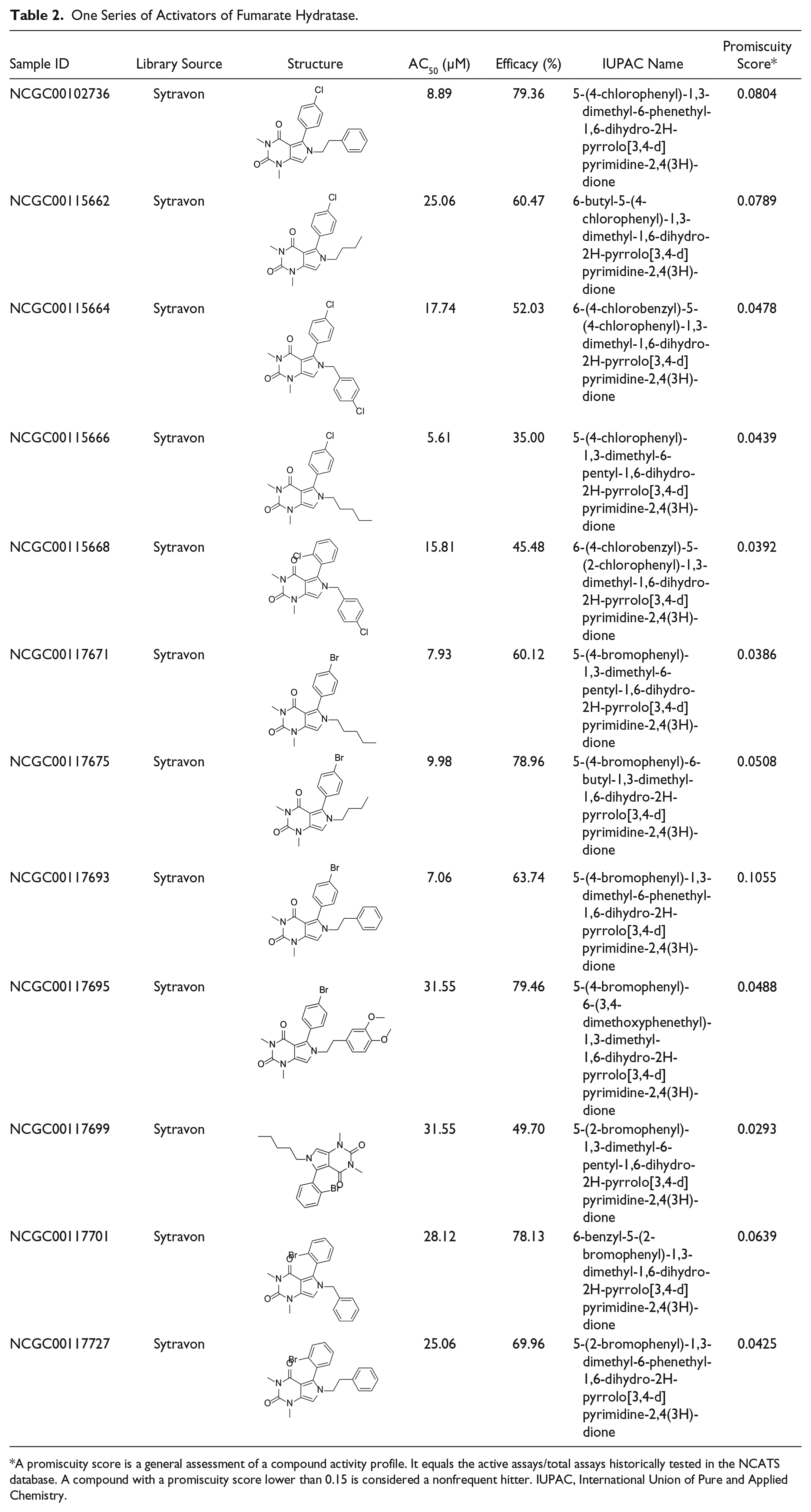
A promiscuity score is a general assessment of a compound activity profile. It equals the active assays/total assays historically tested in the NCATS database. A compound with a promiscuity score lower than 0.15 is considered a nonfrequent hitter. IUPAC, International Union of Pure and Applied Chemistry.
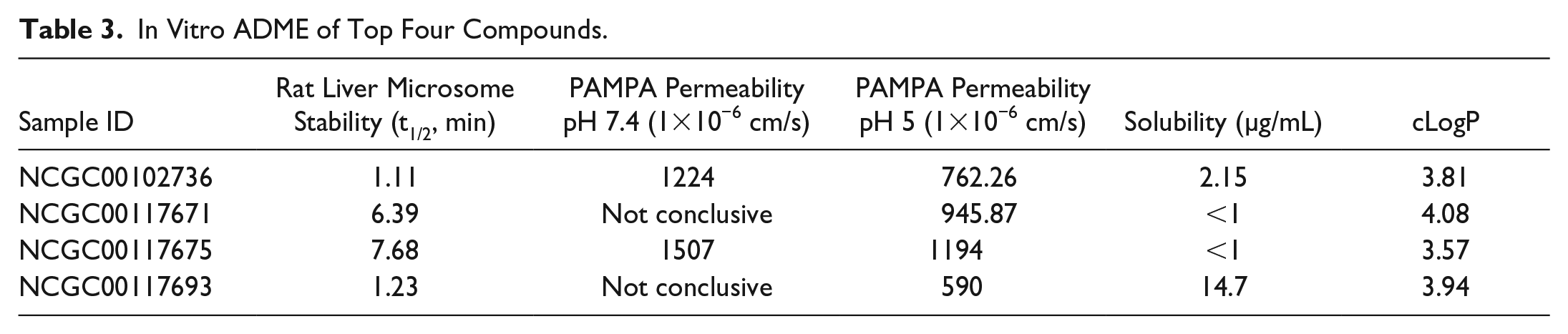
In Vitro ADME of Top Four Compounds.
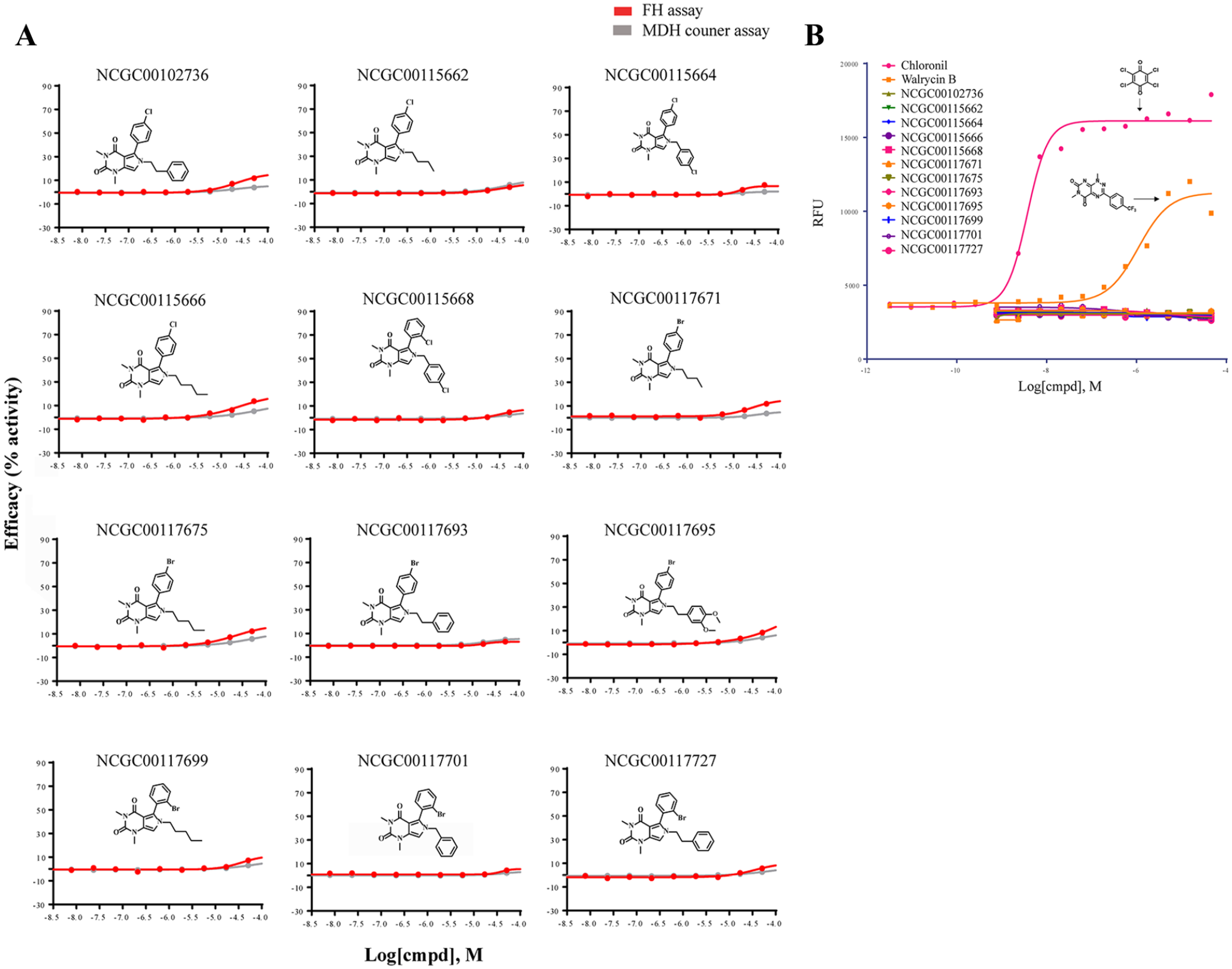
Evaluation of the series of activators on fumarate hydratase (FH) activity without fumarate and reduction–oxidation (redox) activity in the Amplex assay. (
Artefactual liabilities of screening hits can be responsible for the apparent activation, such as the ability to undergo redox cycling. The activator chemotype we identified contains a pyrimidine-2,4(3H)-dione motif. This is not expected to participate in redox chemistry, but it is prudent to de-risk such artefactual activity even if it is not expected. This is especially true when coupled assays such as the one described here use oxidoreductive conversions between NAD+ and NADPH to facilitate assay readout. To examine whether compounds have redox activity, the Amplex Red assay was performed. In the assay, two compounds with weak (walrycin B) and strong (chloranil) redox activity were used as positive controls. None of the hit compounds displayed detectible redox activity (
We performed MST as a biophysical assay to directly demonstrate binding of activators to FH.
24
Two compounds with high potency, NCGC00102736 (AC50 = 8.89 µM) and NCGC00117675 (AC50 = 9.98 µM), were evaluated. The dissociation constant (Kd) of NCGC00102736 was found to be 18.3 µM, and of NCGC00117675 was 23.3 µM (
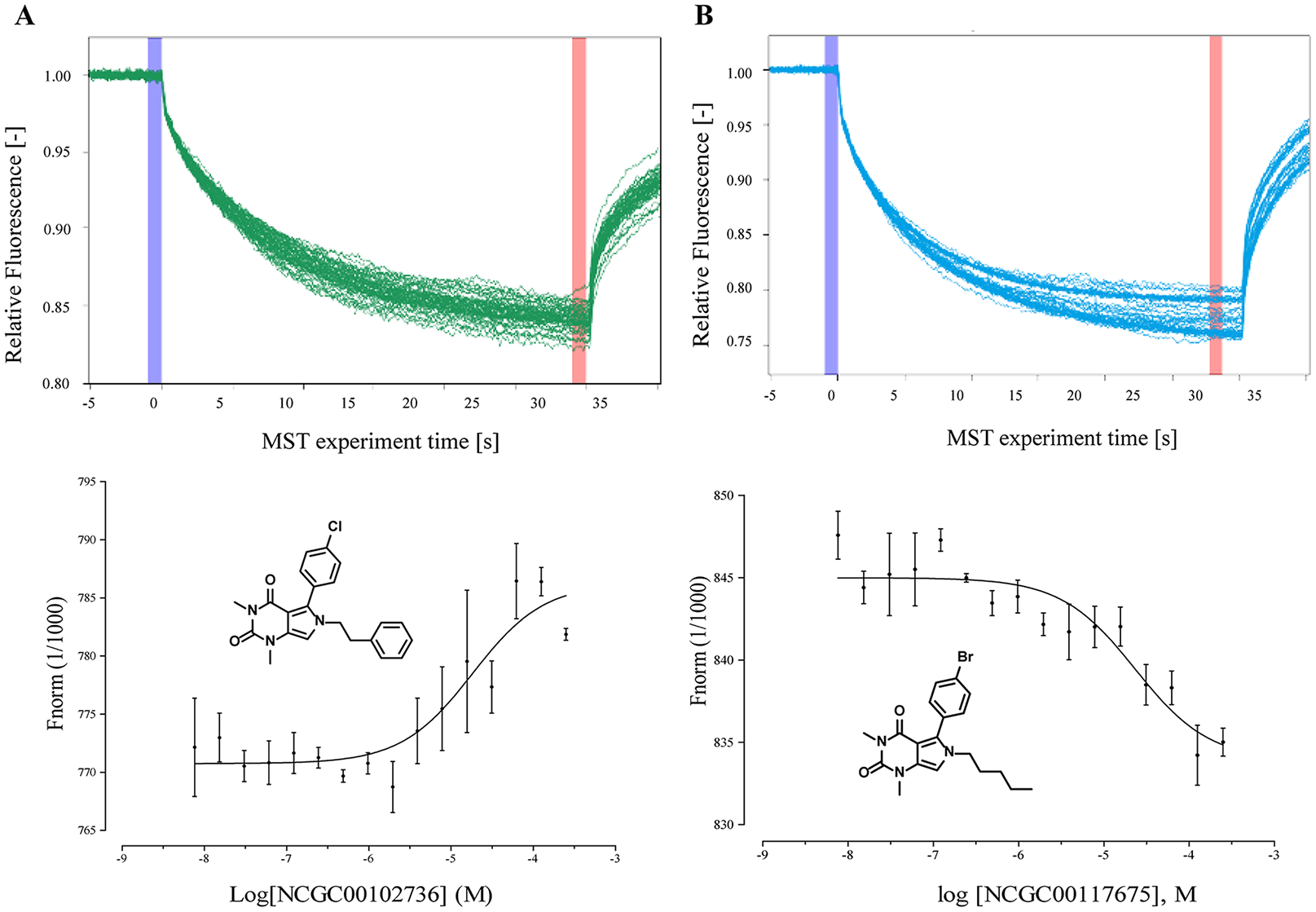
Target engagement of top two compounds with fumarate hydratase (FH). The binding of two compounds with the fumarase enzyme was evaluated by a microscale themophoresis (MST) assay. (Top) MST traces of titrations of NCGC00102736 (
In Vitro ADME Properties of FH Activators
To evaluate the physicochemical and pharmacokinetic properties of the FH activator series, we selected the four most potent compounds and tested them in a series of in vitro ADME assays. Using automated high-throughput in vitro assays, we examined the rat microsomal metabolic stability, permeability, and solubility in those compounds (
Evaluation of Prior Art Compounds and Artifacts
Because two inhibitors of FH were reported previously, we evaluated the activity of those inhibitors in our human FH assay. We resynthesized compound
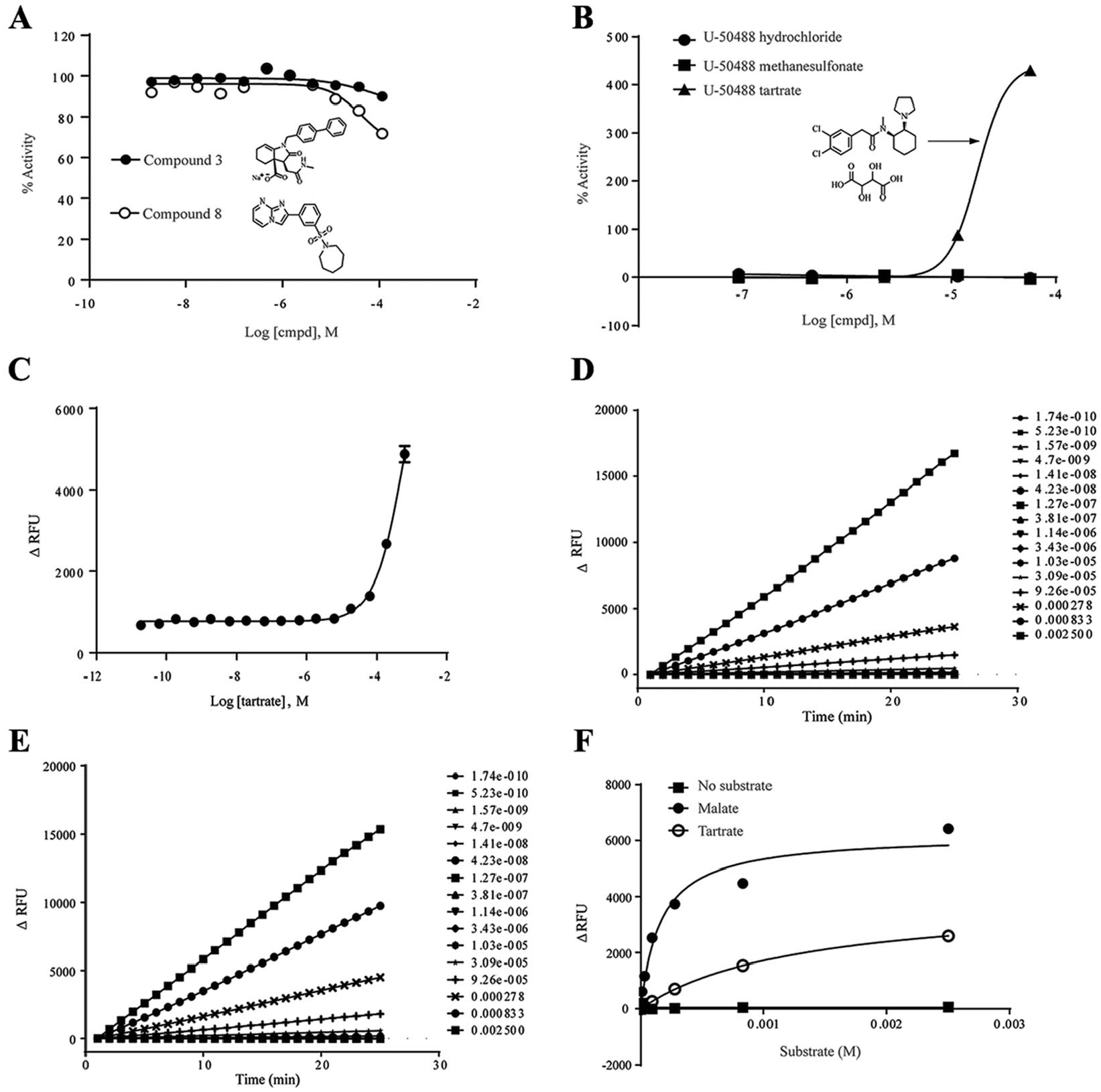
Evaluation of prior art compounds and artifacts. (
During our HTS campaign, we identified an interesting false-positive compound, U-50488, which is a highly selective κ-opioid agonist. There are three salt forms of U-50488 in our collection. We found that the tartrate salt of U-50488 showed very high activator activity. However, the hydrochloride and methanesulfonate salts of U-50488, which are also in our library, showed no activator activity (
Discussion
In our efforts to identify small-molecule probes for FH, we developed a qHTS FH assay and screened a collection of small-molecule libraries. After several rounds of screening and counterscreening, only one series of activators met all activity criteria in our study. Those compounds were not substrates of FH, and they demonstrated direct binding to FH protein. This activator series enhanced the FH activity in the presence of its substrate. Our results suggest that these compounds are allosteric modulators of FH; however, further work is needed to optimize activity and confirm their mechanism of activation.
The series identified as FH activators is a group of phenyl-pyrrolo-pyrimidine-diones (
Pan-assay interference compounds (PAINS) are the compounds shown as false positives in high-throughput screens. 28 To exclude fluorescent compounds, we used change of fluorescence (ΔRFU) as the readout. If compounds are fluorescent, the signal should not change throughout the time (15 min in our assay), and the value of ΔRFU should be close to zero. To filter out the compounds interacting with coupled enzymes in the assay (MDH and diaphorase), a MDH assay was used as a counterassay to triage these false-positive compounds. The MDH counterassay may, however, triage compounds that inhibit or activate both FH and MDH. As a common substrate for both FH and MDH, malate should be able to bind to the active sites of both enzymes, which suggests a structural similarity in active sites between two enzymes. The inhibitors that competitively bind to the same active sites will be filtered out in the MDH counterassay. As such, nonselective competitive inhibitors would be prone to exclusion by assay format. No inhibitors were identified in the current study after screening a collection of 57,037 compounds. In a similar assay format, Kasbekar 15 also didn’t identify any competitive inhibitors for tbFH after screening a collection of 479,984 compounds. Instead, two inhibitors that bind to an allosteric regulatory site were found.
In summary, we developed a quantitative high-throughput screening FH assay and screened a collection of 57,037 small molecules. A series of phenyl-pyrrolo-pyrimidine-diones were identified as activators of human FH, which can serve as a starting point for further optimization and development as probes for the enzyme.
Supplemental Material
DS_873559_Supplementalinformation – Supplemental material for Identification of Activators of Human Fumarate Hydratase by Quantitative High-Throughput Screening
Supplemental material, DS_873559_Supplementalinformation for Identification of Activators of Human Fumarate Hydratase by Quantitative High-Throughput Screening by Hu Zhu, Olivia W. Lee, Pranav Shah, Ajit Jadhav, Xin Xu, Samarjit Patnaik, Min Shen and Matthew D. Hall in SLAS Discovery
Footnotes
Acknowledgements
We thank Monica Kasbekar and Craig J. Thomas for kindly providing the detailed protocol of their fumarase assay; Paul Shinn, Danielle van Leer, and Zina Itkin for their assistance with compound management; Sam Michael, Carleen Klumpp-Thomas, and Jameson Travers for their assistance with automation; Michael Ronzetti and Bolormaa Baljinnyam for technical support of the microscale thermophoresis assay; and Mark Henderson for technical support of the Amplex Red assay.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the intramural research program of the National Center for Advancing Translational Sciences (NCATS).
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
