Abstract
AlphaGOS®, an alpha-galacto-oligosaccharides product, is a mixture of bi-, tri- and tetrasaccharides derived from oligosaccharides in the raffinose family of oligosaccharides (RFOs), naturally occurring plant-derived sugars. RFOs are alpha α-1,6-linked chains of D-galactose attached to the 6-position of D-glucose and differ from the currently commercially available beta-galacto-oligosaccharides products in the chirality and glyosidic bonds. In order to determine the safety of AlphaGOS, rats were given 2000 mg AlphaGOS/kg/day daily via gavage over 90 days. Daily assessments of the animals showed no adverse clinical signs. No adverse treatment-related changes in feed consumption, body weight, clinical chemistry or hematology were noted. There were no adverse treatment-related changes in organ weights, gross or histopathology. Given these findings, it can be concluded that the no observed adverse effect level for AlphaGOS is greater than 2000 mg/kg/day.
Introduction
Historically, galacto-oligosaccharides (GOS) have referred to non-digestible oligosaccharides (NDOs) consisting of 1–7 galactose units linked via β(1-2), β(1-3), β(1-4), or β(1-6) glycosidic bonds to either a terminal glucose or galactose.
1,2
They are formed through the enzymatic reaction of β-galactosidase with lactose to form oligosaccharides with β
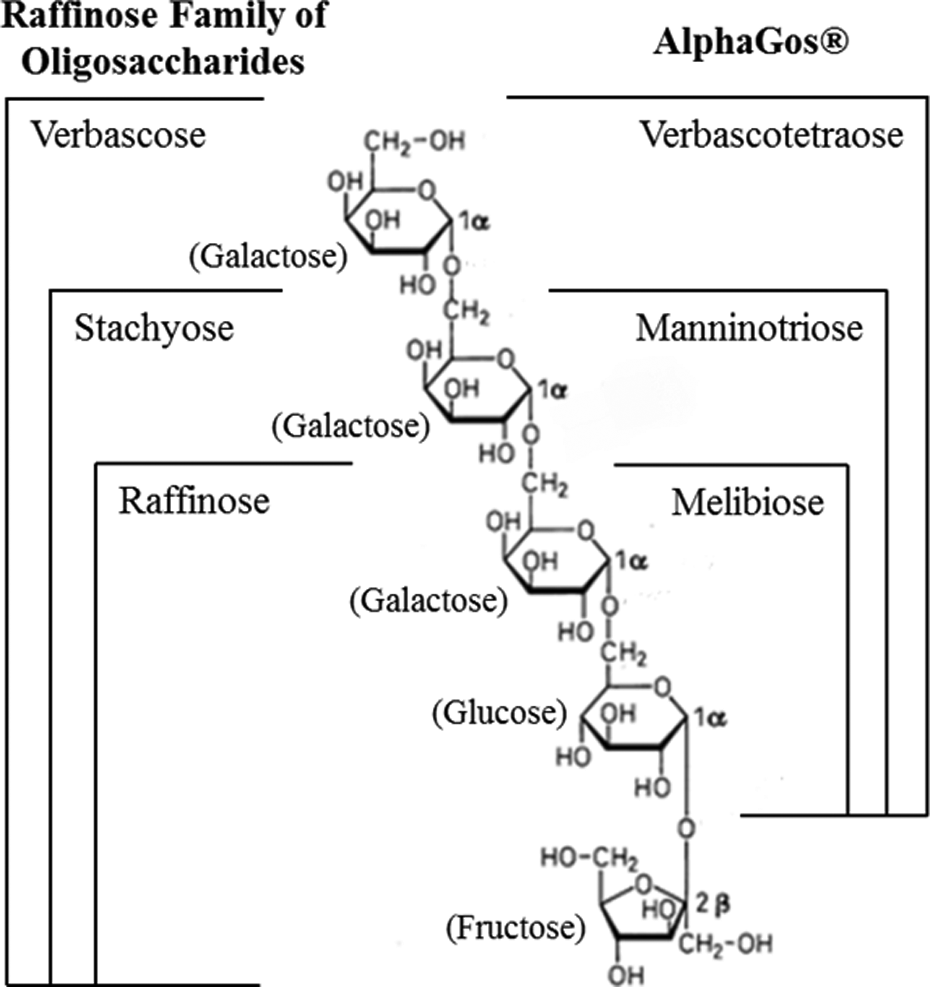
The sugars in AlphaGOS® are derivatives of the raffinose family of oligosaccharides found in many plants. The terminal fructose has been enzymatically removed to produce the resulting sugars.
The α-1,6 glycosidic bonds are hydrolyzed by α-galactosidase. Because humans do not produce this enzyme, the α-GOS product, AlphaGOS, like RFO, passes through the upper gastrointestinal tract without being hydrolyzed and reaches the large intestine intact.
6
AlphaGOS is fermented by microflora in the large intestine. Raffinose and other GOS of the RFO have been shown to increase the numbers of the beneficial bacteria
In order to establish the safety of the α-GOS product, AlphaGOS, as an ingredient in selected food products, a 90-day toxicology study was conducted in Sprague Dawley rats.
Materials and methods
Study design and study animals
A 90-day rat toxicology study was completed by CiToxLAB France under good laboratory practices (GLP) conditions and in accordance with OECD guideline #408.
Sprague Dawley rats were obtained from Charles River Laboratories, Calco, Italy. Upon receipt, two same-sex animals, from the same group, were housed per cage. Prior to starting the treatment, animals were allowed to acclimate to study environment for 9 days (temperature: 22 ± 2°C; humidity: 50 ± 20%; light/dark cycle: 12 h/12 h). Rats were 5–6 weeks old and had a mean body weight of 171 g and 139 g for male rats and female rats, respectively. Ad libitum feed and water were provided to the animals.
Ten male and 10 female rats were either given 2000 mg/kg/day AlphaGOS or vehicle (reverse osmosis-treated drinking water) in the morning via gavage for 90 days. Animals were monitored twice daily for mortality and once daily for aberrant clinical signs. Detailed clinical examinations (skin, fur, eye, mucous membrane changes; occurrence of secretions and/or excretion; autonomic activity, changes in gait, posture and response to handling; clonic or tonic movements), individual body weight and feed consumptions (per cage) were recorded weekly. On week 12 of treatment, animals underwent a functional observation battery (assessment of reactivity to manipulation and different stimuli and motor activity) and ophthalmological exam. Fasting (at least 14 h ) urine and blood samples were collected following the completion of the treatment period, and animals were killed for a full macroscopic and microscopic post-mortem examination. Organs weighted at the completion of the study include adrenals, brain (medulla/pons, cerebellar and cerebral cortex), epididymides, heart, kidneys, liver, ovaries (including oviducts), spleen, testes, thymus and uterus (with horns and cervix). The following tissues were collected and preserved: adrenals, aorta, brain (medulla/pons, cerebellar and cerebral cortex), cecum, colon, duodenum, epididymides, esophagus, eyes (with Harderian glands), femoral bone (with articulation), gut-associated lymphoid tissue, heart, ileum, jejunum, kidneys, liver, lungs (with bronchi), lymph nodes (mandibular and mesenteric), mammary glands, ovaries (including oviducts) pancreas, pituitary gland, prostate (dorsolateral and ventral), rectum, salivary glands (sublingual and submandibular), sciatic nerve, seminal vesicles (including coagulation gland), skeletal muscle, skin, spinal cord (cervical, thoracic and lumbar), spleen, sternum (with bone marrow), stomach (with forestomach), testes, thymus, thyroids (with parathyroids), tongue, trachea, urinary bladder, uterus (horns and cervix) and vagina. All organs, except for the eyes, femoral bone, skeletal muscle and tongue, were examined microscopically.
Test article
AlphaGOS (α-GOS product) was supplied by Olygose (Venette, France) and was dissolved in water treated by reverse osmosis. Test article composition was analytically verified and supplied to the testing laboratory with a certificate of analysis. Test article and vehicle only were delivered at a volume of 10 ml/kg/day. Test article in solution was stable for 2 weeks under refrigerated conditions protected from light and prepared accordingly.
Statistical analysis
Statistical analysis was conducted using either Citox (D.7) software (body weight, feed consumption, haematology, blood biochemistry and urinalysis) or PathData (6.2d2) (organ weight). Specific statistical tests are specified where applicable in the text.
Results
The α-GOS product was well tolerated throughout the study. There were no mortalities during the trial. Test article did not affect feed consumption in either male or female rats when compared to control rats. No test article-related toxicologically significant differences were observed in the clinical examinations, body weights, haematology, clinical chemistry, urinalysis, organ weights, gross or histopathology.
Clinical and ophthalmological examination
Animals were assessed for a variety of clinical signs of distress over the course of the study. Chromodacryorrhea was observed in one male and one female each receiving the α-GOS product. While the chromodacryorrhea was only seen in animals receiving the α-GOS product, chromodacryorrhea is observed in laboratory-housed Sprague Dawley rats and was not considered to be related to the α-GOS product consumption. In the control group, five male rats displayed no abnormalities in fur appearance throughout the study. Of the five control male rats that did exhibit some fur abnormalities, there were five cases of scabbing (lasting 3–4 days) and one case of thinning hair (lasting from day 43 until killed). Seven of the 10 male rats receiving test article had no signs of abnormal fur; there were 2 cases of scabbing (lasting 14 or 26 days) and 1 case of thinning hair (lasting 14 days). Six of the 10 female control rats showed no signs of abnormal fur. Scabs presented on two control female rats (lasting 1–9 days) and there was one case of alopecia (lasting a day). Eight female rats receiving test article presented with no fur abnormalities. The two female rats that did present with fur abnormalities exhibited thinning hair and alopecia from 71 days to 72 days, respectively, until termination.
At the 12-week functional observational battery (FOB), both sexes in the treatment and control groups were normal for touch escape, reactivity to handling, touch response, auditory startle reflex, tail pinch response, forelimb grip strength, gait, posture and breathing. No animal in the study showed any evidence of salivation, lacrimation, piloerection, tremors, convulsions, ataxia, hypotonia, stereotypy or any excessive or abnormal defecation/urination. Stress-induced grooming was noticed in one male rat from both the control and α-GOS product groups and two control female rats and one female given α-GOS product. As this behaviour was seen in both control and α-GOS product groups and is also part of normal behaviour, it was not attributed to the test item.
In both male rats and female rats, the calculated mean landing foot splay was slightly higher in the control group. Landing foot splay measurements were taken twice and a mean was calculated for each rat. In male rats, the higher mean landing foot splay in the control group was mainly due to an isolated animal for which both assays gave high values, leading to a higher mean when compared to other animals in the same group. In female rats, this was attributed to highly variable individual measurements in some control group animals, which increased the mean.
Body weight
The α-GOS product did not affect the body weights of the rats compared to control (Table 1). However, the body weight change was statistically lower in the treated female rats compared to the control female rats (7 g vs. 12 g, respectively) between days 64 and 71. As this finding was only observed during 1 week of feeding and did not significantly affect total body weights, it was considered incidental and not toxicologically relevant.
Body weight of rats (g).a–c
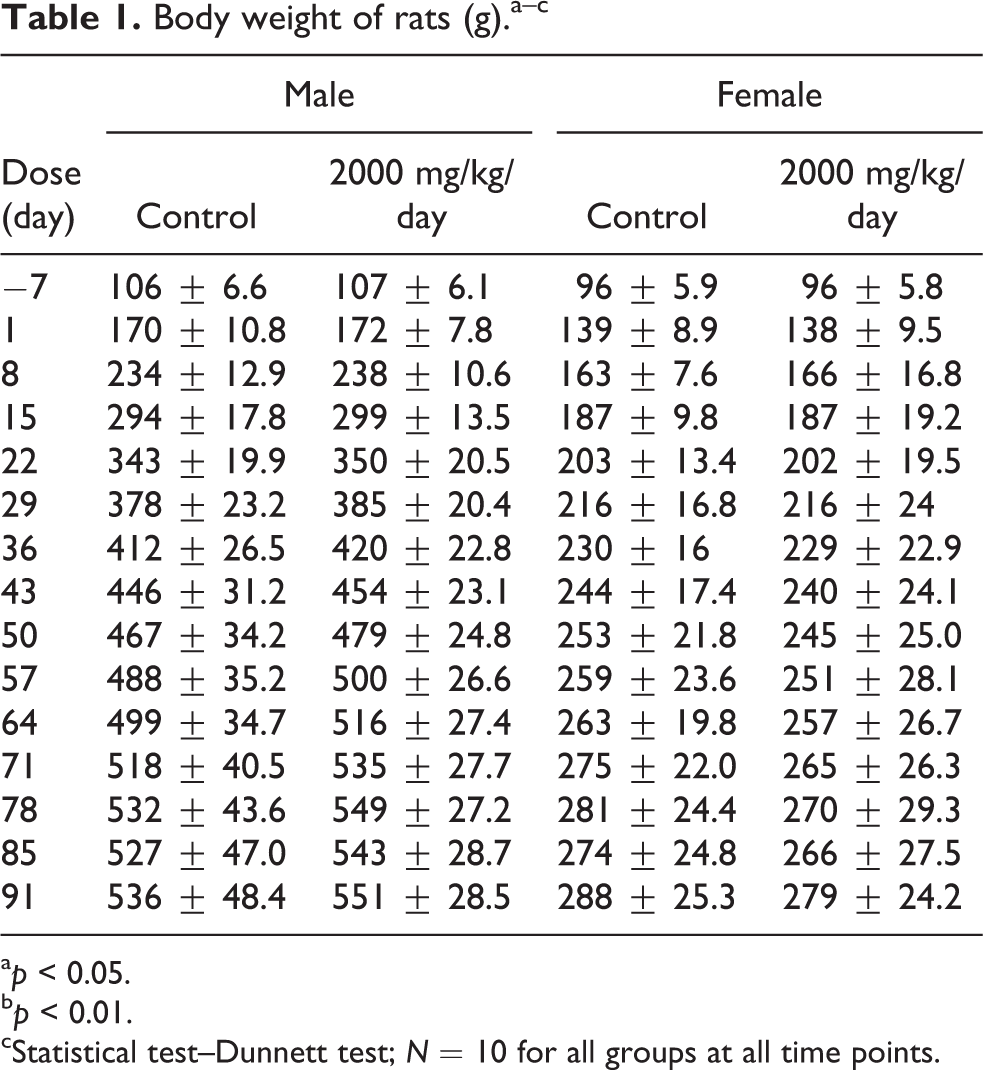
a
b
cStatistical test–Dunnett test;
Haematology
Haematology parameters (Table 2) were not adversely impacted by test article administration. The female rats receiving the α-GOS product had a statistically significant increase in mean corpuscular haemoglobin concentration (MCHC) compared to the control group (34.2 g/dL vs. 33.8 g/dL,
Haematology.a,b
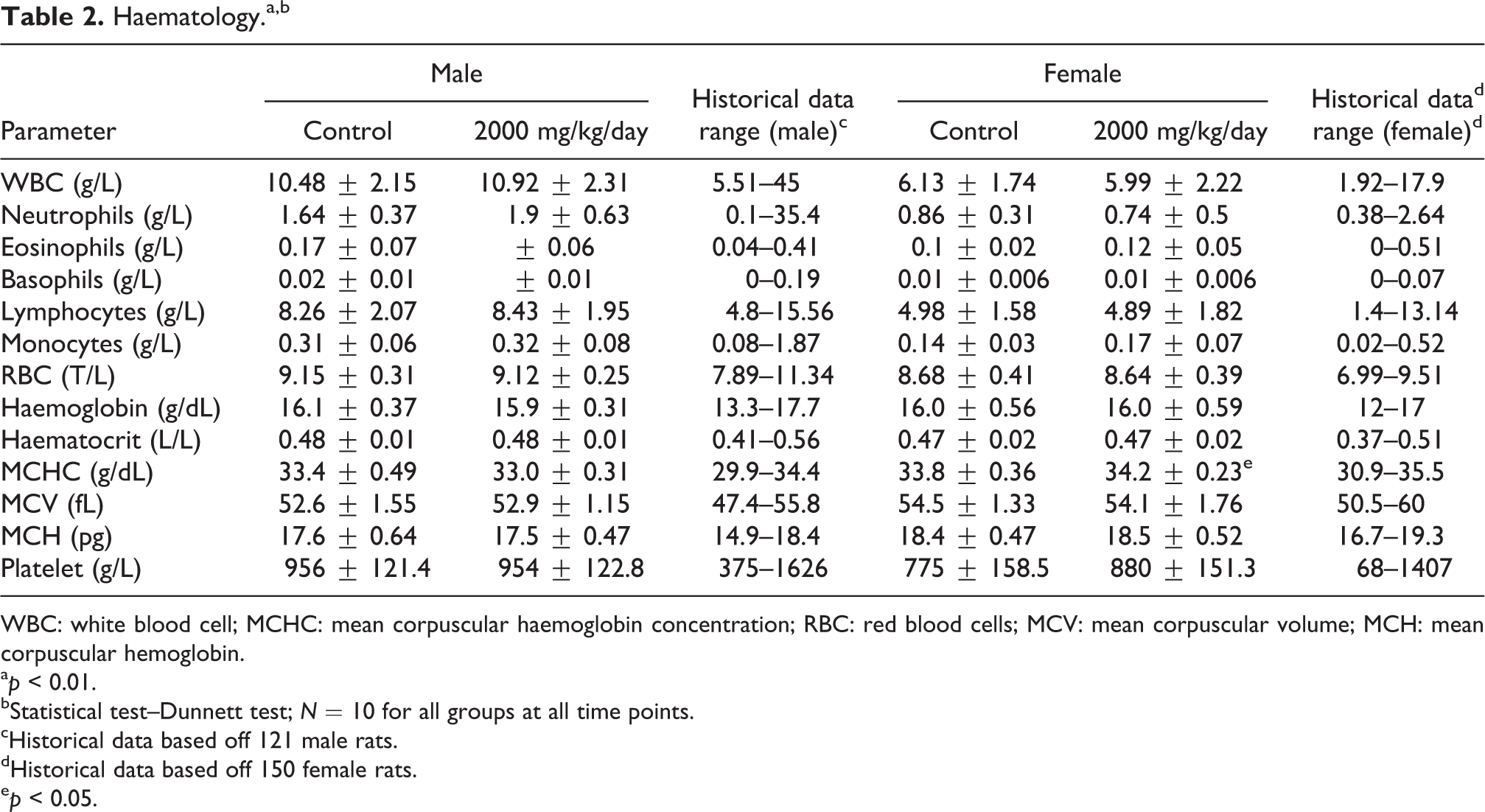
WBC: white blood cell; MCHC: mean corpuscular haemoglobin concentration; RBC: red blood cells; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin.
a
bStatistical test–Dunnett test;
cHistorical data based off 121 male rats.
dHistorical data based off 150 female rats.
e
Clinical chemistry
In male rats from the α-GOS product-treated group, blood glucose level (Table 3) was higher than in the control group (6.91 mmol/L vs. 5.99 mmol/L,
Blood chemistry.a
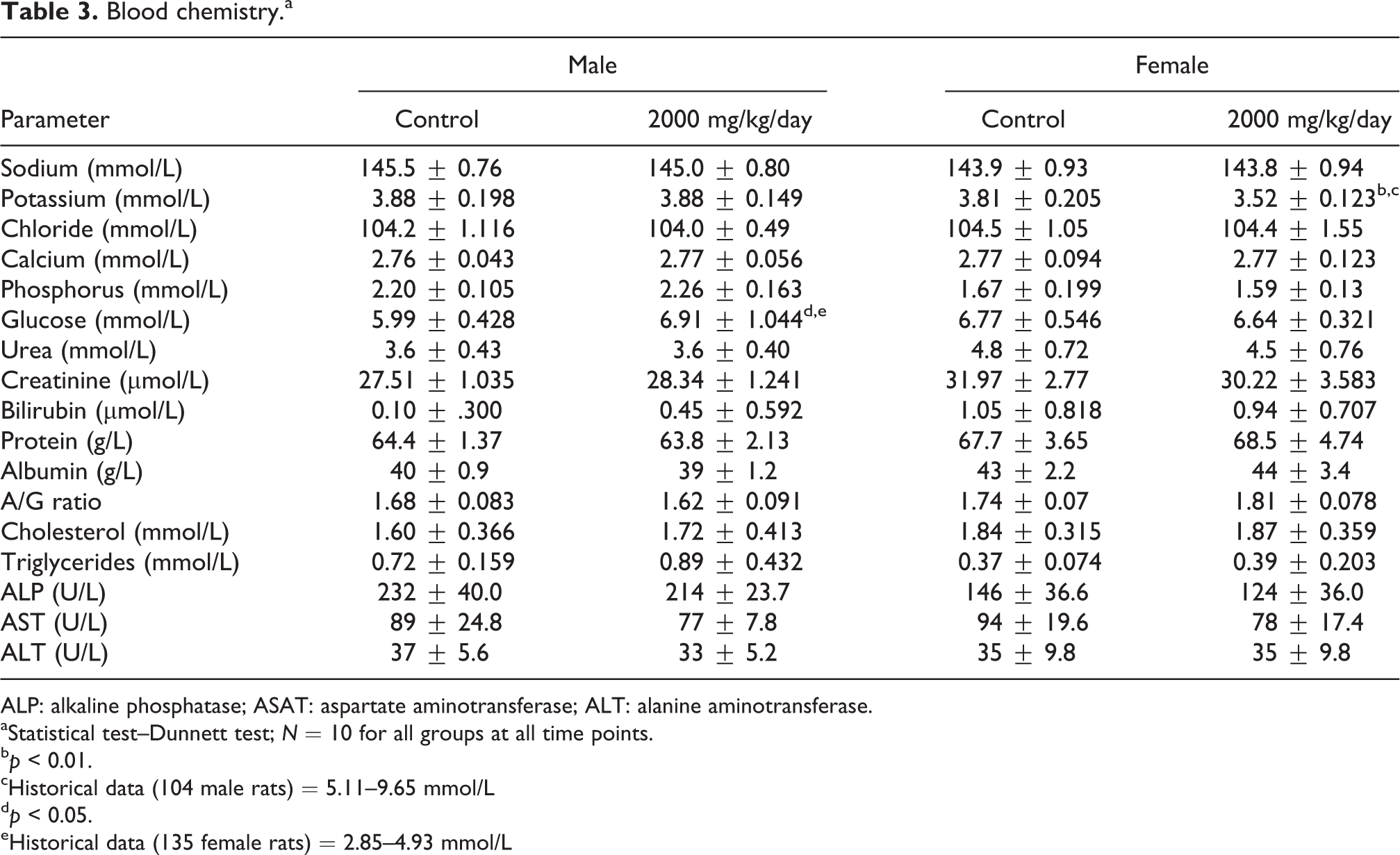
ALP: alkaline phosphatase; ASAT: aspartate aminotransferase; ALT: alanine aminotransferase.
aStatistical test–Dunnett test;
b
cHistorical data (104 male rats) = 5.11–9.65 mmol/L
d
eHistorical data (135 female rats) = 2.85–4.93 mmol/L
Urine analysis
In male rats of the treated group, the urine of 3 of 10 animals tested positive for nitrites. A few white blood cells were also observed in the urine of one male rat receiving the α-GOS product. However, these animals lacked any kidney pathology at histopathologic exam and did not display any clinical symptoms or organ weight changes.
Organ weights
The absolute and relative weights of the testes of rats receiving 2000 mg/kg/day of the α-GOS product were significantly increased when compared to controls (Table 4). The absolute liver weight in female rats was not changed between test and control groups; however, the liver weight relative to body weight was significantly increased in the female rats receiving the α-GOS product compared to controls. This was not a consistent finding across genders, as there was no significant finding in the liver weights (absolute or as a percent of total body weight) in the male rats receiving α-GOS product compared to the control group. No other differences in absolute or relative organ weights were noted between treated and control groups.
Organ weights.a
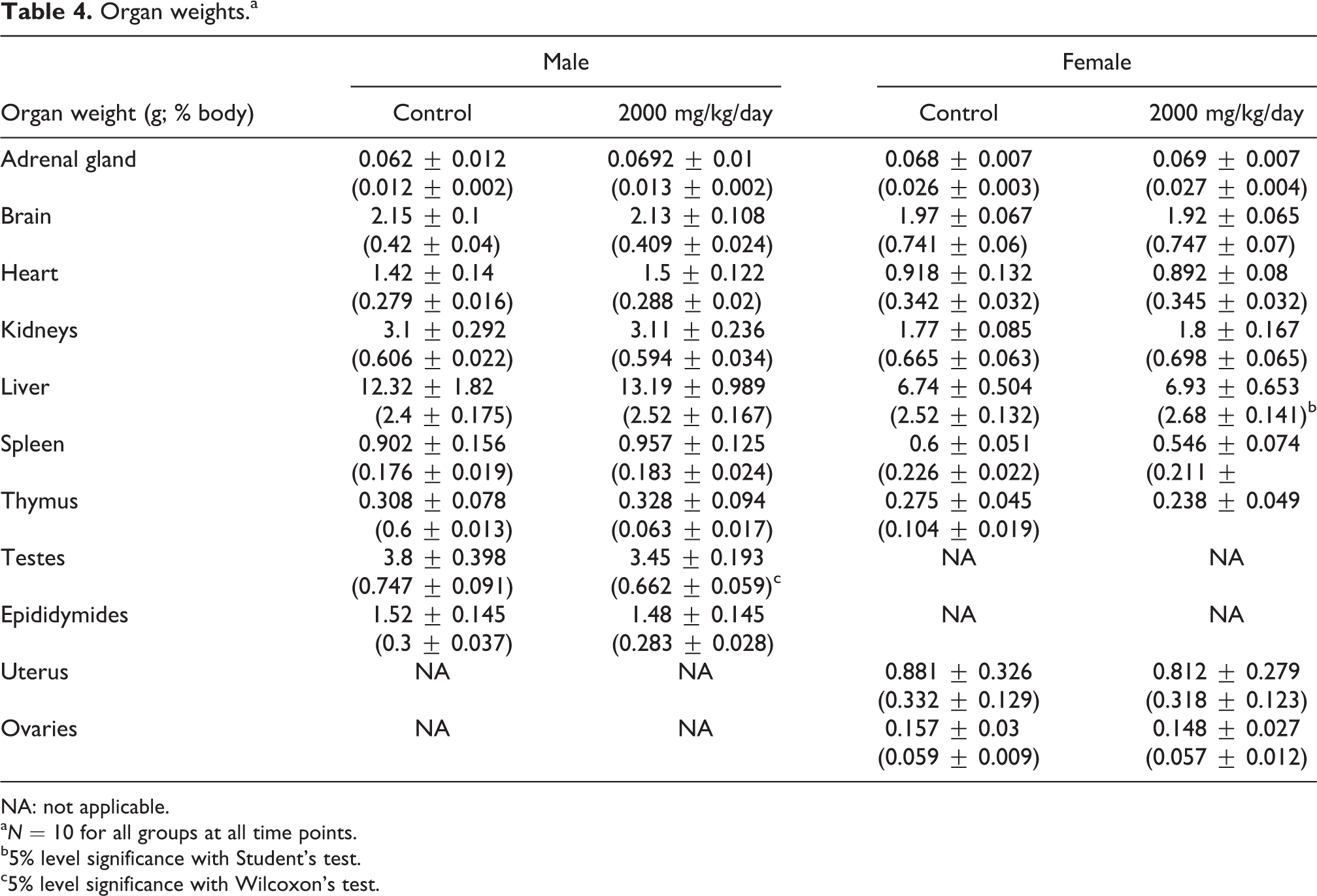
NA: not applicable.
a
b5% level significance with Student’s test.
c5% level significance with Wilcoxon’s test.
Histopathology
No adverse findings in gross or histopathology were attributed to test article. Slight lymphoid hyperplasia was observed in the spleen of a single male treated with the test article. As this observation may be occasionally seen in animals of this age, this isolated observation was considered not to be treatment related. Other changes were observed at a similar incidence and/or severity between both control and treated groups and were considered to be part of the normal background commonly seen in animals of this age.
Discussion
Daily oral (gavage) administration of AlphaGOS at 2000 mg/kg/day to Sprague Dawley rats for 13 weeks was well tolerated, and no clinical signs of toxicity were observed during the
A statistically significant reduction in body weight change was noted in treated female rats compared to control female rats; however this finding was only observed during 1 week of feeding, did not significantly affect total body weights and was not present in the male animals. Therefore, this finding was considered incidental and not toxicologically relevant.
There was a statistically significant increase in MCHC in female rats receiving AlphaGOS compared to the control. In view of the low magnitude of this change, and given that both MCHC values are within historical limits for these rats, this isolated finding was not considered biologically relevant. While blood glucose levels for male rats receiving AlphaGOS were higher than in the control group, female rats did not show a similar increase. Potassium levels were slightly lower in AlphaGOS-treated female rats compared to control animals. However, male rats showed no significant changes in potassium levels. Additionally, all blood glucose and potassium values were within the limits of historical data and not considered clinically adverse. The lack of significant changes in clinical chemistries across both sex groups corroborates the conclusion that any changes were not likely to have been test article related.
While the liver increased as a percentage of body weight in female rats receiving AlphaGOS, the absolute weight of the livers did not differ significantly. Additionally, this change was only observed in the female rats and no change in liver weight was seen in treated male rats. There were no adverse effects seen in clinical chemistries and no corroborating findings in histopathology that would indicate an adverse effect on the liver. Therefore, the effect noted in the relative liver weights in female rats was not considered toxicologically significant. The testes of the male rats treated with AlphaGOS were significantly increased. However, the absence of a similar trend in the epididymis, a lack of adverse findings in histopathology, and corroboration that the weight of the testes was within historical limits (0.74–4.416 g) indicate that this finding is not an adverse effect related to test article intake. The slight lymphoid hyperplasia observed in the spleen of one test article rat is occasionally seen in animals of this age. As this was an isolated observation, it was considered not to be test article related.
Like beta-galacto-oligosaccharides (β-GOS), RFO and AlphaGOS are NDOs.
5,10,11
The fact that the oligosaccharides reach the gut intact is the basis for their use as prebiotics. Raffinose and soy-based oligosaccharides have been shown to be sufficient sugar sources for multiple
β-GOS has been recognized as safe (GRAS) in the United States for use in general feed and infant formulas, 19 –28 approved for use in infant and follow-up formulas in the European Union 29 and Australia/New Zealand 30 and labelled as a feed for specific health use in Japan. 31 AlphaGOS is an alternative source for oligosaccharides in food and infant formula. European Food Safety Authority (EFSA) has opined that α-GOS contained in AlphaGOS is a non-digestible carbohydrate for which there is adequate scientific evidence to support a health claim related to reduction of postprandial glycemic responses. 32
This 90-day oral toxicological rat study has established that AlphaGOS is safe and well tolerated under the conditions of the assay. The no observed adverse effect level exceeds 2000 mg/kg/day.
Footnotes
Acknowledgement
The authors would like to thank Dr Roy Forster from CiToxLAB for careful reviewing of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Claire Kruger and Nicole Beauchamp are employees of ChromaDex Spherix Consulting, a business unit of ChromaDex, Inc. ChromaDex is a public company that was mandated and financed by Olygose to write the manuscript of the study presented in the publication. Virginie Modeste is an employee of CiToxLAB, a private company that provides research services and that was mandated and financed by Olygose to conduct the trial presented in the publication. Claire Kruger, Nicole Beauchamp and Virginie Modeste declared no conflicts of interest. Fanny Morel-Despeisse and Eric Chappuis are employees of Olygose, the company who mandated and funded the study presented in this manuscript and preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
