Abstract
The objectives of this study were to increase PG exposure above concentrations tested by Suber et al. and use systems toxicology analysis of lung tissue to understand molecular events. Sprague Dawley rats were exposed to filtered air (sham), propylene glycol/water (PG/W; 90:10) or a propylene glycol/vegetable glycerin/water (PG/VG/W; 50:40:10) reference. The reference group was added at the high dose to observe any changes that might be associated with a carrier more in line with e-vapor products. Macroscopic examinations and terminal organ weights revealed no observations associated with exposure to PG/W or reference. Food consumption and body weights were unaffected by PG/W or reference when compared to sham. No exposure related alterations were observed in serum chemistry, hematology, coagulation, urinalysis or BALF cytology and clinical chemistry. Although clinical observations of dried red material around the nose in the high dose PG/W group were reported, histopathology showed no nasal hemorrhaging which was previously reported by Suber et al. Non-adverse PG/W and reference related findings of minimal mucous cell hyperplasia were noted in nasal cavity section II. No other exposure-related findings were noted in the primary or recovery necropsies. A systems toxicology analysis on lung tissue showed no statistically significant differentially expressed transcripts or proteins compared to the sham group. The endpoints measured from the PG/W high dose group did not differ significantly from those in the more common carrier PG/VG/W. As anticipated, exposure to PG aerosols was slightly irritating but well tolerated. Accordingly, the highest PG exposure (5 mg/L, 6 hrs/day) was regarded as the NOAEC, corresponding to a PG delivered dose of 1,152 mg/kg/day in rats.
Introduction
Smoking cessation remains the most effective approach to minimize smoking related diseases, however alternative nicotine-delivery products such as electronic nicotine delivery systems (ENDS) that do not generate combustion by-products may offer substantially reduced risk to those who otherwise continue to smoke. 1 –7 The aerosol generated from ENDS is usually comprised of nicotine, a liquid carrier and flavors. Propylene glycol (PG) constitutes one of the main components of the liquid along with vegetable glycerin (VG). PG and VG, alone or as a mixture, are used as a vehicle to deliver the nicotine and flavors in ENDS formulations.
The toxicity and safety of PG through oral, dermal and parenteral routes in several animal species has been extensively studied and reviewed. 8,9 In humans, PG has a mean half-life of 2.3 hours following parenteral exposure and is metabolized into lactic acid, carbon dioxide and water. 10,11 PG is widely used as an excipient in the pharmaceutical industry, as an active ingredient in ophthalmic demulcent, and as a food preservative. 12 As a food additive, the US Food and Drug Administration (FDA) (21 CFR §184.1666), the Flavor and Extract Manufacturers Association, and the Joint FAO/WHO Expert Committee on Food Additives has determined that PG is generally recognized as safe (GRAS) up to 2%. JECFA established an ADI for PG of 0–25 mg/kg/day. 13
PG has also been tested for inhalation toxicity. For instance, Suber et al. exposed Sprague Dawley (SD) rats to nose-only inhalation of nebulized PG up to 2.2 mg/L total particulate matter (TPM) for 90 days. 14 There were no notable changes in the respiratory functions compared to the control group. However, they reported an increase in the number and the mucin content of goblet cells in the nasal passages as well as clinical observations of nasal hemorrhaging and ocular discharge. The nasal hemorrhaging was attributed to mucosal dehydration due to the hygroscopic property of PG. 14 More recently, Werley et al. exposed SD rats once to PG using capillary aerosol generators at concentrations of 14.4, 30.5, and 44.9 mg/L for 4 hours. 15 Clinical observations included an initial 5–10% decrease in body weight during recovery days 1–3 and exposure-related bleeding around the eyes and nose at recovery day 7. The body weights returned to normal by day 7. No mortalities were observed at any of the test concentrations; therefore, the LC50 was reported to be greater than 44.9 mg/L. 15 In a follow-up 7-day study performed at the same laboratory, SD rats were exposed to either 20.8 or 41.0 mg/L PG aerosol for 4 h/day for 7 consecutive days and demonstrated no clinical signs of bleeding around the nose or eyes. 15 Lastly, Phillips et al., conducted a 90-day nose-only study with SD rats that were exposed to nebulized PG/VG with and without nicotine. No adverse effects were reported with PG/VG alone. Up-regulation of proteins CYP1A1 and FMO3 in the lungs, as well as gene expression changes in hepatic metabolic enzymes were attributed to the addition of nicotine, consistent with prior reported effects. 16
The main objective of the current study was to evaluate potential toxicity of PG (PG/Water, 90/10, %V/V) in SD rats using the standard OECD 413 study design and conducted in accordance with Good Laboratory Practice (GLP). Inhalation exposure limits have been recommended for PG based on the endpoint of nasal hemorrhaging reported by Suber et al. 17,18 In order to achieve the highest dose of PG possible, VG was not included in the test article formulation. The rats were exposed on a 5 day/week basis to 5 mg/L TPM of PG/W, with the daily exposure dose adjusted based on the exposure duration: 1 (low), 3 (mid), or 6 (high) hours/day. A reference group of PG/VG/W (50:40:10, %V/V) was added at the high dose only (6 hours/day to 5 mg/L TPM) to observe any effects associated with an e-vapor carrier more in line with available products. In addition to the standard OECD toxicological endpoints, a systems toxicology analysis (non-GLP) was performed on the lungs of females.
Materials and methods
Experimental design
This 13-week subchronic repeated-dose inhalation study was conducted in general accordance with the OECD 413 guideline using male and female SD rats to test the potential toxicological effects of a PG/W (90:10, %V/V) mixture. The PG/W test groups were exposed to a fixed aerosol concentration of 5 mg/L for 1, 3 or 6 hours. The sham control group was exposed for 6 hours to conditioned filtered air and a reference group was exposed to a 5 mg/L aerosol of PG/VG/W (50:40:10, %V/V) for 6 hours. Ten male and 10 female rats per group were allocated for the measurement of the standard OECD endpoints. An additional 10 females were added to each group for assessment of systems toxicology endpoints. The females used for systems toxicology were exposed concomitantly with the study animals evaluated for standard OECD endpoints. Five animals of each sex were allocated to the 6-week recovery groups.
The study was conducted in accordance with FDA GLP except for the plasma PG analysis and the systems toxicology arm. The determination of propylene glycol in rat plasma and the systems toxicology arm were performed using qualified methods.
Test materials
United States Pharmacopeia (USP) grade PG and VG were purchased from Fisher Scientific. All formulations were prepared at the testing facility (Charles River Laboratories Ashland, Ashland, OH) using ultrapure water. The formulations were prepared weekly and stored at room temperature. Throughout preparation and sampling, the formulations were stirred continuously. Uniformity and stability of the formulations were assessed at the first, fourth, eighth and final preparations using a validated gas chromatography method with flame ionization detection.
Animals and group allocation
Male and female Crl: CD Sprague Dawley rats (∼ 4 weeks old) were obtained from Charles River Laboratories, Inc., Raleigh, NC. The testing facility (Charles River Laboratories Ashland, Ashland, OH) is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International and animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council, 2011). Animals were randomly assigned to groups based on body weight stratification in a block design and were approximately 7 weeks of age at the start of the exposures. Basal diet (Certified Rodent Lab Diet® 5002 (meal), PMI Nutrition International, LLC) and water were provided ad libitum throughout the study except during removal for daily exposures or sample collection. Animals were fasted overnight before performing clinical pathology blood collection on animals scheduled for necropsy. Room conditions were set within the 413 guidelines at 23ºC ± 2º and 50% ± 20% humidity on a 12-hour light/dark cycle starting at 6:00 am daily.
Aerosol generation and atmosphere characterization
Test and reference article aerosols were generated using a capillary aerosol generator (CAG) mounted within a Teflon delivery box. A detailed description of the CAG devices and their operation has been reported previously. 19 –24 The benchtop CAG consists of a metal block with channels cut into the interior plate allowing pumped fluid to enter the heated block and a vapor jet to exit. A thermocouple was inserted into the block and controlled using a Digi-Sense Temperature controller. The thermocouples were set to approximately 190°C or 280°C for the PG/W and PG/VG/W, respectively. The CAG vapor jet rapidly cools in the surrounding air and condenses into a fine particulate aerosol prior to introduction into the conventional nose-only system (CNOS). The CNOS tower consists of a central plenum or exposure manifold where the aerosol enters. 25 Animal confinement tubes with one end protruding into the aerosol can be arranged around the plenum allowing for multiple simultaneous exposures. Aerosol flows past the nose of the animals and the dynamic flow prevents re-breathing. The CNOS was designed and fabricated by Charles River. To maintain the CNOS temperature, the diluted aerosol was passed through an EXAIR Vortex Cooling tube.
CNOS aerosol concentrations were measured daily using standard gravimetric methods. Aerosol mass samples were collected from the nose port on pre-weighed 44-mm Cambridge glass-fiber filter pads and weighed to obtain the exposure concentration, TPM in air (TPM mg/L). The 44-mm pads were recommended in CORESTA Method 81 and later in ISO 20768. At least three times weekly, PG content of the PG/W and PG/VG/W exposure atmospheres was analyzed from filter samples collected during an animal exposure for 5 minutes at approximately 250 mL/minute using a validated method. Briefly, filters were extracted in isopropanol with 1,4-butanediol and quinoline internal standards. The extract solution was filtered through a 0.45 µm filter and diluted as needed. Samples were analyzed using a GC-FID with a split ratio of 15:1. Separation was performed on a DB-ALC1 column (30 m x 0.32 mm ID, 1.8 µm film thickness) with the temperature program of 90ºC for 1 minute, ramp at 15ºC/minute to 120ºC then ramp at 40ºC/minute to 280ºC and hold for 2 minutes. Propylene glycol and glycerin eluted at approximately 4 and 5.5 minutes, respectively. The aerosol particle size was determined gravimetrically using a 7-stage stainless-steel cascade impactor and expressed as the mean mass median aerodynamic diameter (MMAD) and the mean geometric standard deviation (GSD).
Animal exposures and daily observations
Animals were exposed via nose-only inhalation on a 5 day per week basis for 13-weeks. For the mid and high dose groups, the daily exposure duration was increased from 1, 2, and 3 hours over the first 3 days to help acclimate the animals to the daily exposure regimens. All animals were observed twice daily for mortality or moribundity. Clinical examinations were performed prior to exposure and within an hour post-exposure. On non-exposure days and during the recovery period, animals were observed once daily. Individual body weights were collected prior to exposure start, weekly during the study period, and on the day of necropsy. Food consumption was recorded weekly.
In-life biological measurements
Plasma propylene glycol analysis
Blood samples were collected from 5 animals/sex/group during Study Weeks 4 and 12 within 5 minutes following exposure. Blood was collected via a jugular vein into chilled tubes containing K
For the determination of PG in rat plasma, plasma was spiked with an internal standard (1,2-propane-d6-diol) and derivatized. Analytes were separated from plasma using a liquid-liquid extraction. Chromatography was performed on a Waters Acquity® UPLC BEH Shield RP18 1.7-µm 2.1 × 50 mm column and analytes were detected using an Applied Biosystems/MDS Sciex API 5500™ triple quadrupole mass spectrometer in the positive ion mode. PG was quantitated over the concentration range of 1000 to 100,000 ng/mL using a 0.025-mL sample.
Carboxyhemoglobin (COHb)
Blood samples from 5 animals/sex/group were collected immediately after exposure during weeks 5 and 11. Blood was collected from the jugular vein into tubes containing sodium heparin, kept on ice and processed within 120 minutes. Percent COHb was determined using an IL 682 Co-Oximeter (Instrumentation Laboratory SpA, Bedford, MA)
Respiratory physiology
For measurement of respiratory function parameters, 8 animals/sex/group were placed in a single-chamber, head-out, neck-sealed plethysmograph (Buxco Electronics, Inc.). The system consisted of single-chamber restraint plethysmographs equipped with a nose-only adaptor, DP45 pressure transducers (Validyne Engineering Corporation) calibrated on each day of collection and interfaced to the Life Science Suite™ Physiology Platform (P3). Respiratory frequency, tidal volume, and calculated minute volume were collected for at least the first 35 minutes of the exposure. Only the last 5 minutes of data were used for analysis and reporting. The first 30 minutes of data collection allowed the animals to acclimate to the exposure atmosphere and respiratory restraint and were not utilized for data analysis.
Terminal biological measurements
Bronchoalveolar lavage (BAL)
BAL was performed on the left lung of all animals designated for standard endpoints. The lung was lavaged 3 times with a lavage solution consisting of Hank’s Balanced Salt Solution without calcium, magnesium, or phenol red, on ice. The lavage solution volume was based on the final nonfasted body weight. The first lavage remained in the lung for approximately 1 minute and the BAL fluid (BALF) was collected and stored on ice until processed. The lung was lavaged two additional times. The BALF was isolated in a refrigerated centrifuge and the supernatant fluid from the first lavage was transferred to a sealable vial and stored on ice until analyzed for lactate dehydrogenase, total protein, and alkaline phosphatase. After the BALF centrifugation, the cell pellets obtained from all 3 lavages were combined and resuspended for counting. Total cell counts were obtained using a hemocytometer with cell viability assessed by trypan blue exclusion. For differential counts, cytospin preparations of 1 or more volumes of cell suspension were made in duplicate and one slide from each volume was stained. BALF differential cell counts of 200 cells/slide were performed and cells were identified as macrophages, neutrophils, lymphocytes, eosinophils, or basophils.
Lung preparation for systems toxicology endpoints
Following 13-weeks of exposures, five females per group were deeply anesthetized by an intraperitoneal injection of sodium pentobarbital and perfused in situ with 0.9% saline. The left lung was collected, frozen on dry ice, wrapped in foil, and stored frozen (−65°C to −85°C) until processing. After 6-weeks of recovery, an additional 5 females per group were anesthetized and the lungs prepared. To minimize RNA degradation, surfaces and surgical tools used for sample collection were treated with RNaseZap® (Ambion).
Left lung tissue samples were randomized and then pulverized using a cryoPREP® Impactor (Covaris). All samples were kept frozen during preparation and were subsequently stored at −80ºC until processed. The pulverized lung samples were processed for RNA isolation for transcriptomics analysis, and for protein isolation by multiplexed isobaric Tags for Relative and Absolute Quantitation (iTRAQ®; Ab Sciex, Framingham, MA).
Lung transcriptomics
Total RNA was isolated using the miRNeasy® mini kit (Qiagen, Hilden, Germany). The quantity of isolated RNA was determined with a NanoDrop 1000 instrument (Thermo Fisher Scientific, Waltham, MA) and the RNA quality was determined with a 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA). All lung samples had an RNA integrity value greater than 6 and were used for further analysis.
RNA was prepared for hybridization using the High Throughput 3´ In Vitro Transcription PLUS (HT 3´ IVTPLUS) kit (Affymetrix, Santa Clara, CA). Samples were hybridized on the GeneChip® Rat Genome 230 2.0 arrays (Affymterix, Santa Clara, CA). Raw CEL files were processed using the custom Chip Description File environment Rat2302_Rn_ENTREZG v19.0.0 (rat2302rnentrezgcdf) 26 and normalized using frozen robust microarray analysis (fRMA). 27 Quality controls, including log-intensities, normalized unscaled standard error (NUSE), relative log expression (RLE), median absolute value RLE (MARLE), and pseudo-images as well as raw image plots, were performed with the affyPLM package (Bioconductor suite). Following the quality control procedures, raw p values were generated for contrasts between sham controls and test groups with the Limma package, 28 and adjusted using the Benjamini-Hochberg false discovery rate (FDR) multiple test correction. 29 The raw data were deposited in the ArrayExpress public repository with accession number E-MATB-10006.
Lung proteomics
Protein was isolated from lung samples using tissue lysis buffer and precipitated with acetone. Following resuspension, the protein concentration was determined using the Bradford assay. Approximately 50 µg of protein were processed using the iTRAQ 8-plex procedure according to the manufacturer’s instructions (AB Sciex, Farmingham, MA). The samples from the main phase and the recovery group were analyzed in separately defined iTRAQ experiments—with a common reference mix sample shared between all iTRAQ analysis sets of both iTRAQ experiments. Separately for each phase, each multiplexed iTRAQ replicate set consisted of one sample of each of the 5 exposure groups; with 5 separate iTRAQ sets for the 5 replicates. In addition, a pooled reference mix was included in each iTRAQ set.
Samples were analyzed in random order using an Easy nanoLC 1000 instrument (Thermo Fisher Scientific, Waltham, MA) connected online to a Q Exactive mass-analyzer (Thermo Fisher Scientific). Peptides were separated on a 50-cm Acclaim™ PepMap™ 100 C18 LC column (2-µm particle size; Thermo Fisher Scientific) at a flow rate of 200 nL/min, with a 200-min gradient from nanoLC buffer A to 40% acetonitrile with 0.2% formic acid.
For further analysis of the quantitative changes in the proteome, iTRAQ spectrum-level quantification data was filtered for a q-value < 0.01 and for “unique” quantification results, as defined by the Proteome Discoverer software. A global variance stabilizing normalization (VSN) was performed with the respective Bioconductor package in R. 30,31 Each iTRAQ reporter ion set was normalized to its median, and protein expression values were calculated as the median of these normalized peptide-level quantification values. 32
For the detection of differentially expressed proteins, raw p values were generated for contrasts between sham controls and test groups from a moderated t-statistic using the empirical Bayes approach. 33 The Benjamini–Hochberg FDR method was used to correct for multiple testing effects. Proteins with an adjusted p value < 0.05 were considered differentially expressed and further processed in the R statistical environment. 34 The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD022362.
Network perturbation amplitude (NPA) for lung transcriptomics
Using a two-layer hierarchical causal model, lung network perturbations following exposures were quantified. The upper layer is the functional backbone and represents the actual network model that consists of causal molecular interactions and signaling pathways regulated in the lung. The lower layer represents gene expression changes that are a result of known upstream biological interactions of the functional backbone. Thus, perturbations in the upper functional backbone are inferred from measuring changes in the underlying transcriptome. 35,36 Once the NPA is quantified, three statistics were computed to determine the significance of the amplitude. Confidence intervals and associated p values were computed with companion statistics to assess the accuracy of downstream gene assignments, O, and the importance of the cause-and-effect relationship, K. A network was considered to be perturbed if the three values (the p value for the experimental variation, and the companion statistics, O and K) were below 0.05. 36
The biological impact factor (BIF) was derived by combining the significantly perturbed networks into a holistic value. 35,37 For each exposure comparison, the δ value (−1 to 1) indicates how similar the underlying network perturbations are with respect to a reference condition (indicated as REF, which has the largest BIF, in this study the 3 hour PG/W exposure). The similarity and directionality are deduced from the correlation of the nodes of a significant impacted pathway.
Necropsy
Prior to the scheduled necropsy, rats were fasted overnight while maintained in a metabolism cage for urine collection. At necropsy, blood from 5 animals per sex per group was collected for hematology, serum chemistry, and coagulation parameters. The necropsy consisted of a complete internal and external examination, including all orifices. Any gross lesions were examined and recorded, and a careful gross examination of the respiratory tract was performed. Tissues and organs were collected and placed in 10% neutral-buffered formalin or Davidson’s solution for eyes and testes. Microscopic examination of haematoxylin-eosin stained paraffin sections was performed on all standard endpoint tissues and organs specified in the OECD guidance from the sham control, and both high dose groups. Microscopic examination of the eyes, respiratory tract and nasal cavities from all groups was performed. Following decalcification, six cross-sections of the nasal cavities were prepared as described by Morgan and Mery et al. 38,39 The six sections provide an additional section close to the nares and the olfactory bulb. In addition, historical controls at the testing facility and previous studies by the testing facility used the nasal sectioning described by Morgan and Mery et al. Histopathological evaluations were performed by a board-certified veterinary pathologist. Severity grades were categorized as minimal, mild, moderate, marked and severe.
Statistics
For the statistical analyses of the standard OECD endpoints, values were subjected to a parametric one-way analysis of variance (ANOVA). 40 If the results of the ANOVA were significant (p < 0.05), Dunnett’s test was applied to the data to compare the PG exposure groups to the sham control group. 41
Results
Exposure characterization
The nose port target concentration for all groups was achieved, and the variability (represented by the SD) was less than 10 percent except for the low dose group (Table 1). The mean TPM was 5 mg/L and the standard deviations ranged from 0.22 to 1.29. The nose port particle size mean mass median aerodynamic diameter (MMAD) ranged from 1.7 to 2.6 µm with mean geometric standard deviations (GSD) of 1.55–1.96. The mean nose port PG concentration was within 12% of the target for all exposure groups. In accordance with the OECD 413 guideline, during the animal exposures, the temperature was between 19ºC and 25ºC and the relative humidity was between 30% and 70%.
Exposure characterization at the nose port.
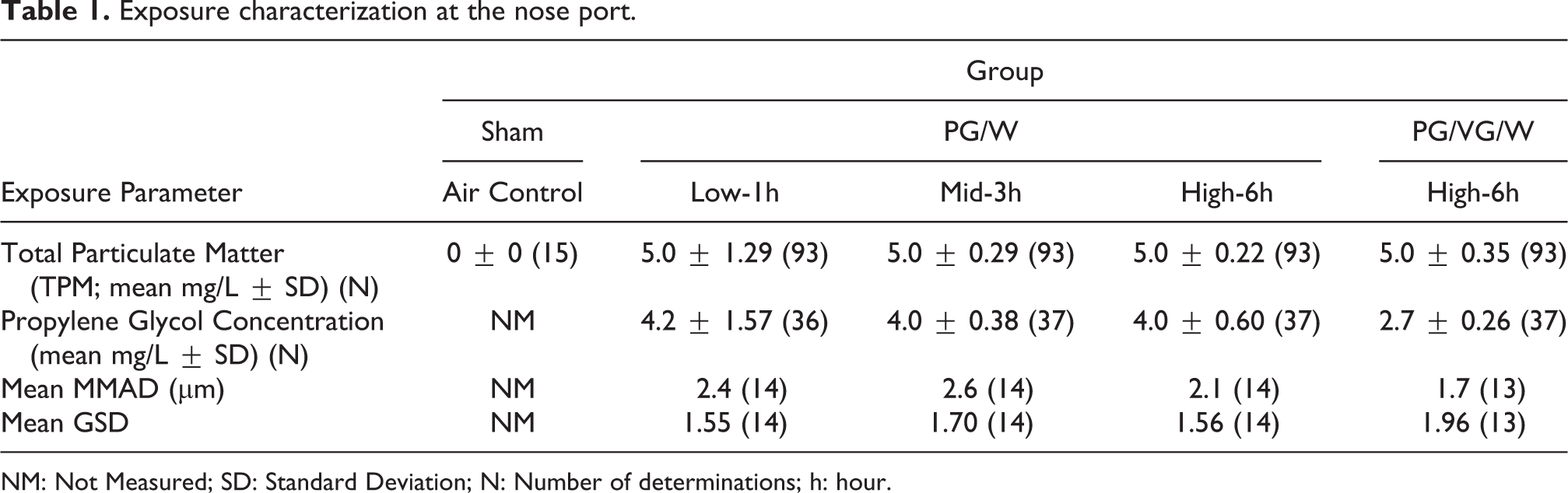
NM: Not Measured; SD: Standard Deviation; N: Number of determinations; h: hour.
In-life observations
There were no unscheduled deaths considered to be directly related to PG/W or PG/VG/W exposures. Non-exposure related early deaths included one male from the PG/W 3-hour exposure group and one female from the PG/W 1-hour exposure group. Postmortem examination showed no macroscopic or microscopic findings that were related to PG/W exposure. Of note, microscopic examination of the respiratory tract for both animals was unremarkable. The cause of death for both was undetermined.
During post-exposure (<1 h) clinical observations, dried red material was noted around the nose and occasionally around the eyes (recorded as “dried red material around” the affected area in the data capture system). This clinical sign did not correlate with microscopic findings in the nasal cavity. No other significant clinical observations were reported, and food consumption and body weights were unaffected in the exposure groups compared to the sham control.
No statistically significant changes in respiratory frequency were noted during weeks 5 or 12 for any comparisons in either sex. Week 5 comparisons were conducted for pooled sexes and a statistically significant decrease in tidal volume (23%) and corresponding minute volume (36%) were noted for the PG/VG/W reference group when compared to sham control (Table 2). The decreases in the PG/VG/W reference group were considered an exposure effect, however, this effect was not observed at week 12. When the week 12 comparisons were conducted for pooled sexes, a statistically significant decrease in minute volume was noted for the 3-hourPG/W group when compared to the sham control (Table 2). The overall impact of PG aerosol exposures compared to the sham control appear minimal and without a clear pattern.
Biomarkers of exposure and respiratory function parameters. Bold text indicates values are statistically significantly different.
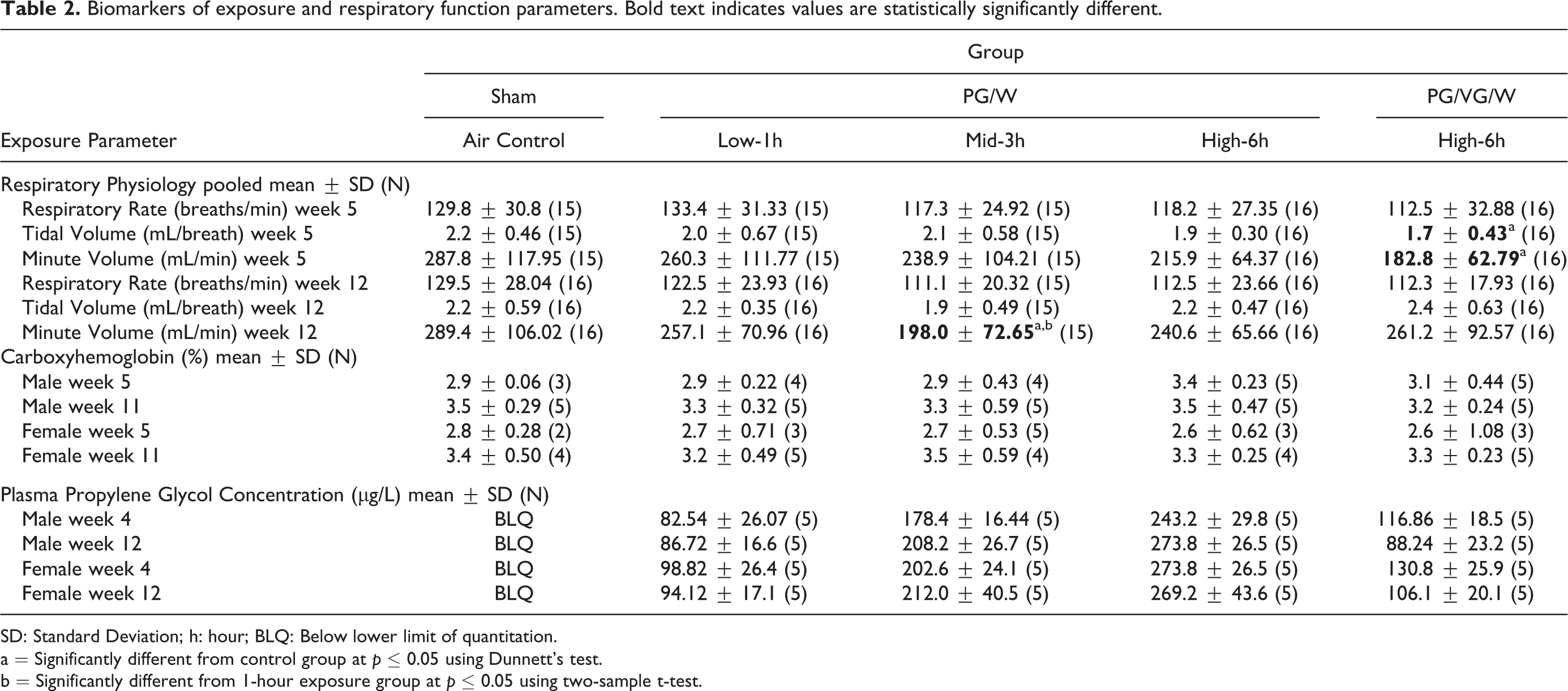
SD: Standard Deviation; h: hour; BLQ: Below lower limit of quantitation.
a = Significantly different from control group at p ≤ 0.05 using Dunnett’s test.
b = Significantly different from 1-hour exposure group at p ≤ 0.05 using two-sample t-test.
As expected, carboxyhemoglobin was unaffected by exposure to either PG/W or PG/VG/W. For both males and females, postexposure plasma PG concentration increased with increasing exposure duration to PG aerosols, while the sham control showed levels below the limit of quantitation. As expected from the difference in aerosol PG concentration, the plasma PG concentration of the PG/VG/W (50:40:10, 6-hour) group was roughly half of that of the PG/W (90:10, 6-hour) exposure group (Table 2).
Standard OECD toxicological endpoints
Following 13 weeks of exposure, animal bodyweights and food consumption were unaffected by exposure to the PG aerosols. Absolute lung weights were comparable between the exposed groups and sham control for both sexes (Table 3). For males, the PG/W Low (1 h) and PG/W Mid (3 h) groups showed a statistically significant decrease in the relative lung weights (normalized to body weight) compared to the sham control. However, this effect was not observed in the PG/W High (6 h) group. There were no other significant differences in terminal organ weights when compared to the sham control. The body and organ weights were also overall comparable between the PG/W High (6 h) and the reference (PG/VG/W, 6 h) groups.
Absolute and relative organ weights at 13-week necropsy. Bold text indicates values are statistically significantly different.
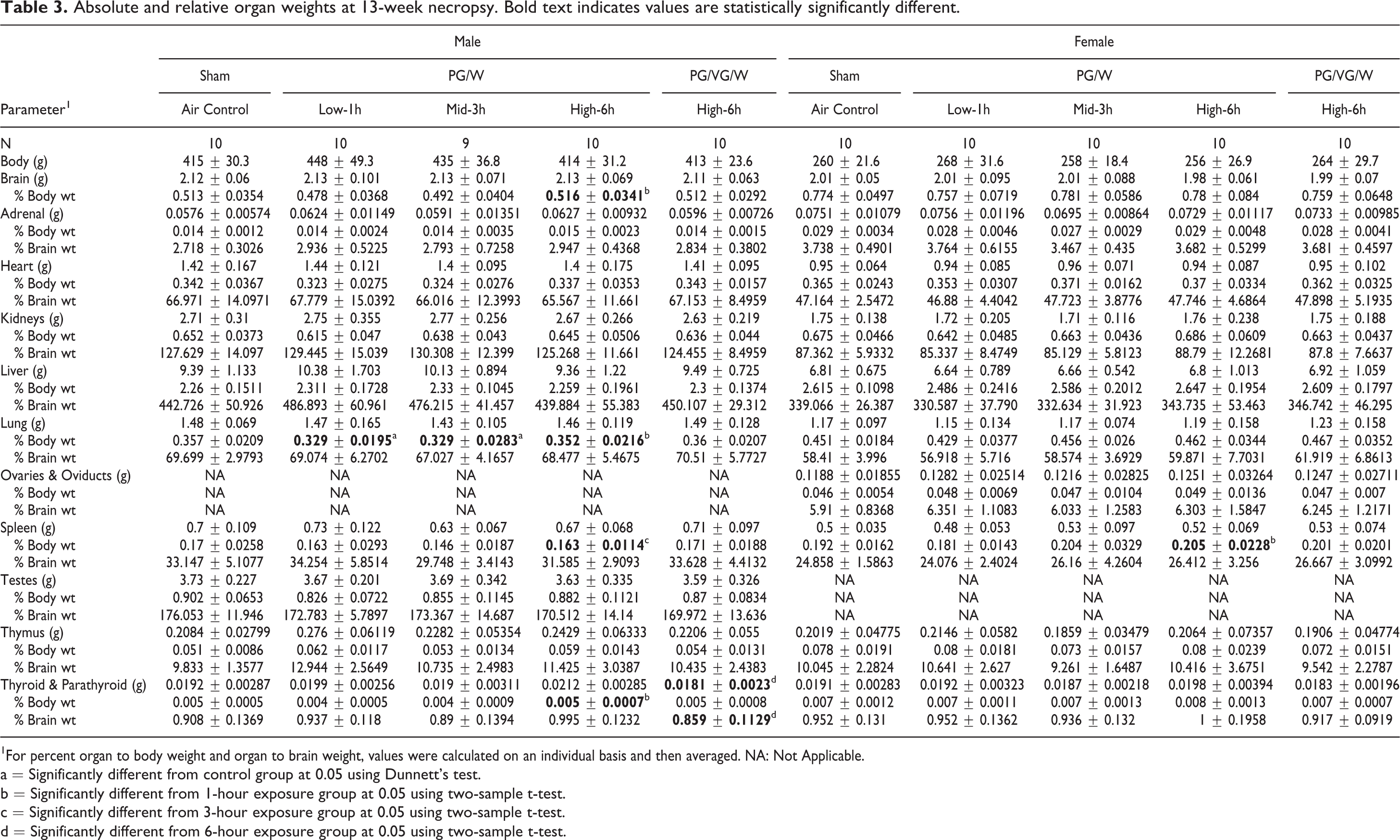
1For percent organ to body weight and organ to brain weight, values were calculated on an individual basis and then averaged. NA: Not Applicable.
a = Significantly different from control group at 0.05 using Dunnett’s test.
b = Significantly different from 1-hour exposure group at 0.05 using two-sample t-test.
c = Significantly different from 3-hour exposure group at 0.05 using two-sample t-test.
d = Significantly different from 6-hour exposure group at 0.05 using two-sample t-test.
Following 13 weeks of exposure to PG/W and PG/VG/W in the respiratory tract, there were dose-related histological changes in the nasal level II (minimal mucous cell hyperplasia, PG/W: incidence 8 out of 10 with a severity of 1; Table 4). The mucous cell hyperplasia was characterized by increased numbers of closely packed goblet cells with areas of pseudostratification or intraepithelial gland formation that minimally thickened the nasal mucosa. The exposure effect was not well defined in the males as half the sham controls also showed minimal mucous cell hyperplasia, while in females, the dose-related response was more pronounced with a very low incidence in the sham control (1/10). One female in the PG/VG/W group showed minimal hemorrhage in nasal level II. All other respiratory tract endpoints were comparable for the PG/W High and the reference groups. Following 6 weeks of recovery phase, the nasal mucous cell hyperplasia in both PG/W and PG/VG/W had resolved in both sexes (incidence and severity were equivalent to or less than sham).
Respiratory tract histopathology following 13-week exposures.
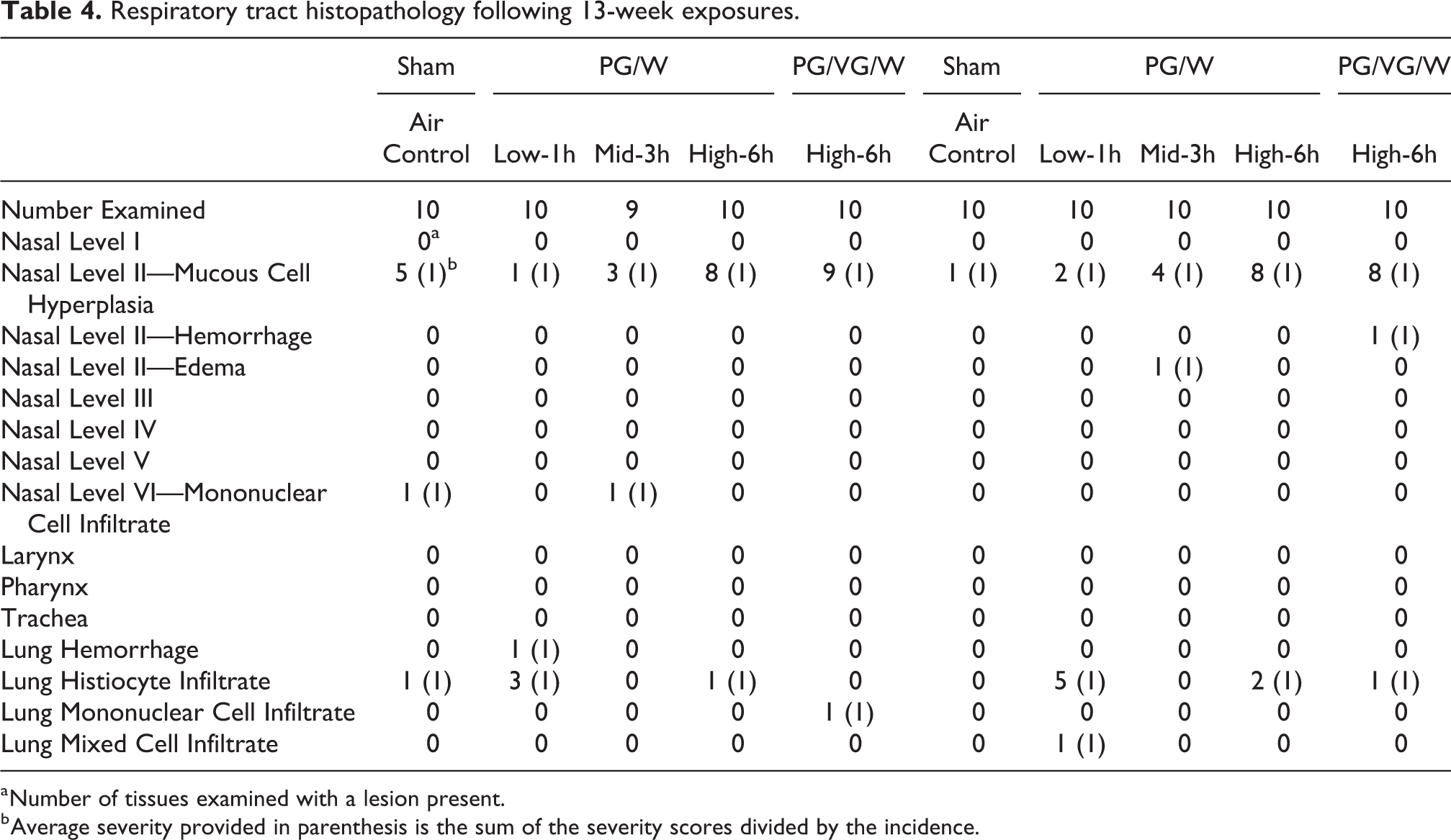
a Number of tissues examined with a lesion present.
b Average severity provided in parenthesis is the sum of the severity scores divided by the incidence.
In the lung, there were no notable histopathological changes in the PG-exposed groups compared to the sham controls in either sex or between the PG/W and PG/VG/W groups (Table 4). The lack of pulmonary inflammation was consistent with the lack of changes in the enzymes and cytology parameters of the BALF (Supplemental). No alterations were found in hematology, coagulation, serum chemistry, and urinalysis parameters between the exposed groups and the sham controls (Supplemental).
Lung transcriptomics and proteomics
A systems toxicology analysis was performed on the lungs of female rats from all groups. In Figure 1, the differential gene and protein expression are shown as volcano plots of the log fold change with statistical significance. When compared to the sham control, there were no statistically significant changes in differentially expressed genes or proteins under the applied threshold (FDR-adjusted p value less than 0.05). These findings are consistent with previous inhalation studies in rats with PG/VG mixtures albeit tested at lower aerosol mass. 16
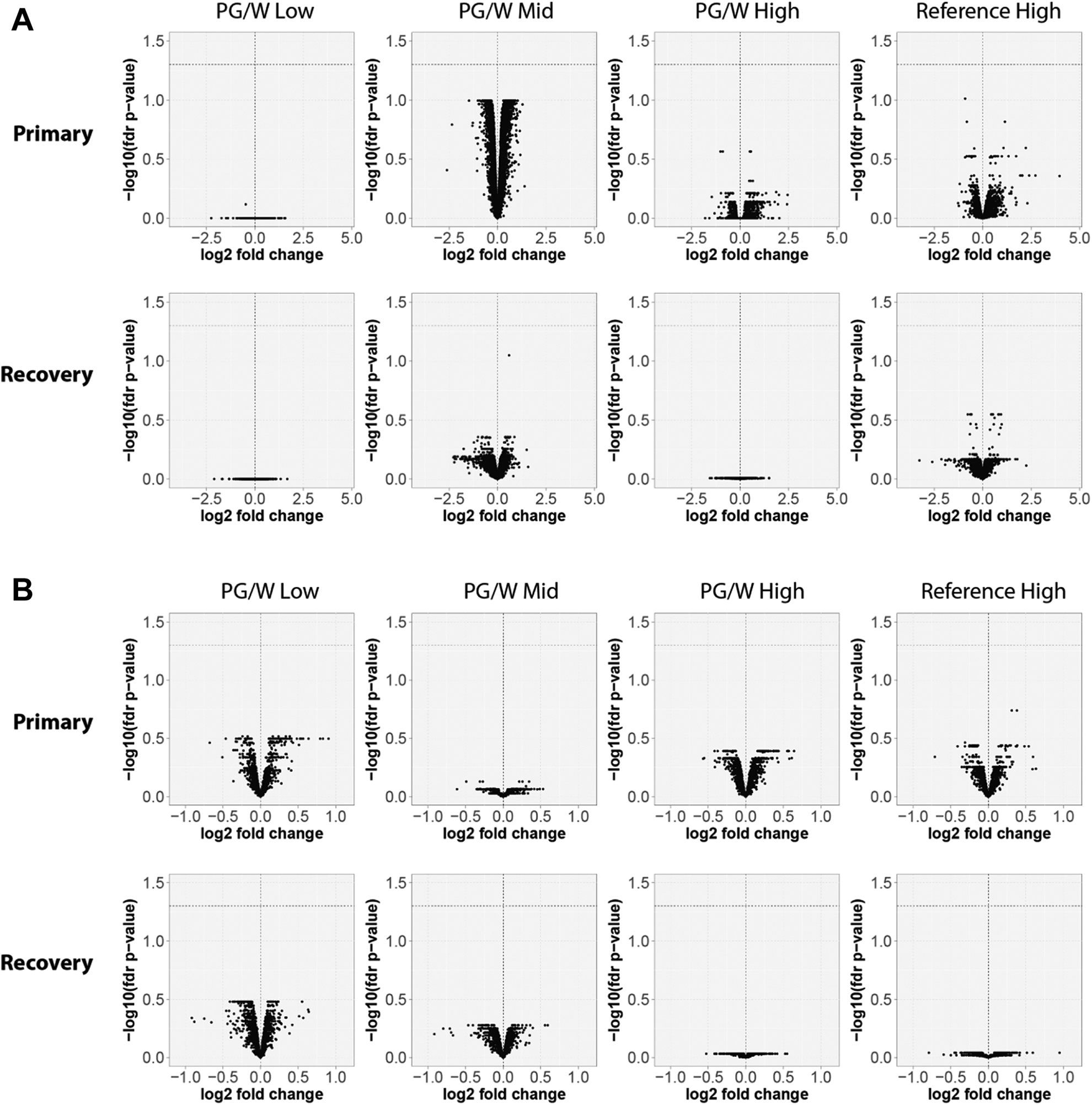
Lung volcano plots representing the systems response profiles at the primary and recovery necropsies. The expression change, calculated as the log2 fold-change, is plotted on the x-axis and the statistical significance (false discovery rate (fdr) <0.05, indicated by a dotted horizontal line), proportional to the negative log10-adjusted p value, is plotted on the y-axis. Yellow and blue dots would highlight genes that are statistically significantly up- and down-regulated, respectively (no statistically significant differentially expressed genes or proteins were identified). Panel (A). Gene expression analysis. Panel (B). Protein expression analysis.
In addition to comparisons of gene expression at the individual level, the aggregate gene expression patterns were used to evaluate perturbations in the upstream regulating networks that include inflammation, tissue repair, cell fate, proliferation and stress. In Figure 2A, the impacted networks and perturbation amplitude are presented in a heatmap of Network Perturbation Amplitude (NPA) that has been normalized to the sham control. In Figure 2B, significant NPA scores were further summarized into a bar plot that compares the exposure group showing the highest perturbations (REF, the PG/W 3-hour group) relative to the other exposure groups. In the lungs, the PG/W 3-hour exposure resulted in the greatest perturbation of biological networks. The δ value indicates the perturbation direction and how closely related the underlying networks are to the reference. A value of 1 indicates that networks are perturbed by the same mechanism. These measures suggest the PG/W 1-hour exposure group was perturbed to a lesser degree than the 3-hour group, and these changes were driven by some shared mechanisms. The 6-hour PG/W exposure showed the least biological impact and these changes were driven through different mechanisms than the lower exposure groups. The PG/VG/W 6-hour exposure demonstrated a similar magnitude of biological impact as the 1-hour PG/W group, but the mechanisms driving the PG/VG/W responses were not shared with the 3-hour PG/W exposure.
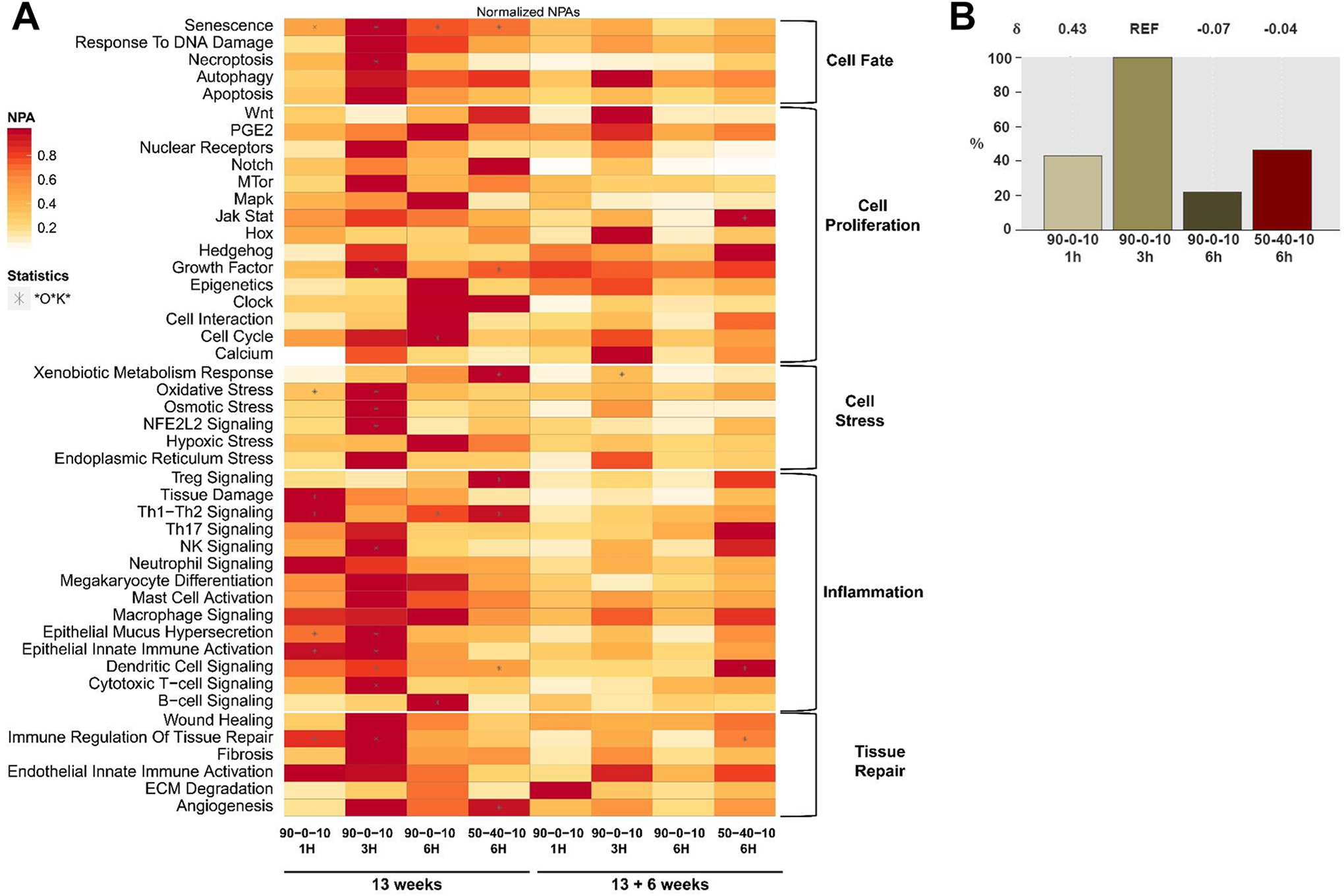
Normalized Network Perturbation Amplitude (NPA) and Biological Impact Factor (BIF) from the Lung. (A) The heatmap of normalized NPA scores from the lungs of female rats provides a summary from subnetworks. For each network (rows), the NPA scores are normalized to the maximum value across all comparisons (columns), i.e. only provide information on the relative but not the absolute amplitude of the effect (red, high; white, low relative impact). A network is considered perturbed if, in addition to the significance of the NPA score with respect to the experimental variation, the two companion statistics, O and K, derived to inform on the specificity of the NPA score with respect to the biology described in the network, are significant. An * indicate the O and K statistic p values are below 0.05 and significant with respect to the experimental variation. (B) Biological impact factor (BIF) analysis. The percentages give the relative biological impact, which is derived from the cumulated network perturbations caused by the treatment relative to the reference (defined as the treatment comparison showing the highest perturbation). Only the significant network perturbations are summarized further into this single number; hence any component entering the score is significant. For each treatment comparison, the δ value (−1 to 1) indicates how similar the network perturbations are with respect to the reference (i.e. REF). A value of 1 indicates that all the networks are perturbed by the same mechanism. Exposures were for 1, 3 or 6 hours (H) to PG/W (90-0-10) or PG/VG/W (50-40-10, Reference).
Discussion
This 13-week subchronic nose only inhalation study demonstrated that daily exposures to PG up to 5 mg/L TPM for 6 hrs/day did not induce biologically meaningful adverse effects in rats. There were no biologically meaningful adverse effects when PG/W exposures were compared to either the sham or the PG/VG/W reference. Thus, the effects observed did not result in impairment of functional capacity to maintain homeostasis or the capacity to respond to an additional challenge. Considering the organ weights in the PG/W and PG/VG/W High dose groups, the only statistically significant difference observed was in the thyroid and parathyroid of the males. The significance of this difference is unknown. The only notable histological finding was minimal mucous cell hyperplasia in nasal level II but this route of entry irritation response resolved following 6 weeks of recovery. In agreement with the lack of findings in the standard toxicological endpoints, a systems toxicology analysis of the lung showed no meaningful changes in differentially expressed genes or proteins between the exposed groups and the sham controls. Transcriptomic data were used in a network perturbation analysis that showed several networks that were significantly impacted with the greatest biological impact occurring in the 3-hour PG/W exposure. However, NPA scores only provide a relative readout of mechanistic changes, without giving direct insights into the absolute magnitude of the biological effects. As seen in a previous e-vapor aerosol inhalation study in mice by Lee et al., the amplitude of e-vapor effects was much lower than the effects after cigarette smoke (3R4F) exposure. 42
Although the PG/W 6-hour exposure group was anticipated to show a similar pattern and higher impact in the network perturbations, this was not the case. The 1-hour and 3-hour groups did show an increase in the number of networks perturbed and, based on the BIF δ value, the underlying pathway mechanisms were partially shared. At these time points, the animals may have been adapting to the exposures resulting in greater impact on the shared networks. In contrast, the 6-hour exposure groups showed fewer networks perturbed and these networks shared fewer pathways with the 1 and 3-hour groups. Suggesting the observed pattern in the NPA reflects the overall weakness of the effects.
Previous studies by Suber et al. and Werley et al. described nasal hemorrhaging in rats after exposure to PG aerosols. 14,15 In the Suber study, nasal hemorrhaging appeared during the second week of exposure and persisted throughout the study. Interestingly, Suber et al. reported a similar trend in ocular discharge that was not described as hemorrhaging. Werley et al. also reported clinical observations of bleeding from the nose and around the eyes 7-days post-exposure in an acute single dose rat inhalation study, but not in a 7-day repeat exposure study with an equivalent aerosol concentration and exposure time. In the current study, we observed clinical observations of red material around the nose and occasionally around the eyes. For the PG/W groups, there were no microscopic findings in the nose suggesting nasal hemorrhage. This suggests the red material around the nose was likely Harderian gland secretions.
The rat Harderian gland covers a large portion of the eye’s posterior and primarily functions to lubricate the eye. 43 The ducts open into the conjunctival sac and the secretions eventually enter the nose and throat via the naso-lacrimal duct. 43,44 In rats, the gland produces various porphyrins that give the appearance of bloody tears or chromodacryorrhea. 45 The Harderian gland secretions increase in response to stress (pain, illness or restraint) and may dry around the eyes and external nares resembling crusts of blood. 46,47 PG is a respiratory irritant in animal models and in humans. 48 –50 Thus, the red material reported around the nose and eyes in the current study was likely Harderian gland secretions resulting from restraint and irritation in the nose and eyes from aerosol exposures. In the current study, addition of water in the PG/W (90/10) aerosol may have reduced the propensity of PG to cause irritation-related bleeding from the nose and eyes. It is also possible that the clinical observations of red material around the nose (Harderian gland secretions) may have been mischaracterized as blood in previous studies. An additional assessment (e.g., microscopic examination of a sample smear) may help determine the identity of the red discharge.
In the Suber et al. study, microscopic evaluation of the nasal cavity showed a thickening of the respiratory epithelium described as an increase in the number of goblet cells or as an increase in the mucin content of the goblet cells. Similarly, in the nasal cavity following 13-weeks of PG aerosol exposures, we observed mucous cell hyperplasia characterized by increased numbers of closely packed goblet cells that minimally thickened the nasal mucosa. The severity of the lesions was minimal and without a clear dose-related increase, considered an adaptive response and deemed non-adverse. 51 –53 Although absolute lung weights in the high PG exposure females were significantly decreased in the Suber et al. study, we did not observe any statistically significant decreases in absolute organ weights compared to the sham controls or the reference, PG/VG/W, and there were no microscopic findings in the lung related to the PG exposures.
The systems toxicology results from lung tissue in this study are comparable to the previous studies that have demonstrated minimal effects of PG/VG exposures in the lung. 16,42 Phillips et al. tested three concentrations of PG/VG aerosols up to 1.52 mg/L PG and 1.89 mg/L VG (6 hours/day 5 days/week) in rats and found no clear effects on measured respiratory and hepatic endpoints that included histopathology, inflammation, and analyses of the transcriptome and proteome. Also, hematology and clinical chemistry endpoints did not show significant and consistent effects. Similarly, Lee et al. examined the effects of a PG/VG/Nicotine mixture following 3 weeks of exposures (41 ug/L nicotine, 4 hrs/day) in mice and observed minimal impact on the standard and systems toxicology endpoints. In addition, as compared to the cigarette comparator, the PG/VG mixtures demonstrated significantly lower impact on all the endpoints measured. 42 Although Phillips et al. and Lee et al. used different test materials and exposure regimens, the collective observations support minimal irritation effects associated with inhaled PG aerosols.
This 13-week subchronic nose only inhalation study established an exposure NOAEC of 5 mg/L for up to 6 hours for a 90:10 PG/W mixture. Based on the exposure, a delivered dose (DD) can be calculated as
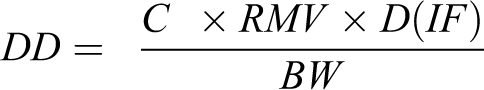
where C = concentration of substance in air (mg/L); RMV = respiratory minute volume (0.2 L/min was used); D = duration of exposure (min); IF = proportion by weight of particles that are inhalable by the test species; BW = bodyweight (0.25 kg was used). 54,55 Since the MMAD was less than 3 µm for all exposures, the IF was assumed to be 100%. 55 The measured PG concentration in the 6-hour exposure group was 4 mg/L. Thus, for PG, the daily delivered dose for the 6-hour exposure group was 1,152 mg/kg. Using body surface area scaling as recommended by the Center for Drug Evaluation and Research (CDER) and dividing the rat delivered dose by 6.2, the estimated delivered human equivalent dose (HED) is 185.8 mg/kg. 56 Based on the daily dose that was demonstrated to be without adverse effects in the 90-day study, a delivered dose of 11 g PG/day would not be expected to result in toxicological concern in a 60 kg individual.
In conclusion, following 13-weeks of exposure, the PG exposures were well tolerated and demonstrated minimal physiological, histopathological or molecular changes which is in line with previous studies. 14 –16,42
Supplemental material
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211021072 - Thirteen-week nose-only inhalation exposures of propylene glycol aerosols in Sprague Dawley rats with a lung systems toxicology analysis
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211021072 for Thirteen-week nose-only inhalation exposures of propylene glycol aerosols in Sprague Dawley rats with a lung systems toxicology analysis by T Langston, J Randazzo, U Kogel, J Hoeng, F Martin, B Titz, E Guedj, T Schneider, B Prabhakar, J Zhang, M Oldham and KM Lee in Toxicology Research and Application
Footnotes
Acknowledgement
The authors would like to thank the following individuals for contributing their expertise: At PMI, Karine Baumer, Elodie Maluenda, Dariusz Peric, David Bornand, and Remi Dulize for their technical assistance in the preparation of RNA samples and arrays and Sophie Dijon, Catherine Nury and Manuel Hernandez for their technical assistance with iTRAQ. The authors thank MC Peitsch and N Ivanov for their continued scientific support and sponsorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by Altria and in part by PMI (for the preparation, analysis and interpretation of OMIC endpoints). The authors thank MC Peitsch and N Ivanov for their continued scientific support and sponsorship.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
