Abstract
During a Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) meeting, the in vitro toxicity testing Sub-Group (IVT SG) met to discuss the evolving field of aerosol exposure research. Given the diversity of exposure parameters and biological endpoints being used, it was considered a high priority to investigate and contextualise the responses obtained. This is particularly driven by the inability to compare between studies on different exposure systems due to user preferences and protocol differences. Twelve global tobacco and contract research companies met to discuss this topic and formulate an aligned approach on how this diverging field of research could be appropriately compared. Something that is becoming increasingly important, especially in the light of more focused regulatory scrutiny. A detailed and comprehensive survey was conducted on over 40 parameters ranging from aerosol generation, dilution and data analysis across eight geographically independent laboratories. The survey results emphasise the diversity of in vitro exposure parameters and methodologies employed across the IVT SG and highlighted pockets of harmonisation. For example, many of the biological protocol parameters are consistent across the Sub-Group. However, variables such as cell type and exposure time remain largely inconsistent. The next steps for this work will be to map parameters and system data against biological findings and investigate whether the observed inconsistencies translate into increased biological variability. The results from the survey provide improved awareness of parameters and nuances, that may be of substantial benefit to scientists in intersecting fields and in the development of harmonised approaches.
Highlights
Results emphasise the complexity and diversity of the in vitro exposure environment
The data show pockets of commonality that can be exploited for harmonisation purposes
However, variables such as cell type and exposure time remain largely inconsistent
The data will help researchers establish parameters in line with the global users
Results will help drive proficiency and inter-laboratory trials and ultimately validation
Introduction
The analysis of tobacco smoke has been conducted in vitro to support continued product assessment for over 40 years. 1,2 Current trends for in vitro tobacco smoke analysis have centred on the need to assess the complete tobacco smoke aerosol and not a fractionated portion. This is for several reasons: 1) known smoke toxicants reside in both particulate and vapour phases of cigarette smoke 3 –5 and some toxicants may be omitted from analysis when using smoke fractions; 2) the need to better mimic human product use and ultimately exposure 6 ; 3) aerosol technology facilitates air-liquid interface (ALI) exposure and the use of a variety of different cell types including primary 3D differentiated human lung tissue; and, 4) 3D lung tissue combined with aerosol exposure systems offer the most advanced and physiologically relevant way to model mechanisms of tobacco associated diseases in vitro. The real advantage of aerosol exposure systems are that they offer the researcher a variety of possibilities to customise the exposure set-up and modify experimental parameters, thus providing a novel, versatile and unique tool, potentially leading to more accurate in vitro to in vivo and human extrapolations.
Aerosol exposure systems have been refined over the last 10 years and are commonly employed by the tobacco industry and in tobacco related and inhalation based research and can be broken down into three functional parts. The smoking machine or aerosol generator, the dilution system (if present) and a module or multi-well plate that houses the cell system. The diversity of exposure systems, modules and plate formats means that this environment is complex with many possible component combinations. Laboratories have either manufactured their own, known as bespoke systems, purchased commercially available systems, or opted to combine both commercially available with bespoke, to create a hybrid approach. When a cell system is combined with the experimental exposure set-up, the result is normally a unique combination, that few laboratories share. The variability and uniqueness of the various systems hinders or prevents comparison of data and inter-laboratory efforts. At this stage either a unified approach is required or a strategy to read-across systems and biological data. This becomes even more important as government agencies such as the United States (U.S.) Food and Drug Administration (FDA), through the Centre for Tobacco Products (CTP) are regulating tobacco products and data generated on these systems could be used in a regulatory submission. An industry-aligned in vitro aerosol exposure approach could have significant advantages to these government agencies and to the companies submitting data, thus strengthening their key messages and the data available. The CTP has several initiatives for evaluating and understanding tobacco products, including; 1) understanding the diversity of tobacco products; 2) reducing toxicity and carcinogenicity of tobacco products and 3) understanding adverse health consequences of tobacco use (www.fda.gov/TobaccoProducts). Currently few if any attempts have been made to harmonise the whole smoke in vitro aerosol environment or to create an aligned approach. Some studies have focused on the characterisation of in vitro exposure systems. Such approaches could be considered as pre-validation efforts. However, to achieve true validation of these approaches, the methodologies must go through formal interlaboratory comparisons. No studies have evaluated the consistency of a biological response in a true-interlaboratory effort. This is in part due to the diversity of the environment. In order to create consistent results across systems and laboratories, an aligned methodology must be employed. Currently there are multiple methods, protocols, system nuances, and different systems all being utilised. To achieve harmonisation, the environment requires a detailed assessment and a strategy with a forward-thinking approach. To give an understanding of the complexity of the current whole smoke in vitro aerosol exposure environment, Table 1 gives a snapshot of some studies dating back over the last 10 years as an example of what exposure systems are being used, how they are being characterised As shown in Table 1, the diversity in the systems is only matched by the variety of techniques and how each of these systems have or have not been characterised and how the biological responses have been presented.
In vitro aerosol studies characterising dose within and comparing between and across systems.
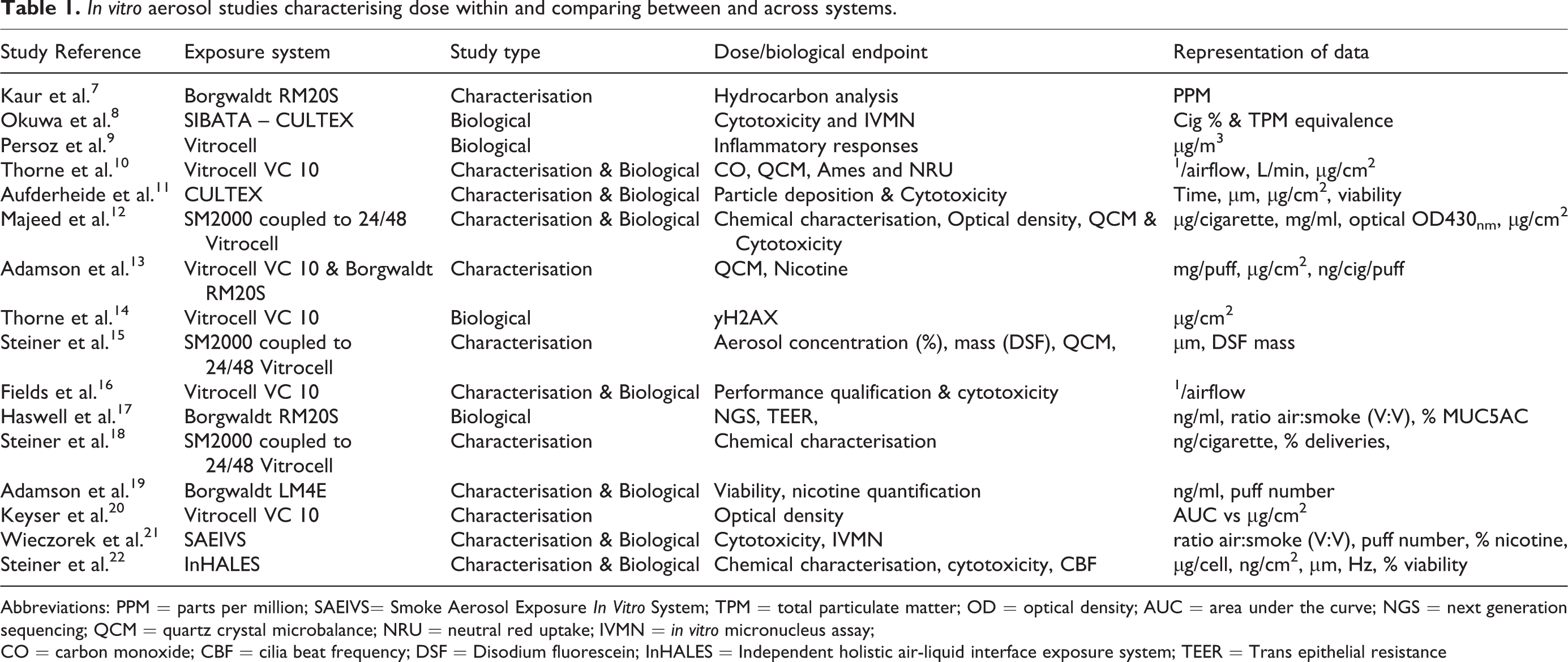
Abbreviations: PPM = parts per million; SAEIVS= Smoke Aerosol Exposure In Vitro System; TPM = total particulate matter; OD = optical density; AUC = area under the curve; NGS = next generation sequencing; QCM = quartz crystal microbalance; NRU = neutral red uptake; IVMN = in vitro micronucleus assay;
CO = carbon monoxide; CBF = cilia beat frequency; DSF = Disodium fluorescein; InHALES = Independent holistic air-liquid interface exposure system; TEER = Trans epithelial resistance
The Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) was founded in 1956 with the purpose to promote international cooperation in tobacco scientific research. Currently, CORESTA is represented by over 158 member organisations in more than 37 countries (https://www.coresta.org/). Scientific work within CORESTA is governed by a Board through the charge of a scientific commission, that orients and coordinates the scientific work. The scientific work is split into four major tobacco related study groups: 1) Agronomy & Leaf Integrity, 2) Phytopathology & Genetics, 3) Smoke Science, and 4) Product Technology. The In Vitro Toxicity Testing Sub-Group (IVT SG), resides within the Smoke Science study group. Its remit is the scientific study of emissions from, and exposure to tobacco related products, which includes the development of specific chemical and biological methods and investigation of means to assess exposure and use (https://www.coresta.org/). The goal of CORESTA is to promote alignment in technologies and/or cooperation in the development of techniques. The assessment of current exposure technologies relevant to in vitro analysis, which includes aerosol exposure technologies, resides within the IVT SG’s remit.
During a 2016 CORESTA meeting, the IVT SG met to discuss the developing field of aerosol exposure research and to assess its current status, with potential harmonisation in mind. Given the current aerosol exposure environment and the diversity of techniques and combination of exposure parameters and biological endpoints being deployed, it was considered a high priority to establish a strategy to assess these systems and contextualise the responses obtained. The aim of the meeting was to establish an approach to accurately survey the environment and to use the information to help direct harmonisation approaches. The working hypothesis is that these systems and their application are too nuanced to appropriately compare and to harmonise. The rationale for this hypothesis is that a previous reported but unpublished CORESTA whole aerosol study, demonstrated that although generating similar cytotoxicity data, there was no direct read-across between studies (https://www.coresta.org/sites/default/files/technical_documents/main/IVT_TF_Report_Smoke_Air_Liquid_Interface.pdf).
Twelve global companies with expertise in in vitro aerosol research met to discuss this topic and formulate an aligned approach. A detailed and comprehensive survey was conducted on over 40 parameters ranging from aerosol generation, dilution, biological methodology, data analysis and dosimetry approaches, across eight independent laboratories. The results from the survey are detailed here and it is the consensus of the IVT SG that awareness of exposure systems, parameters, methodology nuances and data analysis may be of substantial benefit to scientists in intersecting fields of research, new scientists and laboratories starting out in this area of research and those already acclimatised to it. This approach not only clarifies the current environment and nuances, but facilitates future discussions and investigations into the following areas: Comparison of aerosol exposure options available and those currently in use across the tobacco and intersecting industries (pharmaceutical aerosol delivery, 3Rs approaches, environmental and tobacco smoke exposure) Possible alignment of exposure and biological parameters Expression of in vitro aerosol data Comparison of in vitro data and responses across systems Comparison of in vitro data within the same systems across multiple exposure parameters The use of dosimetry techniques to facilitate exposure, systems and data comparisons The complexity and diversity of this environment and future challenges, which include the movement into the in vitro aerosol assessment of next generation tobacco products, including heated tobacco products (HTPs) and e-cigarettes (or otherwise known as Electronic Nicotine Delivery Systems (ENDS)). This topic is in its infancy, as systems are evolving to meet new challenges.
Materials and methods and results
Study design
To start a simple whole aerosol approach (cytotoxicity) method was selected to start the harmonisation discussion and give the best chances of success in developing an aligned approach.
Laboratories within the CORESTA IVT SG consisting of British American Tobacco (BAT), Covance Laboratories Ltd., Imperial Brands (IB), Japan Tobacco (JT), Korean Tobacco & Ginseng Corporation (KT&G), RAI Services Company (RAISC), Lorillard Tobacco (Lorillard, Inc., the parent company of Lorillard Tobacco Co. was acquired by Reynolds American Inc. (RAI) in June, 2015 and certain assets were transferred to ITG Brands, LLC) and Zhengzhou Tobacco Research Institute (ZTRI) were surveyed and supplied data on their independent exposure setups. The survey covered all aspects of the setup including exposure systems, biological parameters, analysis of data and dosimetry tools. Laboratories were encouraged to supply information on Kentucky reference cigarettes (2R4F or 3R4F), using 2-dimensional cytotoxicity approaches (Table 2).
Study design.

NRU = Neutral red uptake assay; MTT = (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction) NS = not supplied.
* = System precursor to The Smoke Aerosol Exposure In Vitro System (SAEIVS). 21
The following criteria were used for data (parameter inclusion) and were based on establishing parameters for a single (and simple) biological endpoint – cytotoxicity. Data on reference cigarettes only (2R4F or 3R4F) Cytotoxicity data on 2D approaches in line with OECD protocols Cell must be compatible with ALI (or equivalent approaches) Data must be generated using ISO smoking conditions Any exposure system and module combination would be considered as CORESTA does not endorse any one system or approach.
A more detailed breakdown of the parameters used, exposure systems and methodologies can be found in later Tables.
Exposure systems
A variety of exposure systems with a diverse range of experimental setups and customisation options were utilised. For a more in-depth understanding of the Vitrocell, Burghart and Borgwaldt, their uses and application in the cigarette smoke testing environment please refer to the following references and reviews. 7,21,23 –29 The exposure environment is evolving, new exposure systems are being developed and systems being adapted, one such adaptation is the BT020 used in this study in Laboratory 7. Since the survey, this system has been upgraded and published as the Smoke Aerosol Exposure In Vitro System (SAEIVS), as described by Wieczorek et al. 21
A breakdown of the experimental setups and exposure parameters is shown in Table 3.
Exposure system parameters.
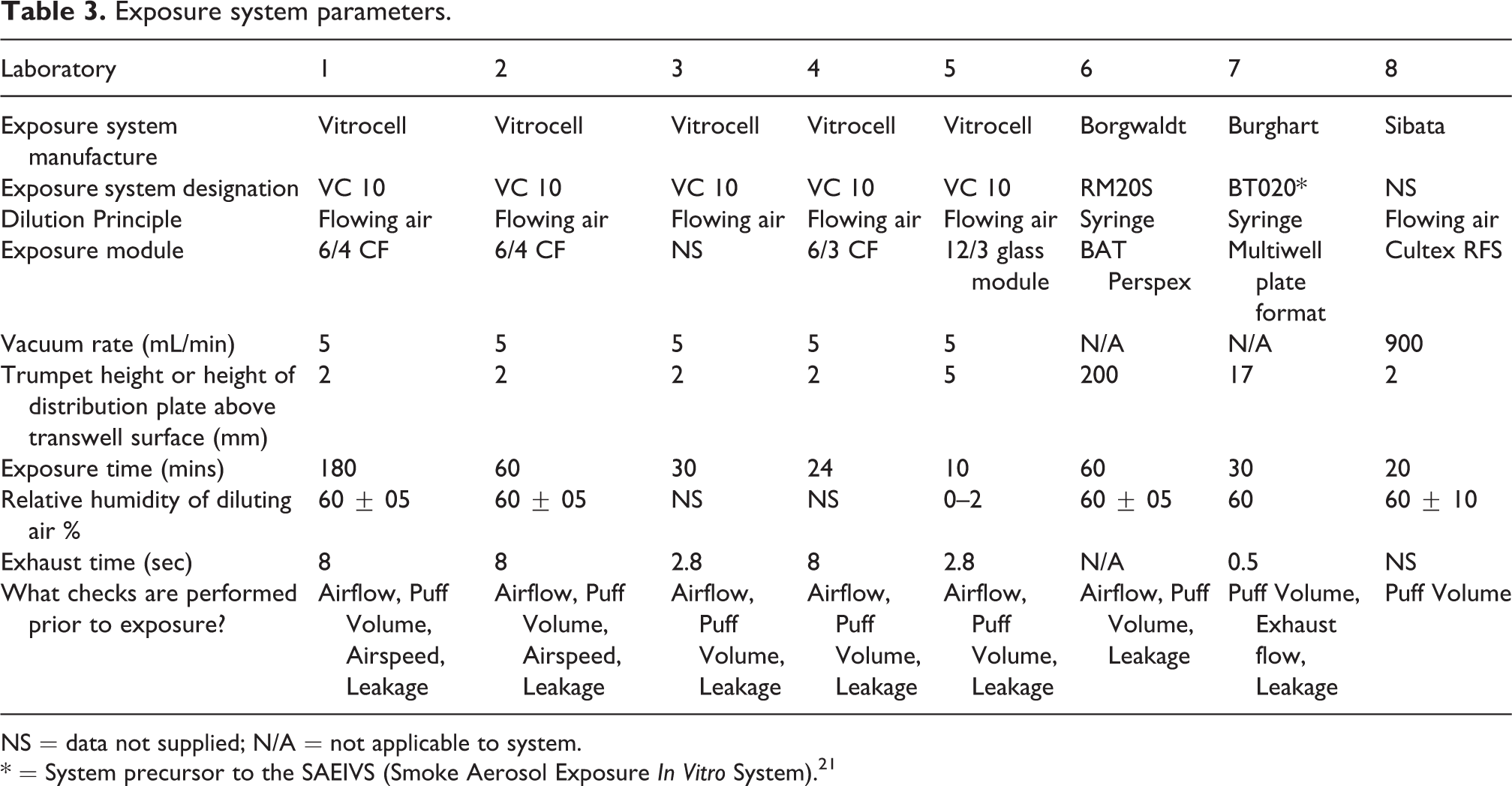
NS = data not supplied; N/A = not applicable to system.
* = System precursor to the SAEIVS (Smoke Aerosol Exposure In Vitro System). 21
Cell lines and culture conditions
Each laboratory has developed its own in vitro aerosol cytotoxicity assay with unique parameters and cell lines. The rationale for the choice of cell lines was not discussed. Rather, it was considered advantageous that multiple different cell lines have been deployed alongside aerosol exposure systems. The only stipulation was that cells are compatible with an air liquid interface (ALI) exposure or a multi-well plate exposure format. In developing and optimising exposure conditions, the following cell lines were used across the IVT SG for cytotoxicity assessments, BALB/c 3T3, CHO, CHO-K1, A549, BEAS-2B, NCI-H292 and HepG2. Cells were obtained from respective cell banks and established culture stocks and were routinely checked/ karyotyped. For those laboratories that have published work on their cell line for use with whole aerosol exposure, the details of the culture conditions can be found in the following references. 16,30 –34 A full breakdown of cell types, exposure conditions, media and culture conditions can be found in Table 4.
Culture conditions.
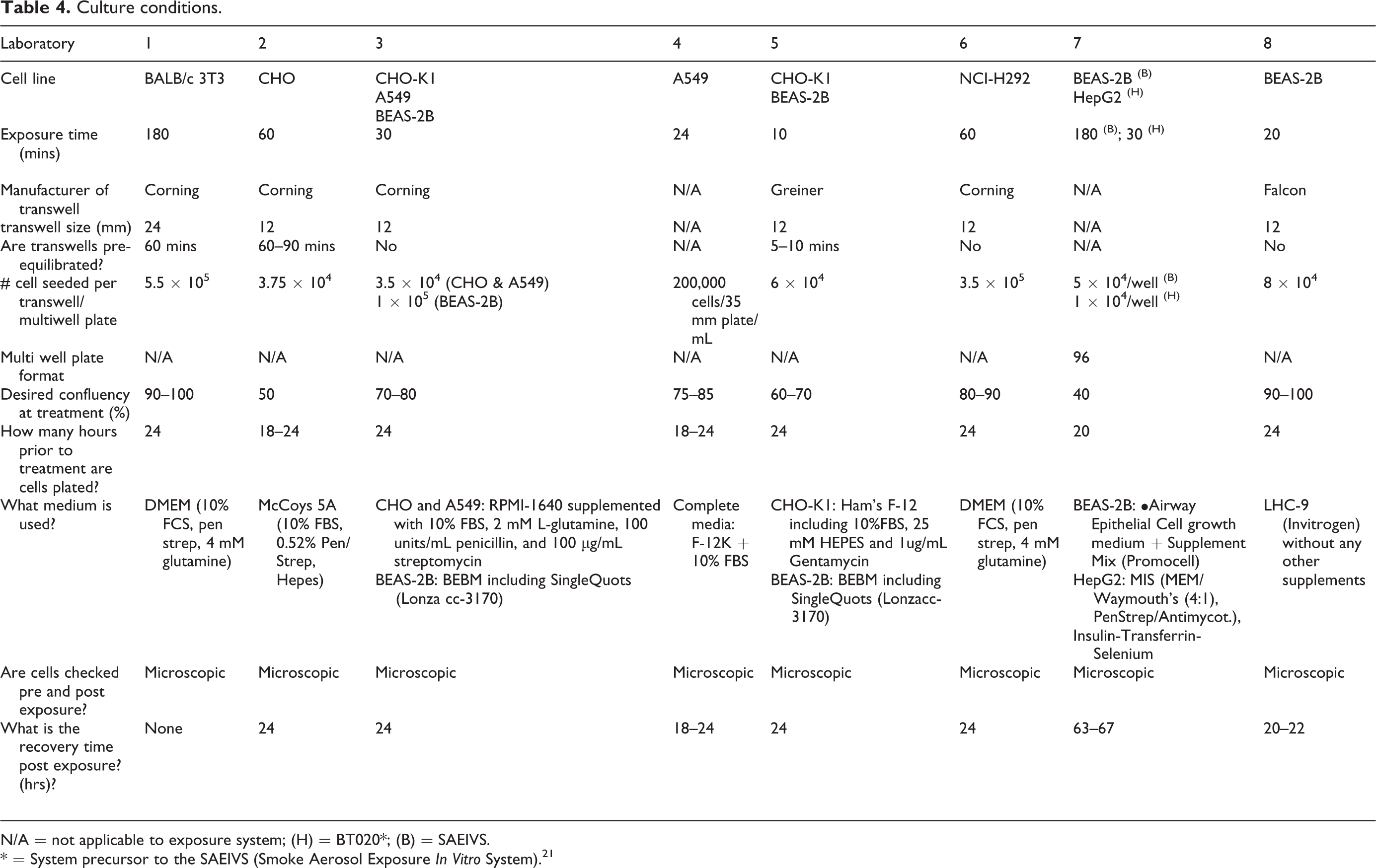
N/A = not applicable to exposure system; (H) = BT020*; (B) = SAEIVS.
* = System precursor to the SAEIVS (Smoke Aerosol Exposure In Vitro System). 21
Cytotoxicity assay conditions and aerosol exposure
Either the neutral red uptake assay (NRU) or MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction) assay was employed by all laboratories for the determination of cytotoxicity. The conditions of each assay and exposure can be found in Table 5.
NRU exposure conditions.
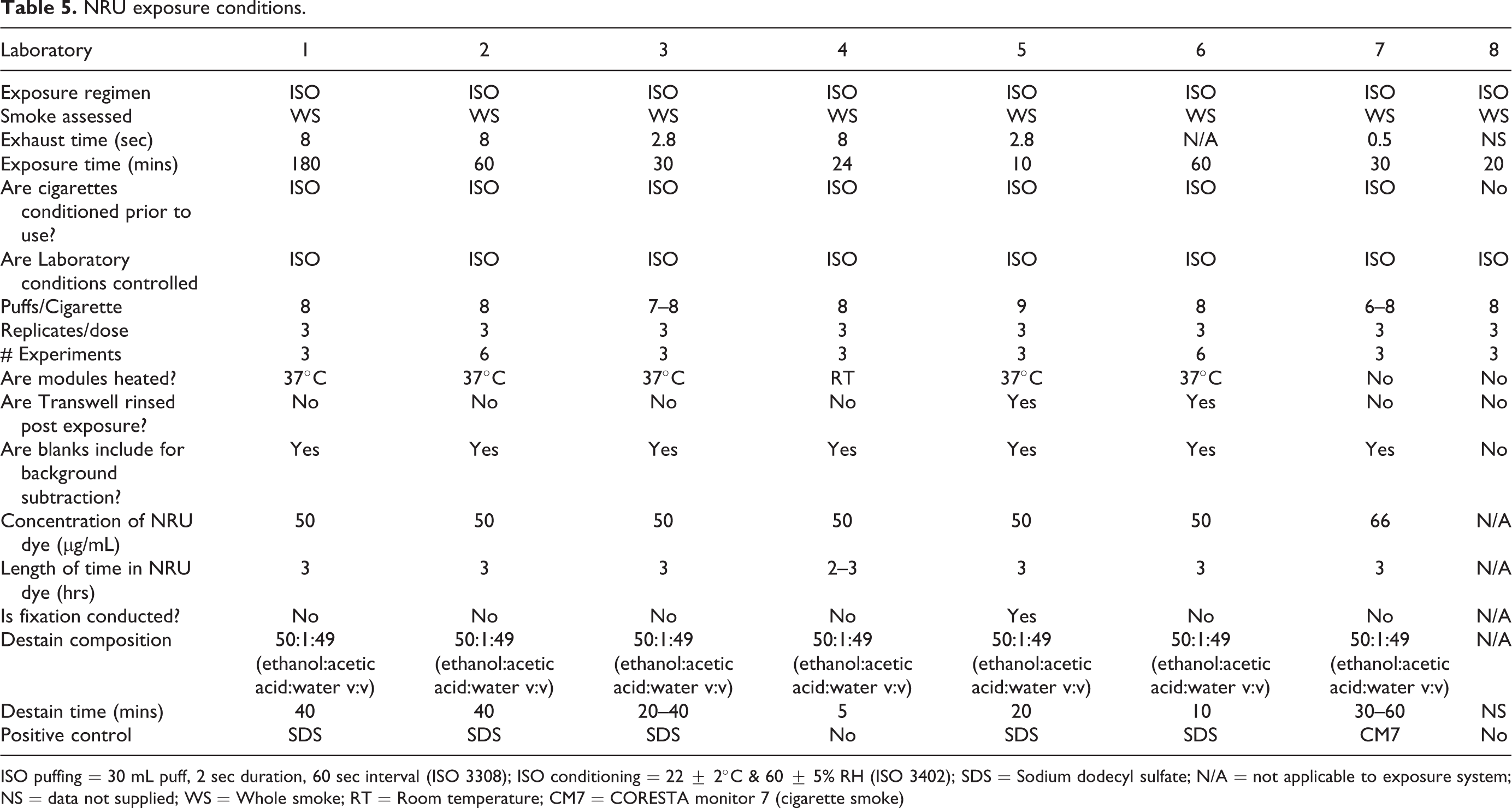
ISO puffing = 30 mL puff, 2 sec duration, 60 sec interval (ISO 3308); ISO conditioning = 22 ± 2°C & 60 ± 5% RH (ISO 3402); SDS = Sodium dodecyl sulfate; N/A = not applicable to exposure system; NS = data not supplied; WS = Whole smoke; RT = Room temperature; CM7 = CORESTA monitor 7 (cigarette smoke)
Dosimetry
Few dosimetry techniques were discussed during the meeting because the application of these approaches was not consistent in terms of how they are being used to support data analyses. A workshop hosted by the Institute for In Vitro Sciences was held (4–6 April 2016, Bethesda, Maryland, USA) met to discuss this topic and to elucidate how dosimetry methods are being applied to whole aerosol exposure systems. 24 The outcomes of this workshop paralleled the discussions here. These activities all support the identification, validation, and dissemination of robust in vitro methods for the evaluation of tobacco products and their constituents. 24 Several clear distinctions came out of discussions, for example, irrespective of dose method, tools were used in the following ways by either all, some or none of the laboratories; 1) as a means to characterise the exposure system prior to experiments; 2) to calibrate the dilution system; 3) an in-line QC method to determine dose delivery and consistency of dose; 4) as a method interpret/analyse the data.
It was clear from discussions that more work and alignment of these techniques was required before any harmonised approach could be discussed, or before these methods could be employed in any significant manner in a future proficiency/ring-trial. In terms of techniques used, the following were discussed as methods applied to ascertain dose in line with the approaches above; carbon monoxide quantification, quartz crystal microbalances, nicotine quantification and nicotine dose matching, optical tar measurements, hydrocarbon analysis and TPM quantification. The approaches discussed were entirely consistent with those outlined in Table 1. Additionally, the positioning of these quantification tools within the exposure system varied for each study. The location of sampling can have a significant impact on the levels of captured materials, as they transit through the exposure system. This creates an additional problem in alignment of sampling location as well as methodology used to sample and what it’s being used to measure. Finally, using these tools to provide a measure of dose, is a very simplistic means of calculating what the actual biological material receives, and must be appropriately caveated. At the exposure interface, there are a number of aerosol and particle interactions and parameters that can affect deposition and the composition of what is actually interacting with the cell surface. 35 In order to accurately model biological dose within these systems, appropriate aerosol dynamics and interactions must be considered. Commonly, in vitro aerosol exposures are characterised, by the composition and/or concentration of the aerosol generated, but not by the delivered aerosol fraction. 18 To calculate aerosol delivery to the exposure interface, requires understanding of the partitioning of its constituents between the aerosol phases, and various coefficients for the gaseous compounds, which in most cases are unknown or poorly understood, 18 leaving simplistic dose tools the only practical option.
To increase the complexity a variety of methods were identified for the presentation of the data. For example, data was presented as a function of dilution (depending on the unique exposure setup), function of dose (nicotine, TPM, Mass), on a puff-by-puff basis or even on a per-cigarette basis and as a percentage of delivered smoke. The diversity of how data is presented is captured in Table 1. In order to better compare between studies and across laboratories, a more consistent method to present the data is required. Often this requires upfront consideration as the dose tools used to help normalise the data, are measurements captured during the experimental phase. Therefore, any joint interlaboratory approach needs to not only lock down the exposure and biological system, but the dosimetry (tool, location and sampling parameters) methodology too.
Discussion
This is the first study to comprehensively survey over 40 parameters from aerosol generation, dilution, biological methodology, data analysis and dosimetry approaches, across eight independent laboratories, using a cytotoxicity endpoint.
The first major observation from the survey, is that irrespective of exposure system, all laboratories qualify their system prior to experimentation. Measurements of airflow, leak tests and puff volumes are standard across all laboratories. This ensures that the system is both well maintained and calibrated prior to in vitro experimentation and should be considered a minimum and ‘best practice’ for in vitro aerosol exposures. The second observation is that the majority of laboratories represented within the IVT SG are using a Vitrocell based exposure system, coupled with a Vitrocell exposure module. Upon initial inspection, this does grant some alignment opportunities and many of the confounding cross-laboratory challenges are instantly removed. However, the real challenge is to ensure that all systems and data can be compared regardless of setup. This is especially important for laboratories utilising different commercially available setups and laboratories that are using bespoke systems not even discussed here, or even hybrid systems coupled together from multiple sources still wanting to participate in inter-laboratory activities. For those using the Vitrocell setup, some common place parameters came out of this survey, for example, the trumpet height. The trumpet within the Vitrocell module is set above the cell surface and for most, the distance is a consistent 2 mm. Altering the height of the trumpet will affect aerosol delivery and diffusion interactions, thus creating a potentially different result. An outcome from this survey should be that a standard 2 mm trumpet height should be employed for laboratories starting out, unless otherwise investigated and assessed. In addition to the trumpet height the exhaust time is set from the outcome of the puffing regimen, and here we have seen a consistent eight seconds for most laboratories. However, few laboratories using the Vitrocell system have reported a shorter 2.8 second exhaust time. Such variables will need investigating to examine the effect of exhaust airflow on the resulting data. For those laboratories using alternative systems, the story is somewhat more complex, as commonalities are harder to define. As smoking machines are made up of three functional parts (aerosol generator, dilution and transit and dose delivery to a cell system), the most obvious place to start to align studies using different systems is the smoking head, where products are puffed. Here alignment could be as simple as an agreed set of pre-tests, as described above or an established smoking regimen, for which several international standards exist. 36 –38 However, it is believed that the data between studies can be compared given using a consistent measure of dose and expressing the data as a function of that dose. Several recent studies have demonstrated this concept successfully, by bridging data across studies and systems. 19,39 This concept is especially important in the world of next generation tobacco products, such as heated tobacco products and e-cigarettes (ENDS), due to their increasing popularity. Exposure systems are diversifying even further from the ones investigated here, so establishing a strategy to compare between studies and systems is even more important.
Although not discussed, the choice of cell type is also driven by a unique set of selection criteria established by each laboratory. The survey demonstrates that cell choice was the most diverse variable investigated. From the eight laboratories, eight different cell lines were identified, some overlap existed between cell types and laboratories, but often even within laboratories, multiple cell types are used depending on the question being investigated. BEAS-2Bs were used by four of the eight laboratories and represented the most consistent cell line, followed by CHO variants, used in three of the eight laboratories. Finally, the remaining labs were split between A549s, BALB/C, HepG2 and NCI-H292. This observation creates some difficulties in establishing a common set of conditions. Clearly, there is not one cell line choice running through the survey that fits all laboratories, and shifting cell lines is not readily feasible, as laboratories have optimised for the choice of cell line over years of testing. Changing cell line would invalidate all the historical data that each laboratory may have. Any strategy for comparing across independent studies must therefore factor in the variability of cell line choice and its impact on results, both in variability and the outcome. A recent study 39 examined just this concept, and demonstrated that different continuous cell lines BALB/C and H292 gave the same response in cytotoxicity on different whole aerosol exposure systems when normalised for delivered dose (mass µg/cm2 and nicotine µg/ml). The study further demonstrated that two different 3D cell systems (MucilAirTM and EpiAirwayTM) gave the similar results when normalised to mass and nicotine (mass µg/cm2 and nicotine µg/ml) for a cytotoxicity endpoint. However, the results between the 2D and 3D test systems were significantly different. This study potentially demonstrates, on a simplistic endpoint such as cytotoxicity, the choice of cell line becomes less relevant, but what appears more important is the structure of the cell type (monolayer vs. differentiated). As complexity in studies increase, the choice of cell line will become a far more important variable. These results reassure this survey in that the difference between the surveyed monolayers may not be as wide as originally anticipated. Only following up with biological analysis on a ‘harmonised’ approach will the response of cell type on biological variability be known. Despite cell differences across the laboratories, cell culture practices highlighted some key commonalities between laboratories. For example, the majority of the laboratories conducted exposures on a confluency of 80–100% irrespective of cell type. Cells were seeded on the transwell between 18–24 hours prior to exposures. Most laboratories conducted a cytotoxicity measure following a 24 hour recovery and all laboratories microscopically examined the transwell/plate following aerosol exposure. Such parameters might be aligned due to most labs following an establish and well tested OECD protocol. 40 Using such protocols like OECD and adapting for whole aerosol exposure represents the easiest way for alignment and potential harmonisation. This approach also gives the best opportunity for validation of these systems. However, complications arise when methods are relatively new and no such protocol exist.
In addition to the already observed commonalities, the majority of laboratories conditioned the cigarettes prior to use in accordance with an ISO standard. This standard approach was maintained in the laboratory where conditions were controlled in accordance with the same ISO standard. Cigarettes were smoked according to a prescribed set of conditions, also established as an ISO standard. 38 Interestingly though, the exposure modules were not always heated to 37°C. Presumably, as exposures are short, heating the module was deemed less important to the overall outcome of the study, compared to other parameters. In an inter-machine comparison study, the authors compared exposure systems through their delivered dose. They observed that machines in conditioned laboratories gave a more consistent, less variable dose compared to those in an unconditioned laboratory, although the overall results between systems were comparable. 41
Finally, although some commonalities exist between the various setups examined in this survey when assessing tobacco smoke, it remains largely unclear whether these parameters will remain consistent when new nicotine and tobacco products, such as heated tobacco products and e-cigarettes (ENDS) are assessed. One approach would be to harmonise systems and methodologies for tobacco smoke from combustible products and utilise this methodology with prescribed changes for the assessment of HTPs and e-cigs (ENDS), thus maximising the harmonisation. However, this remains only theoretical until such an approach is explored.
This study focused on surveying the current in vitro whole aerosol exposure environment to assess potential areas of commonalities with a view of creating, if possible, a harmonised protocol. Although at this stage a harmonised protocol seems unlikely, clearly there are some areas of commonality that can be incorporated into a next stage proficiency trial.
We have only explored how we conduct our exposures, and the nuances associated with aerosol exposure. Next steps must include an experimental phase with aligned dosimetry approaches so we can better map the data against dose and in a more coordinated testing approach. This becomes especially important for exploring new technologies such as heated tobacco products and e-cigarettes (ENDS). Such a testing strategy must include these new technologies at the fore-front of discussions. But as many of the techniques used to assess these products are still being developed/refined, starting with simple cigarette approaches seems a feasible and practical place to start. Additionally, cytotoxicity is only one biological assay and other whole aerosol techniques are established. Such whole aerosol approaches include but are not limited to, Ames, IVMN, NGS, DNA damage (Comet, yH2AX), cytokine analysis and functional endpoint analysis such as cilia beat frequency, active area and transepithelial resistance. 14,17,30,42 –45 A follow-up study could also survey the endpoints being used and investigate areas of potential commonality here. Finally, it is important to note that this survey is not endorsing any one in vitro exposure platform, and that all systems have their advantages and disadvantages as detailed in various reviews. 25 –27 More importantly, it is fundamental to be able to compare across systems and to use the diverging systems to better understand the environment and to elucidate new disease mechanisms. Rather than consolidating under one in vitro exposure platform, it is essential to ensure that each system is appropriately characterised in terms of their chemical deliveries. Ultimately it doesn’t matter how the aerosol is delivered or by what system, rather understanding what is being delivered and ultimately dosed is key.
In terms of next steps, the following proposals from this work can be made. First step is to conduct a preliminary proficiency trial across exposure systems with as many parameters locked down across exposure and the biological assay as possible, utilising a consistent measure of dose, such as nicotine normalisation. Locked parameters would include, consistent cleaning and maintenance procedures, with puff volume and leak tests conducted prior to assessments. A consistent biological assay (NRU) and cell culture parameters. Locked parameters around the exposure system depends on the system being used. For example, if using a Vitrocell based system, trumpet height, exposure time and aerosol dilutions can be all be aligned based on this survey. If using alternative systems, it becomes a little more challenging to align, due to system differences and this is where dosimetry approaches will be key to align exposure rather than individual system parameters. However, such information will give valuable evidence on how to bridge between exposure systems moving forward, which is of paramount importance as the environment is diverse in exposure system options and permutations. Complete cell line alignment across all laboratories may not occur within the initial proficiency trial; however, valuable information would be obtained on the parameters that are driving (or not driving) increased laboratory and/or system variability. The next step would be to investigate the responses obtained from the preliminary proficiency trail and amend with a refined protocol. For example, a protocol harmonised around cell lines to see if variability (if observed) could be reduced. Step 3 would be to investigate other biological approaches, which include additional genotoxicological methodologies (Ames) and more sophisticated 3D cultures and disease mechanistic approaches. Any alignment approach must be step-wise and considered based on the historical evidence and the increasing complexity of study design.
Conclusions
The survey results emphasise the diversity of in vitro exposure parameters and methodologies employed across the IVT SG. Pockets of harmonisation already exist. For example, many of the biological protocol parameters are consistent across the Sub-Group. However, variables such as cell type and exposure time remain largely inconsistent. The key next steps for this work will be to map parameters and system data against biological findings and investigate whether the observed commonalities and inconsistencies translate into increased biological variability or vice versa. Analysing the data will give a better understanding of how data are presented and interpreted and how data may be more accurately aligned between laboratories irrespective of the lack of harmonised protocols. In order to understand the environment in its completeness, other biological endpoints and parameters must also be considered. This information will allow the Sub-Group to rationalise experimental techniques and find areas of consensus within protocols, with an ultimate goal of harmonisation if possible or applicable.
Such approaches as detailed here will be invaluable in determining in vitro whole smoke ‘best practices’ and ensuring that a consistent cross-industry approach is considered and applied, to meet increasing global regulation and the evolution of testing approaches for next generation tobacco and nicotine products.
Footnotes
Abbreviations
Definitions
Air-liquid interface – the interface at which cells are exposed on the apical surface to aerosol while being maintained basally by appropriate media.
Control air-flow – the airflow rate that the control cells are exposed to. For exposure systems that have multiple dilution steps, diluting airflow (L/min) and vacuum rate (mL/min/well), this should be the flow over the exposure interface (mL/min).
Dilution system – the part of the system that creates the required smoke dilution and delivers it to the exposure module/chamber, where cells are housed.
Dilution principle – the principle in which dilution occurs within an aerosol exposure system, this is normally through two main routes. 1) diluting airflow, which creates a turbulent aerosol mixture and 2) Syringe, where the dilution occurs within a syringe and is step-wise and sequential.
Distribution plate – a plate attached to the module or exposure system that facilitates efficient aerosol delivery to the exposure interface.
Dosimetry – The approach of measuring dose within an exposure system to either characterise exposure, the exposure system delivery or amount the cell is exposed to.
Exhaust time – the time required for the puff of an aerosol to be exhausted from the syringe (or piston) to the dilution system or exposure module.
Exposure interface – the cellular interface where the exposing aerosol interacts with the cell surface.
Exposure module – can also be known as exposure chamber. The module in which cell cultures are housed. This can take many forms and traditionally has been limited to air-liquid interface-based technology, where cells are supported on permeable membranes. However, with customisation and the requirement for high throughput, this now include standard multi-well plates, which ‘dock’ into an exposure system.
Trumpet – a device specific to the CULTEX® and Vitrocell® exposure systems, that sits within the exposure module and facilitates efficient aerosol delivery to the exposure interface.
Piston/syringe – the part of the exposure system that creates the puff. This can also be part of the dilution system in some cases.
Smoking head – the part of the exposure system that puffs the cigarette.
Vacuum rate – the negative pressure applied via a vacuum pump to an exposure system to actively sub sample the aerosol from the dilution system through the exposure module (mL/min/well).
Whole smoke – constitutes the cigarette smoke aerosol generated by the in vitro exposure system, consisting of both a vapour and particulate phase interacting.
Authors’ note
Betsy Bombick has subsequently retired from RAI.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Thorne and Marianna Gaca are employed by British American Tobacco (BAT). Roman Wieczorek is employed by Imperial Brands. Robert Leverette and Betsy Bombick are (were) employed by Reynolds American Inc (RAI). RAI is a wholly owned subsidiary of BAT. Mark Ballantyne is employed by Covance Laboratories. Toshiro Fukushima and Kei Yoshino are employed by Japan Tobacco. Xiang Li is employed by Zhengzhou Tobacco Research Institute and Han-Jae Shin is employed by the Korean Tobacco & Ginseng Corporation. The work was facilitated by CORESTA. Each laboratory independently funded their respective work and contributions. BAT collated the data and drafted the manuscript supported by all authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
