Abstract
β-Fructofuranosidase (β-D-fructofuranoside fructohydrolase; EC 3.2.1.26) is used in the production of fructo-oligosaccharides that are commonly used by the food industry as prebiotics for their purported health benefits. The β-fructofuranosidase discussed herein is obtained from a novel source organism that is a non-genetically modified strain of Aspergillus brunneoviolaceus, which phylogenetically belongs to the Aspergillus section Nigri. The safety of β-fructofuranosidase was evaluated in a series of toxicology studies as prescribed by Tier 1 toxicity testing by the European Food Safety Authority, including an evaluation of the mutagenicity and genotoxicity potential using the in vitro bacterial reverse mutation and mammalian chromosomal aberration assays, as well as systemic toxicity in a 90-day oral subchronic toxicity study in Sprague-Dawley rats. β-Fructofuranosidase was demonstrated to lack mutagenic or genotoxic potential based on the results of the in vitro assays due to absence of increased revertant colonies in the bacterial reverse mutation test and incidence of chromosome aberrations in the chromosomal aberration assay. Administration of β-fructofuranosidase by gavage at doses up to 1200 mg total organic solids (TOS)/kg body weight/day for 90 days did not elicit any systemic toxic effects in rats based on a lack of adverse effect in any study parameter, and therefore the no-observed-adverse-effect level of β-fructofuranosidase was concluded to be 1200 mg TOS/kg body weight/day, the highest dose tested. The results of the toxicology studies on β-fructofuranosidase from A. brunneoviolaceus demonstrate this species to be a safe and suitable source of enzymes for use by the food industry.
Introduction
β-Fructofuranosidase (β-D-fructofuranoside fructohydrolase; EC 3.2.1.26), also known as invertase, catalyzes the hydrolysis of beta-fructosyl bonds occurring at the terminal non-reducing residues of beta-
Historically, β-fructofuranosidase (invertase) was obtained from various microbial sources, including Aspergillus niger, Aspergillus japonicus, Rhizomucor miehei, and Saccharomyces cerevisiae.10,11 This enzyme is currently permitted for use in food production in a number of global jurisdictions, including Australia, New Zealand, Canada, China, France, and South Korea, thus highlighting its commercial importance.12–15 The Joint FAO/WHO Expert Committee on Food Additives (JECFA) assigned an acceptable daily intake of “not specified” to invertase obtained from S. cerevisiae due to the low toxicity of the enzyme and the resultant margin of safety based on its proposed uses in food production, suggesting that the use of this enzyme in food production does not pose any safety concerns to final consumers. 16 This acceptable daily intake for invertase is consistent with other enzymes utilized by the food industry, such as alpha-amylase, maltogenic amylase, and chymotrypsin.17–19 Evidently, there is a substantial history of use of β-fructofuranosidase obtained from microbial sources by the food industry.
A species of Aspergillus, Aspergillus brunneoviolaceus, was isolated from a food source based on its ability to produce high quantities of β-fructofuranosidase and is currently under investigation as a novel source for the commercial production of β-fructofuranosidase.20,21 A. brunneoviolaceus is phylogenetically related to Aspergillus fijiensis and has been considered to be synonymous with A. fijiensis.22,23 A. brunneoviolaceus (syn. A. fijiensis) belongs to Aspergillus Section Nigri, which are commercially important species, including A. niger, employed by the enzyme industry to produce enzymes and organic acids.24–26
Food enzymes obtained from microbial sources, such as those within Aspergillus Section Nigri, have the potential to contain mycotoxins or other toxic secondary metabolites that may be produced by the source organism.26–28 The strain of A. brunneoviolaceus described herein has been demonstrated to lack the ability to produce toxic mycotoxins and secondary metabolites, thus demonstrating its suitability as a source organism for enzymes used by the food and beverage industry. 27
Given the utility of β-fructofuranosidase in the production of various food ingredients, including FOS, and the increasing market demands for the inclusion of FOS in food for nutritional benefits, there is potential, although likely minimal, for residual amounts of enzyme and associated impurities originating from the fermentation process to be present in the final FOS foodstuffs. Despite the permittance of use of β-fructofuranosidase (invertase) from different microbial sources in various global jurisdictions, no toxicological information on β-fructofuranosidase produced from A. brunneoviolaceus was identified in the scientific literature. Therefore, a series of toxicology studies were undertaken to evaluate the potential genotoxic and systemic toxicological risk of residual amounts of β-fructofuranosidase in final foodstuffs and any potential chemical or microbial impurities derived from A. brunneoviolaceus. All studies were conducted in accordance with appropriate Organisation of Economic Co-operation and Development (OECD) test guidelines and in compliance with the principles of good laboratory practice, and comply with the Tier 1 toxicity testing criteria prescribed by the European Food Safety Authority.29,30
The findings of these studies support the safety of use of β-fructofuranosidase produced from A. brunneoviolaceus in the manufacture of FOS and related food products.
Materials and methods
Test material
Shin Nihon Chemical Co. (Anjo, Aichi, Japan) provided the liquid enzyme concentrate (Lot No. 160927T3) of β-fructofuranosidase isolated from A. brunneoviolaceus. The same manufacturing lot was used in all studies and compositional analysis of the test batch demonstrate it to meet the purity criteria for enzyme preparations as established by JECFA 31 and the Food Chemicals Codex. 32 The fructofuranosidase activity of the enzyme concentrate was analyzed to be 158800 U/mL or 120 mg total organic solids (TOS) per mL and was stable as validated by consistent enzyme activity and ultraviolet absorbance (280 nm) throughout the study period in the 90-day study when stored in airtight containers at −28.9 to −20.7°C.
The enzyme concentrate of β-fructofuranosidase was obtained by solid-state fermentation of non-genetically modified A. brunneoviolaceus strain FTF1. This strain was selected based on its ability to produce high levels of β-fructofuranosidase activity, cell viability, and suitability of the species for industrial production, as well as its lack of mycotoxin production. The production strain was deposited at the Biological Resource Center, National Institute of Technology and Evaluation.
Genotoxicity studies
Bacterial reverse mutation test (Ames test)
The bacterial reverse mutation test was conducted in accordance with OECD Test Guideline No. 471. 33 The Ames test was performed using Salmonella typhimurium strains TA100, TA1535, TA98, and TA1537 and Escherichia coli strain WP2uvrA. Microsomal fractions (S9) were obtained from the livers of phenobarbital- and 5,6-benzoflavone-induced male Sprague-Dawley rats and used for metabolic activation purposes. The following compounds served as the positive controls in studies conducted without metabolic activation: 0.01 μg/plate 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (Lot No. LKF6772, Wako Pure Chemical Industries) for TA100 and WP2uvrA and 0.1 μg/plate for TA98, 0.5 μg/plate sodium azide (Lot No. YSR7529, Wako Pure Chemical Industries) for TA1535, and 80 μg/plate 9-aminoacridine hydrochloride (Lot No. 09820CEV, Sigma-Aldrich) for TA1537. In studies conducted with metabolic activation, 0.5 μg/plate 2-aminoanthracene (Lot No. TLH6618, Wako Pure Chemical Industries) served as the positive control for TA98, 1.0 μg/plate for TA100, 2.0 μg/plate for TA1535 and TA1537, and 10 μg/plate for WP2uvrA. Distilled water served as the negative control.
Preliminary and concentration range-finding studies were conducted to establish appropriate concentrations and to evaluate potential mutagenicity prior to initiation of the main study. Based on the findings of the preliminary and concentration range-finding studies, the main study was conducted with concentrations of 375, 750, 1500, 3000, 6000, and 12000 μg TOS/plate, equivalent to 496, 993, 1985, 3970, 7940, and 15880 U/plate, respectively. Each concentration was tested in triplicate. In all tests, each plate was prepared as follows. One hundred microliters (100 μL) of the test article, negative control, or positive control were added to a test tube. For tests without metabolic activation, 500 μL of 0.1 mol/L sodium-phosphate buffer (pH 7.4) was added, while the tests with metabolic activation, 500 μL of S9 mix was added. Next, 100 μL of each bacterial suspension was added to each test tube, and mixtures were pre-incubated (120 strokes/min) in a shaking water bath (MM-10, Taitec) for 20 minutes at 37°C. After the pre-incubation period, 2 mL of top agar was added to each test tube and the contents were mixed then plated. The plates were incubated in an incubator (ILL-60, Ikeda Scientific) for 48 hours at 37°C. Each plate was macroscopically observed for precipitation or any change at the start of the pre-incubation period and the time of colony counting. Growth condition of the bacterial background was observed under a stereoscopic microscopic (40X) for bacterial growth inhibition by the test substance. Revertant colonies were counted with an automated colony analyzer (CA-11, System Sciences), correcting for the area and count loss. The results were considered positive if the mean number of revertant colonies was at least twice that of negative control colonies and the increase in revertant colonies was dose-dependent or reproducible. Statistical analysis of the data was not conducted. This is in accordance with OECD Test Guideline 471.
In vitro mammalian chromosomal aberration test
The in vitro mammalian chromosomal aberration test was conducted in accordance with OECD Test Guideline No. 473 34 using short-term and continuous treatment assays. Blood samples collected from two healthy, non-smoking volunteers were cultured in an incubator (ILL-60, Ikeda Scientific) with 4.4 mL of complete culture medium, comprised of sterilized, inactivated fetal bovine serum mixed with penicillin-streptomycin mixture, and 0.25 mL of phytohemagglutinin M (PHA) (Lot No. 1891743, Life Technologies) for 48 hours at 37°C. Next, the lymphocytes were collected by centrifugation (195 × g, 5 minutes) and the supernatant was removed, followed by treatment with the negative or positive control or β-fructofuranosidase concentrate under the following conditions: a 3-hour short-term assay in the absence of metabolic activation followed by a 21-hour expression period, a 3-hour short-term assay in the presence of metabolic activation followed by a 21-hour expression period, and continuous treatment for 24 hours in the absence of metabolic activation. Distilled water served as the negative control. Mitomycin C (MMC) (Lot No. 581AFI, Kyowa Hakko Kirin) served as the positive control at concentrations of 0.5 and 0.25 μg/mL in short-term assay in the absence of metabolic activation and in the continuous treatment assay, respectively. Cyclophosphamide (Lot No. 4431, Shionogi) was used as the positive control at concentrations of 8.00 and 12.50 μg/mL in the short-term assay in the presence of metabolic activation. The S9 microsomal fractions were obtained as previously described for the bacterial reverse mutation test.
In the short-term assay, human lymphocytes were incubated with β-fructofuranosidase concentrate for 3 hours at final concentrations of 1500–12000 μg TOS/mL, equivalent to 1985–15880 U/mL, in the absence of metabolic activation and 750–12000 μg TOS/mL, equivalent to 993–15880 U/mL, in the presence of metabolic activation. In the 24-hour continuous assay, the lymphocytes were incubated with β-fructofuranosidase concentrate at final concentrations of 93.8–3000 μg TOS/mL, equivalent to 124–3970 U/mL, in the absence of metabolic activation. The concentrations were chosen based on a preliminary dose-finding study to determine mitotic index and cell growth inhibition (data not shown). All control and β-fructofuranosidase concentrate treatments were conducted in duplicate.
Following the 3-hour treatment period in the short-term assays, the supernatant was removed from samples by centrifugation (195 × g, 5 minutes) and the cells were rinsed once with fresh medium, followed by incubation in tubes for another 21 hours at 37°C with 4.75 mL of fresh medium and 0.25 mL of PHA.
To inhibit mitosis in the metaphase, 0.1 μg/mL of colcemid solution (Lot No. 1813289, Life Technologies) was added to each tube 2 hours prior to preparation of the slides for examination. After incubation, the culture medium was removed by centrifugation (195 × g, 5 minutes) and the lymphocytes were exposed to a hypotonic treatment consisting of 4 mL of a 75 mmol/L potassium chloride solution at room temperature for 15 minutes. Then, the cells were fixed twice with a 4 mL room temperature 3:1 mixture of methanol and acetic acid, followed by cell suspension in the fresh fixative mixture in a refrigerator overnight. To check the cell density, one chromosome slide per tube was prepared. After confirmation, one drop of each cell suspension was added to a slide using a metaphase spreader (HANABI), and three slides per tube were prepared for chromosome analysis. The slides were dried and stained with 1.2% Giemsa solution (Lot No. HX57878704, Merck) diluted with 0.01 mol/L sodium phosphate buffer (Buffer tablets pH 6.8, Lot No. TP1305174, Merck) for 12 minutes. The slides were then rinsed with water and dried.
The highest concentration resulting in inhibition of mitosis of 50% or less was set as the highest concentration to be assessed for chromosome aberrations. Based on the calculated mitotic index for each group, microscopic examinations for chromosome aberrations were carried out in the short-term test in the absence and presence of metabolic activation at concentrations of 3000, 6000, and 12000 μg TOS/mL, equivalent to 3970, 7940, and 15880 U/mL, respectively. In the continuous treatment test, microscopic examinations were carried out at concentrations of 375, 750, and 1500 μg TOS/mL, equivalent to 496, 993, and 1985 U/mL, respectively. Microscopic evaluation of the selected concentrations involved examination of 150 metaphase cells per tube for the following chromosomal aberrations (chromosome- or chromatid-type aberrations): chromosome and chromatid gaps, breaks, exchanges, and other aberrations. In addition, the number of polyploid cells (69 chromosomes or more) was counted by observing 150 cells (300 metaphases) for each concentration.
Each assay was examined macroscopically for precipitation of the test article at the beginning of the study and at the end of the treatment.
For statistical analyses, a one-side Fischer’s exact test with a significance level of 2.5% was used to determine the significant difference in the incidence of aberrant cells between each test group. Concentration-dependency was not analyzed as no significant differences were observed in the results of the Fisher’s exact test. A response was considered to be positive if the incidence of cells with chromosomal aberrations was statistically significantly increased in more than one of the test concentrations compared to the negative control, a significant dose-response trend was observed, and the observed incidence was greater than the acceptable range calculated from the historical data of the negative control group. The final evaluation was made on the total incidence of aberrant cells minus the number of cells with only gaps.
90-day repeated-dose toxicity study
The 90-day repeated oral dose toxicity study was conducted in accordance with OECD Test Guideline No. 408. 35
Preparation of dosing formulations
Dosing formulations of the β-fructofuranosidase concentrate were prepared by serial dilution of the thawed stock solution (120 mg TOS/mL or 15880 U/mL) with distilled water to obtain concentrations of 13.3 and 40 mg TOS/mL, equivalent to doses of 133 (lose-dose group) and 400 mg TOS/kg body weight/day (mid-dose group), respectively. The undiluted stock solution, equivalent to 1200 mg TOS/kg body weight/day, served as the tested dose for the high-dose group. In terms of enzymatic activity, the doses correspond to 175959, 529200, and 1587600 U/kg body weight/day, for the low-dose, mid-dose, and high-dose groups, respectively. The prepared solutions were divided for daily use and stored in the refrigerator (2.5–7.8°C) until use. The dose formulations were confirmed to be stable under refrigerated conditions (1–9°C) in airtight, light-resistant containers for 8 days at concentrations of 0.240–120 mg TOS/mL.
Animals and treatment
Forty-five male and 45 female 4-week-old Crl:CD (SD) [SPF] rats were obtained from Charles River Laboratories Japan, Inc. Animals were housed individually in wire-mesh cages hung in an automatic water flushing breeding rack under the following conditions: temperature 20–26°C, 35–70% relative humidity, ventilation 12 times or more per hour, and 12 hour light/dark cycles. Animals were allowed access to pellet diet CRF-1 (Oriental Yeast) and drinking water ad libitum and observed daily for general health throughout the 8-day acclimation period, and body weight was measured on the day of receipt (Day 8) and the final day of the quarantine and acclimation period (Day 1). No abnormalities in clinical signs and body weight were observed during the acclimation period. Two males and five females were excluded from the study prior to group assignment due to evident congenital defects observed in the ophthalmological examination conducted on Days 2 and 1. The remaining animals were randomly assigned into groups (10/sex/group) based on body weights using a computer system package for safety studies (LATOX-F/V5, FFC) on the first day of dosing (Day 1). The body weight of the animals used in the study ranged from 156 to 182 g and 132 to 162 g for males and females, respectively. The body weight variations of the assigned animals were within 20% of the mean weight of each sex. All animals were 5 weeks of age at the start of the treatment period.
The doses of β-fructofuranosidase concentrate were selected based on the results of a 2-week repeated dose-finding study in rats. In the dose-finding study, animals were administered doses of 75, 300, or 1200 mg TOS/kg/day. No treatment-related toxicological effects on general condition, body weight change, feed consumption, and necropsy findings were reported at the highest dose of 1200 mg TOS/kg body weight/day (data not shown). As a result, doses for the 90-day study were selected as follows: 1200 mg TOS/kg body weight/day (high-dose level), 400 mg TOS/kg body weight/day (mid-dose level), and 133 mg TOS/kg body weight/day (low-dose level). The control animals received distilled water (vehicle solvent) at a dosing volume of 10 mL/kg body weight/day, equivalent to the treatment groups. The test article and the vehicle control were administered via gavage using a plastic syringe and Teflon gastric tube. The dosing solutions were prepared once or more per 7 days at concentrations of 13.3, 40, and 120 mg TOS/mL for the low-dose (133 mg TOS/kg body weight/day), mid-dose (400 mg TOS/kg body weight/day), and high-dose (1200 mg TOS/kg body weight/day) groups, respectively.
Clinical observations, body weights, food consumption and ophthalmology
The general condition of each animal was observed and recorded twice per day, before and after dosing, and once before necropsy (Day 91). Functional observation battery (FOB) tests, including analyses for sensorimotor function, grip strength, and locomotor activity were conducted in all animals in the final week of the study period. Detailed observations were conducted for home cage observation, responses on removal from cage, and behavior in open field once before dosing and weekly thereafter. Following the FOB tests, the locomotor activity of each animal was assessed using a cage-stationary-type activity-measurement system (LOCOMO LS-7, Melquest) every 1 minute for 1 hour. Body weight of the animals was measured every 7 days from the initiation of the study (Day 1) until Day 90, and on the day of necropsy (Day 91). Body weight gain from Day 1 to 90 was calculated. The amount of food provided to animals was weighed on the days of body weight measurement and the remaining food was measured on the subsequent body weight measurement day. Mean daily food consumption (g/day) was calculated for each animal based on the difference between the amount of food provided and amount of food remaining. Microscopic observations of the estrous cycle in female rats were conducted using vaginal smear preparations obtained from the animals prior to daily dosing from Day 85 until necropsy (Day 91). Ophthalmological examinations were carried out in all the animals before the initiation of the dosing period (Day 2 and 1) and in the final week of the dosing period (Day 86). Following observations for appearance and light reflex, the pupils were dilated to examine the anterior part of the eyeballs, optic media, and fundus oculi (Lot No. M514151, Mydrin P®, Santen Pharmaceutical).
Clinical pathology and urinalysis
Hematological and blood chemical examinations were conducted in all surviving animals on the day of the scheduled necropsy (Day 91), while urinalysis was performed during the final week. Animals were fasted overnight and blood samples were collected in tubes containing an anticoagulant (ethylenediaminetetraacetic acid dipotassium salt) the following day from the abdominal aorta of each animal under isoflurane anesthesia. Blood samples were analyzed with a Hematology System (ADVIA120, Bayer) for the standard hematological parameters as recommended by the OECD Guideline No. 408. 32
Plasma samples were prepared from the blood samples collected into tubes containing an anticoagulant (3.2% sodium citrate solution) by centrifugation at 1700 × g for 13 minutes at room temperature. A coagulation analyzer (STA Compact, Roche) was used to measure the prothrombin time and activated partial thromboplastin time in the plasma samples.
Serum samples were prepared from blood samples and collected into tubes containing a Gel and Clot activator (Venoject II, Terumo) to analyze blood chemistry parameters. Samples were centrifuged at 1700 × g for 7 minutes at room temperature and analyzed for the following parameters using an automatic analyzer (Hitachi 7170, Hitachi): total protein, glucose, triglyceride, total cholesterol, blood urea nitrogen, creatinine, total bilirubin, aspartate aminotransferase, alanine aminotransferase, γ-glutamyl transpeptidase, calcium, and inorganic phosphorus. Serum concentrations of sodium, potassium, and chloride were analyzed using an electrolyte analyzer (EA07, A&T). Albumin ratio, α1-globulin ratio, α2-globulin ratio, β-globulin ratio, γ-globulin ratio, albumin/globulin ratio, albumin concentration, α1-globulin concentration, α2-globulin concentration, β-globulin concentration, and γ-globulin concentration in the serum samples were measured using an electrophoresis analyzer (Epalyzer 2 plus, Helena Laboratories).
During the final week of the study period, animals were placed in an individual urine-sampling cage with food and water, and fresh urine samples were collected within 3 hours after urination and 24 hours after urination (pooled urine). The following parameters were analyzed in fresh urine using Ames test strips (N-Multistix, SG-L, Siemens Healthcare Diagnostics) and an automatic strip reader (CLINITEK Advantus, Siemens Healthcare Diagnostics: pH, occult blood, ketone bodies, glucose, protein, bilirubin, and urobilinogen. The 24-hour urine samples were centrifuged at 400 × g for 5 minutes following examination of the volume and color of urine. An electrolyte analyzer (EA07, A&T) was used to analyze the supernatant for electrolyte concentrations (sodium, potassium, and chloride), and an osmotic pressure analyzer (AUTO&STAT™ OM-6030, Arkray Factory) was used to evaluate the osmotic pressure. The total daily excretion value of each electrolyte was calculated from the urinary volume. Microscopic examination of the urinary sediments was performed followed by staining of the sediments by the Sternheimer method.
Pathology
All animals were terminated following blood sample collection by exsanguination under isoflurane anesthesia and subjected to pathological examination (organ weights, macroscopic, and histopathology). Organs weights were measured as specified in the OECD Test Guideline 408. The relative organ weights were calculated using absolute body weights and the final body weights recorded on Day 91. Histopathological examination of organs and tissues from all animals are consistent with those specified in OECD Test Guideline No. 408. Organs and tissues were fixed in an adequate volume of 10% neutral buffered formalin solution. The testes and eyes (including optic nerve and Harderian glands) were pre-fixed in formalin-acetic acid solution and Davidson’s solution, respectively, followed by fixation of both organs in 10% neutral buffered formalin solution. The left and right lungs were fixed by immersion into the fixative solution. All fixed organ and tissue samples were prepared for histopathological examination by embedding in paraffin, sectioning, and staining with hematoxylin and eosin. Specimens of the control and high-dose groups were examined microscopically, and all types and severity of histopathological findings were recorded according to INHAND.
Statistical analysis
Bartlett’s test for homogeneity of variances was used to analyze the following parameters: body weight, body weight gain, food consumption, metric FOB data (grip strength and locomotor activity), hematology, blood chemistry, urinalysis (volume, osmotic pressure, and electrolytes), and absolute and relative organ weights. Homogenous data (not significantly different) and heterogenous data (significantly different) were analyzed using Dunnett’s multiple comparison test and Steel’s test, respectively, to evaluate the statistically significant differences between the control group and each test substance-treated group. The significance of Bartlett’s test was analyzed at the 5% level of significance, while Dunnett’s test and Steel’s test were analyzed at the 5% and 1% level of significance using a two-sided analysis.
Results
Genotoxicity studies
Bacterial reverse mutation test (Ames test)
Results of bacterial reverse mutation test (concentration-finding study using pre-incubation method).
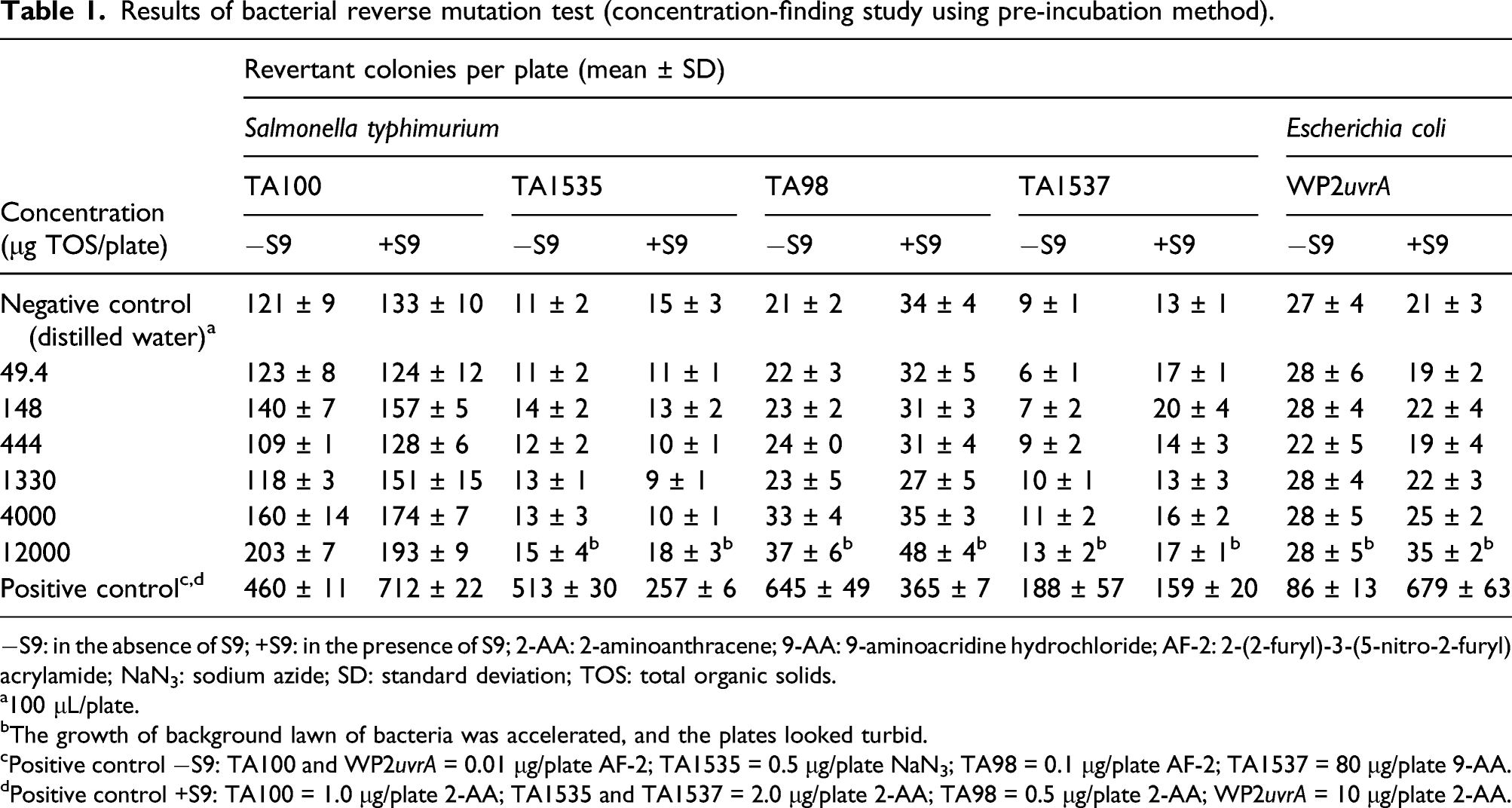
−S9: in the absence of S9; +S9: in the presence of S9; 2-AA: 2-aminoanthracene; 9-AA: 9-aminoacridine hydrochloride; AF-2: 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; NaN3: sodium azide; SD: standard deviation; TOS: total organic solids.
a100 μL/plate.
bThe growth of background lawn of bacteria was accelerated, and the plates looked turbid.
cPositive control −S9: TA100 and WP2uvrA = 0.01 μg/plate AF-2; TA1535 = 0.5 μg/plate NaN3; TA98 = 0.1 μg/plate AF-2; TA1537 = 80 μg/plate 9-AA.
dPositive control +S9: TA100 = 1.0 μg/plate 2-AA; TA1535 and TA1537 = 2.0 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA; WP2uvrA = 10 μg/plate 2-AA.
Results of bacterial reverse mutation test (main study using pre-incubation method).
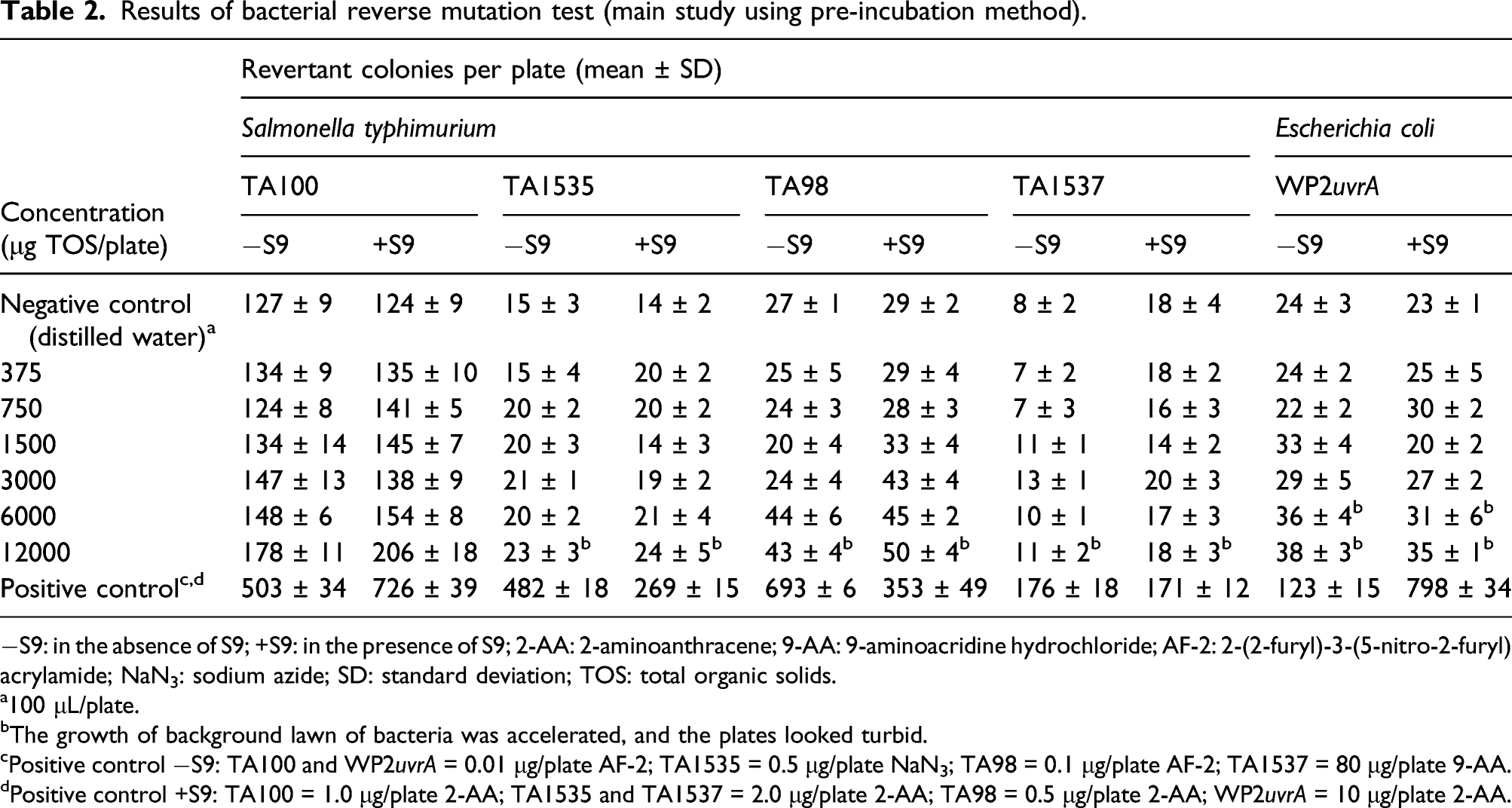
−S9: in the absence of S9; +S9: in the presence of S9; 2-AA: 2-aminoanthracene; 9-AA: 9-aminoacridine hydrochloride; AF-2: 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; NaN3: sodium azide; SD: standard deviation; TOS: total organic solids.
a100 μL/plate.
bThe growth of background lawn of bacteria was accelerated, and the plates looked turbid.
cPositive control −S9: TA100 and WP2uvrA = 0.01 μg/plate AF-2; TA1535 = 0.5 μg/plate NaN3; TA98 = 0.1 μg/plate AF-2; TA1537 = 80 μg/plate 9-AA.
dPositive control +S9: TA100 = 1.0 μg/plate 2-AA; TA1535 and TA1537 = 2.0 μg/plate 2-AA; TA98 = 0.5 μg/plate 2-AA; WP2uvrA = 10 μg/plate 2-AA.
In vitro mammalian chromosomal aberration test
Results of in vitro mammalian chromosomal aberration test.
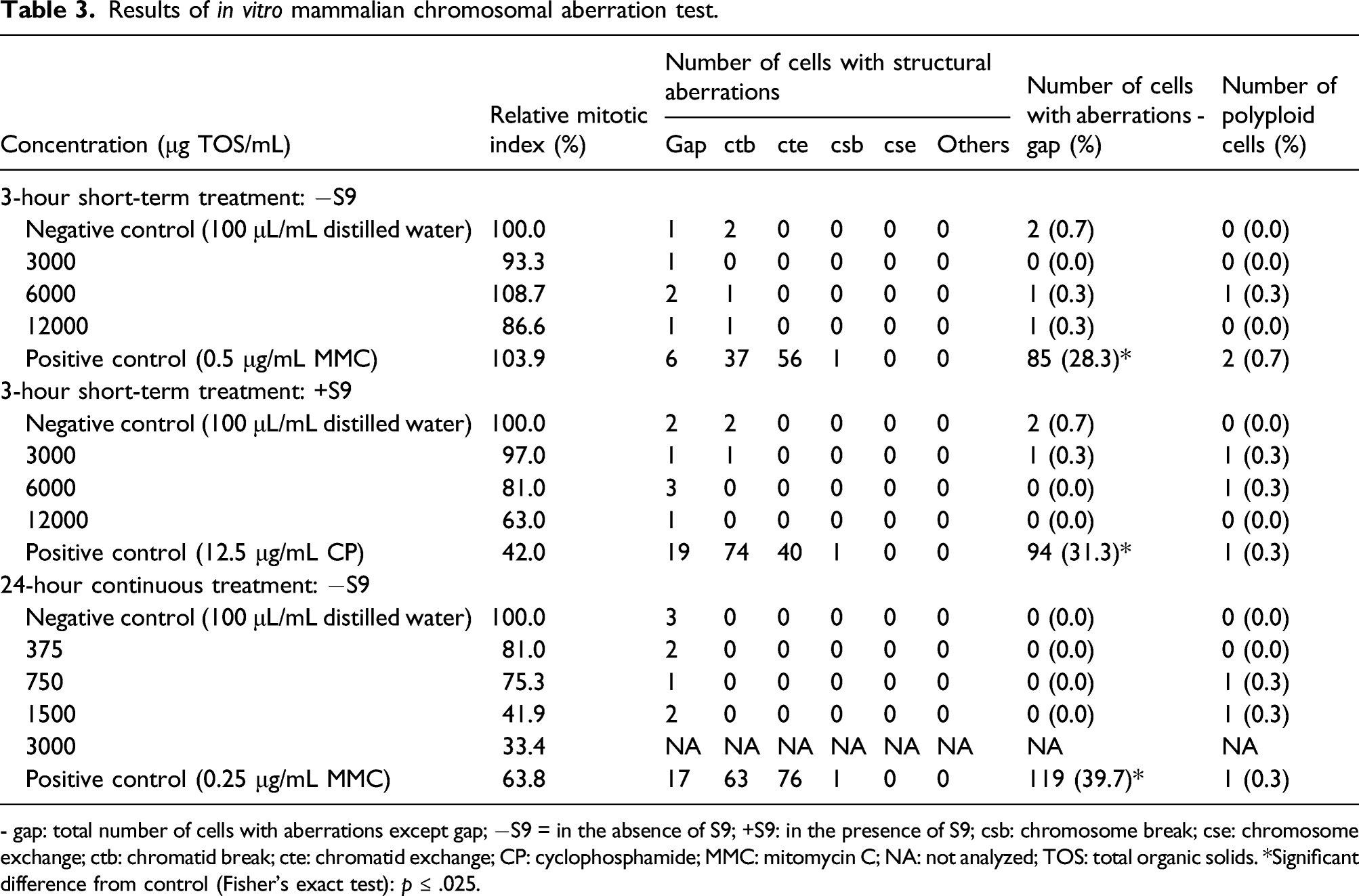
- gap: total number of cells with aberrations except gap; −S9 = in the absence of S9; +S9: in the presence of S9; csb: chromosome break; cse: chromosome exchange; ctb: chromatid break; cte: chromatid exchange; CP: cyclophosphamide; MMC: mitomycin C; NA: not analyzed; TOS: total organic solids. *Significant difference from control (Fisher’s exact test): p ≤ .025.
In the continuous assay, there was no statistically significant difference in the incidence of cells with chromosomal aberrations between the negative control group (0.0%) and the groups treated with the β-fructofuranosidase concentrate (0.0% at concentrations of 375, 750, and 1500 μg TOS/mL) (Table 3). The incidences of polyploid cells were 0.0, 0.3, and 0.3% at β-fructofuranosidase concentrations of 375, 750, and 1500 μg TOS/mL, respectively, and were not significantly different from the incidence in the negative control group (0.0%). In the positive control group treated with MMC, the incidence of chromosomal aberrations was statistically significantly higher (39.7%) compared to the negative control group. The relative mitotic index at the highest evaluated dose of 1500 μg TOS/mL was 41.9%.
90-day repeated-dose toxicity study
Clinical observations, body weights, food consumption, and ophthalmology
All animals except one male in the mid-dose group survived the 90-day study period until scheduled termination. This male rat was found dead on Day 33 and exhibited no changes in general condition upon necropsy. Throughout the dose administration period, toxicological effects related to the test article treatment were not observed in any of the groups upon clinical observation. One female in the control group displayed abnormal gait on Day 89 after dosing and swelling of the hindlimb on Day 90 before dosing. No treatment-related findings were observed for food consumption, ophthalmology, detailed observations, frequencies of defecation and urination, grip strength, or locomotor activity in any of the dose groups throughout the 90-day period.
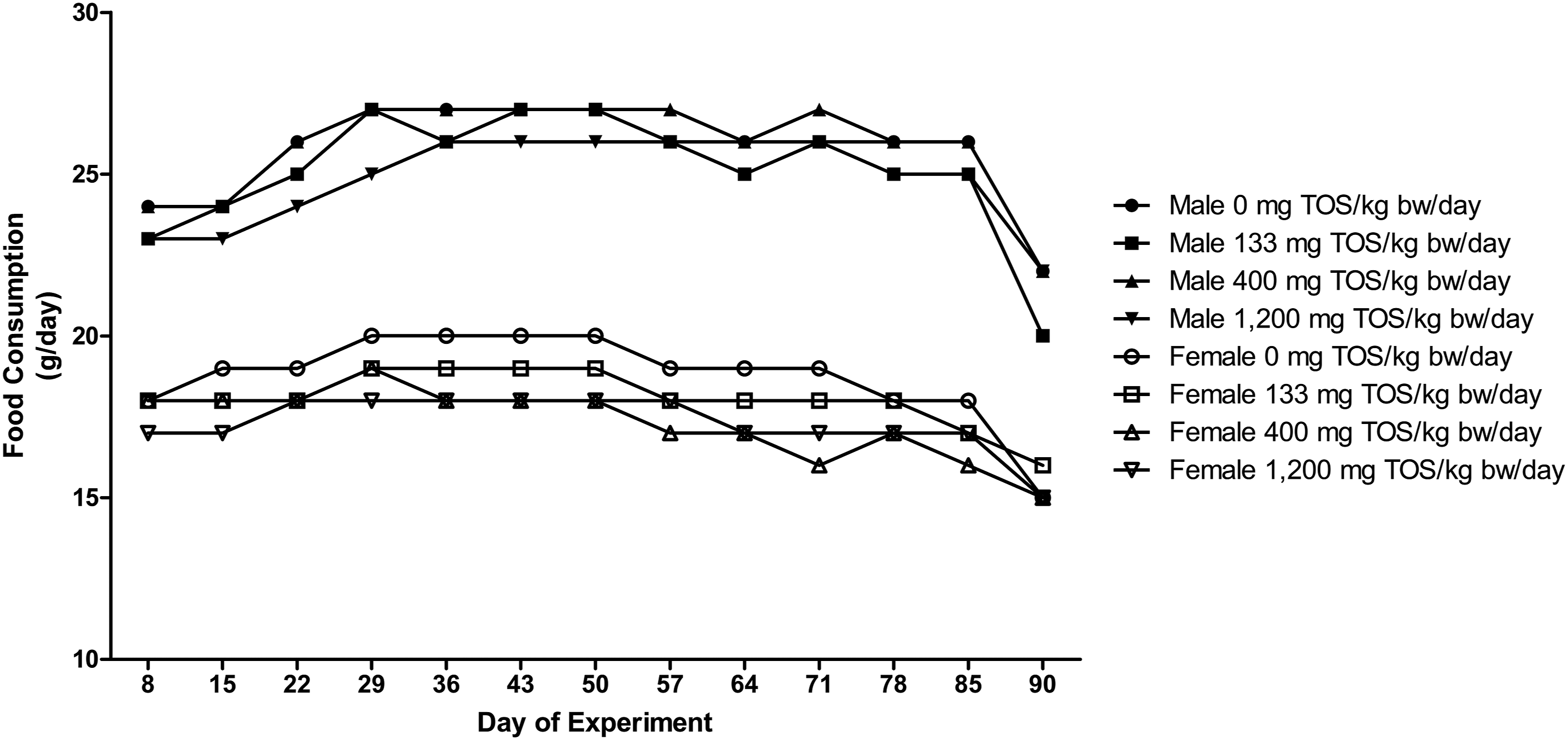
No significant differences in body weights and body weight gains (Figure 1) or food consumption (Figure 2) were observed in any animal during the dosing period. None of the selected animals showed abnormalities in estrous cycles in any of the test groups or control group from the observation period of Day 85–91. Body weight of male and female rats administered the β-fructofuranosidase concentrate by gavage for 13 weeks (N = 10). Food consumption of male and female rats administered the β-fructofuranosidase concentrate by gavage for 13 weeks (N = 10).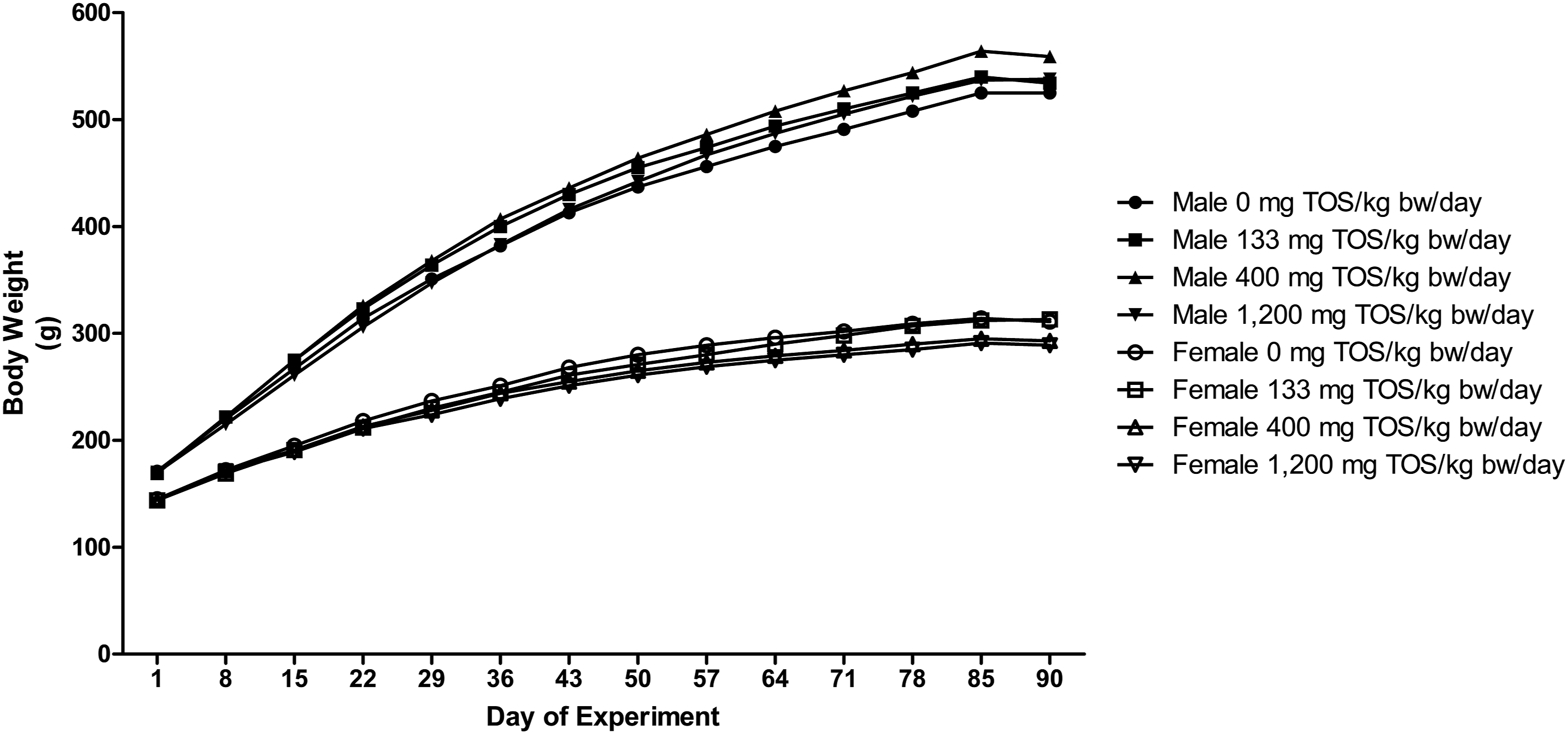
Clinical pathology and urinalysis
Hematology values for male and female rats administered the β-fructofuranosidase concentrate by gavage for 13 weeks.
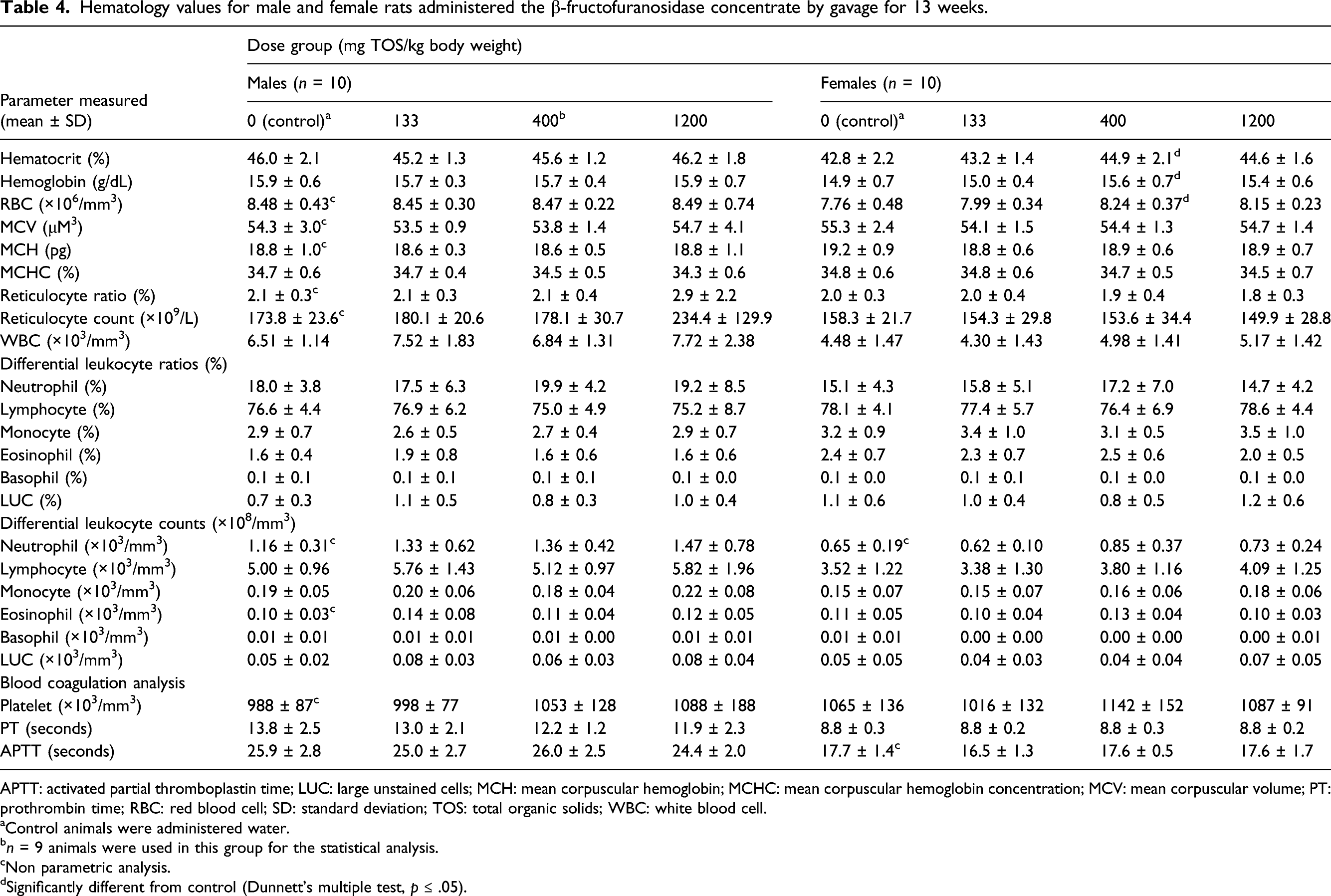
APTT: activated partial thromboplastin time; LUC: large unstained cells; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; MCV: mean corpuscular volume; PT: prothrombin time; RBC: red blood cell; SD: standard deviation; TOS: total organic solids; WBC: white blood cell.
aControl animals were administered water.
bn = 9 animals were used in this group for the statistical analysis.
cNon parametric analysis.
dSignificantly different from control (Dunnett’s multiple test, p ≤ .05).
Selected blood chemistry and urinalysis values for male and female rats administered the β-fructofuranosidase concentrate by gavage for 13 weeks.
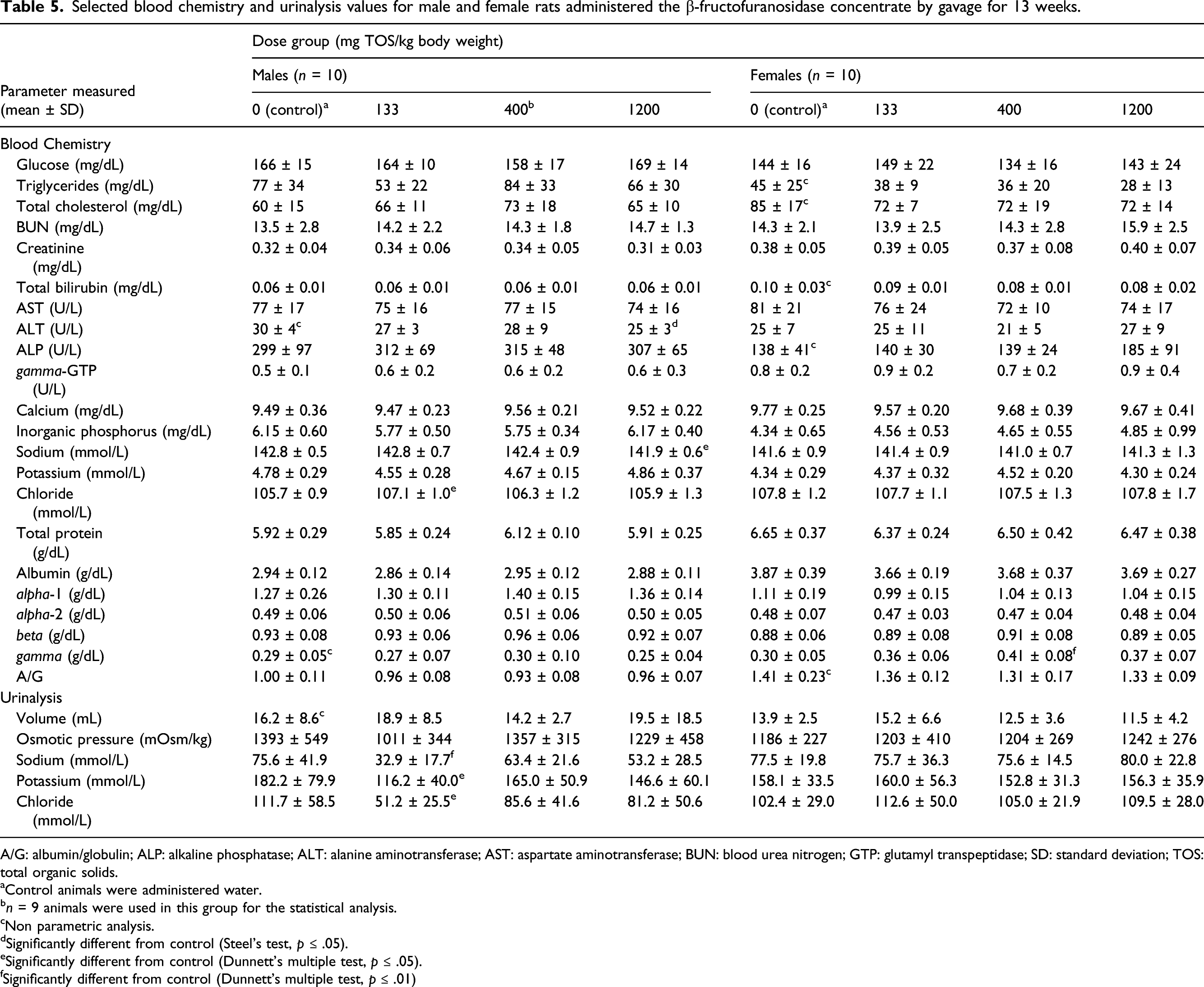
A/G: albumin/globulin; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BUN: blood urea nitrogen; GTP: glutamyl transpeptidase; SD: standard deviation; TOS: total organic solids.
aControl animals were administered water.
bn = 9 animals were used in this group for the statistical analysis.
cNon parametric analysis.
dSignificantly different from control (Steel’s test, p ≤ .05).
eSignificantly different from control (Dunnett’s multiple test, p ≤ .05).
fSignificantly different from control (Dunnett’s multiple test, p ≤ .01)
Pathology
Absolute and relative organ weights of male and female rats administered the β-fructofuranosidase concentrate by gavage for 13 weeks.
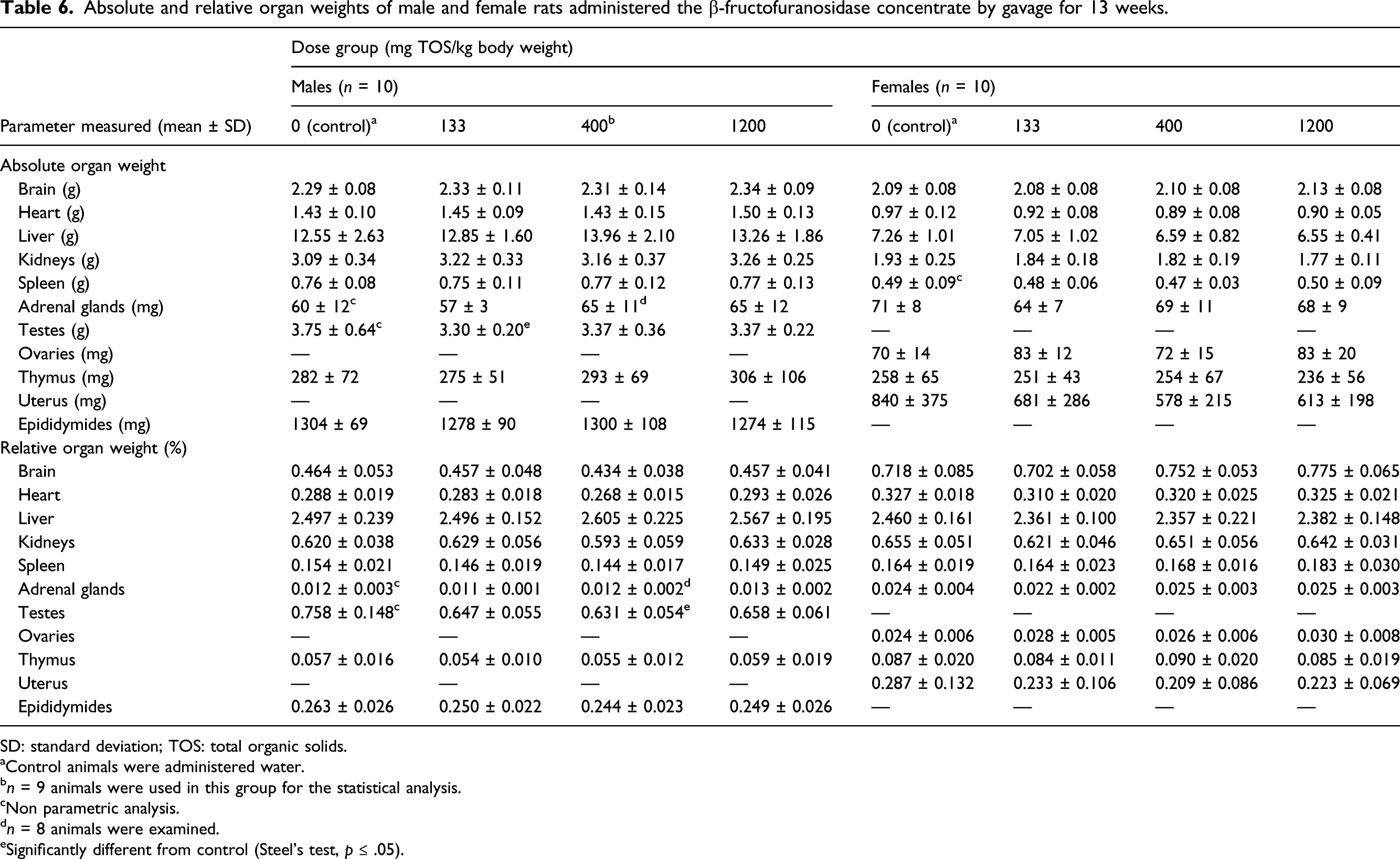
SD: standard deviation; TOS: total organic solids.
aControl animals were administered water.
bn = 9 animals were used in this group for the statistical analysis.
cNon parametric analysis.
dn = 8 animals were examined.
eSignificantly different from control (Steel’s test, p ≤ .05).
Macroscopic examination of the dead male in the mid-dose group revealed “reddish” lungs and retention of food in the esophagus. Upon histopathological examination, congestion and edema of the lungs were found in the same animal, in addition to focal hemorrhage in the thymus, increased hematopoiesis in bone marrow, and inflammatory changes in the kidney. Brown patches on the surface of lungs were identified in two males each in the 400 and 1200 mg TOS/kg/day groups, which was attributed to aggregation of macrophages in the histological examination. The following findings were observed in various groups: diverticulum in small intestine, nodule on small intestine, hepatodiaphragmatic nodules, mass in the abdominal cavity, cyst on kidneys, focal depression on kidneys, and defect of the adrenal gland. A fracture in the hindlimb and abnormal gait were observed in one female of the control group.
Discussion and conclusion
The safety of a liquid concentrate of β-fructofuranosidase obtained from non-genetically modified A. brunneoviolaceus was investigated in a series of toxicology studies involving evaluation of the potential genotoxicity and systemic toxicity of the enzyme. A bacterial reverse mutation test and mammalian chromosomal aberration test were conducted to evaluate the mutagenicity and clastogenicity potential of β-fructofuranosidase, respectively. In the preliminary study of the bacterial reverse mutation test, the number of revertant colonies in the tested concentrations increased 2-fold or more compared to the negative control, only in one strain (TA1537), in the presence and absence of metabolic activation. This finding was not observed in the concentration-range finding study or the main study, suggesting that it is not reproducible, and likely not indicative of mutagenicity of the enzyme concentrate. Moreover, the absence of germ contamination in the test samples was confirmed by sterility testing. The enzyme did not inhibit the growth of the bacterial background lawn and precipitation of the enzyme was not observed that would obscure the background lawn. 36 The bacterial background and revertant colonies were distinguished by stereomicroscopy. In the in vitro genetic toxicity tests, no biologically significant increases in the number of revertant colonies of any tested bacterial or yeast strains, or statistically significant increased incidences of chromosomal aberrations, were observed at concentrations of up to 12000 μg TOS/plate or 12000 μg TOS/mL, respectively, suggesting that β-fructofuranosidase does not have mutagenic or clastogenic potential. Under the conditions of these two assays, it is likely β-fructofuranosidase from A. brunneoviolaceus does not have any genotoxic potential.
The systemic toxicity of β-fructofuranosidase was investigated in a 90-day repeated-dose oral toxicity study in Sprague-Dawley rats. With the exception of one male in the mid-dose group that was found dead on Day 33, all animals survived the testing period. The male animal that was found dead did not exhibit any adverse conditions upon necropsy and the death was attributed to congestive edema of the lungs, likely due to improper gavage dosing. This death was considered to be incidental and not a result of administration of the liquid concentrate of β-fructofuranosidase. No treatment-related findings were observed in any of the clinical parameters, and statistically significant changes in body weight or body weight gain, or food consumption, were not observed in any test animal (Figures 1 and 2).
Female animals administered 400 mg TOS/kg/day exhibited minimally higher hematocrit and hemoglobin concentrations and red blood cell count compared to the control animals. These findings were not considered to be treatment related, as similar findings were not observed in the high-dose group, suggesting the absence of a dose-response relationship. Similar changes in hematocrit and hemoglobin concentration and red blood cell count were not observed in male animals receiving β-fructofuranosidase at similar doses, indicating these findings to be unilateral.
The only statistically significant changes in organ weights were decreased absolute testes weight in the low-dose group and decreased relative testes weight in the mid-dose group. These changes were considered unrelated to treatment, as there was no dose-response (high dose unaffected), the magnitude of change was small, and there were no histopathological correlates.
The findings from macroscopic and pathological examinations were considered to be spontaneous and incidental as they were focally distributed, were observed only in a single animal, or were not dose-dependent.
The findings from the series of toxicological studies indicate that the β-fructofuranosidase enzyme concentrate produced from A. brunneoviolaceus is non-genotoxic and is without significant toxicity. Given that the material is largely proteinaceous, this finding is not surprising and is consistent with the studies of other enzymes produced from other microorganisms, including Aspergillus oryzae and A. niger.37,38 While the current data support the safety of the β-fructofuranosidase enzyme concentrate produced from A. brunneoviolaceus, it is typical that the safety assessment of enzyme products give consideration to the source organism in addition to the inherent hazard profile of the enzyme.27,39 No published scientific opinions regarding the safety of A. brunneoviolaceus were identified. However, the synonymous species A. fijiensis is listed as a source organism for numerous enzymes in various global jurisdictions, such as Canada and Australia and New Zealand.13,40 It is known that A. brunneoviolaceus phylogenetically lies within the Aspergillus Section Nigri, which contains the commercially important A. niger.24,25 The safety of A. niger for the production of food ingredients and enzymes is well established in the scientific literature and has been extensively reviewed by the United States Food and Drug Administration and JECFA, who have collectively raised no safety concerns with the use of this microorganism in food production.41,42
A. brunneoviolaceus has been screened for the production of common secondary metabolites and mycotoxins, including aflatoxin B1, B2, G1, and G2, fumonisin B1 and B2, sterigmatocystin, zearalenone, ochratoxin A, and T-2 toxin by incubation up to 14 days in darkness at 25°C on Czapek yeast autolysate agar, malt extract agar, and yeast extract sucrose agar. These substances were analyzed by high-performance liquid chromatography as described by Frisvad et al. 43 and demonstrated to be below each respective minimal detection limit, ranging from 1 to 50 μg/kg, indicating that A. brunneoviolaceus does not produce mycotoxins or toxic secondary metabolites of concern. Therefore, considering that the source organism does not produce these compounds of concern, there is limited source of contamination in the β-fructofuranosidase product. In addition, commercial enzyme preparations are manufactured in accordance with GMP in which the final product must meet the specifications for enzyme preparations established by JECFA. 31 These specifications ensure the absence of chemical contaminants such as mycotoxins and other toxic secondary metabolites, including aflatoxins, fumonisins, ochratoxin A, T-2 toxin, sterigmatocystin, and zearalenone, that are known to be produced by fungal species.
In summary, the results of the toxicological studies conducted with a liquid concentrate of β-fructofuranosidase produced from A. brunneoviolaceus demonstrate its safety for use in food production, being non-genotoxic and showing no evidence of any toxic effects in a 90-day study that utilized doses of up to 1200 mg TOS/kg body weight/day. Information on the source organism also indicate it to be a likely safe source for producing β-fructofuranosidase. There is no indication that the source microorganism will produce significant amounts of toxic metabolites or mycotoxins.
Footnotes
Acknowledgements
Trung D. Vo, Jwar Meetro, Barry Lynch, Shahrzad Tafazoli, Akio Ichihara and Go Chikamatsu were responsible for preparation of the manuscript. All authors read and approved the content of the manuscript. Intertek Health Sciences Inc. received financial support from Shin Nihon Chemical Co. to assist in the preparation of the manuscript. Shin Nihon Chemical Co. provided editorial review and not the scientific content of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Trung D. Vo, Jwar Meetro, Barry Lynch, and Shahrzad Tafazoli are employees of Intertek Health Sciences Inc. and received financial compensation from Shin Nihon Chemical Co. for their consulting services in preparation of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from Shin Nihon Chemical Co. for their consulting services in preparation of the manuscript.
