Abstract
Tobacco harm reduction is increasingly recognized as a promising approach to accelerate the decline in smoking prevalence and smoking-related population harm. Potential modified risk tobacco products (MRTPs) must undergo a rigorous premarket toxicological risk assessment. The ability to reproducibly generate, collect, and use aerosols is critical for the characterization, and preclinical assessment of aerosol-based candidate MRTPs (cMRTPs), such as noncombusted cigarettes, also referred to as heated tobacco products, tobacco heating products, or heat-not-burn (HNB) tobacco products. HNB tobacco products generate a nicotine-containing aerosol by heating tobacco instead of burning it. The aerosols generated by HNB products are qualitatively and quantitatively highly different from cigarette smoke (CS). This constitutes technical and experimental challenges comparing the toxicity of HNB aerosols with CS. The methods and experimental setups that have been developed for the study of CS cannot be directly transposed to the study of HNB aerosols. Significant research efforts are dedicated to the development, characterization, and validation of experimental setups and methods suitable for HNB aerosols. They are described in this review, with a particular focus on the Tobacco Heating System version 2.2. This is intended to support further studies, the objective evaluation and verification of existing evidence, and the development of scientifically substantiated HNB MRTPs.
Keywords
Introduction
The efforts to reduce the harm caused by smoking have traditionally been focused on preventing smoking initiation and promoting smoking cessation. 1,2 More recently, tobacco harm reduction has emerged and is increasingly recognized as a promising complementary approach to accelerate the decline in smoking prevalence and hence the smoking-related population harm. 3 Tobacco harm reduction aims at decreasing total morbidity and mortality, without the complete elimination of tobacco and nicotine use, by providing adult smokers who would otherwise not quit with less harmful products delivering levels of nicotine comparable to cigarettes. 4
The Family Smoking Prevention and Tobacco Control Act granted the US Food and Drug Administration (FDA) authority to regulate the manufacturing, distribution, and marketing of tobacco products, including of modified risk tobacco products (MRTPs). An MRTP is defined by the Act as any tobacco product that is sold or distributed for use to reduce harm or the risk of tobacco-related disease associated with commercially marketed tobacco products. 5 The US FDA has provided a draft guidance document outlining a framework to assess novel tobacco and nicotine products as MRTPs. 6 Candidate MRTPs (cMRTPs), as actually used, should (1) significantly reduce harm and the risk of tobacco-related disease to individual tobacco users and (2) benefit the health of the population as a whole, taking into account both the users of tobacco products and people who do not currently use tobacco products. 7
A number of modified and reduced-risk assessment frameworks have been proposed. 8 –11 cMRTP reduced-risk assessment is by definition comparative. cMRTPs provide an alternative to continued cigarette smoking, and as such, the aerosols generated by these products need to be evaluated in comparison with cigarette smoke (CS). The framework proposed by Philip Morris International (PMI) is a seven-step assessment program providing five levels of evidence, which, taken together, form the totality of the evidence necessary to evaluate a cMRTP (Figure 1). 11 Assessing the risk of smoking-related diseases that can take decades to develop and coming to sound scientific conclusions on the risk reduction potential of a cMRTP require the totality of the evidence to be considered rather than isolated studies or endpoints.
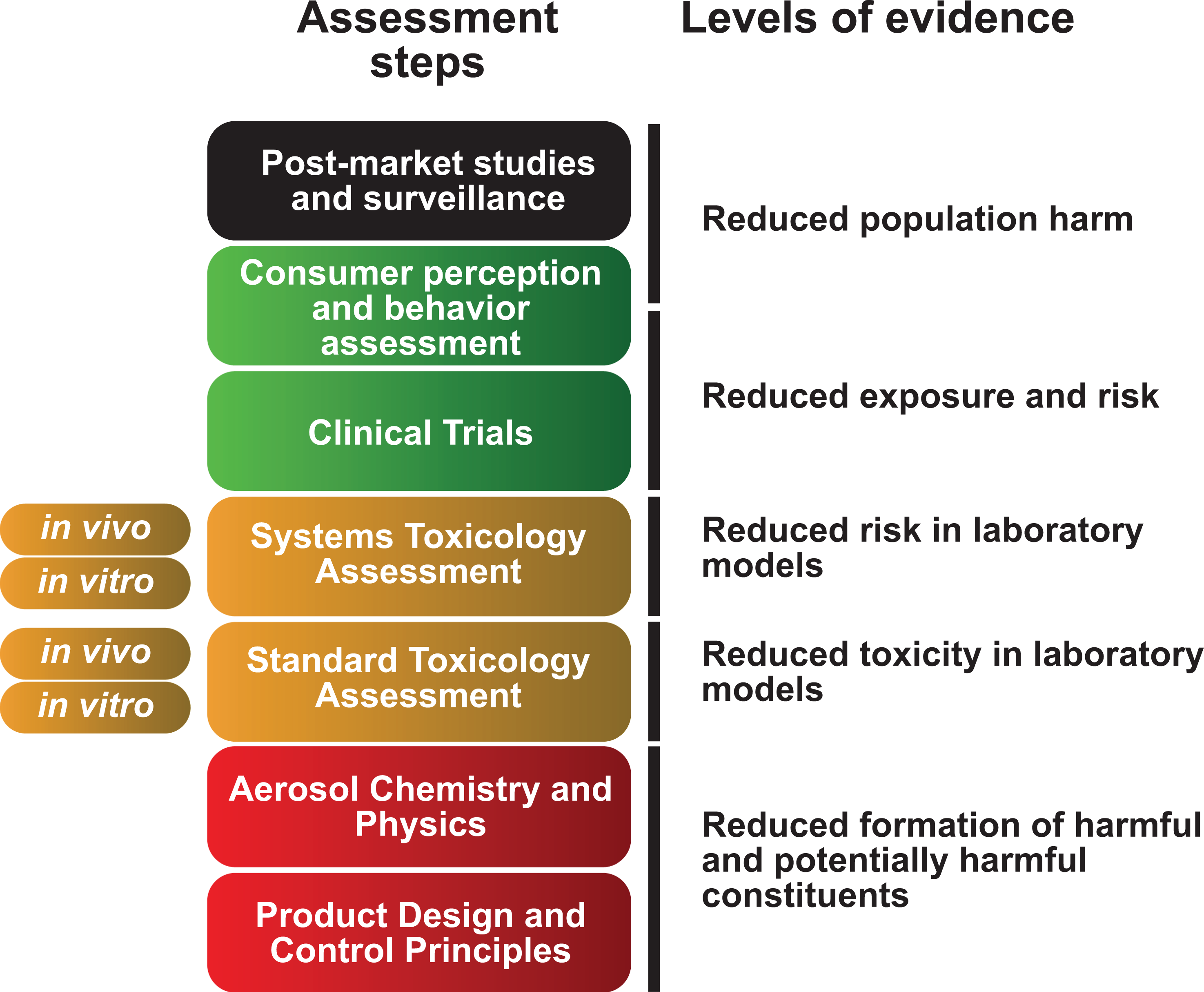
A proposed framework for the toxicological assessment of cMRTPs. cMRTP: candidate modified risk tobacco product.
One category of cMRTPs generate an aerosol by heating tobacco instead of burning it. These products are variably referred to as noncombusted cigarettes, heated tobacco products (HTPs), tobacco heating products (THPs), or heat-not-burn (HNB) tobacco products (thereafter HNB tobacco products). The ability to generate, collect, and use cMRTP aerosols is an essential prerequisite to their in vitro and in vivo assessments, the core of the scientific substantiation of cMRTPs. The aim of studies using cMRTP aerosols is to show that the aerosol (1) delivers significantly fewer and lower amounts of harmful and potentially harmful constituents (HPHCs) than CS and does this consistently throughout the product life cycle, (2) is significantly less toxic than CS in multiple standard in vitro and in vivo toxicology assessments, and (3) shows evidence of reduced risk in in vitro and in vivo systems toxicology studies, while not presenting new hazards.
In this review, we focus on the development, validation, and characterization of methods to generate, collect, and use aerosols from HNB tobacco products for their assessment as cMRTPs. The transparent sharing of protocols, methods, and data is necessary for the objective evaluation and verification of the evidence and will support further studies and the development of scientifically substantiated HNB MRTPs. 12
Aerosol assessment: The challenges
Differences in aerosol formation and characteristics between cigarettes and HNB tobacco products
Aerosols generated by HNB tobacco products are qualitatively and quantitatively different from mainstream CS. Furthermore, unlike cigarettes, which constitute an established, broadly homogeneous product category, HNB tobacco products are a more heterogeneous product category.
This constitutes technical and experimental challenges comparing HNB tobacco products with cigarettes as well as among themselves. It should not be assumed that what is known about mainstream CS generation, dilution, and delivery to in vitro and in vivo exposure systems will apply to HNB aerosols. This review focuses on mainstream CS and HNB aerosol, as, by design, HNB tobacco products do not emit sidestream aerosol. Mainstream smoke/aerosol is the smoke/aerosol released during puffing at the mouth end of the product for inhalation. Sidestream CS is the smoke that is released at the lit end of a cigarette.
Cigarette smoke
CS is a complex and dynamic aerosol consisting of solid and liquid particles, called the particulate phase, suspended in a mixture of vapors and carrier gases, the gas phase (commonly referred to as the gas–vapor phase (GVP)). It is generated by complex and overlapping processes of combustion, pyrolysis, pyrosynthesis, distillation, sublimation, and condensation. 13,14 Because combustion takes place during smoking, a substantial amount of solid particles are released and transferred in the mainstream smoke. A study estimated that approximately 1012 solid particles of median-size diameter smaller than 100 nM were generated in the mainstream smoke of one 3R4F Kentucky reference cigarette over 11 puffs. The solid particles were shown to be composed mainly of carbon-based material and oxygen. 15 CS contains more than 6000 constituents of various chemical classes. 16 Public health authorities have classified approximately 100 of them as causes or potential causes of smoking-related diseases, such as lung cancer, cardiovascular diseases, and chronic obstructive pulmonary diseases. 17,18 Some constituents (e.g. carbon monoxide (CO) and several aldehydes) are found primarily in the GVP, while others, like nicotine, polycyclic aromatic hydrocarbons (PAHs), and tobacco-specific nitrosamines (TSNAs), predominate in the particulate phase. Some constituents (e.g. phenol and the cresols) are partitioned between phases. 16
Freshly generated CS is highly concentrated, with particle number densities of 109–1010 particles/cm3. 19 This high particle number density results in rapid coagulation as the smoke ages. As the number of particles decreases, there is an increase in the average particle diameter.
Aerosols generated by HNB tobacco products
HNB tobacco products generate a nicotine-containing aerosol by heating tobacco instead of burning it.
The dominant constituents in HNB aerosols, including water, glycerin, nicotine, and tobacco flavors, are generated by evaporation and distillation processes.20–22 Combustion is significantly reduced or eliminated, and tobacco pyrolysis is reduced to a minimum, thus reducing the formation of many constituents identified as toxicants. 21,23 –25
The aerosol of a HNB tobacco product, the Tobacco Heating System version 2.2 (THS 2.2), was shown to consist exclusively of liquid droplets formed by a homogeneous nucleation process at the relatively low operating temperature. In contrast, CS contains a large number of solid carbon-based nanoparticles produced during combustion. 15
While cigarettes and HNB tobacco products deliver similar yields of total particulate matter (TPM), its composition is quantitatively and qualitatively different. The TPM of CS contains about 30% water, while the TPM of a HNB tobacco product contains 75% water. Conversely, the TPM generated by an HNB product contains approximately 20% of other constituents (including about 10% glycerol) as compared to >60% in CS TPM (including 5% glycerol). The chemical composition of the GVP of HNB aerosols is also much less complex than that of CS, with 10-times lower abundance of HPHCs. 21,26
Physical and chemical dynamics of aerosols
The physical and chemical behavior of CS and HNB aerosols is highly variable, both temporally and spatially. For this reason, the design and construction of aerosol generation and exposure systems are likely to alter an aerosol’s characteristics (e.g. its particle size distribution (PSD) or the partitioning of its constituents in the solid, liquid, and gas phases). This makes the study of these aerosols a challenging domain. To achieve meaningful results when testing aerosols, a key requirement is to understand and potentially control the processes influencing the aerosol delivery, including aerosol aging, sampling, and deposition in the system. These requirements apply to analytical characterizations as well as to in vitro and in vivo testing.
PSD is a key measure of the physical characteristics of an aerosol, as it is a crucial determinant of its potential for deposition in the respiratory tract, along with the respiratory tract geometry and respiratory physiology. 27 –32 PSD is commonly parametrized by its mass median aerodynamic diameter (MMAD) and geometric standard deviation (GSD), with an assumption of log-normality of the distribution. 33 The MMAD is the aerodynamic diameter for which the particles larger than that diameter constitute one-half of the total mass and particles smaller than that diameter constitute the other half. The GSD describes the spread of the distribution, analogously to how the standard deviation describes the spread in a normal distribution. Studies measuring aerosol particles size for both CS and HNB aerosol showed similar MMADs ranging from 0.6 to 0.9 µm and GSDs ranging from 1.3 to 3.2. 21,34
Particle coalescence (or coagulation), condensation/evaporation, and deposition (or filtration) are the key mechanisms that may profoundly change the nature of a liquid aerosol in experimental settings. 35 –37
Coalescence is the process of clustering and merging of particles because of their collisions, leading to increases in size of newly created particles and to decreases in particle number density due to their merger. It can be linked to the relative motion of the colliding particles, mainly driven by diffusion (Brownian coalescence), but it can also be attributed to convection phenomena (e.g. coalescence caused by turbulence).
Condensation and evaporation describe, respectively, an increase or decrease in particle size/mass occurring due to liquid–gas phase transitions and the movement of molecules at the interface. Evaporation may lead to complete conversion of liquid particles into gas, thus affecting PSD both in terms of particle size and number density.
Primary mechanisms of deposition include gravitational sedimentation, inertial impaction, and diffusion. 27 Sedimentation and impaction are aerodynamic effects that are negligible for very small particles and increase with increasing particle size above 1 µm. Diffusion is negligible for large particles. Deposition of particles occurs all along the flow path of an aerosol exposure system or of the respiratory system. This works as a particle size-dependent filtration mechanism that decreases the particle number density along the system. Lucci et al. used computational fluid dynamics to simulate the transport and deposition of a polydisperse multi-species aerosol in in vitro exposure systems at the air–liquid interface (ALI) described in the “Aerosol exposure systems for in vitro studies: Exposure systems at the ALI” section. 38 Results, verified by comparisons with available experimental data, indicated that, within the recommended operating conditions, aerosol deposition was driven mainly by size-dependent sedimentation and diffusion mechanisms.
The following examples using dilution air illustrate how these mechanisms may interfere with the experimental study of aerosols in vitro and in vivo.
For measurements of PSD, aerosol dilution is often used to diminish particle number density within the operational limits of many types of PSD measurement instruments and may result in erroneous PSD determinations. Dilution has an impact on evaporation (i.e. the phase transition mechanism). This is particularly true for complex liquid mixtures for which the effect will depend on the mixture itself, including the concentrations of various mixture constituents. For example, volatile and semi-volatile constituents, such as flavor compounds (e.g. menthol), can evaporate from the liquid phase to the GVP due to dilution. Caution should therefore be applied when comparing the PSD of undiluted and diluted aerosols.
Dilution is also often used when exposing tissues and animals to CS and HNB aerosols in order to obtain different target concentrations that may trigger effects, preferably in a dynamic range (i.e. low effect with low concentration and higher effect with a higher concentration), while avoiding too many concentrations at no effect, maximal effect, or in saturation.
These processes will be of varying magnitude depending on the flow rate, PSD, and complexity of the aerosol composition. Moreover, diluting an aerosol by adding dilution air also increases the volumetric flow rate and, consequently, the velocity of the diluted aerosol that may trigger additional deposition/filtering of particles along the system. Depending on the PSD, this increase in velocity may change the aerosol sampling efficiency and particle deposition characteristics in the system, 39 as the aerosol travels through regions of different geometric shapes and dimensions under various flow conditions during transportation from the generation site to the exposure setup. During the entire journey, the aerosol experiences a variety of conditions and may undergo dynamic evolution. This is why it is not sufficient to characterize the aerosol initially at the generation site, but it must be characterized after transportation at the exposure site in the in vitro or in vivo aerosol exposure systems.
Reference and test products: Description, sourcing, sampling, and conditioning
3R4F and 1R6F reference cigarettes
HNB tobacco products offer an alternative to continued cigarette smoking, and as such, they should be evaluated compared with cigarettes. To date, the 3R4F reference cigarette is the most frequently used reference in nonclinical HNB studies. Jaccard et al. assessed whether the chemical composition and toxicity of the 3R4F was sufficiently similar to those of commercially available cigarette products worldwide to warrant such use. The study showed that the 3R4F cigarette was sufficiently representative of commercially available cigarettes purchased between 2008 and 2016 in 14 markets worldwide to be a reasonable comparator for the assessment of HNB aerosols in these studies. 40
Reference cigarettes are cigarettes designed to be representative of a certain segment of marketed cigarettes in terms of blend and of tar, nicotine, and CO (TNCO) deliveries. They are designed to have a long shelf life in order to allow the replication and comparison of experiments over several years and across laboratories, which is the reason why they do not include added volatile flavorings. They are produced under normal production tolerances so that their variability is similar to commercial products, and they are made in quantities large enough for the same reference cigarette to be used over a time period of several years. Reference cigarettes have been manufactured and provided by the University of Kentucky Center for Tobacco Reference Products (CTRP, Lexington, Kentucky, USA) since 1968. They are widely used as a consistent and uniform standard in many aspects of research and provide a basis for comparing data in different laboratories. The 3R4F reference cigarette, in use since 2008, is the third version of a full-flavor, filtered, American blended cigarette delivering about 9.4 mg tar/cigarette in 9.0 puffs under standard International Organization for Standardization (ISO) machine smoking conditions (ISO 3308:2000 41 ). 42 In 2014, CTRP obtained a service agreement with the FDA to produce a certified reference cigarette. Fifty million of the first certified reference cigarette, 1R6F, were manufactured in March 2015. The 1R6F reference cigarette is similar to 3R4F, and a certificate of analysis has been issued indicating an ISO tar yield of 8.6 mg in 7.5 puffs. 43,44
Reference cigarettes are packed in soft packs of 20 cigarettes. Prior to use, they should be conditioned for at least 48 h and up to 10 days at 22 ± 1°C and 60 ± 3% relative humidity (RH), according to ISO standard 3402. 45
HNB tobacco products
Several HNB tobacco products are currently commercially available in a number of markets worldwide.
One category of products heats tobacco sticks inserted into a device with an electronically-controlled heating element outside or inside the tobacco rod. Products based on this design include
Another category of products uses a carbon-based heat source at the tip of the product, which transfers heat to a tobacco plug. These include Revo, a rebranded version of Eclipse (R.J. Reynolds Tobacco Company, Winston-Salem, North Carolina, USA) and TEEPS (Philip Morris Products S.A.), designated as CHTP in research.
A third category of products, sometimes referred to as hybrid products, produce an aerosol from a nontobacco source and pass it through a tobacco plug to absorb flavor and nicotine.
53
This review focuses on the first category of products and, moving forward, THS 2.2 is used as a representative example of its product category. The other product categories are outside the scope of the review and are not further discussed.
Tobacco Heating System version 2.2
The THS 2.2 has been previously described and its components are shown in Figure 2, 21 Briefly, THS 2.2 has three distinct components: (1) a tobacco stick, (2) a tobacco stick holder, and (3) a charger (Figure 2(a)). The tobacco stick (Figure 2(b)) contains a tobacco plug (Figure 2(c)), a transfer section, and a mouth piece assembled with an overwrapping of cigarette paper. The tobacco stick holder contains the electronically-controlled heating element, a rechargeable battery, and the heating control software. The tobacco stick is inserted into the tobacco stick holder, which heats the tobacco plug. The holder is programmed to finish heating after a maximum period of 6 min or after 14 puffs (whichever is reached first). After the heating process, the front part of the tobacco stick holder is pulled out to extract the tobacco stick. The charger is used to recharge the tobacco stick holder after each use.
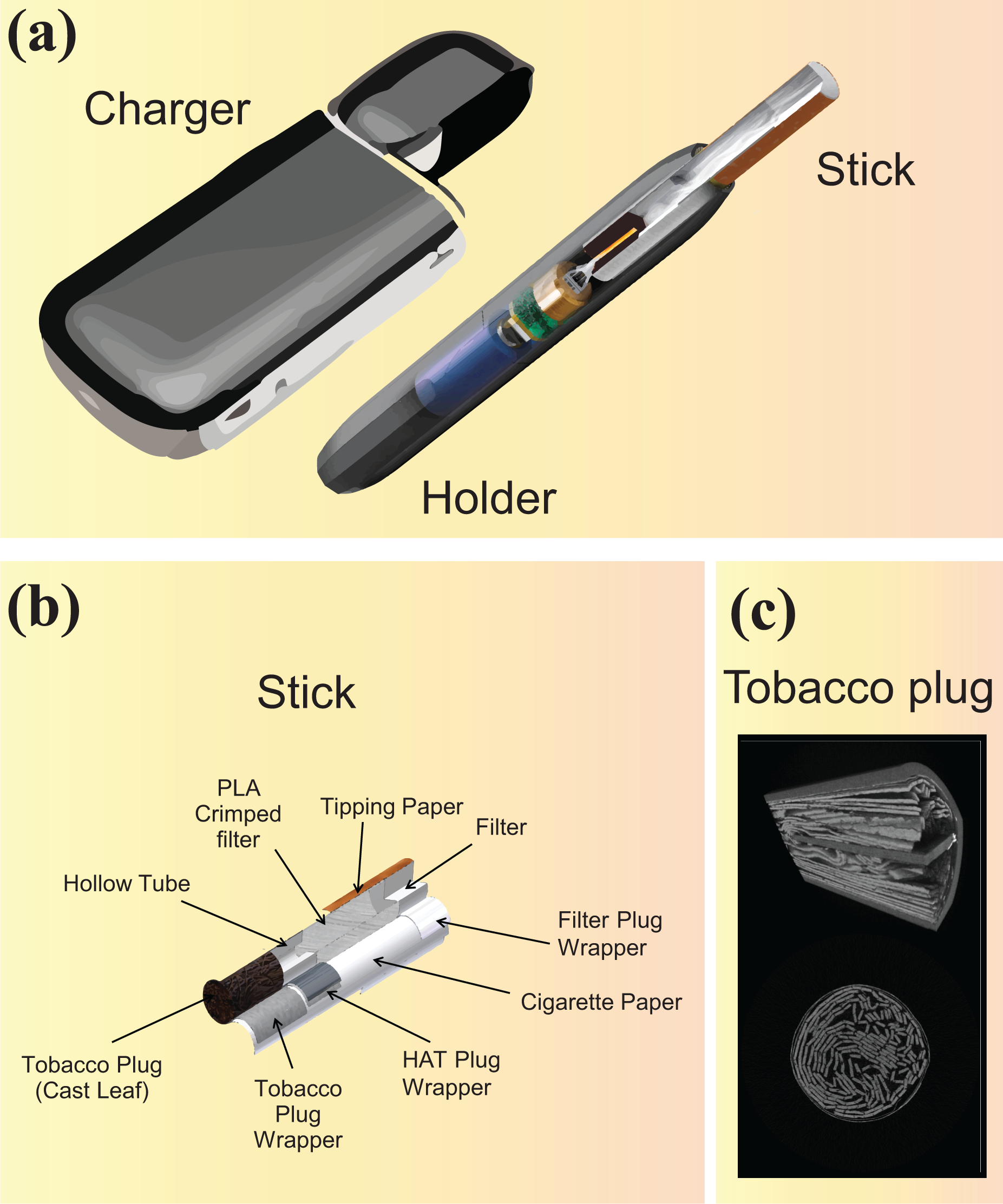
THS 2.2 product characteristics. (a) The three components of the THS 2.2 product. (b) Cross-sectional view of the tobacco stick. (c) Tobacco plug. HAT: hollow acetate tube; PLA: polylactic acid; THS: Tobacco Heating System.
THS 2.2 tobacco sticks are manufactured by Philip Morris Products S.A. For experimental purposes, THS 2.2 tobacco sticks are conditioned for at least 48 h and up to 10 days at 22 ± 1°C and 60 ± 3% RH, according to ISO standard 3402. 45
Puffing regimes
While no puffing regime can be representative of a broad range of human use patterns, standardization is key to provide a basis enabling meaningful comparisons between different products. To this end, the objective of a standard puffing regime is to provide a set of conditions under which the product emissions can be compared with those of other products in a meaningful manner.
The development of smoking machine regimes for cigarettes has been the subject of much debate and has been extensively discussed elsewhere and reviewed most recently by Klus et al. 54
Table 1 provides a comparison of the main cigarette smoking regimes that have been developed over time.
Overview of cigarette smoking regimes.
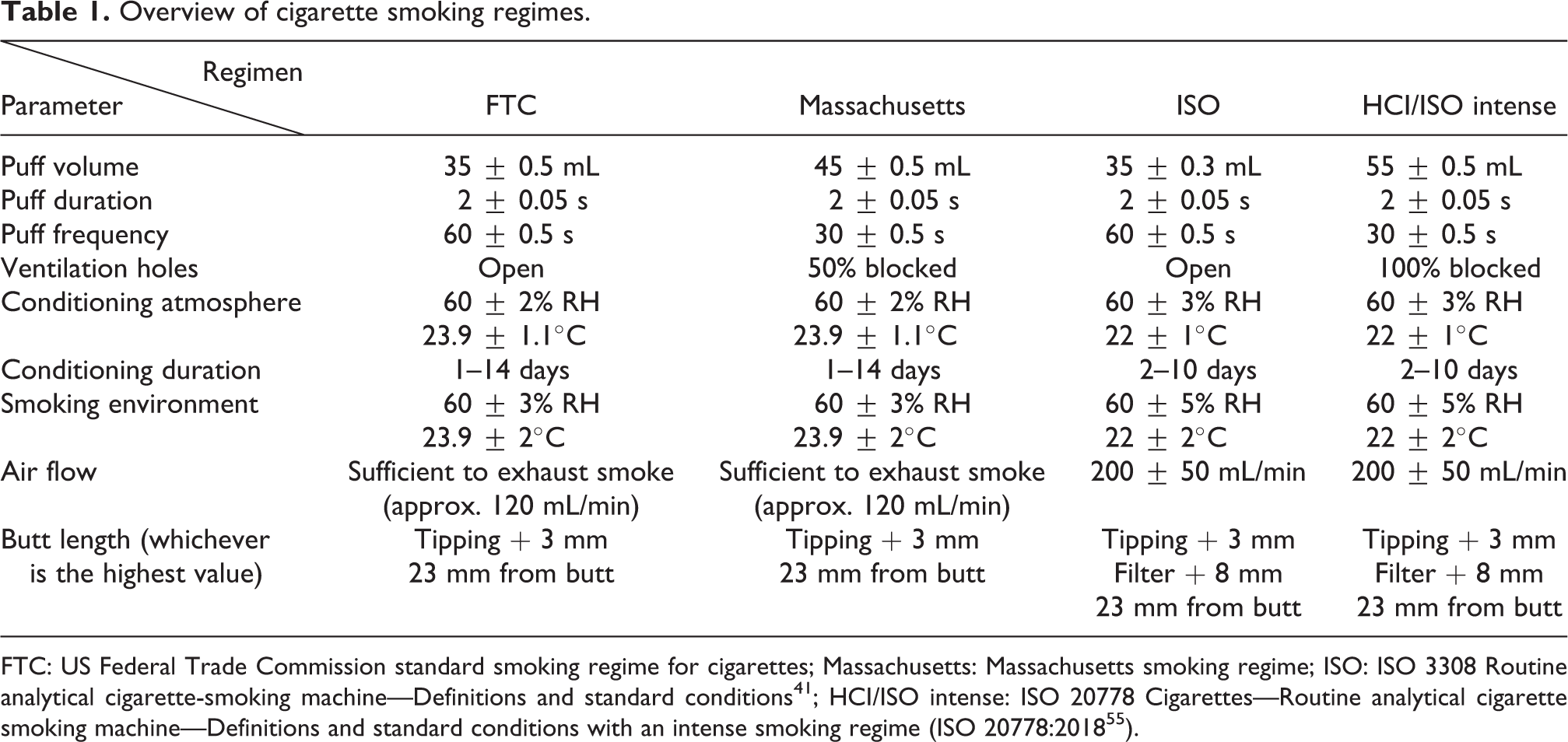
FTC: US Federal Trade Commission standard smoking regime for cigarettes; Massachusetts: Massachusetts smoking regime; ISO: ISO 3308 Routine analytical cigarette-smoking machine—Definitions and standard conditions 41 ; HCI/ISO intense: ISO 20778 Cigarettes—Routine analytical cigarette smoking machine—Definitions and standard conditions with an intense smoking regime (ISO 20778:2018 55 ).
The World Health Organization Study Group on Tobacco Product Regulation (TobReg) recommended using the Health Canada Intense (HCI) machine-smoking regime, now the ISO intense regime (HCI/ISO intense), for product characterization and hazard assessment. 3
While THS 2.2 aerosol can be generated according to the ISO and HCI/ISO intense smoking regimes, some requirements cannot be fulfilled for technical reasons. These parameters may also differ for other HNB products and will need to be addressed in future standards. For the generation of THS 2.2 aerosol: (1) air velocity control around the tobacco stick is not relevant, as the tobacco stick is enclosed within the tobacco stick holder; (2) the butt length requirement is not applicable, as the sticks are not consumed by combustion and remain the same size throughout use; (3) there is no ignition of HNB tobacco sticks by an external lighter (the aerosol collection starts after a 20-s preheating period); (4) the puff count depends on the puffing frequency and is limited by the total heating time of the system, fixed at 6 min (according to this, 12 puffs can be reached when the THS operates under the HCI/ISO intense puffing regime but is limited to 6 puffs when the aerosol generation is performed under the ISO puffing regimen); (5) vent blocking does not apply, as the tobacco stick filters are not perforated.
The HCI/ISO intense regime has been widely used in research, mainly because it provides the most relevant comparative basis to evaluate the composition of HNB aerosols with CS. 56 Moreover, according to recent puffing topography measurements with two different commercial HNB tobacco products, adult smokers’ behavior data were shown to be similar to the puff volume and duration of the HCI/ISO intense smoking regime. 51,57 However, the debate on the applicability and adaptation of existing smoking standards to HNB products for regulatory purposes has yet to take place.
The chemical composition of THS 2.2 aerosol was assessed upon generation with a broad range of different puffing regimes, including the ISO and HCI/ISO puffing regimes, as well as regimes using longer puff volumes, durations, intervals, and higher numbers of puffs. 21 For 49 HPHCs at study, the maximum yield and the yield obtained with the HCI/ISO puffing regime were compared to identify HPHCs for which the standard protocol may underestimate yields. Despite substantial differences in the regimes, the ratios of maximum yield to HCI/ISO yield were less than 2 for 42 of 49 analyzed compounds.
HNB aerosol generation systems and methodologies
Key milestones in the development of aerosol generation systems and methodologies
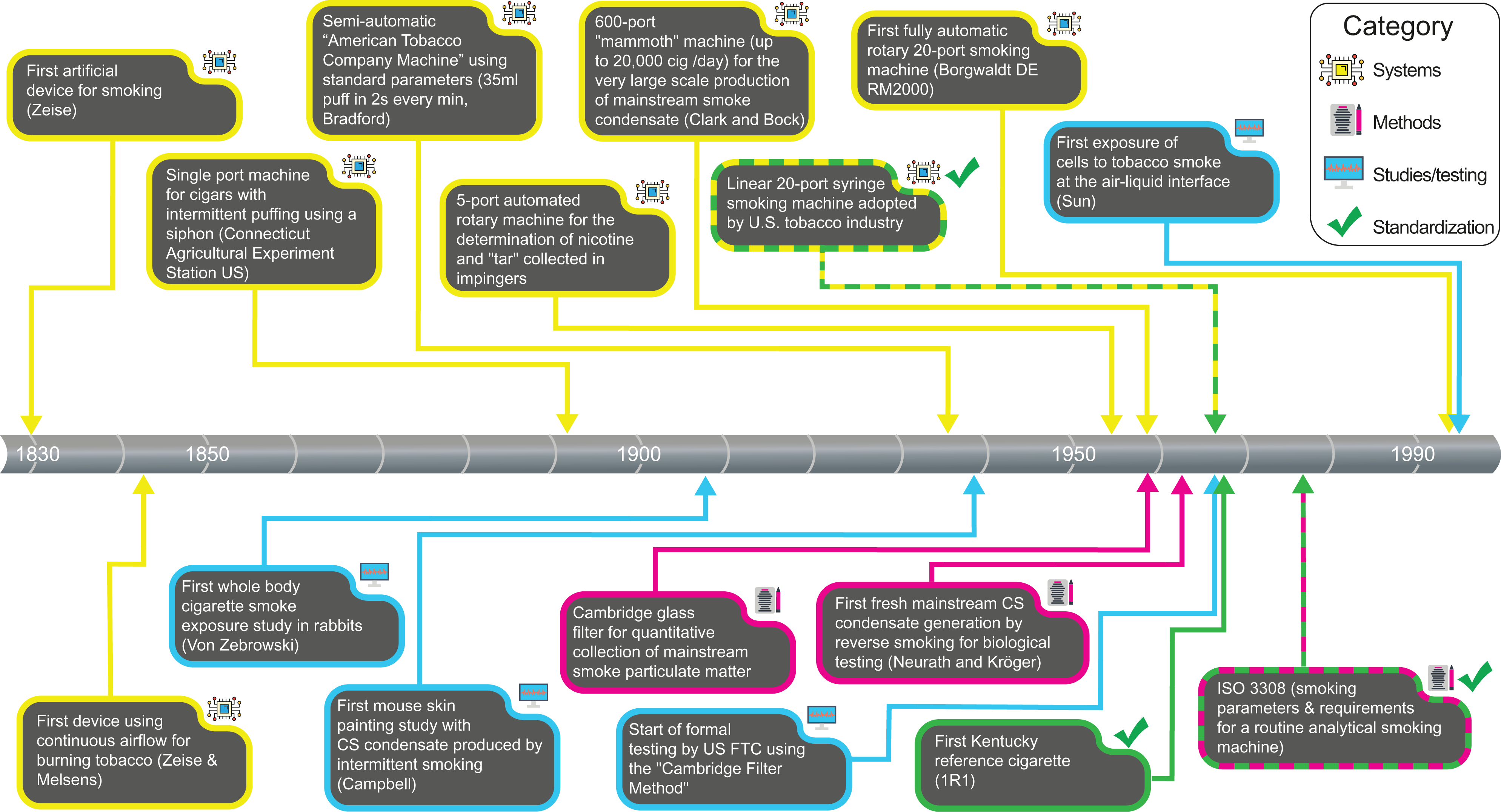
Klus et al. 54 thoroughly reviewed the development of methods and devices for the generation of CS, from the first simple setups in the 1840s to the sophisticated and fully automated smoking machines available today.
Figure 3 provides an overview of key milestones in the development of these systems and methods.
HNB aerosol generation: Experimental setups and methods
The rapid development of HNB products has posed new challenges and required the development of new or modified experimental setups and methods for aerosol generation. Indeed, as previously described, HNB tobacco products are not consumed as cigarettes and they constitute a more heterogeneous product category. Notably, HNB aerosols are qualitatively and quantitatively different from CS.
HNB aerosol generation with linear smoking machines
The original linear smoking machines for CS generation
The first machines allowing smoking of several cigarettes simultaneously or alternately were linear smoking machines. 54 In a linear smoking machine, individual smoking ports are each coupled to a separate syringe or pump, which draws air through the tobacco product placed in a holder connected to that smoking port. In use, mainstream smoke is drawn and collected individually per smoking port, coupled to an aerosol trapping system such as a Cambridge filter pad (CFP) holder or an impinger (“HNB aerosol collection, fractionation, and analysis” section). Linear smoking machines allow stick-to-stick or puff-by-puff collection of aerosols for direct analysis or trapping and are therefore better suited for product chemical characterization (e.g. TNCO yield determination) or for the analysis of HPHCs. A key advantage is that the connection from the smoking port to the trapping system is short, thereby avoiding significant changes in aerosol due to surface interference or aging.
The 20-port linear smoking machine modified for THS 2.2 aerosol generation
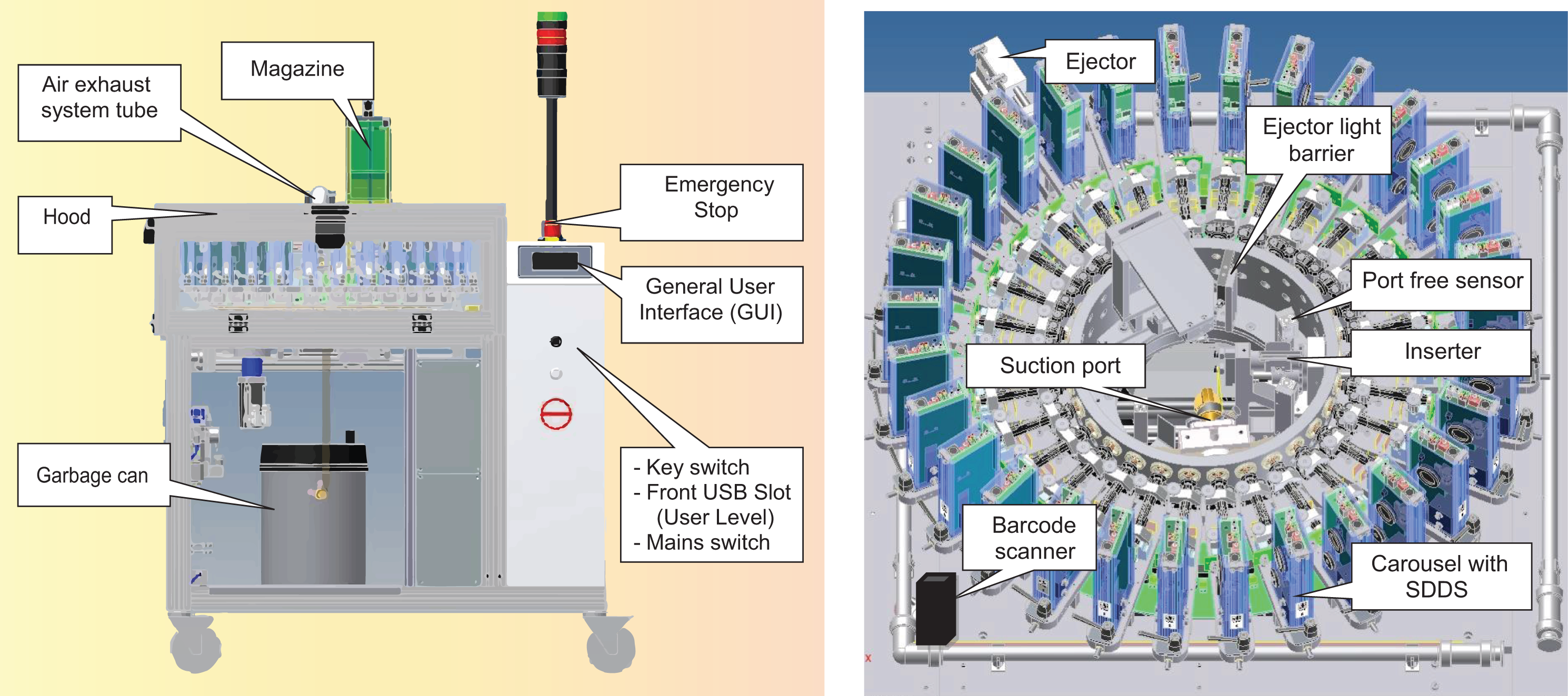
Linear smoking machines with up to 20 ports are commercially available for the analytical smoking of cigarettes. The 20-port linear smoking machine type LX20 (Borgwaldt KC GmbH, Hamburg, Germany) is described in Figure 4. Twenty individual smoking ports are equipped with 20 individually controlled syringes allowing the use of up to 20 different regimes, including all of the standard smoking regimes. When several regimes are used, these can be randomized across the 20 ports.

Modified linear smoking machine for THS 2.2 aerosol generation. THS: Tobacco Heating System.
For THS 2.2 aerosol generation, individual supports made of an aluminum adaptor plate and a Plexiglas adaptor bracket are used to support the tobacco stick holders. An alignment tool, including a stopper, is used to align the support with the smoking port and insert the tobacco stick at the correct distance.
Tobacco stick holders are activated either individually or, when an activation bar is used, concurrently. In both cases, activation is by pressing the central start switch for 4 s. When an activation bar is used, smoking ports 5–7 and 14–16 are used to fit the bar and may not be used for THS 2.2.
At the end of a smoking run, tobacco stick holders are removed manually from the smoking ports, and the tobacco sticks are removed by opening the tobacco stick extractor.
HNB aerosol generation with rotary smoking machines
The original rotary smoking machines for CS generation
The first prototypes of rotary smoking machines were developed in the mid-1950s as a means to improve the automation of cigarette machine smoking and allow continuous, large-scale aerosol generation. 54
In the current designs of rotary smoking machines for conventional cigarettes, ports for inserting cigarettes are arranged horizontally on the outside of a rotating ring/carousel, which rotates in steps to make one turn in a defined period of time. A puff is drawn whenever a port holding a cigarette reaches a position facing the central suction port connected to the smoke pump, resulting in the generation of a near continuous stream of mainstream smoke. Sidestream smoke generated during the nonpuffing interval for each cigarette is either directed into an exhaust air stream or collected for sidestream smoke studies. 66,67 Commonly, the aerosol generated from all ports is collected cumulatively in a central trap. However, smoke collection from individual products is also possible. Unlike in linear smoking machines, in rotary smoking machines, there is a physical separation between the smoking ports and the programmable dual-port syringe pump (PDSP) that generates the suction. Although the connective tubing is made from inert materials, there is an increased risk for smoke/aerosol deposition or condensation.
Rotary smoking machines are ideally designed for continuous, large-scale aerosol generation for in vivo exposure studies. By extension, in order to bridge and compare in vivo and in vitro studies, rotary smoking machines are often also used for in vitro studies. They may also be used to generate sufficient amounts of aerosol (e.g. for large-scale production used in mouse dermal tumorigenicity studies or for the analysis of low-yield chemicals). 68,69
In the following sections, we describe the SM 2000 rotary smoking machine in its original (for cigarettes) and modified (for THS 2.2) versions. These smoking machines are routinely used for studies comparing THS 2.2 with cigarettes, including in vitro whole aerosol exposure studies and in vivo inhalation studies.
The SM 2000 rotary smoking machine for CS generation
The SM 2000 for CS generation (SM 2000 CS) is a fully automated 30-port rotary smoking machine equipped with a PDSP and an active sidestream exhaust. It was designed by Philip Morris Research Laboratories (PMRL, Köln, Germany) and is manufactured and commercialized by Burghart Messtechnik (Weidel, Germany). 70
The PDSP is a pump made of two identical pump modules consisting each of a linear piston pump in a glass cylinder. The pump modules are moved by a stepper motor and connected through a shared aerosol valve. The PDSP is used to draw an aerosol at the valve inlet and deliver it at the valve outlet. Alternating operation of each pump module allows for a range of puffing regimes, from single puffs with long intervals to a quasi-continuous aerosol flow. 71
The fully automated operation of SM 2000 allows for continuous mainstream smoke generation over several hours. In the 30-port configuration, closing time between two puffs from two subsequent cigarettes may be down to less than 0.04 s, thereby achieving the shortest possible flow interruption between two puffs. The SM 2000 CS offers different ways to execute smoke runs and covers most of the common standard cigarette smoking regimes. The operation parameters of the PDSP, such as puff profile, volume, or duration, can also be adjusted to use alternative smoking regimes for research.
The modified SM 2000 for THS 2.2 aerosol generation (SM 2000 THS 2.2)
The SM 2000 THS 2.2 is a modification of the SM 2000 CS specifically designed by PMI R&D (Neuchâtel, Switzerland) to take into account the THS 2.2 product specificities (i.e. a tobacco stick, a tobacco stick holder, and a rechargeable electronic device, as described in the “Tobacco Heating System version 2.2” section).
The tobacco sticks are inserted into the 30 cigarette ports together with their tobacco stick holders. Each tobacco stick holder is connected to a smoking device docking station (SDDS) providing the energy for recharging the tobacco stick holder battery and acting as an electrical interface between the individual tobacco stick holder and the smoking machine controller. Each SDDS consists of a main electronic board with a power supply for recharging and a communication board for interacting with the smoking machine controller (Figure 5).
Schematic views of SM 2000 THS 2.2. (a) Front view. (b) Top view. THS: Tobacco Heating System.
The insertion and extraction of a tobacco stick into and out of a tobacco stick holder is performed automatically by the smoking machine. An inserter pushes the tobacco stick into the tobacco stick holder. An ejector pulls the head of the tobacco stick holder and grabs and releases the butt. The tobacco stick holder heating process is initiated by the smoking machine remotely and controlled by the tobacco stick holder over the preheating and heating periods.
During an aerosol generation session, the 30-port carousel is divided into 2 virtual groups of 15 ports: one virtual group is grouping the even ports, the other the odd ports. One group of SDDS is puffing while the other group is preparing for the next puffing process. The suction port is only available for the puffing group, while the insertion and ejection unit is available for the group in the preparation phase.
In order to ensure constant environmental conditions and to effectively remove heated air, the smoking machine should operate with an inflow of clean air of 25–50 L/min and an exhaust air flow of 50–100 L/min (approximately double the inflow).
Due to the high water content of THS 2.2 undiluted aerosol, condensation may occur when the aerosol comes into contact with materials at a lower surface temperature than the aerosol temperature. In order to avoid major condensation effects, the surfaces of the suction port, the PDSP, and all tubes conveying the aerosol up to the dilution system are stabilized at 41°C by using a water warming system (heated water jacket).
Prior to an experiment, THS 2.2 sticks are conditioned for at least 48 h and up to 10 days at 22 ± 1°C with an RH of 60 ± 3%, in line with ISO standard 3402. 45 Before the start of aerosol generation, a predefined amount of conditioned test items are taken out of the conditioning chamber and placed into the magazine of the smoking machine.
For each tobacco stick, 12 puffs are drawn according to the HCI/ISO intense smoking regime (1 puff every 30 s, for 6 min) with the minor deviations previously described (“Puffing regimes” section).
For in vitro exposures at the ALI, aerosol is usually generated continuously. Mathis et al. optimized the duration of exposure for organotypic bronchial cell cultures based on their sensitivity to CS exposure, likely to have a larger impact than THS 2.2 on the endpoints of interest. The authors exposed normal human bronchial epithelial (NHBE) cells at the ALI with 3R4F CS for 7, 14, 21, and 28 min and found that 28 min induced the highest concentration of secreted matrix metalloproteinase-1, an indicator of airway cell responsiveness. 72
For in vivo exposures, animals (mice and rats) are exposed to the aerosol for up to 6 h/day for 28 days (subacute toxicity studies 73 ), 90 days (subchronic toxicity studies 74 ), or up to 18 months (chronic inhalation toxicity and tumorigenicity studies 75 ).
HNB aerosol collection, fractionation, and analysis
For many applications, spanning routine yield measurements, HPHC quantification and in vitro toxicity testing, the collection of aerosol fractions constitutes an indispensable requirement. Figure 6 shows the experimental design for the collection of, and in vitro exposure to, different aerosol fractions, including whole aerosol, solid and liquid particles (commonly referred to as TPM), and the GVP.
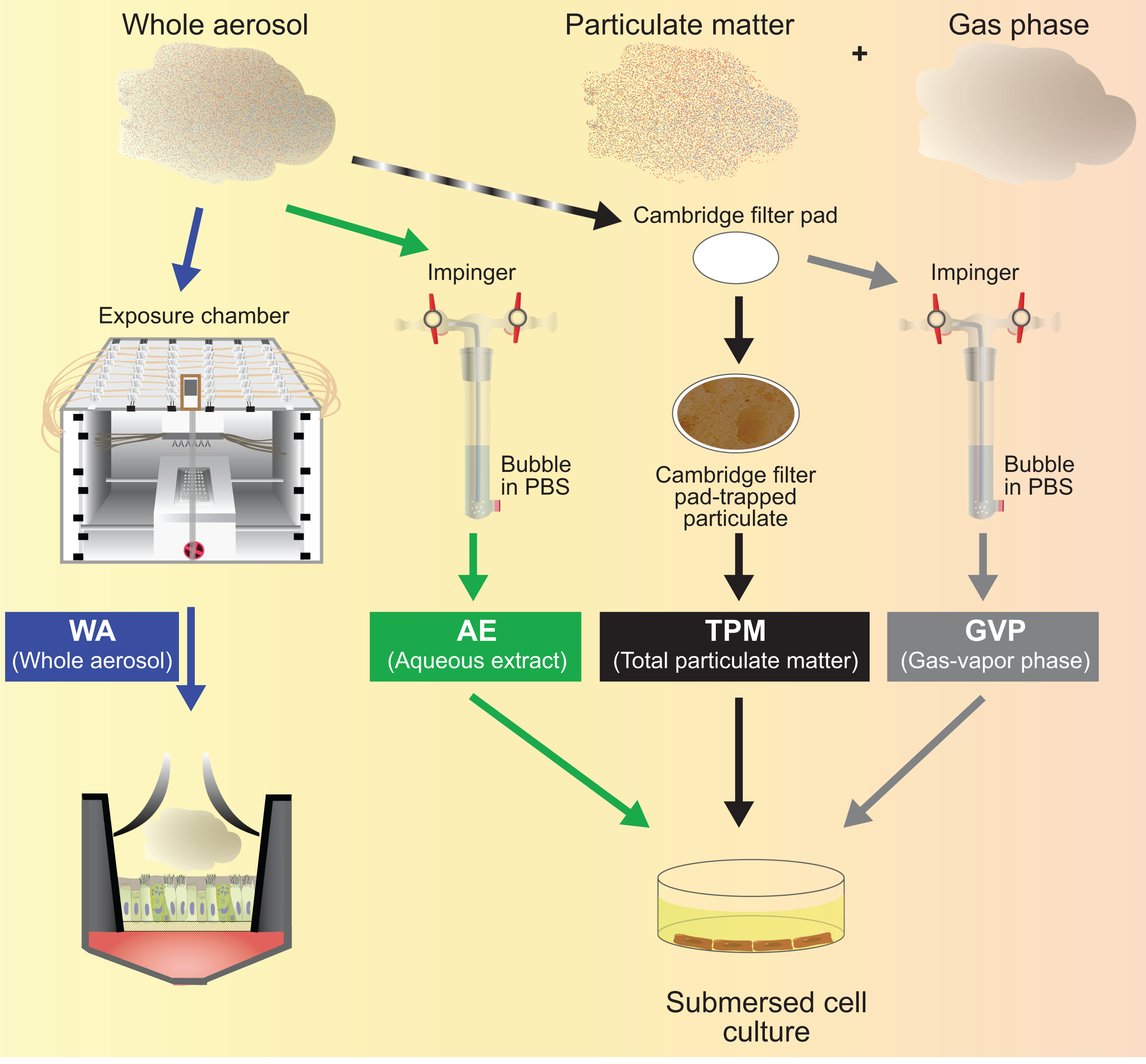
Experimental design for the collection of, and in vitro exposure to, whole aerosol, AE, TPM, and GVP fractions. AE: aqueous extract; TPM: total particulate matter; GVP: gas–vapor phase.
The collection and fractionation process, while widely used, has several limitations. Methods used to trap aerosols may exert profound effects on their physical and chemical composition. No single method can efficiently trap all of the constituents present in the solid, liquid, and gas phases. The analysis of the individual fractions might underestimate the overall toxicity resulting from dynamic evolution and interactions between the phases. Stability of the collected materials prior to analysis has to be ascertained. The solvents used might react with constituents of the aerosol fraction or exert biological activity themselves. For in vitro experiments using submerged cell cultures, the aerosol needs to be collected in a biocompatible solvent, which can then be applied to the cells.
Furthermore, standard trapping and extraction procedures have been developed for combustible CS. Given the qualitative and quantitative differences between CS and HNB aerosols, the applicability of these methods to capture comparable samples from different HNB tobacco products has yet to be fully determined, thus analytical characterization and validation of the trapping has to be performed.
TPM collection using CFPs
The TPM fraction is the most commonly used fraction in in vitro toxicological assessment. TPM contains key mainstream smoke constituents and, in particular, most of the nicotine yield and major carcinogens, such as PAHs and TSNAs. Besides, TPM is more stable than other fractions, and it can be stored refrigerated for longer periods of time.
ISO standard 4387 for the determination of TPM and nicotine-free dry particulate matter (NFDPM) specifies a method for TPM collection by drawing the aerosol through a CFP and eluting the trapped TPM with a solvent (ISO 4387:2000 76 ).
Prior to use, 44 mm diameter CFPs (Borgwaldt) are conditioned for at least 12 h at 22 ± 1°C and RH of 60 ± 3%. One CFP is placed into a standardized filter pad holder. The combination of the CFP and filter holder is weighed before and at the end of trapping for the determination of TPM by gravimetry (mg/test item), which is determined as the difference between the weight after and the weight before sample collection, divided by the number of test items aerosolized. The total quantity of material trapped depends on the sample collection duration and flow rate. Flow rate measurements used for the collection first need to be calibrated using certified flow meters.
For extraction, the CFP is removed from the filter pad holder and placed into a centrifugal tube device with an integrated filtration membrane (0.45 µm) together with a suitable solvent, which is also used for sample dilutions. TPM is extracted by shaking the tube on a rotary shaker for 30 min at 400 rpm prior to centrifugation at 1200 rpm for 5 min.
In situ CFP extraction methodology to obtain more accurate water and tar values
ISO standard 4387 is widely referred to in product regulations mandating ceilings on, or the reporting of, TNCO values. It was developed for cigarettes and cannot be used as is for the determination of TPM and, by extension, NFDPM in high-water content aerosols, such as HNB aerosols. 22 Ghosh and Jeannet showed that errors in water content determination may occur due to water losses at several stages, including when the CFP holder is opened to remove the filter pad, when the filter pad is manually handled, and due to adsorption by the plastic filter pad holder. This resulted in inaccurate values for the water content and, as a consequence, erroneous and overestimated values for NFDPM. 77 In order to obtain more accurate NFDPM values, the authors developed a method for the in situ syringe extraction of TPM from a CFP in a closed filter holder. The principle is to avoid any of the abovementioned water losses by extracting the loaded filter pad while kept in the CFP holder, which is hermetically sealed by two caps. This is achieved by flushing the extraction solvent numerous times through the hermetically sealed CFP holder by means of an in situ extractor. The in situ methodology showed a significantly more complete water recovery, with 24% and 19% higher water yields compared to those obtained using the standard ISO methodology under ISO and HCI/ISO intense smoking regimens, respectively. This resulted in more accurate NFDPM values, reduced by 50% and 42% for ISO and HCI/ISO intense smoking, respectively, compared to the standard ISO methodology values.
TPM exposure concentrations in vitro
When TPM preparations are used for in vitro applications, the highest amount of test substance to be used depends on its cytotoxicity and solubility in the final treatment mixture, which may be determined in a preliminary experiment.
Cytotoxicity may be detected either by a reduction in the number of revertant colonies, a diminution of the background lawn, or the number of surviving cells in a treated culture. The cytotoxicity of a substance may be altered in the presence of metabolic activation systems.
Insolubility should be assessed as precipitation in the final mixture under the actual test conditions and evident to the unaided eye. For noncytotoxic substances that are not soluble at testing concentration, one or more concentrations tested should be insoluble in the final treatment mixture. No precipitate should interfere with the assay.
Collection of whole aerosol and GVP aqueous extracts using impingers
Aqueous extracts (AEs) can be trapped by bubbling the aerosol through a specific volume of liquid medium (cell culture medium or phosphate-buffered saline (PBS)) in an impinger (wash bottle) (Figure 6). Optimization of the trapping methods for novel in vitro assays is described in the “Optimization of aerosol fraction trapping for in vitro mechanistic assays in submersed cell cultures” section. The method allows the trapping of water-soluble chemicals from both the GVP and (partially) the particulate phase. If the impinger is placed downstream of a CFP, a GVP extract will be collected (Figure 6). The content of the CFP is either discarded (for GVP collection) or combined and extracted with the extraction solution of the impinger(s) (for combined TPM and GVP AE collection).
The impinger (or the CFP followed by the impinger) is connected at the sampling point of the aerosol generation machine, usually located before the PDSP pump (see “Optimization of aerosol fraction trapping for in vitro mechanistic assays in submersed cell cultures” section). When used with THS 2.2 aerosol, the impinger is connected behind the temperature-controlled insulation kit.
Aerosol condensate collection
Aerosol condensate collection using cold traps
A cold trap is usually an empty glass impinger cooled in a dry ice and acetone or methanol mixture (−78°C). The low temperatures cause the aerosol and ice particles to form a mat at the bottom of the trap. All phases may be collected as a condensate in a cold trap. 78
Aerosol condensate collection using impaction traps and electrostatic traps
Impaction and electrostatic traps are other methods of collecting CS condensate (CSC) that are not currently routinely used in research of HNB aerosols, as further work is ongoing to optimize their use to that effect.
An impaction trap consists of a round-bottomed flask with an injector tube reaching the bottom of the flask. The distance between the orifice of the injector tube and the bottom is usually smaller than 1 mm. The smoke is directed through the injector tube at velocities depending on the diameter of the orifice. A velocity of 200 m/s is achieved with diameters from 0.25 to 0.50 mm. At this velocity, CSC is effectively deposited at the bottom of the flask. 69
The use of electrostatic traps or precipitators is long established in the field of CS research. 79 Electrostatic CS trapping systems act by placing a positive electrode in the center of a glass tube traveled through by CS. The tube is surrounded by a cylindrical negative electrode, usually made of stainless steel. The electrodes supply a variable voltage creating an electrical field. Charged CS particles are attracted to the inner glass tube surface and are subsequently removed by a solvent. 54,80
Aerosol HPHC analyses
The first level of harm reduction substantiation is to demonstrate reduced formation of HPHCs by HNB products (Figure 1). 8,11,52,81
Most standard test methods for the quantitative determination of tobacco product emissions have been developed for CS and only for a very limited number of constituents. ISO standards have been established for assessing basic smoke parameters, such as nicotine, 82 water, 83,84 and TPM and NFDPM, 76 as well as for the quantification of specific constituents, such as CO, 85 benzo[a]pyrene, 86,87 menthol, 88 or TSNAs. 89 Standardized methodologies and inter-laboratory trial results are also available for other constituents as industry standards through CORESTA, the Centre for Scientific Research Relative to Tobacco, with a number of methods presently in development phase within the ISO technical committee in charge of tobacco and tobacco products, such as for carbonyls and volatile compounds. 56
The relative abundance of HPHCs in CS and their amenability to measurement using routine analytical equipment has facilitated the development of standard procedures. The relatively low levels of constituents in HNB aerosols is a significant technical challenge to the development of efficient analytical methods.
Lists of constituents to be reported for CS have been developed by several regulatory authorities. These lists include the Health Canada list of HPHCs, 90 the TobReg list, 91 and the FDA full 92 and abbreviated lists, 93 which include 44, 39, 93, and 18 HPHCs, respectively. In its Draft Guidance for Modified Risk Tobacco Product Applications, the FDA recommends that applicants conduct analyses to determine levels of the full FDA HPHC list. 7 However, with the growing diversity of HNB tobacco products, the question of which constituents are more relevant to characterize and compare HNB tobacco products with cigarettes has yet to be debated at the international level, taking into account the major differences between these products and within each product category.
A list of 58 constituents has been selected for the evaluation of THS 2.2 according to the following criteria: (1) constituents that are measurable with existing ISO testing methods, (2) constituents that have been identified by regulatory and other relevant authorities, (3) constituents with established biomarkers of exposure (BoExp), and (4) other constituents deemed relevant. The list contains all 18 HPHCs subject to reporting on the FDA’s abbreviated list, all priority constituents identified by Health Canada, International Agency for Research on Cancer (IARC) Group I carcinogens that are measurable in CS, and all priority toxicants identified by TobReg. 21
The systems used to trap aerosol fractions for selected HPHC analyses are described in Table 2. Impingers or micro-impingers are loaded with defined quantities of different solvents depending on the chemical nature and subsequent use of the collected material. Glass beads or frits may be used to increase the surface area contact between the solvent and the aerosol and therefore improve the trapping efficiency. Additional parameters, such as the number of tobacco sticks, number and size of impingers (normal vs. micro-impingers), and the experimental temperature, are also adapted in function of the chemical category of interest.
Aerosol trapping systems for the determination of selected HPHCs in aerosols generated according to HCI/ISO intense.
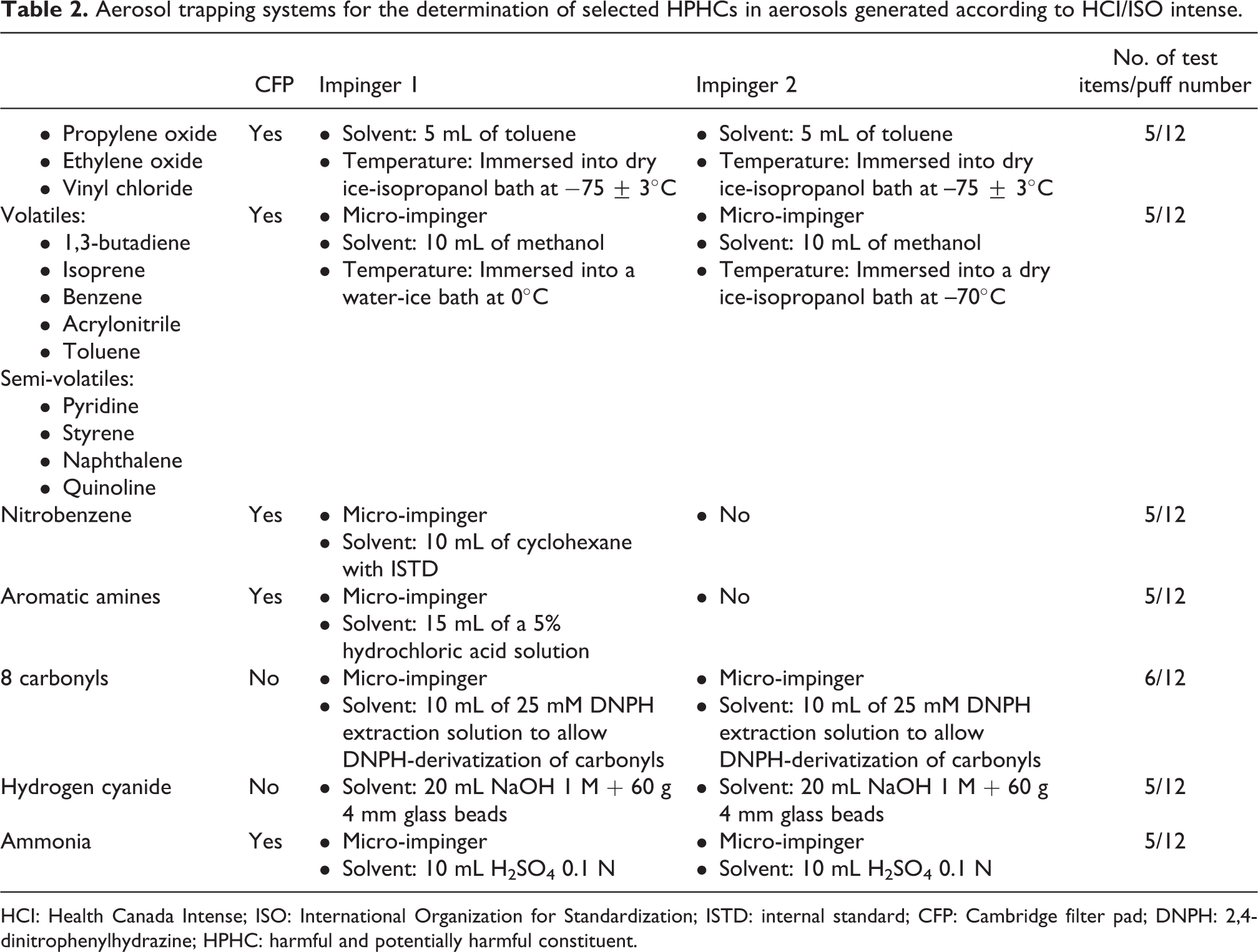
HCI: Health Canada Intense; ISO: International Organization for Standardization; ISTD: internal standard; CFP: Cambridge filter pad; DNPH: 2,4-dinitrophenylhydrazine; HPHC: harmful and potentially harmful constituent.
Determination of PSD
As described in the “Physical and chemical dynamics of aerosols” section, the PSD is a key physical characteristic of an aerosol, as it determines the potential for deposition of its particulate matter in the respiratory tract. PSD determination is therefore a key element of aerosol characterization in an experimental setting. The generic term “particle” is used in the following sections, but it should be borne in mind that different particles are present in CS (liquid droplets and solid particles) and in HNB aerosols (mostly liquid droplets).
PSD determination using the PIXE cascade impactor
Cascade impactors are the most commonly used instruments to measure aerosol PSD, and they are a reference method for inhaled pharmaceutical products. 94,95
A cascade impactor allows the separation of particles present in an aerosol into known size ranges by drawing the sample through a series of progressively finer nozzles. The aerosol is accelerated through a nozzle toward an impaction plate. The cutoff size is reduced by decreasing the nozzle size. By reducing the diameter of the nozzle, the velocity of the airflow increases. Particles are collected on the different impaction plates according to their aerodynamic diameter. Larger particles with greater inertia impact on the impaction plate and are collected, while smaller particles turn with the air around the collection surface.
The cascade impactor operates with a vacuum pump to draw the aerosol of interest through the impactor at the required flow rate for a defined period. Each impactor plate is weighed before and after flushing the impactor with the aerosol to determine the amount of particles collected on a given impactor plate. From the mass of particulate matter on each stage, the MMAD and GSD of the aerosol can be determined. 96,97
PMI uses the PIXE cascade impactor (PIXE International Corp., Tallahassee, Florida, USA) in HNB aerosol research because of its low flow rate, which minimizes the amount of dilution air required. The model consists of nine impactor stages with stainless steel or plastic impactor plates. This device allows the separation of the aerosol into 10 particle diameter ranges: >16, 16–8, 8–4, 4–2, 2–1, 1.0–0.5, 0.5–0.25, 0.25–0.12, 0.12–0.06, and <0.06 µm. These 10 intervals are intended to provide adequate resolution to characterize environmental aerosols of interest to human exposures.
The experimental setup for PSD determination using a PIXE cascade impactor for product characterization is represented in Figure 7. 21
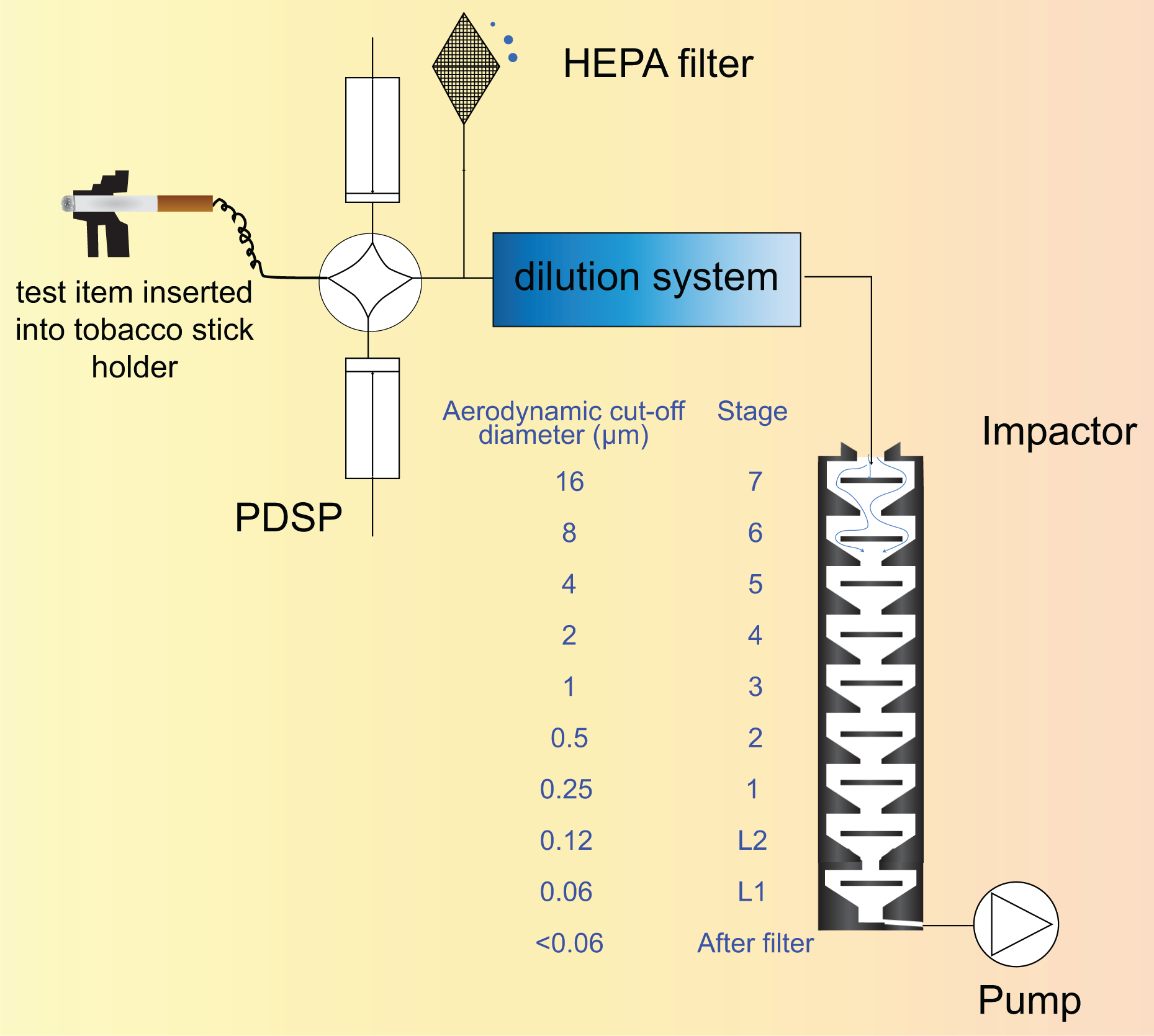
Experimental setup for aerosol particle size determination using PIXE. HEPA: High efficiency particulate air.
A test item or sampling port is connected to the inlet of a PDSP (Burghart Messtechnik GmbH, Wedel, Germany). The outlet of the PDSP is connected to a glass T-junction that allows aerosol dilution and transfer to the PIXE cascade impactor. The outlet of the PIXE cascade impactor is connected to a pump (Vacuubrand GmbH + CO KG, Wertheim, Germany).
Dilution of the aerosol may be needed to minimize the effects of coagulation, which can result in significant and rapid changes in the PSD compared to the undiluted aerosol.
The PIXE impactor is operated at a sampling flow rate of 1 ± 0.1 L/min to achieve the specified particle diameter cut points. The sampling duration depends on the PSD and concentration of the aerosol and is determined in order to avoid overload of any impactor plate.
Impactor plates are weighed before and after the aerosol sampling. The calculation of MMAD and GSD is done separately for each replicate using a log-normal mono-modal fitting distribution of the normalized mass fraction versus the respective midpoint particle diameter for each stage. 21
PSD determination using the Aerodynamic Particle Sizer®
The Organization for Economic Co-operation and Development (OECD) guidelines recommend using cascade impactors for PSD determination. However, they also provide that other devices or physical principles may be used if equivalence to the cascade impactor can be shown or when required by the nature of the test material. 98
PMI has assessed and validated the commercially available Aerodynamic Particle Sizer® (APS™) model 3321 (TSI). Typically, the equivalence of the PIXE and APS™ is verified before the start of a study, and if equivalence is established, the APS™ will be used. For in vivo studies involving THS 2.2 aerosol, equivalence between the APS™ and PIXE was established based on differences between the measures by the two instruments of ±0.2 µm (MMAD) and ±0.3 (GSD).
The APS™ is a time-of-flight mass spectrometer (TOFMS) that measures the velocity of aerosol particles in an accelerating airflow through a nozzle. 99 Figure 8 shows the APS™ experimental setup and aerosol sample flow path. The aerosol flow path is in a continuous downward direction to minimize particle losses. The aerosol is drawn into the inlet and is immediately split into a sample flow, through the inner nozzle, and a sheath flow, through the outer nozzle. The sheath flow is filtered and controlled by the sheath flow pump reunited with the sample flow at the accelerating orifice nozzle, confining sample particles to the center stream and accelerating the airflow around the particles. Small particles reach a higher velocity than larger particles, which lag behind due to inertia.
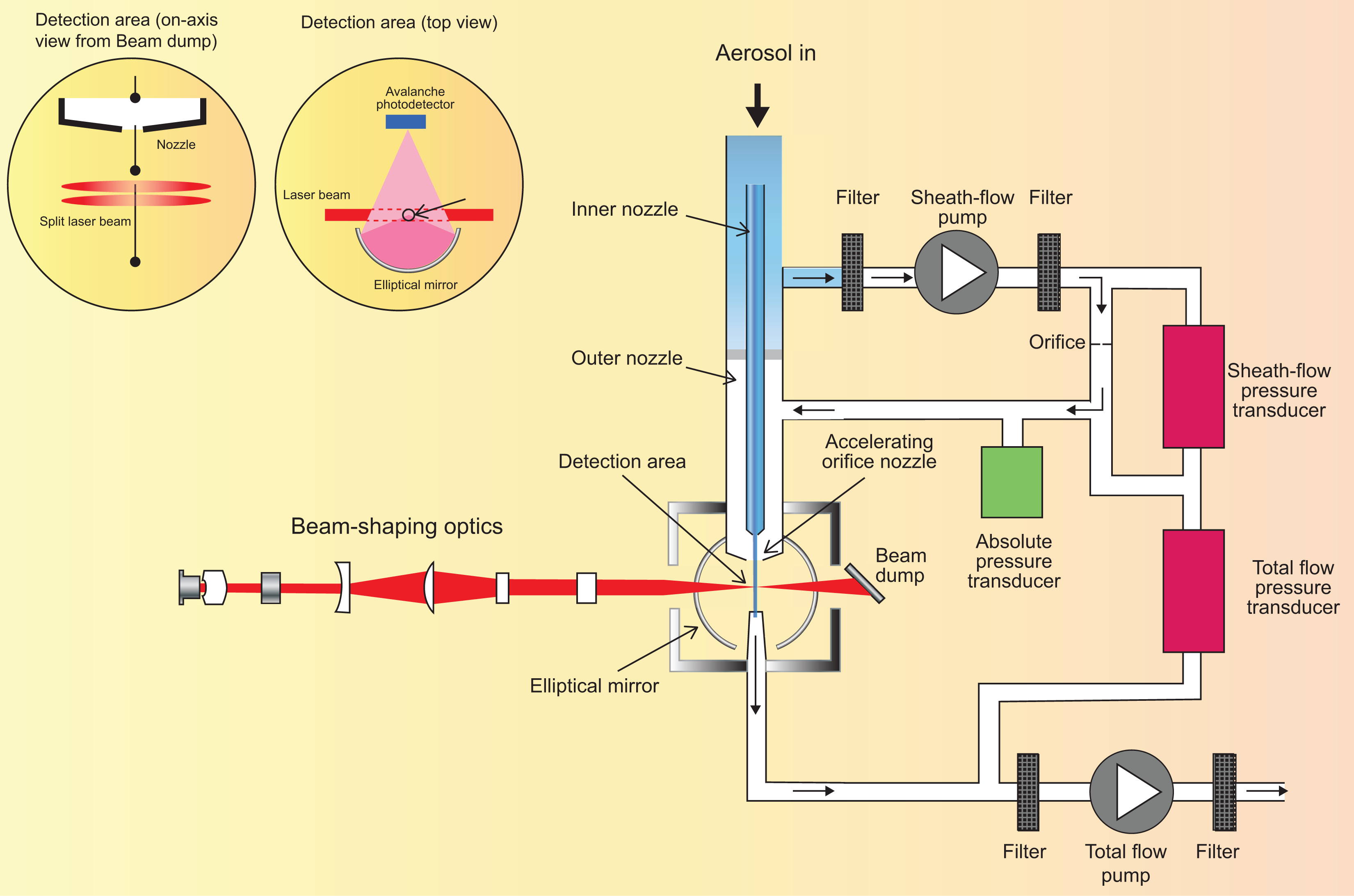
APS™ experimental setup and aerosol flow-through. APS™: Aerodynamic Particle Sizer®.
Particle velocity is then measured in the optics chamber by passing the particles through two overlapping laser beams separated by about 200 µm. An elliptical mirror, placed at 90° to the laser beam axis, collects scattered light onto an avalanche photodetector. A particle passing through both beams generates one signal with two crests. The time-of-flight between the two crests is related to the velocity and hence the aerodynamic diameter of the particle. This measurement detects particles in the range of 0.5–20 µm in densities up to 1000 particles/cm3 At higher densities, errors due to coincidence will increase. Particles generating only one crest (phantom particles) or more than two crests (coincidence error) are not used in building the size distribution calculation. The APS™ also records the height of the peaks, allowing a secondary determination of particle size based on a light-scattering technique. This measurement detects particles in the range of 0.5–20 µm.
The aerosol should be diluted to a particle concentration of <1,000 particles/cm3 to minimize particle coincidence. When this is not possible, the aerosol will be diluted to <10,000 particles/cm3 to be within the concentration limits of the APS™. The diluter uses a capillary tube, the size of which determines the dilution ratio. Dilution factors of 20–10,000 can be achieved using a combination of different capillary sizes. The undiluted aerosol is drawn through the diluter from the APS™. The diluter operates in a closed system in which a small sample of aerosol is diluted with filtered air from the original air sampled. This closed system upholds the integrity of the aerosol by maintaining the same temperature, RH, and elemental composition throughout the sampling process. The flow through the diluter is drawn from the APS™. The flow through the detection area is 5 ± 0.2 L/min. Sampling duration depends on aerosol density and should allow a total particle count of >10,000 particles to reduce the standard error and improve the accuracy of the measurements.
Aerosol exposure systems for in vitro studies
In recent years, driven by the effort to “replace, reduce, and refine” the use of animals in experiments (the “3Rs” principles) 100 and by the vision and strategy for toxicity testing in the 21st century, 101 toxicological assessment of environmental agents has witnessed a strategic shift. Advances in cellular and molecular biology are facilitating a paradigm shift in research, moving away from animal testing to human-relevant in vitro alternatives complemented with computational toxicology, systems biology, and pharmacokinetic modeling. 102,103
Animal-free models are composed of primary cells or cell lines cultured in two dimensions (e.g. NHBE cells) or three dimensions (e.g. nasal, oral, gingival, and bronchial cultures that represent the different airway epithelia). A systems toxicology approach is used to gain mechanistic knowledge by combining advanced analytical and computational tools. In systems toxicology, quantitative systems-wide molecular changes in the context of an exposure are measured, and a causal chain of molecular events linking exposures with adverse outcomes is deciphered. Mathematical models are then built to describe these processes in a quantitative manner. The integrated data analysis leads to the identification of how biological networks are perturbed by the exposure and enables the development of predictive mathematical models of toxicological processes. 102
These new in vitro testing strategies require the development of new aerosol preparations and exposure techniques, ranging from the use of biocompatible solvents for aerosol fractions to complex whole aerosol exposure systems.
Aerosol exposure systems for in vitro studies: Submersed cell cultures
The in vitro toxicological testing of chemical compounds or mixtures of compounds commonly relies on dissolving a test material in an adequate solvent. This solution is then applied to the biological test system (e.g. a cell culture or a bacterial culture) at relevant concentrations.
Likewise, tobacco smoke assessment in vitro has traditionally relied on isolating CS fractions and assessing them in submersed cell cultures systems. Typically, the TPM fraction is captured on a CFP and eluted in dimethyl sulfoxide or ethanol, and AEs of whole smoke or GVP fractions are obtained by bubbling the aerosol through cell culture media or PBS, as previously described.
These in vitro toxicology tests have several advantages: they are rapid and relatively inexpensive, and they allow the screening of a multiplicity of rapidly evolving products. Additionally, the toxicity of individual components, such as additives and flavors, may be examined in these assays.
Standard toxicity assessment of cigarettes has traditionally relied on a battery of in vitro cytotoxicity, mutagenicity, and genotoxicity assays, including the neutral red uptake (NRU) assay, the Ames assay, the in vitro micronucleus test, and the mouse lymphoma assay (MLA). 104 THS 2.2 has also been evaluated using these assays. 21 In addition to these long-established tests, new in vitro assays are used to test the progress toward toxicity reduction by using physiologically-relevant biological models of the respiratory tract and human cardiovascular system. Endpoints are selected that are mechanistically linked to smoking-related chronic diseases and related to BoExp or early biological effects. 105
However, ultimately, assays using submersed cell cultures cannot, or can only to a limited extent, simulate the interaction between a native aerosol and human tissues. 106 –108 First, as previously discussed, the aerosol trapping and elution processes may profoundly change the physical and chemical properties as well as the bioactivity of the test material. Second, 2-D cell cultures cannot reproduce the spatial organization (cell–cell interaction and cell polarity) of relevant human tissues, in particular the human airway epithelia. Finally, medium covering the cells may impede the access of test materials to cell surfaces, bringing into play inherent solubility and diffusion properties with variable effects on the different smoke constituents. 109
Optimization of aerosol fraction trapping for in vitro mechanistic assays in submersed cell cultures
The optimization of trapping methods is critical for the adequate interpretation and comparison of in vitro exposure assays. 110 With the development of in vitro mechanistic assays using submersed cell cultures for HNB research, PMI has assessed the impact of various parameters on the efficiency of trapping of 3R4F and THS 2.2 TPM and AEs with a view to optimize them, namely test item number and trapping solvent volume (for TPM and AE) as well as sample collection point, trapping temperature, and surface contact material and area (for AE) (data not published).
For TPM, the efficiency of trapping was assessed by nicotine concentration, measured using a gas chromatography (GC) with a flame ionization detector (FID). For whole aerosol AE, it was assessed by the levels of eight carbonyls (acrolein, formaldehyde, acetone, crotonaldehyde, propionaldehyde, methyl-ethyl-ketone (MEK), butyraldehyde, and acetaldehyde) per test item, quantified by liquid chromatography (LC) with electrospray ionization mass spectrometry.
Table 3 describes the optimal systems and parameters for trapping 3R4F and THS 2.2 TPM and AEs in biocompatible solvents for testing in submersed cell cultures.
Trapping systems and parameters for the trapping of 3R4F and THS 2.2 TPM and AE.

3R4F: 3R4F Kentucky reference cigarette; AE: aqueous extract; THS: Tobacco Heating System; TPM: total particulate matter; CFP: Cambridge filter pad; PDSP: programmable dual-port syringe pump; PBS: phosphate-buffered saline; CS: cigarette smoke.
In the experiments described here, 3R4F CS and THS 2.2 aerosol were generated according to the HCI/ISO intense smoking regimen using rotary smoking machines, as these machines are used for in vitro and in vivo exposure studies. A stepwise approach was used. For TPM collection, the optimal number of sticks was first assessed by CFP saturation, as assessed by breakthrough on a second CFP. Filter saturation was reached with the fifth stick of THS 2.2, while for 3R4F, no breakthrough occurred up to 10 cigarettes. The lowest variability in nicotine concentration between the experimental replicates was obtained with the cumulative puffing of four THS 2.2 sticks and six 3R4F cigarettes. For elution, no difference in nicotine concentration in THS 2.2 and 3R4F TPM was observed when CFPs were eluted in 5 mL ethanol either by shaking or by using syringe-based extraction; hence, the less time-consuming shaking method is preferred.
Due to the physical separation between the smoking ports and the PDSP, the best collection point minimizing smoke/aerosol deposition or condensation was assessed. For whole smoke AE collection, the best point of collection for both THS 2.2 and 3R4F was upstream of the PDSP (behind the temperature-controlled insulation kit for THS 2.2), as it was optimal for all carbonyls, except propionaldehyde.
The optimal condition for trapping AE was using an ice-cold impinger filled with ice-cold PBS for both 3R4F and THS 2.2, as it was optimal for all carbonyls (except formaldehyde, which resides in the TPM fraction). The use of glass beads to increase the surface area contact between the solvent and the smoke/aerosol improved the trapping efficiency of five of eight carbonyls for 3R4F (formaldehyde, MEK, acetaldehyde, crotonaldehyde, and acetone). For THS 2.2, it was concluded that glass beads were not necessary, as they only slightly improved the trapping efficiency of four carbonyls, while in the absence of glass beads, the trapping efficiency of acrolein and butyraldehyde was much better, and that of MEK and crotonaldehyde was slightly better. Further optimization of the trapping method was investigated by varying the number of sticks (4, 6, 8, or 10 3R4F cigarettes and 8, 10, 15, or 20 THS 2.2 sticks) and the volume of the trapping solution (20, 25, 30, 36, or 40 mL PBS). The optimal PBS volume and number of test items, as determined by the levels of nicotine and eight carbonyls, were 36 mL PBS and 6 cigarettes for 3R4F, corresponding to approximately 1.8 puffs/mL, and 40 mL PBS and 10 sticks for THS 2.2, corresponding to 3 puffs/mL.
For in vitro uses, TPM and AE stock solutions must be prepared fresh prior to each experiment. They are diluted in cell culture medium and used for cellular exposure within 30 min of generation.
In vitro mechanistic assays using submersed cell cultures
More than 10 different assays have been established for HNB research, allowing the measurement of more than 15 different cellular parameters in cellular types representing the respiratory and the cardiovascular systems. 111 –117 The description of these assays is outside the scope of this review, and the studies can be consulted for full descriptions of the assay principles, study designs, and study endpoints. By way of example, Figure 9 shows the study design for a functional in vitro adhesion assay of THS 2.2 AE compared with 3R4F AE. 111
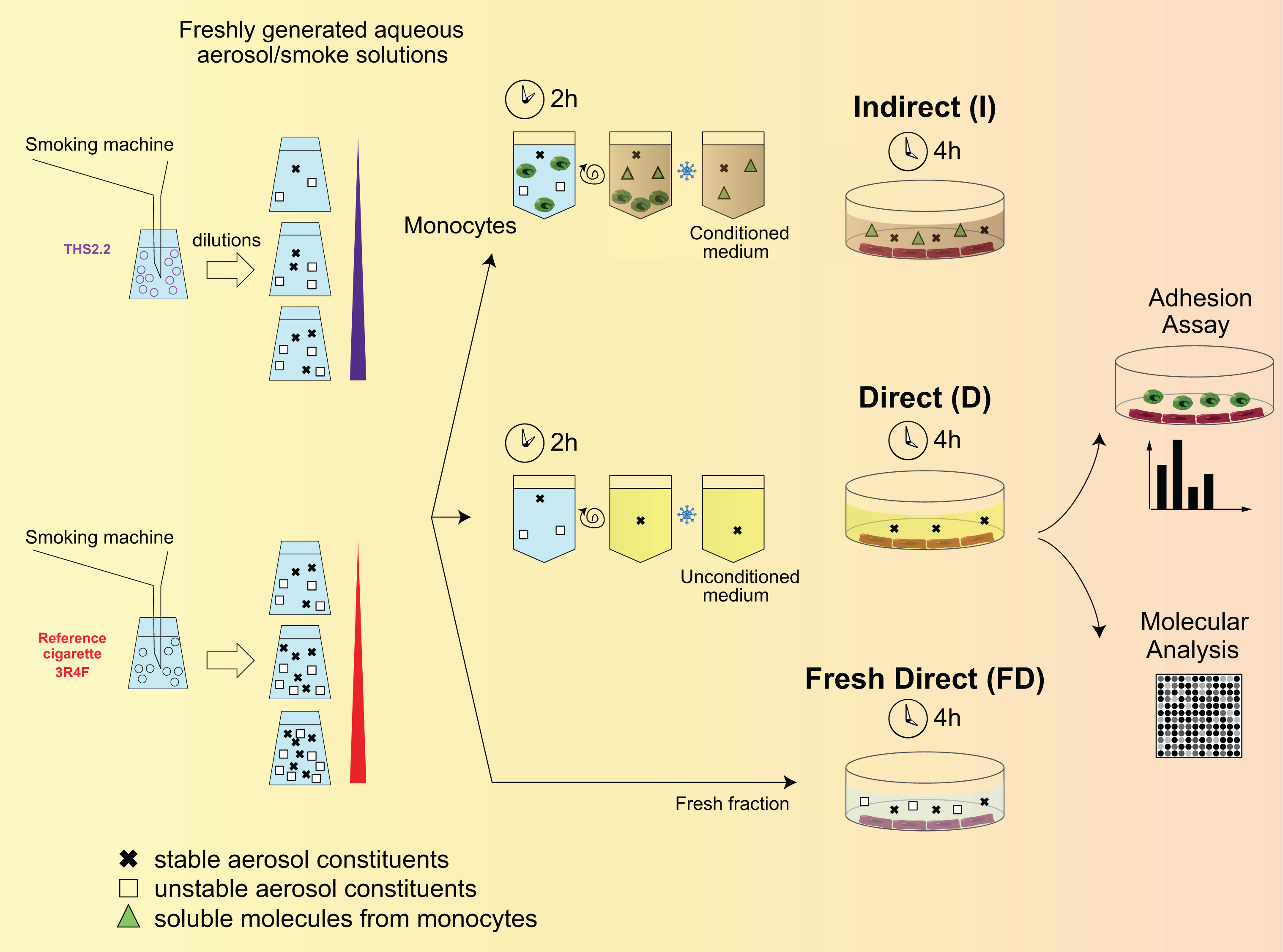
Functional in vitro adhesion assay of THS 2.2 aerosol AE compared with 3R4F CS AE. AE: aqueous extract; THS: Tobacco Heating System.
Dose-range-finding studies using real-time cell analysis
In order to determine the doses of aerosol fractions to be tested in vitro, dose-range-finding experiments are conducted. Such studies can be carried out using impedance-based real-time cell analysis (RTCA). RTCA uses tissue culture plates with sensor gold micro-electrodes covering the bottom area of each well. In the absence of cells, electric current flows freely through culture medium, completing the circuit between the electrodes (Figure 10(a)). The presence of the cells on top of the electrodes affects the local ionic environment at the electrode/solution interface, leading to an increase in the electrode impedance. The larger the number of cells attached to the electrodes, the larger the increases in electrode impedance. In addition, impedance readings are dependent on the quality of the cell interaction with the electrodes. For example, increased cell adhesion or spreading will lead to a larger change in electrode impedance. Thus, electrode impedance can be used to monitor cell viability and cell number. 118 –120 Dose–response curves for cell viability percentage may be plotted to calculate EC50 values (Figure 10(b)). RTCA can be used with several preparations relevant for HNB toxicity analysis, such as aerosol fractions or ingredients added to tobacco.
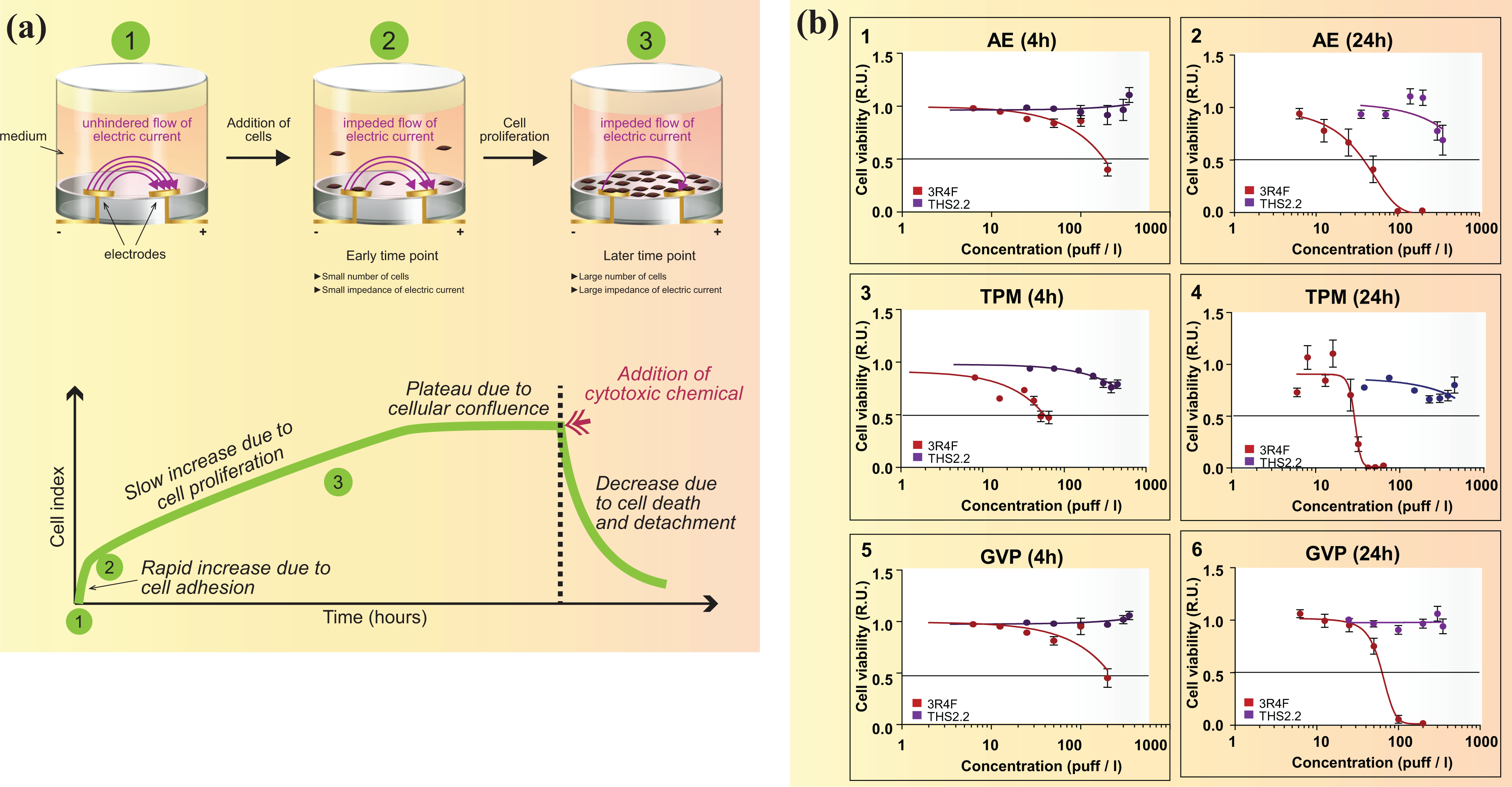
(a) RTCA. (b) Dose-range-finding study: Cell viability measured using RTCA in NHBE cells exposed for 4 or 24 h to AE (1 and 2), TPM (3 and 4), or GVP (5 and 6) fractions from 3R4F CS or THS 2.2 aerosol.
Aerosol exposure systems for in vitro studies: Exposure systems at the ALI
Whole aerosol exposure systems at the ALI
Technical progress has allowed the development of complex, three-dimensional, organotypic cell cultures that mimic the physiology of human tissues. Exposure of these advanced cellular models to aerosols or gases can be conducted at the ALI. Cells are seeded on porous membranes in culture inserts and are supplied with medium from their basal side, while their apical side is in contact with air. 108,121,122
Exposure systems at the ALI are more physiologically relevant than submersed exposure systems for the study of tissues exposed to airborne substances, as they better mimic the organization, functionality, and exposure of the human tissue counterparts. In addition, they allow exposure to whole aerosols, bringing the cells in direct contact with the GVP as well as the particulate phase. 123
However, they represent considerable methodological challenges. Aerosol constituents must be transferred from the aerosol to the cell surface to exert an effect on the biological test system. Due to the complexity of the physical processes governing aerosol transport, dilution, and deposition, the physicochemical characteristics of the aerosol delivered to the biological test system cannot be predicted based on the characteristics of the aerosol generated. Determining the actual delivery efficiency of these exposure systems is therefore critical for the interpretation and comparison of exposure experiments.
A complete exposure system requires an aerosol generator, a dilution/distribution system, and an exposure chamber that houses the biological culture.
A few exposure systems are presently available that enable CS and HNB aerosol exposure of cell cultures at the ALI, including commercial and bespoke systems. 124 Each system offers unique advantages and disadvantages.
The VITROCELL® 24/48 exposure system (VC 24/48)
The VC 24/48 (VITROCELL Systems GmbH, Waldkirch, Germany) has been characterized and is used for HNB aerosol exposure studies at the ALI. The system is schematically represented in Figure 11. A cultivation base module houses the cell cultures. Up to 48 wells can be exposed simultaneously in individual exposure chambers grouped into 8 rows of 6 replicate positions. The cultivation base module is placed on a heating plate to maintain adequate temperature for the cells.
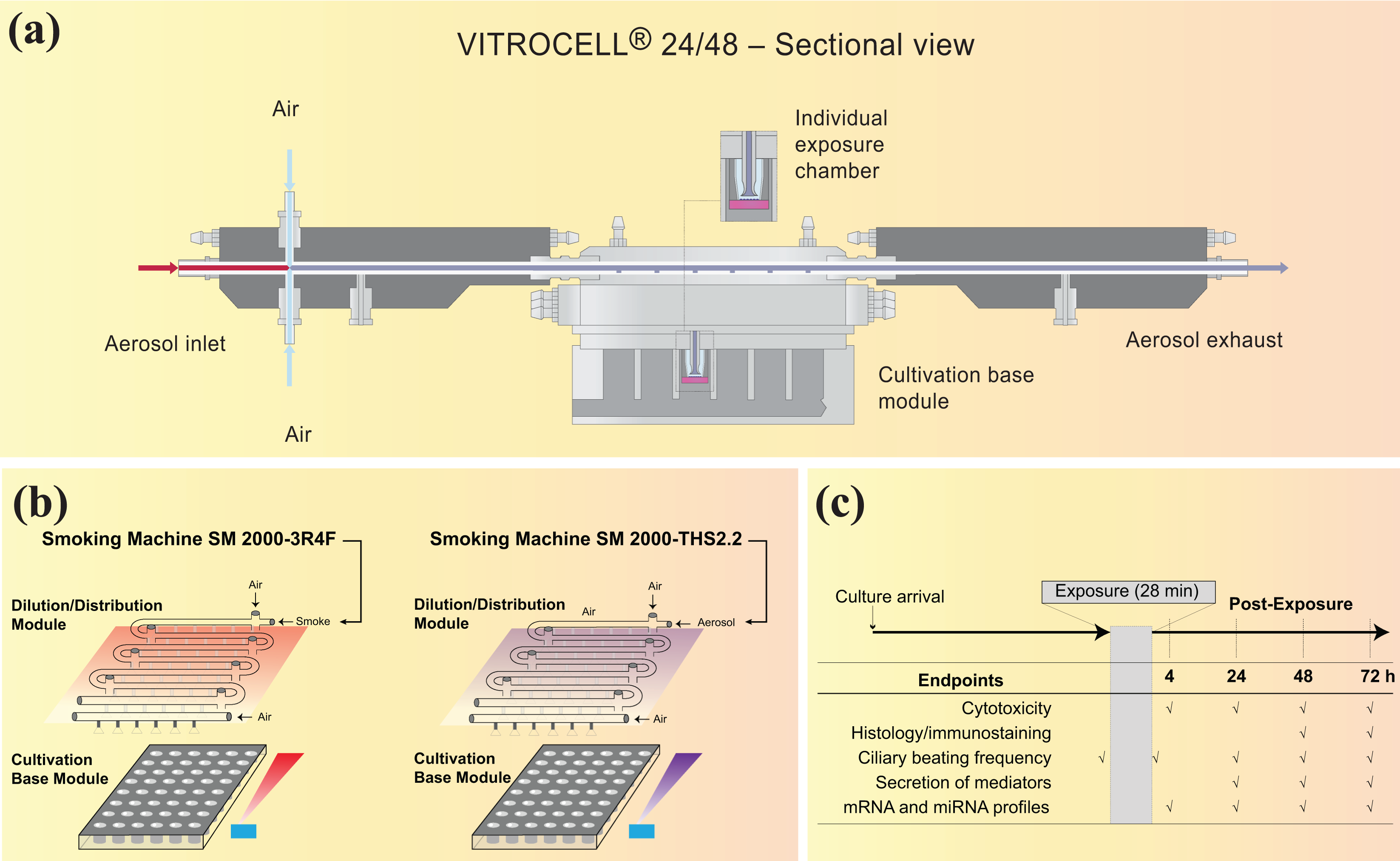
VC 24/48. (a) Schematic cross-sectional view of one row showing the dilution/distribution module, the cultivation base module, and individual exposure chambers (
The test aerosol passes through a dilution/distribution pipe located on top of the base module. The pipe has a diameter of 6 mm and goes back and forth over the eight rows. At the beginning of each row, the aerosol can be diluted, resulting in a total of eight dilutions that can be tested simultaneously (one per row). Under standard operating conditions, only seven dilutions are tested, while the eighth row is used for negative control exposure to clean air only and is not connected to the aerosol supply. Each dilution system consists of two opposing dilution pipes of modifiable diameter attached to the aerosol distribution pipe in a double-tee junction. The aerosol is diluted, generally to target nicotine concentrations, with filtered, conditioned fresh air (60 ± 5% RH) entering the dilution/distribution system through inlets at the beginning of each row. A VITROCELL® Humidification Station (VITROCELL Systems GmbH) connected to the air inlets is used for the humidification of the dilution air and the control air (sham). Flow rates ranging from 0 to 3.0 L/min are used for aerosol dilutions.
The aerosol is partially sampled by negative pressure, applied by a vacuum pump connected to the cultivation base module, into each individual exposure chamber through trumpet-shaped delivery systems protruding from the dilution/distribution system into the wells. Each 3 mm diameter trumpet is connected to the dilution/distribution system at a 90° angle and projects 2 mm toward the well. The sampled aerosol flows through a trumpet and passes over the biological sample, where it partly deposits, before the rest of the aerosol is exhausted from the system. The unsampled part of the aerosol continues along the main pipe. 39 The flow rate through individual trumpets should be 2 ± 0.2 mL/min. In order to confirm system tightness, the airflow leaving the system must equal the dilution airflow entering the system, minus the airflow passing the exposure trumpets (target value ± 10%). At the end of each dilution row, one trumpet allows the sampling and real-time monitoring of aerosol mass deposition by quartz crystal microbalances (QCM), as described in the “Online monitoring of particle mass deposition using QCM” section.
The dilution/distribution system and cultivation base module are housed in a climatic chamber.
Depending on the aerosol at study, a VC 24/48 is connected to an SM 2000 CS (for CS exposure) or an SM 2000 THS 2.2 (for THS 2.2 aerosol exposure). For a comparative study, independent generation and exposure setups are used in parallel. The aerosols are generated as previously described and are delivered to the VC 24/48 dilution/distribution system through a fluoroelastomer tube (ISO-Versinic®, Saint-Gobain, Courbevoie, France). The distance to the cell cultures is kept as short as possible in order to maintain the integrity of the aerosol prior to exposure. To account for system memory effects, the first exposure run on each day of exposure is conducted with clean air and PBS-loaded culture inserts in rows 1 and 7 only. 72
Cells are grown on cell inserts (pore size of 0.4 µm) and then maintained to complete differentiation for durations and in conditions depending on the cell type. Once differentiated, cultures are incubated in 24-well plates. Before exposure, cell culture medium is removed from the apical side of each insert.
Cell culture inserts and dummy stainless steel cell culture inserts with 100 µL PBS are placed into the cultivation base module, heated to 37°C, and the loaded cell cultivation base is placed into the climate chamber. Usually, each row is loaded with three cell culture and three dummy inserts, for three replicates per aerosol concentration. Once exposure is complete, cell culture inserts are placed into 24-well cell culture plates, with 0.7 mL of prewarmed culture medium per well, and into the incubator (37°C, 5% CO, 90% RH) prior to analyses.
To verify proper aerosol generation and delivery to the system and to assess the level of exposure of cells, nicotine aerosol concentration (by trapping in EXtrelut® 3NT columns, see “Determination of nicotine in the aerosol by trapping on EXtrelut® 3NT columns” section) and the deposition of carbonyls in PBS (see “Determination of carbonyls deposited in PBS” section) are determined in separate experimental runs.
The VITROCELL® 24/48 exposure system: Characterization
Understanding how an in vitro aerosol exposure system delivers a test aerosol to the biological test system is a crucial prerequisite for the interpretation and comparison of exposure experiments and relies on detailed exposure system characterization. Given the different properties of aerosols generated by different products, system performance should be characterized for each type of aerosol. Key parameters include achievable dose delivery, dosing accuracy (the accurate translation of aerosol dilution to aerosol delivery), physicochemical composition of delivered aerosol, uniformity of aerosol delivery to replica positions, and the stability of system performance across individual repetitions (reproducibility).
Exposure system characterization can be approached in two different ways: by comparing the aerosol entering the system (or a part of the system) with the aerosol leaving the system (or a part thereof) or by direct quantification of aerosol deposition. The former approach allows measuring aerosol evolution and deposition online, simultaneously with the exposure of the biological test systems. However, it assumes sufficient measurement sensitivity to detect differences and suffers from low spatial resolution, as the exact location within the system at which the observed changes arise cannot be determined. Direct quantification of aerosol deposition does not suffer from these limitations. However, although it can be performed online, it is usually performed after exposure or in separate experiments specifically dedicated to system characterization, 124 –126 with the obvious limitation that actual experimental conditions are not the ones determined.
System characterization can be done using cell cultures or a surrogate matrix, such as PBS or culture medium. Large-scale production of organotypic cell cultures is expensive. Because of the low complexity, the stability, the absence of metabolic activity, and the absence of transepithelial transport, the gross aerosol deposition during PBS exposure experiments can be determined more easily and more accurately than by the post-exposure chemical analyses of living cells. Therefore, surrogate matrices are generally preferred.
Majeed et al. characterized VC 24/48 for use with CS. 127 They assessed and compared aerosol deposition of different CS concentrations as determined by three different approaches: (1) a WST-1 colorimetric assay, based on the previous observation that WST-1 is reduced upon exposure to CS, presumably because of CS high oxidant concentration; (2) the determination of eight carbonyls trapped in PBS; and (3) QCM-determined particle mass deposition. The authors found that a given CS concentration reduced WST-1 diluted in Dulbecco’s Modified Eagle Medium, as measured by changes in optical density (OD) at 430 nm, in all culture inserts to a similar level. They also observed concentration-dependent changes in OD with different dilution flow rates. Trapped carbonyl concentrations were well correlated with the applied CS concentration. The particle mass deposition measured by QCMs attached to different rows of the dilution/distribution system was also found to be similar at the fixed CS concentration. The authors concluded that the VC 24/48 is well suited for CS exposure of cells growing at the ALI.
Yet, Steiner et al. demonstrated that the delivery efficiency of individual CS constituents may vary by several orders of magnitude and that, consequently, characterization of the aerosol used in in vitro exposures does not necessarily give an accurate description of the aerosol fraction that ultimately enters and interacts with the biological test system. 128 The authors provided a large set of delivery efficiencies that describe the conversion of an applied dose of CS to a delivered dose during in vitro exposure. These delivery efficiencies provide a simple dose metric tool that allows the characterization and comparison of aerosol exposure experiments with respect to the composition of the aerosol presented to the biological test system.
Sampling and monitoring aerosol exposure and delivery to the VC 24/48 exposure chamber
Online monitoring of particle mass deposition using QCMs
The monitoring of mass deposition using QCMs relies on the resonant oscillation frequency of piezoelectric crystals, which changes with the mass adhering to the crystal. As particles deposit on the crystal’s surface its mass loading, and thus its natural oscillation frequency, changes. 126 The proportionality between a change in the resonant frequency and the adherent mass is described by the Sauerbrey equation (equation (1)):

Δ
QCMs are capable of quantifying changes in particle load in real time and within the nanogram range. 129
During an exposure session, tobacco stick-to-tobacco stick patterns should be visible in the reported mass deposition, identical in all rows, and in line with the aerosol generation. The mass deposition in the different rows should also reflect the aerosol dilution applied in the corresponding rows.
Determination of nicotine in the aerosol by trapping on EXtrelut® 3NT columns
For the determination of nicotine concentrations in the diluted aerosol, aerosol samples are drawn from the VC 24/48 dilution/distribution system through EXtrelut® 3NT columns (Merck, Zug, Switzerland) during separate aerosol generation runs. 127 EXtrelut® 3NT columns use a chemically inert, wide-pore, and highly pure diatomaceous earth-based solid phase. The method allows the determination of nicotine present in both the GVP and particulate phase. EXtrelut® 3NT columns are also used to determine nicotine concentration in animal exposure chambers, as described in the “Determination of aerosol uptake” section. Figure 12 shows the experimental setup for trapping nicotine using EXtrelut® 3NT columns with the VC 24/48.
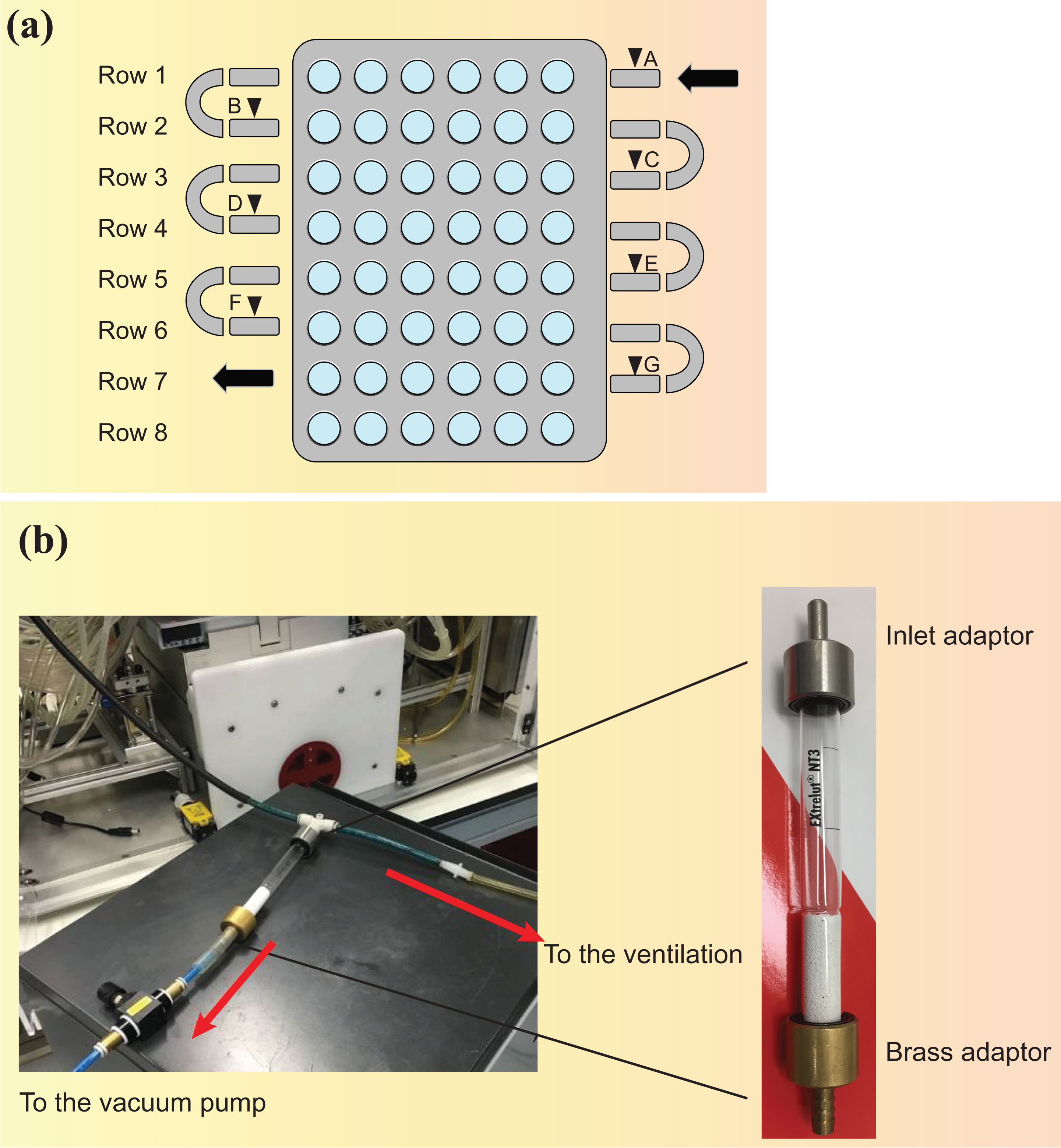
Experimental setup for trapping nicotine using EXtrelut® 3NT columns with the VC 24/48. (a) Layout of the cultivation base plate module, dilutions points (black triangles) and EXtrelut® 3NT sampling points (black arrows). (b) View of an EXtrelut® NT 3 columns setup.
Aerosol trapping on EXtrelut® 3NT columns cannot be done simultaneously while exposing cell cultures because an amount of aerosol is removed from the VC 24/48. Hence, dedicated exposure sessions must be carried out. The VC 24/48 is prepared as for a regular experiment, except that 10 mL deionized water is added to each row of the cultivation base module (no cell culture insert). For each dilution, three replicates are sampled from the dilution/distribution system.
The diluted aerosol is sampled from the dilution module prior to each dilution point (Figure 12(a)). An EXtrelut® 3NT column is connected via a needle valve (Aarlborg, 0–10 L/min brazen valve), a column adaptor, and a vacuum pump (Figure 12(b)). Trapping starts after 1 ± 0.5 min of aerosol generation, once the VC 24/48 is equilibrated. The aerosol flow is drawn through the EXtrelut® 3NT column by negative pressure applied downstream of the column for 5 min. Sampling flow rates of 0.1–1 L/min are applied depending on the aerosol dilution (low flow rate for high aerosol concentration) and are adjusted to not affect the flow through the system. A controlled, defined volume flow rate passes the EXtrelut® 3NT column, and excess aerosol coming from the system is passively removed through the exhaust system.
After trapping, EXtrelut® 3NT columns are sealed on both ends with parafilm and stored at 4 ± 1°C until analysis. The nicotine trapped on the column is eluted on the same day with 3 × 5 mL of a 5% (v/v) mixture of triethylamine in
Determination of carbonyls deposited in PBS
Aerosol deposition can be determined in specific PBS exposure experiments. PBS provides a good model for the thin film of an aqueous matrix commonly covering the surface of cell cultures. Furthermore, due to its low complexity, stability, and the absence of metabolic and transport activities, PBS provides an easy, accurate, and cost-effective surrogate method to determine gross aerosol deposition.
Carbonyl compounds are an important category of GVP chemicals and an important class of marker compounds for in vitro and in vivo exposure to CS. Their high volatility makes these harmful constituents difficult to analyze. The most common approach is to prepare stable derivatives of trapped carbonyls with 2,4-dinitrophenylhydrazine (DNPH), which can be analyzed by high-performance liquid chromatography (HPLC).
The method described here allows the quantification of 8 carbonyl compounds (formaldehyde, acetaldehyde, acetone, acrolein, propionaldehyde, crotonaldehyde, MEK, and butyraldehyde) in aerosols trapped in PBS solutions. 130 It has been validated over wide ranges of concentrations and has allowed the quantification of the carbonyl compounds in aerosols generated by HNB tobacco products, where the content of these harmful molecules is substantially reduced compared with CS. The number of cigarettes/tobacco sticks or the PBS volume can be modified to ensure a proper quantification of these very low-level compounds.
Carbonyl trapping in the VC 24/48 exposure chamber is carried out in a dedicated experiment. These blank runs are conducted for the same duration as the study cell exposure runs. Three to four replicates are generated on different days.
Dummy cell culture inserts are placed in the cultivation base module and filled with 18.5 mL of PBS per row so that the liquid surface in each well is at a similar height and distance from the delivery trumpet as the apical side of a typical cell culture. The first exposure run on each day of exposure is conducted with clean air instead of aerosol to account for any deposited aerosol on internal system parts. Immediately after exposure, aliquots of exposed PBS are collected from each cell culture insert and pooled row-wise into silanized amber glass vials. Samples are stored at −80°C until chemical analyses are performed.
An aliquot of PBS-exposed sample is incubated with 3:2 (v/v) of a 15 mM DNPH solution (> 90.0% purity, Acros Organic) for 30 min at room temperature. Chemical derivatization is then quenched by addition of 1:20 (v/v) pyridine after 30 min. In the case of unsaturated aldehydes, such as acrolein and crotonaldehyde, the derivatization reaction might produce bi- and tri-derivatized adducts. Quenching with pyridine at 30 min allows stabilizing the sample solutions while avoiding the formation of poly-derivatized carbonyl adducts.
A 500 µL of derivatized sample is transferred to a liquid chromatography–mass spectrometry (LC-MS) glass vial with 485 µL acetonitrile and 15 µL internal working standard solution containing 24 µg/mL each of acetone-d6-DNPH (ISTD for formaldehyde, acetaldehyde, and acetone), propionaldehyde-d2 (ISTD for acrolein, propionaldehyde, and crotonaldehyde), and MEK-d5-DNPH (ISTD for MEK and butyraldehyde). Samples are diluted with a DNPH/PBS/pyridine solution prior to injection with higher dilutions for samples with higher expected levels of carbonyls, such as 3R4F samples.
LC is performed with an Agilent 1200 series (Agilent Technologies, Santa Clara, California, USA) HPLC coupled to a Sciex Triple Quad 5500 mass spectrometer (AB Sciex, Framingham, Massachusetts, USA) with an electrospray ionization probe operating in negative mode. Separation of the aldehydes is carried out in the isocratic mode on a Chromolith Speedrod RP-18e HPLC column (Merck KGaA, Darmstadt, Germany) using water, acetonitrile, tetrahydrofuran, and isopropanol (59:30:10:1, v/v/v/v) at a flow rate of 2.5 mL/min (column set at 40°C and equipped with a post-column splitter 1:6 before entrance into the LC-MS). LC-MS detection is performed in multiple reaction monitoring mode. The carbonyl compound concentration, expressed as µg/test item, is calculated using an external calibration curve.
Online aerosol characterization within exposure systems using soft ionization TOFMS (Photonion)
Offline chemical determination methods have several limitations. As, by definition, they are not carried concurrently with the actual studies, the representativeness and reproducibility of the generation and exposure is not guaranteed. They are time-consuming given the multiple process steps required, including trapping, extraction, and measurement. Hence, there is a need for the development of methods for sampling and analyzing complex aerosols online during exposures in close proximity to the biological test system.
Photon ionization (PI) TOFMS is an established technology for online, puff-resolved characterization of organic compounds in CS. 131,132 The method uses a combination of PI and TOFMS to accelerate and separate ions by mass. Known aerosol constituents can be identified and quantified based on their molecular mass. Many constituents can be detected in real time with single-puff resolution in CS.
The single PI TOFMS (Photonion GmbH, Schwerin, Germany) can be used with the VC 24/48 for the online analysis of THS 2.2 aerosol. The diluted aerosol is sampled from the dilution/distribution system, as shown in Figure 13. A heated (up to 300°C) sampling capillary of 180 µm inner diameter is inserted into the tube and samples the aerosol at a rate of 3–5 mL/min. The samples are ionized by vacuum ultraviolet light of 120–160 nm (ionization energy of approximately 10.3 eV) and enter the TOFMS by linear extraction. Mass spectra are reported at a frequency of 1 Hz, and the covered mass range is 10–2,000 m/z. Absolute quantification is based on compound-specific cross-sections (ionizabilities) relative to toluene, determined using 100 ppm reference gas. PI results in only limited fragmentation of analytes, hence known aerosol constituents can be identified and quantified based on their molecular mass. Yet, the risk of biased quantification in the presence of isobaric molecules should be addressed (e.g. by the identification of mass fingerprints for compounds for which fragmentation occurs). 133
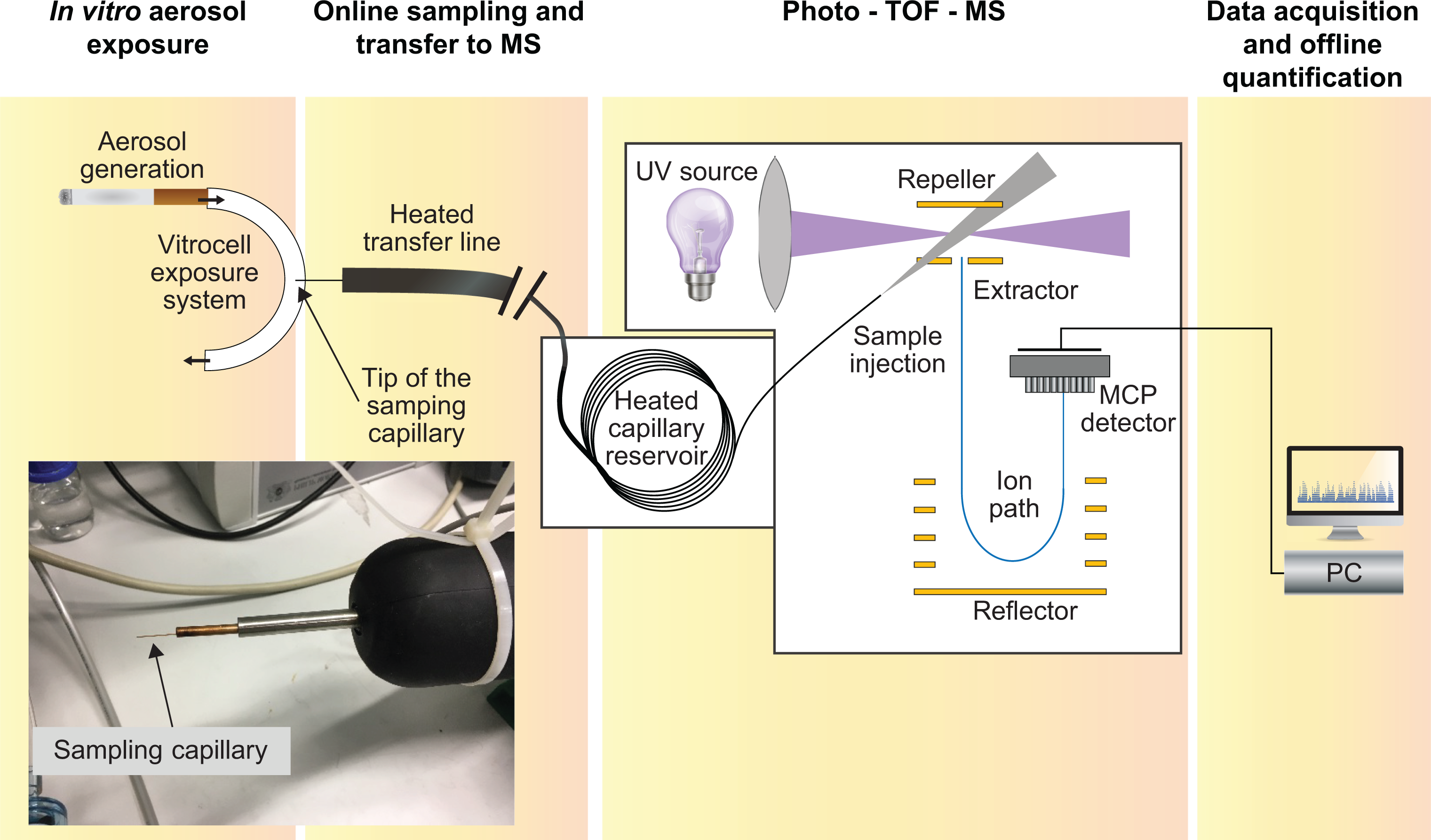
Schematic representation of the Photonion experimental setup. MCP: microchannel plate.
The VITROCELL® 24/48 exposure system: Validation
The VC 24/48 exposure system is being validated for the exposure process of three-dimensional, organotypic cell culture inserts with CS and with aerosols generated from HNB tobacco products and e-liquids. The strategy for the validation of a complex workflow such as the VC 24/48 exposure process requires a structured approach and splitting the process into small entities which can be managed independently. These include such diverse processes as operator training, standard operating procedures creation and updates, the qualification of the aerosol generators or of the aerosol trapping methods. Computerized systems and equipment involved in the execution of analytical methods and exposure process activities have to be qualified or validated before the start of the activities.
System validation usually uses three specific testing protocols: Installation Qualifications to verify that systems have been properly installed and that the configuration is correct; Operational Qualifications to verify that systems perform as expected; and Performance Qualifications or User Acceptance Testing to verify that systems perform tasks in real-world conditions.
Previously validated processes are used for the validation of the VC 24/48 exposure system, including, for example, aerosol characterization processes, such as the ones described in the “Determination of nicotine in the aerosol by trapping on EXtrelut® 3NT columns” section (quantification of nicotine in diluted and undiluted aerosols) or “Determination of carbonyls deposited in PBS” section (determination of eight carbonyls trapped in PBS), as well as a set of validated bioanalytical methods to support criteria for validity and reproducibility.
Aerosol exposure systems for animal inhalation studies
Despite great progress in the development of complex in vitro surrogate models for in vivo inhalation studies, such as described earlier in this review, these systems cannot yet fully mimic the complex physiological systems of whole living organisms. Animal studies are therefore still needed, and rodent inhalation studies are a key element of cMRTP assessment, as clinical trials with smokers can be initiated when it has been demonstrated from the animal studies that the cMRTP has equal or lower toxicity than cigarettes, and that there are evidences of reduced risk of the cMRTP aerosol compared with that of CS.
Animal use in cMRTP assessment is restricted to situations where there are no other alternatives available, or if required by regulation. Therefore, the development of nonanimal alternatives is a priority. All procedures involving animals carried out by PMI and described in the following sections are performed in an American Association for the Accreditation of Laboratory Animal Care-accredited, Agri-Food & Veterinary Authority of Singapore-licensed facility with approval from an Institutional Animal Care and Use Committee and are performed in compliance with guidelines set by the National Advisory Committee for Laboratory Animal Research Guidelines. 134
Animal inhalation studies – Study design principles and key requirements
OECD Test Guidelines (TG) provide a set of internationally accepted study designs, methods, and endpoints for assessing, identifying, and characterizing toxicity by the inhalation route. The main OECD guidelines applicable to cMRTP aerosol research are guidelines for acute (OECD TG 403:2009 135 ), subacute (OECD TG 412:2018 73 ), subchronic (OECD TG 413:2018 74 ), and chronic inhalation toxicity studies (OECD TG 452:2018 136 ) as well as for combined chronic toxicity/carcinogenicity studies (OECD TG 453:2018 75 ).
A challenge with the standard toxicological endpoints recommended by OECD guidelines is that they are often detectable only after substantial damage has already occurred and therefore can lack sensitivity. 137 Furthermore, they provide limited mechanistic insights into changes that occur upon HNB aerosol exposure compared with CS exposure.
Thus, in order to increase sensitivity and better understand underlying mechanisms, cutting-edge research is complementing standard OECD-recommended endpoints with systems biology approaches using transcriptomics, proteomics, and lipidomics. 102,138 These “OECD plus” endpoints allow the detection of early events in disease etiology and of differential effects between CS and cMRTP aerosol exposure. Analyses are performed either by using organs and body fluids collected for assessing OECD endpoints or as additional study arms on animals exposed concomitantly with the animals used for the determination of the OECD-recommended endpoints. Eventually, knowledge gained using systems toxicology could be applied to reduce the need for animal studies.
In vivo inhalation studies require exposure systems with the capability of consistently producing and delivering large amounts of aerosols at concentrations that are stable during prolonged exposure periods and with appropriate physical properties to enable efficient inhalation and uptake.
Dynamic single-pass flow-through exposure systems
Inhalation exposure systems can be either static (no airflow) or dynamic (airflow). In a static inhalation exposure system, a given quantity of test material is introduced into a closed system and allowed to mix with the trapped air. These systems are limited by depletion of oxygen, accumulation of waste, and loss of test agent over time, and they are generally unsuitable for most inhalation studies under current standards. Today, most inhalation exposure systems are of the dynamic type. In a dynamic, single-pass, flow-through exposure system, the test atmosphere is continuously delivered to, and removed from, the animal chamber. The test material is not depleted, and waste does not accumulate. These systems allow for long-term inhalation studies with a large number of animals. However, they consume larger quantities of test material than static systems and require more complex experimental setups. 139
Figure 14 shows the key elements of a dynamic exposure system (Figure 14(a)) illustrated by an actual experimental setup (Figure 14(b)), further described in the following sections.
(a) Key elements of a dynamic exposure system. (b) Experimental setup for a rodent inhalation study. NOEC: nose-only exposure chamber; WBEC: whole-body exposure chamber.
Aerosol generation
For in vivo inhalation studies, aerosol generators should be capable of continuously producing large amounts of aerosols in a reproducible manner over the long term, as described in the “HNB aerosol generation systems and methodologies” section.
Depending on the volume of the exposure chamber and the target aerosol concentration, one or several generators are needed for one exposure chamber.
Aerosol dilution
The aerosol is diluted with conditioned filtered air to obtain the target exposure concentration (generally expressed in target nicotine concentration) and volumetric flow rate required by the chamber. In order to avoid particle growth, dilution takes place as close as possible after generation (i.e. immediately after the PDSP).
Aerosol transportation
In order to safeguard the required environmental and safety conditions in the animal exposure room, aerosol generation and animal exposure are carried out in separate rooms (the control and exposure rooms, respectively). The aerosol must therefore be transported from one room to the other. To minimize the risk of chemical and physical changes during transportation, and to allow visual inspection for deposition and condensation, the transportation tubing is made of borosilicate glass. Tubes of different diameters (15, 25, and 40 mm diameter) are used to handle different aerosol flow rates (≤50 L/min, ≤250 L/min and ≤500 L/min, respectively). A higher flow velocity provides better mixing and less deposition but potentially increases losses due to impaction at areas where there is a pressure drop (e.g. bends). The transportation tube length must be kept to a minimum and its route the straightest possible. The transportation routes should be similar for all the animal exposure setups used in parallel in a given study to avoid experimental artifacts.
Exposure chamber
Various types of chambers have been developed to expose experimental animals via the inhalation route. These include whole-body exposure chamber (WBECs), head-only exposure chambers, nose-only exposure chamber (NOECs), and lung-only (intratracheal) and partial-lung exposure systems. 27 OECD TGs expressly mention WBECs and NOECs. 98 Exposure chambers are mainly constructed of inert materials, such as stainless steel, glass, and plastics, to minimize chemical and physical alteration of the aerosol.
Air flow rate
The air supply to the chamber must be operated at an adequate flow rate, substantially exceeding the ventilation requirements of the animals, to ensure adequate air changes and oxygen availability and to avoid rebreathing, suffocation, and temperature and humidity build-up. The number of required air changes depends on the type of exposure chamber used. 98
Figure 15 shows the key parameters used to determine the dilution flow requirements of a study.
Key parameters used to determine the dilution flow requirements of a study (air flow at chamber inlet,
Typical values of air flow at chamber inlet (
Typical values of air flow at chamber inlet (

FPC: flow-past chamber; NOEC: nose-only exposure chamber.
a
The flow from each generator,
Chamber equilibration
After an initial buildup of the test material in the chamber, a stable equilibrium concentration is approached and maintained. 140 The time to reach 95% equilibrium is a function of the chamber volume, the chamber flow rate, and the type of aerosol. The time to reach equilibrium differs for gas, liquid, and solid phase components. Constituents that exist predominantly in the GVP (e.g. CO) tend to require shorter durations to reach the equilibrium concentration and display better temporal and spatial homogeneity compared to multiphase constituents, which exist predominantly in the liquid or solid phase or are partitioned between the two phases.
This is due to the higher depositional losses associated with non-GVP constituents in the aerosol due to their tendency to impact, coagulate, and deposit. Hence, the concentration ratio of test aerosol components in large exposure chambers could be altered, with relative disproportionation of some components. 141,142 Due to their different characteristics, CS and HNB aerosols are likely to be differently impacted.
The time to reach equilibrium should be kept as short as practically feasible in relation to the total duration of exposure or be adequately taken into account. The duration of an exposure usually spans the time that the test chemical is generated. This takes into account the time required to attain chamber equilibration (
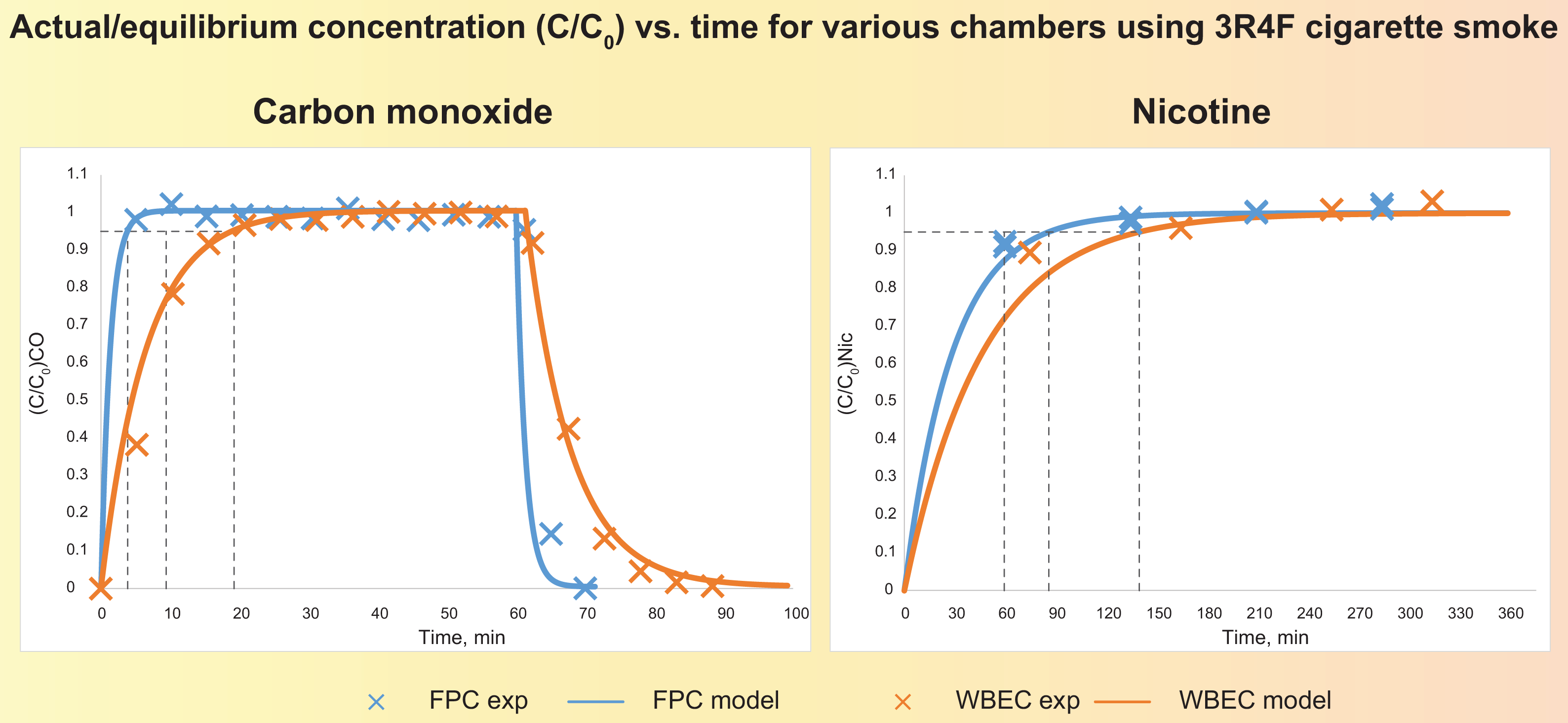
Concentration time curves for 3R4F CS CO and nicotine in various chambers. FPC: flow-past chamber; 3R4F: 3R4F Kentucky reference cigarette; CS: cigarette smoke.
Adaptation period
At the beginning of a study, animals are allowed a period to adapt to the aerosol (dose adaptation or dose acclimatization). Adaptation is typically performed by applying time escalation, concentration escalation, or a combination of both, to eventually achieve the nominal dose (time × concentration). The choice is depending on the study objective, test item, animal strain, and type of exposure chamber. For time adaption, animals are exposed to the final target concentration for increasing exposure durations over a period of 1–2 weeks. For concentration adaption, typically applied to avoid irritation-induced respiratory depression, 144 animals are exposed during the full exposure duration to increasing test concentrations in the chamber over a period of 1–2 weeks. 145 When tube restraint is used in NOECs, a tube restraint adaptation period is also recommended. 74
Test atmosphere sampling and monitoring
Test atmosphere characterization is critical for understanding and interpreting data from animal inhalation exposure experiments. Achieving the target concentration and the target PSD is of primary importance, as is their day-to-day reproducibility. OECD TGs provide that an individual measurement in the chamber should not deviate from the mean chamber concentration by more than 10% for gases or by more than 20% for liquid or solid aerosols. 98 In practice, in THS 2.2 inhalation studies, this requirement is fulfilled when the daily mean measurement falls within 15% of the study mean.
Sampling for analytical characterization is carried out at sampling ports close to the animal breathing zone at various frequencies, depending on the parameter and the exposure group. Typical sampling and analytical conditions are described in the “Test atmosphere sampling and characterization” section and include determination of TPM, nicotine, CO, carbonyls, and PSD.
The temperature (22 ± 3°C), RH (30–70%), 98 CO, dilution air flow, and pressure are continuously monitored and controlled using field instruments coupled to a supervisory control and data acquisition system (SCADA) (e.g. Wonderware by Aveva, Lake Forrest, California, USA). Data are processed (Application Server) and stored in a database (Historian Server) backed up daily (Figure 17).
Data acquisition and processing system. I/O: input/output.
Aerosol exhaust
The test atmosphere exiting the chamber is filtered twice, using a F7 fine dust filter conforming to EN779 (Trox® Teknik) followed by an activated carbon filter, before being released into the environment. The particulate filter removes solid and liquid particles, while the activated carbon filter removes gaseous compounds.
Exposure chambers
The following sections describe WBECs and NOECs used in THS 2.2 aerosol research. The referenced studies are published and provide full descriptions of study designs, animal procedures, and study endpoints.
Whole-body exposure chambers
Whole-body exposure systems employ chambers in which test animals are free to move, in groups or isolated. Their entire body is immersed in the test atmosphere.
The advantages of WBECs over NOECs include the ability to expose large numbers of animals simultaneously with limited daily animal procedures (no insertion and removal). The animals are unrestrained, which results in minimal stress. While feed is withdrawn during the daily exposure period to minimize oral uptake of deposited aerosol constituents, animals have access to water.
On the downside, WBECs require large quantities of test materials and air throughput. Nonhomogeneous distribution of concentration and particle size is more likely to occur within a large WBEC chamber than in NOECs. 146 Animals are exposed to the test material via other, confounding routes of exposure, including transdermal uptake following fur deposition and surface contact, oral uptake from grooming, and ocular exposures. 147 When several animals are housed together, they may huddle, leading to varying uptake of the test material, and filtration may occur as rodents breathe through own or mates’ fur. 27 The test atmosphere can be contaminated by the animal’s activities and excreta.
WBECs come in a wide variety of sizes and designs, from very large, room-sized chambers hosting up to 200 animals to small chambers that hold one animal. 54 In most WBECs, the test atmosphere is introduced at the top of, and exhausted below, the exposure zone. In the following sections, we describe two WBECs characterized and validated for use in murine inhalation studies of THS 2.2 aerosol.
800-L 24-cage WBEC for mouse inhalation studies
The 800-L, 24-cage WBEC is a stainless steel and glass inhalation chamber that can house up to 8 mice per cage for a total of 192 mice (Figure 18). The chamber has been developed and widely used by PMI for CS inhalation studies before being adapted to the study of HNB tobacco products.

800-L, 24-cage WBEC and sampling ports. WBEC: whole-body exposure chamber.
The aerosol is delivered to the WBEC using three vertical glass pipes running at the back of the chamber. Each vertical glass pipe has eight holes. Each hole is aligned to the top of a cage to deliver the aerosol directly into the cage. The holes on the vertical glass pipe are offset by 45° sideways to prevent the aerosol exiting the glass pipes from impacting the front door of the WBEC perpendicularly, thereby reducing losses through impaction and deposition. To prevent artifacts arising from any potential spatial inhomogeneity, the position of the cages in the chamber is rotated typically once per week.
Eight sampling ports are available at the back of the WBEC, in the middle row, to collect samples for online monitoring and aerosol characterization. The aerosol flow rate delivered to the 800 liter WBEC after accounting for sampling is ≥133 L/min to sustain ≥10 air changes per hour. 98 The WBEC is operated under a positive pressure of 20 to 60 Pa to help ensure that the entire volume of the WBEC is filled by the test material, thereby improving spatial homogeneity.
Typically, mice are whole-body exposed for 6 h/day, 5 days/week. In order to avoid excessive CO exposure with CS, intermittent exposure may be used, whereby mice are exposed 6 × 1 h to CS with breaks of 30 to 60 min in between, during which animals are exposed to fresh filtered air.
Nose-only exposure chambers
NOECs are designed to expose test animals via the inhalation route while preventing confounding exposures via noninhalation routes. OECD TGs recommend that NOECs be used in inhalation toxicity studies whenever possible. The test animals are held in exposure tubes attached to a central chamber so that only the nose is exposed to the test atmosphere. The exposure tubes are preferably made of glass, which provides efficient transfer of the animal body heat to the surroundings and allows animal observation. An adjustable, air tight back restraint (plunger) is used to prevent both the animal from backing out and aerosol leaks (Figure 19(c)). The deposition of aerosol particles on the skin and fur of the animals is limited, and aerosol uptake through dermal absorption or ingestion (grooming) is minimized.
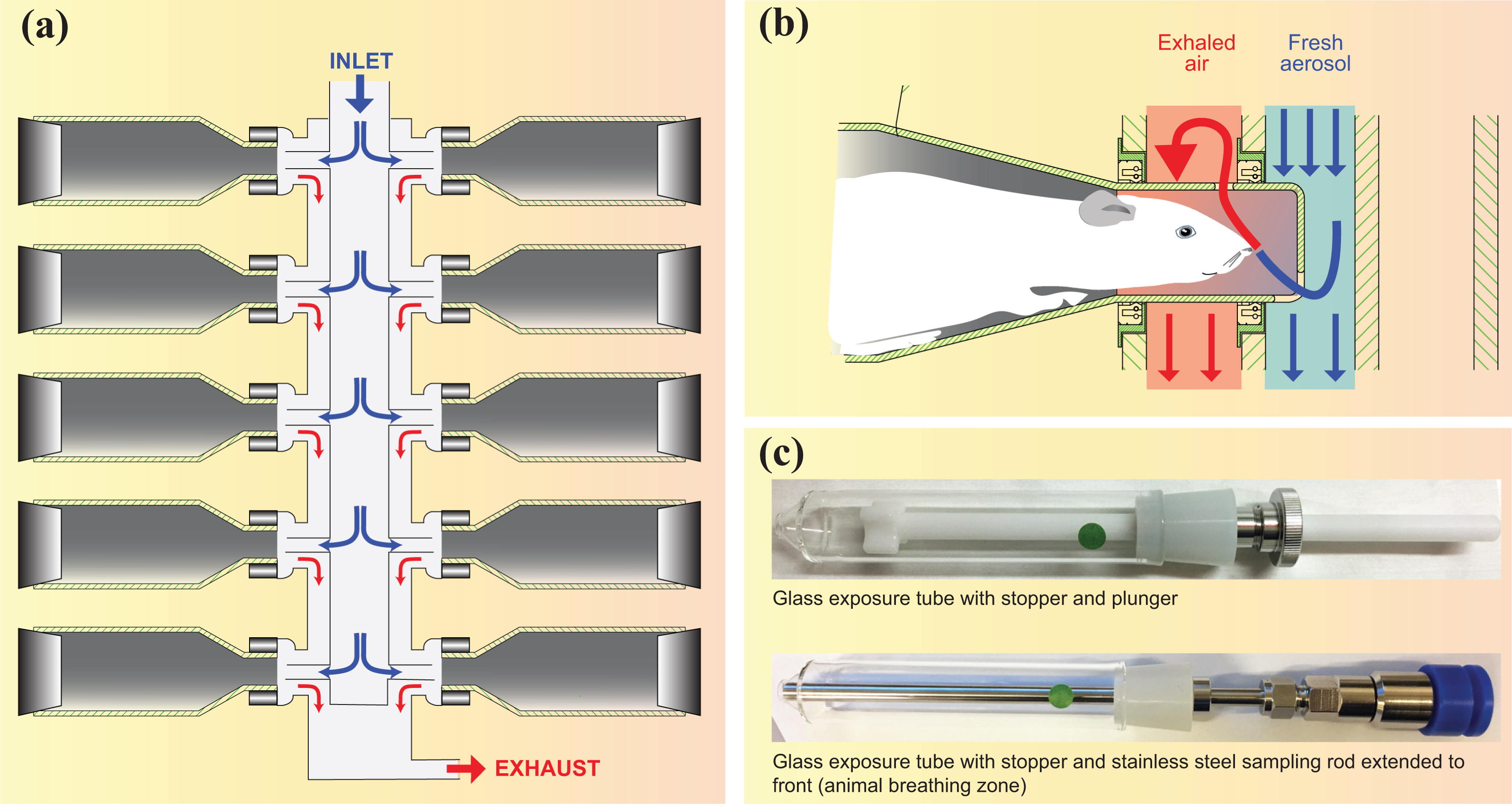
(a) Schematic of the principle of a double-plenum FPC. (b) Air circulation at the animal breathing zone of the FPC-232 (“64-Port nose-only flow past exposure chamber for rat inhalation studies” section). (c) Exposure and sampling tubes for the FPC-232 NOEC. FPC: flow-past chamber; NOEC: nose-only exposure chamber.
On the downside, the animals may be subject to higher stress levels in NOECs due to them being restrained and having no access to water throughout the daily exposure period. However, the confounding effect of stress on toxicity endpoints in inhalation studies using NOECs has yet to be demonstrated for each type of test item. Little difference in toxicity has been observed when comparing head-only to whole-body exposures. 139 Apart from differences in food and water intake, it was concluded that mode of exposure-associated differences in cardiovascular endpoints and respiration did not confound the results of long-term toxicity studies. 148 One key limiting factor for the use of NOECs is that daily animal procedures are highly labor intensive, de facto limiting the number of animals that can be used in a study.
A continuous flow of air is supplied to the chamber. Early chambers were conceived with a single central plenum chamber. Animals would inhale from the plenum and exhale back into it. Rebreathing could therefore occur, and the downstream animals could inhale air exhaled from the upstream animals, thereby getting a lower, or variable, concentration of the test material, although this could be partially compensated by increasing the aerosol flow through the chamber. In order to minimize these problems, PMI uses double-plenum systems with an inner and an outer plenum built on the so-called “flow-past” model first described by Cannon et al. 149 (Figure 19(a)). The test atmosphere flows vertically downward through an inlet to the inner plenum and is distributed horizontally through the exposure ports and each animal’s breathing zone. Unused aerosol and exhaled air are cleared away with the excess airflow into the outer plenum and then to the chamber exhaust (Figure 19(b)). For NOECs, it is recommended that the minimum volumetric flow rate supplied at each nose port is five times the animal maximum respiratory minute volume. 150
In use, the pressure in the inner plenum is higher than the pressure in the outer plenum to maintain a flow direction from the inner to the outer plenum.
Due to the smaller chamber volume and smaller internal surface areas of a NOEC, the time to attain chamber equilibrium is typically shorter, and the amount of test article needed is minimized compared to large WBECs. Precisely controlled concentrations can be delivered to the breathing zone of each animal, and concentration in the exposure chamber can be changed rapidly. NOECs also offer less risk of test material chemical instability (e.g. through reaction with excreta or humidity).
NOECs allow for the sampling of the test atmosphere at the breathing zone of the animals. Closed exposure tubes are fitted with hollow stainless steel sampling rods that extend to the front of the exposure tube to replicate where the animal’s nose would be during exposure so that the samples are representative of what the animal would inhale (Figure 19(c)).
There are a number of commercially available and bespoke NOECs. The following sections describe two types of double-plenum NOECs: a 64-port NOEC FPC for the exposure of rats and a 60-port NOEC for the exposure of mice.
64-Port nose-only flow past exposure chamber for rat inhalation studies
The FPC-232 was designed by PMI (Patent EP2095791A1) and is manufactured by Geraetebau Insul Simsheuser GmbH, Insul, Germany. It is a double-plenum, multilevel, modular chamber made of anodized aluminum for rat inhalation studies.
Typically, the exposure tower is configured with 8 levels of 8 exposure ports each, for a total of 64 ports. Sampling ports are installed in the exposure ports of the middle row of the tower (row 4 from top). Up to five ports are used for the sampling and real-time monitoring of parameters such as temperature and chamber pressure.
The volume of the inner chamber is around 4 L, and that of the outer chamber is around 12 L. The aerosol flows vertically downward through an inlet to the inner plenum and is distributed horizontally through the exposure ports.
Each exposure tube passes through the opening of the exposure port in the outer and inner plenum and extends at least partially into the inner plenum. The exposure tube comprises an exposure inlet in a portion of the exposure tube in the inner plenum and an exposure outlet in a portion of the exposure tube in the outer plenum. Test aerosol flows into the inner plenum, into each exposure tube via the exposure inlet, and out of the exposure tube, with exhaled air, via the exposure outlet into the outer plenum. An air flow of 100 L/min is used to maintain an aerosol flow of 1.2–2 L/min per exposure port. When atmosphere is withdrawn for sampling, the air flow should be adjusted accordingly.
The inner plenum is operated under a slight positive pressure of 10–30 Pa, and a pressure differential of 0.5 to 2 Pa is maintained between the inner and outer plenums to ensure that the aerosol flows from the inner to the outer plenum and not in the reverse direction. The pressure is maintained using a suitably sized orifice and an open coupling placed at the outlet of the outer plenum that connects to the exhaust line. The gap across the open coupling is set at a distance that prevents the exhaust line pressure from actively pulling on the FPC. Pressure fluctuations in the exhaust line therefore do not influence the inner and outer plenum pressures relative to the environment. This helps to maintain a stable inner plenum pressure and differential pressure between the inner and outer plenums. The animals are rotated daily across tube positions to prevent artifacts in the data arising from any potential spatial inhomogeneity.
Urine may be collected during exposure, either discontinuously or continuously, using a modified exposure tube including a urine collector to collect urine flowing away from the animal.
60-Port NOEC for mouse inhalation studies
The 60-port NOEC is a stainless steel, double-plenum, multilevel modular nose-only exposure system for murine exposures (CH Technologies, Westwood, New Jersey, USA, Patent 5297502). It consists of individual tiers of 12 exposure ports each. In a typical configuration with 5 tiers and 60 ports, the volume of the inner chamber is around 6 L, and that of the outer chamber is around 8 L. The aerosol flows vertically downward through an inlet (16.5 mm diameter in a standard configuration) to the inner plenum and is distributed horizontally through the exposure ports. It exits from the outer plenum through the bottom of the NOEC.
The 60-port NOEC has not yet been used in THS 2.2 research. Notably, though, Lucci et al. assessed the aerosol flow characteristics in the chamber using computational flow dynamics and estimated potential particle size-dependent nonuniformities in aerosol sampling at the exposure ports. 151 Figure 20 shows the flow through a single tier of a 60-port NOEC (Figure 20(a)), a visualization of the computed air flow velocity inside the inner plenum (Figure 20(b)), and computed particle density in an exposure port (Figure 20(c)).
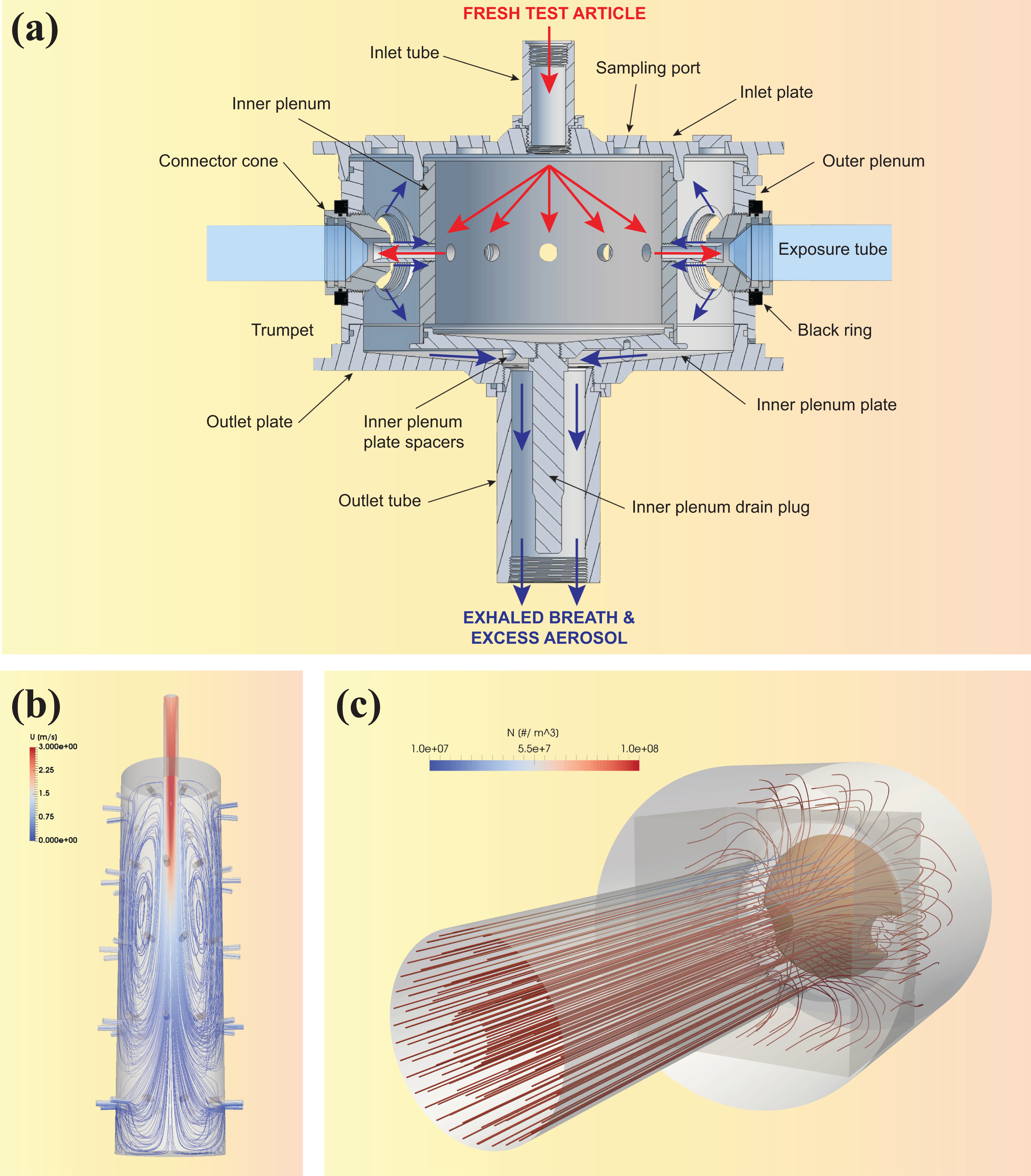
60-Port NOEC. (a) Illustration of flow through the NOEC using a single tier. (b) Computed air flow velocity (
The model showed some port variability in air flow velocity, with relatively higher flow through the ports of the lower tiers due to recirculation of the aerosol after reaching the bottom of the inner plenum. Port variability did not result in significant variability in particle density. The model also predicted sedimentation of larger particles (>3 µm) inside the exposure ports. However, as the effect was minor and applied to particles beyond the size range recommended for rodent inhalation studies, the authors considered that this sedimentation should not influence the dosimetry of inhaled aerosols. Furthermore, the daily rotation of animals across tube positions should prevent artifacts in the data arising from any actual spatial inhomogeneity.
Test atmosphere characterization and animal exposure assessment
Test atmosphere sampling and characterization
OECD TGs provide that actual chamber concentrations should be measured at least three times during each exposure day for each exposure level. In case this is not feasible due to limited air flow rates or low concentrations, it is considered that one sample per exposure period is acceptable. 73,74,98
Table 5 presents typical conditions for the characterization of the test atmosphere in an exposure chamber. Sampling is carried out at the sampling locations previously described for the WBECs and NOECs.
Test atmosphere characterization: Method of determination, sampling parameters, and duration.
TPM: total particulate matter; PSD: particle size distribution; CO: carbon monoxide; CFP: Cambridge filter pad; GC: gas chromatography; HPLC: high-performance liquid chromatography; DNPH: 2,4-dinitrophenylhydrazine; APS™: Aerodynamic Particle Sizer®.
Determination of aerosol uptake
Even though animals may be exposed to the same test atmosphere, the amount of material that they inhale and that is deposited on their biological tissues can still differ among individuals because of differences in respiratory physiology and anatomical characteristics.
Hence, aerosol uptake by the animals should be monitored to demonstrate physiologically relevant exposure and provide a basis for data interpretation and comparison.
Biomarkers of exposure offer the potential to provide quantitative evidence of the presence of HPHCs, or their metabolites, in the body. The US National Academy of Sciences’ Institute of Medicine has defined a BoExp as “a constituent or metabolite that is measured in a biological fluid or tissue that has the potential to interact with a biological macromolecule; sometimes considered a measure of internal dose.” 4 Ideally, BoExp should: be specific to the source of exposure compound, with other sources being minor or nonexistent; be correlated with exposure dose; be easily detectable and accurately measurable using reliable, reproducible, precise analytical methods; and reflect a specific toxic exposure or be a reliable surrogate of exposure to HPHCs. BoExp may also be of interest for facilitating the dosimetric extrapolation from animals to humans.
BoExp for the particulate and GVP constituents of CS and HNB aerosols are typically used, such as steady-state plasma nicotine metabolites and carboxyhemoglobin (COHb) concentration and urinary metabolites of representative aerosol constituents. In addition, respiratory measurements may be performed by plethysmography, including respiratory frequency, tidal volume, and peak inspiratory flow.
A typical sampling and analytical schedule in a subchronic rat inhalation study is described in Table 6.
Sampling and analytical schedule for the determination of aerosol uptake in a 90-day subchronic rat inhalation study.

COHb: carboxyhemoglobin; HPLC: high-performance liquid chromatography.
Dosing, normalization, and extrapolation
Dosing rationales for in vitro assays
Exposure doses in nonclinical studies should ideally span realistic human equivalent doses. However, toxicological assessment guidelines may require that markedly toxic doses are also included, in particular in in vitro assays. In case the assay endpoint has low sensitivity and/or the test items have low biological activity, this may lead to unrealistically high concentrations of constituents compared with those occurring in an exposed organism. In vitro data must be interpreted very carefully with regard to in vivo health risks if the effects are observed at concentrations far beyond the systemic or local concentrations that can be achieved in an organism. For example, in vitro nicotine concentrations in the millimolar range are used in in vitro studies, while cigarette smokers have blood plasma levels below 600 nM and saliva levels below 50 µM 152 (see “Animal to human extrapolation” section). Moreover, most in vitro assays investigate acute exposure effects, while smoking-related diseases develop upon chronic exposures to acutely subtoxic concentrations.
For functional in vitro assays using cell types that are systemically exposed to inhaled or ingested substances, such as fibroblasts, endothelial cells, muscle cells, or glandular epithelial cells, it is reasonable to test doses corresponding to the nicotine concentration ranges observed in the body fluid and to also include some moderately higher levels (e.g. 3-fold and 10-fold). A similar rationale can be applied to organotypic epithelial cultures that are exposed to various media, such as ambient air (respiratory tract epithelium), saliva (oral epithelium), ingested materials (gastrointestinal tract), or urine (kidney and bladder epithelia). The tested doses can be selected based on corresponding in vivo concentrations or on surface doses observed with normal and exaggerated use levels. In air-exposed epithelial cells, both the deposited surface dose (g/m2) and the aerosol concentration (ppm, g/m3) may be relevant. The deposited concentration per volume of airway surface liquid, as can be determined from lung lavage, can also be leveraged for dose extrapolations.
For standardized assays, such as the regulatory assays described in the “Aerosol exposure systems for in vitro studies: Submersed cell cultures” section, test doses are dictated by design. For example, in an NRU assay, test doses should cover the relevant concentration range around the IC50 (i.e. the test substance concentration producing 50% inhibition of the endpoint measured (cell viability)). For in vitro mutation assays, such as the MLA and thymidine kinase assays, OECD TG 490 suggests that the highest concentration should aim to achieve between 10% and 20% of relative total cell growth. Therefore, results from such assays indicate comparative potencies rather than reflect the consequences of realistic exposure conditions.
Dose-range-finding studies
Before initiating comparative assessments of CS and HNB aerosol, it is highly recommended to perform dose-range-finding studies in order to understand the dynamic range of respective biological effects. Because the aerosol from HNB products is very different from CS, it is very likely that the biological effects are qualitatively and quantitatively different. It has been shown that up to orders of magnitude higher concentrations of HNB aerosols are needed to show similar biological activities, if any, as CS. 153
For in vitro studies, a dose-range-finding study allows the identification of the upper concentration that can be assessed without confounding effects due to cytotoxicity. Typically, the initial evaluation of toxicity can be performed using an RTCA system, such as the impedance-based system described in the “Dose-range-finding studies using real-time cell analysis” section. The determination of an exposure-response relationship will usually require at least three graded concentrations and a fresh-air control. Dose-range-finding studies also allow the identification of the parameters that will best characterize the test aerosol toxicity.
For in vivo studies, a maximum tolerated exposure dose is determined using protocols of escalating exposure concentration at the nominal daily exposure concentration up to the desired test atmosphere concentration of one or more aerosol reference compounds (e.g. nicotine). Alternatively, an escalation of the daily exposure duration at the nominal concentration can be chosen to achieve the full dose (concentration × time). The latter procedure may be applied if the inhalant causes some sensory irritation that may lead to respiratory depression, or if the initially lower uptake due to sensory uptake does not compromise the aim of the study. This maximum tolerated exposure level (used as a surrogate for maximum tolerable dose) is an exposure level at which some acceptable toxicity is observed in target organs while undue stress, moribundity, and mortality do not harm the animals, nor hamper the study. 73
In in vivo CS inhalation studies, the limiting factor is the concentration of CO delivered by CS, and exposure concentrations should be limited to those that elicit COHb levels in animals below 50%. Scientific literature shows that a wide range of biological effects can be observed at such exposure levels and that the inclusion of lower concentrations in a concentration-response approach allows the assessment of the dynamic range of many effects. 154 –156 CO accumulation typically limits exposure to 3R4F CS to a maximum of 6 h/day in rats and 6 × 1 h/day in mice. Thus, for shorter exposure durations, higher test atmosphere concentrations can be used compared to longer exposure durations.
Given the fact that HNB product aerosols contain much less CO, the limiting factor for in vivo inhalation studies is the nicotine concentration in the test atmosphere combined with the exposure duration. For 6-h exposures in rats, nicotine test atmosphere concentrations should be limited to 50 µg/L, while for mice, the maximal tolerated exposure levels are usually lower (e.g. 26–30 µg/L) for extended daily exposure durations 156 –161
Normalization
Frequently applied normalization parameters in conventional CS toxicity comparisons, such as machine-measured TPM or CO, are not meaningful for the normalization of HNB aerosols in comparison with CS because, by design, HNB aerosols mostly yield under-proportionally low CO concentrations in the GVP and proportionally higher TPM concentrations due to high yields of water, glycerol, and/or other aerosol carriers forming liquid particles. Different approaches have therefore been proposed and used to draw comparisons among the wide variety of available products.
Comparing products on a per article/consumable basis rests on the assumption, or verification with clinical trials or post-market surveillance, that the number of consumables per day is the same for cigarettes and HNB tobacco products. However, this has yet to be demonstrated. For example, a 5-day randomized, controlled study of THS 2.2 consumption in confinement showed that on average, users switching from cigarettes to THS sticks consumed about two THS sticks less per day than continuing cigarette smokers. 57
Comparing products on a per-puff basis necessitates an estimate of the number of puffs per day for both the cigarettes and the HNB tobacco product. 56,162
Using nicotine concentration as a normalization parameter in comparative studies of CS constituents has been recommended by the TobReg (2nd report). 3 HNB tobacco products aim at delivering similar amounts of nicotine per puff to the consumer as a puff of CS would deliver. Comparisons of additional delivered compounds would be best, providing additional in situ quantification.
Whenever possible, and for the ease of comparison and statistical procedures, it is recommended in in vivo studies to cover a concentration range of nicotine in the test atmospheres for both CS and HNB aerosols, preferably with at least one matching nicotine exposure concentration for both product types.
Animal to human extrapolation
In clinical studies, exposure is usually measured as the number of cigarettes or HNB sticks used per day. The reference BoExp is the amount of nicotine and its metabolites (particularly cotinine) in blood and urine. The BoExp to HPHCs can then be related to the nicotine exposure, and reductions in HPHC exposure levels can be normalized for equal nicotine levels to compensate for differences in the nicotine yields of cigarettes and HNB tobacco products as well as differences in individual smoking behavior. Ranges of reported nicotine concentrations in blood, saliva, and maternal milk in users of various nicotine-containing products are provided in Table 7.
Ranges of nicotine concentrations measured in body fluids upon exposure to nicotine-containing products.
To extrapolate from animal studies to humans, the simple matching of nicotine plasma levels would lead to an under-dosing due to differences in the organism’s size, anatomy, and pharmacodynamics. For example, typical plasma nicotine levels ranged from 110 to 212 ng/mL in ApoE−/− mice whole-body exposed to 3R4F CS (600 µg TPM/L, 28–29 µg nicotine/L, 3 × 1 h/day) 159,160 and from 260 to 307 ng/mL in rats nose-only exposed to 3R4F CS (273 to 330 µg TPM/L, 21.6–23 µg nicotine/L, 6 h/day). 34,156,181 These values are up to eight times higher than the plasma levels found in human smokers. The cotinine-to-nicotine ratios in rat plasma samples from the abovementioned studies ranged from 1.7 to 2.7, while the mean cotinine-to-nicotine ratios in plasma samples from human smokers ranged from 13.4 to 16.4, 164,182,183 thus indicating differences in the kinetics of nicotine metabolism between rodents and humans that may contribute to the higher plasma levels observed in rodents.
Hence, the human equivalent concentration approach can be used instead to extrapolate a biologically active exposure concentration in an animal inhalation experiment to the equivalent concentration for humans. Depending on the endpoint of interest, the conversion factor may be based on body surface area (for systemic toxicity) or on lung surface area (for local toxicity). 184
Because similar biomarkers can be measured in in vitro, in vivo, and human clinical studies, a more direct comparison of potential adverse events at similar plasma levels can be made between species and tissues. The use of systems biology approaches using transcriptomics, proteomics, and lipidomics for a more mechanistic understanding can amplify the insight from this more direct comparison.
Conclusion
The recent emergence and rapid growth of HNB tobacco products has been accompanied by the development and adaptation of testing methods that take into account products’ specificities, and has fostered innovation with development of systems biology approaches using transcriptomics, proteomics, and lipidomics. The FDA has provided a draft guidance document outlining a framework to assess novel tobacco products as MRTPs. 7 The international harmonization of methods is needed to avoid the generation of data which are, at best, difficult to compare and, at worst, may result in misleading comparisons and conclusions. This review was intended to provide detailed information that may help with replicating studies and conclusions in experiments conducted by different groups on the same products. Such transparent sharing of protocols, tools, and data with a wide audience of scientists in academia, industry working groups, and regulatory bodies is intended to facilitate the alignment of best experimental practices and promote understanding of best methodologies. This, in turn, will lead to the formulation of standards specifically relevant to HNB tobacco products.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB, DG, JH, AK, SM, CM. BP. MCP, FR, WTT and PV are employees of Philip Morris International. AM and WKS were contracted and paid by Philip Morris International to contribute to the writing of this review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Philip Morris International is the sole source of funding and sponsor of this project.
