Abstract
Cigarette smoking causes serious diseases, including lung cancer, atherosclerotic coronary artery disease, peripheral vascular disease, chronic bronchitis, and emphysema. While cessation remains the most effective approach to minimize smoking-related disease, alternative non-combustible tobacco-derived nicotine-containing products may reduce disease risks among those unable or unwilling to quit. E-vapor aerosols typically contain significantly lower levels of smoke-related harmful and potentially harmful constituents; however, health risks of long-term inhalation exposures are unknown. We designed a 7-month inhalation study in C57BL/6 mice to evaluate long-term respiratory toxicity of e-vapor aerosols compared to cigarette smoke and to assess the impact of smoking cessation (Cessation group) or switching to an e-vapor product (Switching group) after 3 months of exposure to 3R4F cigarette smoke (CS). There were no significant changes in in-life observations (body weights, clinical signs) in e-vapor groups compared to the Sham Control. The 3R4F CS group showed reduced respiratory function during exposure and had lower body weight and showed transient signs of distress post-exposure. Following 7 months of exposure, e-vapor aerosols resulted in no or minimal increase in pulmonary inflammation, while exposure to 3R4F CS led to impairment of lung function and caused marked lung inflammation and emphysematous changes. Biological changes observed in the Switching group were similar to the Cessation group. 3R4F CS exposure dysregulated the lung and nasal tissue transcriptome, while these molecular effects were substantially lower in the e-vapor group. Results from this study demonstrate that in comparison with 3R4F CS, e-vapor aerosols induce substantially lower biological responses including pulmonary inflammation and emphysematous changes, and that complete switching from CS to e-vapor products significantly reduces biological changes associated with CS in C57BL/6 mice.
Keywords
Introduction
Cigarette smoking (CS) causes serious diseases associated with chronic inflammation in the lung including pulmonary carcinoma, bronchitis, and chronic obstructive pulmonary disease (COPD). 1,2 CS also causes or exacerbates various diseases beyond the lung such as atherosclerotic coronary artery disease, peripheral vascular disease, rheumatoid arthritis and Crohn’s disease, associated with chronic inflammatory processes. 3,4 Globally, COPD is one of the leading causes of mortality and morbidity next to ischemic heart disease and cerebrovascular disease. 5 CS is considered the predominant cause of COPD in industrialized countries. 5 –10 CS induces pulmonary inflammation, airway obstruction and remodeling, and emphysematous changes which are typical characteristics of COPD. 1,6,8,11,12
While cessation remains the most effective approach to minimize smoking-related diseases, 13 alternative non-combustible tobacco-derived nicotine-containing products may offer reduced disease risk to those who otherwise continue to smoke. For example, e-vapor products are considered a potentially reduced risk product (RRP) which deliver nicotine without many of the harmful combustion products. 14 –16 Though toxicological investigations of e-vapor aerosols are relatively recent, evidence from the literature indicates that e-vapor products likely offer significantly lower disease risk potential than combustible tobacco products. 17 –19 Moreover, early clinical evidence indicates that harm reduction could be expected in smokers who completely switch from cigarettes to e-vapor products. 20 –23 This is likely due to the reduction or elimination of smoking-related harmful and potentially harmful constituents 24 when compared to CS. 14
With the reduced risk potential and perception that e-vapor products can mimic sensory aspects of cigarette smoking, use of e-vapor products has dramatically increased in popularity in the last several years. 20,25 –27 E-liquids are typically composed of aerosol formers (carrier), nicotine, and flavor mixtures in varying proportions. Most flavors used in e-liquids are generally recognized as safe (GRAS) for oral consumption; however, their toxicity when inhaled is largely unknown. With respect to the use of e-vapor products as RRPs, two opposite viewpoints are currently being debated. On one hand, some studies have reported potential adverse effects of e-vapor aerosols, for instance showing that e-vapor aerosol exposure affected lipid homeostasis and caused immune impairment in the lungs, independent of nicotine, in a mouse model. 28 In addition, Ghosh et al. 29 reported equally elevated neutrophil elastase matrix metalloproteinase (MMP)-2 and MMP-9 activities in both vapers’ and smokers’ bronchoalveolar lavage (BAL) fluid (BALF) relative to non-smokers, suggesting that e-vapor aerosols are not safer alternatives to smoking. In contrast, large number of studies have demonstrated reduced risk potential of e-vapor products in vitro and in vivo. 30 –33 Also, in vivo inhalation studies with carriers (propylene glycol [PG]/vegetable glycerol [VG]) in the presence and absence of nicotine have shown only limited biological effects as compared to vehicle controls. 34 –36 The reduced risk potential of e-vapor products was also supported by clinical data in smokers who switched to vaping, which showed rapid improvement in vascular endothelial functions 37 and respiratory symptoms. 21
Consequently, these mixed results warrant a thorough and a robust study on e-vapor aerosols in terms of absolute (relative to Sham Control) and relative (relative to CS) toxicity endpoints. Also, the long-term health risk of e-vapor aerosols as well as in the context of switching from combustible CS to e-vapor products, especially in the context of respiratory toxicity, is largely unknown.
In this study, we performed a chronic inhalation (4 hours/day, 5 days/week, up to 7 months) study in C57BL/6 mice using an e-vapor product and a combustible reference cigarette (3R4F) using a Switching and Cessation study design. We used chronic exposure to 3R4F CS in C57BL/6 mice to induce pulmonary inflammation and emphysematous changes associated with COPD 38,39 as a reference to evaluate the reduced risk potential of e-vapor aerosols on systemic and COPD-related endpoints. In addition to standard toxicological endpoints (in-life observations, biomarkers of exposure [BOEs], respiratory function, and histopathological evaluation), underlying molecular changes (transcriptomics of target tissues, and biomarkers of pulmonary inflammation and tissue damage) were evaluated over time. The collective results from this study demonstrate that in comparison with CS, e-vapor aerosols induce substantially lower biological responses, including pulmonary inflammation and emphysematous changes, and complete switching from CS to e-vapor products significantly reduces biological changes associated with CS in C57BL/6 mice.
Materials and methods
Test materials
A commercial e-vapor product (MarkTen® device [version 2.6.8]; “Test Red”) was supplied by Altria Client Services, LLC. The Test Red formulation was composed of aerosol formers (PG and VG), ∼4% nicotine by weight, and flavors (non-menthol) (Supplementary Table S1). 3R4F reference cigarettes (Lexington, KY) were purchased from the University of Kentucky. 40 HEPA-filtered air at the testing facility (Battelle, West Jefferson, OH) was used for Sham Control.
Test systems
General procedures for animal care and housing met the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) recommendations and requirements stated in the “Guide for Care and Use of Laboratory Animals” (National Research Council [NRC]) and approved by the Institutional Animal Care and Use Committee (IACUC). Female C57BL/6 mice were received from Charles River Kingston (Stone Ridge, NY). Female mice were chosen because of higher susceptibility to develop emphysema. 39
During the in-life phase, bacterial culture testing was conducted to evaluate potential infection of Streptococcus agalactiae (Streptococcus species beta-hemolytic group B; GBS): Abnormal lesions observed during unscheduled sacrifice or scheduled necropsy were collected for possible culture. In addition, fecal samples were collected at months 4 and 7 prior to the scheduled sacrifice and subjected to PCR analysis for GBS. The light/dark cycle was maintained at 12 hours of light and dark cycles starting at 6:00 AM each day, and during the study the housing room conditions were maintained at a room temperature of 20.6°C to 23.9°C and a mean relative humidity of 50%. Mice had ad libitum access to water and certified feed (Purina Certified Rodent diet, LabDiet 5002), except when removed from the home cage for daily exposures or sample collection.
Exposure system and characterization
Test atmosphere was generated using CH Technologies (Westwood, NJ, USA) JB-series 2090 and 2080 cigarette smoking machines. 41 Cigarette smoke or e-vapor aerosols were delivered to the mice through a nose-only exposure system at a mean nose-port temperature and relative humidity ranging from 22.1°C to 22.5°C and 52% to 57%, respectively. The nose-only (carousel) exposure units contained five tiers, each with 16 animal exposure ports (a total of up to 80 ports/carousel). The exposure ports were connected to a delivery manifold, which transferred aerosol to the animal breathing zone, and to an outer concentric manifold that drew the exhaled and excess smoke to an exhaust duct. The Sham Control nose-only carousel was supplied with HEPA-filtered building air, and nose-port temperature and humidity were controlled to the same specifications as above. During exposures, mice were restrained in polycarbonate tubes, with an o-ring seal between the nose portion of the tube and the nose port of the exposure carousel, allowing mice to breathe the test atmosphere with minimal whole-body surface contamination. The back portion of the tube was closed with a plastic cap.
The modified Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) Reference Method 81 regimen (55/30/5: a 55 ± 0.3 mL puff volume, a puff every 30 seconds, a 5-second puff duration) was used to generate e-vapor aerosol for 130 puffs/cartridge. Mainstream smoke from 3R4F cigarette was generated using a modified Health Canada Intense regimen (55/30/2: a 55 ± 0.3 mL puff volume, a puff every 30 seconds, a 2-second puff duration, and a near-square puff profile) for eight puffs/cigarette. Nose-port exposure concentrations were checked daily based on the total aerosol collection mass or the total particulate matter (TPM) gravimetrically (on 47 mm Cambridge filters). During exposure, carbon monoxide (CO) concentration was continuously monitored, temperature and relative humidity were recorded at least every 30 minutes, and particle size (mass median aerodynamic diameter [MMAD] and geometric standard deviation [GSD]) were measured monthly. Total concentrations (filters and sorbent tubes) of nicotine, PG, and glycerol for e-vapor aerosols, and nicotine and glycerol for 3R4F CS, were analyzed during months 1, 3, 4, 6, and 7. Aerosol samples were collected using bubblers containing a derivatization solution (which included 2,4-dinitrophenylhydrazine) and analyzed for carbonyls (formaldehyde, acetaldehyde, acrolein, propionaldehyde, and crotonaldehyde) during months 1, 3, 4, 6, and 7 using methods described elsewhere. 31
Study design
Female C57BL/6 mice (∼10 weeks old) were randomly assigned based on body weight to one of five exposure groups: Sham Control, 3R4F CS, Test Red, Switching, and Cessation, as shown in Table 1. Each group included five cohorts for various endpoints: (1) confirmation of exposure (COE) cohort (15/group), (2) BAL cohort (10/group), (3) histopathology (HISTO) cohort (12/group), (4) transcriptomics/proteomics (OMICS) cohort (10/group) and (5) lung function cohort (12/group) (Details of group allocations are included in Supplementary Table S2).
Study design.
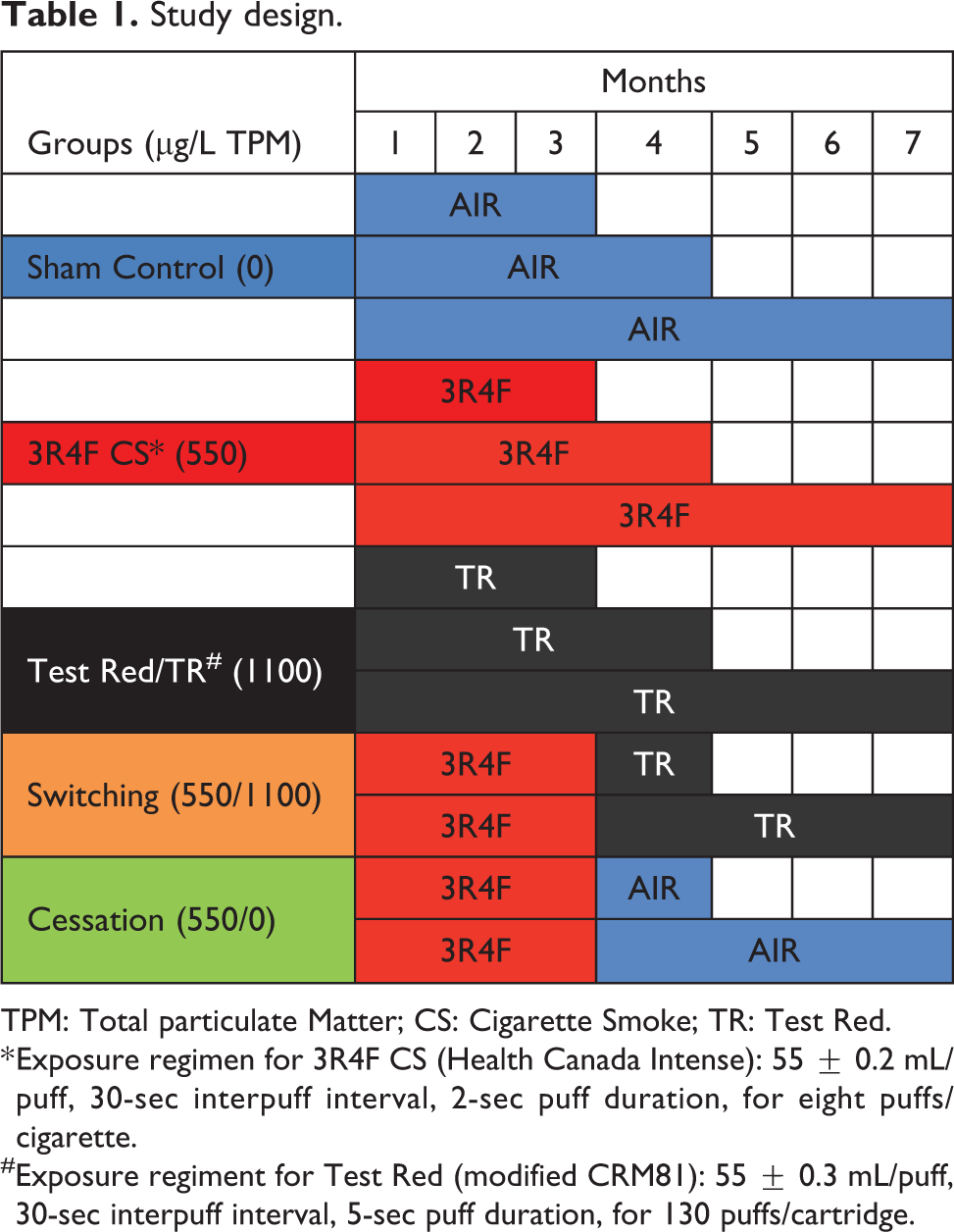
TPM: Total particulate Matter; CS: Cigarette Smoke; TR: Test Red.
* Exposure regimen for 3R4F CS (Health Canada Intense): 55 ± 0.2 mL/puff, 30-sec interpuff interval, 2-sec puff duration, for eight puffs/cigarette.
#Exposure regiment for Test Red (modified CRM81): 55 ± 0.3 mL/puff, 30-sec interpuff interval, 5-sec puff duration, for 130 puffs/cartridge.
Mice were approximately 11 weeks old at the start of exposure. To acclimatize mice with nose-only exposure settings, during quarantine and prior to exposure start, mice were acclimatized to nose-only exposure restraint tubes while breathing room air for an increasing daily duration of 1, 2, 3, 4, and 5 hours over a period of 5 consecutive days. Mice were exposed to 3R4F CS (550 µg/L TPM) or e-vapor aerosols (Test Red; 1100 µg/L TPM) via nose-only inhalation using the same polycarbonate tubes used during acclimatization, up to 4 hours/day, 5 days/week, for up to 7 months. During the first 2 weeks of exposure, exposure duration gradually increased from 0.5 hour (Day 1, 2), to 1 hour (Day 3, 4), to 2 hours (Day 5, 6), to 3 hours (Day 7, 8), and then to 4 hours (starting on Day 9 and continued up to month 7). The daily duration was set to the maximum (4 hours/day) recommended for nose-only inhalation exposures in mice. 42
After the first 3 months of exposure, groups of 3R4F CS mice were subjected to exposures of: (1) Test Red aerosol (“Switching”) or (2) filtered air (“Cessation”), while a group of mice continued to be exposed to 3R4F CS (Table 1).
Mice were examined for in-life observations (clinical signs, body weights, and food consumption) throughout the study. At months 3, 4, and 7, cohorts were evaluated for various biological endpoints (Supplementary Table S2), summarized below.
Biomarkers of exposure (BOEs)
Plasma nicotine, cotinine, and PG
At week 2 and months 3, 4, and 7, mice were anesthetized with isoflurane and blood samples were collected within 10 minutes post-exposure. Whole blood samples were analyzed for carboxyhemoglobin (COHb) using an OSM3 Hemoximeter (Radiometer America Inc., Brea, CA). The remaining blood from these samples was centrifuged for at least 10 minutes at ∼1,500 to 2,000 × g, and resulting plasma samples were analyzed for nicotine, cotinine, and PG using high-performance liquid chromatography coupled with tandem mass spectrometry (HPLC-MS/MS).
Urinary BOEs
Urine was collected in metabolism cages (two mice/cage; with the collection tubes immersed in crushed ice) overnight beginning immediately post-exposure until the following morning, (approximately 20 hours) at week 2 and months 3, 4, and 7. Animals had access to water ad libitum but not food during the urine collection. Urine volume was recorded, specimens were centrifuged, and the supernatant was analyzed for creatinine. Remaining samples were stored frozen at −85°C to −60°C for further analysis of nicotine, trans-3′-hydroxycotinine, nornicotine, nicotine-1′-oxide, cotinine N-oxide, and 3-hydroxypropyl mercapturic acid (acrolein metabolite).
Respiratory physiology and lung function
Respiratory rate (RR), tidal volume (TV), and minute volume (MV) were measured over the first hour of exposure (six/group) at months 3, 4, and 7. Prior to data collection, mice were placed in nose-only restraint tubes and then subsequently placed in volume displacement plethysmographs, which had a pneumotach attached to a port. The pneumotach signals were detected by a variable reluctance transducer, amplified, and recorded by the BioSystemXA data acquisition software (version 2.5.2; Buxco Electronics/Data Sciences International, St. Paul, MN).
Lung function was measured using the Flexivent system (SCIREC, Montreal, Canada) at months 3, 4, and 7. Mice were weighed and sedated with an initial dose of a mixture of ketamine (50 mg/kg) and xylazine (5 mg/kg) via the intraperitoneal (IP) route, followed by another dose of ketamine (100 mg/kg), xylazine (10 mg/kg), and acepromazine (3 mg/kg) via the IP route. Once the surgical plane of anesthesia was reached, mice were subjected to tracheotomy and placed on the Flexivent instrument and ventilated mechanically. Mice were then injected with a muscle paralytic (pancuronium bromide, approximately 0.8 μg/g, IP) to minimize inspiratory efforts against the ventilator. A positive end-expiratory pressure was set at approximately 3 cm H2O and the following lung function parameters 39 (based on the perturbations listed below) were measured:
Snapshot: the tracheal dynamic resistance (R), dynamic compliance (C), and dynamic elastance ([E]; 1/C); Quick Prime: the Newtonian resistance, approximating airway resistance (Rn), tissue damping or resistance (G), and the tissue elastance (H), and tissue histeresivity (G/H); Pressure-volume (PV) loop: the quasi-static compliance (Cst) and quasi-static elastance (Est), pressure-volume curves, estimate of inspiratory capacity (A), shape parameter (K), and area of the PV loop (Hysteresis).
Bronchoalveolar lavage fluid (BALF)
At scheduled termination, the mice from the BAL cohort (Supplementary Table S2) were anesthetized with an IP injection of barbiturate, and whole blood samples were collected via the retro-orbital sinus. After blood collection, BALF was collected as described elsewhere. 31 Briefly, a total of five 1 mL washes were performed for each mouse. Two tubes were collected for each mouse, with tube 1 containing the first wash, and tube 2 containing washes 2 through 5. The tubes were centrifuged at 300 × g for 5 minutes. The supernatant from the first wash (Tube 1) was collected and stored at −85°C to −60°C until analysis of inflammatory mediators and gelatinolytic MMP activity. The cell pellets from both BALF tubes were combined and assessed for cytology.
BAL cytology
The combined cell pellets from each animal were resuspended in 1 mL of saline containing 1% CD16/CD32 antibody (BD Bioscience, San Diego, CA) for flow cytometry. Fluorescence-missing-one (FMO) controls were generated from whole blood of C57BL/6 mice. Cells were washed with BD Stain Buffer and resuspended in 2 mL of 1× Fix Lyse Solution (eBioscience, San Diego, CA) to lyse the red blood cells and fix the cells prior to incubation in the dark at room temperature for 15 minutes. The cells were further incubated at 2°C to 8°C in the dark for up to 48 hours before analysis. Flow cytometry data were acquired on an LSR Fortessa using FACSDIVA (Version 8.0.1; BD Biosciences, San Jose, CA). Appropriate gating strategy was adopted to target populations based upon bimodal responses and FMO stains. Absolute counts of immune cells and their subtypes were reported.
BALF (first wash) analyses
The BALF first wash supernatant was analyzed for (1) Multi-Analyte Profiling (MAP) of pro-inflammatory mediators including cytokines, (2) MMP activity, and (3) a selection of proteins using targeted Parallel Reaction Monitoring (PRM). MAP was performed with the Luminex platform by using the Mouse Cytokine/Chemokine panel (catalog number: MCYTOMAG-70K-PMX-25, Merck KGaA, Darmstadt, Germany) and Mouse CVD panel (catalog number: MCVD1MAG-77K-07, Merck KGaA, Darmstadt, Germany) kits following the manufacturer’s instructions. MMP activity was measured by a gelatinolytic activity assay using fluorescence-labeled gelatin (EnzChek® Gelatinase/Collagenase Assay Kit; Invitrogen, Karlsruhe, Germany) following the manufacturer’s protocol. PRM was used to measure the absolute amounts of 15 selected proteins: AL3A1, ApoA1, CATD, CATS, CXCL15, FETUA, FIBB, IC1, ILEUA, ICAM-1, PERM, SFTPD, SODE, UTER and vWF. Briefly, 70 µl of samples were processed in randomized order using Filter Aided Sample Preparation protocol. Specific details of PRM are given as supplementary information (Materials and methods).
Histopathology
The mice from HISTO cohort mice (Online Supplementary Table S2) were necropsied at months 3, 4, and 7. Animals were weighed prior to necropsy, sedated with a mixture of 70% CO2/30% O2, and exsanguinated. Each necropsy included examination of the external surface of the body and all orifices, the cranial, thoracic, abdominal, and pelvic cavities and their contents, and collection of different tissues (Supplementary Table S3A).
The tissues collected at necropsy were fixed in 10% neutral buffered formalin (NBF), and eyes with optic nerve were first fixed in Davidson’s fixative and then placed in 10% NBF. The lungs were inflated with 10% NBF through tracheal administration at ∼25 cm of water at constant pressure. The following organs were weighed: adrenal glands (paired and weighed together), brain, heart, kidneys (paired and weighed together), liver, lungs, and thymus. Organ weights and the terminal body weight were used to calculate both the organ to body weight and the organ to brain weight ratios. All gross lesions noted at necropsy and selected fixed tissues at scheduled necropsies (Online Supplementary Table S3B) were embedded (paraffin), sectioned, stained (with hematoxylin and eosin), and examined by a board-certified veterinary pathologist, and an external peer-review was also performed. Microscopic findings were graded semi-quantitatively with the average severity grades such as: minimal (Grade 1: the least detectable lesion); mild (Grade 2: an easily discernible lesion); moderate (Grade 3: a change affecting a large area of the represented tissue); and marked (Grade 4: a lesion regarded maximal changes).
Transcriptomics and proteomics
The methods used for transcriptomics analyses have been previously described. 31 Briefly, all OMICS cohort animals were weighed and anesthetized by IP administration of a barbiturate. Blood was collected, and mice were whole-body perfused with cold saline via trans-cardiac perfusion. Tissues (nasal epithelium and lung) were collected, and efforts were made to minimize RNA degradation, including the use of RNase-free collection tubes and petri dishes, cleaning surfaces, and dissection tools with RNaseZap® reagent (Ambion, CA, USA). Tissues were dissected and collected within 15 minutes of perfusion, and then frozen in liquid nitrogen. Cryocut left lung slices and nasal epithelium tissues were processed for isolation of total RNA, including microRNAs, by using the miRNeasy mini Kit (Qiagen, Hilden, Germany). The quantity and integrity of the isolated RNA were determined using a NanoDrop 1000 instrument (Thermo Fisher Scientific, Waltham, MA), and 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA), respectively. Only samples with an RNA integrity number of 6 or greater were considered optimal for subsequent gene expression profiling.
For mRNA analysis, 100 ng of total RNA were reverse-transcribed to cDNA using the Affymetrix® HT 3′ IVT PLUS kit (Thermo Fisher Scientific). The labeled cRNA was hybridized to a GeneChip Mouse Genome 430 2.0 Array (Thermo Fisher Scientific). The arrays were then washed and stained on an FS450 DX GeneChip® Fluidics Station (Thermo Fisher Scientific) and scanned using a 3000 7G GeneChip® scanner (Thermo Fisher Scientific). The raw data were deposited in the ArrayExpress public repository with accession number E-MTAB-9597.
For analysis, raw data (CEL files) were background-corrected, normalized, and summarized using frozen-robust microarray analysis. 43 After a quality control procedure, 44 raw p-values were generated for the Sham Control versus treatment comparisons using the limma package in R, 45 and adjusted using the Benjamini–Hochberg false discovery rate (FDR) multiple test correction. 46 FDR-adjusted p-values <0.05 were considered significant.
Using the causal biological network models, 47 along with network perturbation amplitude (NPA) algorithms, fold changes in gene expression were translated into differential values for each network, which were captured as NPA Scores. 48 These NPA values from all applied network models were aggregated together to generate a Biological Impact factor (BIF) 48 –50 to assess impact of exposure on biology. For comparisons of BIF, the values were expressed as the relative BIF, i.e. values were normalized as a percentage of the maximum BIF. In addition, genes coding for surfactants were analyzed for gene expression, and gene set enrichment analysis was performed to investigate lipid metabolism-specific changes in gene expression by searching in the Reactome data base for lipid associated pathways. Aspects of lipid metabolism currently annotated in Reactome include lipid digestion, mobilization, and transport; fatty acid, triacylglycerol, and ketone body metabolism; peroxisomal lipid metabolism; phospholipid and sphingolipid metabolism; cholesterol biosynthesis; bile acid and bile salt metabolism; and steroid hormone biosynthesis.
Lung tissues from the OMICS cohort mice were assessed for proteomic profiles as previously described. 39 Briefly, frozen tissue samples were homogenized, followed by extraction and processing using the iTRAQ® 8-plex labeling procedure in accordance with the manufacturer’s instructions (AB Sciex, Framingham, MA, USA). iTRAQ® analysis sets were assigned to consist of one sample replicate from each of exposure groups, with eight separate iTRAQ® sets corresponding to the eight replicates. Additionally, one pooled reference mix with all samples combined was included in each iTRAQ® set. Consequently, an Easy-nLC 1000 instrument connected online to a Q-Exactive MS mass-analyzer (Thermo Fisher Scientific, Waltham, MA) was used to analyze iTRAQ® sample sets.
For identification and quantification of proteins, mass spectra obtained were compared against a mouse reference proteome set (UniProt, version July 2014, canonical isoforms only) using Proteome Discoverer software (version 1.4, Thermo Fisher Scientific). Further processing of peptide-level quantification data was done in the R statistical environment. The peptide-level quantification data were filtered for a q value <.01 and for “unique” quantification results, as defined by Proteome Discoverer software. Bioconductor package in R was used for a global variance-stabilizing normalization. 51,52 Each iTRAQ® reporter-ion set was normalized to its median, and protein expression values were determined as the medians of normalized peptide-level quantification values. 53 For the statistical analysis to determine differentially expressed proteins, a linear model was fitted for each exposure condition and the respective control groups. Moderated t-statistics with the empirical Bayes approach 54 was used to calculate p-values for control versus exposure conditions, and proteins with a Benjamini–Hochberg FDR-adjusted p-value <0.05 were considered differentially expressed. The KEGG database was used as the protein/gene set collection. 55 Significance was assessed by protein (Q1) and sample (Q2) permutation. 56 For each test, the p-values were adjusted using the Benjamini–Hochberg procedure. The mass spectrometry proteomics data have been deposited in the database of the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD019918.
Statistics
Results were considered significantly different for a specific comparison if p < 0.05. Statistical comparisons included: all treatment groups compared to the Sham Control group; the 3R4F CS group compared to the Test Red, Cessation, and Switching groups; the Cessation group compared to the Switching group; and the Test Red group compared to the Cessation and Switching groups. Statistical comparisons on in-life observations (body weights, food consumption, respiratory physiology), Flexivent, COE, BALF flow cytometry (absolute and percent values), and organ weight data were performed using the Provantis system from Instem (Philadelphia, USA).
Data were analyzed for treatment effects by parametric or nonparametric analysis of variance (ANOVA). Normality was determined by the Shapiro-Wilk’s test, and homogeneity of variances was determined by Levene’s test. Data were log-transformed if needed to meet parametric assumptions. For parametric data determined to be normally distributed and homogenous among groups, an ANOVA F-test was used to determine differences among group means. If the ANOVA F-test was significant, then tests for differences between the control and each of the comparison groups were conducted using Dunnett’s post-hoc test. For nonparametric data that were not normally distributed and/or non-homogenous, a Kruskal-Wallis test was used. If the Kruskal-Wallis test was significant, then tests for differences between the control and each of the comparison groups were conducted using Wilcoxon tests and the Bonferroni-Holm method to correct for multiple comparisons.
Histopathology data (incidence and severity scores) were exported from Provantis and the comparisons were performed in SAS (v9.4). A two-sided Fisher’s Exact Test was used to compare the incidence rates between each pair of groups at each time points. In addition, the incidence rates were compared using Cochran-Mantel-Haenszel (CMH) test across time points (months 3, 4, 7, and recovery) to determine if the rates cumulatively resulted in a statistically significant difference. For each endpoint, the distribution of severity scores for each exposure group was compared across exposure groups via a two-sided Kolmogorov-Smirnov Exact Test at each time point to determine if the distribution of scores was significantly different.
For the mechanistic endpoints (MAP, MMP activity, and PRM) in BALF (first wash), treatment comparisons were performed as previously described. 31 Briefly, comparisons were done using t-tests within a linear mixed model framework for each endpoint. To stabilize the variability of outcomes, the targeted endpoints were first transformed to the logarithmic scale prior to statistical modeling. Results were shown using boxplots.
Results
Exposure characterization
The inhalation atmosphere (Supplementary Table S4) was well-controlled over 7 months of exposure and the results were overall comparable to prior studies. 31 For the 3R4F CS exposure, the overall mean TPM concentration was 540 µg/L (within 2% of the target concentration of 550 µg/L TPM), with a % relative standard deviation (RSD) of 3%. For Test Red e-vapor aerosol exposures, the mean TPM was 1091 µg/L (within 1% of the target concentration of 1100 µg/L TPM), with a % RSD of 3%. Although the Test Red TPM concentration was two-fold higher than the 3R4F CS TPM, the Test Red nicotine concentrations were ∼30% lower (29.4 µg/L) compared to 3R4F CS (42.1 µg/L). As explained in the BOE section, we set the nose-port nicotine levels to be higher for 3R4F CS exposures because of CS-specific suppression of respiratory function. CO was only detected in the 3R4F CS group whereas PG was only detected in e-vapor aerosol exposures.
Despite higher nose-port TPM compared with 3R4F CS, e-vapor aerosol had significantly diminished levels of carbonyls. Compared to 3R4F CS, the concentrations of acrolein, acetaldehyde, and propionaldehyde in e-vapor aerosol exposures were over 94%–99% lower. The particle size (MMAD 0.7–1.1, GSD 1.4–1.5) for both the Test Red aerosols and 3R4F CS were within the respirable range for rodents. 57 The overall mean nose-port temperature and supply air relative humidity parameters were within acceptable limits of 22°C ± 2°C and 55% ± 15%, respectively.
In-life observations
There was no significant impact on mortality related to exposures throughout the in-life phase (7 months of exposure plus 9 weeks of recovery). After a 2-week exposure acclimation and until the end of in-life phase, the mean survival rates were high and similar among groups (96% to 99%); there were a total of 21 early removals composed of 2, 5, 3, 6, and 5 mice from the Sham Control, 3R4F, Test Red, Switching, and Cessation groups, respectively.
Following daily exposures, the 3R4F CS group showed the most notable clinical findings (e.g., hunched posture, head tilt, rough coat) at higher frequency and incidence than other e-vapor aerosol and Sham Control groups. In addition, a majority of mice in the 3R4F CS groups also displayed decreased activity, tremors, and were cold to touch which depicted clinical signs of distress and were mostly noted as post-exposure observations (Supplementary Table S5). After mice were switched to Test Red (Switching) or filtered air (Cessation) following 3 months of 3R4F CS exposures, the above CS-related signs of stress were only observed infrequently.
The 3R4F CS group also displayed consistently lower body weight throughout the study compared to all other exposures and Sham Control (Supplementary Figure S1A). After the first 3 months of exposure, the mean body weights for Switching and Cessation groups gradually increased and reached comparable levels to Sham Control/Test Red groups following ∼21 weeks of exposure. The Test Red group mean body weights tended to mirror the Sham Control throughout the study, generally within 3% of the Sham Control group (Supplementary Figure S1A). Concurrent with changes in body weights, the food consumption showed a similar trend for all exposure groups, with lowest mean food consumption in the 3R4F CS group (Supplementary Figure S1B). Overall, in-life observations showed most notable changes in the 3R4F CS group, with none to minimal changes in Test Red, and clear signs of reversal on switching to Test Red (Switching group) or filtered air (Cessation group).
Among a total of 21 early removals, 6 mice showed lesions related to GBS infection at necropsy (1 of Sham Control, 2 of 3R4F CS, 1 of Test Red, 1 of Switching and 1 of Cessation). An additional 10 mice were identified with GBS based on microbial culture obtained at scheduled sacrifices (months 4 and 7). To confirm the GBS infection, fecal PCR was performed in all mice at months 4, 7, and recovery sacrifices (Supplementary Table S6). Fifty mice out of the 490 tested (10%) were positive for GBS: 2/100 (2% in Sham Control), 8/98 (8% in 3R4F CS), 17/99 (17% in Test Red), 20/96 (21% in Switching) and 3/97 (3% in Cessation). However, not all affected mice were symptomatic or were accompanied with tissue findings; only 14% (7/50) showed gross and/or culture results consistent with GBS. Identification for GBS was regarded as a background finding under the standard pathogen-free designation by the animal supplier (Charles River Laboratories, Stone Ridge, NY). Given the overall low number of mice with GBS lesions and the associated distinctive microscopic changes, the study pathologist and the peer-review pathologist concluded no impact to the interpretation of the study results.
Biomarkers of exposure (BOEs)
Both plasma and urine samples were collected and analyzed for BOEs (Table 2). Reflecting high concentrations of CO in the test atmosphere of the 3R4F CS group, blood COHb levels ranged from 42% to 48%, whereas Sham Control and both e-vapor (Test Red and Switching) groups showed only background levels (≤2.3%). Plasma PG was detected only in e-vapor aerosol exposed groups. As presented in Supplementary Table S4, despite the fact that exposure nicotine concentrations were ∼30% higher for the 3R4F CS group compared to the e-vapor group, the plasma nicotine and cotinine levels of the 3R4F CS group were overall two- to three-fold lower than all e-vapor (Test Red and Switching) groups (Table 2).
Biomarkers of exposure.
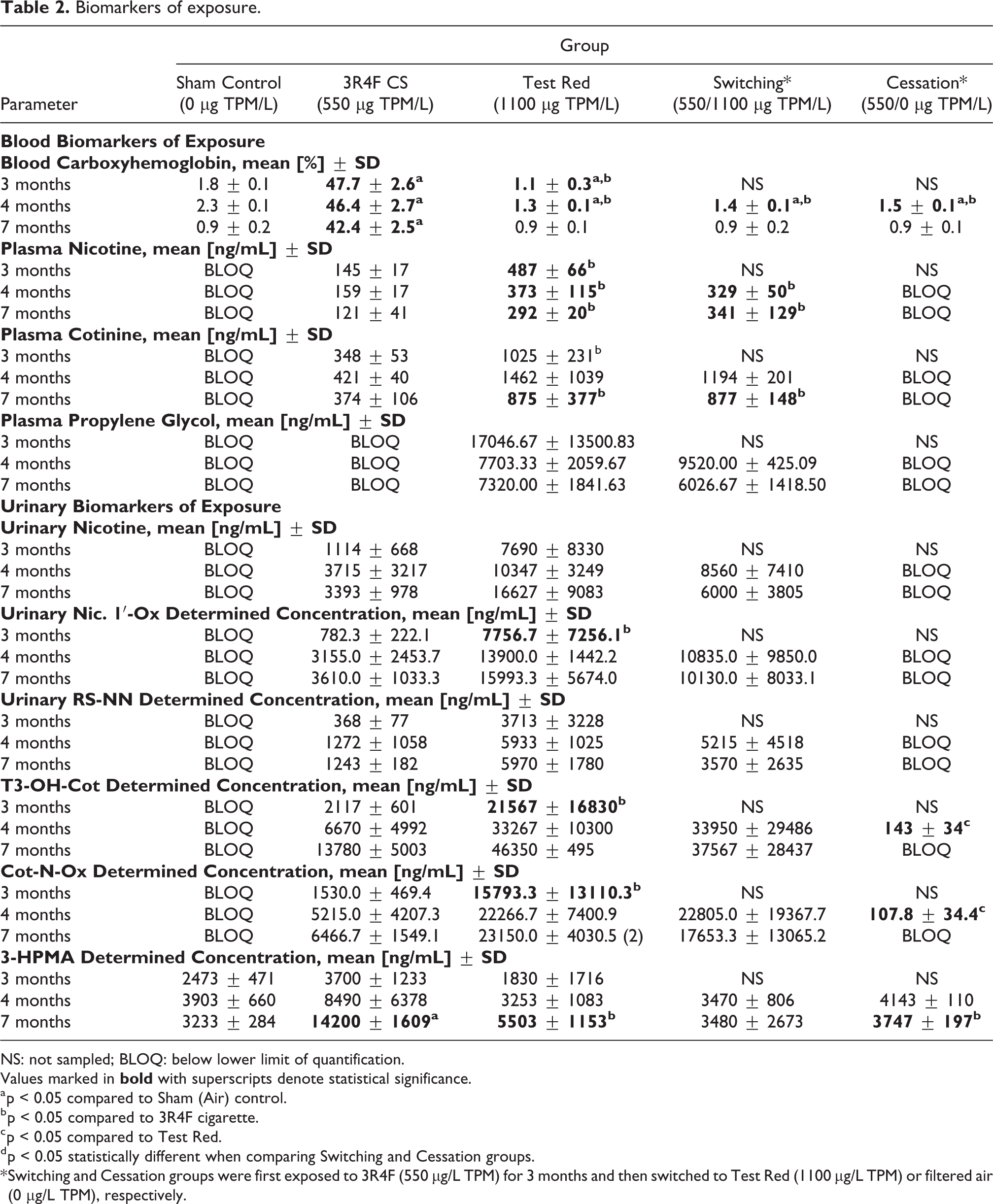
NS: not sampled; BLOQ: below lower limit of quantification.
Values marked in
a p < 0.05 compared to Sham (Air) control.
b p < 0.05 compared to 3R4F cigarette.
c p < 0.05 compared to Test Red.
d p < 0.05 statistically different when comparing Switching and Cessation groups.
* Switching and Cessation groups were first exposed to 3R4F (550 µg/L TPM) for 3 months and then switched to Test Red (1100 µg/L TPM) or filtered air (0 µg/L TPM), respectively.
Urine analysis of nicotine and nicotine metabolites (nicotine 1-oxide; R, S-nornicotine, trans-3′-hydroxycotinine, and cotinine N-oxide) concurred in trend with plasma nicotine and cotinine levels with higher mean levels in both e-vapor (Test Red and Switching) groups at all time points as compared to 3R4F CS groups. In contrast to urinary levels of nicotine and metabolites, 3-HPMA, a urinary biomarker for acrolein exposure, was present in all groups including the Sham Control; however, the 3R4F CS group had consistently higher levels than Sham Controls with up to >4-fold higher mean levels at month 7, while all e-vapor (Test Red and Switching) groups had similar or slightly higher (<1.7-fold) levels than Sham Control.
Respiratory physiology
Respiratory function (RR, TV, and MV) was measured over the first hour of exposure and the MV and the actual TPM were used to calculate the estimated total inhaled mass (TIM) (Table 3). The 3R4F CS group showed significant and consistent suppression in RR up to ∼45% compared to Sham Control. The TV was not significantly different between the Sham Control and any of the exposure groups at all time points. Consistent with the decline in RR, MV was significantly lower (up to 56%) in the 3R4F CS group as compared to Sham Control (Table 3; Online Supplementary Figure S2). Overall, the Test Red group, as well as the Switching and Cessation groups, showed comparable respiratory function to the Sham Control. Because of the difference in MV, the resulting TIM was approximately three- to four-fold higher for e-vapor groups (Test Red and Switching) compared to the 3R4F CS group.
Respiratory physiology.
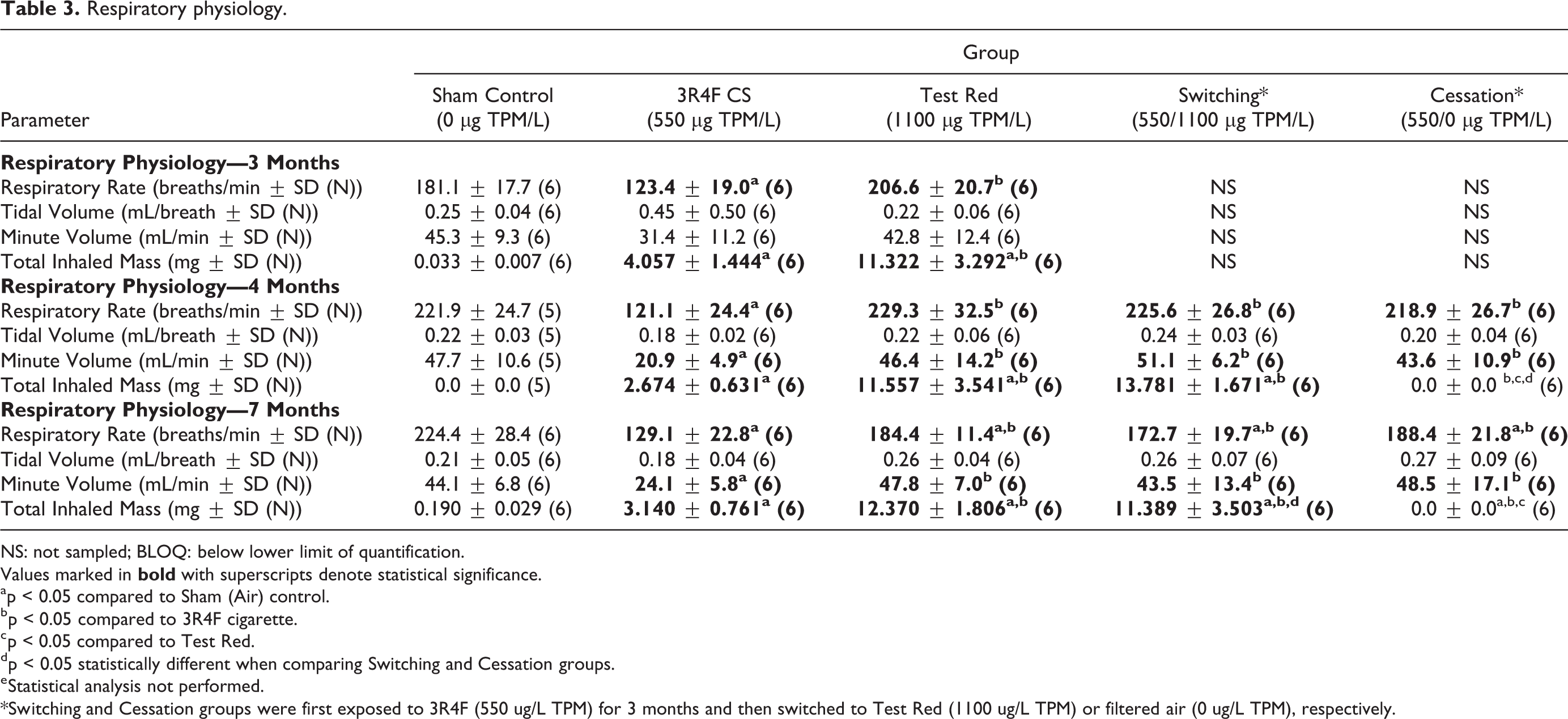
NS: not sampled; BLOQ: below lower limit of quantification.
Values marked in
a p < 0.05 compared to Sham (Air) control.
b p < 0.05 compared to 3R4F cigarette.
c p < 0.05 compared to Test Red.
d p < 0.05 statistically different when comparing Switching and Cessation groups.
e Statistical analysis not performed.
* Switching and Cessation groups were first exposed to 3R4F (550 ug/L TPM) for 3 months and then switched to Test Red (1100 ug/L TPM) or filtered air (0 ug/L TPM), respectively.
Lung function by Flexivent
After confirming exposure, downstream effects on lung function were investigated using Flexivent. Exposure to 3R4F CS resulted in an upward and leftward shift for both inflation and deflation curves at all time points compared to the Sham Control (Figure 1(a)). The lung volumes at the maximum pressure in 3R4F CS group were persistently larger than Sham Control, which was consistent with emphysematous changes observed in mice after chronic exposure to cigarette smoke. 39 In contrast, apart from a slight shift up and left at month 4, the PV curve for Test Red group overlapped with Sham Control at months 3 and 7. The PV curves in Switching and Cessation groups showed the transition from closely overlapping with 3R4F CS group at month 4 to shifting away, falling between the 3R4F CS group and the Sham Control at month 7.
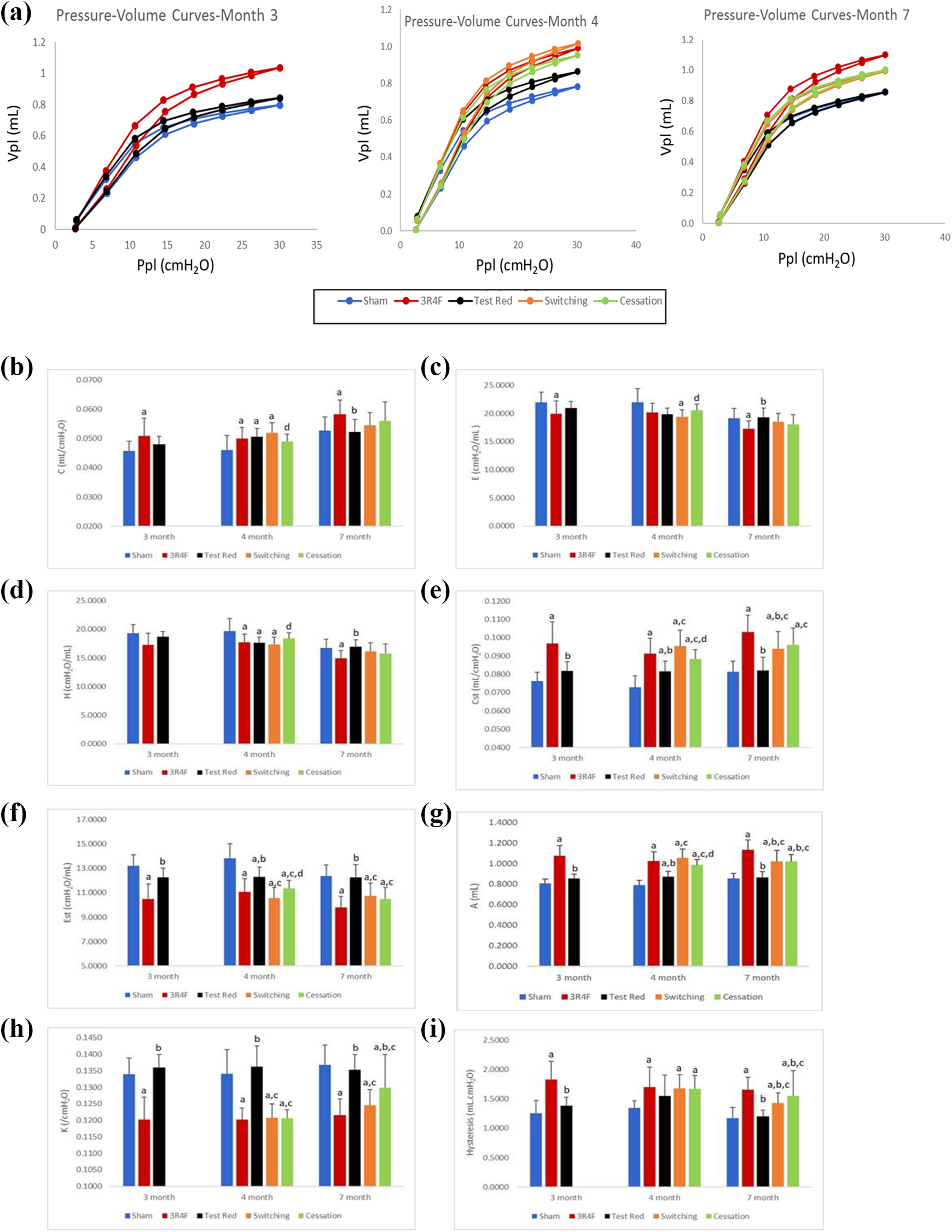
Lung function measurements in lung function cohort animals were done using Flexivent at months 3, 4, and 7. (a) Pressure-volume curves at 3, 4, and 7 months (left, middle, and right panels respectively) after exposure. Mean values of pressure (Ppl) and volume (Vpl) derived from PV loop perturbations were plotted. For other parameters shown in Figure 1(b) to (i), data are presented as mean ± SD. (b) Dynamic compliance, (c) dynamic elastance, (d) tissue elastance, (e) quasi-static compliance (Cst), (f) quasi-static elastance (Est), (g) estimate of inspiratory capacity, (h) shape parameter, and (i) area of the PV loop (hysteresis) derived from PV loop perturbations were plotted. N = 11 to 12 mice used to generate these data and comparisons with p < 0.05 were considered statistically significant. ap < 0.05 compared to Sham Control (Air) control; bp < 0.05 compared to 3R4F CS cigarette; cp < 0.05 compared to Test Red; dp < 0.05 statistically different when comparing Switching and Cessation groups.
Further, the significant increase in dynamic compliance, quasi-static compliance, estimate of inspiratory capacity and hysteresis (area of PV loop) along with decreased dynamic elastance, tissue elastance, quasi-static elastance, and shape parameter K in the 3R4F CS group at all time points indicated lung changes concurring with emphysema. These parameters for Test Red group mostly overlapped with Sham Control with exceptions at month 4; however, the difference was no longer observed at month 7 (Figure 1(b) to (i)). The Switching and Cessation groups mostly mirrored the 3R4F CS group after 1-month post switching or cessation (month 4); however, by month 7 some functional parameters (dynamic compliance, dynamic elastance, and tissue elastance) had returned to levels approximating with Sham Control (Figure 1). Other parameters (quasi-static compliance, quasi-static elastance, estimate of inspiratory capacity, shape parameter K, and area of PV loop/hysteresis) did not fully return to levels of the Sham Control indicating reversibility but not complete recovery by 4 months after switching or cessation. A full list of lung function parameters is shown in Supplementary Table S7.
BALF parameters
BALF cytology data demonstrated a clear sign of pulmonary inflammation in the 3R4F CS group (Figure 2(a) to (h)). These changes often included increased absolute leukocyte counts which were also reflected in an overall increase in the number and/or activation status of multiple cell types, CD3+ T cells, B-cells, macrophages (activation status only), dendritic cells, neutrophils, and eosinophils/basophils/mast cells (Figure 2(a) to (h)). Macrophage absolute counts (total, and alveolar) were often decreased in the 3R4F CS group (Supplementary Table S8), apparently contradicting increases in BALF macrophages often reported in literature. 38,39,58 This could be associated with a much higher number of dead cells in the 3R4F CS group than other groups. In contrast, 7 months of e-vapor (Test Red) aerosol exposures induced only minimal changes (increased number and activation of macrophages; Supplementary Table S8) without invoking infiltration of other leukocytes. While most changes in BALF cytology showed similar reversibility for Switching and Cessation groups, the number of activated macrophage/dendritic cells tended to fall between 3R4F CS and the Test Red groups by month 7, reflecting incomplete reversal and residual effects of 3R4F CS exposure (Figure 2(d) and (f)).
To better understand the activation status of macrophages, we further characterized as either M1 or M2 based on their polarization status using month 7 samples. M1 macrophages are associated with inflammatory activation, whereas M2 macrophages are associated with physiological processes such as wound healing. 59 Flow cytometric analysis of CD45+/F4/80+/CD68+/MHCII+ cells depicting M1 macrophages showed that the 3R4F CS-exposed group had a macrophage population skewed to the M1 phenotype (Supplementary Figure S3). This was in concurrence with other BALF cytology changes (Supplementary Table S8) supporting increased pulmonary inflammation in the 3R4F CS group. In contrast, while other exposure (Test Red, Switching or Cessation) groups showed increased numbers of macrophages, they were M2 types similar to Sham Controls.
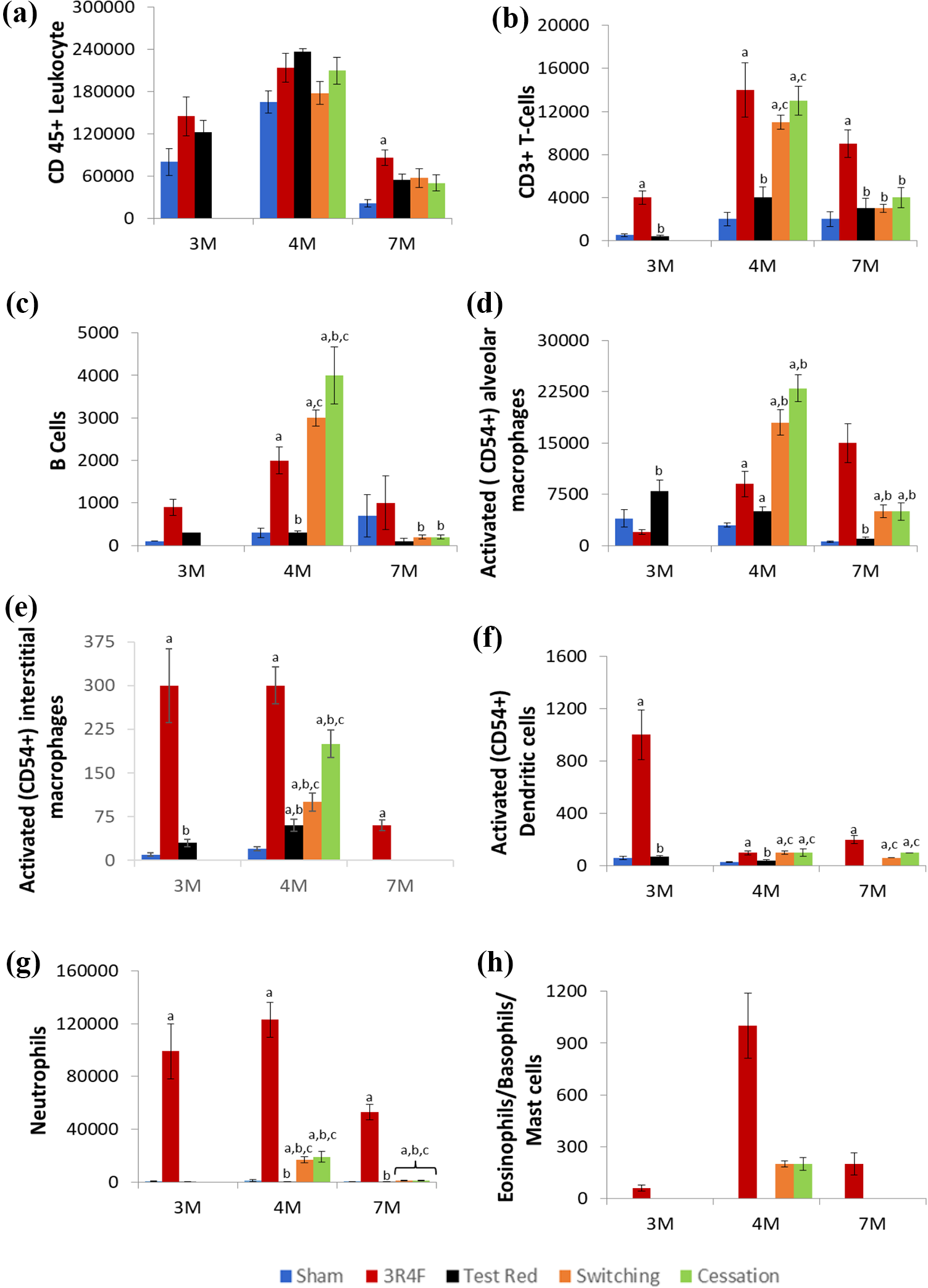
Immuno-phenotyping of bronchoalveolar lavage fluid (BALF) cells by flow cytometry. BALF cells were identified and quantified by flow cytometry. Data are expressed as mean of absolute cell count per mL ± SE for (a) total leukocyte counts, (b) CD3+ T cells, (c) B cell counts, (d) activated (CD54+) alveolar macrophages, (e) activated (CD54+) interstitial macrophages, (f) activated (CD54+) dendritic cells, (g) neutrophils count, and (h) eosinophil/basophil/mast cells count. N = 10 mice used to generate these data and comparisons with p < 0.05 were considered statistically significant. ap < 0.05 compared to Sham Control (Air) control; bp < 0.05 compared to 3R4F CS cigarette; cp < 0.05 compared to Test Red.
Consistent with increased inflammatory cell counts in the BALF, the exposure to 3R4F CS elicited a significant increase in the levels of many inflammatory mediators (Figure 3(a); Supplementary Table S9). Specifically, inflammatory chemokines, tumor necrosis factor (TNF)α, macrophage inflammatory protein (MIP) 1a, 1b, monocyte chemoattractant protein (MCP) 1, platelet-factor 4-type cytokine (KC), interferon gamma-induced protein (IP)-10, and G-CSF and protease (pro-matrix metalloprotease [MMP]-9) were consistently upregulated in the 3R4F CS group relative to the Sham Control group at all time points. This was a clear contrast to very few changes observed in the Test Red group compared to Sham Control. In the Cessation and Switching groups, KC and G-CSF were upregulated after 4 and 7 months, whereas TNFα was upregulated only at the 4-month time point. In general, the Cessation and Switching groups exhibited similar changes in BALF chemokines (Figure 3(a)).
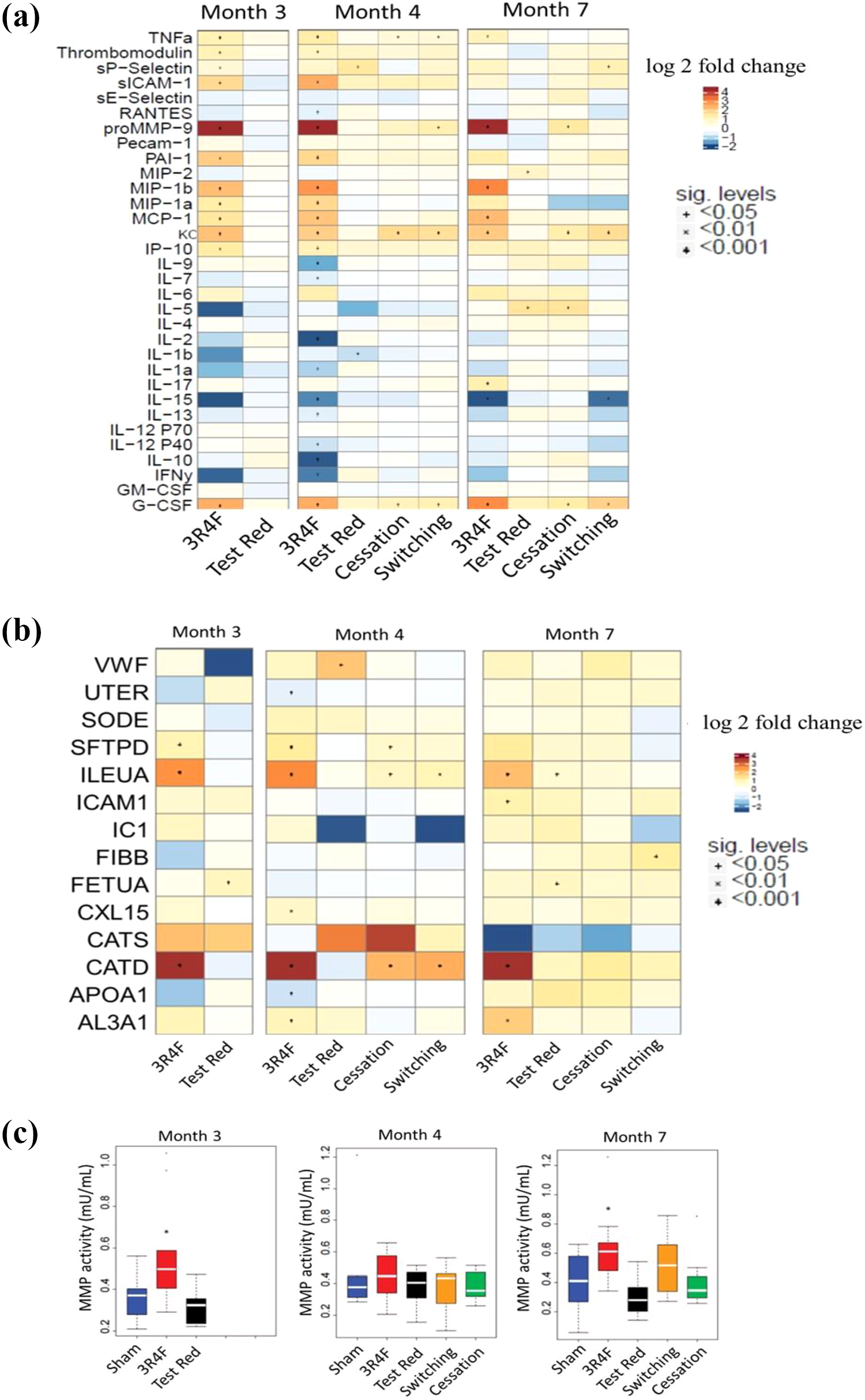
Inflammatory mediators in bronchoalveolar fluid (BALF). (a) Cytokine profiling in BALF: the color scale reflects the magnitude of the estimated differences among the groups (log2-fold change), while the text symbols (star, cross, and bullet) correspond to p-values resulting from testing for significant differences between the groups at P < 0.05, P < 0.01, and P < 0.001 significance levels, respectively. Warm tones ranging from beige to red depict increasing values in endpoints in the treatment groups compared with the Sham Control, while cool tones ranging from sky-blue to deep blue depict decreasing values for the targeted endpoints in the treatment groups compared with the Sham Control. (b) Targeted proteomics analysis in BALF. The color scale and significance symbols are the same as in Figure 3(a). (c) Boxplots of matrix metalloproteinase activity in BALF. Horizontal lines inside the boxplots indicate medians. Boxes include 50% of the observed data. The asterisk indicates a significant difference compared with Sham Control. All abbreviations can be found in Supplementary Table S9.
To complement BALF chemokine analysis, targeted proteomics was performed by PRM. Compared with the Sham Control, most changes were consistently observed with the 3R4F CS group, with consistent upregulation in cathepsin D (CATD) and Serpinb1 (ILEUA) (Figure 3(b)). Test Red aerosol exposure showed changes in fetuin-A (FETUA) levels at months 3 and 7 with inconsistent changes in von Willebrand factor (VWF) at month 4 and ILEUA at month 7. In general, Cessation and Switching groups showed upregulation in ILEUA and CATD levels at month 4; however, these changes were not evident at month 7. Altogether, concurring with the MAP findings, PRM analysis indicated the most notable changes in BALF protein markers in the 3R4F CS group and fewer changes in inflammatory mediators in the Test Red, Switching, and Cessation groups.
The gelatinolytic MMP activity in BALF collected from the 3R4F CS group was significantly higher at months 3 and 7 as compared to Sham Controls. There was no significant difference in the MMP activity in the Test Red, Switching, and Cessation groups compared with the Sham Control at any of time points tested (Figure 3(c)).
Organ weights
Following 3, 4, and 7 months of exposure, the most notable changes in the organ weights, absolute as well as relative to body and brain weights, were observed with the 3R4F CS group (Figure 4; Supplementary Table S10). The 3R4F CS group had significantly higher relative (to body or brain weight) lung weights compared to Sham Control and Test Red groups at months 3 and 4. At month 7, the 3R4F CS group had significantly higher lung (absolute and relative) weights compared to Sham Control, Test Red, Switching, and Cessation groups. In addition, the 3R4F CS group showed a significant decrease in absolute and relative liver weights compared to all exposure groups, and a significant decrease in absolute and relative adrenal gland weights at month 4 compared to Sham Control. None of these organs had any significant changes in absolute or relative weights in Test Red, Switching or Cessation groups compared to Sham Control.
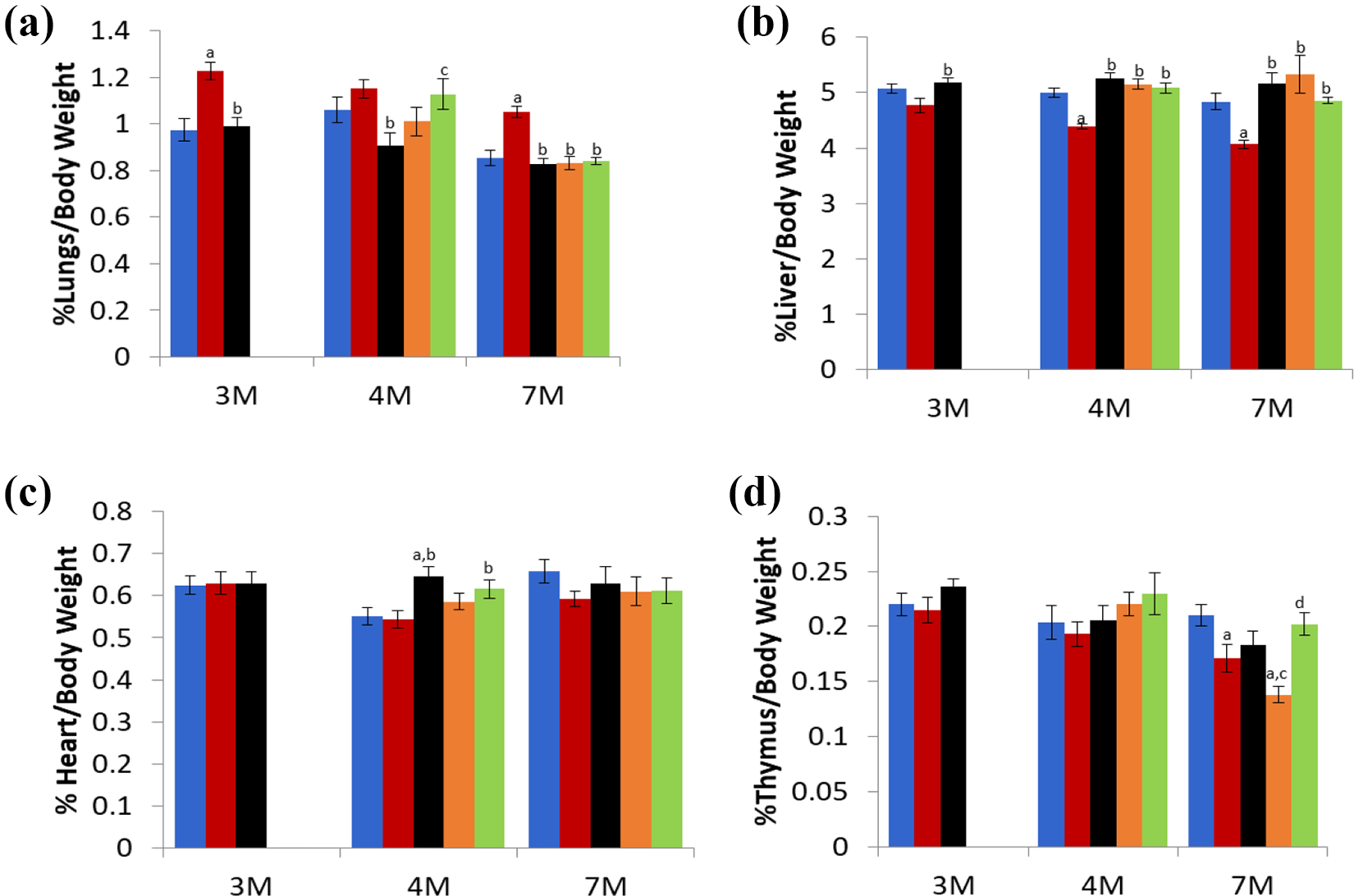
Percent organ weights relative to body weights for (a) lungs, (b) liver, (c) heart, and (d) thymus. Values are expressed as mean ± SE for N = 11 to 12. Comparisons with p < 0.05 were considered statistically significant. ap < 0.05 compared to Sham Control (Air); bp < 0.05 compared to 3R4F CS cigarette; cp < 0.05 compared to Test Red; dp < 0.05 statistically different when comparing Switching and Cessation groups.
There were other organ weights displaying sporadic differences without a clear trend between the exposed groups and Sham Control. In thymus, a significant decrease in absolute and relative weights was observed in the 3R4F CS and Switching groups only at month 7. The Test Red group showed significantly higher heart (absolute and relative) weight at month 4 only.
Histopathology
Histopathological findings for respiratory tissues are listed in Table 4. As observed with changes in organ weights, the 3R4F CS groups had the most notable and consistent changes in nose, larynx, trachea, and lungs compared to Sham Control and e-vapor groups.
Histopathology incidence and severity.
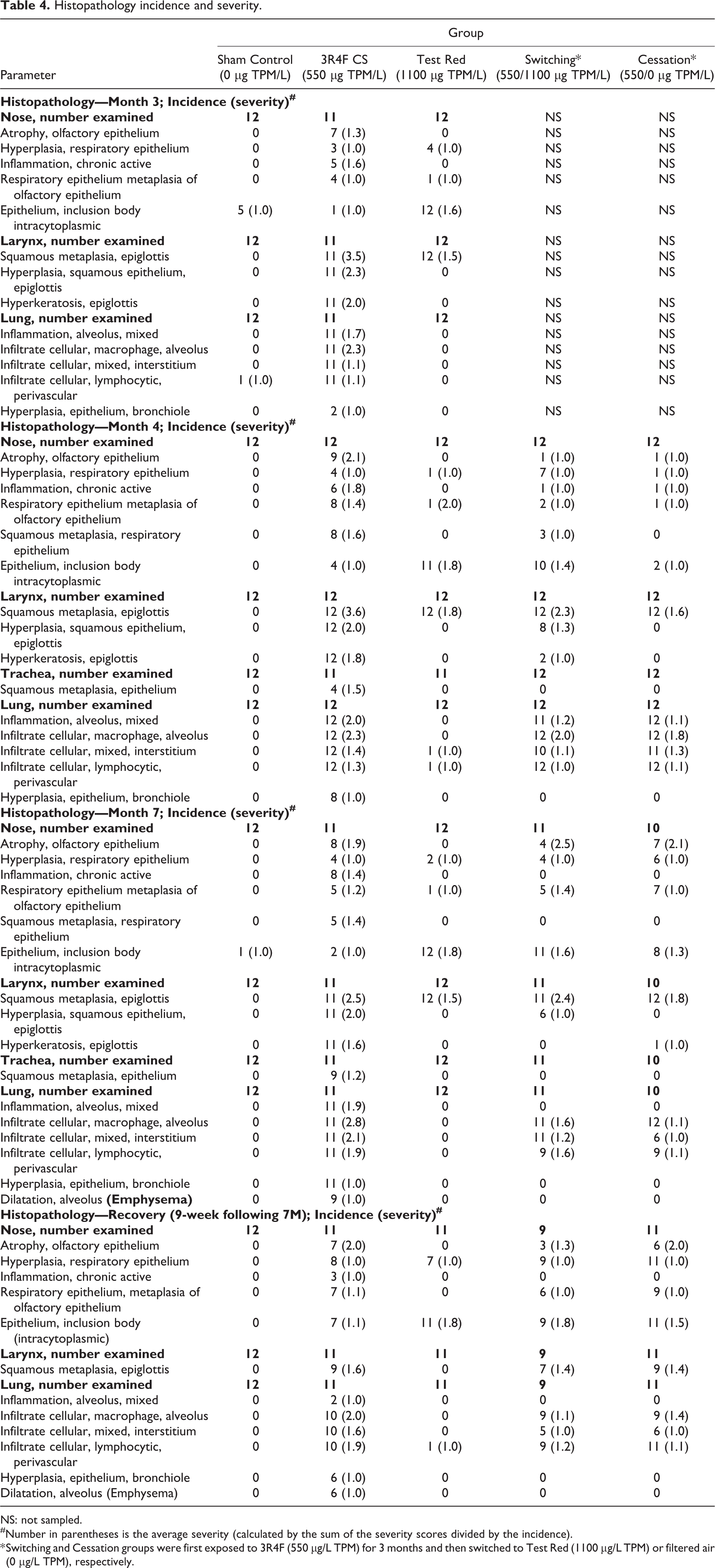
NS: not sampled.
#Number in parentheses is the average severity (calculated by the sum of the severity scores divided by the incidence).
* Switching and Cessation groups were first exposed to 3R4F (550 µg/L TPM) for 3 months and then switched to Test Red (1100 µg/L TPM) or filtered air (0 µg/L TPM), respectively.
In the nose, 3R4F CS exposure elicited olfactory epithelium atrophy, hyperplasia in respiratory epithelium, focal metaplasia in respiratory to olfactory epithelium, and chronic active inflammation within the mucosal and underlying submucosa of nasal cavity at months 3, 4, and 7. The majority of these findings were often only seen, or of increased severity, in the 3R4F CS group, while nasal respiratory epithelial hyperplasia and intracytoplasmic epithelial inclusions were also observed in other exposure (Test Red, Switching, and Cessation) groups compared to Sham Control. Following 9 weeks of recovery after 7 months of exposure, most of the nasal lesions persisted in all exposure groups with slightly lower incidence and/or severity when compared to month 7, indicating incomplete recovery from aerosol exposures.
Histopathological assessment of the larynx showed the most prominent findings (severity and incidence) in the 3R4F CS group, such as hyperplasia, squamous metaplasia, and hyperkeratosis (a superficial surface layer of stratified/flattened densely packed keratin) at the base of the epiglottis at months 3, 4, and 7 (Figure 5(a)). Squamous metaplasia at the base of the epiglottis was also consistently observed in the Test Red group, although at lower severity than the 3R4F CS group (Table 4). Hyperplasia of squamous epithelium was seen in the Switching group at months 4 and 7 but at lower severity and incidence as compared to the 3R4F CS group. The Switching group showed lower incidence of hyperkeratosis at month 4, which was absent at month 7. Following 9 weeks of recovery, the previously CS-exposed (3R4F CS, Switching, and Cessation) groups showed decreases in most laryngeal lesions (lower incidence and severity) but did not completely recover. In contrast, the Test Red group showed full recovery (no incidence) of laryngeal squamous metaplasia at the base of the epiglottis after 9 weeks of recovery.
Histopathological evaluation of trachea showed minimal to mild squamous metaplasia at months 4 and 7 in the 3R4F CS group. Following 9 weeks of recovery, the tracheal lesions were absent in the 3R4F CS group, indicating complete recovery. All other groups including Test Red, Switching or Cessation did not show any notable histopathological findings in trachea at any time points.
Up to 7 months of 3R4F CS exposure elicited the most prominent changes (incidence and severity) in lungs, most notably pulmonary inflammation with cellular infiltration, bronchiolar epithelial cell hyperplasia, and dilatation of the terminal/respiratory bronchioles and adjacent alveoli, indicative of pulmonary emphysema (Figure 5(b); Table 4). For most of these changes, the incidence was 8 to 12/12, and severity was minimal to moderate. In contrast, apart from a few sporadic findings (interstitial mixed cell infiltrates and perivascular lymphocytic infiltrates; 1/12 at 4 months), the Test Red exposure did not elicit any significant histopathological changes in the lung compared to Sham Control. The Switching and Cessation groups displayed similar incidence to lung findings for the 3R4F CS group at month 4, but the severity of lung lesions consistently decreased by month 7. Lung lesions between Cessation and Switching groups were largely comparable, suggesting that the e-vapor (Test Red) aerosol exposure had only minimal effect on histopathological findings and that most changes in Switching or Cessation groups were likely attributed to residual effects of 3R4F CS exposure.
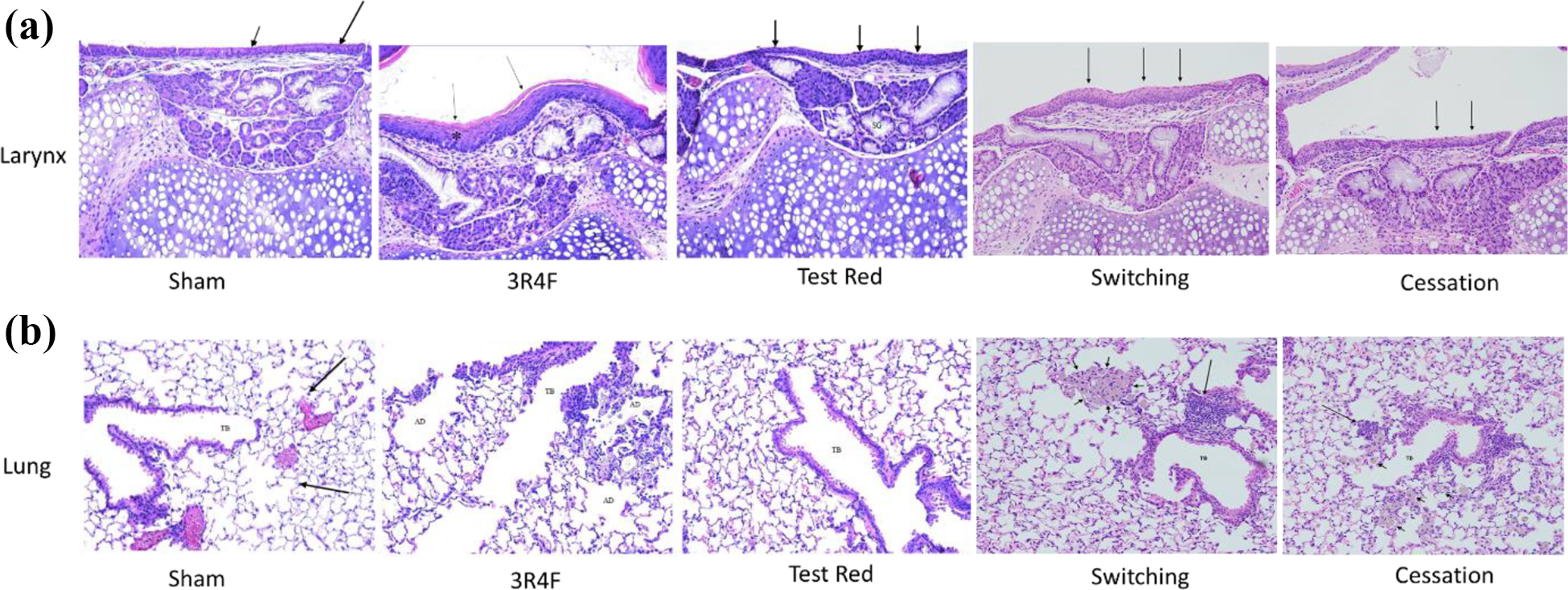
H&E staining showing representative images from mice exposed for 7 months (N = 11 to 12). (a) Larynx. Sham panel shows ciliated (long arrow) epithelial lining at the base of the epiglottis. 3R4F panel shows clear signs of metaplasia (*) accompanied by hyperkeratosis (arrow). Test Red panel shows squamous metaplasia at the base of epiglottis. Switching and Cessation panels show minimal squamous metaplasia (arrows) at the base of the epiglottis. (b) Lung. Sham panel shows very few alveolar macrophages (arrows) which were deemed within normal limits. 3R4F panel shows terminal bronchiolar (TB) region with evidence of alveolar dilation in regions showing inflammation. Test Red panel shows terminal bronchioles devoid of inflammatory cells with only fewer alveolar macrophages. Switching panel shows terminal bronchiolar region with evidence of residual inflammation (long arrow) and a small aggregate of pigmented macrophages (short arrows). Cessation panel shows terminal bronchiolar region with evidence of residual inflammation (long arrow) and a few scattered pigmented macrophages (short arrows). Images were taken at 20× magnification.
Transcriptomics and proteomics
Up to 7 months of 3R4F CS exposure triggered the differential expression of thousands of genes in the lung compared to Sham Control, with the highest number of differentially expressed genes (DEGs) at month 7 (Figure 6(a) and (b)). This was in a stark contrast to findings in the e-vapor (Test Red) group which did not show any significant increase in DEGs compared to Sham Control at any time point. After cessation or switching, the number of DEGs gradually decreased over months 4 and 7, and by month 7, there were 114 DEGs in the Cessation group, whereas only 10 DEGs were observed in the Switching group compared with the Sham Control (Figure 6(a) and (b)).
As described in the methods section, these changes in gene expression were applied to causal biological network models 47 and NPA algorithms to infer differential values for each network model. 48 These network models capture mechanistic details of various biological processes relevant to lung, which contribute to cellular fate (CFA), cellular proliferation (CPR), cellular stress (CST), inflammation (IPN), tissue repair and angiogenesis (TRA) processes. The impact on network models perturbed in each exposure group is shown as a heatmap of NPA scores (Figure 6(c)). These NPA values from all applied network models were aggregated to generate a BIF to assess the overall impact of exposure on biology of the lungs. 31 This causal biological network approach, based on transcriptomics data from the lung, showed the highest impact (expressed as BIF) in the 3R4F CS group at all timepoints (Figure 6(d)), whereas the relative biological impact factors (RBIFs—relative to the CS at month 7, BIF set as 100%) for the Test Red group remained close to baseline, indicating a significantly lower impact of e-vapor aerosol exposure. A weak but significant impact was observed in the Cessation and Switching groups at months 4 and 7 (Figure 6(d)). The overall BIF shown in Figure 6(d) illustrates the collective impact of 3R4F CS exposure on all the network models (CFA, CPR, CST, IPN, and TRA) tested and highlights the minimal biological impact of e-vapor aerosol exposure and significant recovery from 3R4F CS exposure in the lung upon Cessation and Switching.
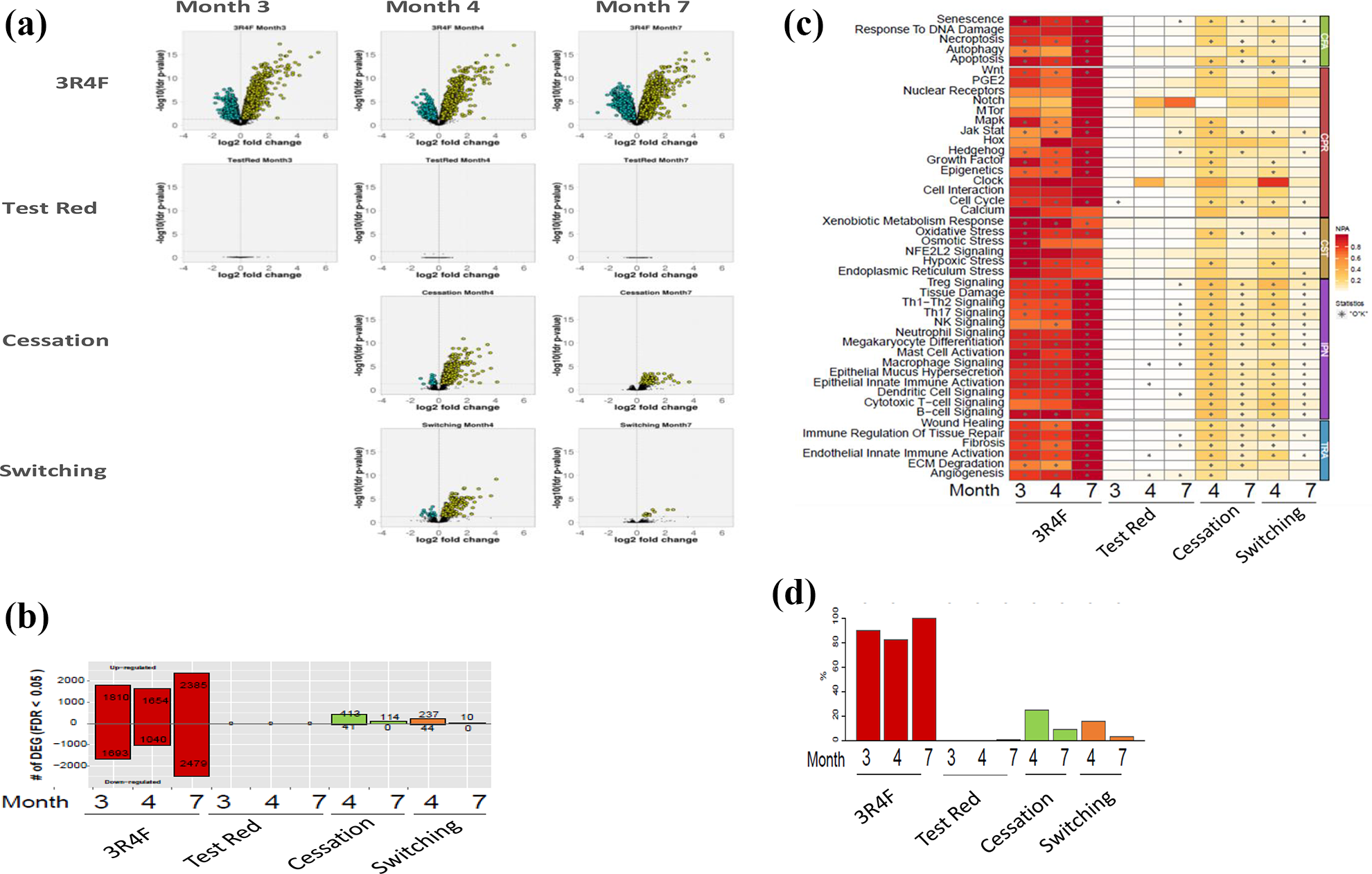
Lung transcriptomics. (a) Volcano plots for differentially expressed genes. For each gene, the gene expression change, calculated as the log2-fold change, is plotted on the x-axis, and the statistical significance, proportional to the negative log10-adjusted p-value, is plotted on the y-axis. Yellow and cyan dots highlight genes that are statistically significantly up- and downregulated, respectively. (b) Number of differentially expressed genes. (c) Heatmap of the network perturbation amplitude (NPA) scores across comparisons (normalized to the maximum NPA) for each network which is significantly impacted in at least one contrast. A network is considered as impacted if, in addition to the significance of the NPA score with respect to the experimental variation, the two companion statistics (O and K) derived to inform on the specificity of the NPA score with respect to the biology described in the network, are significant. *O and K statistic p-values < 0.05 and NPA significantly non-zero. (d) The BIF values are derived for each contrast (e.g., treatment vs. sham) from the aggregated NPA scores for networks applicable to the set of contrasts. The contrast showing the highest perturbation is set to 100%. CFA: cell fate; CPR: cell proliferation; CST: cell stress; IPN: inflammatory processes; TRA: tissue repair and angiogenesis.
Lung transcriptomics data were further analyzed to assess any potential impact of 3R4F CS and Test Red aerosol exposure on lipid metabolism. We looked at genes coding for surfactants (Sftpa1, Sftpb, Sftpc, and Sftpd), Prdx6 (a lysosomal-type phospholipase A2 for surfactant lipid), Abca3 (a transporter important for surfactant lipid secretion), and Slc34a2 (a marker of alveolar type II cells that secrete surfactant) (Supplementary Figure S4A). While most of these genes coding for surfactants showed a significant increase in expression, especially at month 7 in the 3R4F CS group, none showed significant changes in the Test Red group at any time point. Further gene set enrichment analysis on lipid metabolism-related gene sets (Supplementary Figure S4B) clearly indicated significant impact in the 3R4F CS groups at all time points, whereas the results showed no significant impact in lipid metabolism pathways in the Test Red, Switching, and Cessation groups relative to the Sham Control group.
The proteomic analysis of lung tissue showed a significant and increasing number (338, 348, and 612 proteins at 3, 4, and 7 months, respectively) of differentially regulated proteins in the 3R4F CS group, while the results showed no such changes (FDR-adjusted p-value < 0.05) in the Test Red group at any time point (Figure 7(a) and (b)). The Cessation and Switching groups showed significantly lower (compared to the 3R4F CS group) abundance of proteins at month 4 with further decline at month 7. The protein sets that were significantly affected by exposure were identified using the KEGG compendium. 55 Considering lung tissue specificity of Network models used for gene expression, which is not typical for KEGG compendium, only some pathways (example: Xenobiotic metabolism) overlapped between KEGG and network analyses.
Consistent with the above findings, the 3R4F CS exposures induced the most notable changes in multiple pathways including upregulation of proteins contributing to cell adhesion and antigen processing (Supplementary Figure S5; Figure 7(c)), whereas only minimal changes were observed at month 4 in Test Red group. The pattern of change was visually similar but at much lower intensity in the Switching or Cessation groups at month 4, which further declined to baseline at month 7 (Supplementary Figure S5). For some of the pathways, the enrichment analysis indicated an opposite directionality for the regulation of protein sets in response to Test Red exposure at 4 months compared with 3R4F exposure (Figure 7(c)).
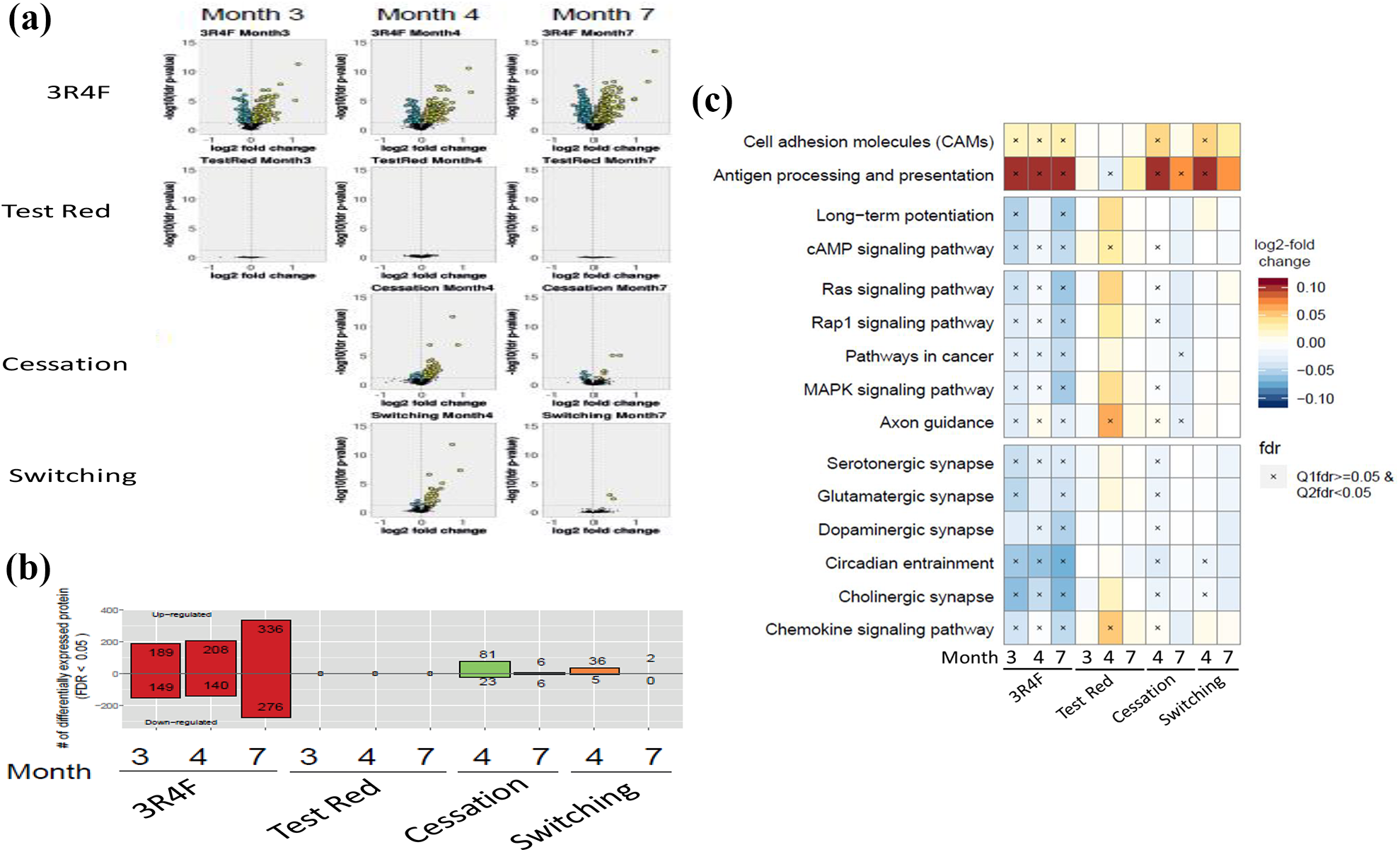
Lung proteomics. (a) Volcano plots of regulated proteins. For each protein, the change in protein expression, calculated as the log2-fold change, is plotted on the x-axis, and, the statistical significance, proportional to the negative log10-adjusted p-value, is plotted on the y-axis. Yellow and cyan dots highlight proteins that are statistically significantly up- and downregulated, respectively. (b) Bar graph of regulated proteins in each study group compared with Sham Control group. (c) Selected protein sets regulated in the 3R4F CS, Test Red, Cessation, and Switching groups in the lung. Significance marked as “+ indicates Q1 significance but not Q2 significance, × indicates Q2 but not Q1 significance, and * indicates Q1 and Q2 significance.”
Consistent with the gene expression data above, 3R4F CS exposure upregulated surfactant proteins (SFTPA1 and SFTPD) in the lung, while Test Red aerosol exposure showed no significant changes in any surfactant or surfactant-related proteins (Supplementary Figure S6). Altogether, lung transcriptomics and proteomics results showed that chronic CS exposures induced persistent molecular changes (e.g., inflammation, cell proliferation, tissue repair and stress, and increases in pulmonary surfactant expression) associated with chronic lung diseases. In contrast, chronic e-vapor (Test Red) aerosol exposures induced only minimal changes similar to baseline. Additionally, switching to e-vapor aerosol exposures after 3 months resulted in similar decreases in biological endpoints, indicative of significant recovery from 3R4F CS exposure that were comparable to cessation in the mouse lung.
In addition to lung tissue, respiratory nasal epithelium (RNE) tissues were also evaluated for gene expression. Transcriptomics analysis of data from RNE showed that the number of DEGs was near 10,000 in the 3R4F CS group at all timepoints, while there were very small changes (62 DEGs only at month 4) in the Test Red group (Supplementary Figure S7A). The Cessation and Switching groups showed similar DEGs in the RNE at month 4 compared to the 3R4F CS group, with a slight decrease at month 7 (Supplementary Figure S7A). The NPA analysis showed a strong perturbation of the majority of biological processes in the RNE of the 3R4F CS group, whereas the impact was nearly absent in the Test Red group at all time points (Supplementary Figure S7B). The Cessation and Switching groups showed notable BIFs above the Sham Control or Test Red groups, but they had significantly lower BIFs than the 3R4F CS group (Supplementary Figure S7C). Altogether, the RNE transcriptomics data showed the most changes in the 3R4F CS group and minimal changes in the Test Red group, with signs of significant but not complete recovery from effects of 3R4F CS exposure upon cessation and switching.
Discussion
The purpose of this study was to characterize the toxicological profile of an e-vapor product (Test Red) in terms of chronic lung diseases, specifically emphysematous changes, compared to combustible 3R4F CS. In addition, the study investigated the impact of complete switching to an e-vapor product (Test Red) in comparison to the filtered air (Cessation) after 3 months of exposure to 3R4F CS. Also, biochemical, functional, and structural changes depicting COPD have been reported in C57BL/6 mice with chronic exposure to cigarette smoke, 39 but there are no comparable mouse nose-only inhalation studies on e-vapor aerosols.
In this study, the e-vapor aerosol exposure groups were subjected to two-fold higher exposure (1100 µg/L) in terms of aerosol mass or TPM compared to the 3R4F CS group (550 µg/L). Despite higher TPM, the concentrations of all five measured carbonyls at nose-port were over 94% lower in Test Red aerosols, consistent with the vast majority of reports showing substantially low levels of carbonyls with e-vapor aerosol exposure. 60 –62 Even though few investigators reported e-cigarette generated carbonyls as a contributing factor for pulmonary inflammation, 63,64 it is increasingly evident that device control such as voltage/temperature of heating coil 65,66 can cause unintended (e.g., dry puffing) high levels of carbonyl generation. The nicotine concentration in the test atmosphere was selected based on the maximum tolerated exposure levels (∼500–600 µg/L TPM) in the C57BL/6 mouse model of nose-only smoke inhalation. 38 Mice exposed to 3R4F CS showed respiratory function depression with a decline in RR and therefore MV, while the Test Red group had comparable respiratory function with the Sham Control. This resulted in approximately three- to four-fold higher TIM in the Test Red group compared to the 3R4F CS group, resulting in higher levels of BOEs (nicotine and metabolites in plasma and urine). These findings aligned with the previous study showing substantially higher plasma levels of nicotine and cotinine in e-vapor aerosol exposed mice despite similar aerosol nicotine levels at nose-port. 31 It is also noted that plasma levels of nicotine in the Test Red group in this study (average ranging 292–487 ng/mL) were substantially higher (over 20-fold) than plasma levels typically reported in cigarette smokers or e-vapor product users. 67 Consistent with aerosol constituent results, BOEs for CO (blood COHb) and acrolein (urinary 3-HPMA) were at background levels in Test Red groups. This is in part due to simpler chemistry and substantially lower harmful constituents in e-vapor aerosols. 61
Lung function analysis demonstrated that the 3R4F CS group had the largest changes in lung function across all time points. These changes indicated that continuous exposure to 3R4F CS smoke led to physiological changes in pulmonary function which accompanied histological evidence of inflammation and emphysematous changes in the lung. These COPD-related functional changes (increased compliance and inspiratory capacity and hysteresis [area of PV loop] along with decreased quasi-static elastance) were already significant after 3 months of exposures in the 3R4F CS groups. These changes in lung function are one of the diagnostic parameters in COPD patients 68,69 and they are also associated with elastase-induced animal models of emphysema. 70 In contrast, lung function parameters for the Test Red group after 7 months of exposure overlapped with Sham Control indicating no emphysematous changes.
Accompanying the lung function changes in the 3R4F group were persistent pulmonary inflammation as reflected through an influx of inflammatory cells and increased inflammatory mediators/cytokines in BALF parameters. These changes often included increased absolute leukocyte counts as well as an overall increase in the number and/or activation status of multiple cell types such as CD3+ T cells, B-cells, macrophages (activated/M1 phenotype), dendritic cells, neutrophils, and EBM (Eosinophils/Basophils/Mast cells). The infiltrated immune cells, in particular activated alveolar M1 macrophages and neutrophils, secrete various pro-inflammatory cytokines, growth factors, and elastolytic enzymes including various MMPs, which can remodel lung parenchyma and trigger emphysematous changes. 71 The changes in BALF cytology and inflammatory mediators (such as TNFα, MIP1a, MIP1b, KC, IP 10, and pro-MMP-9) observed in the present study were in concurrence with previous chronic CS inhalation studies. 38,39,58 Also, the upregulation of pro-MMP-9 accompanied with overall increased MMP activity as shown in this study has been linked to alveolar destruction and emphysematous changes. 39,72 Altogether, the analyses of infiltrated cells, inflammatory mediators, and MMP activity confirm persistent inflammation, and in part explain emphysematous changes in the lungs of 3R4F CS-exposed mice. In contrast, most BALF parameters for the Test Red group after 7 months of exposure were comparable to the background levels in Sham Controls and did not elicit changes indicative of pulmonary inflammation.
Biomarkers of effects observed in BALF parameters support the definitive histopathological findings in the lung of the 3R4F CS group. Up to 7 months of 3R4F CS exposures induced pulmonary inflammation (increased number of alveolar macrophages, interstitial mixed cell infiltrates, perivascular lymphocytic infiltrates, and bronchiolar epithelial cell hyperplasia; all time points), and dilatation of the terminal/respiratory bronchioles and adjacent alveoli (emphysematous changes at month 7 and recovery time points). These findings correlated with the inflammatory response noted in the BALF (cellular infiltration, inflammatory mediators, and overall increases in MMP activity contributing to emphysematous changes) and likely contribute to the changes in lung function and increased lung weights. In contrast, 7 months of exposure to Test Red aerosol did not elicit significant histopathological changes in lungs and were comparable to Sham Control.
Additional histopathological findings related to Test Red aerosol and/or 3R4F CS exposure were limited to upper respiratory tract tissues (nose, larynx, and trachea). Most of these findings were often seen, or of increased incidence and/or severity, in the 3R4F CS group. Nasal respiratory epithelial hyperplasia (at month 3) and intracytoplasmic epithelial inclusions (at all time points) were also observed in the Test Red group, likely related to irritation from hygroscopic (PG, VG) aerosol exposures. 34,36 While similar signs of respiratory irritation were observed in a clinical setting, 22 further investigation is warranted to investigate the chronic impact of e-vapor aerosols on nasal lesions (respiratory epithelial hyperplasia and intracytoplasmic epithelial inclusion).
In the larynx, squamous metaplasia was seen in both 3R4F CS and Test Red groups at months 3, 4, and 7 with complete reversal only in the Test Red group after 9 weeks of recovery. The Test Red aerosol exposure related nasal epithelial changes and squamous metaplasia of the larynx in this study concur with previous rodent studies showing similar upper respiratory tract changes with exposure to PG. 36,73 Altogether, histopathological evaluations indicated that Test Red aerosol exposure induced fewer histopathological changes in respiratory tract tissues with no indication of COPD-related changes as typically seen with 3R4F CS exposure at month 7.
To obtain mechanistic insights on the underlying molecular events, the lungs were evaluated by transcriptomics and proteomics analyses at each time point. The lung transcriptomics analysis demonstrated robust upregulation of pathways contributing to pulmonary inflammation in response to 3R4F CS exposure; in contrast, Test Red aerosol exposure elicited only minimal changes in inflammatory pathways. Further evaluations of cell stress networks showed upregulated xenobiotic metabolism, oxidative, osmotic, and hypoxic stress pathways in 3R4F CS group, whereas no such changes were evident in the Test Red group. Oxidative stress is well recognized as one of key contributing factors in CS-induced pulmonary diseases including COPD. 74,75 A previous meta-analysis showed a large increase in 8-iso-prostaglandin F2α levels, a reliable marker for oxidative stress, in various diseases, including respiratory tract disorders. 76 Also, upregulation in xenobiotic metabolism can lead to more active metabolic intermediates which can further increase oxidative stress and emphysematous changes. 77 Oxidative stress can in turn lead to upregulation of nuclear factor-κB, inducing release of pro-inflammatory cytokines 78,79 which can further augment pulmonary inflammation. These collective findings provide reference points of underlying mechanistic changes at the molecular level for the CS-induced pulmonary inflammation and emphysematous changes. In contrast, the Test Red group showed minimal changes in the cellular perturbation network, as previously observed in a shorter (3-week) study. 31 Similarly, in a 90-day nose-only inhalation study in which rats were exposed to PG/VG mixtures, with and without nicotine, 34 showed only limited gene expression changes in the lungs.
Recently, Madison et al. 28 reported that e-vapor aerosols could impair metabolism of pulmonary surfactants, which has been associated with accumulation of intracellular lipids and deposition of excess lipids in the alveolar space. Accordingly, we performed the secondary analysis of lung transcriptomics data focusing on lipid metabolism. Many genes coding for surfactants (Sftpa1, Sftpd, and Sftpc) were significantly upregulated in the 3R4F CS group, especially at month 7, whereas none of these showed significant changes in the Test Red group. Additional gene set enrichment analysis showed no significant impact of Test Red exposure on lipid metabolism pathways. Subsequent proteomics analysis of surfactant proteins (SFTPB, SFTPA1, SFTPC, and SFTPD) further confirmed gene expression findings.
The above results clearly demonstrated minimal impact of e-vapor aerosol compared to 3R4F CS exposures on systemic and COPD-related endpoints. Complete switching to e-vapor aerosol exposure after initial exposure to 3R4F CS for 3 months showed a clear and consistent trend of reversal based on in-life, functional, structural, and mechanistic endpoints. Some parameters in Switching and Cessation groups showed complete reversal compared to the 3R4F group, with changes similar to Sham Control (e.g., mean body weights, PV curves, relative organ weights) at month 7 (approximately 4 months after switching or cessation). There were also parameters showing partial recovery indicating residual effects of 3R4F CS exposure (e.g., lung histopathology, BALF parameters, transcriptomics and proteomics, clinical observations), likely requiring longer than 4 months to revert to near baseline. These findings showing switching being comparable to cessation on COPD endpoints were similar with another study with heated tobacco products. 39
In conclusion, results from this study demonstrate that in comparison with CS, Test Red aerosols induce substantially lower biological responses, including pulmonary inflammation and emphysematous changes, and complete switching from CS to e-vapor products could significantly reduce biological changes associated with CS in the C57BL/6 mouse model of smoking-related COPD. The observations from this study suggest that pulmonary effects from inhalation of e-vapor aerosol can be expected to be significantly lower compared to continued smoking.
Supplemental material
Supplemental Material, sj-pdf-1-tor-10.1177_2397847321995875 - A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products
Supplemental Material, sj-pdf-1-tor-10.1177_2397847321995875 for A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products by Ashutosh Kumar, Ulrike Kogel, Marja Talikka, Celine Merg, Emmanuel Guedj, Yang Xiang, Athanasios Kondylis, Bjoern Titz, Nikolai V Ivanov, Julia Hoeng, Manuel Peitsch, Joshua Allen, Amit Gupta, Anthony Skowronek and K Monica Lee in Toxicology Research and Application
Supplemental material
Supplemental Material, sj-pdf-2-tor-10.1177_2397847321995875 - A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products
Supplemental Material, sj-pdf-2-tor-10.1177_2397847321995875 for A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products by Ashutosh Kumar, Ulrike Kogel, Marja Talikka, Celine Merg, Emmanuel Guedj, Yang Xiang, Athanasios Kondylis, Bjoern Titz, Nikolai V Ivanov, Julia Hoeng, Manuel Peitsch, Joshua Allen, Amit Gupta, Anthony Skowronek and K Monica Lee in Toxicology Research and Application
Supplemental material
Supplemental Material, sj-pdf-3-tor-10.1177_2397847321995875 - A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products
Supplemental Material, sj-pdf-3-tor-10.1177_2397847321995875 for A 7-month inhalation toxicology study in C57BL/6 mice demonstrates reduced pulmonary inflammation and emphysematous changes following smoking cessation or switching to e-vapor products by Ashutosh Kumar, Ulrike Kogel, Marja Talikka, Celine Merg, Emmanuel Guedj, Yang Xiang, Athanasios Kondylis, Bjoern Titz, Nikolai V Ivanov, Julia Hoeng, Manuel Peitsch, Joshua Allen, Amit Gupta, Anthony Skowronek and K Monica Lee in Toxicology Research and Application
Footnotes
Acknowledgments
The authors acknowledge Sam Harbo (while employed at Battelle) for his valuable contribution in conducting the study, Bill Gardner (Altria) and Patrick Vanscheeuwijck (Phillip Morris International) for their technical feedback. The authors would like to thank PMI leads: Sam Ansari and Edouard Dargaud for the sample management; Keyur Trivedi and Petros Kanellos (contracted) for lung slicing; Remi Dulize, David Bornard, Pauline Duvoisin for target preparation and chip hybridization; Dariusz Peric, Mehdi Auberson for the RNA isolation; Maica Corciulo for MAP analysis; Manuel Hernandez, Fabio Maranzano, Catherine Nury for iTRAQ proteomics experiments; Thomas Schneider and Edina Kishazi for PRM proteomics experiments and MMP activity assays.
Declaration of conflicting interests
The author(s) declare that there is no conflict of interest. The authors Ashutosh Kumar and K Monica Lee are employees of Altria Client Services LLC. Ulrike Kogel, Marja Talikka, Celine Merg, Emmanuel Guedj, Yang Xiang, Athanasios Kondylis, Bjoern Titz, Nikolai V Ivanov, Julia Hoeng, and Manuel Peitsch are employees of Philip Morris International. Joshua Allen, Amit Gupta, and Anthony Skowronek are employees of Battelle.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by Altria and in part by Philip Morris International (for the analysis and interpretation of omics endpoints).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
