Abstract
A framework for the assessment of novel next-generation tobacco and nicotine products with the potential to reduce health risks compared with cigarettes should integrate scientific studies incorporating nonclinical, clinical, and population studies approaches. These products should have lower emissions and exhibit reduced biological effects compared with those of cigarettes, ideally approaching those of smoking cessation. The products should also be acceptable cigarette alternatives for current smokers, while not appealing to nonsmokers or former smokers. Validating harm reduction and promoting regulatory acceptance of the assessment methods require a collaborative approach by industry, independent reviewers, the public health community, and regulatory agencies. This article summarizes the science and approaches applied and considered to substantiate tobacco harm reduction in the light of regulatory requirements, presented at the 53rd Congress of the European Societies of Toxicology, 2017.
Keywords
Introduction
Cigarette smoking is the leading cause of preventable disease and premature mortality worldwide and exerts a considerable economic burden, which has been reported to be as high as 15% of national health-care expenditures in some countries. 1 The goal of reducing the harm caused by smoking has traditionally relied on two pillars: (1) preventing smoking initiation and (2) promoting smoking cessation. While these two pillars are, and will continue to be, crucially important in reducing the prevalence of smoking, smoking cessation has proven difficult for many smokers. Consequently, there is an unmet need for additional methods to reduce the harm caused by cigarette smoking among current smokers.
Tobacco harm reduction is increasingly recognized as a promising approach to accelerate the decline in smoking prevalence and hence smoking-related population harm. It was defined by the US Institute of Medicine (IoM) as “decreasing total morbidity and mortality, without the complete elimination of tobacco and nicotine use.” The IOM referred to “potentially reduced-exposure products (PREPs) as having reductions in exposure to one or more tobacco toxicants.” 2 With recent advancements in the development of novel next-generation tobacco and nicotine products such as tobacco-heated products and e-cigarettes, McNeil and Munafò proposed a “risk continuum” 3 : tobacco- and/or nicotine-containing products, including oral smokeless tobacco (ST) and e-cigarettes, were placed on a continuum of risk, with cigarettes positioned at the high-risk end and nicotine replacement therapies (NRTs) at the lower risk end.
The US Food and Drug Administration (FDA) has provided a draft guidance document (FDA, 2012) outlining a framework to assess novel tobacco and nicotine products as modified-risk tobacco products (MRTPs), replacing the PREP terminology. 4 Based on that draft guidance, a number of product assessment frameworks have been published, proposing nonclinical, clinical, and population studies to assess the risk reduction potential of novel tobacco and nicotine products both at the individual and population levels. 5 –8
Next-generation tobacco products are being adopted by smokers looking for alternatives to help reduce the use of cigarettes or replace them entirely. These alternatives to cigarettes are likely to be less harmful because they are designed and operated differently from cigarettes, generating different aerosols that emit significantly lower levels of toxicants or harmful and potentially harmful constituents (HPHCs) than cigarettes. 8 It is the HPHCs generated by burning tobacco, and not nicotine that are the primary cause of smoking-related diseases (ref). Hence, nicotine delivery products that do not combust tobacco or do not contain tobacco are essential tools in tobacco harm reduction. 9,10
Effective harm reduction at the population level requires that a significant number of smokers actually adopt the available MRTPs, meaning that MRTPs must be designed to minimize product risk while maximizing product acceptance by smokers (i.e. motivating users to switch to reduced-risk alternatives). Therefore, an MRTP must meet two conditions: (1) it must significantly reduce the risk of disease compared with cigarettes and (2) it must be acceptable to smokers and encourage them to switch, while neither appealing to nonsmokers nor being perceived as an alternative to quitting. 2,8
To ensure that candidate MRTPs fulfill these two conditions, they must undergo a rigorous premarket scientific assessment program that includes chemical characterization, nonclinical research, clinical research, and consumer perception and behavior studies. 8 Following market introduction, their postmarket assessment should include studies designed to regularly assess how the MRTPs are used as well as their long-term health effects. 2,8
The nonclinical assessment of candidate MRTPs requires a broad range of studies that altogether provide a comprehensive picture of the biological effects of a candidate’s MRTP in comparison with those of cigarettes and cessation. This involves the use of in vitro toxicology and biological models of the human cardiovascular system 11,12 and respiratory tract 13 –15 and may require inhalation studies conducted in vivo. Across all studies, end points reflective of the complex, multidimensional etiology of smoking-related diseases should be measured and include markers of HPHC exposure, exposure response (xenobiotic metabolism), oxidative stress, DNA damage, and inflammation. 16 –18 Similarly, clinical studies should include markers of HPHC exposure and exposure response, as well as clinical risk markers that are affected by smoking and diminish following cessation; a study design incorporating composite primary end points may also be considered. 19
Designing a program to assess candidate MRTPs involves several challenges. These include (1) the selection of biological systems that adequately mimic key aspects of the biology affected by smoke exposure, (2) the selection of the most meaningful and appropriate markers and end points that are both affected by smoking and are reversed following cessation, (3) the selection of exposure modalities and dosimetry in nonclinical studies that would be representative of human exposure, 14 (4) the characterization and identification of the animal biology that best reflects human biology in the context of smoking and cessation, and (5) how to use experimental data to bridge product variants. Recently, at the 53rd Congress of the European Societies of Toxicology, five presentations were given at a symposium dedicated to assessing candidate MRTPs, validating harm reduction, and promoting regulatory acceptance of the assessment methods. A summary of each presentation follows.
Heated tobacco products: Nonclinical and clinical assessment
Manuel C Peitsch, Philip Morris International (PMI), Neuchâtel, Switzerland
The presentation focused on the previously described risk framework and multistep approach to the scientific assessment of candidate MRTPs and discussed the role of systems toxicology in MRTP risk assessment. 8
The risk framework for candidate MRTP assessment is defined by epidemiological evidence. 8 The harm and risk of smoking-related disease is caused by exposure to the HPHCs emitted by combustible cigarettes. Epidemiological studies have provided overwhelming evidence that the risk of smoking-related disease rises in a dose- and time-dependent manner with continued exposure to HPHCs, as illustrated by the red line in Figure 1. Epidemiological studies have also demonstrated that harm and the risk of tobacco-related disease can be dramatically reduced by smoking cessation (green line in Figure 1). Smoking cessation effectively eliminates the cause of smoking-related disease development and progression and allows, over time, for a normalization of the molecular, cellular, and tissue functions affected by smoking. Smoking cessation is the best way for a smoker to reduce the risk of harm and smoking-related disease and is the gold standard for MRTP assessment. 20 Therefore, a significant reduction in harm and disease risk should be achieved by products that significantly reduce or eliminate exposure to HPHCs (yellow lines). Furthermore, the closer the biological effects of a candidate MRTP are to those induced by cessation, the higher its potential to reduce the harm and risk of tobacco-related disease. 8
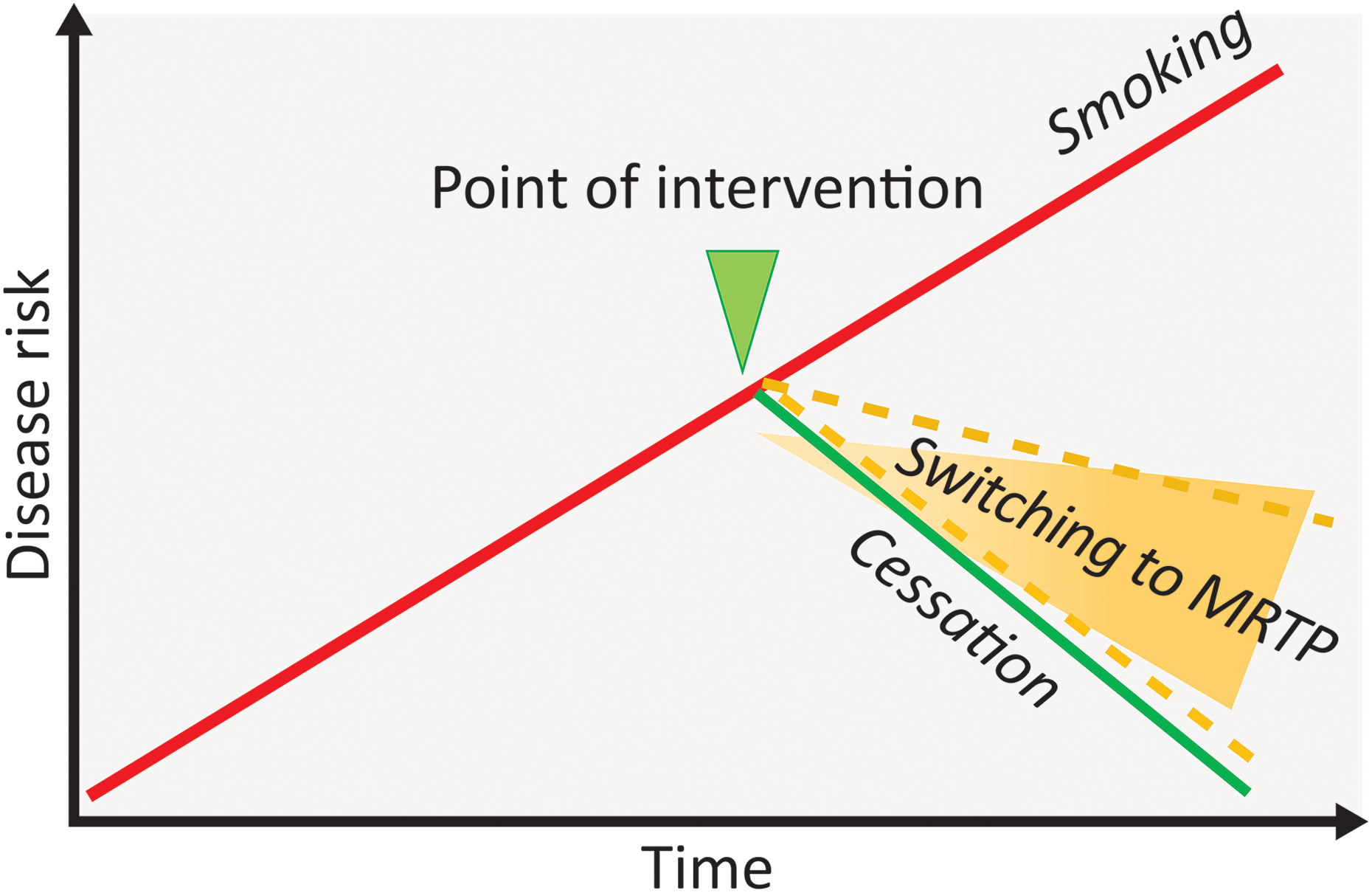
Framework for assessing MRTPs. Conceptual depiction of the cumulative risk of smoking (red line) and the effect of cessation (green line) over time. These represent the two boundaries for the assessment of a candidate MRTP (yellow lines). The upper boundary compares switching to an MRTP and continuing to smoke cigarettes; the lower boundary compares switching to an MRTP and smoking cessation. The straight lines used in this figure are for illustration purposes only, as the accumulation of disease risk and the reduction upon cessation or switching to an MRTP follow different trajectories for specific diseases and health outcomes. Reproduced with permission from Smith et al. 8 MRTPs: modified-risk tobacco products.
To assess candidate MRTPs, we have developed a generally applicable assessment program integrating seven steps of assessment. This assessment program provides five levels of evidence, which, taken together, form the totality of the evidence necessary to evaluate whether a candidate MRTP significantly reduces harm and the risk of tobacco-related disease to individual smokers and benefits the population as a whole (Figure 2). 8
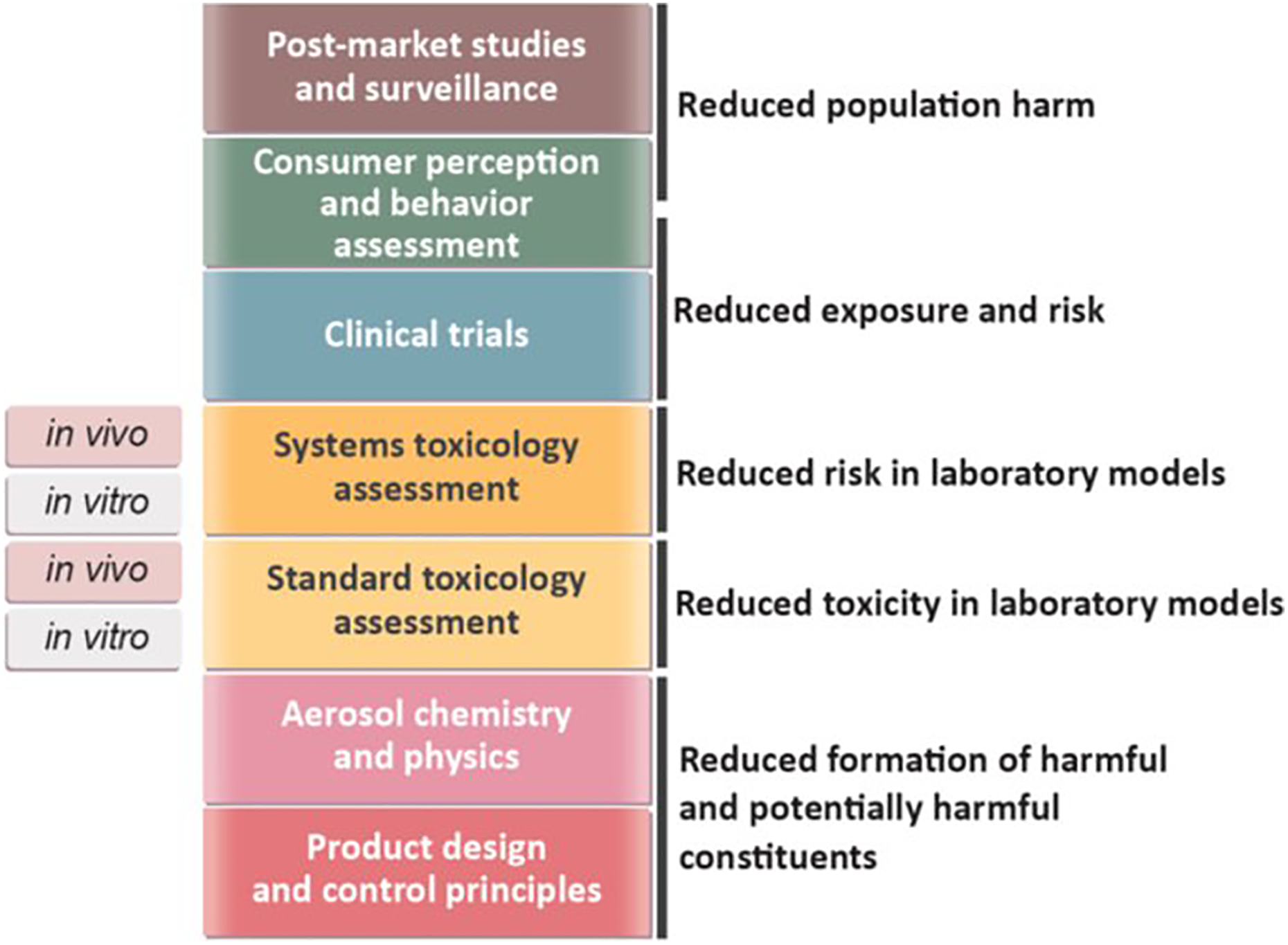
MRTP assessment program. Seven steps of assessment yield five levels of evidence. Taken together, these levels of evidence provide scientific support to demonstrate that a novel product significantly reduces harm and the risk of tobacco-related disease to individual smokers, and benefits the health of the population as a whole (both smokers and nonsmokers). Reproduced with permission from Smith et al. 8 MRTP: modified-risk tobacco product.
In the absence of premarket epidemiology data, systems toxicology can provide a solid basis for risk assessment. The primary objective of Philip Morris International’s (PMI) systems toxicology assessment was to evaluate whether use of the candidate MRTP, the Tobacco Heating System (THS), reduced the risk of disease in laboratory models. The secondary objective was to evaluate whether switching from cigarette smoke exposure to THS aerosol exposure led to positive changes that approached those caused by smoking cessation in laboratory models.
Accordingly, PMI conducted systems toxicology studies employing several in vitro and in vivo systems, including animal models of disease. These studies provided comparative data on the effects of THS aerosol on a broad range of disease-associated mechanisms affected by cigarette smoke, 21 –23 including inflammation, cell stress, cell proliferation, DNA damage, and apoptosis. 22,24 In vitro systems toxicology studies were conducted with organotypic buccal, bronchial, and nasal epithelial tissue cultures exposed to whole reference cigarette smoke and whole THS aerosol at the air–liquid interface, 25 while in vivo switching studies used murine models of cardiovascular and chronic obstructive pulmonary disease (COPD). 23
The presentation then summarized key results showing that the reduced emission of HPHCs in THS aerosol leads to a reduced impact on all biological mechanisms affected by cigarette smoke in vitro, 25 and that switching from cigarette smoke exposure to THS aerosol exposure reduces the risk of disease in laboratory models in a way that approaches the effects of cessation. 23
The MRTP assessment program also includes clinical studies to assess (1) the nicotine pharmacokinetics of a candidate MRTP, 26 (2) the reduction in HPHC exposure among study participants who switch from cigarette smoking to THS use, 27 –29 and (3) the effect of switching on clinical risk markers. Results of several clinical studies have been published previously. 30 The critical roles of perception, behavior, and postmarket studies were also described. 8
Scientific peer review is an important component of quality control in science and is typically managed by the editorial office of scientific journals. However, substantiation of risk reduction must be assessed by regulators and the public health community to determine the impact of the product. Independent verification of methods, tools, and study outcomes requires both data transparency and an anonymous, crowd-based approach. This includes publishing in peer-reviewed journals, providing study plans and reports for independent review, and sharing methods and data. Toward this end, the INTERVALS platform (http://intervals.science) and the sbvIMPROVER program (https://www.sbvimprover.com/) developed by PMI were introduced. The sbvIMPROVER program allows the conduct of crowd-sourced verification of methods and study results. This approach aims to provide a measure of quality control of industrial research and development by independent verification of methods and results. 31 While this approach was first developed to verify methods and tools in systems biology, it was later extended to include anonymous in-depth reviews of studies through a double-blind process established by SciPinion (https://scipinion.com). The INTERVALS platform allows proactive sharing of the data from assessment studies to enable data analysis by independent third parties. 32 This platform prototype was built to share results from in vivo inhalation studies and in vitro studies conducted by PMI. 10,21
Nonclinical and clinical assessment of an e-cigarette product
Christopher Proctor, British American Tobacco, London, UK
Over the last decade, there has been a substantial increase in the use of e-cigarettes, resulting in the availability of a large variety of devices and accompanying e-liquids. E-cigarette design has evolved rapidly, and products can vary from “cigalike” devices that resemble cigarettes to open-tank systems, closed-tank systems, and box modular units. E-cigarettes are devices that heat an e-liquid (typically containing nicotine, propylene glycol, glycerol, and flavors) to generate a simple aerosol that reportedly contains substantially lower levels of toxicants than cigarette smoke, although both products deliver nicotine in a similar manner. 33,34
E-cigarettes have the potential to be a reduced-risk product (RRP), and there is a growing consensus on their potential to support tobacco harm reduction. 7 Public Health England 35 has stated that e-cigarettes are approximately 95% less harmful than smoking, the UK Royal College of Physicians has supported the use of e-cigarettes as a substitute for smoking, 36 and Cancer Research UK has stated that e-cigarettes are significantly safer than tobacco products. 37 As this new product category evolves, e-cigarette product standards too are evolving to safeguard consumers from poorly manufactured products, 38,39 supported by quality design and appropriate testing.
British American Tobacco (BAT) has outlined a three-step process (emissions, exposure, and risk) to support an assessment framework for evaluating the risk profile of e-cigarettes at the individual and population levels. 7 The nonclinical studies (first step) assess product design stability (useful life and shelf life) and chemical and physical characteristics (quantification of aerosol components and environmental emissions). Clinical studies (second step) assess individual exposure and risk using pharmacokinetics, consumer behavior, and biomarkers of exposure. The population studies (third step) assess the risk relative to cigarettes on a population level and include consumer perception studies and postmarket surveillance and measurement of biomarkers of biological effect. The use of in silico and in vitro models can support the assessment across all steps, including in vitro regulatory toxicology assays (e.g. the Ames test and neutral red uptake assay), in vitro disease models, computational toxicology (margins of exposure, mode of action studies, and physiologically based pharmacokinetics models), and a systems science approach (biological perturbations measured using transcriptomics). In each phase, international standards and approved methods are applied whenever possible. Moreover, the implementation of alternative methods is emphasized to reduce and potentially eliminate the need for animal testing. 7
Using a commercially available closed-system e-cigarette, BAT has conducted a series of studies as outlined in the assessment framework. Chemical characterization showed that toxicant levels in e-cigarette aerosol were reduced by 92–99% compared with levels in cigarette smoke. 34,40 In vitro toxicological assays, 41 –43 disease modeling techniques, 44 –47 and toxicogenomic evaluations 48,49 have demonstrated significant reductions in the impact of e-cigarette aerosol compared with that of smoke from a reference cigarette. 31 Studying consumer puffing behavior ensures that laboratory machine-testing data are similar to real-world use, and a recent study reported that this can vary across a variety of e-cigarette products. 50 Two pharmacokinetic studies assessed nicotine uptake in the blood of naïve and experienced e-cigarette users. Naïve users were found to have reduced nicotine uptake with e-cigarettes in comparison with cigarettes, while experienced vapers had similar nicotine uptake with e-cigarettes in comparison with cigarettes. 51 Clinical studies can evaluate changes in biomarkers of exposure and effect in smokers who switch to e-cigarettes, 52 and studies have shown that switching from smoking to e-cigarettes reduces exposure to toxicants (Figure 3). 53 Population modeling can assess the potential population health effects of e-cigarettes in the absence of epidemiology, supporting postmarket surveillance. In a recent study using publicly available data and a systems dynamic modeling approach, an overall public health benefit effect was observed with the introduction of e-cigarettes over a 50-year period. 54
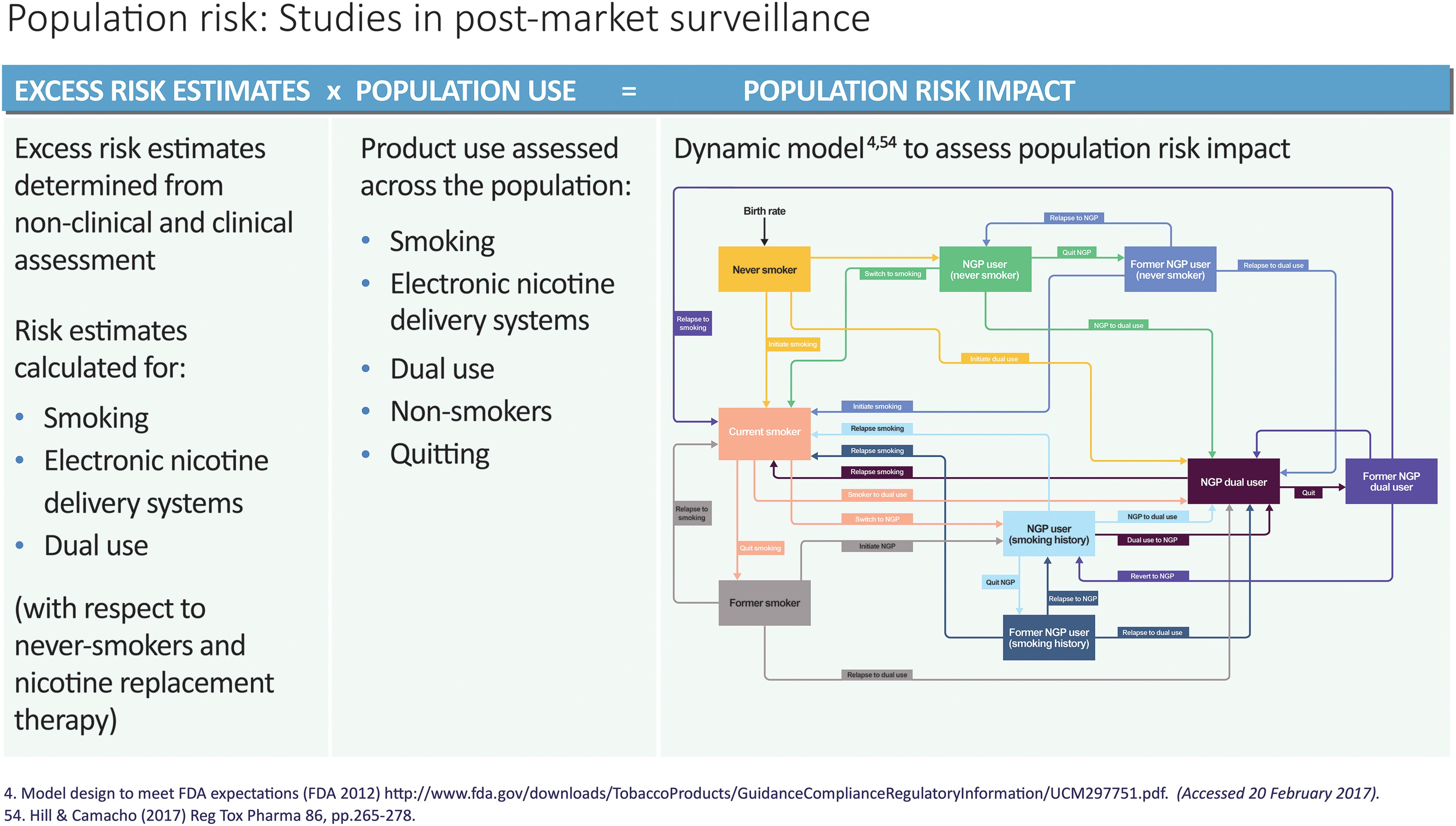
Population model illustrating the introduction of an e-cigarette to market. Use status (nonsmoker, smoker, former smoker, dual e-cigarette, and cigarette user) are represented as stocks and the probabilities of changing user status as transitions or flows. Excess risk estimates can be determined from nonclinical and clinical assessment for each of the stocks. To calculate the impact on population health, subjects would be surveyed once the product had been on the market for 3–6 months, and placed into the stocks as defined in the model. 54 NGP: next-generation product.
With the pace of e-cigarette innovation and shorter product lifecycles, the ability to maintain product and safety standards should match the incremental improvements. Using a bridging framework, as adopted by the pharmaceutical industry using a “biosimilars” approach, would enable this. 55 Foundation data sets collected on the original product variant (“reference”) can be modified on a “need” basis to allow bridging to the new variant (“similar”).
In summary, developing toxicological screens for consumer safety across the wide range of e-cigarette devices and liquids has become particularly important and is leading to the evolution of product standards. The proposed framework can generate foundational data sets on an original reference product meeting regulations, public health, and consumer needs while supporting product innovation, enabling data bridging between product variants.
Pathway to regulatory submission: The Swedish Match story
Thord Hassler, Swedish Match North America, Richmond, VA, USA
The moist powder tobacco products known as snus offer a reduced risk compared with dry snuff, moist snuff, or chewing tobacco; they are also more discreet and do not require spitting. Swedish Match is the first (and to date, only) company to receive a Premarket Tobacco Application (PMTA) order from the US FDA for modified-risk, moist powder tobacco products. The order was based on product standard and quality control procedures, product chemistry, and toxicological data. The company also has a pending MRTPA. If approved, this would be the first tobacco product deemed as lower risk by the FDA. Approval of both processes requires a finding by the FDA that the candidate product is “appropriate to the protection of the public health.” 56 The PMTA is mandatory for a recently introduced product to remain on the market, and the MRTPA allows a company to make advertising claims for a product that has been determined to be of modified risk.
In the PMTA decision document (the Technical Project Lead (TPL) report), the FDA acknowledged the significance of the “Swedish experience” (referring to extensive Swedish longitudinal cohort studies) but emphasized that the “top-line reasons” for awarding the PMTA related to how the products are manufactured, resulting in low levels of nitrosamines (N-nitrosonornicotine (NNN) and nicotine-derived nitrosamine ketone (NNK)) and other HPHCs. The first of the reasons cited in the TPL report is that the products are “produced with a voluntary, proprietary standard using acceptable manufacturing processes”; this standard is the company’s GOTHIATEK® product standard. 57
GOTHIATEK is a voluntary quality standard developed by Swedish Match that sets maximum levels for selected constituents in smoke-free tobacco products (Table 1). These constituents include metals, polycyclic aromatic hydrocarbons, tobacco-specific nitrosamines, aldehydes, pesticides, and plant-derived toxins. Finished products should not exceed the maximum allowable levels of these constituents, whether they originate in the environment or the tobacco plant. These standards are lower than the maximum levels prescribed by the Swedish Food Act or the World Health Organization recommendations for ST products. In addition, GOTHIATEK outlines standards for raw materials, manufacturing, and consumer package labeling.
Standards for maximum levels of harmful and potentially harmful constituents in tobacco products.a
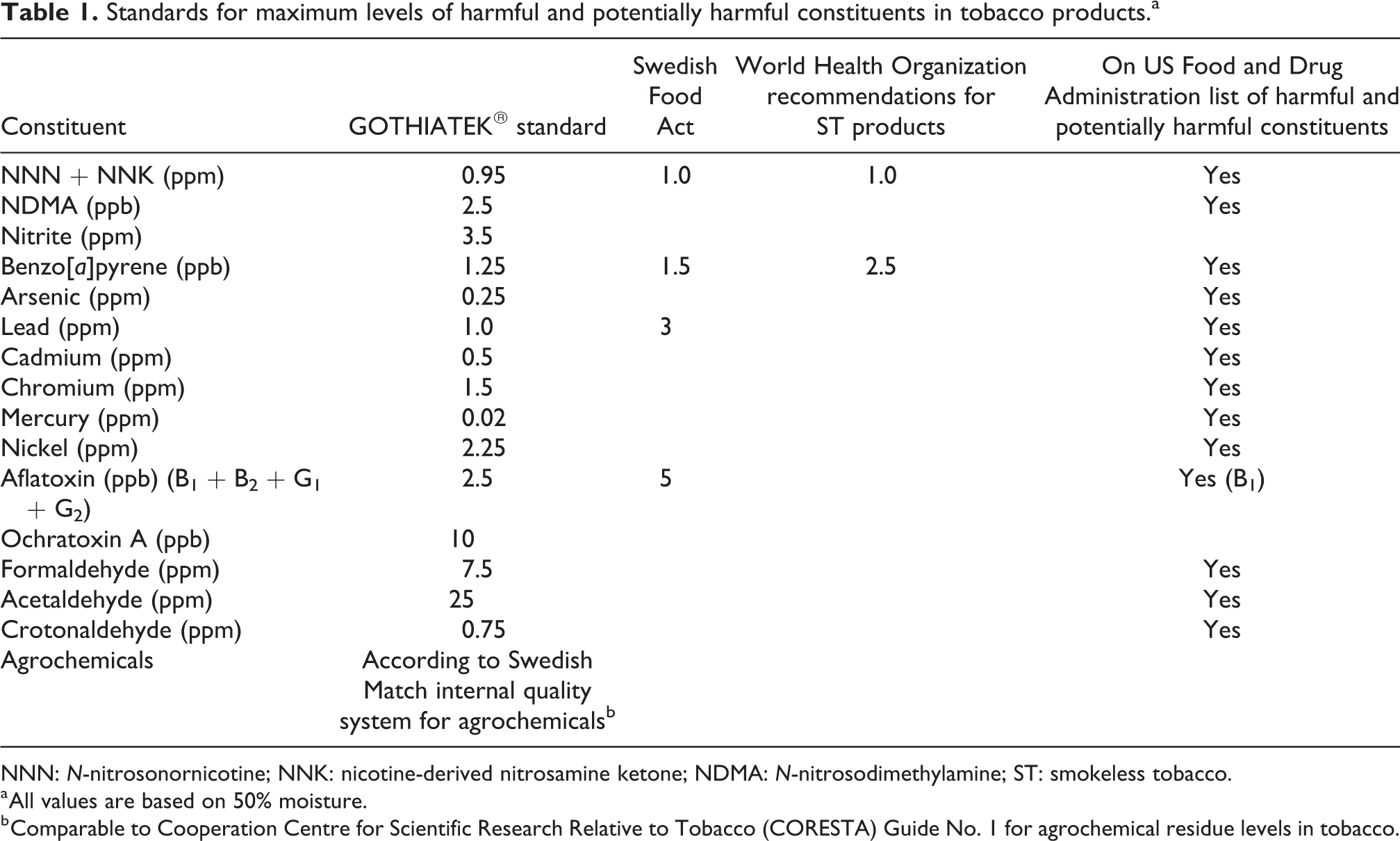
NNN: N-nitrosonornicotine; NNK: nicotine-derived nitrosamine ketone; NDMA: N-nitrosodimethylamine; ST: smokeless tobacco.
a All values are based on 50% moisture.
b Comparable to Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) Guide No. 1 for agrochemical residue levels in tobacco.
The TPL report emphasized the importance of nitrosamines in determining the risk profile of a product, stating that they are among the most carcinogenic constituents in tobacco products, and reduction of nitrosamine levels could reduce cancer risk. The report highlights that “The proposed products contain significantly lower levels of NNN and NNK compared to over 97% of the ST (smokeless tobacco) products currently on the US market.” 57 According to the FDA, this results in significant risk reduction: “Assuming persons who would have used other US ST products use these products instead, an individual using these products with reduced NNN levels could decrease the excess cancer risk by 90% compared to use of moist snuff (market share: 82%), 67% compared to use of chewing tobacco (market share: 15%), 38% compared to use of US-style snus, and 92% compared to use of dry snuff.”
The FDA has stated an interest in pursuing product standards for HPHCs, and in January 2017 introduced a draft rule proposing an NNN limit in ST products. 58 The draft rule specifically cites GOTHIATEK and describes how it establishes limits for NNN and other HPHCs. Thus, it is reasonable to assume that GOTHIATEK will be instrumental as the FDA considers how to regulate HPHCs through a product standard approach.
The GOTHIATEK ethos also provided the foundation for Swedish Match’s research into low-risk, novel products with substantially lower levels of HPHCs. The company anticipates preparing PMTAs for such new products, which would require the submission of substantial scientific evidence, and perhaps most importantly, comparing the new product to a product already on the market. Swedish Match will compare new products to the snus products that have already successfully completed the PMTA process and demonstrate that the HPHC levels in the new products are comparable with, or lower than, those in snus.
Substantiating the harm reduction potential of reduced-risk products (RRPs)
Riccardo Polosa, Dipartimento di Medicina Clinica e Sperimentale, University of Catania, Catania, Italy
The World Health Organization’s Framework Convention on Tobacco Control recognizes harm reduction as an integral part of a comprehensive approach to reducing tobacco smoking. 59 To integrate harm reduction strategies into existing tobacco control policies and to accelerate the decline in smoking prevalence, widespread access to RRPs is paramount.
Tobacco harm reduction refers to policies seeking to prevent or reduce the damage caused by smoking in people unable or reluctant to stop, rather than aiming at complete abstinence from tobacco use. 60 Tobacco harm reduction is based on the concept that smokers smoke for nicotine but die from tar. 61 In fact, nicotine is unlikely to contribute significantly to the development of smoking-related diseases. 62
Morbidity and mortality from smoking is known to result from cellular and physiological events sustained by exposure to high levels of thousands of combustion products (including toxic chemicals and carcinogens) in cigarette smoke, rather than from nicotine itself. Although nicotine may not be absolutely harmless, at concentrations typically found in tobacco products, it is relatively safe, 63 is not a carcinogen, 64 and does not contribute to respiratory or cardiovascular disease. 18 Removing combustion using alternative products is the key to harm reduction; the level of harm reduction depends on the mechanisms of the delivery system. Consequently, noncombustible nicotine sources (e.g. NRTs, ST, e-cigarettes, and tobacco heating products) are likely to produce far fewer toxicants. Noncombustible nicotine sources have been proposed as alternatives to conventional cigarettes for smoking harm reduction 65 ; across the risk spectrum of nicotine-containing products, they feature the lowest level of risk. 10 Although not risk-free, studies of RRPs have shown substantial reductions in exposure to a wide range of chemicals in smoke. 27,66,67
Figure 4 shows a matrix of reduced-risk nicotine delivery systems, which can be divided into four groups: heated pure nicotine-based products (e.g. e-cigarettes), unheated pure nicotine-based products (e.g. nicotine gum), heated tobacco-based products (e.g. vaporizers), and unheated tobacco-based products (e.g. chewing tobacco).

Matrix of reduced-risk nicotine delivery systems.
It is important to note that exposure reduction may not always translate to risk reduction or harm reversal. Even for the well-established benefits of quitting, such as a reduction in coronary heart disease, the risk reduction is not significant until several years after a smoker quits. 68 In addition, a general lack of response in health effect indicators to a reduction in toxicant exposure has been highlighted in clinical studies when switching from conventional cigarettes to an RRP prototype. 16 Investigating changes in robust and validated health effect indicators in smokers with preexisting disease may help to substantiate the harm reduction potential of RRPs. 69
This approach is particularly relevant for smoking-related diseases such as COPD. There is now emerging evidence to suggest that substitution of conventional tobacco cigarettes with noncombustible nicotine sources (e.g. e-cigarettes) can ameliorate clinical and functional outcomes in patients with COPD. 70 The challenge lies in identifying the risk reduction indicators of adequate sensitivity and reproducibility. Some biomarkers of potential harm shown to be effective in assessing the impact of tobacco exposure are white blood cell counts and levels of high-density lipoprotein, high-sensitivity C-reactive protein, and serum cotinine. 17 Comparative studies with routine physiological measures, such as blood pressure, spirometry, and walking tests, can also assist in discerning indicators of risk reduction.
Identifying appropriate biomarkers of risk reduction is of particular importance for future trials, as the implementation of multiple end points must be carefully considered when designing a study. For data to be statistically robust, adjustments for multiplicity must be considered, which may require very large sample sizes to draw meaningful conclusions. Alternatively, a study may instead utilize a composite primary end point. 19
Building pathways for regulatory acceptance of alternative methods for tobacco product assessment
Erin Hill, Institute for In Vitro Sciences, Gaithersburg, MD, USA
The Institute for In Vitro Sciences (IIVS) was founded in 1997 as a nonprofit laboratory to promote the use and acceptance of in vitro methods for toxicology. IIVS staff have substantial experience in optimizing, standardizing, and validating in vitro methods and have been successful in bringing together industry and regulators to identify nonanimal testing approaches that meet industry needs and regulatory requirements. One such collaboration resulted in an in vitro approach to assess the eye irritation potential of registered pesticide products through the US Environmental Protection Agency. 71 The success of this collaboration rested on the ability of the industry to engage with the regulatory authorities to outline a program that would be useful to both parties. Additionally, this program serves as an example of a regulatory agency accepting a “fit-for-purpose” validation study that met the agency’s specific requirements.
Regulations for conventional and next-generation tobacco products in the United States, and the potentially large numbers of animals that could be used to meet regulatory requirements, led IIVS to expand its laboratory capabilities and outreach initiatives to help identify useful in vitro respiratory methods and approaches to assess the risk associated with these products. Given the current climate in which collaboration among tobacco companies is often difficult, and where opportunities to engage with the FDA’s Center for Tobacco Products (CTP) are limited, IIVS created a workshop series to promote the identification of in vitro approaches for tobacco products. To date, the workshops have focused on in vitro COPD models 72 and dosimetry tools 14 for inhaled tobacco products. The workshops explored in detail the topics such as inflammation and oxidative stress, ciliary dysfunction and ion transport, goblet cell hyperplasia and mucus production, and dosimetry considerations necessary for in vitro modeling. Most importantly, the workshops brought together stakeholders representing regulatory agencies, academia, and industry to explore how in vitro methods could be used in the context of the research priorities articulated by the CTP. 73
The workshops revealed that a number of in vitro methods were available and in use by industry to model respiratory events (Figure 5). Three commonly used end points—ciliary beating frequency, mucus production, and goblet cell hyperplasia—have been recommended by workshop participants for further investigation and optimization in proof-of-principle multi-laboratory studies that would employ commercially available, reconstructed human airway models. 72
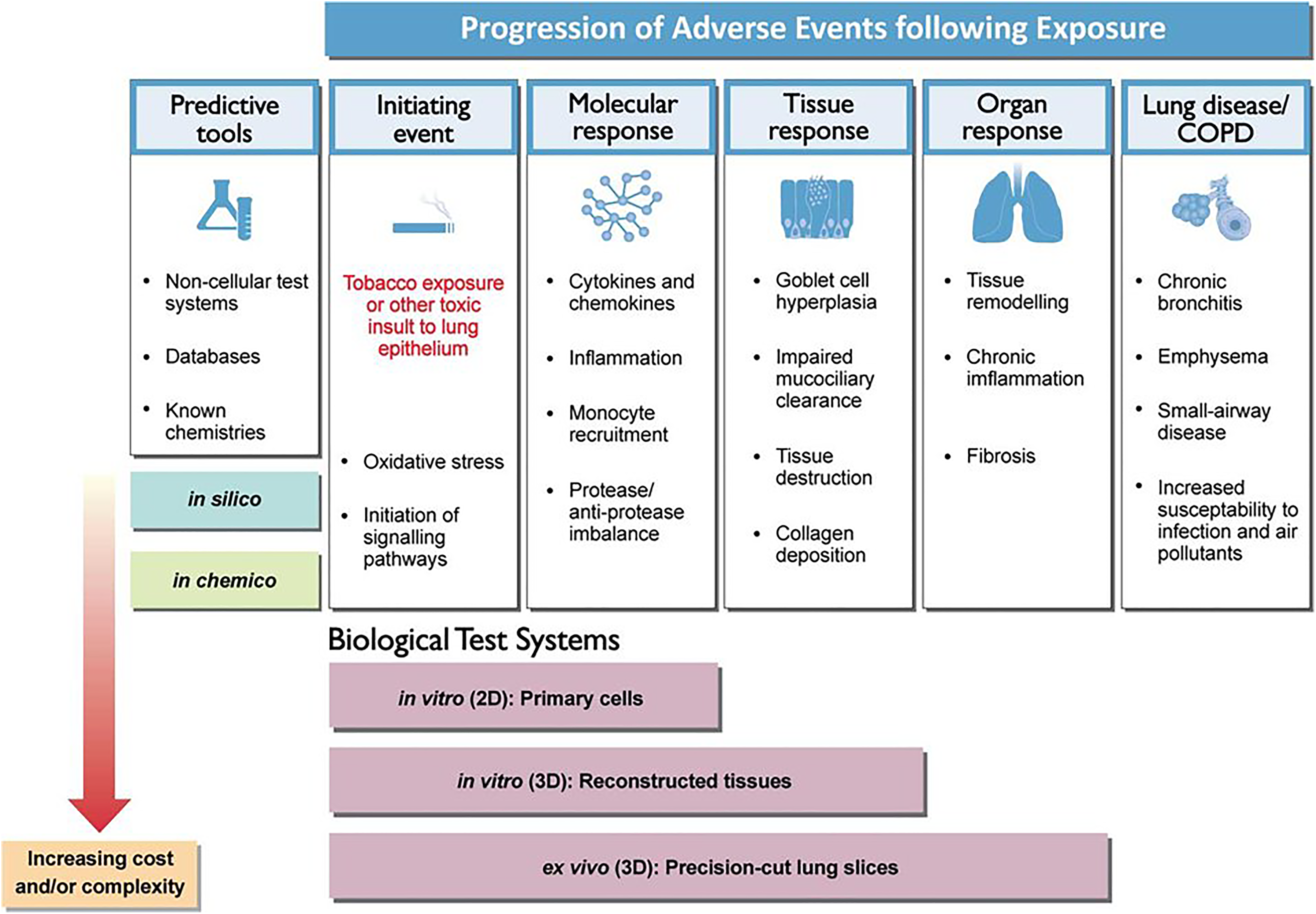
Adverse respiratory events and choice of model systems.
Although US tobacco regulations do not currently require animal testing of next-generation tobacco products, it is possible that the FDA may request animal testing to demonstrate harm reduction. Animal models are often limited in their ability to predict effects in humans, and those used to assess conventional tobacco products may not be applicable to next-generation tobacco products. US regulatory authorities and industry are actively looking for ways to incorporate more predictive, mechanistic, nonanimal models into toxicity testing and the regulatory review process. 74 Collaborative approaches involving industry and regulatory scientists, with large input from the scientific community, will be required to standardize in vitro methods to make them suitable for regulatory acceptance, thereby maximizing their use in tobacco regulations.
Conclusions
Tobacco harm reduction is possible with the adoption of MRTPs, and assessment strategies that incorporate nonclinical, clinical, and systems toxicology sciences can yield robust data to meet scientific, public health, and regulatory objectives. Standardizing the assessment methodology would optimize data collection and interpretation and facilitate data reporting for regulatory purposes. Independent verification would require a collaborative approach by the scientific community and regulatory bodies; this is critical to the development of a comprehensive framework for risk assessment.
Footnotes
Authors’ note
Manuel C Peitsch and Julia Hoeng are employees of Philip Morris International. Christopher Proctor and Marianna Gaca are employees of British American Tobacco (Investments) Ltd., Group Research and Development. Thord Hassler is employee of Swedish Match European Division, Gothenburg, Sweden. Erin Hill is employed by IIVS which accepts contributions from the tobacco industry. Riccardo Polosa's research in harm reduction has not been supported by the tobacco industry.
Acknowledgements
The authors thank Dean Meyer, PhD, of Edanz Medical Writing for providing writing support, which was funded by Philip Morris International.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Hayes received fees from Philip Morris International for his contribution (critical review for intellectual content) to this manuscript. Institute for In Vitro Sciences provides in vitro testing services and accepts contributions from a variety of industries, including tobacco companies.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
