Abstract
Introduction
For in vivo inhalation studies, aerosol dosimetry links aerosol exposure and deposited dose within the respiratory tract. Aerosol dosimetry programs like the Multiple Path Particle Deposition model utilize respiratory tract geometry, respiratory physiology, and aerosol properties to predict particle deposition within the respiratory tract. A challenge to wider use of these dosimetry programs for in vivo inhalation studies is the lack of species-specific or strain specific respiratory tract anatomy, and in some cases, strain specific respiratory physiology data. The objective of this work was to develop aerosol dosimetry predictions for the in vivo human disease models of chronic obstructive pulmonary disease (COPD; C57BL/6 mice) cardiovascular disease (CVD; ApoE-/- mice), and lung cancer (AJ mice) using the Multiple Path Particle Deposition dosimetry model.
Methods
Microcomputed tomography derived tracheobronchial geometry data were combined with available pulmonary geometry data for C57BL/6, ApoE-/-, and AJ mice and incorporated into the Multiple Path Particle Deposition dosimetry model. Mouse strain specific respiratory physiology literature values were used as input into the Multiple Path Particle Deposition dosimetry model.
Results
The resulting particle deposition predictions compared well with the very limited strain specific experimental particle deposition data. Since multiple tracheobronchial geometries and thus pulmonary anatomies were used for C57BL/6 and ApoE-/- mice, an estimate of intrastrain variability in predicted particle deposition was obtained.
Discussion and Conclusions
The intrastrain variability in predicted particle deposition for C57BL/6 and ApoE-/- mice was always smaller in the tracheobronchial compared to pulmonary airways. The differences in reported respiratory physiology values for C57BL/6 mice resulted in greater intrastrain variability in predicted particle deposition than observed with the different tracheobronchial geometries and pulmonary anatomies. These mouse strain specific aerosol dosimetry predictions can provide insight into the delivered dose to specific respiratory tract regions that can be correlated with in vivo endpoints.
Keywords
Introduction
Aerosol dosimetry provides the critical link between aerosol exposure and the deposited dose within the respiratory tract for in vivo inhalation studies. Without accurate aerosol dosimetry, interpretation of in vivo study results is difficult, especially when the target organ is the respiratory tract or a specific region in the respiratory tract (e.g., nasal, oral, laryngeal, tracheobronchial, alveolar duct or alveoli). This difficulty occurs because the biomarkers of exposure that are usually measured in urine and/or serum (e.g., rarely in exhaled breath, etc.) represent temporally averaged deposited doses that have occurred throughout the duration of exposure and respiratory tract. Biomarkers of exposure also incorporate the temporal aspect of the absorption, distribution, and for some chemicals metabolism. By combining respiratory tract anatomy, animal physiology, and aerosol characteristics, aerosol dosimetry models like the Multiple-Path Particle Dosimetry (MPPD) model can predict total (entire respiratory tract), regional (head, tracheobronchial, and alveolar) and local (nasal, oral, groups of tracheobronchial airways) deposited doses within the respiratory tract1–3 that can be correlated to in vivo study results. For example, MPPD dosimetry predictions in Balb/c mice for iron oxide, silver, and C-60 fullerence nanoparticles and C-60 fullerene micron sized particles correlated well with the retained dose from in vivo inhalation studies. 4
One major limitation to wider use of aerosol dosimetry models is the lack of species-specific or strain specific respiratory tract anatomy. Acquisition of the necessary respiratory tract anatomy has evolved from various manual digitization techniques5–7 to non-invasive scanning methods (e.g., magnetic resonance imaging, computed tomography- CT. 8 Recently, micro-CT techniques were used to obtain the tracheobronchial tract anatomy of several mouse strains,9–12 including those mouse strains used as in vivo models of human disease, such as chronic obstructive pulmonary disease (COPD; C57BL/6 mice), cardiovascular disease (CVD; ApoE-/- mice) and lung cancer (AJ mice). Numerous inhalation studies have used the C57BL/6 mouse as a model of COPD,13–18 the ApoE-/- mouse (derived from the C57BL/6 mouse and is apolipoprotein E-deficient) as a model of CVD19–23 and the AJ mouse as a model of lung cancer.24–27 Unlike the other two mouse strains, the C57BL6 mouse when not exposed to inhaled toxicants (e.g., mainstream tobacco smoke), is considered a wildtype mouse that does not exhibit features of COPD. Since the focus of this work is particle deposition predictions for inhaled toxicants studied in inhalation toxicology, we refer to the C57BL/6 mouse as a human disease model of COPD.
The purpose of this work was to develop aerosol dosimetry predictions within the mouse lower respiratory tract (tracheobronchial and alveolar airways) using the MPPD dosimetry model for the in vivo human disease models of COPD (C57BL/6 mice), CVD (ApoE-/- mice), and lung cancer (AJ mice). These mouse strain specific aerosol dosimetry predictions will provide insight into the delivered dose to specific respiratory tract regions that can be correlated to in vivo endpoints.
Methods
Tracheobronchial and pulmonary airway anatomy
Tracheobronchial (TB) airway geometry was developed based upon micro-CT derived airway dimensions (airway length, diameter, branch angle and inclination to gravity) from five C57BL/6, four ApoE-/-, and three A/J young adult mice in situ replica lung casts.
12
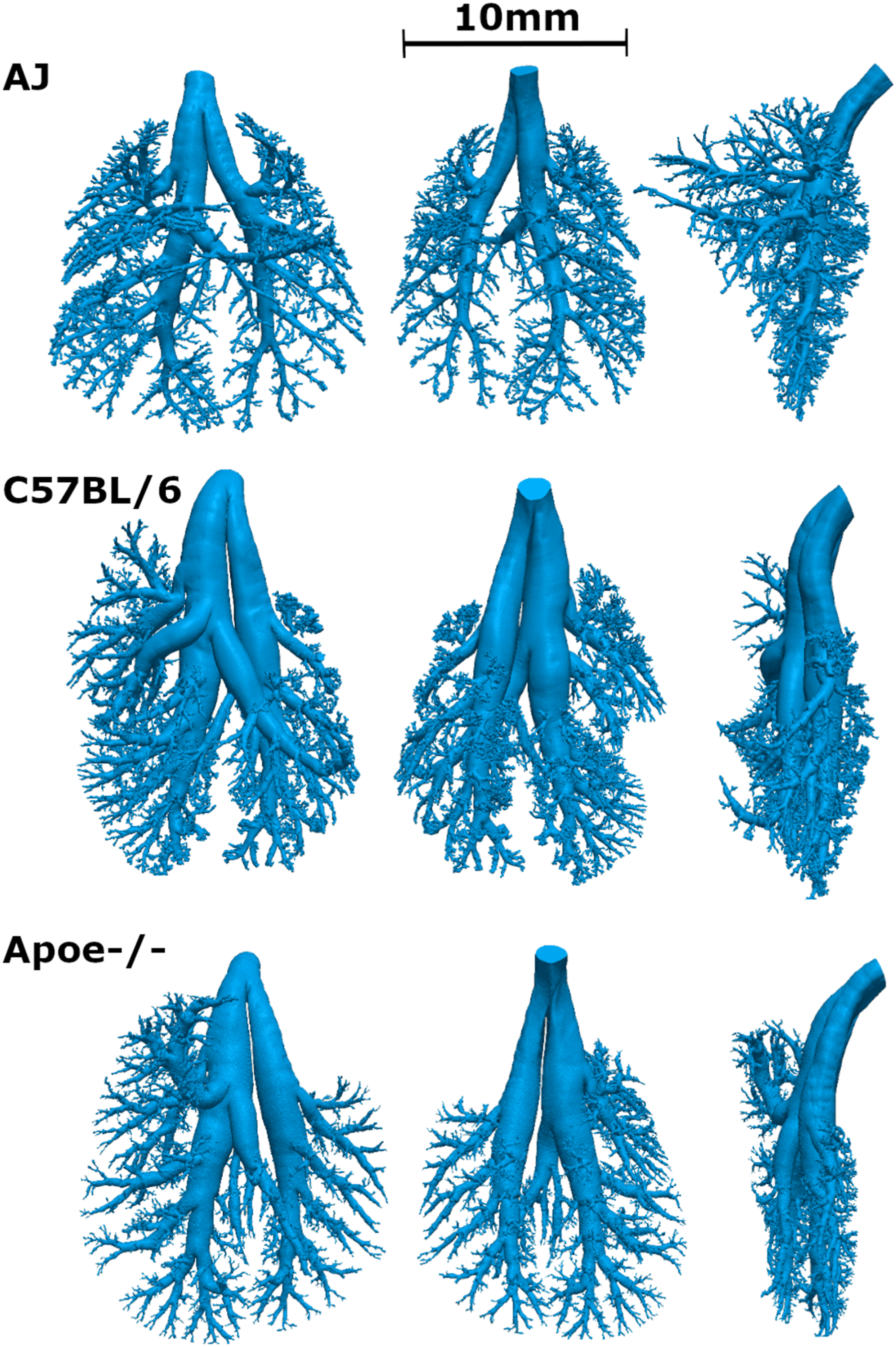
An asymmetric TB model was developed for each cast from each mouse strain, meaning unique average airway dimensions were used for each airway until they reached a terminal bronchiole (Figure 1). Each mouse strain had five lung lobes (apical or cranialis; medial or medius; cardiac or cardialis; diaphragmatic or caudalis; and left or sinister). Airway dimensions obtained from scanned images of the TB tree were rescaled based on the TB volume for each mouse strain (Table 1). TB volume is proportional to the total lung volume of each mouse strain. Thus, when TB volume for other mouse strains was not found in the literature, it was extrapolated from the ratio for the C57BL/6 mouse. The average airway value from all animals per strain as well as the standard deviation of the value was used to provide an estimate of the variability within each mouse strain. Reconstructed views of ventral (left), dorsal (center), and side (right) of lung casts for each mouse strain (Adapted from
12
). Mean and range of mouse strain specific values used in the MPPD model. N = number of mice used for mean measurement value. aUses unrealistic assumptions that alveoli are spheres and are of constant size. Newer lung lobe terminology- Apical = Cranialis; Medial = Medius; Cardiac = Cardialis; Diaphragmatic = Caudalis; and Left = Sinister. cNo respiratory physiology data obtained using restrained double plethysmography. dUsing technique of.
28
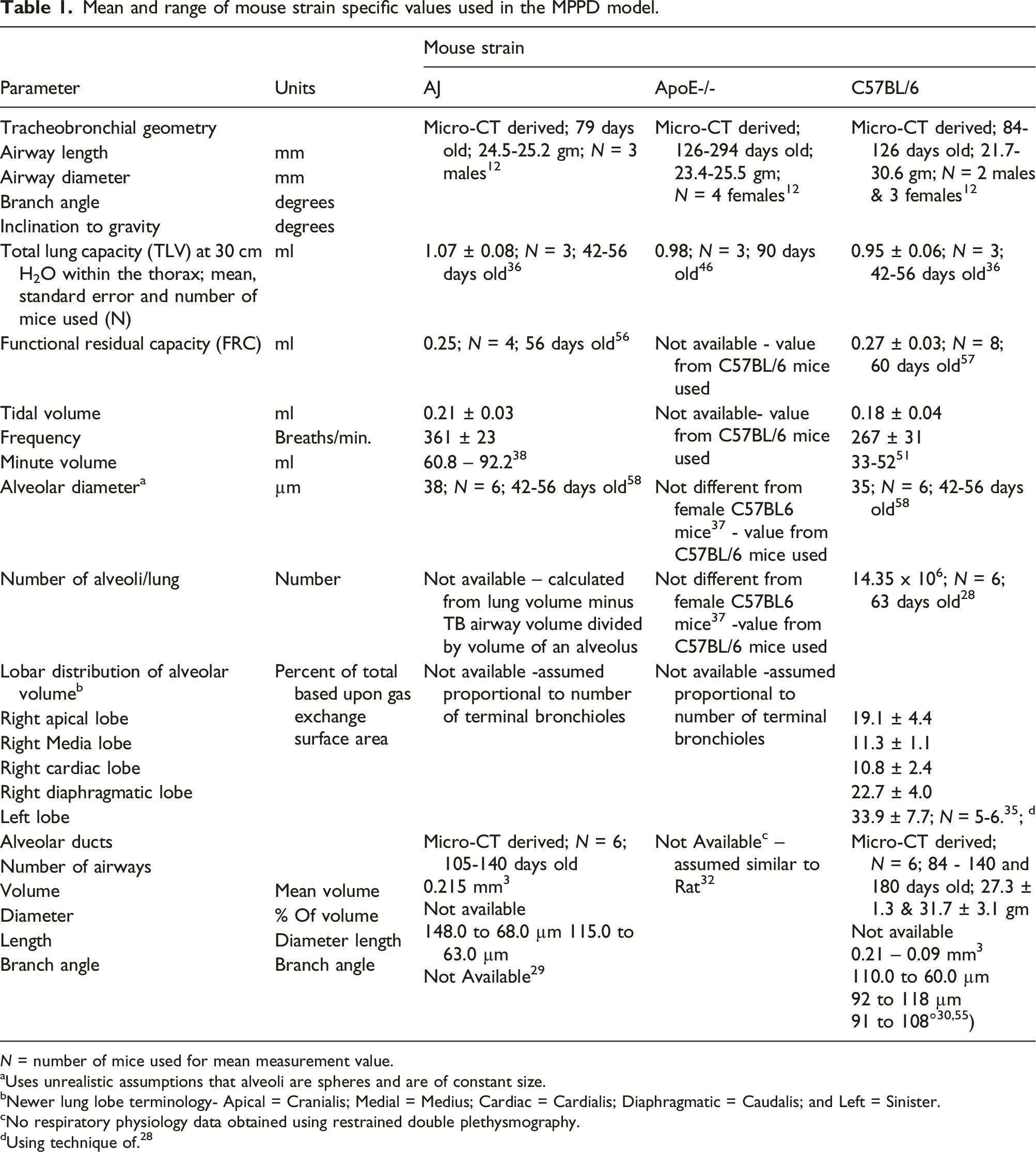
Pulmonary anatomy (alveolar ducts and alveoli) for each mouse strain was obtained from the literature (Table 1). For C57BL/6 and AJ mice, alveolar duct dimensions in several acini reported by Puliyakote et al.29,30 were used because they utilized the recommendations of the American Thoracic/European Respiratory Society for quantitative assessment of lung structure. 31 For both C57BL/6 and AJ mice a 7-generation alveolar duct tree was constructed to match the number of alveolar duct airways and alveolar duct volume reported by Puliyakote et al.29,30 using the reported mean alveolar duct length, diameter, and branch angles. Since there was no data for ApoE-/- mice, we assumed the alveolar geometry tree to be similar between rats and mice and an eight-generation alveolar tree was constructed following the approach of Yeh et al. 32 for rats. Since no measurements of the gravity angles were provided, an average of 60° angle was assumed. 32 For C57BL/6 mice the number of alveoli was obtained from Pozarska et al., 28 because they conducted a longitudinal study from postnatal day 5 to over 22 months of age, utilized the recommendations of the American Thoracic/European Respiratory Society for quantitative assessment of lung structure 31 and design-based stereology.33,34 Subsequent work from the same group 35 provided the lobar distribution of the number of alveoli for age matched C57BL/6 mice at postnatal day 14, which was assumed not to change as a function of age for our young adult C57BL/6 mouse model. For ApoE-/- and AJ mice the number of alveolar were calculated (Table 1) and lobar distribution was assumed to be consistent with the number of terminal bronchioles. Alveolar diameter values for each mouse strain were obtained from the literature (Table 1) but are based upon two unrealistic assumptions: that alveoli are spheres and are of constant size. Since the age and weight (and thus size) of the “young” C57BL/6 mice used by Puliyakote et al. 30 were different from the C57BL/6 mice used to construct the tracheobronchial tree, the pulmonary airway dimensions and alveoli volume were rescaled to match the size for the size of the TB tree. The constructed pulmonary anatomy was then attached to the distal end of each terminal bronchiole of the TB tree to create a complete lung geometry for each mouse strain.
Respiratory physiology
Respiratory physiology values for total lung capacity (TLC) and functional residual capacity (FRC) were also obtained from the literature (Table 1). As noted by Soutiere and Mitzner, 36 the use of the TLC is based upon it’s use in humans where it is defined as the lung volume at the end of “maximal” voluntary inspiration. Obviously, mice are unable to perform such a maneuver, so we used literature values that measured TLC and FRC in anesthetized AJ and C57BL/6 mice 36 from realistic inspiratory pressure-volume curves that corresponded to pressures (≤30 cm H2O) used to obtain pulmonary anatomical values. In this work (similar to, 36 TLC is used in a “vernacular” sense to indicate maximum lung capacity. Since only female ApoE-/- mice tracheobronchial airway geometry was obtained by Oldham et al., 12 the female ApoE-/- mouse pulmonary data of Massaro and Massaro, 37 indicating no difference from age and weight matched female C57BL/6 mice was used.
Tidal volume and respiratory frequency values for each mouse strain were also obtained from the literature (Table 1). We chose tidal volume and frequency values from studies using restrained double plethysmography since this represents what typically occurs in nose-only inhalation studies. Use of tidal volume and respiratory frequency results from unrestrained or single plethysmography techniques result in significantly higher respiratory frequency values. 38 If these better represent the experimental situation, they can be changed within MPPD. The range of minute volumes were calculated from the tidal volume and respiratory frequency values in Table 1.
Multiple-path particle dosimetry
The MPPD dosimetry model1,39 was extended to different strains of mice by using mouse strain-specific models for breathing rates, and lung geometry and volumes. The MPPD model is based on 1D formulation of airflow and aerosol transport in the lung during a complete breathing cycle, which includes inhalation, pause and exhalation. The asymmetric tracheobronchial structure and dichotomous pulmonary airways are approximated as straight, cylindrical tubes. 4 Lung ventilation is based on uniform expansion and contraction of airways during inhalation and exhalation, 40 moves the aerosols through lung airways while being deposited by external forces of gravitational settling, Brownian diffusion for ultrafine and nanoparticles, and inertial impaction and sedimentation for larger particles. These deposition mechanisms are based upon conservation of mass and momentum of particles and are treated as sink terms in the governing transport (convection-diffusion) equations to determine deposition in each airway and region of the respiratory tract. Particle deposition is represented by deposition fraction per site or region, which is the amount of deposition divided by the total amount of inhaled particles.
Based upon recent Organization for Economic Cooperation and Development (OECD) (OECD Guidelines 39,412, and 413,41–43 guidelines for inhalation toxicology studies that specify geometrical and mass median aerodynamic particle diameters for mice should not exceed 2 microns with a geometric standard deviation of less than 3, predicted aerosol deposition results are shown for geometrical and mass median aerodynamic monodisperse particle diameters from 0.01 to 2 microns. This particle size range is also consistent with nose-only exposure of CF-1 mice that showed 15.8% of 1.0 µm and only 3.6% of 3.36 µm particles deposited distal to the larynx. 44 The original reported aerodynamic resistance diameter reported by Raabe et al., 44 was converted to aerodynamic diameter via Raabe et al. 45
Results
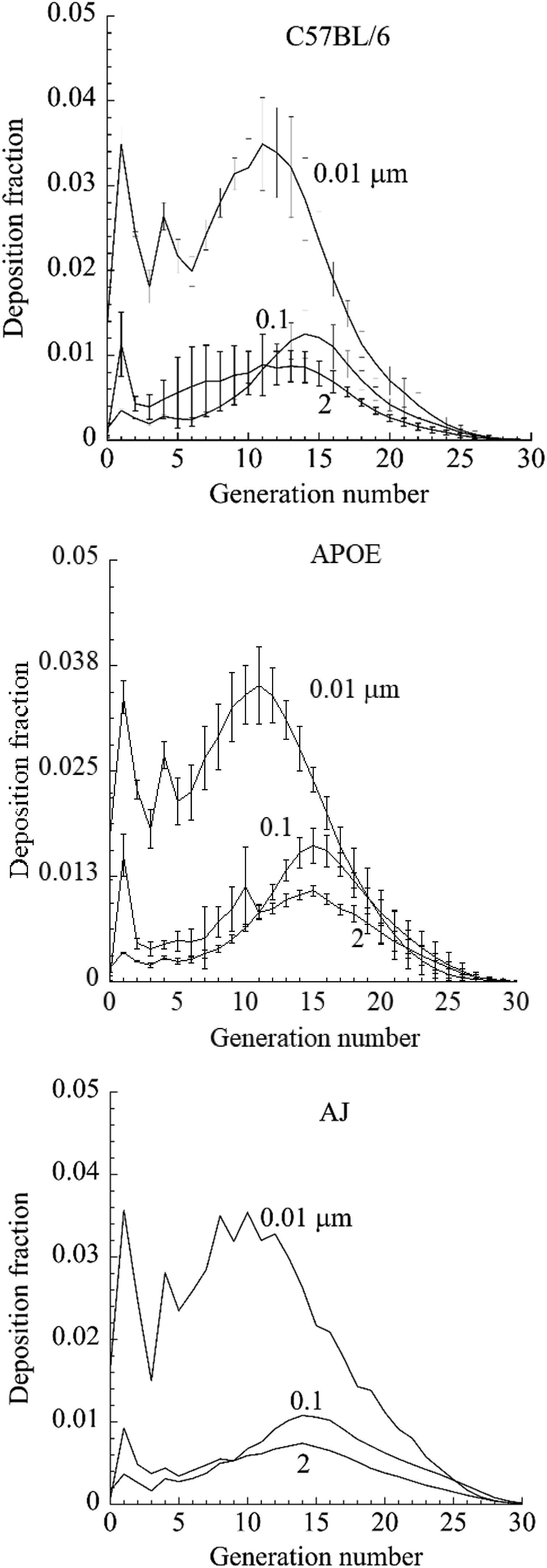
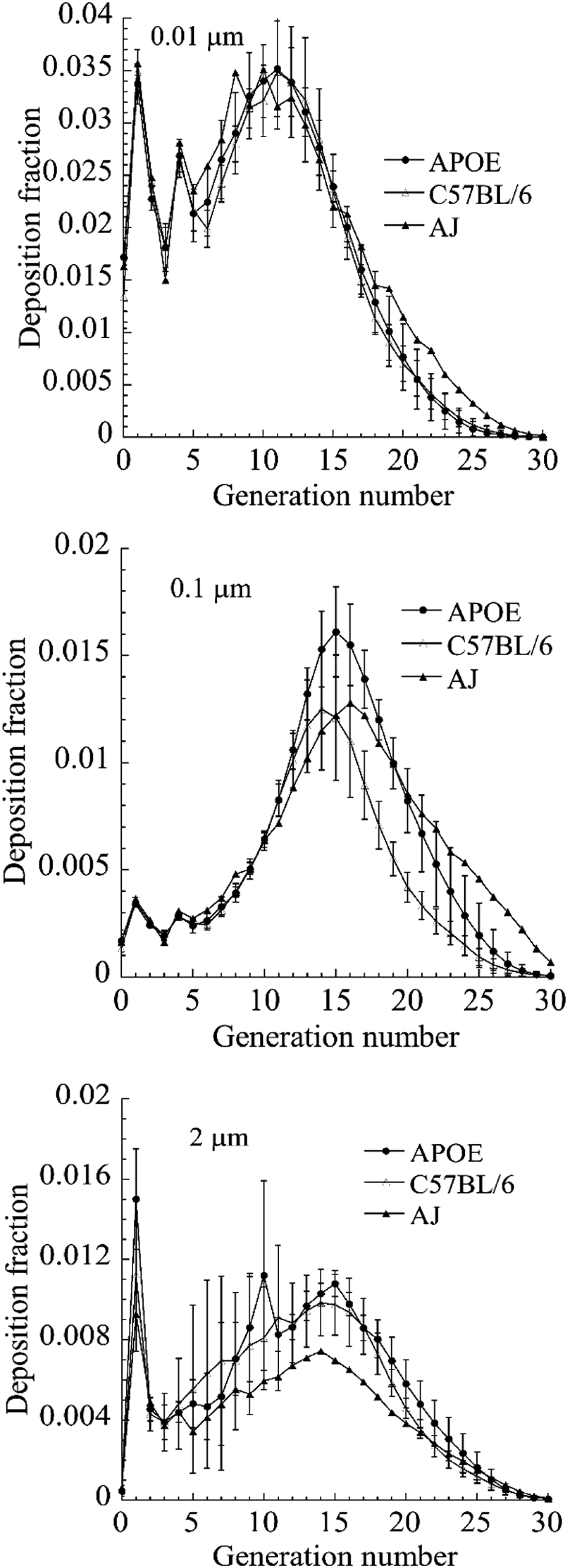
The individual tracheobronchial and pulmonary anatomy files used in this work are in supplemental tables 1-20. The mean and range of predicted particle deposition fraction (intrastrain variability) due to mean and standard deviation in tracheobronchial and pulmonary airway geometry in each mouse strain resulted in different aerosol deposition fraction predictions (Figure 2). There was no intrastrain variability for the AJ mouse since only a single in situ lung cast was suitable for complete micro-CT measured tracheobronchial geometry. For both C57BL6 and ApoE-/- mice the intrastrain variability in predicted deposition fraction was less in TB than pulmonary airways. A similar pattern of variability in predicted deposition fraction as a function of airway generation occurred for both C57BL/6 and ApoE-/- mice strains although variability was more slightly more pronounced for the C57BL/6 mice (Figure 3). The intrastrain TB airway geometry variability combined with the differences in pulmonary anatomy and respiratory parameters resulted in little to no difference in predicted particle deposition fraction between the three different strains of mice (Figure 4). Predicted deposition fraction (mean ± SD) versus particle diameter for the C57BL/6, ApoE-/-, and AJ mice for the tracheobronchial (TB), pulmonary (PUL) and the combined lower respiratory tract (LRT) airways. Predicted deposition fraction (mean ± SD) versus airway generation for 0.01, 0.1 and 2 μm diameter monodisperse particles for C57BL/6, ApoE-/-, and AJ mice. Comparison of predicted particle deposition for C57BL/6, ApoE-/-, and AJ mice for 0.01, 0.1, and 2 µm diameter monodisperse particles.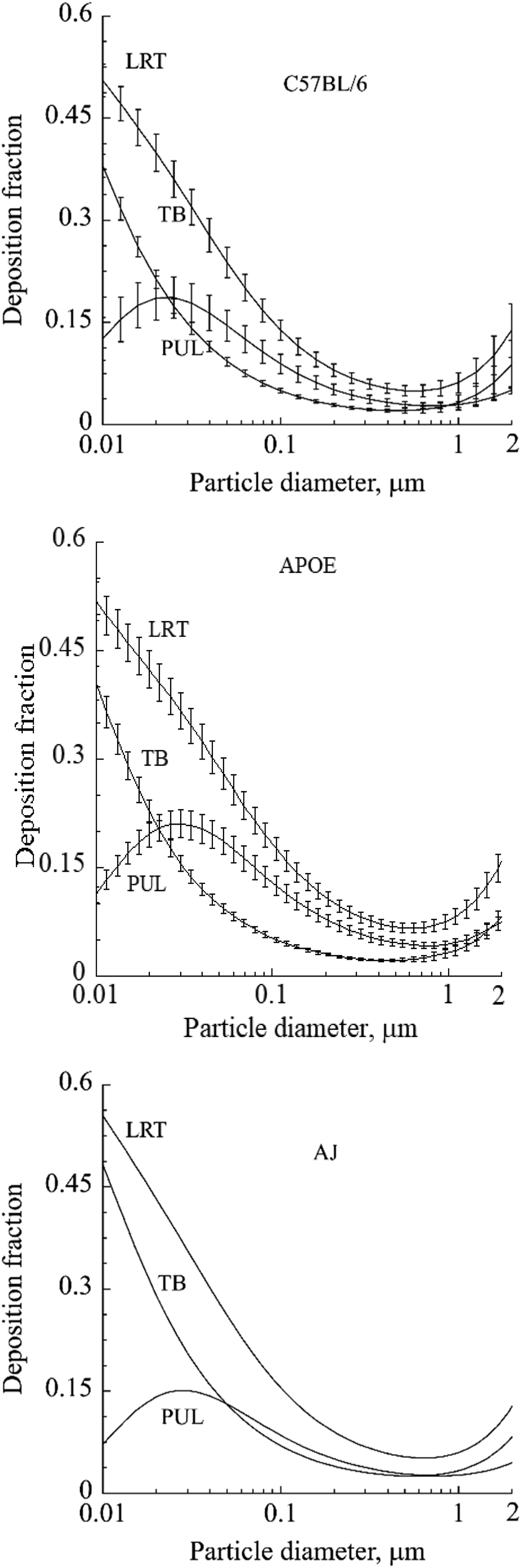
Discussion
The larger intrastrain variability seen in the predicted particle deposition fractions for C57BL/6 mice compared to ApoE-/- mice, despite having more C57BL/6 TB geometries, is likely due to the larger body mass range covered by those TB geometries (21.7-30.6 gm for C57BL/6 vs 23.4-25.5 gm for ApoE-/-; Table 1). The C57BL/6 TB geometries were also obtained from both male (N = 2) and female (N = 3) mice while those for ApoE-/- mice were only obtained from females (N = 4). The use of only female ApoE-/- mice in the convenience sample of Oldham et al 12 was fortuitous since Massaro and Massaro, 46 found that male ApoE-/- mice have impaired alveologenesis resulting in fewer and larger alveoli. This difference for male ApoE-/- mice manifests between 30 and 90 days of age and results in unique respiratory parameters in adult mice. 46
The variability in body mass for the convenience sample used by Oldham et al., 12 was compared to the variability in body mass typically seen in mouse inhalation studies. Body mass variability in the convenience sample of Oldham et al., 12 for the C57BL/6 mice was approximately 18% compared to approximately 4% for the ApoE-/-mice. The common practice in mouse inhalation studies is to use a stratified randomization scheme designed to achieve similar group mean body mass prior to exposure. Results from mouse inhalation studies has shown that when the test article is not toxic, the variability in body mass at the end of exposure is approximately 5% 26 - 5 months,14,15,18,20,47; however as test article toxicity increases the variability in body mass also increases, up to 25% 26 – 9 months.17,22,27,48
Beichel et al.,
10
and Bauer et al.,
11
reported relative overall percent particle deposition and measured respiratory physiology data for eight C57BL/6 mice. Four of these C57BL/6 mice were exposed to 1 µm and the other four were exposed or 2 µm diameter fluorescent particles. Using their reported respiratory physiology data10,11 in MPPD, our predicted percent particle deposition results for 1 and 2 µm diameter particles in C57BL/6 mice were consistently a little higher but within the experimentally measured standard deviation of overall percent particle deposition (Figure 5). Experimentally measured particle deposition using either ApoE-/- or AJ mice were not found in the literature. In CF-1 mice, the deposition fraction distal to the larynx of 15.8% for 1 µm particles reported by Raabe et al.,
44
is greater than the predict particle deposition results for all three mouse strains in this work (C57BL/6, ApoE-/-, and AJ), however there is an assumption of resting ventilation in this comparison.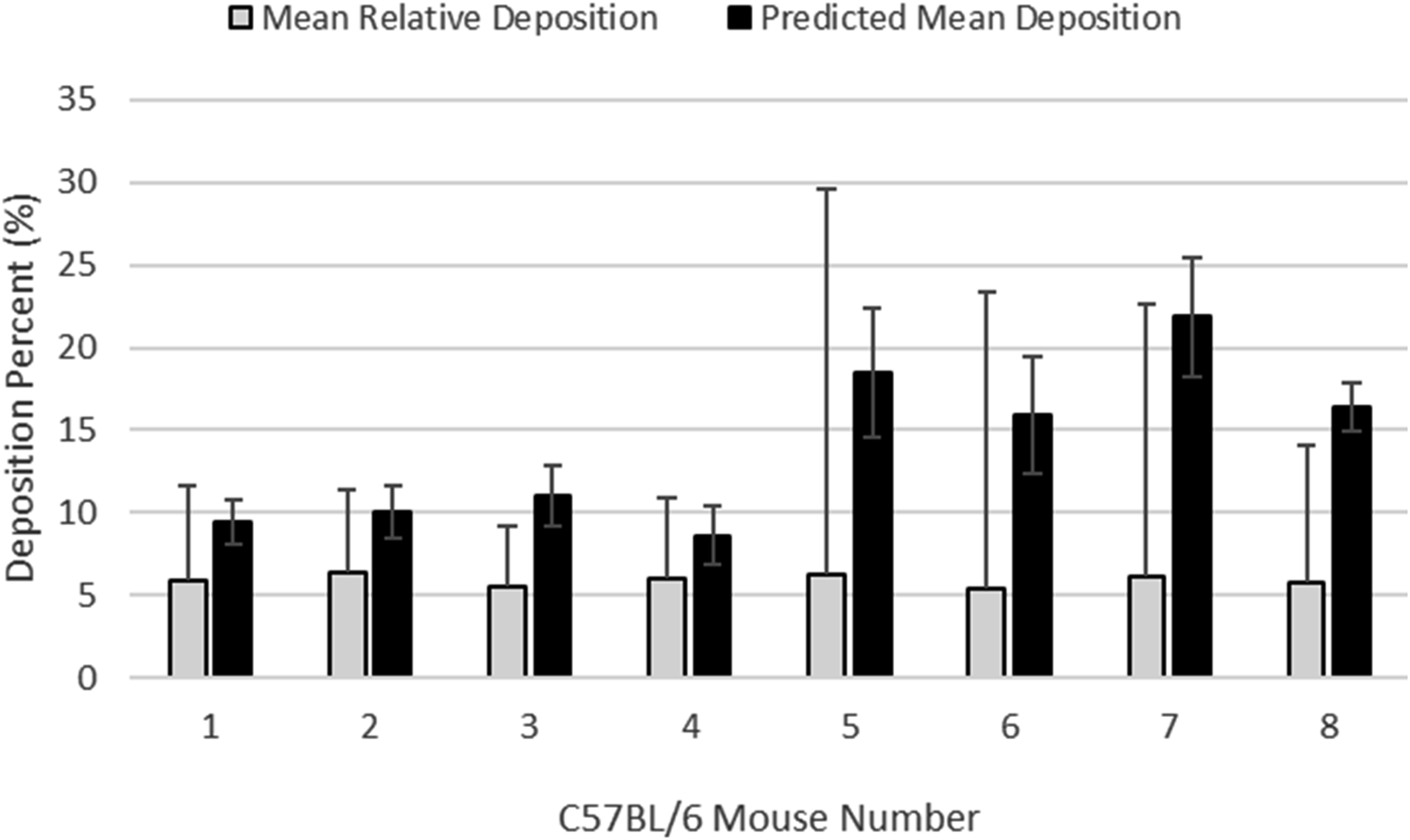
One of the limitations of this work is that Oldham et al.,
12
used a convenience sample of mice that did represent the entire mouse life-span that are used in in vivo inhalation studies. Another limitation is that a single set of respiratory physiology parameters were selected for each mouse strain. Using the C57BL/6 median TB and pulmonary geometry with other published values of minute volume (i.e.,
49
MV = 100 mL,
50
; MV = 6.23 mL,
51
; MV = 48.2 mL,
51
; MV = 27.2 mL,
52
; MV = 27.75 mL
53
; [lean and obese mice], MV = 23.3 and 29.5 respectively,
54
; MV = 43.0
17
) resulted in a large amount of intrastrain variability in predicted particle deposition fraction (Figure 6). The intrastrain variability in predicted particle deposition fraction from use of the various reported respiratory physiology parameters was larger than the intrastrain variability in predicted particle deposition fraction due to differences in TB and pulmonary geometries. Another limitation is that the MPPD dosimetry model is a 1D model that cannot implement differences in alveolar duct dimensions that have been noted in acinar dimensions on the mouse lung surface compared to deeper within the mouse lung.29,30 For C57Bl/6 mice, differences in acinar dimensions as a function of age reported by Vasilescu et al.,
55
and Puliyakote et al.,
29
were not implemented in MPPD since only two ages (12 and 90 weeks) of male mice and only two lung lobes were studied. Finally, it was not possible to predict particle deposition throughout the respiratory of these three strains of mice due to a lack of strain specific extrathoracic airway geometries (nasal through larynx). Depending on the differences in particle deposition resulting from these mouse strain specific extrathoracic airways, any differences in total predicted respiratory tract deposition between these three mouse strains could be eliminated, minimized, or exacerbated. Predicted particle deposition fraction in C57BL/6 mice using minute volumes reported in different studies.
In conclusion, tracheobronchial and pulmonary geometries for the C57BL/6, ApoE-/-, and AJ mice strains that are used as in vivo models of COPD, cardiovascular, and lung cancer, respectively, were created and incorporated into the MPPD dosimetry software. Strain specific representative respiratory physiology data was used to provide particle deposition predictions that correlated well with the limited existing experimental data for these strains of mice.
Supplemental Material
Supplemental Material - Predicted aerosol dosimetry for mouse models of chronic obstructive pulmonary disease, cardiovascular disease and lung cancer
Supplemental Material for Predicted aerosol dosimetry for mouse models of chronic obstructive pulmonary disease, cardiovascular disease and lung cancer by Michael J. Oldham, Francesco Lucci and Arkadiusz K. Kuczaj in Toxicology Research and Application
Footnotes
Acknowledgements
The authors would like to thank Bahman Asgharian and Owen Price for incorporating the respiratory tract anatomies and physiology data into MPPD and providing the deposition predictions for each mouse strain.
Author Contributions
All authors conceived the project. FL, AKK, and MJO provided known lung anatomy and physiology data for incorporated into MPPD. MJO wrote the first draft and all authors reviewed, edited, and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Oldham Associates, LLC, and Philip Morris International.
Data Availability Statement
The data in this article is based upon outputs from the Multiple Path Particle Deposition dosimetry model using the mouse geometry provided in the supplemental materials. The Multiple Path Particle Deposition dosimetry model is available for download free of charge at the Applied Research Associates, Inc. website (![]() ).1–4
).1–4
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
