Abstract
TrueBroc® Glucoraphanin-Rich Broccoli Seed Extract (TrueBroc®), is a hot water extract of Brassica oleracea var. Italica Plenck (Brassicaceae) seeds. The broccoli seeds are non-genetically modified and are tested and selected for their specific glucosinolate content. TrueBroc® contains greater than 99% of the aliphatic glucosinolate glucoraphanin and its precursor glucoerucin in the glucosinolate fraction. A battery of toxicology assays was conducted on TrueBroc® which includes in vitro and in vivo genotoxicity studies, an acute oral toxicity study in rats, a subchronic toxicology study in rats. The result from the subchronic rat study establishes a NOAEL of 1500 mg/kg/day.
Introduction
TrueBroc® is a hot water extract of Brassica oleracea var.iItalica Plenck (Brassicaceae) seeds. The broccoli seeds are non-genetically modified and are tested and selected for their specific glucosinolate content. Glucosinolates (GSLs) are a structural class of plant secondary metabolites that are characteristic of the members of the family Brassicaceae, whose members include cruciferous vegetables such as broccoli, cabbage, watercress, Brussels sprouts, cauliflower, turnip, mustard, radish, and horseradish. Major classes of glucosinolates can be grouped into four categories according to their sidechain (“R-”) group which consists of either an aliphatic group (Class 1), an allyl (unsaturated) group (Class 2), an aromatic (e.g., phenylethyl) group (Class 3) or an indole group (Class 4) (GRAS Notice (GRN) 496). 1 TrueBroc® contains, in the glucosinolate fraction, greater than 99% of the Class 1 aliphatic glucosinolates glucoraphanin and glucoerucin and the overall glucosinolate level and profile are similar to those found in broccoli sprouts and mature broccoli. The remaining 0.4% of the glucosinolates in TrueBroc belong to class 2 (sinigrin, gluconapin, epigoitrin, and progoitrin), Class 3 (gluconasturtiin), and Class 4 (glucobrassicin). In addition, analytical characterization of the non-glucosinolate portion of the extract shows that it contains components that are widely found in foods, such as small organic acids, low molecular weight phenolics, and simple sugars. All of the processing aids and final product additives (maltodextrin, silicon dioxide) used in broccoli seed extract comply with 21 CFR and Food Chemicals Codex monographs.
Glucosinolates are hydrolyzed by the enzyme myrosinase to isothiocyanates. Myrosinase, a ß-thioglucosidase, in the intact plant is physically separated from its substrate, however, following plant tissue injury, such as biting or chewing or cutting, glucoraphanin comes in contact with the enzyme. The production process for TrueBroc®, however, includes a critical step that allows for heat penetration of the seed that inactivates the endogenous myrosinase enzyme which would convert the glucosinolates to their respective isothiocyanates (e.g., glucoraphanin to sulforaphane).2–5 The inactivation of myrosinase in the production of the TrueBroc® product is an important consideration in the evaluation of safety of its ingestion because glucoraphanin is chemically stable and biologically inert and the inactivation of the myrosinase reduces conversion of glucoraphanin to the active isothiocyanate, sulforaphane. Certain human gut flora microbes exhibit myrosinase-like activity and hydrolysis of the glucosinolates in TrueBroc® to isothiocyanates occurs to a small extent. Without exogenous myrosinase, on a population basis, only about 10% of glucoraphanin is converted to sulforaphane by the bacterial microflora.3,6
TrueBroc® has been determined to be Generally Recognized As Safe (GRAS) as an ingredient in teas, energy drinks, thirst quenching drinks, and non-carbonated flavored waters at intakes up to 554 mg/person per day (GRAS Notice (GRN) 496). 1 The safety of intake of TrueBroc® in GRN 496 relied on a determination that the amounts of glucosinolates from the proposed uses (up to 554 mg/day) were safe because they were equivalent to or less than exposure from normal diet and consistent with global authoritative dietary recommendations. In addition, these safe levels of intake were corroborated by a critical review of human and animal studies documenting the metabolic fate, physiologic activity, and lack of adverse health effects associated with ingestion of levels of glucosinolates exceeding the proposed levels of ingestion.
GRN 496 did not include results from toxicology studies nor a derivation of an Acceptable Daily Intake (ADI) on the basis of extrapolation from toxicology data. A publication subsequent to GRN 496 provided the results for TrueBroc® from three genotoxicity experiments, including an Ames, in vivo mouse micronucleus, and in vivo mouse sperm abnormality assay in addition to an acute oral toxicity test and 30-day feeding study in rats. 7 In this publication, authors note that no genotoxicity was observed in these studies, the LD50 was >10 g/kg/day and no toxicologically significant effects were noted at 3.0 g/kg/day, the highest dose tested, at 30 days of administration in rats.
The objective of this paper is to summarize the results from a battery of toxicology assays conducted on TrueBroc® which includes in vitro and in vivo genotoxicity studies, an acute oral toxicity study in rats, a 90-day subchronic toxicology study in rats. The result from the subchronic rat study establishes a NOAEL of 1500 mg/kg/day.
Materials and methods
Test material
The test substance, identified as TrueBroc® is characterized as an aqueous extract of B. oleracea var. Italica Plenck seeds that are concentrated and spray dried with maltodextrin and silicon dioxide. The primary constituents of TrueBroc® are the broccoli glucosinolates glucoraphanin and glucoerucin, protein, ash, moisture, a minor amount of fat, and traces of other glucosinolates. Additives used for technological purposes include maltodextrin and silicon dioxide. The batch of TrueBroc® used in the following toxicology studies contained 11.28% glucoraphanin content. Homogeneity and concentration verification of the glucoraphanin content of test article was performed using a validated HPLC method.
Study compliance
The following studies were performed at Product Safety Laboratories (Dayton, NJ), an Organization for Economic Co-operation and Development (OECD) good laboratory practices (GLP) and AAALAC accredited facility using OECD-compliant methodologies: bacterial reverse mutation test (OECD No. 471, 1997), mammalian erythrocyte micronucleus test (OECD No. 474, 2016), acute oral toxicity test in rats (OECD No. 425, 2008), range-finding toxicity study in rats (OECD No. 407, 2008), and a subchronic oral toxicity study in rats (OECD No. 408, 2018). The in vitro mammalian chromosome aberration test in Chinese hamster V79 cells was performed at Eurofins Munich in compliance with OECD methods (OECD No. 473, 2016).
Bacterial reverse mutagenicity (ames assay) (OECD No. 471, 1997)
The Ames test was conducted with TrueBroc® at levels of 1.58, 5.0, 15.8, 50, 158, 500, 1580, and 5000 μg/plate using strains of Salmonella typhimurium (S. typhimurium) and Escherichia coli (E. coli). The assay was conducted in both the presence and absence of metabolic activation using rat liver S9 purchased from Molecular Toxicology, Inc. The positive controls used for the Ames test strains without metabolic activation were as follows: 15 μg/mL sodium azide (S. typhimurium TA100 and TA1535), 10 μg/mL ICR 191 acridine (S. typhimurium TA1537), 60 μg/mL daunomycin (S. typhimurium TA100 and TA1535), and 25 μg/mL methyl methanesulfonate (MMS) (E. coli WP2 uvrA). The positive control for all tester strains used with metabolic activation was 100 μg/mL 2-aminoanthracene (2AA). The test substance was found to be soluble in sterile water, which was used as the vehicle control.
The initial (main) test followed the plate incorporation method; plates were prepared in triplicate at each experimental point. The confirmatory test employed the pre-incubation modification of the plate incorporation test. The study design for the confirmatory, test, including strains and dose levels was the same as for the initial (main) test.
Toxic effects of the test substance are indicated by the partial or complete absence of a background lawn of non-revertant bacteria or a substantial dose-related reduction in revertant colony counts compared with lower dose levels and concurrent vehicle control taking into account the laboratory historical control range.
For each experimental point, the Mutation Factor (MF) was calculated by dividing the mean revertant colony count by the mean revertant colony count for the corresponding concurrent vehicle control group. Results were considered positive if MF ≥ 2 for strains TA98, TA100, and WP2 uvrA or MF ≥ 3 for strains TA 1535 and TA 1537, with mean value(s) outside the laboratory historical control range. Otherwise, results were considered negative. The increase must be dose-related and/or reproducible, that is, increases must be obtained at more than one experimental point (at least one strain, more than one dose level, more than one occasion or with different methodologies).
Mammalian erythrocyte micronucleus test (OECD No. 474, 2016)
Based on a maximum tolerated dose of 5000 mg/kg, TrueBroc® was diluted in distilled water and administered by oral gavage to male and female Swiss albino (ICR) mice (n = 5/sex/group) at 1250, 2500, and 5000 mg/kg/day. A dose volume of 10 mL/kg was used to reduce the viscosity of the test substance formulations. Mice were 7–8 weeks old at the start of the study. Animals were group-housed in cages conforming to the size recommendations in the latest Guide for the Care and Use of Laboratory Animals 8 Distilled water was used as the negative (vehicle) control and cyclophosphamide monohydrate supplied by Sigma-Aldrich was used as the positive control; the positive control group was treated at 5 mL/kg. Animals were dosed with TrueBroc® or negative control on Days 1 and 2. The positive control was administered on Day 2 only.
All animals were observed at least twice daily for viability. Cage-side observations of all animals were performed daily during the study. Observations of the test animals were conducted at intervals appropriate to assess the onset and termination of adverse effects; the first observation was made approximately 30 min following administration. Animals were also observed during the first several hours post-dosing on Days 1 and 2 for the negative control and test substance-treated groups. Blood samples were collected from all groups, by cardiac puncture while animals were anesthetized, at 44–48 h after last treatment. Blood was processed for micronucleus analysis; the target was a minimum of 5000 immature erythrocytes per animal. Data generated (proportions of reticulocytes, micronucleated reticulocytes, and number of micronucleated immature erythrocytes observed (MIE)) were compared using paired t-tests to determine the expected changes resulting from treatment with the positive control compared to the negative control, and then the limit dose compared to the negative control. A positive result was defined as a statistically significant increase in MIE compared with the negative control group. Any such increase would be dose-related with mean values and at least two individual animal values outside the laboratory control range.
In vitro mammalian chromosome aberration test in Chinese hamster V79 cells (OECD No. 473, 2016)
The in vitro mammalian chromosome aberration assay consisted of a solubility test, a pre-experiment and two main experiments. The solubility test and pre-experiments were conducted prior to Experiments I and II to determine the compatibility of the test article with the culture medium, the cytotoxicity of the test article with and without metabolic activation, and the concentrations to be used in Experiments I and II. A pre-experiment was conducted under identical conditions as the main Experiment I. The following concentrations were tested with and without S9 mix: 10, 25, 50, 100, 250, 500, 1000, 2500, and 5000 μg/mL TrueBroc®.
In the first main experiment, Chinese hamster V79 cells were treated with different concentrations of TrueBroc® with and without S9 metabolic activation for 4 h. The S9 liver microsomal fraction was obtained from Trinova Biochem GmbH, Giessen, Germany. In the second main experiment, V79 cells were treated with different concentrations of TrueBroc® without metabolic activation for 21 h. On the basis of data and observations from the pre-experiment, the following concentrations were selected for the main Experiment I: 250, 500, 1000 (-S9 at 4 and 21 h) and main Experiment II: 250, 500, 1000, 2500 (-S9 at 21 h).
The positive controls ethylmethanesulfonate (supplied by Sigma-Aldrich) (600 and 900 μg/mL) and cyclophosphamide (supplied by Sigma-Aldrich) (0.83 μg/mL) were used. Due to observed precipitation and cytotoxicity at doses greater than 1000 μg/mL in the first main experiment, this experiment was performed with 250, 500, and 1000 μg/mL TrueBroc®. Similarly, precipitation and cytotoxicity were observed in the second main experiment at doses greater than 2500 μg/mL; therefore, the second main experiment was performed with 250, 500, 1000, and 2500 μg/mL TrueBroc®.
Chromosomes were prepared 21 h after start of treatment with the test item. The treatment intervals were 4 h with and without metabolic activation (experiment I) and 21 h without metabolic activation (experiment II). 150 metaphases per culture were scored for structural chromosomal aberrations. Due to high numbers of cells with chromosome aberrations reported for the positive controls, the number of metaphases scored was reduced to 175 (900 μg/mL EMS), 200 (0.83 μg/mL CPA), and 150 metaphases (600 μg/mL EMS) in experiment I and II.
All slides, including those of positive, negative and (if necessary) solvent controls were independently coded before microscopic analysis. If observed, structural chromosomal aberrations, including breaks, fragments, deletions, exchanges, and chromosomal disintegration were recorded. Gaps were recorded as well but not included in the calculation of the aberration rates. To describe a cytotoxic effect the relative increase in cell count (RICC) was determined.
A positive result was determined if any of the experimental conditions examined: (a) at least one of the test concentrations exhibited a statistically significant increase compared with the concurrent negative control; (b) the increase was dose-related when evaluated with an appropriate trend test; (c) any of the results were outside the 95% control limits of the historical negative control data. When all of these criteria are met, the test chemical is then considered able to induce chromosomal aberrations in cultured mammalian cells in this test system.
Acute oral toxicity test in rats (OECD No. 425, 2008)
An acute oral toxicity test was conducted with 10–11 week-old female Sprague-Dawley rats. Prior to each dosing, experimentally naive rats were fasted overnight by removing the feed from their cages. During the fasting period, the rats were examined for health and weighed (initial). The test substance was administered by gavage as a 40% w/w mixture in distilled water. An initial limit dose of 5000 mg/kg was administered to one healthy female rat by oral gavage. Due to the absence of mortality in this animal, two additional females received the same dose level. Because these animals survived, no additional animals were tested.
The animals were observed for mortality, signs of gross toxicity, and behavioral changes approximately 30 min post-dosing, during the first several hours post-dosing and at least once daily thereafter for 14 days after dosing. Observations included gross evaluation of skin and fur, eyes and mucous membranes, respiratory, circulatory, autonomic and central nervous systems, somatomotor activity and behavior pattern. Particular attention was directed to observation of tremors, convulsions, salivation, diarrhea, and coma. Individual body weights of the animals were recorded prior to test substance administration (initial) and again on Days 7 and 14 (terminal) following dosing. Necropsies were performed on all animals at terminal sacrifice at the end of the 14-day observation period. Tissues and organs of the thoracic and abdominal cavities were examined.
Range-finding toxicity study in rats (OECD No. 407, 2008)
A range-finding toxicity study utilizing gavage administration was conducted in CRL:Sprague Dawley CD® IGS rats to establish the doses for a subsequent subchronic toxicity study of TrueBroc®. Forty healthy rats (20 males and 20 females), aged 7–8 weeks, were randomized into test groups of five males and five females per group. Dose levels of 0 (vehicle control, distilled water), 750, 1500, and 3000 mg/kg/day of TrueBroc® were selected. The high-dose was the maximum recommended dose in accordance with current guideline requirements for sub-acute and sub-chronic repeat dose administration and was not expected to cause marked toxicity. The mid- and low-dose levels were selected to derive a dose-response for any effects observed. Each dose was prepared prior to the daily dosing. The animals were individually housed and observed daily for viability, signs of gross toxicity, and behavioral changes at least once daily during the study, and weekly for a battery of detailed observations. Body weights were recorded two times during the acclimation period (including prior to administration on Day 1), and on Days 8 and Day 15 (prior to sacrifice). Individual food consumption was also recorded on Days 1, 8, and 15. Food efficiency was calculated and reported for selected intervals. All animals were subjected to a gross necropsy at study termination (Day 16).
Subchronic oral toxicity study in rats (OECD No. 408, 2018)
Eighty (80) adult Crl: Sprague-Dawley CD® IGS rats between 7 and 8 weeks of age (40 males and 40 females) were distributed into four groups (10/sex/group). Dose levels of 750, 1500, and 3000 mg/kg/day as well as a vehicle control (distilled water), were selected and administered for at least 90 days (i.e., 95 days for male rats and 96 days for female rats). An appropriate amount of the vehicle control or test substance was administered daily (7 days/week) via oral gavage to each rat. The test substance was administered as 75, 150, or 300 mg/mL w/v mixtures in distilled water. Samples of the neat test substance were collected at the beginning (Test Day 1) and at the end of the dosing phase of the study (Test Day 96) and analyzed to evaluate stability. Samples were also collected from the dose formulation mixtures to verify homogeneity (Test Day 1) and dose concentration verification (Days 43 and 96) using a validated high performance liquid chromatography method.
Animals were conditioned to the housing facilities for 6 days prior to testing. Rats were group-housed in cages which conform to the size recommendations in the most recent Guide for the Care and Use of Laboratory Animals 8 with 12-h light/dark cycles and control of temperature between 19 and 22°C. and humidity between 15 and 70%. 2016 Certified Envigo Teklad Global Rodent Diet® (Envigo Teklad, Inc.) was stored in a dedicated temperature and humidity monitored feed storage site and available ad libitum during acclimation and throughout the study, except when animals were fasted for clinical sample collections and terminal sacrifice. Filtered tap water was available ad libitum from an automatic watering access system.
During the acclimation period, the eyes of all rats being considered for study were examined by focal illumination, indirect ophthalmoscopy and, when indicated, slit-lamp microscopy.
Clinical observations
Both eyes of all animals on study were examined by focal illumination, slit lamp biomicroscopy and indirect ophthalmoscopy prior to study initiation and near completion of the dosing phase of the study for the surviving animals on Test Day 93.
All animals were observed at least twice daily for viability. Cage-side observations of all animals were performed daily during the study. Prior to the first treatment with the test substance on Test Day 1, and weekly thereafter, a detailed clinical observation was conducted while handling the animal. Potential signs noted included, but were not limited to: changes in skin, fur, eyes, and mucous membranes, occurrence of secretions and excretions and autonomic activity (e.g., lacrimation, piloerection, pupil size, and unusual respiratory pattern). Likewise, changes in gait, posture, and response to handling, as well as the presence of clonic or tonic movements, stereotypies (e.g., excessive grooming, repetitive circling), or bizarre behavior (e.g., self-mutilation, walking backwards) were also recorded.
Body weight and body weight gain
Individual body weights were recorded two times during acclimation. Test animals were weighed on Test Day 1 (prior to study start) and weekly thereafter (intervals of 7 days ±1). The animals were weighed prior to sacrifice to calculate organ-to-body weight ratios. Body weight gain was calculated for selected intervals and for the study overall.
Food consumption and food efficiency
Individual food consumption was measured and recorded to coincide with body weight measurements. Food efficiency was calculated.
Clinical pathology
Clinical pathology was performed on all surviving animals at the end of the study. Animals were fasted overnight prior to blood collection. Blood samples were collected from the sublingual vein or vena cava/abdominal aorta, under isoflurane anesthesia.
Hematology: the following parameters were measured: hematocrit; platelet count; reticulocyte count; hemoglobin concentration; red blood cell count; white blood cell and differential; mean corpuscular hemoglobin; red cell distribution width; leukocyte count; mean corpuscular volume; mean corpuscular hemoglobin concentration was calculated.
Coagulation: the following parameters were measured: activated partial thromboplastin time; prothrombin time.
Clinical Chemistry: the following parameters were measured: albumin; alkaline phosphatase, bilirubin (total); blood creatinine; calcium; chloride; cholesterol (total); fasting glucose; globulin; inorganic phosphorous; lipoprotein (high density); lipoprotein (low density); potassium; serum alanine aminotransferase; serum aspartate aminotransferase, serum protein (total); sodium; sorbitol dehydrogenase; triglycerides; urea nitrogen.
Urinalysis: the following parameters were measured: bilirubin; blood; color; clarity; glucose; ketone; microscopic urine sediment; pH; protein (total); quality; specific gravity; volume; urobilinogen.
Thyroid Hormone Assessment: the following parameters were measured: Triiodothyronine (T3), Thyroxine (T4), and Thyroxine Stimulating Hormone (TSH).
Terminal sacrifice and histopathology
At terminal sacrifice, all surviving animals were euthanized by exsanguination under isoflurane anesthesia. All animals in the study (including decedents) were subjected to a gross necropsy, which included examination of the external surface of the body, all orifices, musculoskeletal system, and the cranial, thoracic, abdominal, and pelvic cavities, with their associated organs and tissues. All gross lesions were recorded. • The following tissues (of all animals sacrificed by design) were weighed wet as soon as possible after dissection to avoid drying: adrenals (combined); brain; epididymides (combined); heart; kidneys; liver; ovaries with oviducts (combined); spleen; testes (combined); thymus; uterus. • The following tissues were weighed at least 24 h after preservation in 10% neutral buffered formalin: prostate and seminal vesicles with coagulating gland (combined); thyroid/parathyroid; pituitary. • The following organs and tissues from all animals were preserved in 10% neutral buffered formalin for possible future histopathological examination: accessory genital organs (prostate and seminal vesicles); ileum with Peyer’s patches; rectum; jejunum; salivary glands (sublingual, submandibular and parotid); adrenals; kidneys; all gross lesions; larynx; skeletal muscle; aorta; liver; skin; bone (femur); lungs; spinal cord (3 levels: cervical, mid-thoracic, and lumbar); bone marrow (from femur and sternum); lymph node mandibular; lymph node mesenteric; spleen; brain (sections including medulla/pons, cerebellar, and cerebral cortex); mammary gland; sternum; nasal turbinates; stomach; nose; thymus; cecum; ovaries; thyroid; cervix; oviducts; trachea; colon; pancreas (with islets); urinary bladder; duodenum; parathyroid; uterus; esophagus; peripheral nerve (sciatic); vagina; Harderian gland; pharynx; heart; pituitary gland. • The following organs and tissues from all animals were preserved in modified Davidson’s fixative and then stored in ethanol, for possible future histopathological examination: eyes; optic nerve; testes; epididymides.
Histological examination was performed on the preserved organs and tissues of the main test animals from both the control and high-dose groups (Groups 1 and 4, respectively) as well as from any animal that died during the course of the study. In addition, the liver, nasal turbinates, and nose from all animals sacrificed by design were analyzed.
Statistical analysis
Statistical analysis was performed on all data collected during the in-life phase of the study as well as organ weight data and clinical pathology results. The use of the word “significant” or “significantly” indicates a statistically significant difference between the control and the experimental groups. Significance was judged at a probability value of p < 0.05. Male and female rats were evaluated separately. For all in-life endpoints, treatment and control groups were compared using a two-way analysis of variance (ANOVA).
If warranted by sufficient group sizes, all endpoints with single measurements of continuous data within groups (e.g., organ weight, relative organ weight, etc.) were evaluated for homogeneity of variances 9 and normality. 10 Where homogeneous variances and normal distribution was observed, treatment and control groups were compared using a one-way analysis of variance (ANOVA). When one-way analysis of variance was significant, a comparison of the treated groups to control was performed with a multiple comparisons test (e.g., Dunnett’s test). Where variances were considered significantly different, groups were compared using a nonparametric method (e.g., Kruskal–Wallis non-parametric analysis of variance 11 ). When non-parametric analysis of variance is significant, a comparison of treated groups to control was performed (e.g., Dunn’s test 12 ).
Means and standard deviations were calculated for all quantitative clinical pathology parameters. Bartlett’s test for homogeneity and Shapiro–Wilk test for normality was conducted. If the preliminary test was not significant, One-way analysis of variance followed with Dunnett’s test was completed. If the preliminary test was significant, log transformations of the data to achieve normality and variance homogeneity could be used. If the log transformation were to fail, a non-parametric method (Kruskal–Wallis non-parametric analysis of variance) would be used. When non-parametric analysis of variance was significant, a comparison of treated groups to control.
Results
Bacterial reverse mutagenicity (ames assay) (OECD No. 471, 1997)
Ames assay results.
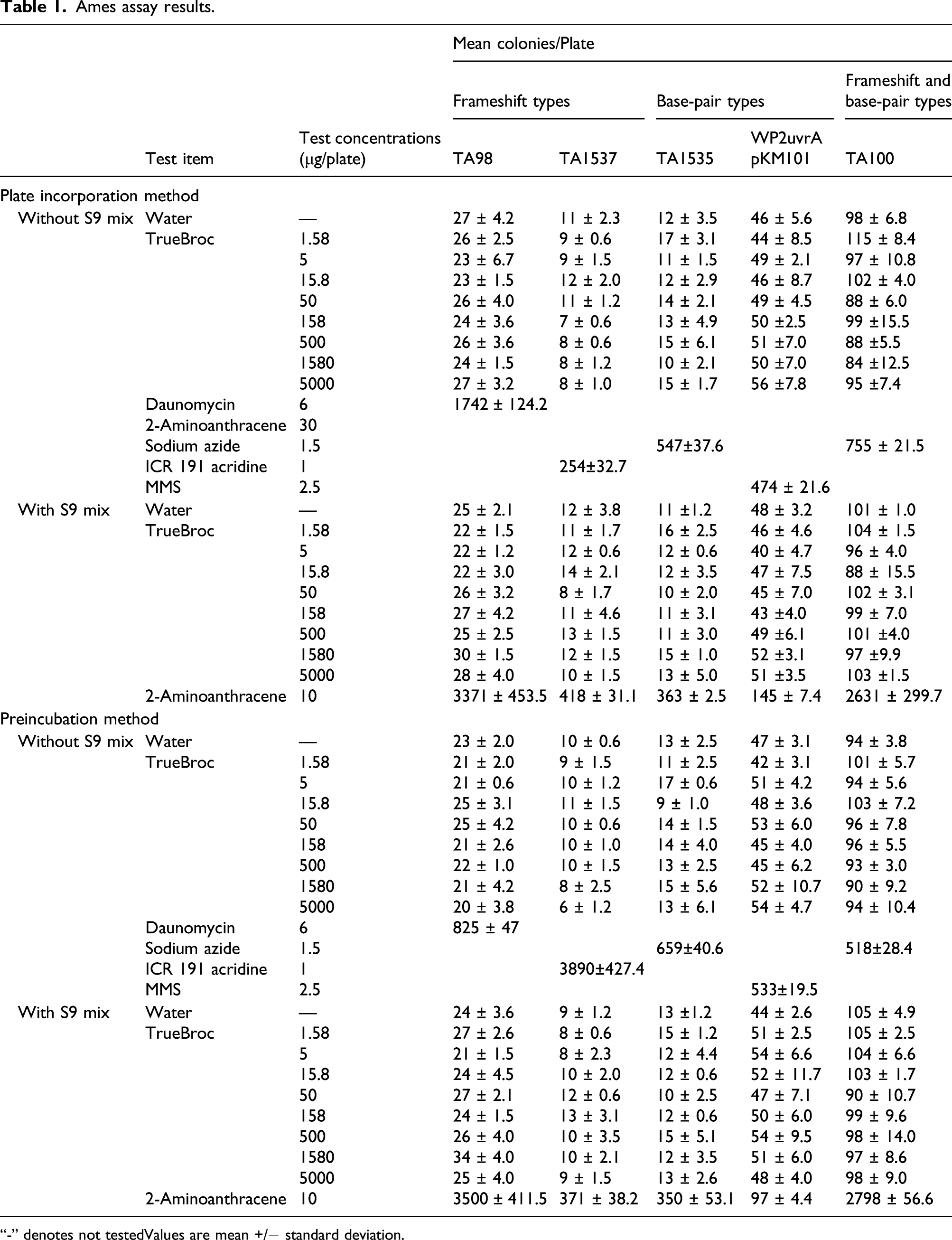
“-” denotes not testedValues are mean +/− standard deviation.
Mammalian erythrocyte micronucleus test (OECD No. 474, 2016)
In Vivo micronucleus flow cytometry results summary.
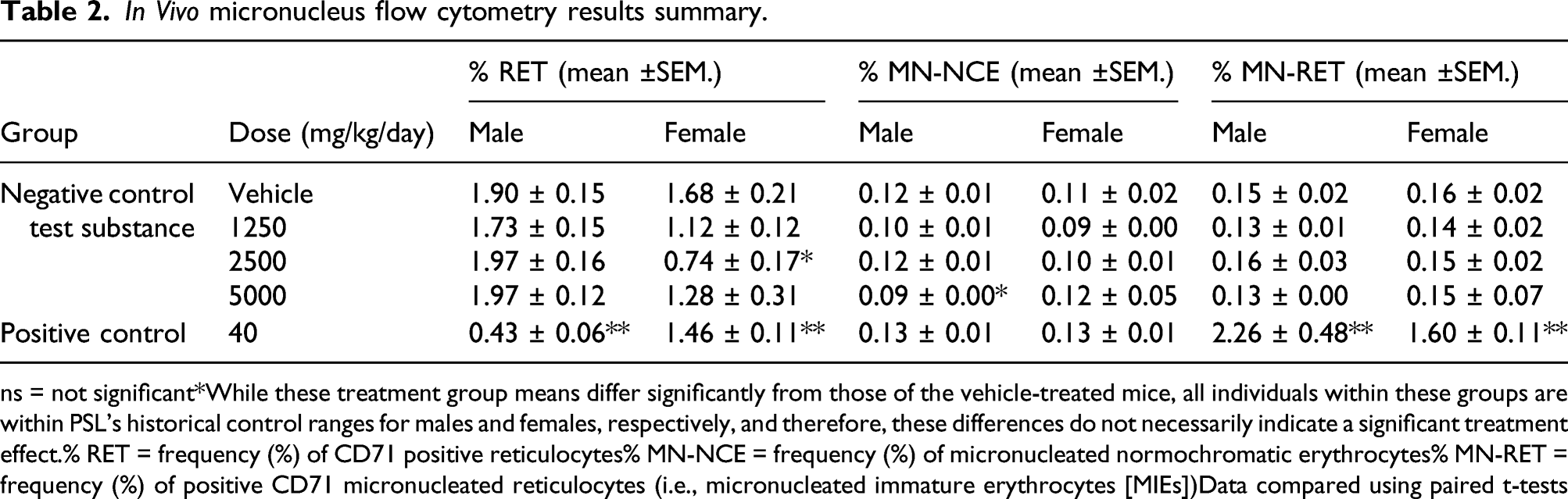
ns = not significant*While these treatment group means differ significantly from those of the vehicle-treated mice, all individuals within these groups are within PSL’s historical control ranges for males and females, respectively, and therefore, these differences do not necessarily indicate a significant treatment effect.% RET = frequency (%) of CD71 positive reticulocytes% MN-NCE = frequency (%) of micronucleated normochromatic erythrocytes% MN-RET = frequency (%) of positive CD71 micronucleated reticulocytes (i.e., micronucleated immature erythrocytes [MIEs])Data compared using paired t-tests
TrueBroc® did not induce micronucleus formation in the immature erythrocytes in mice at doses up to 5000 mg/kg/day; therefore, under the conditions of this study, TrueBroc® is not considered to be genotoxic.
In vitro mammalian chromosome aberration test in Chinese hamster V79 cells (OECD No. 473, 2016)
The highest dose group evaluated in the pre-experiment was 5000 μg/mL. The RICC (relative increase in cell count which was calculated by the increase in cell number of the test groups compared to the control groups) was used as a parameter for toxicity. Based on the results of the pre-experiment, the highest concentration selected for experiments I and II was 5000 μg/mL, as it showed no precipitation and achieved 55 ± 5% cytotoxicity (reduction in RICC to 45 ± 5% of the concurrent negative control).
Summary: Experiment I, without and with metabolic activation.
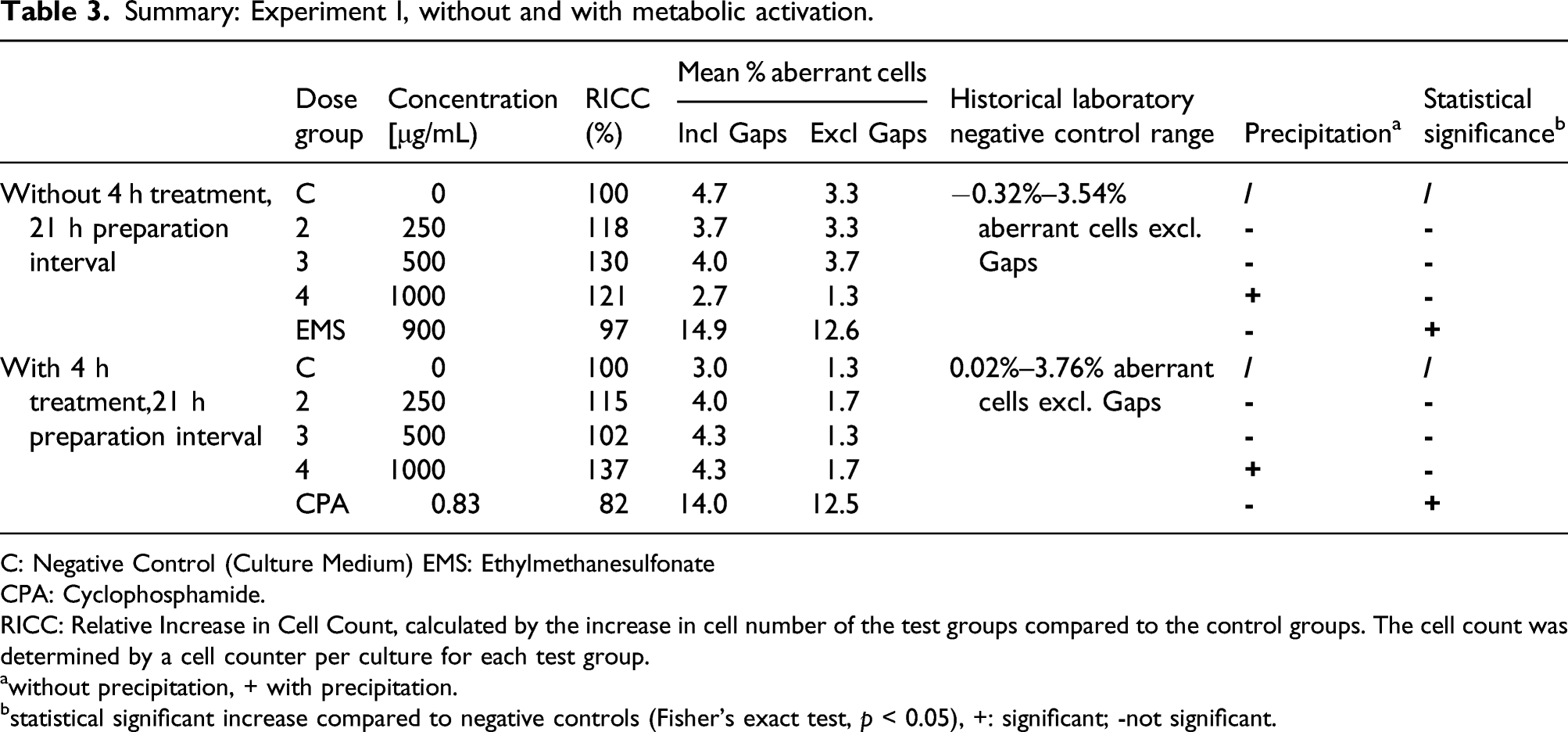
C: Negative Control (Culture Medium) EMS: Ethylmethanesulfonate
CPA: Cyclophosphamide.
RICC: Relative Increase in Cell Count, calculated by the increase in cell number of the test groups compared to the control groups. The cell count was determined by a cell counter per culture for each test group.
awithout precipitation, + with precipitation.
bstatistical significant increase compared to negative controls (Fisher’s exact test, p < 0.05), +: significant; -not significant.
Summary: Experiment II, without metabolic activation.
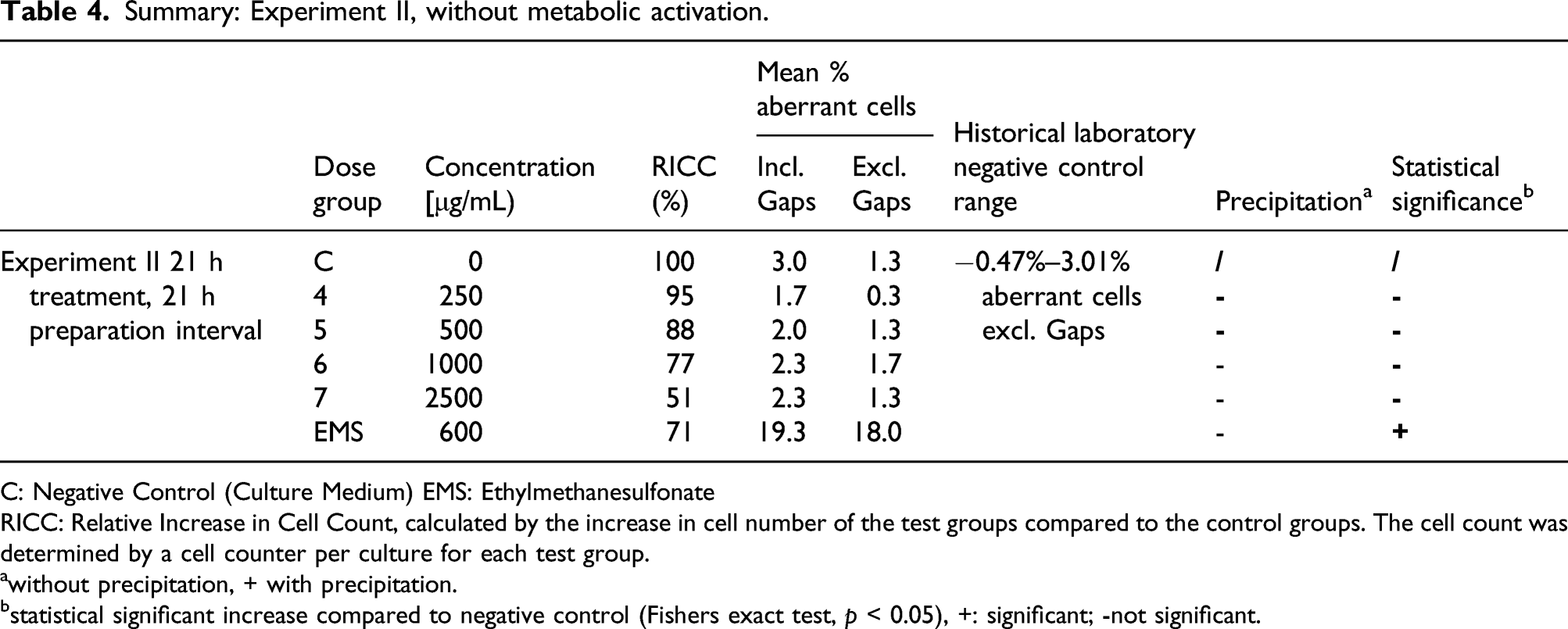
C: Negative Control (Culture Medium) EMS: Ethylmethanesulfonate
RICC: Relative Increase in Cell Count, calculated by the increase in cell number of the test groups compared to the control groups. The cell count was determined by a cell counter per culture for each test group.
awithout precipitation, + with precipitation.
bstatistical significant increase compared to negative control (Fishers exact test, p < 0.05), +: significant; -not significant.
TrueBroc® did not induce structural chromosomal aberrations in the V79 Chinese Hamster cells in the experimental conditions used in this study; therefore, TrueBroc® was determined to be non-clastogenic.
Acute oral toxicity test in rats (OECD No. 425, 2008)
All animals survived test substance administration, gained body weight, and appeared active and healthy during the study. There were no signs of adverse clinical effects, or abnormal behavior. No gross abnormalities were noted for any of the animals. Under the conditions of the study, the acute oral LD50 of TrueBroc® is greater than 5000 mg/kg of body weight in female Sprague-Dawley rats.
Range-finding toxicity study in rats (OECD No. 407, 2008)
There were no mortalities during the study and no clinical observations attributable to test article administration. There were no test article-related changes in body weight or body weight gain in the test groups; values for these parameters were comparable to the control group. Although there was no change in food consumption observed in the male rats in any group, females receiving 1500 and 3000 mg/kg/day had a statistically significant decrease in mean daily food consumption on Days 1–8, 8–15, and Days 1–15, respectively, compared to control and 750 mg/kg/day group. The decrease in food consumption did not result in any other corresponding adverse effects for the in-life endpoints, and therefore was not considered to be toxicologically relevant. There were no observations in gross pathology examination attributed to test article. Under the conditions of the study, and based on the toxicological endpoints evaluated, there were no test substance-related adverse changes. Male and female rats are expected to tolerate dose levels up to 3000 mg/kg/day (the highest dose tested) in a study of longer duration.
Subchronic oral toxicity study in rats (OECD No. 408, 2018)
The overall stability of neat test substance beyond the duration of this study was determined to be 99.4%. Based on the overall stability, homogeneity, and concentration verification analysis, animals were considered to have received their targeted dose levels.
Clinical observations
All surviving animals evaluated on Test Day 93 were normal upon visual inspection. There were no test substance-related mortalities during this study. One low-dose male animal was found dead after the blood draw, but prior to sacrifice, on Day 96. Slight moist rales on Days 50–53 was exhibited in this animal during the study, which corresponded to an observation of moist rales for respiratory pattern in detailed observations, however, there was no evidence that the moist rales contributed to the death of the animal, based on gross necropsy results. One high-dose male animal was found dead on Day 81. Subsequent necropsy on this rat determined that the death was due to oral gavage trauma. These two animal deaths were considered incidental and not test substance-related.
Body weight and body weight gain
Mean weekly body weights for female rats in all test groups were comparable to the control group throughout the study (Figure 1). No statistically significant differences were observed between any of the test substance administered groups and the control. Mean daily body weight gain for female rats in the test groups were generally comparable to control throughout the study, with the exception of significant decreases in the low- and high-dose groups on Days 29–36 (p ≤ 0.05) (data not shown). Body weights of female rats.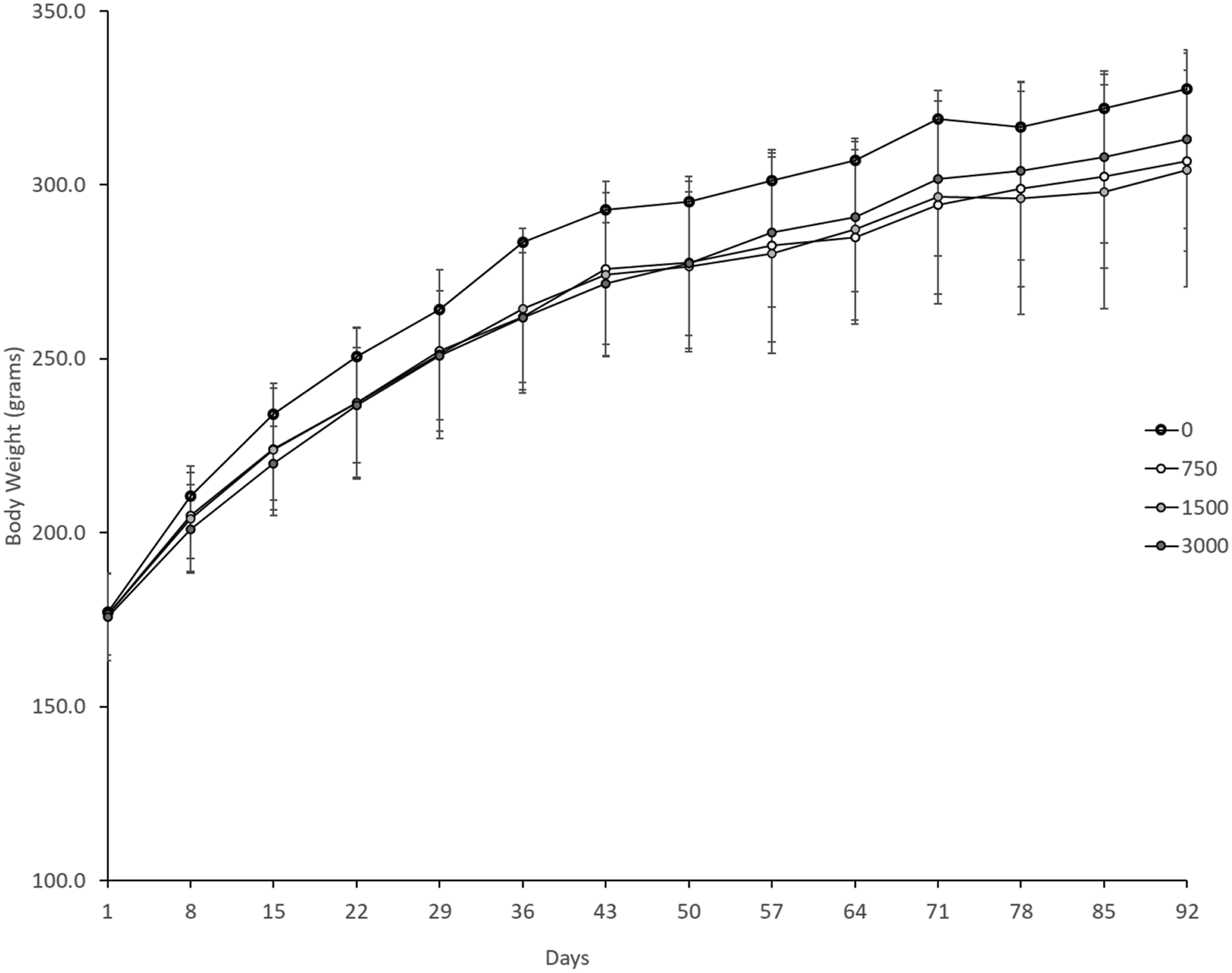
Significant decreases in body weights of male rats were noted in mid-dose animals (p ≤ 0.05) on Days 50, 64, 71, and 85 and in high-dose animals (p ≤ 0.05–0.01) on Days 29 and 43–92 (Figure 2). Significant body weight reductions at the mid-dose were sporadic and less than 10%. Mean daily body weight gain for male rats in the low-dose group was comparable to controls; significant decreases in body weight gain were noted in the mid-dose group on Days 22–29, 36–43, and for the study overall (Days 1–92) and in the high-dose group on Days 22–29 and for the study overall (Days 1–92) compared to controls (p ≤ 0.001–0.01). The mean ± standard deviation of daily body weight gain in males for days 1–92 for control, low-, mid- and high-dose groups was 4.10 ± 0.42, 3.91 ± 0.43, 3.60 ± 0.55, and 3.36 ± 0.39 g/day, respectively. The significant decrease in body weight gain observed for the mid-dose males and high-dose males was greater than 10%. Body weights of male rats.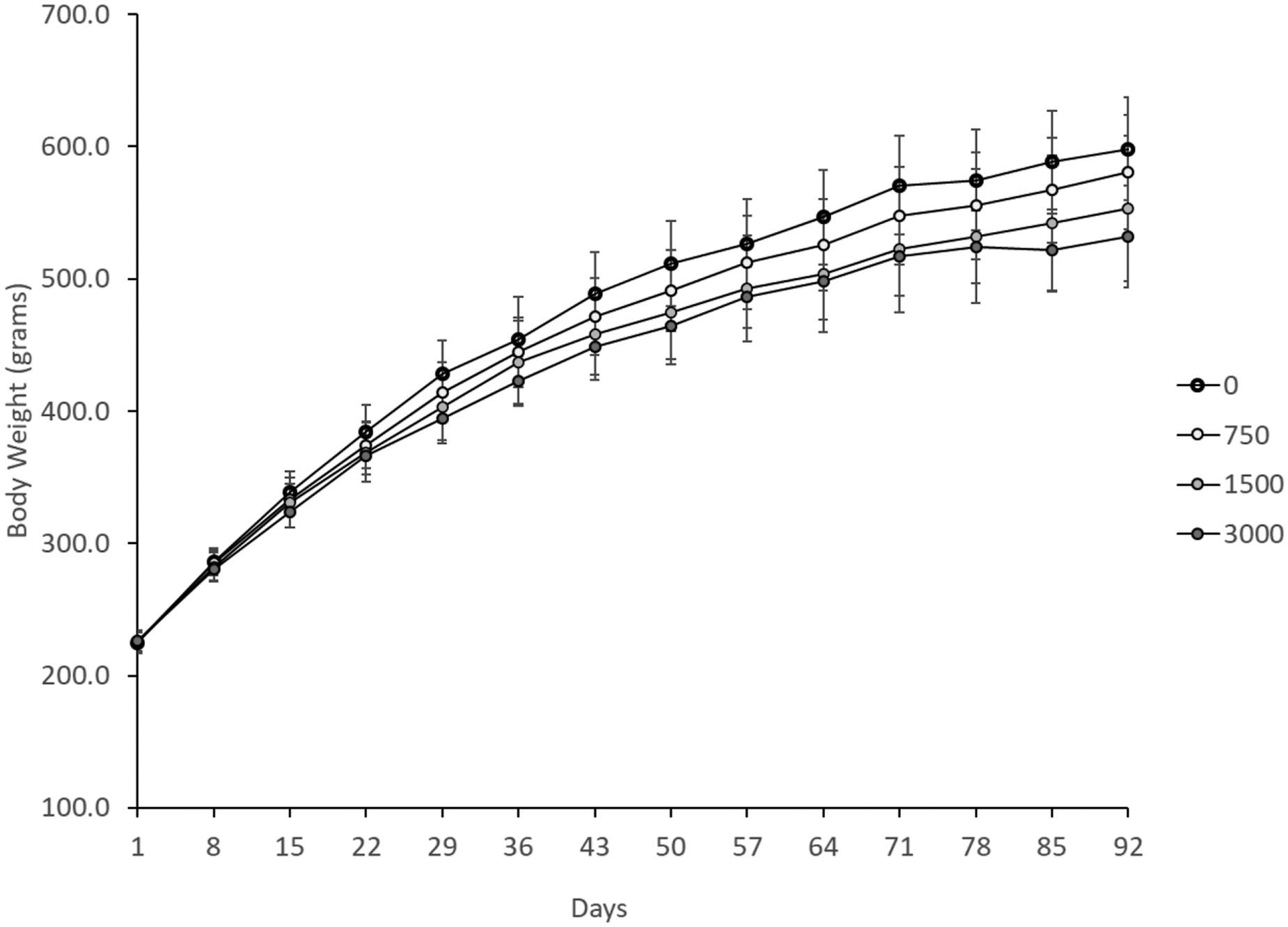
Food consumption and food efficiency
Significantly different mean daily food consumption observed in male rats.
Anova and Dunnett: *p < 0.05, **p < 0.01, ***p < 0.001 Anova and Dunnett (Rank): †p < 0.01, ‡p < 0.001.
Anova and Dunnett (Log): §p < 0.001.
Mean daily food consumption for female rats in treated groups was comparable to controls with the exception of significant decreases in the low-dose group on Days 50–57 and in the high-dose group on Days 1–8 and 22–29. The statistically significant differences were not considered test substance-related or adverse, as there was no dose-dependent relationship, and the differences were not consistently observed throughout the study. Furthermore, there were no statistically significant differences observed in food efficiency (data provided in Supplementary Table 1b).
Clinical pathology
Significantly different hematology parameters.
Anova and Dunnett *p < 0.05.
Kruskal–Wallis and Dunn: **p < 0.01.
Coagulation: There were no test substance-related changes in coagulation parameters. The significant and dose-dependent increases of APTT values in males (control, low-dose, mid-dose, high-dose of 15.56 ± 0.93, 16.67 ± 0.84, 16.76 ± 0.58, 17.11 ± 1.39, respectively) was of minimal magnitude (<10%) and was not associated with any clinical signs or microscopic changes. This effect was also not observed in any of the female rats.
Significantly different clinical chemistry parameters.
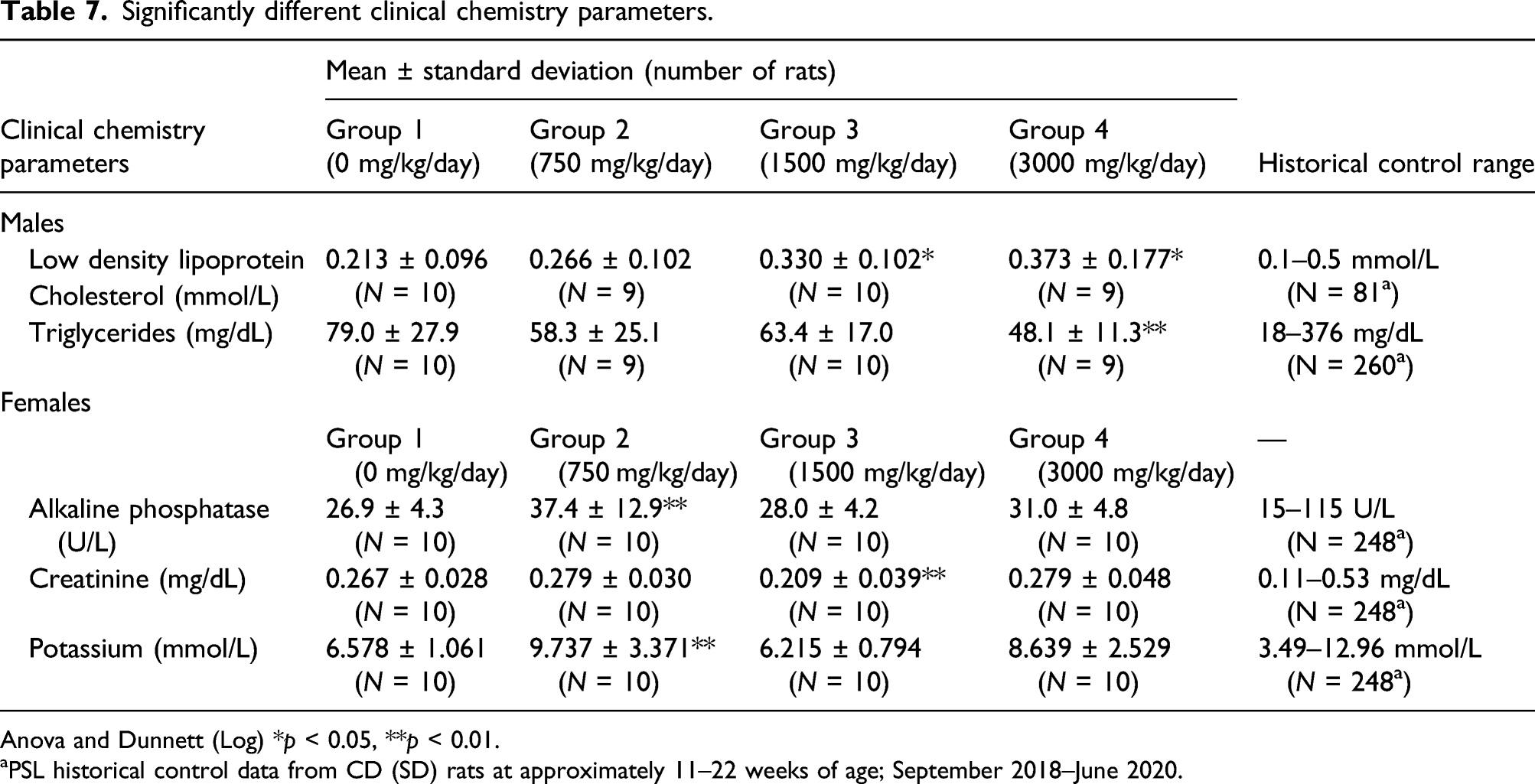
Anova and Dunnett (Log) *p < 0.05, **p < 0.01.
aPSL historical control data from CD (SD) rats at approximately 11–22 weeks of age; September 2018–June 2020.
Urinalysis: There were no test substance-related changes in urinalysis parameters (data not shown). Statistically significant differences were observed in urine pH in high-dose group males (6.80 ± 0.26, 6.70 ± 0.35, 6.60 ± 0.32, and 6.28 ± 0.26 in control, low, mid, and high-dose groups, respectively). The change in pH was minimal in magnitude and considered to be incidental. A statistically significant increase in urine volume of the high dose group females was also seen. The change, without any pathological correlate, is not considered to be adverse (data not shown).
Thyroid hormone assessment
There were no test substance-related changes in T3, T4 and thyroid stimulating hormone levels at all the dose levels tested. Increased TSH concentration in high-dose males and females. Decreased T3 in females and T4 in males at mid- and high-dose groups were not associated with any microscopic changes in the thyroid glands of high-dose animals and were within historical control values (data provided in Supplementary Table 2).
Terminal sacrifice and histopathology
There was no test substance-related mortality. One male animal in the high-dose group was found dead on Day 81. Macroscopic and microscopic findings and the subsequent case of death were determined to be secondary to gavage-related trauma and unrelated to test substance. One male animal in the low-dose group died during the blood draw immediately prior to sacrifice on day 96. This death was due to human error in anesthesia administration.
No test substance-related macroscopic observations were noted at scheduled termination.
Microscopic findings such as esophageal myofiber degeneration and/or regeneration were considered incidental, procedural-related, of the nature commonly observed in this age and strain of rat, and/or had a similar incidence across the control and treatment groups and were therefore considered unrelated to administration of test substance. Acute inflammation of the nasal meatuses was observed in both sexes (mild to marked in males at the mid- and high-doses; moderate to marked in females at low-, mid- and high-doses). This finding was characterized by abundant intraluminal neutrophils admixed with variable amounts of proteinaceous fluid, hair shaft fragments, and/or plant material. Occasionally, acute inflammation was also present within the adjacent turbinate mucosa with resulting foci of erosion/ulceration (not given a separate diagnosis). This finding was considered consistent with gavage-related reflux rather than a direct test substance-related respiratory effect or technical gavage error.
Test article microscopic observations were noted in one male from the high-dose group. Inclusions were of uncertain composition but were interpreted as a degenerative change, particularly in the context of concurrently elevated liver enzyme values (aspartate aminotransferase and alanine aminotransferase) and increased liver weight in this animal. Due to the constellation of liver findings in this animal that were suggestive of organ functional impairment (histopathologic, clinicopathologic, and organ weight alterations), the midzonal hepatocellular vacuolation was considered adverse.
Organ weights
Significantly different organ weights.
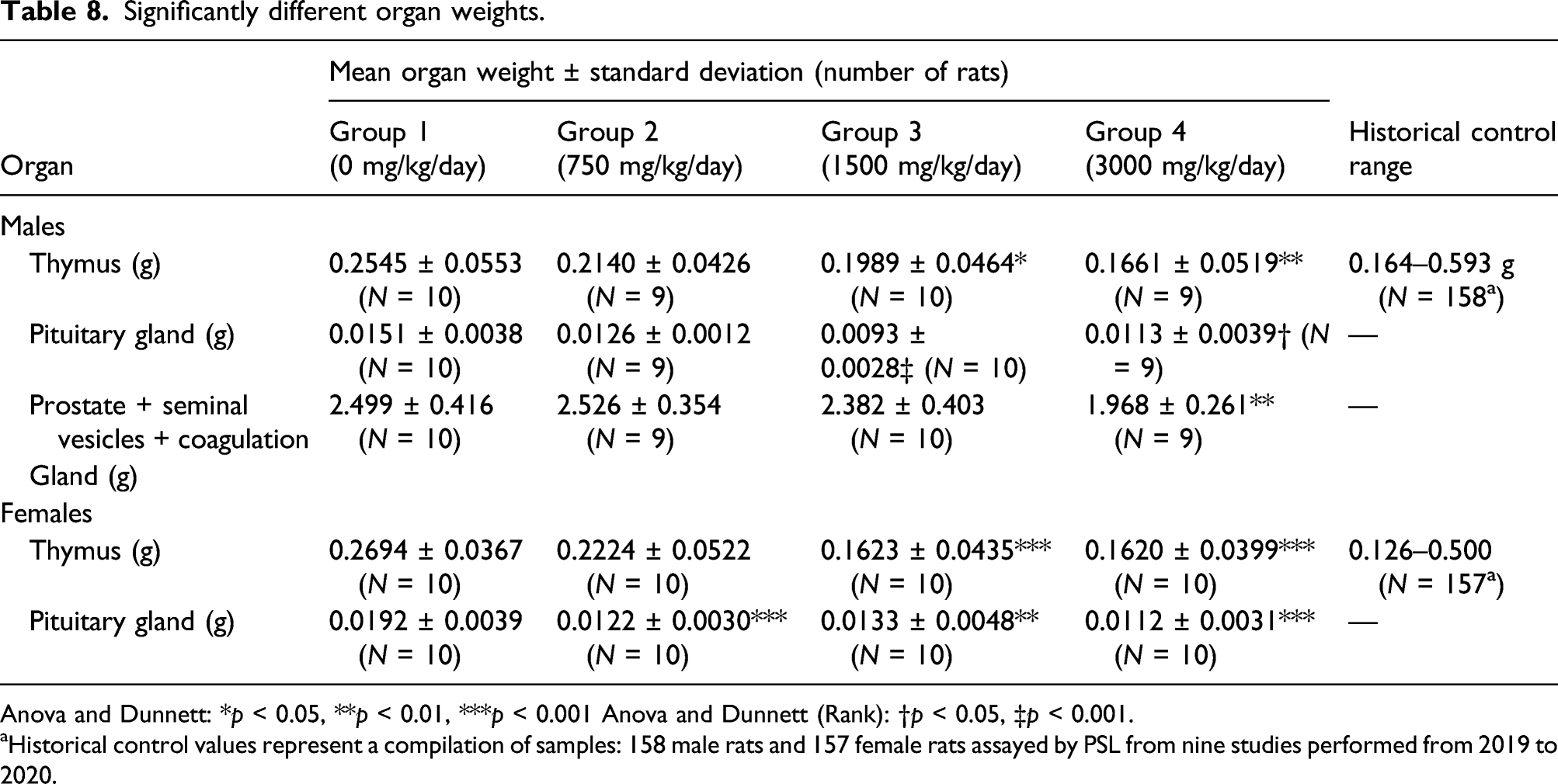
Anova and Dunnett: *p < 0.05, **p < 0.01, ***p < 0.001 Anova and Dunnett (Rank): †p < 0.05, ‡p < 0.001.
aHistorical control values represent a compilation of samples: 158 male rats and 157 female rats assayed by PSL from nine studies performed from 2019 to 2020.
Significantly different organ-to-body weights.
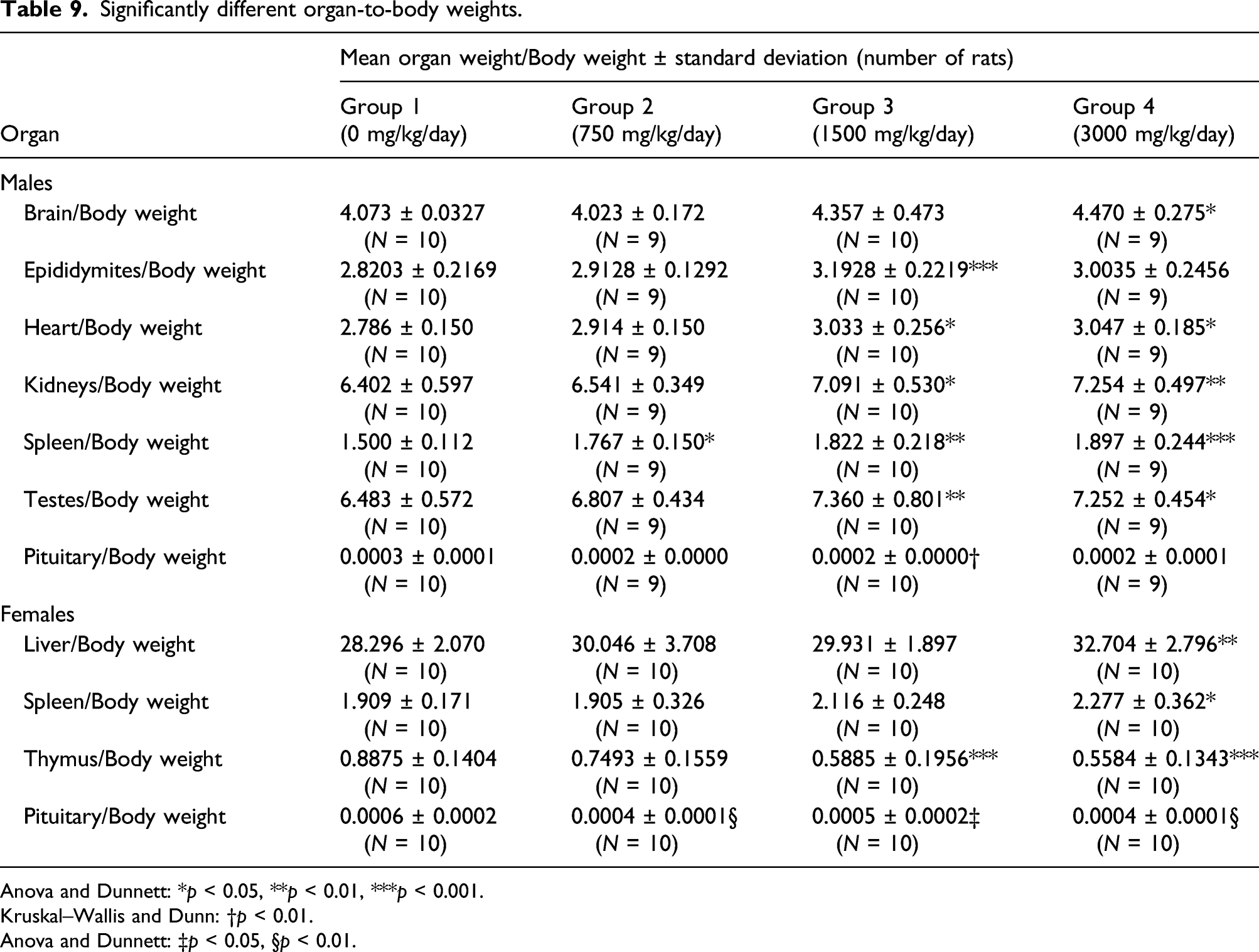
Anova and Dunnett: *p < 0.05, **p < 0.01, ***p < 0.001.
Kruskal–Wallis and Dunn: †p < 0.01.
Anova and Dunnett: ‡p < 0.05, §p < 0.01.
Significantly different organ-to-brain weights.
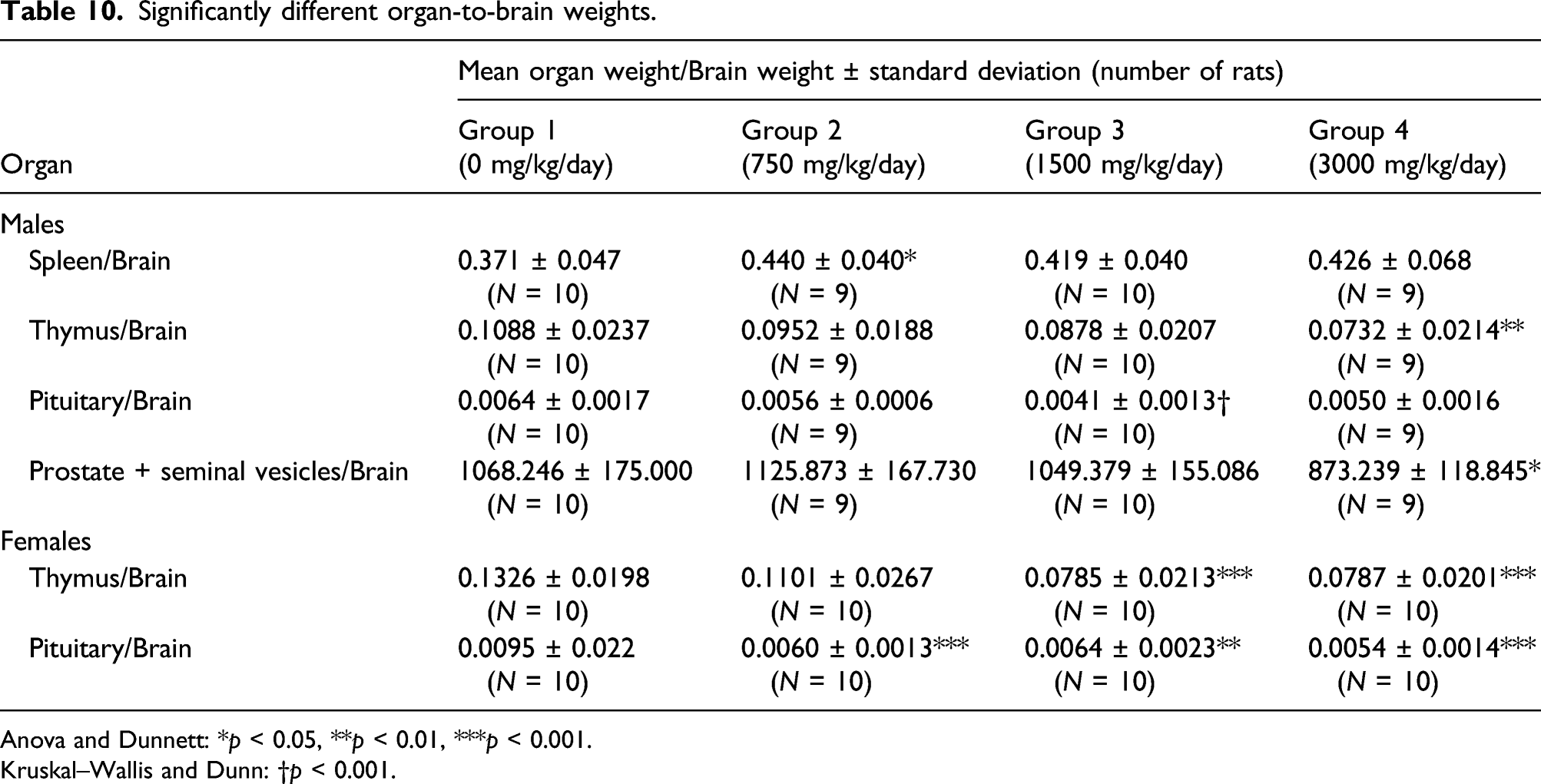
Anova and Dunnett: *p < 0.05, **p < 0.01, ***p < 0.001.
Kruskal–Wallis and Dunn: †p < 0.001.
Under the conditions of the study and based on the toxicological endpoints evaluated, the no observed-adverse-effect-level (NOAEL) for the oral administration of TrueBroc® was determined to be 3000 and 1500 mg/kg/day for female and male Sprague Dawley rats, respectively.
Discussion
TrueBroc® has been demonstrated to be safe for human consumption through in vitro and in vivo genotoxicity studies and toxicology studies in rats. TrueBroc® was not genotoxic at doses up to 5000 μg/plate, as assessed by a bacterial reverse mutation test (Ames assay), mammalian erythrocyte micronucleus test in mice, and an in vitro chromosome aberration assay in Chinese Hamster V79 cells. TrueBroc® was determined to have an LD50 of greater than 5000 mg/kg in an acute oral toxicity test in rats.
A 90-days subchronic oral toxicity study in Sprague Dawley rats determined a no-observed adverse effect level (NOAEL) of 3000 and 1500 mg/kg/day in female and male rats, respectively. Clinical observations noted in this study were considered incidental, transient, of minimal incidence, without occurring in a dose-dependent manner, and of no toxicological significance.
Statistically significant differences in body weights in female rats did not display a dose–response relationship; therefore, this difference was considered incidental, possibly related to a transient decrease in food intake and not adverse or test substance-related. Significant reductions in body weight in high dose male rats were considered adverse. Significant body weight reductions in male rats at the mid-dose were sporadic, less than 10% and not considered adverse.
No significant or adverse reductions in body weight gain were seen in treated females. A decrease in body weight gain in both the mid-dose and high-dose males was greater than 10% (Days 1–92 mean daily body weight gain was 4.10, 3.91, 3.60, and 3.36 mg/kg/day, respectively, for control, low-, mid-, and high-dose males). In high-dose males, the reduction in body weight gain was associated with a significant reduction in food efficiency, however, this was observed during only one of the 13 weeks in the study. The reduction in body weight gain in high dose males was considered treatment related and high dose. The decrease in body weight gain in the mid-dose males, although greater than 10%, was not associated with a reduction in food efficiency. Therefore, the significant decrease in body weight gain observed in the mid-dose males which was correlated with a reduction in food intake, may have been related to a palatability issue as there were no correlating adverse outcomes in clinical signs, hematology, clinical chemistry, food intake, feed efficiency, clinical chemistry, urinalysis, organ weights, hematology, gross or histopathology. Therefore, the body weight gain reduction in mid-dose males was not considered an adverse test article related change.
Changes in hematology parameters were not considered to be related to test article. The increases in absolute large unstained cells, absolute lymphocytes, and white blood cell counts in the high-dose in males and females were associated with moderate to marked acute inflammation of the nasal meatuses in rats from these groups and are consistent with gavage-related reflux rather than a direct test substance-related respiratory effect or technical gavage error.
Increased LDL cholesterol in mid- and high-dose males and decreased triglycerides in high-dose males were within historical control ranges and were not considered to be test substance-related. Clinical literature has shown glucoraphanin to decrease the levels of LDL. 13 The magnitude of the increase in LDL cholesterol in the high-dose male group was microscopically associated with hepatocellular cytoplasmic vacuolation (fatty change) in the liver of one male and was indicative of alterations in lipid metabolism in that animal.
The increased TSH concentration at the high-dose in males and females, decreased T3 in females and T4 in males at the mid-dose and high-dose were considered an incidental change of no toxicological significance as these changes were not associated with increases in thyroid organ weight or with any microscopic changes in the thyroid glands of high dose animals and were within historical control values. Additionally, in considering the risk of thyroid dysfunction to humans based on the findings of animal studies, known species difference between rodents and humans should be taken into consideration. In rodent toxicology studies, quantitative physiological differences in the thyroid gland and the HPT axis exist between rodents and humans.14–19 For this reason, in cases of findings involving the thyroid axis, dose adjustment methodology for extrapolation to humans has little merit because the functional aspects of the HPT axis in the rat or human do not scale across species in a simple allometric manner.
Investigation of organ weights noted reductions observed in thymus weights in both male and female rats in the mid- and high-dose groups. The thymus weights were within historical control ranges and are likely due to glucocorticoids being produced from stress that the animals experienced at high-dose levels. This reduction did not display a dose-dependent relationship, and there were no other differences that would indicate an adverse effect on the thymus. These decreases in thymus weight were not considered toxicologically relevant.
Histopathologic analysis revealed acute inflammation of the nasal meatuses in both sexes (mild to marked in males at ≥1500 mg/kg/day; moderate to marked in females at ≥750 mg/kg/day). Gavage-related reflux is sometimes only observed in test substance-dosed groups; this can be due to physicochemical properties of the test substance formulation, increased concentration of test substance resulting in precipitation, increased viscosity, and/or irritancy 20 and is not considered a test article related adverse finding.
The NOAEL in female rats was based on the lack of treatment-related adverse findings in all parameters measured. The NOAEL in male rats was based on a significant reduction in body weights in males at the high-dose.
Conclusion
In GRN 496, an intake of up to 554 mg/day was determined to be safe because exposure to the glucosinolates in the product was equivalent to or less than exposure from normal diet and consistent with global authoritative dietary recommendations. In addition, safety was supported by a critical review of human and animal studies documenting the metabolic fate, physiologic activity, and lack of adverse health effects associated with ingestion of the levels of glucosinolates in the product. Subsequent toxicology studies published by 7 further supported the safety of the product for human ingestion. This current battery of toxicology assays conducted on TrueBroc® demonstrates that under the conditions of the assays, it is not genotoxic. The result from the subchronic rat study establishes a NOAEL of 1500 mg/kg/day.
Supplemental Material
sj-pdf-1-tor-10.1177_23978473211068834 – Supplemental Material for Toxicological assessment of truebroc® glucoraphanin-rich broccoli seed extract
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211068834 for Toxicological assessment of truebroc® glucoraphanin-rich broccoli seed extract by Claire L Kruger, Jennifer Symonds and Dietrich Conze in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Spherix Consulting Group, Inc. is a private company that was authorized and financed by Brassica Protection Products LLC to write this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Brassica Protection Products LLC sponsored and financed the studies presented in this article. Brassica Protection Products LLC did not influence the findings or interpretation of data in any way.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
