Abstract
Chemotaxis is the directional movement of cells in response to a chemical stimulus and is vital for many physiological processes, including immune responses, tumor metastasis, wound healing, and blood vessel formation. Therefore, modulation of chemotaxis is likely to be of therapeutic benefit. Hence, a high-throughput means to conduct chemotaxis assays is advantageous for lead evaluation and optimization in drug discovery. In this study, we have validated a novel approach for a higher-throughput, label-free, image-based IncuCyte chemotaxis assay encompassing various cell types, including T cells, B cells, mouse Th17, immature and mature dendritic cells, monocyte THP-1, CCRF-CEM, monocytes, neutrophils, macrophages, and MDA-MB-231. These assays enable us to visualize chemotactic cell migration in real time and perform kinetic cell motility studies on an automated platform, thereby allowing us to incorporate the quantitative studies of cell migration behavior into a routine drug discovery screening cascade.
Keywords
Introduction
Chemotaxis is the movement of an organism or cells in response to a chemical stimulus. Over the past 100 years, research has illustrated the importance of chemotaxis in physiological processes, such as the recruitment of immune cells to the sites of infection, organ development during embryogenesis, wound healing and tissue repair, and tumor cell metastasis. 1 Chemotaxis not only affects tumor cells but also helps to shape the tumor microenvironment. 2 The directional migration to a chemokine/growth factor source for most cells forms a dynamic network in the tumor microenvironment. In the context of the tumor microenvironment, several major events are regulated by the chemotaxis network, including immune evasion, angiogenesis, invasion, and dissemination. 2
Given the importance of chemotaxis in cancer, immune disease, and fibrosis, the development of therapeutics targeting chemotaxis is a strategy and opportunity, not only for the development of new drug candidates, but also for safety assessment.
Chemokines are small-molecular-weight (8–14 kDa) chemoattractant proteins that signal by binding to G-protein-coupled, seven-transmembrane-spanning receptors. 3 Chemokines have well-defined roles in directing cell movements necessary for the initiation of T-cell immune responses. 4 Chemokines are also needed to attract monocytes and immature dendritic cells (iDCs) to sites of inflammation, direct maturing antigen-presenting cells (APCs) to lymphatic vessels, and bring T cells and APCs together within the draining lymphoid organ. 5
Traditionally, the Boyden chamber assay has been used to measure chemotaxis, which was originally introduced by Steven Boyden in the 1960s for the analysis of leukocyte chemotaxis. 6 The Boyden chamber assay is based on a chamber of two medium-filled compartments separated by a microporous membrane. In general, cells are placed in the upper compartment and the chemotactic agents are present in the lower compartment. After an appropriate incubation time, the membrane between the two compartments is fixed and stained; thus, the number of cells that have migrated to the lower side of the membrane is determined. 6
Several challenges have been found with the Boyden chamber assay for both biology and throughput ( Suppl. Table S1 ). The Boyden chamber membrane has many more pores, and it requires a high cell density. The cell movement cannot be excluded from a random gravity-dependent gradient. In addition, the Boyden chamber is an end-point assay; the rate of migration cannot be measured, nor can cell morphology be observed. The Boyden chamber requires fixing and staining of the cells, and the chemical gradient has a short life. Furthermore, it is not amenable for automation and has no integrated data quantitation. 7
Recently, Essen BioScience launched the IncuCyte chemotaxis module, a novel, integrated system for real-time visualization and automated analysis of chemotactic cell migration in a 96-well format within a tissue culture incubator. 8
In comparison with the Boyden chamber system, the IncuCyte chemotaxis module has significant advantages. IncuCyte’s proprietary plate has fewer pores constructed on the membrane and requires much fewer cells in the assay. It not only is beneficial for the study of primary cells, but also allows monitoring of cell migration on a horizontal surface prior to the vertical axis, which is a more physiologically relevant movement independent of gravity. IncuCyte technology provides kinetic measurement of chemotaxis and enables the detection of cell morphology. Unlike the Boyden chamber, IncuCyte investigates cell migration on biologically relevant surfaces. Furthermore, the label-free IncuCyte chemotaxis module has integrated data algorithms and is feasible for automation. Most importantly, the chemotactic gradient can be stable for more than 72 h in the IncuCyte chemotaxis module.
In this article, we present our validation results of the IncuCyte technology for both adherent and suspension cells, including T cells, iDCs and mature dendritic cells (mDCs), monocytes, and CCRF-CEM and MDA-MB-231 cells for their migration behaviors directed by an array of chemokines, and their relevant chemokine receptors, including MIP-1α/CCR1 (CC chemokine receptor-1), MCP-1/CCR2, and SDF-1α/CXCR4. We also share our efforts for the installment of a fully automated chemotaxis platform for the analysis of the structure–activity relationship (SAR) for drug discovery efforts.
Materials and Methods
Reagents
SDF-1α, MIP-1α, and MCP-1 were purchased from R&D Systems (Minneapolis, MN). Phytohemagglutinin (PHA) and AMD3100 were from Sigma-Aldrich (St. Louis, MO). All Bristol-Myers Squibb (BMS) test compounds were made internally at BMS. Granulocyte-macrophage colony-stimulating factor (GM-CSF), interleukin (IL)-2, and IL-4 were purchased from Peprotech (Rocky Hill, NJ). Lipopolysaccharide (LPS) was from Invivogen (San Diego, CA). Anti-human CD3, CD4, CD8, CD25, CD83, CD86, and FoxP3 antibodies were from eBioscience (San Diego, CA).
Cells Culture
The monocyte THP-1, CCRE-CEM, and MDA-MB231 cell lines were purchased from American Type Culture Collection (ATCC, Manassas, VA). The monocyte THP-1, CCRE-CEM, and MDA-MB-231 were cultured in RPMI-1640 medium (Invitrogen, Carlsbad, CA) supplemented with 10% heat-inactivated fetal bovine serum (FBS, Sigma-Aldrich) and 1% Pen/Step (Invitrogen). CCRE-CEM and MDA-MB-231 also have 10 mM of HEPES (Invitrogen), and monocyte THP-1 has 55 µM of 2-mercaptoethanol (Invitrogen) as additives.
Primary Cell Preparation
DC preparation: iDCs were differentiated from monocytes (Biological Specialty Corporation, Colmar, PA) with the addition of a cocktail of GM-CSF (100 ng/mL) and IL-4 (200 ng/mL) for 6 days at 37 °C, 5% CO2. Cytokines were replenished on day 3 by adding fresh medium. For maturation, iDCs were exposed to 1 µg/mL of LPS (Invivogen, cat. tlrl-eblps) for 1 day. mDCs were stained using anti-CD83 and anti-CD86 to confirm the purity of the preparation.
PHA blast preparation: Peripheral blood mononuclear cells (PBMCs) were isolated from human whole blood. After 3 days of stimulation with 5 µg/mL of PHA (Sigma-Aldrich, cat. L1668), T cells were further expanded using IL-2 (20 ng/mL) for 4 days. Immunofluorescence staining of anti-CD3, CD4, and CD8 antibodies confirmed the mixed population of the T cells in the PHA blast preparation.
Treg preparation: CD25-positive selection was done using fresh human CD4+ T cells (Biological Specialty Corporation). Tregs (CD4+ CD25+) were expanded using the Miltenyl kit (cat. 130-091-301, San Diego, CA) for 10–14 days. Tregs were frozen using 90% FBS and 10% DMSO. The isolated Tregs were stained with the following antibodies: anti-CD4 AF 488, anti-CD25 PE-Cy 7, and anti-FoxP3 APC (eBioscience, San Diego, CA) and analyzed on an Intellicyte iQue Screener Plus flow cytometer (Albuquerque, NM).
IncuCyte Chemotaxis Assay
Matrigel coating: The IncuCyte ClearView migration plate (Essen BioScience, cat. 4582) consists of three parts: lid, insert, and reservoir ( Suppl. Fig. S1A ). Prior to the assay, the Matrigel (Corning, cat. 354234) was thawed overnight on ice at 4 °C. The migration plate, pipette tips, and reagents were prechilled to 4 °C. Matrigel (50 μg/mL) was prepared in RPMI (Life Technologies 11875-085, Waltham, MA) plus 10% FBS (Sigma-Aldrich F2442). To coat both sides of the membrane, 20 μL of Matrigel was applied to the insert wells and 150 μL of Matrigel was added to the reservoir wells; then the insert was gently placed into the reservoir plate containing coating matrix ( Suppl. Fig. S1B ). The migration plate was placed at 37 °C for 30 min. After that, the migration plate was moved to room temperature for an additional 30 min.
Test compound preparation: Test compounds were serially diluted in DMSO with threefold dilutions for 11 concentration points ranging from 1.69 × 10−7 to 10−2 M.
Chemokine preparation: MIP-1α, MCP-1, and SDF-1α were reconstituted in PBS to a concentration of 100 µg/mL or according to the manufacturer’s instruction. To measure chemokine-mediated cell migration, MIP-1α, MCP-1, and SDF-1α were serially diluted in the assay buffer for a threefold, 11-point concentration–response curve.
Cell preparation: For suspension cells, including THP-1, DCs, CCRF-CEM, or T cells, the cell density was measured using the Cellometer Auto M4 (Nexcelom Bioscience, Lawrence, MA) according to the manufacturer’s manual. The cell suspension was centrifuged at 1200 rpm for 5 min, and the cell pellet was resuspended in assay medium (RPMI [Life Technologies 11875-085] with 0.5% FBS [Sigma-Aldrich F2442]) at a density of 8.4 × 104 cells/mL.
For adherent MDA-MB-231 cells, complete cell culture medium DMEM (Life Technologies 11995) plus 10% FBS (Sigma-Aldrich F2442) was aspirated from the T150 flask and the cells were gently rinsed twice with dPBS (without Ca2+, Mg2+, Life Technologies 10010). The cells were harvested using trypsin (Gibco, 25300054), and the cell count was performed using Cellometer Auto M4 according to the manufacturer’s manual. The appropriate cell suspension was centrifuged at 1200 rpm for 5 min. The cell pellet was resuspended in assay buffer (DMEM + 2.5% FBS) at the density of 1.7 × 104 cells/mL.
Chemotaxis measurement with compound test: After Matrigel coating, 200 μL of dPBS was added to a new reservoir plate. Immediately prior to cell addition, the insert was taken out and it was gently placed into the new dPBS reservoir plate. Twenty microliters of Matrigel from the insert wells was aspirated out using a multichannel pipette. Sixty microliters of cells was then loaded into the insert well for a cell density of 5000 cells/well for suspension and 1000 cells/well for surface attachment cells MDA-MB-231. For compound inhibition studies, 100 nL of serial diluted compounds was transferred onto the insert well using the Echo Liquid Handling Platform (Labcyte, Sunnyvale, CA). The cells and compounds were incubated at room temperature for 30 min. Meanwhile, the optimal concentration of the chemoattractant was made in assay medium (RPMI with 0.5% FBS) and 200 μL of the chemoattractant or control medium was added onto the appropriate wells in a new reservoir plate. After incubation, the insert plate containing the cells was carefully transferred into the reservoir plate containing the chemoattractant ( Suppl. Fig. S1B ). The IncuCyte ClearView cell migration plate was subsequently placed into the IncuCyte ZOOM instrument, and repeat scanning (10×) for every 30 min was scheduled over a 24 h period according to the manufacturer’s manual.
Chemotaxis data quantification and analysis: Whole-well images of cells on both sides of the membrane either from the top or from the bottom of the ClearView migration plate were captured at user-defined intervals. All images were processed using automated algorithms to quantify cell areas on each side of the membrane. Directed cell migration can be reported as either an increase in area on the bottom side of the membrane for adherent cells that migrated down the pores to the reservoir wells, or a decrease in area on the top side of the membrane for nonadherent cells or adherent cells ( Suppl. Fig. S1C ).
Results were plotted using the raw signal count; percent inhibition was calculated based on the average signal of the chemoattractant-stimulated (positive) and unstimulated (negative) controls. Duplicate results were plotted to generate a dose–response curve. The IC50 is defined as the concentration of test compound corresponding to 50% inhibition derived from the 11-point fitted curve as determined using a four-parameter logistic regression model. The coefficient of determination (R2) was used for determining the degree of linear correlation of variables in regression analysis and was calculated using defined equations in Microsoft Excel. 9 The Z′ value was used to measure the assay quality.
Results and Discussion
Monocyte THP-1 Chemotaxis
CCR1 is a class A G-protein-coupled receptor (GPCR), 10 expressed by several cells of the immune system, including monocytes and macrophages, DCs, osteoclasts, T lymphocytes, and neutrophils. 10 CCR1 binds several chemokine ligands such as MIP-1α, RANTES, and leukotactin-1. Chemokine–CCR1 interactions mediate leukocyte activation and migration, which plays a critical role in the host response to pathogens, as well as in several inflammatory conditions. 10
Preclinical studies in rodents demonstrated that CCR1 receptor blockade represents a viable strategy for the development of therapies for inflammatory disorders. 11 The BMS CCR1 program aimed to discover a small-molecule antagonist for the treatment of rheumatoid arthritis (RA). 10 The Boyden chamber assay has been used to evaluate the CCR1 inhibitor in the monocyte THP-1 background. 10
For the proof-of-concept study of evaluating the IncuCyte technology for application to chemotaxis studies, MIP-1α-stimulated THP-1 chemotaxis was conducted. MIP-1α titration showed a typical bell-shaped dose-dependent response, with cell migration returning to control levels at higher concentrations of chemoattractant. 12 MIP-1α displayed a compatible EC50 of 0.2 nM compared with the historical Boyden chamber data of 0.1 nM ( Fig. 1A ).
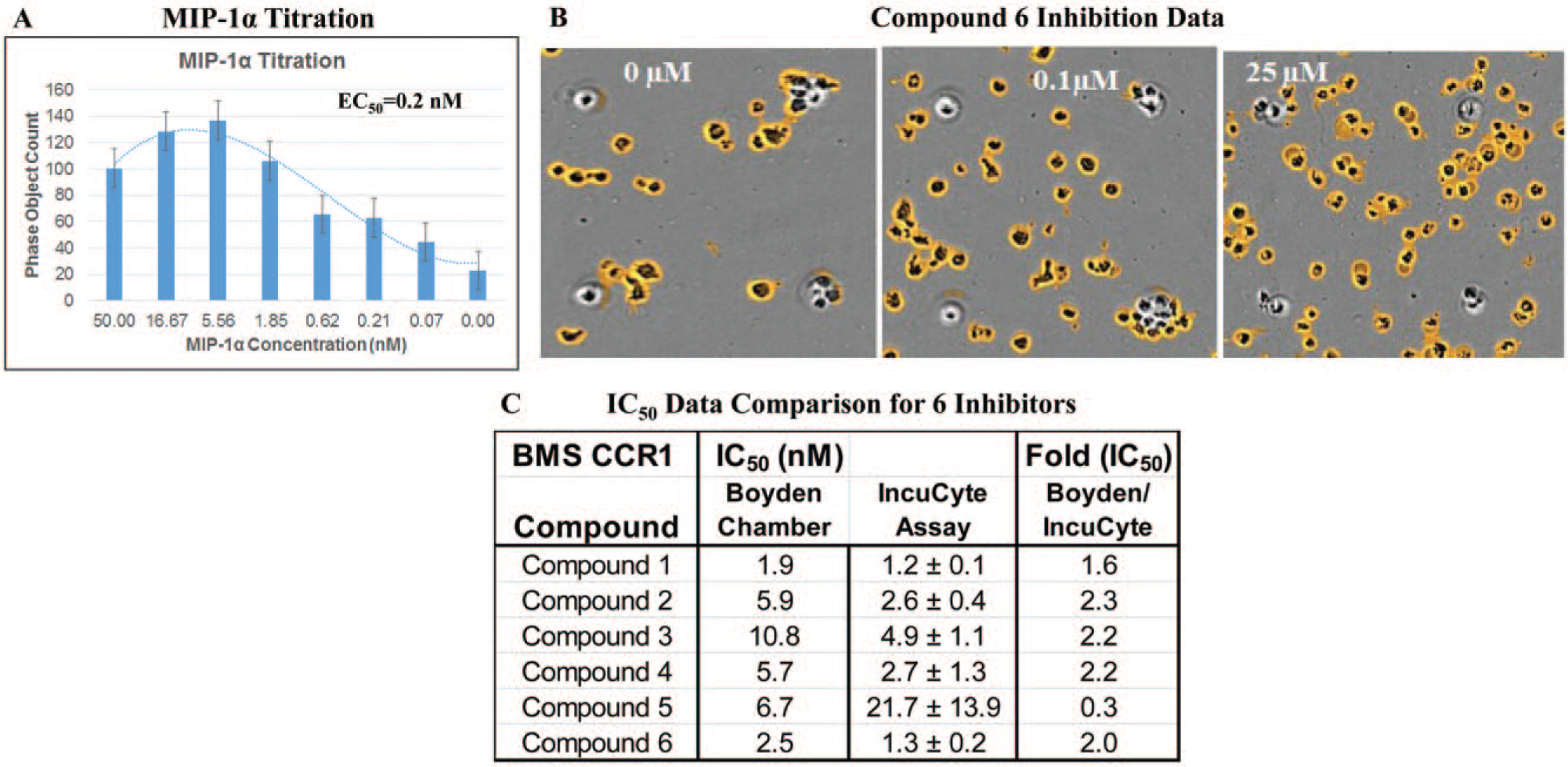
MIP-1α/CCR1-mediated THP-1 chemotaxis assay. (
For compound inhibition, more THP-1 cells on the top well were observed at higher concentrations of a CCR1 antagonist compound 6, indicating the inhibition of the THP-1 migration at higher concentrations of compound 6 ( Fig. 1B ). IC50 data of six CCR1 compounds obtained from IncuCyte chemotaxis are within a threefold difference of those from the historical Boyden chamber ( Fig. 1C ). Data represented mean ± SD (n = 3). Pharmacology data of CCR1 agonist and antagonists demonstrated good alignment comparing IncuCyte chemotaxis with Boyden chamber data.
Dendritic Cell Chemotaxis
DCs are the most potent APCs and are essential for the initiation of an immune response due to their unique ability to take up and process antigen, translocate to lymph nodes, and present processed antigen to naive T cells. 13 The maturation phenotype of the DC is central to immune regulation and polarization of Th1/Th2 responses. 14 Many chemokines, chemokine receptors, and GPCRs are implicated in these various aspects of DC biology. 15
Studies have shown iDC express CCR1, CCR2, CCR5, CXCR1, CXCR2, and CXCR4, while mDCs express CCR7 and CXCR4. 16 To shed light on the emerging significance of various members of the chemokines and chemokine GPCRs in DC biology, we explored CCR1, CCR2, and CXCR4, as well as their ligand MIP-1α, MCP-1, and SDF-1α, respectively, for their role in mediating chemotaxis in the DCs.
MIP-1α and MCP-1-Mediated iDC Chemotaxis
MIP-1α and MCP-1 showed a dose-dependent response in the iDC chemotaxis with EC50 values of 0.16 and 5.6 nM, respectively. No significant chemotactic response was observed for both MIP-1α and MCP-1 in the mDCs, indicating that the expression of CCR1 and CCR2 is distinct for iDCs ( Fig. 2A,B ).
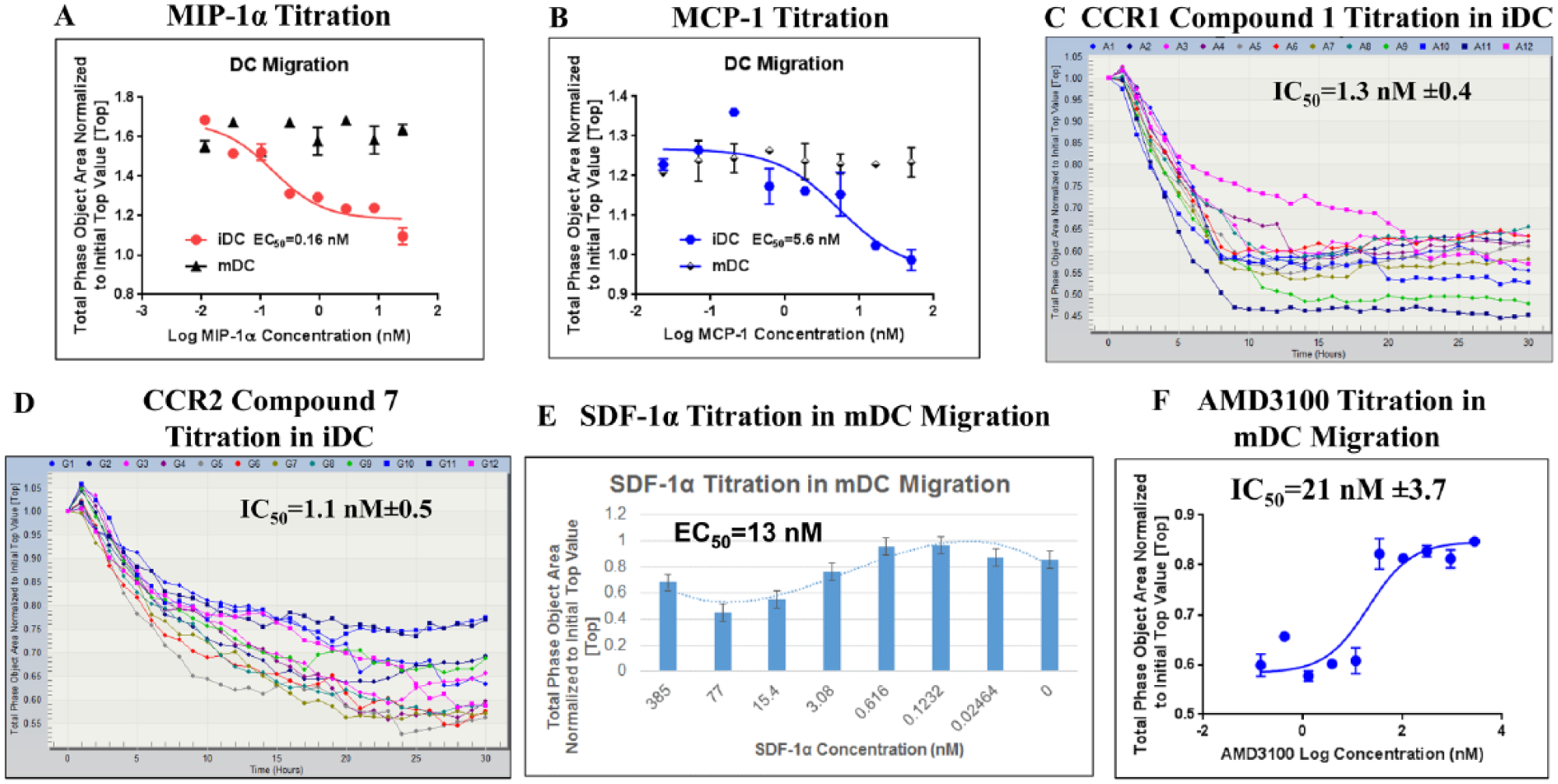
DC chemotaxis assay. (
A CCR1 inhibitor compound 1 was tested in the iDC migration assay in the presence of 4 nM MIP-1α. A CCR2 antagonist compound 7 was profiled in the MCP-1 (20 nM)-mediated iDC migration assay. Compounds 1 and 7 showed an IC50 of 1.3 and 1.1 nM, respectively, in the IncuCyte chemotaxis assay, and these data displayed good correlation compared with historical Boyden chamber data (1.9 and 1.7 nM, respectively) ( Fig. 2C,D ).
SDF-1α-Mediated mDC Chemotaxis
The continuous migration of DCs and T cells into secondary lymphoid organs such as lymph nodes represents a fundamental element of immune surveillance and homeostasis. mDCs migrate through the afferent lymphatic vessels to the lymph nodes, where they regulate the initiation of the immune response. 17 SDF-1α is one of the key chemokines directing the mDC migration through CXCR4 from sites of inflammation to T-cell zones of draining lymph nodes. 18 SDF-1α/CXCR4-mediated mDC migration was reported in vivo from the skin to regional lymph nodes. 18
SDF-1α-mediated mDC chemotaxis exhibited a bell-shaped dose-dependent response, 19 with an EC50 of 13 nM ( Fig. 2E ). An IC50 of 21 nM was obtained for AMD3100 ( Fig. 2F ), a small-molecule specific antagonist of the CXCR4 receptor that competitively binds and prevents the interaction of the receptor with SDF-1α. 20
MDA-MB-231 Chemotaxis
Many types of tumor cells have been found to express high levels of CXCR4. 21 In addition, tumor metastasis target tissues (lung, liver, and bone) express high levels of the CXCR4 ligand SDF-1α, which allows tumor cells to directionally migrate to target organs via an SDF-1α/CXCR4 chemotactic gradient. 22
Breast cancer is the most common malignancy with metastases at distant sites, which is the main cause of cancer-related death among Western women. 23
CXCR4 was found to be the most abundant chemokine receptor in breast cancer. 20 The expression of CXCR4 in breast tumors has been correlated with poor prognosis, increased metastasis, and poor treatment outcomes. 22 Immunofluorescence staining of CXCR4 in normal breast tissue shows that CXCR4 expression levels are low, whereas CXCR4 staining in primary tumors and lymph node metastases from the same breast cancer patient showed elevated CXCR4 protein levels. 24
MDA-MB-231 is a human breast cancer cell line with high metastatic potential. It has high levels of CXCR4 expression with very weak SDF-1α expression. 24 Therefore, targeting SDF-1α/CXCR4 migration in MDA-MB-231 could have significant implications for treating breast cancer. Focusing on the SDF-1α/CXCR4 axis in regulating the metastatic behavior of tumor cells could allow us to gain insight for the molecular mechanisms that are essential in the morbidity and mortality of breast cancer.
SDF-1α showed a dose-dependent bell-shaped response in the MDA-MB-231 chemotaxis assay with an EC50 of 22 nM ( Fig. 3A ). In SDF-1α-stimulated MDA-MB-231 chemotaxis, AMD3100 displayed an IC50 of 82 nM ( Fig. 3B ). AMD3100 was evaluated for cytotoxicity with an MTS assay, and no discernable effect was observed.
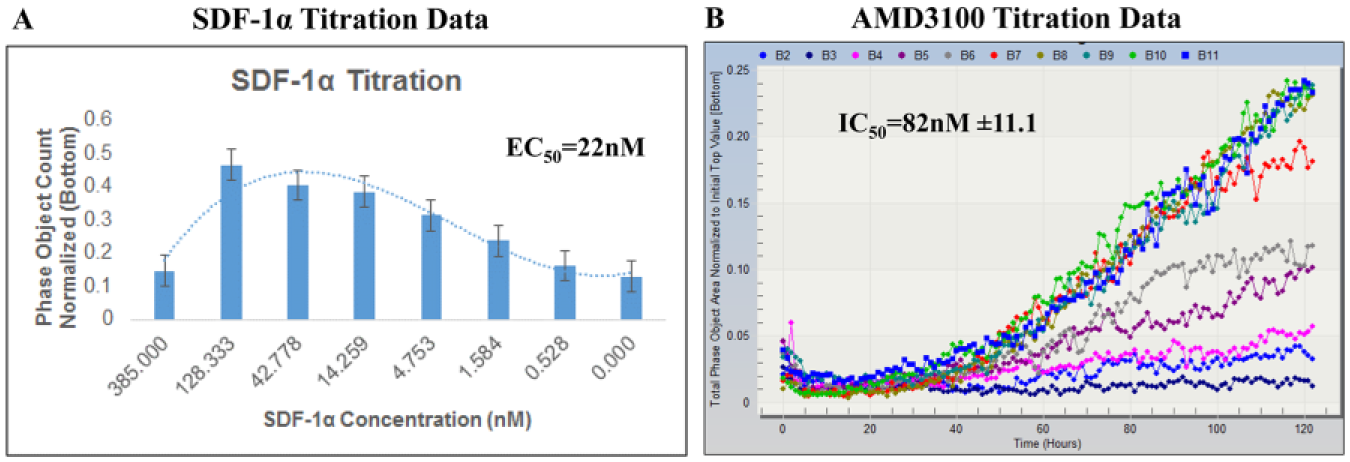
MDA-MB-231 migration assay profile. (
The in vitro chemotaxis assay data described here, combined with other published results, demonstrated that the SDF-1α/CXCR4 interaction in regulating MDA-MB-231 migration can be used as a measurement of breast cancer metastasis. 24
T-Cell Migration
Activated T-Cell Migration
Efficient migration of leukocytes from peripheral blood into and within tissues is essential to an optimal immune response. 25 T lymphocytes are prominent drivers of many autoimmune diseases; the regulation of T-cell trafficking involves the coordination of chemokines and specific chemokine receptors, which provide directional cues critical to T-cell trafficking, migration into inflammatory sites, retention of hematopoietic precursors in the bone marrow, and anatomic compartmentalization of lymphocyte subpopulations in secondary lymphoid organs. 26
Studies showed that the chemokine receptor CXCR4 is constitutively expressed in T lymphocytes and regulates T-cell migration along gradients of the chemokine SDF-1α. 27 Studies also showed that both SDF-1α and CXCR4 “knockout” mice exhibited critical defects in leukocyte generation and hematopoiesis, leading to embryonic and neonatal fatalities. 28 In RA, increased amounts of SDF-1α mRNA were found in RA synoviocytes. 29 In addition, elevated CXCR4 expression by synovial memory T cells was reported. 29 Furthermore, small-molecule antagonists and CXCR4 knockout mice both exhibited reduced joint inflammation. 30
To gain an understanding of the SDF-1α/CXCR4 interaction and its role in inflammation, we explored SDF-1α-mediated T-cell migration.
SDF-1α-mediated chemotaxis was observed in PHA blasts with an EC50 of 33 nM ( Fig. 4A ). AMD3100 displayed an IC50 of 99 nM ( Fig. 4B ) in the SDF-1α-stimulated PHA blast migration assay.
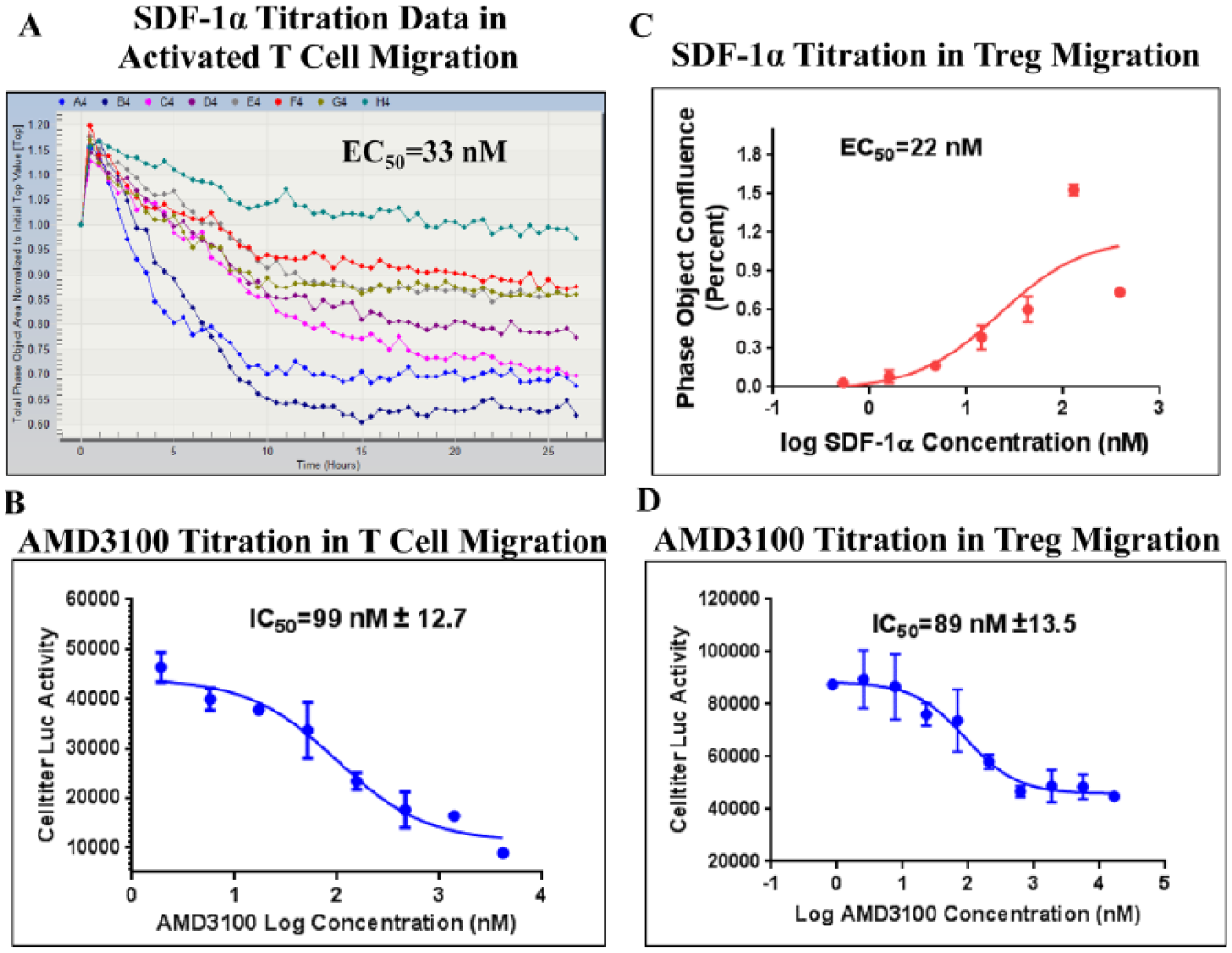
SDF-1α/CXCR4-mediated T-cell chemotaxis assay profile. (
Although several other autoimmune disorders, such as systemic lupus erythematosus, uveitis, and multiple sclerosis, demonstrated aberrant CXCR4/SDF-1α-mediated inflammatory responses, 31 the mechanism of CXCR4/SDF-1α interaction is still not entirely elucidated. Further investigation of the CXCR4/SDF-1α chemokine receptor axis and its relevant signatures in autoimmune pathogenesis may provide novel approaches for therapeutic intervention. 32
Treg Migration
In recent years, immunotherapy using Treg cells has emerged as a novel approach for anticancer therapy. 33
CD4+ CD25+ regulatory T cells (Treg) are a subset of CD4+ T cells with the functional ability to suppress effector T-cell immune responses. Tregs play a vital role in immunologic self-tolerance, antitumor immune responses, and transplantation. 34 Increased Treg cells have been found in patients with hepatocellular cancer (HCC), ovarian cancer, and gastric and esophageal carcinomas. Experimental tumor models show that removal of CD25+ T cells alters the immune response to tumors both in vitro and in vivo. 35
Studies have demonstrated chemokine receptor CXCR4 expression on Tregs, 36 which enables migration of Treg cells toward the chemokine SDF-1α in tumor microenvironments. 37 The SDF-1α/CXCR4 interaction may therefore facilitate Treg accumulation in tumors. 38 Furthermore, CXCR4 antagonism was found to selectively reduce the intratumor FoxP3+ T-cell infiltration in comparison with CD8+ T cells. 39 As a result, the SDF-1α/CXCR4-mediated Treg migration became part of the mode of action (MOA) study for the BMS CXCR4 program, which was seeking a CXCR4 antagonist for antitumor treatment. 39
SDF-1α exhibited an EC50 of 22 nM in mediating Treg chemotaxis ( Fig. 4C ). AMD3100 displayed an IC50 of 89 nM ( Fig. 4D ) in the SDF-1α-stimulated Treg migration assay.
In summary, our data showed that the SDF-1α/CXCR4 axis played a role in regulating T-cell (for both PHA blasts and Tregs), mDC, and MDA-MB-231 migration. Taken together, these chemotaxis study data showed the different biological properties of SDF-1α/CXCR4 and its potential implications for autoimmune diseases, cancer, and tumor cell motility in metastasis. Further characterization of the SDF-1α/CXCR4 axis may provide a better understanding of the mechanism of action toward the progression of these diseases, which would warrant further investigation to develop anti-inflammation and antimetastatic drugs.
IncuCyte Chemotaxis Assay Reproducibility in CCRF-CEM Cells
A SDF-1α-mediated CCRF-CEM IncuCyte chemotaxis assay was developed, and SDF-1α presented an EC50 of 18 nM ( Fig. 5A ). Ten CXCR4 antagonists were tested through the CCRF-CEM migration assay using SDF-1α as the stimuli; the IC50 data exhibited the same rank order for these 10 compounds comparing the CCRF-CEM IncuCyte chemotaxis data with the Boyden chamber data. A correlation plot displayed a R2 value of 0.80 ( Fig. 5B ), supporting good alignment between the Boyden chamber and IncuCyte assays.
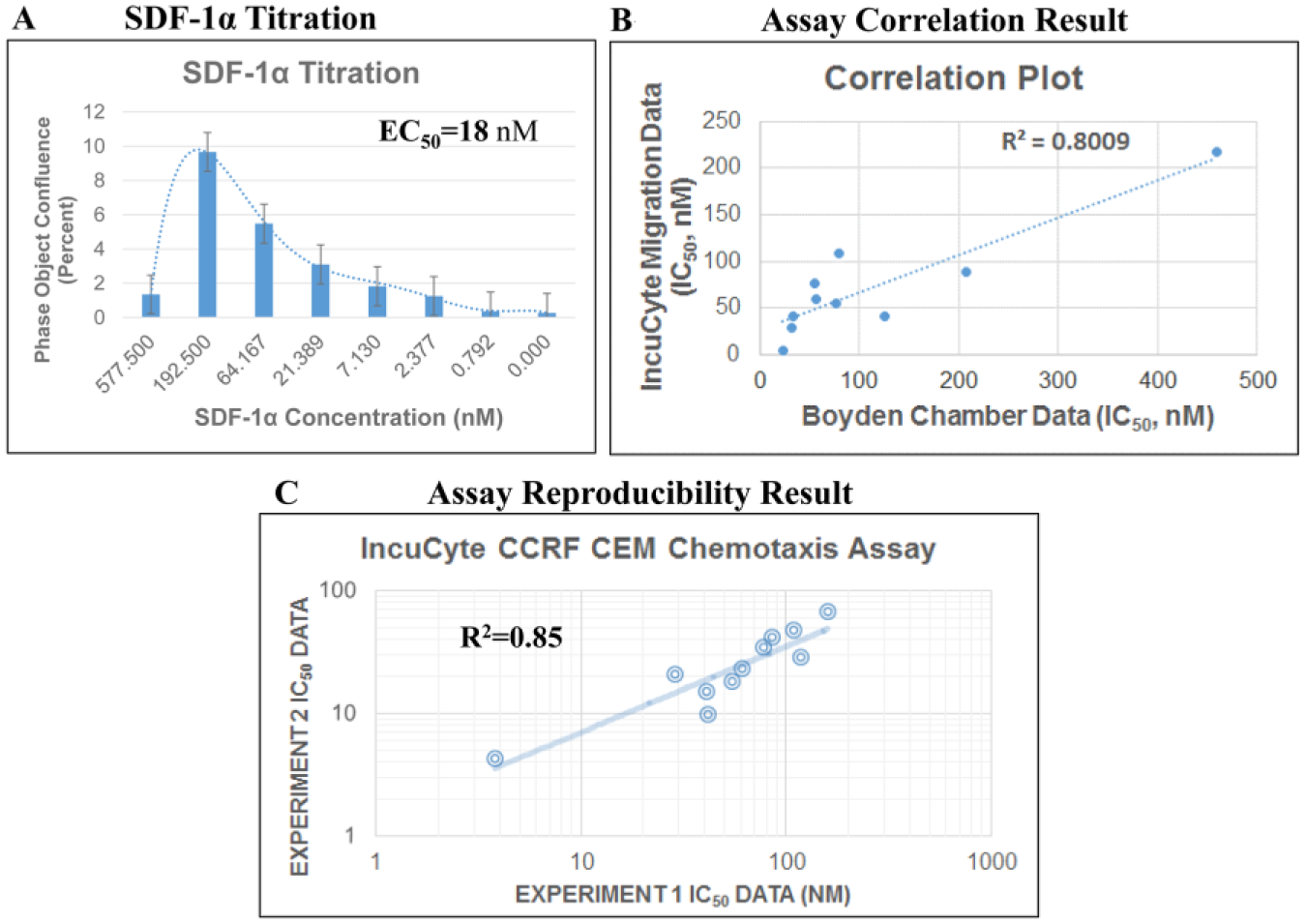
CCRF-CEM chemotaxis screening assay. (
Two independent IncuCyte CCRF-CEM chemotaxis assays were performed on different days; IC50 data from the n = 1 test correlated well with n = 2 data, indicating good assay reproducibility with a R2 value of 0.85 ( Fig. 5C ).
Automated IncuCyte Chemotaxis Assay and Assay Process Implementation
Lead optimization is one of the most critical steps during the early phase of drug discovery. Its efficiency directly affects the capability to turn the most promising compounds into early candidate nominations (ECNs) for clinical development. 40 Chemotaxis data have become high demand in several therapeutic areas in BMS. However, IncuCyte chemotaxis assays involve many steps, including matrix protein coating, aspirating, reagent addition, and plate transfer, which resulted in the inherent challenges for scaling up the throughput of compound testing using the manual mode. To address these challenges, we utilized lab automation to incorporate a highly efficient process for the establishment of an automated chemotaxis platform.
The integrated automated IncuCyte migration system has components such as an Echo 550 (Labcyte), an acoustic liquid dispenser for compound dosing, and a CyBi-FeliX (Analytik Jena AG, Jena, Germany) for Matrigel aspiration. Also, the IncuCyte migration platform is centered by BMS Thermo Work-Station3 (WS3), which consists of Multidrop Combi (Thermo Scientific) for reagent dispensing, as well as a Spinnaker Robot (Thermo Scientific). The Spinnaker Robotic System has a Spinnaker Mover, a built-in imaging bar code reader, an integrated regrip station, and a microplate hotel for plate moving and assay connection. Additional components include a Cytomat incubator (Thermo Scientific), IncuCyte Zoom, and HP computer with Thermo Scientific Momentum software for robot scheduling and execution ( Fig. 6A ).
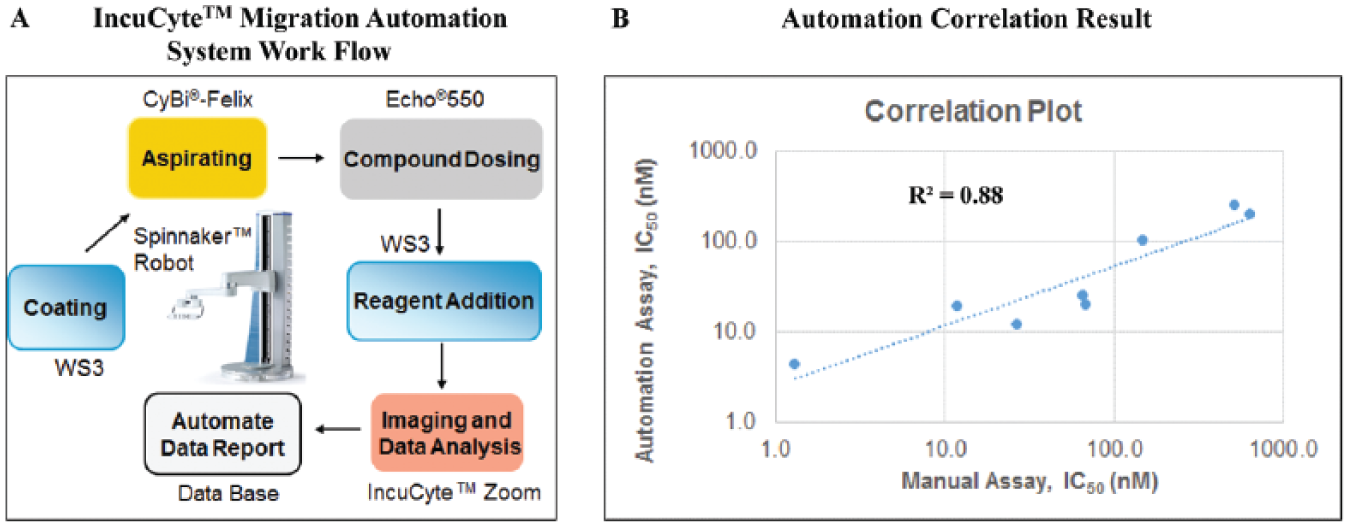
Leveraging automation system to support migration panel screening. (
To validate the automated IncuCyte migration system, an SDF-1α-mediated Treg chemotaxis assay was conducted. At the completion of an assay plate, the data are saved by the Zoom software on the computer. Subsequently, the data are transferred into the BMS in-house IT tool Toolset and the CurveMaster utility, where IC50 data and curves were produced. 9
IC50 data for 8 CXCR4 compounds showed good alignment comparing the automated and manual SDF-1α-mediated Treg IncuCyte chemotaxis assays. These two sets of data agreed well in the correlation plot with a R2 of 0.88. A Z′ value of 0.6 was obtained ( Fig. 6B ), indicating good robustness and precision comparing the automation assay with the manual assay. 9
We have demonstrated that the IncuCyte chemotaxis platform is a quantitative and reproducible approach for measuring chemotaxis for both adherent and suspension cell types. This platform allows the kinetic measurement of cell migration toward chemotactic gradients in real time on a physiologically relevant matrix surface. It provides movies and images with morphological and phenotypic insights.
The IncuCyte chemotaxis platform enabled MOA studies of drug targets for their chemotaxis profiling in multiple cell types, including T cells (Treg, PHA blasts), iDCs and mDCs, and monocyte THP-1 and MDA-MB-231 cells. In addition, B-cell, mTh17, monocyte, neutrophil, macrophage, A549, and NHLF (normal human lung fibroblast) migration have been validated (data not shown). An IncuCyte migration panel has been launched at BMS to support disease research in immunology, oncology, immuno-oncology, cardiovascular, and fibrosis.
The automated IncuCyte chemotaxis platform facilitated the incorporation of quantitative studies of cell migration behavior into a routine drug discovery screening workflow for SAR/SLR (structure–liability relationship) support of multiple drug discovery programs, which enabled lead optimization screening capability.
Footnotes
Acknowledgements
The authors would like to acknowledge Daniel Appledorn, Maria Tikhonenko, Courtney Shin, and Lindy Oclair at Essen BioScience for their help in setting up the IncuCyte chemotaxis module at BMS. We are appreciative of the contribution of Rob Hutchins, Scott Manolopoulos, and Shawn Silverstein for the creation of compound plates for experimentation. In addition, we recognize the efforts of Kristina Tess and Khushbu Thumar for the cell culture support. We thank Jenny Xie, Murali Gururajan, John Hynes, Chris Kitson, Maria Mauricio, Sivakumar Pitchumani, Susan Wee, Gretchen Schroeder, Neelam Srivastava, and the BMS Immunology, Oncology & Fibrosis Discovery Teams for supplying compounds for this study. Also, we would like to express our gratitude to Daniel Maloney, Yi Fan, and Mark Fereshteh for their support during this validation effort.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
