Abstract
Introduction
Recently,
The purpose of these studies was to assess the toxicity of
Materials and methods
E. gracilis strain Eu029
Phylogenetic analysis of EG029
Total DNA was extracted from the cells of cultured EG029. The 16S rDNA region was then sequenced as described.
9
Briefly, this sequence and several sequences of
Bacterial reverse mutagenicity (Ames assay)
Bacterial reverse mutation assays were performed by Biotech Co., Ltd, (Chungcheongbuk-do, South Korea) in compliance with OECD guidelines (ENV/MC/CHEM(98)17) and good laboratory practice (GLP) for nonclinical laboratory studies (notification no. 2005-79 Korea Food and Drug Administration). Sodium azide, 2-nitrofluorene, 2-aminoanthracene, and 9-aminoacridine were obtained from Sigma-Aldrich (St Louis, Missouri, USA). 2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide was obtained from Wako, Japan. S9 and cofactor A were purchased from Oriental Yeast Co., Ltd (Tokyo, Japan).
The mutagenicity of EG029 was determined using the preincubation method. EG029 was diluted in water at 313, 625, 1250, 2500, and 5000 µg/plate incubated with tester strains
Bacillus subtilis rec assay
Japan Food Research Laboratories Chitose Labs (Hokkaido, Japan) conducted this test in accordance with previously published methods.
10
Briefly, an 8-mm paper disk was soaked for 24 h at 4°C with 313, 625, 1250, 5000, and 10,000 µg EG029/disk, 1 µg/disk Kanamycin (negative control), or 0.l µg/disk mitomycin C (positive control) diffused in 50-µL DMSO (vehicle).
In vivo micronucleus assay
The in vivo micronucleus assay was conducted by Asahi Food & Healthcare Co., Ltd (Tokyo, Japan). The methods for this test were described in guidance on genotoxicity tests of pharmaceuticals (notification no. 1604 issued by the Evaluation and Licensing Division, the Pharmaceutical and Medical Safety Bureau of Japan (PMSB) dated on November 1, 1999). Six-week old male Crlj: CDI (ICR) mice (Charles River Laboratories Japan, Inc.) were administered two daily doses of either 0, 1000, 2000, and 2500 mg/kg/day EG029 or vehicle control (water) in 10 mL/kg via oral gavage. Mitomycin C (Kyowa Hakko Kirin, Co., Ltd, Tokyo, Japan) was administered via one intraperitoneal injection to serve as a positive control. Each treatment condition had five male animals per group. Body weight was measured prior to each administration and 24 h post final administration. Animals were euthanized 23–24 h following the final administration of test substance and bone marrow cells (BMCs) in both femurs were washed with fetal calf serum (GIBCO, Gaithersburg, Maryland, USA). Excess serum was removed from BMC and cells were smeared onto two microscope slides per animal. After being dried over night, they were fixed in methanol. Samples were stained with 0.005% acridine orange solution (Wako Pure Chemical Industries, Ltd, Richmond Virginia, USA) and washed with 1/15 mol/L phosphate buffer (pH 6.8), covered with a glass cover and sealed with enamel. Samples were analyzed under fluorescence microscope (BX50: BX-FLA, Olympus Optical Co, Ltd, Tokyo, Japan) at a magnification of 1000×. All samples were scored blinded. The frequency of micronucleus occurrence per 1000 immature red blood cells from each animal was calculated. Additionally, the percentage of immature red blood cells per 250 red blood cells from each animal was calculated. The test item was considered toxic if the polychromatic erythrocyte/total erythrocyte ratio was less than that in the vehicle control group.
To evaluate the frequency of micronucleated immature red blood cells between the negative control group and other test groups, a conditional binomial test (Kastenbaum and Bowman test) was performed. To evaluate the measured values of body weight and the frequency of immature red blood cells between the negative control group and other test groups, a homoscedastic Student’s
Acute oral toxicity test
Japan Food Research Laboratories Tama Laboratory (11-10 Nagayama 6-chome, Tama-shi, Tokyo) performed this test in accordance with OECD guidelines for the testing of chemicals 420 (2001). Five-week old female ICR mice (Japan SLC, Inc., Japan) were acclimatized and monitored for abnormalities for 1 week, with five animals housed per cage. Mice were provided feed and water ad libitum, except for fasting 4 h prior to treatment. Five mice per group were administered either 5000 mg/kg EG029 or water by gavage and monitored for 14 days. The animals were observed frequently on the administration day and once daily on the following day and later. Body weight was monitored prior to treatment and at days 7 and 14 post-gavage and a
Subchronic toxicity study
A 13-week subchronic toxicity study in 6-week old male and female Sprague-Dawley (SD) rats was conducted by Biotoxtech Co., Ltd (Chungcheongbuk-do, South Korea) in compliance with the GLP Regulation for Nonclinical Laboratory Studies notification no. 2005-79 (Korea Food and Drug Administration) and the OECD Principles of GLP (ENV/MC/CHEM(98)17) guidelines. Rats were housed two to three animals per cage during the quarantine and acclimation period of 6–7 days and one animal per cage during the study. Prior to dosing, the rats were randomized by body weight into three groups (
Developmental toxicity
A developmental toxicity test (Hokudo Co., Ltd, Japan) was performed under the guidelines described in “Revision of guideline for detection of toxicity to reproduction for medical products,” notification no. 1834 issued by PMSB. Male and female SD rats aged 9 and 11 weeks, respectively, were obtained from Charles River Laboratories, Japan, and were acclimatized for 16 days before being mated. Successful mating was observed by vaginal plug and considered pregnancy day 0, upon which female rats were singly housed and provided feed and water ad libitum throughout the study. Twenty-two pregnant female rats were obtained for each treatment group, consisting of either 1000 mg/kg EG029 suspended in water at 100 mg/mL or the same volume of water as vehicle control. Pregnant females were treated once daily by oral gavage from day 7 to day 17 of pregnancy. General appearance in all animals was observed twice daily during treatment and once daily before treatment. Body weight of all animals was measured once daily on day 0, day 3, and every day between days 7 and 20 of pregnancy. For all animals, the amount of daily feed consumption was calculated on days 1, 3, 7, 10, 14, 17, and 20 of pregnancy. Animals were euthanized by exsanguination under ether anesthesia on day 20 of pregnancy.
After macroscopic observation, the following tissues were fixed and stored in 10% neutral buffering formalin liquid: the brain (the cerebrum and cerebellum), pituitary gland, thyroid gland (including parathyroid), adrenal gland, spleen, heart, liver, lungs (including the bronchi), kidneys, ovaries, uterus (bilateral uterine horns and cervix), vagina, and mammary glands. During the necropsy of maternal animals, the ovaries and uterus were removed and the weight of pregnant uterus was measured. The uterine wall was dissected to observe the conditions of uterus, fetus, and placenta. Fetuses and their placentas were removed and the weight of the fetuses was measured and sex was noted. The number of corpora lutea, implantations, live fetuses, dead fetuses, and resorbed fetuses was recorded. The rate of implantation for each maternal animal was calculated by the following method: the implantation rate (%) = (number of implantations/level of corpus luteum in pregnancy) × 100.
All the surviving fetuses were examined for external abnormalities, and approximately half of surviving fetuses from each maternal animal were fixed with Bouin’s solution for visceral examination, while the other half was washed with water after their organs were removed, and fixed with 80% ethanol for skeletal examination. The head was examined for abnormalities macroscopically in accordance with Wilson’s method. 11 Visceral examination under stereomicroscope was performed under Nishimura’s microdissection method. 12 After the examination was completed, fetuses were stored individually. For skeletal examination, all fetuses in each group were fixed with 80% ethanol and stained with alizarin red S, dipped in glycerol solution and transparent skeletal specimens were prepared. Ossification was measured (number of ossifications in each vertebral body of the cervical spine, thoracic vertebrae, lumbar vertebrae, sacrum, sternebra, metacarpals, and metatarsals) using a stereomicroscope to examine abnormalities and variation.
Mean values and standard deviations were calculated for the body weight of maternal animals, increase in body weight, amount of feed consumption, weight of pregnant uteruses, number of corpora lutea in pregnancy, number of implantations, surviving fetuses, dead embryos and fetuses, body weight of surviving fetuses, weight of placentas, and progression of ossification. The variance was analyzed in an
To evaluate the implantation rate, the fetal survival rate, the embryonic and fetal mortality, the gender ratio and the occurrence of fetuses having abnormalities or variations, the significant difference between the control and EG029 treatment was determined in the Wilcoxon rank sum test, using one maternal animal as the sample unit. In all the test methods,
Results
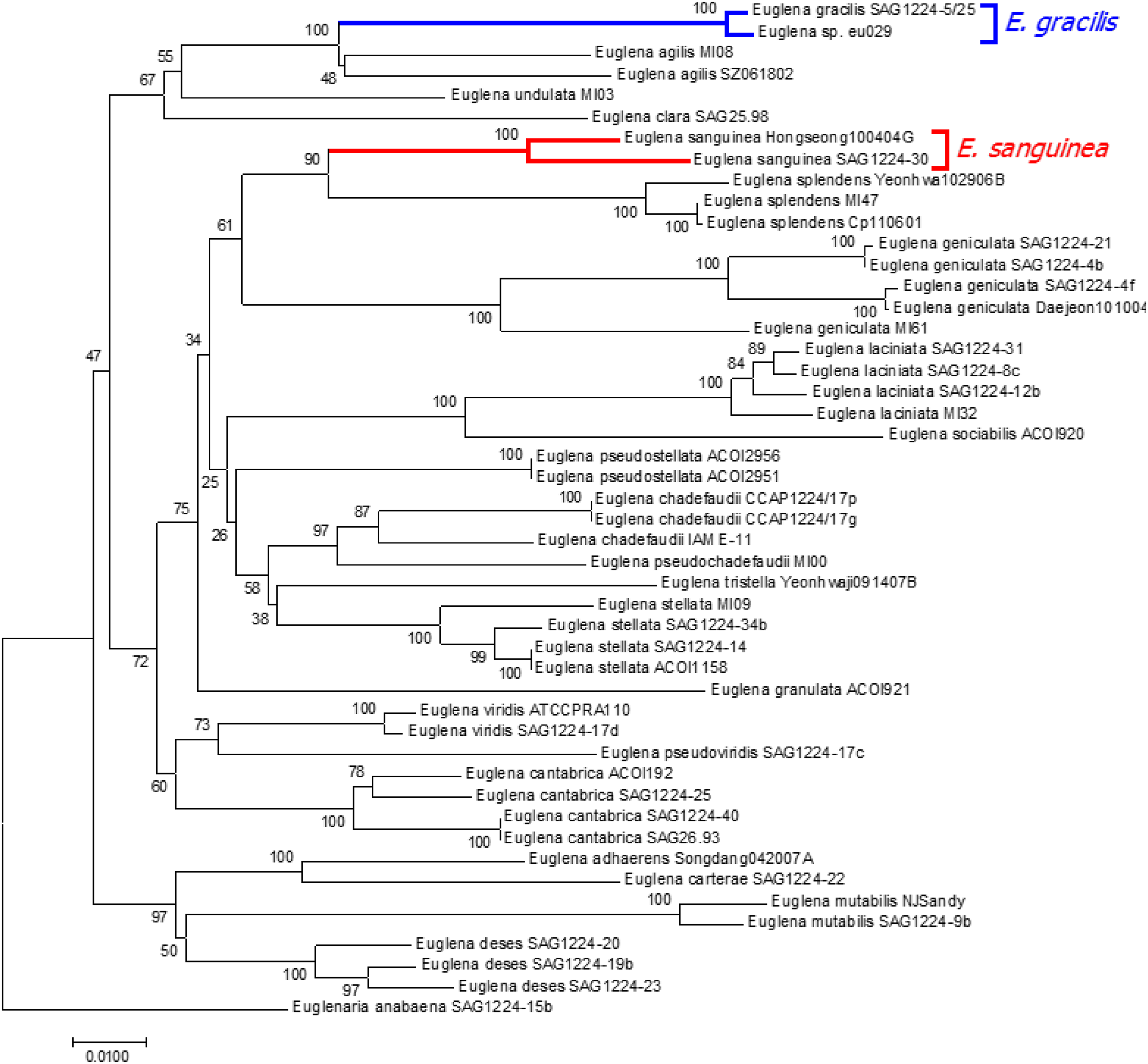
Dendrogram of EG029
Ribosomal DNA sequencing was used to verify that EG029 was a strain of the species
Bacterial reverse mutagenicity (Ames assay)
EG029 was not cytotoxic at any of the doses used compared to vehicle control. EG029 treatment did not increase the number of revertant colonies in any of the base-pair substitution strains or frame-shift strains, while the positive controls for each strain of bacteria increased the number of revertant colonies, demonstrating the validity of the assay. EG029 treated with S9 mix found no increase in the number of revertant colonies (Table 1). Thus, EG029 and its metabolites are not mutagenic in this assay up to a dose of 5000 µg.
Ames analysis shows EG029 elicits no genotoxic effects.a
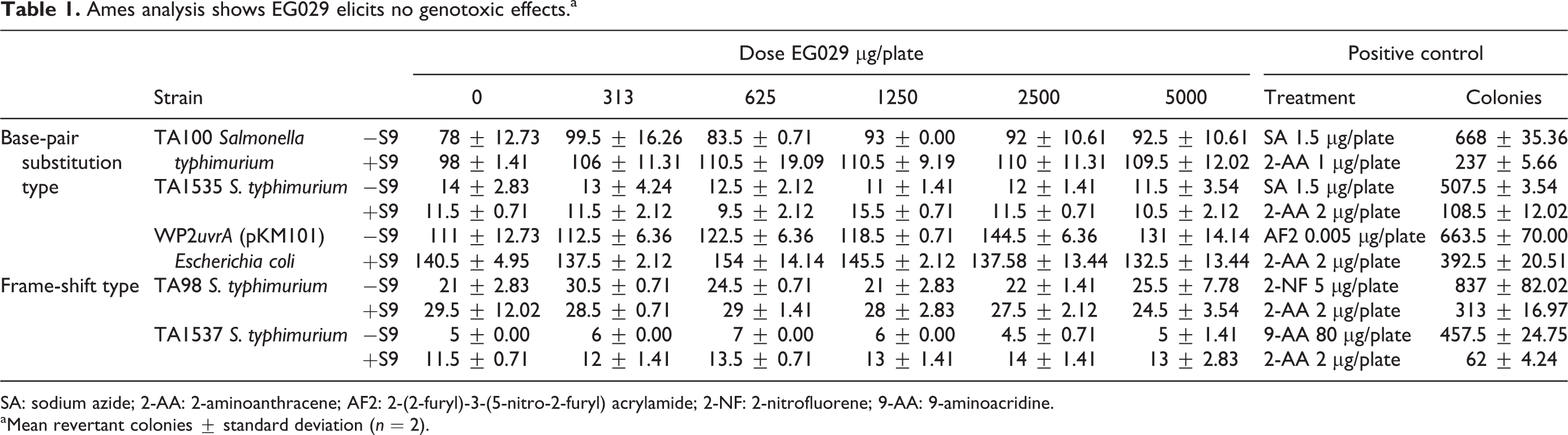
SA: sodium azide; 2-AA: 2-aminoanthracene; AF2: 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; 2-NF: 2-nitrofluorene; 9-AA: 9-aminoacridine.
aMean revertant colonies ± standard deviation (
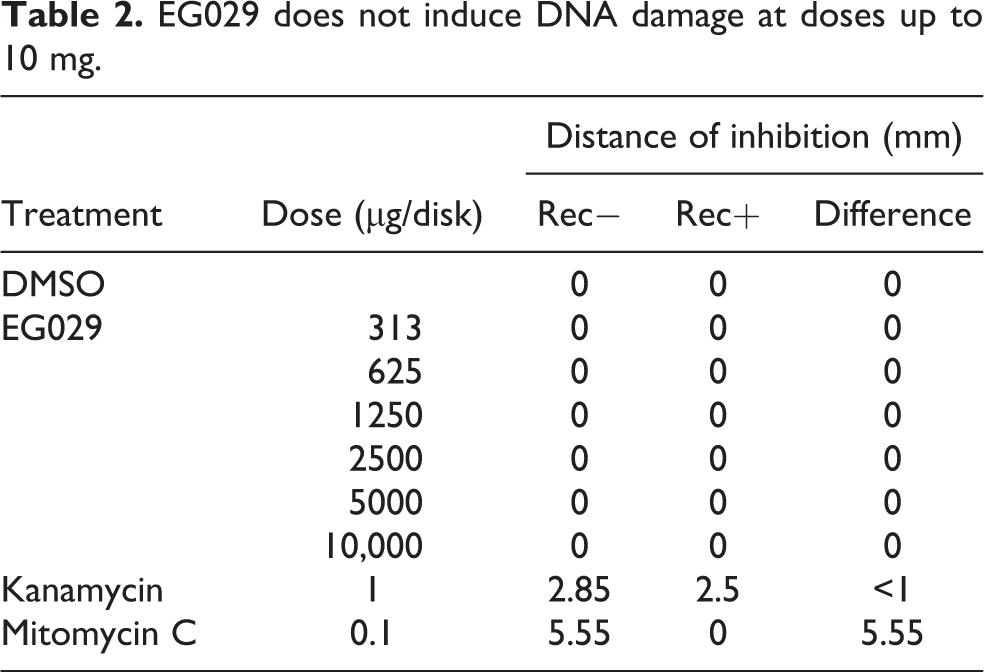
B. subtilis rec assay
Rec−
EG029 does not induce DNA damage at doses up to 10 mg.
In vivo micronucleus assay
After two treatments of daily oral gavage of EG029 at 500, 1000, 2000, or 2500 mg/kg, no increase in the frequency of micronucleated immature red blood cells was observed in any of the ICR mice administered EG029. The mice treated with the water control were observed to have a similar rate of micronucleated immature red blood cells at all doses of EG029, while the positive control mitomycin C caused a statistically significant increase in micronuclei compared to the negative control (Table 3). During the experiment, animals were reported as having a normal appearance and behavior with no adverse effects due to treatment. EG029 was therefore determined to not be clastogenic.
In vivo chromosomal aberration with EG029 exposure.
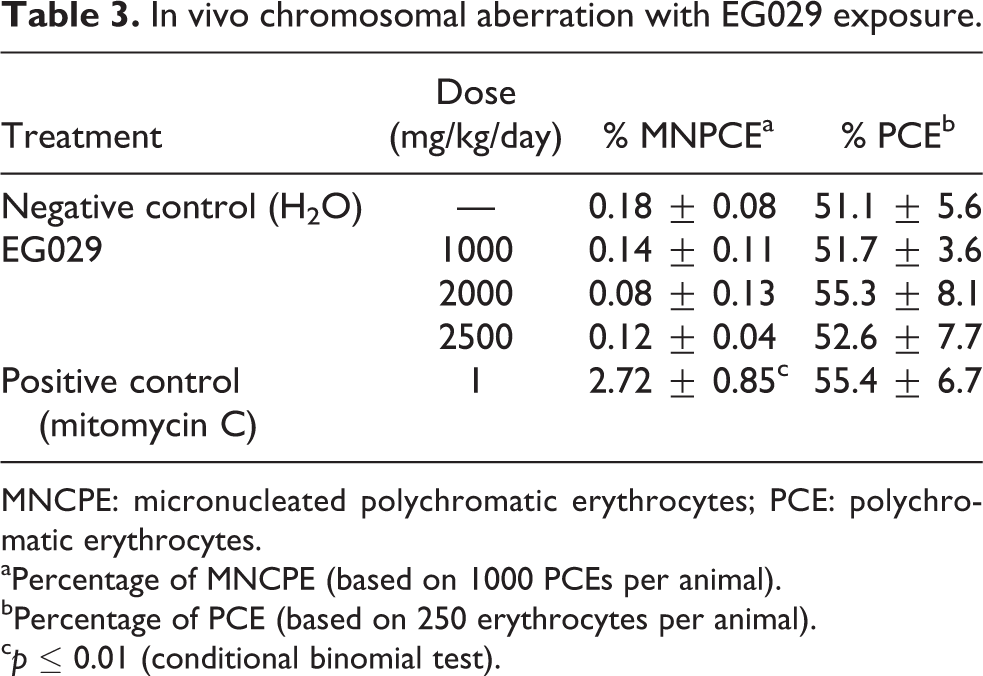
MNCPE: micronucleated polychromatic erythrocytes; PCE: polychromatic erythrocytes.
aPercentage of MNCPE (based on 1000 PCEs per animal).
bPercentage of PCE (based on 250 erythrocytes per animal).
c
Acute oral toxicity test
To assess the acute toxicity of EG029 in mammals, a single dose of 5000 mg/kg EG029 was administered to adult female ICR mice by oral gavage followed by 14 days of monitoring. No change was observed in body weight at either 7 days or 14 days after EG029 administration compared to the control group (data not shown). EG029 did not lead to any mortalities and no abnormalities were observed upon necropsy. These data indicate that the LD50 of EG029 is greater than 5000 mg/kg in female mice after a single dose via oral gavage.
Subchronic toxicity study
A 13-week study of daily EG029 treatment by oral gavage was performed in male and female SD rats. No treatment-related mortality or abnormal clinical observations were noted for the 200 and 1000 mg/kg treatment groups. No differences were observed in body weights between treated rats (200 and 1000 mg/kg groups) and control animals (Figure 2). Feed consumption was monitored weekly. No changes were observed in feed consumed in males or females treated with either 200 or 1000 mg/kg EG029 compared to control.
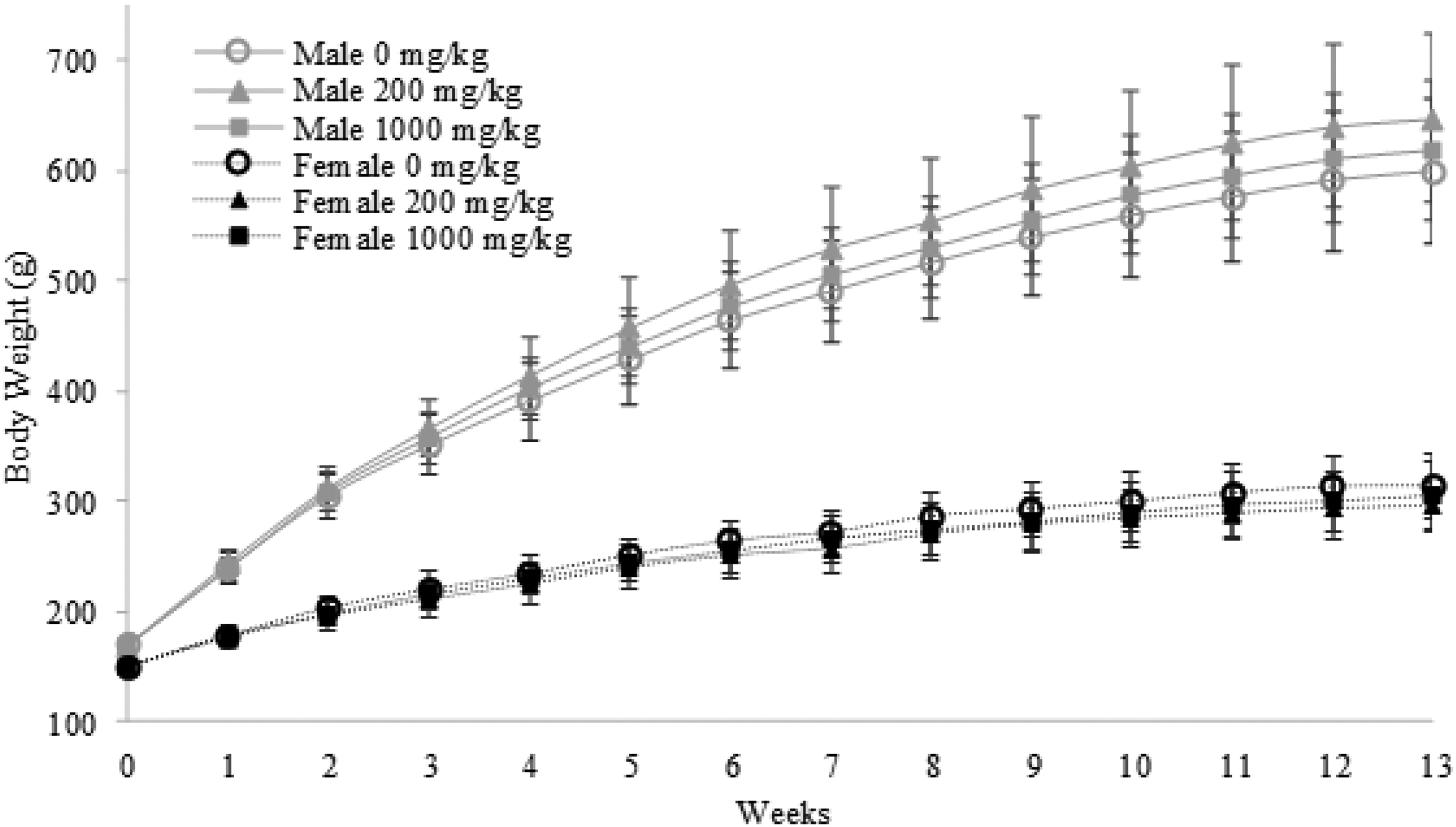
EG029 in diet does not affect body weight during 13-week study at either 200 or 1000 mg/kg in both male and female rats.
No clinically significant changes were observed in the hematological parameters. There was a slight decrease in Mean Cell Hemoglobin Concentration (MCHC) in the females dosed with 200 mg/kg EG029 (34.1 ± 0.4) compared to vehicle (34.6 ± 0.5; Table 4). This decrease was not dose dependent and was not observed in the male rats receiving EG029. Although this decrease was statistically significant, the difference between control and 200 mg/kg EG029-treated females was very small and within the historical limits at the testing facility and were, therefore, considered independent of EG029 treatment. There was a small, but significant increase in the percentage of eosinophils observed in the male rats treated with 1000 mg/kg EG029 (1.5 ± 0.6) compared to the control group (1.0 ± 0.2; Table 4). Considering the small increase in the percentage of eosinophils and the absence of any other increase in white blood cells in both male and female rats, it is unlikely that this difference is a response to the EG029 supplementation. Furthermore, all eosinophil percentages were within the observed range of physiological variation (0.4–2.6% eosinophils) for control rats at from this testing facility.
Hematological parameters of 13-week oral repeated dose toxicity study in male and female SD rats.a
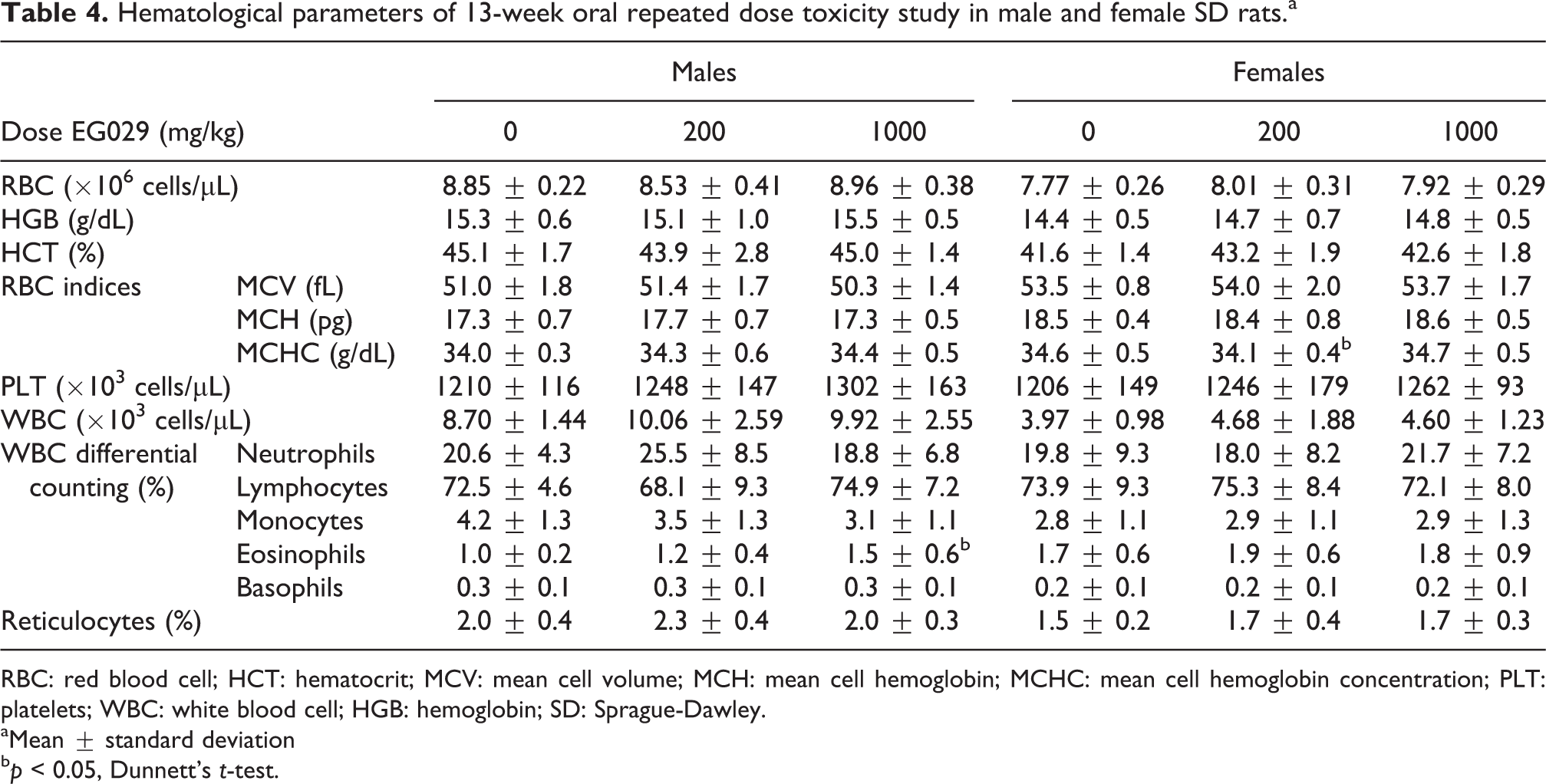
RBC: red blood cell; HCT: hematocrit; MCV: mean cell volume; MCH: mean cell hemoglobin; MCHC: mean cell hemoglobin concentration; PLT: platelets; WBC: white blood cell; HGB: hemoglobin; SD: Sprague-Dawley.
aMean ± standard deviation
b
The test substance produced a statistically significant increase in ALT in males in the 200 mg/kg group (35.4 ± 8.5) compared to male control rats (24.8 ± 3.5; Table 5). The modest increase in ALT was still well below the threshold of abnormal ALT, considered to be three to four times greater than control, and was not dose dependent. No other blood chemistry values were significantly different than control male and female rats. Although there were small, statistically significant differences in the treatment groups compared to control regarding MCHC, eosinophils and these results were not attributed to treatment because they each occurred in only one gender and there was no dose response across the groups.
Blood chemistry values of 13-week oral repeated dose toxicity study in male and female SD rats.a
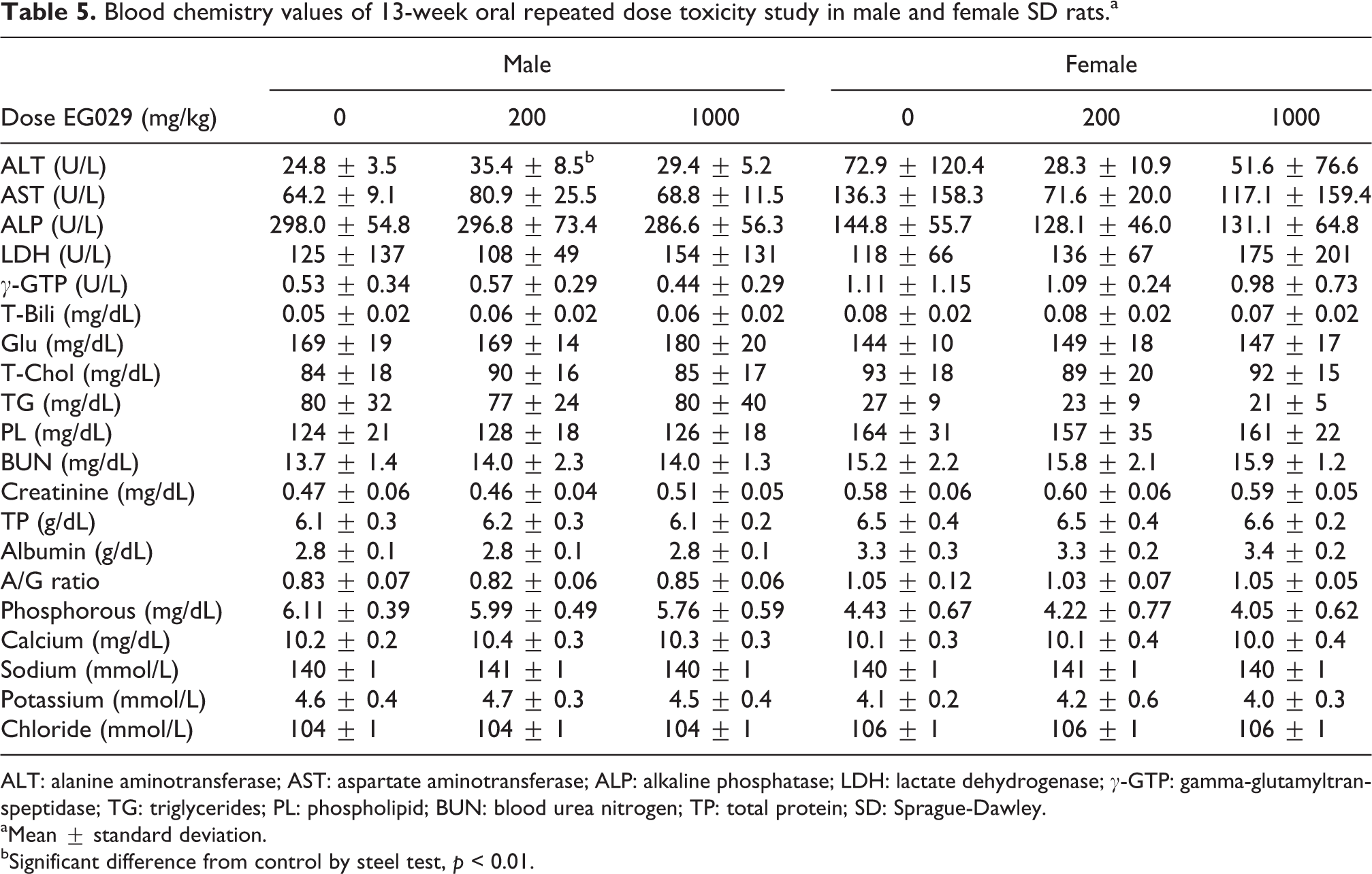
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; γ-GTP: gamma-glutamyltranspeptidase; TG: triglycerides; PL: phospholipid; BUN: blood urea nitrogen; TP: total protein; SD: Sprague-Dawley.
aMean ± standard deviation.
bSignificant difference from control by steel test,
Supplementation of EG029 in male and female rats did not affect urine pH, protein, or glucose concentration. Bilirubin and urobilirubin were not detected in any of the animals in the study. Ketone bodies (5 mg/dL) in the urine were detected in a single male and female rat in each of the EG029 treatment groups, and two male rats in the 200 mg/kg group had blood in their urine. However, these findings were not significantly different from control animals; therefore, the supplementation of EG029 did not produce urinalysis results outside of the normal range.
No statistically significant changes in the relative organ weights were observed in any EG029 treatment groups (Table 6). Upon necropsy, one male rat supplemented with 200 mg/kg EG029 showed signs of an enlarged spleen, which was likely due to increased hematopoiesis. Histopathology was carried out on the male and female rats in both the control and the 1000 mg/kg EG029 groups. Cell infiltrate was found in the heart of one male rat in the 1000 mg/kg group and in the spleen of one control male. Heart and spleen histology was unremarkable for all other animals. Additionally, there were no significant histological findings for the colon, liver, lung, rectum, sternum, and stomach of in any of the animals, and the uterus and ovaries were normal for all female animals.
Relative organ weights (g organ/100 g body weight) of 13-week oral repeated dose toxicity study in SD rats.a,b
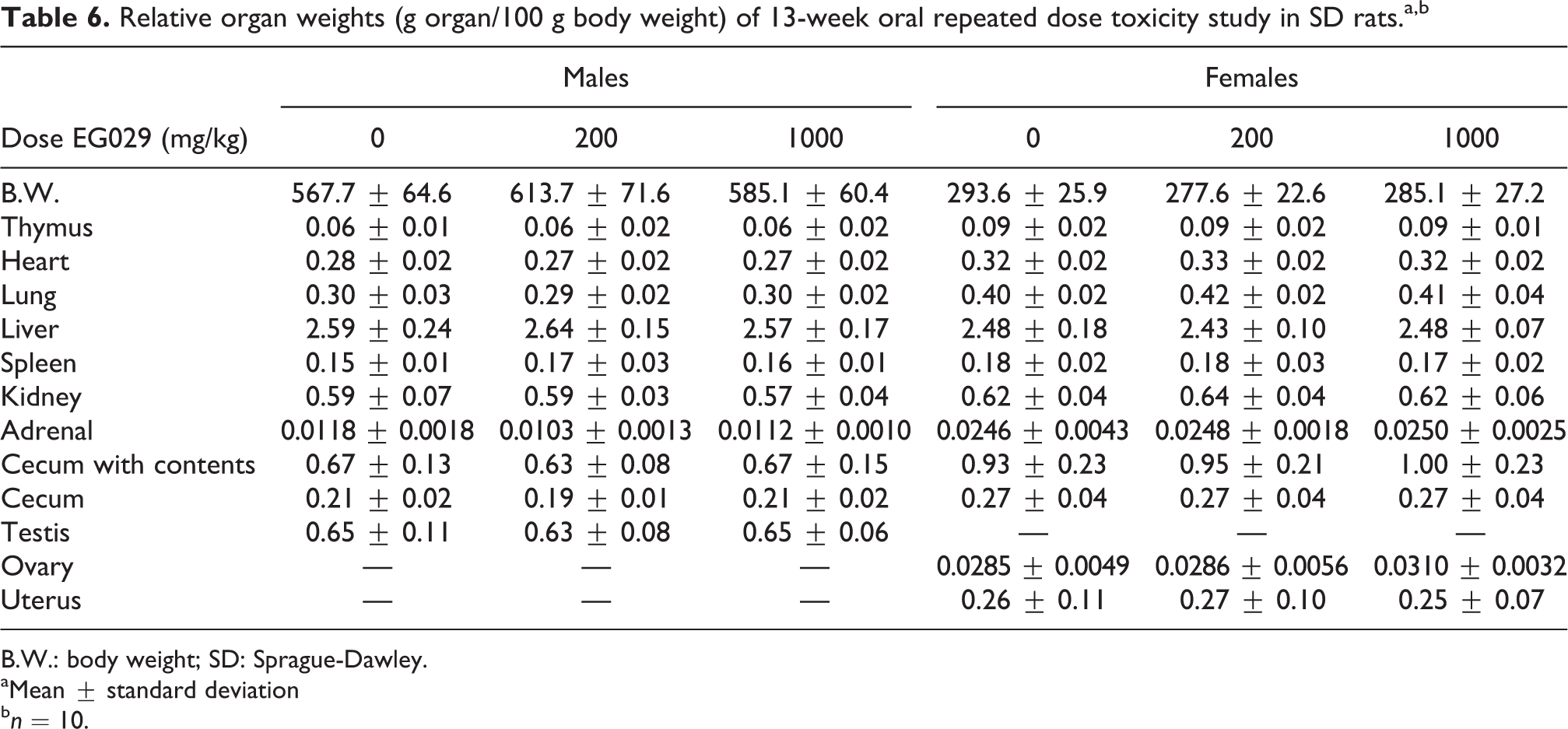
B.W.: body weight; SD: Sprague-Dawley.
aMean ± standard deviation
b
Developmental toxicity test
A developmental toxicity test was performed to assess if EG029 supplementation during pregnancy would have adverse effects on either the pregnant females or the developing fetuses. Pregnant female rats were treated with 1000 mg/kg EG029 daily from day 7 to day 17 of pregnancy, and body weight and feed consumption were monitored. EG029 supplementation did not affect body weight of pregnant females (Figure 3(a)), although feed consumption on day 20 of pregnancy in the EG029 group (29.4 g) was significantly lower than the control (31.8 g; Figure 2(b)). However, since supplemented dams continued to gain weight normally through day 20 of pregnancy, the difference was considered incidental and not related to test article. No abnormal findings in the dams were detected upon necropsy.
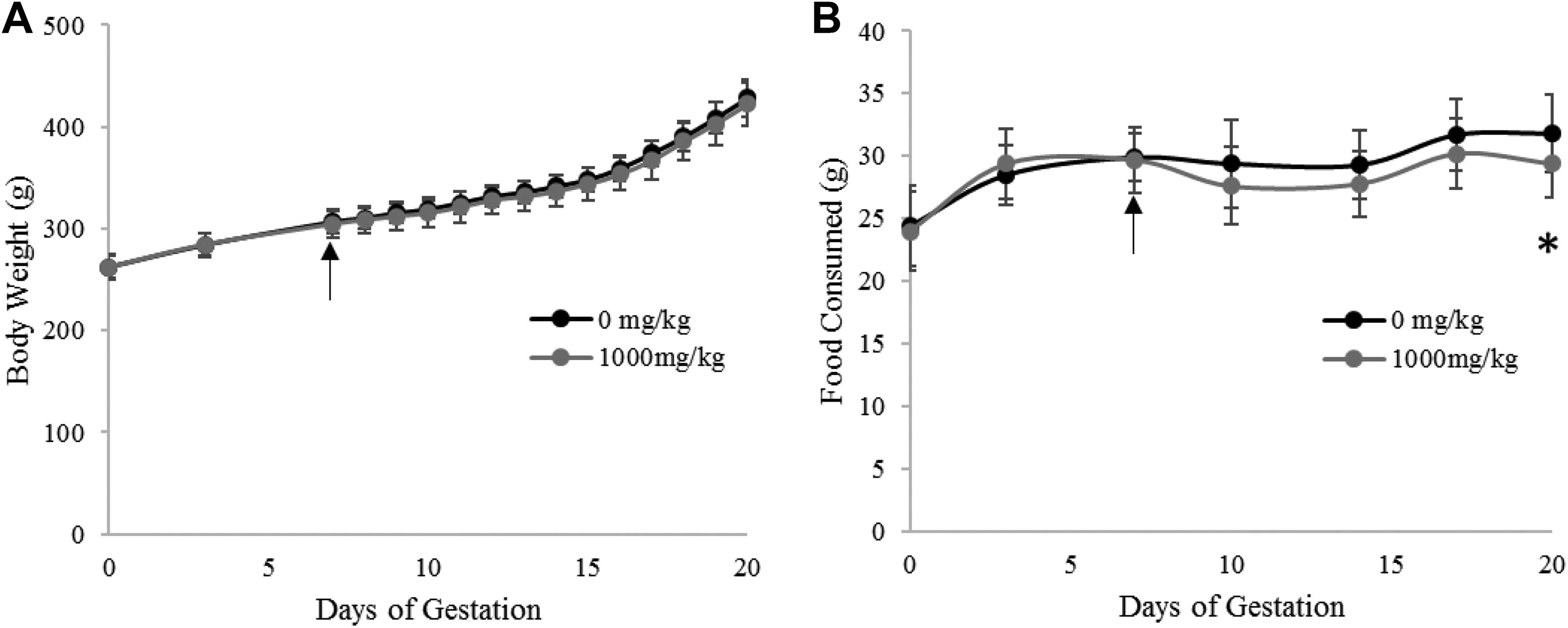
Daily oral gavage of 1000 mg/kg EG029 did not affect either maternal health or fetal development during pregnancy in rats. Arrows mark start of daily oral gavage of EG029. *
Oral gavage of EG029 in pregnant females did not affect the number of implantations or the frequency of dead or resorbed embryos/fetuses. EG029 supplementation did not affect the sex ratio of males to females in the live fetuses or their body weights at day 20 of pregnancy (Table 7). No external changes, skeletal anomalies, or visceral anomalies were found in fetuses in either the control or EG029 group (Online Supplementary Table S1).
Embryofetal development in pregnant rats with daily oral gavage of EG029.a
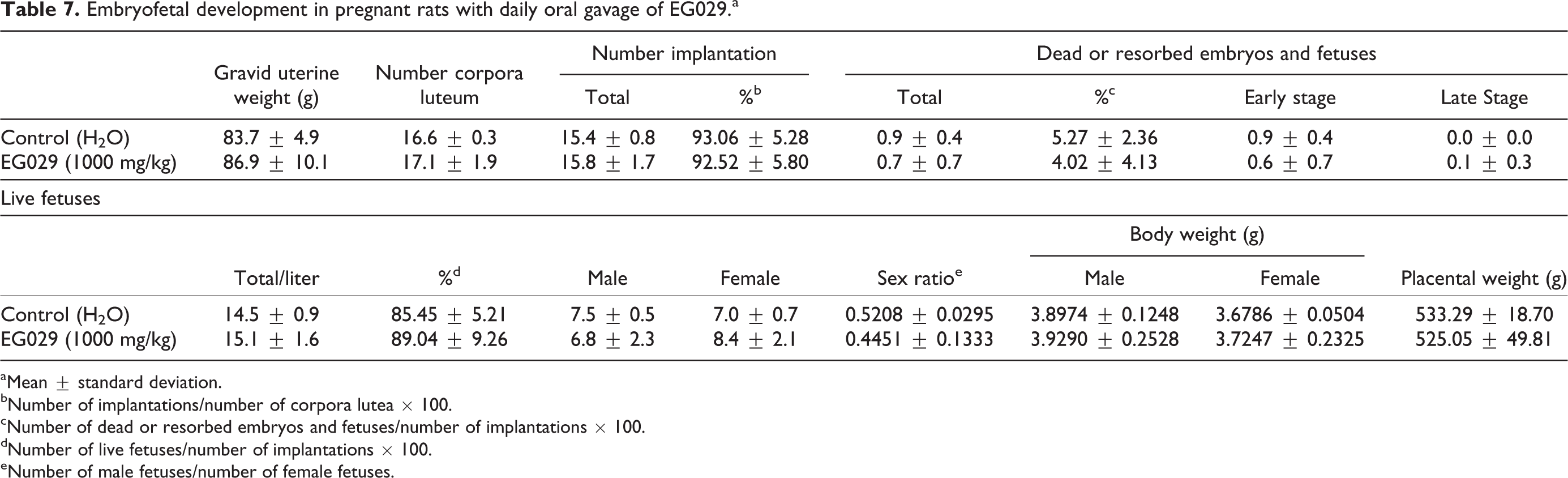
aMean ± standard deviation.
bNumber of implantations/number of corpora lutea × 100.
cNumber of dead or resorbed embryos and fetuses/number of implantations × 100.
dNumber of live fetuses/number of implantations × 100.
eNumber of male fetuses/number of female fetuses.
Thymic remnant variation was observed in one fetus in the control group (of 141 total fetuses) and one fetus in the treatment group (of 172 total fetuses; Table 8). The incidence of fetal skeletal variation was not significantly different between treated and control groups (Table 9). One litter from the EG029 group had an increase in the number of observed 14th ribs and sacrilization of lumbar vertebrae. Constriction of thoracic vertebral body was observed in four maternal animals and lumbar ribs were observed in seven to nine maternal animals in both groups. Sacralization of the 14th rib and lumbar vertebra was only found in the EG029 group. These variations were observed at a similar incidence compared to control litters. Fetal skeletal ossification was not affected by maternal EG029 exposure (Table 10). EG029 supplementation during pregnancy did not adversely affect either the dams or F1 generation.
Fetal visceral variation from pregnant rats administered daily oral gavage of 1000 mg/kg EG029.a

aMean ± standard deviation.
bNumber of fetuses showing variations/number of fetuses observed × 100.
Fetal skeletal variation of F1 fetuses of pregnant rats administered daily EG029.a
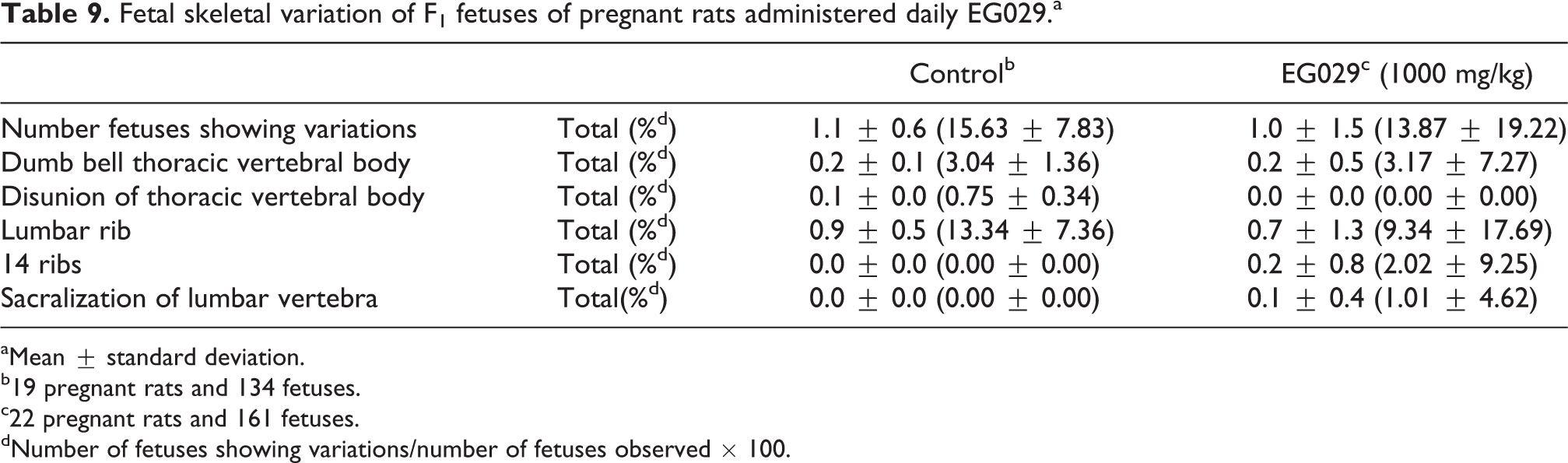
aMean ± standard deviation.
b19 pregnant rats and 134 fetuses.
c22 pregnant rats and 161 fetuses.
dNumber of fetuses showing variations/number of fetuses observed × 100.
Fetal skeletal ossification of F1 fetuses of pregnant rats administered daily EG029.a
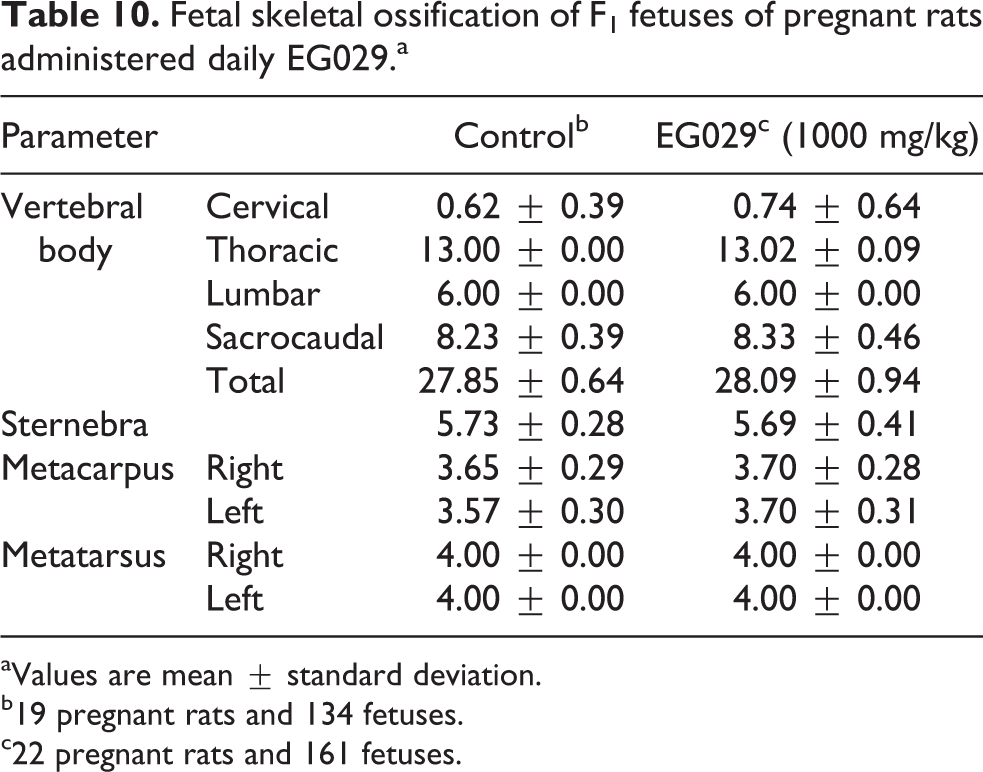
aValues are mean ± standard deviation.
b19 pregnant rats and 134 fetuses.
c22 pregnant rats and 161 fetuses.
Discussion
The purpose of these studies was to assess the toxicity of
Compared to other microalga like spirulina and
By dry weight,
Euglena supplementation may also provide additional health benefits other than basic nutrition. The 1,3-beta glucan, paramylon, when linearized, has an immune-activating effect on rats through inducible nitric oxide synthase (iNOS) and Nuclear Factor κ B (NFκB) signaling and has been proposed to help patients with immunodeficiency.
15
Another study found that, when compared to isolated paramylon, 2% Euglena powder in the animal feed can decrease symptoms of diabetes in a type-II diabetes mellitus rat model. The rats in this model typically develop diabetes within 4 months of life, including increased blood glucose levels, increased appetite, obesity, and insulin resistance. Rats given Euglena in the diet had decreased incidence of obesity, decreased fasting glucose levels, and decreased feed consumption, while control rats had no change in diabetes indices.
17
These findings suggest that
Conclusion
The safety of EG029 was evaluated in genotoxicity assays, single and subchronic dose toxicology assays, and a developmental toxicity test. An acute dose of 5000 mg/kg showed no signs of toxicity in female mice (LD50 > 5000 mg/kg). Rats supplemented with 1000 mg/kg/day of EG029 during a 90-day, subchronic study showed no adverse effects when compared to the control animals (No Observed Adverse Effect Level [NOAEL] > 1000 mg/kg/day). The 1000 mg/kg/day of EG029 also produced no adverse effects on dams or their fetuses when supplemented during days 7–17 of pregnancy. Taken together, these data support the safety of
Supplemental material
Supplementary Material, Supplementary_Table_1 - Toxicological assessment of Euglena gracilis strain Eu029 shows no adverse effects in vivo and in vitro
Supplementary Material, Supplementary_Table_1 for Toxicological assessment of
Footnotes
Acknowledgments
The authors would like to thank Fumihiko Yoshino and Osamu Iwata of euglena Co. Ltd in Japan for their advice on this project and also GLOBIZZ CORP., Los Angeles, for their coordinative role and contributions to the completion of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Claire Kruger, Jennifer Symonds, and Nicole Beauchamp are employees of ChromaDex Spherix Consulting, a business unit of ChromaDex, Inc. ChromaDex is a public company that was mandated and financed by euglena Co. to write the manuscript of the study presented in the publication. Takuto Takeuchi, Koji Yamada, Kohei Atsuji, Yuta Asayama, and Kengo Suzuki are employees of euglena Co. Ltd, the company who mandated and funded the study presented in this manuscript and preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
