Abstract
Iron is an essential requirement for the survival and virulence for bacteria. The bacterial ferrous iron transporter protein B (FeoB) functions as a major iron transporter in prokaryotes and has an N-terminal domain (NFeoB) with homology to eukaryotic G-proteins. Its GTPase activity is required for ferrous iron uptake, making it a potential target for antivirulence therapies. Here, two assay strategies relying on different spectroscopic readouts are described to monitor NFeoB GTPase activity. The first one is the colorimetric-based platform that utilizes a malachite green reagent to monitor phosphate production from GTP hydrolysis. The absorbance change directly relates to the GTPase activity of NFeoB. The assay was further improved by the addition of Tween-20 and miniaturized in a 384-well plate format with a 10 µL assay volume. The second format is a luminescence-based platform, measuring the GTP depletion by using a modified GTPase-Glo assay from Promega. In this platform, the luminescence signal correlates to the amount of GTP remaining, allowing for the direct calculation of GTP hydrolysis by NFeoB. The colorimetric platform was tested in a high-throughput manner against a custom-assembled library of a~2000 small molecules and was found to be simple, cost-effective, and robust. Additionally, the luminescence-based platform demonstrated its capability as an orthogonal assay to monitor GTPase activity, providing a valid and convenient method to filter false hits. These two assay platforms are proven to offset the limitations of each platform while enhancing overall quality and success rates.
Introduction
Iron is an essential requirement for the survival and virulence for nearly all bacteria. 1 To shuttle iron, bacteria commonly use iron transport proteins that allow them to obtain free or chelated iron from their environments. Among these, the ferrous iron transporter protein B (FeoB), which is a transmembrane protein, functions as a major iron transporter in prokaryotes, particularly in anoxic environments. Notably, FeoB contains a C-terminal transmembrane anchor and an N-terminal domain that closely mimic eukaryotic G-proteins.2,3 The cytosolic G-protein domain is required for ferrous iron uptake in vivo, but it is not clear whether the GTPase activity is providing the energy for iron transport 4 or is playing a role in regulating ferrous iron transport in response to cellular iron status. 5 Because FeoB plays a role in the virulence of some pathogenic bacteria and is widely conserved in most prokaryotes, the N-terminal cytosolic domain (NFeoB) presents a good target for therapeutic intervention. Finding new small molecules that selectively target the GTPase activity of NFeoB may disrupt iron transport sufficiently to block growth and virulence within the host. However, there are no reported high-throughput screening (HTS) platforms for monitoring NFeoB activity, and there are no reported specific inhibitors of NFeoB activity. Thus, we sought to develop a new platform that is capable of identifying novel inhibitors of the GTPase activity of NFeoB in a high-throughput manner.
As illustrated in Figure 1 , NFeoB GTPase activity can be determined either by the production of phosphate or GDP or by the depletion of GTP. Commonly, the detection of inorganic phosphate is performed with a traditional malachite green (MG)-based colorimetric assay 6 (denoted as “colorimetric platform”). While the colorimetric platform has been widely employed in compound screening, sensitivity and miniaturization have been the major bottleneck to advancing this technology into HTS campaigns. The other significant drawback of utilizing the colorimetric platform is the assay interference due to the inherent spectroscopic characteristics of small compound libraries, resulting in false hits. Here, we describe two assay platforms to monitor the GTPase activity of NFeoB, which can be employed in a complementary manner to allow for a cost-effective primary screen, with a sensitive and robust counterscreen. First, optimization of a simple mix-and-read colorimetric platform in a 384-well format by tweaking buffer composition and miniaturization for robust HTS application is presented. This optimized platform was validated in a pilot screen of approximately 2000 small molecules.
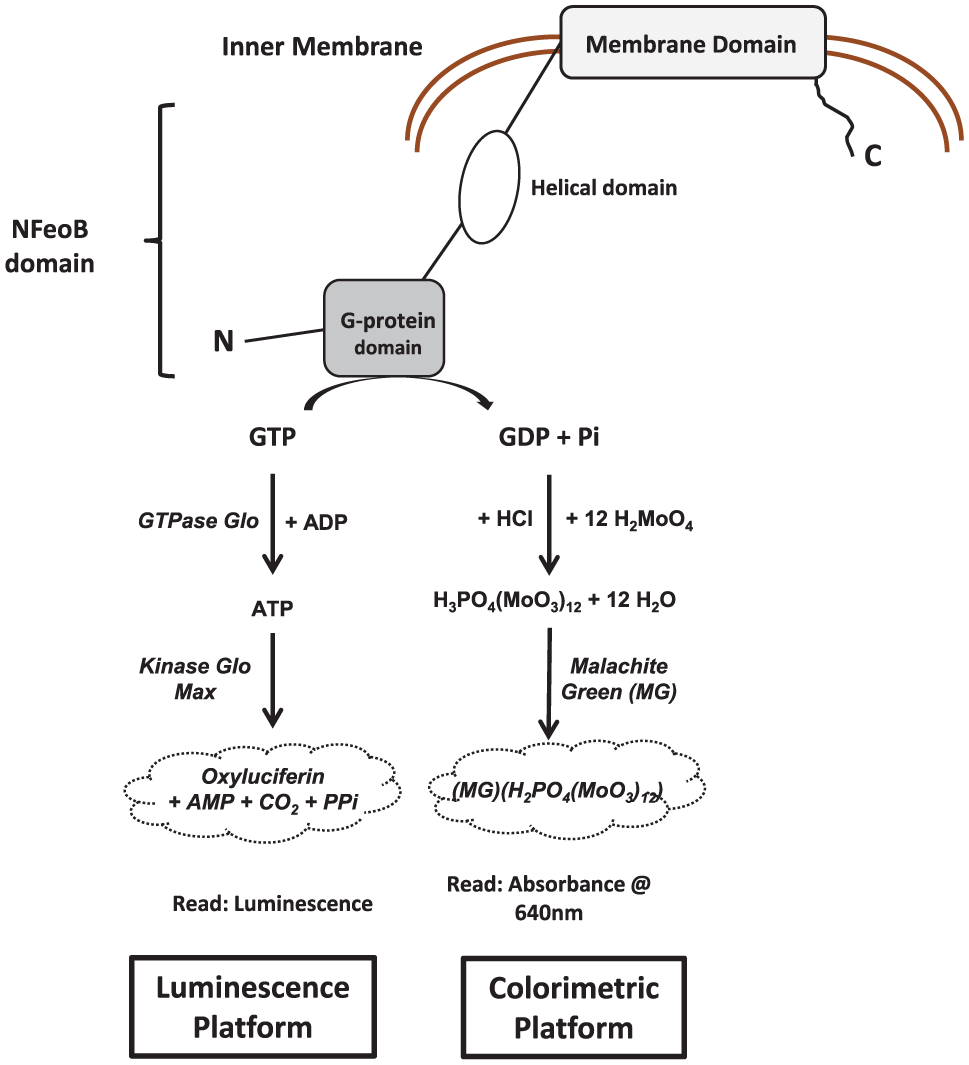
Assay principle of NFeoB GTPase activity. The amount of GTP hydrolyzed after the GTPase activity is measured by either (1) production of free phosphate via complexation with malachite green reagent colorimetrically or (2) depletion of GTP via coupled enzymatic reaction that generates luminescence. The malachite green signal is proportional to NFeoB activity, while the luminescence signal is inversely proportional to NFeoB activity.
Next, we present a new luminescence-based platform that monitors the depletion of GTP, as an alternative to the colorimetric platform to resolve compound interference. The assay platform utilizes commercial GTPase-Glo 7 (denoted as “luminescence platform” in Fig. 1 ), originally designed to monitor GTP up to 5 µM. The luminescence readout is known to be much more sensitive than a colorimetric detection platform, and a major benefit is its instant adaptation to biochemical assays. However, the protocol of GTPase-Glo required revision to meet the dynamic range of the GTPase activity of NFeoB and was subsequently validated for compound screening. This new luminescence platform demonstrated its potential to monitor GTPase activity of NFeoB with extended dynamic range. It also proved its capability as an orthogonal assay to discriminate false hits from the colorimetric platform. In summary, we present a convenient approach to the high-throughput investigation of compounds that modulate NFeoB GTPase activity, which will be useful in developing a new approach to combating bacterial pathogenesis.
Materials and Methods
Reagents and Materials
All reagents and chemicals were purchased from Sigma-Aldrich (St. Louis, MO), unless otherwise stated. Ultrapure GTP (cat. R0461), malachite green oxalate (cat. 41349-0250), and ammonium molybdate (cat. 448540050) were purchased from Thermo Fisher Scientific (Waltham, MA).
NFeoB was expressed and prepared as described in Marlovits et al.
4
with modification. In detail, pET21a-based expression vectors containing the cytosolic N-terminus of FeoB from
Colorimetric GTPase Activity Assay
The GTPase activity assay was initially established using NFeoB purified from
The room temperature stability of the assay was evaluated by varying enzymatic reaction time. In detail, the assay mixture after adding GTP was incubated for 0, 20, 60, 90, and 120 min before adding MG solution. Enzyme kinetics were evaluated by varying GTP concentrations and measuring the amount of phosphate generated after 120 min of incubation. Unless otherwise stated, further assay validation was performed in an assay buffer using 1 µM NFeoB and 390 µM GTP for 120 min.
Validation of Colorimetric GTPase Activity Assay
A full plate assay validation was conducted by plating columns 2–23 of 384-well plates with positive controls that contained NFeoB and GTP in an assay buffer, whereas columns 1 and 24 were plated with negative control containing only substrate in an assay buffer. First, 5 µL of protein solution containing 2 µM NFeoB in assay buffer (columns 2–23) or assay buffer alone (columns 1 and 24) was dispensed using a Janus Automated Workstation (PerkinElmer, Waltham, MA). Second, 100 nL of 100% DMSO was dispensed using an ECHO 550 Acoustic Dispenser (Labcyte, Sunnyvale, CA). The reagents were allowed to mix for 1 h at room temperature. Third, 5 µL of 780 µM GTP diluted in assay buffer was dispensed with the MicroFlo Select Bulk Dispenser (BioTek). The assay was performed and read as described above while the final DMSO concentration was 1%. Interplate variation was measured as described above, but in two separate plates on two separate experiments, using freshly prepared assay mixtures.
Luminescence-Based GTPase Activity Assay
A luminescence-based platform was developed by modifying the GTPase-Glo assay (Promega, Madison, WI, cat. V7681) with the Kinase Glo Max assay (Promega, cat. V6071). The assays were carried out in white, 384-well, flat-bottom, low-volume microplates (Corning, cat. 3825) with 10 µL assay volume. The dynamic range of GTPase-Glo was investigated by varying the concentration of GTP up to 1 mM, diluted in an assay buffer according to the manufacturer’s protocol. For the GTPase activity of NFeoB, the enzymatic reaction was conducted as described above in a 10 µL assay volume with 1 µM NFeoB and 390 µM GTP. After 120 min of incubation at room temperature, 10 µL of modified GTP Glo I reagent containing 6 mM ADP was added to stop the reaction. This reaction was allowed to incubate for 60 min at room temperature, before adding 20 µL of Kinase Glo Max detection solution. The DMSO tolerance was examined by varying the fractional percent of DMSO up to 10%. The percent conversion of GTP was calculated based on a control assay that included 390 µM ATP in an assay buffer.
Primary Compound Screening
Compound screening was performed using the colorimetric assay as described above for assay validation, but with compounds instead of DMSO. More than 2000 bioactive compounds, which were custom-assembled, were chosen and compounds were screened at a final concentration of either 10 or 100 µM, with the DMSO concentration maintained at 0.1% or 1%. This unique collection is composed of small molecules with known activities against more than 100 kinases and other important therapeutic targets, representing a useful panel of compounds for developing leads against novel NTP-dependent proteins based on their off-target activities.
Hit Validation
A total of 11 compounds selected from the primary screen were screened again at 100 µM using both colorimetric and luminescence-based GTPase activity assays. Compound interference with MG was also conducted by assaying them in the presence of 60 µM potassium phosphate instead of NFeoB and GTP in the colorimetric assay. Two compounds that exhibited comparable inhibition activity in both the colorimetric and luminescence assays were chosen for dose–response validation using colorimetric assay at final concentrations of 0, 25, 50, and 100 µM. Additionally, the substrate-dependent inhibition activity of those two hits was examined at three concentrations of GTP (50, 390, and 700 µM) using the colorimetric assay.
Statistical Analysis
The initial velocities and turnover rate were calculated according to eqs 1 and 2, respectively, where
A compound’s potency was calculated according to eq 4, Standard Curves, Four Parameter Logistic Curve from SigmaPlot 13.0 (Systat Software, San Jose, CA), where


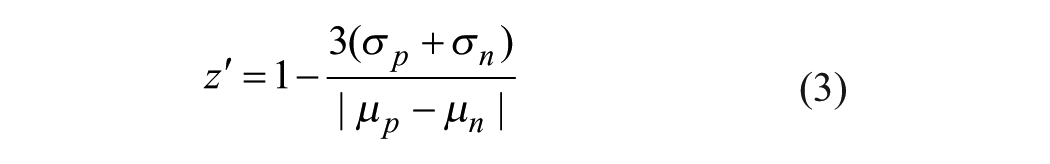
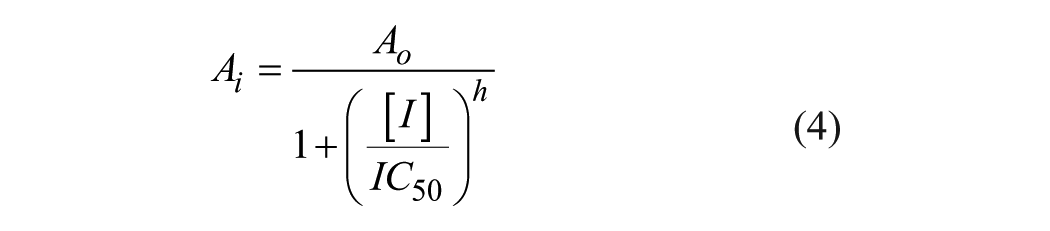
Results and Discussion
Colorimetric GTPase Activity Assay Optimization
Initially, the enzymatic assay was performed in a 40 µL reaction in a 96-well plate, using an assay buffer without Tween-20. The reaction was quenched using 150 µL of MG, prepared in the absence of Tween-20. A phosphate standard curve was used to back-calculate GTP conversion as a guide of assay performance. Since detergents are useful in reducing nonspecific interactions and improving liquid handling performance, the effect of detergent on the colorimetric platform was first investigated by monitoring the absorbance spectra of MG with phosphate standard solution in the presence and absence of 0.01% Tween-20. As shown in Figure 2A , the peak at 640 nm corresponding to a complex of MG with phosphate increased ~40% in the presence of 0.01% Tween-20, while the peak at 440 nm corresponding to MG itself was not affected. This result is in accordance with the previous reports that the use of polyvinyl alcohol 10 or detergents 11 improved solubility of the MG complex and ultimately signal stability. A dose–response curve with phosphate standard in Figure 2B also showed that the addition of Tween-20 did not alter the linearity up to 250 µM phosphate in addition to the signal enhancement. Since compounds are often dissolved in DMSO, we examined whether the presence of 1% DMSO had any interference on the colorimetric platform. As shown in Figure 2C , the addition of 1% DMSO did not alter the linearity even up to 500 µM phosphate. These results support the claim that assay additives do not alter the colorimetric assay protocol. Instead, the addition of Tween-20 enhanced the signal strength, improving the dynamic range.
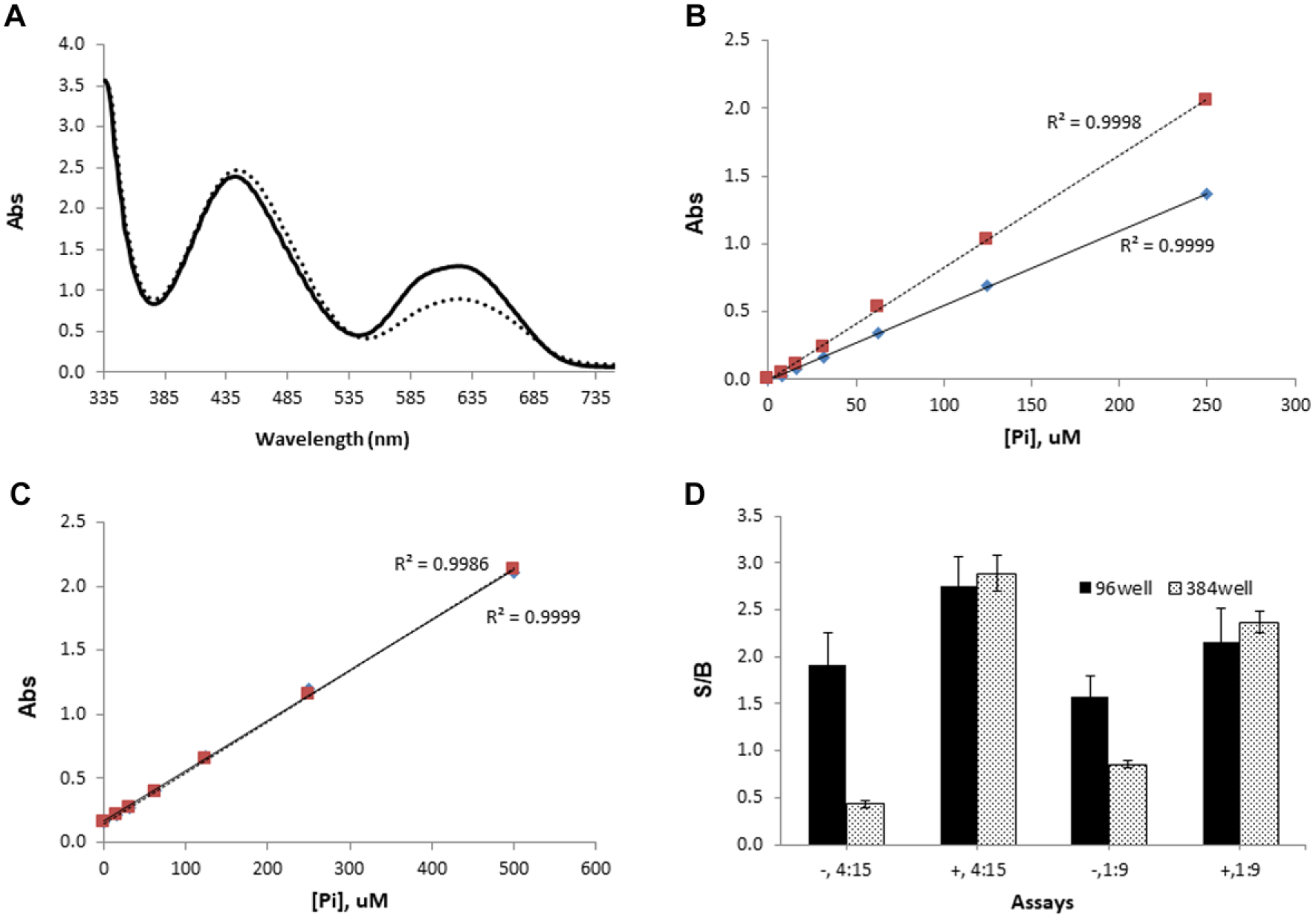
Effect of assay additives on malachite green-based colorimetric platform. (
Due to the relatively narrow dynamic range of the colorimetric platform, we explored whether the ratio of assay mixture to MG could expand the dynamic range further. Using a phosphate standard solution, it was observed that the ratio of phosphate standard to malachite green (Pi/MG) altered the linear detection range but not the dynamic range. Conclusively, the linearity increased to 1 mM phosphate when a ratio of 1:9 was used, while the absorbance signal was linear between 0 and 250 µM phosphate at a 1:4 ratio (data not shown). Furthermore, we tested the effect of both Pi/MG and the presence of Tween-20 when the assay was miniaturized from a 96-well format to a 384-well format with assay volumes from 40 µL to 10 µL. As shown in Figure 2D , the signal-to-background (S/B) ratio was higher in the presence of Tween-20 regardless of Pi/MG. Also, the S/B ratio was consistent regardless of miniaturization if Tween-20 was present. However, the 384-well format with lower assay volume exhibited at least a two times decreased S/B ratio in the absence of Tween-20, indicating that Tween-20 is critical for assay miniaturization. This presents a useful modification, since successful miniaturization of this assay enables a more cost-effective approach to HTS. Because NFeoB’s GTPase activity resulted in approximately 60 µM phosphate in 2 h, which corresponds to ~15% conversion of GTP to GDP, a 1:4 ratio was chosen for further experiments in order to linearly detect phosphate generation. This is consistent with a similar HTS screening assay utilizing malachite green detection in a 384-well format that utilizes higher enzyme concentrations. 12 However, we demonstrate here that the detection range can be easily extended by simply modifying the ratio of the reaction to malachite green from 1:4 to 1:9.
The room temperature stability of the GTPase activity was evaluated by varying the incubation time before the addition of MG in the presence of Tween-20. The phosphate production from NFeoB was proportional to assay time ( Fig. 3A ) in the presence of Tween-20 at room temperature, resulting in an S/B ratio of approximately 2.0 after 2 h of enzymatic reaction. This result demonstrated that the GTPase activity of NFeoB is tolerant at room temperature at least up to 2 h. Additionally, we confirmed that the presence of Tween-20 or DMSO in assay buffer did not alter the GTPase activity (data not shown). Based on these results, 0.01% Tween-20 in both the MG detection reagent and assay mixture was chosen with an enzymatic reaction time of 2 h for further assay development.
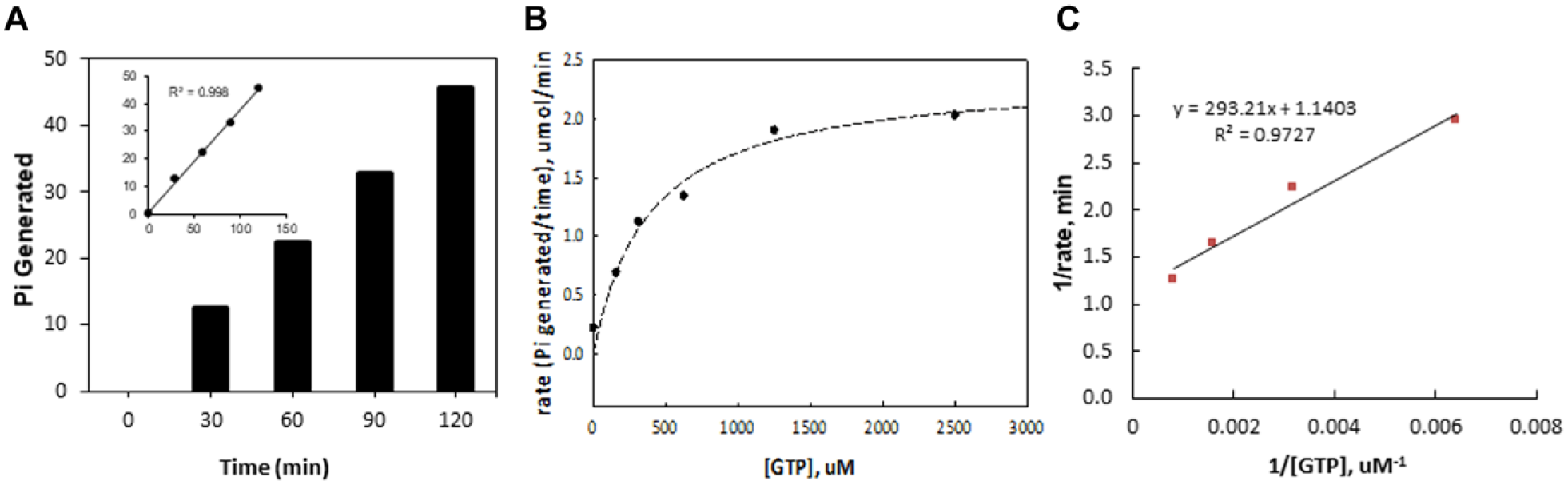
Colorimetric GTPase activity assay performance. (
Following the serial optimization and miniaturization, assay kinetics was next examined. As shown in
Figure 3B,C
, the Michaelis–Menten constant,
To ensure that the assay is robust enough for HTS, a full plate validation using a 10 µL assay volume in a 384-well plate was conducted. Two validation experiments were conducted on different days with freshly prepared assay mixtures for each experiment. The averaged values for the S/B ratio, signal-to-noise (S/N) ratio, and z′ factor were 1.5, 53.3, and 0.69, respectively, proving the robustness of the assay. These results prove that the assay gives excellent separation between positive and negative controls across the plate (intraplate) and between the plates (interplate) and is suitable for high-throughput compound screening.
Luminescence-Based GTPase Activity Assay
Because many compounds may interfere with colorimetric absorbance detection, or may interact with the MG complex, it is important to have an orthogonal detection strategy to identify false positives.
While an HTS platform utilizing a fluorescent phosphate sensor has been reported, 13 the potential for spectroscopic interference was considered too high, as many small-molecule compounds have intrinsic fluorescence. Additionally, an orthogonal assay that measures the substrate remaining (GTP) or a different reaction product (GDP) allows for better confirmation of the screening results. Sensitive and robust high-throughput bead-based measurements of GTP 14 and fluorescence polarization methods that measure GDP15,16 have been reported; however, these require specialized instrumentation that are not commonly found in research laboratories, and these specialized reagents can be prohibitively expensive for smaller labs or new investigators with limited funding. Thus, a luminescence-based platform was sought to visualize the remaining GTP, as this would provide a sensitive and complementary detection platform to the colorimetric platform used in primary screening.
As illustrated in
Figure 1
, the luminescence signal is inversely proportional to the NFeoB’s GTPase activity. Promega’s GTPase-Glo assay was first considered, but it was found that this technology could not be easily adapted to measure NFeoB’s GTPase activity because the detection limit of the Glo kit (5 µM GTP)
7
was much lower than the NFeoB’s calculated
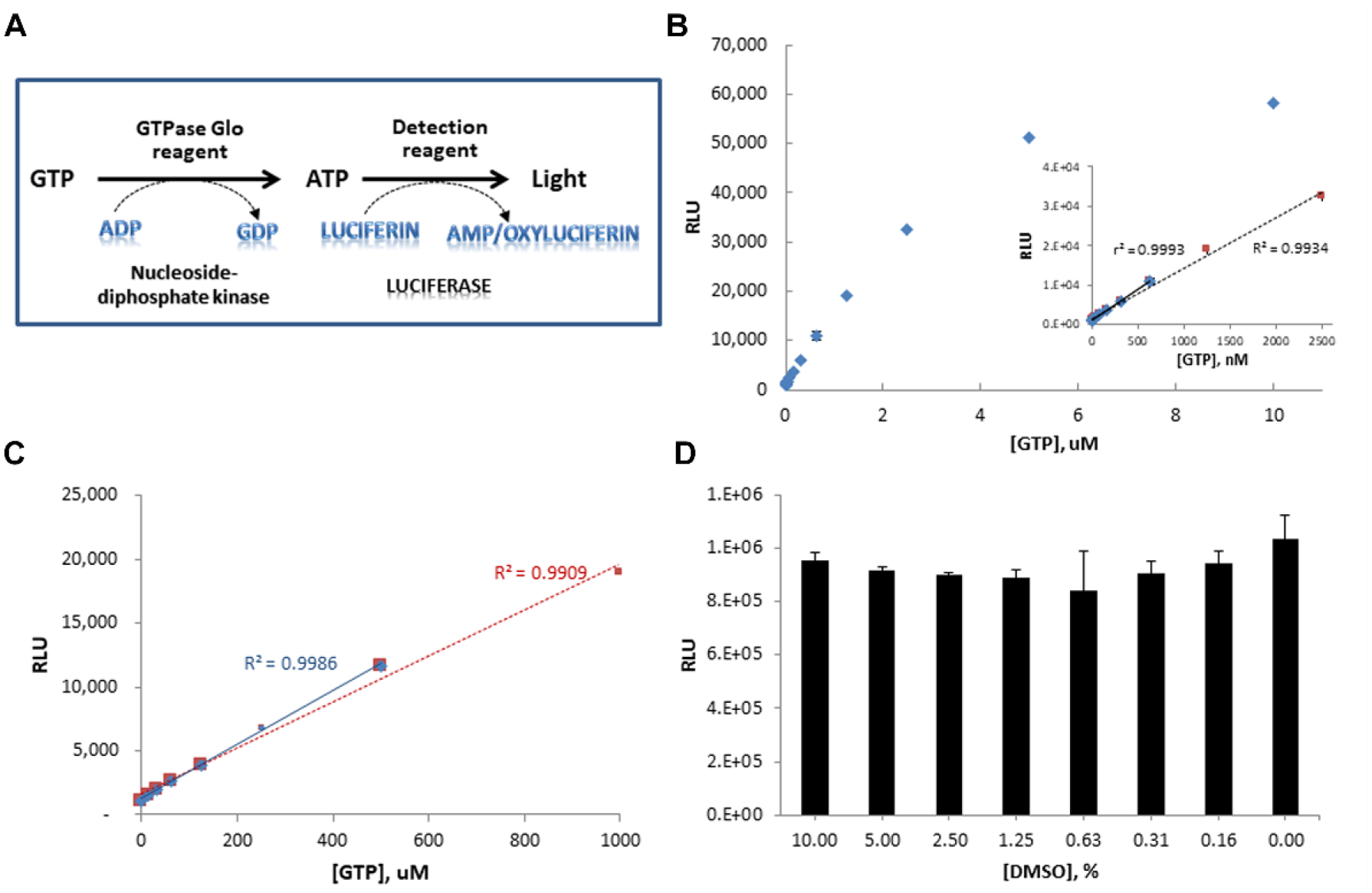
Luminescence-based detection platform for monitoring NFeoB GTPase activity. (
Next, the modified GTPase-Glo assay was validated for the detection of GTPase activity of NFeoB using the same enzymatic reaction protocol optimized in the colorimetric assay. The resulting luminescence signal corresponded to the depletion of ~65 µM GTP. This value is comparable with the value measured by the colorimetric platform, proving the reliability of the luminescence-based platform to monitor the NFeoB GTPase activity. The S/B ratio and z′ calculated based on phosphate production resulted in 13.31 and 0.83, supporting the potential of this assay to validate compound activity. The tolerance test of this new detection platform against DMSO showed its stability up to 10% DMSO ( Fig. 4D ). While further modification of this new luminescence-based strategy can improve the robustness of this platform to be suitable for practical application to HTS campaigns, this assay still provides a convenient alternative to colorimetric detection to monitor the GTPase activity of NFeoB. While surface plasmon resonance (SPR) and thermal melt assay are good alternative methods to verify the binding kinetics of inhibitors or the protein stability change by inhibitor binding, they would not provide direct information on the GTP hydrolysis activity of FeoB. Additionally, chromatographic analysis measuring conversion of 32P-GTP has been utilized to confirm compound inhibition in a similar screening system, 12 but this requires the use of expensive instrumentation and it generates hazardous waste. The distinctive advantages of the luminescence-based GTPase-Glo kit as an orthogonal assay over the other assays are that (1) it provides direct information on GTP hydrolysis activity; (2) it can be readily adapted to an HTS platform; and (3) its application allows for a miniaturized scale and produces less waste. In this regard, colorimetric and luminescence-based detection platforms were chosen for primary screening and hit validation, respectively.
Primary Screening and Hit Validation
A primary screen was performed using the colorimetric detection platform with a custom-assembled library. The averaged S/B ratio, S/N ratio, and z′ factor were 1.90 ± 0.03, 55.1 ± 0.02, and 0.75 ± 0.04, respectively. Initial screening at 10 µM did not identify any potent hits. Rescreening at a final concentration of 100 µM identified 11 compounds over 35% inhibition against the GTPase activity of NFeoB.
Eleven compounds were tested again in both the colorimetric and luminescence detection platforms in parallel at 100 µM. As summarized on Figure 5A , 6 compounds out of 11 failed to reproduce their activity in the colorimetric platform, indicating that they were false hits due to the systematic error. Among the remaining five compounds (compounds 2, 6, 7, 8, and 11), three compounds were inactive in the luminescence assay, suggesting that they were false hits due to assay interference in primary screening. Two compounds (compounds 2 and 8) were confirmed as reproducible using the colorimetric assay, while comparable activities in both platforms were observed.
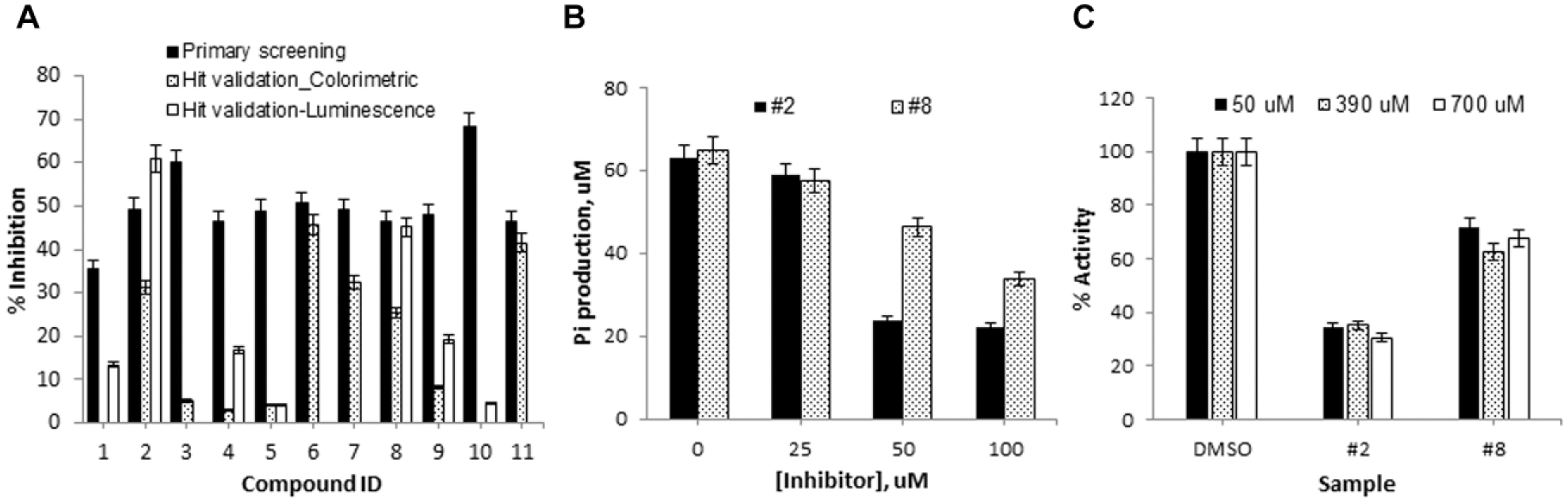
Hit validation. (
As shown in Figure 5B , a dose–response experiment using the colorimetric assay demonstrated that both compounds showed dose-dependent inhibition activities with IC50 values estimated at ~30 and 100 µM for compounds 2 and 8, respectively. The mode of inhibition mechanism was examined at low, medium, and high substrate concentrations using the colorimetric detection platform. As shown in Figure 5C , their inhibitory activity was not altered at any substrate level, indicating that their inhibitory mechanism is noncompetitive and both compounds inhibit the GTPase activity allosterically. CID1067700 and 3-methoxybenzamide, which are known inhibitors against Ras GTPases or FtsZ GTPase, respectively, did not inhibit the GTPase activity of VcNFeoB17,18 (data not shown). Nonhydrolyzable GTP-gamma-S would be used as a control compound, although it only represents a competitive inhibition. 19 Compounds 2 and 8 have been known as an inhibitor or an agonist of specific signaling cascades in humans. However, there are no reports regarding an effect of these compounds on a bacterial system. Further evaluation using in vitro cell-based and in vivo assays is undergoing.
To initially eliminate false hits due to colorimetric assay interference, a counterscreen can be performed by replacing substrate and enzyme with 60 µM phosphate standard. However, a different detection platform is more desirable to securely discriminate false hits considering the weak sensitivity and absorbance that may arise from the compounds themselves. Here, we have presented two detection platforms that measure either the product formation (MG) or substrate depletion (luminescence) and are individually optimized to a convenient mix-and-read protocol that can be read on a plate reader. The advantages of establishing two platforms for NFeoB relying on different detection strategies are (1) the same enzymatic assay protocol can be adapted instantly and (2) it can efficiently eliminate false hits due to spectroscopic interference. We demonstrated that the commercial GTPase-Glo assay can be amended for the biochemical GTPase assay where the
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Cancer Prevention and Research Institute of Texas (CPRIT), grant RP160657, to K.N.D. and by NIAID, grant R01AI091957, to S.M.P.
