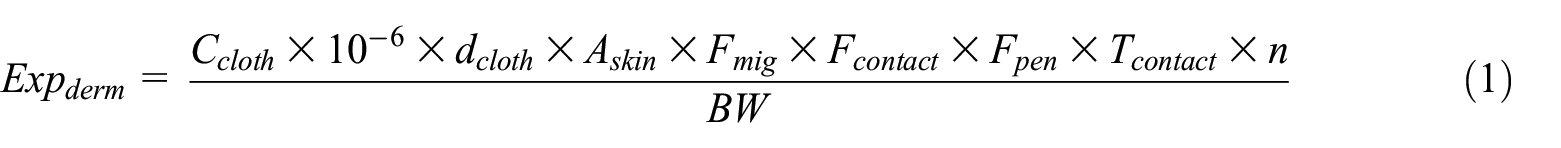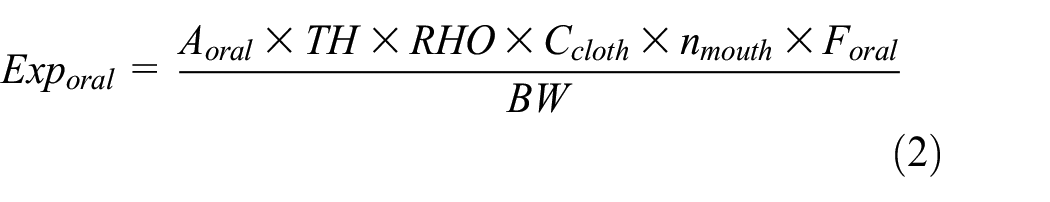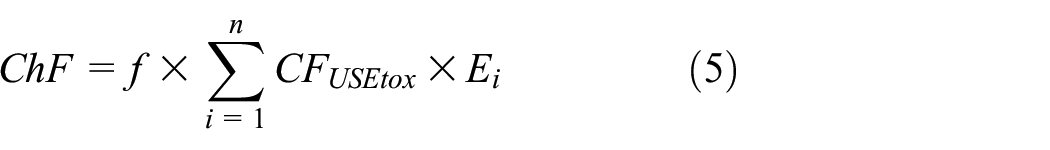Abstract
The production and processing of textile and apparel products involve the use of various chemicals. These chemicals, such as alkylphenol ethoxylates (APEOs), are released during washing and wearing and may be potentially harmful to humans and the aquatic environment. The primary objective of this research was to investigate the effect of laundering on nonylphenol ethoxylates (NPEOs) and octylphenol ethoxylates (OPEOs) in textiles. We performed washing experiments on nine unwashed textile samples. The results showed that the levels of NPEOs and OPEOs were significantly reduced after washing compared to the initial levels, and the release rates during washing ranged from 63% to 98%. However, it may not be possible to completely remove residual APEOs from clothing by washing. The total exposure dose to infants wearing unwashed textiles was approximately 2.7–50.1 times that of washed textiles, with oral exposure accounting for 35.7–53.5% of the total exposure dose. Furthermore, nine samples had a chemical footprint after washing of up to 114 PAF·m3·day. Given these findings, meticulous attention should be directed toward auxiliary selection and the formulation of eco-friendly alternatives throughout the textile production lifecycle.
Introduction
Alkylphenol ethoxylates (APEOs), important non-ionic surfactants, find extensive application in household detergents, industrial detergents, wetting agents, dispersants, emulsifiers, solubilizers, and foaming agents across various industries including textiles, leather, printing and dyeing, metal treatment, plastic, rubber, and paper. Among these, nonylphenol ethoxylates (NPEOs) constitute approximately 80–85%, while octylphenol ethoxylates (OPEOs) constitute approximately 15%. 1 In China alone, the production of APEOs reached approximately 69,000 tons in 2020, with 60% of this volume ultimately finding its way into aquatic environments through various pathways. 2 Due to significant use and release, APEOs may have potential impacts on humans through direct contact with consumer products and on the environment through wastewater discharges and sludge disposal. Various APEO metabolites are ubiquitous in many environmentally relevant matrices such as wastewater treatment plant influent and effluent, river water, seawater, surface water, groundwater, drinking water, sediment and soil, food drinking water, and human tissue samples.3,4 Many studies have highlighted that these small molecule metabolites of APEOs, including 4-alkylphenols (AP), (4-alkylphenoxy) acetic acid (AP1EC), (4-alkylphenoxy) ethoxy acetic acid (AP2EC), 4-alkylphenol ethoxylate (AP1EO), and 4-alkylphenol diethoxylate (AP2EO), are persistent, bioaccumulate and toxic. Some are already considered priority substances (e.g., octylphenol [OP]) or listed as priority hazardous substances in the Water Framework Directive of the European Union (e.g., nonylphenol [NP]).
The textile industry stands as one of the largest sectors globally, annually furnishing nearly 100 million metric tons of new products to the market. 5 Research indicates that clothing constitutes the most extensive surface area among indoor materials, closely followed by household textiles such as upholstery and curtains. 6 Regrettably, as the textile industry continues its upward trajectory, a multitude of chemicals are employed, leading to the release of various chemicals. These substances, whether intentionally or unintentionally added, may persist in the final product at notable concentrations. 7 Textiles have been found to harbor a diverse array of chemicals, including alkylphenol ethoxylates, perfluoroalkyl and polyfluoroalkyl substances, aromatic amines, and brominated flame retardants. 8 The origins of APEOs in textiles are multifaceted. Emulsifying agents utilized in spinning processes facilitate the solubilization of lubricants in water, while those employed in dyeing and printing processes aid in the dissolution and stabilization of dyestuffs in water. Wetting and deaeration agents enhance dye liquor wetting or dye absorption, carriers facilitate dye application, and leveling agents ensure uniform dye distribution. In addition, detergents utilized throughout various stages of textile manufacturing serve to eliminate impurities. 9 APEOs have long been utilized as surfactants, owing to their exceptional emulsifying, and dispersing properties, enabling the formulation of highly stable emulsions or dispersion concentrates. Renowned for their efficacy, versatility, and cost-effectiveness, APEOs find widespread application across diverse industrial domains.
However, owing to their detrimental impacts, numerous countries and regulatory bodies have implemented laws and standards over time to curb the usage of textiles, footwear, and related products. Notably, in 2012, the OEKO-TEX® Association introduced the OEKO-TEX® STANDARD 100, marking the first instance of imposing limit requirements on APEOs in textiles. Subsequent to this, in 2019, the European Union (EU) enacted a prohibition on the utilization of NPEOs in textiles. This directive includes a transitional phase of 5 years, during which textiles containing NPEOs exceeding 0.01% (equivalent to 100 mg/kg) are barred from entering the EU market. Furthermore, the 2024 iteration of the OEKO-TEX® STANDARD 100 further restricts the total concentration of OP and NP to 10.0 mg/kg and the combined concentration of NP, OP, NPEOs, and OPEOs to 100.0 mg/kg. Despite regulatory measures, studies reveal continued usage of APEOs in textile processing across many nations. Residual APEOs may persist on items until reaching end consumers, with the potential for release into the environment during washing. Investigations by the Danish Environmental Protection Agency (2013) and the UK Environment Agency (2013) indicate that 25–99.9% of APEO content is removed after a single wash, with more than 99.9% of NPEOs released after two washes.10,11 Given the non-ionic nature of APEOs, rendering them readily soluble in water, repeated washing is likely to eliminate all APEOs, regardless of textile type. 12 When APEOs enter the aquatic environment, they can pose a threat to aquatic organisms, thereby affecting the ecosystem. According to the U.S. EPA (2010), NPs and NPEOs in freshwater ecosystems have the potential to adversely affect aquatic species at all trophic levels that are exposed to them. Ultimately, the presence of hazardous and toxic chemicals may cause irreversible adverse effects on human health and ecosystems. 13 Furthermore, APEOs can permeate the human body through dermal contact and other pathways, thereby posing risks to human health.
The objective of this research was to investigate the effect of laundering on NPEOs and OPEOs in textiles and to evaluate the release of these chemicals into household wastewater. For this purpose, the nine textile samples were collected from a third-party laboratory to determine the concentration and distribution of NPEOs and OPEOs, to assess removal by the washing process, to estimate the dose of APEOs exposed to the skin when the garments were worn, and to quantify the environmental impacts caused by washing. This study is the first to combine human exposure model and chemical footprint with the aid of washing experiments to assess the human health and ecological impacts of APEOs. Our research can inform public health policy and environmental protection, as well as provide a basis for chemical risk management and chemical contaminant database development.
Materials and Methods
Chemicals and Reagents
The chemicals and reagents used included methanol (HPLC grade), acetonitrile (HPLC grade), octylphenol standard (OP, CAS no. 140-66-9), nonylphenol standard (NP, CAS no. 25154-52-3), octylphenol ethoxylate standard (OPnEO, CAS no. 9002-93-1) and nonylphenol ethoxylate standard (NPnEO, CAS no. 9016-45-9) were purchased from ANPEL Laboratory Technologies (Shanghai) Inc. (Shanghai, China).
Textile Samples
Supplemental Table S1 presents a summary of the primary characteristics of the nine textiles utilized in this investigation. Textiles containing APEOs were obtained from a third-party laboratory. All samples were meticulously stored in airtight, light-proof plastic bags at room temperature to ensure consistent conditions.
The washing experiments comply with the international standard Textiles—Domestic washing and drying procedures for textile testing (ISO 6330: 2021, procedure 4N). Each of the nine samples was placed in a laboratory washing machine from Wenzhou Fangyuan Instrument Co., Ltd. (Y089CA, Zhejiang, China), equipped with steel vessels measuring 515±5 mm in diameter, 335±10 mm in height, and with a volume of 65 L, with enough polyester fiber ballast (without the target chemical) to accompany the wash so that the total wash load was 2.0 kg. The water used in the washing cycle was treated using a pure water treatment system from Guangdong Yangsheng Construction Engineering Co., Ltd (R0-500 L/H, Guangdong, China). A 4N wash program was selected and the amount of standard detergent added per wash cycle was 15.4 g ECE Detergent 98 (linear sodium alkylbenzene sulfonate, ethyloxylated fatty alcohol C12-18, sodium soap, foam inhibitor, sodium aluminum silicate zeolite 4A, sodium carbonate, sodium salt of a copolymer from acrylic and maleic acid, sodium silicate, carboxymethylcellulose, diethylene-triamine penta, sodium sulfate and water), 4 g sodium perborate tetrahydrate and 0.6 g tetra-acetylethylenediamine (TAED) from SDC Enterprises Limited (England). The textiles were subjected to washing at 40±3 °C for 60 min, encompassing washing and spinning processes. Immediately after the selected washing program, the specimen and dummy load were removed and dried at 40–60 °C in the dryer from Wenzhou Fangyuan Instrument Co., Ltd (YG 741, Zhejiang, China). After the first washing cycle, the textiles were repeated using the same procedure. The amount of washed APEOs was determined using scissors to randomly cut fabrics from the samples after washing and drying were completed. Due to the high original APEO contents of the samples, they were not measured every time after washing.
Extraction and Analysis
To mitigate the potential heterogeneity of contamination arising from the processing of the measured textile samples, three sections were randomly selected from the samples, cut into approximately 5 × 5 mm pieces, and then mixed homogeneously. These fragments were thoroughly mixed to ensure homogeneity across the sample. The sample preparation methodology employed for the determination of APEOs and APs in textiles was adapted from the standard Textiles—Method for the detection and determination of alkylphenol ethoxylates (ISO 18254-1: 2016). In summary, approximately 1.0 g of each textile sample was accurately weighed and transferred into 50 mL glass centrifuge tubes. Extraction was carried out by adding 30 mL of methanol to each tube. Subsequently, sonication at 70 °C for 60 min facilitated the extraction process. The resulting solvent was collected, filtered through a 0.22 μm nylon filter, and transferred to another glass tube. Quantification of APEOs and APs was conducted utilizing liquid chromatography−mass spectrometry (LC-MS/MS) equipped with a C18 chromatography column (4.6 × 150 mm × 5.0 μm; SCIEX, the United States). The chromatographic column temperature was maintained at 40 °C, with a flow rate of 250 μL/min. Both atomization gas and collision gas utilized were high-purity nitrogen.
Quality Assurance and Quality Control
The laboratory was meticulously cleaned to meet the strict standards of trace analysis. To ascertain the concentrations of APEOs and APs, peak areas of standards were employed with an external calibration method that underwent validation at five distinct concentration levels. The signal-to-noise ratio ≥ 3 is defined as the instrument detection limit, which in this study ranged from 0.05 to 1.00 μg/mL. 14 When the detected concentration exceeded the linear range, it was diluted appropriately and retested. All calibration curves demonstrated correlation coefficients greater than 0.997. To verify the accuracy of the instrumental methods, spiked blank, and matrix-spiked samples were utilized. The recoveries of the target chemicals in spiked matrices varied from 85.1% to 107.2%.
Exposure Assessment
It is assumed that exposure via inhalation will be irrelevant as APEOs have very low vapor pressure and, therefore, very few APEOs can be expected to be released from textiles into the air. The other routes of exposure, skin contact and ingestion (i.e., by mouth by children sucking on clothes corners), are considered relevant exposure routes. Because they do not have entirely developed biological systems, infants are particularly vulnerable to environmental pollutants.
The dermal exposure dose for infants (<12 months old) was assessed, which can be obtained from Equation (1) following the European Chemical Agency (ECHA) guidance on information requirements and chemical safety assessment. 15
where Expderm corresponds to dermal exposure (mg/kg-day), Ccloth is the concentration of NPEOs and OPEOs in clothing (mg/kg), 10−6 is a conversion factor (kg/mg) and dcloth is the cloth surface density (mg/cm2). Askin is the infants’ skin area covered by the clothing (cm2), assumed to be 1602.5 cm2. 16 Fmig is the fraction of substance that migrates to the skin each day (1/day). Given the absence of available information regarding NPEO and OPEO migration from textiles to skin, a migration factor of 0.02 was utilized, following the criteria of the Bundesinstitut für Risikobewertung. 17 Fcontact is the fraction of contact area for skin (dimensionless), assumed to be 1. 18 Tcontact is the duration of the clothing skin contact (day), assumed to be 1. 18 n is the number of events per day (1/day), also assumed to be 1. 19 Fpen represents the penetration rate of chemicals into the body, assumed to be 5%. 17 BW is the body weight (kg), assumed to be 7.31 kg. 16
Oral exposure is assumed to occur as a result of infants sucking on the textile, and it is here calculated from the following equation, which is the equation that the tool ECETOC TRA (http://www.ecetoc.org/tra) uses for estimating oral exposure 20 :
where Exporal corresponds to oral exposure (mg/kg-day). Aoral is the area of the textile that the person has put in his mouth (cm2), assumed to be 10. nmouth is the number of times per day that the person has sucked on the textile, assumed to be 1. TH is the layer thickness (m), assumed to be 0.0001. RHO is the density of fabric, assumed to be 1000 kg/m3. Foral is the fraction of NPEOs released by oral contact and it is set at 1. 12
Chemical Footprint
The chemical footprint (ChF) theory integrates chemicals, humans, and ecology by evaluating the fate and toxicity mechanisms of chemical pollutants released into the ecosystem and characterizing the environmental impact of chemical pollutants in terms of their toxicity signature factors and quantities. 21 In 2013, Sala and Goralczyk, based on the chemical footprint theory, combined with the USEtox model recommended by the UNEP-SETAC toxicity, quantitatively characterized the environmental loads generated by chemical pollutants through the toxicity characterization factor in the model. 22 The USEtox model quantifies the impacts of environmental loads by calculating the characteristic factor through simulation of the process of diffusion of a substance into the environment and the impacts on human beings or the ecology after exposure, as shown in equation (3):
where FF denotes the fate factor (day), which represents the duration of exposure of a chemical pollutant to the environment. XF denotes the exposure factor, which in ecotoxicity represents the degree of availability of a chemical pollutant to an organism (dimensionless), and in human toxicity represents the rate at which a chemical pollutant is transferred from the environment to the human body through a series of exposure pathways (day−1). EF denotes the effect factor, representing the different toxic effects of a chemical pollutant on the human body and ecology. The toxicity data used to calculate CF are HC50 data related to EF, which is calculated as follows:
where HC50 is the hazardous concentration of a chemical, indicating the concentration (kg·m−3) at which 50% of species are exposed above the chronic EC50 of the chemical. EC50 is the half-effective lethal concentration, indicating the effective concentration at which 50% of species show a response (e.g. mortality) in a laboratory test. The USEtox model uses the EC50 to determine the relative ecotoxicity of chemicals because it is less affected by experimental conditions. To ensure biodiversity, the USEtox model uses algae, crustaceans, and fish to simulate producers, primary consumers, and secondary consumers in the food chain, and the HC50 is obtained by calculating the geometric mean of the EC50 for a single species from organisms at three trophic levels.
In 2014, Bjørn et al. 23 calculated the chemical footprint of chemicals with the help of the USEtox model and chemical footprint theory. The accounting method of the chemical footprint is shown in equation (5):
where ChF represents the ecological chemical footprint (in PAF·m3·day), f denotes the correction factor between the USEtox model and ChF, with a predefined value of 290, Ei signifies the mass (in kg) of substance i discharged into fresh water.
It is important to mention that this study only focused on the washing process. During this process, chemicals from textile products are released into the aquatic ecosystem within the system boundary. To calculate CF, the USEtox model requires specific input data about the substances, including their chemical identities and associated toxicity. The Estimation Programs Interface Suite was used to estimate physicochemical parameters, including molecular weight (MW), octanol−water partition coefficient (KOW), organic carbon−water partition coefficient (KOC), Henry’s law coefficient at 25°C (KH), vapor pressure at 25°C (Pvap), solubility at 25°C (Sol), and degradation rate in water (KdegW), for the chemical substances. 24 The physicochemical properties of the chemical pollutants are available on the US Environmental Protection Agency’s CompTox chemical board (https://CompTox.epa.gov). HC50 values were predicted for NPEOs based on previous studies using a GBM model with the number of trees = 100 and learning rate = 0.1. To simplify accounting, the declared unit is nine textiles (1 kg per textile). It is assumed that all chemicals released during the wash cycle were discharged into the aquatic environment.
Results and Discussion
Occurrence of Chemicals in Unwashed Textiles
In the investigation of the nine textile samples for their content of APEOs and APs, the concentrations of each compound in the various textiles are summarized in Supplemental Table S2. The results showed that the concentration of APEOs in these samples exceeded the limit requirements of the OEKO-TEX® STANDARD 100, with the highest concentration of the samples reaching 137.6 times the limit requirements. The total concentrations of NPEOs and OPEOs ranged from 183.2 to 1376.0 mg/kg across the study samples, with an arithmetic mean concentration of 513.1 mg/kg. NPEOs were found to be the most abundant of the target analytes, ranging from 182.0 to 1366.0 mg/kg, with an arithmetic mean concentration of 510.4 mg/kg. OPEOs exhibited an arithmetic mean concentration of 2.73 mg/kg. The detection concentration range of sample OPEOs is LoD-10.0 mg/kg. None of the nine clothing samples exhibited detectable amounts of APs.
Since the samples in this study were designed to study the effects of laundering of NPEOs and OPEOs in textiles, we compared the concentrations of NPEOs in textiles from previous studies, as shown in Supplemental Table S3. Notably, in 30–40% of the textile samples, either no or low concentrations (<10 mg/kg) of NPEOs were detected. At these low levels, it is not likely there is intentional use of NPEOs in the manufacturing process. Possible explanations for this include that the NPEO content could arise from contaminated water in the manufacturing process or contamination by other fabrics during transport or storage. Conversely, approximately 50% of the analyzed samples exhibited concentrations ranging from 10–20 up to 400–500 mg/kg (with an average of 100–150 mg/kg), indicative of deliberate usage of NPEOs in the manufacturing process. Moreover, around 5% of the samples revealed NPEO concentrations exceeding 500−1000 mg/kg. These elevated levels could potentially stem from the use of dyestuffs contaminated with high concentrations of NPEOs during the fabric dyeing process. A rough estimation based on information gives an average concentration of 10–100 mg/kg (between the geometric and arithmetic mean). Different textile raw materials have different requirements for the printing and dyeing process, and in turn have different needs for the auxiliaries required in the printing and dyeing process. Fan et al. obtained the detection ratios of APEOs in textiles of different compositions by summarizing and analyzing the textiles of different compositions. The results indicated that cotton and polyester fabrics exhibited the highest detection rates, approximately 45.5%. 25 This was followed by pure nylon fabrics, which had a detection rate of 40.5%, and pure wool fabrics, with a detection rate of 37.5%. The likelihood of detecting APEOs in various raw materials, ranked from highest to lowest, is as follows: pure polyester fabric = pure cotton fabric > pure nylon fabric > pure wool fabric.
The previous studies suggest that AP concentrations are probably not contained in textile articles at considerable concentrations. However, populations are exposed to NPs directly or as metabolites of NPEOs through a variety of everyday products (e.g. detergents, cleaners, pesticides, food packaging, and cosmetics). 26 Women and children are particularly at risk as small children crawling on floors can be exposed to NPEOs and NP present in floor and carpet cleaners.
Chemicals Release During Washing
Figure 1 shows the concentration of APEOs in textiles after multiple washing cycles, revealing a significant decrease in APEO content after washing compared to the initial levels. The removal of NPEOs after three washes ranged from 11.5 to 97.3% and the removal of OPEOs ranged from 59.8 to 100%. This shows how important washing clothes before use is, regarding a minimization of the risk from use. After 20 washes, four of the nine samples still did not meet the requirements of OEKO-TEX® STANDARD 100 (2024 Edition) for APEO content (total APEOs and APs < 100 ppm). In addition, after multiple washes, we found that the concentrations of these four samples did not show a significant declining trend. Therefore, we can draw a preliminary conclusion that washing may not completely remove residual APEOs from clothing.
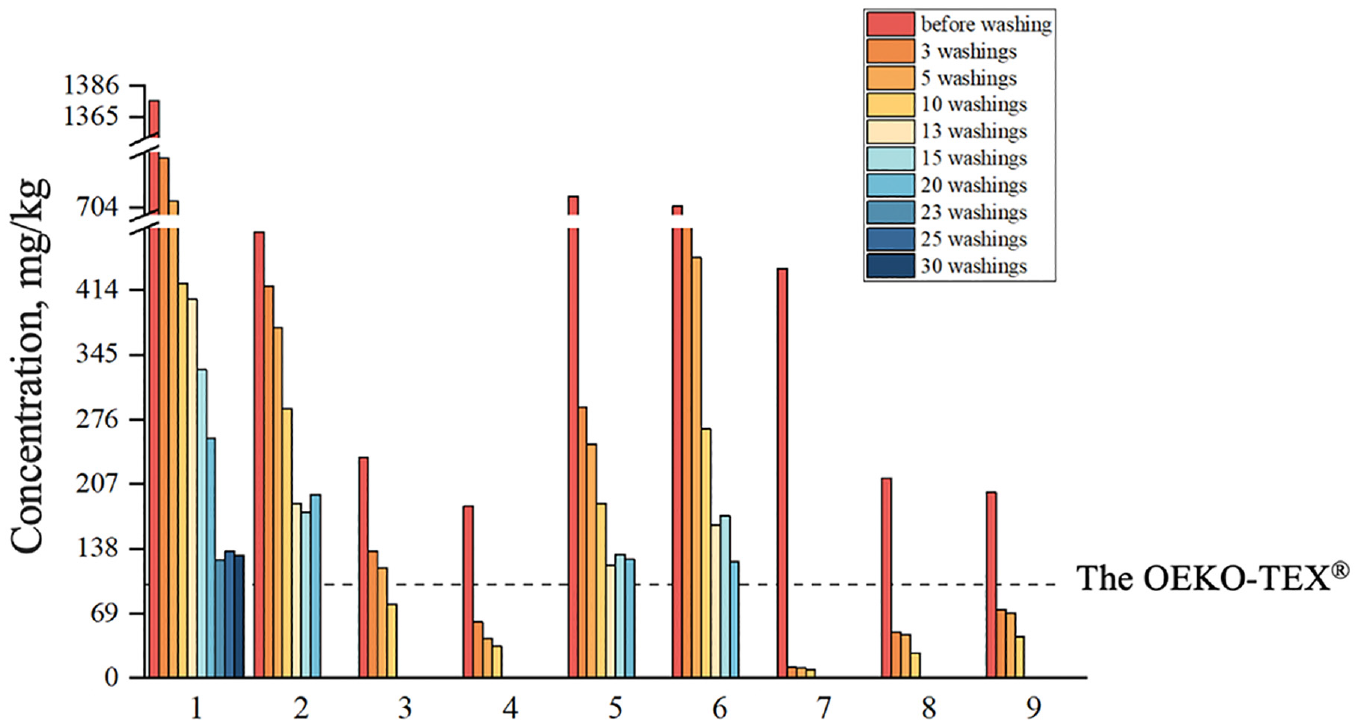
Concentrations (mg/kg) of APEOs (total NPEOs and OPEOs) in the textile samples.
The percentage distribution of APEOs released into the laundry water after the completion of all washing experiments and the residual APEO on textiles are shown in Figure 2. From Figure 2 we can conclude that the release of APEOs from the washing process chemicals ranged from 63 to 98%, with an arithmetic mean of 81%. More soluble chemicals, such as APEOs, are more likely to be released into the laundry water through washing. In a study by the Danish EPA (2013), a single wash removed 25–99.9% of the NPEOs content, while in a survey by the UK EPA (2013), more than 99.9% of the NPEOs were released after two washes.10,11 How much NPEOs are released during the washing process differs between different types of textiles and NPEO contents in the fabric.
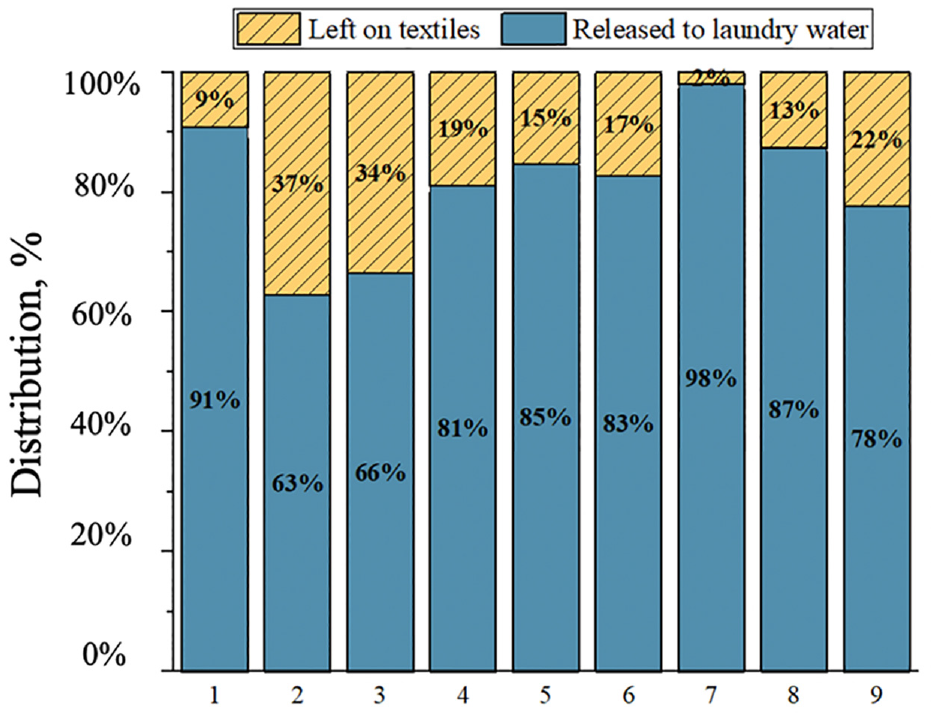
Percentage distribution of APEOs released to laundry water and remaining on textiles.
With a total global fiber production of 116 million tons in 2022, the average concentration of NPEOs in textiles is assumed to be 53 mg/kg, resulting in a total of 6148 tons of NPEOs. 9 Calculation based on NPEO with eight ethoxy units (where the NP/NPEO ratio is 2:5) gives 2459 tons of NPequ annually. Based on the release rate of 81% for NPEOs in our study and the release rate of 82.3% for NP in the literature, it is estimated that the global amounts of NP and NPEOs discharged to wastewater treatment plants as a result of the laundering of textiles are 2024 tons and 4980 tons. 9 Based on the release rate, it is possible to estimate the amount of textiles released into the environment as a result of washing. High release rates mean that more NPEOs and NPs may be released into the environment. Knowledge of these release estimates is essential for the development of effective environmental protection policies. Overall, these data provide a basis for assessing the potential environmental impacts of NPs and NPEOs, which can help guide future environmental protection measures and policy formulation.
Exposure Assessment
APEOs in clothing can enter an infant’s body through dermal absorption and the oral cavity. Figure 3 shows the exposure doses of APEOs via dermal absorption and oral ingestion for infants wearing unwashed and washed clothing. Total dermal absorption and oral ingestion of APEOs from unwashed textiles in infants ranged from 4.19×10−3 to 3.94×10−2 mg/kg/day, and the total exposure dose of washed APEOs ranged from 2.00×10−4 to 3.77×10−3 mg/kg/day. Oral ingestion by infants accounted for 35.7–53.5% of the total exposure dose. The total exposure dose for infants wearing unwashed textiles is approximately 2.7–50.1 times that of washed textiles.
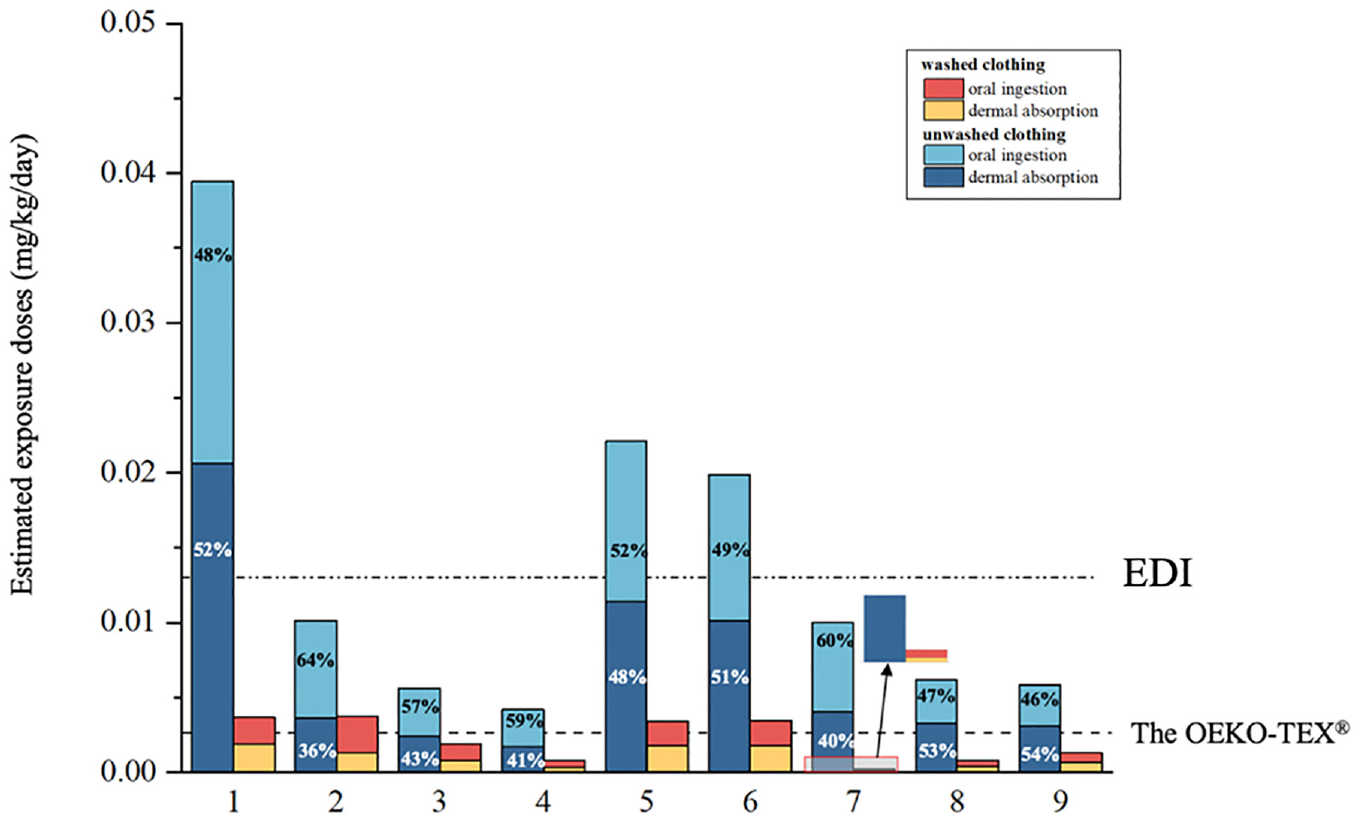
The exposure doses of APEOs via dermal absorption and oral ingestion for infants wearing unwashed and washed clothing.
The limit value set by OEKO-TEX® (100 mg/kg) was used to calculate the exposure dose for the same scenario. The exposure doses calculated by OEKO-TEX® and the health limit values (EDI) given by the Institute of Food Safety and Toxicology Danish Veterinary and Food Administration (DVFA) were compared with our estimated exposure doses. 27 Based on the health limit values provided by DVFA, three of the nine unwashed samples had calculated exposure doses exceeding the limit value requirement, with exposure doses ranging from 1.99×10−2 to 3.94×10−2 mg/kg/day. All samples after washing met the health limit values provided by the DVFA.
In the previous study, calculations were made of the average daily uptake of NP and NPEOs when a 2-year-old child (10 kg) wears either unwashed or washed clothes. The report shows that the total amount of NPEOs ingested by 2-year-old children due to wearing unwashed new T-shirts is 3.39×10−3 mg/kg/day, with a total amount of NPs ingested of 1.21×10−3 mg/kg/day. After one wash, the total amount of NPEOs ingested is 2.54×10−3 mg/kg/day, with a total amount of NPs ingested of 9.07×10−4. This result is similar to the exposure dose calculated in this study.
Chemical Footprint
Based on the results shown in Figure 4, it can be concluded that the total ChF produced by the nine samples after washing was 114 PAF·m3·day, with Sample 1 contributing the largest chemical footprint with 36.6 PAF·m3·day, or 32% of the total ChF, followed by Sample 5 at 19.5 PAF·m3·day, or 17% of the total ChF. The main reason for this result is the high initial residual concentration of APEOs in the samples and therefore the large chemical footprint after washing. In Figure 4, it is shown that NPEOs account for more than 99% of the total ChF and are the most dominant contaminants in APEOs. However, NPEOs may not necessarily have such a high percentage in other types of textiles due to differences in production processes and the use of additives. Since there are no studies quantifying the ecological impacts of APEOs in textiles after washing, we compared the chemical footprint results from unpublished articles. The 1000 kg polyester fiber product produces about 140 PAF·m3·day from NPEOs at the dyeing and finishing stage, while the nine samples (9 kg) in this study produced a chemical footprint of 114 PAF·m3·day. This indicates that the chemical footprint produced by the washing of the exceeding samples is quite large, and the ecological impacts on the environment should not be ignored.
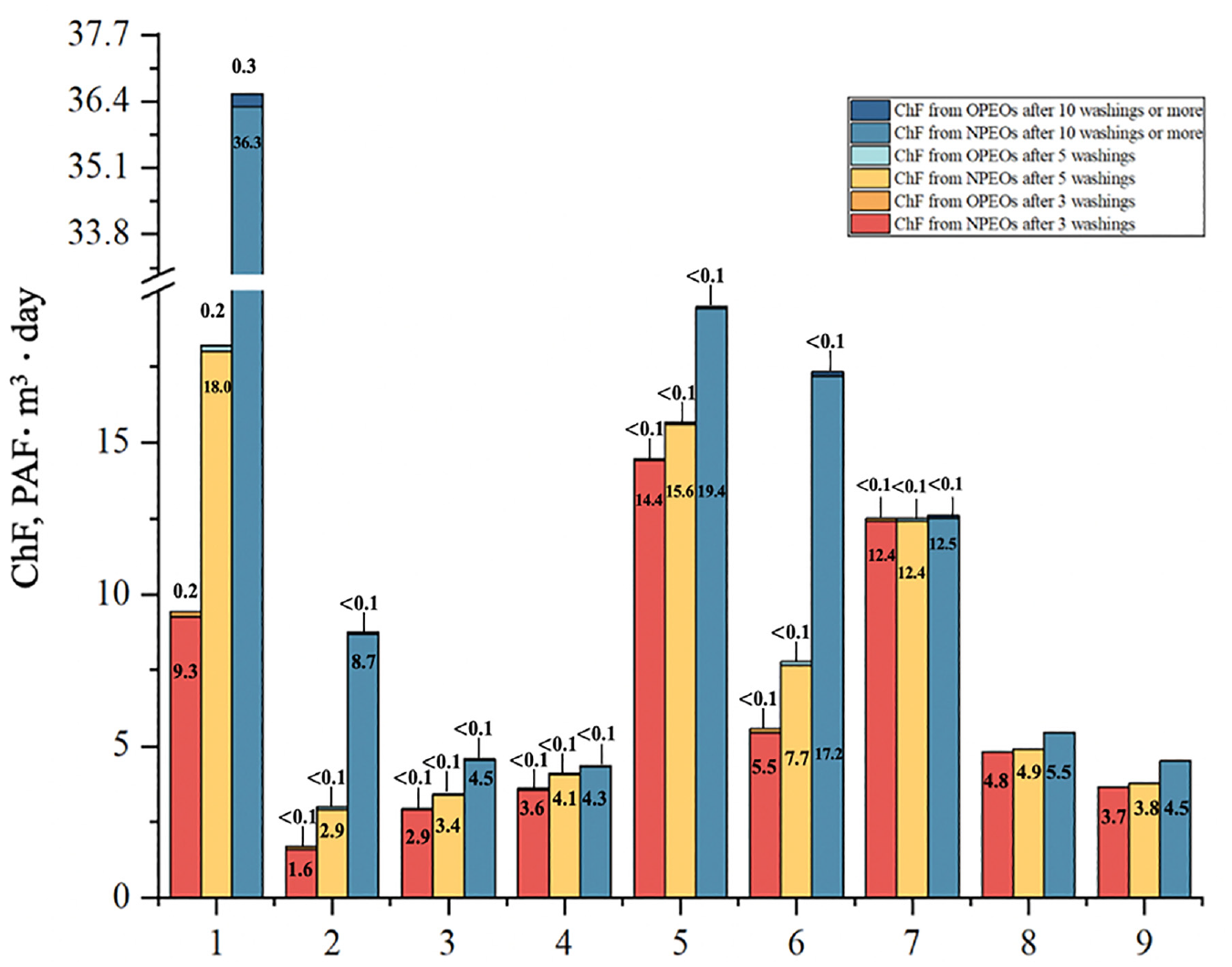
Chemical footprint of NPEOs and OPEOs in the washing process.
Conclusions
In this study, we quantified the human health and ecological impacts of APEOs for the first time by combining human exposure model and chemical footprint approaches. We investigated the occurrence of APEOs in nine samples with initial concentrations ranging from 183.2 to 1376.0 mg/kg. We subjected the samples to 3, 5, 10, and even more washing experiments, which yielded a release rate of 63–98% of APEOs, and washing did not completely remove APEOs from the textiles. This study points out that washing may be one of the important sources of release of APEOs into the environment. Chemicals in textiles through dermal contact may be a potential route of chemical exposure for humans, and the potential route of oral ingestion by infants cannot be ignored. Our study can inform public health policy and environmental protection components and provide a basis for future guidelines and database development.
Supplemental Material
sj-docx-1-aat-10.1177_24723444241288273 – Supplemental material for From Clothing to Laundry Water: Assessing Human Exposure and Chemical Footprint of Alkylphenol Ethoxylates
Supplemental material, sj-docx-1-aat-10.1177_24723444241288273 for From Clothing to Laundry Water: Assessing Human Exposure and Chemical Footprint of Alkylphenol Ethoxylates by Zhaoxia Guo, Yeju Cheng, Xiang Ji, Yue Sun, Feng Li, Qizheng Li, Lisha Zhu and Laili Wang in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ24E030006, Science Foundation of Zhejiang Sci-Tech University (ZSTU) under Grant No. 22202009-Y.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.