Abstract
Textile production involves using various chemicals that may remain in clothing and pose health risks. Alkylphenol ethoxylates are non-ionic surfactants which may be involved in various textile printing and dyeing auxiliaries due to their excellent emulsification, wetting and decontamination abilities. This study aimed to determine the dermal exposure doses and health risks of alkylphenol ethoxylates in 10 unwashed textile samples for infants, toddlers and pregnant women. The concentrations of alkylphenol ethoxylates and alkylphenols (APs) were determined by liquid chromatography–mass spectrometry (LC-MS/MS). The total concentrations of alkylphenol ethoxylates (nonylphenol ethoxylates and octylphenol ethoxylates) in 10 textile samples were 113.21–1431.18 mg/kg, among which nonylphenol ethoxylates with ethoxy groups of 2–10 were the dominant chemicals. The highest dermal exposure dose was found in the toddler group, which was 1.27 and 4.14 times higher than those in the infant and pregnant women groups. When the maximum concentration was used for risk assessment based on tolerable daily intake, the hazard indices (HI) for toddlers and infants were estimated to be 10.62 and 8.37, which may pose a higher risk to human health. Our assessment was specifically focused on new garments before washing. The garments containing alkylphenol ethoxylates can be washed to significantly reduce the exposure risk. The results of our study offer important information on human exposure to alkylphenol ethoxylates in textiles and clothing and provide a scientific basis for planning risk management measures for alkylphenol ethoxylates.
Introduction
There are approximately 16,000 textile chemicals worldwide, with an annual production of 6.4 million tons. 1 According to current studies, the required amount of chemicals per kilogram of textiles and clothing throughout their lifecycle is 1.5–6.9 kg. 2 Chemicals adhering to textiles and clothing may be dislodged during daily washing and wearing, and enter the human body through three pathways, namely, inhalation, ingestion and dermal absorption, thereby endangering human health. Moreover, when these chemicals enter the environment, where they may inhibit the metabolism of aquatic organisms and destroy the self-purification ability of water bodies, thus affecting the ecological balance.3–6 Accordingly, the United Nations Environment Programme has identified the overuse of chemicals and the release of chemical pollutants as one of the major environmental issues affecting human survival and development. 7
Alkylphenol ethoxylates (APEOs) are non-ionic surfactants, a mixture of homologous compounds with different degrees of polymerization and multiple isomers. They may be used in textile printing and dyeing process due to their excellent emulsifying, wetting, permeability, washing and decontaminating effects.8,9 The most common APEOs on the market are nonylphenol ethoxylates (NPEOs), which account for more than 80% of the total, followed by octylphenol ethoxylates (OPEOs), which account for more than 15% of the total, and dodecyl ethoxylates (DPEOs) and nonylphenol ethoxylates (DNPEOs), each of which account for about 1% of the total.10,11 The degree of polymerization of the product is characterized by the average number of ethoxy groups (EO). APEOs with different EO chain lengths are used for different purposes. For NPEOs, when the EO is 1–3, they can be used as oil-soluble emulsifiers. When the EO is 3–6, it has a certain wetting and permeation ability towards oil. When the EO is greater than 7, it becomes soluble in water and slightly insoluble in oil, making oil-in-water emulsifiers. When the EO continues to increase to 9–10, it exhibits good emulsification and wetting functions, and has strong washing and decontamination ability. As the EO continues to increase, the surface tension gradually increases while the wetting ability decreases. When the EO is greater than 15, it can be used as an emulsifier and detergent in strong electrolyte solutions. 12 The above discussion indicates that the differences in EO endow NPEOs with complex and diverse properties to meet various needs.
The hazards of APEOs to ecosystems and organisms mainly involve toxicity, biodegradability and environmental hormones. 9 Previous studies have shown that APEOs are highly toxic to aquatic organisms, while NPEOs are close to being highly toxic to fish.10,11 Compared to other non-ionic surfactants, APEOs are deemed the least biodegradable. The European Union requires environmentally friendly surfactants to have an initial biodegradation rate greater than 80%, while the degradation rates of APEOs are only required to be 0–9%. 13 APEOs also have a similar effect to estrogen. Biodegradation products of APEOs, such as nonylphenol (NP), octylphenol (OP), OPEO1 and NPEO1, are also environmental hormones that affect normal hormone secretion levels in the human body. When APEOs enter the human body, they produce estrogen-like effects that result in diminished male characteristics and reduced fertility.14,15 In addition, APEOs undergo side reactions during the production and polymerization process, producing ethane and ethylene oxide, which are recognized as carcinogens. 10 APEOs have a strong irritant effect on the eyes and skin of the human body, which can cause discomfort and even related inflammation. 10 In 1993, Council Directive 793/93/EC was issued requiring the assessment of the environmental risks of NP and NPEOs. The US Environmental Protection Agency (US EPA) also added NP and OP to its list of 70 environmental hormones in 1997. In 2001, the Scientific Committee on Health, Environmental, and Emerging Risks (SCHEER) recognized the existence of a risk and requested that it be reduced. In 2003, the European Parliament and the Council of the European Union adopted a resolution (2003/53/EC) restricting the use and release of NPEOs. The Annex XIV of Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH) in 2017 has also formally included OPEOs in the range of substances that require authorization for use. The EU2016/26 regulation amended the REACH Annex XVII restriction on nonylphenol ethoxylates (NPEOs) and stipulated that from 2021, washable textiles containing NPEOs at a limit of ≥0.01% would not be allowed to be placed on the market. Zero Discharge of Hazardous Chemicals (ZDHC) Manufacturing Restricted Substances List (MRSL) V3.1 listed two alkylphenols (APs (NP and OP)) and their respective APEOs (NPEOs and OPEOs) having formulation limits of Sum = 100 mg/kg and Sum = 250 mg/kg, respectively. Standard 100 by OEKO-TEX®, one of the world’s most recognized eco-textile labels for harmful substances, is updated annually. The 2024 version of the OEKO-TEX® STANDARD 100 reduces the limit to 10.0 mg/kg for OP and NP and 100.0 mg/kg for NP, OP, NPEOs and OPEOs.
APEOs in textiles have been listed as restricted substances in international regulations for decades. APEOs could be intentionally or unintentionally added during various processes in textile manufacturing, particularly in the dyeing and printing process. 9 Although China, the European Union, the United States and other countries have stipulated that APEOs should not be used in detergents, a small number of companies are still violating the law by using APEOs in their laundry products. In addition, some textile manufacturers are unaware of whether their chemical products contain APEOs. Due to their ecological hazards, many scholars have studied the toxic effects of APEOs in the environment, but fewer studies have been conducted on textiles.16–20 Cai and Wang 15 conducted a study on the content and detection value of APEOs in textiles and the results showed that the detection frequency of NPEOs was 30% and that of OPEOs was 1%, which indicated that the main component of APEOs detected in textiles was NPEOs. In addition, in the textile samples in which APEOs were detected, most of the detected values were concentrated in the range of 0.5–100 ppm, and the detected values of APEOs in a very small number of textile samples were greater than 1000 ppm. Fan et al. 13 conducted a statistical analysis of the results of down samples sent by enterprises and the results showed that the detection frequencies of NPEOs and NP were 60.08% and 0.20%, respectively, which also indicated that NPEOs were the main component of APEOs. Moreover, the percentage of samples that failed was 9.76%, with 2.76% of samples having a detection value higher than 1000 mg/kg. Clothing worn all day may lead to skin absorption of APEOs through direct skin contact; however, the possible pathways of exposure to these compounds have not been fully studied, especially when considering vulnerable groups in the population.21,22
Human exposure to APEOs is a public health concern. However, more information is needed on concentrations of APEOs and APs in textile products and on dermal exposure assessment. Considering the potential effects on human health, risk assessment of APEOs in clothing is necessary to ensure the safety of consumers, mainly vulnerable groups. In this article, we aim to determine the concentrations of APEOs and APs in textiles, assessing the dose and health risks of dermal exposure to these chemicals in pregnant women, newborns and toddlers. The novelty of our study offers important information on human exposure to APEOs in textiles and clothing and provides a scientific basis for planning risk management measures for APEOs.
Materials and Methods
Sample Collection
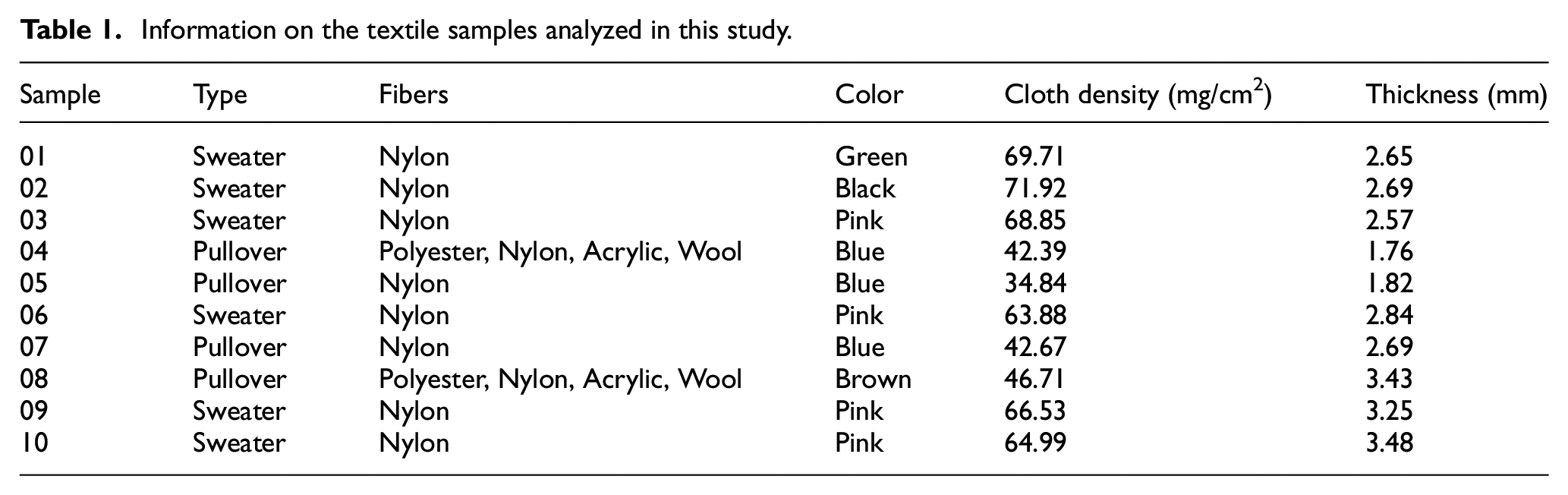
A total of 10 unwashed textile samples were collected, mainly in woven and knitted fabrics, and the relevant information on the samples is shown in Table 1. Prior to analysis, all samples were sealed in plastic bags and stored at room temperature away from light. To minimize inhomogeneous contamination of textile samples caused by processing, three different parts of each sample were taken randomly, and the textile sample was cut into small pieces of approximately 5 mm × 5 mm and then mixed homogeneously.
Information on the textile samples analyzed in this study.
Chemicals and Reagents
Alkylphenols and alkylphenol ethoxylates standards—nonylphenol ethoxylates (NPEOs, CAS 9016-45-9), octylphenol ethoxylates (OPEOs, CAS 9002-93-1), nonylphenol (NP, CAS 620-92-8) and octylphenol (OP, CAS 77-40-7)—were purchased from Anpel (Shanghai, China). Methanol was purchased from Xihua (Guangdong, China). The stock and working solutions were prepared in HPLC-grade methanol and stored at 4°C to avoid degradation.
APEOs and APs Chemical Analysis
The sample preparation methodology for determining APEOs and APs in textiles was adapted from Textiles—method for the detection and determination of alkylphenol ethoxylates (ISO 18254-1: 2016). Briefly, approximately 1.0 g of each textile sample was weighed and placed in 50 mL glass centrifuge tubes. The extraction was performed by adding 30 mL of methanol. After sonication at 70°C for 60 min, the solvent was collected and filtered through a 0.22 m nylon filter and transferred to another glass tube.
APEOs and APs were quantified using liquid chromatography–mass spectrometry (LC-MS/MS) equipped with a C18 chromatography column (SCIEX, USA). The temperature of the chromatographic column is 40°C, and the flow rate is 250 L/min. Both atomization gas and collision gas are high-purity nitrogen.
Quality Assurance and Quality Control
All reagents used in the experiment are guaranteed reagents. The overall cleanliness of the laboratory meets the requirements of trace analysis. The experimental containers were soaked in 25% nitric acid solution for 24 h, then rinsed with ultra-pure water, and dried in an electric thermostatic drying oven before use. The concentrations of APEOs and APs were quantified using an external calibration method based on the peak areas of the composite standards. The linearity was tested at five different concentration levels. The correlation coefficients of the quantification curves were all greater than 0.9995. The accuracy of the instrumental method was checked using method blanks and spiked blanks as well as matrix-spiked samples. The recoveries of the target chemicals in the spiked matrices were 84.7–103.4%.
Dermal Exposure and Human Health Risk Assessment
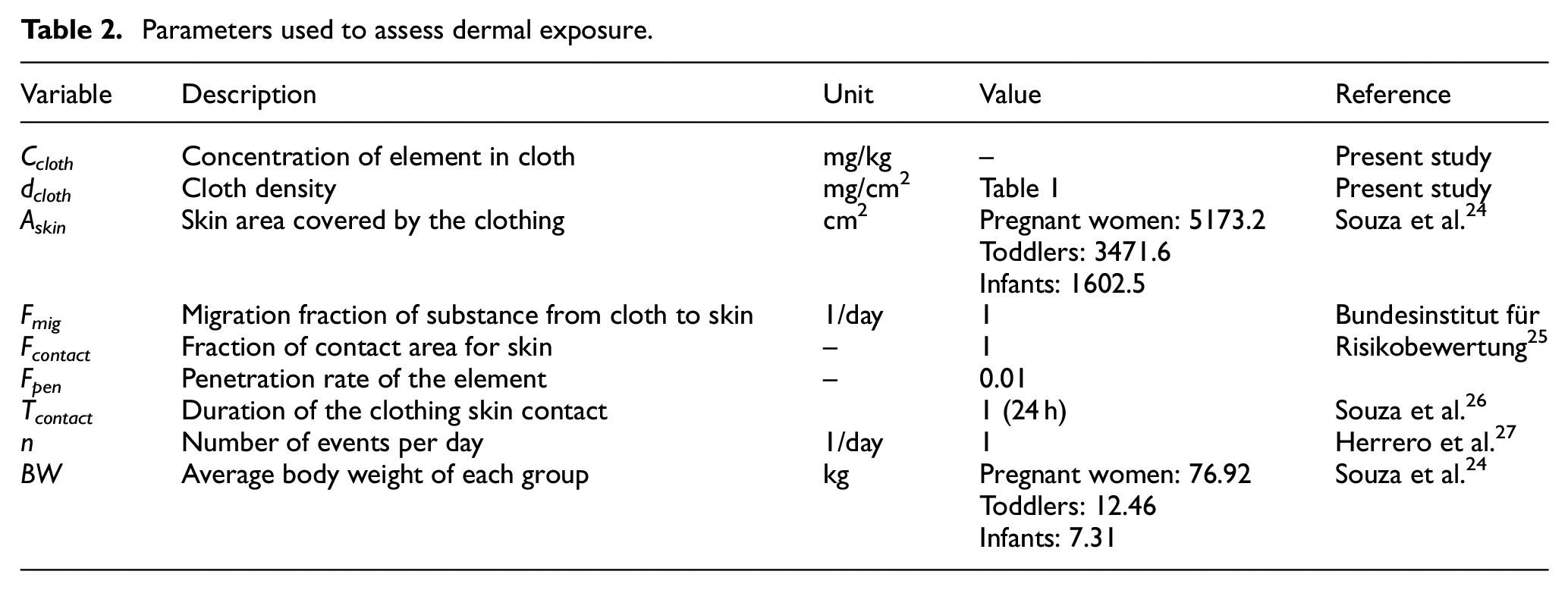
The concentrations of APEOs and APs in textiles can be used to estimate human exposure and health risks. In this study, only dermal contact was considered as a route of exposure to APEOs and APs, which come from clothing–skin contact. Equation (1) was used to calculate the dermal absorption. Based on the European Chemical Agency (ECHA) guidance on information requirements and chemical safety assessment, 23 NPEOs and OPEOs were selected for dermal exposure and health risk assessment based on the detection of APs and APEOs in the samples. Exposures were estimated for pregnant women, infants (12 months) and toddlers (12–36 months):

where Expderm corresponds to dermal exposure (mg/kg-day), Ccloth is the concentration of APEOs in clothing (mg/kg), 10−6 is a conversion factor (kg/mg), dcloth is the density of the clothing (mg/cm2), Askin is the skin area covered by the clothing (cm2), Fmig is the fraction of substance migrating to the skin per day (1/day), Fcontact is the fraction of contact area for skin (dimensionless), Fpen is the penetration rate of the element (dimensionless), Tcontact is the duration of the clothing skin contact (day), n is the number of events per day (1/day), and BW is the body weight (kg). The dermal exposure parameters are summarized in Table 2.
Parameters used to assess dermal exposure.
The risk assessment describes the nature and scope of the population health risks, and presents uncertainty, including non-carcinogenic and carcinogenic risks. Based on sample testing and toxicological data, we analyzed only the non-carcinogenic risk of APEOs. Non-carcinogenic risks were assessed using a hazard quotient (HQ), defined as the quotient between the predicted exposure and the corresponding Reference limit value, calculated using the following equations:
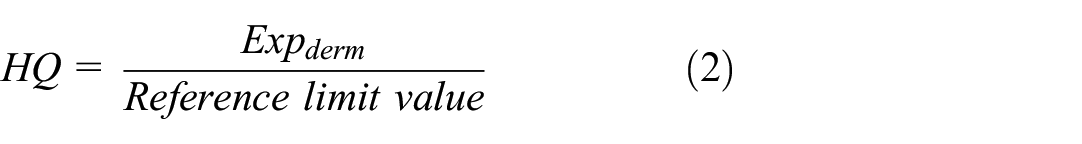
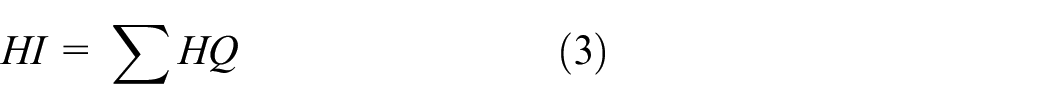
The reference limit value is tolerable daily intake (TDI) established by the Danish Veterinary and Food Administration (DVFA) or the reference dose (RfD) recommended by the US EPA. Due to the lack of RfD values for certain chemicals in the relevant databases, a Conditional Toxicity Value Predictor (CTV) based on the quantitative structure–activity relationship (QSAR) model was used to predict the reference dose of substances.28,29
Results and Discussion
Occurrence of APEOs and APs in Clothing
The concentrations of APEOs and APs in commercial textiles are given in Table 3. APs were not detected in all samples. The total concentrations of APEOs (NPEOs and OPEOs) were 113.21–1431.18 mg/kg. The highest median concentration was observed for NPEO4–6, followed by NPEO9–10 and NPEO7–8. The maximum concentration of NPEO2–3 was found to be 214.55 mg/kg, second only to NPEO4–6. However, the occurrence of APEOs might depend on the use of APEOs-based surfactants, the intensity of the later rinsing and any substantivity that an APEOs might have for a given textile fiber.
Concentrations (mg/kg) of APEOs and APs in 10 textile samples in this study.
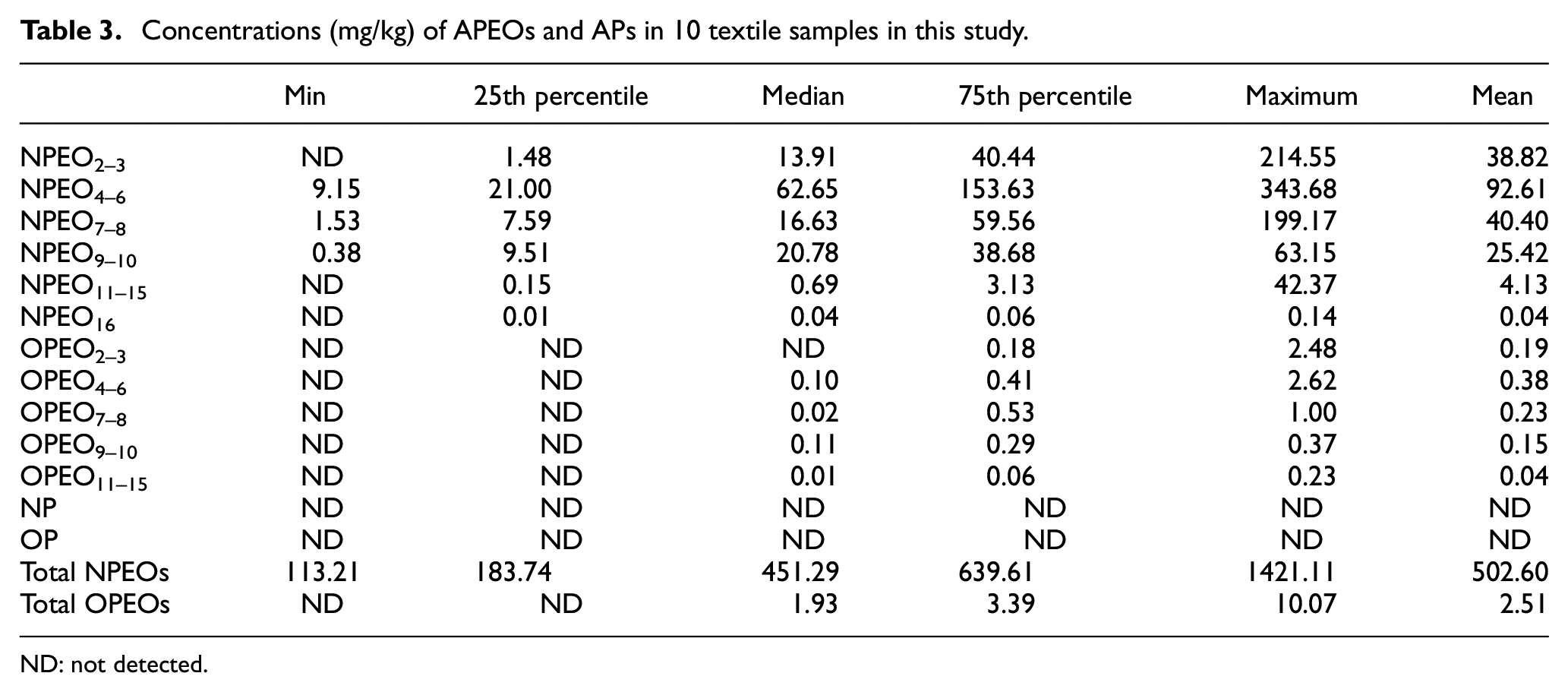
ND: not detected.
Figure 1(a) and (b) depicts the profiles of APEOs contamination in the sampled clothing. NPEO4–6, NPEO9–10, NPEO7–8 and NPEO2–3 were the most abundant chemicals, representing medians of 54.51%, 18.08%, 14.47% and 12.10% of the total concentrations, respectively. Other APEOs contributed less than 1% to the total concentrations. In addition, based on the mean concentrations, NPEO4–6, NPEO7–8, NPEO2–3 and NPEO9–10 were also the most abundant chemicals, contributing 45.75%, 19.96%, 19.18% and 12.56% of the total concentrations, respectively. NPEO11–15 accounted for 2.04% of the total concentration, while other APEOs accounted for a total of 0.51%. The difference in ethoxy groups endows APEOs with complex and varied properties to meet various needs. Since these compounds can enter the production process at different stages, it is not possible to identify a single source of contamination.
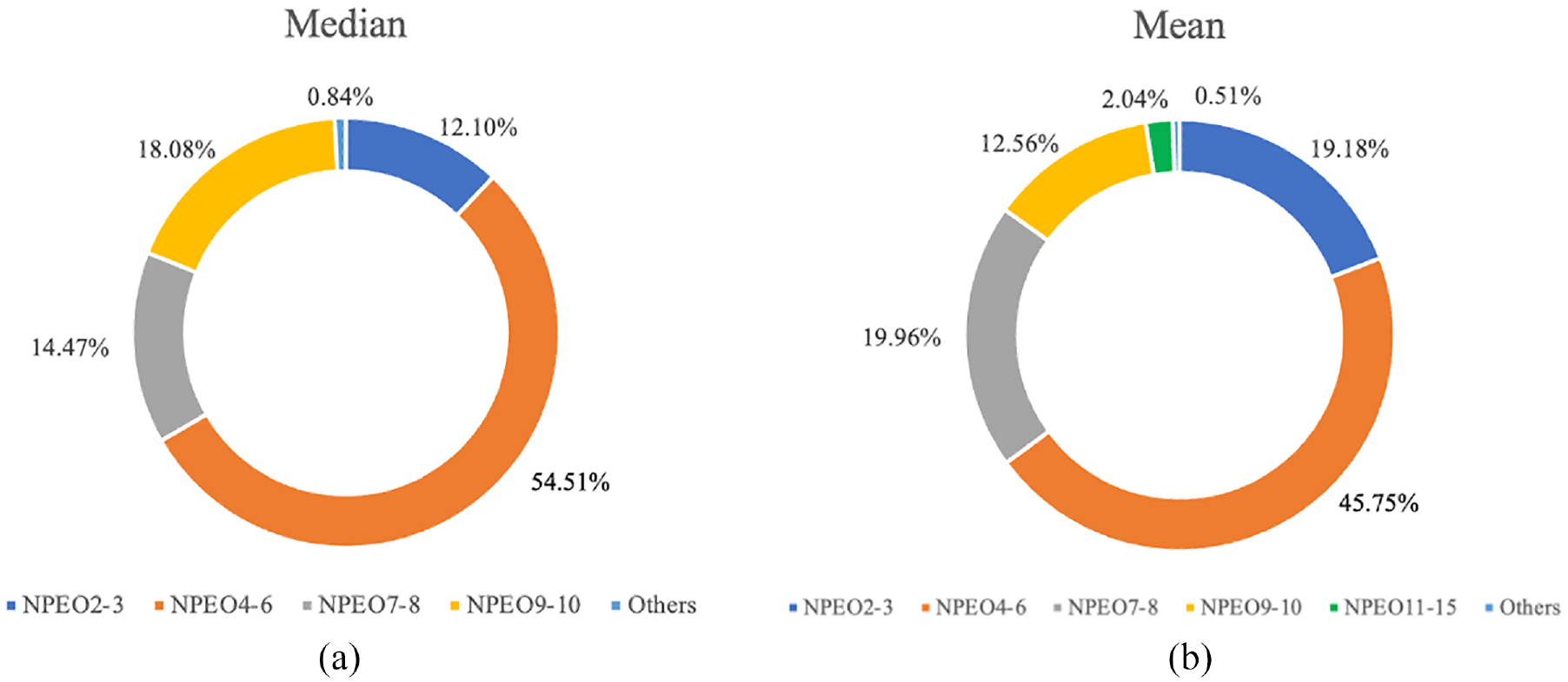
Composition profile of APEOs in 10 textile samples, based on the median concentrations (a), and the mean concentrations (b).
Exposure and Risk Assessment
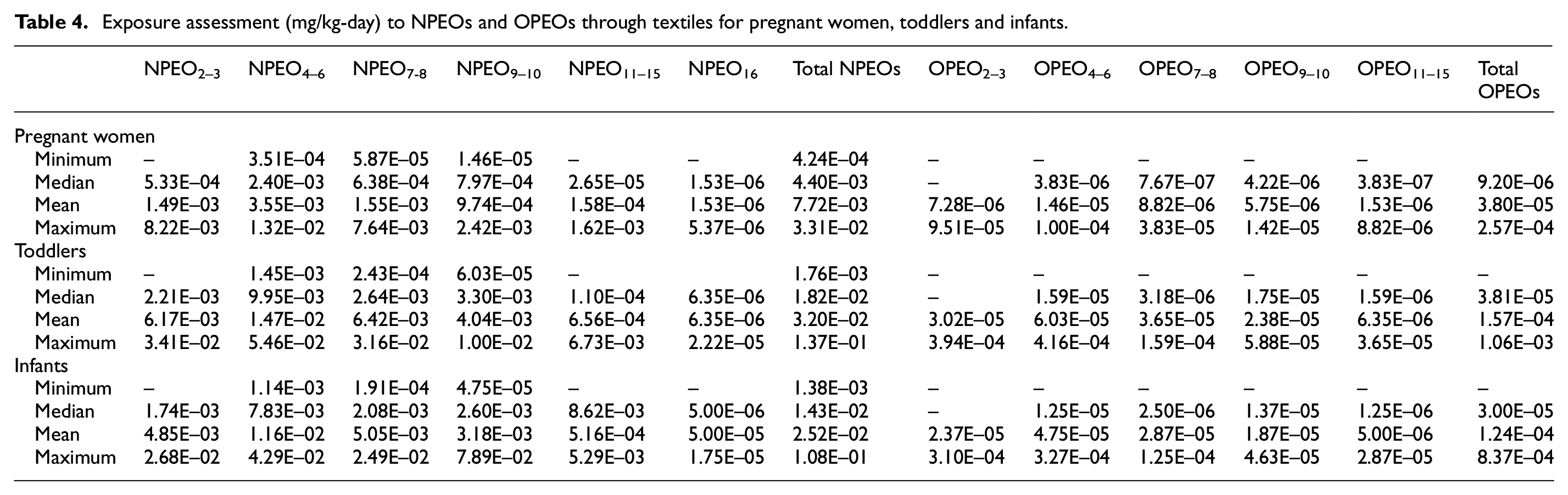
Concentrations of APEOs measured in textiles were used to estimate human dermal exposure and the associated health risks for the three susceptible groups (pregnant women, toddlers and newborns), considering the duration of the clothing skin contact (24 h) and the total amount of clothes used (instead of considering only one item of clothing). The doses (mg/kg-day) of exposure to NPEOs and OPEOs through textiles for pregnant women, infants and toddlers are listed in Table 4. The total Expderm of APEOs for the three susceptible groups from clothing ranged from 4.24E–04 mg/kg-day to 3.34E–02 mg/kg-day, from 1.76E–03 mg/kg-day to 1.38E–01 mg/kg-day and from 1.38E–03 mg/kg-day to 1.09E–01 mg/kg-day, respectively. The highest dermal exposure dose was found in the toddler group, which was 1.27 and 4.14 times higher than those for the infant and pregnant women groups, respectively. It is worth noting that toddlers have a larger skin area in relation to their body weight, which might result in a greater risk of dermal exposure to chemicals. Among all APEOs, NPEO4–6 had the highest contribution for toddler exposure to APEOs from textiles, with a median value of 9.95E–03 mg/kg-day (mean 1.47E–02 mg/kg-day), followed by NPEO9–10 and NPEO7–8 (3.30E–03 mg/kg-day and 2.64E–03 mg/kg-day, respectively).
Exposure assessment (mg/kg-day) to NPEOs and OPEOs through textiles for pregnant women, toddlers and infants.
We hypothesized that these chemicals have the same mechanism of action for the cumulative exposure risk assessment. In this study, the cumulative risk was applied to estimate the susceptible groups’ exposure to APEOs based on the HQ. Figure 2 shows the hazard index (HI) of dermal exposure to NPEOs and OPEOs for the three vulnerable groups based on two different reference limit values. Only NPEOs had the TDI value required to estimate the HQ, which was assumed to be the same as the OPEOs. Considering the lack of RfD and TDI values for certain chemicals, some new approaches should be considered shortly by regulatory agencies worldwide. QSAR models, surrogate approaches and read-across approaches can be useful in predicting the toxicity of chemicals for which data are lacking, thereby reducing the need for animal experiments in safety testing. To capitulate in short, these modern approaches can help prioritize chemicals for which dose-response data are most urgently needed.30,31
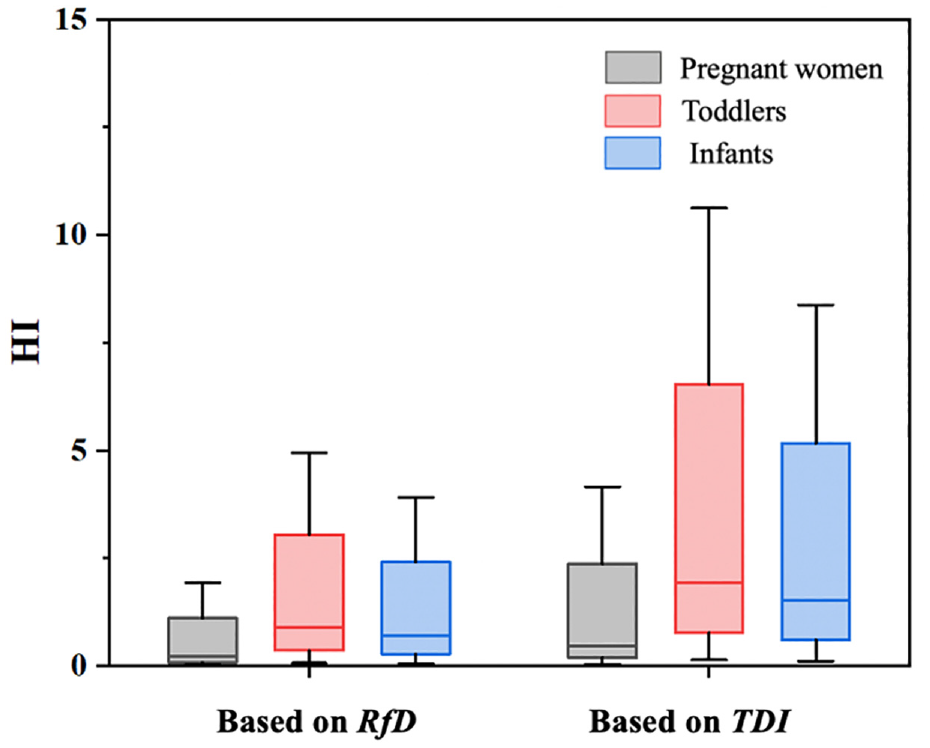
Non-cancer risks of dermal exposure to NPEOs and OPEOs through textiles based on RfD and TDI. The horizontal lines represent the 25th percentiles −1.5IQR, 50th, and 75th percentiles +1.5IQR, and the boxes represent the 25th and 75th percentiles.
Chemicals are hazardous to human health when the HI value exceeds 1. As shown in Figure 2, the median HI values for pregnant women, toddlers and infants based on RfD were 0.22, 0.90 and 0.70, respectively. In addition, the median HI values for the three vulnerable groups were 0.47, 1.94 and 1.53, respectively, based on TDI. When the maximum concentration was used for risk assessment, based on TDI, the HI values for toddlers and infants were estimated to be 10.62 and 8.37, respectively, which may pose a higher risk to human health. Therefore, the non-carcinogenic risk of APEOs should be taken seriously.
Furthermore, it is important to note that only dermal exposure to APEOs was considered in the assessment of health risks in this study. However, the role of other potential routes of exposure should not be neglected. Other recent studies have pointed to the important role of textiles in airborne contaminants, with dermal exposure also being of particular importance. 32 Moreover, for the toddlers and newborns, the exposures through oral sucking of fabrics can be substantial in certain circumstances and is likely to be the dominant route of exposure to chemicals. 32 Inhalation may also be an important route of exposure in some cases. However, available information on the exposure doses by these routes is still limited, making accurate comparisons of these routes difficult.
Conclusions
Based on the scientific literature, this is the first study focusing on the assessment of the health risks of APEOs in textiles for pregnant women, newborns and toddlers. Although the results of the study are preliminary and rudimental, it provides substantial evidence of the occurrence of APEOs in textiles and clothing, aligning with important information for the risk assessment of APEOs. In this study, the health risks of 10 unwashed textile samples were evaluated based on the human exposure model, and the following conclusions were drawn: NPEOs are likely to be the major contaminants in clothing containing APEOs. Considering the large ratio of skin area to body weight, young children appeared to be at greater risk of exposure compared to pregnant women and infants. The non-carcinogenic risk to infants and young children from NPEOs in multiple samples was close to the risk threshold, which should be taken seriously. APEOs are increasingly attracting widespread attention in the textile and other industries due to their ecological hazards. Various alternatives have been developed, such as primary alcohol ethoxylates (AEOs), straight-chain dodecylbenzene (LAB) and alkyl polyglucosides (APGs), and the use of APEOs is decreasing. It is important to recognize that our study discusses the exposure risk of new clothes before they are laundered, and that most of the APEOs present will be lost as the clothing is laundered. Therefore, we recommend that people wash their clothing before wearing new ones, thereby substantially reducing the exposure risk of APEOs. In general, this study presents important information that textiles are a source of exposure to APEOs to people. These findings indicate the need for further studies on the sources and exposure levels of chemicals present in textiles. The regulatory authorities should join up with various industry associations to expedite the development of mandatory standards that strictly limit APEOs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors are grateful to the Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ24E030006, Science Foundation of Zhejiang Sci-Tech University (ZSTU) under Grant No. 22202009-Y.
