Abstract
The US Food and Drug Administration (FDA) Division of Food Contact Notifications (DFCN) performs infant lifestage-specific exposure and safety assessments during the premarket review of FCNs proposed for use with infant formula or human milk. DFCN previously identified two protocols that may be best suited to support infant safety: the multigenerational developmental and reproductive toxicology (Gen-DART) protocol and the juvenile animal study (JAS) protocol. The Gen-DART protocol tests chemical exposure from prior to mating through one or two generations in rodents, while JAS protocols test a chemical during a specific developmental window. At FDA, Gen-DART studies are currently recommended to support the safety of food additives (FAs), including food contact substances, across lifestages (from conception through adulthood). JAS protocols are recommended in the nonclinical safety assessment of drugs seeking pediatric indications. To improve our recommendations regarding the use of either Gen-DART or JAS protocols for the infant safety assessment of food contact materials, we created a database of FDA-reviewed Gen-DART or JAS. Our database contains information from 41 Gen-DART studies (38 FAs) and 51 JAS (29 drugs). Both protocols can detect toxicity relevant to infant safety assessment, such as developmental toxicity in neurological, endocrine, reproductive, immunological, or skeletal systems. Selection of either protocol will depend on the amount of information available for the chemical under study. The Gen-DART protocol may be preferred when limited data on the mode of action or target organ of the chemical are available. However, if these data are available, a JAS may provide the best assessment of potential juvenile toxicity.
Keywords
Introduction
The US Food and Drug Administration (FDA) Center for Food Safety and Applied Nutrition (CFSAN), Office of Food Additive Safety (OFAS), Division of Food Contact Notifications (DFCN) conducts a separate infant-specific exposure and safety assessment for food contact materials intended to contact human milk or infant formula. Infant food contact materials include feeding articles (e.g. bottles, nipples) and containers (e.g. powdered or liquid infant formula canisters, human milk storage bags). The premarket infant-specific assessments are performed in a case-by-case manner considering the infant-specific exposure factors and specific biological differences between infants and other lifestages. 1 Ultimately, an infant safety assessment requires analysis of various types of toxicity data, often including the extrapolation of findings from adult animals to predict outcomes in juvenile and neonatal animals. Occasionally, the available data may not adequately address a concern for infant safety, and FDA may recommend toxicity testing to support the safety of the product.
A variety of developmental and reproductive toxicology (DART) protocols are used in regulatory toxicology settings. These range from short-term tests of a specific developmental stage (e.g. prenatal toxicity study) to more elaborate tests that study the effects of a test article in animals prior to conception, during in utero and postnatal (PN) development and through mating of the offspring (e.g. two-generational DART study). In contrast, juvenile animal study (JAS) protocols can be tailored to include specialized end points relevant to known effects of the test article and include a customized exposure paradigm targeted to the developmental stage in the animal model that most closely matches the proposed pediatric population. We previously reviewed the most common DART protocols used for regulatory decision-making and concluded that the multigenerational DART (Gen-DART) and JAS protocols may provide the most useful information for infant-specific safety assessments. 2,3
Both Gen-DART and JAS protocols are recommended to support the safety of FDA-regulated products during premarket or nonclinical review. Currently, Gen-DART protocols are recommended by US FDA/CFSAN/OFAS to characterize the DART potential of FAs, including food contact materials. The FDA Redbook provides specific recommendations for the conduct of a two-generational DART study to assess the potential for an FA to elicit reproductive or developmental toxicity across lifestages. 4 The JAS protocol is recommended by US FDA Center for Drug Evaluation and Research (CDER) for the nonclinical safety assessment of drugs for pediatric indications. 5 While CFSAN/OFAS does not currently recommend JAS protocols to support the safety of infant food contact materials, we recognized that there may be a role for this protocol in the OFAS paradigm. Therefore, we partnered with US FDA CDER to evaluate the JAS protocol for use in infant-specific safety assessments for food contact materials.
Overall, this project completed two objectives. We evaluated Gen-DART studies submitted to FDA for the premarket safety assessment of FAs across lifestages for the specific information these studies provide for infant safety assessment. Second, we assessed a DART protocol not currently recommended by OFAS/DFCN for potential inclusion in the infant-specific safety assessment of food contact materials. Our work provides the first analysis of Gen-DART and JAS protocols for use in infant safety assessment. Both study designs can provide useful information for infant safety assessment and, in some cases, may provide the most conservative point of departure to set protective human health standards. However, study selection will depend on the amount and type of information available on the chemical under review in the context of the estimated infant-specific exposure.
Methods
Study selection, inclusion criteria, and exclusion criteria
Gen-DART studies
To be considered for inclusion, Gen-DART studies had to be submitted during the FDA premarket review of FAs through the Food Additive Petition or Food Contact Notification processes. FAs have been reviewed by FDA for over 50 years, and Gen-DART protocols have changed substantially over this time. We evaluated all types of Gen-DART studies for inclusion, including continuous breeding protocols and one-, two-, three-, and six-generation reproduction studies. However, we restricted the studies to those that included assessment of offspring during the preweaning period. Some protocols followed FDA Redbook or Organisation for Economic Co-operation and Development guidelines, but some studies were conducted prior to the establishment of these guidelines. Studies conducted prior to the establishment of Good Laboratory Practice regulations were included if sufficient individual animal data were available to evaluate the study quality. Overall, 91 Gen-DART studies with PN measurements were identified and compared against exclusion criteria. Eighteen studies were eliminated because of gross deficiencies that limited study interpretation (e.g. excessive mortality, too few animals per dose group, insufficient end points), 12 studies were excluded because they were not reviewed in the premarket setting, 7 studies did not have enough data or protocol details available for evaluation, 6 studies employed less than three doses, 4 studies did not include PN end points through weaning (e.g. termination instead at postnatal day (PND) 0 or 4), and 3 studies employed a nonoral route of administration. Overall, a total of 41 Gen-DART studies associated with 38 FAs were included in our database.
JAS studies
To be considered for inclusion, the JAS had to be submitted in an investigatory new drug (IND) or new drug application (NDA) in support of a pediatric indication. FDA CDER provided a list of 65 INDs or NDAs that included at least one JAS. From this list, five drugs had ongoing JAS at the time and were excluded. Eight of the remaining 60 drugs were excluded because they contained duplicate JAS since the same JAS was used to support more than one pediatric drug indication. Another consideration was the time and resources needed to harvest toxicological data from textual memoranda. We needed to balance time and resource investment with our ability to meet the aims of this project. Ultimately, 51 JAS associated with 29 drugs were harvested. To maintain confidentiality of the drug data, drugs will not be identified by name or chemical abstracts registry number (CASRN) but will instead be referred to by a randomized identity letter.
Quality control
The harvested information for each study was evaluated to ensure that the captured information accurately represented the protocol details and FDA’s interpretation of the effects observed in the study.
Database design and population
We stored data harvested from Gen-DART, JAS, 90-day repeated-dose subchronic (SC) toxicity, and peri- and postnatal (PPN) toxicity studies in Microsoft Access (Microsoft, 2010). We extracted metrics associated with data source, chemical information, study design, and toxicological effects (Table 1). For each study, the overall lowest observed adverse effect level (LOAEL) and no-observed adverse effect level (NOAEL) were recorded (“study LOAEL” or “study NOAEL”). Additionally, the LOAEL and NOAEL specific to each toxicological effect (“effect LOAEL” or “effect NOAEL”) were recorded. In some cases, only NOAELs or LOAELs (not both) were observed.
Examples of database end points.a
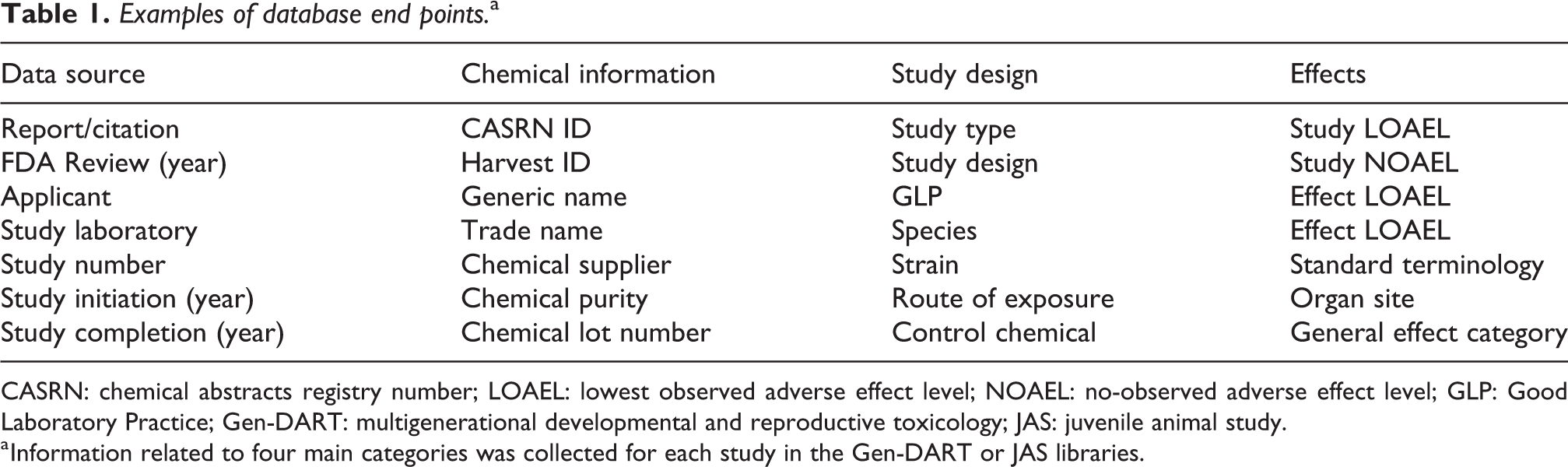
CASRN: chemical abstracts registry number; LOAEL: lowest observed adverse effect level; NOAEL: no-observed adverse effect level; GLP: Good Laboratory Practice; Gen-DART: multigenerational developmental and reproductive toxicology; JAS: juvenile animal study.
a Information related to four main categories was collected for each study in the Gen-DART or JAS libraries.
Data analysis
Affected systems
There are several areas of particular interest for infant safety assessment, specifically, the potential for a chemical to elicit neurological, endocrine, immunological, skeletal, or reproductive toxicity. 2 We tracked the effects in Gen-DART and JAS as potential alerts for developmental neurotoxicity, endocrine toxicity, immunotoxicity, skeletal toxicity, or reproductive toxicity, respectively. Neurological alerts included clinical signs related to neurological activity (e.g. bruxism, ataxia, hypo/hyperactivity, altered body temperature, abnormal secretions, etc.), effects in the functional observational battery or behavioral assays, changes in brain weight, effects in white matter, and eye effects (e.g. retinal histopathology). Endocrine alerts included effects on hormone levels, effects on endocrine organ weights, histopathological changes in endocrine organs, effects on hormonally regulated developmental milestones (e.g. pinna unfolding, testicular descent, balano-preputial separation, vaginal patency), estrous cycle parameters, gestation length, and dystocia. In addition, specific effects on local endocrine-active cells (e.g. chief cells in the stomach) were considered endocrine alerts. Reproductive toxicity alerts overlapped with most endocrine toxicity alerts; exceptions included reproductive toxicity alerts of abnormal maternal care and decreased pup viability at birth, which were considered reproductive but not endocrine alerts. Immune alerts included weight changes in immune organs, histopathology in immune organs, specialized immune end points (e.g. T cell-dependent antigen response), and effects on white blood cells. Skeletal alerts included effects on bone density, longitudinal growth, diametric growth, or histopathology, and effects on bone marrow (also an immunotoxicity alert).
Effect categorization
Each toxicological effect in our study library was harmonized to a preferred terminology. 6 This harmonization allowed us to merge synonymous effects such as “acoustic startle response” and “auditory startle response.” Each harmonized effect was placed into broader general effect categories to facilitate comparisons across study protocols. The general effect categories were organ toxicity, body weight, clinical signs, behavior, developmental milestones, reproductive function, hematology, mortality, clinical chemistry, feed and water intake, urinalysis, and multicomponent physiological systems such as the immune, endocrine, and cardiovascular systems.
Toxicokinetics (JAS database only)
To evaluate age-related differences in internal exposure, blood area under the curve (AUC) or CMAX levels were compared in juvenile (≤PND 21) and adult rats (≥PND 43) at the same external dose. Toxicokinetic (TK) data in the adult animals were recorded from the general toxicity studies found in IND and NDA reviews. Juvenile AUC was available for all nine drugs, but adult TK for the same doses as used in the JAS was available for only four SC studies. In the other cases, adult TK was available, but the doses used in the SC study were different; therefore, juvenile and adult exposure could not be compared at the same dose. For the five remaining studies, the AUC measured at the end of dosing in the JAS was considered “adult AUC” if the juvenile animal age at the end of dosing was
Results
Objective 1: Utility of Gen-DART protocols for infant-specific safety assessment
Study library
The Gen-DART library contains a total of 41 Gen-DART studies associated with 38 direct and indirect FAs (Table 2). The majority of studies were two-generational studies (59%). For studies that tested multiple generations, we captured data from P0 and F1 generations only. Additionally, we harvested information from only F1A pups from study protocols that bred maternal animals successively to produce multiple litters per generation.
Gen-DART study library.

Gen-DART: multigenerational developmental and reproductive toxicology; FA: food additive.
Table 3 provides the FA identity number, chemical name, CASRN, and examples of regulated uses for each additive in the Gen-DART library. Our library contains a variety of direct additives and food contact substances, ranging from nonnutritive sweeteners to antimicrobial food contact substances.
Chemical identity, abbreviation or example trade name, and example of regulated use of FAs tested in Gen-DART studies.a
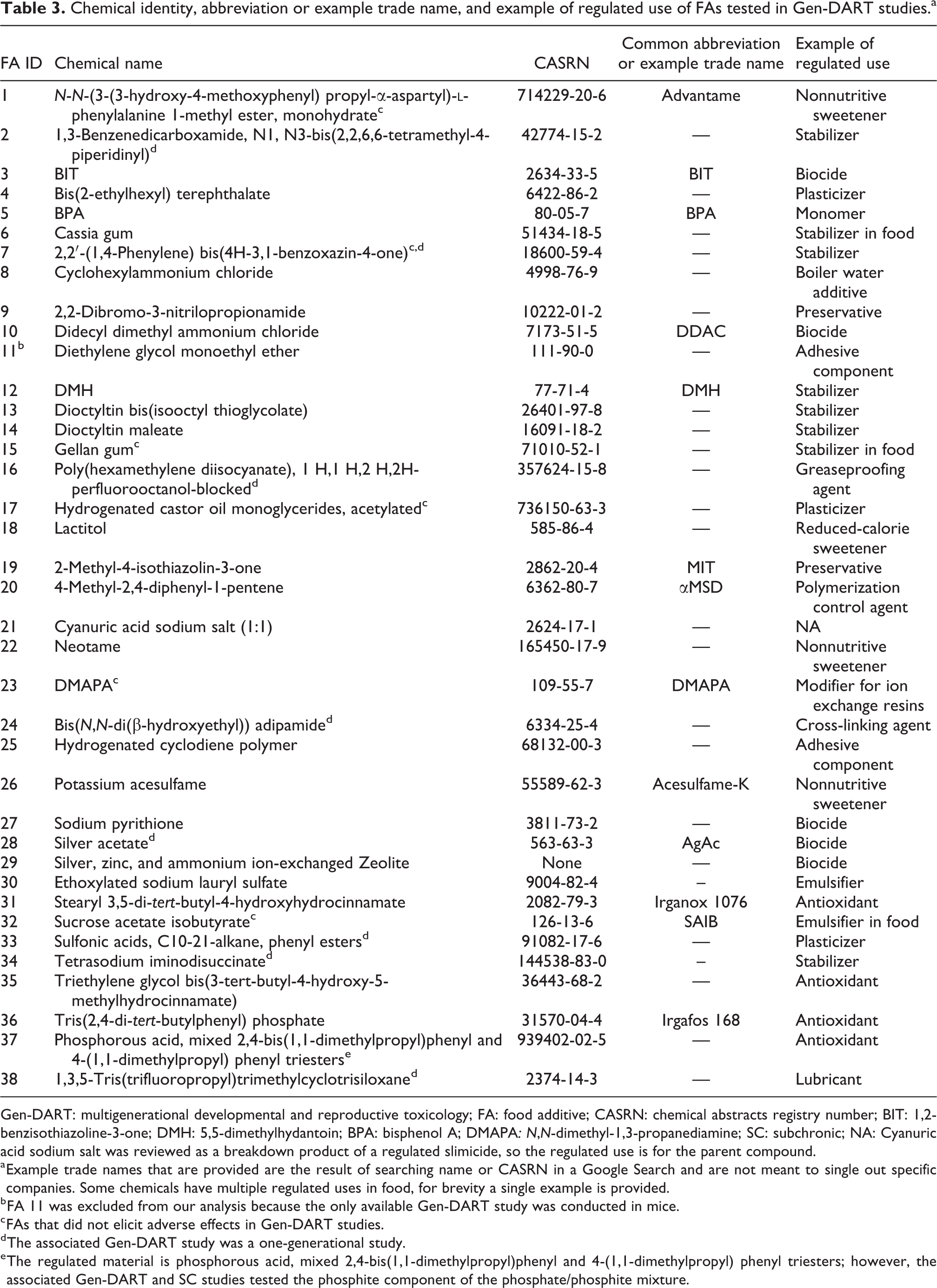
Gen-DART: multigenerational developmental and reproductive toxicology; FA: food additive; CASRN: chemical abstracts registry number; BIT: 1,2-benzisothiazoline-3-one; DMH: 5,5-dimethylhydantoin; BPA: bisphenol A; DMAPA: N,N-dimethyl-1,3-propanediamine; SC: subchronic; NA: Cyanuric acid sodium salt was reviewed as a breakdown product of a regulated slimicide, so the regulated use is for the parent compound.
a Example trade names that are provided are the result of searching name or CASRN in a Google Search and are not meant to single out specific companies. Some chemicals have multiple regulated uses in food, for brevity a single example is provided.
b FA 11 was excluded from our analysis because the only available Gen-DART study was conducted in mice.
c FAs that did not elicit adverse effects in Gen-DART studies.
d The associated Gen-DART study was a one-generational study.
e The regulated material is phosphorous acid, mixed 2,4-bis(1,1-dimethylpropyl)phenyl and 4-(1,1-dimethylpropyl) phenyl triesters; however, the associated Gen-DART and SC studies tested the phosphite component of the phosphate/phosphite mixture.
Not every Gen-DART study in our library was included in comparative or cross-study analyses. Two chemicals, 1,2-benzisothiazoline-3-one and 5,5-dimethylhydantoin, were tested in two independent Gen-DART studies in rats that met the study inclusion criteria. In comparative analyses, we used the study that provided the lower study LOAEL. All but two of the Gen-DART studies were conducted in rats; one bisphenol A and one diethylene glycol monoethyl ether study were conducted in mice. The mouse studies were not used in comparative analyses. Six chemicals did not elicit adverse effects on either the P0 or the F1 generation; therefore, these studies did not detect an LOAEL. Due to lack of adverse effects, these studies were not used in comparative analyses examining effects.
Affected systems
Our previous publication identified several areas of particular interest for infant safety assessment, specifically, potential neurological, endocrine, immunological, skeletal, and reproductive toxicity. 2 We tracked effects in Gen-DART studies that occurred in these systems as potential alerts for developmental neurotoxicity, endocrine toxicity, immunotoxicity, skeletal toxicity, or reproductive toxicity, respectively. Of these five systems, the endocrine and reproductive systems were the most commonly affected at the study LOAELs in Gen-DART studies (Table 4). Since alerts for these systems are highly interrelated (see Methods section), the similar frequencies are not surprising. While endocrine or reproductive effects were never the only effects occurring at the LOAEL, there were cases where the immune and neurological systems were the sole system affected at the study LOAEL. In these cases, the observed immunological effects were altered immune response, histopathological changes in the spleen, and increased spleen weights. For the neurological system, behavioral effects such as altered motor activity and learning and memory were observed at the study LOAELs. Skeletal toxicity was not detected in any of the studies in our Gen-DART library. Gen-DART studies tend to have limited end points related to skeletal toxicity. For example, bone density and morphometry are not routinely measured in Gen-DART studies. Therefore, the lack of effects on the skeletal system may be due to protocol limitations or a lack of toxic action on bone by these FAs.
Gen-DART protocols detect effects in systems considered important for infant safety assessment.a
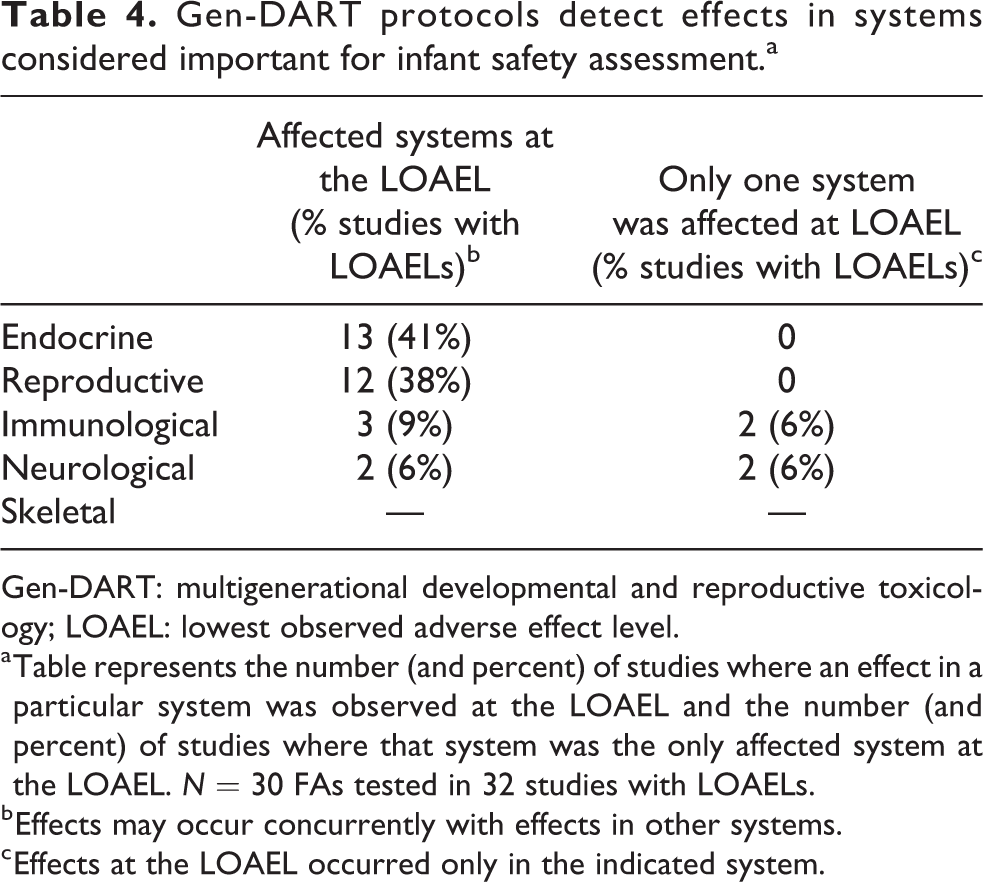
Gen-DART: multigenerational developmental and reproductive toxicology; LOAEL: lowest observed adverse effect level.
a Table represents the number (and percent) of studies where an effect in a particular system was observed at the LOAEL and the number (and percent) of studies where that system was the only affected system at the LOAEL. N = 30 FAs tested in 32 studies with LOAELs.
b Effects may occur concurrently with effects in other systems.
c Effects at the LOAEL occurred only in the indicated system.
General effect categories
A total of 335 adverse effects were recorded in our Gen-DART library (32 studies with LOAELs). Most effects (195 effects; 58%) were observed at the study LOAELs. To evaluate which effects were most common at the LOAEL, we assessed all effects occurring at the LOAELs in rat Gen-DART studies. We also assessed the percentage of studies that observed effects at the LOAEL in each general effect category (Table 5). Of all observed effects, the most commonly affected categories at study LOAELs were body weight, organ toxicity, and reproductive function in the parental and offspring generations.
Effects distribution by general effect category in P0 and F1 generation at Gen-DART study LOAELs.a
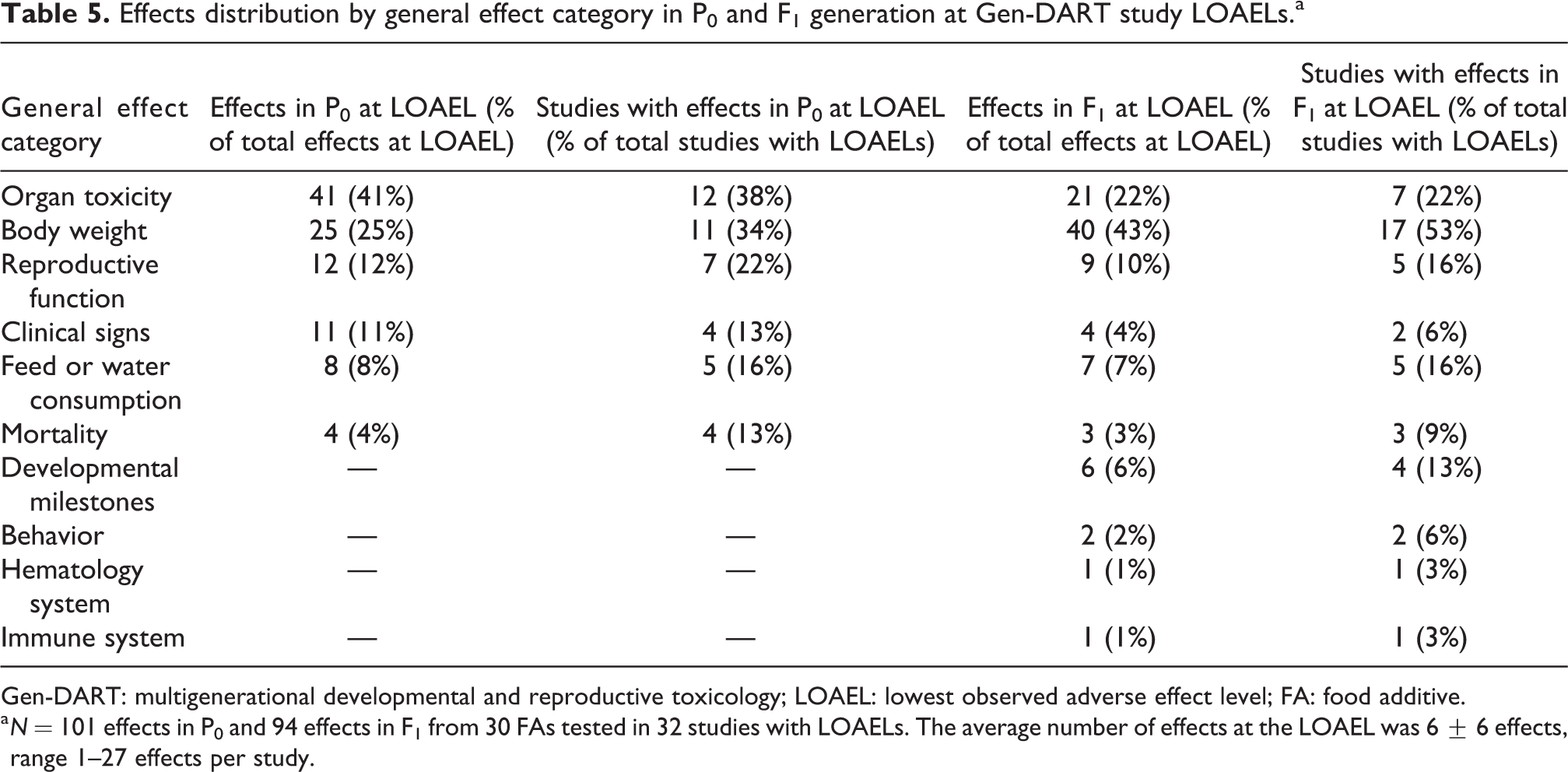
Gen-DART: multigenerational developmental and reproductive toxicology; LOAEL: lowest observed adverse effect level; FA: food additive.
a N = 101 effects in P0 and 94 effects in F1 from 30 FAs tested in 32 studies with LOAELs. The average number of effects at the LOAEL was 6 ± 6 effects, range 1–27 effects per study.
In terms of study incidence, generally the same top three categories were affected with the addition of feed or water consumption matching the study incidence of the reproductive function in the F1 generation. In some cases, a single study had more than one effect in a general effect category at the study LOAEL. For example, the body weight category includes both altered body weight and altered body weight gain, which often occur concurrently. A variety of organs were affected at the study LOAELs and are further detailed in the next section and Table 6.
Effects distribution by organ toxicity in P0 and F1 generation at Gen-DART study LOAELs.a

Gen-DART: multigenerational developmental and reproductive toxicology; LOAEL: lowest observed adverse effect level; FA: food additive.
a N = 41 organ toxicity effects in P0 rats and 21 organ toxicity effects in F1 rats from 30 FAs tested in 32 studies with LOAELs.
Organ toxicity
Organ toxicity was one of the most commonly affected categories in both the P0 and F1 generations at the study LOAELs (62 effects). Organ toxicity may be indicated by changes in organ weight (absolute or relative to brain or body weight), histopathological changes, or ultrastructural changes (e.g. gross dilation of ureters). Overall, organ toxicity was more frequently observed in P0 rats than in F1 rats. The most commonly affected organs in the P0 rats were kidney (39%) and liver (22%), whereas liver (38%) and stomach (24%) were the most commonly affected organs in the F1 generation (Table 6). When we evaluated the percentage of studies with organ effects, the same top organs were affected in both generations with the addition of the spleen matching the study incidence of the liver and stomach for the F1 rats (Table 6).
Cohort-specific toxicity
One important question for developmental toxicology is whether juvenile animals are more sensitive to chemical exposures than adult animals. Gen-DART studies provide the unique opportunity to compare adult and juvenile responses to chemicals within the same study. Despite this advantage, it is still difficult to determine whether juveniles or adults were more sensitive to a test substance due to questions regarding juvenile exposure, especially in the preweaning period. Current and historical Gen-DART guidelines do not routinely include direct dosing to preweaned pups. None of the studies in our library included direct dosing to preweaned pups. Further, none of the Gen-DART studies in our library included TK assessments in juvenile or adult animals. Therefore, we do not know the internal exposure of the F1 pups and conclusions about sensitivity can only be informed by the external doses.
Considering these limitations, we focused on cases where toxicity was only observed in only one cohort, either P0 or F1 animals. Additionally, we focused on cases where the study NOAEL or LOAEL was determined solely by effects in a single cohort (P0 or F1).
Our analysis shows that the LOAEL of Gen-DART studies is not often determined by developmental effects. We observed cohort-specific toxicity with similar frequency in P0 and F1 generations. There were six cases (19%) where the study LOAEL was determined by the P0 generation and five cases (16%) where the F1 generation determined the study LOAEL (Table 7). We also observed that in four of the six studies where the P0 generation determined the study LOAEL, there were no effects on the F1 cohort (67%) and in the cases where F1 generation drove the study LOAEL, there were no effects in the P0 cohort in 20% of studies (one of the five cases).
Cohort-specific toxicity.
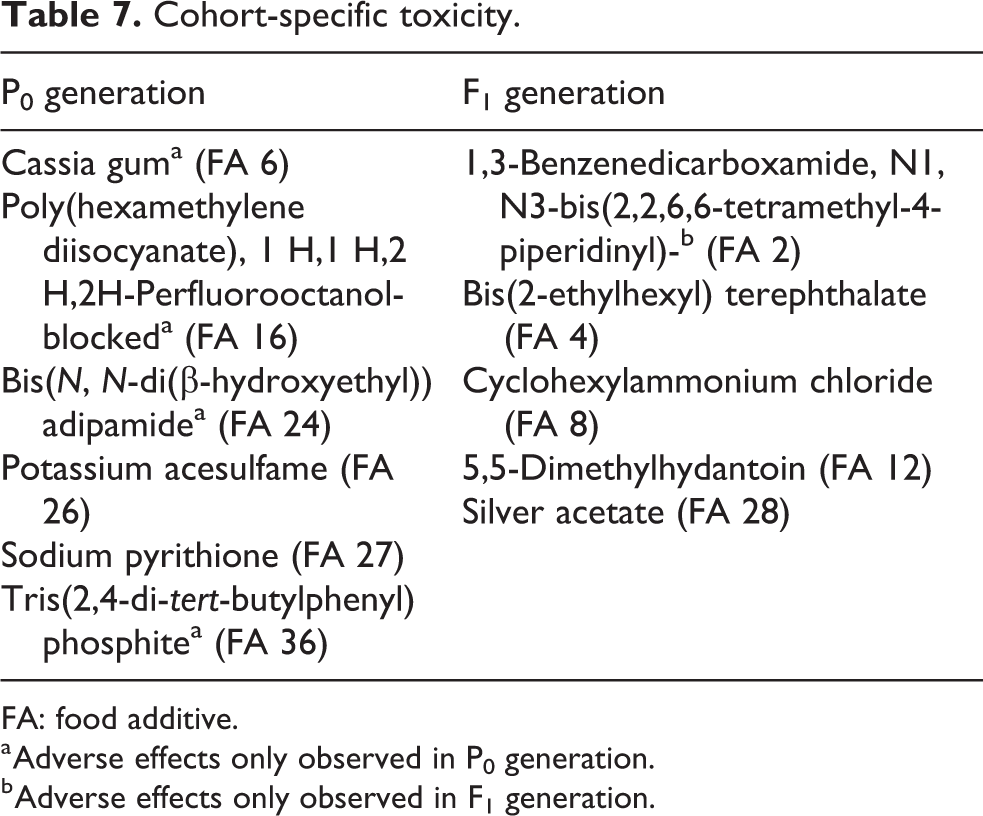
FA: food additive.
a Adverse effects only observed in P0 generation.
b Adverse effects only observed in F1 generation.
In the five cases where the F1 generation determined the study LOAELs, the effects included decreased pup body weights or body weight gains (FAs 2, 4, and 12), decreased feed intake and litter size (FA 8), decreased pup survival, decreased pup body weight, decreased stomach weight relative to body weight, histopathological changes in the spleen, and altered immune response (FA 28). Together these studies revealed potential effects on offspring growth, survival, and target organ effects observed at lower doses in the F1 generation than in the P0 generation.
Gen-DART and SC cross-study comparison
In DFCN, safety assessment focuses on identifying the lowest (or most conservative) point of departure in available toxicology studies to use in the extrapolation to an estimated safe level for human exposure. Repeated-dose toxicity studies are preferred to use in these extrapolations. We were interested to assess whether the Gen-DART study provided a more conservative point of departure than repeated-dose toxicity studies conducted with adult animals. In DFCN, the repeated-dose SC toxicity study is the most common in vivo toxicity protocol submitted in premarket notifications. Therefore, comparing the Gen-DART study to SC toxicity protocol allowed the most comparisons between a Gen-DART study and a repeated-dose toxicity study conducted with adult animals. We compared points of departure, adverse effects, affected organs, and affected biological systems in both study designs.
To compare Gen-DART and SC toxicity studies, we required that both studies were tested in the same species (rat) and used the same exposure route (oral). A total of 20 FAs were tested in 20 corresponding SC studies that fit these criteria. In the majority of cases, the administration was the same across studies (e.g. oral gavage Gen-DART and oral gavage SC study). In four cases (FAs 19, 21, 34, and 36), the same exposure route (oral) was used, but the method of administration differed (e.g. diet vs. oral gavage). We recognize that comparing oral gavage to dietary administration may introduce some variability or uncertainty in the comparison. However, we did not observe any general trend showing that one route produced different or higher toxicity than another. Furthermore, excluding these comparisons did not alter the outcome of our analyses.
Point of departure comparison
We provide the LOAELs and NOAELs for each study in Table 8.
Summary table of Gen-DART and SC paired study comparisons.a
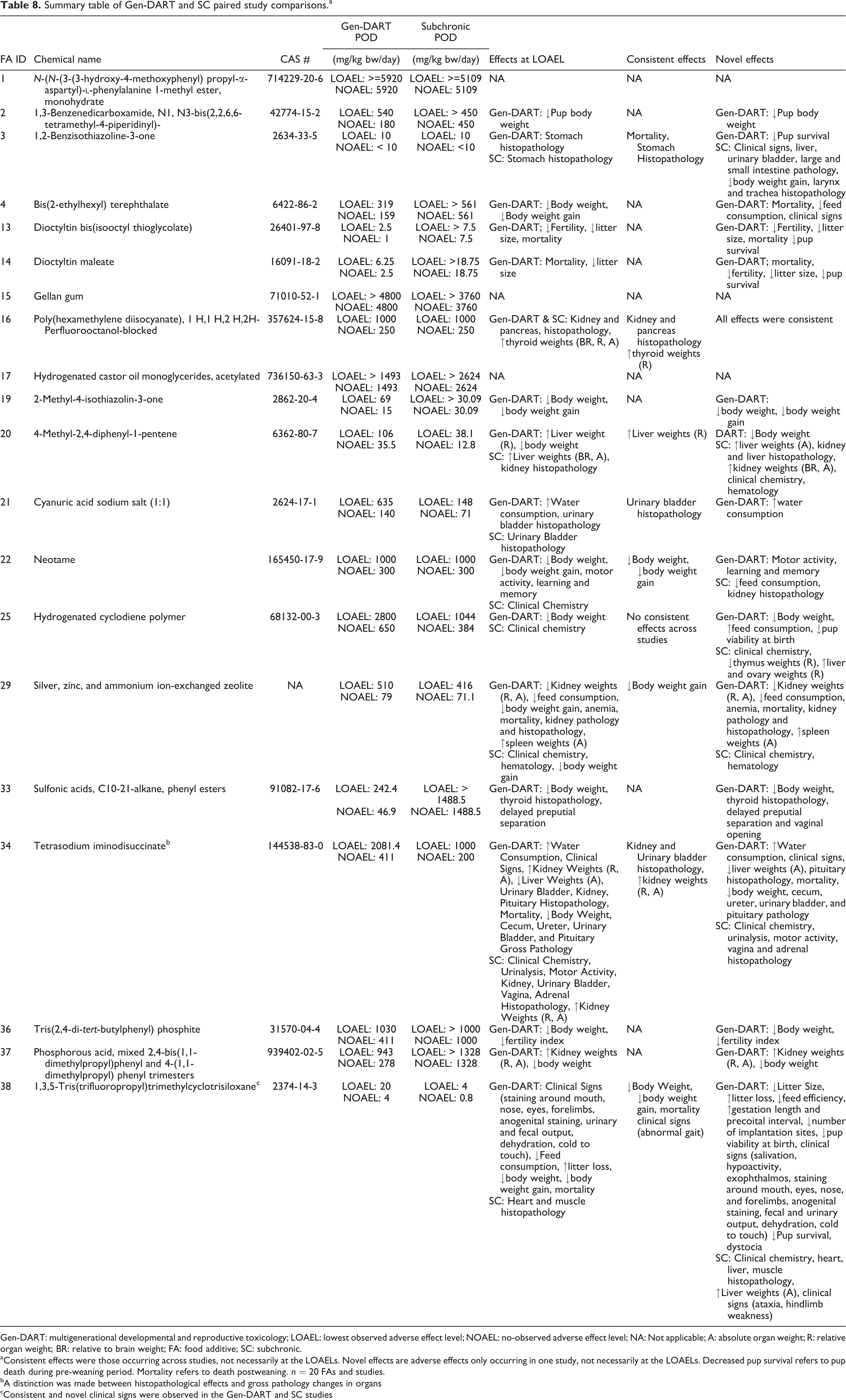
Gen-DART: multigenerational developmental and reproductive toxicology; LOAEL: lowest observed adverse effect level; NOAEL: no-observed adverse effect level; NA: Not applicable; A: absolute organ weight; R: relative organ weight; BR: relative to brain weight; FA: food additive; SC: subchronic.
a Consistent effects were those occurring across studies, not necessarily at the LOAELs. Novel effects are adverse effects only occurring in one study, not necessarily at the LOAELs. Decreased pup survival refers to pup death during pre-weaning period. Mortality refers to death postweaning. n = 20 FAs and studies.
bA distinction was made between histopathological effects and gross pathology changes in organs
cConsistent and novel clinical signs were observed in the Gen-DART and SC studies
There were seven cases where we could not determine which study had the lowest LOAEL due to dose-selection considerations. In four cases, the highest dose tested in the SC study was lower than the high dose in the Gen-DART study. For example, for FA 2 (1,3-benzenedicarboxamide, N1, N3-bis(2,2,6,6-tetramethyl-4-piperidinyl)-), the LOAEL for the Gen-DART study was 540 mg/kg body weight (bw)/day and for the SC study, an LOAEL was not determined with a high dose of 450 mg/kg bw/day. In this case, the SC study may not have tested a dose high enough to observe adverse effects. In the remaining three cases, dose spacing in one study was wider than the other study. For example, for FA 34 (tetrasodium iminodisuccinate), the doses tested in the Gen-DART study were 0, 105, 411, and 2081 mg/kg bw/day compared to the SC study dose levels of 0, 50, 200, and 1000 mg/kg bw/day. In both study types, the study LOAEL was the highest dose tested, but the Gen-DART study had a higher LOAEL possibly driven by large dose spacing. The Gen-DART LOAEL may have been equal to or lower than the SC LOAEL, if the dose spacing was narrower.
In cases where dose spacing did not limit POD comparisons, the Gen-DART studies provided the most conservative study LOAEL in 5 of the 20 cases (25%) and the SC study provided the most conservative study LOAEL in 2 of the 20 cases (10%). There were three cases where both the Gen-DART and SC study provided the same study LOAEL. There were three cases where we could not determine which study type had the most conservative LOAEL because the highest dose tested was the NOAEL.
Overall, neither the Gen-DART nor the SC study consistently provided the most conservative study LOAEL. In cases where an LOAEL was identified in both study types, the ratio between LOAELs was less than a factor of 10 (range 1–5). In cases where the Gen-DART identified an LOAEL but the SC study did not, the ratio between studies was between >1 and >6.
In the five cases where the Gen-DART study provided the most conservative study LOAEL, the effects observed included decreased body weights and body weight gains (FAs 4, 33, and 37), mortality, decreased fertility and litter size (FAs 13 and 14), histopathological changes in the thyroid and delayed preputial separation (FA 33), and increased kidney weights (FA 37).
When we compared study NOAELs, we found that the Gen-DART studies provided the most conservative study NOAEL in 9 of the 20 cases (45%), whereas the SC provided the most conservative NOAEL in 3 of the 20 cases (15%). There were five cases where both the Gen-DART and SC study provided the same or similar NOAEL, two cases that were undetermined due to dose spacing, and one case where both study types observed adverse effects at the first dose tested, resulting in no NOAELs for comparison (Table 8). In most cases, the NOAEL ratios were less than 10. However, in the case of FA 33, sulfonic acids, C10-21-alkane, and phenyl esters, the ratio between the Gen-DART and SC NOAELs was >32 (>1488.5 mg/kg bw/day ÷ 242.4 mg/kg bw/day; Table 8). In this case, the highest dose tested in the SC study did not elicit any adverse effects, whereas the middle dose tested in the Gen-DART study was the study LOAEL. Effects observed at the Gen-DART LOAEL included decreased body weight, altered developmental milestones, and histopathological changes in the thyroid. Therefore, it is possible that the juvenile F1 pups were particularly susceptible to FA 33.
Overall, the Gen-DART studies provided the more conservative NOAEL when compared to the SC studies. The median ratio was 2, range 1 to >32.
Gen-DART and SC effect comparison
Table 8 provides an overall summary of the PODs and effects that occurred in Gen-DART and SC studies for each FA. We focused on effects used to define the LOAEL, consistent effects that were seen in both study types at any dose, and novel effects that were only observed in one study type at any dose. The same FA may have both consistent and novel effects observed in the Gen-DART or SC studies.
Effects at LOAEL
The most common effects at Gen-DART LOAELs were decreased body weight or body weight gains, adverse clinical signs, and organ toxicity (largely restricted to the kidney, liver, and stomach). The SC studies detected a higher frequency of systemic toxicity at the LOAELs, such as changes in clinical chemistry and urinalysis, and organ toxicity.
Consistent effects
Eight FAs observed consistent effects in both the Gen-DART and SC studies. Identical adverse effects (kidney and pancreas histopathology and increase in thyroid weights) and PODs were observed in both studies for FA 16. The general effect categories affected in both study types were body weight (FAs 22, 29, and 38), organ toxicity (FAs 3, 20, 21, and 34), clinical signs (FA 38), and mortality (FA 3).
Organ toxicity consistently affected in both study types included stomach histopathology (FA 3), increased liver weight relative to body and brain weights (FA 20), urinary bladder histopathology (FA 21), increased kidney weights absolute and relative to body weights, and kidney and urinary bladder histopathology (FA 34).
Novel effects
There were eight FAs where toxicity was only observed in the Gen-DART study, whereas adverse effects were not seen in the paired SC study at any dose level. The novel effects observed in the Gen-DART studies were effects on body weights (FAs 2, 19, 33, 36, and 37), developmental milestones (FAs 13, 14, and 33), reproductive function (FAs 13, 14, and 36), mortality (FAs 4, 13, and 14), organ toxicity (e.g. histopathology changes in the thyroid, and kidney weights; FAs 33 and 37), and clinical signs and feed consumption (FA 4).
In other cases (n = 9), both studies elicited adverse effects, but the effects were different in each study. Therefore, the effects in the Gen-DART study were distinct (or novel) from those observed in the SC study. In the Gen-DART studies, the novel effects were reproductive function (FAs 22, 25, and 38), organ toxicity (e.g. the kidney, spleen, liver, seminal vesicles, testes histopathology, and weights changes; FAs 29 and 34), feed/water intake (FAs 25, 29, and 34), developmental milestones (FA 3), and behavior (FA 22). In the SC studies, the unique effects included organ toxicity (e.g. the liver, kidney, urinary bladder, small intestine weights, and histopathological changes; FAs 3, 20, 22, 25, 34, and 38), clinical chemistry (FAs 20, 25, 29, 34, and 38), hematology (FAs 20 and 29), clinical signs (FAs 3 and 38), urinalysis (FA 34), and behavior (FA 34).
Summary
Overall, when compared to the SC study, the Gen-DART studies generally provided more conservative study NOAELs and performed similarly to the SC study when we compared LOAELs. In some cases, dose spacing affected the identification of the lowest POD across studies. General effect categories affected in each study type were generally expected based on the designs of each protocol. For example, the Gen-DART studies uniquely detected the effects on reproductive function and developmental milestones, end points that are not assessed in SC studies. In some cases, the Gen-DART study identified organ toxicity that was not observed in adult animal, suggesting novel toxicological targets in the juvenile animals.
Comparing LOAELs can provide some information about juvenile versus adult sensitivity because this comparison is focused on doses at which effects occurred. Our comparison of LOAELs did not reveal a general trend that F1 or P0 was more sensitive to test article exposure than animals in SC studies.
The only case that showed evidence that the juveniles responded to a much lower exposure to the test article than adults was FA 33, with an NOAEL ratio of >32. In this case, there were no adverse effects observed in the SC study at any dose level. The doses used in both the Gen-DART and SC study overlapped to some degree (Gen-DART: 46.9, 242.4, and 1242.2 mg/kg bw/day; SC: 68.7, 282.6, and 1488.5 mg/kg bw/day) and the absence of toxicity in the SC study was not solely due to inadequate dosing. Instead, it is likely that the juveniles were more sensitive to the effects of the test article than adults. The LOAEL in the Gen-DART study was defined by decreased body weight and delayed developmental milestones (preputial separation and vaginal opening) in the PN F1 pups and body weight and histopathological changes in the thyroid in the P0 generation.
Objective 2: Utility of JAS protocols for infant safety assessment
Study library
Our JAS library contains 51 JAS from 29 drugs reviewed in either INDs or NDAs submitted to US FDA CDER. The majority of studies were conducted with rats (Table 9). Study duration and age at treatment initiation varied based on the test species and the age of the intended pediatric population. Some drugs were tested in more than one JAS. These JAS each tested a separate question; for example, JAS 1 for drug Q tested for effects on reproductive function, while JAS 2 tested for effects on neurodevelopment. Therefore, these JAS are not replicates but are independent studies. Ideally, both end points would have been tested in the same JAS, as outlined in FDA’s guidance. 5
JAS study library.a
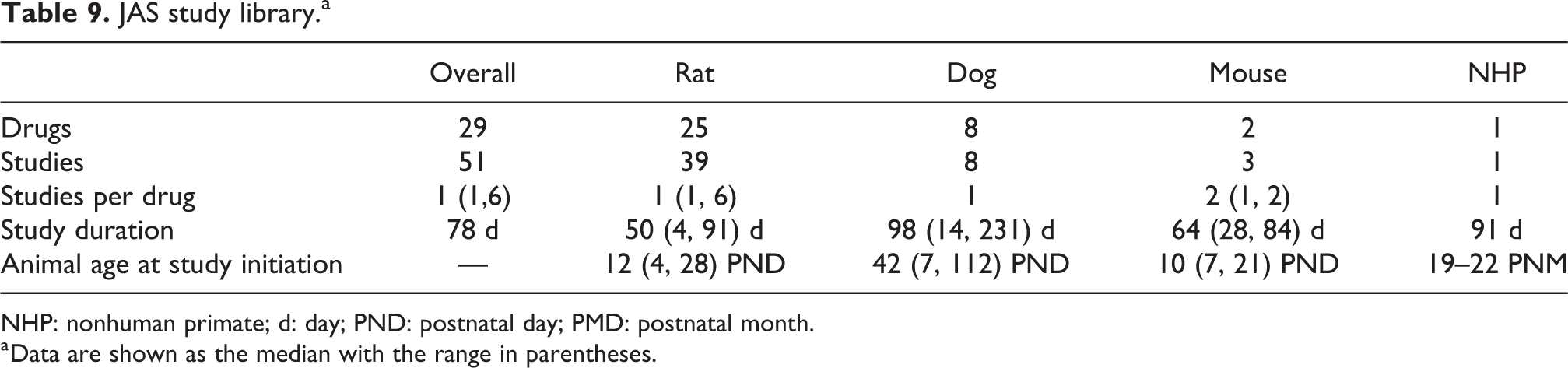
NHP: nonhuman primate; d: day; PND: postnatal day; PMD: postnatal month.
a Data are shown as the median with the range in parentheses.
JAS are conducted in non-rodent species only when the pharmaceutical target or mode of action is better tested in non-rodents, or there is another scientific basis to deviate from the rodent model. In DFCN, Gen-DART studies are exclusively conducted in rodent species, predominately the rat. For the purposes of evaluating the JAS protocol for use in the DFCN infant safety assessment paradigm, we restricted our analysis to JAS conducted in rats.
Fifteen of the 29 drugs had central nervous system (CNS)-focused indications like epilepsy, major depressive disorder, and attention-deficit hyperactivity disorder. Seven drugs had metabolic- and endocrinology-related indications like types I and II diabetes; six drugs had gastroenterology indications like irritable bowel syndrome and constipation. One drug had a urological indication.
Affected systems
Our analysis of alerts for neurotoxicity, endocrine toxicity, reproductive toxicity, immunotoxicity, or skeletal toxicity showed that JAS routinely detect toxicity in these systems (Table 10). The neurological and endocrine systems were most commonly affected at the JAS study LOAELs.
JAS protocols detect effects in systems considered important for infant safety assessment.a
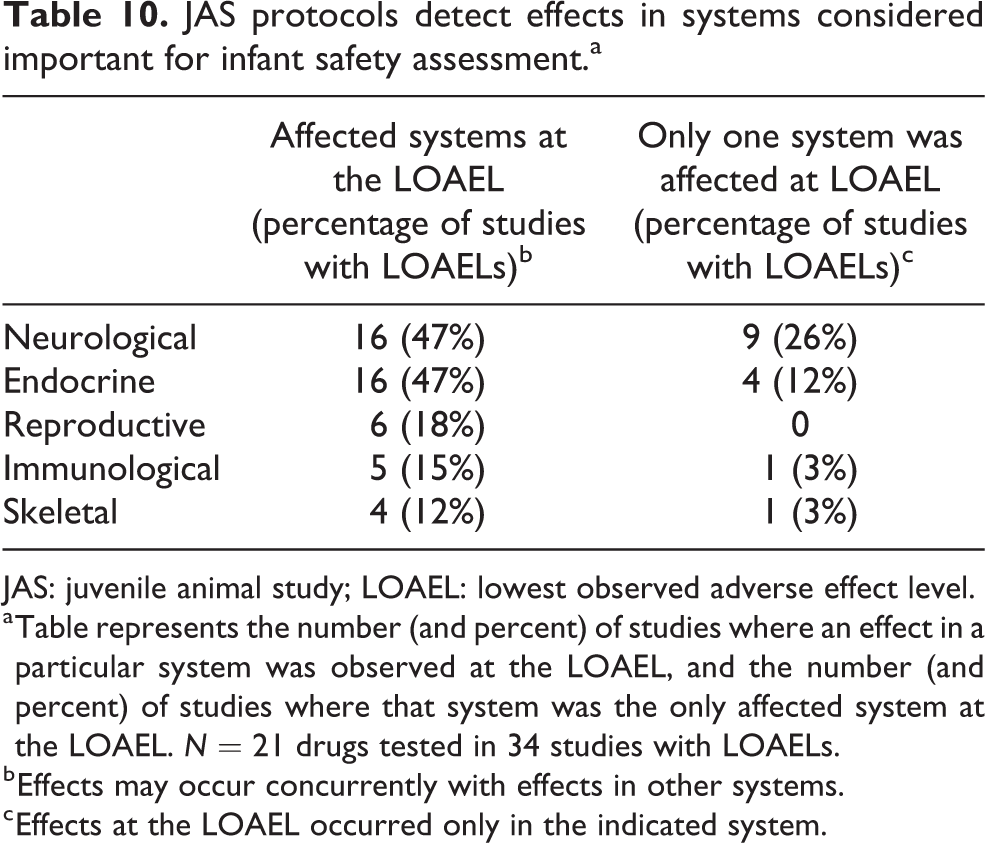
JAS: juvenile animal study; LOAEL: lowest observed adverse effect level.
a Table represents the number (and percent) of studies where an effect in a particular system was observed at the LOAEL, and the number (and percent) of studies where that system was the only affected system at the LOAEL. N = 21 drugs tested in 34 studies with LOAELs.
b Effects may occur concurrently with effects in other systems.
c Effects at the LOAEL occurred only in the indicated system.
Unlike the Gen-DART library, the JAS library included studies showing alerts for skeletal toxicity. The JAS protocol is better powered to detect effects on skeletal toxicity than the Gen-DART protocol because specialized end points related to bone growth can be included in the study design. For example, JAS protocols may include measurements of bone density, diametric growth, and longitudinal growth that are not included routinely in Gen-DART protocols.
Similar to the Gen-DART library, there were cases where only one of the five systems we examined affected the study LOAEL (Table 10). The neurological system was most frequently the sole system affected at the LOAEL, followed by the endocrine, skeletal, and immunological systems. Examples of alerts solely observed at the study LOAEL include altered motor activity and histopathological changes in the brain (neurological), decreased crown-rump length and altered developmental milestones (endocrine), decreased lymphocytes and immune response (immune), and decreased ulna growth and histopathological changes in the bone (skeletal). All the reproductive alerts at the study LOAEL occurred concurrently with effects in other systems.
General effect categories
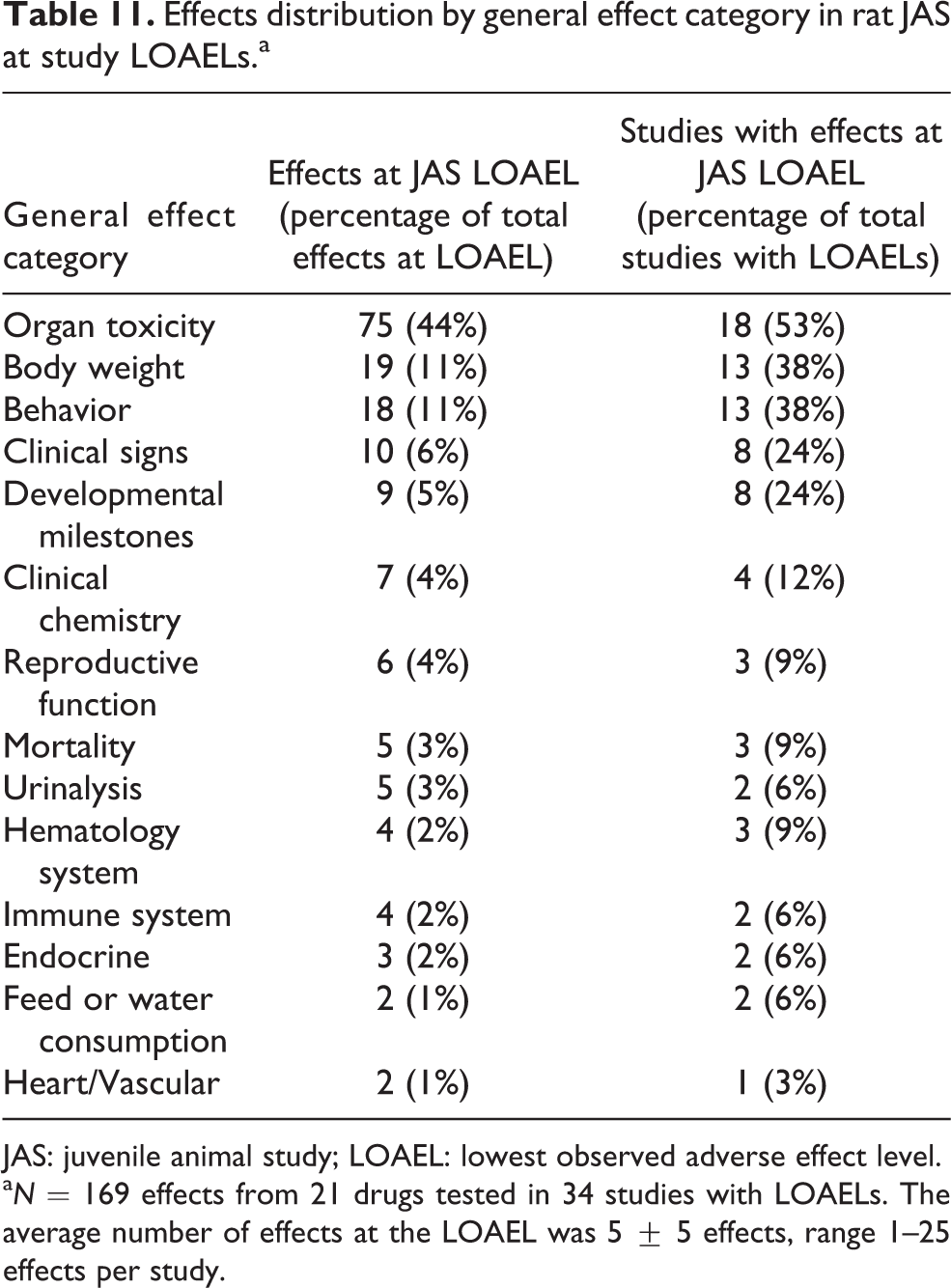
A total of 397 adverse effects were observed in the rat JAS (39 studies). Many effects (169 effects, 43%) were observed at the study LOAELs. Similar to the Gen-DART analysis, we assessed all effects occurring at the LOAELs in rat JAS. We also assessed the percentage of studies that observed effects at the LOAEL in each general effect category (Table 11). Organ toxicity, body weight, and behavioral effects were the most commonly affected categories in the JAS.
Effects distribution by general effect category in rat JAS at study LOAELs.a
JAS: juvenile animal study; LOAEL: lowest observed adverse effect level.
a N = 169 effects from 21 drugs tested in 34 studies with LOAELs. The average number of effects at the LOAEL was 5 ± 5 effects, range 1–25 effects per study.
Similar to the Gen-DART studies, the same top three categories were affected when we examined the study incidences at the LOAELs. There were cases where a study had more than one effect in a general effect category at the study LOAEL. For example, we sometimes observed multiple effects in the behavioral category that occurred concurrently at the LOAEL, such as altered motor activity and altered learning and memory. A variety of effects were observed in different organs detailed further in Table 12.
Effects distribution by organ toxicity in rat JAS at study LOAELs.a
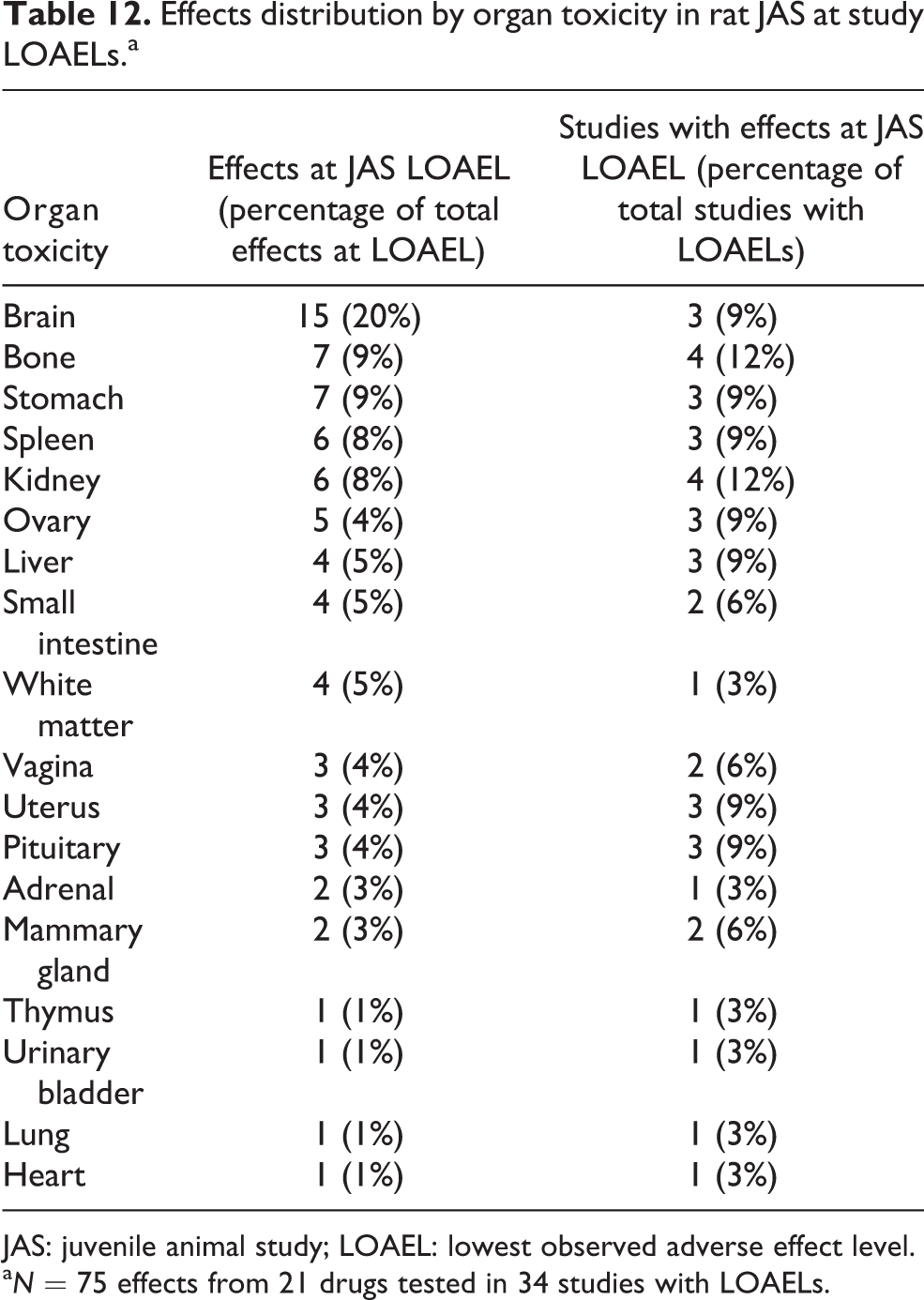
JAS: juvenile animal study; LOAEL: lowest observed adverse effect level.
a N = 75 effects from 21 drugs tested in 34 studies with LOAELs.
Organ toxicity
Similar to the Gen-DART library, organ toxicity was one of the most common types of toxicity in rat JAS (75 effects). The brain (20% of LOAEL effects), bone (9% of LOAEL effects), and stomach (9% of LOAEL effects) were the most commonly affected organs, which were consistent with the pharmaceutical targets of the drugs (Table 12).
The top three affected organs differed slightly in terms of study incidences compared to total effect incidence. The kidney and bone were affected at the LOAEL in 12% of studies, followed by the brain and stomach (but also the spleen, ovary, liver, uterus, and pituitary gland; all 9% of studies). All three of the JAS that observed toxicity in the brain at the LOAEL were for drugs with CNS indications. Twelve of the 15 incidences were observed in one study which included numerous related effects such as altered absolute and relative brain weights with accompanying histopathological changes.
JAS, SC, and PN toxicity cross-study comparison
While the Gen-DART design allows comparison between juvenile and adult toxicity within a single protocol, the JAS only examines juvenile toxicity. Therefore, we compared the JAS to SC studies to compare juvenile versus adult toxicity. We also compared JAS to other DART protocols that studied effects in F1 pups, specifically the PPN toxicity assay and multigenerational studies, to learn what additional information is provided by the JAS compared to other DART protocols. We grouped these studies together as alternative measures of PN testing. In cases where a drug had more than one study per protocol, the study with the most conservative (lowest) LOAEL was selected for the cross-study point of departure comparison.
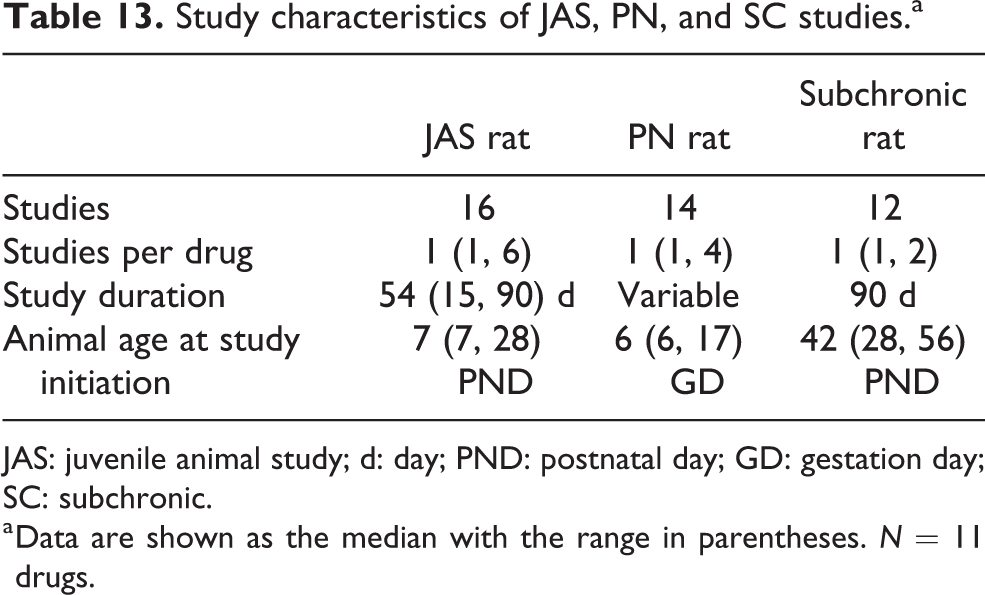
To be included in the cross-study analysis, the drug had to be tested in the same species (rat) and the same route of exposure (e.g. oral, subcutaneous) for all studies. We identified 11 drugs that had been tested in concordant 90-day SC and PN studies (two-generational or PPN; Table 13). In all but two cases, the same routes of administration were used. For drugs A and Q, the route of exposure was the same (oral) but used a different method of administration (e.g. oral gavage vs. diet).
Study characteristics of JAS, PN, and SC studies.a
JAS: juvenile animal study; d: day; PND: postnatal day; GD: gestation day; SC: subchronic.
a Data are shown as the median with the range in parentheses. N = 11 drugs.
Point of departure comparison
We provide the LOAELs and NOAELs for each study in Table 14.
We found that the JAS provided the most conservative study LOAEL in four cases (36%), whereas the PN study provided the most conservative study LOAEL in five cases (45%). None of the SC studies provided the most conservative study LOAEL when compared to the JAS and the PN studies. There was one case, drug Q, where the JAS and the PN had the same study LOAEL. In the case of drug E, none of the three study designs observed a study LOAEL (Table 14). In most cases, the ratio between the LOAELs was equal or less than a factor of 10 (range 2–10). In the case of drug K, the ratio was greater than 500 based on the PN study LOAEL of 0.004 mg/kg bw/day compared to the JAS and SC studies that did not observe effects even at a high dose of 2 mg/kg bw/day. Hypoactivity, decreased feed consumption, and decreased body weights were observed in the pregnant dams at the study LOAEL of the PN study, but the absence of any effect in the JAS or SC study suggests that maternal animals may have been more sensitive to the test article than juvenile or adult (nonpregnant) animals.
Summary table of JAS, SC, and PN studies cross-study effect analysis.a
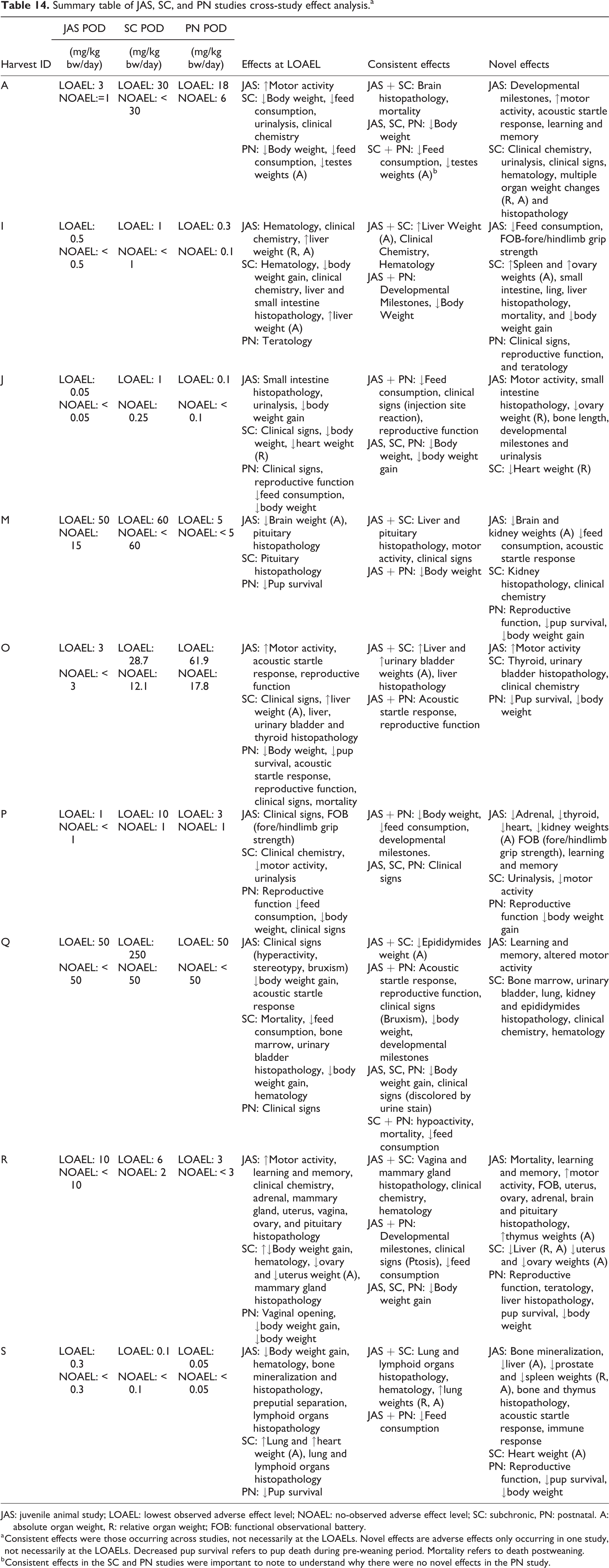
JAS: juvenile animal study; LOAEL: lowest observed adverse effect level; NOAEL: no-observed adverse effect level; SC: subchronic, PN: postnatal. A: absolute organ weight, R: relative organ weight; FOB: functional observational battery.
a Consistent effects were those occurring across studies, not necessarily at the LOAELs. Novel effects are adverse effects only occurring in one study, not necessarily at the LOAELs. Decreased pup survival refers to pup death during pre-weaning period. Mortality refers to death postweaning.
b Consistent effects in the SC and PN studies were important to note to understand why there were no novel effects in the PN study.
Generally, when comparing the LOAELs of all three study designs, the JAS and the PN study performed similarly and both protocols provided a lower LOAEL than the corresponding SC study.
When we compared study NOAELs, the JAS provided the most conservative NOAEL in 3 of the 11 cases (27%). For drug K, the PN study may have provided the most conservative NOAEL (<0.004 mg/kg bw/day) and for drug E, the PN and SC studies provided the same study NOAEL, both lower than the JAS NOAEL (Table 14). We could not determine which protocol provided the most conservative NOAEL in six cases because at least two study types did not observe a study NOAEL. For those six drugs, adverse effects were observed at the lowest dose tested in multiple protocols. Generally, most of the NOAEL ratios ranged from 1 to 6, with the exception of drug K. Since the lowest dose tested in the PN study was the study LOAEL, the fold difference would be greater than 500.
Toxicokinetics
The juvenile rats had a higher internal exposure than the adult rats in 2 of the 9 (22%) drugs, a lower exposure in 4 of the 9 (44%) drugs, and similar exposure as the adults in 3 of the 9 (33%) drugs (Table 15). A higher juvenile internal exposure suggests that the juveniles may not be more sensitive to the drug, particularly if we see a similar response in both juveniles and adults. Instead, the juveniles may be exhibiting TK differences that allow greater accumulation of the drug in the blood. If adverse effects are observed at a lower internal exposure in juveniles than adults, it may suggest that the juveniles are more sensitive to the drug because they are responding to a lower blood level, possibly due to a toxicodynamic difference. Finally, a similar internal exposure to adults may suggest that that the juveniles did not exhibit a TK or a toxicodynamic difference and responded similarly to the administered drug. These data show that juvenile TK is often different from TK in adult animals. This is an important finding because it suggests that without TK measurements, it may be hard to predict whether the TK profile is similar to juvenile and adult animals. Furthermore, an understanding of whether juveniles are more sensitive than adult animals is not possible without also knowing the internal exposures for both.
Juvenile and adult rat PK comparison.a
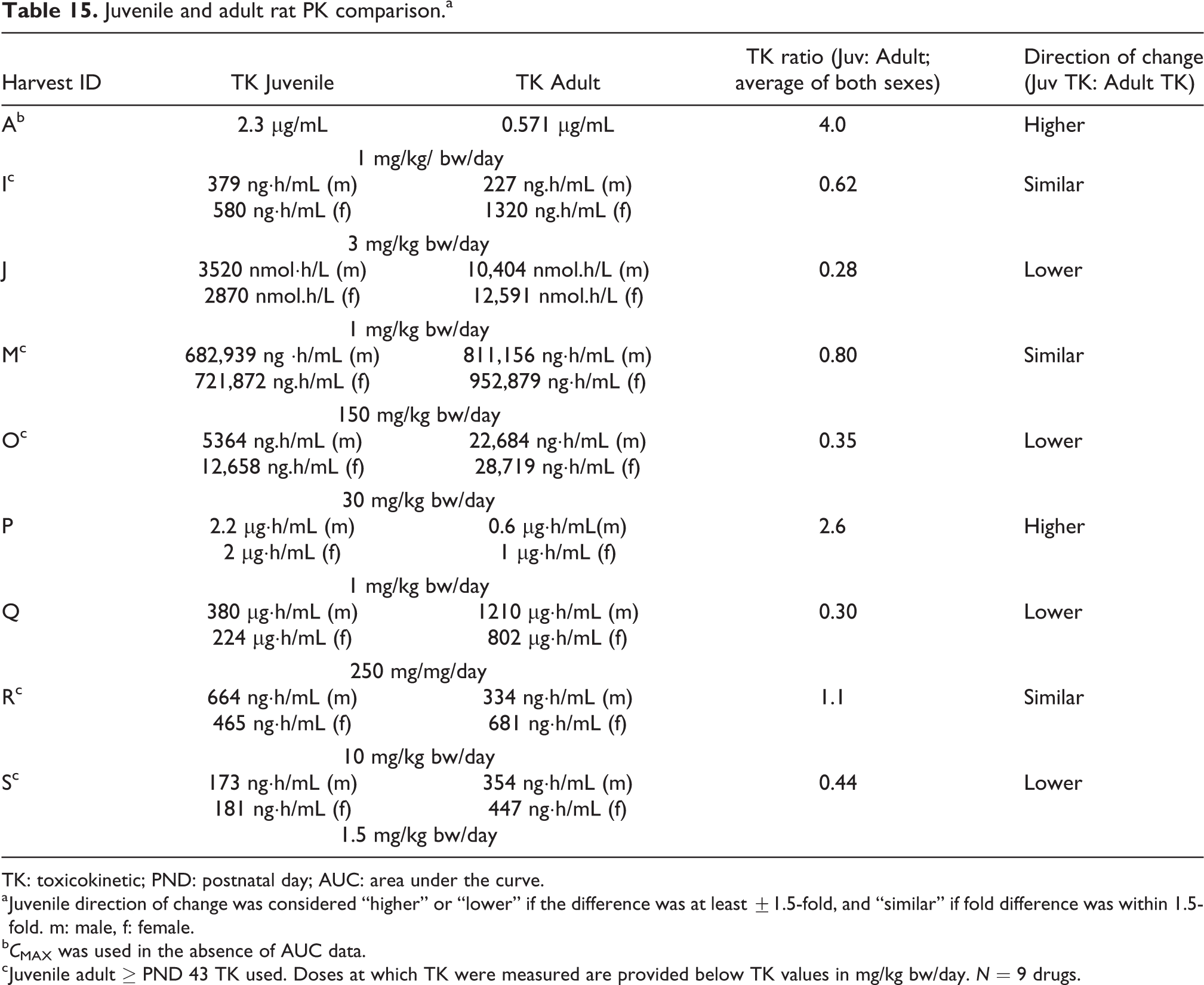
TK: toxicokinetic; PND: postnatal day; AUC: area under the curve.
a Juvenile direction of change was considered “higher” or “lower” if the difference was at least ±1.5-fold, and “similar” if fold difference was within 1.5-fold. m: male, f: female.
b C MAX was used in the absence of AUC data.
c Juvenile adult ≥ PND 43 TK used. Doses at which TK were measured are provided below TK values in mg/kg bw/day. N = 9 drugs.
Cross-study effect analysis
Drugs K and E were eliminated from the effect analysis because one or more study type did not observe a study LOAEL and therefore had no effects to compare with the other study types. As a result, a total of nine drugs were available for our cross-study effect analysis.
Effects at LOAEL
At the study LOAELs, the JAS commonly observed organ toxicity (e.g. effects on the brain in drug M and reproductive organs in drug R) and behavioral effects that affected motor function and acoustic startle response (drugs A, O, P, Q, and R). The PN studies commonly observed effects on body weight (drugs A, J, O, P, and R), adverse clinical signs (drugs J, O, P, and Q), and altered developmental milestones (e.g. pup survival in drugs M, O, and R). The SC studies often observed organ toxicity (e.g. liver urinary bladder, ovary histopathological, and weight changes in drugs I, J, M, O, Q, R, and S) and body weight effects (drugs A, I, J, Q, and R) at the LOAEL (Table 14).
Consistent effects
Some consistent organ toxicity effects were seen in JAS and SC studies, which included histopathological or weight changes in the liver, lung, urinary bladder, pituitary, lymphoid organs, brain, and reproductive organs. Consistent effects on feed consumption, clinical signs, reproductive function, and body weight effects were seen in JAS and PN studies. Body weight and clinical signs were often affected in all three study types (drugs A, J, P, Q, and R).
Novel effects
The JAS revealed novel effects in all nine drugs in the cross-study comparison. The novel information provided by the JAS included effects on developmental milestones, behavior, organ toxicity (e.g. histopathological and weight changes in the small intestine, kidney, ovary, adrenal, thyroid), and feed consumption. In general, the novel effects seen in SC and PN studies were consistent with their respective protocols. SC studies provided novel information on urinalysis, clinical chemistry, hematology, clinical signs, and organ toxicity effects (e.g. histopathological changes in the bone marrow, urinary bladder, lung, kidney, and epididymides in drug Q), while PN studies provided novel information on adverse effects on reproductive, teratologic, clinical signs, body weight, and developmental milestones (drugs I, M, O, P, and R).
Summary
Overall, when compared to the PN and SC studies, the JAS provided the most conservative study NOAELs and performed similarly to the PN study when we compared the study LOAELs. In the cross-study effect comparison, we observed that the JAS provided novel information in all drugs that included effects on developmental milestones (e.g. sexual maturation), behavior (e.g. motor activity, acoustic startle response, learning and memory), and organ toxicity (e.g. histopathological and weight changes in the small intestine and kidney).
Generally, findings observed in the JAS, SC, and PN studies were consistent to the protocol design. The JAS observed more organ toxicity and behavioral effects, the PN studies detected more reproductive, feed or water intake and teratological effects, and the SC study observed more clinical chemistry, hematology, urinalysis and immunotoxic effects.
We observed cases where the juvenile TK parameters of internal exposure were less than, greater than, or equal to adult levels. This information allowed us to understand whether the juvenile animals exhibited potential differences in TK or toxicodynamic factors compared to adults.
After comparing the effects observed in the JAS, PN, and SC studies, we found that the SC study was toxicologically consistent with the JAS in 4/9 (44%) drugs, and the PN study was toxicologically consistent with the JAS in 1 of the 9 (11%) drugs. For drug J, even though the PN study observed consistent effects in body weight, feed consumption, and changes in the reproductive function, the JAS observed several additional novel effects including developmental milestones, behavior, urinalysis, and organ toxicity in the small intestine, ovary, and bone length changes. For this reason, we determined that neither the SC nor PN studies were toxicologically consistent with the JAS.
The case of drug J demonstrates the added value of the JAS. Specialized neurobehavioral, skeletal, and behavioral end points in the JAS allowed detection of toxicity that was not observed in adult animals even though all three study types had the same high dose (1 mg/kg bw/day).
Discussion
This article summarizes the first assessment of the toxicological effects observed in FDA-reviewed Gen-DART studies on FAs and JAS on drugs to determine the utility of these protocols for infant food contact safety assessment. Comprehensively, we indexed the toxicological effects observed in 41 Gen-DART studies, 51 JAS, and 52 additional studies associated with the same chemicals (e.g. SC studies, PN studies), for a total of 144 repeated-dose toxicity studies. Both the Gen-DART and JAS protocols can be useful for infant safety assessment, but the level of information available for a specific chemical may dictate which protocol will be preferred.
The objectives of this work were twofold: First, we sought to understand what information the protocols historically used by OFAS to assess DART provided for infant-specific safety assessments of food contact materials. Second, we sought to determine whether the newer JAS protocol, not currently recommended by OFAS, could be useful in the DFCN infant-specific safety assessment paradigm. The overarching goal of this work was to provide more quantitative advice regarding DART protocol selection to support the safety of infant food contact materials.
Due to the confidential business information in FDA premarket submissions, we are unable to provide some details regarding the studies in our data library. For example, we can provide names and CASRNs for regulated FAs or food contact substances. However, we are prohibited from sharing any information that may reveal drug identity in this publication. Despite these limitations, we believe that our article can provide helpful information to industry stakeholders who may be considering approaches to address the infant safety of food contact materials.
There are a small number of similar assessments on the performance of Gen-DART or JAS protocols in the context of regulatory toxicology. In a study comparing 47 reproductive toxicants and 75 nonclassified toxicants, the ratio between the NOEL/NOAEL derived in a 90-day study and that derived in a two-generation study was less than 10 for most substances, and the geometric mean was 1.9 (for reproductive toxicants) and 1.3 (for nonclassified toxicants). 7 The authors indicated that in the cases where the differences in NOEL/NOAELs were larger than 10-fold, there was a large dose spacing in the two-generation assay that may have contributed to the lower NOAEL. We also observed an effect of dose spacing on cross-study comparisons between the Gen-DART and SC protocols. However, in the single case where the NOAEL ratio between the Gen-DART and the SC study exceeded 10× (FA 33), dose spacing was not a factor. Instead, the animals in the Gen-DART study (FA 33) showed treatment-related adverse effects (LOAEL of 242.4 mg/kg bw/day), while the animals in the SC study did not respond to the test article at any tested dose (up to 1488.5 mg/kg bw/day).
Two papers were published that described the effects observed in over 240 JAS. 8,9 These publications described the findings from JAS conducted by the pharmaceutical industry and voluntarily submitted to the International Life Science Institute Health and Environmental Science Institute Developmental and Reproductive Toxicology Committee. Similar to our data library, the data set used to inform these publications included predominately rodent JAS, with relatively fewer non-rodent studies. Similar to our library, the most frequent drug indication in their JAS was for the CNS. One major difference is that our analysis provides greater detail regarding the toxicological effects observed, organs affected, and categorical toxicity. Additionally, these publications do not perform cross-study comparisons. Instead, the authors report whether a given finding may have been “predicted” from other data. This approach is limited by the fact that insufficient details are provided about individual drugs that allow a transparent understanding of how the predictive ability was determined. Ultimately, conclusions about whether a given finding could be predicted before doing a toxicology study are highly subjective and affected by retrospective bias. Our analysis suggests that in all of our cross-study comparisons, novel or unique information was provided by the JAS that was not obtained in other study protocols. Further, the JAS provided a lower LOAEL in many cases. These comparisons show the utility of the JAS for providing new information about potential juvenile sensitivity or toxicological effects. However, findings in JAS that suggest a lack of concern for juvenile sensitivity may provide a reassurance of safety that is just as important in regulatory decision-making as the observation of toxicological effects. Currently, the ability to predict developmental toxicity from modeling, computational toxicology, in vitro testing, or read-across approaches is imprecise at best; until this ability expands, we remain cautious in our ability to definitively predict the developmental toxicity of chemicals intended for use as consumer products in the absence of confirmation studies conducted in vivo.
One limitation of our work is the relatively small sample size of the cross-study comparisons. Gen-DART studies are large, resource-intensive studies and are generally not conducted unless recommended by regulatory authorities or undertaken to address specific safety questions. Usually an SC study has also been conducted for a chemical tested in a Gen-DART or JAS protocol, but sometimes the route of exposure does not match between the two study designs (e.g. inhalational vs. oral exposure). This was the most common reason for the reduced number of comparisons in the cross-study assessment for both FAs and drugs. Although the number of cross-study comparisons was limited, we were still able to make important observations about the general consistency of toxicological effects observed across study designs as well as novel or unique information provided in the Gen-DART or JAS protocols compared to other study designs.
There are some important differences between the types of chemicals used as FAs and those used as drugs. In general, FAs, particularly direct additives, exhibit low potential for toxicity in animals. NOAELs and LOAELs are generally high and can routinely be greater than 1000 mg/kg bw/day. In contrast to direct FAs, food contact chemicals are not purposeful additions to food. For example, they are often industrial manufacturing precursors, unreacted starting materials, biocides, or low-molecular-weight oligomers that migrate at low levels into food from food contact materials. These types of chemicals may exhibit higher toxicity than direct additives but do not show targeted pharmacological effects that are the hallmark of drugs. Since drugs exhibit designed pharmacology, they tend to show targeted effects on specific organ systems consistent with that pharmacology. This difference between FAs and drugs is somewhat illustrated by our data on organ effects. FAs in our database tend to elicit adverse effects in liver, kidney, urinary bladder, and stomach (Table 3), while the drugs in our database elicited adverse effects in brain, bone, and stomach (Table 6) consistent with their intended pharmacology (neurological, endocrine, and gastroenterology indications).
Despite these generalized differences between chemical categories (direct FAs, food contact chemicals, and drugs), we can evaluate the JAS protocol tested with drugs for use in food contact safety assessment because it allows us to see how the protocol performs with chemicals that elicit toxicity. Direct FAs and food contact chemicals are much less biologically active than drugs. However, if we seek to evaluate whether a food contact chemical elicits specific effects in a developing system, this may be best tested in a JAS.
The JAS protocol accommodates information on mode-of-action information, target organ information, and findings observed in preceding repeated-dose toxicity studies conducted in adult animals. This information can be used to include specialized end points relevant to the known toxicity associated with the drug. These specialized end points may enhance the sensitivity of the study to provide a lower POD than the preceding studies associated with that drug, as observed with drug J in our database. Further, the inclusion of PK/TK measurements provides insight regarding internal exposure that is currently not routinely measured in Gen-DART studies.
In contrast, the Gen-DART study can provide novel information on the effects of a test substance on reproductive function and developmental milestones that are not tested in other study designs currently recommended for the safety assessment of FAs, including food contact substances. Gen-DART protocols have the unique advantage of potentially revealing differences in the response of adult and juvenile animals to test article exposure within the same study. Additionally, the Gen-DART study may reveal target organ toxicity in F1 animals that is not seen in adult animals tested in SC studies, possibly due to developmental toxicodynamic or TK factors.
Our analyses suggest that JAS may be useful for infant safety assessment of food contact materials when specific mechanistic information is available; namely, mode-of-action information and target organ or receptor information. However, this level of information is generally lacking for environmental chemicals, including food contact substances and food contaminants. There are a couple of options to address these data gaps. First, mechanistic studies in in vitro or alternative models may provide information regarding putative mode of action or molecular targets of understudied chemicals. Findings from these studies may need to be confirmed in in vivo assays to increase confidence in the validity of the in vitro or alternative test findings. Dose range finding studies (DRFs) in juvenile animals may be modified to include end points to resolve questions regarding toxicological target or mode of action; however, protocol modifications need to be carefully considered to ensure that the number of animals (usually low in a DRF) is adequate to power the study.
For chemicals that lack mode of action and molecular or organ target information, the Gen-DART protocol may be best suited to assess the safety of chemicals for infants. Gen-DART protocols cover all lifestages (excepting elderly stages) and include measurements in systems that are particularly sensitive to early life disruption by chemicals, such as the reproductive, endocrine, neurological, skeletal, and immunological systems. Gen-DART protocols can include special cohorts to provide additional information regarding specific end points, such as developmental neurotoxicity, if justified based on hazard identification or other information. It may also be possible to include specialized end points that have been shown to be particularly useful or sensitive in JAS (e.g. bone mineral density measurements) or have been identified in in vitro or alternative models, to increase the sensitivity of Gen-DART protocols. Again, protocol modifications should be dictated by known or suspected hazards associated with the chemical in question and balanced with the need to conduct adequately powered studies. There are other ways to enhance the Gen-DART protocol to reduce uncertainty regarding study interpretation. For example, routinely incorporating TK end points in the Gen-DART study could greatly enhance our ability to determine the extent of juvenile exposure and whether the juveniles were more sensitive to the test article than adults. Additionally, as more developmental studies include TK end points, more data will be available for in silico modeling, such as physiologically based pharmaco(or toxico)kinetic modeling (PBPK or PBTK). Ultimately, refinement of PBPK or PBTK modeling for developmental lifestages could greatly enhance regulatory decision-making by reducing uncertainty regarding chemical disposition in potentially susceptible populations.
Some effects seen in an SC study may prompt a recommendation of a Gen-DART or JAS. In particular, effects observed in any of the five systems of particular interest for infant food contact safety (reproductive, endocrine, neurological, immune, or skeletal systems) may need to be followed up in either a Gen-DART or a JAS study. Protocol selection will likely be driven by the amount of information available for the chemical under study (see above) as well as the estimated infant-specific exposure to the migrating chemical. 1
We conclude that both the Gen-DART and JAS protocols can be useful for the infant safety assessment of food contact materials. Specialized DART testing may not be recommended in every case, depending on the infant-specific exposure and known properties of the chemical under review. The US FDA offers prenotification consultations (PNCs) for notifiers considering submitting an FCN. We encourage prospective notifiers to use the PNC process, particularly when there may be questions regarding toxicological study design for infant food contact safety assessments.
Footnotes
Acknowledgements
The authors gratefully acknowledge the feedback of the OFAS/DFCN Infant Toxicology Working Group and the CDER/OND PTCC Pediatrics Subcommittee. We thank Dr. Darren Fegley of CDER/OND for his contributions to group discussions during JAS data analysis. Dr. Abigail Miller of OFAS/DFCN curated chemical name, CASRN, and intended use. Dr. Kirk Arvidson and Dr. Patra Volarath of OFAS/DFCN and Dr. Chihae Yang (Altamira) provided helpful suggestions regarding database design and population. Mr. Ayomide Igun contributed to this project by harvesting data associated with Gen-DART studies during the summer of 2017.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
