Abstract
E-vapor products (EVP) have become popular alternatives for cigarette smokers who would otherwise continue to smoke. EVP research is challenging and complex, mostly because of the numerous and rapidly evolving technologies and designs as well as the multiplicity of e-liquid flavors and solvents available on the market. There is an urgent need to standardize all stages of EVP assessment, from the production of a reference product to e-vapor generation methods and from physicochemical characterization methods to nonclinical and clinical exposure studies. The objective of this review is to provide a detailed description of selected experimental setups and methods for EVP aerosol generation and collection and exposure systems for their in vitro and in vivo assessment. The focus is on the specificities of the product that constitute challenges and require development of ad hoc assessment frameworks, equipment, and methods. In so doing, this review aims to support further studies, objective evaluation, comparison, and verification of existing evidence, and, ultimately, formulation of standardized methods for testing EVPs.
Highlights
E-vapor products research is complex and rapidly evolves in application of various assessment methods.
Various experimental setups and methods for e-vapor products assessment are provided and reviewed.
Formulation of standardized methods and development of assessment frameworks are required.
Introduction
E-vapor products (EVP) have become popular alternatives for cigarette smokers who would otherwise continue to smoke. 1 In one of the most comprehensive reviews of the evidence on the health effects of EVPs to date, the United States (US) National Academies of Sciences (NAS) stated that “there is conclusive evidence that completely substituting EVPs for combustible tobacco cigarettes reduces users’ exposure to numerous toxicants and carcinogens present in combustible tobacco cigarettes.” 1 Likewise, a recent systematic review of published empirical research literature found that studies indicate that EVPs pose substantially less harm to smokers than cigarettes. 2
An ongoing debate exists in the public health community about the long-term health effects of EVPs and their value as less harmful sources of nicotine or as smoking cessation aids. 1,3 The NAS pointed out the lack of long-term data from repeated inhalation exposures and recommended to “conduct long-term (2-year) animal studies, using inhalation exposure to e-vapor, to better understand disease risks from inhaling reactive carbonyl compounds and other potentially toxic constituents of e-vapor, including flavoring chemicals and additives.” 1 The NAS report 1 provides recommendations for the improvement of current research and highlights gaps that are a priority for the future research. This situation provided the motivation for our review to contribute to the optimization and standardization of the existing and future technologies.
EVP research is challenging and complex, mostly because of the numerous and rapidly evolving technologies and designs as well as the multiplicity of e-liquid flavors and solvents available on the market. The lack of manufacturing standards also adds uncertainty to the relevance of research findings because of variations in product quality. Furthermore, the absence of standardized assessments makes research data difficult to evaluate, interpret, and compare.
These considerations highlight an urgent need to standardize all stages of EVP assessment, from reference product production to e-vapor generation methods and from physicochemical characterization methods to nonclinical and clinical exposure studies 1,4 –9
The objective of this review is to provide a detailed description of selected experimental setups and methods for EVP aerosol generation and collection and exposure systems for in vitro and in vivo assessment. The focus herein is on the specificities of the product that constitute challenges and require development of ad hoc assessment frameworks, equipment, and methods. In so doing, this review aims to support further studies, objective evaluation, comparison, and verification of existing evidence, and, ultimately, formulation of standardized methods for testing EVPs.
E-vapor products are not risk-free and the systematic toxicological evaluation of residual or novel health risks as postulated by the public health community is the reason and motivation for this publication, which aims at an optimization and standardization of the equipment and technology to render such investigations meaningful and comparable. Any reviewing, discussion, or prioritization of the potential health risks is out of the scope of this technology review article and readers interested in these topics are kindly referred to the numerous publications on this topic, such as references, 1,3,5,7,8,10 –12 and many more.
E-vapor products
E-vapor product devices
EVPs are battery-powered devices that generate an aerosol for inhalation by heating a liquid formulation (the “e-liquid”) to temperatures reported as ranging from 40°C to 300°C. 6,10,13 Most EVPs use a wick and coil system to transport and heat the e-liquid. The wick draws the e-liquid into the coil-heating element, where it is heated to form a supersaturated vapor, which is subsequently cooled by an air flow to form an aerosol through a thermal nucleation process.
The aerosol generated by EVPs is often called “e-vapor,” which is the terminology used in this manuscript to distinguish it from the aerosols generated by other tobacco products, such as cigarette smoke (CS), or aerosols from other electronic nicotine delivery products (ENDP).
EVPs have been on the market for a little more than a decade. Yet, the product category has experienced, and continues to experience, a rapid pace of innovation, with evolving technology and novel product designs being constantly added to the marketplace. Overall, products have moved from disposable or rechargeable closed systems with prefilled cartridges (“cigalikes” resembling cigarettes) to open systems featuring a prominent, refillable tank and a separate battery (Figure 1). A distinctive feature of EVP product evolution is the capacity of new-generation products to deliver more power than earlier designs. EVP batteries operate at voltages ranging from 3 to 6 V, and many devices allow the user to adjust the voltage. Resistance of the coils also varies, with values commonly ranging from 1.0 to 6.5 Ω. 6 Together, voltage and resistance determine the EVP power output. For example, a device with a battery voltage of 4.2 V and coil resistance of 1.5 Ω is expected to deliver 11.8 W, in contrast to an approximately 4 W for a typical cigalike device. 14 Increased power delivery influences operating temperature and may affect the generation, quality, and chemical constituent yields of e-vapor. 15

Evolution of EVPs, by product generation and characteristics.
E-liquids
E-liquids are typically composed of aerosol formers and varying flavoring substances, with or without nicotine. 6,16
Aerosol formers
The most common aerosol formers used in EVPs are propylene glycol (PG) and vegetable glycerol (VG). 1
PG and VG are classified as “generally recognized as safe” (GRAS) substances by the US Food and Drug Administration (FDA) when used as food additives. While this GRAS certification indicates that these ingredients are safe for oral ingestion in food products, it does not attest that they are safe for inhalation. 1 PG and VG are also commonly used in cosmetics and in a variety of topical, oral, and some intravenous pharmaceutical preparations.
Inhaled PG and VG are considered to be generally nontoxic. 11 The few existing studies to date on this topic have found no adverse effects on the lungs, kidney, or reproductive system in animals or humans. 17 –21 EVP users inhale significant amounts of PG and VG over a long-term period. Such lung exposures are very different from those employed for therapeutic purposes and should not be a priori regarded as harmless, and the safety of PG and VG intake through the inhalation route, in amounts relevant to EVP consumption, should be assessed. This is especially relevant because studies have shown that PG and VG may be thermally degraded during aerosolization within the EVP, resulting in the formation of potentially toxic byproducts such as formaldehyde, acetaldehyde, and acrolein. 10,22 –24 Two recent 90-day inhalation studies in rats have shown that combined PG/VG exposure relevant for EVP users does not result in any toxicological concerns. 18,25
Nicotine
Not all e-liquids contain nicotine. The nicotine concentrations in commercially available e-liquids usually range from 0 to 36 mg/mL, with reports of nicotine levels up to 50 mg/mL. 1,15
The nicotine yield per puff is determined only in part by the nicotine content of the e-liquid: Under machine-puffing conditions, the nicotine yields in an e-vapor generated with a given EVP device are proportional to the e-liquid nicotine concentration and will increase with increasing e-liquid nicotine concentrations. Yet, the type of device, delivered power, and individual puffing behavior have been shown to be more important determinants of nicotine yields per puff and can result in vastly different deliveries from e-liquids with similar nicotine concentrations. 1,15,26
As nicotine is derived from the tobacco plant, e-liquids may contain tobacco-related toxicants, often in trace amounts, such as tobacco-specific nitrosamines. Yet, when evaluated, these were found to be at much lower levels than in cigarettes. 27,28
Flavorings
The use of flavorings is an important element in the development of EVPs in order to increase product acceptance by adult smokers and enable them to switch away from cigarettes. It has been estimated that some 8000 flavored e-liquids are currently available in the market and that more than 200 new flavors are added every month. 10,22,29 Their total proportion in e-liquids has been reported to be typically in the range of 1–4%. 22
Most flavoring substances used in e-liquids are classified as GRAS. The US Flavor and Extract Manufacturers’ Association (FEMA) issued a statement warning that the FEMA GRAS list applies to flavor chemicals for food only, and these flavors should not be considered as safe for use by inhalation without further testing. 30 A well-known example is chronic exposure to high levels of diacetyl, a butter flavoring processed in popcorn factories, which has been shown to lead to subclinical decline in lung function, airway obstruction, and, eventually, life-threatening, irreversible bronchiolitis obliterans, or “popcorn lungs.” 31 –33
The potential health risks of flavoring substances in EVPs are gaining significant attention because of the limited toxicological information on their inhalational use. Flavors are also used in cigarettes, and the weight of evidence indicates that flavors do not increase the toxicity of CS relative to flavor-free CS. This includes recent evidence developed to fulfill the EU Tobacco Products Directive (TPD) regulatory requirement for comprehensive studies on a priority list of ingredients. 34 –36 There is an urgent need for similar regulatory requirements for e-liquid manufacturers to generate toxicological data to support the use of flavors and other e-liquid ingredients at intended use levels. Emerging data from subchronic inhalation studies indicate that, relative to flavor-free e-vapor, the presence of flavors does not increase the biological effects of e-vapor on the respiratory tract or cardiovascular system in Apoe− /− mice 37 (Wong et al., in prep). Furthermore, a recent 90-day inhalation study in which rats were exposed to nebulized flavor mixtures showed no inhalational or systemic toxicity of the mixture. 25 Further investigation is required to confirm whether flavorings, when aerosolized at temperatures such as those used in commercial EVPs, are not converted into toxic substances. Moreover, the carcinogenicity of inhaled flavor mixtures should be investigated.
E-vapor
Physicochemical properties of e-vapor
E-vapor (aerosol) is generated by heating an e-liquid, which then forms a supersaturated vapor, which is subsequently cooled by an air flow to form an aerosol through a thermal nucleation process. E-vapor, like ENDP aerosols, is not generated by combustion, which makes these two aerosols fundamentally different from CS. Yet, e-vapor is qualitatively and quantitatively different from CS as well as ENDP aerosols, and its dynamic behavior is, in many aspects, not comparable to that of either of these aerosols. 38 In particular, e-vapor is much more dynamic and prone to evolution.
E-vapor particle number density is comparable to that of CS and has been reported to be in the range of 1–6 × 109 particles/cm3. 39 –41 Yet, e-vapor is not as chemically complex as CS, neither in its particulate nor gas–vapor phase (GVP). While CS contains more than 6000 constituents of multiple chemical classes, 42 including combustion-generated carbon nanoparticles, e-vapor contains mainly PG, VG, water, preservatives, and, if added to the e-liquid, nicotine and flavoring substances. The potential presence of thermal decomposition byproducts is highly dependent on the type of product and use parameters. 27,43
Because e-vapor constituents are volatile or semi-volatile, they partition between liquid and gas phases. E-vapor is highly affected by dilution, as the volatiles may partially or totally evaporate, which affects phase partitioning and particle size distribution (PSD).
Particle size distribution is a key physical characteristic of an aerosol, as it determines the deposition pattern of its particles along the respiratory tract. 44 Various particle size distributions have been reported, with sub-micrometer to micrometer median particle diameters. 40,41,45 –47 These PSDs suggest deposition patterns in the respiratory tract comparable to those of CS and ENDP aerosols, provided they don’t undergo critical changes while passing through the airways into the lungs. The PSD of e-vapor has been shown to be influenced by the type of generating device, delivered power, puffing behavior, and composition of the e-liquid. 14,47,48 When studying the effects of e-vapor in a laboratory context, it is notably the PSD, together with other aerosol properties, that may be affected by the experimental setup and parameters (such as dilution and sampling location) used for measuring it and that may lead to the discordant outcomes observed in the literature.
Byproducts of e-vapor generation
The release of potentially toxic substances by EVPs has been reported in the scientific literature. These substances may originate from poor-quality products and product failures, such as e-liquid contamination or uncontrolled device manufacturing. Potentially toxic substances may also result from overheating of the coil in the wick-and-coil system. As most EVPs are power-controlled and not temperature-controlled, the temperature of the heating coil may vary significantly depending on the available e-liquid amount for evaporation. At a low e-liquid level, the wick is insufficiently wetted, and the energy delivered to the coil is more than that required to evaporate the liquid, leading to the wick-and-coil system overheating. Several studies have shown that puffing on an EVP when the e-liquid level is low can drastically change the chemical composition of the e-vapor and generate potentially toxic thermal degradation byproducts of the vaporized ingredients (often described as the “dry puff” phenomenon). 49
Such potentially toxic byproducts can also be generated as a result of user-driven changes in delivered power to the device or individual puffing patterns that impact complex heat and mass transfer processes required for aerosol generation. For example, carbonyl generation has been reported experimentally as a result of the puffing regimen, device setup, 50,51 voltage supply, or heating coil temperature. 52 –55 At high temperatures and with frequent puffing, PG and VG have been suggested to undergo thermal degradation to form acetaldehyde, formaldehyde, acrolein, and other potentially toxic carbonyls. 43,54,56 The presence of byproducts of other classes has also been reported, such as glycidol as a byproduct of VG. There is a critical need to develop testing standards that set forth which puffs, or block of puffs, are relevant for testing, as these will have an important influence on chemical e-vapor composition. In parallel, an internationally agreed list of e-vapor constituents for product characterization and toxicity assessment should be established. Notably, findings from laboratory studies need to be carefully interpreted in light of evidence showing that EVP users will rarely operate their devices under extreme conditions because of the resulting adverse taste of the aerosol. 53
Challenges and principles of EVP testing
Toxicity assessment of EVPs faces many challenges. At the outset, there is no regulatory and scientific consensus on the appropriate testing framework or study endpoints for evaluating e-liquid and e-vapor toxicity across brands and in comparison to other tobacco products or nicotine replacement products. 5 Furthermore, EVP development and scientific research progress at a different pace. While EVP devices, their technology, and e-liquids evolve rapidly, preclinical and clinical toxicity studies take time. Currently adequate toxicity studies may rapidly become obsolete or, at best, limited. The lack of manufacturing standards and the resulting lack of consistency of finished products and goods also add uncertainty to the relevance of research findings.
In its guidance for the industry on the premarket tobacco product application (PMTA) for electronic nicotine delivery systems (ENDS), the US FDA outlines the type of information that can be provided to support the view that permitting the marketing of a new EVP would be appropriate for the protection of public health. 57 This includes, inter alia, nonclinical and clinical studies, the relative risk of the new tobacco product for both users and nonusers compared with other tobacco products on the market, and the chemical and physical identity and quantitative levels of the emission of aerosols under the range of operating conditions and use patterns. Scientific reviews of flavors should also be included, covering, for example, toxicological analyses of flavoring substances, chemistry analyses, clinical studies, and literature reviews.
A number of risk assessment frameworks have been proposed in the scientific literature. Some proposals focus on the assessment of e-liquids. For example, Costigan and Meredith proposed the selection of e-liquid constituents with certain purity criteria and exclusion of ingredients known to be carcinogenic, mutagenic, and reprotoxic. 58 The authors also suggested the exclusion of known respiratory sensitizers. 59 More recently, the use of the Genomic Allergen Rapid Detection assay was proposed to determine the sensitizing potential of e-liquid ingredients. 60
Other frameworks propose screening e-liquids before moving on to assessment of their aerosols. For example, Iskandar et al. (see Figure 2) proposed a multilayer systems toxicology framework for systematic in vitro assessment of e-liquids to complement standard toxicology approaches for assessing the cytotoxic and genotoxic potential of e-liquids and trapped e-vapors. 9 Briefly, the first layer of the framework is aimed at screening e-liquids for potential toxicity by using relevant two-dimensional (2D) primary human cell culture systems. The composition of the test e-liquids should be carefully designed to allow generation of broadly relevant toxicological data. This includes using ranges of PG/VG and nicotine concentrations as well as representative flavors and use levels that can be selected. E-liquids associated with a considerable reduction in cell viability are further assessed in the second layer by toxicity-related mechanistic investigations in culture systems like those used in the first layer. Finally, the third layer of the framework focuses on toxicity-related mechanistic investigation of the corresponding aerosols by using three-dimensional (3D) airway culture systems grown at the air–liquid interface (ALI), which is relevant to the inhalation route.
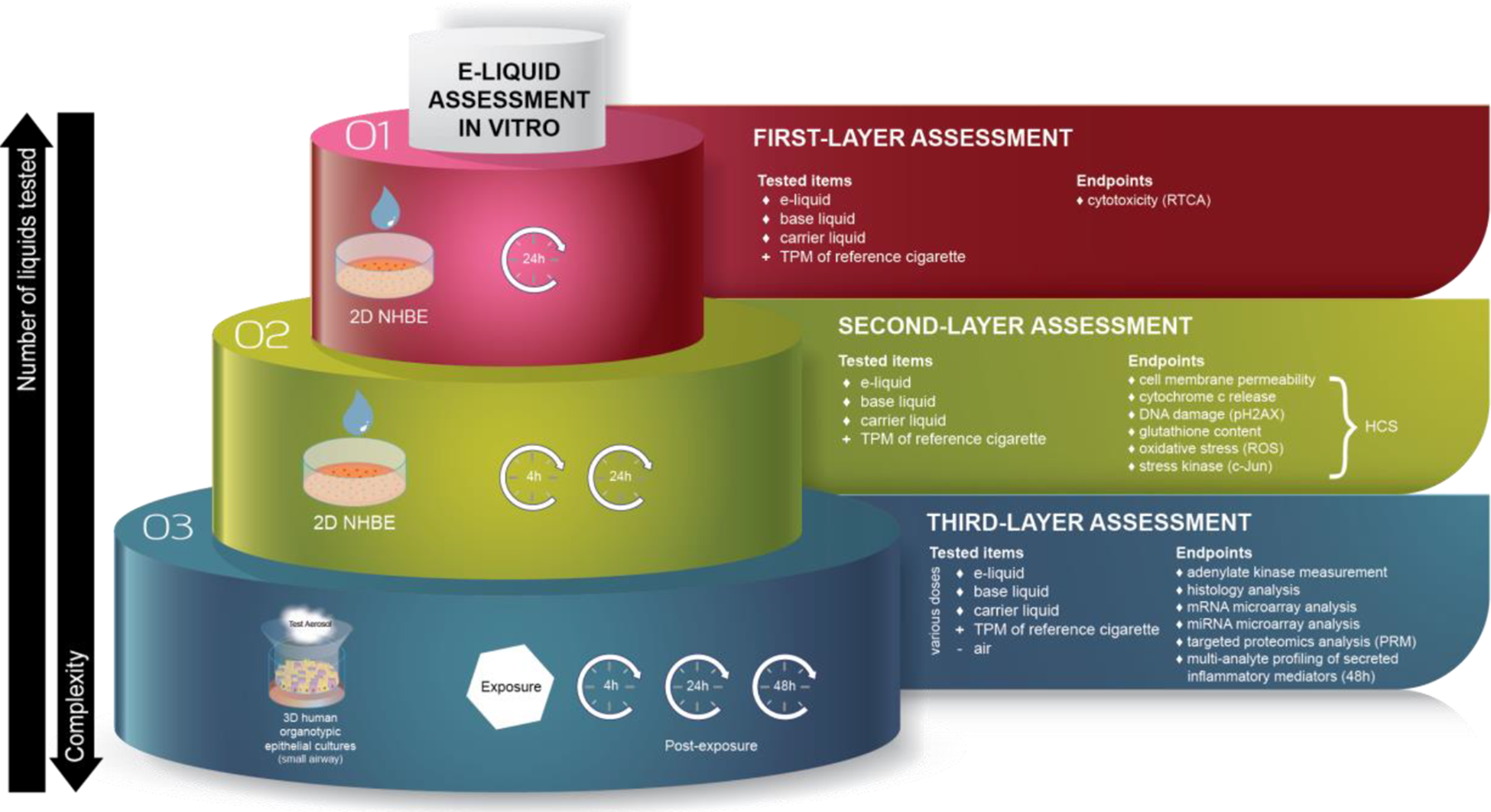
Three-layer assessment framework of e-liquids and their aerosols. Source: adapted from Iskandar et al. 9
In line with the effort to “replace, reduce, and refine” the use of animals in experiments (the “3R” principles) and with the vision and strategy for toxicity testing in the 21st century, 61 there is an emphasis in e-liquid and e-vapor assessment on high-throughput in vitro methods using relevant test systems. Yet, in its 2018 report, the NAS pointed out the lack of long-term data from repeated inhalation exposures and recommended long-term (2-year) animal studies involving inhalational exposure to EVP aerosol to better understand the disease risks from inhaling e-vapor. 1
Testing e-liquids vs. testing e-vapors
Current proposed EVP assessment frameworks focus diversely on testing e-liquids, testing e-vapors, or a combination of both.
In reality, the human respiratory system is exposed to e-vapors and not e-liquids. Yet, e-liquid testing is interesting, as it constitutes a relatively cheap, faster, and less labor-intensive screening opportunity. 62 This is particularly relevant for initial toxicity assessment of flavoring substances, given their huge numbers.
The extent to which e-liquid testing may provide relevant insights into the toxicity of e-vapors is being investigated, although research in this area has yet to be systematized. Chemical reactions may result in the formation of new substances during heating and aerosolization. Several studies have shown that the chemical composition of an e-vapor is similar to, but not the same as, the composition of the e-liquid used to generate it. For example, Herrington and Myers demonstrated distinct analyte profiles for e-liquids and their aerosols. 63 Notably, formaldehyde, acetaldehyde, acrolein, and siloxanes were found in the aerosols but not in the e-liquids. 63 Also, volatile substances from e-liquids, such as flavorings, are unlikely to be present in the e-vapor in the same amounts and proportions. 64
Several studies have compared the results of in vitro e-liquid and e-vapor testing and found both similarities and differences. Behar et al. screened 36 e-liquids in liquid and aerosol forms for cytotoxicity by the MTT (3-(4,5-dimethylthiazol2-yl)−2,5-diphenyltetrazolium bromide) assay in human pulmonary fibroblasts, lung epithelial cells (A549), and human embryonic stem cells (H9) and showed that the cytotoxicity of an e-liquid and its aerosol were correlated about 74% of the time. 62 Sassano et al. found that aerosolization did not change the relative toxicity of e-liquids when assessed in HEK293T, human bronchial epithelial cells (HBEC), and human airway smooth muscle (HASM) cells, except in the case of one flavor, which, the authors suggested, indicated that this phenomenon was unique to this particular flavor. 65 Rowell et al. found that the liquid and vapor forms of commercial e-liquids induced similar dose-dependent decreases in CALU3 cell viability and proliferation. 66 Iskandar et al. found that information obtained by using e-liquids in submersed 2D cultures provided relevant indications on the potential toxicity of the corresponding aerosols in 3D cultures at the ALI. 67
These findings warrant further investigation of the extent to which e-liquid toxicity data can be used to predict e-vapor toxicity. Product standard requirements will improve product quality and decrease the risk of contamination by, or generation of, unwanted byproducts, therefore increasing the predictability of e-liquid data. Yet, even if the data can be bridged, any complete risk assessment of EVPs should ultimately include both e-liquid toxicity data and evaluation of heated and aerosolized e-liquids by using their corresponding EVP devices.
Testing aerosols generated by EVP devices vs. aerosols generated by aerosol generators
Different approaches may be used to generate e-vapors for characterization and toxicity assessment. Each method has its specific pros and cons and is better suited for a specific purpose—for example, puff-by-puff analysis or continuous, large-scale generation of e-vapor for inhalation studies.
While it may be argued that aerosol generation from an EVP device with a relevant, standardized puffing regimen is more representative of human consumption, it has several limitations. Given the wide variety of EVP types and devices, any particular device may not be representative of other devices. 68 Furthermore, EVP devices may increase the toxicity of the aerosol, for example, by the release of metal ions. 1,7,12,69,70 The battery can run out of power, its level of power delivery may vary from one puff to the next, or it may provide a false reading of the applied power. Operating the devices, including charging, refilling, and cleaning, is labor intensive and a limiting factor when it comes to long-term inhalation studies. According to Werley et al., approximately 27,000 EVPs are required for a 90-day rodent inhalation study. 21 Hence, for large-scale production of e-vapor, the use of laboratory nebulizers or aerosol generators simplifies the logistics and removes parameters that could potentially affect reproducibility. An approach that uses nebulizers does not, however, help study the effects of substances that may be generated through thermal treatment of the e-liquid; in contrast, larger scale aerosol generators may be more suitable, as long as the temperature used for aerosolization is representative of that in EVPs.
The physicochemical characteristics of aerosols generated by these different approaches will vary, and these differences may result in different toxicity profiles. Therefore, physicochemical characterization of the aerosol should always be part of product assessment.
Standard EVPs and standard e-liquids for research
In tobacco research, the reference cigarettes produced by the University of Kentucky Center for Tobacco Reference Products since 1968 have provided much needed reference products for tobacco manufacturers, government agencies, and research institutions. Similar reference products are not yet available in the EVP field.
Current EVP toxicity studies generally use cigarettes as a reference. Although the use of these products is well justified for comparative analyses, availability of reference EVP and e-liquid products would allow benchmarking of research data against a relevant, consistent, and reproducible reference. 71
The large heterogeneity in EVP designs, performance, and consumption parameters as well as the huge variety of e-liquid formulations have hampered the definition and development of a reference standard for EVPs and e-liquids. 72
In 2014, the US National Institute of Drug Abuse (NIDA) launched a competitive innovation research program to develop a Standardized Research E-Cigarette (SREC). The US-based EVP company NJOY was awarded the contract and has developed an SREC to facilitate clinical EVP research in line with FDA requirements. The NJOY SREC will remain available for an extended period of time and can be used as a bridging element in various studies aimed at evaluating EVPs as reduced-risk products.
The French Standardization Association’s (AFNOR, Part 3) Experimental Standard for EVPs and e-liquids proposes the use of e-liquids with standard compositions for evaluating the impact of EVP devices on aerosol emissions. 73 Two reference flavored e-liquids are proposed, with two PG and VG concentration variants and one flavoring preparation, in order to match the diversity of the e-liquids normally consumed in Europe. The higher VG test e-liquid (48% VG and 38% PG wt/wt) is intended for assessment of potentially toxic substances defined by the standard, namely: diacetyl, formaldehyde, acetaldehyde, acrolein, and metals and inorganic substances (lead, antimony, arsenic, nickel, chromium, and cadmium). The higher PG test e-liquid (63% PG and 24% VG wt/wt) is intended for determination of nicotine yields and stability.
Puffing regimens for EVPs
Human topography studies indicate that EVP usage depends greatly on product design and on the individual consumer. Overall, EVP users have been observed to have a longer puff duration than cigarette smokers. EVP consumers have also been reported to use a wide range of puffing parameters, which may significantly influence e-vapor yields and toxicity profiles. 74 It is questionable whether a single puffing regimen is appropriate for all EVP types.
So far, a variety of puffing regimen parameters have been used in research, which makes it difficult to evaluate and compare study findings. Standardization is urgently required.
In a 2016 guidance to industry, the US FDA recommends using both non-intense and intense regimens for evaluating EVP emissions and testing e-liquids with low and high emission devices, but it does not specify the actual parameters. 57
International Organization for Standardization (ISO) Standard 20768:2018 defines the technical requirements, parameters, and standard conditions for routine analytical vaping of vapor products, including EVPs. 75 The standard was developed on the basis of the Cooperation Centre for Scientific Research Relative to Tobacco’s (CORESTA) recommended method 81 (CORESTA Recommended Method 81; CRM 81) for machine puffing of EVPs, 76 which aimed at reflecting the puff volume and duration of typical cig-a-like EVP users. 77 ISO 20768:2018 and CRM 81 specify a puff volume of 55 mL, a 3-s puff duration, and one puff every 30 s (55/30/3). A square-shaped profile is recommended, given that a minimum airflow velocity is required for device activation in puff-activated devices. The devices are activated by airflow draw or button.
ISO 20768/CRM 81 may be regarded as reflecting non-intense use. CORESTA has published a guidance on which criteria could be considered for intense use (CORESTA Guide N° 22 78 ). If topography data for the device under study are available, it is recommended to use these as a basis for determining intense usage scenarios. In other cases, variations in the following, interdependent parameters should be considered: puff duration, volume, frequency, profile, and number; battery charge; heating element age; voltage and ventilation settings; and device orientation. The guidance stresses that puffing regimens unrepresentative of typical consumer behavior should be avoided, as they may result in the generation of byproducts which would not occur in real life, as users would experience a self-limiting unpleasant taste. 49
E-vapor generation
E-liquid nebulization
Nebulization (atomization) is often used to convert a liquid into an aerosol. Nebulization is widely used in drug delivery devices to administer active pharmaceutical ingredients into the lungs. 79 Nebulization does not involve heating and, therefore, differs from the aerosol generation process of an EVP device. An aerosol produced by nebulization contains unaltered main e-liquid constituents, and toxicity studies using nebulization will, therefore, provide information on these constituents, but not on substances potentially generated by the thermal treatment of e-liquids.
The Collison nebulizer
The Collison nebulizer was first described in the scientific literature by Collison in 1935. 80 Since then, it has become a recognized technique for nebulizing liquids and suspensions and is commonly used in aerosol generation research.
The Collison nebulizer was shown to be appropriate for delivering nicotine to rodents via inhalation. 81 Thereafter, it has been used in 28- and 90-day rat inhalation studies of nicotine-containing solutions and was shown to be able to continuously generate stable and reproducible aerosols over extended periods. 18,82
In a Collison nebulizer, high-velocity air is used to aspirate a liquid via negative pressure into a siphon and shear it through one or several orifices into small liquid particles, a process known as atomization (Figure 3). Commercial Collison nebulizers (BGI, Butler, NJ, USA; now CH Technologies, USA) exist with 1, 3, 6, or 24 orifices to produce the high-speed jet stream. The mass output of the nebulizer is directly proportional to the number of jets present in the nebulizer.
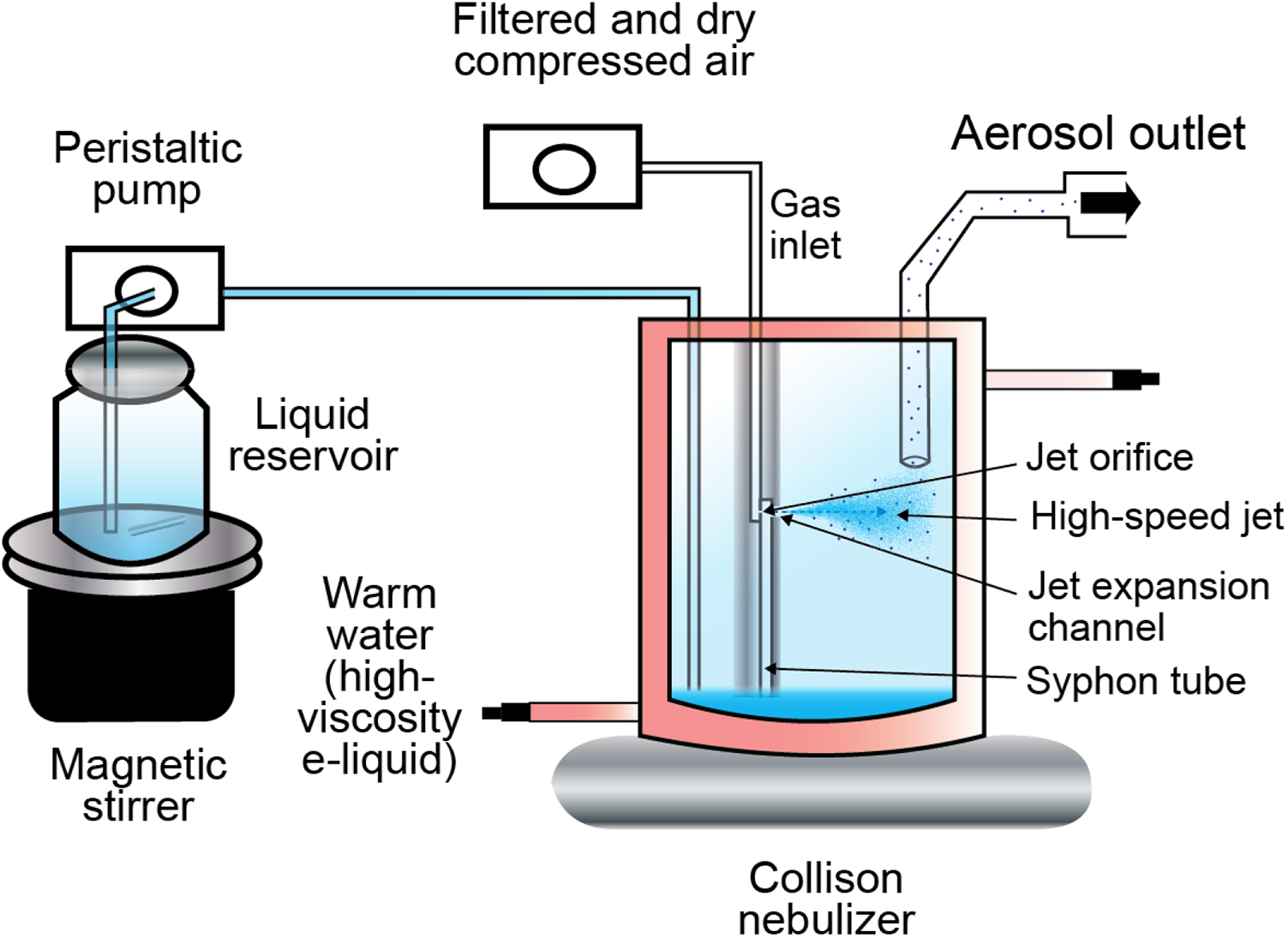
Schematic view of the experimental setup of a Collison nebulizer.
The liquid particles produced by this process often have a wide size distribution. To remove larger particles, the jet stream is directed toward the wall of the nebulizer chamber, where large particles with sufficient mass are deposited and drained back to the nebulizer reservoir. Smaller liquid particles, with size ranges suitable to reach the lungs, leave the reservoir through the aerosol outlet.
A peristaltic pump is used to provide the nebulizer reservoir with a constant supply of e-liquid from an external reservoir to maintain its homogeneity. E-liquid solutions are stored away from light, at a controlled temperature of 2–8°C, but under uncontrolled humidity conditions. They are equilibrated at room temperature prior to nebulization.
E-liquids typically have high viscosity owing to the presence of PG and VG, and their constituents have different densities. For these reasons, the solution in the external reservoir must be continuously stirred to avoid liquid stratification and maintain a homogeneous feed to the nebulizer.
For high-concentration or high-viscosity solutions, the nebulizer is warmed to 30°C with a water heater in order to decrease viscosity and generate aerosols of appropriate concentrations and particle sizes. The nebulizer should also be warmed when the aerosol is used for cell culture exposure, at a temperature matching the exposure chamber temperature (usually 36.6–37°C). This helps minimize the changes in aerosol properties during transportation and delivery.
Vaping machines
A number of smoking machines have been developed for the study of CS, from the first simple setups in the 1840s to the sophisticated and fully automated smoking machines available today. 83
The rapid development of ENDP products has posed new challenges and necessitated the development of new or modified experimental setups and methods for ENDP aerosol generation. 84 Likewise, new and adapted machines need to be developed to generate e-vapors from EVP devices. 85 These need to fulfill many specific requirements.
Given the immense variety of EVPs, vaping machines should allow the use of a variety of designs by means of custom-designed adaptors. They should also allow the possibility of puff- and switch-activation of the device and of orienting the product at various degrees, which is necessary for many devices that require an orientation other than horizontal to facilitate efficient wicking of the e-liquid. EVP batteries can be unreliable and discharge. Therefore, the experimental setup should provide external power supply to the EVP and control the energy supplied to the coil. 86 A vaping machine should be enclosed and supplied with air under conditioned humidity and temperature (e.g., 60% relative humidity at 22°C) to ensure that consistent results can be obtained regardless of environmental conditions. Finally, it should offer the possibility of executing standard but also ad hoc vaping regimens to replicate real-world, human puff profiles.
Programmable single syringe pumps
The simplest laboratory vaping machine is a programmable single syringe pump, a linear piston pump with a glass cylinder. The pump is moved by a stepper motor and generates an aerosol by drawing on an EVP device at the valve inlet and delivers the aerosol at the valve outlet. The outlet can be connected to a trapping system or an exposure system.
Figure 4 shows the experimental setup for continuous generation of e-vapor from two EVP devices by using a programmable single syringe pump. 87
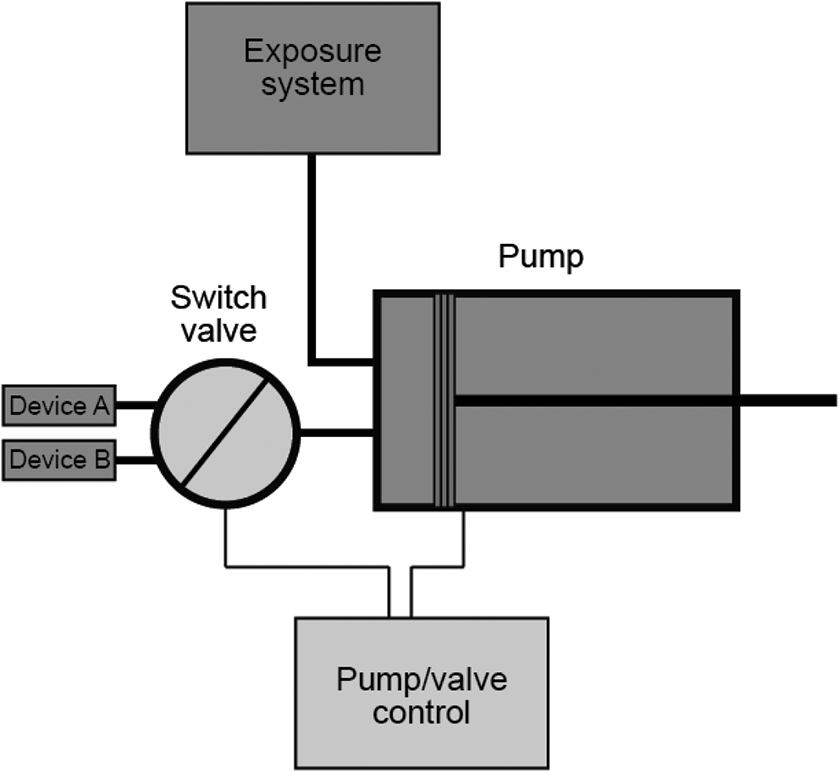
Experimental setup for generation of an aerosol from a single type of EVP. “Device A” and “Device B” refer to two devices of the same kind. Both devices are connected to a switch valve, allowing for alternation of aerosol generation from the two devices.
Two devices of the same kind are connected to one programmable single syringe pump. A pinch valve, synchronized with the pump, is installed between the two devices and the pump to allow alternated activation of the two devices. When used to generate aerosols in accordance with the ISO 20768/CRM 81 puffing regimen, this setup can generate one 3-s puff every 15 s and a 30-s inter-puff interval for each cartridge, for a total of 4 puffs/min. The maximum puff count that can be generated from a given device depends on the type of the device.
Vaping machines
An increasing number of multichannel laboratory vaping machines are commercially available. The EVP vaping machine series CETI (Cerulean, UK) is one example of a multichannel linear vaping machine for aerosol trapping and characterization (Figure 5). The machine can be used with puff- and switch-activated EVP devices loaded and removed manually. The puff profile (square or sinusoidal), volume, duration, interval, total number of puffs, and number of conditioning puffs per device can be user-defined, including for use in accordance with ISO 20768/CRM 81. The machine also offers the possibility of adjusting the angle at which the device is held and vaped, at 15° intervals between +90° and −90°. Each channel can be combined with a Cambridge filter pad (CFP) holder and/or an impinger for e-vapor fraction collection.

Eight-channel EVP vaping instrument CETI8 (Cerulean).
Capillary aerosol generator (CAG)
The capillary aerosol generator (CAG) generates an aerosol from e-liquids by processes that mimic the physical and chemical processes of aerosol generation by an EVP device. The key advantage of the CAG is that it can be used for continuous production, over several hours, of a controlled aerosol similar to e-vapor. Therefore, it is ideal for in vivo inhalation studies.
The CAG was developed by Philip Morris, Inc. and further refined by Virginia Commonwealth University. 88,89 The CAG produces a stream of well-controlled aerosol by heating and vaporization of a liquid, followed by nucleation and condensation of the vapor. The physical process of aerosol generation from liquid mixtures in the CAG is complex and follows various stages with varying thermodynamic conditions. The thermodynamic, physical, and chemical properties and conditions of the mixture, such as surface tension, temperature, saturation, equilibrium vapor pressure, and gas phase concentrations of the constituents are important to obtain controlled and continuous aerosol delivery at the required particle number density and PSD.
Figure 6 shows the experimental setup and key elements of a CAG.

Capillary aerosol generator (CAG). A. Experimental setup. B. CAG capillary and heating elements. C. Cross-sectional schematic view of the CAG.
The CAG described here is a bench-top laboratory device consisting of a stainless steel heated capillary connected to a temperature controller and to a liquid reservoir via a peristaltic pump.
E-liquid solutions are prepared and stored as for the use with a Collison nebulizer. They are stirred throughout the aerosol generation process to maintain homogeneity. The liquid is supplied via a pump to the capillary at a controlled flow rate.
The capillary (160 mm; 21 G; stainless-steel) is housed in an aluminum block, where it is heated by heating elements embedded in the aluminum block. The temperature is set to mimic the temperature of the heating coil during puffing of the EVP, typically 250–275°C. 90
The liquid pumped through the capillary is heated, evaporated, and exits the tip of the capillary as a supersaturated, hot vapor. The vapor is immediately cooled down by mixing with a cooler, filtered air stream around the capillary outlet. This leads to homogeneous nucleation of vapors and condensational growth of the generated nuclei to form an aerosol. The flow rate of the filtered air supplied at this point has a strong influence on the PSD. 88
An additional, cooling air flow circulates along the capillary housing and then along the capillary heating blocks. This air flow prevents vapor from flowing back and coming into contact with the hot surface, thereby avoiding the formation of unwanted degradation products such as carbonyls (Figure 6C).
When used for in vivo inhalation studies, the CAG-generated aerosol is diluted to the target concentration with conditioned, filtered air. The aerosol is delivered via glass tubing to the exposure chamber. 37
The CAG has been shown to produce an aerosol similar to that generated by EVP devices. Werley et al. compared an aerosol generated by a prototype EVP and a CAG in terms of chemical composition, chemical byproducts, particle size measurements, and port-to-port variability in a nose-only exposure chamber. 91 They concluded that the use of the CAG was well justified on the basis of the similarity of the aerosol with that generated by the EVP device and given the improved logistics, consistency, and study throughput of the CAG. 91 When using the CAG to generate an aerosol intended to be representative of the aerosol generated by a specific EVP device, it is important to measure specific chemicals in order to demonstrate similarity and the validity of the approach.
The CAG has been validated prior to its implementation in in vitro and in vivo studies. The impact of various parameters has been assessed, including liquid mixture supply ranging from 0.2 to 1.0 mL/min, capillary temperature ranging from 200 to 300°C, and air streams of 10 to 100 L/min. A capillary temperature of 250°C and an air stream of 10 L/min resulted in a mass median aerodynamic diameter (MMAD) of 1.5 µm and a geometric standard deviation (GSD) of 2 (PMI, data not published). For in vivo inhalation studies, a capillary temperature of 250°C and an air stream of 18 L/min, followed by dilution with 260 L/min of filtered air, led to an MMAD of approx. 1 µm and a GSD of 1.4 when measured in the exposure chamber by using a PIXE cascade impactor.
Characterization of e-liquids: Stability and osmolarity
Any study of e-liquids should include chemical characterization to identify which, if any, ingredient increases the toxicity of the mixture. Apart from added ingredients, e-liquids should be screened for potential contaminants and impurities.
Any experiment should also carefully take into account two factors that may potentially affect results and lead to incorrect interpretations: changes in e-liquid composition over time and e-liquid osmolarity, the latter being particularly important when conducting in vitro studies. The following sections describe approaches for assessing and controlling these elements.
Stability study
Some e-liquids have been shown to be chemically unstable, with reactions occurring between their ingredients (such as vehicles and flavors) to form new substances. The resulting new substances may have toxicological properties that differ from those of the initial ingredients. 92
Stability studies are necessary for assessing the rate and magnitude of change of e-liquid constituents in different environments. Once product stability and the associated conditions that limit product stability have been determined, a product shelf life can be assigned for commercial e-liquids. In research, stability studies are key to supporting the adequate design of studies, particularly long-term studies (such as inhalation studies), which require large volumes of e-liquids. In order to address the limited stability of some e-liquids, preblends of increased stability may be prepared and stored and mixed into e-liquid test solutions just before use. Stability studies are used to determine the composition of these preblends and the frequency at which preblends and e-liquid test solutions will need to be prepared during the study. They are also used to determine the level of degradation that should be taken into account for calculation of exposure levels.
In its “Technical Guide for Designing E-Vapour Product Stability Studies,” CORESTA provides guidance for designing formal stability studies as may be required, for example, by regulators. 93 The Committee recommends that stability studies should be performed on each individual e-liquid, unless a bracketing approach is applied, for example, as outlined by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use in its guidance on bracketing and matrixing designs for stability testing of drug. 94 Bracketing is the design of a stability study in which only samples on the extremes of certain design factors are tested at every time point. The design assumes that the stability of any intermediate level is represented by the stability of the extremes tested.
A stability study was performed to assess the stability of 38 flavoring substances routinely used in commercial e-liquids in different environments (PMI, data not published): (i) neat flavoring substance; (ii) 6 high-concentration (up to 20 times the final concentration in the e-liquid) preblends of 2 to 9 flavoring substances each; and (iii) e-liquid test solutions prepared by combining multiple preblends. For preparation of flavor stocks, flavoring substances were initially dissolved in PG and, when needed, ethanol to improve the solubility of some compounds. Flavor stocks were then mixed and added at desired concentrations to a base solution composed of PG, VG, water, and, when present, nicotine at 2.0
lavor solutions containing nicotine. Hence, it was recommended that all test e-liquids be made fresh at least every 3 days and stored at refrigerated conditions (4°C) to ensure the stability of flavoring substances.
Osmolarity testing
The osmolarity of e-liquids, which often exceeds the normal physiological range (290–310 mOsm/L), is another challenge for assessment of e-liquids in in vitro cell-based assays. Studies have highlighted that osmolarity can be a confounding factor in the toxicity assessment of e-liquids. Gonzalez-Suarez et al. measured the osmolarity of solutions containing different concentrations of PG, VG, and nicotine. A concentration-dependent increase in osmolarity was observed with all PG/VG mixtures. The osmolarity values were higher with mixtures containing PG than with mixtures containing only VG. A solution of 2% PG in cell culture media was shown to have an osmolarity of around 600 mOsm, indicating the potential to induce hyperosmotic shock in normal human bronchial epithelial cells. Nicotine alone did not have an effect on osmolarity, and nicotine at 1.8% did not increase the osmotic concentration of PG/VG mixtures. 95 Iskandar et al. measured cell viability by real-time cellular analysis (RTCA) in normal human bronchial epithelial cells incubated for 24 h with 3R4F total particulate matter (TPM), a commercial, flavored e-liquid (MESH Classic Tobacco; Philip Morris International, Switzerland), and its base liquid (39% PG, 39% VG, and 1.8% nicotine, wt/wt). The study found that the cytotoxicity of e-liquids was directly correlated to their osmolarity, with no significant difference between the commercial e-liquid and its base liquid. 67 Therefore, the osmolarity of e-liquids should be systematically measured.
Osmolarity is measured with an osmometer, a device that measures the osmotic strength of a solution. Different models exist that rely on different techniques, including vapor pressure osmometers, membrane osmometers, and freezing point depression osmometers, which are the most commonly used. A freezing point depression osmometer determines the osmolality by measuring the freezing point of aqueous solutions. The freezing point depression in comparison to pure water is used as a direct measure of the osmotic concentration. 95
The instrument (e.g., a Löser micro osmometer type OM806; Löser Messtechnik, Berlin, Germany) is calibrated before use with distilled water (0 mOsm) and with 300 and 900 mOsm calibration solutions (Vogel Gmbh & CO., Fernwald-Annerod, Germany). Sample tubes are filled with 100 µL of test solution and inserted into the measuring head of the instrument. The osmolality value of a sample is determined with a precision of ±1 mOsm.
Characterization of e-vapors
Chemical characterization of e-vapors
There is an increasing body of scientific literature on the chemical characterization of e-vapors. Studies assess the presence of harmful and potentially harmful constituents (HPHC) commonly present in CS as well as potential toxicants specific to e-vapors.
A recent systematic review of the scientific literature on carbonyl emissions in e-vapors found that a large diversity of methodologies are used, including different puffing regimens, aerosol sampling and analytical methods, as well as reported units of measurement. 96 This diversity, which is more generally observed in e-vapor research, hampers comparisons and makes it difficult to determine the accuracy of the findings.
The ISO Technical Committee on Vape and vapor products (ISO/TC 126/SC 3) is developing analytical methods for measurement of e-vapor product emissions, including nicotine, carbonyls, and metals. Meanwhile, the methods used to analyze e-vapor constituents are mostly adapted from the methods developed for CS. Yet, their applicability to e-vapor, which is qualitatively and quantitatively different from CS, has yet to be determined through appropriate method validation and interlaboratory collaborative trials. Notably, these methods are often not sensitive enough to detect the relatively low levels of constituents in e-vapors, many of which are several orders of magnitude lower than in CS.
Given the large and expanding variety of EVP products, methods should allow rapid aerosol screening as an alternative to the classical, time-consuming aerosol sampling and analysis methods. Furthermore, it is critical to assess the emissions on a puff-by-puff basis over the full lifecycle of EVPs. 97 Indeed, the types and levels of emissions may change drastically depending on the quantity of e-liquid remaining. A key limitation of many studies is that they focus only on the first 20, 50, or 100 puffs, whereas HPHC levels may increase dramatically later, with decreasing levels of e-liquid being parallel to increasing coil temperature. This adds increased complexity to the in vitro testing, where the results may vary greatly depending on which puff block was collected for testing.
Furthermore, an internationally agreed list of e-vapor constituents that would be relevant for product characterization and comparison with CS has yet to be developed. In the US, the FDA recommends reporting the levels of 33 constituents in e-liquids and e-vapors to provide the Agency with relevant data to assess whether the marketing of an EVP would be appropriate for the protection of public health. 98 The list includes constituents that, to the FDA’s current thinking, could potentially cause health hazards depending on their level, absorption, or interaction with other constituents. The FDA has also proposed the addition of 19 toxicants to the existing list of HPHCs to cover ENDS such as e-liquids and EVP aerosols. In Europe, the 2018 Technical Report of the European Committee for Standardization (CEN) “Electronic cigarettes and e-liquids—Constituents to be measured in the aerosol of vaping products” (CEN/TR 17236:2018) provides a list of constituents proposed for the purpose of regulatory submission under Directive 2014/40/EU. 99 This list is not intended to be comprehensive, but represents the default minimum requirement in the view of the CEN.
The following sections describe the development of new or adapted methods for offline and online determination of the key e-vapor constituents: PG, VG, flavors, and carbonyls.
Offline methods
Determination of PG and VG in the liquid and gas–vapor phases
A collaborative study under CORESTA has led to the establishment of CRM 84 for determination of PG, VG, nicotine, and water in trapped e-vapor. 100 The method has been shown to be appropriate for quantification of analytes of interest in e-vapor. The repeatability and reproducibility of the method have been assessed in accordance with ISO 5725-2:1994. 101 CRM 84 is based on the determination of PG and VG in the aerosol collected mass (ACM) trapped on a CFP.
Because of the presence of PG and VG in both the liquid and GVP of a diluted aerosol, a modified trapping method has been developed that uses aerosol passage through a 44-mm CFP to collect ACM, followed by its bubbling through an impinger containing 16 mL of 2-propanol to collect the GVP. The volume of 2-propanol in the impinger can be altered to allow for mass transfer across the gas–liquid interface and/or to ensure that the results fall within the calibrated range of the GC.
After aerosol sampling (45 min at 0.4 L/min), the loaded CFP is transferred to a glass bottle. In parallel, the solution in the 2-propanol impinger is transferred into a 25-mL volumetric flask. Then, 1.875 mL of 4 mg/mL internal standard (ISTD) n-heptadecane in 2-propanol is added to the volumetric flask and topped up to 25 mL with 2-propanol.
The solution in the volumetric flask is transferred to the glass bottle containing the CFP for analyzing the PG and VG trapped both in the impinger and on the CFP. Typically, the concentrations of PG and VG trapped in the 2-propanol impinger are below the limits of quantification for this method. The glass bottle is then vortexed for 2 min, and the contents are mixed by using a rotary shaker for 30 min at 400 rpm and centrifuged for 5 min at 1200 rpm/290 rcf. The solution has been determined to be stable for 1 month when stored at 4–8°C.
Then, 1 mL of the solution is transferred into an amber glass vial and analyzed by using a GC system (Agilent Technologies) fitted with a flame ionization detector and DB-WAXetr GC column (30 m × 0.25 mm; 0.5-µm film thickness; J&W Scientific Inc., Folsom, CA, USA).
Quantification is based on a series of calibration standards containing N-heptadecane as the internal standard, along with USP testing specification-grade PG (≥99.5%; Sigma-Aldrich, St. Louis, MO, USA) at 0.16–1.6 mg/mL and VG (≥99%; Sigma-Aldrich) at 0.2–2 mg/mL. 18
Untargeted screening of volatiles and semi-volatiles in e-liquids, e-vapors, and e-vapor fractions
A method for untargeted, qualitative screening of flavors has been developed for screening of e-liquids, e-vapors, and e-vapor fractions (PMI, data not published). It is based on GC coupled with high-resolution mass spectrometry (GC–HR-MS) by using headspace and/or liquid injection modes.
Specifically, a GC system (Agilent 7890A instrument) equipped with a J&W DB-624 ultra-inert column (30-m × 0.25-mm internal diameter; 1.4-µm film thickness; Agilent Technologies, Basel, CH) is used. Detection is performed with a 7200A quadrupole with time-of-flight (TOF) accurate mass spectrometer system (Agilent Technologies, Santa Clara, CA) operating in full-scan mode, scanning mass-to-charge (m/z) values ranging from 22 to 500 in the positive-electron ionization mode.
The combination of exact mass measurement of eluted substances at specific retention times together with experimental versus predicted linear retention indexing values allows an increased confidence level for compound identification.
For screening aerosols, the ACM and GVP fractions are trapped by using a 44-mm CFP, followed by an impinger containing methanol cooled to −75 ± 5°C. The samples should be analyzed as soon as possible, preferably within a day after sample generation. If this is not possible, the samples should be frozen at −80°C and analyzed directly after thawing and homogenization by mixing.
Acquired data are first processed by qualitative analysis and the Qualitative MassHunter software (Agilent Technologies) for automatic peak finding, mass spectral deconvolution, and compound identification by matching both accurate-mass measurements and experimental retention time information against a personal compound database library containing data for more than 500 reference standards acquired under similar analytical conditions.
In the second step, the raw data files are processed by unknown analysis and the MassHunter Unknown Analysis software, and the peaks remaining unidentified after the first step are submitted for a National Institute of Standards and Technology library search.
Non-targeted differential screening is realized by using the Mass Profiler Professional and/or Progenesis QI software to highlight differences between two or more samples.
Proposed putative compound hits are confirmed by purchasing reference standards, if commercially available.
Determination of flavors trapped on EXtrelut® NT 3 cartridges
Knowledge about the actual transfer of flavors contained in e-liquids into the e-vapor is key to understanding their potential toxicity. However, the small quantities of individual flavors in the aerosol and their huge variety constitute a significant challenge for their identification.
The utility of EXtrelut® NT 3 cartridges (Merck, Zug, Switzerland) for trapping flavor in e-vapors has been assessed (PMI, data not published). EXtrelut® NT 3 cartridges use a chemically inert, wide-pore, highly pure diatomaceous earth-based solid phase and are commonly used to trap the nicotine present in the gas and particulate phases of nicotine-containing aerosols. 84
The trapping, retention, and extraction efficiency of EXtrelut® NT 3 cartridges has been assessed by using a selection of 38 flavoring substances commonly used in e-liquids (PMI, data not published). Figure 7 shows the experimental design used to assess the capacity of EXtrelut® NT 3 cartridges to trap, retain, and release these flavors. With an undiluted aerosol, the method showed higher efficiency than traditional trapping techniques using CFP and microimpingers (PMI, data not published).
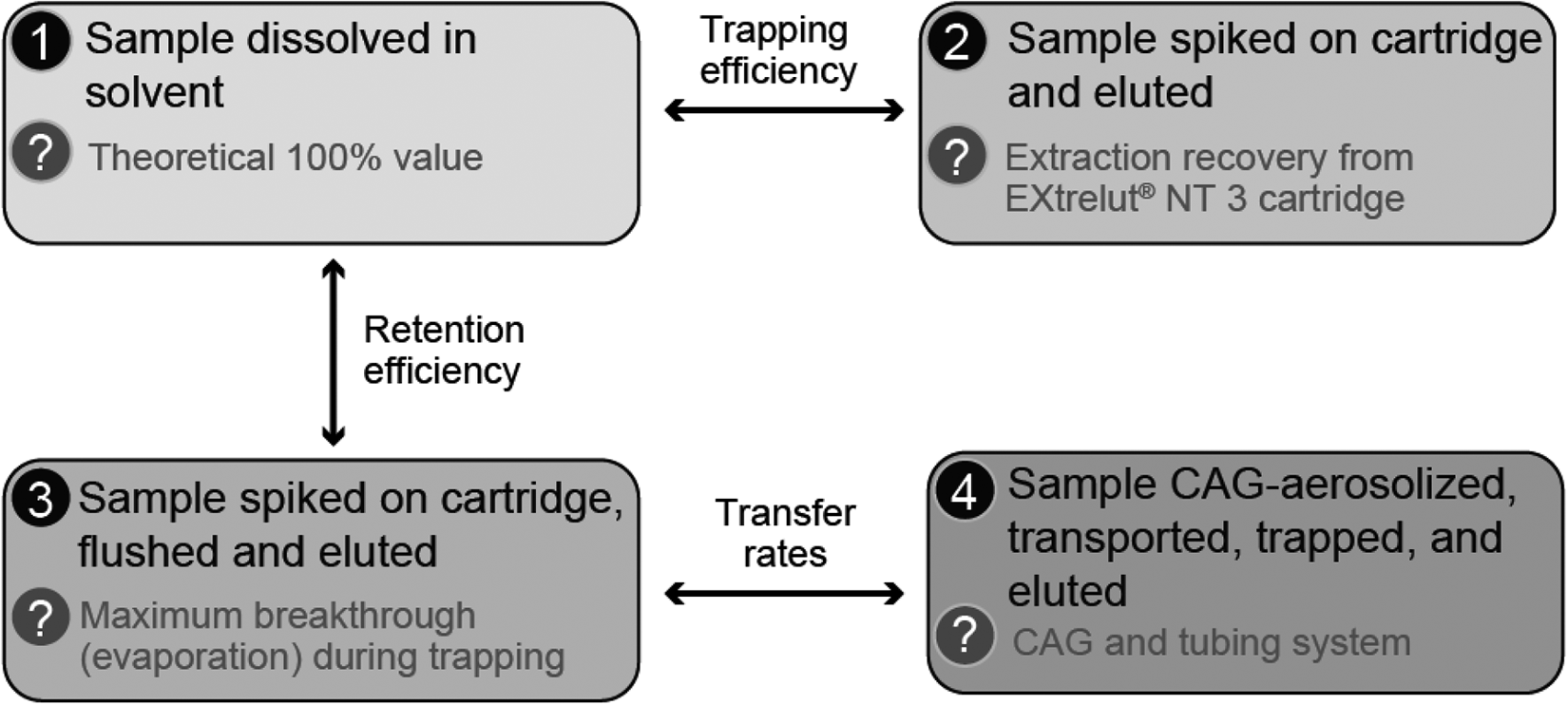
Experimental design for assessing the capacity of EXtrelut® NT 3 cartridges to trap, retain, and release 38 test flavors.
The method was tested with high-, medium-, and low-concentration flavor solutions. However, because of the limited number of replicates and data points within the calibration curve range for the medium- and low-concentration solutions, they are not described here.
The trapping efficiency of, and recovery from, EXtrelut® NT 3 cartridges for flavors was first assessed by determining and comparing the concentrations of the test flavors present in (i) the test flavor solution (theoretical 100% value) (Figure 7, trapping condition n° 1) and (ii) the eluate from an EXtrelut® NT 3 cartridge spiked with the test flavor solution (Figure 7, trapping condition n° 2). EXtrelut® NT 3 cartridges were preconditioned with 2 mL of 2-propanol and spiked with 50 µL of freshly prepared flavor solution dissolved in 1 mL of 2-propanol. Elution of the columns was performed shortly after trapping with 20 mL 2-propanol and internal standard. The flavors in the test flavor solution or in the EXtrelut® NT 3 cartridge eluate were determined by GC–HR-MS and quantified by using external calibration curves.
Three inter-day replicates were analyzed for each test flavor solution. For 84% of the flavors, the mean recovery was good (defined as recovery >90%). The lowest mean recovery rate was 84%. A mean recovery rate higher than 100% (±10%) was observed for one flavor, and this is thought to be attributable to human error.
The second experiment assessed the retention efficiency of EXtrelut® NT 3 cartridges by comparing the concentrations of the test flavors present in (i) the test flavor solution (theoretical 100% value) and (ii) the eluate from an EXtrelut® NT 3 cartridge spiked with the test flavor solution and flushed with conditioned air (22 ± 2°C and 60 ± 5% relative humidity) at 0.7 L/min during 30 min prior to extraction with 2-propanol until recovery of 20 mL (Figure 7, trapping condition n°3). The experiment was expected to demonstrate the maximum loss of flavor that can be expected owing to non-retention or evaporation and probably overestimates potential losses.
For 63% of the flavors, the mean retention efficiency was good (defined as recovery >85% after flushing). For 16% of the flavors, the mean recovery rates were between 70% and 85%, indicating some losses during the process. For the remaining flavors, recovery rates below 50% indicated that the EXtrelut® NT 3 cartridges were not suitable for efficiently trapping and retaining these substances.
The third experiment assessed the transfer rates of flavors from the flavor solution to the aerosol and to an inhalation exposure chamber by determining the ratios of the concentrations of the test flavors present in (i) the eluate from an EXtrelut® NT 3 cartridge spiked with the test flavor solution and flushed and (ii) the eluate from an EXtrelut® NT 3 cartridge used to trap the aerosol after the aerosol was passed through 3.85 m of glass piping (Figure 7, trapping condition n°4).
For 71% of the flavors, the mean recovery rates were >60%. The other flavors had mean recovery rates between 45% and 60%, except for one flavor with a mean recovery rate lower than 45%. The transfer rates ranged from 113% to 58%, with a median transfer rate of 73%. Recoveries higher than 100% may be explained by differences in the trapping conditions, that is, continuous loading of the compound by CAG aerosolization versus single loading of the compound by spiking an EXtrelut® NT 3 cartridge.
To evaluate which parameter mainly influenced the results, the recovery rates obtained with the CAG-generated aerosol were divided by those obtained by spiking and eluting the flavor solution. A result close to 100% would indicate that the compound is correctly aerosolized by the CAG system and transported without loss in the tubing. A result lower than 100% would indicate that the compound is either not appropriately aerosolized by the CAG system or that there is a loss during transportation.
This gross estimation indicated that one-third of the study flavors were efficiently aerosolized by the CAG and transported through the tubing, as demonstrated by transfer rates above 85%. Altogether, up to half of the flavors showed aerosolization and transfer rates higher than 70%. Yet, these estimates should be considered with caution given the important difference between continuous loading of flavor substances from CAG-generated aerosol versus single loading by spiking of a stock solution on the EXtrelut® NT 3 cartridge.
Online methods
Offline chemical determination methods, such as those described in the previous section, have several limitations. By definition, they are not performed concurrently with the actual studies, and the representativeness and reproducibility of the generation and exposure processes are not guaranteed. These methods are time-consuming, given the multiple steps required, including trapping, extraction, and measurement. These methods require the accumulation of a certain number of puffs and are not suitable for puff-by-puff analyses. Hence, they are limited in their capacity to rapidly screen fast-evolving EVPs and e-liquids, and there is a need for development of methods for sampling and analyzing complex aerosols online.
Online analysis using Fourier-transform infrared spectroscopy (FTIR)
Fourier-transformed (FT) infrared (IR) spectroscopy may be used for online, puff-by-puff quantification of key e-vapor constituents. FTIR is a form of IR spectroscopy which allows simultaneous, rather than individual, real-time measurement of the infrared frequencies of multiple gas phase substances in complex mixtures thanks to the use of an interferometer.
The IR source produces broadband IR radiation, which is modulated in the interferometer. The modulated IR radiation passes through the sample cell, where the sample gas absorbs certain wavelengths of the IR radiation. The detector detects the transmitted IR radiation. The signal is digitized by an A/D converter. The computer performs a mathematical Fourier transformation on the digitized modulated signal and generates a spectrum.
FTIR allows substantial gains in signal-to-noise ratio, resolution, speed, and detection limits. FTIR spectroscopy has been widely used for trace gas analysis of the atmosphere. 102 It has also been used for analysis of CS. 102 The method has been adapted for online quantification of multiple e-vapor constituents on a puff-by-puff basis and at critical moments, such as at the end of battery life or end of e-liquid content.
Figure 8 shows an experimental setup which uses a GasmetTM portable gas analyzer for real-time quantification of a diluted e-vapor. The GasmetTM analyzer incorporates an FTIR spectrometer, a temperature-controlled sample cell, and signal processing electronics.
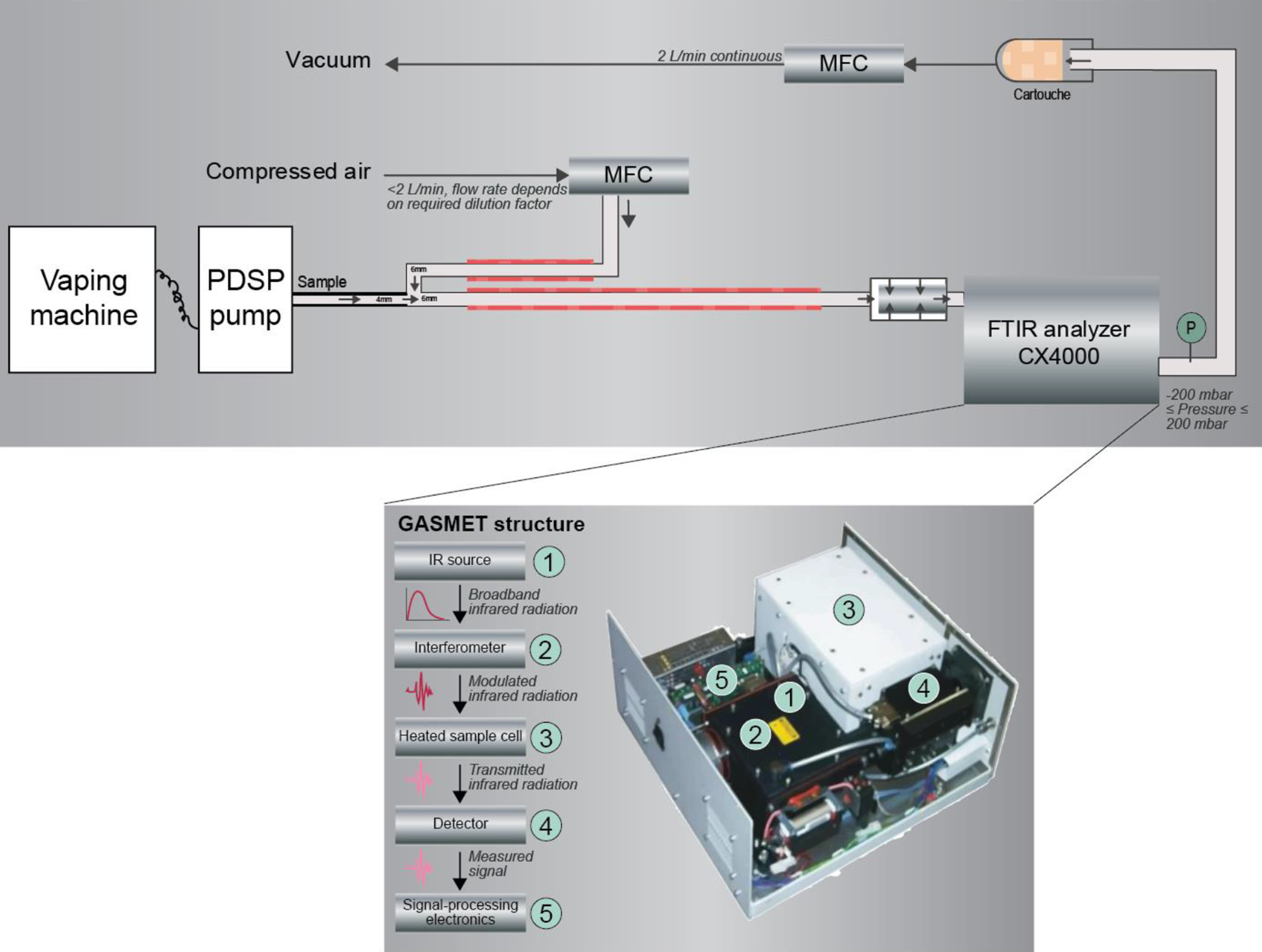
Experimental setup for online analysis of diluted e-vapor constituents by FTIR spectroscopy. IR, infrared; MFC, mass flow controller; PDSP, programmable dual syringe pump. E-vapor is generated with a vaping machine in accordance with ISO 20768/CRM 81.
The tubes that transport the aerosol to the analyzer are heated to 180°C to allow the mass transfer of liquid particles into the gas phase, thereby minimizing loss by particle deposition. Full vaporization of liquid particles is also needed for accurate quantification, as the FTIR is designed for gas analyses. It is critical that the temperature needed for vaporization does not trigger substance modifications. For substances with boiling points higher than 180°C, such as VG (boiling point of 290°C), lower recovery should be expected because of deposition due to condensation in the system.
A vacuum pump pulls the aerosol through the gas analyzer, usually at a flow rate of 2 L/min. The FTIR spectrometer collects data over a wavenumber range of 4200–900 cm−1, with a spectral resolution of 0.5 cm−1.
The FTIR, which measures signals in the order of 1 s, also allows puff-by-puff analysis of the aerosol generated by a single EVP by using a programmable single syringe pump. However, the dynamic response of the FTIR is limited by the volume of the sampling unit (approximately 400 mL, as compared with a puff volume of 55 mL according to ISO 20768/CRM 81).
The FTIR results were shown to be in good agreement with standard liquid and aerosol analytical results. The EVP yields of selected constituents (nicotine, VG, PG, and water) could be quantified in a diluted aerosol with approximately ±10% differences compared with the results of ACM filter analysis. 103
Photon ionization time-of-flight mass spectrometry
Photon ionization (PI) TOF MS is an established technology for online, puff-resolved characterization of organic substances in CS. 104,105 The method uses a combination of photo ionization (PI) and time-of-flight mass spectrometry (TOFMS) to accelerate and separate ions by mass. Known aerosol constituents can be identified and quantified in real time on the basis of their molecular mass.
Figure 9 shows the experimental setup for online chemical characterization of an e-vapor in an in vitro exposure system at the ALI.
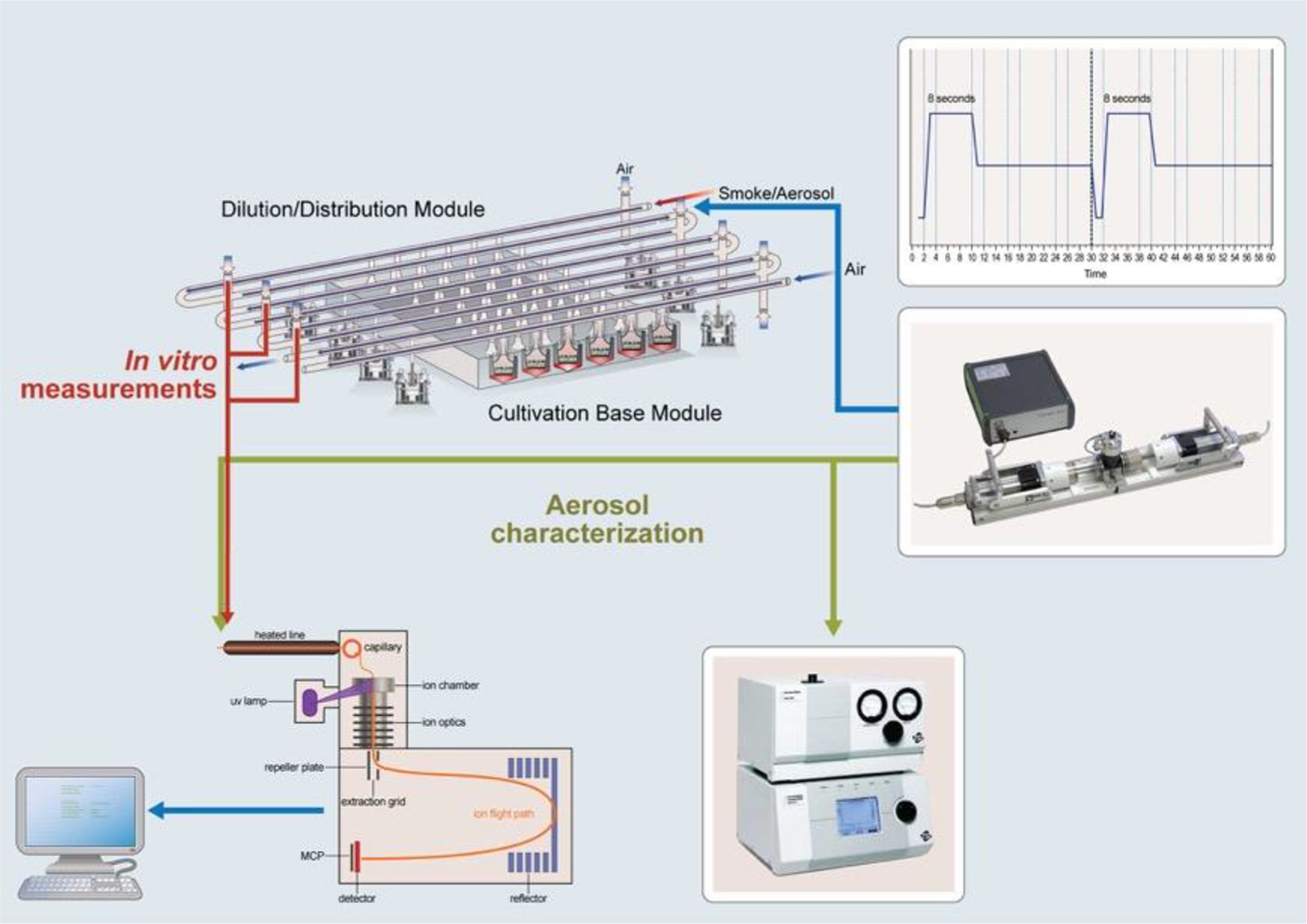
Experimental setup for online characterization by PI-TOFMS of an e-vapor in an exposure system at the ALI. Source: Frege et al. 106
The blue arrow represents the aerosol entering the Vitrocell system after generation. The green lines represent the measurements for characterization of the primary aerosol. The red arrow represents the sampled aerosol from the in vitro system.
The setup described here uses a programmable dual syringe pump (PDSP; Burghart Messtechnik GmbH, Wedel, Germany) to generate e-vapor from two EVP devices. The e-vapor is distributed to an exposure system at the ALI, the Vitrocell® 24/48 exposure system (VC24/48, Vitrocell® Systems GmbH, Waldkirch, Germany). The e-vapor is sampled and characterized by using a single PI-TOFMS (Photonion GmbH, Schwerin, Germany).
The diluted aerosol is sampled at a flow rate of 2–5 mL/min from the dilution/distribution system through a 3-m capillary of 180-µm inner diameter. Sampling is perpendicular to the direction of the aerosol flow. The capillary is heated (up to 300°C) to ensure mass transfer of the liquid particles to the gas phase and avoid mechanical blockage of the capillary flow in case of aerosol deposits along it.
The sample is then ionized by a neodymium-doped yttrium aluminum garnet (Nd: YAG) laser with third-harmonic generation vacuum ultraviolet light at 10.3–10.5 eV (118–160 nm). After ionization, the ions are guided through a set of lenses toward the TOF region, where they are detected according to their m/z ratio. Mass spectra are reported at a frequency of 1 Hz, and the covered mass range is 10–2000 m/z. Absolute quantification is based on compound-specific cross-sections (ionizabilities) relative to toluene at a concentration of 100 ppm.
Photon ionization results in only limited fragmentation of analytes; hence, known aerosol constituents can be identified and quantified on the basis of their molecular mass. Yet, the risk of biased quantification in the presence of isobaric molecules should be addressed, for example, by identification of mass fingerprints for substances for which fragmentation occurs.
Physical characterization of e-vapors—Particle size distribution
PSD is a key physical characteristic of an aerosol, as it determines the potential for deposition along the respiratory tract. 44 PSD determination is, therefore, a key element of aerosol characterization in an experimental setting.
Measurement of the PSD of e-vapors can be particularly challenging because of the concentration and volatile nature of these aerosols. Prior to PSD measurement, some dilution of the e-vapor is necessary to adapt to the flow rate and/or particle number density limits of the equipment used. E-vapors are highly affected by dilution, as the volatiles may partially or totally evaporate, which biases size estimates toward smaller values, thus causing underestimation of mass distributions 14,48,107,108
Two instruments are routinely used to determine PSD: the PIXE cascade impactor and the Aerodynamic Particle Sizer® (APS™). Their use for PSD measurement of ENDP aerosols has been described previously. 84
Cascade impactors are the most commonly used instruments for measuring aerosol PSD, and they are a reference method for inhaled pharmaceutical products. 109,110 A cascade impactor uses particle inertia to size fractionate an aerosol. The aerosol is drawn through a series of progressively finer nozzles toward impaction plates. By reducing the diameter of the nozzle, the velocity of the airflow increases. Particles are collected on the different impaction plates according to their aerodynamic diameter. Larger particles with greater inertia impact on the impaction plate and are collected, while smaller particles follow air streamlines around the collection surface. Inertial methods such as cascade impactors are not affected by particle number density, as long as this does not result in overloading of the impaction plates. Yet, they do require a constant flow and, if this constant flow is not matched to the e-vapor generation flow, dilution of the aerosol is required. Additionally, because the impactor stages must be analyzed gravimetrically, mass losses due to evaporation of volatiles must be taken into consideration. 46
The APS™ measures the time of flight at defined velocity of aerosol particles in an accelerating airflow through a nozzle. 111 Small particles reach a higher velocity than larger particles, which lag behind because of inertia. Particle velocity is measured in an optics chamber by passing the particles through two laser beams separated by about 200 µm. A particle passing through both beams generates one signal with two crests. The time of flight between the two crests is related to the velocity and, hence, the aerodynamic diameter of the particle. This measurement detects particles in the range of 0.5–20 µm in densities up to 1000 particles/cm 3 . At higher densities, errors due to coincidence will increase. At the particle number densities observed in e-vapors, coincidence counting is a concern. Coincidence counting occurs when two particles are too close together and are counted as one particle. To minimize coincidence counting, e-vapor should be diluted; but, dilution will affect the PSD of the aerosol because of evaporation of the volatiles and semi-volatiles.
In vitro toxicity testing in submersed cell cultures
Standard toxicity assessment of CS has traditionally relied on a battery of in vitro cytotoxicity, mutagenicity, and genotoxicity assays using non-human cell lines, including the neutral red uptake (NRU) assay, Ames assay, in vitro micronucleus test, and mouse lymphoma assay (MLA). 112 These in vitro genetic toxicology assays are susceptible to producing erroneous results, especially false positives, under extreme conditions such as increases in osmolality and pH. These results can be misinterpreted as biologically relevant. Other factors, including high test concentrations, liver microsome activation systems, or lysosomal nuclease release, potentially confound the performance and predictivity of in vitro assays that detect genetic damage. 113 Therefore, research on EVPs is turning to new in vitro assays being developed to complement the traditional battery of in vitro assays and that use physiologically relevant biological models of the respiratory tract and cardiovascular system. To test the progress toward toxicity reduction, endpoints are selected that are mechanistically linked to smoking-related chronic diseases. 9
For relatively inexpensive and rapid screening of e-liquids and e-vapor fractions, 2D airway cell culture systems (i.e., monolayers of uniform epithelial cells with incomplete differentiation) are appropriate. They are easier to handle, less expensive, and more suitable for large-scale studies than 3D cell culture systems such as airway cell cultures differentiated into a pseudostratified airway epithelium comprising basal, ciliated, and mucus-producing cells. These cells will be better suited for testing at the ALI. Primary human bronchial epithelial cells have been shown to be useful and relevant 2D cultures for airway pathophysiology toxicity screening. 114 –116 The potential effects of e-liquids and e-vapor fractions on cardiovascular disease risk have been assessed in primary human coronary artery endothelial cells. 117
The flavor toolbox approach
One of the key challenges of EVP testing is the number and variety of flavoring substances that can be used in potentially endless combinations. The use of in vitro toxicity testing in submersed cell cultures has been recommended for streamlining the testing process. 9 A testing strategy has been proposed that uses 2D primary human lung epithelial cells. 118 This “flavor toolbox” approach aims at systematic screening of a large number of flavoring substances for potential toxicity prior to additional testing of selected identified substances for confirming their safety. Ultimately, it aims at identifying acceptable substances and use levels and forming a toolbox of flavors that can be used in e-liquids.
The flavor toolbox approach is based on a read-across principle, whereby data obtained for a data-rich substance (the source) can be used for data-poor substances (the targets) which are considered similar enough. 118 To that effect, flavoring substances of interest are clustered into chemical groups of structurally related substances expected to exhibit similar metabolic and biological properties, e.g. by using the flavor groups defined by the European Food Safety Authority (EFSA). 119 At least one source, or flavor group representative (FGR), is selected for each group. The FGR is the chemical predicted to display the highest toxicological effect within the group on the basis of physicochemical properties, available/predicted toxicological data (i.e., lethal dose 50%, predicted lethal concentration 50%, predicted ocular irritancy, predicted developmental toxicity, and predicted chronic lowest-observed-adverse-effect level), and estimated usage levels for the different flavoring substances.
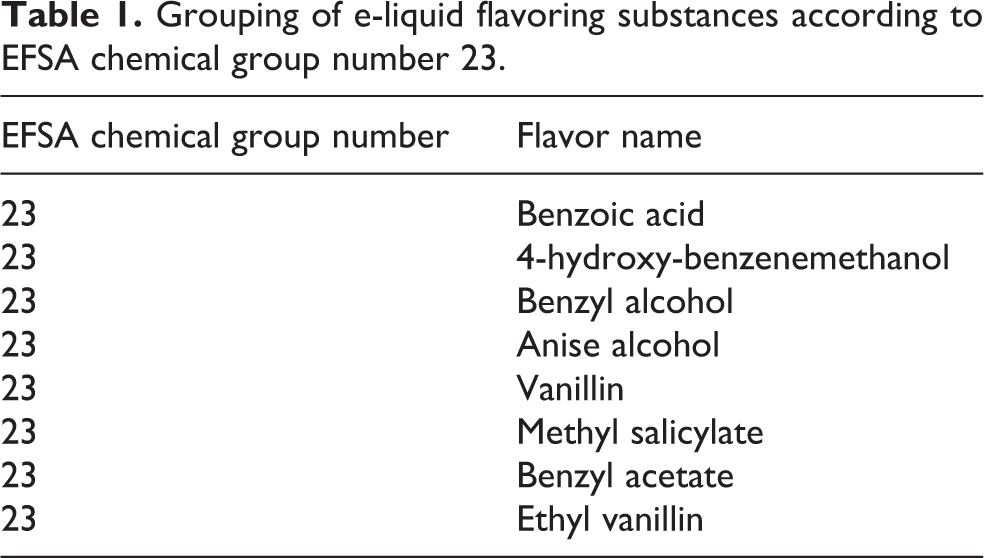
Table 1 shows one example of the grouping of commercially used e-liquid flavoring substances in EFSA chemical group 23 (benzyl alcohol/aldehydes/acids/esters/acetals) and the identified FGR (in bold).
Grouping of e-liquid flavoring substances according to EFSA chemical group number 23.
In order to address potential synergistic and/or antagonistic interactions, flavorings should be tested both individually and in combination. This approach also allows unmasking of interactions in flavor mixtures that may mask or dampen the effect of a flavoring substance. Marescotti et al. proposed an iterative method based on a single flavoring substance exclusion strategy. 118 If a flavor mixture displays increased toxicity relative to a flavor-free base solution, new mixtures are generated by removing one flavoring substance at a time. By assessing these mixtures, it should be possible to identify which flavoring substance contributes to the observed increase in cytotoxicity. This iterative flavor assessment workflow is described in Figure 10. In this example, the toxicity assays used included (1) RTCA, (2) evaluation of a panel of phenotypic high-content screening (HCS) endpoints, and (3) a combination of gene expression analysis (GEX) and computational models to gain a deeper understanding of the changes occurring at the molecular level. 118 The cytotoxicity of the mixture is first assessed by RTCA and compared with the cytotoxicity of the flavor-free base solution containing PG, VG, and nicotine. If no increased cytotoxicity is observed, the mixture is further assessed by HCS and GEX to collect further evidence. The concentration of each flavoring substance should be selected on the basis of actual maximum use levels and include low, medium, and high concentrations. In case a flavor cannot be dissolved in the PG, VG, and nicotine base solution at the target concentration, it should be tested at the highest soluble concentration.
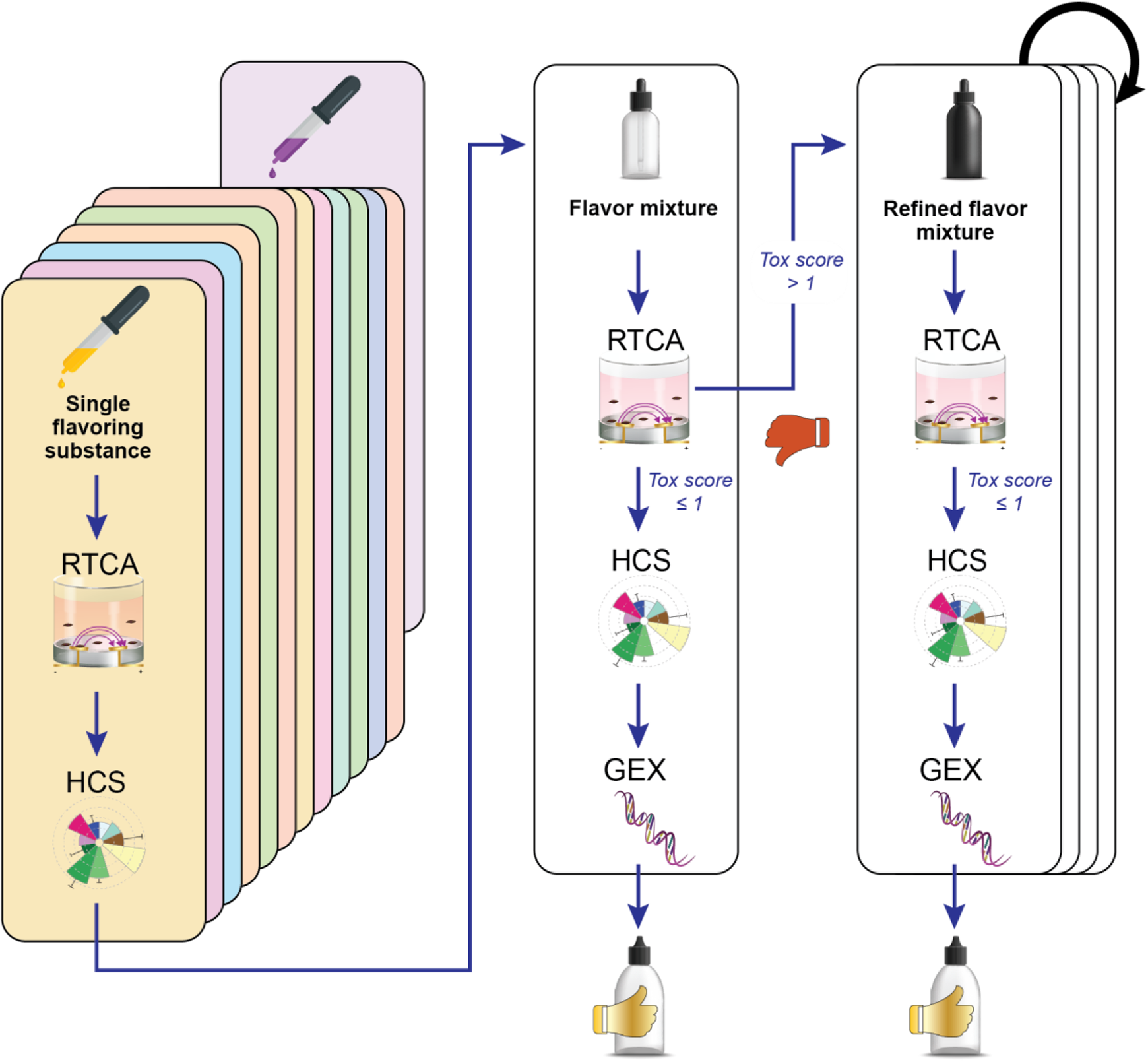
Iterative flavor assessment workflow. RTCA, real-time cell analysis; HCS, high-content screening; GEX, gene expression.
The ToxTracker® assay for screening of e-liquid flavors
New assays are being developed that have the potential to complement the traditional battery of in vitro cytotoxicity, mutagenicity, and genotoxicity assays. One example is the ToxTracker® assay (Toxys, Leiden, Netherlands), a panel of six green fluorescent protein (GFP)-based mouse embryonic stem (mES) reporter cell lines, which can be used to identify the biological reactivity and potential carcinogenic properties of compounds in a single test (PMI, data not published). ToxTracker® consists of a panel of six different mES GFP reporter cell lines representing four distinct biological responses that are associated with carcinogenesis—that is, general cellular stress, DNA damage, oxidative stress, and unfolded protein response. The assay is a relatively new addition to the battery of classical genetic toxicology endpoints and is currently under validation by the European Union Reference Laboratory for Alternatives to Animal Testing (EURL ECVAM).
The technical performance and reproducibility of the ToxTracker® assay was assessed by using a selection of flavoring substances commonly used in commercial e-liquids. The flavorings were assessed either individually or as constituents of two e-liquid formulations (high and medium concentrations) containing PG, VG, and nicotine. Individual test flavor solutions were prepared as 1 ± 0.01 M stock solutions in dimethyl sulfoxide (DMSO) (or ethanol, in case of solubility problems) within a week of the planned dates for in vitro exposure. They were stored in amber glass vials at 3 ± 2°C in the dark. Cell culture and treatment were performed by following the procedures recommended by Toxys in the Toxtracker® protocol. The exposure doses (low, medium, and high) were determined in a dose-range-finding assay. For single flavoring substances, doses within the acceptable range of toxicity for performing a valid ToxTracker® assessment were identified. For complex e-liquid solutions, the maximum acceptable cytotoxicity of each solution was used to determine the top dose. A minimum of two independent experiments were performed for each Toxtracker® cell line with and without the S9 metabolizing fraction of Aroclor-treated rat liver. Throughout the exposure, the microplates were covered with a vapor barrier to minimize the loss of volatile material while still allowing CO2 exchange. The cells were exposed for 24 h, washed with phosphate-buffered saline (PBS), and detached from the microwell plate. The intact cells in suspension were assessed for GFP expression by using a high-throughput Becton Dickinson FACS Canto II cytometer with the Facs Diva 8 software. The threshold for a negative test outcome was a change in the GFP signal less than 2-fold above the background of the solvent control, while a positive result was a value greater than a 2-fold change above background. 120
The Toxtracker® assay performed successfully and was able to discriminate among the different test solutions. A range of responses were obtained. Some compounds were uniquely positive in the presence of the S9 metabolizing fraction, indicating a need for metabolic activation of a progenotoxin. Other compounds tended to possess a similar broad positive profile across the mechanistic reporter cell lines. Some compounds failed to elicit positive responses with any of the ToxTracker® reporter systems and were, therefore, not considered to possess genotoxic or non-genotoxic carcinogen potential. Finally, some compounds were determined to be negative in the ToxTracker® assay main endpoint (genotoxic DNA damage) but were positive in several of the potential mechanistic pathway endpoints.
The assay could be leveraged in the future to screen an extended library of flavoring substances and complex e-liquid formulations. As the ToxTracker® system does not provide mechanistic information on classical endpoints (clastogenicity vs. aneugenicity), further verification and validation of results is warranted.
Collection of e-vapor fractions for in vitro toxicity testing in submersed cell cultures
In vitro cigarette smoke (CS) assessment has traditionally relied on isolating CS fractions and assessing them in submersed cell cultures systems. Typically, the total particulate matter fraction (TPM) is captured on a Cambridge filter pad (CFP) and eluted in dimethyl sulfoxide (DMSO) or ethanol, and aqueous extracts and gas–vapor phase (GVP) fractions are obtained by bubbling the aerosol through cell culture media or PBS. This approach has been extended and adapted to the study of ENDP aerosols and e-vapors. 84
The aerosol collection and fractionation process has several limitations. The methods used for trapping aerosols may exert profound effects on the physical and chemical composition of the aerosols. Accordingly, the composition of the trapped aerosol fractions should be assessed and compared with that of the e-liquid by, e.g. fingerprint analysis or differential screening. No single method can efficiently trap all of the constituents present in the solid, liquid, and gas phases. The analysis of individual fractions might underestimate the overall toxicity resulting from dynamic evolution and interactions among the phases. The stability of the collected materials prior to analysis has to be ascertained. The solvents used might react with constituents of the aerosol fraction or exert biological activity themselves. For in vitro experiments in submersed cell cultures, the aerosol should be collected in a biocompatible solvent, which can then be applied to the cells. Furthermore, standard trapping and extraction procedures have been developed for CS. Given the qualitative and quantitative differences between CS and e-vapors, these methods need to be optimized to capture comparable samples from these products. Test item number, trapping solvent volume, sample collection point, and trapping temperature may all influence the efficiency of trapping a given fraction from a given aerosol. 84,121
Table 2 shows the parameters for collecting comparable aerosol fractions from 3R4F CS, aerosol from an ENDP, the Tobacco Heating System 2.2 (THS 2.2; Philip Morris Products S.A., Neuchâtel, Switzerland), and e-vapor from P4M3 version 1.0 (P4M3 v 1.0, Philip Morris International, Switzerland) (PMI, data not published).
Trapping systems and parameters for trapping aerosol fractions from 3R4F, THS 2.2, and P4M3 v 1.0. Source: van der Toorn et al. 122
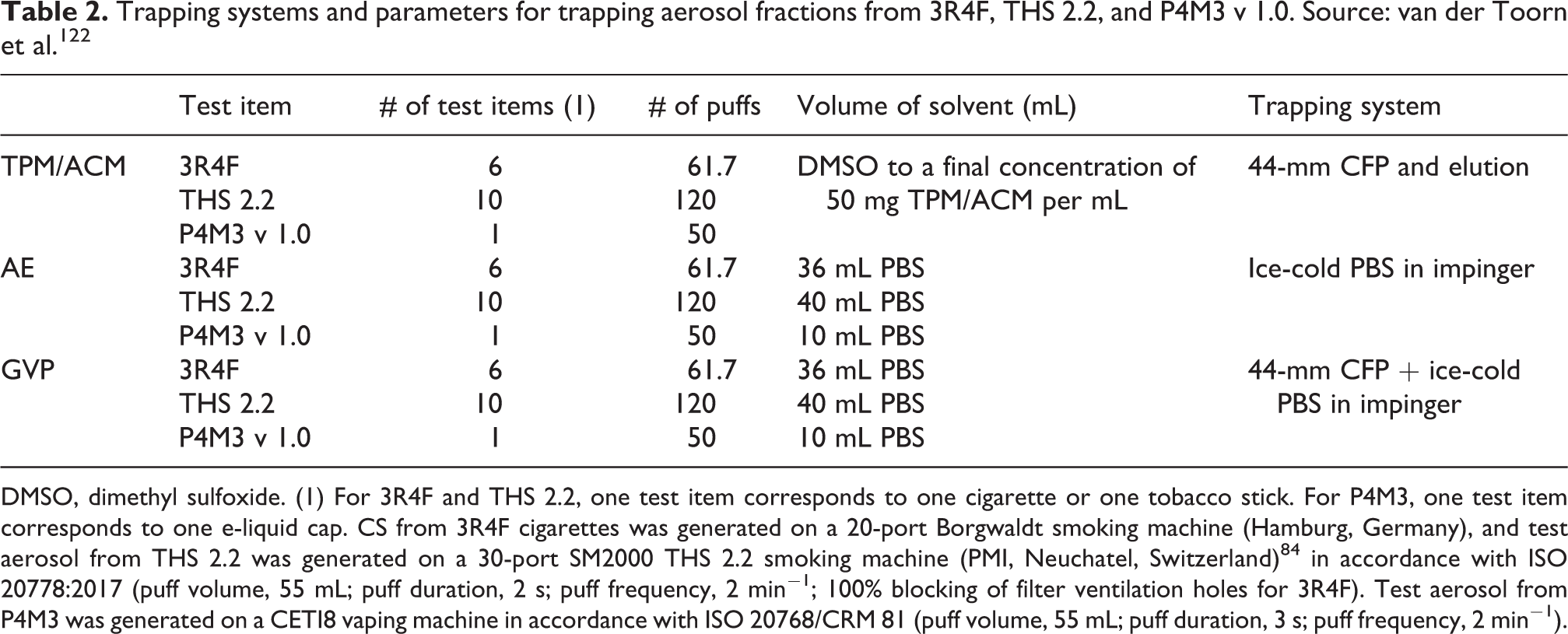
DMSO, dimethyl sulfoxide. (1) For 3R4F and THS 2.2, one test item corresponds to one cigarette or one tobacco stick. For P4M3, one test item corresponds to one e-liquid cap. CS from 3R4F cigarettes was generated on a 20-port Borgwaldt smoking machine (Hamburg, Germany), and test aerosol from THS 2.2 was generated on a 30-port SM2000 THS 2.2 smoking machine (PMI, Neuchatel, Switzerland) 84 in accordance with ISO 20778:2017 (puff volume, 55 mL; puff duration, 2 s; puff frequency, 2 min−1; 100% blocking of filter ventilation holes for 3R4F). Test aerosol from P4M3 was generated on a CETI8 vaping machine in accordance with ISO 20768/CRM 81 (puff volume, 55 mL; puff duration, 3 s; puff frequency, 2 min−1).
In vitro toxicity testing at the ALI
ALI exposure systems have been continuously developed since the 1970s, 83 and various commercial or in-house-designed systems are currently applied in a range of research areas. 123 Researchers are increasingly using exposure systems at the ALI to study aerosols such as CS, ENDP aerosols, and e-vapors. 124 –127 Exposure systems at the ALI are more physiologically relevant than 2D submersed cell exposure systems for studying tissues exposed to airborne substances. The 3D cell cultures exhibit characteristics of a pseudostratified epithelium and contain mucus-secreting goblet cells, columnar ciliated cells, and basal cells, similar to the in vivo epithelial tissues. 124,128 –130 In addition, exposure at the ALI mimics exposure of the human respiratory tract tissues.
The VITROCELL® 24/48 exposure system has been characterized and validated for use with CS and THS 2.2 aerosol (Figure 11). 84
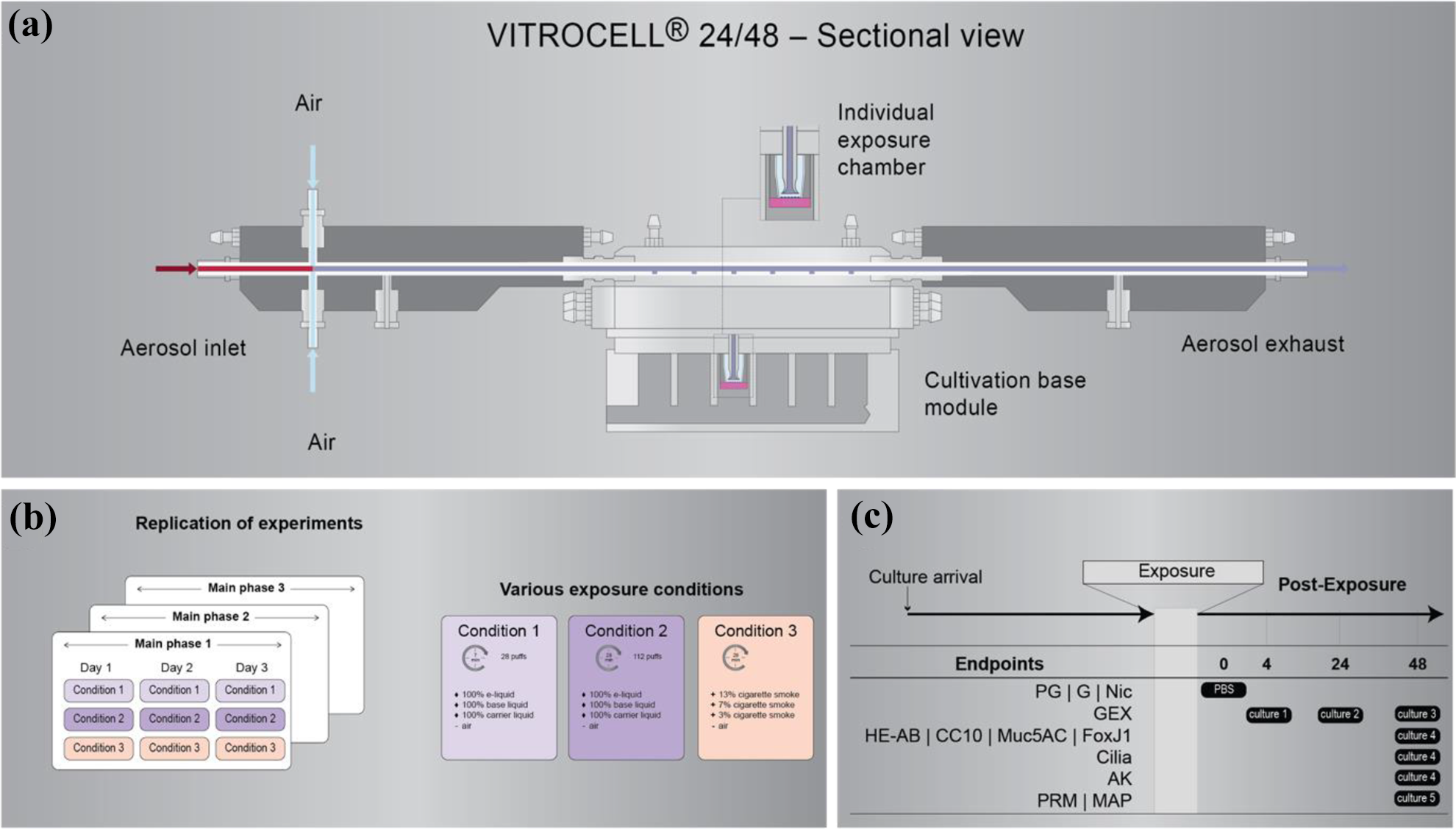
Schematic representation of the VITROCELL® 24/48 exposure system (A) and example of an experimental study design (B, C).
The system consists of a climatic chamber where aerosol can be delivered to human organotypic cultures under controlled temperature and humidity conditions. For studying e-vapor aerosols, the VITROCELL® 24/48 exposure system is connected to an e-vapor generation machine, such as a programmable single syringe pump, Collison nebulizer, or CAG. E-vapor is generated as previously described and delivered to the VITROCELL® 24/48 exposure system dilution/distribution system through a fluoroelastomer tube (ISO-Versinic®, Saint-Gobain, Courbevoie, France). The distance from the generation to the exposure chamber is kept as short as possible to minimize aerosol evolution. Up to 48 cell culture inserts, grouped into 8 rows of 6 replicate positions, can be exposed simultaneously under controlled temperature and humidity conditions. The test aerosol passes through a dilution/distribution pipe located on top of the base module that hosts the inserts. At the beginning of each row, the aerosol can be diluted with conditioned pure air to produce a total of eight distinct dilution values, which can be tested simultaneously (one per row). Under standard operating conditions, only seven dilution values are tested, while the eighth row is used for negative control exposure to clean air only and is not connected to the aerosol supply. The aerosol is partially sampled—by negative pressure applied by a vacuum pump connected to the cultivation base module—through trumpet-shaped protrusions from the dilution/distribution system into each cell culture insert. At the end of each line, one trumpet allows the sampling and real-time monitoring of aerosol mass deposition by quartz crystal microbalances (QCM). 131
The Vitrocell® 24/48 exposure system: Characterization for use with e-vapors
Despite ongoing efforts, limited information and few guidelines are available for characterizing and validating commercial exposure systems. 123,132 Flexibility in the design of exposure experiments, unspecified aerosol type-related application, and poorly defined maintenance of the exposure equipment can potentially result in non-reliable and highly variable results. Particular attention must, therefore, be given to understanding how and to what extent the aerosol is modified by the experimental protocol or system before it reaches the exposure site. An appropriate link between the generated and delivered aerosol must be established from the exposure–dose perspective.
The VITROCELL® 24/48 exposure system has been characterized for use with e-vapors. Observed aerosol dynamics during aerosol transportation, dilution, and sampling at the exposure trumpets warrant that the optimal system settings be empirically determined before conducting a study, to achieve accurate and controlled dose delivery with minimal variation.
Steiner et al. assessed the deposition of a fluorescently labeled liquid aerosol generated by a condensation monodisperse aerosol generator (TSI 3475, TSI, Shoreview, MN, USA) in the cell culture inserts. 133 The study showed a higher variability in delivery with the liquid aerosol than when the system is used with CS or ENDP aerosols. Globally, it was found that the aerosol delivery to a position of choice in a repetition of choice can reasonably be expected to lie within the range of the average delivery to all positions exposed to the same aerosol concentration ± 25%. Serial dilution resulted in a clearly discernible dose–response; yet, the aerosol concentration and delivery did not generally change by the same factor. The study also showed that QCMs could not report accurate mass deposition of the tested liquid aerosol. The mass of the liquid deposits, depending on the thickness of the deposited layer and viscosity of the material, may be underestimated because of viscous energy dissipation into the liquid layer. 134,135
Frege et al. investigated the evolving aerosol inside the VITROCELL® 24/48 exposure system by using online single-photon ionization MS (SPI-MS) to measure in real time three main constituents of a test aerosol (PG, VG, and nicotine). 106 The aerosol was generated from the EVP P4M3 v 1.0 by using a programmable dual-port syringe pump. The concentrations of PG, VG, and nicotine were measured in the aerosol sampled at the end of each delivery line and at the QCM port in three exposure protocols: (i) no dilution, (ii) gradually increasing dilution at each line, and (iii) single-dilution applied only once at the beginning of the line. The measured concentrations were compared with expected values calculated from the concentrations measured in the aerosol generated by the programmable dual-port syringe pump and calculated expected changes due to differential phase partitioning, selective sampling, and deposition. The changes in the aerosol as dilution was applied showed not only a reduction in the concentrations of the traced substances but also selective sampling due to evolution of the aerosol and phase partitioning of the substances that formed the aerosol. Consequently, the authors recommended that comparative in vitro assessment studies should be conducted with attention to both dilution rates and their actual application in the study design, as these two factors exert direct effects on the delivered doses.
Determination of e-vapor delivery and deposition in the VC24/48 exposure chamber (PBS exposure experiments)
The data presented in the previous section highlights the key importance of monitoring actual aerosol delivery to and deposition in the individual VITROCELL® 24/48 exposure chambers. One approach is to measure the deposition of selected aerosol constituents in PBS, either in independent PBS exposure experiments or as part of the experiment, thereby allowing in situ monitoring.
PBS is used as an easy, accurate, and cost-effective surrogate to organotypic cell cultures. PBS provides a good model for the thin film of the aqueous matrix that commonly covers the surface of cell cultures. Furthermore, because of the low complexity and stability of PBS and its inability for metabolic activity and transepithelial transport, gross aerosol deposition can be determined more easily and more accurately during PBS exposure experiments than by post-exposure chemical analysis of living cells. 84
PBS deposition can be used to determine PG, VG, and nicotine deposition in the VITROCELL® 24/48 exposure chambers after exposure to e-vapors. Dummy stainless steel inserts, each filled with 100 µL of MgCl2- and CaCl2-free PBS (Sigma Aldrich, Buchs, Switzerland), are placed in the cultivation base module. Usually, for in situ assessment, each row is loaded with three cell culture and three dummy inserts in randomized positions, for three replicates per aerosol concentration. Immediately after exposure, aliquots of exposed PBS are collected from each steel insert and pooled row-wise into silanized amber glass vials. These liquid samples are stored at −80 °C until chemical analyses targeting quantification of the constituents present in the aerosol exposed liquid.
Figure 12 shows the concentrations of PG, VG, and nicotine deposited in PBS after 7 or 28 min of exposure to undiluted e-vapors generated by a P4M3 v 1.0 EVP device with a Classic Tobacco flavor e-liquid and a base e-liquid containing 39.1% PG, 39.1% VG, and 1.8% nicotine as well as a carrier e-liquid with the same proportions of PG and VG but no nicotine. Exposure to various dilutions of 3R4F CS is used in comparison.
Concentrations of PG, VG, and nicotine deposited in the VITROCELL® 24/48 exposure chamber.
Boxplots show the concentrations of deposited PG, VG, and nicotine in exposed PBS samples placed in the VITROCELL® 24/48 exposure chamber.
The concentrations of PG and VG deposited in PBS were comparable in samples exposed for the same duration to e-vapors generated from the three e-liquid formulations, which contained identical proportions of PG and VG. Increasing deposited PG and VG concentrations were observed with increasing durations of exposure. In contrast, the concentrations measured following exposure to 3R4F CS were minimal at all exposure durations.
The concentrations of deposited nicotine also increased with the duration of exposure to nicotine-containing aerosols. The nicotine concentrations detected following exposure to the nicotine-free carrier aerosol were around the lower limit of the analytical measurement and comparable to those following exposure to air.
PBS exposure experiments can also be used to determine deposited carbonyl concentration. Carbonyls are an important research topic for modified risk products. Carbonyls might contribute to smoking-related morbidity and mortality and may occur in e-vapor as thermal degradation byproducts. 96
The method described here allows the determination of eight carbonyls trapped in PBS by liquid chromatography (LC) with electrospray ionization MS. An aliquot of a PBS-exposed sample is incubated with a 3:2 (v/v) 15 mM 2,4-dinitrophenylhydrazine (DNPH) solution (>90.0% purity, Acros Organic) for 30 min at room temperature. Chemical derivatization is then quenched by addition of 1:20 (v/v) pyridine after 30 min. Quenching at 30 min allows stabilization of the sample solutions with pyridine while avoiding the formation of poly-derivatized carbonyl adducts.
A 500-µL aliquot of derivatized sample is transferred to an LC–MS glass vial with 485 µL acetonitrile and 15 µL of an internal standard (ISTD) solution containing 24 µg/mL each of acetone-d6-DNPH (ISTD for formaldehyde, acetaldehyde, and acetone), propionaldehyde-d2 (ISTD for acrolein, propionaldehyde, and crotonaldehyde), and methyl ethyl ketone-d5-DNPH (ISTD for methyl ethyl ketone and butyraldehyde). For samples with higher expected levels of carbonyls, such as 3R4F CS, the samples are diluted with a DNPH/PBS/pyridine solution prior to injection with higher dilutions.
LC is performed with an Agilent 1200 series (Agilent Technologies, Santa Clara, CA, USA) high-performance LC (HPLC) system coupled to a Sciex Triple Quad 5500 mass spectrometer (AB Sciex, Framingham, MA, USA) with an electrospray ionization probe operating in negative mode. Aldehyde separation is performed in the isocratic mode in a Chromolith Speedrod RP-18e HPLC column (Merck KGaA, Darmstadt, Germany) by using water, acetonitrile, tetrahydrofuran, and 2-propanol (59:30:10:1, v/v/v/v) at a flow rate of 2.5 mL/min (column set at 40°C and equipped with a post-column splitter 1:6 before entrance into the LC–MS). LC–MS detection is performed in the multiple-reaction monitoring mode. Carbonyl compound concentrations, expressed as µg/item, are calculated by using an external calibration curve.
In vivo assessment
Despite significant progress in the development of complex in vitro surrogate models for in vivo inhalation studies, such as those described earlier in this review, these systems cannot yet fully mimic the complex physiological systems of whole living organisms.
Animal studies are still needed, and rodent inhalation studies are a key element of toxicity assessment of e-vapor products. A well-designed inhalation exposure study can help assess the potential toxicity of inhaled products at elevated aerosol exposure concentrations, repeated exposures, and elevated doses over a significant proportion of an animal’s lifetime and provide a thorough evaluation of the impact upon all exposed tissues and organs.
A literature search shows a very limited number of inhalation studies on e-vapors to date 21,91,136,137 and a few studies on the biological effects of inhaled PG 20,138,139 and VG. 19,37,140 In its 2018 report, the NAS pointed out the lack of long-term data from repeated inhalation exposures to e-vapor and recommended to “[c]onduct long-term (2-year) animal studies, using inhalation exposure to e-cigarette aerosol, to better understand disease risks from inhaling reactive carbonyl compounds and other potentially toxic constituents of e-cigarette aerosol, including flavoring chemicals and additives. These studies should include two controls: combustible tobacco smoke-exposure animals and those exposed to ambient air. Endpoints evaluated should include clinical outcomes and biomarkers relevant for, at a minimum, cancers, cardiovascular diseases, and respiratory diseases and other relevant clinical outcomes.” 1
E-liquid preparation and stability
In vivo inhalation studies require a stable supply of large volumes of e-liquids, which is the first challenge for such studies to be conducted. E-liquids can be chemically unstable, with reactions occurring between their ingredients (such as vehicles and flavors) to form new substances. 92 For this reason, it is of paramount importance to perform a stability study, to devise the proper schedule for preparation of preblends and test e-liquid solutions.
For example, for a 2-year inhalation study of 38 flavorings, 6 preblends were needed to ensure maximum stability. In total, about 64 batches were needed for each preblend, which were manufactured every 2 weeks. Test e-liquid solutions were prepared from these preblends three times per week, as the solutions displayed sufficient stability over 3 days. In total, an estimated 400 batches of test e-liquid solutions will have to be prepared over the course of the study. This example illustrates the complexity of conducting long-term inhalation studies and the critical need for carefully planning such studies.
Continuous, long-term aerosol generation
The use of laboratory aerosol generators, such as the Collison nebulizer or CAG, is particularly suited for long-term inhalation studies, as it simplifies the logistics and removes parameters that could potentially affect reproducibility. As previously described, an aerosol produced by nebulization contains unaltered main e-liquid constituents and will not provide information on substances potentially generated by the thermal treatment of e-liquids. A CAG generates an aerosol intended to be representative of the aerosol generated by a specific EVP device when its temperature is set to mimic the temperature of the heating coil during puffing of that EVP, typically 250–275°C.
The CAG has been shown to be capable of delivering a homogeneous and stable aerosol within target concentrations in a 6-month mouse exposure study in an 800-L, 24-cage whole-body exposure chamber (WBEC). 37 The aerosol was generated from e-liquid stock solutions at 250–275°C by using the setup parameters described in Figure 13, for 3 h per day, 5 days per week, for a maximum exposure duration of 6 months as per study design. PG, VG, nicotine, and guaiacol (selected representative flavoring substance) were monitored daily in the test atmosphere. The MMAD and GSD of the PSD in the test atmosphere exposed to the e-vapor were comparable with those in the test atmosphere exposed to CS and were within the respirable range 37 (Wong et al., in prep).
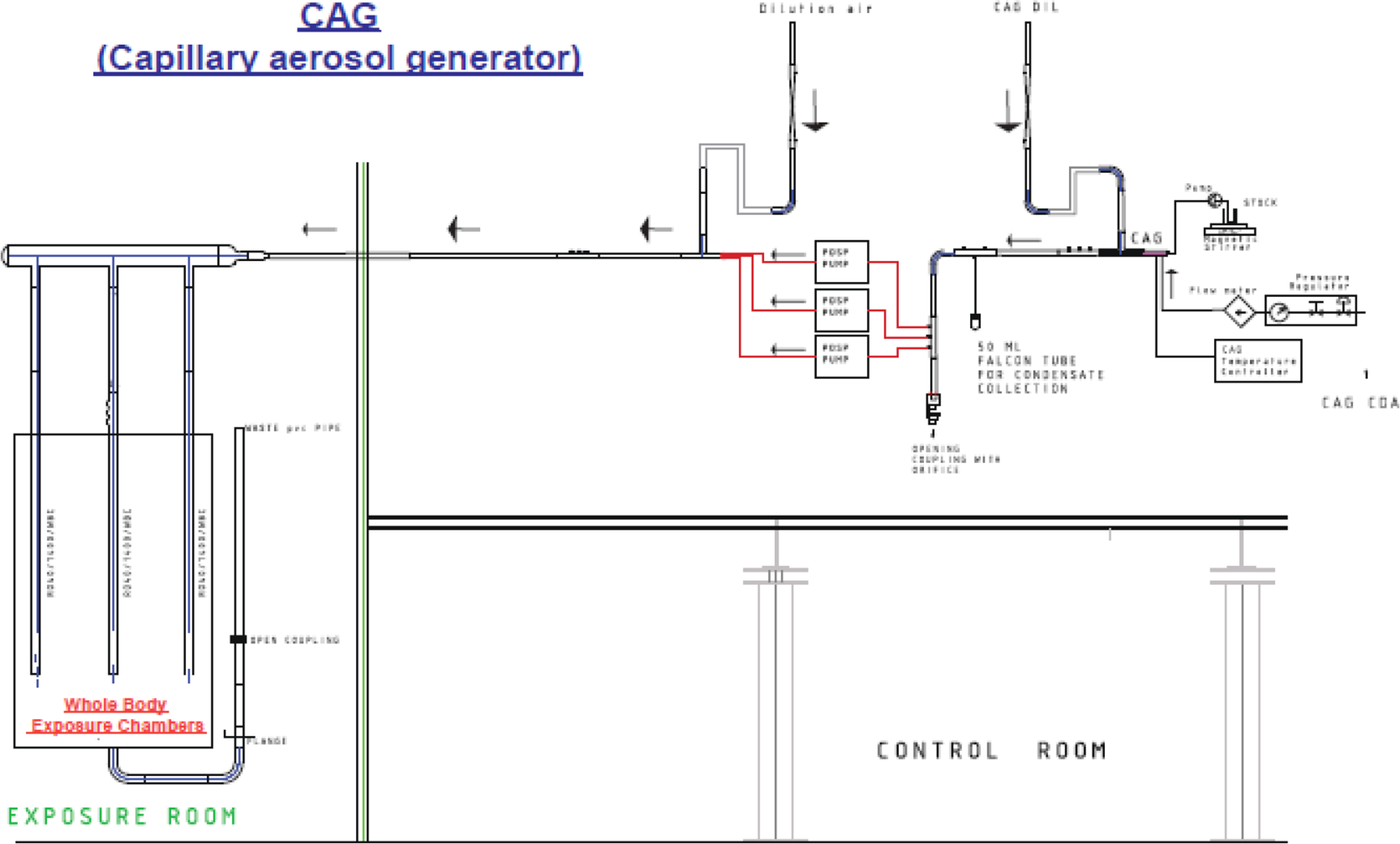
Exposure system consisting of a CAG for aerosol generation and a WBEC for mouse whole-body exposure. Source: Szostak et al. 37
Similarly, a 6-jet Collison nebulizer (BGI) has been shown to reproducibly deliver target concentrations of e-liquid constituents PG, VG, and nicotine at the nose ports of a 32-port nose-only exposure chamber (NOEC) (FPC-132, Patent EP2095791A1; Geraetebau Insul Simsheuser GmbH, Insul, Germany) in a 90-day subchronic, nose-only inhalation study in rats exposed for 6 h per day, 5 days per week as per designed exposure study. 17
E-vapors for inhalation studies can also be generated by vaping machines using EVP devices. An exposure system consisting of a rotary vaping machine and an NOEC was used for a 3-week repeated-dose nose-only inhalation study in female C57BL/6 mice. The test product was a commercially available EVP, MarkTen® (version 2.6.8, Nu Mark), with three different e-liquids formulations. 141 Aerosols were generated in accordance with CRM 81 for up to 4 h/day, 5 days/week, for 3 weeks. Each cartridge was puffed on for up to 180 puffs. The test atmosphere was well controlled during the 3-week exposures. Given the good performance of the system in the 3-week study, the same system has subsequently been used in a chronic inhalation (4 h/day, 5 days/week, 7 months) study in C57BL/6 mice to evaluate the long-term respiratory toxicity of MarkTen® e-vapor aerosols in comparison to the reference 3R4F CS (Kumar et al., in prep).
Chamber equilibration
A stable equilibrium concentration in the exposure chamber is approached and maintained after an initial buildup of the test material. 142 The time to reach 95% equilibrium is a function of the chamber volume, chamber flow rate, and type of aerosol. Owing to their different physicochemical characteristics, different aerosols, such as CS, ENDP aerosols, and e-vapors, behave differently. For example, constituents that exist predominantly in the GVP tend to require shorter durations to reach the equilibrium concentration and display better temporal and spatial homogeneity. Constituents in the liquid or solid phase experience higher depositional losses because of their tendency to impact, coagulate, and deposit. Hence, a relative disproportionate distribution of some constituents due to their partitioning between phases and related depositional losses may occur. 143,144
FTIR was used to determine the chamber equilibration in a 60-port NOEC (CH Technologies, NJ, USA) (PMI, data not published). The chamber is a stainless steel chamber consisting of 5 tiers of 12 exposure ports each. 84 In this experiment, all 60 exposure ports were loaded with glass exposure tubes closed with stoppers and plungers.
The aerosol was generated by a CAG at 250°C, from an e-liquid solution containing weighted mass percentage of the following constituents: PG (23.9%), VG (56%), nicotine (4%), preservatives (0.9%), flavors (0.12%), and water (15.1%).
The FTIR was installed at the glass pipe upstream of the NOEC, and it sampled the atmosphere at a flow rate of 2 L/min to allow continuous monitoring of PG, VG, and nicotine concentrations. The data were compared with measurements obtained by using offline methods. The FTIR findings showed that it took approximately 1 h for the nicotine and PG concentrations to reach 95% of the equilibration concentration. This was likely because of the time required for saturating the surfaces of the tubing and pipes used for aerosol generation and delivery. In line with this finding, it was found that the nicotine and PG concentrations in the atmosphere collected at the nose-ports were lower 30–60 min after the start of exposure than at later time points (i.e., more than 150 min after the start of exposure). For VG, the concentrations measured by FTIR were more than 10% lower than those measured by offline methods, likely because of deposition by condensation within the FTIR setup.
Test atmosphere sampling and characterization
Test atmosphere characterization is critical for understanding and interpreting data from animal inhalation exposure experiments. Achieving the target concentration and PSD is of primary importance, as is their day-to-day reproducibility. Organization for Economic Co-operation and Development (OECD) Test Guidelines provide that an individual measurement in the chamber should not deviate from the mean chamber concentration by more than 10% for gases or by more than 20% for liquid or solid aerosols. 145
The test atmosphere is sampled at sampling ports close to the animal breathing zone at various frequencies, depending on the parameter and exposure group. Typical sampling and analytical conditions are described in Table 3.
Test atmosphere sampling and characterization: Method of determination, sampling parameters, and duration.

ISTD, internal standard; GC: gas chromatography; CFP: Cambridge filter pad; DNPH, 2,4-dinitrophenylhydrazine; LC–MS/MS, liquid chromatography–tandem mass spectrometry; APS, aerosol particle sizer.
Determination of animal aerosol uptake
Even though animals may be exposed to the same test atmosphere, the concentrations of materials that they inhale and that reach their respiratory tract can still vary among individuals because of differences in behaviors, respiratory physiology, and individual anatomical specificities. Hence, aerosol exposure and uptake by the animals should be monitored to demonstrate physiologically relevant exposure and provide a basis for data interpretation and comparison.
Typically, exposure measurements include blood nicotine, cotinine, and carboxyhemoglobin (COHb) levels, plasma PG levels, and urine levels of aerosol constituent metabolites.
Blood samples (target volume, approximately 500 µL/mouse) are collected within 15 min post-exposure from the retro-orbital venous plexus under isoflurane anesthesia by using uncoated (i.e., no heparin) polyethylene tubes containing ethylenediaminetetraacetic acid. Blood samples are kept on wet ice until centrifugation for at least 10 min at ∼1500 to 2000 × g to obtain plasma. The plasma samples can be stored at ≤−70°C until use. COHb levels are measured in whole blood by spectrophotometry.
Nicotine and cotinine levels are measured in plasma by LC–MS/MS with atmospheric pressure chemical ionization. Prior to analysis, the plasma samples are spiked with D3-nicotine and D3-cotinine. PG is determined in plasma by LC–MS/MS. VG measurements are not relevant because of the presence of endogenous glycerol in plasma.
Urine is collected during and after exposure, for a total period of approximately 24 h. Samples collected during and after exposure are pooled with urine collected by rinsing the cage with approximately 100 µL water. The aliquots are stored at ≤−70°C until analysis.
Determination of nicotine metabolites (trans-3′-hydroxycotinine, norcotinine, cotinine, nicotine-N′-oxide, and nornicotine) in urine is based on their derivatization with 1,3-diethyl-2-thiobarbituric acid. The colored derivatives are separated by HPLC and detected photometrically at 532 nm.
Depending on the study design, additional biomarkers may be evaluated, such as 3-hydroxypropylmercuric acid (HPMA), exposure marker of acrolein; S-phenylmercapturic acid (SPMA), exposure marker of benzene; 2-cyanoethylmercapturic acid (CEMA), exposure marker of acrylonitrile; and 8-hydroxy-2′-deoxyguanosine (8-OH-dG), marker of DNA damage.
Conclusion
EVP research is challenging and complex, mostly because of the numerous and rapidly evolving technologies and designs as well as the multiplicity of e-liquid flavors and solvents that are available on the market. There is an urgent need for international harmonization of methods to avoid generating data that are, at best, difficult to compare and, at worst, may result in misleading comparisons and conclusions. This review was intended to provide detailed descriptions of experimental setups and methods for generation, exposure, and collection of e-vapor for in vitro and in vivo assessment. Such transparent sharing of protocols, tools, and data with a wide audience of scientists in academia, industry working groups, and regulatory bodies is intended to facilitate the alignment of best experimental practices and promote understanding of best methodologies. This, in turn, will lead to the formulation of standards specifically relevant to e-vapor products.
Footnotes
Declaration of conflicting interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors except A.M. and W.K.S. are employees of Philip Morris International. A.M. and W.K.S. were contracted and paid by Philip Morris International.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Philip Morris International is the sole source of funding and sponsor of this research.
