Abstract
Introduction
WS-5 is a flavor ingredient that provides a cooling sensation similar to that of menthol but without the characteristic menthol flavor. In the available toxicological evaluations of WS-5 there was no data specific to inhalation exposure.
Methods
A 90-days nose-only inhalation study was conducted using Sprague Dawley rats exposed to Filtered air, propylene glycol/glycerin (40/60 by weight; vehicle control), or one of three WS-5 concentrations (0.2, 0.4, or 0.8% by weight) to assess the inhalation toxicity of WS-5. The study design was consistent with the Organization for Economic Development and Cooperation test guideline 413 and included a 28-day and 6-week recovery sacrifices, in addition to the 13-week assessment.
Results
Food consumption and body weights were unaffected by WS-5 exposure compared to vehicle control or Filtered air. No WS-5 exposure related alterations were observed in serum chemistry, hematology, coagulation, urinalysis or bronchoalveolar lavage fluid cytology and clinical chemistry. Macroscopic examinations and terminal organ weights revealed no observations associated with exposure to WS-5.
Discussion
Histopathology findings of alveolar hemorrhage and mixed cell infiltration at the 28-day necropsy were either not present in the WS-5 exposed groups or were present at similar frequencies in the vehicle control and/or Filtered air groups at the 13-weeks necropsy and thus, were not considered to be vehicle control or WS-5 related.
Conclusion
Based on the absence of adverse effects, the no-observed-adverse effect concentration was considered to be 2.5 mg/L of aerosolized 0.8 w% WS-5 that achieved a mean measured exposure concentration of approximately 18 μg WS-5/L
Introduction
WS-5 has a chemical name of N-((Ethoxycarbonyl)methyl)-p-menthane-3-carboxamide with a CAS Number of 68489-14-5 (FEMA number 4309) and was developed in the 1970s by Wilkinson Sword, Ltd as an alternative to menthol for toiletries and cosmetic products. 1 Subsequently, WS-5 and similar compounds have been called synthetic cooling compounds and have been used in numerous different types of consumer products. 1 In comparison to menthol, WS-5 has four times the cooling strength with a similar duration of cooling effect, 1 but without the characteristic menthol taste.2,3 Cell receptor binding studies indicate that the cooling effect of WS-5 occurs via the transient receptor potential (TRP) channels, specifically TRPA-1 and TRPM-8. 4
Recently, several authors have identified various synthetic cooling compounds in Electronic Nicotine Delivery Systems (ENDS) and noted the lack of inhalation toxicology data for these chemicals.2,5–7 The available toxicology data on synthetic cooling compounds, including WS-5, has been summarized by the European Food Safety Authority,8–10 Flavor Extract Manufacturers Association and Joint FAO/WHO Expert Committe on Food Additives.11–13 None of these summaries and toxicological evaluations of synthetic cooling compounds included inhalation toxicology studies. Recently Wu et al., 14 published results of a 28-days nose-only inhalation study using an ENDS device for aerosol generation with an e-liquid containing 10% WS-23. The study used an Organization for Economic Cooperation (OECD) compliant study design and was conducted in accordance with Good Laboratory Practice guidelines. Although there were sporadic statistically significant differences in a few endpoints among the three WS-23 dose groups compared to the vehicle control group, the authors determined a No Observed Adverse Effect Level (NOAEL) to be at least 342.85 mg/m3, the highest dose tested. 14 The objective of this study was to evaluate the toxicity of WS-5 in a 90-day nose-only rodent inhalation study.
Materials and methods
The 90-day nose-only inhalation study was conducted using the standard OECD guideline 413 15 study design (90-days [13-weeks] of exposure with a 28-day [4-weeks] time point followed by 42-day [6-weeks] of recovery) and was conducted in compliance with U.S. Good Laboratory Practices (21 CFR Part 58) by a contract research organization (Charles River Laboratories, Ashland, Ashland, OH). This study is being published in accordance with the Animal Research: Reporting of In Vivo Experiments guidelines. 16
Animals
All care and use of Sprague-Dawley rats (Crl:CD[SD]) were performed in facilities that were accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC International), and all animal experiments were approved by the Charles River Ashland Institutional Animal Care and Use Committee. Male and Female (nulliparous) Sprague-Dawley rats bred under specific pathogen-free conditions were obtained from a commercial supplier (Charles River Laboratories, Raleigh, NC). Animals were individually identified via subcutaneous transponders and housed two to four animals (same sex) per polycarbonate cage (Bed-o’ Cobs®, ScottPharma Solutions) during non-exposure periods. Certified rodent diet (PMI Nutrition International, LLC) and fresh drinking water (municipal tap water, reverse osmosis and ultraviolet irradiated) was supplied ad libitum to each cage. Enrichment items (e.g., treats, gnawing device, and/or nesting material) were provided in each cage. A 12-hr. Light/12-hr. Dark cycle was used for the animal rooms. Laboratory air was fresh filtered and conditioned (20–26°C; 30–70% relative humidity). The animal housing set-up was in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council, 2011). Animals were acclimated for a minimum of 6 days to laboratory conditions and were subsequently acclimated to the nose-only exposure tubes over 4 days. No food or water was available during acclimation to the nose-only exposure tubes or inhalation exposures. Animals were approximately 7 weeks of age at the beginning of exposures.
Test article
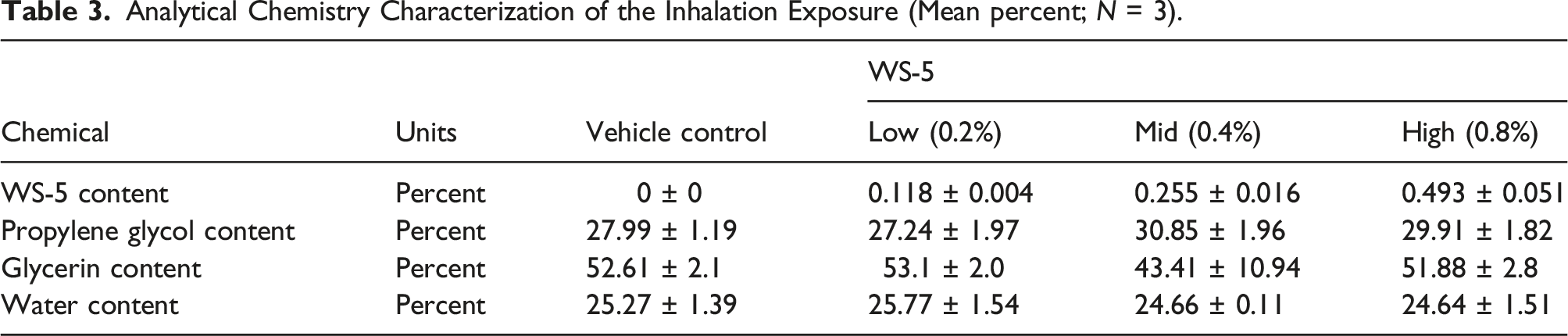
WS-5 is a white powder with a molecular weight of 269.38, a melting point of 80–82°C, a boiling point of 151°C and a vapor pressure of 0.00023 Pa at 25°C. WS-5 is soluble in propylene glycol (11.5 gm/100 gm at 20°C) and insoluble in water.17,18 Based upon these physical data, WS-5 was expected to be in the particle phase of the aerosol. A single lot of WS-5 (100% purity; Vigon, International, Inc.) that was within its shelf-life period was used for all WS-5 formulations. Each of the WS-5 formulations consisted of a base of a single lot of USP grade propylene glycol and USP grade, Food Chemical Codex compliant glycerin that were mixed in a 40%/60% ratio (w/w). WS-5 was added to the 40%/60% base of propylene glycol/glycerin at 0.2%, 0.4%, and 0.8% on a weight basis (w/w). At the target aerosol concentration of 2.5 mg/L this equates to 5.0, 10.0, and 20.0 μg/L of WS-5 for the low, mid and high exposure groups, respectively. The highest inclusion level was limited by solubility of WS-5, so all WS-5 formulations were heated to 38 ± 1.2°C in a water bath and stirred during use in their aerosol generation systems to maintain WS-5 solubility. The vehicle control consisted of the base formulation; 40%/60% (w/w) propylene glycol/glycerin without any WS-5. When not being used for aerosol generation, formulations were stored at room temperature (18–24°C). A 40-day room temperature stability study of the 0.2% and 0.8% WS-5 formulations was performed. WS-5 concentration was measured using a validated gas chromatograph with a flame ionization detector (GC-FID) technique. After 40-days the mean post-storage WS-5 concentrations were 96.3% and 99.0% of the pre-storage values (N = 2), respectively. These results were within the a-priori acceptance criteria of ±10% of the pre-storage value. Vehicle control and WS-5 formulations were made weekly.
Exposures
Overall study design.
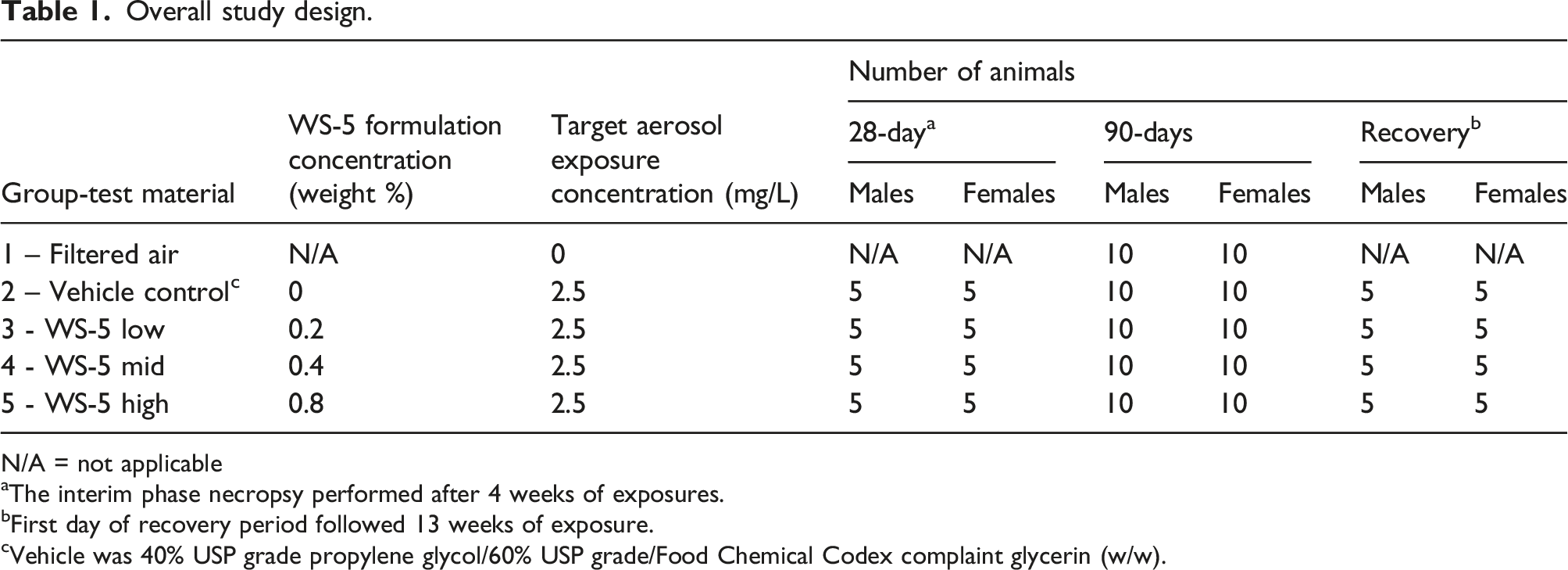
N/A = not applicable
aThe interim phase necropsy performed after 4 weeks of exposures.
bFirst day of recovery period followed 13 weeks of exposure.
cVehicle was 40% USP grade propylene glycol/60% USP grade/Food Chemical Codex complaint glycerin (w/w).
To assess exposure to vehicle control and WS-5 formulations, validated analytical chemistry assays were used to measure WS-5, propylene glycol and glycerin using GC-FID and water using Karl Fischer Titration analysis in the formulations and filter samples collected twice weekly from the nose-ports of the nose-only inhalation towers used for each group. Actual exposure concentrations were determined gravimetrically at least five times daily by collecting aerosol on pre-weighed 44 mm glass fiber filters (Whatman, GE Healthcare, UK) and dividing the accumulated mass by the sample volume. The aerosol particle size distribution (mass median aerodynamic diameter – MMAD and geometric standard deviation – GSD) was determined weekly using a cascade impactor (InTox, Moriarty, NM; model 02-001-A in brass).
Biological endpoints
Twice daily in-life observations (before and after exposure with animals in their cages) were performed on exposure days and once per day on non-exposure days. Detailed clinical observations were conducted weekly for all exposed animals. Body weights (non-fasted) and feed consumption were measured weekly for each animal upon arrival through termination. Ophthalmic examinations were performed by a board-certified veterinary ophthalmologist prior to exposure, during week 4 and 13 of exposure, and near the end of the recovery period.
Confirmation of exposure was performed by measuring the amount of propylene glycol in rat plasma using a validated analytical assay 28 for all exposure groups, except Filtered Air, on days 3, 28, and last day of exposure. Blood samples were obtained within 10 min post exposure from the recovery group animals (5 animals/sex/group) via jugular vein. An anticoagulant tube (K2EDTA) was used to collect the plasma, which was gently mixed and centrifuged within 2-h of collection. The resultant plasma was transferred to a polypropylene tube and stored at −70°C until analysis.
Respiratory physiology measurements (tidal volume and frequency) were measured twice during the study (days 43–50 and 82–90) using a single-chamber restraint plethysmograph (Buxco Electronics, Inc., Wilmington, NC). After 42-days of exposure five animals/sex/group from the vehicle control and high (0.8%) WS-5 groups were assessed. In the last week of exposure (days 82–90), five animals/sex/group in all exposure groups except Filtered Air were evaluated. Baseline measurements started 10 min prior to exposure and measurements continued for 30 min of exposure with only the last 15 min used for data analysis. Data acquisition logging rate was set to 5 s and respiratory data were sampled at 500 Hz. Any data indicating more than 350 breaths/minute or a tidal volume of greater than 5.0 mL was considered physiologically unlikely and excluded from analysis. Minute volume was calculated as the product of the measured tidal volume and respiratory frequency.
Terminal endpoints
Animals were fasted for at least 8 h prior to blood collection while housed in metabolism cages for urine collection. Blood was collected via the jugular vein and placed into tubes with potassium K2EDTA for hematology, and tubes with no anticoagulant for clinical chemistry. Blood was collected from anesthetized animals (using isoflurane via inhalation) into tubes with sodium citrate for coagulation parameters at the time of euthanasia via the vena cava. The euthanasia was completed by exsanguinating the anesthetized animals.
Urinary volume, pH, and specific gravity as well as glucose, ketone and protein concentration were measured. The hematology endpoints included red blood cell indices (mean corpuscular hemoglobin concentration, mean corpuscular volume, red blood cell and reticulocyte count and hematocrit), platelet count, mean platelet volume, white blood cell count, differential count, as well as coagulation parameters (function included prothrombin time, activated partial thromboplastin and fibrinogen). Clinical chemistry endpoints included: Total Protein, Albumin, Globulin, Albumin/Globulin ratio, Alkaline phosphatase, Alanine aminotransferase, Aspartate aminotransferase, Gamma glutamyltransferase, Cholesterol, Creatine kinase, Creatine, Glucose, Triglycerides, Total bilirubin Direct and Indirect bilirubin, Sodium, Calcium, Potassium, Chloride, Phosphorus and Urea Nitrogen.
Three bronchoalveolar lavages (BALF) were performed on the right lung of all animals after lung weight was recorded and before the lungs were inflated with fixative. The supernatant of the first lavage was evaluated for protein concentration, lactate dehydrogenase, and alkaline phosphatase enzyme activities using automated methods. The cell pellets from the three lavages per animal were combined and resuspended for counting. Total cell counts were obtained using a hemocytometer (Hausser Scientific, Pa.). For differential counts (cytology), at least 200 cells per slide (cytospin preparation) were identified (if possible) as macrophages, neutrophils, lymphocytes, eosinophils, basophils, or others.
Macroscopic evaluation for gross lesions was conducted on all animals at all necropsies (Supplemental Table 1). Organ weights (absolute) were obtained for all organs (Supplemental Table 1) at each necropsy (after 28 and 90 days of exposure and following the 42-days of recovery). Paired organs were weighed together. Organ weight to final (fasted) body weight and organ weight to brain weight ratios were calculated from animals at each scheduled necropsy. After 29 and 90 days of exposure histopathological evaluation was performed on respiratory tract tissues (including six different sections of the nose from oropharynx to nasopharynx, larynx, trachea, main carina-intersection of trachea and two major bronchi, extrapulmonary bronchus, and lung) from all groups and organs from the high WS-5 dose (0.8%) group (Supplemental Table 1) by a board-certified veterinary pathologist using a 5-level severity grade scale of minimal, mild, moderate, marked and severe. If histopathological examination of the high WS-5 dose (0.8%) group after 90-days of exposure revealed concerns, organs from the low (0.2%) and mid (0.4%) WS-5 groups were examined. After recovery, histopathology was only performed on animals exhibiting gross lesions and on potential target tissues.
Statistical analysis
The use of statistical comparisons between WS-5 groups and vehicle control was performed to indicate where toxicologically significant differences were most likely to be present. A statistical difference between groups may or may not be a toxicologically significant difference, depending upon expert interpretation which included analysis of factors such as dose-response and coherence among findings. The 5% level (p < 0.05) for statistical significance was used throughout the evaluations. SAS®, ver. 9.4 (SAS International, Cary, NC) was used for statistical analysis.
In general, means, standard deviations (or % coefficient of variation or standard error, when deemed appropriate), ratio, percentages, numbers, and/or incidences were reported depending on the dataset.
The pairwise comparisons for statistical analysis were: Filtered Air versus Vehicle Control; Low dose WS-5 (0.2%) vs. Vehicle Control; Mid dose WS-5 (0.4%) vs. Vehicle Control; and High dose WS-5 (0.8%) vs. Vehicle Control. Any group with less than three observations were excluded from analysis. Levene’s test was used to assess the homogeneity of group variances. The groups were compared using an overall one-way ANOVA F-test if Levene’s test was not significant or the Kruskal-Wallis test if it was significant. If the overall F-test or Kruskal-Wallis test was significant, then pairwise comparisons using two sided tests were conducted using Dunnett’s or Dunn’s test (equivalent to Wilcoxon Rank-Sum test in Nevis 2012 tables), respectively.
Respiratory physiology data were tabulated within each summary time interval and the arithmetic mean (Mean), number of subjects (N), least squares mean (LS Mean), and standard error of the LS Mean (LSM SE) determined. The effect of treatment during each analysis segment was evaluated using an analysis of covariance. Factors in the model included treatment and the covariate (average of 10 min prior to exposure). If the treatment main effect was significant, step-down adjusted t-tests were used to compare each active treatment to the vehicle control treatment using the overall segment means. Step-down simulated adjustment was used to adjust t-tests for pair-wise comparisons. All pair-wise comparisons were evaluated at the 0.05 level of significance, using two-tailed tests.
Results
Characterization of inhalation exposures
Characterization of inhalation exposures (mean ± SD).
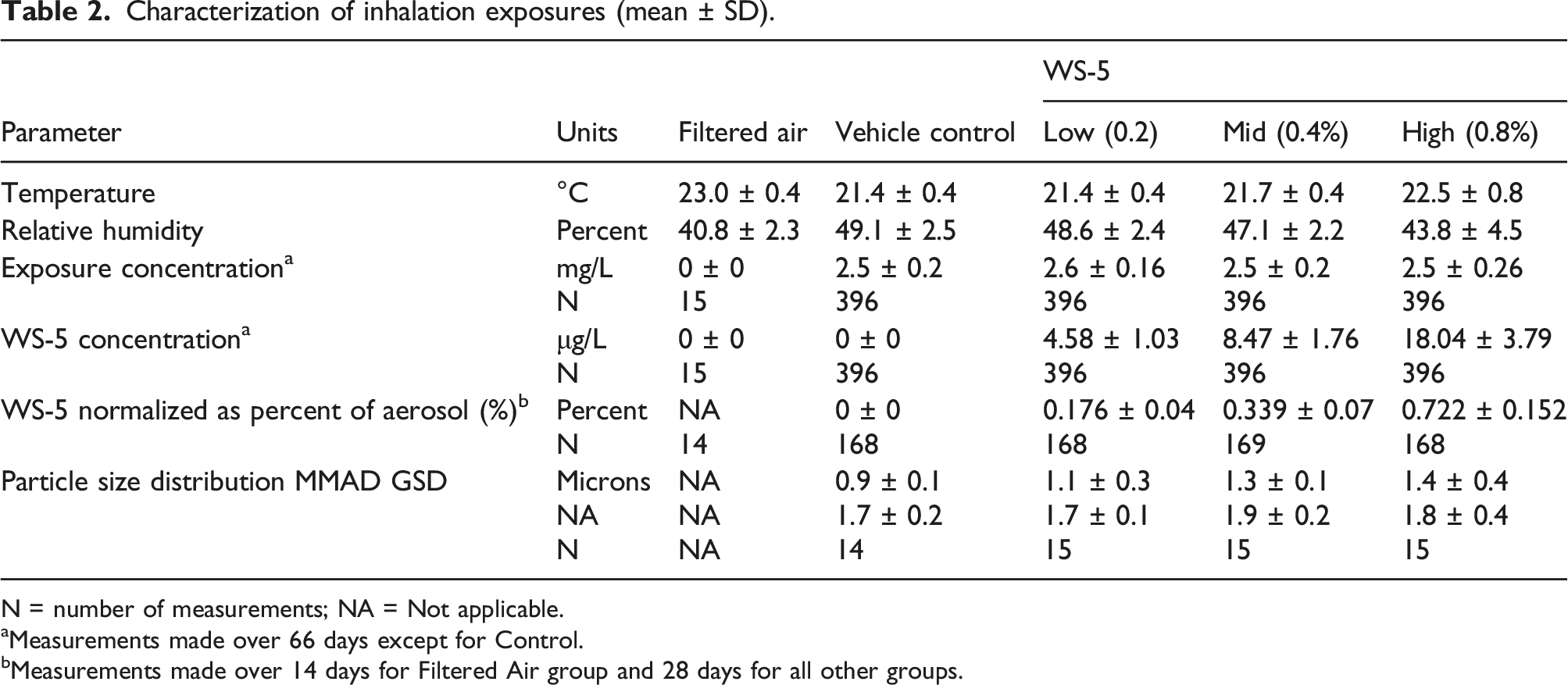
N = number of measurements; NA = Not applicable.
aMeasurements made over 66 days except for Control.
bMeasurements made over 14 days for Filtered Air group and 28 days for all other groups.
Analytical Chemistry Characterization of the Inhalation Exposure (Mean percent; N = 3).
Rat Plasma Propylene Glycol Concentrations (μg/ml) on Days 3, 28 and 91 of Exposures. All values represent the mean of five replicates unless otherwise noted.
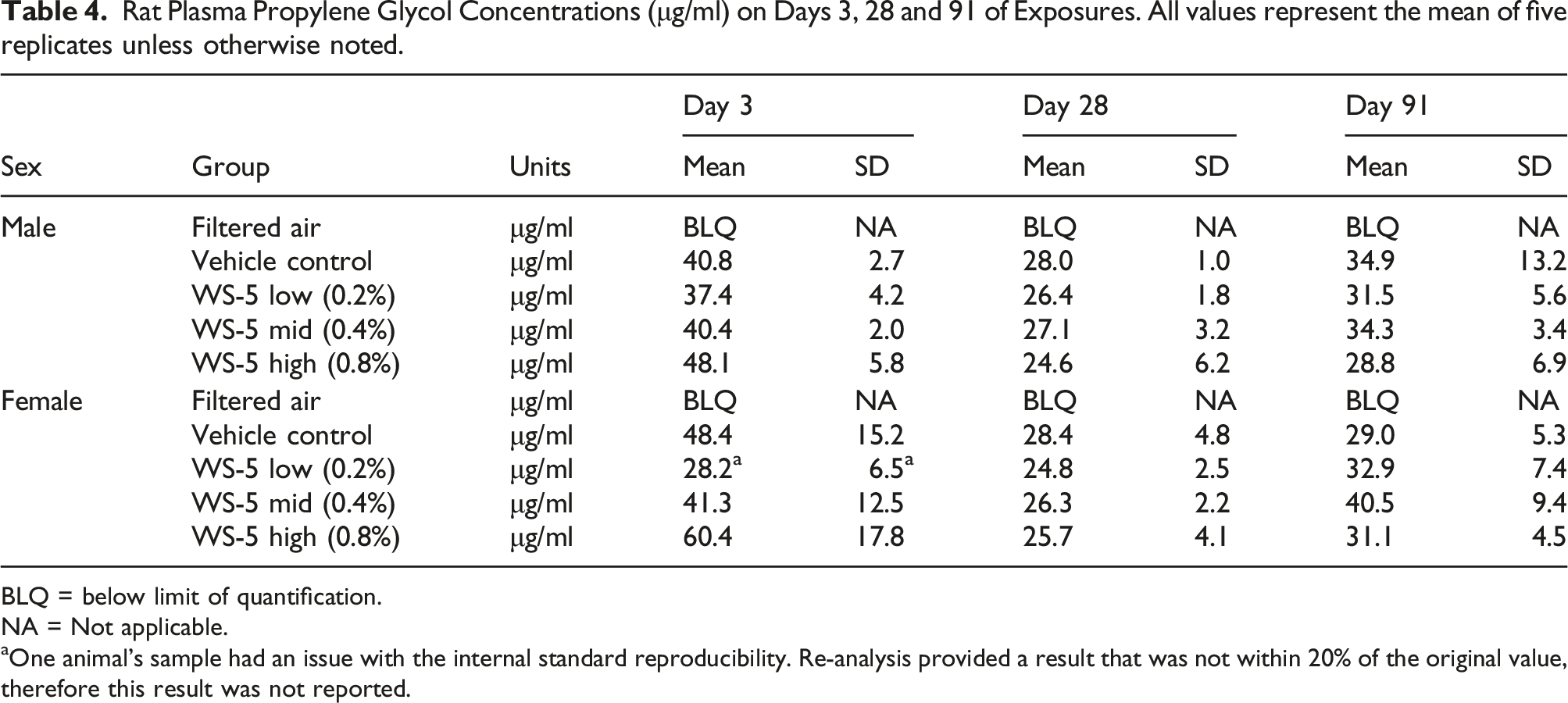
BLQ = below limit of quantification.
NA = Not applicable.
aOne animal’s sample had an issue with the internal standard reproducibility. Re-analysis provided a result that was not within 20% of the original value, therefore this result was not reported.
Mortality/morbidity/in-life observations
All animals survived to their scheduled necropsies. There were no WS-5-related clinical observations. All clinical observations noted were considered incidental as they were either, only noted in a single animal, were of similar incidence in Filtered Air, vehicle control, and WS-5-exposed animals, and/or were common findings for laboratory rats of this age. No ophthalmic lesions indicative of toxicity were observed in any of the WS-5 exposed groups. All ophthalmic findings observed were typical in prevalence and appearance for laboratory rats of this age and strain.
The only statistically significant difference in body weights were for female rats in the recovery group that had been exposed to the high level (0.8%) of WS-5 (Figure 1). These differences were considered to be due to biological variability and not WS-5-exposure related due to lack of a dose-response trend with the mid and low dose groups (0.4% and 0.2% WS-5, respectively). The body weight data were consistent with the food consumption data (Supplemental Tables 2 and 3). Mean bodyweight as function of exposure day for males (top panel) and females (bottom panels).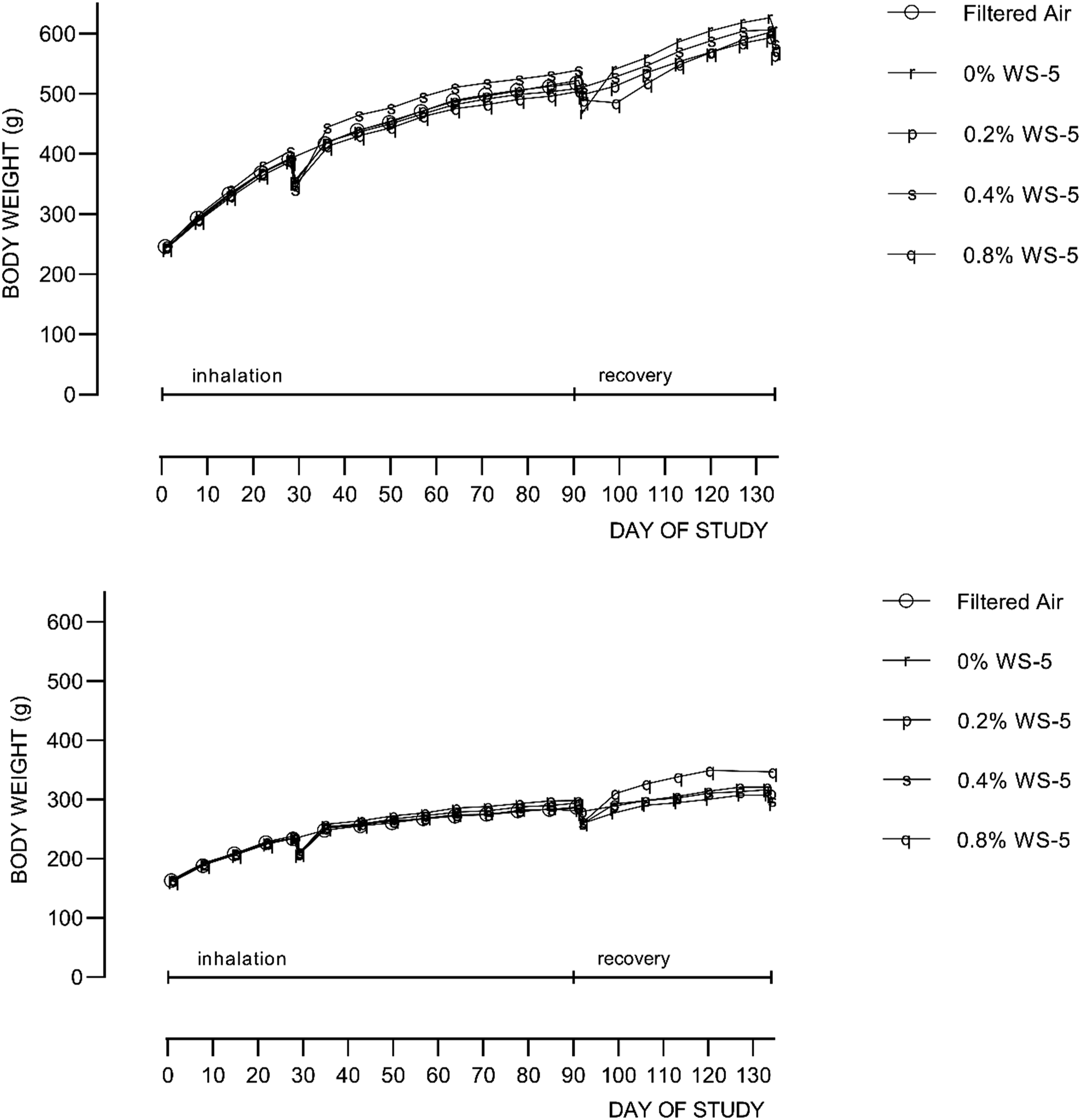
Mean (±SE) respiratory physiology measurements with Least Squares Mean (LS Mean ± SE) with five replicates unless otherwise noted. Bold values are statistically significantly different than Vehicle Control.
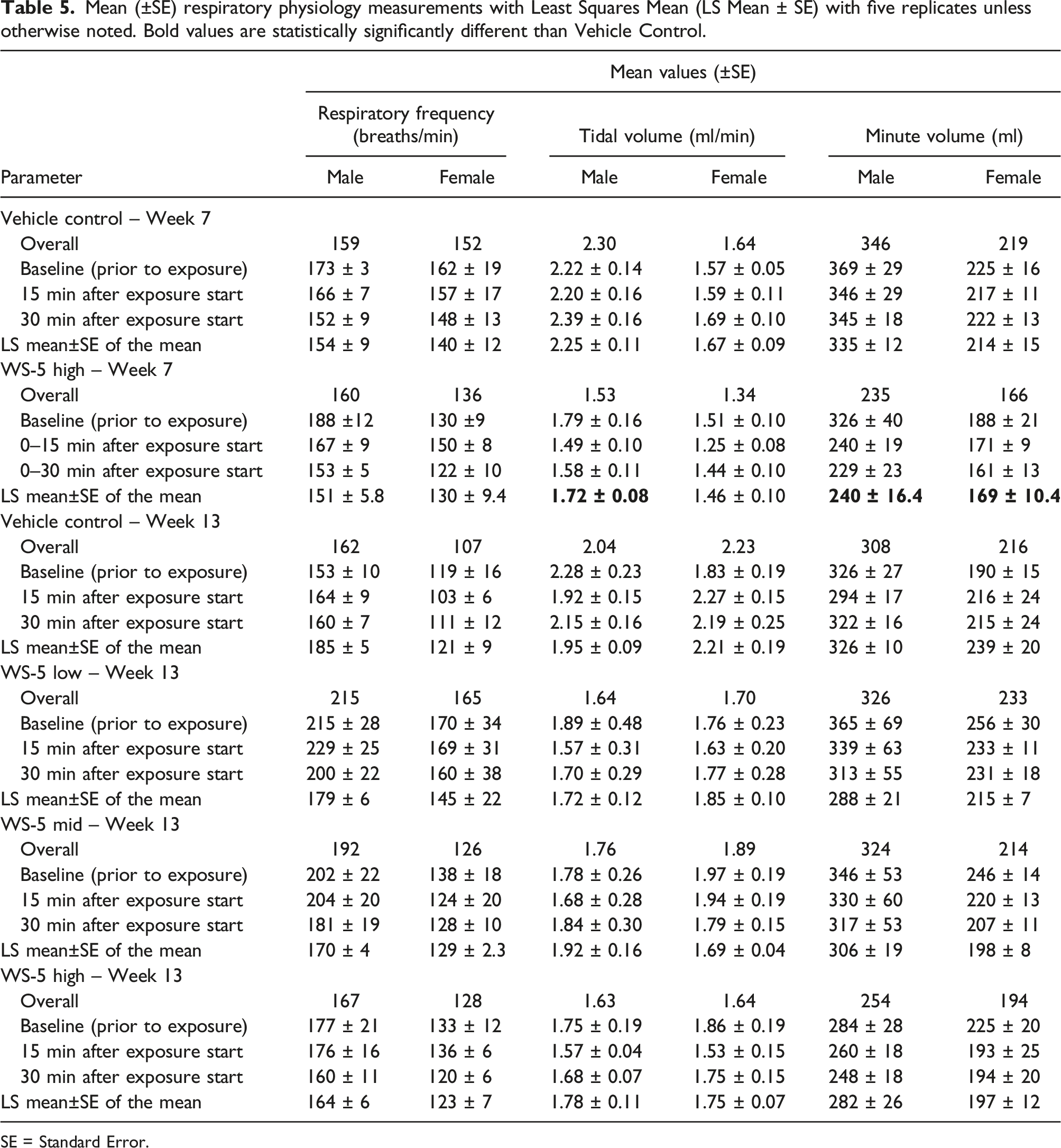
SE = Standard Error.
Terminal endpoints
Although there were several differences (statistically significant and non-statistically significant) in hematology, clinical chemistry, and urinalysis, these were sporadic in nature, consistent with biological variation with no consistency across doses, sexes or time points (Data are presented in the Supplemental Tables 4-17). For example, there was a statistically significant difference in eosinophil levels measured in Male rats at after 28 days of exposure for the low WS-5 (0.2%) exposure group compared to vehicle control, with no dose response evident in the mid or high WS-5 exposure groups (0.4 and 0.8%, respectively). There was also a non-statistically significant lower neutrophil count in the high WS-5 (0.8%) group females compared to vehicle control at after 28 days of exposure, but these were considered unlikely to be associated with WS-5 exposure due to a few higher individual animal counts in the vehicle control group and no dose response relationship. There was also a single female rat in the high WS-5 (0.8%) exposure group after 28 days of exposure with a moderately decreased platelet count with minimal (1+) platelet clumping, however due to the single incidence and presence of platelet clumps, the decreased platelet count was considered unrelated to WS-5 exposure. As another example of a non-statistically significant difference, two female rats in the high WS-5 (0.8%) exposure group were observed with increased urine volume and markedly decreased urine specific gravity. These changes lacked correlating clinical chemistry findings, were most consistent with water contamination of the urine sample and were therefore considered unrelated to WS-5 exposure.
There were no statistically significant changes in BALF cytology, total protein, lactate dehydrogenase, or alkaline phosphatase for any WS-5 exposure group at any necropsy (Supplemental Tables 18-23). After 90 days of exposure and recovery necropsies, many of the BALF cytospin smears collected contained numerous extracellular bacteria. This was consistent with contamination at necropsy; however, the presence of bacteria did not compromise the cytological evaluation.
No statistically significant differences in absolute organ weight, organ weight normalized to body or brain weight were found at the necropsy after 28-days of exposure (Supplemental Tables 24-41). After 90-days of exposure, there were two statistically significant differences from vehicle control group mean value for female rats in the absolute weight of the thyroid/parathyroid, one for the Filtered Air group and the other for the low WS-5 (0.2%) exposure group, however there was no difference between these two group mean values. There was also no dose response with the mid or high WS-5 exposure group (0.4 and 0.8%, respectively) and no correlation with histopathology (Supplemental Table 31). When organ weights were normalized to body or brain weight, the statistically significant difference shifted to the mid WS-5 (0.4%) exposure group for the thyroid/parathyroid, again with no dose response and no correlation to histopathology (Supplemental Table 33). At the recovery necropsy, there were no statistically significant differences in absolute thyroid/parathyroid weight or when normalized to body weight or brain weight (Supplemental Tables 37, 39, and 41). There was a statistically significant decrease in brain weight when normalized to body weight for females in the high WS-5 (0.8%) exposure group, however there was no dose response nor any correlation to histopathology (Supplemental Table 39). Therefore, the few organ weight differences observed were considered incidental and unrelated to exposure to WS-5 at any of the necropsies.
Summary of respiratory tract histopathology data after 28-days of exposure in rats.
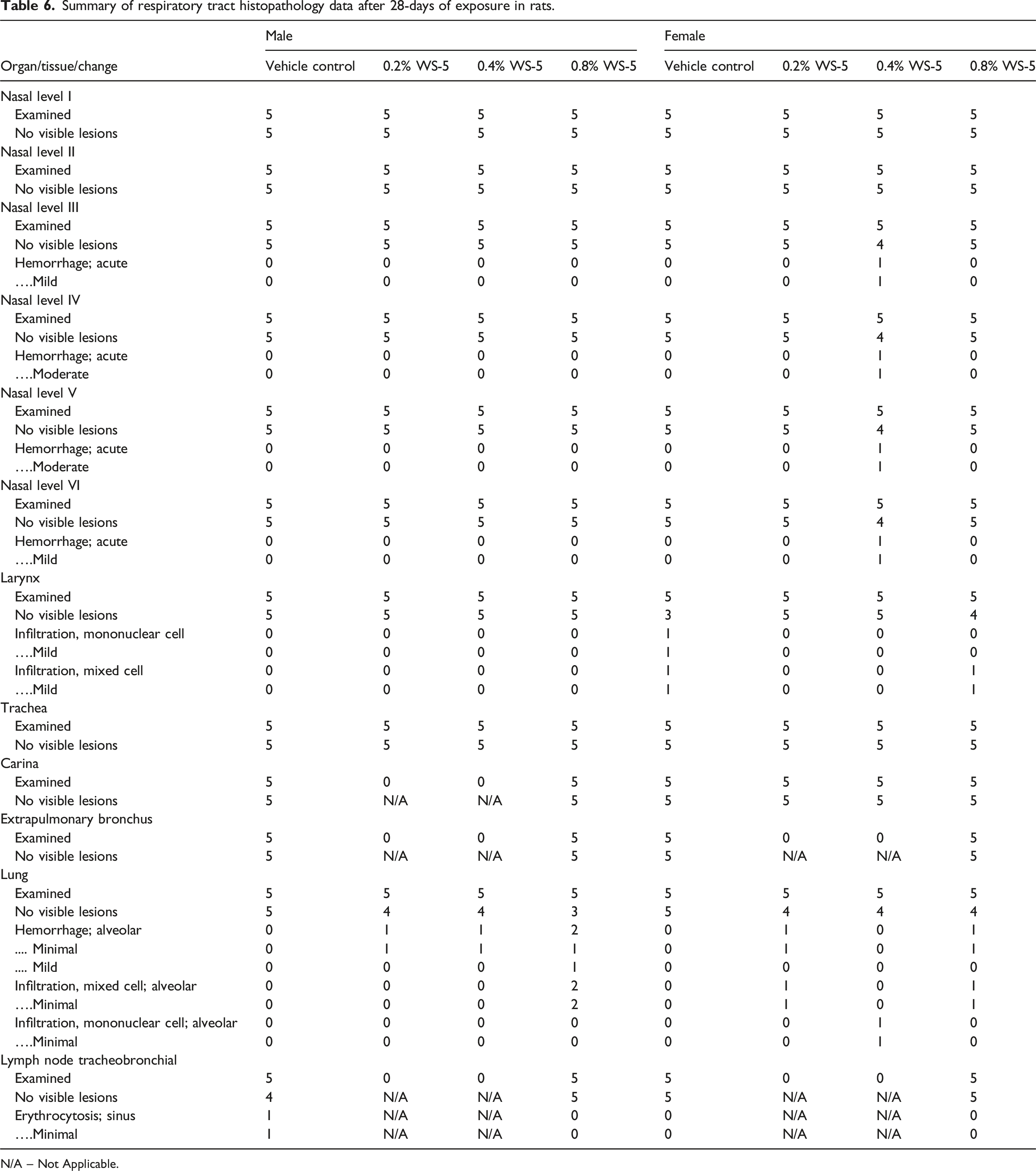
N/A – Not Applicable.
Summary of respiratory tract histopathology data in rats after 90-days of exposure.
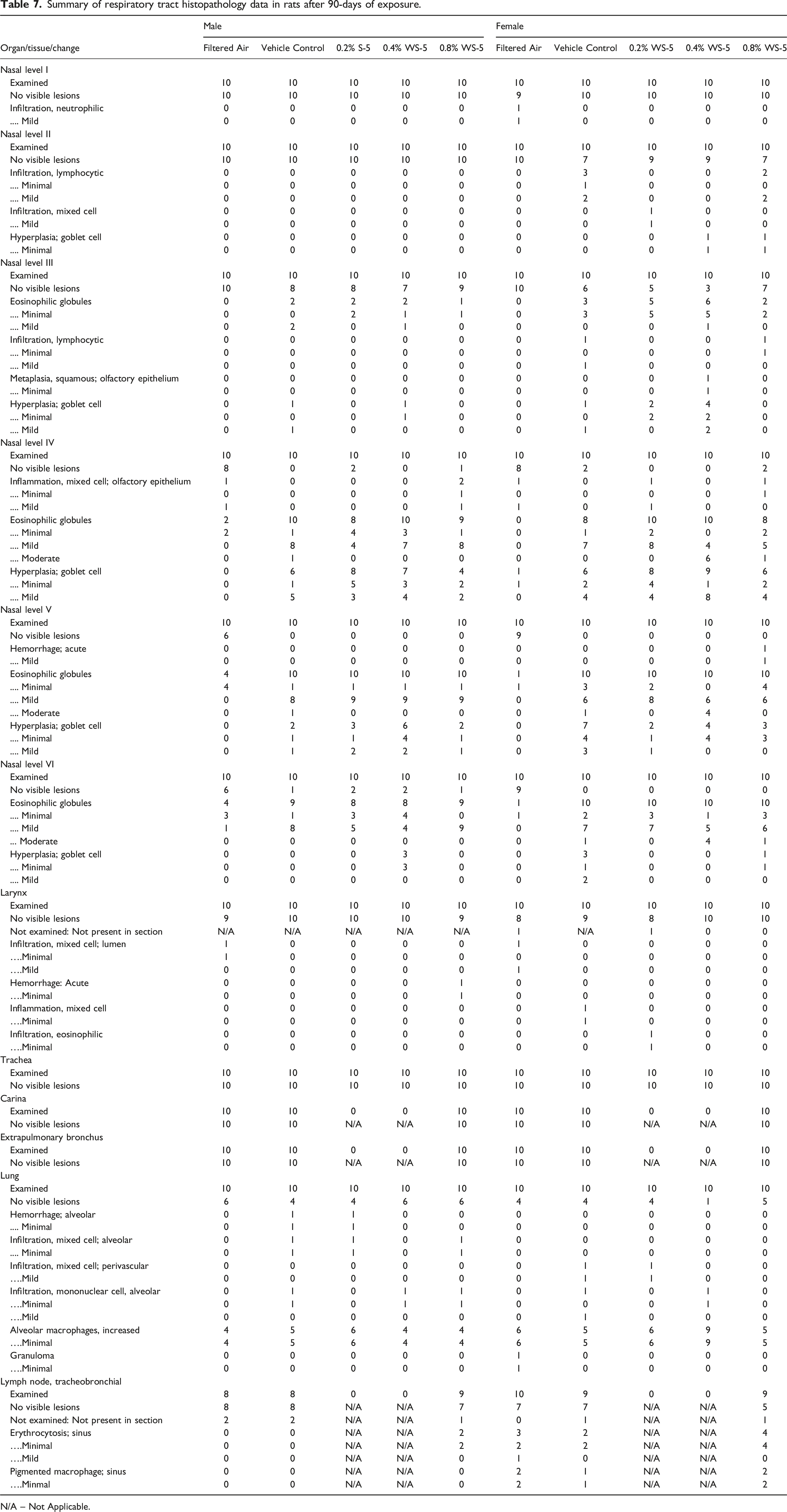
N/A – Not Applicable.
Summary of histopathology data in rats at recovery.
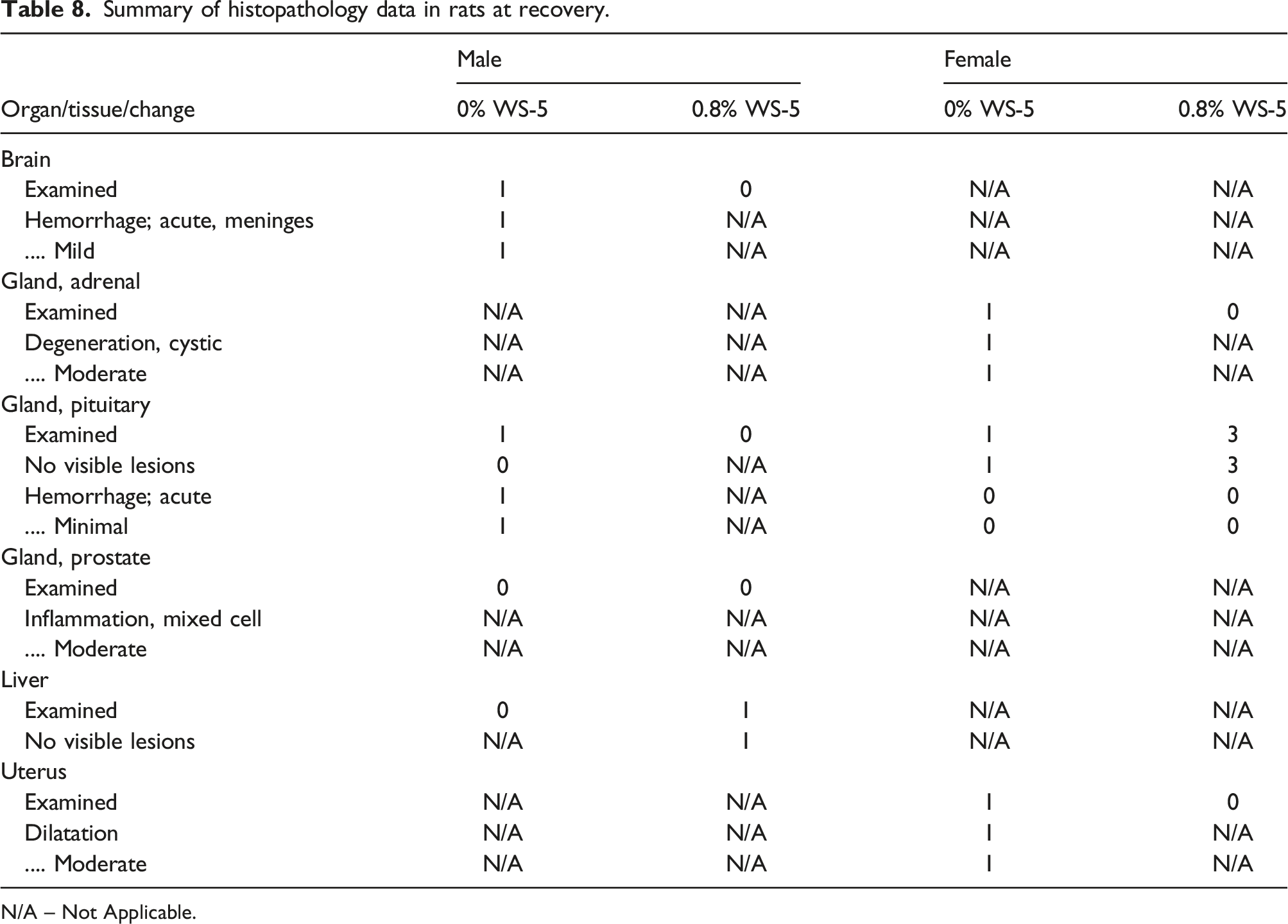
N/A – Not Applicable.
Discussion
The measured exposure WS-5 concentrations being within 4% of the target exposure concentrations and the similarity in the particle size distribution among the exposure groups facilitates comparison of biological responses among the groups. The amount of water measured in the analytical aerosol measurements was not surprising due to use of humectants (propylene glycol and glycerin) in the vehicle control and WS-5 formulations, the humidified dilution air used in inhalation studies, and the rapid water absorption of ENDS aerosols recently reported by. 35 There was no statistically significant difference in measured rat plasma propylene glycol levels between groups within time points. Although sporadic statistically significant differences from vehicle control were noted for some endpoints in the WS-5 exposed groups (e.g., high dose WS-5 male tidal volume; etc.), no dose-response or consistency in response between sexes was noted. Addition of WS-5 at up to 0.8% resulted in no WS-5-related clinical observations or effects on body weight, food consumption, respiratory physiology, hematology, coagulation, clinical chemistry, urinalysis, bronchoalveolar lavage fluid parameters, organ weights, ophthalmic, or macroscopic observations.
The histopathology finding of alveolar hemorrhage and mixed cell infiltration after 28-days of exposure were either not present in the WS-5 exposed groups or were present at similar frequencies in the vehicle control and/or control groups after 90-days of exposure. Histopathology findings of alveolar hemorrhage and mixed cell infiltration are not unique to this study and have been reported in control and test article groups in previous inhalation studies with equivalent incidences,26,28,36 therefore, they were not considered to be vehicle control or WS-5 related. Thus, no WS-5-related histopathology findings were noted at any necropsy. This 13-week subchronic nose-only inhalation study demonstrated that daily exposures to WS-5 up to 2.5 mg/L TPM for 6 h/day did not induce biologically meaningful adverse effects in rats.
One of the limitations of this study is that measurements of WS-5 in rat plasma were not performed, meaning the bioavailability of WS-5 via the inhalation route could not be calculated. This study did measure one of the three formulation constituents, propylene glycol using a validated analytical method to confirm exposure. The lack of toxicity from exposure to WS-5 observed in this study might be due to a lack of WS-5 bioavailability from the inhalation route, or a lack of toxicity from WS-5 at the concentrations used in this study.
In summary, administration of WS-5 by nose-only inhalation to Crl:CD(SD) rats at a target concentration of 2.5 mg/L of aerosolized formulations containing 0, 0.2, 0.4, and 0.8 w% (corresponding to 0, and approximately 5, 10, and 20 μg WS-5/L) for 6 h per day for 5 days per week for 13 weeks was well tolerated at all WS-5 concentrations. Based on the absence of adverse effects, the no-observed-adverse effect concentration (NOAEC) was considered to be 2.5 mg/L of aerosolized 0.8 w% WS-5 that achieved a mean measured concentration of approximately 18 μg WS-5/L.
Supplemental Material
Supplemental Material - Evaluation of the toxicity of WS-5 in a 90-days nose-only exposure in sprague-dawley rats
Supplemental Material for Evaluation of the toxicity of WS-5 in a 90-days nose-only exposure in sprague-dawley rats by Michael Jerome Oldham, Rahat Wadhwa Desai, James Randazzo, Guy Lalonde and Roxana Weil in Toxicology Research and Application.
Footnotes
Acknowledgments
Acknowledgements: The authors would like to thank the staff at Charles River Laboratories for a well-conducted study and Maleeha Agha for assistance with data presentation.
Author Contributions
MJO and RW conceived the study, RW obtained funding for the study, MJO, RWD, JR, and GL designed the study, RWD monitored the study, JR was the study director, MJO wrote the first draft and all authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
All authors are employees of Juul Labs, Inc. or the contract research organization that was compensated for performing the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by Juul Labs, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
