Abstract
Purine nucleosidase (EC 3.2.2.1) catalyzes the N-riboside hydrolysis of purine nucleosides to D-ribose and a purine base. This enzyme may be used in the production of beer and other alcoholic beverages to reduce the purine content of these products. Purine nucleosidase was obtained from Aspergillus luchuensis naturally occurring in grain sources. The safety profile of purine nucleosidase is not well documented in the scientific literature, and a series of toxicological studies were undertaken to investigate the safety of its use in food production. Purine nucleosidase from A. luchuensis was non-mutagenic and non-clastogenic in a standard Ames test and in vitro mammalian chromosome aberration assay. Administration of purine nucleosidase in a 90-day subchronic toxicity study in Sprague-Dawley rats did not elicit adverse findings on any hematology, clinical chemistry, urinalysis, organ weight, or histopathological parameter at doses up to 1700 mg total organic solids (TOS)/kg body weight/day, the highest dose tested. The results suggest purine nucleosidase to lack systemic toxic effect. The no-observed-adverse-effect level was concluded to be 1700 mg TOS/kg body weight/day. The results of the toxicology studies support the safety of purine nucleosidase from a non-genetically modified strain of A. luchuensis when used in food production.
Introduction
Purine nucleosidase (purine β-ribosidase, purine nucleoside hydrolase, purine ribonucleosidase, EC 3.2.2.1) catalyzes the irreversible N-riboside hydrolysis of purine nucleosides to yield D-ribose and a purine base.1,2 Pyrimidine nucleosides and pyrimidine nucleotides, nicotinamide adenine dinucleotide, nicotinamide adenine dinucleotide phosphate, and nicotinamide mononucleotide are not substrates of this enzyme.1,2 Many variants of nucleosidase have been isolated from numerous sources including bacteria,2–4 fungi, 5 protozoans,6,7 and other higher organisms.8–10
A search of the literature did not yield any indication of widespread commercial use of purine nucleosidases. A number of patents for the use of various purine nucleosidases in food production were identified, predominantly in the manufacture of beer.11–14
Purine nucleosidases are of interest to the food technology sector due to their ability to catabolize nucleosides and ability to generate ribose from nucleoside substrates. A purine nucleosidase obtained from Aspergillus luchuensis isolated from grain sources is currently under investigation for potential commercial application by the food industry. This strain was phylogenetically identified as A. luchuensis through the calmodulin and β-tubulin genes. A. luchuensis is a member of the Aspergillus Section Nigri and is commonly considered synonymous with Aspergillus foetidus, Aspergillus kawachii, and Aspergillus acidus due to similarities in their rDNA-ITS, β-tubulin, and calmodulin sequences.15,16 These organisms also share similar morphology to Aspergillus niger and Aspergillus tubingensis, both of which are commonly used as fermentation organisms in the food industry. 16 The whole genome of A. luchuensis (A. luchuensis strain NBRC 4314) has been sequenced17,18 and shows close homology with A. niger with an average nucleotide identity of 93%. 15
A. luchuensis is part of the Aspergillus group known as “black koji molds,” which are traditionally used in the fermentation process to provide enzymes that macerate or saccharify rice to produce koji and are subsequently used in the production of rice-based alcoholic beverages such as awamori, shochu, and makgeolli that are commonly consumed in Japan and Korea.16,19 A. luchuensis has been identified in fermented Pu-erh tea 20 and meju and nuruk in Korea. 16 A. luchuensis has been noted for its importance in Japanese enzyme technology. 21 Despite its classification as filamentous fungi, A. luchuensis does not appear to produce mycotoxins.16,22 The European Food Safety Authority has considered the food enzyme xylanase produced from a genetically modified A. luchuensis Inui strain (RF7398) 23 to be safe under its conditions of use. The history of use of A. luchuensis by the food industry and lack of production of mycotoxins and toxic secondary metabolites demonstrate its suitability as a source organism for enzymes.
As few, if any, purine nucleosidases have been utilized extensively in commercial food manufacturing settings, there are no safety data on any variants of purine nucleosidases (EC 3.2.2.1) in the published literature. To evaluate the possible toxicological risk of residual amounts of purine nucleosidase that could be present in food manufactured with purine nucleosidase from A. luchuensis, a series of toxicology studies were conducted in accordance with standardized guidelines by the Organisation for Economic Co-operation and Development (OECD). These included genotoxicity studies (in vitro bacterial reverse mutation and mammalian chromosome aberration assays) and a 90-day oral subchronic toxicity study in Sprague-Dawley rats. The findings of these studies support the safety of purine nucleosidase produced from A. luchuensis when used in food production.
Materials and methods
Test article
Shin Nihon Chemical Co. (Anjo, Aichi, Japan) provided a liquid concentrate of purine nucleosidase obtained from a non-genetically modified strain of A. luchuensis. The test article used in all studies was obtained from the same manufacturing batch (Lot No. 170925T) and met specification criteria established for the product based on internationally recognized purity criteria for enzyme preparations.24,25 The enzyme concentrate contained a total organic solids (TOS) content of 16% and an enzyme activity of 4798 U/g nucleosidase activity, equivalent to 170 mg total organic solids (TOS) per milliliter. Throughout the 90-day study period, the test article was stored in airtight containers at −28.6 to −20.4°C and remained stable under these conditions as verified by consistent enzyme activity and ultraviolet absorbance (280 nm).
The purine nucleosidase was obtained by solid-state fermentation of A. luchuensis strain PZ-1. Selection of the production strain was based on its capacity for the production of high levels of nucleosidase activity, its cell viability, and its suitability for industrial production due to its lack of mycotoxin production. The A. luchuensis production strain was deposited at Biological Resource Center, National Institute of Technology and Evaluation.
Genotoxicity studies
Bacterial reverse mutation test (Ames test)
The bacterial reverse mutation test was conducted in accordance with OECD Guideline No. 471 26 and OECD Good Laboratory Practice (GLP). 27 The test was conducted using the pre-incubation method and a “treat-and-wash” method. The “treat-and-wash” method is employed to exclude false positive results that may arise due to the presence of free amino acids (e.g., histidine) in the test substance, which can enhance the overall bacterial growth, including background bacteria and the number of revertant colonies. The “treat-and-wash” method is considered an acceptable method to account for any “false positives” that may otherwise be observed using standard methods such as pre-incubation.28,29
The bacterial reverse mutation test was performed with four Salmonella typhimurium strains (TA100, TA1535, TA98, and TA1537) provided by Dr. Bruce N. Ames (University of California) and one Escherichia coli strain (WP2uvrA) provided by National Institute of Health Sciences of Japan. The microsomal fractions (S9) were purchased from Oriental Yeast Co., who produces from the livers of male Sprague-Dawley rats following induction by phenobarbital and 5,6-benzoflavone. The protein content of the S9 fraction was 19.6–21.3 mg/mL, while the microsome fraction contained a cytochrome P450 content of 2.44–2.54 nmol/mg, cytochrome b5 content of 0.474–0.499 nmol/mg, NADPH-cytochrome c reductase content of 206–232 nmol/mg/min, and aniline hydroxylase content of 103–107 nmol/mg/hour. The negative control used in all tests was distilled water. The positive controls used in the pre-incubation method studies without S9 metabolic activation were as follows: 0.01 μg/plate 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (AF-2) (Lot No. LKF6772, FUJIFILM Wako Pure Chemical) for TA100 and WP2uvrA and 0.1 μg/plate for TA98; 0.5 μg/plate sodium azide (Lot No. YSR7529, FUJIFILM Wako Pure Chemical) for TA1535; and 80 μg/plate 9-aminoacridine hydrochloride (9-AA) (Lot No. 09820CEV, Sigma-Aldrich) for TA1537. In studies conducted with the pre-incubation method with S9 metabolic activation, the positive control was 2-aminoanthracene (2-AA) (Lot No. TLH6618, FUJIFILM Wako Pure Chemical), used at a dose of 0.5 μg/plate for TA98, 1.0 μg/plate for TA100, 2.0 μg/plate for TA1535 and TA1537, and 10 μg/plate for WP2uvrA. The positive controls used in the studies with the “treat-and-wash” method in the absence of S9 metabolic activation were as follows: 0.02 μg/plate AF-2 for TA100 and 0.05 μg/plate for TA98, 0.5 μg/plate 4-nitroquinoline N-oxide (Lot No. N4RDF, Tokyo Chemical Industry) for TA1535, and 5 μg/plate 9-AA for TA1537. In the studies conducted with the “treat-and-wash” method with S9 metabolic activation, the positive control was 2-AA at doses of 0.5 μg/plate for TA98, 1.0 μg/plate for TA100, and 2.0 μg/plate for TA1535.
Preliminary concentration range-finding studies were conducted for both the pre-incubation and the “treat-and-wash” methods to determine appropriate test concentrations and to assess mutagenic potential prior to initiation of the respective main studies. A confirmative study was also conducted using the “treat-and-wash” method, due to conflicting results between the preliminary study and the main study, in order to verify the reproducibility of results (data not shown). Test concentrations were selected based on serial dilution of the stock solution of the enzyme in accordance with OECD Guideline 471.
Based on the findings of the preliminary concentration range-finding studies, the main study using the pre-incubation method was conducted with concentrations of 266, 531, 1060, 2130, 4250, 8500, and 17000 μg TOS/plate. Each concentration was tested in triplicate. The highest test concentration was the maximum dose available from the stock solution (17000 μg TOS/plate). For plate preparation, 100 μL of either the test substance, negative control, or positive control was placed into test tubes. Next, 500 μL of S9 mix was added for tests with metabolic activation, while tests without metabolic activation involved the addition of 500 μL of 0.1 mol/L sodium-phosphate buffer (pH 7.4). The bacterial suspension (100 μL) was added to each test tube, followed by pre-incubation in a shaking water bath (MM-10, Taitec) for 20 min at 37°C at 120 strokes per minute. Following pre-incubation, the contents of each test tube were mixed with 2 mL of top agar, plated, and then incubated for 48 h at 37°C in an incubator (SSV-R11DA, Ikeda Scientific). For the “treat-and-wash” method, purine nucleosidase concentrations of 1060, 2130, 4250, 8500, and 17000 μg TOS/plate were tested in triplicate. Plate preparation followed the same protocol as the pre-incubation method with the following exceptions: pre-incubation was carried out for 90 min and the contents of each test tube underwent a washing step following pre-incubation. In brief, 10 mL of washing solution (2.5% nutrient broth: 0.1mol/L sodium phosphate buffer; 1:7 v/v) was added to each test tube mixture and centrifuged (LC-122, Tomy Seiko) at 3000 r/min for 10 min. The supernatant was removed and the sediment was re-suspended in washing solution (500 μL) then mixed with 2 mL of top agar prior to plating. All studies were performed with and without S9 metabolic activation in strains TA100, TA1535, and TA98. Strain TA1537 was performed only in the absence of S9 metabolic activation.
Plates were observed macroscopically for any changes (including precipitation of the test article) at the start of the treatment and the time of colony counting. Revertant colonies were counted with an automated colony analyzer (CA-11, System Sciences), and the condition of bacterial background growth was monitored under a stereoscopic microscope (40X) for inhibition by the test substance. If the mean number of revertant colonies was observed to be at least two times that of the negative control colonies and the increase in revertant colonies was dose-dependent or reproducible, the result was considered positive. Statistical analysis of the data was not conducted in accordance with OECD Guideline 471.
In vitro mammalian chromosomal aberration test
The in vitro mammalian chromosome aberration test was conducted in accordance with OECD Guideline No. 473 30 and OECD GLP. Blood samples collected from two non-smoking, healthy volunteers were mixed with 0.25 mL of phytohemagglutinin M (Lot No. 1898839, Life Technologies) and 4.4 mL of a prepared culture medium (mixture of inactivated fetal bovine serum, RPMI 1640 liquid medium, and penicillin-streptomycin), and cultured in an incubator (ILL-60, Ikeda Scientific) for 48 h at 37°C. Tubes were inclined and mixed gently by inversion every 10–12 h throughout the incubation period. Following incubation, the tubes were centrifuged (1000 r/min, 5 min) to collect the cultured human lymphocytes. The lymphocytes were treated with either the negative control, positive control, or purine nucleosidase under the following conditions: two short-term assays (consisting of 3-h treatment in either the presence or absence of metabolic activation followed by a 21-h expression period) and one continuous treatment for 24 h in the absence of metabolic activation. The S9 microsomal fractions were obtained as previously described for the bacterial reverse mutation test. The negative control was distilled water. The positive control in the short-term assay in the absence of metabolic activation and in the continuous treatment assay was mitomycin C (Lot No. 581AFI, Kyowa Hakko Kirin), at concentrations of 0.5 and 0.25 μg/mL, respectively. In the short-term assay in the presence of metabolic activation, the positive control was 12.5 μg/mL of cyclophosphamide (Lot No. 4444, Shionogi). All assays were conducted in duplicate.
Preliminary tests were conducted to establish mitotic index and cell growth inhibition in order to determine appropriate concentrations for use. Purine nucleosidase concentrations of 69.6, 174, 435, 1090, 2720, 6800, and 17000 μg TOS/plate were designated for use in the range-finding studies under the same conditions as the short-term (3 h; −S9 and +S9) and continuous treatment (24 h) for the main study assays. Test concentrations were selected based on serial dilution of the stock solution of the enzyme in accordance with OECD Test Guideline 473.
In the short-term assays, purine nucleosidase was incubated with the cultured human lymphocytes at final concentrations of 1700–17000 μg TOS/mL in the absence of metabolic activation and 850–17000 μg TOS/mL in the presence of metabolic activation. After the treatment period, the tubes were centrifuged and the supernatant was removed. The lymphocytes were rinsed with fresh culture medium. Next, 4.75 mL of fresh complete culture medium and 0.25 mL of phytohemagglutinin M (PHA) were added, and the mixtures were incubated for another 21 h at 37°C. In the continuous assay, purine nucleosidase was incubated with the lymphocytes for 24 h at final concentrations of 7.59–121 μg TOS/mL.
A 0.1 μg/mL solution of colcemid (Lot No. 1898682, Life Technologies) was added to each tube 2 h prior to slide preparation in order to inhibit mitosis in the metaphase. After incubation, tubes were centrifuged at 1000 r/min for 5 min to remove culture medium, and the lymphocytes underwent a 15-min hypotonic treatment following the addition of 4 mL of a 75 mmol/L potassium chloride solution at room temperature. Next, 4 mL of a 3:1 mixture of methanol and acetic acid at room temperature was added twice to fix the cells, then 5 mL of the fixative solution was added to the cell-pellet to prepare a cell suspension, followed by overnight refrigeration. Cell density was verified via the preparation of one chromosome slide. Following verification, one drop of each cell suspension was applied to a slide, and two chromosome slides were prepared per plate. Following complete drying of the slides, 1.2% Giemsa staining solution (Lot No. HX69072204, Merck) diluted with 0.01 mol/L sodium phosphate buffer (Buffer tablets pH 6.8, Lot No. TP1305174, Merck) was applied and stained for 12 min. Slides were then rinsed with water and dried.
The highest purine nucleosidase concentration assessed for chromosome aberrations was the highest dose resulting in mitosis inhibition of 50% or more. Microscopic examinations for chromosome aberrations were conducted based on the respective calculated mitotic indices at the following concentrations: 6800, 13600, and 17000 μg TOS/mL in the short-term assays (in both the absence and presence of metabolic activation) and 15.2, 30.4, 60.7, and 121 μg TOS/mL in the continuous treatment assay. At each concentration, 150 metaphase cells per tube were inspected microscopically for chromosome and chromatid gaps, breaks, exchanges, and other aberrations. Three hundred metaphases at each concentration were also observed to determine the number of polyploid cells.
The significant difference in the incidence of aberrant cells between each group was determined using a Fisher’s exact test with a one-sided significance level of 2.5%. Concentration-dependency was not analyzed, as there was an absence of significant differences in the results of the Fisher’s exact test. Results were considered positive if a statistically significant increase in incidence of cells with chromosomal aberrations was observed in one or more of the test concentrations compared to the negative control, there was a significant dose-response trend, and the observed incidence was greater than the acceptable range calculated for the negative control group (using historical data of the control).
90-day repeated-dose toxicity study
The 90-day repeated oral dose toxicity study was conducted in accordance with OECD Guideline No. 408 31 and OECD GLP. This study was conducted in compliance with the Act on Welfare and Management and Standards Relating to the care and Management of Laboratory Animals and Relief of Pain. This study was reviewed and approved before initiation by the Institutional Animal Care and Use Committee.
Preparation of dosing formulations
The purine nucleosidase dosing formulations were prepared in accordance with OECD Guideline No. 408 by 3-fold serial dilution of the thawed stock solution (170 mg TOS/mL) with distilled water to achieve concentrations of 18.9 and 56.7 mg TOS/mL, equivalent to doses of 189 (lose-dose group) and 567 mg TOS/kg body weight (bw)/day (mid-dose group), respectively. The concentration for the high-dose group was the undiluted stock solution, equivalent to 1700 mg TOS/kg bw/day. After being divided for daily use, the prepared solutions were stored in a refrigerator (4.0–8.2°C) until use. Stability testing confirmed the dose formulations to be stable in airtight, light-resistant containers for 4 days under refrigerated conditions at concentrations of 0.340–170 mg TOS/mL.
Animals and treatment
Forty-five male and forty-five female 4-week-old Crl:CD(SD) [SPF] rats were obtained from Charles River Laboratories Japan, Inc. Upon arrival and during the 8-day acclimation period, animals were monitored daily for general health condition and body weights were measured on the day of arrival (Day −8) and the last day of the quarantine and acclimation period (Day −1). No abnormalities in clinical signs or body weights were observed during the acclimation period. Ophthalmological examinations were also conducted on Days −3 and −2 during this period. Two females were excluded from the study prior to group assignment due to evidence of congenital defects. On the first day of dosing (Day 1), a computer system package for safety studies (LATOX-F/V5, FFC) was used to randomly assign animals into groups (10/sex/group) based on body weights. The body weight of the study animals ranged from 142.8 to 161.5 g and 119.7 to 140.6 g for males and females, respectively.
The doses of purine nucleosidase were selected based on the results of a 2-week repeated dose-finding study in rats. In the 2-week study, no treatment-related toxicological effects on general condition, body weight change, food consumption, or necropsy findings were reported in animals administered purine nucleosidase at doses of 106, 425, or 1700 mg TOS/kg bw/day (data not shown). As a result, for the 90-day study, the maximum dose of 1700 mg TOS/kg bw/day was selected for the high-dose level, and 567 mg TOS/kg bw/day and 189 mg TOS/kg bw/day were selected for the mid- and low-dose levels, respectively, based on 3-fold dilution ratios. The test article and the vehicle control were administered via gavage using a plastic syringe and Teflon gastric tube. The control animals received 10 mL/kg bw/day distilled water (vehicle solvent), which was an equivalent dosing volume to the treatment groups. The dosing solutions were prepared once or more per 7 days at concentrations of 18.9, 56.7, and 170 mg TOS/mL for the low-dose (189 mg TOS/kg bw/day), mid-dose (567 mg TOS/kg bw/day), and high-dose (1700 mg TOS/kg bw/day) groups, respectively.
Clinical observations, body weights, food consumption, and ophthalmology
The general condition of each animal was monitored and recorded twice per day (before and after dosing) and once before necropsy (Day 91). Beginning prior to dosing and continuing weekly thereafter, detailed observations were performed for home cage observation, responses on removal from cage, and behavior in open field. In addition, sensorimotor function, grip strength, and locomotor activity were assessed as part of functional observation battery (FOB) tests in the final week of the study period. Body weight of the animals was measured every 7 days from the initiation of the study (Day 1) until Day 90, and on the day of necropsy (Day 91). Total body weight gain was also calculated from Day 1 to 90. Mean daily food consumption (g/day) was calculated for each animal based on the difference between the amount of food provided and amount of food remaining on each day, measured following the same schedule as body weight. Ophthalmological examinations were performed on all animals before the initiation of the dosing period (Day 3 and 2) and in the final week of the dosing period (Day 86 and 90). General appearance of the eye and light reflex were observed under normal conditions, followed by dilation of the pupil using a mydriatic (Lot No. M514151, Mydrin P®, Santen Pharmaceutical) to examine the anterior part of the eyeballs, optic media, and fundus oculi.
Clinical pathology and urinalysis
Hematological and blood chemical examinations were conducted in all surviving animals on the day of the scheduled necropsy (Day 91), while urinalysis and microscopic observation of the estrous cycle were performed during the final week. Blood samples were collected from the abdominal aorta of each animal in tubes containing an anticoagulant (ethylenediaminetetraacetic acid dipotassium salt). Each animal was fasted overnight and was subject to anesthesia with isoflurane during sampling. Blood samples were analyzed using a Hematology System (ADVIA120, Bayer) for the following hematological parameters: hematocrit, hemoglobin, red blood cell count, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, reticulocyte ratio, reticulocyte count, platelet count, white blood cell count, differential leukocyte ratios, leukocyte count (neutrophil, lymphocyte, monocyte, eosinophil, basophil), and large unstained cell count.
Plasma samples were prepared from the blood samples and collected into tubes containing an anticoagulant (3.2% sodium citrate solution). Samples were prepared by centrifugation at 1700 x g for 13 min at room temperature and analyzed for prothrombin time and activated partial thromboplastin time with a coagulation analyzer (STA Compact, Roche).
Serum samples were also prepared from the blood samples via centrifugation at 1700 x g for 7 min at room temperature following collection into tubes containing a Gel and Clot activator (Venoject II, Terumo) for blood chemistry parameters. Samples were analyzed for the following parameters using an automatic analyzer (Hitachi 7170, Hitachi): total protein, glucose, triglyceride, total cholesterol, blood urea nitrogen (BUN), creatinine, total bilirubin, aspartate aminotransferase, alanine aminotransferase, γ-glutamyl transpeptidase, calcium, and inorganic phosphorus. Serum concentrations of sodium, potassium, and chloride were analyzed using an electrolyte analyzer (EA07, A&T). An electrophoresis analyzer (Epalyzer 2 plus, Helena Laboratories) was used to analyze the following parameters: albumin ratio, α1-globulin ratio, α2-globulin ratio, β-globulin ratio, γ-globulin ratio, albumin/globulin ratio, albumin concentration, α1-globulin concentration, α2-globulin concentration, β-globulin concentration, and γ-globulin concentration.
For urinalysis, samples of urine were collected within 3 h after urination (fresh urine) and 24 h after urination (pooled urine). Ames test strips (N-Multistix, SG-L, Siemens Healthcare Diagnostics) and an automatic strip reader (CLINITEK Advantus, Siemens Healthcare Diagnostics) were used to analyze the following parameters in fresh urine: pH, occult blood, ketone bodies, glucose, protein, bilirubin, and urobilinogen. The 24-h urine samples were examined for volume and color and then centrifuged for 5 min at 400 x g. An electrolyte analyzer (EA07) was used to analyze the supernatant for electrolyte concentrations (sodium, potassium, and chloride), and osmotic pressure was evaluated using an osmotic pressure analyzer (AUTO&STAT™ OM-6030, Arkray Factory). The total excretion value of each electrolyte was determined from the urinary volume. The Steinheimer method was used to stain the urinary sediments in preparation for microscopic examination. The urine sediments were examined for erythrocytes, leukocytes, squamous cells, transitional epithelial cells, renal tubular epithelial cells, casts, fat globules, mucous threads, and crystals.
Microscopic observations of the estrous cycle in female rats were conducted on vaginal smear preparations obtained from the animals prior to daily dosing from Day 85 until necropsy (Day 91).
Pathology
Following blood sample collection, all animals were euthanized via exsanguination under isoflurane anesthesia and subject to pathological examination (organ weights, macroscopic and histopathology). The following organs of all animals in all groups were weighed: brain, heart, liver, kidneys, spleen, testes, adrenal glands, ovaries, thymus, uterus, and epididymides. The final body weights recorded on Day 91 and absolute organ weights were used to calculate relative organ weights. Histopathological examination was conducted organs and tissues from all animals consistent with those specified in the OECD Guideline No. 408. All organs and tissues were fixed in an adequate volume of 10% neutral buffered formalin solution. The testes and eyes (including optic nerve and Harderian glands) were pre-fixed in formalin-acetic acid solution and Davidson’s solution, respectively, followed by fixation of both organs in 10% neutral buffered formalin solution. The left and right lungs were fixed by dropping infusion of fixative solution. All fixed organ and tissue samples were embedded in paraffin, sectioned, and stained with hematoxylin and eosin for histopathological examination. Specimens of the control and high-dose groups were examined microscopically, and all histopathological findings and their severity were recorded.
Statistical analysis
The following parameters were analyzed using Bartlett’s test for homogeneity of variances: body weight, body weight gain, food consumption, metric FOB data (grip strength and locomotor activity), hematology, blood chemistry, urinalysis (volume, osmotic pressure, electrolytes), and absolute and relative organ weights. To evaluate the statistical significance of the differences between the control group and each test substance-treated group, Dunnett’s multiple comparison test was used to analyze homogenous data (not significantly different) and Steel’s test was used to analyze heterogenous data (significantly different). The significance of Bartlett’s test was analyzed at the 5% level of significance and the other tests were analyzed at the 5% and 1% level of significance using a two-sided analysis.
Results
Genotoxicity studies
Bacterial reverse mutation test (Ames test)
Results of bacterial reverse mutation test (concentration-finding study using pre-incubation method).
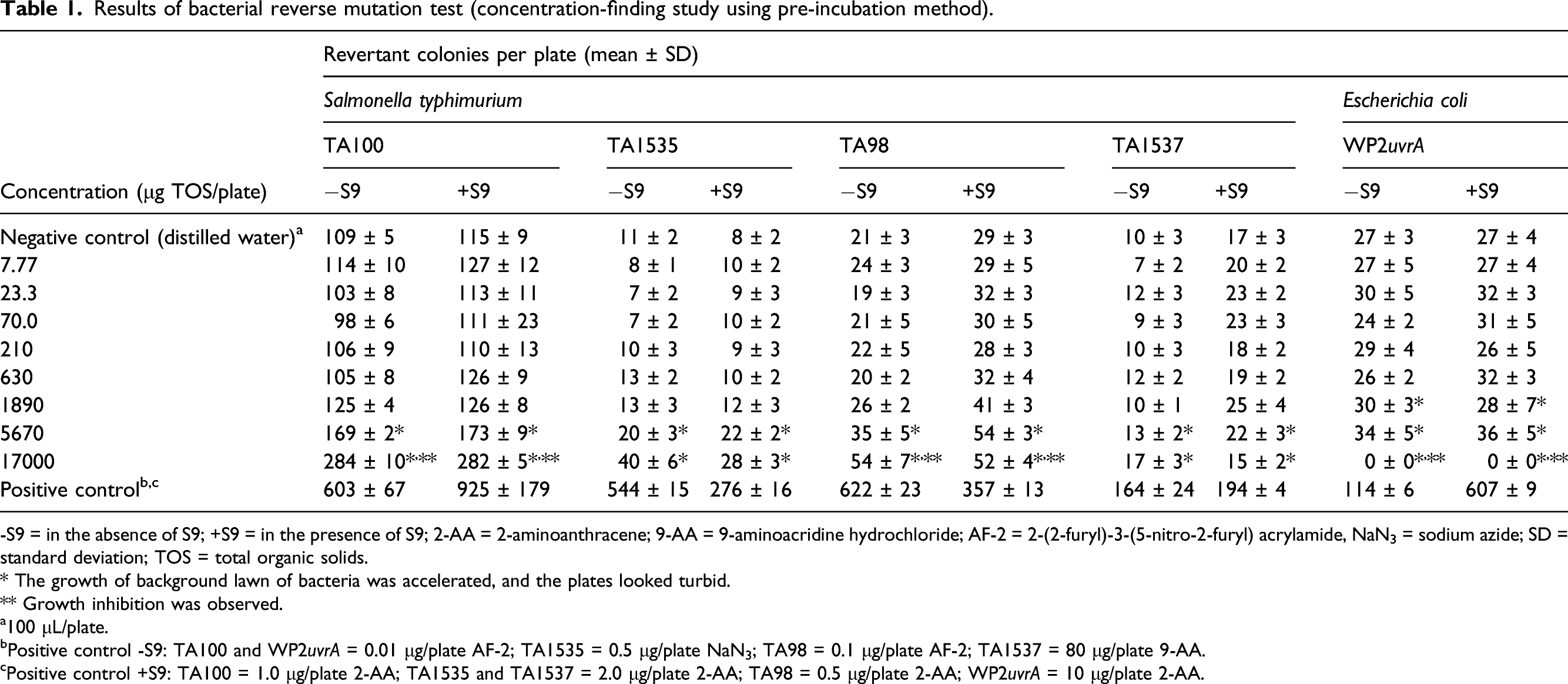
-S9 = in the absence of S9; +S9 = in the presence of S9; 2-AA = 2-aminoanthracene; 9-AA = 9-aminoacridine hydrochloride; AF-2 = 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide, NaN3 = sodium azide; SD = standard deviation; TOS = total organic solids.
* The growth of background lawn of bacteria was accelerated, and the plates looked turbid.
** Growth inhibition was observed.
a100 μL/plate.
bPositive control -S9: TA100 and WP2uvrA = 0.01 μg/plate AF-2; TA1535 = 0.5 μg/plate NaN3; TA98 = 0.1 μg/plate AF-2; TA1537 = 80 μg/plate 9-AA.
cPositive control +S9: TA100 = 1.0 μg/plate 2-AA; TA1535 and TA1537 = 2.0 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA; WP2uvrA = 10 μg/plate 2-AA.
Results of bacterial reverse mutation test (main study using pre-incubation method).
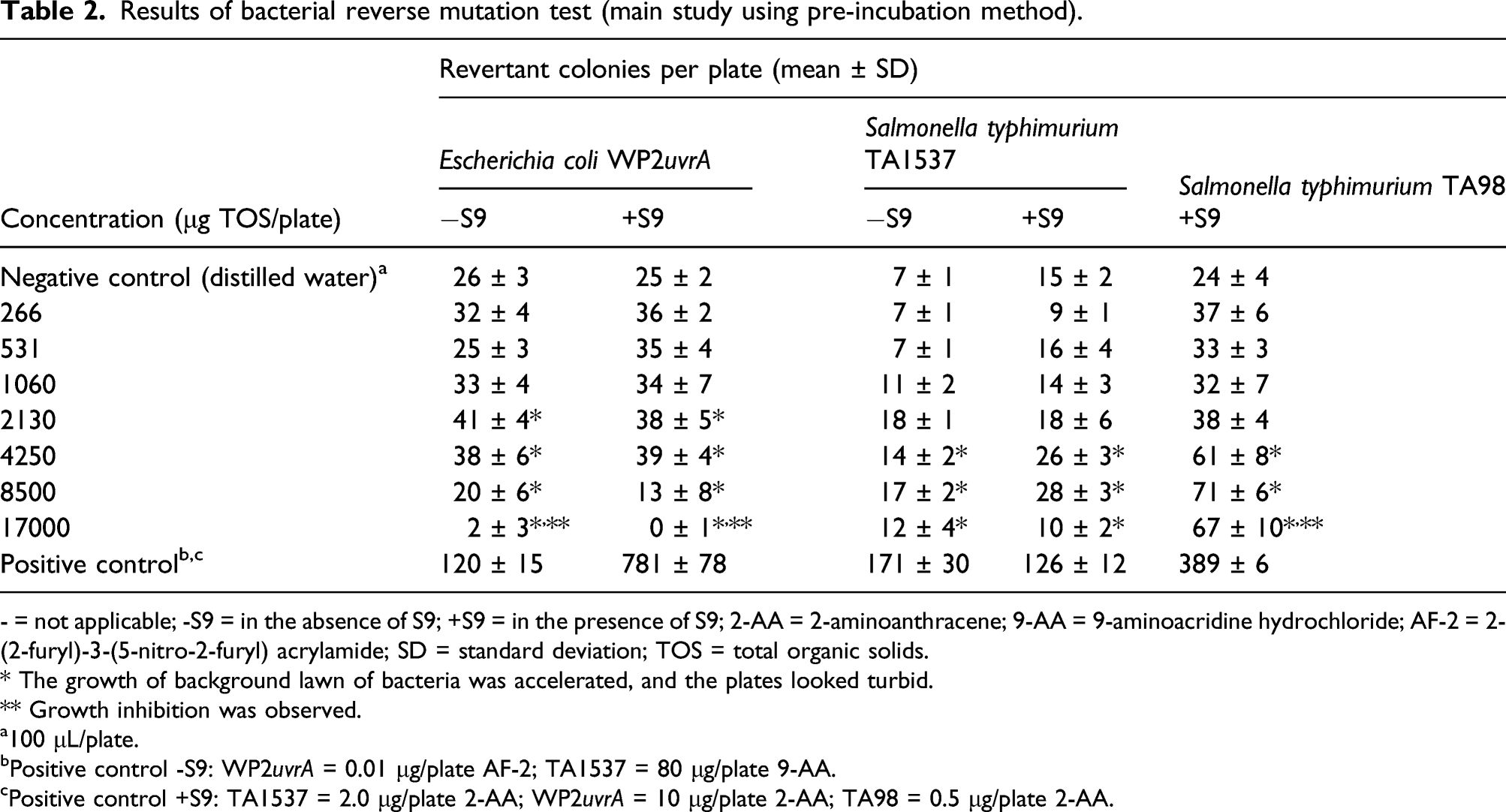
- = not applicable; -S9 = in the absence of S9; +S9 = in the presence of S9; 2-AA = 2-aminoanthracene; 9-AA = 9-aminoacridine hydrochloride; AF-2 = 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; SD = standard deviation; TOS = total organic solids.
* The growth of background lawn of bacteria was accelerated, and the plates looked turbid.
** Growth inhibition was observed.
a100 μL/plate.
bPositive control -S9: WP2uvrA = 0.01 μg/plate AF-2; TA1537 = 80 μg/plate 9-AA.
cPositive control +S9: TA1537 = 2.0 μg/plate 2-AA; WP2uvrA = 10 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA.
Results of bacterial reverse mutation test (concentration-finding study using the “treat-and-wash” method).
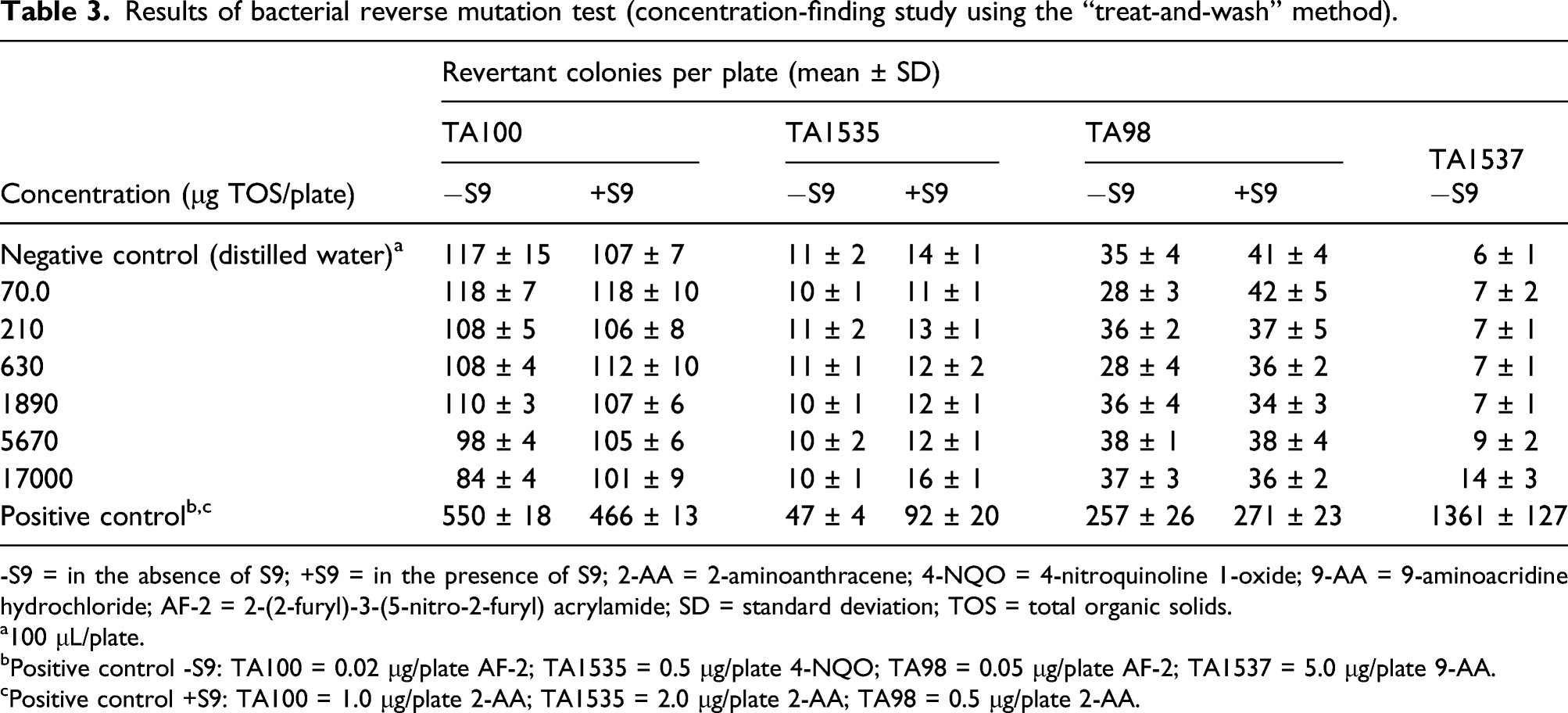
-S9 = in the absence of S9; +S9 = in the presence of S9; 2-AA = 2-aminoanthracene; 4-NQO = 4-nitroquinoline 1-oxide; 9-AA = 9-aminoacridine hydrochloride; AF-2 = 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; SD = standard deviation; TOS = total organic solids.
a100 μL/plate.
bPositive control -S9: TA100 = 0.02 μg/plate AF-2; TA1535 = 0.5 μg/plate 4-NQO; TA98 = 0.05 μg/plate AF-2; TA1537 = 5.0 μg/plate 9-AA.
cPositive control +S9: TA100 = 1.0 μg/plate 2-AA; TA1535 = 2.0 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA.
Results of bacterial reverse mutation test (main using the “treat-and-wash” method).
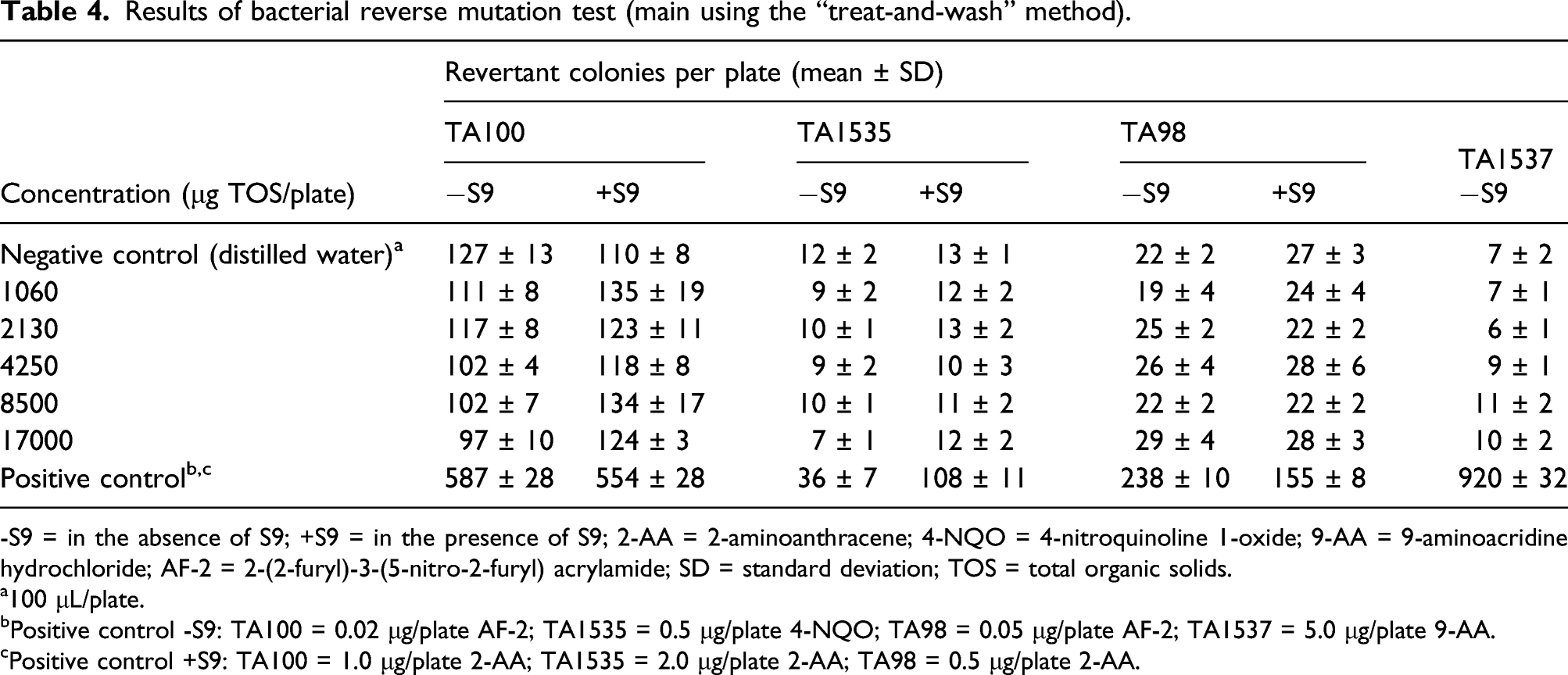
-S9 = in the absence of S9; +S9 = in the presence of S9; 2-AA = 2-aminoanthracene; 4-NQO = 4-nitroquinoline 1-oxide; 9-AA = 9-aminoacridine hydrochloride; AF-2 = 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; SD = standard deviation; TOS = total organic solids.
a100 μL/plate.
bPositive control -S9: TA100 = 0.02 μg/plate AF-2; TA1535 = 0.5 μg/plate 4-NQO; TA98 = 0.05 μg/plate AF-2; TA1537 = 5.0 μg/plate 9-AA.
cPositive control +S9: TA100 = 1.0 μg/plate 2-AA; TA1535 = 2.0 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA.
In vitro mammalian chromosomal aberration test
Results of in vitro mammalian chromosome aberration test.
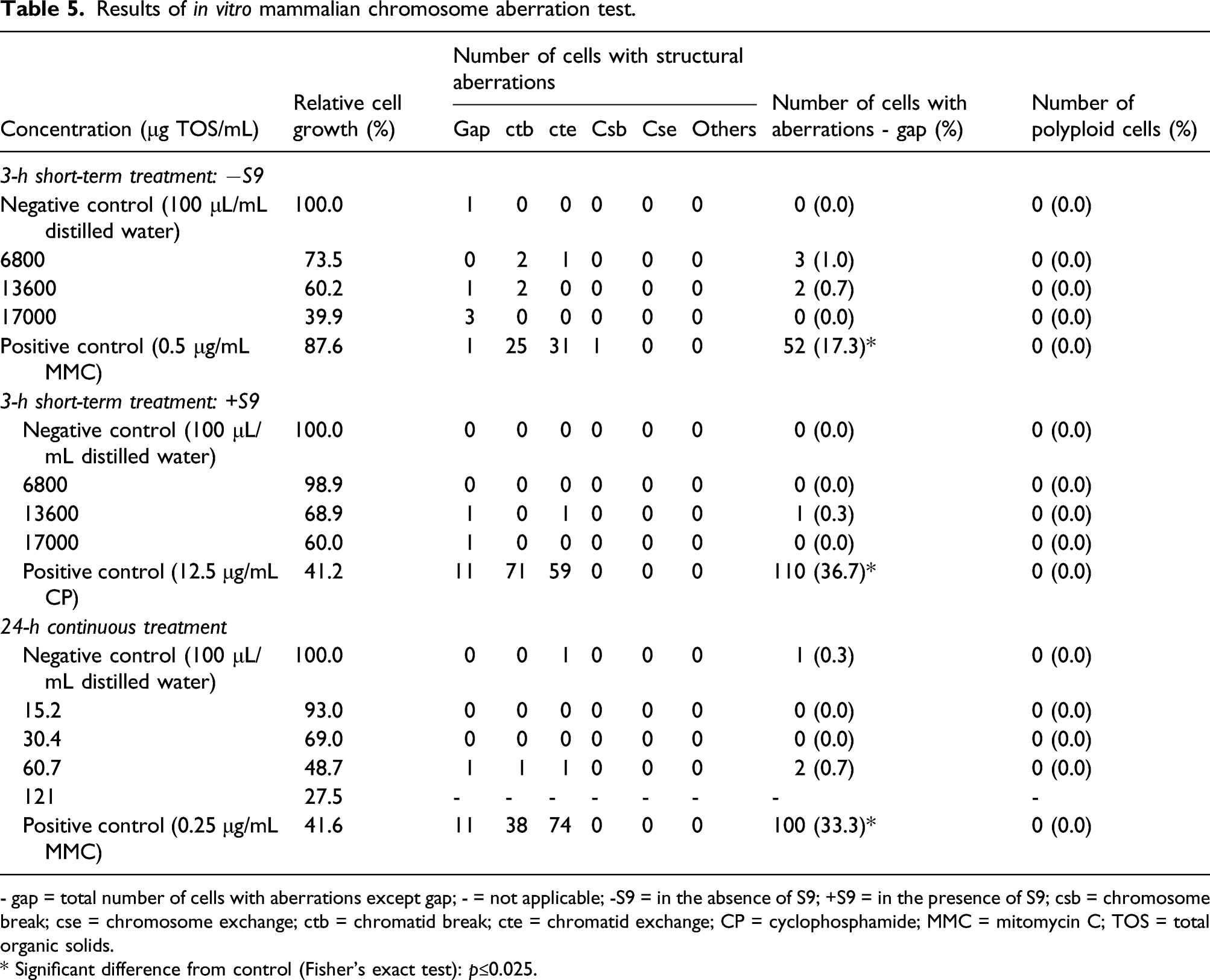
- gap = total number of cells with aberrations except gap; - = not applicable; -S9 = in the absence of S9; +S9 = in the presence of S9; csb = chromosome break; cse = chromosome exchange; ctb = chromatid break; cte = chromatid exchange; CP = cyclophosphamide; MMC = mitomycin C; TOS = total organic solids.
* Significant difference from control (Fisher’s exact test): p≤0.025.
In the 24-h continuous assay, the incidences of cells with chromosomal aberrations in groups treated with purine nucleosidase were 0.0, 0.0, and 0.7% at concentrations of 15.2, 30.4, and 60.7 μg TOS/mL, respectively, and were not significantly different compared to the negative control groups (0.3%) (Table 5). The incidence of chromosomal aberrations was significantly higher in the mitomycin C positive control group (33.3%) compared to the negative control group. The incidence of polyploid cells was 0.0% in all purine nucleosidase groups and negative control groups. The relative mitotic index was 48.7% at the highest examined concentration of 60.7 μg TOS/mL. No precipitation of purine nucleosidase was observed during the study period.
90-day repeated-dose toxicity study
Clinical observations, body weights, food consumption, and ophthalmology
Animals in all groups did not show any changes in general condition throughout the dosing period. On Day 89, one male in the mid-dose group bit off a piece of the gastric tube affixed to the syringe and ingested it. Due to a decrease in its body weight and food consumption on Day 85, the animal was terminated on Day 86. Throughout the 90-day period, no significant differences were observed in any of the groups for reactivity tests, grip strength, frequencies of defecation and urination, locomotor activity, detailed observations, or ophthalmology.
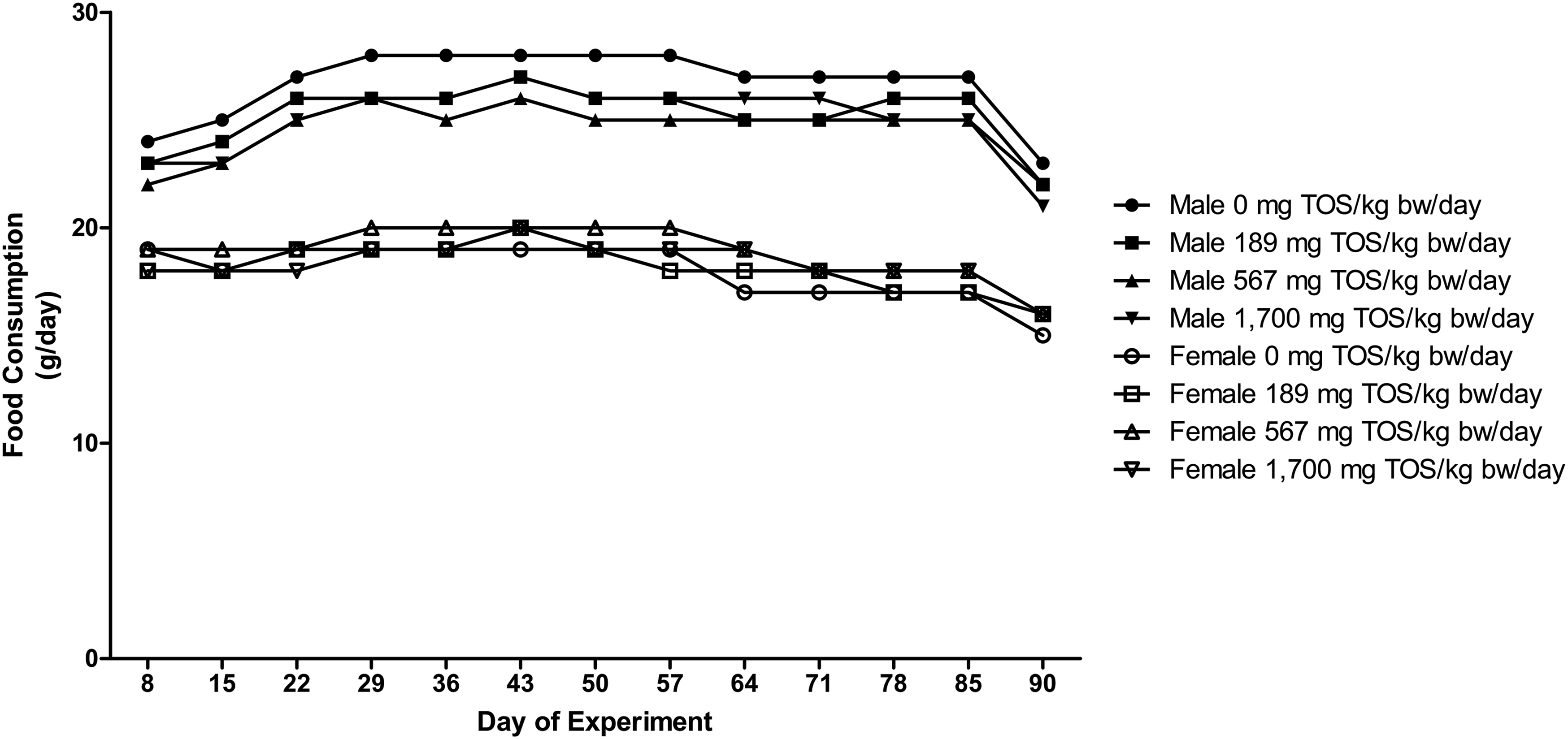
No statistically significant differences were observed in the body weight and body weight gain (Figure 1) or food consumption (Figure 2) between the control and purine nucleosidase test groups during the dosing period. None of the selected animals showed abnormalities in estrous cycles in any of the test groups or control group from the observation period of Day 85–91. Body weight of male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks. Food consumption of male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks.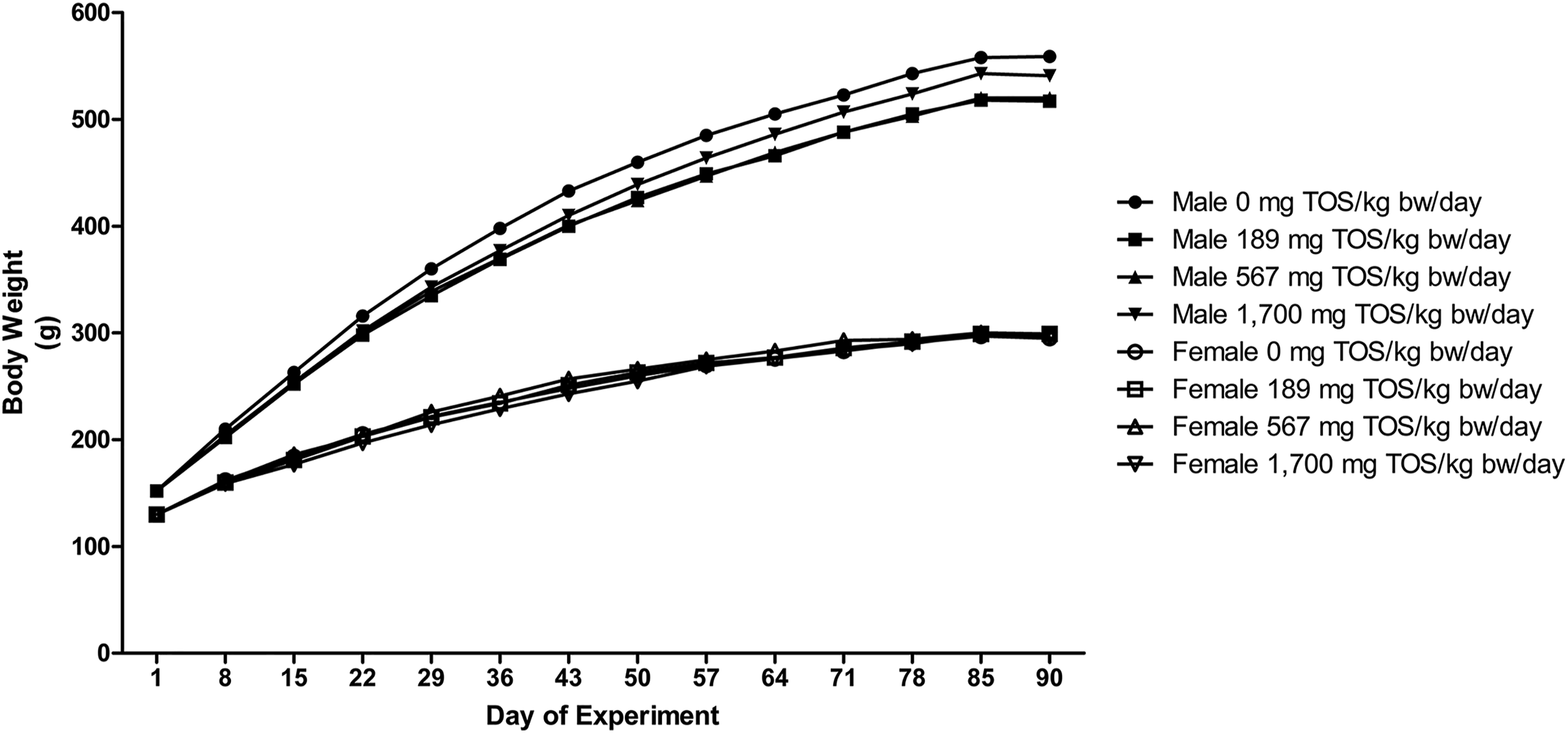
Clinical pathology and urinalysis
Hematology values for male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks.
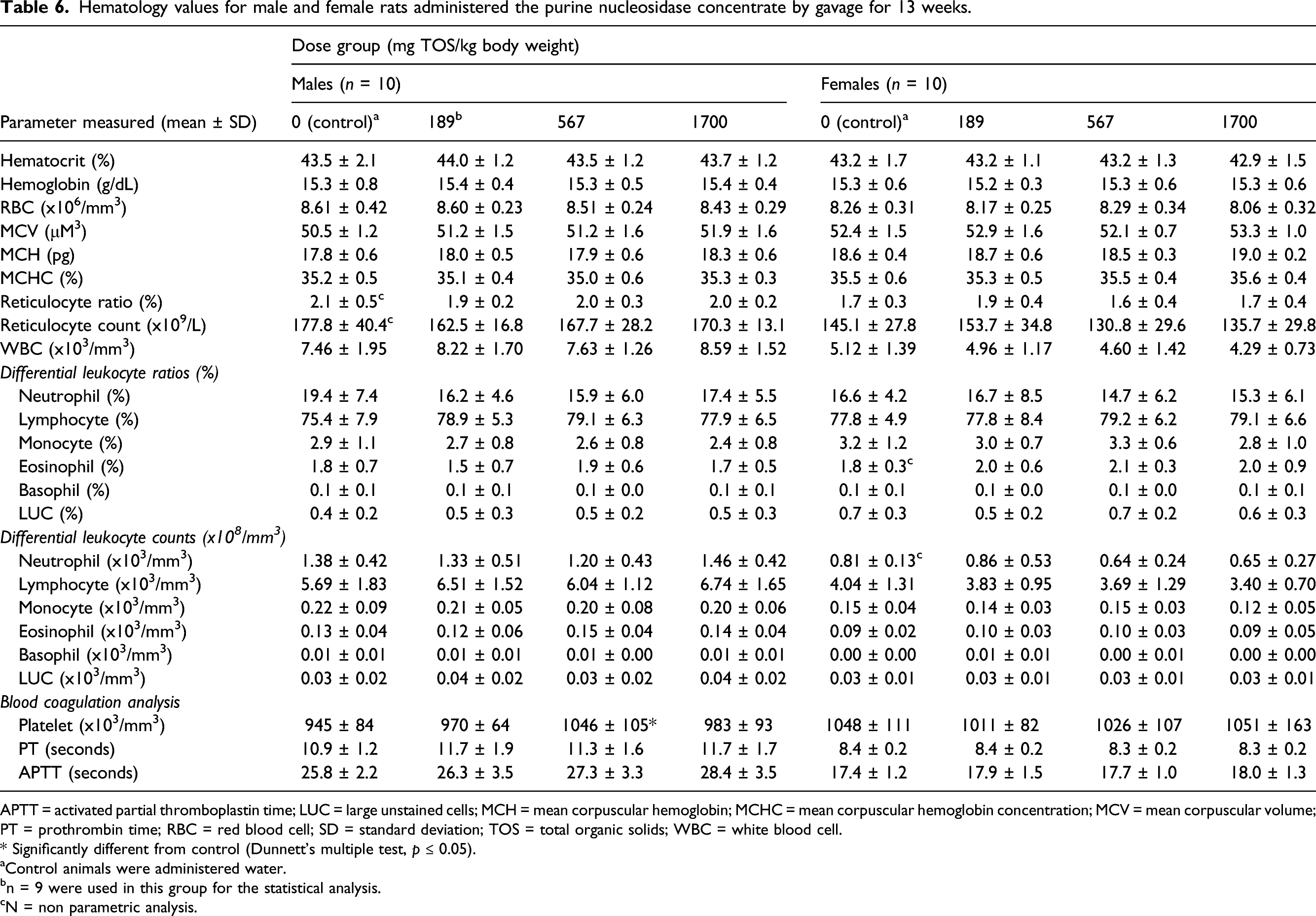
APTT = activated partial thromboplastin time; LUC = large unstained cells; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; PT = prothrombin time; RBC = red blood cell; SD = standard deviation; TOS = total organic solids; WBC = white blood cell.
* Significantly different from control (Dunnett’s multiple test, p ≤ 0.05).
aControl animals were administered water.
bn = 9 were used in this group for the statistical analysis.
cN = non parametric analysis.
Selected blood chemistry and urinalysis values for male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks.
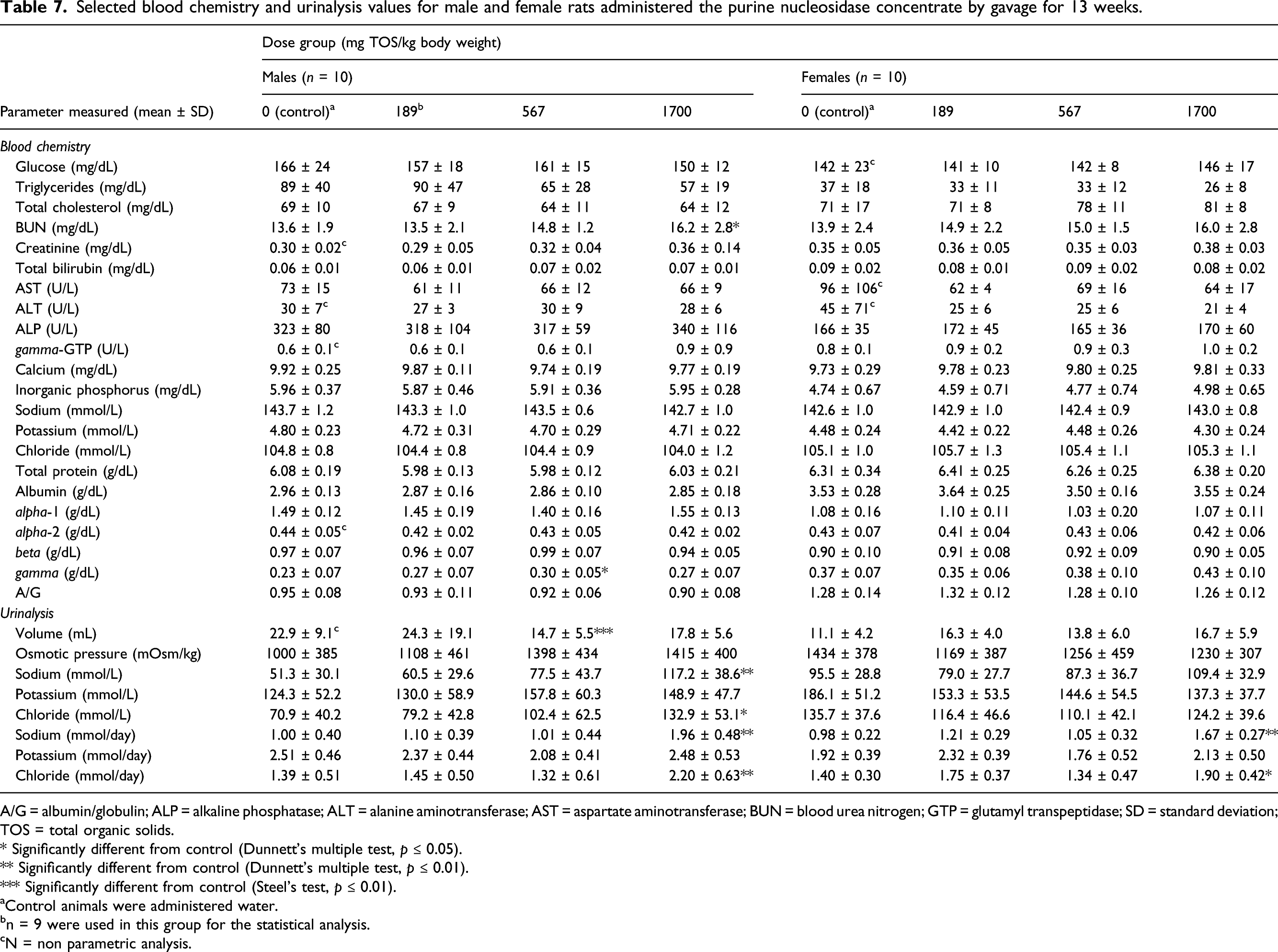
A/G = albumin/globulin; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; GTP = glutamyl transpeptidase; SD = standard deviation; TOS = total organic solids.
* Significantly different from control (Dunnett’s multiple test, p ≤ 0.05).
** Significantly different from control (Dunnett’s multiple test, p ≤ 0.01).
*** Significantly different from control (Steel’s test, p ≤ 0.01).
aControl animals were administered water.
bn = 9 were used in this group for the statistical analysis.
cN = non parametric analysis.
Summary of select urinalysis parameters for male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks.
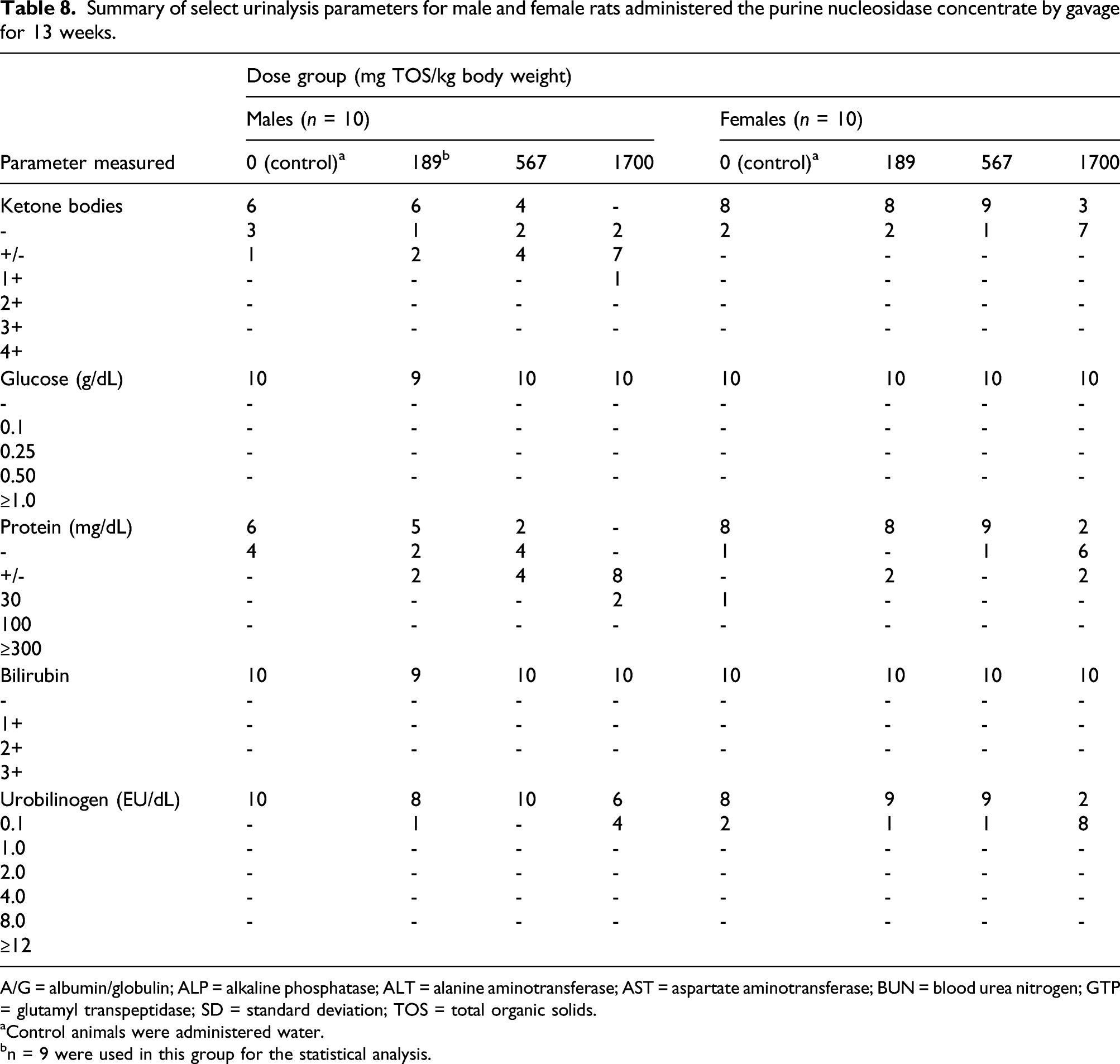
A/G = albumin/globulin; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; GTP = glutamyl transpeptidase; SD = standard deviation; TOS = total organic solids.
aControl animals were administered water.
bn = 9 were used in this group for the statistical analysis.
Pathology
Absolute and relative organ weights of male and female rats administered the purine nucleosidase concentrate by gavage for 13 weeks.
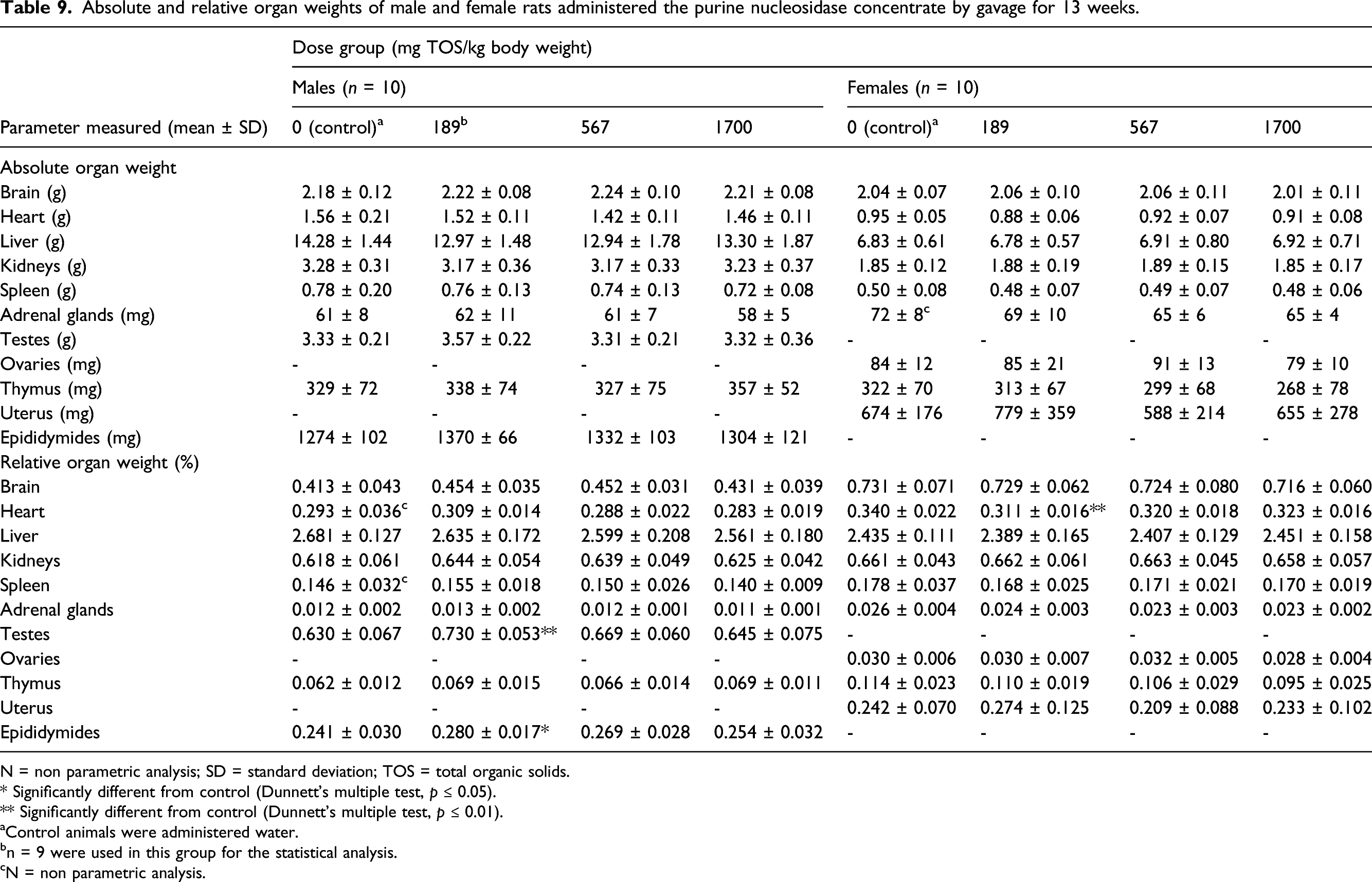
N = non parametric analysis; SD = standard deviation; TOS = total organic solids.
* Significantly different from control (Dunnett’s multiple test, p ≤ 0.05).
** Significantly different from control (Dunnett’s multiple test, p ≤ 0.01).
aControl animals were administered water.
bn = 9 were used in this group for the statistical analysis.
cN = non parametric analysis.
Necropsy of the male from the low-dose group terminated on Day 86 due to ingestion of the stomach tube revealed focal depressions in the kidney upon macroscopic examination. The swallowed segment of the tube was found in the esophagus. Upon macroscopic examination of animals from the control and high-dose groups, the following spontaneous lesions were identified: brown patches on the surface of lungs; white patches on the surface of the stomach; cysts and focal depression in the kidneys; brown discoloration in the testis; and dilated lumen in the uterus. None of the observed lesions were deemed to be treatment-related at the scheduled necropsy as the incidence of the findings were similar between the control and high-dose animals (between 1 and 2 animals per group), minor in severity, and were not associated with any changes in the clinical chemistry, hematology, or urinalysis of the respective animals.
Upon histopathological examination, the following lesions were observed in the control group and high-dose group at varying incidence rates: cardiomyopathy; hemorrhage; macrophage aggregation in the lungs; inflammatory changes in the lungs, pancreas, kidneys, and prostate; microgranuloma in the liver; cysts, mineralization, tubular regeneration in the kidneys; atrophy of seminiferous tubules in the testes; decrease in the number of spermatozoa in the epididymides; dilated lumen in the uterus; and cysts in the pituitary gland.
Discussion and conclusion
A purine nucleosidase has been obtained from a non-genetically modified strain of A. luchuensis isolated from a food source and evaluated for safety for use in food manufacture. The safety of this potentially useful industrial food enzyme was investigated in a series of genotoxicity and mutagenicity bioassays, including a bacterial reverse mutation test, mammalian chromosomal aberration test, and a 90-day repeated-dose oral toxicity study in rats.
The enzyme induced more than 2-fold increases in revertant colonies at the higher concentrations tested in each of the concentration range-finding and main assays in the Ames test using the pre-incubation method. These responses were noted in the presence of accelerated growth of the bacterial lawn. In contrast, no evidence of mutagenicity or accelerated bacterial growth was observed using the “treat-and-wash” modification at the same enzyme concentrations of up to 17000 μg/plate. The “treat-and-wash” method removes peptides and amino acids (e.g., histidine) that are known to produce false positives. 29 On this basis, the observed increase in revertant colonies using the pre-incubation method was attributed to the presence of peptides or free amino acids; thus, the purine nucleosidase was considered to be non-mutagenic in the bacterial reverse mutation assay. The “treat-and-wash” method was employed in bacterial reverse mutation tests on other enzymes intended for food production which support the conclusion of a non-mutagenicity of purine nucleosidase under the conditions of this assay.32–34 The lack of genotoxic potential of purine nucleosidase from A. luchuensis was further confirmed in the in vitro mammalian chromosome aberration assay in which no evidence of clastogenic effects were observed up to the highest concentrations tested.
The results of the 90-day repeated-dose oral toxicity study in rats were unremarkable. There were no treatment-related adverse effects on clinical signs, FOB observations, body weights, food consumption, estrous cycle, or on the results of ophthalmological, hematological, blood chemistry, organ weight, and gross and histopathological analyses.
The few statistically significant findings in the hematology, clinical chemistry, and urinalysis findings were considered to be spontaneous and unrelated to purine nucleosidase treatment given the low magnitude of the changes, lack of dose-responses and lack of replication in both sexes. In the blood chemistry examination, BUN concentration of males in the 1700 mg TOS/kg bw/day group was statistically increased relative to controls; however, this change was judged as a non-adverse given the absence of changes in the histopathology and macroscopic examination, with the incidence rates similar between the control and high-dose animals, as well as a lack of such finding in females. It is noted that the increase in BUN concentration (approximately 16 mg/dL) in both sexes were within the reported historical control range of 10–17 mg/dL for Sprague-Dawley rats. 35 Furthermore, the increase in BUN concentration could be attributable to a slightly increased protein intake in the diet, due to oral administration of the enzyme, a proteinaceous substance. This effect is often observed in animals with increased protein intake in the diet. 36 Thus, such changes are not considered relevant to low level exposure to protein associated with human intakes of the enzyme. Considering the foregoing, the reported changes in BUN are not considered to be toxicologically relevant.
The concentrations of sodium, potassium, and chloride tended to increase in the urine of male animals in dose-related manner; however, this was associated with a corresponding trend to decreasing urine volumes and increased urine density in these animals. In the urinalysis, increased total excretion values of sodium and chloride were observed in both sexes in the 1700 mg TOS/kg bw/day group. This change was not considered adverse since the increased excretion was related to the sodium chloride content (1.3%) of the enzyme preparation. Similar findings were not observed in the low-dose or mid-dose groups. There were no effects on electrolytes in the hematological analyses. The urinalysis results also revealed that the numbers of ketone body- and protein-positive males, and urobilinogen-positive animals tended to increase in the high-dose group (1700 mg TOS/kg bw/day) of both sexes. However, no findings indicative of abnormal glycol-metabolism, inadequate food intake or other changes related to ketosis were identified. Moreover, there were no reported histopathological lesions that are indicative of renal disorders. Kidney cysts and focal depressions were observed in the control and high-dose animals, however; the incidence rates (1–2 animals in the control and high-dose groups) were within the historical control range for this strain of rat, suggesting that these findings are typical for this rat strain. 37 Therefore, the changes in the urinary parameters were not considered to have been adversely affected by treatment.
The results of the 90-day toxicity study indicate that purine nucleosidase produced from A. luchuensis had no adverse effect up to the highest dose tested of 1700 mg TOS/kg bw/day. The no-observed adverse effect level (NOAEL) was, therefore, established at 1700 mg TOS/kg bw/day. The findings of this study are consistent with the results of a 90-day study conducted with xylanase, another commercially important food enzyme, obtained from A. luchuensis Inui in which a NOAEL of 1,000 mg TOS/kg bw/day was reported based on lack of adverse effects following oral administration of the enzyme for 90 23 -days to Wistar rats. These findings suggest that enzymes obtained from this species (A. luchuensis) are safe for use in food production.
In considering the safety of the production microorganisms, A. luchuensis is very closely related to the industrially important A. niger, 15 a species that has been considered to raise no safety concerns over its decade-long use by the food industry.38,39 A. luchuensis is a member of the Nigri Section. 16 This Aspergillus species also does not appear to produce mycotoxins. 16 The A. luchuensis PZ-1 strain has been screened for the production of common toxic secondary metabolites and mycotoxins, including aflatoxin B1, B2, G1, and G2, sterigmatocystin, fumonisin B1and B2, zearalenone, ochratoxin A, and T-2 toxin. The production organism was found to be free of these toxins, consistent with other safety conclusions from the European Food Safety Authority on enzymes from A. luchuensis for use in food processing. 23
In conclusion, the results of the toxicological studies presented above support the safety of purine nucleosidase for use in food processing. In addition, the data available on the production enzyme indicate it to be of no safety concern.
