Abstract
Many flame retardants (FRs) are brominated, and many of them are considered toxic, persistent and/or bio accumulative. A switch to non-halogenated alternatives for textile finishing is considered beneficial, but there is little knowledge about their environmental impact, their health risks during the production process and those in their final use.
A conventional and an alternative FR (based on dialkyl phosphono carboxylic acid amine and ammonium sulfamate; urea and organophosphonate PO(OH)2-R-PO(OH)2, respectively) were applied to cotton fabrics that could be in touch with the skin in their final use. Burning and thermal behaviors and dermal toxicity when exposed to skin rubbing are evaluated. Fabrics treated show that both FR products act as FRs. The increase in the thermal stability of the tissue that causes an increase in the decomposition temperature is a relevant effect associated with the application of the conventional FR in the tissue, while the most important effect of the alternative FR lies in the increase of the residue at 600°C, associated with the generation of fewer volatiles, which has an impact on the intensity of combustion. The study of the dermal toxic potential of FR-treated cotton bed sheets showed that the two tested products do not penetrate into the skin when applied under the conditions used and were safe when in contact with the skin. Moreover, the alternative FR seems to be safer when in contact with the skin considering also its higher concentration in the textile.
Keywords
Polymers and textiles can be found in every facet of human everyday life particularly as household items and apparels, even though most of them are inherently flammable. Polymers can be categorized as naturally occurring polymers (cotton, silk, wood, wool, etc.) or synthetic (polyester, polypropylene, polyethylene, etc.). Many materials used in building interiors are flammable, such as curtains, upholstered furniture, bed items, cushions, etc. Cotton bed linens have been widely used in hospitals, residences, etc., due to their excellent properties. 1 However, their high flammability is a problem in some cases. Cotton is a cellulosic fiber, composed of cellulose as the main polymer (85–90%). Its limiting oxygen index (LOI) is only about 18%, which makes it easy to burn, and once ignited, the flame will spread rapidly, so it is very easy to cause a fire. 2 Flame retardant (FR) applications play a crucial protective role in reducing fire risk by inhibiting ignition, or once ignited, suppressing the spread of fire and/or delaying the time to flashover, and hence enabling people to have sufficient time to escape or to be rescued from fire hazards. In the case of textiles, FRs are applied to make them self-extinguishing or to reduce the flammability. The phenomenon involves many factors, such as ignitability, ease of extinction, flammability of the volatile products generated, smoke obstruction, flame spread, rate of heat release, smoke toxicity, etc.
There are some strategies to impart flame retardancy to polymers and/or textiles, namely polymer composites, polymer chain modification or polymer coating (surface finishing), which is the one most used for cotton. 3 There are many classification methods for FRs, the most common of which is divided by FR elements, such as halogen, phosphorus nitrogen, silicon, metalhydroxide, etc. 2 However, FRs are often categorized according to their mechanism of action. FRs can restrict flame spread via a physical action (by fuel dilution, endothermic cooling or the nanoparticle physical barrier effect) or via chemical action (by flame inhibition, charring, intumescence or melt dripping). Some FRs exhibit almost only physical actions; however, chemical mechanisms are often accompanied with several physical actions to retard fire spread or stop the combustion process. 3
Conventional FRs are, for example, organohalogen compounds (chloroparaffins, polybrominated diphenyl ether/ethane and bromobisphenols) or formaldehyde-releasing processes such as Pyrovatex®, which are very effective as FRs. 4 Several of these halogenated compounds have been phased out in the past due to their persistence or toxicity. It is suggested to avoid the use of non-restricted halogenated and formaldehyde-releasing FR materials as this might lead to regrettable substitution. However, users of FRs want to have a clear view of the offered alternatives. Therefore, research on alternatives, such as those based on phosphorus and nitrogen, are finding acceptance on the market.5 –9
Phosphorus-containing FRs have gradually replaced halogen-based FRs because of their high FR efficiency, low toxicity and being environmentally friendly. They cover a wide range of inorganic and organic compounds and include both reactive products, which are chemically bound to the polymer material, as well as additive products, which are integrated into the material by physical mixing only. 10 They have a broad range of applications and a good fire safety performance. Phosphorus-based FRs mainly use the condensed phase flame. The most important phosphorus-containing FRs are phosphate esters, phosphonates and phosphinates. 11 For example, N-methylol dimethylphosphonopropionamide (MDPA, Pyrovatex® process) is among the most efficient FRs used for cotton. To improve the FR performance and durability, MDPA needs a crosslinking agent such as trimethylol melamine (TMM). 12 As formaldehyde is a starting product to produce TMM, high levels of formaldehyde are introduced, so other eco-friendly finishes for cotton are being studied.13,14
Nitrogen compounds are a small but rapidly growing group of FRs that are in the focus of public interest concerning environmentally friendly FRs. Their main common advantages are their low toxicity, their solid state and, in the case of fire, the absence of dioxin and halogen acids as well as their low evolution of smoke. 9 However, N-based FRs alone have a lower intrinsic fire retardant performance.
Nitrogen compounds are mostly used in combination with phosphorous-based FRs due to their synergistic effect. They possess the advantages of both FRs, playing a FR role both in the condensed phase and gas phase; they not only have high FR efficiency, low toxicity and less smoke emission, but are also safe and environmentally friendly. 2 Several authors have recently report new strategies in this line.15,16
In this study a conventional FR and an alternative phosphorous-nitrogen-based FR have been applied to cotton fabrics that could be in touch with the skin in its final use (bed linen). Burning and thermal behaviors and dermal toxicity when exposed to skin rubbing are evaluated.
Specifically, a formulation based on dialkyl phosphono carboxylic acid amine (DPCAA) is used as a conventional FR that has good performance and high washing fastness, although it leads to the release of formaldehyde.17,18 Thermal degradation and the release of water produced by the action of heat leads to the formation of phosphoric acid that polymerizes, forming a layer of polyphosphoric acid that, by carbonization, becomes a glassy layer, thus inhibiting “pyrolysis” (decomposition and flammable gases release), which is necessary to fuel the combustion. In this way it blocks the transfer of heat to the unburned areas of the material, slowing down the spread of fire.
As an alternative FR, a compound with ammonium sulfamate, urea and organophosphonate PO(OH)2-R-PO(OH)2 is assessed, as this is one of the few commercially available treatments identified that could make cotton and blends with up to 50% polyester flame retardant. The active components react with cotton, leading to sulfation and phosphorylation of the cellulosic substrate. 19 The phosphoric acid acts as a sulfation catalyst and a phosphorylating agent is capable of reacting with any cellulose substrate that has reactive hydroxyl groups, maintaining good mechanical properties of the treated cotton. Upon ignition, sulfur releases sulfuric acid, allowing the cellulose material to degrade at temperatures below the ignition temperature, nitrogen forms non-combustible gases and phosphorus inhibits the formation of levoglucosan during heating and also forms the compound P2O5, which provides a barrier effect and protects the underlying combustible material. It retards the decomposition of cotton and can release large amounts of ammonium, nitrogen and water vapor during combustion. 20
Most of the work on flammable fabrics focuses on the evaluation of the facility of ignition, the rate and extent of flame spread, the duration of flame propagation and heat release and the heat of combustion. 11 The LOI, also called the oxygen index (OI), denotes the minimum concentration (vol%) of O2 that will sustain flaming combustion. Simple ignition tests represent other usual approaches for assessing flammability. Flame spread and the cone calorimetry test are used as bench scale tests and the micro-combustion calorimeter (MCC) has recently been standardized and exploited for evaluating flammability polymers. 21 Further, actual end application (bed linen) fire tests have been performed: the most used EN ISO 12952-1 & 2 (cigarette and match-flame equivalent test) have been selected, together with the commonly used BS 7175 (crib 5).
The fabric samples treated with FRs were also characterized by thermal analysis techniques TGA (thermogravimetric analysis) and DSC (differential scanning calorimetry). 17 Since cotton sheets will be in contact with the skin, it is important to evaluate the possible dermal toxicity of FRs. Therefore, the dermal toxic potential of compounds applied to cotton has also been studied, using the percutaneous absorption technique in Franz cells according to the Organisation for Economic Co-operation and Development (OECD) methodology. 22 So far, no studies have been found on FR release from a textile to the skin when it is in contact with the textile. However, there is increasing evidence that suggests that dermal absorption is a potentially significant pathway in human exposure to FRs.23,24 In this work, the margin of safety (MoS) of the cotton FR-treated fabrics (bed linen) is calculated after percutaneous absorption tests were performed using an in vitro methodology with Franz diffusion cells.
Therefore, the main aim of this work is to determine the feasibility of the use of an alternative FR, compared to a conventional one in terms of flame retardancy, thermal assessment and dermal toxicity. This last property will be calculated using MoS as a risk characterization to demonstrate the non-toxic dermal use of the alternative FR in textile finishes.
Materials and methods
Flame retardant treatments
The fabrics (bed linen) treated with FRs are the following.
CO: 100% cotton fabric, non-treated (112 g/m2). CO-FR-conv: 100% cotton fabric of 112 g/m2 finished at the lab level by padding (1 bar, 5 rpm, wet pick-up 75%) with 500 g/l of a commercial formulation based on DPCAA, 20 g/l crosslinker (Quecodur DM 70, an etherified melamine formaldehyde resin), 20 g/l softener (Finistrol NVN) and 20 g/l of phosphoric acid 85%. The fabric is dried for 30 seconds at 110°C and cured for 3 minutes at 160°C. Afterwards, the fabric is rinsed with cold water, neutralized with 7 g/l sodium carbonate and 2 g/l detergent (Laventin LNB) at 60°C, treated with 2 g/l sodium carbonate and 5 g/l hydrogen peroxide at 60°C and rinsed with cold water. The followed procedure was set by the supplier of the FR formulation. The crosslinking reaction increased the dry weight of the fabric by 0.72%, on average. The industrial companies participating in the FLAREX project did not have the necessary equipment to safely apply formaldehyde-releasing formulations, hence the conventional FR was only applied at the laboratory scale (A4 scale). CO-FR-alter: 100% cotton fabric of 112 g/m2 finished by padding (1.5 bar, 5 rpm, wet pick-up 79%) with a water-based formulation (100%) based on ammonium sulfamate, urea and PO(OH)2-R-PO(OH)2 (proprietary formulation), followed by 200 seconds drying at 160°C. After drying, the fabric is washed with 10 wt% (NH3)2HPO4 and 10 wt% peracetic acid of 15% for 2 hours at 60°C (ratio 1:15–1:5), followed by a cold rinse. The followed procedure was set by the supplier of the FR formulation. The crosslinking reaction increased the dry weight of the fabric by 10.7%. This application was done at the industrial scale (ca. 100 running meters).
Flame retardancy efficiency
The burning behavior of all fabrics was evaluated according to EN ISO 12952-1 & 2:2010. 25 A mineral wool fiber pad was used as the testing substrate. The match-flame equivalent was placed on top of the bed sheet and cigarettes were placed on top and in between the top surface and second fold of the bed sheet. The BS 7175:1989 standard 26 was also executed. Also, in this case, a mineral wool fiber pad was used as the testing substrate. The crib 5 was positioned above the test specimens. Prior to all fire test, the fabrics were washed five times at 60°C following EN ISO 6330:2000 + A1:2008 and ISO 6330:2012 27 (washing method 2A, drying method E).
Physical properties
The tensile strength and elongation of the fabrics were assessed according to EN ISO 13934-1:2013 28 and the tear strength according to EN ISO 13937-2:2000. 29 Abrasion and pilling resistance were assessed following EN ISO 12947-2:2016 30 with a pressure of 12 kPa and EN ISO 12945-2:2000 31 with a pressure of 6.5 N/cm2, respectively. Air permeability was tested according to ISO 9237:1995 32 and water vapor permeability according to EN ISO 11092:2014. 33 Dimensional change was evaluated following EN ISO 5077:2008 34 after five washing cycles at 60°C (EN ISO 6330:2000 – method 2A) and subsequent tumble drying after each washing cycle.
The effect of different influences on the color fastness of the fabrics, being rubbing (EN ISO 105-X12:2016 35 ), light (ISO 105-B02:2014 36 ), perspiration (ISO 105-E04:2013 37 ) and domestic and commercial laundering (ISO 105-C08:2010 38 ), was evaluated.
Thermal analyses
The thermal analysis of the FR products themselves, FR-conv and FR-alter, was performed, as well as analysis of the untreated textiles and the textiles treated with these FRs.
The DSC analysis was conducted with a Mettler-Toledo DSC-823 apparatus using samples of about 3 mg placed in micro-punched aluminum pans for internal pressure control, in order to let the water and other volatiles be completely removed from the testing pans. 39 Tests were conducted from 30°C to 500°C at 10°C/min under 50 mL/min of N2 flux.
The TGA was performed in a Mettler-Toledo TGA/SDTA 840 using samples of about 10 mg placed in completely open aluminum pans. Tests were conducted from 25°C to 600°C at 10°C/min under a N2 flux of 60 mL/min and O2 flux of 60 mL/min. 39
Percutaneous absorption for dermal security
In order to calculate the MoS, firstly, permeation (percutaneous absorption) experiments were performed to determine the distribution of FR in the different layers of the skin after 24 hours of skin–textile contact, using Franz-type diffusion cells (3 mL, 1.86 cm2, Lara-Spiral, Courtenon, France) (Figure 1).40,41 For FR-conv, the compound DPCAA was analyzed and, for FR-alter, ammonium sulfamate (ASU) was quantified, evaluating unreacted ASU.
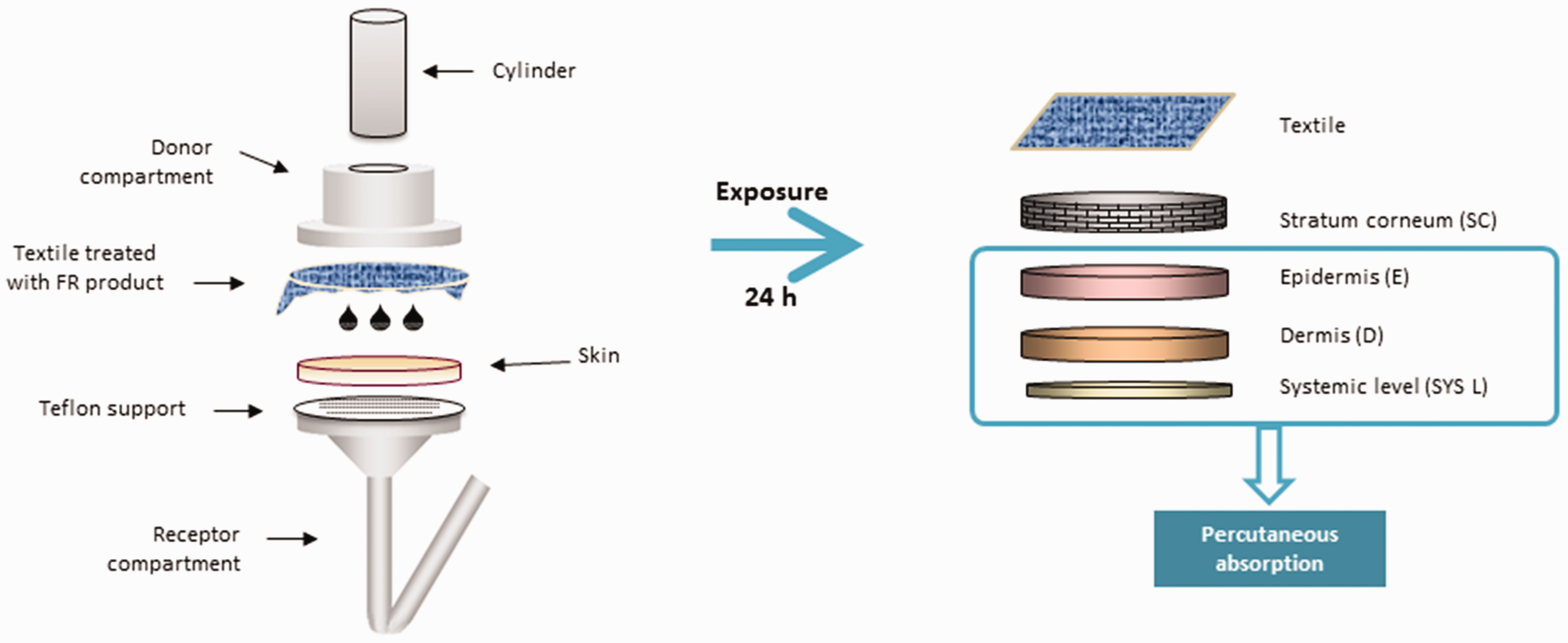
Diagram of percutaneous absorption with textiles. FR: flame retardant.
Porcine skin was used and dermatomized up to a thickness of 500 ± 50 µm (Dermatome GA630, Aesculap, Germany). A punch was used to obtain skin discs (2.5 cm internal diameter) that fit into the cell.
A magnet for magnetic agitation was inserted in the recipient and filled with receptor fluid. The receptor fluid was an aqueous solution in the ASU case and phosphate-buffered saline solution at pH 7.4 (containing 0.9% NaCl, 0.02% KCl and 0.8% phosphate buffer) in the DPCAA experiment.
The cells were placed in a thermostatic water bath ensuring a skin surface temperature of 32°C while undergoing magnetic agitation. The integrity of the skin samples was evaluated by measuring transepidermal water loss (TEWL) with a Tewameter TM300 (Courage & Khazaka, Cologne, Germany). A minimum of three diffusion cells were used for each experimental test. The FR-treated textiles were applied to the skin. A small amount of distilled water was added between the textile and the skin to ensure close contact. In addition, a constant pressure (125 g/cm2) was applied with a steel cylinder (Figure 1).
After 24 hours, the receptor fluid was collected and the different layers of skin were separated and the fabric was collected along with the top of the cell. The stratum corneum was obtained by eight strippings with adhesive strips (D-Squame, Cuderm Corporation, Dallas, USA). The epidermis was separated from the dermis after heating the skin to 80°C for a few seconds.
FR compounds were extracted from the different samples to be analyzed (textiles, SC tapes, epidermis and dermis) using the appropriate solvents. Fabric samples applied with FR-conv at the laboratory level, as well as an untreated fabric, were extracted with methanol (Merck, Darmstadt, Germany) and the alternative FR-treated sample, industrially applied, was extracted with p-hydroxybenzoic acid (5.8 mM, pH 9.4)/methanol (97:3), both supplied by Merck (Darmstadt, Germany). The receptor fluids were diluted and analyzed directly. Both compounds were analyzed by high-performance liquid chromatography with photodiode-array detection (HPLC-DAD) in order to obtain the analytical parameters. Analytical conditions and the results obtained are indicated in Table 1.
High-performance liquid chromatography (HPLC) analytical conditions and parameters

DPCAA: dialkyl phosphono carboxylic acid amine; HPLC-DAD: high-performance liquid chromatography with photodiode-array detection; LOD: limit of detection; LOQ: limit of quantification; RT: room temperature.
According to the “Guidelines for in vitro methods to assess the percutaneous absorption of cosmetic ingredients” adopted by the SCCNFP (Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers, European Commission),22,42 the amounts of the substance found in the epidermis (with the exception of the stratum corneum) and the dermis are considered as absorbed, as well as the amounts of the penetrated substance found in the receptor fluid. The amounts retained by the stratum corneum at the time of sampling (24 hours) are not considered to be absorbed percutaneously and therefore do not contribute to the systemic dose at that time.
The results are expressed as µg/cm2 and as a percentage of the dose of the compound FR applied. In all cases, the overall recovery is calculated and should be at least 100 ± 15%.
Once the percutaneous absorption (permeation) of each FR compound has been determined, the MoS can be calculated through Equation (1):

The NOAEL is obtained from a toxicological evaluation based on oral administration of the active ingredient in animals that can last up to 90 days; it is called subchronic toxicity. The unit of the critical dose is given in mg of compound/kg body weight (bw)/day.
The SED (mg/kg bw/day) is calculated as follows:

It is obvious that for a given NOAEL value, if the percutaneous absorption of a substance is low, the SED will also be low and consequently the range will be high and therefore appropriate. To achieve a very acceptable MoS, the substance must be non-toxic and have a high NOAEL value. In addition, this compound must not be easily absorbed through the skin to obtain a low SED value. Therefore, its MoS may be well above the threshold of 100. 42
Results and discussion
Cotton fabrics were treated by padding with two different types of FRs: one conventional FR, (DPCAA, CO-FR-conv), and another as a more ecological and non-hazardous alternative, ASU + urea + PO(OH)2-R-PO(OH)2 (ASU, CO-FR-alter). The weight gains of the fabrics after crosslinking of the FRs are respectively 0.72% owf (lab scale) and 10.7% owf (industrial scale). It appeared that impurities were present on the cotton fabric used at the lab scale. This explains the low weight gain after the FR treatment. The impurities were removed, influencing the weight of the fabric and potentially leading to low linkage of the DPCAA at the lab scale. Further optimization was deemed necessary, but was not possible to execute within the course of this project.
Flame retardant efficiency
After five domestic washings at 60°C (as required by EN ISO 12952-1 & 2 25 when no washing conditions are specified), the burning behavior was assessed and is presented in Table 2, numbers 1–3. The bed linen is not prone to ignition with a smoldering cigarette (EN ISO 12952-1). The match-flame equivalent test (EN ISO 12952-2) and crib 5 test (BS 7175) 26 were executed in twofold, and both flaming times are mentioned. The non-treated fabric passed the EN ISO 12952-2, but failed the more severe crib 5 test. As result of the low linkage of FR-conv, no significant difference in FR behavior was observed compared to the untreated fabric. Applying FR-alter on the cotton bed linen (CO-FR-alter) leads to an afterflame time of around 30 seconds, well below the limit of 120 seconds. Also, the crib 5 test was passed, showing a clear improvement of the FR behavior of the bed linen when treated with the alternative FR compared to the non-treated fabric. The crib 5 test has also been executed on a non-washed sample (Table 2, number 4), showing similar flame times. This shows that the alternative FR treatment is resistant to washing.
Results of the fire tests on the non-treated and treated cotton bed linen
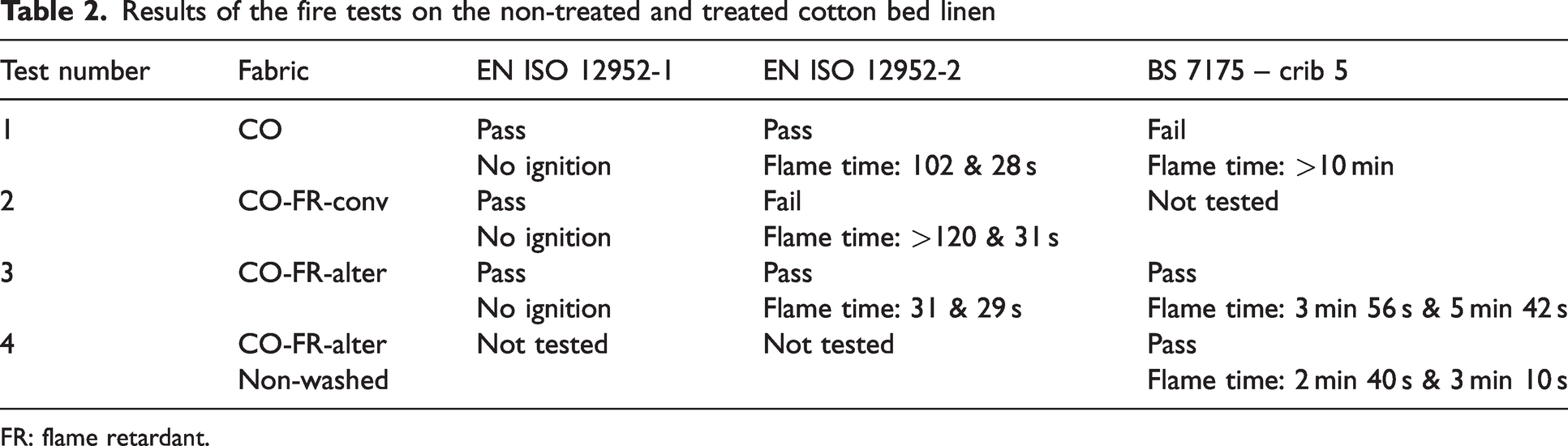
FR: flame retardant.
Physical properties
The physical properties of the cotton fabric treated with the alternative FR at the industrial scale were compared with the untreated fabric. Results of the performed physical tests are summarized in Table 3.
Results (averages) of the physical tests on the FR-alter treated and non-treated cotton bed linen
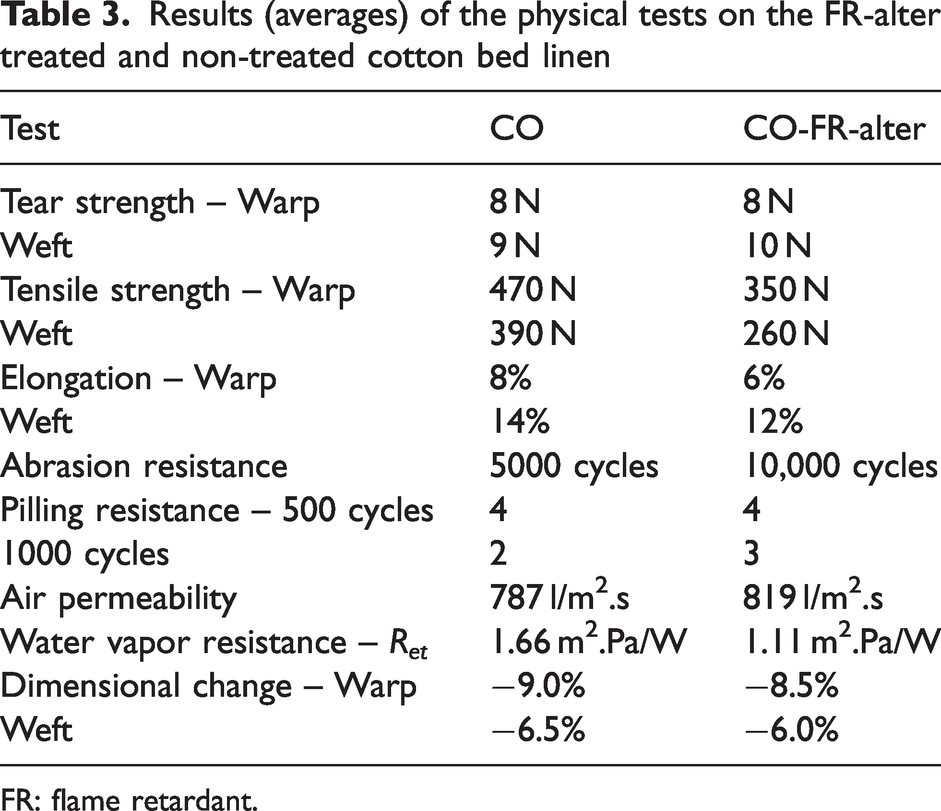
FR: flame retardant.
The FR-alter treatment of the cotton fabric did not significantly impact the tear strength, water vapor resistance, air permeability or dimensional change after five washings at 60°C. The abrasion and pilling resistance are slightly improved by the FR treatment. On the other hand, the tensile strength dropped by 25% in the warp direction and 33% in the weft direction. Also, the elongation of the fabric slightly decreased after the treatment with FR-alter.
Furthermore, color fastness were also assayed on the different non-treated and FR-treated fabrics. The FR treatment did not affect the color fastness to rubbing: before and after the FR treatment the cotton fabric scored 5. The color fastness to light of the untreated cotton fabric was significantly improved after the treatment: it increased from a rating of 2 to 6–7. The poor light resistance of the untreated fabric (it turns orange upon xenon irradiation) is caused by the presence of impurities. These impurities are removed during the FR treatment, leading to a vast improvement of the color fastness to light. No impact was seen from the FR treatment on the color fastness to domestic and commercial laundering: the rating of “change in color” remained at 4 and that of “staining on cotton and polyester” remained at 4–5. As bed linen will come into contact with human sweat, the color fastness to alkaline and acid perspiration was also assessed: the FR treatment did not affect the staining on cotton, prior to and after FR treatment the rating was 5. The change in color (both for contact with alkaline and acid solutions) was improved after the FR treatment: the rating increased from 4 (non-treated cotton) to 5 after the FR treatment. So, to conclude, the FR-alter treatment has no or a positive effect on the color fastnesses.
Thermal analyses
Treated cotton fabrics have been analyzed using the TGA and DSC techniques. TGA is a technique that evaluates the sample mass variation as a function of temperature or time while the sample is subjected to a heating program. The analysis of the fabrics treated with FR provides information on the formation of the final residue (%). DSC provides precise information on different thermal properties of the separate components from the different textiles and FR analyzed: melting and decomposition temperatures and fusion enthalpies, crystallinity and absorption indices and heat emission. In the case of fabrics treated with FR, it is interesting to know the onset temperature of decomposition, which should be as high as possible and the heat emission as low as possible.
Figure 2 shows the behavior of the original cotton fabric. The DSC curve shows a first endothermic peak at 122.9°C, which corresponds to the evaporation of the moisture contained in the fiber and, from 256.9°C, a notable exothermic peak begins, centered at 346°C, which is caused by the reactions of cellulose dehydration and degradation that yields to the formation of carbonaceous products, levoglucosan and highly inflammable products of low molecular weight, which contribute to the pyrolysis of the cellulose.43,44 The TGA curve shows the mass losses that occur with temperature:44,45 the initial loss corresponds to water evaporation, followed by the degradation stage of amorphous cellulose that impairs its mechanical properties, which is linked to a slight loss of mass. The amorphous part is the first that degrades at low temperature, and it is difficult to see since the loss of mass is small in front of the crystalline one that occurs at high temperature. The main stage of decomposition in nitrogen atmosphere occurs in the range of 300–350°C where the rapid mass loss is attributed to the crystalline cellulose degradation, leading to the formation of pyrolysis products, mainly levoglucosan, and other combustible gases. At temperatures close to 400°C the combustion of carbonaceous products occurs, which competes with the formation of levoglucosan, carbonization reactions, dehydration, decarboxylation and the emission of water, carbon dioxide, carboxyl and double bond carbonyl derivatives. The carbon content of the decomposition products increases and carbonaceous residues are formed. Different behavior was found for thermal decomposition in oxygen atmosphere (red line). When tested in oxygen atmosphere, a sudden loss of mass at 274°C caused by the instantaneous combustion of the degraded crystalline cellulose is observed.
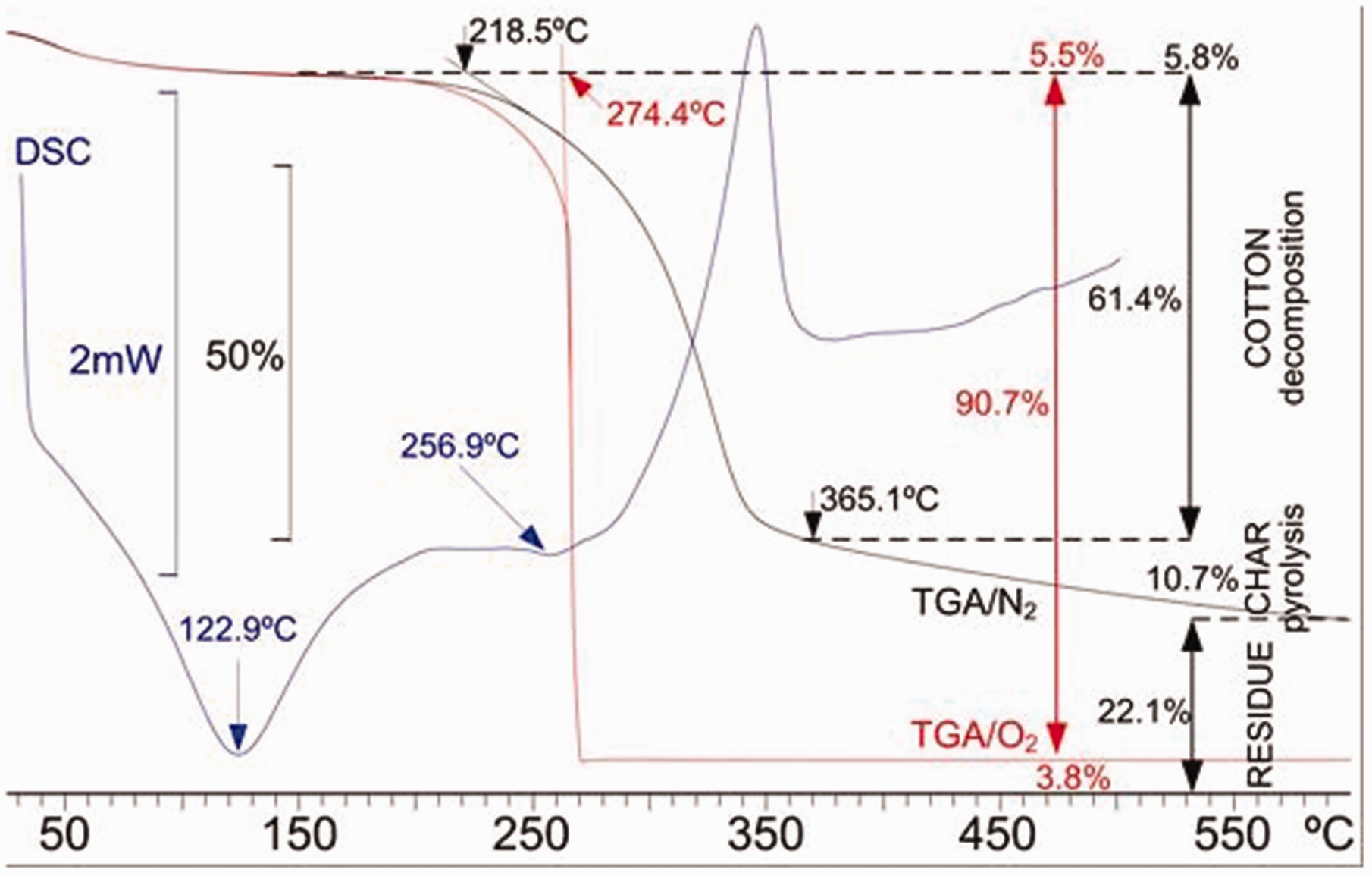
Thermogravimetric analysis (TGA) in O2 atmosphere (red), TGA in N2 atmosphere (black) and differential scanning calorimetry (DSC) (blue) of 100% untreated cotton linen fabric (color online only).
The two FR products used are FR-conv and FR-alter. Both are analyzed by DSC and TGA.
Figure 3 shows the DSC curves of FR-conv (black) and non-treated cotton linen fabric, CO (blue) and that treated with FR-conv (red). FR-conv shows small transitions with endothermic and exothermic reactions of very small energy in all ranges of temperature. It is a fair thermally stable product. If the DSC curve of FR-conv-treated linen fabric reflects the superimposition of the DSC plots of the linen fabric and FR-conv, no chemical reaction between the two components has been detected. The DSC plot of the linen fabric treated with FR-conv (CO-FR-conv) does not reflect the transitions of the linen fabric or FR-conv. The first endotherm of moisture evaporation is greatly shifted from 122.9°C to 75.8°C, and the high exotherm caused by cellulose decomposition is greatly reduced in energy, ascending the onset temperature of decomposition from 256.9°C to 281.3°C, being the peak temperature shifted from 346°C to 350°C, and shortening the final temperature of decomposition from 380°C to 360°C. The application of FR-conv on the linen fabric causes a great reduction in the emission of caloric energy when the sample is heated.
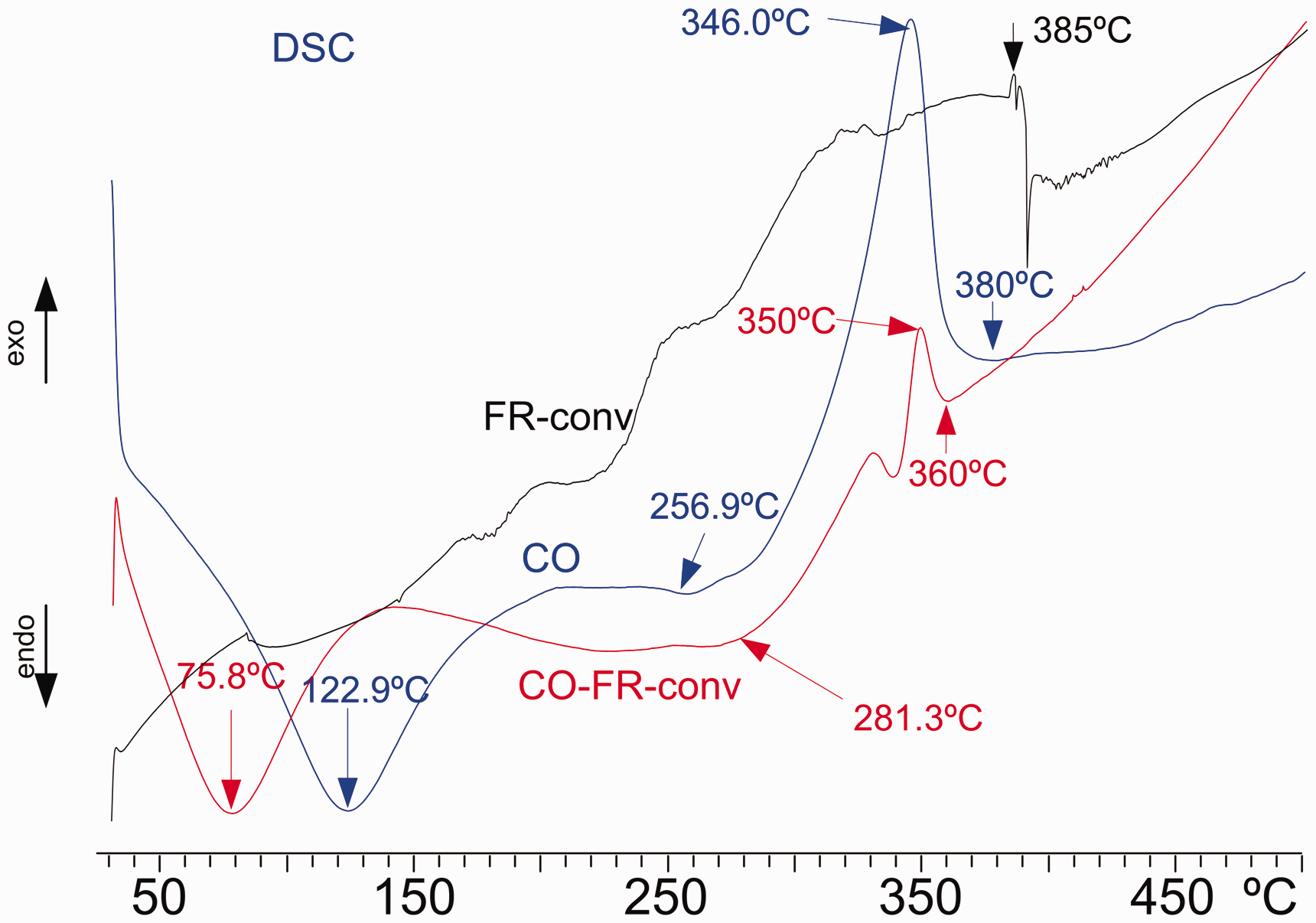
Differential scanning calorimetry (DSC) plots of FR-conv (black), untreated cotton linen fabric (CO) (blue) and cotton linen fabric treated with FR-conv (CO-FR-conv) (red) from 25°C to 500°C (color online only). FR: flame retardant; DPCAA: dialkyl phosphono carboxylic acid amine.
Figure 4 presents the TGA plots of FR-conv (blue) and those of non-treated cotton linen fabric, CO, (black) and that of the fabric treated with CO-FR-conv (red). The small mass losses caused by temperature on FR-conv can be associated with the evaporation/sublimation of the different components leading to a final residue of 42.7% at 600°C. The application of FR-conv on cotton fabric shifts the losses of mass to greater temperatures; the onset temperature of decomposition grows from 218.5°C to 301.4°C, the loss of mass due to thermal decomposition of cellulose is slightly reduced from 61.4% to 60.2% and a slight increase in the final residue from 22.1% to 23.5% at 600°C is observed, which can be attributed to the formation of a higher amount of non-flammable volatiles. The application of FR-conv shifts the loss of mass plot given by the TGA to greater temperatures.
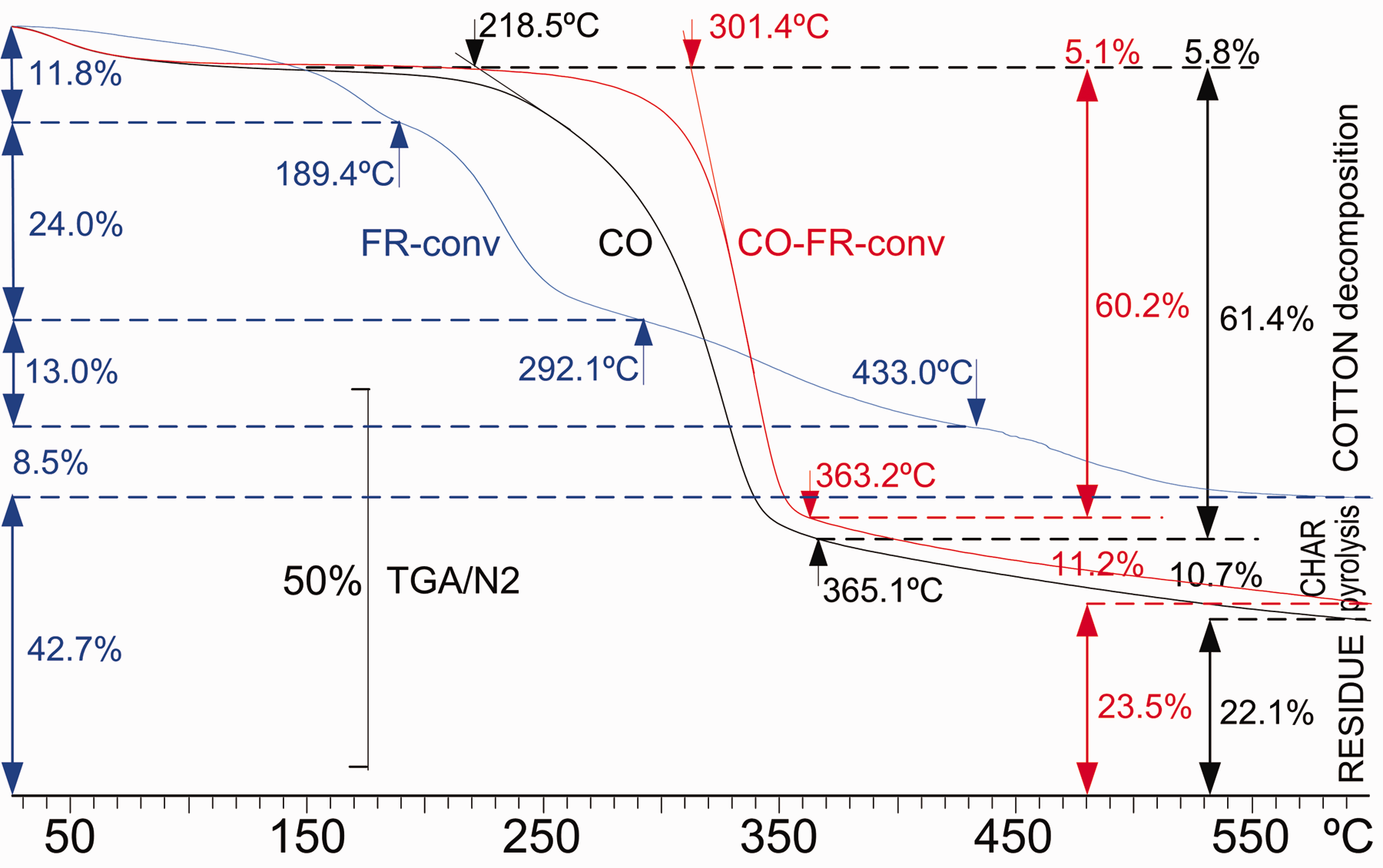
Thermogravimetric analysis (TGA) plots of FR-conv (blue), untreated cotton linen fabric (CO) (black) and cotton linen fabric treated with FR-conv (CO+FR-conv) (red) from 25°C to 600°C in N2 atmosphere (color online only). FR: flame retardant; DPCAA: dialkyl phosphono carboxylic acid amine.
Figure 5 shows the DSC curves of FR-alter, the untreated cotton fabric, CO and the cotton treated CO-FR-alter. The DSC of FR-alter presents two small exo-endo transitions in the areas of 250°C and 420°C, presenting a fairly stable product with temperature. Nevertheless, the great modification in the DSC plot of the linen fabric reveals that FR-alter reacts with cellulose, 39 shifting down and reducing the evaporation peak attributed to moisture, and yielding to a decomposition of the weak thermally stable FR-alter-cellulose component explained by the great endotherm at 238.3°C, followed by a small exotherm at 251.6°C, probably caused by the formation of a more thermally stable FR-alter-cellulose component. The great decomposition exotherm of the cellulose of the cotton linen fabric has completely disappeared.
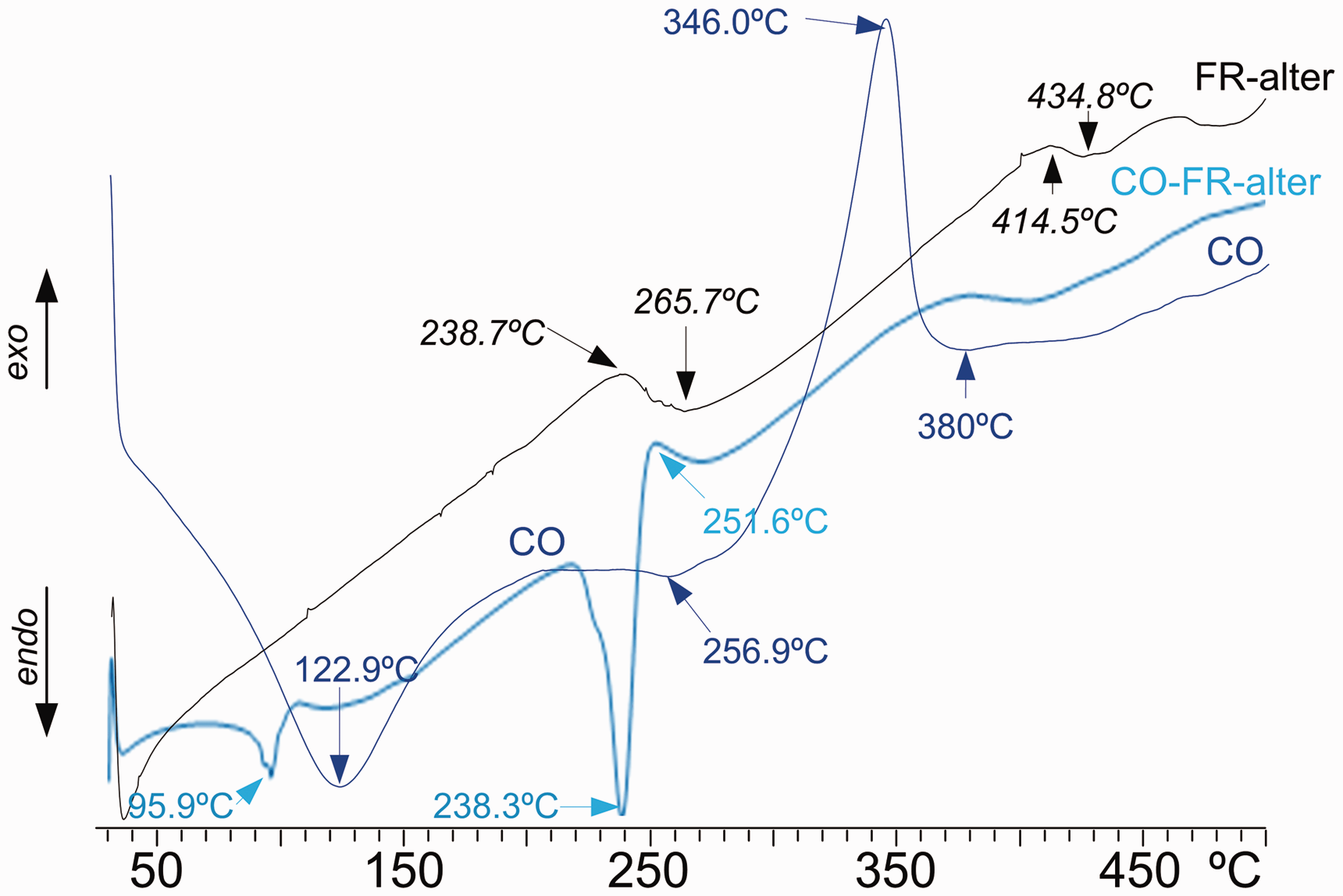
Differential scanning calorimetry plots of the alternative FR (FR-alter) (black), untreated cotton linen fabric (CO) (blue) and cotton linen fabric treated with the alternative FR (CO-FR-alter) (light blue) from 25°C to 500°C (color online only). FR: flame retardant; ASU: ammonium sulfamate.
As regards the TGA plots shown in Figure 6, the thermal stability of FR-alter observed in the DSC is confirmed by the TGA, where practically no loss of mass occurs up to 350°C. At 436.6°C, which follows the second DSC endo-exo transition, the loss of mass accounts for 10%, which progressively increases leading to a final residue of 13.9% at 600°C.
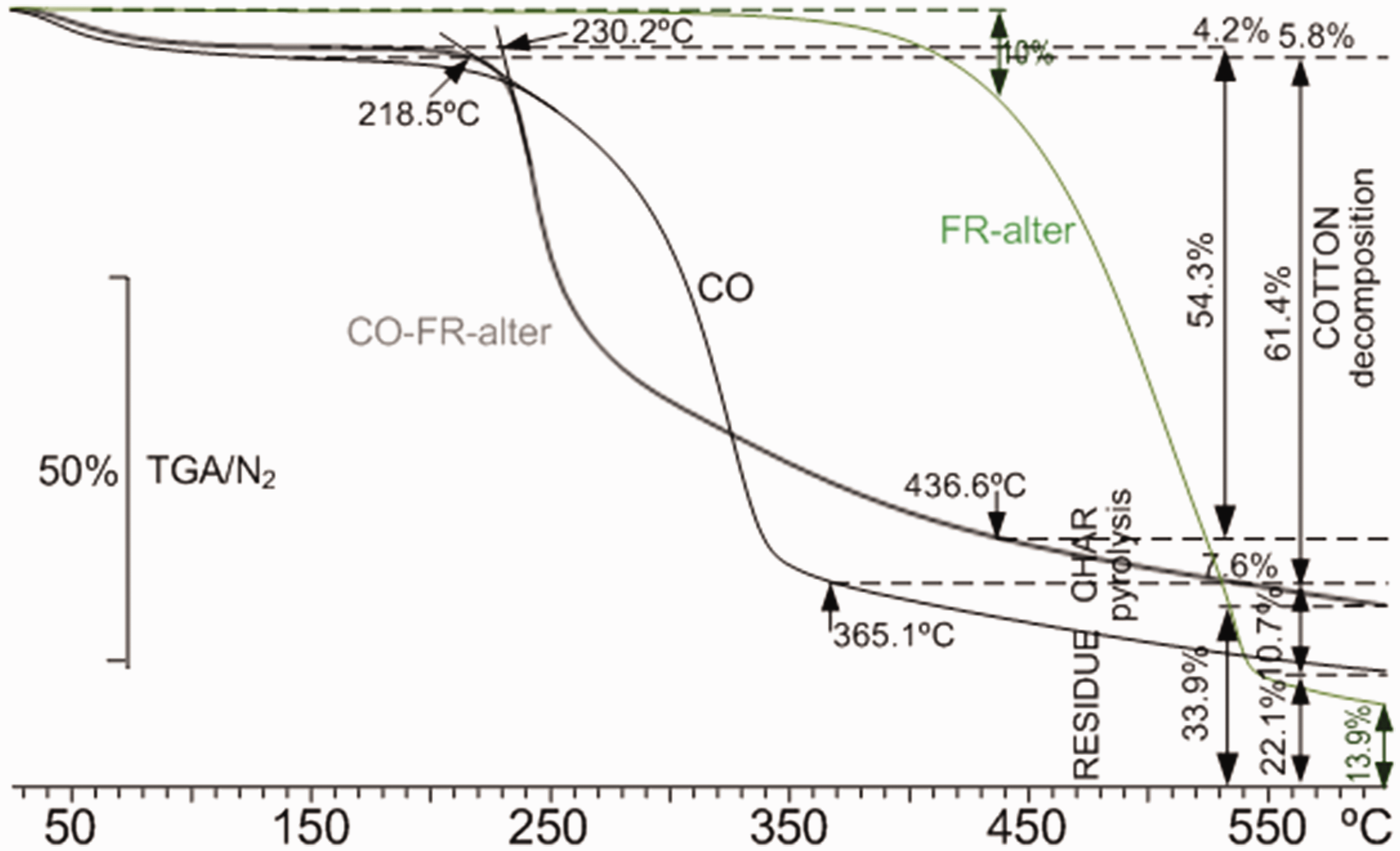
Thermogravimetric analysis (TGA) plots of FR-alter (green), untreated cotton linen fabric (CO) (black) and cotton linen fabric treated CO-FR-alter (grey) from 25°C to 600°C in N2 atmosphere (color online only). FR: flame retardant; ASU: ammonium sulfamate.
The TGA plots of the cotton linen fabric and that treated with FR-alter are in accordance with the thermal transitions reflected by the DSC. The strong decomposition endotherm with peak temperature at 238.3°C is in accordance with the great loss of mass observed in the TGA that begins at 230.2°C. This means that the reaction between FR-alter and cellulose opens its structure, leading to a less thermally stable component that is transformed into non-flammable volatile. This can be explained by both the greater loss of mass of the CO+FR-alter sample from 230°C to 251.6°C and the small thermal energy release illustrated by the small exotherm at 251.8°C in Figure 5, when compared with the greater flammability of the volatiles of the untreated fabric that shows a great exotherm at 346.0°C in Figure 5 as a consequence of the greater loss of mass, especially from 325°C. The shorter loss of mass of the FR-alter treated fabric from 325°C can be justified by the more thermally stable FR-alter-cellulose component that contributes to the 11% greater amount of the final residue when compared with that of the untreated one. At approximately 325°C, the TGA plots of both the untreated CO and CO-FR-alter cross, and from that temperature, the FR-alter treated one shows a shorter loss of mass which can be explained by the more thermally stable FR-alter-cellulose component shown by the DSC exotherm at 251.6°C.
The derivative of the TGA plots enables one to identify the temperature at which the decomposition of cellulose ends: 365.1°C for untreated CO and 435.6°C for CO-FR-alter, which is in accordance with the more stable FR-alter-cellulose formed component.
FRs on textiles are expected to be effective in presence of oxygen. Figure 7 shows the effect of the FRs on the TGA of bed linen cotton fabric in pure O2 atmosphere.
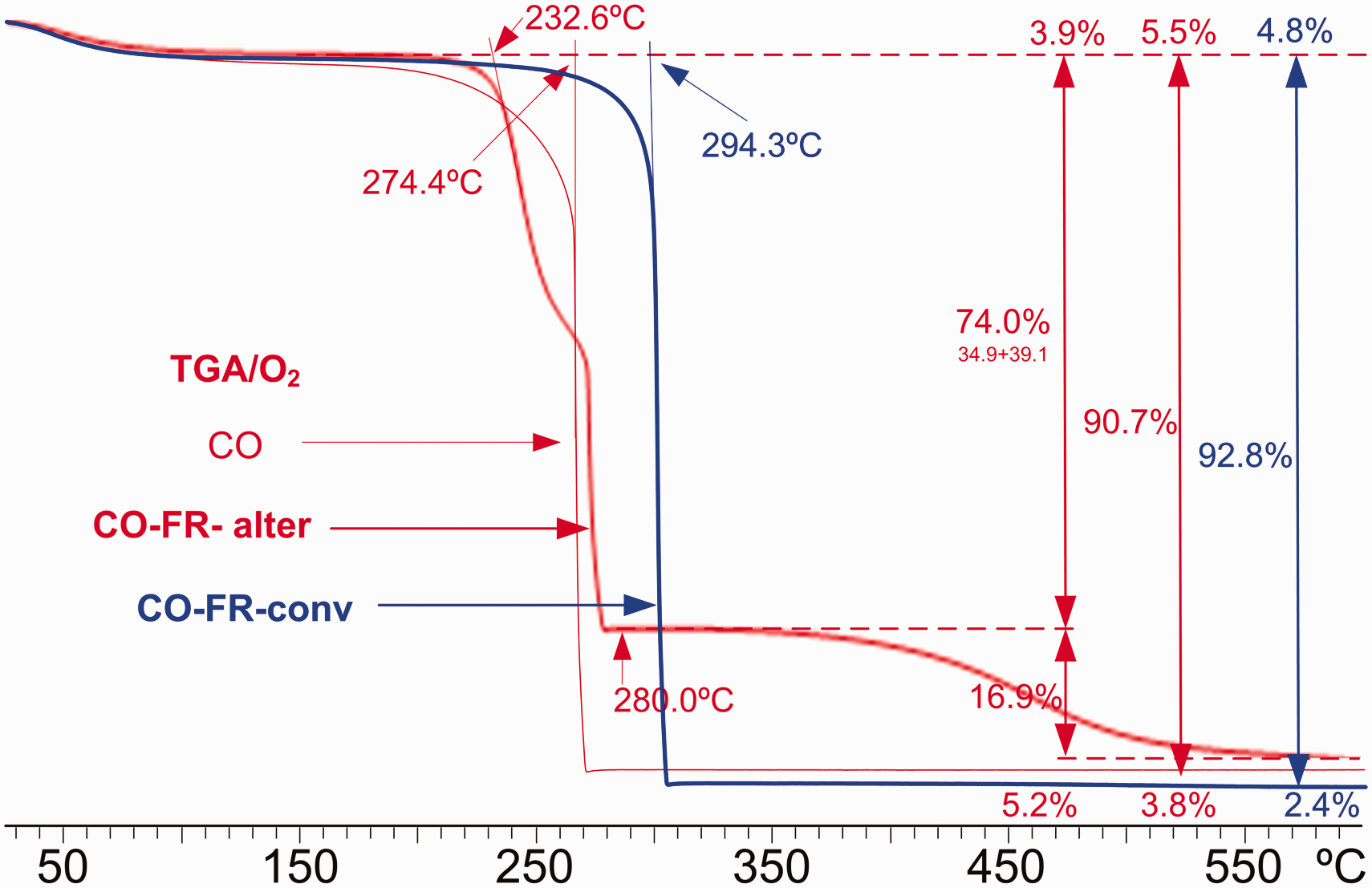
Thermogravimetric analysis (TGA) plots of untreated cotton linen fabric (CO) (red) and that treated with conventional FR (CO-FR-conv) (blue) and with alternative FR (CO-FR-alter) (dark orange) from 25°C to 600°C in O2 atmosphere (color online only). FR: flame retardant; ASU: ammonium sulfamate; DPCAA: dialkyl phosphono carboxylic acid amine.
The untreated cotton linen fabric shows the highest loss of mass due to moisture (5.5%) followed by the loss of mass explained by the degradation of the amorphous and crystalline cellulose that accounts for 90.7% of mass loss at 274.4°C, leading to a final residue of 3.8% at 600°C.
The treatment with the FR-conv shows a similar decomposition plot by temperature, but shifted by 20°C. The loss of mass due to moisture (4.8%) is followed by the loss of mass explained by the degradation of cellulose (92.8%), leading to a final residue of 2.4% at 600°C. The FR-conv effects are mainly based on the formation of more non-flammable compounds in oxygen atmosphere that are reflected by the increase of 20°C to reach the whole decomposition of cellulose.
The application of FR-alter greatly modifies the decomposition plot by temperature. As expected, its application drives the decomposition to lower temperatures, reducing the damage caused by heat, 39 while the formation of a more thermally stable non-flammable products results in components that are progressively volatilized from 280°C to 600°C, accounting for a 16.9% mass loss. The initial loss attributed to moisture is 3.9% and the decomposition of the less stable FR-alter-cellulose component accounts for a 74% in mass that is volatilized in two steps from 232.6 to 280°C, which are in accordance with the DSC plot. The final residue at 600°C is the highest (5.2%).
The application of FR to cotton fabrics can improve the thermal stability of the fabric by increasing the decomposition temperature and/or increasing the final residue by decreasing the fraction of mass that can be transformed into flammable volatiles. The application of FR-conv leads to an increase of decomposition temperature and that of FR-alter leads to the decomposition of cotton in successive stages that limits the emission of volatiles and end up generating a greater volume of final waste. The action of these retardants will be more effective the more stable they are with the temperature, since their presence acts in the carbonization and dehydration reactions, inhibiting the reaction that leads to the production of levogucosan, reducing the generation of combustible products and increasing the formation of H2O, CO2 and other volatiles. 9 Table 4 presents a summary of the results that can be considered most relevant to the effectiveness of the applied FRs. The increase in the thermal stability of the tissue that causes an increase in the decomposition temperature is a relevant effect associated with the application of FR-conv in the tissue, while the most important effect of FR-alter lies in the increase of the residue at 600°C, associated with the generation of fewer volatiles, which has an impact on the intensity of combustion.
Summary of thermogravimetric analysis (TGA) results for cotton fabrics

FR: flame retardant.
Percutaneous absorption
Percutaneous absorption of the two textiles treated with the different FRs was performed to determine the possible dermal toxicity. The procedure for assessing the risks to human health of a product (in our case, a FR product) is to compare the exposure levels to which populations are or may be exposed with the exposure level at which no toxic effects are expected. The risk evaluation of a chemical consists of four basic steps: hazard identification, hazard characterization (dose response assessment), exposure assessment and risk characterization. The modification of the incidence and severity of the effects that may affect a human population due to actual or expected exposure to a chemical may include the probability of the risk (MoS).
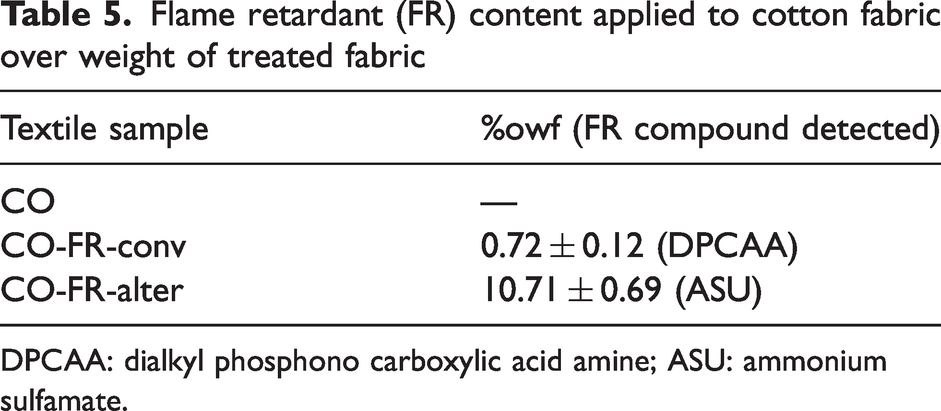
Fabric samples applied with FR-conv (approximately 2 × 2cm2, ∼58 mg textile material) at the laboratory level, as well as an untreated fabric (CO), were extracted with methanol and the CO-FR-alter sample, industrially applied, was extracted with p-hydroxybenzoic acid (5.8 mM, pH 9.4)/methanol (97:3); all extracts were analyzed by HPLC-DAD. The content of the compound from FR-conv (DPCAA) and from FR-alter (ASU) was calculated according to the previously performed calibration curves; in the case of ASU, the remaining unreacted ASU is the active principle detected. The textile sample, its reference and the amount of FR compound detected in the samples are detailed in Table 5. It is important to remark on the much higher concentration of the CO treated with FR-alter, since this high amount was optimized during the first studies 43 to give enough FR properties.
Flame retardant (FR) content applied to cotton fabric over weight of treated fabric
DPCAA: dialkyl phosphono carboxylic acid amine; ASU: ammonium sulfamate.
The efficacy of the solvent extraction and the evaluation of possible interference with the skin were verified in different compartments: stratum corneum, epidermis and dermis. After 24 hours, the FRs were extracted with the appropriate solvent and analyzed by HPLC-DAD. The recoveries obtained for the FR products tested were acceptable.
In order to calculate the MoS, permeation (percutaneous absorption) experiments were performed to determine the distribution of FRs in the different layers of the skin, after 24 hours of skin–textile contact, using Franz-type diffusion cells. After extraction, samples were analyzed to determine the presence of the two FRs in the different fractions: textile (T), stratum corneum (SC), epidermis (E), dermis (D), receptor fluid (RF) and the total percutaneous absorption, which is the sum of active components in the epidermis, dermis and receptor fluid (PA).
Percutaneous absorption experiments of the treated cotton fabrics were then performed, obtaining the results shown in Figure 8; after 24 hours of exposure, the distribution of the analyzed compounds DPCAA and ASU in the different skin compartments (stratum corneum, epidermis and dermis), as well as the excess in the tissue and its content in the receptor fluid, were determined. The total amount of recovery is acceptable. In the case of CO-FR-conv, most of the active component was found in the remaining textile (86%), while there was 14% in the stratum corneum. No amount of DPCAA was detected in the internal layers of the skin (epidermis, dermis and systemic level). As a result, the absorbed compound that contributes to the percutaneous dose is below the limit of detection (1.855 µg/cm2).
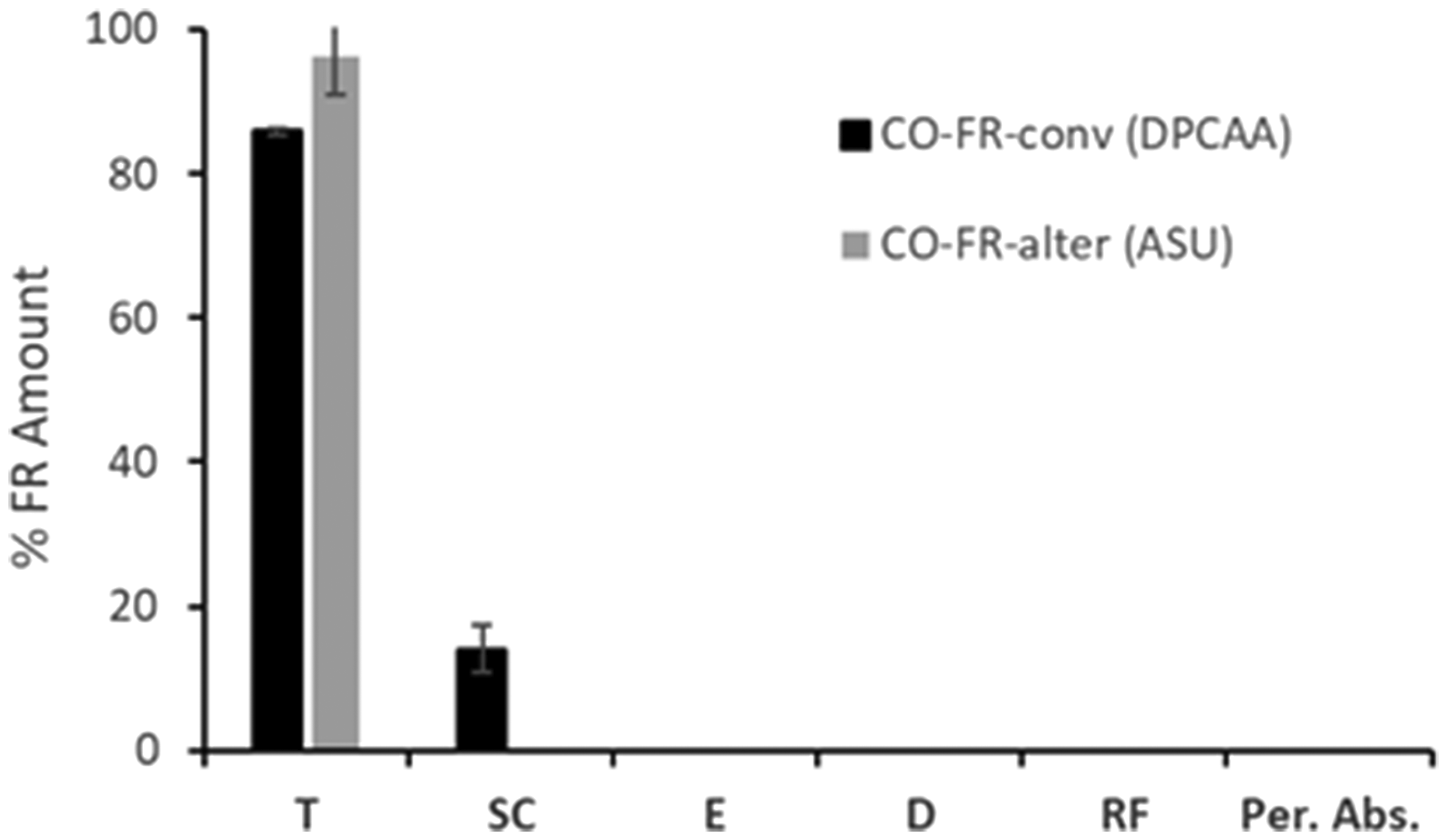
Percutaneous absorption of FR-treated cotton fabrics. FR: flame retardant; ASU: ammonium sulfamate; DPCAA: dialkyl phosphono carboxylic acid amine; T: textile; SC: stratum corneum; E: epidermis; D: dermis; RF: receptor fluid.
The alternative FR percutaneous absorption profile shows only the presence of ASU in the remaining textile (93%). This active component was not detected in the stratum corneum nor in the inner skin when the treated cotton fabric is in contact with human skin. Thus, as in the previous case, total percutaneous absorption (epidermis + dermis + receptor fluid) is below the limit of detection (1.102 µg/cm2).
Once the percutaneous absorption is obtained, the MoS can be calculated. To do this, first the NOAEL values must be researched in the existing literature and were found to account for 1000 mg component/kg body weight (bw)/day (Table 6).
Values of the different parameters needed to calculate the margin of safety (MoS)
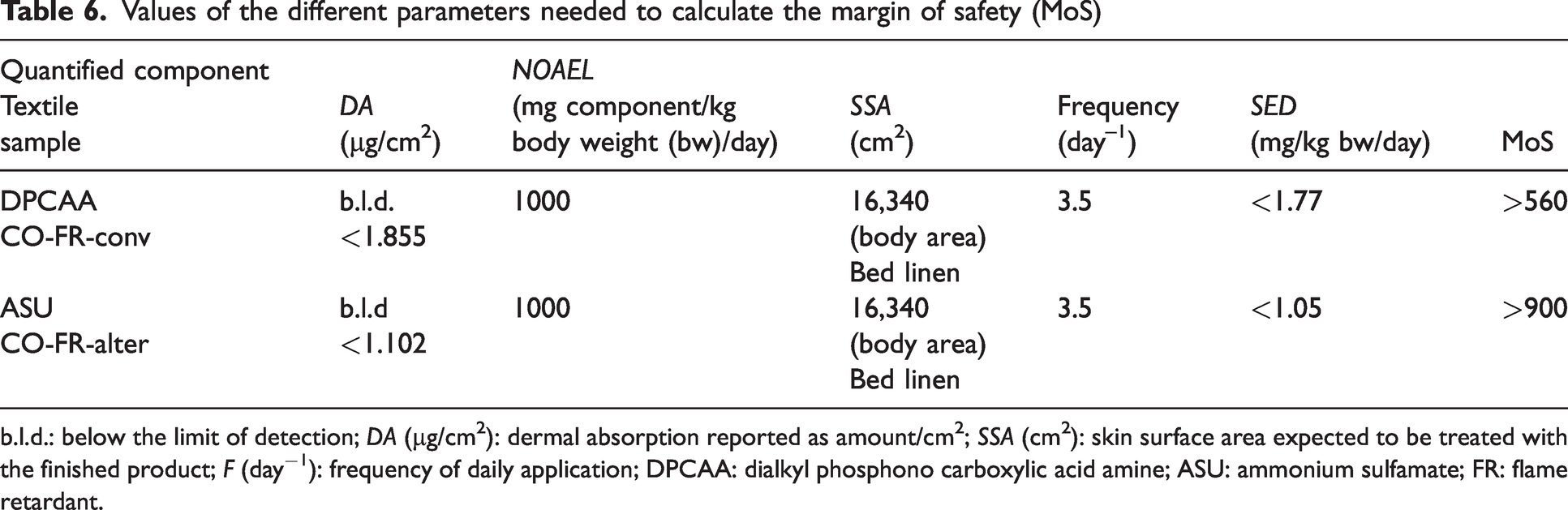
b.l.d.: below the limit of detection; DA (µg/cm2): dermal absorption reported as amount/cm2; SSA (cm2): skin surface area expected to be treated with the finished product; F (day−1): frequency of daily application; DPCAA: dialkyl phosphono carboxylic acid amine; ASU: ammonium sulfamate; FR: flame retardant.
To calculate the SED value (systemic exposure dose) of Equation (2), the following values of the parameters explained in the experiments have been used (see Table 5). The MoS is calculated according to Equation (1), obtaining the values shown in Table 6.
FR finished fabrics with a MoS greater than 100 are safe for human health, so in this study, both FRs applied to a cotton bedding fabric are not potentially toxic to humans in the quantities used to obtain a suitable FR finish. Moreover, FR-alter seems to be safer when in contact with the skin considering also its higher concentration in the textile.
Conclusions
In this study, two FR finishes for 100% cotton bed fabrics were analyzed. A conventional FR based on phosphorus and an alternative FR based on a crosslinking reaction between ammonium sulfamate, urea and PO(OH)2-R-PO(OH)2 were applied to cotton fabrics.
The burning behavior was assessed, where the bed linen is not prone to ignition with smoldering cigarette. Although the non-treated fabric passes EN ISO 12952-1 & 2, it failed the more severe crib 5 test. While no improvement was found for the cotton treated with FR-conv (due to the low linkage of the DPCAA), the cotton treated with FR-alter passes all the flame tests performed.
The thermal behavior of the FRs and the textiles finished with them were evaluated using DSC and TGA. FR-treated fabrics were demonstrated to improve the thermal stability, hence both FR products act as FRs. FR-conv increases the temperature at which decomposition of the cotton fabric begins, and FR-alter increases the amount of final residue, which indicates a decrease in volatile components, reducing the intensity of combustion.
Percutaneous absorption of the two textiles treated with the different FRs were performed to determine the possible dermal toxicity. In the case of CO-FR-conv, most of the active component was found in the remaining textile (86%), while there is a 14% in the stratum. The CO-FR-alter percutaneous absorption profile shows only the presence of ASU in the remaining textile (93%), not in the stratum corneum nor in the inner skin. Notwithstanding that the industrially treated fabric had a higher uptake of FR, it did not lead to an increase in the penetration of the active component in the skin. Then, the dermal toxic potential study of FR-treated cotton bed sheets showed that none of the two products tested penetrate into the skin when applied under the conditions used. Therefore, all FR-treated cotton fabrics, even the more concentrated FR-alter, are safe when in contact with the skin.
Therefore, this study executed on FR-treated cotton bed sheets showed that it is possible to replace the conventional formaldehyde-releasing FR product with a safer alternative product, since both act as FRs. The tested treatments have shown to have no dermal toxicity potential for humans.
Footnotes
Acknowledgements
The present work could not have been performed without the collaboration and contribution of the Service of Dermocosmetic Assessment and the Service of Thermal Analysis and Calorimetry “Josep Carrilla,” both from IQAC-CSIC.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministerio de Ciencia, Innovación y Universidades (RTI2018-09014-B-100 Project) and the EU (Mitigation of environmental impact caused by Flame Retardant textile finishing chemicals. LIFE-FLAREX. Life 16 ENV/ES/000374).
