Abstract
Contract Research Organizations (CROs) conducting toxicology studies on behalf of biopharmaceutical sponsors and others routinely provide supportive care for animals to minimize pain and distress on studies. A large number of guidance documents govern the care of experimental animals, however there is currently no uniform approach on the communication between sponsor and their CRO partners in providing a standard definition of and strategies for administering supportive care in toxicity studies. This survey was conducted by the CRO Outreach Working Group (WG), a part of the 3Rs Translational and Predictive Sciences (TPS) Leadership Group of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ Consortium), to better understand the provision of supportive care on nonclinical studies. The survey aimed to define supportive care strategies, identify alternatives to supportive care, and understand regulatory feedback and implications about supportive care decisions. The survey was distributed to members of the 3Rs Leadership Group of the IQ Consortium and several CRO partners, representing 35 organizations as potential respondents. The results of the survey from 13 respondents provided positive feedback that helped in highlighting the existing best practices for supportive care. Areas of enhancements identified included greater consistency in the inclusion of sponsor veterinarians on project teams for externalized studies, the timing of initiation of supportive care, and increased sharing of regulatory outcomes. Suggested best practices include creating a plan of action for veterinary care prior to study start, and enhancing information sharing regarding expected toxicities from previous study findings. Improved communication regarding supportive care will pave the way for enhanced 3Rs initiatives, refining the existing animal care paradigm and helping to ensure the most ethical toxicology study designs.
Introduction
Ever since the publication of The Principles of Humane Experimental Technique in 1959, the 3Rs of replacement, refinement and reduction in animal usage have been widely adopted as guiding principles across biopharmaceutical industries for humane animal usage in translational research. 1 A major opportunity for refinement in toxicology studies is the assessment of pain and distress in animals and the provision of supportive care. 2,3
Supportive care is routinely provided to animals to minimize pain and distress on toxicology studies that are conducted both internally at biopharmaceutical companies and at Contract Research Organizations (CROs) on behalf of biopharmaceutical sponsors. Supportive care strategies align with the 3Rs and aim to improve animal welfare, and ultimately positively impact data quality. 4
Supportive care strategies can range from simple environmental modifications like changes in husbandry practices to administration of systemic drugs that can significantly impact the research outcome of toxicology studies. 5 Type, duration, and use of supportive care practices vary widely across study types, depending on the severity of clinical signs and evidence of pain and distress exhibited by the study animals. The practices of providing supportive care may vary widely across industry due to a lack of uniform definition and strategies, variability in communication and differences in sponsor preferences. Variability in supportive care decisions among sponsors and between CROs can also be further influenced by species of animals used (rodent vs. non-rodent), type of study (regulated vs. early non-regulated studies). However, the impact of these factors on supportive care strategies and decisions and their wider impact on regulatory submissions largely remains to be delineated. The 3Rs TPS Leadership Group of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ Consortium) created a CRO Outreach Working Group (WG) in 2013. One of the 2018 goals of this WG was to conduct an industry wide survey to better understand the current supportive care strategies that are used across CROs and the biopharmaceutical industry.
Multiple national and international regulatory guidelines clearly define the basic design and conduct of toxicology studies, and there is ample regulatory and academic discourse governing and advising on the compassionate use of animals in biomedical research. 6 –8 A large number of guidance documents govern the care of experimental animals, however there is a lack of literature addressing this important and regulated area of research and currently there are no uniform approaches or guidance’s that can provide insights on how supportive care is defined, used and decisions of providing or opting out of supportive care are communicated between sponsor and their CRO partners. In addition, regulatory feedback and queries associated with supportive care administration are widely undisclosed and may lead to withholding of supportive care measures that can impact the quality of data that are generated. 9 The current survey helps to bridge the two by defining and providing strategies for administering supportive care in toxicity studies.
Survey goals included describing the scope of information sharing between biopharmaceutical sponsors and CRO partners regarding the decision process for provision of supportive care, delineating and defining supportive care strategies, identifying alternative options, and understanding regulatory feedback and implications of supportive care decisions. Information generated from the survey will characterize and collate current best practices across industry, identify gaps and propose best practices that would enhance communication and impact better and more ethical toxicology study design.
Materials and methods
The survey was submitted to biopharmaceutical member companies of the IQ Consortium and Contract Research Organizations (CRO) participating in the IQ 3Rs TPS Leadership Group. The survey invitations were emailed to 35 participating organizations, with responses requested between June 8, 2018 and July 20, 2018. Participation was voluntary and participants were asked to limit the response to one per organization to limit repetition. Several roles are involved in animal testing in preclinical settings in Bio-pharmaceutical and CROs hence survey responses were generally provided by a team of subject matter experts from the participating organizations that may have included veterinarians, study directors, study monitors and regulatory scientists. These individuals were selected by the company management in discussion with the IQ liaison, DruSafe and/or 3R scientists. Based on the diverse expertise, working knowledge and company background responses help to gather informative insights of industry wide practices where uniform practices are not followed. Respondent answers for the survey should not be considered as “individual opinions” as they reflect the actual practices that are provided as supportive care to animals in each individual company and have been carefully deliberated and discussed before submission. An independent third-party secretariat (Drinker, Biddle, & Reath LLP) administered the survey and provided confidential, de-identified responses to the authors.
To define supportive care for the survey, authors proposed the standard supportive care practices that are routinely used on toxicity studies, included in study protocols or a part of the standard operating procedures or internal/external guidelines. These supportive care options were provided as a scaffold to build the final definition of supportive care with additional free text options to add more practices from respondents based on their study experiences. The objectives of the survey questions were to determine how organizations defined “supportive care,” delineate current practices of supportive care, determine who is involved in decision making regarding the administration of supportive care treatments, understand the impact of supportive care treatments on regulatory submissions, and identify gaps present in information sharing and decision making around administration of supportive care.
The intent of the survey was to understand the CRO-sponsor specific issues with respect to supportive care. Since respondents answered questions specifically to CRO conducted studies, assumptions can be made that all sponsors have responded with their experience of toxicology studies at CROs and not internally conducted toxicology studies. The survey was intended for routinely used toxicology animals that includes USDA (dogs, swine, non-human primates and rabbits) and non-USDA (rats and mice) regulated species; however, specific questions on the type of animal species used or referred to while responding was not asked.
Many of the questions sought insights regarding the relationship, sharing of information, and communication between sponsors and CROs. Other questions related to the policies and practices surrounding supportive care administration, types of treatments that are considered to be supportive care, who is involved in the decision-making process regarding the administration of supportive care, and regulatory feedback received after the administration of supportive care on studies.
A link to the survey was sent via SurveyMonkey [https://www.surveymonkey.com/] to all participating organizations. The survey consisted of 26 questions, with subsets of questions that allowed for collection of additional case-specific details (see supplemental data). Additional subsets of questions were directed to biopharmaceutical sponsors or CROs. Survey questions encompassed both regulated and non-regulated early animal studies that were used in regulatory submissions. Fifteen questions offered multiple options with instructions to choose all responses that applied, as well as a free text box to list additional responses if needed to provide further details or to present case studies. The option for responses as text/comments and the inclusion of “other” categories limited data interpretation for some questions. Three questions were open ended, requesting the respondent to provide a case study or other free text answer with the intention of gaining information specific to regulatory feedback received or unique treatment strategies that led to a successful regulatory submission.
Data obtained was de-identified and grouped in three major categories: characterization of supportive care, communication between sponsor and CRO, and impact of supportive care on regulatory submission. Data analysis was based on the number of positive responses to each question, calculated as the number of responses divided by the total number of respondents for each individual question. When appropriate, data was further stratified by type of respondent (biopharmaceutical company vs. CRO).
Results and discussion
Demographics
The survey was distributed to members of the 3Rs TPS Leadership Group of the IQ Consortium and to several CRO partners, representing 35 organizations as potential respondents. Thirteen organizations responded; however for the majority of questions, 2 respondents did not respond, and the number of total respondents is 11 in these cases. Respondents included eight pharmaceutical companies, two biotechnology companies, one small CRO and two large CROs.
Specific demographic and geographic details of the survey respondents were not collected, since the IQ Consortium members who responded to the survey have representatives from pharmaceutical, and biotechnology industry and have wide representations from USA, Europe, and Asia. The authors acknowledge the drawbacks of the survey in terms of low number of respondents, and that most of the questions were sponsor-oriented. However, each response is from organizations (Pharmaceutical or CRO) with tens to hundreds of scientists engaged in toxicology studies, representing a significant proportion of potential respondents. Industry wide surveys with robust participation are difficult to conduct due to the differences in geographical, regulatory, ethical and company based working standards that are non-uniform throughout. Despite the moderate sample size, the survey responses provide substantial data with useful insights of industry-wide trends for supportive care that have largely remained unknown. Additional surveys could be conducted in the future to complete gaps identified by the current survey.
Definition of supportive care
Supportive care that reduces pain and distress in study animals can positively impact animal health and support the generation of high-quality study data. 10 Respondents were asked to define supportive care and identify both general categories of treatment and specific treatments that constitute supportive care at their respective institutions.
When asked to define supportive care practices, respondents were consistent in selecting broad definitions, encompassing both palliative and etiology-based treatment strategies. Most (90%, n = 10) of 11 total respondents defined supportive care as both “Interventions used to improve animal comfort or general wellbeing (palliative care) without treating a specific disease,” and as “Interventions used to mitigate clinical issues and reduce pain and distress.” Eight (72%) of 11 total respondents also included “Interventions used to treat an animal with a specific clinical issue with known etiology.” Additionally, one respondent stated that these interventions are administered on a case by case basis. Responses between sponsor and CROs were very similar.
Types of supportive care
Supportive care may include a variety of pharmaceutical and non-pharmaceutical approaches. 11 Responses were collected to characterize the major categories of treatments that constitute supportive care. 11 of 11 total respondents (Figure 1) considered diet alterations and fluids to be supportive care. 90. % (n = 10) of 11 total respondents considered environmental modification to be supportive care, and 81% (n = 9) of 11 total respondents considered systemic drugs, and topical drugs or veterinary treatment to be supportive care. Additionally, 27% (n = 3) of 11 total respondents considered alternative therapies (e.g. low-level laser treatment) to be supportive care. Most free text responses to questions about definitions and types of supportive care emphasized that decisions are made on a case-by-case basis, with consideration of study impact. Responses between sponsor and CROs were very similar. Further questions delineated the characterization of each specific major category of supportive care.
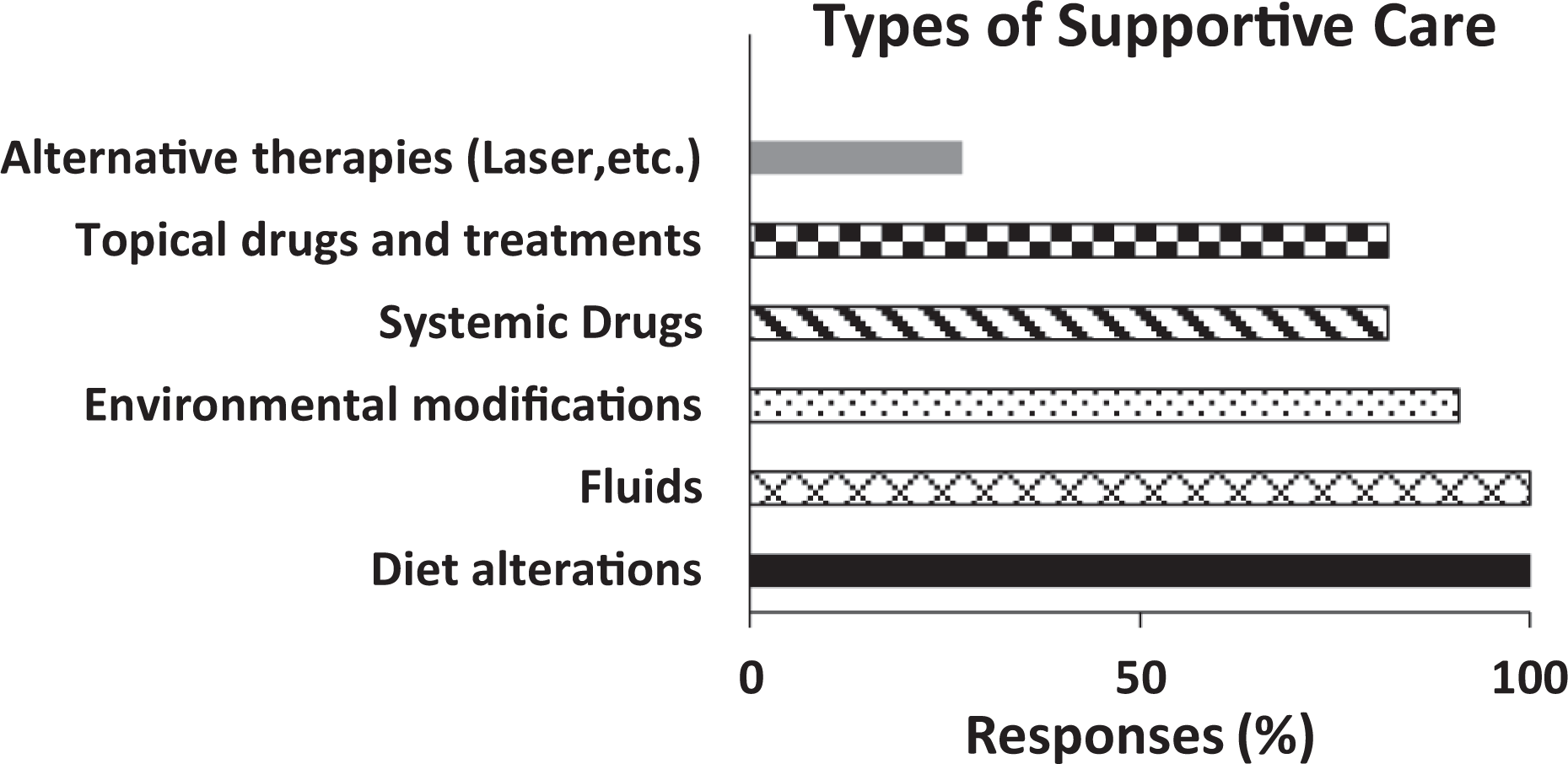
Histogram depicts the treatment modalities considered to be supportive care by respondents. Y axis depicts each reasoning category and X-axis depicts the % is calculated from number of respondents divided by the total number of respondents (n = 11).
Diet alterations
When respondents were asked about specific types of diet alteration they considered as supportive care, all selected one or more modality, consistent with the results from the previous question. Out of 11 total respondents, 100% (n = 11) considered diet gels to be supportive care, 90% (n = 10) considered moistened food or food enrichment, 81% (n = 9) considered palatable diet, and 72% (n = 8) considered hand feeding as supportive care. Interestingly, 45% (n = 5) of respondents used gavage feeding as an atypical method of providing supportive care. One respondent stated that they provide novel enrichments such as frozen items, yogurt and items fit for human consumption to increase palatability for animals. Another respondent stated that the amount of time with access to food was increased for animals to allow access beyond maximum concentration of test item (Cmax) when the resulting test item induced nausea or anorexia in the animals.
Dietary enrichment can positively influence the overall health and wellbeing of study animals and potentially affect the quality of data generated by toxicology studies. 12 Care should be taken while providing food enrichment to ensure it will not interfere with test article related mechanisms and does not mask any test article related changes on body weight or food consumption. Most toxicology studies utilize a standardized diet, specific to the requirements of the species, hence nutritional value and content of each ingredient used for food enrichment should be carefully evaluated to minimize any risk for diet alterations on study endpoints causing dietary imbalances. 13,14 However, dietary modification is a widely accepted supportive care practice used by sponsor organizations and CROs. Most dietary modifications stimulate an animal’s inherent appetite through provision of more appealing diet. This dependence on voluntary consumption may account for the widespread acceptance of this form of supportive care. Gavage feeding is unique in that it bypasses the animal’s desire to eat, however it may allow the detection of toxicities beyond impacts that cause reductions in voluntary food consumption. Supportive care like gavage feeding may include restraint and may result in additional stress; hence careful consideration needs to be given before its implementation.
Environmental modifications
Environmental modification considered to be supportive care by most respondents (of 11 total respondents) included thermal support via supplemental heat (90%, n = 10), supplemental bedding (63%, n = 7), and increased enrichment (90%, n = 10). 27% (n = 3) of respondents reported that an altered light cycle was also considered as supportive care. 45% (n = 5) listed additional forms of environmental modification including restricting access to a portion of the animal’s enclosure, other modifications to the enclosure, changing bedding type, increased rotation of non-edible enrichment items, or altering husbandry, highlighting the diversity of opportunity for supporting study animals within this category.
In contrast with most other forms of supportive care, environmental modifications provide support externally, which may alleviate concerns of interaction with the test article or direct physiologic impacts. These environmental modifications have the ability to positively impact animal health outcomes and support welfare strategies while simultaneously posing a low to negligible risk of confounding study related data or causing data variability. 15
Fluids
90% (n = 10) considered hydrogels and oral rehydration solutions as supportive care, 27% (n = 3) of 11 total respondents considered intraperitoneal fluids and intravenous fluids as supportive care and a single respondent included an alternate water source, such as a bowl or bottle instead of a lixit, as supportive care. All respondents (n = 11 of 11 total) considered subcutaneous fluids as supportive care.
Decreased water intake, and increased fluid loss through emesis, diarrhea, urination, or evaporative cooling can all negatively impact the hydration status of animals on toxicology studies, which can significantly impact the animal’s physiology and welfare. Fluid therapy is supportive but does not correct underlying disease processes. 16 Less invasive methods of fluid administration (oral rehydration solutions, subcutaneous fluids) were the more frequently used methods for providing supportive care than the less frequently used more invasive methods (intravenous, intraperitoneal). This may be due to the more invasive methods requiring additional resources and monitoring and having a marked impact on the animal’s physiology, or because situations in which animals require these more invasive treatments may also constitute humane endpoints. Administration of supplemental fluids helps to maintain animal health without adversely affecting data interpretation. 17
Systemic drugs
There was considerable variability in the perception of systemic drugs as supportive care for study animals. However, all respondents considered at least one type of systemic drug to constitute supportive care when asked about specific types of drugs. This contrasts with previous responses in which only 81% of respondents said that systemic drugs constituted supportive care. 90% (n = 10) of 11 total respondents considered analgesics as supportive care, with one respondent (n = 1 of 11) also considering the use of non-steroidal anti-inflammatory drugs. The next most common categories of drugs considered to be supportive care were gastroprotectants and antibiotics, with 81% (n = 9) of 11 total respondents included in these categories.
With the increase in biologics testing wherein hypersensitivity reactions may be noted, a majority of respondents (63%, n = 7 of 11) considered prophylactic use of antihistamines (e.g. diphenhydramine) to be supportive care when preventing or reducing known hypersensitivity reactions observed in previous dose administration. 18 Since preclinical hypersensitivity has poor concordance with clinical studies, 19 such prophylactic administration can help to obtain a better understanding of the test article mechanisms and toxicity. Pre-approvals of prophylactic treatments are generally done on a case by case basis and can highly vary between sponsors. A minority of respondents (of 11 total respondents) considered steroids and anticonvulsants (9%, n = 1, 27%, n = 3 respectively) as supportive care. Several respondents additionally commented that treatment with systemic drugs was conditional based on a lack of study impact and prior knowledge of test article characteristics, study design consideration and scientific endpoints being collected. In general, there is more widespread acceptability of drugs that are designed to alleviate pain without masking test article effects in comparison to systemic therapeutics that may interfere in data interpretation.
Topical drugs and treatments
When questioned on whether topical drugs and treatments constituted supportive care, 90% (n = 10) of 11 total respondents affirmed topical antibiotics as supportive care, with the majority also considering topical steroids (54%, n = 6), topical emollients (72%, n = 8), and bandaging (81%, n = 9) as supportive care. More than half of respondents (45%, n = 5) consider therapeutic shampoo as supportive care, and two respondents wrote that minor surgical procedures, such as tail amputation and tooth trimming were considered as supportive care.
Topical treatments pose a low risk of interfering with test article provided systemic exposure to the topical treatments are limited and they do not exacerbate potential test article effects or impact of the test article distribution or metabolism.
Other alternative therapies
Pain and distress in preclinical animals are assumed comparable to humans, 20 however unlike humans who are verbal, pain assessment in preclinical animals are more difficult to assess and efforts should be made to reduce them.
Respondents were offered the opportunity to write in alternative treatment modalities they would employ, identifying several alternative therapies such as behavioral assessment, therapeutic laser, massage therapy and heat. One respondent stated that these therapies “may be used on occasion in place of systemic medications for pain relief.” Like other forms of supportive care, alternative therapies are generally used in a study-dependent manner. Use of alternative therapies highlights the range of options that can be employed to positively impact the health and welfare of animals on toxicology studies.
Provision and withholding of supportive care
Animal toxicity in preclinical studies accounts for 20% of drug attrition. 21 Prolonged duration of study, and the impacts of housing, husbandry, dosing, and sample collection, combined with test article toxicity can require the need to provide supportive care to animals. 22 Since provision of supportive care can impact study outcomes, further responses were collected to understand scenarios where supportive care measures are provided to animals on regulated studies following Good Laboratory Practices (GLP) (Figure 2).
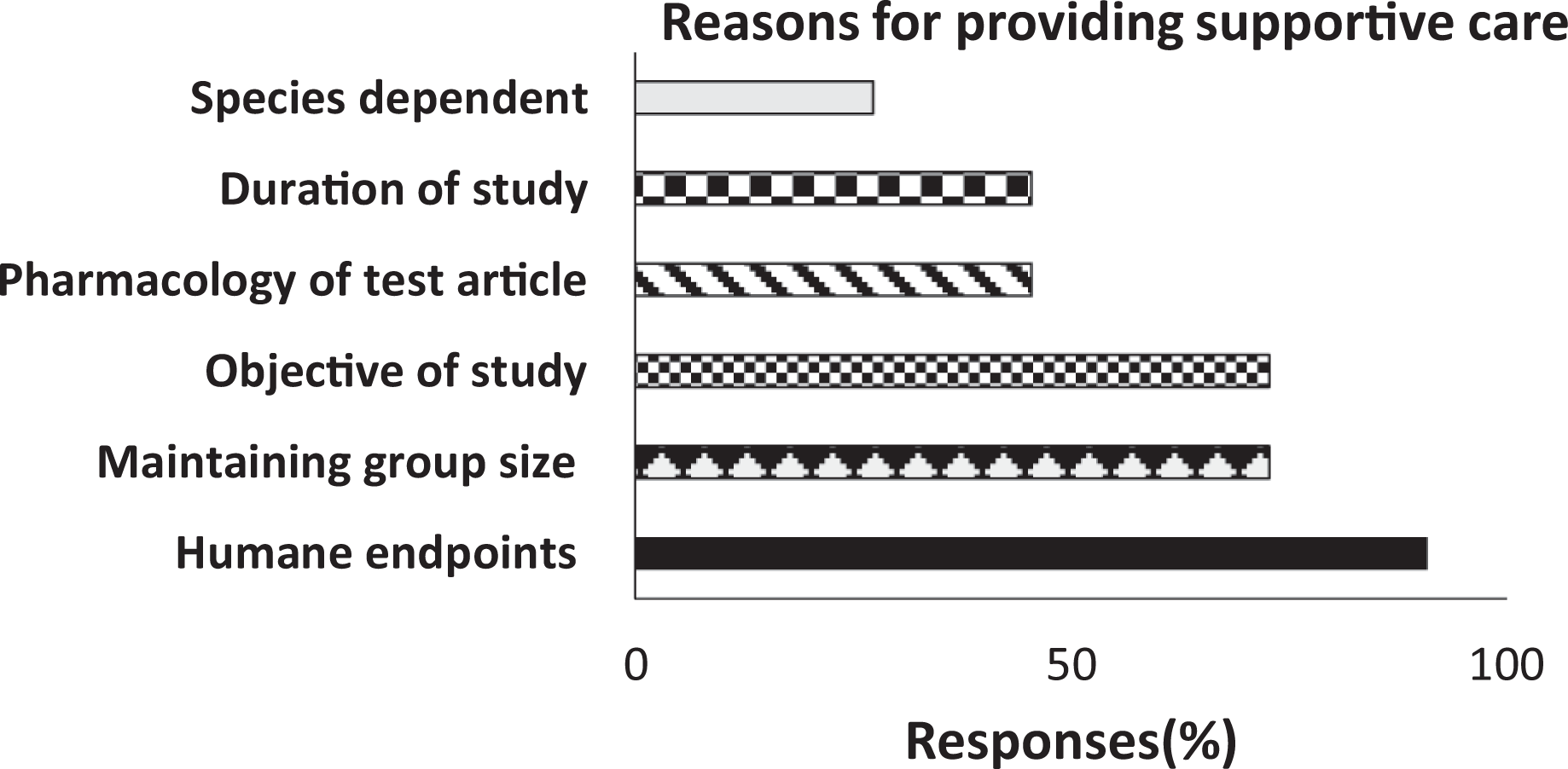
Histogram shows the main reason when supportive care is provided by the CROs to animals on toxicology studies. Y axis depicts each reasoning category and X-axis depicts the % is calculated from number of respondents divided by the total number of respondents (n = 11).
Regulatory agencies have guidance for veterinary interventions as part of adequate veterinary care for animals on study where humane endpoints and scientific objective of the study need to be considered. 23 Notably, 90% (n = 10) of 11 total respondents cited humane considerations as the primary driver for providing supportive care. Other significant factors in providing supportive care were study objectives and the need to ensure that enough animals remain on the study to obtain interpretive data. Respondents also indicated that supportive care provision was dependent on pharmacology of test article (hypoglycemia or convulsions) and duration of study (45%, n = 5 of 11) or species dependent (27%, n = 3 of 11).
Observed clinical signs are instrumental in assessing animal welfare. 24 As such, severity of clinical signs and perception of pain and distress correlate to the respondent’s likelihood of providing supportive care. Respondents were questioned on when supportive care measures are implemented, based on the degree of clinical abnormality present and to what degree those signs indicated pain or distress. None of the respondents employed supportive measures for mild clinical signs (mild weight loss, no change in body condition score). 36% (n = 4 of 11) used supportive care when moderate clinical signs (moderate weight loss, mildly decreased body conditioning score, no additional clinical signs) were present, indicating no to minimal pain or distress. All (n = 11) respondents employed supportive measures for studies with significant clinical signs (marked weight loss, decreased body conditioning score, diarrhea, dehydration, vocalization on abdominal palpation) and at least moderate pain or distress when the primary driver for supportive care was based on humane considerations. 54% (n = 6 of 11) employed supportive care when severe clinical signs (severe weight loss, respiratory distress and labored respiration apart from moderate clinical signs listed earlier), pain, and distress indicating moribundity or nearing death were observed.
Based on the authors experiences, severe clinical signs, pain, and distress in any preclinical study animals warrant immediate veterinary intervention and, rather than focusing on the provision of long-term supportive care, decisions are made to relieve the pain and distress due to humane reasons and elective euthanasia (non-scheduled/early euthanasia) is generally opted. Additionally, information was gathered in the survey to determine supportive care decisions that were species dependent. 64% (n = 7) of 11 total respondents cited that certain species (USDA covered species, canines, and non-human primates) are more readily provided supportive care on GLP studies.
The survey provides overview of reasons for withholding supportive care measure in animals on regulated and non-regulated studies (Figure 3). Withholding supportive care does not account for withholding enrichments as these are essential and required for welfare purposes and provided to all animals routinely. Decision to withhold supportive care could be influenced by humane endpoints, scientific or regulatory aspects. The survey respondents (11 total) indicated that potential interference of supportive care with test article pharmacology (81%, n = 9) and masking of test article toxicity (72%, n = 8) are the main reasons for withholding supportive care measures. In early toxicity studies, where the basic intent is to identify tolerable doses with the minimum number of animals, supportive care administration can significantly impact data interpretation. Other reasons cited by a minority of respondents included clinical signs not warranting treatment from a welfare perspective (45%, n = 5 of 11), lack of appropriate supportive care options (27%, n = 3 of 11), and concerns due to previous or potential regulatory feedback (27%, n = 3 and 18%, n = 2, respectively).

Histogram shows the main rationales for withholding supportive care for animals in toxicology studies as indicated in the survey. Y axis depicts each reasoning category and X-axis depicts the % is calculated from number of respondents divided by the total number of respondents (n = 11).
Elective euthanasia of animals, rather than providing supportive care, was entered by a single respondent as a free text response, indicating the variability in decision making around supportive care. 25 Notably, none of the respondents cited resources, or need to maintain a certain group size for interpretation as reasons to withhold supportive care. Greater information sharing on regulatory feedback on supportive care could enable institutions to make better quality decisions surrounding supportive care.
Decisions for providing or withholding supportive care are largely dependent on effective communication between the sponsors and their CRO partners. While the Attending Veterinarian of a CRO, or their delegate, has ultimate authority to ensure that an animal receives appropriate veterinary care, CRO study directors along with sponsor representatives (including sponsor veterinarians) are actively engaged in these decisions with case-by-case involvement of other team members from CRO and Sponsors. Effective communication for medical management of animals on study is key to minimizing pain and distress and providing quality scientific data. 8,26
Information sharing between sponsors and CRO partners
As more industry wide focus is shifted toward 3R strategies, information sharing between sponsors and CRO partners is critical. 27 This is particularly true with regard to decision making for supportive care administration in toxicology studies that directly impacts the timing of initiation of supportive care.
All respondents (n = 13 total respondents) at biotechnology companies (n = 2 of 2) felt that adequate information about the compound is shared and majority of respondents at sponsor companies (87%, n = 7 of 8) indicated that either complete or partial/relevant information about the compound is shared. Notably, all CROs that responded (n = 2 of 3) and one biopharmaceutical company indicated that adequate information is not shared between the sponsor and CRO to allow for optimal veterinary decision-making. Both CROs provided free text answers stating that this is largely sponsor-dependent. The survey further identified that information sharing between sponsors and CRO partners regarding potential on-target toxicities and clinical signs for any given study was found to be inconsistent and was identified as one of the gaps in communication that warrants further discussion.
Reasons for inconsistent information sharing
The reasoning behind partial or lack of information sharing from biopharmaceutical companies is captured in Figure 4. A major reason for this inconsistency in information sharing was identified to be a lack of details/information by sponsor representatives regarding the test item (50%, n = 2 of 4 total respondents), this could be largely dependent on the stage of drug development as for early studies limited information would be available for the sponsor themselves. Other inconsistency included concerns over intellectual property (25%, n = 1 of 4 total respondents) and assurance required that information sharing would not adversely impact clinical sign observations in order to assure the study remains blinded (50%, n = 2 of 4 total respondents).
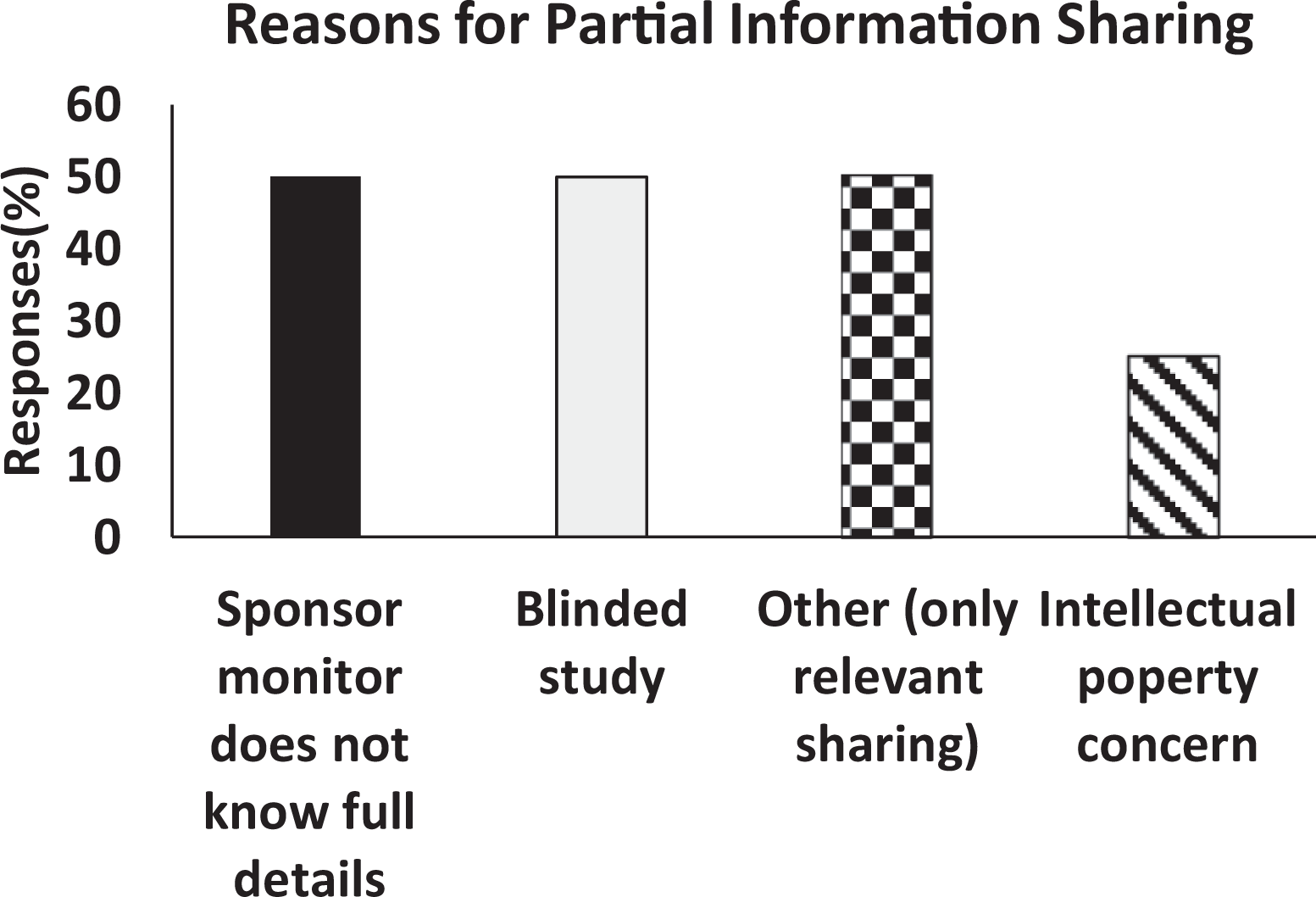
Histogram shows the main reasons of partial information sharing between the CROs and their sponsor partners. X-axis depicts each reasoning category and Y-axis depicts the % is calculated from the number of respondents for that choice divided by the total number of respondents (n = 4).
Preconceived bias against some clinical signs can be introduced with prior information and can impact data interpretation. 28 Gaps identified through free text responses included issues with sponsor transparency, inconsistencies between research groups within a sponsor, late communication to CRO veterinary staff versus the study director, and little alignment across sponsors. These gaps can critically impact supportive care decision making and can potentially impact animal welfare.
Ideal information sharing
The ideal information sharing between sponsor and CRO partner, from a sponsor’s perspective, would help to promote optimal veterinary care, animal welfare, and the 3Rs. Interestingly, two (of three CROs) CRO respondents affirmed sharing of information on target biology, expected toxicity, previous study findings, clinical information, and justification for numbers and study design as part of an ideal relationship. In contrast, all sponsor respondents universally selected sharing of information on expected toxicity and previous study findings (n = 7 of 7 total sponsor respondents) as part of an ideal relationship.
Gosselin et al. and Rovira et al. have both highlighted that for pathology reports the best quality data can only be generated through such effective information sharing. 29,30 Based on the survey findings, the authors suggest sharing expected toxicity findings, any previous study findings with information about target biology, scientific justification for animal number or study design justification, followed by improved routine communication between sponsors and CROs would be considered ideal information sharing between the sponsor and the CRO partner. For toxicity studies that occur during or after phase I or II, additional information about clinical phase outcomes or clinical signs noted would also help for better quality for supportive care decision making.
Two CROs (out of two total CRO respondents) indicated that an ideal sponsor-CRO relationship includes information sharing in all categories, while just 1 of 2 suggested routine communication between the sponsor and veterinary staff. In contrast, 71% (n = 5) of 7 total sponsor respondents said that routine communication between the sponsor and veterinary staff is part of an ideal relationship. The differences in “ideal” relationship between CRO and sponsor respondents are very interesting, with the sponsor respondents only selecting two categories of information that are uniformly considered to be part of an ideal relationship, while CROs felt that information sharing for almost all categories was the ideal. This discrepancy in data sharing perspective between sponsor and CROs can largely arise due to sponsors concern on effective data blinding to prevent bias in interpretations, intellectual property concerns or lack of a need to communicate previous findings (in other species) or any clinical data if available to the CROs. CROs on the other hand can mitigate sponsor-based variations and benefit with an early and consistent information sharing including regulatory feedback sharing from sponsors. The current survey data suggests that sponsors have opportunity to provide more types of information to CROs, and that increased communication and higher inclusion of the sponsors, sponsor veterinary and CRO veterinary staff may benefit externalized studies.
Gaps identified and scope for alignments
Sharing of humane endpoint guidelines and other animal care and welfare standards between sponsor and CRO was also found to be highly variable. 72% (n = 8) of 11 total respondents felt this to be sponsor and CRO dependent, including all the CRO respondents. Respondents indicated that CROs more commonly share their internal guidelines whereas sponsors do not, identifying a potential gap in expectations for animal care and welfare. Additionally, this lack of internal guidelines from a sponsor could be potentially due to lack of in-house veterinarians or vivarium. 42% (n = 3) of 7 total responding sponsors also indicated that sponsor laboratory animal veterinarians are not routinely included as consultants to the project team for external animal studies. Most of the sponsors (71%, n = 5) of 7 total responding sponsors indicated that sponsor veterinarians are primarily engaged in responding to internal animal care and welfare questions and clinical or adverse events as they arise, highlighting an opportunity for earlier contribution by the sponsor veterinarian.
Seven out of eleven respondents (63%) stated that they either provide or use an approved formulary as a tool for improved communication. A veterinary treatment plan was identified as one tool that some CROs use to align with sponsors on anticipated clinical signs and acceptable treatment regimens prior to study initiation. All survey respondents (100%, n = 11 of 11 total respondents) had some form of a veterinary care plan prior to study initiation, with 15% (n = 2 of 11 total respondents) only using standard generic text in the study plan. However, 36% (n = 4) of 11 total respondents include a study-specific veterinary plan in the study plan, and 27% (n = 3 of 11 total respondents) employ a separate veterinary care plan which is not included as a part of the study plan. Write-in responses cite variation between sponsors, and the use of situationally appropriate plans as the need arises.
Animal care staff, animal technicians performing in-life techniques and veterinarians are key in implementing changes and forming a strategic unit for inculcating the 3Rs “culture of care.” 31 These veterinary treatment plans (included as a part of the study plan/protocol or communicated on a case-by-case basis) are drafted with CRO veterinarians in discussion with the sponsor representative and study director and reflect the variety in composition of individuals who contribute based on the data.
Impact of supportive care on regulatory submissions
Use of animals in toxicology studies is critical for making informed decisions that can impact not only human and animal lives but can also significantly impact the environment (manufacturing waste disposal). All investigational new drug and marketing applications include a critical evaluation of all animal studies that support human safety by regulatory agencies across the world. Though most regulatory testing focuses on the refinement and reduction of animals during such studies, 3 one of the major concerns that sponsors have for providing supportive care is masking of any test article effects and their subsequent implications on regulatory submissions. Most respondents did not have any experience with regulatory feedback (positive or negative) about supportive care or clinical treatment provided to animals on GLP studies. Similar surveys between sponsors and CROs have been conducted for benchmarking social housing of non-rodents for safety pharmacology. 32,33
Several respondents shared unique supportive care strategies that were employed with no impact on regulatory submissions. These cases included gavage feeding as supportive care for appetite loss in dogs (animals had >25% body weight loss with no additional clinical signs), animals pre-dosed with high caloric liquid nutritional supplement (in cases where animals had to be dosed in a fed state) that contributed to good clinical condition of the animals, and treatment with fluids, antidiarrheals, probiotics, and fiber in sub-chronic studies involving gastrointestinal signs.
In a single toxicology case study identified in the survey, negative impact on regulatory submission was noted. In a nonhuman primate study, prophylactic administration of systemic antibiotics was provided to all animals due to a decrease in neutrophils seen in a subset of animals. Regulatory feedback stated that prophylactic use of drugs (such as antibiotics) to prevent occurrence of treatment related adverse outcomes may confound study interpretation and should be given only when animals have clinical signs. Subsequently, the respondent does not provide any prophylactic treatment for potential infection based solely on hematology indicators.
Survey respondents were asked about supportive care options that were perceived to be more compatible with regulatory submission and provided options of dosing holidays, early euthanasia, and topical administration and any additional practices (as free text for others) of supportive care that are used on their preclinical studies. Most of the respondents (60%, n = 6) saw opportunities for adoption of refinements in supportive care and clinical treatment practices for GLP study animals, with respondents providing free text responses focusing on opportunities for increased communication between sponsors, CROs, and regulatory agencies. Multiple respondents cited the need for greater information sharing on regulatory feedback to help standardize supportive care practices across species.
Conclusion
The results of the survey from 13 respondents provided positive feedback that helped in highlighting the existing best practices for supportive care. Apart from collating supportive care practices across the industry, the survey suggested a need to provide adequate information at the drafting stage of study design and several potential methods for further improvement in communication between the study sponsors and CROs (Figure 5). The current manuscript highlights the breadth of supportive care options identified by both CROs and sponsors, the lack of literature addressing this important and regulated area of research and is the first step in sharing information cited by many as a way to improve and standardize supportive care practices. Areas for enhancement for improving supportive care outcomes that can lead to improved information sharing between the sponsors and CROs included greater consistency in the inclusion of sponsor veterinarians on project teams for externalized studies, and a sharing of regulatory feedback outcomes both positive and negative outcomes. Other working groups, including veterinary pathologists, have championed adoption of the 3Rs strategies for maximizing animal values and focusing on the regulatory policies. 34 Suggested best practices include having CRO veterinary staff involved in initial study planning meetings, creating a plan of action for veterinary care prior to study start, enhancing information sharing regarding expected toxicities from previous study findings, and sharing guidelines on humane endpoints and animal welfare standards. Improved communication regarding supportive care will pave the way for enhanced 3Rs initiatives, refining the existing animal care paradigm and helping to ensure the most ethical toxicology study designs.
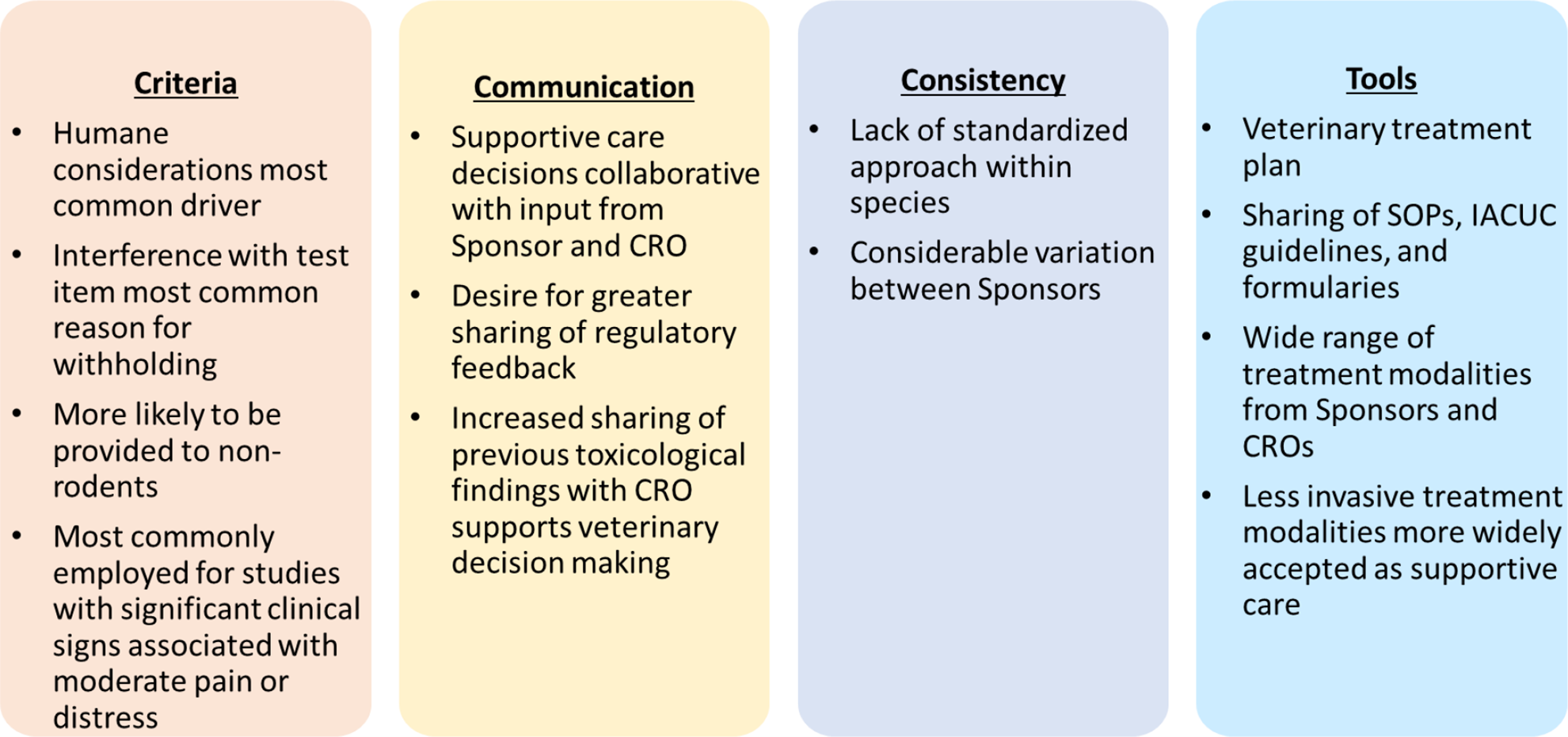
Summarizes the major outcome of the best practices survey on supportive care for preclinical study animals.
Supplemental material
Supplemental Material, sj-pdf-1-tor-10.1177_2397847321999760 - Supportive care for animals on toxicology studies: An industrial best practices survey conducted by the IQ 3Rs TPS CRO Outreach Working Group
Supplemental Material, sj-pdf-1-tor-10.1177_2397847321999760 for Supportive care for animals on toxicology studies: An industrial best practices survey conducted by the IQ 3Rs TPS CRO Outreach Working Group by Smita Salian-Mehta, Jolaine M Wilson, Holly N Burr, Abigail Wolf Greenstein, Kathleen Murray, Wanda West and Nancy Poy in Toxicology Research and Application
Footnotes
Acknowledgments
The authors would like to thank Letty Medina (previously employed at AbbVie Inc), and Amy Kamholz (Covance, WI) for helping in drafting the survey questions. The authors would also thank Alexis Myers from the IQ Consortium for all her help during survey execution and review. The authors extend their heartfelt thanks to IQ 3Rs Translational and Predictive Sciences CRO Outreach Working Group, IQ DruSafe Leadership Group, and all author companies for their help and support for the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This publication was developed with the support of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ, ![]() ). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
