Abstract
The use of electronic vapour products (EVPs) continues to increase worldwide and with advances in cell culture systems, molecular biology and the computational sciences there is also accumulating evidence of their potential reduced toxicity and reduced potential harm when compared to cigarette smoke. To further understand the potential risks and health effects associated with exposure to EVP aerosols we have assessed the cellular and transcriptomic response from a commercially available lung tissue culture system (MucilAirTM) following a single sub-cytotoxic exposure to cigarette smoke and the equivalent nicotine delivered dose of EVP aerosol. The transcriptomic, cellular (cilia beat frequency (CBF) and percent active area (%AA), trans epithelial electrical resistance (TEER), histology) and cytokine release were assessed at 4- and 48- hours following recovery from air, EVP aerosol (8.4% V/V: mybluTM blueberry flavour, 2.4% nicotine) and 3R4F smoke (3.5% V/V: exposure). No pathological changes were observed at either recovery time point from any exposure. Air and EVP aerosol exposure had no effect on CBF, %AA nor TEER at 48 hours. Exposure to cigarette smoke resulted in a decrease in TEER, an increase in CBF and the release of proinflammatory cytokines at both recovery time points. Although the number of significantly expressed genes was minimal following exposure to EVP aerosol, exposure to 3R4F smoke resulted in a significant upregulation of several disease relevant pathways. These data provide evidence that following an acute exposure to EVP aerosol there is significantly less damage to lung cells in culture than the equivalent, nicotine based, dose of cigarette smoke.
Introduction
Cigarette smoke is a dynamic and complex mixture of particulates and gases and contains more than 6000 chemicals, 1 some of which have been classified as harmful or potentially harmful by the US Food and Drug Administration. 2 Currently there are more than 1 billion cigarette smokers worldwide 3 and epidemiological studies have linked exposure to cigarette smoke with diseases such as cancer, cardiovascular disease (CVD) and chronic obstructive pulmonary disease (COPD). 4 Complete cessation of all tobacco and nicotine use is the best course of action cigarette smokers can take to improve their health. However, a growing number of international public health organizations, agencies and governments are clear that encouraging and assisting adult smokers, who are neither interested nor willing to quit smoking, to transition to alternative nicotine products that are substantially less harmful than cigarette smoke, is the next best option. 5 –12
Electronic Vapour Products (EVPs) are battery-operated devices that are designed to deliver an aerosol containing propylene glycol, glycerol and flavourings with or without nicotine. 13,14 These products do not contain tobacco but do simulate the visual, sensorial, and behavioural aspects of conventional smoking through inhalation of the EVP aerosol following heating of the e-liquid. The use of EVPs as an alternative to combustible cigarettes has expanded in recent years and as there is continued debate on the toxicological consequences of EVP aerosol exposure 15 there is the need for a greater scientific understanding of the potential benefits and risks of these products to the adult smoker, especially relative to combustible cigarettes. Although the potential toxicants associated with EVPs is a topic of current scientific interest, 16 according to the Royal College of Physicians, “the hazard to health arising from long-term vapour inhalation was unlikely to exceed 5% of the harm from smoking tobacco”. 17 This opinion has been reaffirmed by Public Health England suggesting that EVPs pose only a fraction of the harm that smoking does and that adult smokers should be encouraged to transition. 18 Evidence also suggests that e-liquids and their aerosols contain fewer and substantially lower levels of toxicants than tobacco smoke and when present, are typically at levels equivalent to tolerances permitted in medicinal products. 19,20 Therefore, exposure to EVP aerosols would be expected to elicit reduced biological responses compared with cigarette smoke exposure. However, while various studies have shown that the reduced levels of toxicants in EVP aerosols also translates into reduced In Vitro and In Vivo toxicity, 21,22 there is evidence that EVPs may induce differential effects to that of cigarette smoke, especially when different flavours are used 15 and as such the potential long-term health consequences of these products remains to be established. 23 –25
The airway epithelium is a structurally complex and functionally important part of the respiratory system. It facilitates the efficient transportation of gases to and from the alveoli; is metabolically active and is involved in a range of important homeostatic functions including, inflammation, immunity, host defence and tissue remodelling; and also acts as a structural barrier to inhaled toxicants such as those from cigarette smoke. 26,27 Early studies 28,29 into the toxicological assessment of the effects of EVP aerosols have mainly focused on routine cytotoxicity studies using submerged cell culture systems. Although these studies have shown that the cytotoxic effects of e-liquids are markedly reduced compared to cigarette smoke condensates, airway cells in adult smokers are directly exposed to whole cigarette smoke or EVP aerosols and so it is important to understand the toxicological impact of these types of exposures on the lung as they are more consumer relevant.
In recent years the use of air-liquid interface cultures, consisting of primary human lung cells cultured and exposed at the air-liquid interface, has begun to explore different exposure regimes, with realistic aerosol and smoke dilutions, flow and humidity, conditions that occur during the vaping and smoking process. 30 –32 Coupled with pathway mapping, the transcriptomic assessment following EVP aerosol exposure and other novel tobacco and nicotine delivery products have demonstrated significantly reduced toxicological impact on air-liquid interface cultures when compared to cigarette smoke. 33,34 Recently we have shown that an acute exposure of an EVP aerosol to organotypic bronchial tissue does not result in any significant toxicity when compared to matched air controls. 34 However, this study did not analyse any molecular pathway-based information that may be involved in cigarette smoke toxicity, the understanding of which may help with the development of future EVPs and the impact on disease relevant pathways. As such the objective of the current study was to explore the transcriptomic response and selected biological pathways in more detail, in addition to the cellular response of these 3D reconstituted human epithelial airway tissue cultures, following an acute and sub-cytotoxic exposure to an EVP aerosol and cigarette smoke.
Materials and methods
Test products
Blueberry flavoured mybluTM EVP (2.78% Blueberry flavour; 2.42% nicotine, 55.82% propylene glycol, 38.98% glycerol [w/w]) is a rechargeable closed “pod” system that was selected for study because it is commercially available (manufactured by Fontem Ventures, USA), it could be purchased from the US market at the time of the study and there was existing data on this type of system. 35,36 The EVPs were stored at room temperature and the batteries were fully charged before use. The comparator combustible reference cigarettes (3R4F) were US-blended king-sized products with cellulose acetate filters, with a reported International Organization for Standardization (ISO) tar yield of 9.4 mg and were obtained from the University of Kentucky (Lexington, KY, USA). The chemical and physical characteristics of the 3R4F smoke has been reported elsewhere. 37,38 3R4F cigarettes were purchased just prior to use and maintained, in their packaging at room temperature. Cigarettes were then removed and conditioned for at least 48 hours in a temperature and humidity-controlled environment (22 ± 1 oC and 60 ± 3% relative humidity) according to the ISO 3402 guideline. 39
Tissue culture
MucilAir™ tissue, a fully differentiated 3D airway epithelium, were purchased from Epithelix Sàrl (Plan-Les-Quates, Switzerland) and produced from a pathology-free, Caucasian male non-smoker (Batch No. MD072001). The tissues were maintained in proprietary MucilAirTM culture medium for a week following arrival which was refreshed every 2–3 days. Just before 3R4F smoke and EVP aerosol exposure, the tissue cultures were placed in Hanks balanced salt solution ([HBSS]: Thermo Fischer Scientific, US) without phenol red or fetal bovine serum. Directly following exposure, the tissue cultures were transferred into fresh HBSS and allowed to recover at 37°C, 5% CO2 in a humidified incubator for 4 or 48 hours.
3R4F smoke and EVP aerosol generation
Machine puffing for both 3R4F smoke and EVP aerosol was conducted on a VC1 smoking machine (VitroCell® Systems) that can deliver a range of 3R4F smoke and EVP aerosol dilutions to cells in culture. Briefly, The VC1 is a single port, single syringe smoking machine with a VITROCELL dilution and exposure module. The dilution was achieved by adding air perpendicular to the aerosol creating a turbulent and homogenous diluted mix which was pumped through to exhaust. Diluted aerosol was sampled and drawn into the exposure module via negative pressure applied by a vacuum pump 40 and where cells cultured on inserts were housed. All VC1 tubing was replaced prior to both 3R4F smoke and EVP aerosol generation and delivery. Both the sample volume (puff volume/regime) and concentration (dilution and sample flow rates) were predetermined before the exposure began. 3R4F cigarettes were smoked to the intense ISO smoking regime according to ISO 20778 41 and with a dilution flow rate of 6000 mL/Min. MybluTM e-cigarettes were puffed to a modification of the ISO 20768 regime 42 with a puff duration of 2 seconds (55 ml puff volume, 30 second puff interval, bell shape puff profile, dilution flow rate of 2400 mL/Min, exhaust duration of 8 seconds and 100% ventilation block), to ensure both EVP aerosol and 3R4F smoke generation, and deposited nicotine were equally matched.
Experimental design
No observed adverse effect level (NOAEL)
Tissue culture inserts were placed in HBSS without phenol red or fetal bovine serum in 4 wells of the VC1 module. Cells at the air-liquid interface were then exposed to 100 puffs of a range of 3R4F smoke dilutions (0.0–12.8% V/V) according to the smoking regime described above. Directly following exposure, the cell cultures were transferred into fresh culture medium and allowed to recover at 37°C, 5% CO2 in a humidified incubator for 48 hours. Cytotoxicity (adenylate kinase release [AK] and cell viability [WST-8]) was then measured and a sub-cytotoxic dilution of 3R4F smoke determined from the dose response curve.
The matching of EVP aerosol nicotine deposition equivalent to 3R4F NOAEL
To determine the dilution of EVP aerosol that delivered an equivalent amount of nicotine as the NOAEL dilution of 3R4F cigarette smoke, 100 puffs from the EVP were generated and delivered to the exposure module wells (n = 4) containing 3 mL of phosphate buffered saline (PBS) and in accordance with Behrsing et al. 40 An equivalent 100 puffs from the 3R4F reference cigarette (10 cigarettes at 10 puffs per cigarette) was also drawn and the smoke delivered to the same number of wells containing the same volume of PBS. The nicotine concentration in both solutions of PBS were then measured (Enthalpy Analytical, Irvine, US) and the ratio of nicotine derived from both exposures was used to determine the dilution of EVP aerosol that would deposit the same concentration of nicotine as the NOAEL 3R4F smoke dilution.
Main study
In the main part of the study, three separate experimental runs were conducted with eight cell cultures per exposure group (air, EVP aerosol and 3R4F smoke). Following exposure, the cultures were allowed to recover for 4 and 48 hours in an incubator (37 ± 1°C, 5 ± 1% CO2) after which four tissues were taken at each recovery time point for the non-destructive measurements of ciliary beat frequency (CBF), percent active area (%AA) and trans epithelial electrical resistance (TEER). One culture from each group was then taken for histological analysis and the remaining three prepared for mRNA extraction. Culture medium was also taken at each recovery time point and from each exposure group and stored at −60°C for later inflammatory cytokine analysis. In addition, 4 inserts per experimental run were maintained in the incubator (non-exposed) and taken for histology (n = 1) and for endpoint analysis (n = 3) 48-hours after culture medium refresh.
3R4F NOAEL cytotoxicity and cell viability measurements
Cytotoxicity was determined by measuring the release of adenylate kinase. Adenylate Kinase, an enzyme involved in energy metabolism and homeostasis is released by cells in culture following injury. 43 This is a non-destructive assay and can be conducted on the same tissue a multitude of times. AK in the culture medium was measured using the ToxiLightTM non-destructive cytotoxicity bioassay kit according to the manufacture’s recommendations (Lonza Walkersville, Inc. Walkersville, MD 21793).
To measure cell viability, a non-toxic water-soluble tetrazolium salt (WST-8) was used. This assay (Dojindo Molecular Technologies, Inc, Rockville, Maryland 20850 USA) is based on the extracellular reduction of WST-8 by NADH produced in the mitochondria resulting in a water-soluble formazan which dissolves directly into the culture medium. 44 Briefly, following recovery from exposure, tissue cultures were moved to 24 well plates containing 350 µL of culture medium and WST-8 buffer (10:1 ratio). 150 µL of the culture medium/WST-8 buffer solution was also added to the apical surface. The cell cultures were incubated (37°C, 5% CO2) for 2 hours after which the apical liquid was removed and added to the basal liquid, mixed and 100 µL measured at 450 nm.
Trans epithelial electrical resistance
Barrier integrity of each tissue was assessed at 4- and 48-hours following recovery by measuring TEER using an EVOM Epithelial Ohm Meter (World Precision Instruments, Sarasota, FL, USA). Prior to TEER measurement, 200 µl of HBSS was added to the apical surface of each tissue culture and TEER readings taken as per the manufacturer’s recommendations. Tissues were classified as having intact barrier function and deemed physiologically normal, at the beginning of the study, when TEER values were equal to or greater than 700 ohms.cm2. 45
Cilia beat frequency and percent active area
Cilia beat frequency and percent Active Area were measured at room temperature (21.5–23.3°C) using the Sisson Ammons Video Analysis (SAVA) system (Ammons engineering, Clio, MI, USA) which comprised of an inverted Nikon Te2000 microscope (Nikon Instruments, Melville, NY) coupled to a high-speed camera (Basler AC645-100 µm). Live cells were visualized at 4× magnification. For each sample a 2.6 second video was recorded at 100 frames per second (260 frames per video). The CBF data were adjusted for normal physiological temperature (37°C) by applying a correction factor according to the linear relationship between CBF and temperature and as published by Sisson et al. 46
Histology
Tissue architecture was assessed using haematoxylin and eosin (H&E) staining. Tissue from each exposure group and from each experimental run were fixed in 10% buffered formalin. These were then embedded in paraffin wax, sectioned and attached to slides using conventional histological techniques. The slices were then stained with H&E to evaluate tissue morphology. Stained slides were imaged using a Lumenera 2 camera and photographs taken of each culture using a x20 objective. The assessment of histomorphology was performed by a board-certified pathologist. Specifically, the tissues were analysed semi-quantitatively and assigned % of resident cells or scores (1–4) based on the severity of change; loss of epithelial cells (%); loss of pseudostratification (Score: 0 = no appreciable loss, 1 = up to 25% loss, 2 = 26–50% loss, 3 = 51–70%, 4 = 71–100%); loss of cilia (%); apoptotic/necrotic cells (Score 0 = no change, 1 = 3–10 cells, 2 = 11–20, 3 ≥ 20) and metaplastic changes (Score 0 = no changes, 1 = early changes, 2 = intermediate, 3 = well-formed squamous morphology).
Inflammatory cytokines
Culture medium was collected from each exposure group and each experimental run 4- and 48-hours following recovery. The culture medium was centrifuged to pellet any debris and the supernatants aliquoted and frozen at −60°C. Aliquots of the culture medium were thawed and the concentrations of secreted inflammatory cytokines (IFN-γ, IL-1β, IL-2, IL-4, IL-6, IL-10, IL-12p70, IL-13, TNF-α) were measured using the Meso Scale Discovery V-PLEX Proinflammatory Panel 1 (Cat. No 15049 D-1, Meso Scale Diagnostics, Rockville, MD) Kit according to the manufacturer’s recommendations.
RNA extraction
After completion of the WST-8 assay, RNA was extracted from tissue culture inserts in Qiazol® reagent. In brief, the apical surface of the tissues were rinsed twice (2 × 200 µl) in cold PBS (calcium and magnesium free). After rinsing, the tissues inserts were place in a 24 well plate on an ice-block and each received 150 µl of Qiazol reagent. The cellular material was then removed by carefully cutting the membrane from the tissue culture inserts and placing them into pre-labelled cryogenic storage tubes. Fresh Qiazol (550 µl) was added and the tubes frozen on dry ice prior to storage at −60°C. RNA was then extracted from a total of 54 tissue culture inserts in TRIzol® reagent according to the manufacturer’s procedure. The resultant RNA was purified via sodium acetate/ethanol precipitation followed by 70% ethanol washes. The RNA was further purified with the mRNeasy Mini kit (Qiagen), quantitated using a Qubit™ 3.0 Fluorometer, the Qubit™ RNA BR Assay Kit and assessed for quality using the Agilent 2100 Bioanalyzer and Agilent RNA 6000 Nano kit.
Whole transcriptome next generation sequencing (NGS) libraries were generated from 400 ng of total RNA template per the manufacturer’s procedure with the Illumina TruSeq® Stranded Total RNA Library Prep Gold Kit with dual indices. An aliquot of each whole transcriptome NGS library was checked for quality and mean fragment size using the Agilent 2100 Bioanalyzer and Agilent DNA 1000 kit. All NGS libraries were quantified using the Qubit™ 3.0 Fluorometer and the Qubit™ dsDNA BR Assay Kit. The KAPA Library Quantification Kit for Illumina platforms was used in conjunction with the ViiA™ 7 Real-Time PCR System to confirm the presence of adapter–ligated libraries. The mean fragment size and quantity were used in the library normalization procedure. All libraries were normalized and multiplexed/pooled with 10 nM in 10 mM Tris pH 8.5 containing 0.1% Tween 20.
cDNA libraries were pooled in batches. Each pooled library was denatured in 0.2 N sodium hydroxide, and then neutralized in 200 mM Tris pH 7.0. The pooled whole transcriptome NGS libraries were further diluted in HT1 buffer to 1.5 pM and loaded into the NextSeq™ 500 System for sequencing using High Output Flow Cells at 2 × 76 cycles.
Gene data analysis
Prior to differential gene expression analysis, 584 RNA-Seq FASTQ data files were pre-processed, followed by a quality assessment of individual reads and samples using FastqQC. On average, the libraries contained 19.2 million read-pairs, which were aligned to the Homo sapiens genome release GRCh38 using STAR aligner. After alignment and quantification, the reads that mapped uniquely to gene sequences were retained as measures of expression per gene. The set of gene counts across samples (the count matrix) was assessed using density plots, principal component analysis and hierarchical clustering to identify potential outliers. However, none were identified, and all samples were retained for downstream analysis. The count data were normalized using trimmed mean of M-values and transformed with voom, resulting in log2-counts per million with associated precision weights.
Nine comparisons were set up to identify differentially expressed features between cell culture groups following 4- and 48-hours recovery from exposure to air, EVP aerosol and 3R4F smoke (1. 4 hr Air vs 48 hr Air; 2. 4 hr EVP aerosol vs 4 hr Air; 3. 48 hr EVP aerosol vs 48 hr Air; 4. 4 hr EVP aerosol vs 48 hr EVP aerosol; 5. 4 hr 3R4F smoke vs 4 hr Air; 6. 48 hr 3R4F smoke vs 48 hr Air; 7. 4 hr 3R4F smoke vs 48 hr 3R4F smoke; 8. 4 hr 3R4F smoke vs 4 hr EVP aerosol and 9. 48 hr 3R4F smoke vs 48 EVP aerosol). An additional two comparisons were conducted between the incubator and air exposure groups (1. 4 hr Air vs Incubator and 2. 48 hr Air vs Incubator).
Gene set enrichment analysis
Gene set enrichment analysis (GSEA) was implemented using the fast-pre-ranked gene set enrichment analysis (fgsea) R package. Each individual GSEA was performed with an ordered list of genes, ranked according to the significance of differentially expressed genes in the exposure group comparisons of interest. The significance was measured using -log10 (p-values) that were subsequently multiplied by 1 or −1 to indicate up- or down-regulated genes, respectively. Ranking of genes according to this metric resulted in a list of genes ordered according to their differential expression in the comparison of interest from most up-regulated to most down-regulated genes. GSEA was then used to determine whether the genes involved in a pathway-of-interest tended to be over-represented at the extremes of this pre-ranked list of genes. The statistical significance (nominal p-value) of the over-representation was evaluated using an empirical phenotype-based permutation test with the number of permutations set to 10,000,000.
Data and statistical analysis
All data and statistical analysis were conducted using Microsoft Excel and GraphPad Prism (Version 8). To determine the NOAEL, a four-parameter logistic regression model was applied to the cytotoxicity data and the dilution of 3R4F smoke at which no cytotoxicity was observed was taken for use in the main study. A linear regression model was applied to the cell viability data to determine the level of viability at the chosen NOAEL. For TEER, CBF, %AA and inflammatory cytokines, comparisons were made between the three experimental runs using a 1-way analysis of variance (ANOVA). A 2-way ANOVA was also conducted to assess; 1. The effect of replicate samples and 2, the effect of air, EVP aerosol and 3R4F smoke exposure, at each time point, compared to each other.
Results
Cytotoxicity and cell viability
There was a sigmoidal dose response relationship between 3R4F smoke exposure and cytotoxicity (R2 = 0.99). A 3R4F smoke dilution of 3.5% was considered as the NOAEL and was taken forward for use in the main part of the study (Figure 1A). There was also a significant (p < 0.0001) linear dose-response relationship between 3R4F smoke exposure and cell viability, with a smoke dilution of 3.5% approximating to 80% tissue viability (Figure 1B).
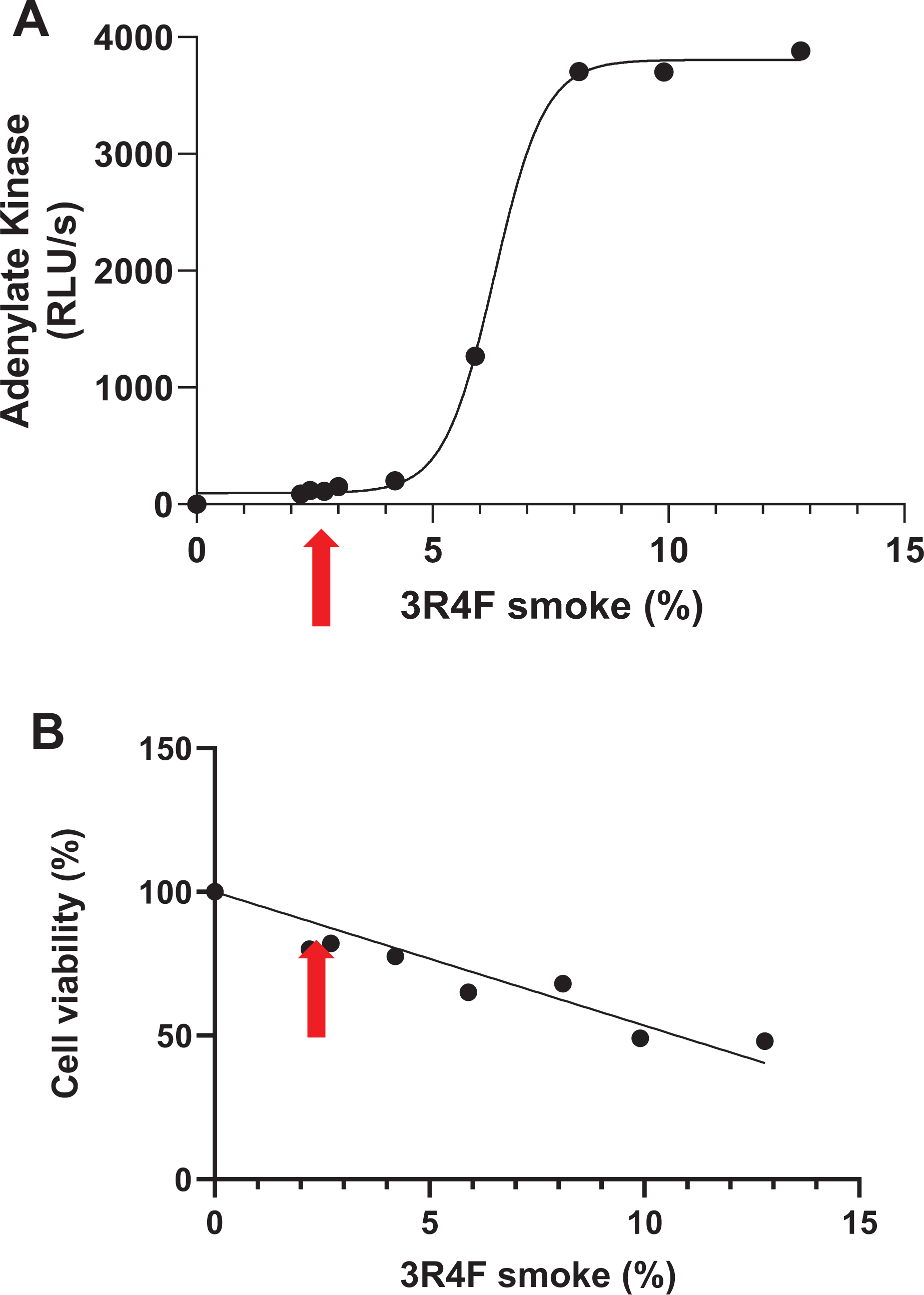
The effect of 3R4F smoke exposure on MucilAirTM culture cytotoxicity (A) and cell viability (B) following 48 hours recovery. Assays were conducted by measuring the concentration of extracellular adenylate kinase (A) for cytotoxicity and formazan production (B) for cell viability. The red arrow relates to 3.5% 3R4F smoke.
Dosimetry
Nicotine is often used as a marker of 3R4F smoke and EVP aerosol delivery. 40 Deposited nicotine in PBS following exposure to 100 puffs of undiluted 3R4F smoke and EVP aerosol was quantified. There was significantly (p < 0.05) more nicotine deposited following 100 puffs of 3R4F smoke (69.5 ± 4.5 µg/ml) than following the same number of puffs generated from the mybluTM device (28.9 ± 4.3 µg/ml). This equates to a dilution of 8.4% for the EVP aerosol to ensure deposition of equivalent nicotine levels to 3.5% 3R4F smoke.
Trans epithelial electrical resistance
The effect of air, EVP aerosol and 3R4F smoke exposure on barrier integrity is shown in Figure 2. No significant difference between the three experimental runs was observed. When compared to incubator controls (818 ± 185 Ohms.cm2) TEER from the air exposed tissues was significantly (p < 0.005) increased following 4 hours (980 ± 43 Ohms.cm2) recovery but returned to control levels by 48 hours (823 ± 90 Ohms.cm2). Following 4- (450 ± 212 Ohms.cm2) and 48-hours (633 ± 315 Ohms.cm2) recovery from 3R4F smoke exposure, barrier integrity was significantly lower than equivalent time matched air and EVP aerosol groups. No significant difference, at either recovery time point, was observed between the EVP aerosol and the equivalent time matched air controls. However, both air and EVP aerosol TEER values at 4 hours (Air: 980 ± 43 Ohms.cm2, EVP: 1006 ± 66 Ohms.cm2) were significantly (p < 0.005) higher than equivalently exposed tissue cultures at 48 hours (Air: 823 ± 90 Ohms.cm2, EVP: 841 ± 51 Ohms.cm2).
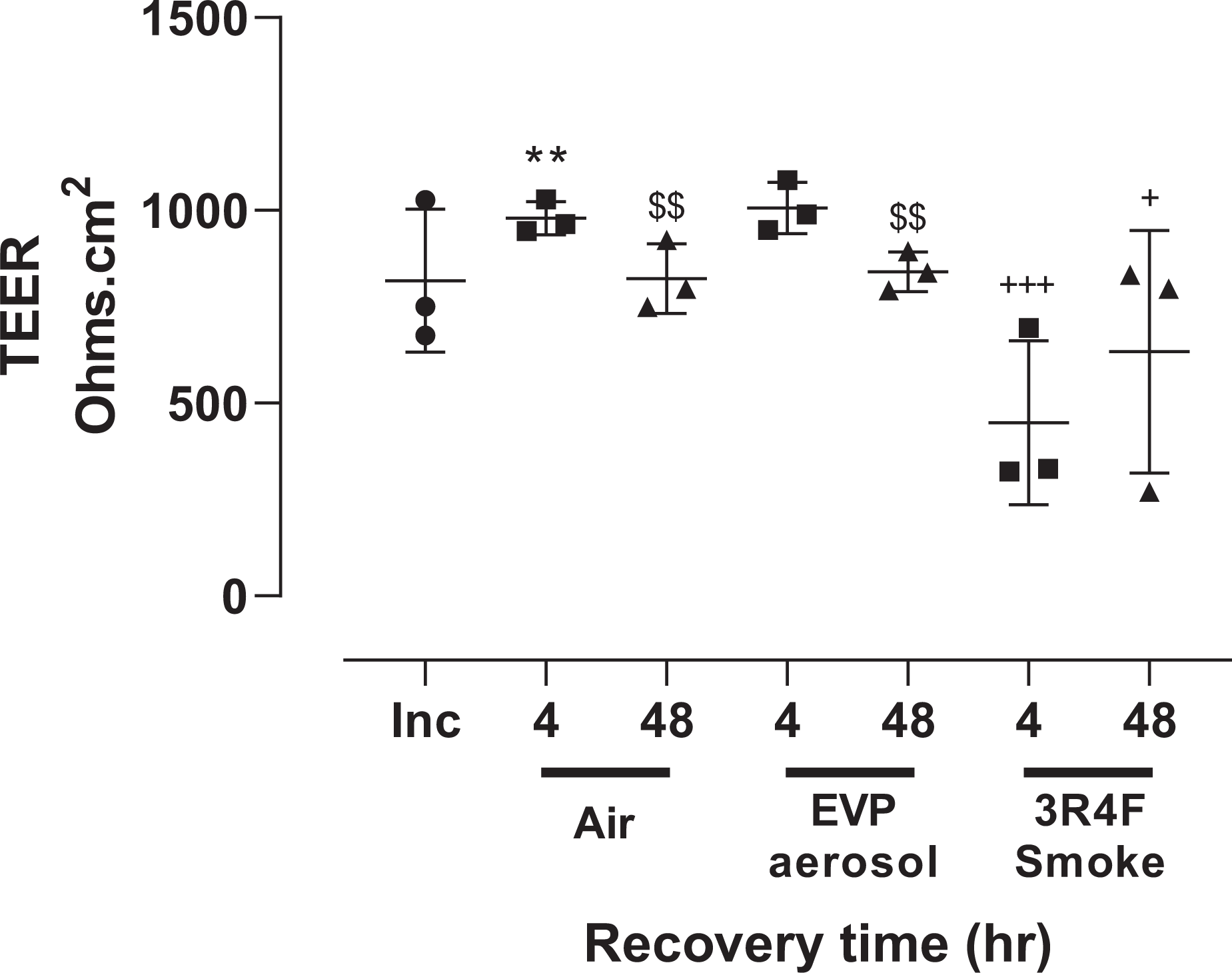
The effect of air, 3R4F smoke and EVP aerosol exposure on barrier integrity 4- and 48-hours following exposure. Data is represented as the mean and standard deviation of 3 experimental runs, each consisting of 4 replicate tissues (n = 12 tissues/time point/exposure group). Symbols refer to; (●) incubator controls, (▪) 4 hour recovery and (▴) 48 hour recovery. ** p < 0.005 compared to incubator control, $$ p < 0.005 compared to equivalent exposure groups at 4 hr and + p < 0.05, +++ p < 0.0005 compared to air and EVP aerosol exposure groups at comparable recovery time points.
Cilia beat frequency and percent active area
The effect of exposure and recovery from air, 3R4F smoke and EVP aerosol exposure on CBF and percentage active area is shown in Figure 3. No significant effect of experimental run or air exposure (compared to incubator controls) was observed for CBF (Figure 3A). Following exposure to 3R4F smoke, CBF significantly (p < 0.005) increased following 4 hours recovery (16.9 ± 0.4 Hz) and remained significantly elevated at 48 hours (16.5 ± 0.8 Hz) when compared to equivalent time matched air (4 hrs:15.8 ± 0.3 Hz, 48 hrs: 15.8 ± 0.5 Hz) and EVP aerosol (4 hrs:15.8 ± 0.2 Hz, 48 hrs: 15.4 ± 0.2 Hz) exposure groups. No significant difference between the air and EVP aerosol exposure groups were observed, nor any significant effect of air, EVP aerosol or 3R4F smoke exposure on percentage active area (Figure 3B).
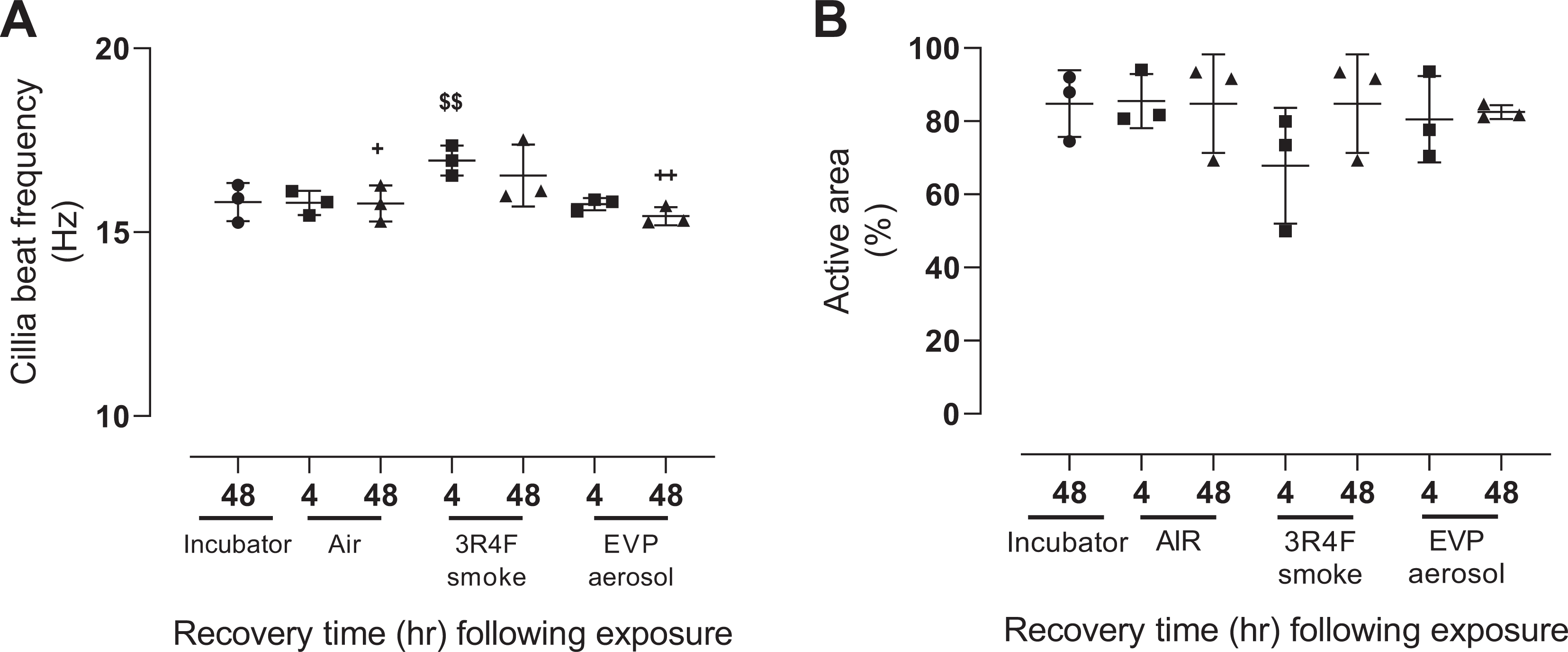
The effect of air, 3R4F smoke and EVP aerosol on cilia beat frequency (A) and percent active area (B) and 4- and 48-hours following recovery from exposure. Data represents mean and standard deviation of three experimental runs each consisting of 4 replicate tissues (n = 12 tissues/time point/exposure group). Symbols refer to; (●) incubator controls, (▪) 4-hour recovery and (▴) 48-hour recovery. + p < 0.05 and ++ p < 0.005 compared to 48 hr 3R4F smoke and $$p < 0.005 compared to time matched air and EVP aerosol groups.
Histology
No pathological changes, including loss of pseudo-stratification, loss of cilia nor any apoptotic, necrotic or metaplastic changes were observed following exposure to 3.5% smoke or the equivalent dose (8.4%) of EVP aerosol at either the 4- or 48-hour recovery time points (Figure 4).
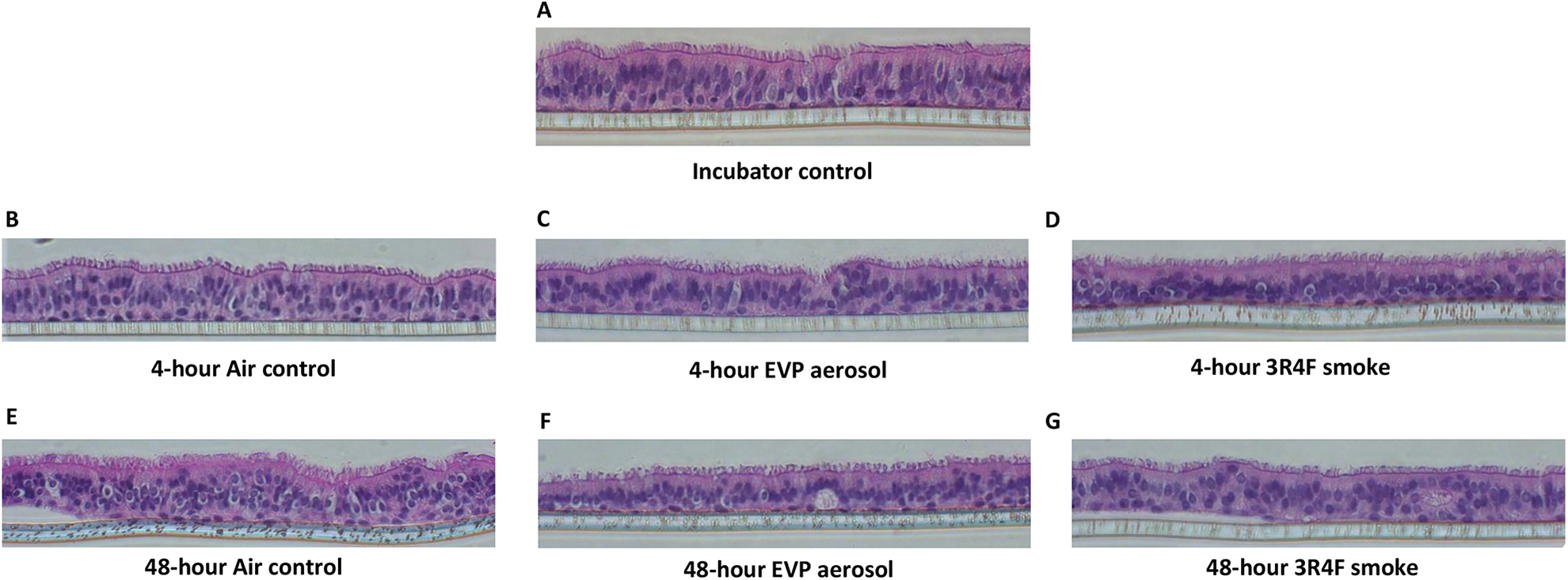
4. Tissue histology at 4- and 48-hours recovery following exposure to air, EVP aerosol and 3R4F smoke. At each time point tissues (n = 4/group) were fixed, paraffin embedded, sectioned and stained with H&E to visualize (×20) tissue structure. Figures are representative images of, incubator controls (A), air controls (B and E), EVP aerosol exposure (C and F) and 3R4F smoke exposure (D and G) following 4- and 48-hours recovery respectively.
Inflammatory cytokine release
Tobacco smoke exposure has been reported to have a strong inflammatory effect on the respiratory epithelium, 47 so a variety of inflammatory cytokines were measured in the culture medium following 4- and 48-hours recovery from air, EVP aerosol and 3R4F smoke exposure (Table 1). No significant effect of experimental run was observed on any of the cytokines measured. When compared to incubator controls, the concentration of all inflammatory cytokines were significantly lower 4 hours following recovery from air exposure. Except for IL-12p70 which was significantly (p < 0.001) elevated, by 48 hours, 8 of the 9 remaining cytokine concentrations were equivalent to concentrations found in the culture medium of the incubator controls. When compared to equivalent time matched air controls, exposure to 3R4F smoke resulted in an increase in the concentration of 6 of the 9 cytokines following 4 hours recovery. By 48 hours all cytokines were significantly increased above their equivalent time matched air controls. No significant effect was observed on the inflammatory cytokine concentrations following EVP aerosol exposure at 4 hours when compared to equivalent time matched air controls. By 48 hours, although 6 of the 9 cytokines had similar concentrations to air controls, IL-13, IL-12p70 and TNFα concentrations were significantly lower.
The effect of air, EVP aerosol and 3R4F smoke exposure on inflammatory mediator secretion following 4- and 48-hours recovery.
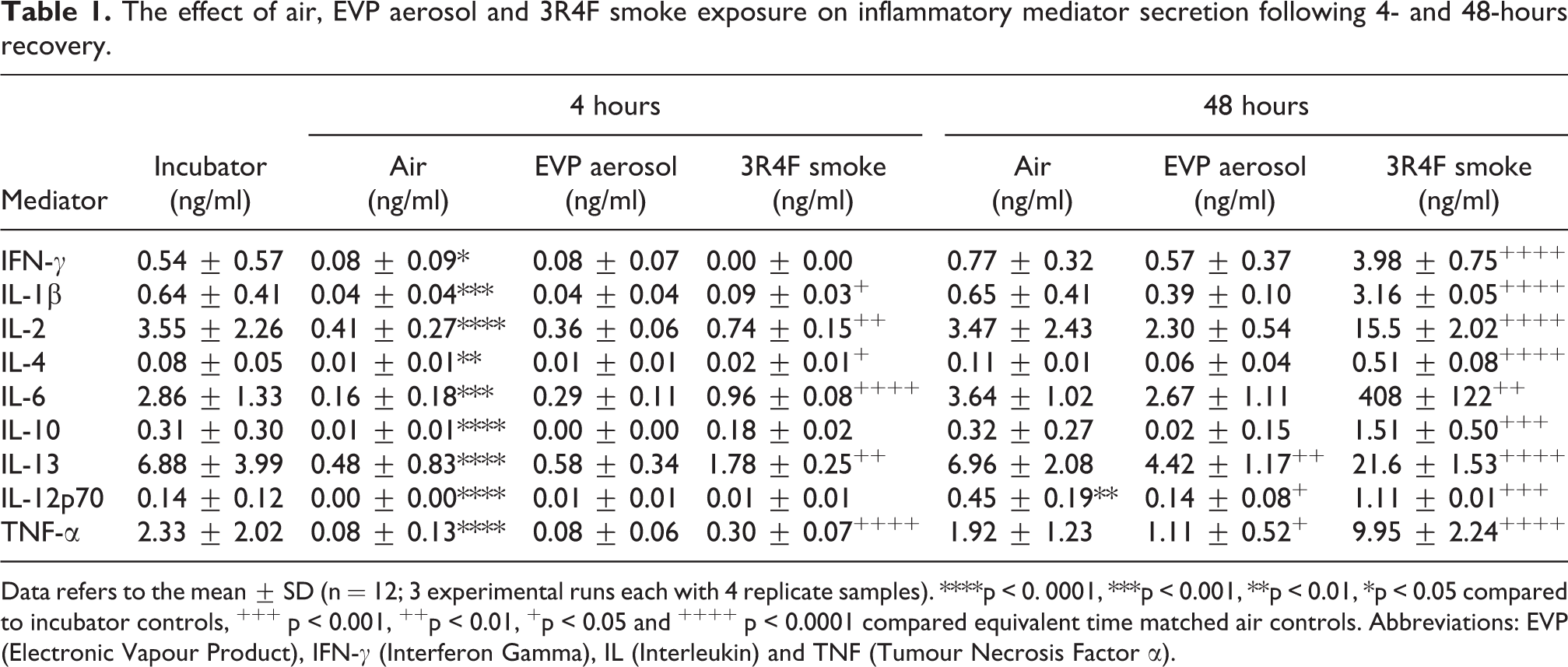
Data refers to the mean ± SD (n = 12; 3 experimental runs each with 4 replicate samples). ****p < 0. 0001, ***p < 0.001, **p < 0.01, *p < 0.05 compared to incubator controls, +++ p < 0.001, ++p < 0.01, +p < 0.05 and ++++ p < 0.0001 compared equivalent time matched air controls. Abbreviations: EVP (Electronic Vapour Product), IFN-γ (Interferon Gamma), IL (Interleukin) and TNF (Tumour Necrosis Factor α).
Gene expression profiling
Exposures were conducted on three separate occasions with three inserts for each condition to give a total of 54 samples. The transcriptomes of these samples were sequenced and measured using RNA-Seq. After alignment, the samples contained between 6.0 and 16.2 million reads that were mapped uniquely to a gene sequence in the human genome (GRCh38).
The differential expression results for six of the comparisons are shown as volcano plots (Figure 5), a heatmap overlap of the number of common differentially expressed genes (Figure 6) and the total number of up and down regulated genes, at two different fold change levels (p < 0.05 FDR and a p < 0.05 FDR with FC ≥ 2) for each comparison (Table 2). There was a significant alteration in gene expression following air exposure when compared to incubator cultures, with 1969 genes significantly up (n = 1097) and down (n = 872) regulated following 4 hours recovery (p < 0.05 FDR). Of these, only 5% (n = 92) were altered 2-fold or greater (p < 0.05 FDR, FC > 2). This gene response would indicate a degree of sensitivity of this cell culture system when air is pulsed onto the surface of the cells. This acute change in gene expression resolved quickly, as there was a >90% reduction (p < 0.05 FDR, FC > 2) in this number following a further 44 hours of recovery. However, 3 of the 92 genes that were significantly expressed at 4 hours remained significantly expressed at 48 hours (Figure 6). For all other subsequent comparisons, analysis was conducted against this background response to air and thus any observed changes in gene expression may likely represent a direct chemical response to exposure. Of the 9794 significantly expressed genes induced by 3R4F smoke exposure following 4 hours recovery (p < 0.05 FDR), 22% (n = 2199) were increased and decreased 2-fold or more. Following a further 44 hours of recovery, there was an 50% reduction in this number (p < 0.05 FDR, FC ≥ 2), with 254 genes significantly expressed at both time points. EVP aerosol exposure resulted in 142 significantly (p < 0.05 FDR) expressed genes at 4 hours, 20% of which were altered significantly altered 2-fold or more (p < 0.05 FDR, FC ≥ 2). At the 48-hour recovery time point, although there was a 57% reduction in this number, none of the 142 genes at 4 hours were observed at the later recovery time point.
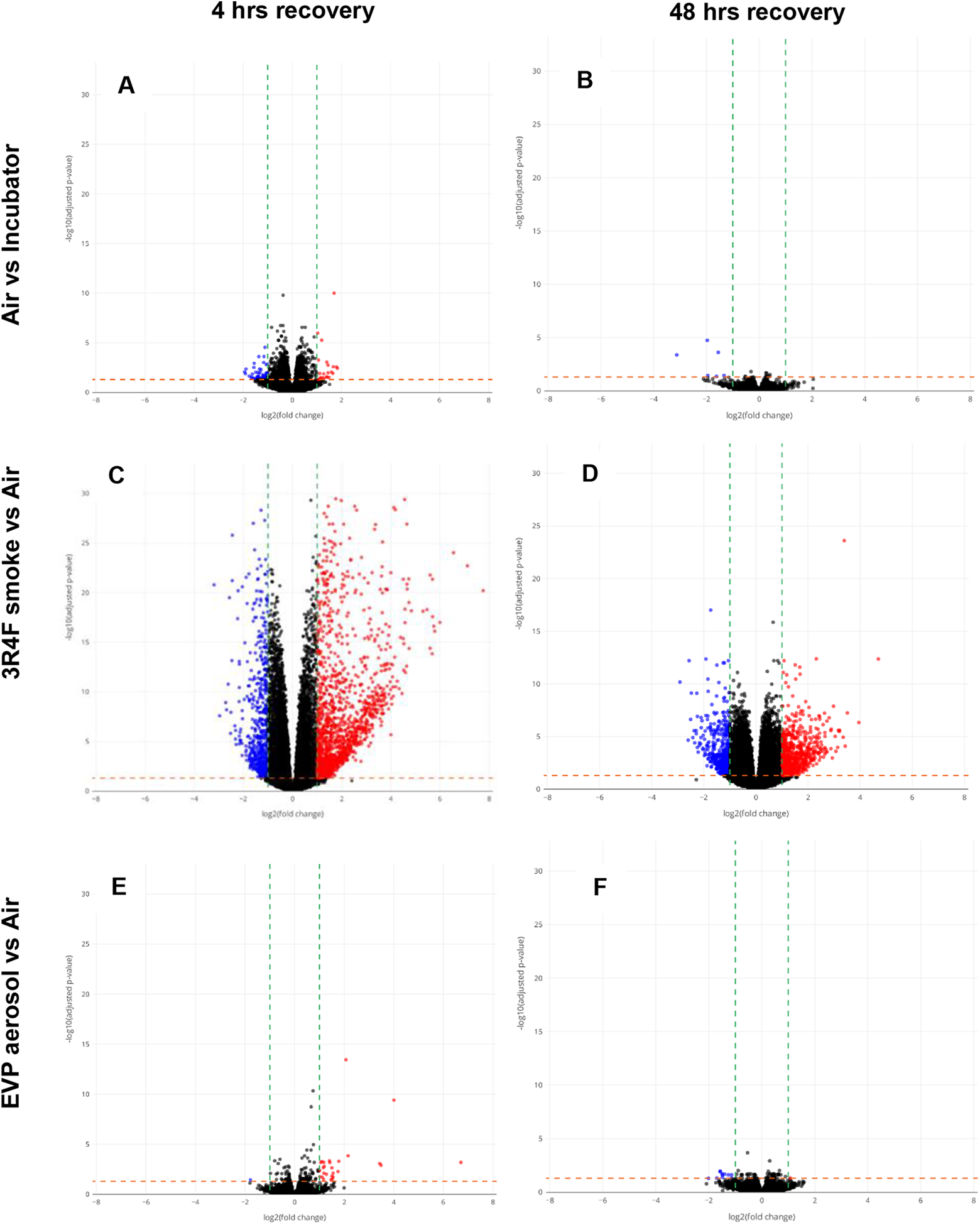
Volcano plots for six RNA-Seq comparisons showing significance as −log10 adjusted p-values against log2 fold change. Plots refer to 4 hr Air vs Incubator (A), 48 hr Air vs Incubator (B), 4 hr 3R4F smoke vs 4 hr Air (C), 48 hr 3R4F smoke vs 48 hr Air (D), 4 hr EVP aerosol vs 4 hr Air (E) and 48 hr EVP aerosol vs 48 hr Air (F). Genes identified as having different levels between samples are represented as red (up-regulated) or blue (down-regulated) dots. Black dots are displayed are a representative subsample of the full dataset. The horizontal orange line represents the applied 0.05 p-value (pFDR) threshold. The vertical green lines show the +1.5 and −1.5 fold change thresholds.
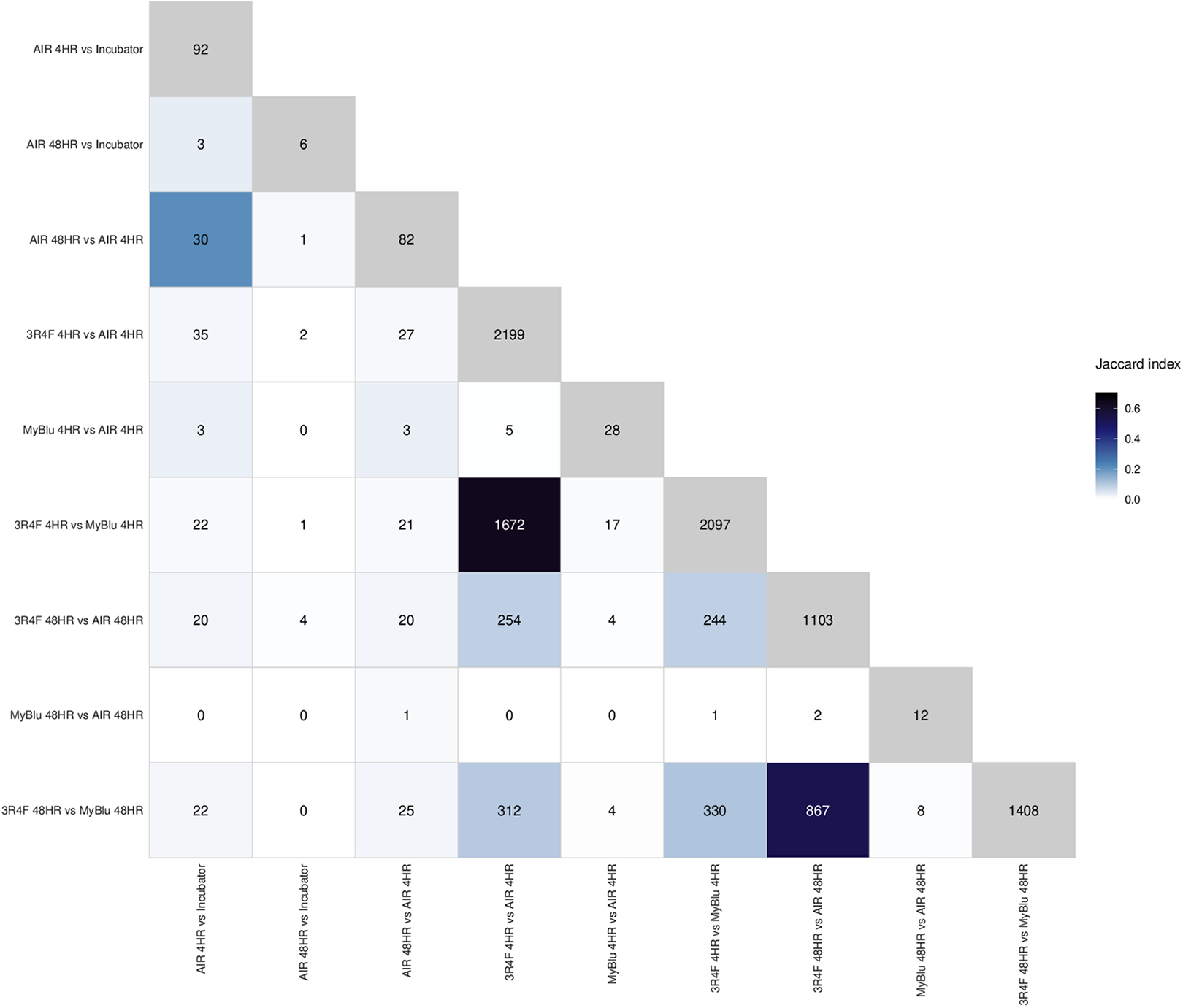
Heatmap overlaps of up- and down-regulated (y-axis) vs. up- and down-regulated (x-axis) genes for each comparison. The numbers on the diagonal represent the total number of significant gene expressions found for each comparison (pFDR < 0.05, FC > 2). The values in the plot represents the number of intersecting significant genes and the colour represents the Jaccard index (the intersection over the union) for the two comparisons under consideration.
Number of differentially significant up and down expressed genes for four comparisons following 4- and 48-hours recovery.
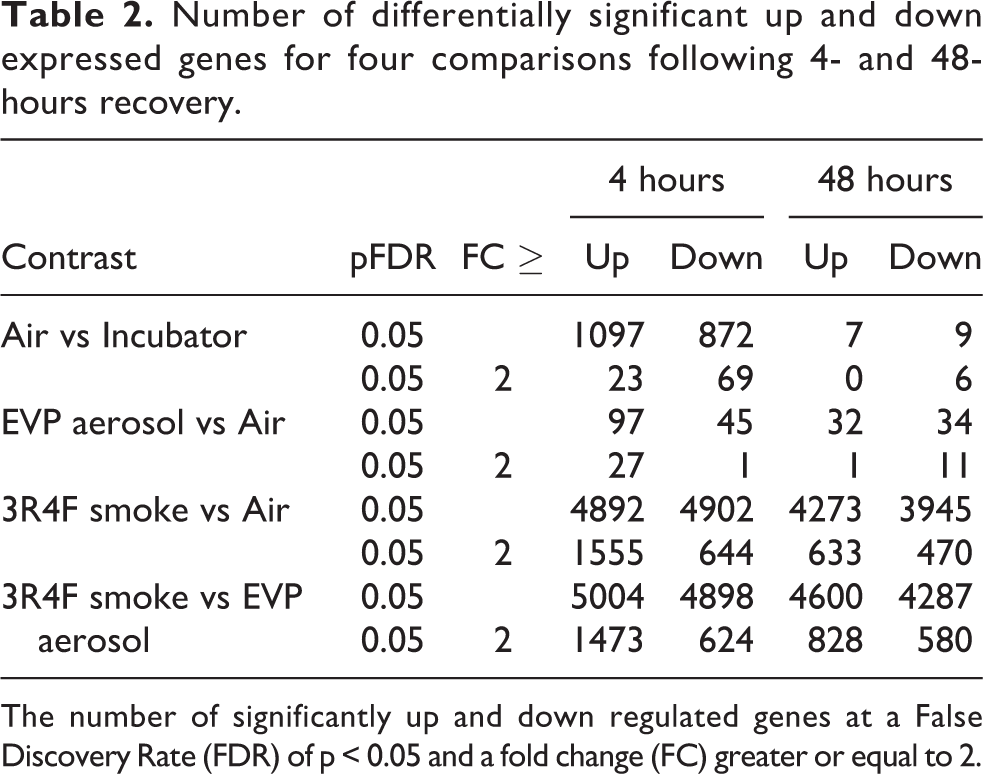
The number of significantly up and down regulated genes at a False Discovery Rate (FDR) of p < 0.05 and a fold change (FC) greater or equal to 2.
There was also a high degree of similarity in the differential gene expression response between 3R4F smoke vs EVP aerosol and 3R4F smoke vs air (Figure 6). At 4 hours recovery there was 64% similarity in the gene expression profile, which at 48 hours remained high at 54%. This may suggest a degree of similarity between the cell culture gene response to EVP aerosol and that following air exposure.
Functional enrichment analysis was also undertaken on the over-represented genes (p < 0.05 FDR and FC ≥ 2). Pathways associated with GO terms, KEGG and Reactome databases were explored, along with the collection of a 174 pathway-focused gene list (SABiociences) involved in specific biological functions and disease processes. The Reactome heatmaps for the top 50 enriched pathways for both up and down regulated genes are shown in Figure 7. This type of functional enrichment analysis reveals an over-representation of features associated with many distinct pathways and GO terms. Although many of the identified pathways were distinct and unique to each database assessed, some common pathways were observed which included cell cycle, cancer, oxidative stress and inflammation. Additionally, the heatmaps demonstrate that very few of the identified pathways were impacted upon following either air or EVP aerosol exposure.
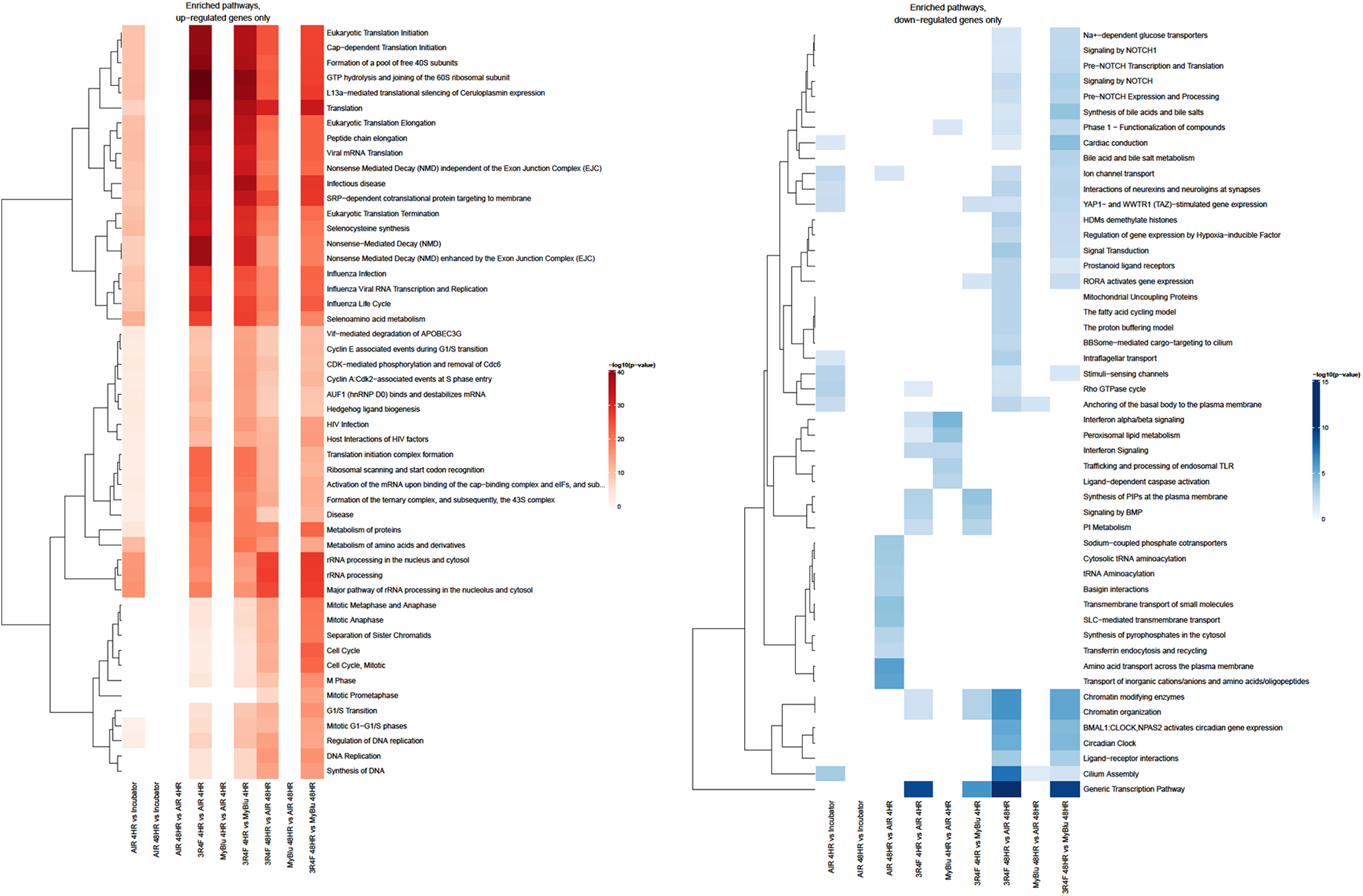
Heatmaps of significantly enriched Reactome pathways. Significant genes (at adjusted p-value < 0.05 and fold change ≥ 2) from each comparison were analysed for enrichment of top 50 Reactome pathways using a hypergeometric test by mapping genes to pathways (if appropriate). Enrichment (p-value < 0.05) was assessed for up- and down-regulated genes separately. Only the top 50 pathways are displayed. Results are shown separately for up-regulated (A) and down-regulated (B) genes and different genes of a given Reactome pathway may be both up- and down-regulated within a single comparison. Colour (red for enriched among up-regulated genes and blue for those enriched among down-regulated genes) is assigned based on the -log10(enrichment p-value), with lighter colours implying less significant enrichment. Hierarchical clustering was applied to pathways (rows). The most significant pathways were clustered according to Euclidean distance using the complete linkage method.
Further functional analysis was then conducted on specific pathways using the SABiosciences gene set list. The heatmaps shown in Figure 8 demonstrate four pathways (Apoptosis, Cell Cycle, Necrosis and p53 signalling) that were significantly impacted upon following 3R4F smoke exposure. A clear response to 3R4F smoke exposure is shown by the degree of significantly up and down regulated genes in each cluster which is replicated with many of the other disease gene sets. Interestingly the pattern of response between 3R4F vs Air and 3R4F vs EVP aerosol, at both the 4- and 48-hour recovery time points are very similar, thereby indicating that EVP aerosol exposure induces a similar response to that elicited by air exposure to cells.
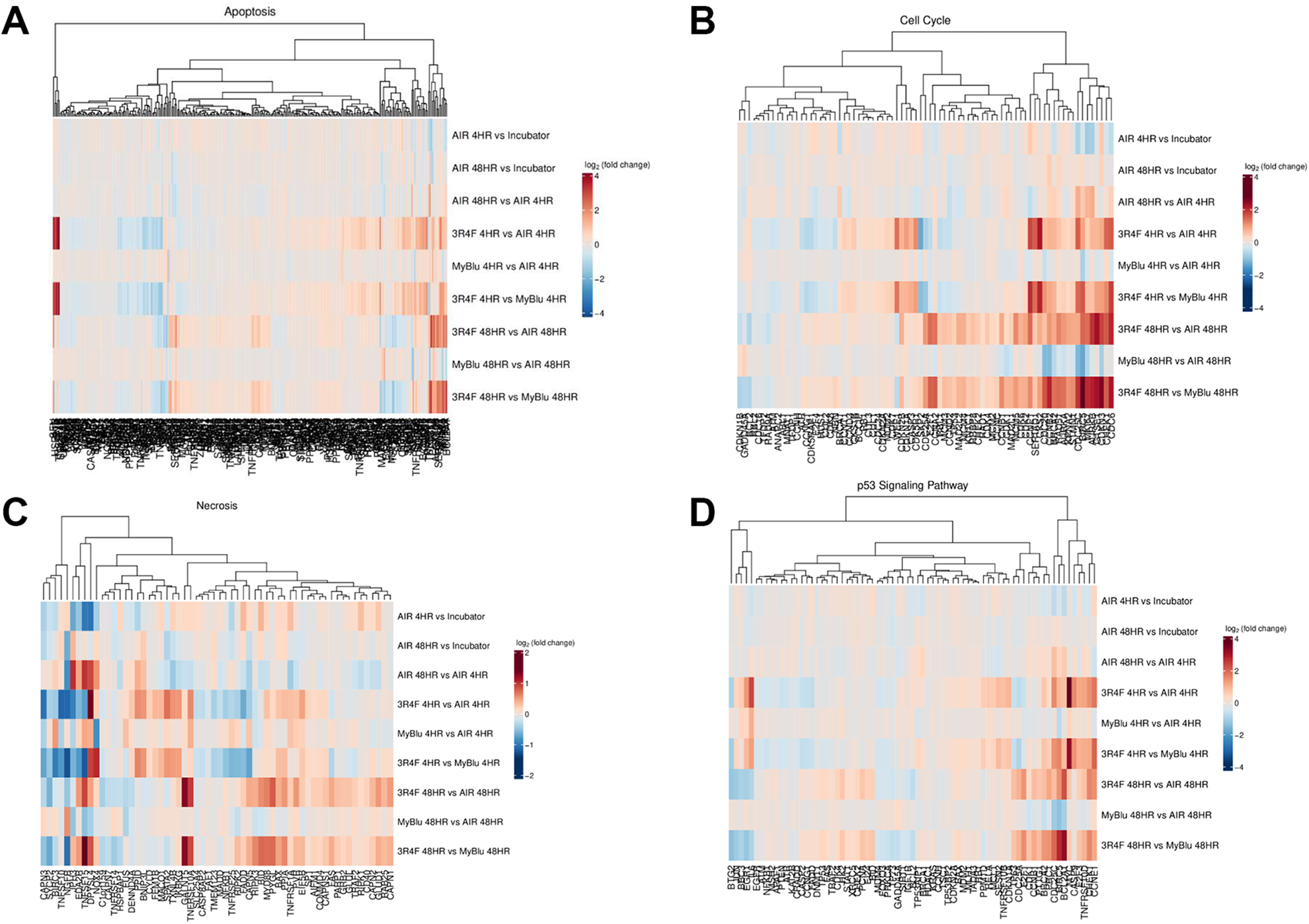
Heatmaps of up and down regulated genes for (A) Apoptosis, (B) cell cycle, (C) Necrosis and (D) p53 signalling. Significant genes (at adjusted p-value < 0.05 and fold change ≥ 2) from each comparison were analysed for enrichment in each pathway. Colour (red for enriched among up-regulated genes and blue for those enriched among down-regulated genes) is assigned based on the −log10(enrichment p-value), with lighter colours implying less significant enrichment. Hierarchical clustering was applied to pathways (rows).
Gene enrichment analysis
While standard enrichment analysis requires a pre-defined statistical significance threshold to define significantly differentially expressed genes, Gene Set Enrichment Analysis (GSEA) identifies whether the genes within a pathway tend to fall in the extremes of a pre-ranked list of genes tested. Specifically, GSEA was performed on the set of detectable genes
from this study and were ranked according to the statistical significance of their differential expression for all the possible comparisons. Focusing primarily on the enrichment of genes involved in five pathways; 1. Cell cycle, 2. Apoptosis, 3. p53 signalling, 4. Necrosis and 5. NF-kappa B signalling, GSEA was performed to evaluate if the gene sets specific to these pathways were over-represented among the most significant and differentially expressed genes from all the comparisons analysed. Figure 9 shows the effect of the differentially expressed gene set on the cell cycle pathway only. None of the five pathway-specific gene sets were enriched among the most differentially expressed genes when 3R4F smoke exposed cultures were compared to air or EVP aerosol exposed cultures at the 4-hour time point. However, by 48 hours, genes involved in Necrosis and Cell cycle were significantly over-represented. Following recovery from exposure to EVP aerosol (compared to 4 hr air), only genes involved in the NF-kappa B signalling pathway appeared to be significantly over-represented and more specifically only in the up-regulated gene set. By 48 hours, this observation was lost, and no gene set involved in the five pathways-of-interest were over-represented.
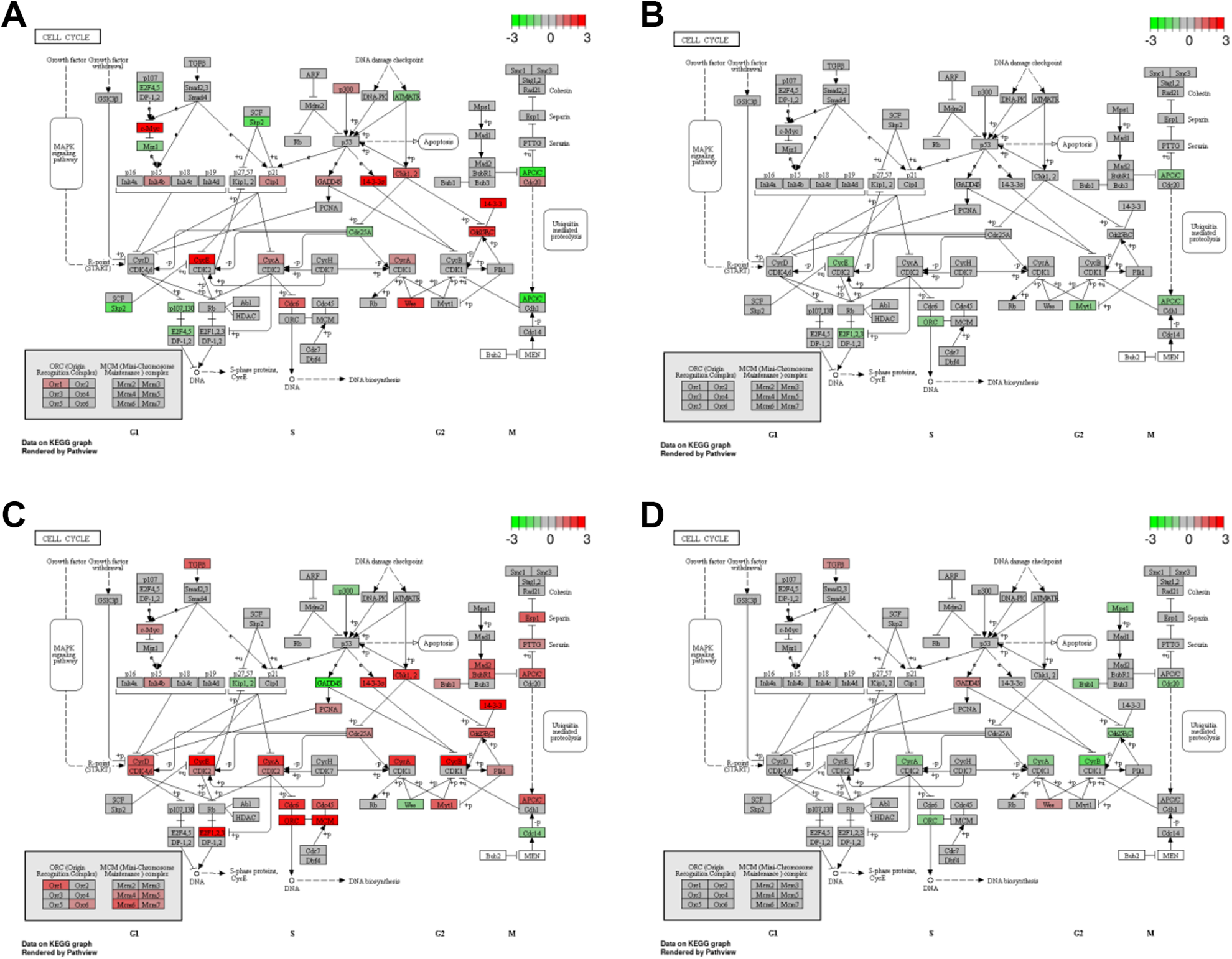
The effect of 3R4F smoke (A and C) and EVP aerosol (B and D) on the cell cycle pathway. Effected pathways are shown at 4 hours (A and B) and 48 hours (C and D) following exposure. Colour (red for enriched among up-regulated genes and green for those enriched among down-regulated genes) is assigned based on the -log10(enrichment p-value), with lighter colours implying less significant enrichment.
Discussion
The objective of this study was to assess the cellular and transcriptomic response of an In Vitro 3D organotypic model of the human airway following an acute, sub-cytotoxic exposure to 3R4F smoke and EVP aerosol. These 3D models have the advantage, over simpler cell culture systems of having a long-shelf life while maintaining their original lung tissue phenotypic and biochemical profile. 48 They also more closely represent human morphological, biochemical and transcriptomic responses seen following cigarette smoke exposure, 49,50 suggesting that these cultures maybe useful tools for the assessment of chemical induced injury to the lung.
Inhaled cigarette smoke consists of toxic gases and particulates that contain thousands of chemicals 1 and over many years, exposure to this mixture can result in the development of a variety of diseases, including COPD. 51,52 Chronic cigarette smoke exposure injures the airway epithelium resulting in a sustained recruitment of immune and inflammatory cells, alterations in the number and type of cells that make up the lung tissue, disruption of the junctions between epithelial cells, 53 the loss of cilia, reduced ciliary beating, and mucus hypersecretion, all resulting in significant tissue destruction, altered repair and potentially life-threatening airflow limitation. The evaluation of the early, sub-cytotoxic smoke-epithelial interactions was conducted to provide insights into some of the initiating responses that can lead to these adverse health effects. However, to ensure accurate and equivalent (dose matching) comparisons can be made between different tobacco related products, the use of common markers is required. To dose match, previous studies have used parameters such as puff number 54 or deposited mass, 55 while others have not considered delivered dose at all. 56,57 As tobacco product users achieve their nicotine dose in different ways 58,59 and as there are fundamental differences in the chemical composition between EVP aerosols and cigarette smoke, 19,20,60 the effect of product comparisons made on puff number and deposited mass may not be the best approach to use. In the current study 3R4F smoke and EVP aerosol were delivered to air-liquid interface cell cultures using the same puff volume, puff number, puff duration and at the same nicotine dose, thus allowing for a more exacting and equal comparison between products to be made. Although this approach generated EVP aerosols using a modified ISO smoking regime 42 with a 2 second rather than a 3 second puff duration and a bell shaped puff profile, this approach is in line with previous studies 61 –63 in which modifications have been made to standardized exposure protocols to ensure equivalent dosing comparisons can be made. Although no smoking regime can capture the human individual variation in puffing parameters the target of this work was to deliver equal doses of 3R4F and EVP, at sub-cytotoxic levels, to allow a direct transcriptomics comparison to be made.
Following exposure to 3.5% 3R4F smoke and 8.4% EVP aerosol, there was no loss of pseudostratification, cilia, or any apoptotic, necrotic or metaplastic change at either the 4- or 48- hour recovery time points, processes that are known to occur at much higher doses, and at much later time points. 64 The use of this sub-cytotoxic dose thus allowed for a cleaner assessment of the effects of both cigarette smoke and EVP aerosol exposure without the complex changes that occur in the presence of necrotic and apoptotic tissue. Barrier integrity (TEER) and CBF, with the corresponding percentage of cellular surface containing cilia, were assessed. 3R4F smoke exposure resulted in a significant reduction in barrier integrity and is consistent with observations in humans, 65 3D organotypic cultures 66,67 and animal studies. 68 This decrease in barrier integrity may increase mucosal permeability and consequently increase the movement of viruses and bacteria across the epithelium, an observation that is seen in nearly 50% of COPD patients during periods of exacerbations. 69 Reduced ciliary beating has also long been recognized to impact mucociliary clearance. 70 However, data on the effect of smoking on CBF has been conflicting. Cigarette smoke exposure can lead to either reduced 71,72 or increased cilia beat frequency when compared to non-smokers. 73 In the current study, the increase in CBF after exposure to 3R4F smoke likely represents the response to a low dose of cigarette smoke or/and an initial, early response before cilia toxicity becomes evident. 74,75 Although CBF and the number of active cilia on the surface of epithelial cells are known to be impacted upon following exposure to a variety of next generation nicotine delivery products, 31 EVP aerosols have minimal effects on mucous transport velocity measurements, a measurement of mucociliary clearance, 76 and little or no effect on CBF when compared to air controls. 33 As in all studies, much of the changes observed are dose dependent. However, at low doses and at doses comparable to 3R4F smoke exposure, where significant effects were seen on TEER and CBF, EVP aerosol exposure had no effect.
These changes following cigarette smoke exposure are known to be driven by inflammatory cytokines released from a variety of sources including the airway epithelia. The airway epithelium is highly dynamic and displays a broad spectrum of activities related to inflammation, immunity, host defence and tissue remodelling. 77 In the current study, air exposure had little effect on cytokine secretion, with IL-12p70, the only mediator elevated significantly above incubator controls following 48 hours recovery. Following cigarette smoke exposure, all 9 cytokines were significantly increased above equivalent air controls at the same 48-hour time point and is consistent with other In Vitro 34,78 and In Vivo 68 exposure studies. Similar changes have also been shown in COPD patients where there is evidence of impaired host defence. 79,80 IL-6 a pleiotropic proinflammatory mediator, was increased more than 100-fold following cigarette smoke exposure and plays an important role in the propagation of chronic inflammation and the accumulation of inflammatory cells at sites of injury. 81 IL-6 has also been implicated in the pathogenesis of COPD and elevated levels are found in the plasma 79 and sputum 82 of these patients, levels which have been shown to correlate with impaired lung function. 82 The anti-inflammatory cytokine IL-10 was also over expressed in the heat maps for 3R4F at 4 and 48 hours when compared to air and EVP exposure, as were IL-4 and IL-13 at 48 hours. This over expressed family of genes also correlated with an increased concentration of cytokines in the cell culture medium. Interleukin 10 (IL-10) has potent anti-inflammatory properties that play a central role in limiting the host immune response to pathogens, thereby preventing damage to the host while maintaining normal tissue homeostasis. 83 The expression of IL-10 is closely linked to TNF alpha production, 84 both of which have been identified in the lungs of patients with acute respiratory distress syndrome (ARDS), with higher lung fluid levels of IL-10 associated with improved survival. 85 Interestingly, exposure of the tissue cultures in this study to an equivalent dose of EVP aerosol had little impact on inflammatory cytokine secretion and by 48 hours all cytokines were consistently lower than their time matched air controls. Although, increased levels of TNF-α, IL-6 and IL-13 has been associated with lung disease, 86 the effect of EVP exposure is more variable with studies showing an increase 78 while others show no effect. 68 The reason for this is unknown, but the variety of exposure methods, doses and the different products used can vary the delivered dose quite significantly and thus vary the tissues response. Further studies on the effect of dose, long term and repeated exposures and the response of different tissue donors are required to fully differentiate the magnitude of the difference observed between these products.
One of the main goals of this study was also to assess the transcriptomic response of these 3D organotypic lung cultures to an acute sub-cytotoxic exposure to both EVP aerosol and cigarette smoke. Results from the air controls demonstrated that this type of tissue culture system is very sensitive to external influences. When compared to untreated incubator controls there was a significant change in transcriptomic regulation with 92 genes significantly altered (pFDR p < 0.05 and FC > 2) 4 hours following air exposure. However, the tissue quickly recovered with a 90% reduction in this number by 48 hours, demonstrating that air exposure can result in significant transcriptional changes, even in the absence of any measurable functional or cellular response. Exposure to 3R4F smoke resulted in a major transcriptomic response at 4 hours, with 2199 significantly over and under expressed genes. Although there was approximately a 50% reduction in this count following a further 44 hours recovery, the major reduction was seen in the number of upregulated genes. EVP aerosol exposure had a limited impact on the transcriptomic response following exposure. This response, which was less than 1% of that following 3R4F smoke exposure reduced, again by approximately 50% by 48 hours. Interestingly, on assessing the 3R4F response relative to the air control and the equivalent EVP aerosol response, a similar number of genes were up and down regulated. On closer inspection the transcriptomic response between the two comparisons were very similar (Figure 6), even at both recovery time points, and may indicate that EVP exposure has a similar response profile to that following air exposure. This further supports the cellular responses seen in this study, in that EVP aerosol exposure, has minimal impact on the tissue when assessed following an acute sub-cytotoxic exposure and at time points during the early period of recovery.
Functional enrichment analysis was undertaken on the over-represented genes (p < 0.05 FDR and FC > 2) using pathways associated with GO terms, KEGG and Reactome databases and demonstrated that many pathways were impacted upon following exposure to 3R4F smoke, including oxidative stress and inflammation. These pathways have been implicated in numerous studies following cigarette smoke exposure, conducted both In Vitro and In Vivo and in tobacco related human lung diseases such as COPD and cancer. Exposure of the tissue culture to an equivalent dose of EVP aerosol resulted in a significantly reduced transcriptomic response compared to cigarette smoke, with those pathways impacted upon by cigarette smoke not being observed in the response to EVP aerosol exposure (Figure 7). In fact, of the >2000 significantly regulated genes observed following 3R4F smoke exposure only 5 were also seen following EVP aerosol exposure. Thus, due to this low response and the minimal overlap of significantly regulated genes, it is not surprising that the pathways impacted upon by 3R4F smoke exposure were not evident following EVP aerosol exposure.
Further gene set enrichment analysis focused on five common pathways that had been impacted on following 3R4F smoke exposure across three different databases (GO terms, KEGG and Reactome) and included cell cycle, apoptosis, p53 signalling, necrosis and NF-kappa B signalling, pathways common in many cigarette smoke related diseases. As before, the enrichment of pathway genes following 3R4F smoke exposure was most evident among the up-regulated genes. Overall, changes to the lung cell gene expression profile following EVP aerosol exposure tended to be small and were associated with pathways mainly at the 4-hour recovery time point having been largely resolved 44 hours later. Exposure to 3R4F smoke on the other hand, had large effects across a broad range of pathways and were mainly found to significantly affect pathway-specific genes after 48 hours recovery. In fact, of the 5 key pathways investigated, genes involved in necrosis and not apoptosis were significantly over-represented, a cellular response that is observed at much higher cytotoxic doses of cigarette smoke. The exact mechanism of necrosis was not determined, as a growing list of triggers have been reported to lead to necrosis including the involvement of cytokines, pathogen-associated molecular patterns (PAMPs), excitotoxins, cell irradiation, DNA damage from alkylating agents or oxidative stress that can all initiate necrotic cell death, showing a developing model of an intricate inter relationship between both necrosis and inflammation. 86 However, even with the evidence that the necrotic pathway has been activated and that cytokines involved in this process were found in the culture medium, no evidence of such damage was observed histologically. Although, the main design concerned a sub-cytotoxic dose, it may be too early for any structural alterations to manifest. Alternatively, the disconnect between the transcriptomic observations and the pathological evidence may be the result of the many checks and balances that occur at all levels of organization, resulting in no damage to the tissue. Further longer recovery time points will be required to assess this. These results therefore suggest that the immunological and genetic/transcriptomic response to EVP aerosol exposure is less extreme and is resolved quickly, whereas the response to 3R4F smoke is much slower, more profound, and may continue even after 48 hours recovery.
The data presented supports the adverse effect of cigarette smoke exposure on the cellular and transcriptomic response of primary human lung cells in culture. Cigarette smoke exposure results in the enrichment of pathways associated with oxidative stress and inflammation, among others, and supports similar observations from both In Vitro studies and studies conducted in humans. In contrast, exposure to an equivalent nicotine dose of EVP aerosol shows minimal effects at all levels of organization. Although EVP exposure induced a similar response to that observed by tissue cultures exposed to air, several pathways were impacted upon and may indicate that EVP exposure can have a different toxicological impact on cells 15 than that observed following cigarette smoke. However, this reduced impact following EVP aerosol exposure has been seen over a very short recovery period and as such further studies are required to address the toxicological effects of repeated exposure and longer recovery periods on this type of tissue culture system. Additionally, the tissue used in the current study was also derived from a single, healthy male donor. Populations show extensive variation in their biological response to injury and as such further studies with several donors of different genders, age and ethnicity, including smokers, would be required to fully ascertain active pathways common to the early response to cigarette smoke and EVP aerosol exposure. Also, additional scope for improvement can be achieved by addressing product use and vaping behaviour in the experimental design as these parameters are known to differ substantially between users. In a recent systematic review 87 of EVP use, it was highlighted that it was difficult to estimate the precise difference in the harmful effects of exposure to vaping when compared to smoking, but it was concluded that EVP aerosols result in exposure to far fewer toxic chemicals and at much lower or trace levels. Further chemical studies will also be required to fully elucidate all the chemicals present in the EVP aerosol and the changes in these compounds that may occur during use across the multitude of products currently on the market. However, even with low or trace levels of inhaled toxicants, EVP aerosol exposure is known to produce mild adverse reactions (e.g., irritation, nausea) and has been associated with a variety of pulmonary presentations ranging from lipoid pneumonia to diffuse alveolar haemorrhage 88,89 in some users and since the summer of 2019, an acute respiratory illness 90 in a small cohort of vapers. The Centre for Disease Control and Prevention (CDC) has termed this disease, e-cigarette or vaping product use-associated lung injury (EVALI) and is primarily associated with males under the age of 35 91 and with those more severely affected demonstrating with pre-existing health conditions. In most of these EVALI cases individuals were found to be vaping unregulated ‘off street’ and illicit cannabis products, usually cannabis oils that contain tetrahydrocannabinol. Testing of both the e-liquids and samples from the airways of these vapers have found the presence of vitamin E acetate (VEA), 92 a known cutting agent for cannabis oils, and as such VEA is currently considered to be a potential toxic agent involved in the development of this type of lung disease. While there is evidence associating VEA with lung injury in animals, 93 the precise mechanism of action that leads to this type of respiratory illness in man is still yet to be fully elaborated. These observations have been invaluable in explaining the role of specific chemicals found in the e-liquids and aerosols of EVPs and in helping prevent the adulteration of these liquids and products. 94 Finally, there is still the lack of good, longitudinal clinical studies to assess the long-term toxicity of these types of products, especially in those groups considered to be at risk. 95 Although there is still much pre-clinical and clinical research to be conducted on these relatively new products, there is early evidence on the clinical improvements in smokers who have switched to EVPs. 96 –98 These new clinical and/or epidemiological studies will ultimately define the potential of these products as tools for smokers who find it difficult to quit and who would otherwise continue smoking. Further, this in vitro study adds to the increasing body of scientific weight-of-evidence supporting the potential role of high-quality EVPs as less harmful products when compared to cigarettes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
