Abstract
Given the complexity of inhaled substances, the aerosol exposure environment has seen diversification and development of setups in conjunction with the evolving in vitro toxicology space. Each laboratory uses its in vitro exposure system differently (different protocols, adaptations, and biological analysis). Unfortunately, as systems diversify, so does the complexity of comparing multiple systems in a “standardized” manner. As yet, no one has compared simply whether these diverse systems can all generate a consistent aerosol stream, which is paramount prior to transit and exposure. This study has compared, at source, aerosol generation (using nicotine as an exposure marker) in nine in vitro whole-aerosol exposure setups (seven different systems) across five distinct geographically independent locations, including the UK, the USA, Switzerland, Germany, and Japan. The results demonstrate that, despite system-wide differences (adaptations, nuances, and application), these systems—when appropriately maintained and used under a prescribed set of established conditions can all generate a consistent and statistically comparable aerosol stream. These data will be invaluable for new researchers and established laboratories, so they may benchmark against this study. Finally, this interlaboratory comparison combined with the wealth of transit and exposure interface data, may help the environment move towards a truly validated and consistent approach to aerosol exposure. Such an approach could be replicated for other aerosolized products, such as e-cigarettes and heated tobacco products.
Keywords
Introduction
Aerosol exposure systems have become commonplace in modern in vitro inhalation toxicology programs and are playing a more important role in bridging in vivo and clinical studies. 1 As their importance increases, so does innovation, which, in turn, drives use and application. This is the current environment where systems are being adapted and deployed for testing a variety of aerosols, including cigarette smoke, electronic cigarettes and heated tobacco product vapor, inhaled pharmaceuticals and environmental exposure. As such, new systems are being developed and old systems adapted for use with these new product categories. Furthermore, data generated on these systems are being used to support product development, as part of regulatory dossiers, for cosmetic claims, and for claim substantiation. 2 –5 Never has it been more important to standardize in vitro aerosol approaches and consider the validation of these systems. Unfortunately, the in vitro aerosol exposure system environment is more diverse than it has ever been. Test products are driving system change and adaptations; but, as these products continue to be innovated, there is increased demand for higher throughput and higher content biological analysis in line with 21st century toxicology approaches. 6 –9 Therefore, each independent setup is likely to be very different from the next, depending on its application.
Irrespective of the system, cigarette smoke in vitro aerosol exposure platforms have three principal components in common. The first is aerosol generation. This is the point at which the aerosol first enters the system. If assessing a consumable product, the aerosol is puffed to a set regimen or standard under standard smoking conditions. For tobacco products (cigarettes), internationally established smoking standards are available to ensure consistent aerosol generation. 10 –13 For e-cigarettes, although CORESTA (Cooperation Centre for Scientific Research Relative to Tobacco) has released a standard, 14 many studies still use their own individual puffing regimens. 15,16 For HTPs, no standard currently exists, but one is currently being developed by CORESTA. Once the aerosol is generated, usually by a piston/syringe, it transfers through the system to be diluted (where dilutions are applied). It is at this stage, post-syringe, that these exposure systems start to diverge. The type of tubing, diameter and length of tubing, dilution principle, and subsampling of the aerosol significantly affect the delivered dose. The final aspect which all systems share is the exposure chamber/module, where cells are housed at the exposure interface. The type of exposure chamber and its individual exposure characteristics and how cells are maintained within the module (for example, at the air–liquid interface, air–agar interface, or in a 96-well plate) all affect the deposition efficiency and, therefore, delivered dose.
Several comprehensive reviews are available on whole-aerosol exposure systems. 17,18 To date, most of the system characterization studies have focused on these three principal components, as they quantify and qualify how individual aerosols pass through the exposure setup and what reaches the exposure interface, which ultimately defines the delivered dose. Few studies have compared within systems and fewer still have compared across systems. Where interlaboratory approaches have been investigated, they have almost exclusively been used to assess across a single platform type; very few (if any) studies have ventured into comparing within and across systems and laboratories because of the difficulty in standardizing approaches and the challenges of assessing diverging setups, as discussed above. Table 1 gives a snapshot of some of the studies conducted.
In vitro aerosol studies characterizing dose within and comparing between and across systems.

As shown in Table 1, most studies have attempted to characterize their individual and unique setups. Where cross-system comparisons have been made, they have been conducted in the same laboratory and not across laboratories in a true interlaboratory approach. If these systems are to be characterized in terms of their setups against other systems in the exposure environment and if they are to be validated as an approach, more interlaboratory studies need to be conducted. 1 Owing to the lack of standardized approaches (i.e., where to conduct the assessments within the system and what to assess), given the differences among the systems, there has been no true interlaboratory study to date that has performed comparison across different systems.
This study has been specifically designed to address this issue and be the first to assess these systems in a true interlaboratory cross-platform approach. In order to do this, the first point of aerosol generation was assessed, which also happened to be a common denominator among systems and one of the three principal components linking all platforms together. From aerosol generation, the systems differ, as aerosol transit, dilution, and delivery characteristics significantly affect the delivered dose. To standardize the approach, reference 3R4F cigarette smoke was exclusively used alongside internationally recognized smoking regimens. The aim of the study was to establish a base dataset to assess whether seven distinctly different in vitro aerosol exposure platforms can generate a cigarette smoke aerosol stream in a consistent manner in line with international smoking standards yields.
Materials and methods
Study design
The study was designed to assess only aerosol generation from multiple exposure systems rather than any of the downstream dilution and delivery processes. The goal was to assess whether these diverging systems could at least be compared from the very first point of aerosol generation, rather than investigating different transit lengths, dilution principles, and exposure characteristics within the module, all of which play an important role in the actual delivered dose and, ultimately, biological response. By comparing the systems at the first logical point of aerosol generation, researchers can investigate one of the confounding variables for in vitro aerosol delivery. This study focuses purely on reference cigarette smoke to compare systems. This study assumes that each system is well maintained (cleaned and appropriately serviced) and calibrated before data generation. Puff volumes and leak tests were performed on each machine as standard before any data were generated.
Table 2 shows the laboratories involved, their systems, serial numbers for traceability, and geographical location. This study involved nine in vitro exposure systems—seven different systems split over five distinct geographical locations (laboratories), all with their own individual exposure characteristics.
Exposure systems, models, locations, serial numbers, CFP position, and tubing lengths.
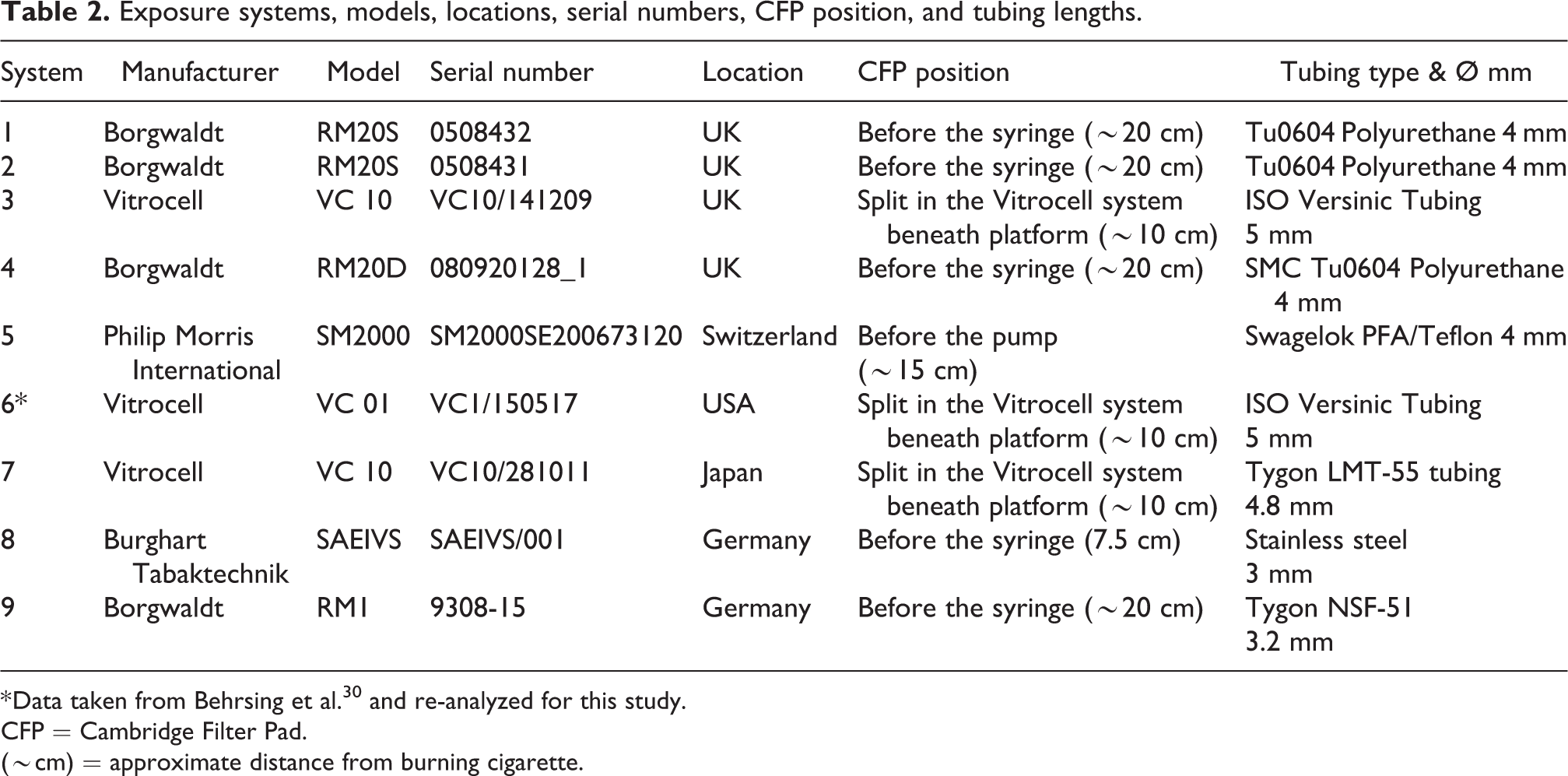
* Data taken from Behrsing et al. 30 and re-analyzed for this study.
CFP = Cambridge Filter Pad.
(∼cm) = approximate distance from burning cigarette.
Smoking regimens/sample generation
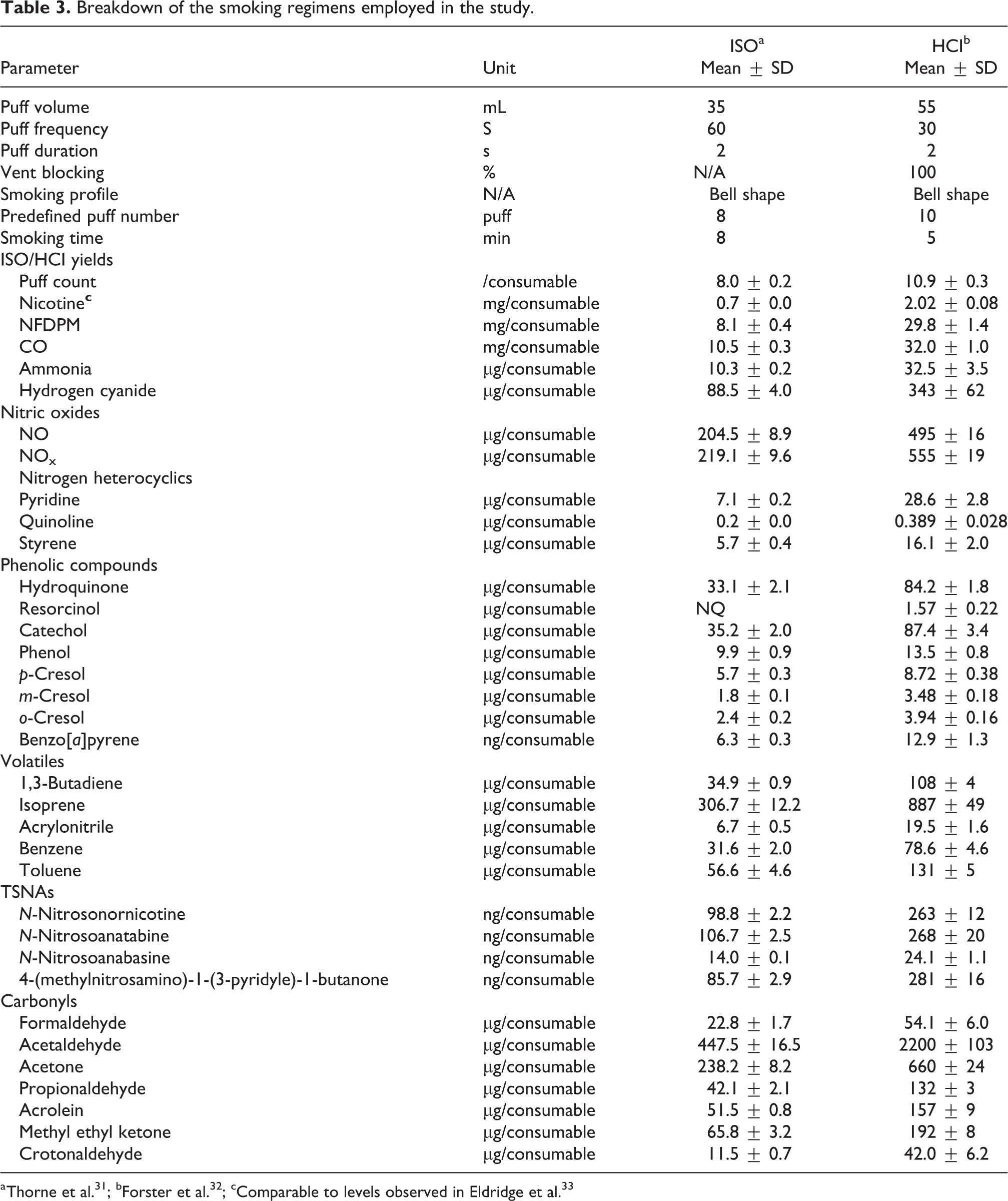
3R4F reference cigarettes were used exclusively and obtained from the University of Kentucky, Lexington, KY, USA. The cigarettes were conditioned in accordance with ISO guidelines (48 h at 22 ± 1°C and 60 ± 3% relative humidity) prior to smoking. ISO and Health Canada Intense (HCI) smoking regimens were employed for standard and intense use to achieve two distinct smoking profiles. 10,11 The two regimens differ in puff volume, puff frequency, and vent blocking, with HCI being the more intense regimen, delivering a bigger puff, more frequently, and with the vent holes blocked. A comparison of the two smoking regimens, the parameters used in the study, and a summary of the delivered analytical yields to demonstrate differences is shown in Table 3.
Breakdown of the smoking regimens employed in the study.
Particulate collection
Puff-by-puff profiles were generated for each system and regimen by capturing the total particulate matter (TPM) on a Cambridge filter pad (CFP) between puffs (30- to 60-s intervals). Under ISO smoking conditions, eight puffs were taken per reference cigarette, and the pads changed every 60 s between puffs. Under HCI smoking conditions, 10 puffs were taken per reference cigarette, and the pads were changed every 30 s between puff intervals. This procedure was repeated on three independent occasions per regimen per system.
For all systems, TPM was collected on a CFP installed between the burning cigarette and the syringe. Owing to system differences/characteristics and the practicality of installing a holder and line-split that can be easily accessed, the CFP holder was installed in a different place in each system. In all instances, the CFP holder was installed as close to the aerosol source as practicably possible and always before the piston or syringe in order to capture maximum aerosol delivery and minimize losses. In some systems, dilution occurs in the piston, which often involves dilution of the aerosol stream and deliberate loss of some aerosol to meet the required dilution steps. Therefore, capturing the aerosol before this step is fundamental to assessing the systems in a robust and consistent manner. Usually, where the aerosol enters the piston is the point where each exposure system starts to differ. Because of the system differences, no single standard position could be identified for every system; therefore, the CFPs were installed in a variety of positions, all as close to the burning cigarette as practicably feasible and always pre-piston.
Exposure systems
Borgwaldt RM20S
The Borgwaldt RM20S and associated ALI exposure chamber was developed by Borgwaldt KC (Borgwaldt KC GmbH, Hamburg, Germany) in conjunction with British American Tobacco. The RM20S is a rotary-based smoking machine. The machine allows either eight different cigarettes to be assessed at one dilution or one cigarette at up to eight different dilutions. Smoke is generated via a syringe, which draws a puff from the cigarette first, followed sequentially by a puff of filtered air to create the required dilution, expressed as the ratio of smoke in air (1: X, volume: volume). For larger dilutions, the puff is drawn and diluted and then partially exhausted into the environment before the next dilution step is activated. This multistep process produces a ratio in the range of 1:3 to 1:600,000 (aerosol: air), which corresponds to 33–0.00017% aerosol in air (v/v). Diluted smoke is exhausted from the syringe at 0.8 L/min into the exposure chamber. The exposure chamber is simplistic in design and comprises a top and bottom made of autoclavable Perspex and stainless-steel fixings. The chamber is designed to allow cell cultures to be exposed basally to fresh medium and apically to aerosol (from a smoke generator) at the ALI. Aerosol is exhausted from the smoke dilutor through a central gas inlet and distributed around the chamber with the aid of a smoke distribution plate. Smoke is displaced and exhausted from the chamber via the positive pressure created by the arrival of the following puff. Cell cultures are supported within the exposure chamber on permeable membranes, facilitating ALI exposure. Membrane sizes can be adjusted within the chamber, so that either three 24-mm, six 12-mm, or eight 6.5-mm permeable membranes or a combination of all three can be exposed to diluted tobacco smoke. The exposure chamber seals via a rubber “O” ring and three quick release locking nuts, which allows the culture inserts to remain under sterile conditions but ensures quick access (Figure 1).

British American Tobacco’s standard exposure chamber and the Borgwaldt RM20S smoking machine. (i) Cigarette smoke generator. (ii) Original four-syringe system. (iii) Four-syringe extension. (iv) Air-flow controller. (v) Cell culture media maintained at 37°C. (a) British American Tobacco’s exposure chamber housed at 37°C, attached to the smoke diluter and culture medium reservoir. 20
Borgwaldt RM20D
Borgwaldt RM20D is a rotary 20-port smoke machine for analytical purposes. It was designed to generate TPM to be captured on a CFP and has more recently been adapted to work as an aerosol generator and has been coupled with the BAT smoke exposure chamber for in vitro experimentation. RM20D has no smoke dilution capability. Aerosol is generated and delivered directly to the exposure chamber, maximizing dose and delivery. Like other smoke machines, RM20D is programmable and can perform smoking to meet a number of recognized smoking regimens. It can be coupled with a variety of trapping techniques, which makes it a versatile smoke engine. Aerosol is generated through a single piston or syringe.
Borgwaldt RM1
Borgwaldt RM1 (later renamed LM1) was developed by Borgwaldt (Borgwaldt KC GmbH) and is a linear one-port mechanical syringe-based smoking machine. RM1 was developed to be a versatile cigarette smoke machine that is customizable and adaptable for a variety of needs. It is programmable with a variety of smoking regimens to meet researchers’ needs.
Vitrocell VC 01/10
The Vitrocell® exposure system (Vitrocell® Systems, Waldkirch, Germany) consists of an aerosol generator (normally termed a smoking robot) combined with an aerosol dilution system and ALI exposure module. VC 10 and VC 01 both have a syringe, which draws the puff and transfers the aerosol to an independent continuous flow dilution system. Different smoke concentrations are achieved by increasing or decreasing the diluting air flow (L/min), typically between 0.25 and 12 L/min. In addition to a diluting air flow, a vacuum subsamples aerosol (via negative pressure) from the dilution system into the module, which docks directly to it. The vacuum flow rate can be set anywhere between 1 and 200 mL/min, depending on the amount of aerosol required to be delivered to the cell system. Diluting airflow rates within this system are usually maintained by mass flow controllers (Analyt-MTC GmbH, Mülheim, Germany) and vacuum rates set by mass flow meters (Analyt-MTC GmbH, Mülheim, Germany). The Vitrocell system can be used with a variety of Vitrocell-based modules. There is little difference between VC 10 and VC 01 other than throughput, with the VC 10 having ten ports and the VC 01 having only one (Figure 2).
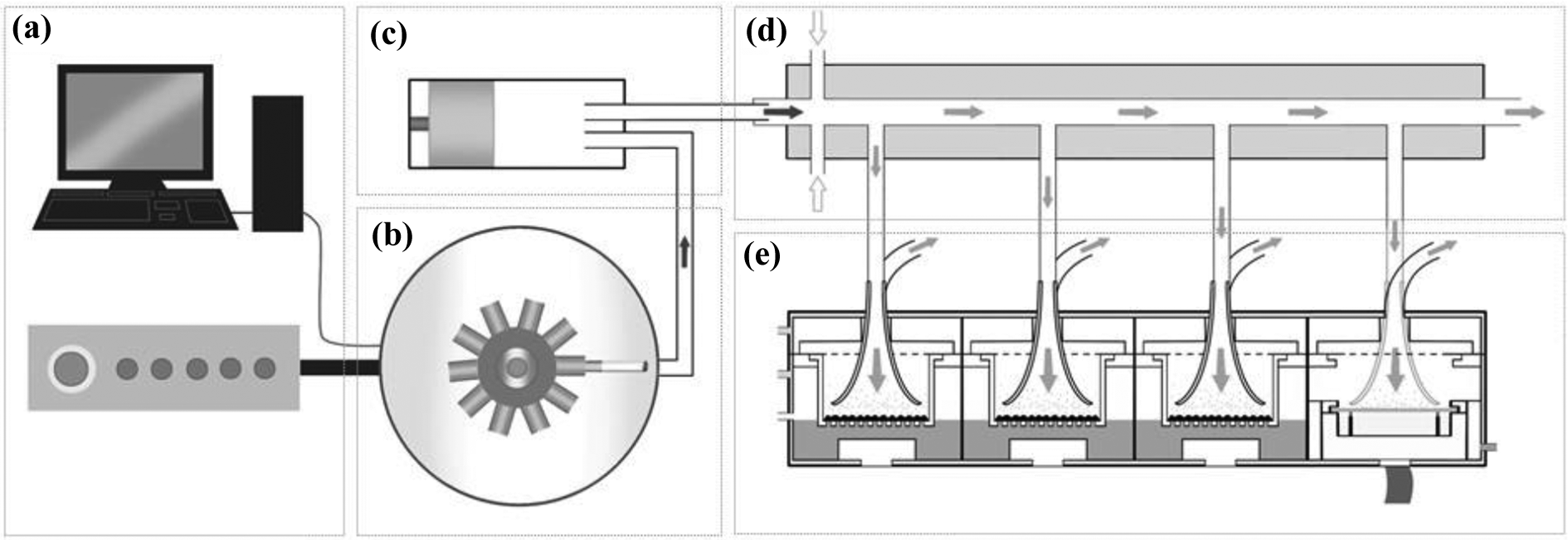
Schematic representation of the major components of Vitrocell VC 10. (a) Computer, software controller, and air-flow controller, which determines the smoking parameters and key machine settings. (b) Smoking head, where cigarettes are smoked, enclosed within an extraction ventilation hood. (c) Piston/syringe, which draws and delivers a puff of mainstream cigarette smoke to the smoke dilution system. (d) Dilution, transit, and delivery of whole smoke occurs in the dilution bar. Continuous diluting air is added perpendicular to the mainstream smoke in the range of 0.2–12 L/min and administered to the dilution bar through smoke air jets. Flow within the dilution system continuously transits through to exhaust. (e)Smoke exposure module, which holds Transwells or agar plates, which are maintained at the ALI or air–agar interface (AAI). Shown here is the Vitrocell® 6/4 CF Stainless Steel module; but other Vitrocell® module variations exist and can be coupled to the exposure system to tailor the experimental setup. Smoke is sampled from the dilution system into the exposure module via negative pressure applied through a vacuum pump (this vacuum rate can be tailored depending on preference and the exposure setup). Smoke is distributed within the exposure module via the smoke “trumpet” inlets and, because of the linear configuration, each culture insert is isolated and receives an independent sample of smoke from the dilution system. 21
Smoke aerosol exposure in vitro system
The Smoke Aerosol Exposure In vitro System (SAEIVS) was designed by Imperial Brands in collaboration with Burghart Tabaktechnik (Wedel, Germany) as a versatile, high-throughput way to expose cells in 96- and 24-multiwell plates (MWP) with transwells at the ALI. The SAEIVS is a five-port smoking machine directly connected to two independent exposure devices. A computer-controlled smoke dilution system allows transfer of undiluted or diluted smoke/aerosol to the exposure devices. The rapid mixing and dilution process uses an exact predefined volume of humidified and filtered air and is performed in a closed system. The two exposure chambers have separate independent dilution systems to allow parallel exposure to the same smoke/aerosol and its gas–vapor phase at different dilution levels. Smoke/aerosol is rapidly delivered to the cells (in about 10 s) to prevent ageing effects. Each well of the plate are provided with an individual smoke inlet and outlet ducts for smoke/aerosol exposure and extraction at the end of the puff. The use of a blanking plate in each exposure chamber enables puff-by-puff dose–response determination for different rows of wells. Furthermore, the separate chambers enable testing of the same product in two different in vitro assays in different MWPs at the same time (Figure 3).
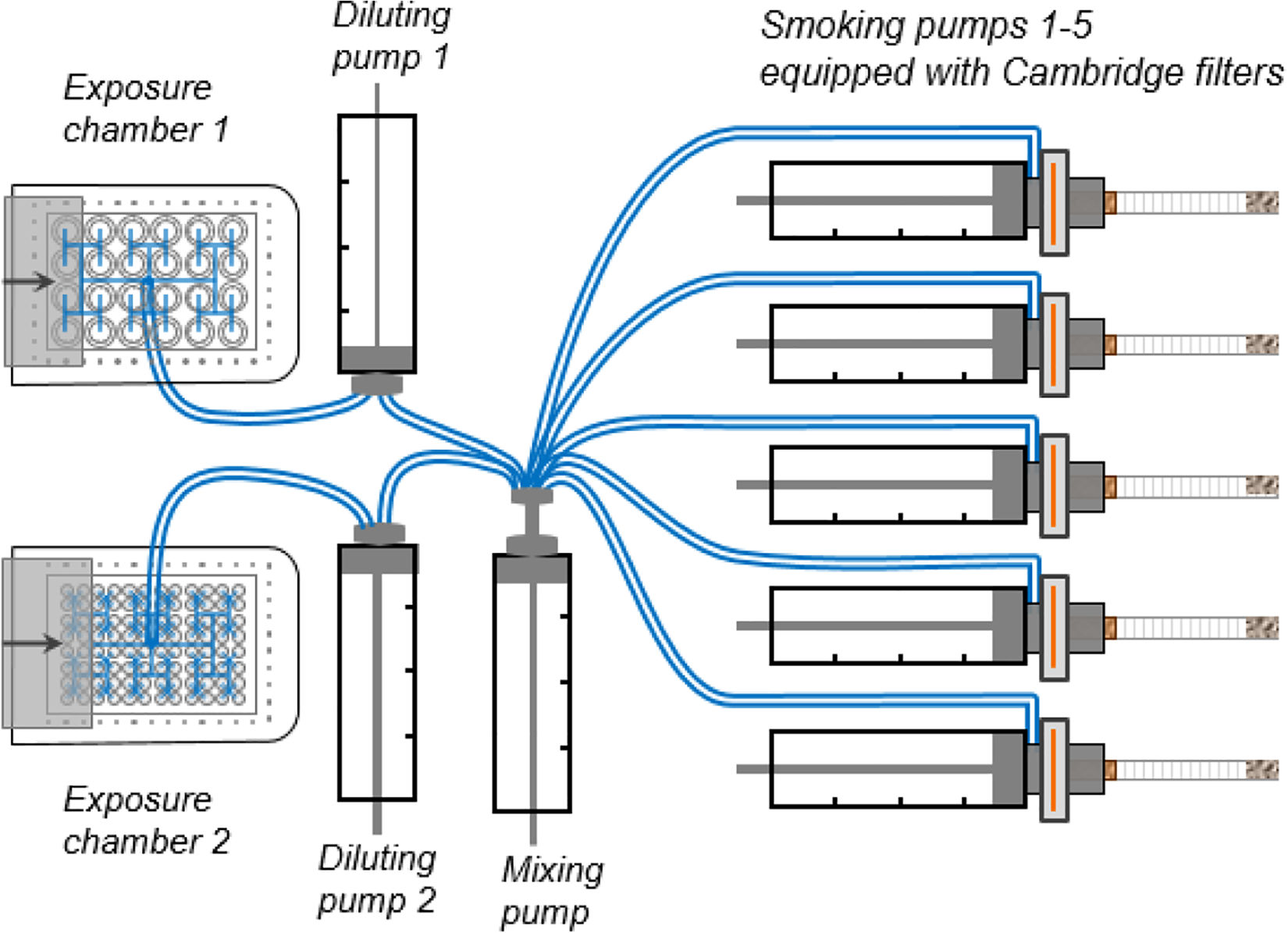
Diagram of the SAEIVS, showing independent dilution pumps, which enables independent dilution in both exposure chambers. A sliding plate enables serial puff exposure to the different rows of wells. 29
Philip Morris SM2000
SM2000 is 30-port rotary-based dual-syringe smoking machine developed and manufactured by PMI (SM 2000, Philip Morris Intl.). It is capable of generating aerosol to meet all standard smoking regimens, including bell and square wave puff profiles. The two pumps in the dual-pump system work in tandem to ensure that a near continuous aerosol stream is generated. In this capacity, while one pump is exhausting the aerosol, the second pump is already taking the next puff. The two pumps are connected by a controller and valve system. The turning of the valve is the only process that interrupts the continuous stream of aerosol. SM2000 offers the flexibility of smoking regimen and controllable exhaust airflows. This system is designed solely for reproducible generation of aerosols. The system itself does not dilute the aerosol stream, leaving the syringes to purely deliver the aerosol in the most efficient manner. In order to deliver the resulting aerosol, the aerosol generator must be coupled with a dilution system (where dilution is required) and exposure modules. The advantage of this setup is that it can be tailored to work alongside any dilution system and exposure module configuration. In its existing configuration, SM2000 is coupled to a Vitrocell® 24/48 smoke dilution and distribution system consisting of an exposure base module and a climatic chamber in which the former two are contained. Smoke passes into the Vitrocell® 24/48 whole-smoke exposure system via the dilution/distribution system. The smoke passing through the dilution system is sampled into the exposure trumpets by negative pressure and streamed over the cell cultures present in the exposure chambers, at a low volume flow rate of 2 mL/min. Eight access points for dilution air allow dilution of smoke in the dilution/distribution system serially and group the 48 exposure chambers into eight groups of six replica chambers. Under normal operation conditions, only seven dilutions are applied, and the eight-dilution row is used for control exposure to clean air. The temperature and relative humidity during the exposure are controlled by the climatic chamber built in around the dilution/distribution system and the exposure base module, through a heating water circuit incorporated into the dilution/distribution system and a humidification system, which maintains stable humidity of the dilution air used for exposure (Figure 4).
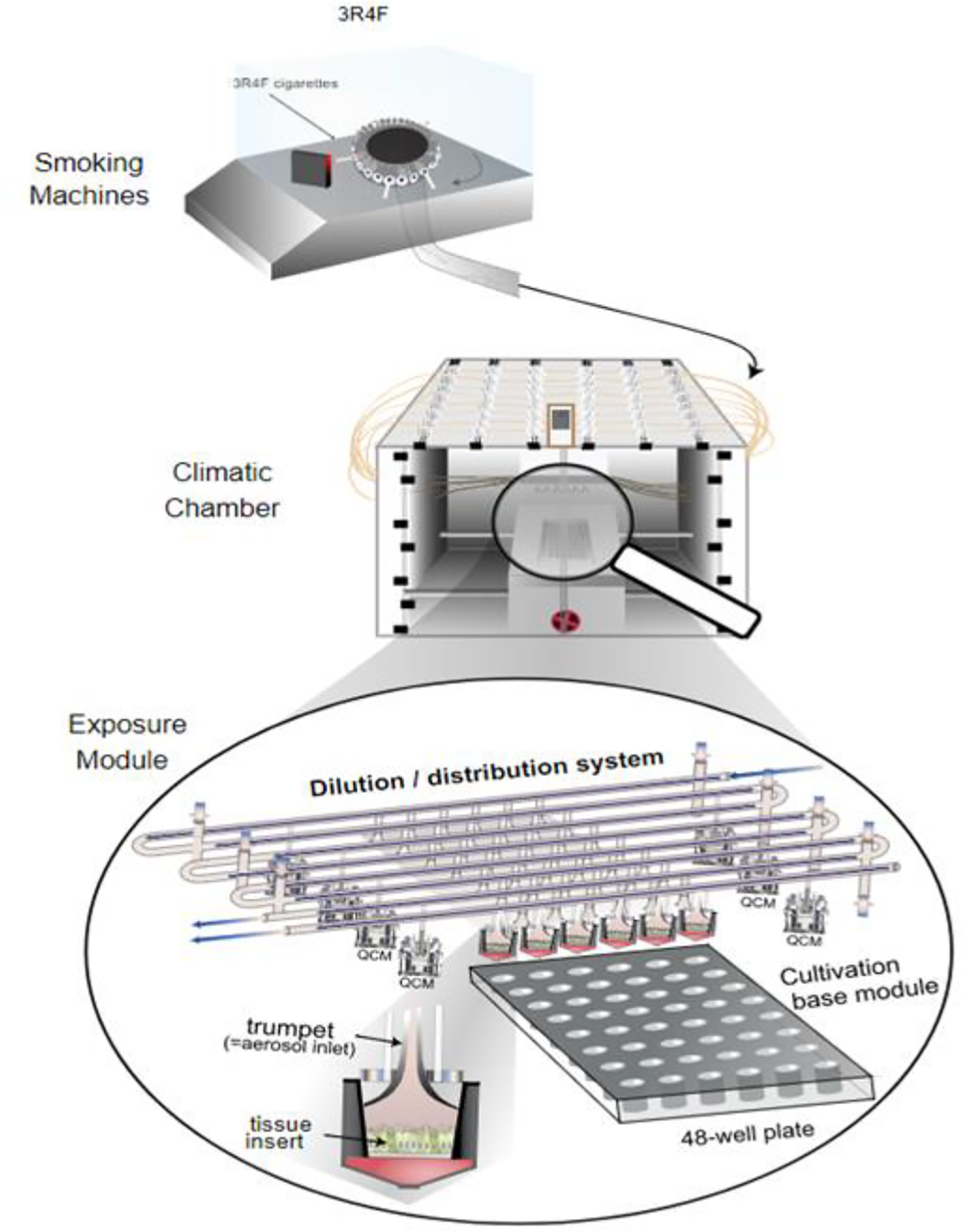
Schematic representation of the Vitrocell® 24/48 whole-smoke exposure system with the smoking machine. 23
Nicotine quantification
Nicotine was analyzed in the UK and/or Germany laboratories. Extracted samples 2 mL of methanol containing pad collected TPM was shipped under ambient conditions in amber vials. Ambient (amber vials) conditions were selected for shipment as a previous nicotine stability data indicated that samples were stable up to 60 days under these conditions. 34 Samples were analyzed with a week of receipt of sample, well within the required 60-day cut off as previously established. Nicotine was captured on CFPs—which were swapped between puffs (60 s for ISO and 30 s for HCI)—and extracted in 20 mL HPLC-grade methanol. The samples were rocked for 20 min at 120 rpm. Following extraction, 1 mL of each extract was transferred to glass gas chromatography (GC) vials and spiked with 10 µL of d4-nicotine standard. The solvent was evaporated in a concentrator (rotary, heated (<30°C) and vacuumed) for approximately 2 h to avoid thermal degradation of the nicotine. The concentrated samples were then resuspended in 5% acetonitrile in water. Nicotine was quantified by ultra-performance liquid chromatography–tandem mass spectrometry (UPLC–MS/MS) against the d4-nicotine standard peak. Where samples were shipped and analyzed, they were vortexed thoroughly and spiked with d4-standard before condensation, resuspension, and quantification. Nicotine quantification was performed by using Genedata, which is a web-based tool for quantifying proteins. It is used to eliminate variance that might occur when samples are quantified “manually” by using Sciex’s Analyst software, which require the operator to select peaks by eye.
Data presentation and statistical analysis
Raw nicotine values were quantified in ng/mL and converted to mg/puff. The raw value was first multiplied by the total extraction volume of solvent per pad (20 mL) and then divided by by 1,000,000 to convert from ng to mg. Minitab 18 was used to graph/present and analyze the results. The figures are split between the ISO and HCI smoking regimens, with all systems clearly denoted. Puff-by-puff profiles are also shown, along with average data for each system/smoke regimen. Global analysis of variance (ANOVA) was used to compare the systems to determine statistical differences.
Results
First comparisons were made by using the ISO smoking condition. The pads were changed between every puff, and a total of eight puffs were collected per cigarette on three independent occasions per system. The data for each system were compared on mg/cig and mg/puff bases. Puff profiles were generated for each system under the prescribed smoking regimen for one reference (3R4F) cigarette. Each system was compared on an mg/cig basis against a reference value obtained for a 3R4F cigarette smoked under ISO conditions, 33 by using a generally accepted limit of 10%, which indicates that the equipment is fit for purpose. 35
In the first instance, the data from each system were compared with an ISO reference value of 0.73 ± 0.02 mg/cig ±10%. In this comparison, all systems were approximately within or equal to the 10% established limits of the machine or measurement error. This demonstrated that, irrespective of where the CFP was installed and the nicotine was captured, all systems were performing as expected when compared with an ISO reference value for 3R4F cigarette smoke. Some variation was observed, but this was to be expected, given the natural variability of the product and the sampling position for each system. In order to get a better idea of the source of variation, the systems were then compared on an average mg/puff basis. In this comparison, the systems were compared to each other by using a statistical approach, an ANOVA with a global linear model. When statistically compared, all systems were deemed to be statistically comparable to one another (p = 0.135). All 9 machines showed consistent delivery when captured nicotine was used as a dose marker and expected levels of 3R4F smoke yields derived under ISO conditions. This was further examined by comparing the puff-by-puff profiles of each system under ISO smoking conditions for the designated 8 puffs. For example, the same profile was observed for each system. The nicotine content started low on the first puff and increased in amount delivered up to the final puff as the tobacco was combusted. In some cases, a slight plateau was observed in nicotine delivery at approximately puff 5; but the general trend was conserved. This puff-by-puff profiling accounts for the variability observed in each system and can be regarded as an artefact of the product and how it is consumed rather than the system (Figure 5).
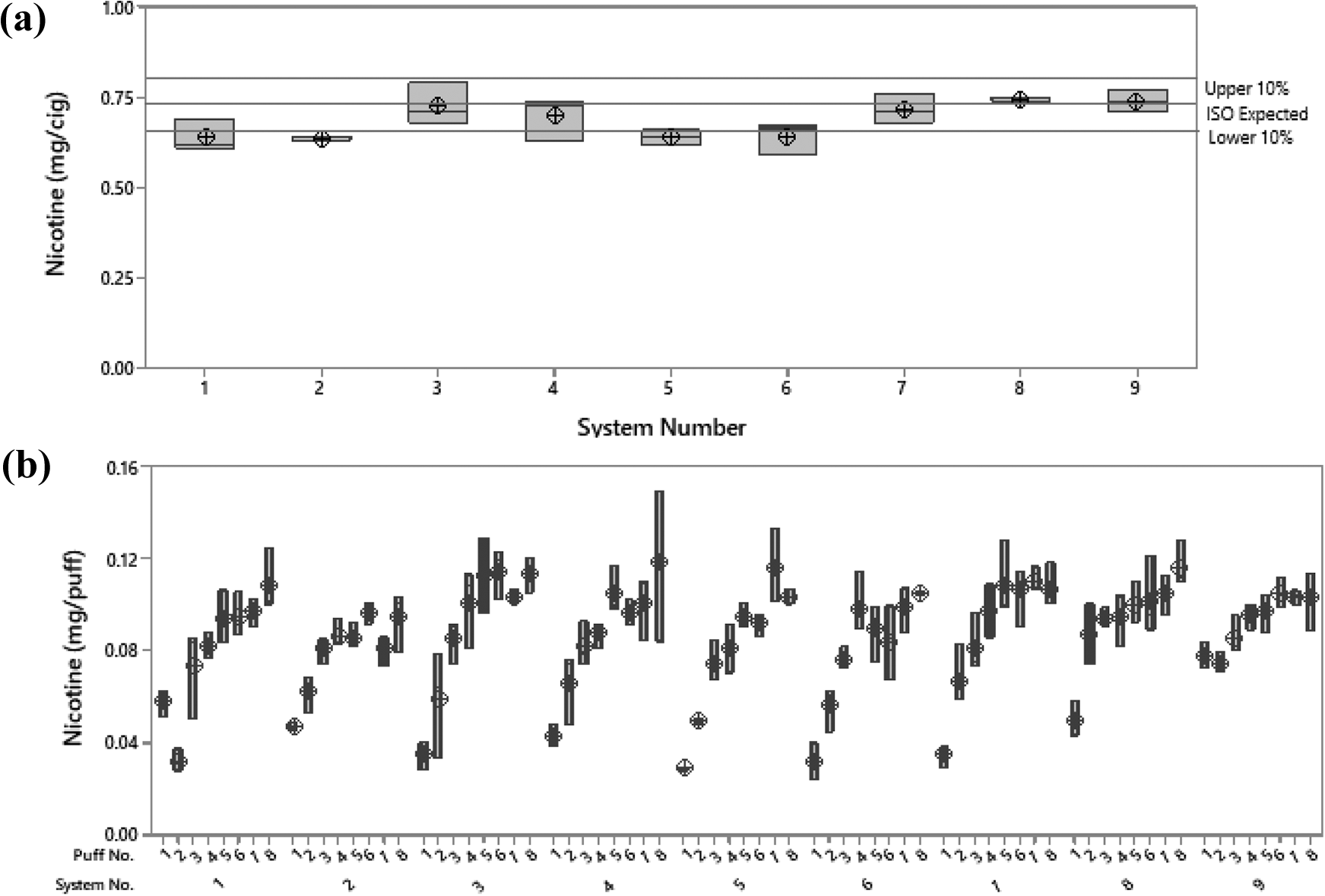
Comparison of systems under the ISO smoking regimen. (a) Average data on an mg/cig basis for all systems, with upper and lower 10% limits calculated on the basis of published ISO analytical data. 33 All systems were comparable (p = 0.135) and performed within a 10% limit of the expected values. (b) mg/puff data for all systems (1–8 puffs) under the ISO smoking condition. All systems showed a comparable puff-by-puff profile.
When an intense (HCI) smoking regimen was investigated, similar responses were observed between systems. All machines were approximately within 10% of an HCI reference value. 33 In this instance, the reference value was 1.97 ± 0.04 mg/cig ±10%, significantly higher than that with the ISO regimen, as expected, owing to the fact that the HCI regimen delivers approximately 2.5-fold the amount of nicotine as the ISO regimen. Again, when compared with each other by ANOVA (global linear model), there were no statistical differences between the systems (p = 0.135). When assessing puff profiles, the same trends were observed between the systems as those observed under the ISO conditions; however, owing to the higher delivery, a greater variation was observed between puffs 1–10 than between puffs 1–8 of the ISO regimen. A clear statistical difference was observed between the ISO and HCO smoking regimens (p = 0.000) (Figure 6).
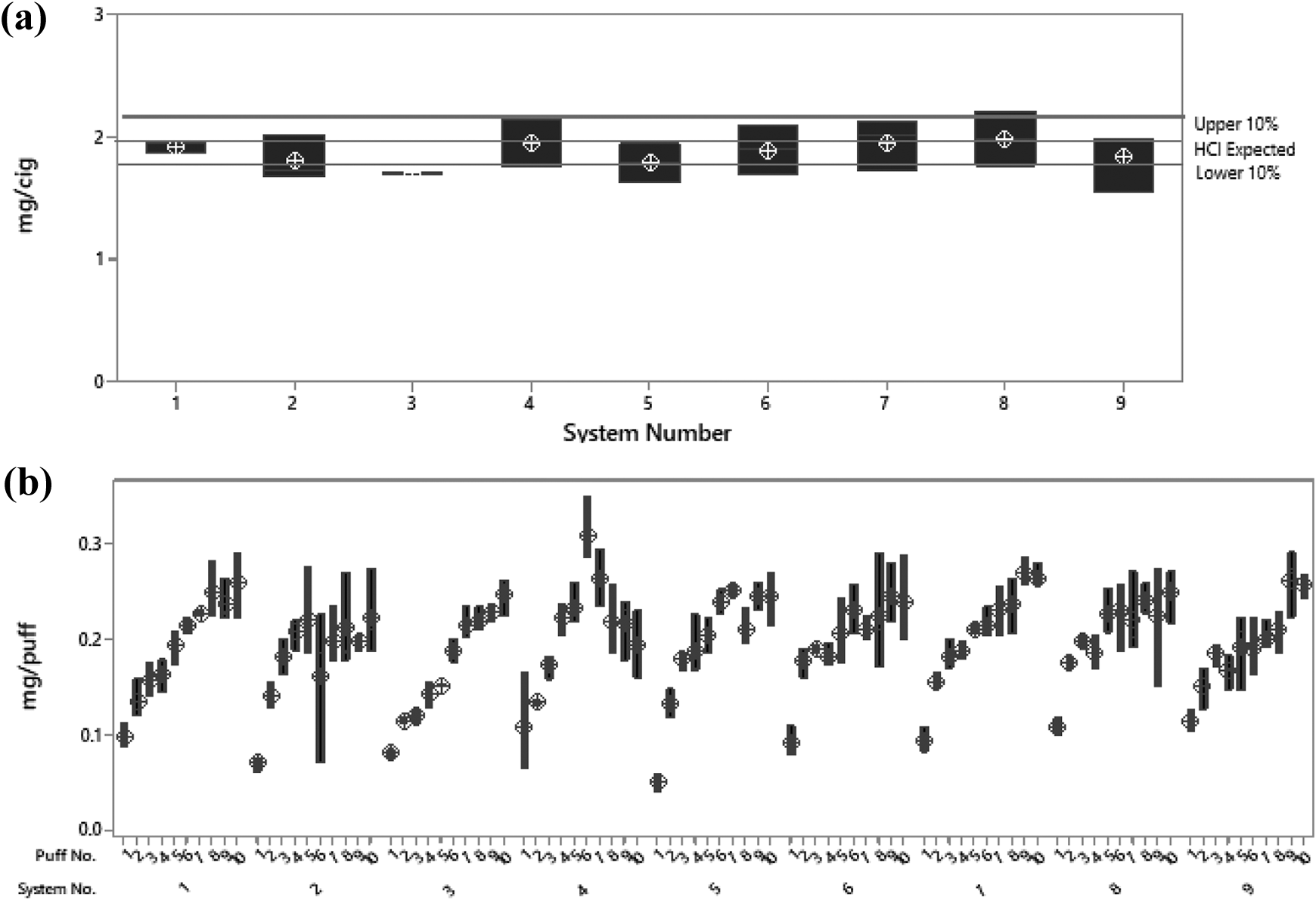
Comparison of systems under the HCI smoking regimen. (a) Average data on an mg/cig basis for all systems, with upper and lower 10% limits calculated on the basis of published ISO analytical data. 33 All systems were comparable (p = 0.135) and performed within a 10% limit of expected values. (b) mg/puff data for all systems (1–10 puffs) under the HCI smoking condition. All systems showed a comparable puff-by-puff profile.
Data from both ISO and HCI condition present a consistent story. However, there does not appear to be a correlation between the systems that performed well under the ISO and HCI smoking regimens. For example, systems 1, 2, 5, and 6 all appear to be on the lower end of the ISO 10% limit; but, when compared across the HCI data, these systems all appear well within the expected 10% tolerances. Conversely, system 3 under the HCI regimen appeared at the lower end of the 10% limits, whereas, under ISO conditions, this system appeared balanced and well within the limits. When comparing both regimens together in terms of average values, it is clear that all systems perform within a reasonable range under the ISO and HCI regimens and that both regimens should be considered for performance evaluation if using cigarettes. However, if we consider how the aerosol is generated and captured by using these systems in this study in comparison with how the published reference value is calculated, there are clear differences that could account for the systems performing, on average, on the lower end of the 10% limits. For example, the published reference value was generated by using an analytical smoke machine with the CFP placed directly behind the cigarette to maximize aerosol capture; in contrast, in our study, the CFP was installed in a convenient position to allow pad changeover and puff-by-puff analysis. Moreover, the analytical approach used for generating the published ISO and HCI values does not include changing pads over between puffs for puff-by-puff analysis. Given the diversity of these in vitro aerosol exposure systems, and that there is no single standardized position for CFP installation across all systems and the requirement for potential puff-by-puff analysis, it is unsurprising that there is an obvious bias towards the lower end of the 10% limits. It is, therefore, reasonable to suggest that these limits are not entirely suitable for analysis of in vitro aerosol exposure systems such as the ones used in this study, and a more realistic value should be used upon which appropriate limits can be set, which takes into account the measurement variability of puff-by-puff analysis and the positional effect of the CFP position. This study has established a new set of limits for analysis and potential future analysis of in vitro aerosol exposure systems, which other researchers can use to cross validate their systems. By taking the average across all systems under both the ISO and HCI smoking regimens, these data can be used to create limits that more accurately cover the diverse range of any aerosol generator and in vitro exposure platform currently in use (Figure 7).
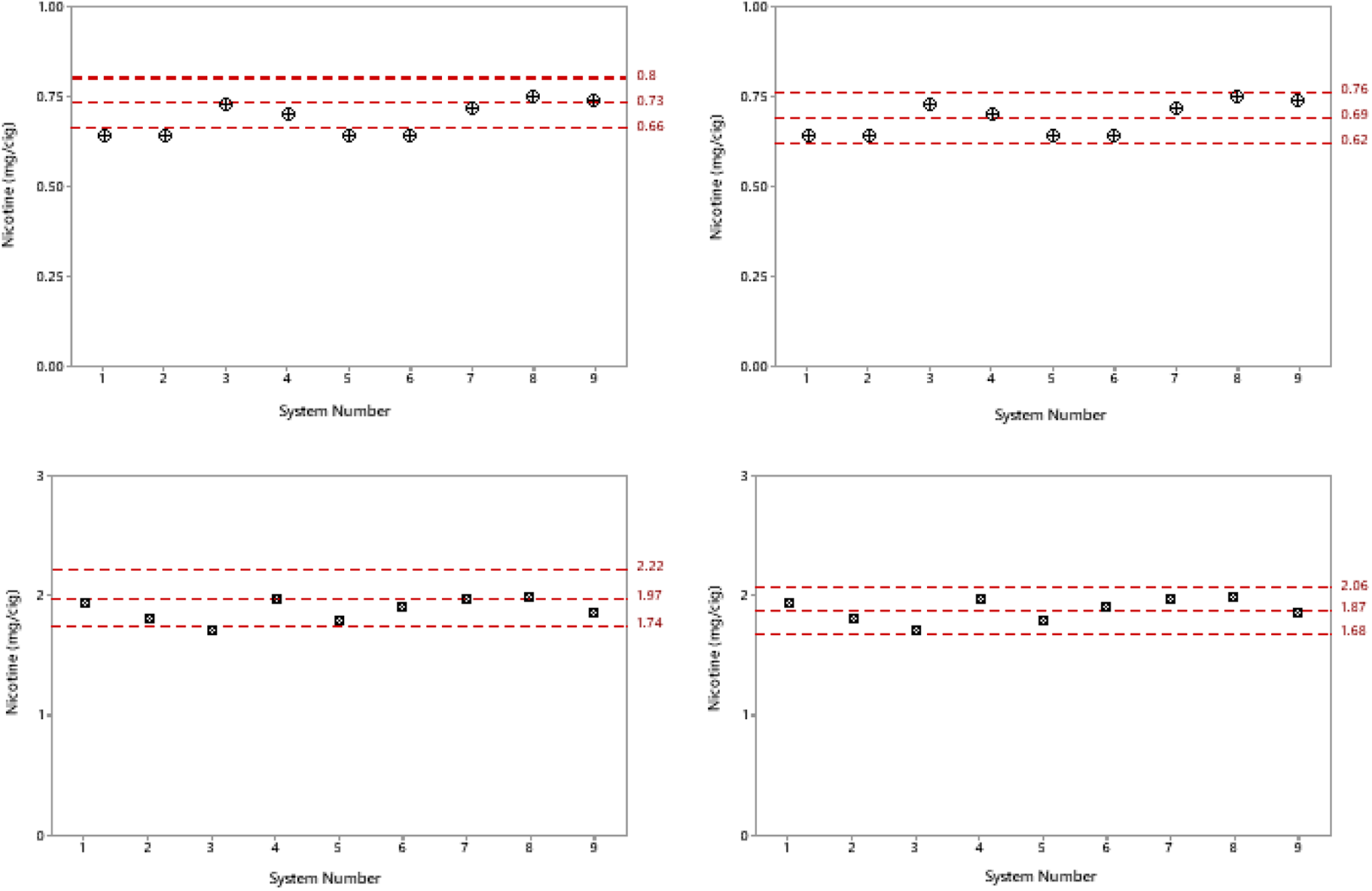
Comparison of systems under the ISO and HCI smoking regimens. (a) and (c) represent the ISO and HCI smoking limits derived by using a published nicotine value. 33 (b) and (d) represent the averaged data taken from this study across all systems, with the same 10% limits applied, but on the basis of the data generated in this study. The average from a published value is reduced for both the ISO and HCI regimens. The ISO average was 0.73 mg/cig and has reduced to 0.69 mg/cig, and the HCI average has been reduced from 1.97 to 1.87 mg/cig. Both reductions account for the losses in transit of the aerosol from the smoking head to CFP in-line capture. The location will depend on the system design and ease of CFP installation and swapping between puffs and the slight loss of aerosol on each occasion. With these new established averages for both the ISO and HCI regimens and the 10% limits, the data-spread and systems appear more balanced and consistent and are well within this 10% threshold. According to these data, the new proposed limits for in vitro aerosol exposure investigations should be 0.69 ± 0.069 and 1.87 ± 0.187 for the ISO and HCI regimens, respectively.
Discussion
These data demonstrate that, irrespective of the in vitro exposure system setup (different systems, position of the CFP and tubing differences), consistent and comparable data can be generated across multiple systems and geographical locations if standard procedures and approaches are employed. For example, this study has used two internationally recognized smoking regimens (ISO and HCI) and captured nicotine pre-syringe to assess aerosol generation. All systems were deemed statistically comparable when compared with each other and fell around a 10% upper and lower limit range when compared against an analytically derived value for the ISO and HCI regimens. The 10% variability was deemed fit for purpose for assessment for mechanical machinery. 35 The data presented here were used to establish a more accurate and realistic reference value which better represents the diversity of current aerosol generation systems. Furthermore, this new reference value can be used by other laboratories looking to characterize their exposure platform against this reference study, to further strengthen these data and support validation efforts.
Despite clear system-to-system differences, all exposure platforms are composed of three principal components. The first component is a smoking head and piston, which enables “aerosol generation.” The second component is the transit if the aerosol from the pistol to the dilution system (if present), termed “transit and dilution,” and the third component is delivery of an aerosol dose to the in vitro exposure chamber/module where the biological material is housed, termed “exposure interface.” Once the aerosol is generated, it enters the exposure system, where individual system differences start to affect the aerosol transit and delivery. The aerosol generation is not only the first common denominator between exposure platforms but also the most comparable and consistent and easiest to assess. Comparison of aerosol generation removes confounding downstream system differences that can make comparisons difficult. Furthermore, if aerosol generation is not consistent between setups, there is little point in assessing dilution, transit, and delivered dose to the exposure interface, as the systems would be generating different aerosols at the starting point of the aerosol’s journey. In the context that all systems use constructionally similar piston pumps, which are operated to generate puffs under standard protocols, the results from this study are unsurprising and reassuring. If the cigarette is puffed by using a set of established parameters, such as puff volume, duration, and profile, as set out in both the ISO and HCI smoking regimens, the resulting aerosol should be comparable, providing the machines are appropriately maintained and calibrated (and used within a standardized ISO conditioning environmen. 12 At the source of aerosol generation, once the smoking regimen is controlled, there are few variables that allow aerosol differentiation. It is only when the aerosol transits through the system, which includes the dilution set and exposure module, that significant aerosol changes occur and are dependent on each individual exposure system. For example, each exposure system handles aerosol dilution slightly differently. Some systems fractionate the smoke stream and subsample, while others dilute on a ratio basis. Some systems do not even use dilution principles at all. All systems have different lengths of tubing and tubing diameters leading from the smoking head to the exposure module. Each system is coupled with a distinct exposure module designed for that system. Each module has its own characteristics, and these also vary between setups. The combination of these variables makes comparison of systems and the resulting data difficult.
When comparing aerosol generation at source, all machines were deemed statistically comparable and all operated within approximately a 10% limit for ISO and HCI delivery of 3R4F reference cigarette smoke, which signifies that the systems are fit for purpose. 35 However, these comparisons must be considered with caution, because the published reference value was defined by using an analytical machine with a standardized position for the CFPs, which was directly behind the cigarette to maximize the transfer and capture efficiencies. This study did not compare standard analytical smoking machines; rather, it compared adapted smoking machines for in vitro aerosol delivery, and, therefore, it is not expected that these machines can exactly replicate the ISO and HCI reference values. For example, in this study, because of system differences, it was not possible to standardize the position of the CFP. The only caveat was that the CFP had to be placed before the piston. This means that the CFP could be directly behind the cigarette or up to a meter down the line. Therefore, it is unsurprising that some of the values are on the lower 10% limit and none are on the upper limit, as this accounts for potential differences in CFP position and losses within the transit line before aerosol capture. However, this study has taken the average across all systems and come up with a new set of values that better reflect the current state of play and the diversity of the in vitro aerosol exposure environment. These values can be applied to other in vitro aerosol exposure systems outside this study by using either a published ISO/HCI value or the measured value generated here for a more realistic, practical, and comprehensive assessment of machine performance.
This study adds significant information to the current literature regarding system characterization and potential validation. Validation is the process of demonstrating that an analytical method is suitable for its intended use via evaluation of method performance under defined criteria. 36 Method validation studies may involve a single laboratory (intralaboratory) or multiple laboratories (interlaboratory). Organizations such as the Association of Analytical Communities (AOAC) and US Environmental Protection Agency (EPA) provide methods that are validated through interlaboratory studies, and the parameters that may be assessed in method validation include precision, defined by reproducibility (R) and repeatability (r), and bias. 36 As such, data generated from method validation studies or standardized protocols will provide information for future regulation or testing standards. Lacroix et al. proposed a validation approach for ALI models. 1 But, their outlined approach did not differentiate between exposure platforms, in vitro models, and the combination of both. Rather, validation was focused on the in vitro approach and the exposure system as a combined “in vitro model”. 1 Here, an approach is proposed that supports the outcomes of Lacroix et al., by simplifying the situation even further for those laboratories involved in pre-validation and potential future validation exercises. The proposal is to view in vitro aerosol exposure platforms as their own distinct entity and establish performance criteria for these systems first, as has been demonstrated here. Of course, the applied in vitro model would itself have to be validated or at least be widely understood and applied in an in vitro aerosol exposure setting. However, there are several good candidates for future studies, such as simple cytotoxicity assays like the neutral red uptake assay, which has undergone a formal validation exercise, albeit under submerged conditions with individual chemicals. 37
To fully “validate” an in vitro exposure system, several key aspects need to be addressed across and within the platforms. Ultimately, researches have to demonstrate that in vitro exposure systems can collectively and consistently generate an exposure, deliver a dose, and match that dose to a biological response (validation of this response, in line with the suggestions of Lacroix et al., against clinical and in vivo settings is subject to validation of the model rather than the exposure platforms). 1 To achieve this, the following steps need to be addressed in both individual system characterizations and across platforms in interlaboratory efforts.
Step 1. Aerosol generation needs to be consistent and controlled, as this study has demonstrated. This is the first and most important step in the process, as, without these comparisons, any subsequent in vitro data generated with these systems only assumes accurate aerosol generation. This study is the first step in assessing different exposure platforms in an interlaboratory effort. The focus needs to be on expanding this reference set and investigating other aerosols such as those from e-cigarette and HTPs.
Step 2. Comparison of delivered dose. This is a little trickier, as the delivered dose is derived by system specifics such as transit tube length, presence or absence of dilution, subsampling, and, ultimately, chamber characteristics. Comparisons can certainly be made within system classes; but, comparison across systems could prove challenging. Interlaboratory studies have been conducted within exposure systems, 19,20,24 and, although studies have performed dose comparisons across platforms, 3,25 these have been individual laboratories comparing across platforms rather than true interlaboratory efforts. The results suggest that different exposure systems can operate within similar ranges and delivered doses can be compared. True interlaboratory efforts are required to confirm these observations.
Step 3. Delivered dose must be contextualized against a biological output to demonstrate that what is being delivered translates into an actual and consistent response within and across platforms. Some activities have been conducted in this space; but, again, there have been no true interlaboratory approaches. Adamson et al. demonstrated that different systems can generate the same response once data were presented as a function of normalized dose. 3 Thorne et al. took this one step further in a case study and demonstrated that delivered dose and biological response can be compared across systems if a consistent measure of exposure was used to normalize data. 27
Underpinning these steps, individual system characterizations which ensure that these systems are fully understood and detailed in regard to their application and nuances must be conducted by each laboratory. To some extent, validation of these systems are ongoing with pre-validation efforts made with individual system characterizations, which appear to be more common place than interlaboratory efforts. A great deal of work is focused on characterizing individual system setups, which includes generation, dilution/transit, and dose delivery to the exposure interface, through a number of analytical and biological techniques. 23,26,28,38 This characterization process usually focuses on the individual system and understanding its setup and delivery across the three functional aspects (generation, transit, and dose delivery). This study, for example, may lead to the identification of a reference setup to which all other systems can be compared at a later stage. Studies focusing on interlaboratory and platform comparisons are few but increasing in their relevance and importance as laboratories move into a regulatory space that required validated systems. So far, these studies have included comparison within systems and not across systems, as done in this study. To really consider the validation of these systems, more cross-platform and interlaboratory techniques need to be considered and applied. Platforms need to be monitored by using a unique identifier and tracked through multiple interlaboratory studies. It is important then that each of these systems and others are followed through a series of interlaboratory efforts to ensure a consistent and reliable dataset across generation, dose delivery, and biological response to support validation exercises.
Finally, this study has only considered cigarette smoke; but the current environment is far more complex than just cigarette smoke. E-cigarettes and HTPs are widely available and being tested by using these and other in vitro aerosol exposure systems. Future studies need to investigate this aspect and form a similar and consistent approach for system validation. The same fundamental principles of an in vitro aerosol exposure system apply—generation, dose delivery, and biological response; but the starting aerosols are significantly different. There are several options here: assume that a validated system for cigarette smoke also applies to e-cigarette and HTP aerosols or validate these systems independent of aerosol type. The latter is far more time consuming but probably the most relevant and accurate. These aerosols are different and should, therefore, be treated as such within these exposure systems.
In terms of future next steps, delivered dose and biological responses will need to be assessed. This has been conducted within a class of exposure system, 19,20,24 but never across multiple divergent systems. This poses some logistical challenges when designing such a study. For example, directly comparing delivered dose, may be challenging as these systems are designed differently and will naturally deliver different amounts of aerosol to the cell system, making direct comparisons redundant. Comparing biological responses requires the in vitro approach and exposure protocol to be sufficiently locked down and aligned for each laboratory, with consistent and robust responses. This includes, aligning parameters of exposure, the cell system and parameters of the in vitro approach between systems wherever possible. To achieve this and accurately compare between systems, a point of reference needs to be established that allows the direct comparison between systems. Each system may deliver different amounts of aerosol, but if the cell system and biological endpoint are consistent then dose can be aligned around a point of departure or an IC50 for example. As mentioned earlier a robust and reliable endpoint needs to be considered, there are several good candidates for future studies, such as the neutral red uptake assay, which has undergone a formal validation exercise, albeit under submerged conditions with individual chemicals, 37 but could provide the ideal assay to start interlaboratory comparisons. Finally, biological endpoints must be coupled with more active dose measurements to facilitate wider system-to-system comparisons.
Conclusions
This study has set out to compare nine distinct in vitro aerosol exposure systems in an interlaboratory approach across multiple laboratories and geographical locations. The study was standardized by using an internationally recognized reference cigarette (3R4F) and two accepted smoking regimens (ISO and HCI). Aerosol generation was selected for comparison, as this represents the first and most consistent denominator between systems. It also is the start of the aerosol journey and, therefore, in many respects, represents the most important part. Without this part of the aerosol story, subsequent comparisons and analyses between systems are fundamentally flawed, as they assume consistent delivery between setups. The results of this study demonstrate just that. Among the nine exposure systems assessed across five geographical locations, no statistical differences were observed across the systems with either smoking regimen, despite clear statistical differences between the regimens. All systems were comparable in terms of their variability, which was mostly attributed to the combustion process of the reference cigarette. Relative to a published ISO and HCI value, the systems all performed in a comparable manner (approximately at 10% variation); however, there was a clear bias at the lower end of the proposed limits, possibly due to the CFP location of the puff-by-puff approach employed. Either way, this led to the establishment of a new reference value more suitable to the capture of aerosol, given the system-to-system differences and differences in CFP capture location. This value, combined with 10% upper and lower limits, suggests that these systems are fit for purpose, and a 10% variance is acceptable for assessment. Other researchers in the field of inhalation toxicology can now use this value to compare and cross-validate their system (no matter what system it is) to this established and optimized dataset. Furthermore, interlaboratory approaches such as the one presented here will help establish these in vitro aerosol exposure systems as validated once all aspects are covered: generated aerosol, delivered dose, and biological response. Future studies will need to investigate a similar interlaboratory cross-platforms approach as that presented here for delivered dose and, ultimately, biological response, if a system is to be truly considered validated.
Footnotes
Acknowledgment
The authors would like to thank Tomasz Jaunky, Andrew Baxter, Anya Terry, and Nicole East for their technical support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Thorne, Jason Adamson, and Marianna Gaca are employees of British American Tobacco (BAT). Sandro Steiner, Stefan Frentzel, Shoaib Majeed, and Julia Hoeng are employees of Philip Morris. Edgar Trelles Sticken, Roman Wieczorek, and Liam Simms are employees of Imperial Brands. Shinkichi Ishikawa, Shigeaki Ito, and Kei Yoshino are employees of Japan Tobacco. Holger Behrsing is employed by Institute for In Vitro Sciences. Each institute funded their research and BAT collated the data. All authors approved the final version and supported writing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
